Abstract
Epidermal stem cells (SCs) residing in the skin play an essential role for epidermal regeneration during cutaneous wound healing. Upon injury, distinct epidermal SCs residing in the interfollicular epidermis and/or hair follicles are activated to proliferate. Subsequently, SCs and progeny migrate, differentiate and restore the epidermis. We review a role of the vitamin D signaling through its receptor of vitamin D receptor (Vdr) in these processes. Vdr conditional knockout (cKO) mouse skin experiences a delay in wound re-epithelialization under low dietary calcium conditions, stimulating our efforts to examine a cooperative role of Vdr with calcium signaling through the calcium sensing receptor in the epidermis. We review the role of vitamin D and calcium signaling in different processes essential for injury induced epidermal regeneration during cutaneous wound repair. First, we discuss their roles in self-renewal of epidermal SCs through β-catenin signaling. Then, we describe epidermal remodeling, in which SCs and progeny migrate and differentiate to restore the epidermis, events controlled by the E-cadherin mediated adherens junction signaling. Finally, we discuss the potential mechanisms for vitamin D and calcium signaling to regulate injury induced epidermal regeneration mutually and interdependently.
Keywords: Vitamin D, Calcium, Stem cells, Epidermis, Regeneration, Wound healing
Core tip: Vitamin D and calcium signaling play critical roles in epidermal stem cells and progeny to regenerate the epidermis during cutaneous wound healing. Their regulation of these processes is mediated at least in part through β-catenin and E-cadherin mediated adherens junction signaling.
INTRODUCTION
Chronic skin wounds are estimated to affect 6.5 million patients in the United States at a cost of over $25 billion[1]. A disproportionate number of these wounds are found in patients suffering from a variety of medical conditions including poor nutrition. A survey of patients with chronic leg ulcers by one of our clinical collaborators, Dr. Gasper, found vitamin D deficiency (25OHD levels less than 20 ng/mL) and decreased serum calcium (below 8.7 mg/dL) in nearly 50% of these patients (unpublished observation with permission). Moreover, chronic kidney disease but without diabetes mellitus or cardiovascular disease led to delayed healing of abdominal surgical wounds correlating inversely with 25OHD levels[2,3].
Adult stem cells (SC) residing in the skin play an important role in the regeneration of the epidermis after wound injury. Understanding the mechanisms regulating these adult SC is central for understanding epidermal regeneration during cutaneous wound healing. Skin epithelia are derived from the ectoderm and differentiate into the interfollicular epidermis (IFE), sebaceous gland (SG) and hair follicle (HF) during the embryonic developmental process. After birth, adult SC residing in the basal layer of the epidermis (eSC), isthmus (iSC) and bulge (bSC) regions of the HF regenerate the IFE, SG and HF, respectively[4-7]. In the epidermis, eSCs produce transient amplifying cells, which leave the basal layer and produce differentiation marker proteins such as involucrin (IVL), keratin 1 (KRT1), loricrin (LOR) and filaggrin (FLG) in a sequential process. In the proximal portion of the HF, iSCs maintain the SG. The bSCs regenerate HFs in a cyclic manner through activation signals initiated by the dermal papilla adjacent to the bSC.
However, when the skin is injured, these stem cells and progeny from all regions of the HF, SG and IFE contribute to regeneration of the epidermis at least initially[4,7] but to a different extent. Ito et al[8] found that bSCs provide around 25% of the newly regenerated epidermal cells by using an inducible Krt15-crePR/R26R transgene, although these cells did not persist during the healing process. Levy et al[9] demonstrated that the SCs from HF infundibulum also contribute to re-epithelialization by using a Shh Cre/R26R transgene, and these cells remained in the newly formed epidermis. However, eSC in the IFE make the greatest and most persistent contribution to epidermal regeneration[4]. Moreover, Langton et al[10] showed that bSC are not required for wound re-epithelialization because epidermis lacking HF as in the paw also re-epithelize the wound but with a delayed rate. The self-renewal of these SCs and their injury induced activation as well as migration and differentiation of progeny are controlled by various signal pathways.
The vitamin D receptor (VDR) is enriched in these stem cells. Its ligand,1,25(OH)2D3, a well-known regulator of epidermal differentiation and proliferation[11] can be produced in these epidermal cells. The epidermis is the major source of vitamin D for the body. Vitamin D is produced from 7-dehydrocholesterol by irradiation with UVB from the sun. As noted keratinocytes in the epidermis metabolize vitamin D to its active ligand 1,25(OH)2D3[11]. Tian et al[12] observed that topical 1,25(OH)2D3 enhanced wound healing. Vitamin D signaling regulates epidermal stem cells by interaction with other signaling molecules during cutaneous wound healing.
β-catenin signaling plays an important role for the maintenance of adult SCs in skin in both the HF and IEF. The role of β-catenin in bSC function has been extensively reported[13,14], but less attention has been paid to its role in eSC function. However, a sensitive probe for β-catenin signaling in the IFE is that of an Axin 2 Cre reporter that demonstrates a role for β-catenin in eSC activation[5,13]. VDR appears to support activation of adult SCs through interaction with β-catenin. VDR binding to β-catenin promotes VDR transcriptional activity and facilitates cell fate determination[15,16]. In the HF, VDR is essential for β-catenin signaling and bSC activation[17,18]. VDR binding to β-catenin in the AF2 domain supports transcription for the bSC marker Krt15 and HF differentiation genes such as PADI3 and S100a3[16]. Our observations also suggest that the same may be true for eSC in the epidermis as keratinocytes lacking Vdr show a blunted wound induced activation of β-catenin target gene expression at the wound edge as described below.
Calcium signaling also is important for cutaneous wound repair through interaction with vitamin D signaling. VDR profoundly affects calcium signaling within the cell including the induction of the calcium sensing receptor (CaSR) and a number of the pathways regulated by both calcium and vitamin D that are involved in epidermal differentiation. CaSR is a seven transmembrane domain, G protein coupled receptor first identified in parathyroid cells[19], that we cloned from keratinocytes[20]. CaSR is essential for the keratinocyte response to calcium[21,22], but like the VDR its role in wound healing has received little attention. The in vivo role of CaSR in calcium signaling in epidermis is demonstrated by a Casr null mice, in which the transmembrane domain and intracellular Casr is deleted from Krt14 expressing keratinocytes[23]. The expression of the CaSR is increased by 1,25(OH)2D3, causing the keratinocyte to be more sensitive to calcium actions[24,25]. Moreover, to our surprise, CaSR deletion reduces VDR expression[23].
Calcium signaling mediates epidermal remodeling after wound injury through adherens junction (AJ) signaling. In skin epithelia, the core molecular components of the AJ are cadherins and their binding partners β-catenin, α-catenin, and p120-catenin. The AJ signaling plays an essential role in epidermal differentiation via its role in activating phospholipase (PLC)γ, that in turn hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) to inositol trisphosphate (IP3) and diacylglycerol (DAG), signaling molecules critical for the differentiation process through their release of calcium from intracellular stores (IP3) and activation of several protein kinase Cs[26]. Formation of the E-cadherin/catenin complex is regulated by both VDR and calcium[26]. The E-cadherin/catenin complex also links to the underlying cytoskeleton via α-catenin that helps form an epithelial sheet in enabling the cell migration required to re-epithelialize the wound in addition to promoting its subsequent differentiation to regenerate the epidermis[27].
In this review, we focus on the roles of vitamin D signaling and calcium through their receptors (VDR, CaSR) in the epidermis for the control of adult SCs and progeny during the epidermal response to wound injury, where a defective response leads to poor wound healing.
VITAMIN D AND CALCIUM SIGNALING IN INJURY INDUCED ACTIVATION OF EPIDERMAL SC THROUGH INTERACTION WITH β-CATENIN SIGNALING
Studies of the role of VDR in skin have previously focused on hair cycling as alopecia (hair loss) is a striking phenotype in global Vdr knockout (KO) mice[28], in which a gradual decrease in bSC has been reported accompanied with impaired β-catenin signaling[18]. Delayed wound healing is reported in these mice, but it was primarily attributed to altered dermal fibroblast function not to alterations in epidermal function[29]. However, our studies showed that VDR has an essential role in epidermal SCs and progeny during cutaneous wound healing. Wound closure is delayed and wound re-epithelialization is retarded in Vdr conditional knockout (cKO) mice in which Vdr is specifically removed from Krt14 expressing epidermal SCs and progeny[30,31]. The number of SCs residing in the IFE as well as the HF is decreased[30] demonstrating impaired self-renewal of these SCs. In addition, Vdr cKO results in blunted SC proliferation and in impaired β-catenin signaling[30] that plays an important role in the epidermal response to wounding. These results are observed only when Vdr cKO mice are maintained on a low dietary calcium. Therefore, we explored the co-operative role of calcium signaling with vitamin D signaling by generating conditional double knockout mice (cDKO), in which both Vdr and Casr are deleted from Krt14 expressing epidermal SCs and progeny[32]. Delayed wound closure and retarded wound re-epithelialization is also observed in these cDKO mice ingesting normal calcium diets. Injury activated SC proliferation is impaired at the wounding edges. In addition, injury induced induction of β-catenin target genes was also blunted in cDKO wounds[32]. These responses are similar to skin wounds in Krt14 specific β-catenin KO mice. These results show that Vdr and Casr are essential for injury-induced SC activation at least in part via stimulation of β-catenin signaling.
VITAMIN D AND CALCIUM SIGNALING IN ADHERENS JUNCTION FORMATION ESSENTIAL FOR EPIDERMAL REMODELING
Our transcriptomic study also revealed that AJ signaling is a top canonical pathway altered in the epidermis of mice that lack both Vdr and Casr (cDKO) in Krt14 expressing keratinocytes[32]. The expression levels of E-cadherin and the levels of the epithelial specific desmosome component desmoglein 1 are decreased in wounds of cDKO mice[32]. In addition, the expression of the IFE early differentiation marker KRT1, middle differentiation marker IVL, and late differentiation marker LOR in the epidermis did not extend across the wound and remained disorganized in the shortened epithelial tongues at the remodeling stage of wound healing of Vdr cKO[30]. The reduction of E-cadherin and differentiation markers are also observed in Casr cKO mice, in which Casr is removed from epidermal SC and progeny[33], in association with a delay in wound closure and reduced wound re-epithelialization. Similarly, cell migration is impaired in an in vitro wound scratch model when VDR[30] , CaSR[33] or both VDR and CaSR[32] are silenced in cultured human keratinocytes. Therefore, vitamin D and calcium signaling are critical for keratinocyte migration and E-cadherin/catenin mediated epidermal differentiation, each essential for epidermal regeneration during wound healing.
POTENTIAL MECHANISMS FOR VITAMIN D AND CALCIUM SIGNALING TO REGULATE WOUND RE-EPITHELIALIZATION
Compensatory and/or interacting aspects of vitamin D and calcium signaling on wound healing exist. We examined two processes; (1) β-catenin signaling, that induces epidermal proliferation to produce the cells that subsequently regenerate the epidermis[4,5], and (2) the E-cadherin/catenin complex that is critical for keratinocyte differentiation as well as migration during wound induced re-epithelialization[34]. Our working model summarizes our findings illustrating the reduction in proliferation due to defects in the nuclear actions of β-catenin and decreased re-epithelialization caused by failure of migration, differentiation and formation of the AJ in the epidermis of mice in which vitamin D and calcium signaling are disrupted (Figure 1). Potential molecular mechanisms by which vitamin D and calcium interact to control migration and proliferation of keratinocytes during wound induced epidermal regeneration are shown (Figure 2). First, VDR induces genes including CaSR and β-catenin regulating differentiation genes in the epidermis and HFs[11]. VDR may facilitate β-catenin binding to its response elements in target genes such as cyclin D1 to promote cell proliferation[16]. VDR also is required for calcium, mediated by the CaSR, to form E-cadherin/catenin complex to stimulate AJ signaling[22]. This is due in part to activation of the Src/Fyn kinases that phosphorylate the catenins facilitating their incorporation into the E-cadherin/catenin complex. This complex provides a reservoir of β-catenin in the membrane. The shift of β-catenin from the nucleus to the membrane is crucial to allow differentiation, in part by reducing its proliferative function in the nucleus.
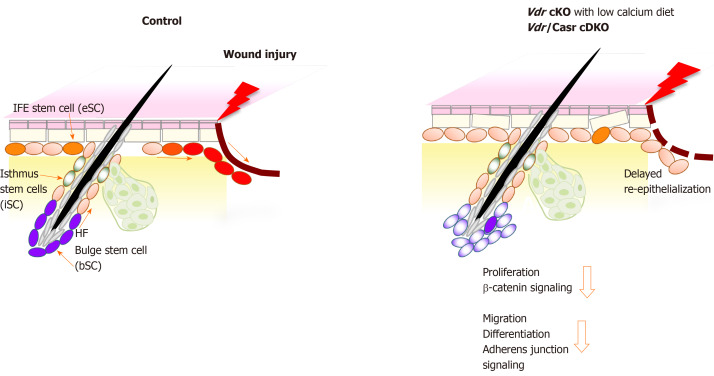
Figure 1.
Schematic model showing that deficiency in both vitamin D receptor and calcium sensing receptor prevents proliferation and migration of keratinocytes thus delaying wound re-epithelialization of the wounded epidermis. The location of the different stem cells (SC) niches in skin is shown; stem cells in the hair follicle bulge (bSC shown in purple color) required for hair cycling, isthmus stem cells (iSC, green) in the junctional zone of the upper hair follicle responsible for sebaceous gland renewal, and epidermal stem cells (eSC, orange) in the interfollicular epidermis (IFE) responsible for epidermal regeneration. Upon injury of normal skin (left panel), these stem cells at the wound edge are activated to proliferate, shifting their normal cell fate to re-populate the disrupted epidermis by migrating to the wound and differentiating to re-epithelialize the wound (red color). In contrast, vitamin D receptor (Vdr) conditional knockout (cKO) mice fed a calcium deficient diet and Vdr/Casr double KO (cDKO) mice show defects in these stem cells that reduce their responses to wounding (right panel). The number of bSC and eSC decreases in Vdr cKO demonstrating defects in their self-renewal. Both Vdr cKO and cDKO mice have decreased injury induced proliferation of these stem cells associated with a reduction in β-catenin signaling. Delayed re-epithelialization is accompanied with defects in migration and differentiation of these stem cells, mediated by decreased AJ signaling. cKO: Conditional knockout; cDKO: Conditional double knockout.
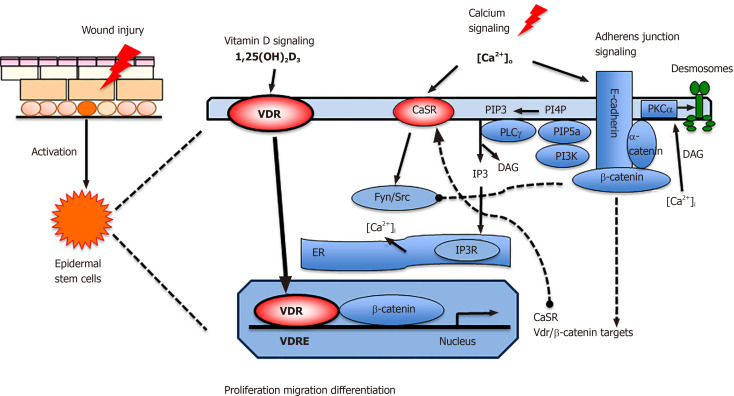
Figure 2.
Proposed model in which vitamin D and calcium signaling mutually regulate β-catenin and AJ signaling essential for wound re-epithelialization. First, vitamin D receptor (VDR) may partner with β-catenin in the nucleus to regulate the expression of β-catenin target genes such as Cyclin D1 to promote proliferation of stem cells when the skin is wounded. Subsequently extracellular calcium [Ca]o in collaboration with VDR stimulates E-cadherin/catenin complex formation to promote keratinocyte differentiation while reducing the proliferative stimulus by sequestering β-catenin in the membrane. The E-cadherin/catenin complex formation is facilitated by the activation of Fyn/Src kinases by the CaSR which phosphorylate the catenins required for their recruitment into the E-cadherin/catenin complex. The E-cadherin/catenin complex not only provides a reservoir of β-catenin in the membrane but also includes a link to the cytoskeleton via a catenin enabling cell migration and differentiation essential for epidermal remodeling during wound re-epithelialization. Moreover, enzymes within the E-cadherin/catenin complex sequentially phosphorylate PIP to PIP3, that activates PLCγ, that in turn hydrolyzes PIP2 to DAG and IP3. The latter stimulates the IP3 receptor in subcellular organelles (ER and Golgi in keratinocytes) to release calcium. DAG, on the other hand along with calcium activates PKCa, the enzyme that activates the AP-1 transcription factors involved in the expression of differentiation markers in keratinocytes as well as phosphorylation of desmoplakin, which alters the desmosomal structure facilitating migration of the keratinocytes to re-epithelialize the wounds. VDR: Vitamin D receptor; CaSR: Calcium sensing receptor; DAG: Diacylglycerol;ER: Endoplasmic reticulum; PLCγ: Phospholipase γ; IP3: Inositol trisphosphate; PIP3: Phosphatidylinositol 3,4,5-trisphosphate; PI3K: Phosphatidyl inositol 3 kinase; PIP5K1a: phosphatidyl inositol 4-phosphate 5-kinase 1α.
The E-cadherin/catenin complex includes α-catenin, that links the complex to the cytoskeleton subsequently enabling cells to migrate. The complex also includes the enzymes phosphatidyl inositol 3 kinase (PI3K) and phosphatidyl inositol 4-phosphate 5-kinase 1α (PIP5K1α)[34,35]. These enzymes sequentially phosphorylate of PIP and PIP2 to PIP3 to activate PLC-γ1 and other signaling molecules such as Akt. PLC-γ1 cleaves PIP2 to form IP3 and DAG; IP3 releases intracellular [Ca]i from intracellular stores, which is essential for the acute response to wounding, and DAG which along with calcium activates PKCα. PKC regulates the activity of AP-1 transcription factors involved in differentiation. Moreover, PKCα phosphorylates desmoplakin, a component of desmosomes, that loosens their intercellular adhesion to enable keratinocyte migration across the wounds[36].
Efficient wound repair is critical for life by restoring the integrity of the skin to prevent the invasion by infectious organisms and other harmful materials and the loss of body fluids. Understanding the mechanisms by which the SC populations in the skin respond to wounding should lead to better therapies to promote more efficient healing. Examining the role of vitamin D and calcium signaling in this process is an important step in this direction. Moreover, our studies address the more general question of tissue regeneration, both pathologic as in chronic wounds and physiologic as in normal wound repair in the skin and other tissues. The roles of β-catenin and E-cadherin signaling in SC as they are regulated by vitamin D and calcium during the response to wounding and epidermal remodeling are central to the wounding response. Further study of the shift in the transcriptional profile during wounding as affected by deletion of Vdr and/or Casr is likely to reveal a better understanding of the molecular mechanisms by which VDR and CaSR sequentially regulate the different aspects of the wounding response with the potential that these results will lead to new approaches to treatment of chronic wounds.
These studies have clinical significance as vitamin D deficiency is linked with poor wound healing[2,3]. Improved wound healing of patients with diabetic foot ulcers with vitamin D supplementation compared to placebo is supported by a randomized clinical trial of oral vitamin D supplementation[37]. Likewise, calcium alginate dressings are superior to other wound care products[38] indicating the clinical role of calcium signaling in wound repair. Like vitamin D signaling, calcium signaling is expected to play an important role for activation, migration and differentiation of the SCs regenerating the epidermis.
CONCLUSION
In summary, we have discussed the role of vitamin D and calcium signaling in epidermal SCs and progeny essential for normal wound re-epithelialization in the epidermis. We propose that vitamin D and calcium promote wound re-epithelialization, through both β-catenin and AJ signaling.
Footnotes
Conflict-of-interest statement: Authors declare no conflict of interests for this article.
Manuscript source: Invited manuscript
Peer-review started: February 25, 2020
First decision: April 22, 2020
Article in press: June 10, 2020
Specialty type: Cell and tissue engineering
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Lalli E S-Editor: Gong ZM L-Editor: A E-Editor: Xing YX
Contributor Information
Yuko Oda, Department of Medicine, University of California San Francisco, CA 94158, United States; Endocrine Research, Veterans Affairs Medical Center San Francisco, CA 94158, United States. yuko.oda@ucsf.edu.
Daniel D Bikle, Department of Medicine, University of California San Francisco, CA 94158, United States; Endocrine Research, Veterans Affairs Medical Center San Francisco, CA 94158, United States.
References
- 1.Sen CK, Gordillo GM, Roy S, Kirsner R, Lambert L, Hunt TK, Gottrup F, Gurtner GC, Longaker MT. Human skin wounds: a major and snowballing threat to public health and the economy. Wound Repair Regen. 2009;17:763–771. doi: 10.1111/j.1524-475X.2009.00543.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Burkievcz CJ, Skare TL, Malafaia O, Nassif PA, Ribas CS, Santos LR. Vitamin D deficiency in patients with chronic venous ulcers. Rev Col Bras Cir. 2012;39:60–63. [PubMed] [Google Scholar]
- 3.Zubair M, Malik A, Meerza D, Ahmad J. 25-Hydroxyvitamin D [25(OH)D] levels and diabetic foot ulcer: is there any relationship? Diabetes Metab Syndr. 2013;7:148–153. doi: 10.1016/j.dsx.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 4.Mascré G, Dekoninck S, Drogat B, Youssef KK, Broheé S, Sotiropoulou PA, Simons BD, Blanpain C. Distinct contribution of stem and progenitor cells to epidermal maintenance. Nature. 2012;489:257–262. doi: 10.1038/nature11393. [DOI] [PubMed] [Google Scholar]
- 5.Lim X, Tan SH, Koh WL, Chau RM, Yan KS, Kuo CJ, van Amerongen R, Klein AM, Nusse R. Interfollicular epidermal stem cells self-renew via autocrine Wnt signaling. Science. 2013;342:1226–1230. doi: 10.1126/science.1239730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jensen KB, Collins CA, Nascimento E, Tan DW, Frye M, Itami S, Watt FM. Lrig1 expression defines a distinct multipotent stem cell population in mammalian epidermis. Cell Stem Cell. 2009;4:427–439. doi: 10.1016/j.stem.2009.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Plikus MV, Gay DL, Treffeisen E, Wang A, Supapannachart RJ, Cotsarelis G. Epithelial stem cells and implications for wound repair. Semin Cell Dev Biol. 2012;23:946–953. doi: 10.1016/j.semcdb.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ito M, Liu Y, Yang Z, Nguyen J, Liang F, Morris RJ, Cotsarelis G. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat Med. 2005;11:1351–1354. doi: 10.1038/nm1328. [DOI] [PubMed] [Google Scholar]
- 9.Levy V, Lindon C, Zheng Y, Harfe BD, Morgan BA. Epidermal stem cells arise from the hair follicle after wounding. FASEB J. 2007;21:1358–1366. doi: 10.1096/fj.06-6926com. [DOI] [PubMed] [Google Scholar]
- 10.Langton AK, Herrick SE, Headon DJ. An extended epidermal response heals cutaneous wounds in the absence of a hair follicle stem cell contribution. J Invest Dermatol. 2008;128:1311–1318. doi: 10.1038/sj.jid.5701178. [DOI] [PubMed] [Google Scholar]
- 11.Bikle DD. Vitamin D metabolism and function in the skin. Mol Cell Endocrinol. 2011;347:80–89. doi: 10.1016/j.mce.2011.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tian XQ, Chen TC, Holick MF. 1,25-dihydroxyvitamin D3: a novel agent for enhancing wound healing. J Cell Biochem. 1995;59:53–56. doi: 10.1002/jcb.240590107. [DOI] [PubMed] [Google Scholar]
- 13.Lien WH, Polak L, Lin M, Lay K, Zheng D, Fuchs E. In vivo transcriptional governance of hair follicle stem cells by canonical Wnt regulators. Nat Cell Biol. 2014;16:179–190. doi: 10.1038/ncb2903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Choi YS, Zhang Y, Xu M, Yang Y, Ito M, Peng T, Cui Z, Nagy A, Hadjantonakis AK, Lang RA, Cotsarelis G, Andl T, Morrisey EE, Millar SE. Distinct functions for Wnt/β-catenin in hair follicle stem cell proliferation and survival and interfollicular epidermal homeostasis. Cell Stem Cell. 2013;13:720–733. doi: 10.1016/j.stem.2013.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shah S, Islam MN, Dakshanamurthy S, Rizvi I, Rao M, Herrell R, Zinser G, Valrance M, Aranda A, Moras D, Norman A, Welsh J, Byers SW. The molecular basis of vitamin D receptor and beta-catenin crossregulation. Mol Cell. 2006;21:799–809. doi: 10.1016/j.molcel.2006.01.037. [DOI] [PubMed] [Google Scholar]
- 16.Pálmer HG, Anjos-Afonso F, Carmeliet G, Takeda H, Watt FM. The vitamin D receptor is a Wnt effector that controls hair follicle differentiation and specifies tumor type in adult epidermis. PLoS One. 2008;3:e1483. doi: 10.1371/journal.pone.0001483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lisse TS, Saini V, Zhao H, Luderer HF, Gori F, Demay MB. The vitamin D receptor is required for activation of cWnt and hedgehog signaling in keratinocytes. Mol Endocrinol. 2014;28:1698–1706. doi: 10.1210/me.2014-1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cianferotti L, Cox M, Skorija K, Demay MB. Vitamin D receptor is essential for normal keratinocyte stem cell function. Proc Natl Acad Sci USA. 2007;104:9428–9433. doi: 10.1073/pnas.0702884104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Brown EM, Gamba G, Riccardi D, Lombardi M, Butters R, Kifor O, Sun A, Hediger MA, Lytton J, Hebert SC. Cloning and characterization of an extracellular Ca(2+)-sensing receptor from bovine parathyroid. Nature. 1993;366:575–580. doi: 10.1038/366575a0. [DOI] [PubMed] [Google Scholar]
- 20.Oda Y, Tu CL, Pillai S, Bikle DD. The calcium sensing receptor and its alternatively spliced form in keratinocyte differentiation. J Biol Chem. 1998;273:23344–23352. doi: 10.1074/jbc.273.36.23344. [DOI] [PubMed] [Google Scholar]
- 21.Tu CL, Oda Y, Komuves L, Bikle DD. The role of the calcium-sensing receptor in epidermal differentiation. Cell Calcium. 2004;35:265–273. doi: 10.1016/j.ceca.2003.10.019. [DOI] [PubMed] [Google Scholar]
- 22.Tu CL, Chang W, Xie Z, Bikle DD. Inactivation of the calcium sensing receptor inhibits E-cadherin-mediated cell-cell adhesion and calcium-induced differentiation in human epidermal keratinocytes. J Biol Chem. 2008;283:3519–3528. doi: 10.1074/jbc.M708318200. [DOI] [PubMed] [Google Scholar]
- 23.Tu CL, Crumrine DA, Man MQ, Chang W, Elalieh H, You M, Elias PM, Bikle DD. Ablation of the calcium-sensing receptor in keratinocytes impairs epidermal differentiation and barrier function. J Invest Dermatol. 2012;132:2350–2359. doi: 10.1038/jid.2012.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ratnam AV, Bikle DD, Cho JK. 1,25 dihydroxyvitamin D3 enhances the calcium response of keratinocytes. J Cell Physiol. 1999;178:188–196. doi: 10.1002/(SICI)1097-4652(199902)178:2<188::AID-JCP8>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 25.Bikle DD, Ng D, Tu CL, Oda Y, Xie Z. Calcium- and vitamin D-regulated keratinocyte differentiation. Mol Cell Endocrinol. 2001;177:161–171. doi: 10.1016/s0303-7207(01)00452-x. [DOI] [PubMed] [Google Scholar]
- 26.Bikle DD, Xie Z, Tu CL. Calcium regulation of keratinocyte differentiation. Expert Rev Endocrinol Metab. 2012;7:461–472. doi: 10.1586/eem.12.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li L, Hartley R, Reiss B, Sun Y, Pu J, Wu D, Lin F, Hoang T, Yamada S, Jiang J, Zhao M. E-cadherin plays an essential role in collective directional migration of large epithelial sheets. Cell Mol Life Sci. 2012;69:2779–2789. doi: 10.1007/s00018-012-0951-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li YC, Pirro AE, Amling M, Delling G, Baron R, Bronson R, Demay MB. Targeted ablation of the vitamin D receptor: an animal model of vitamin D-dependent rickets type II with alopecia. Proc Natl Acad Sci U S A. 1997;94:9831–9835. doi: 10.1073/pnas.94.18.9831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Luderer HF, Nazarian RM, Zhu ED, Demay MB. Ligand-dependent actions of the vitamin D receptor are required for activation of TGF-β signaling during the inflammatory response to cutaneous injury. Endocrinology. 2013;154:16–24. doi: 10.1210/en.2012-1579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Oda Y, Hu L, Nguyen T, Fong C, Zhang J, Guo P, Bikle DD. Vitamin D Receptor Is Required for Proliferation, Migration, and Differentiation of Epidermal Stem Cells and Progeny during Cutaneous Wound Repair. J Invest Dermatol. 2018;138:2423–2431. doi: 10.1016/j.jid.2018.04.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Oda Y, Tu CL, Menendez A, Nguyen T, Bikle DD. Vitamin D and calcium regulation of epidermal wound healing. J Steroid Biochem Mol Biol. 2016;164:379–385. doi: 10.1016/j.jsbmb.2015.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Oda Y, Hu L, Nguyen T, Fong C, Tu CL, Bikle DD. Combined Deletion of the Vitamin D Receptor and Calcium-Sensing Receptor Delays Wound Re-epithelialization. Endocrinology. 2017;158:1929–1938. doi: 10.1210/en.2017-00061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tu CL, Celli A, Mauro T, Chang W. Calcium-Sensing Receptor Regulates Epidermal Intracellular Ca2+ Signaling and Re-Epithelialization after Wounding. J Invest Dermatol. 2019;139:919–929. doi: 10.1016/j.jid.2018.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xie Z, Chang SM, Pennypacker SD, Liao EY, Bikle DD. Phosphatidylinositol-4-phosphate 5-kinase 1alpha mediates extracellular calcium-induced keratinocyte differentiation. Mol Biol Cell. 2009;20:1695–1704. doi: 10.1091/mbc.E08-07-0756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xie Z, Bikle DD. The recruitment of phosphatidylinositol 3-kinase to the E-cadherin-catenin complex at the plasma membrane is required for calcium-induced phospholipase C-gamma1 activation and human keratinocyte differentiation. J Biol Chem. 2007;282:8695–8703. doi: 10.1074/jbc.M609135200. [DOI] [PubMed] [Google Scholar]
- 36.Thomason HA, Cooper NH, Ansell DM, Chiu M, Merrit AJ, Hardman MJ, Garrod DR. Direct evidence that PKCα positively regulates wound re-epithelialization: correlation with changes in desmosomal adhesiveness. J Pathol. 2012;227:346–356. doi: 10.1002/path.4016. [DOI] [PubMed] [Google Scholar]
- 37.Razzaghi R, Pourbagheri H, Momen-Heravi M, Bahmani F, Shadi J, Soleimani Z, Asemi Z. The effects of vitamin D supplementation on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J Diabetes Complications. 2017;31:766–772. doi: 10.1016/j.jdiacomp.2016.06.017. [DOI] [PubMed] [Google Scholar]
- 38.Brenner M, Hilliard C, Peel G, Crispino G, Geraghty R, OʼCallaghan G. Management of pediatric skin-graft donor sites: a randomized controlled trial of three wound care products. J Burn Care Res. 2015;36:159–166. doi: 10.1097/BCR.0000000000000161. [DOI] [PubMed] [Google Scholar]