Abstract
The efficacy of a UV-A light emitting diode system (LED) to reduce the concentrations of aflatoxin B1, aflatoxin M1 (AFB1, AFM1) in pure water was studied. This work investigates and reveals the kinetics and main mechanism(s) responsible for the destruction of aflatoxins in pure water and assesses the cytotoxicity in liver hepatocellular cells. Irradiation experiments were conducted using an LED system operating at 365 nm (monochromatic wave-length). Known concentrations of aflatoxins were spiked in water and irradiated at UV-A doses ranging from 0 to 1,200 mJ/cm2. The concentration of AFB1 and AFM1 was determined by HPLC with fluorescence detection. LC–MS/MS product ion scans were used to identify and semi-quantify degraded products of AFB1 and AFM1. It was observed that UV-A irradiation significantly reduced aflatoxins in pure water. In comparison to control, at dose of 1,200 mJ/cm2 UV-A irradiation reduced AFB1 and AFM1 concentrations by 70 ± 0.27 and 84 ± 1.95%, respectively. We hypothesize that the formation of reactive species initiated by UV-A light may have caused photolysis of AFB1 and AFM1 molecules in water. In cell culture studies, our results demonstrated that the increase of UV-A dosage decreased the aflatoxins-induced cytotoxicity in HepG2 cells, and no significant aflatoxin-induced cytotoxicity was observed at UV-A dose of 1,200 mJ/cm2. Further results from this study will be used to compare aflatoxins detoxification kinetics and mechanisms involved in liquid foods such as milk and vegetable oils.
Subject terms: Biochemistry, Microbiology, Environmental sciences, Diseases, Engineering, Optics and photonics
Introduction
Filamentous fungi invading various feed crops produces toxic secondary metabolites called mycotoxins which possess a serious threat to consumer health1. Aflatoxins are highly cytotoxic and carcinogenic secondary metabolites, produced predominantly by Aspergillus flavus and Aspergillus paraciticus, especially in the tropical and subtropical regions as hot and humid climatic conditions are optimal for mold growth and toxin production2,3. Aflatoxins are difuranocoumarin derivatives formed from the polyketide pathway and include a group of 17 aflatoxins, among which AFB1 is the predominant and is highly toxic4. Aflatoxin contamination in animal feed results in their carry-over in foods of animal origin such as eggs and milk. The major AFB1 metabolite in milk, AFM1, is formed due to the action of hepatic cytochrome P450-dependent polysubstrate monoxygenase enzyme superfamily as a result of the hydroxylation of the fourth carbon of the terminal furan ring. In dairy cows, the biotransformation rate of AFB1 to AFM1 ranges from 0.3 to 6.2%5,6.
Aflatoxins are detrimental upon ingestion, inhalation, and skin contact; and the consequences of aflatoxin infection are collectively called aflatoxicosis. The biotransformation of AFB1 in liver results in the production of AFB1-8,9-epoxide which is highly related to the incidence of hepatocellular carcinoma6,7. The carcinogenicity of AFM1 is approximately one-tenth of that of AFB1 The International Agency for Research on Cancer in 1993 classified AFB1 under Group 1 carcinogen and AFM1 under group 2B carcinogen8; therefore, some international organizations have enacted stringent regulations to mitigate aflatoxin contamination in food and feed. The maximum residue limit (MRL) of aflatoxins for human consumption ranges from 4 to 30 µg/kg9. The European Union has the strictest standards, the AFB1 level should not exceed 2 µg/kg in edible oils and AFM1 levels should not exceed 0.05 µg/kg9. The United States Food and Drug Administration specified maximum acceptable limits of 20 µg/kg for total aflatoxins and 0.5 µg/kg for AFM1 in human food and milk9,10.
Aflatoxin contamination in food and feed can be reduced by various processing methods. For example, aflatoxin contamination during growth and storage of grains can be reduced by employing various pre- and post-harvest technologies. Several aflatoxin decontamination methods include destruction by physical methods including heating at high temperatures; selective separation using adsorbents such as reduced graphene-oxide-gold nanoparticles11; chemical modification using several acids, bases, and oxidizing agents; and biological decontamination using enzymes and fermentation1. Despite their efficacy, each method presents certain challenges such as utilization of chemicals, adverse impacts on the nutritional and sensorial attributes of the food, and difficulties in scale-up, thereby limiting their use in food industry12. Exploration of safe, cost-effective, and novel food processing technologies with an objective of achieving maximum inactivation of aflatoxins with minimal effect on the quality of food helps to address these challenges13. Examples of alternative innovative food processing technologies include electromagnetic irradiation, advanced-packaging materials, and dielectric heating1. Pulsed light technology, which effectively degrade aflatoxins, utilizes broad spectra of white light, that includes ultra-violet, visible and infra-red14. Studies show that the light intensity and the UV spectrum greatly influence the degradation of aflatoxins more than the visible and infra-red spectrum15.
UV irradiation has been demonstrated in literature as an effective physical method to inactivate chemical contaminants, and micro-organisms through photolysis and DNA damage, respectively16–20. This non-thermal technology is efficient in degrading aflatoxins because of their photosensitivity21. Dominant sources of UV treatment such as low pressure and medium pressure mercury lamps were used to degrade aflatoxins. In a study, 98% reduction of AFB1 in water was observed at an UV dose of 4,880 mJ/cm2 using a medium pressure UV lamp which emits irradiation between 200 and 360 nm wavelength16. Similarly, 100% reduction of AFB1 in peanut oil was observed when UV intensity (220–400 nm) of 800 mJ/cm2 was used for 30 min22. In a separate study, irradiation at 365 nm showed nearly 100% degradation of AFM1 in milk after 60 min of exposure to 100 W lamp23. As an alternative to mercury containing traditional UV lamps, alternative sources of UV light such as UV-LEDs and excimer lamps are being studied for their application in food industry as they have several advantages like mercury free, high energy efficiency, constant light intensity and prolonged lifetime24,25.
The presence of mycotoxins in food and feed has been investigated extensively. But few studies reported the occurrence of mycotoxins in surface, ground and wastewaters due to contamination from agricultural fields26,27. Paterson et al., in 1997 first detected the presence of aflatoxins in cold storage water tank28. Aflatoxin B2 was the most often detected mycotoxin present in bottled water followed by aflatoxin B1, aflatoxin G1 and ochratoxin A29. Even though, the levels of aflatoxins found in water is low (ng/L), long term exposure may cause health risk. Until now, no reports are available on UV degradation of aflatoxins in pure water. AFB1 and AFM1 have absorption maxima at 362 nm which elevate their susceptibility for degradation when exposed to light around 362 nm30. Hence, we hypothesize irradiation of aflatoxins using UV-A light at 365 nm could be efficient to degrade aflatoxins in water via photo irradiation. The key points related to application of UVA light in aflatoxin reduction and detoxification include the analysis of kinetics, quantum yield and understanding the cytotoxic behavior of AFB1 and AFM1 degradation products for liver cells. Furthermore, a major issue in many UV studies is that they do not account the absorbance of the test fluid22,23; hence, the present study rectifies this problem by accounting for fluid optics, and corrections for UV fluence gradients.
In this study, a custom-built laboratory scale batch reactor using an UV-A LED source which emits at a peak wavelength of 365 nm was used. This study is carried out to determine the degradation kinetics and the possible degradation mechanism of AFB1 and AFM1 in a pure water, without the hindrance of other biomolecules which may result in UV-A attenuation. This study also assesses the cytotoxicity of UV-A treated samples which contain residual AFB1 and AFM1 and their degradation products in water using human hepatoma cell line (HepG2).
Material and methods
Chemicals and reagents
AFB1 and AFM1 were procured from LKT laboratories, Inc (St. Paul MN, USA). Human hepatoma cells (HepG2; ATCC HB-8065), Eagel’s minimum Essential Medium (EMEM; ATCC 30-2003), and fetal bovine serum (FBS; ATCC 30-2020) was purchased from American Type Culture Collection (ATCC, Manassas, VA).
Preparation of Aflatoxins standards in ultrapure water
Aflatoxin standard solutions were prepared by dissolving AFB1 and AFM1 in methanol. Working solutions of AFB1 and AFM1 with initial concentrations of 1 µg/mL and 2 µg/mL, respectively were prepared in ultrapure water before being exposed to UV-A irradiation. As methanol is found to be toxic to cells, for cytotoxicity studies, AFB1 and AFM1 were dissolved in DMSO, each with initial concentration of 25 µg/mL.
Light emitting diodes irradiation system
A UV-A LED (IRTRONIX, Torrence, CA, USA) which emits at a peak wavelength of 365 nm, mounted on top of a quasi-collimated bench scale reactor, was used to perform the irradiation experiments. Five mL of the test solution was dispensed in a 10 mL beaker and placed above the magnetic stirrer. The test solution was continuously stirred to ensure uniform dose distribution and cold water (4 °C) was continuously circulated to prevent increase in temperature during irradiation at higher doses. Central irradiance incident on the surface of the test solution was measured with the help of a high sensitivity spectrometer (QE Pro series, Ocean Optics, Dunedin, FL, USA). Average irradiance was calculated by taking into account the absorption of the test solution at 365 nm. The absorbance was determined using a double beam Cary100 Spectrophotometer connected to a 6-inch single integrating sphere (Agilent Technology, Santa Clara, CA, USA). The volume-averaged irradiance was evaluated by incorporating corrections factors (reflection factor, petri factor, divergence factor, water factor) as per the standard method described by Bolton and Linden31. Average irradiance was divided by target UV-A dose to obtain specific exposure times. Total UV-A doses of 0, 300, 600, 900 and 1,200 mJ/cm2 were delivered to AFB1 and AFM1 test solutions and each treatment was done in triplicates in a randomized order.
| 1 |
| 2 |
where ‘Incident fluence’ is the incident irradiance at the surface of the liquid, ‘a’ is the absorption coefficient per cm at 365 nm, d is the depth of fluid in the beaker, and ‘L’ is the distance from center of lamp source to the lower meniscus of the surface of the liquid.
HPLC analysis of AFB1 and AFM1
Separation and quantification of irradiated AFB1, AFM1 and their degradation products were carried out using a HPLC System (Shimadzu Scientific Instruments, Columbia, MD, USA) following the method described by Patras et al.16 with slight modification. A reversed-phase C18 column (Phenomenex, CA, USA) with configuration 150 mm × 4.6 mm 2.6 µm, maintained at 37 °C was used as stationary phase. Separation was achieved with the mobile phase consisting of water/acetonitrile/methanol in the ratio of 120:75:30, under isocratic flow mode with a flow rate of 1 mL/min. Aflatoxins were detected using a Shimadzu RF-20A fluorescence detector with the excitation and emission wavelengths set at 365 and 450 nm respectively. The calibrated concentration for the HPLC method validation of AFB1 and AFM1 ranged between 0.25 and 1.0 µg/mL, 0.5 and 2.0 µg/mL for AFB1 and AFM1, respectively.
Aflatoxins degradation analysis LC–MS/MS
The identification of Aflatoxin B1, M1 and the respective degraded products was carried out with an LCMS method using a Shimadzu Prominence XR UHPLC system connected to a Shimadzu LCMS 8040 triple-stage quadrupole mass spectrometer with a chromatographic and mass spectrometric method described previously16. The column used for separation was a Phenomenex Kinetex 2.6 C18 column (50 × 2.1 mm, 2.6 µm). The injection volume was 5 µL. Control and experimental samples were injected, and data acquired in the scan mode to search for degraded products. Once degraded products were identified, corresponding selected ion monitoring (SIM) MS methods were developed for monitoring the parent and degraded product ions: AFB1 (m/z 313), AFB1 degraded products (m/z 303 and m/z 331), AFM1 (m/z 329), and AFM1 degraded products (m/z 347). The dwell time was 10 ms for all SIM events. Control and experimental samples were injected and analyzed. The chromatographic peak areas were determined and compared to controls with Shimadzu LabSolutions V5.89 software (Shimadzu, Columbia, MD, USA).
Cell cytotoxicity analysis
Cell cytotoxicity analysis was carried out using the method described by Patras et al.16 with slight modification. The HepG2 cells (American Type Culture Collection (ATCC); HB-8065) with a cell plating density of 2 × 105 cells per well were seeded in a 12-well plate containing 10% (v/v) fetal bovine serum (FBS) in Eagle’s minimal essential medium (EMEM). After 24 h, the cells were washed thrice with PBS and serum starved overnight in EMEM containing 1% FBS. Following serum starvation, the cells containing 1 mL media were exposed to 10% (v/v) of untreated and treated AFB1 and AFM1 test solutions at a final concentration of 2.5 µg/mL. After 48 h, the cells were measured for viability using XTT assay (ATCC, Manassas, VA) as per the manufacturer’s protocol.
Kinetic modelling and data analysis
Log-linear reduction model available in the GInaFiT tool (a freeware add-in for Microsoft Excel)32 was used to describe the UV-A degradation kinetics of aflatoxins (B1 and M1). This model provides good fit to data in which the inactivation exhibits first order kinetics and goodness of fit parameters including R2, root mean square error, and rate constants were evaluated. The model is given in the following equation,
| 3 |
where ‘C’ is the initial concentration of aflatoxin, ‘C0’ is the concentration of aflatoxin at dose D, ‘k’ is the degradation rate constant and D is the UV-A dose delivered.
For identification purposes, the expression was reformulated as,
| 4 |
A balanced design with three replicates for each treatment was exposed to the selected UV-A treatment. Each sample was independent and assigned randomly to a treatment. One-way ANOVA with Tukey's HSD multiple comparison tests were performed to assess the effects of UV-A in SAS statistical computing environment (SAS, 2016). Data are presented as means ± standard deviation from the mean. Statistical significance was tested at 5 percent significance level.
Results and discussion
In this study, a novel UV-A LED which emits with the peak wavelength of 365 nm was used. The UV-A LED system employed in this study emits peak irradiation at 365 nm. Table 1 shows the characteristics (optical properties) of the test solutions prior to UV-A irradiation. The absorbance of AFB1 and AFM1 were 0.094 and 0.101/cm and ultraviolet transmittance (%) was calculated as 80.5 and 79.3/cm.
Table 1.
Optical properties and treatment parameters of AFB1 and AFM1 in ultrapure water under UV-A radiation.
| Parameters | Aflatoxin B1 | Aflatoxin M1 |
|---|---|---|
| Irradiance (mW/cm2) | 10.26 | 10.26 |
| Absorbance (Au/cm) | 0.094 | 0.101 |
| Transmittance (%T/cm) | 80.5 | 79.250 |
| Exposure time (s) | 39, 78, 117, 156 | 39, 79, 118, 157 |
| Delivered dose (mJ/cm2) | 300, 600, 900, 1,200 | 300, 600, 900, 1,200 |
The absorption spectrum of AFB1 and AFM1 observed in the ultraviolet region of the electromagnetic spectrum and the relative emission spectra of the lamp are shown in Fig. 1. The data show AFB1 and AFM1 strongly absorbs UV-A irradiation at 362 nm (Fig. 1a). The peak emission of the UV-A light source was noticed at 365 nm (Fig. 1b).
Figure 1.
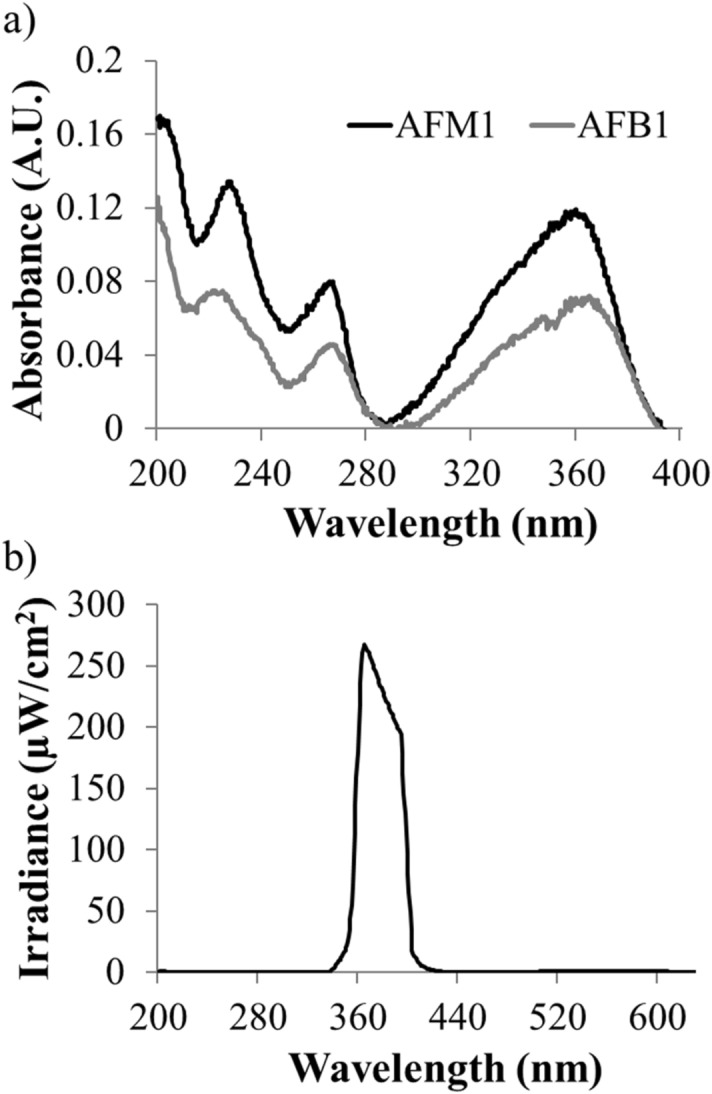
(a) Measured absorption spectra of AFB1 and AFM1 in ultrapure water using Cary100 spectrophotometer (b) Measured spectral irradiance of UV-A LED using Ocean optics QE Pro spectrometer equipped with UV–visible optical fiber.
UV-A degradation kinetics of AFB1 and AFM1
In this study, Aflatoxins solutions AFB1 and AFM1 in ultrapure water were irradiated at different UV-A doses ranging from 0 to 1,200 mJ/cm2. UV-A degradation of AFB1 (C0 = 0.997 ppm) and AFM1 (C0 = 2 ppm) in ultrapure water are compared in Fig. 2.
Figure 2.
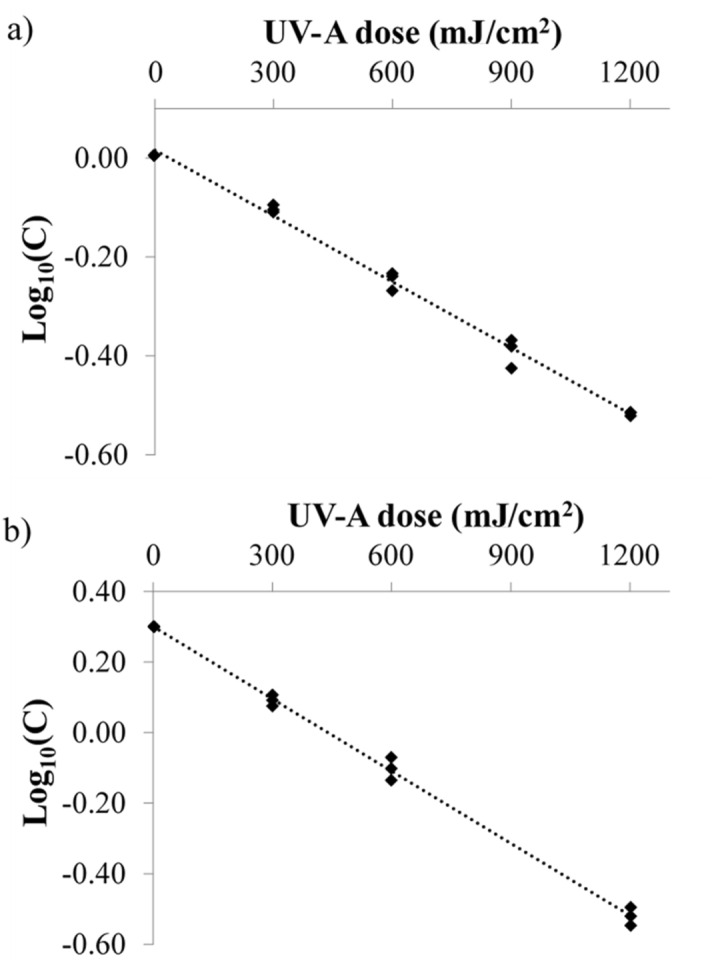
Degradation kinetics of (a) AFB1 (b) AFM1 in ultrapure water at different UV-A dose levels.
The degradation reaction followed first-order kinetics for AFB1 and AFM1 and both were reduced by more than 70%. At UV-A doses of 300, 600, 900 and 1,200 mJ/cm2 AFB1 reduced by 22.0 ± 1.32, 43.9 ± 2.34, 59.6 ± 2.66 and 70.0 ± 0.27 percent respectively. Similarly, a reduction of 36.0 ± 2.70, 57.5 ± 4.10, 76.3 ± 0.90, 84.0 ± 1.95 percent of AFM1 was observed at UV-A doses 300, 600, 900 and 1,200 mJ/cm2 respectively. Log10(C) is plotted against the UV-A doses delivered. Table 2 shows the kinetic parameters of the irradiation experiment. Log linear trend was observed which indicates that the degradation of AFB1 and AFM1 in ultrapure water under UV-A irradiation follows first-order kinetics (R2 > 0.99), given by the Eq. (4). The kinetic rate constant for AFB1 degradation in water was 0.001 cm2/mJ, which is ~ 1.6 times lesser than the kinetic constants for AFM1 (0.0016 cm2/mJ).
Table 2.
Kinetic parameters of AFB1 and AFM1 under UV-A irradiation.
| Parameters | Aflatoxin B1 | Aflatoxin M1 |
|---|---|---|
| R2 | 0.99 | 0.99 |
| Kmax (cm2/mJ) | 0.001 | 0.0016 |
| Half-life (mJ/cm2) | 693 | 433 |
| Quantum yield | 0.0043 | 0.0057 |
Quantum yield is a fundamental photochemical parameter, which explains the photochemical fate of the compound, under controlled conditions where light absorption and change in target compound concentration are properly quantified33,34. Quantum yield is the number of defined events occurring per photon absorbed by the system35. For a photochemical reaction, quantum yield ɸ(λ) at a wavelength λ is given by,
| 5 |
Einsteins absorbed during the UV-A treatment is easily obtained by the following equation;
The energy of a single photon is quantified by the following law:
| 6 |
where h = Planck constant (6.63 × 10−34 J s), c = light speed (3 × 108 m/s), λ = wavelength (m). In our case
| 7 |
Absorbed energy is given by:
| 8 |
| 9 |
Power absorbed is obtained by following equation:
| 10 |
where is surface irradiance (W/m2); is the area (m2), is the absorbance value at 365 nm (base10/m), is the fluid depth (m).
The quantum yields of AFB1 and AFM1 in ultrapure water were determined based on Table 3.
Table 3.
Quantum yield calculation of AFB1 and AFM1 in ultrapure water under UV-A radiation.
| UV-A dose (mJ/cm2) | Irradiance (W/m2) | Treatment time (s) | Absorbed energy (J/m3) | Einsteins absorbed (E/m3) | Aflatoxin (mol/m3) | Quantum yield |
|---|---|---|---|---|---|---|
| AFB1 | ||||||
| 300 | 102.6 | 39 | 79,236 | 0.24 | 0.0025 | 0.0105 |
| 600 | 102.6 | 78 | 158,473 | 0.48 | 0.0018 | 0.0038 |
| 900 | 102.6 | 117 | 237,709 | 0.73 | 0.0013 | 0.0018 |
| 1,200 | 102.6 | 156 | 316,945 | 0.97 | 0.0010 | 0.0010 |
| Average | 0.0043 | |||||
| AFM1 | ||||||
| 300 | 102.6 | 39 | 84,171 | 0.26 | 0.0032 | 0.0039 |
| 600 | 102.6 | 79 | 170,500 | 0.52 | 0.0022 | 0.0026 |
| 900 | 102.6 | 118 | 254,671 | 0.78 | 0.0014 | 0.0014 |
| 1,200 | 102.6 | 157 | 338,842 | 1.03 | 0.0010 | 0.0010 |
| Average | 0.0057 | |||||
The average quantum yields of AFB1 and AFM1 in ultrapure water were found to be 4.25 × 10−3 and 5.74 × 10−3 respectively. The quantum yield of AFM1 in ultrapure water is 1.35 times more than that of the quantum yield of AFM1. Comparing the UV-A photolysis rate constants, and quantum yield, it is found that AFM1 is more susceptible to UV-A photolysis than AFB1.
Under UV-A radiation, AFB1 acts as a photosensitizer and results in the formation of reactive oxygen species through the involvement of triplet excited state, by either Type I or Type II mechanism. In Type I mechanism, electron transfer occurs from triplet aflatoxin to molecular oxygen resulting in the formation of superoxide anion radical. In Type II mechanism, energy is transferred from triplet aflatoxin to molecular oxygen leading to singlet oxygen formation. These reactive species react with the aflatoxins and form oxidized products36. Various authors have studied the degradation kinetics of aflatoxins. For example, Patras et al. observed 98% reduction of AFB1 with a UV dose of 4,880 mJ/cm2 in ultrapure water when irradiated using medium pressure lamp which emits irradiation between 220 and 400 nm16. Similarly, Liu et al. used UV lamp emitting at 220–400 nm with an irradiance of 800µw/cm2 and observed thorough reduction of AFB1 in peanut oil after 30 min of UV exposure, the degradation followed first order kinetics22. Mao et al. used a UV-A lamp (lamp power = 100 W; irradiance = 55–60 mW cm2) which emits at 365 nm to treat AFB1 in peanut oil, the authors observed ≈96% reduction after 30 min of UV-A exposure (UV dose equivalent = 108,000 mJ/cm2)37. Diao et al. tested a continuous flow UV-A reactor (lamp power = 36 W; irradiance = 6.4 mW/cm2; flow rate = 0.55 L/min) to treat AFB1 in peanut oil at 365 nm and observed 88.74% reduction after 40 min of UV-A exposure38. It should be noted that the authors did not report the UV-A doses; a common discrepancy in many studies. From the above studies, it is quite evident that optical based techniques (i.e. irradiation) can degrade the aflatoxins efficiently due to their photosensitivity.
AFB1 and AFM1 degradation products
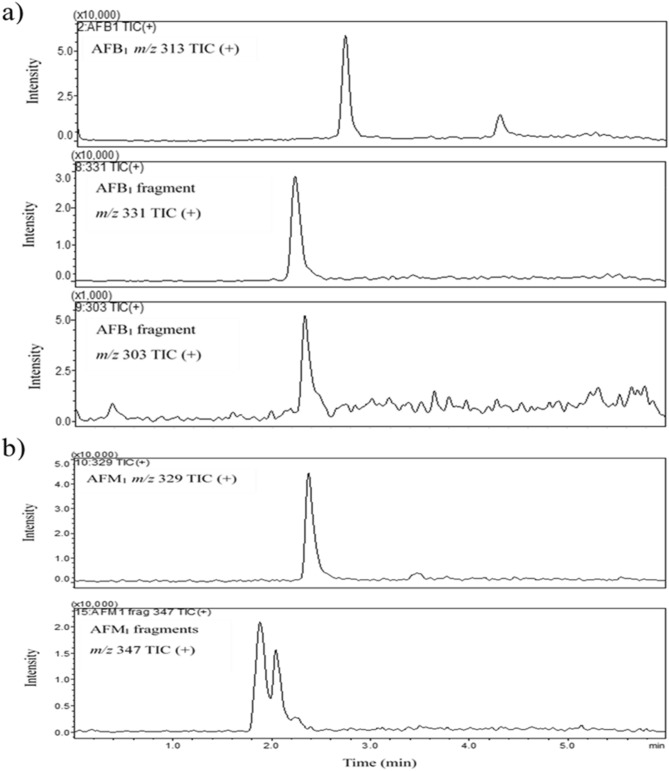
LC–MS scans were used to search for degraded products of AFB1 and AFM1 in water after UV-A irradiation, and are given in Fig. 3.
Figure 3.
LCMS single ion monitoring (SIM) total ion chromatograms (TIC) of (a) AFB1 and (b) AFM1 samples treated at UV-A dose 1,200 mJ/cm2.
LC–MS single ion monitoring (SIM) chromatographic peaks were analyzed as a qualitative approach to approximate the amount of degraded products produced by determining the areas of (SIM) chromatographic peaks. Since authentic standards of the degraded products are not readily available, a quantitative approach is not possible. When compared to controls, LC–MS peak areas for AFB1 and AFM1 decreased and the peak areas of the degraded products increased significantly with increase in the UV-A dose. Representative SIM chromatograms and the LC–MS peak areas for AFB1 and AFM1 are presented in the supplemental information (Figure S1).
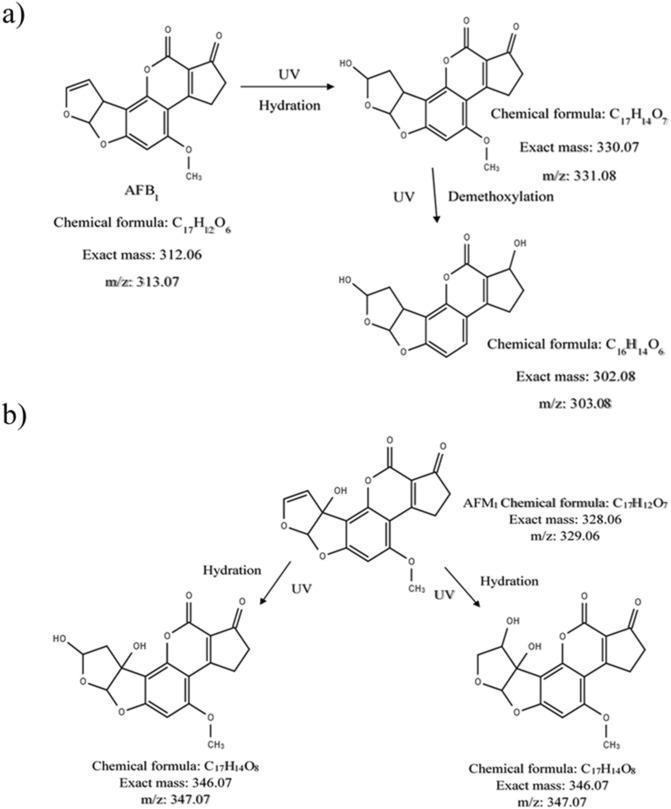
The structure of the degraded products of AFB1 and AFM1 was identified and the possible degradation pathway is proposed based on them, as given by Fig. 4. Two degradation products, P1 and P2 were observed for AFB1 in water. Hydration and demethoxylation were the main fragmentation pathways of AFB1. The double bond equivalence (DBE) of AFB1 is 12. The photolysis product C17H14O7 (P1, m/z 331.08) was the result of hydration on the double bond of terminal furan ring of AFB1, with DBE of 11 which is one less than AFB1. The structure of product C17H14O7 (P1, m/z 331.08) is similar to that of Aflatoxin B2A, whose toxic potential is comparatively less than AFB1, and is inactive with respect to toxicity to ducklings and is non-lethal to chick embryos39,40. Product C16H14O6 (P2) (m/z 303) was the result of hydration on furan ring and demethylation on the side chain of benzene. The DBE of P2 is 10, which is 2 less than AFB1. The AFB1 degraded products had retention times of 2.2 and 2.3 min.
Figure 4.
Proposed UV-A light degradation mechanism of (a) AFB1 and (b) AFM1 in ultrapure water.
Two degraded products with similar chemical formula C17H14O8 (m/z 347.07) were observed for AFM1 and were found to be structural isomers of each other. Hydration was observed to be the main fragmentation pathway which occurred at the double bond of terminal furan ring. The DBE of both the products were found to be 11, which is one less than AFM1 (DBE of AFM1 = 12). The AFM1 degraded products had retention times of 1.8 and 2.0 min. The DBE of all the photolysis products of both AFB1 and AFM1 were found to be less than that of their parent molecules, implying that double bond addition reactions occurred.
The UV-A irradiation results in the formation of reactive species, which react with AFB1 and AFM1 resulting in photolysis. Hence, the structure of all photolysis products formed due to the reaction of free radicals with AFB1 and AFM1 is almost similar to that of their parent molecules41. The double bond in the terminal furan ring and the lactone ring in the coumarin moiety are considered to be the most toxicological sites of AFB142. While AFB1 is not mutagenic, it is bioactivated by undergoing epoxidation of double bond in the furan ring results in the formation of AFB1-8,9-epoxide and it is the key active site for its toxic and carcinogenic activities as the aflatoxin-DNA and the aflatoxin-protein interactions occur6,7. From the proposed structure of the degradation products, it is shown that the double bond in the terminal furan ring is removed in the degradation products of both AFB1 and AFM1. Hence, based on quantitative structure–activity relationships, it is evident that the toxicity of the photolysis products is reduced compared to the toxicity of AFB1 and AFM1.
Cytotoxicity analysis
Aflatoxins are reported to reduce cell survival in various cultured cells, especially HepG2 cells as liver is the target organ of aflatoxins43,44. Several literature studies have demonstrated aflatoxins‐induced oxidative stress damage, and its effect in hepatotoxicity45–47. AFB1 can induce reactive oxygen species (ROS) to cause oxidative stress, also cause genetic alterations prone to DNA damage and alter mitochondrial permeability46,48,49. Liu et al. (2016) reported precise mechanism of AFB1 induced on hepatotoxicity in primary broiler hepatocytes, AFB1 impaired mitochondrial functions by inducing reactive oxygen species and oxidative stress resulting in the activation of caspase-3 and caspase-9-induced apoptosis through mitochondrial signal pathway in addition to maintaining proper redox balance50. On the other hand, AFM1 is a detoxification product of AFB1, and AFM1 showed only 10% of mutagenicity when compared to AFB151. The metabolic fate of AFM1 resulted to be similar to that of AFB1, with the difference that AFM1 represents a poorer substrate for epoxidation, thus explaining the differences in genotoxicity potencies. Moreover, it has been reported that Cytochrome P450 (CYP) activation is not required to AFM1 to exert cytotoxic effects52.
The efficiency of the UV treatment in reducing the toxic potential of AFB1 and AFM1 was studied using in vitro cell culture methods. The cell viability was assessed using XTT [2,3-bis-(2methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5carboxanilide] assay, and the results were given in Fig. 5.
Figure 5.
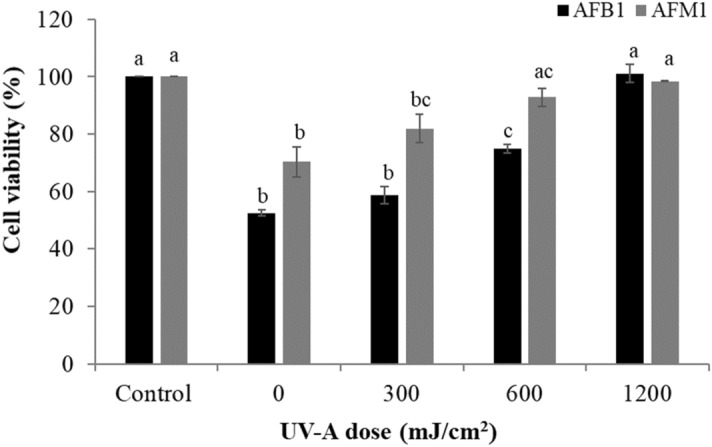
Cytotoxic effect of untreated and UV-A treated ultrapure water consists of (a) AFB1 (b) AFM1 on human hepatoma HepG2 cells. Results are expressed as mean percentage ± SD of two separate experiments. Levels connected by different letters are significantly different at p < 0.05.
The cells were exposed to the test samples for 48 h. With an increase in the UV-A dose from 0 to 1,200 mJ/cm2, the cell viabilities increased from 52.5 ± 1.1 to 101.1 ± 3.2% in the samples treated with AFB1 respectively. Similarly, when the UV-A dose increased from 0 to 1,200 mJ/cm2, the cell viabilities increased from 70.42 ± 5.25 to 98.44 ± 0.25% in the samples treated with AFM1 respectively. The difference in cell viability between the treated and the control samples were found to be statistically significant. As the doses increased from 0 to 1,200 mJ/cm2, there is a significant decrease in the concentration of AFB1 (p value 0.001) and AFM1 (p value 0.0027), and thereby increase in cell viabilities was observed. Also, it is worth mentioning that there is no significant difference between the negative control and the maximum applied dose of 1,200 mJ/cm2 for both AFB1 (p value 0.76) and AFM1 (p value 0.899), clearly demonstrating the efficiency of UV-A degradation.
Conclusion
The current research clearly demonstrated the efficiency of UV-A light in photolysis of AFB1 and AFM1 in ultrapure water. The results show that AFB1 and AFM1 were significantly reduced with increase in UV-A dose. A reduction of 70% and 84% were observed at 1,200 mJ cm−2 respectively. Likewise, cytotoxicity analysis of UV-A treated samples using HepG2 liver cells show increase in cell viability as the dose increases from 0 to 1,200 mJ/cm2 and no cytotoxicity was observed at UV-A dose of 1,200 mJ/cm2. These results confirm that the degraded AFB1 and AFM1 products of UV-A photolysis in water are safe without any effect on cell viability. Overall the results revealed that efficient degradation of AFB1 and AFM1 using UV-A irradiation at 365 nm could be a potential approach to reduce their levels in contaminated water. Due to its lipophilic nature, milk and edible vegetable oils are typically contaminated by aflatoxins hence could be reduced by UV-A light. Further studies will be conducted to assess the efficiency of this technology on reduction of AFB1 and AFM1 levels, quality (nutritional and sensory) and safety of the food products (milk and edible vegetable oils). Since AFB1 and AFM1 have absorbance maxima at 365 nm and milk and edible vegetable oils have absorbance minima at 365 nm, this permits efficient degradation of AFB1 and AFM1 by improving light penetration while potentially having less impact on the nutritional and sensory quality of food. UV-A dose response curves of aflatoxins will be generated using a continous flow UV system, continuous flow reactors are significantly more desirable for industrial food processes.
Supplementary information
Acknowledgements
This project is funded under the Agriculture and Food Research Initiative (Food Safety Challenge Area), USDA, Award Numbers; 2015-69003-23117 and 2018-38821-27732. The authors would like to thank Ms. Judy Stanley for all her dedication and hardwork. She was a remarkable scientist. We will remember her forever.
Author contributions
Conceived and designed the experiments: J.S., A.P. and B.P. Performed the experiments: J.S., B.P., M.J.V., and R.R.B. Analyzed the data: J.S., A.P. B.P., M.J.V., and R.R.B. Wrote the paper: J.S. and B.P. Revised the paper: A.P., B.P., M.J.V., and R.R.B.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ankit Patras, Email: apatras@tnstate.edu.
Brahmaiah Pendyala, Email: bpendyal@tnstate.edu.
Supplementary information
is available for this paper at 10.1038/s41598-020-70370-x.
References
- 1.Pankaj SK, Shi H, Keener KM. A review of novel physical and chemical decontamination technologies for aflatoxin in food. Trends Food Sci. Technol. 2018;71:73–83. doi: 10.1016/j.tifs.2017.11.007. [DOI] [Google Scholar]
- 2.Asao T, et al. The structures of aflatoxins B and G1. J. Am. Chem. Soc. 1965;87:882–886. doi: 10.1021/ja01082a031. [DOI] [PubMed] [Google Scholar]
- 3.Rustom IYS. Aflatoxin in Food and Feed: Occurrence, Legislation and Inactivation by Physical Methods. Food Chemistry. Amsterdam: Elsevier; 1997. [Google Scholar]
- 4.Dhanasekaran D, Shanmugapriya S, Thajuddin N, Panneerselvam A. Aflatoxins and aflatoxicosis in human and animals. Aflatoxins Biochem. Mol. Biol. 2011 doi: 10.5772/22717. [DOI] [Google Scholar]
- 5.Kamkar A, Fallah AA, Mozaffari Nejad AS. The review of aflatoxin M1 contamination in milk and dairy products produced in Iran. Toxin Rev. 2014;33:160–168. doi: 10.3109/15569543.2014.922580. [DOI] [Google Scholar]
- 6.Marin S, Ramos AJ, Cano-Sancho G, Sanchis V. Mycotoxins: occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013;60:218–237. doi: 10.1016/j.fct.2013.07.047. [DOI] [PubMed] [Google Scholar]
- 7.Samarajeewa U, Sen AC, Cohen MD, Wei CI. Detoxification of aflatoxins in foods and feeds by physical and chemical methods. J. Food Prot. 1990;53:489–501. doi: 10.4315/0362-028X-53.6.489. [DOI] [PubMed] [Google Scholar]
- 8.International Agency for Research on Cancer (IARC). Some naturally occurring substances: food items and constituents, heterocyclic aromatic amines and mycotoxins. in Aflatoxins 245–295 (1993).
- 9.Udomkun P, et al. Innovative technologies to manage aflatoxins in foods and feeds and the profitability of application—a review. Food Control. 2017;76:127–138. doi: 10.1016/j.foodcont.2017.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iqbal SZ, Jinap S, Pirouz AA, Ahmad Faizal AR. Aflatoxin M1 in milk and dairy products, occurrence and recent challenges: a review. Trends Food Sci. Technol. 2015;46:110–119. doi: 10.1016/j.tifs.2015.08.005. [DOI] [Google Scholar]
- 11.Guo W, et al. Reduced graphene oxide-gold nanoparticle nanoframework as a highly selective separation material for aflatoxins. Sci. Rep. 2017 doi: 10.1038/s41598-017-15210-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Iram W, Anjum T, Iqbal M, Ghaffar A, Abbas M. Mass spectrometric identification and toxicity assessment of degraded products of aflatoxin B1 and B2 by Corymbia citriodora aqueous extracts. Sci. Rep. 2015;5:14672. doi: 10.1038/srep14672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Prietto L, et al. Post-harvest operations and aflatoxin levels in rice (Oryza sativa) Crop Prot. 2015;78:172–177. doi: 10.1016/j.cropro.2015.09.011. [DOI] [Google Scholar]
- 14.Wang B, et al. Effectiveness of pulsed light treatment for degradation and detoxification of aflatoxin B<inf>1</inf> and B<inf>2</inf> in rough rice and rice bran. Food Control. 2016;59:461–467. doi: 10.1016/j.foodcont.2015.06.030. [DOI] [Google Scholar]
- 15.Moreau M, et al. Application of the pulsed light technology to mycotoxin degradation and inactivation. J. Appl. Toxicol. 2013;33:357–363. doi: 10.1002/jat.1749. [DOI] [PubMed] [Google Scholar]
- 16.Patras A, et al. Effect of UV irradiation on aflatoxin reduction: a cytotoxicity evaluation study using human hepatoma cell line. Mycotoxin Res. 2017;33:343–350. doi: 10.1007/s12550-017-0291-0. [DOI] [PubMed] [Google Scholar]
- 17.Chandra S, et al. Patulin degradation and cytotoxicity evaluation of UV irradiated apple juice using human peripheral blood mononuclear cells. J. Food Process Eng. 2017;40:1–9. doi: 10.1111/jfpe.12586. [DOI] [Google Scholar]
- 18.Gopisetty VVS, et al. Impact of UV-C irradiation on the quality, safety, and cytotoxicity of cranberry-flavored water using a novel continuous flow UV system. LWT. 2018;95:230–239. doi: 10.1016/j.lwt.2018.04.042. [DOI] [Google Scholar]
- 19.Pendyala B, Patras A, Gopisetty VVS, Sasges M, Balamurugan S. Inactivation of bacillus and clostridium spores in coconut water by ultraviolet light. Foodborne Pathog. Dis. 2019;16:704–711. doi: 10.1089/fpd.2019.2623. [DOI] [PubMed] [Google Scholar]
- 20.Pendyala B, Patras A, Ramaswamy R, Gopisetty VVS, Sasges M. Evaluation of UV-C irradiation treatments on microbial safety, ascorbic acid, and volatile aromatics content of watermelon beverage. Food Bioprocess. Technol. 2019 doi: 10.1007/s11947-019-02363-2. [DOI] [Google Scholar]
- 21.Van der Zijden ASM, Koelensmid WB, Boldingh J, Barrett CB, Ord WO, Philp J. Aspergillus flavus and Turkey X disease: isolation in crystalline form of a toxin responsible for Turkey X disease. Nat. Int. J. Sci. 1962;196:1048–1050. [Google Scholar]
- 22.Liu R, et al. Photodegradation of aflatoxin B1 in peanut oil. Eur. Food Res. Technol. 2011;232:843–849. doi: 10.1007/s00217-011-1452-6. [DOI] [Google Scholar]
- 23.Yousef AE, Marth EH. Degradation of aflatoxin M 1 in milk by ultraviolet energy. J. Food Prot. 1985;48:697–698. doi: 10.4315/0362-028X-48.8.697. [DOI] [PubMed] [Google Scholar]
- 24.Koutchma T. Advances in ultraviolet light technology for non-thermal processing of liquid foods. Food Bioprocess. Technol. 2009;2:138–155. doi: 10.1007/s11947-008-0178-3. [DOI] [Google Scholar]
- 25.Antonio-Gutiérrez, O. T., López-Díaz, A. S., López-Malo, A., Palou, E. & Ramírez-Corona, N. UV-C Light for Processing Beverages: Principles, Applications, and Future Trends. Processing and Sustainability of Beverages (Elsevier, 2019). 10.1016/b978-0-12-815259-1.00007-0.
- 26.Gromadzka K, Waśkiewicz A, Goliński P, Świetlik J. Occurrence of estrogenic mycotoxin—Zearalenone in aqueous environmental samples with various NOM content. Water Res. 2009 doi: 10.1016/j.watres.2008.11.042. [DOI] [PubMed] [Google Scholar]
- 27.Laganà A, et al. Analytical methodologies for determining the occurrence of endocrine disrupting chemicals in sewage treatment plants and natural waters. Anal. Chim. Acta. 2004 doi: 10.1016/j.aca.2003.09.020. [DOI] [Google Scholar]
- 28.Paterson RRM, Kelley J, Gallagher M. Natural occurrence of aflatoxins and Aspergillus flavus (Link) in water. Lett. Appl. Microbiol. 1997 doi: 10.1111/j.1472-765X.1997.tb00012.x. [DOI] [PubMed] [Google Scholar]
- 29.Mata AT, et al. Bottled water: analysis of mycotoxins by LC-MS/MS. Food Chem. 2015 doi: 10.1016/j.foodchem.2014.12.088. [DOI] [PubMed] [Google Scholar]
- 30.Jubeen F, Bhatti IA, Khan MZ, Zahoor-Ul-Hassan H, Shahid M. Effect of UVC irradiation on aflatoxins in ground nut (Arachis hypogea) and tree nuts (Juglans regia, prunus duclus and pistachio vera) J. Chem. Soc. Pak. 2012;34:1366–1374. [Google Scholar]
- 31.Bolton JR, Linden KG. Standardization of methods for fluence (UV dose) determination in bench-scale UV experiments. J. Environ. Eng. 2003;129:209–215. doi: 10.1061/(ASCE)0733-9372(2003)129:3(209). [DOI] [Google Scholar]
- 32.Geeraerd AH, Valdramidis VP, Van Impe JF. GInaFiT, a freeware tool to assess non-log-linear microbial survivor curves. Int. J. Food Microbiol. 2005;102:95–105. doi: 10.1016/j.ijfoodmicro.2004.11.038. [DOI] [PubMed] [Google Scholar]
- 33.Reina AC, et al. Photochemical degradation of the carbapenem antibiotics imipenem and meropenem in aqueous solutions under solar radiation. Water Res. 2018;128:61–70. doi: 10.1016/j.watres.2017.10.047. [DOI] [PubMed] [Google Scholar]
- 34.Challis JK, Hanson ML, Friesen KJ, Wong CS. A critical assessment of the photodegradation of pharmaceuticals in aquatic environments: defining our current understanding and identifying knowledge gaps. Environ. Sci. Process. Impacts. 2014;16:672–696. doi: 10.1039/c3em00615h. [DOI] [PubMed] [Google Scholar]
- 35.Bolton JR, Mayor-Smith I, Linden KG. Rethinking the concepts of fluence (UV dose) and fluence rate: the importance of photon-based units—a systemic review. Photochem. Photobiol. 2015;91:1252–1262. doi: 10.1111/php.12512. [DOI] [PubMed] [Google Scholar]
- 36.Netto-Ferreira JC, Heyne B, Scaiano JC. Photophysics and photochemistry of aflatoxins B1 and B 2. Photochem. Photobiol. Sci. 2011;10:1701–1708. doi: 10.1039/c1pp05103b. [DOI] [PubMed] [Google Scholar]
- 37.Mao J, et al. A structure identification and toxicity assessment of the degradation products of aflatoxin B 1 in peanut oil under UV irradiation. Toxins (Basel) 2016;8:332. doi: 10.3390/toxins8110332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Diao E, et al. Safety evaluation of aflatoxin B1 in peanut oil after ultraviolet irradiation detoxification in a photodegradation reactor. Int. J. Food Sci. Technol. 2015;50:41–47. doi: 10.1111/ijfs.12648. [DOI] [Google Scholar]
- 39.Wogan GN, Edwards GS, Newberne PM. Structure-activity relationships in toxicity and carcinogenicity of aflatoxins and analogs. Cancer Res. 1971;31:1936–1942. [PubMed] [Google Scholar]
- 40.Lillehoj EB, Ciegler A. Biological activity of aflatoxin B2a. Appl. Microbiol. 1969;17:516–519. doi: 10.1128/AEM.17.4.516-519.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang F, et al. Structure elucidation and toxicity analyses of the radiolytic products of aflatoxin B 1 in methanol-water solution. J. Hazard. Mater. 2011;192:1192–1202. doi: 10.1016/j.jhazmat.2011.06.027. [DOI] [PubMed] [Google Scholar]
- 42.Lee LS, Dunn JJ, DeLucca AJ, Ciegler A. Role of lactone ring of aflatoxin B1 in toxicity and mutagenicity. Experientia. 1981;37:16–17. doi: 10.1007/BF01965543. [DOI] [PubMed] [Google Scholar]
- 43.Van Vleet TR, Watterson TL, Klein PJ, Coulombe RA. Aflatoxin B 1 alters the expression of p53 in cytochrome p450-expressing human lung cells. Toxicol. Sci. 2006;89:399–407. doi: 10.1093/toxsci/kfj039. [DOI] [PubMed] [Google Scholar]
- 44.Liu Y, Du M, Zhang G. Proapoptotic activity of aflatoxin B1 and sterigmatocystin in HepG2 cells. Toxicol. Rep. 2014;1:1076–1086. doi: 10.1016/j.toxrep.2014.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yener Z, Celik I, Ilhan F, Bal R. Effects of Urtica dioica L. seed on lipid peroxidation, antioxidants and liver pathology in aflatoxin-induced tissue injury in rats. Food Chem. Toxicol. 2009 doi: 10.1016/j.fct.2008.11.031. [DOI] [PubMed] [Google Scholar]
- 46.Shi D, et al. Protective effects of selenium on aflatoxin B1-induced mitochondrial permeability transition, DNA damage, and histological alterations in duckling liver. Biol. Trace Elem. Res. 2015 doi: 10.1007/s12011-014-0189-z. [DOI] [PubMed] [Google Scholar]
- 47.Shi J, et al. Distinct response of the hepatic transcriptome to Aflatoxin B 1 induced hepatocellular carcinogenesis and resistance in rats. Sci. Rep. 2016 doi: 10.1038/srep31898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Theumer MG, et al. Subchronic mycotoxicoses in Wistar rats: assessment of the in vivo and in vitro genotoxicity induced by fumonisins and aflatoxin B1, and oxidative stress biomarkers status. Toxicology. 2010 doi: 10.1016/j.tox.2009.12.007. [DOI] [PubMed] [Google Scholar]
- 49.Mary VS, Theumer MG, Arias SL, Rubinstein HR. Reactive oxygen species sources and biomolecular oxidative damage induced by aflatoxin B1 and fumonisin B1 in rat spleen mononuclear cells. Toxicology. 2012 doi: 10.1016/j.tox.2012.08.012. [DOI] [PubMed] [Google Scholar]
- 50.Liu Y, Wang W. Aflatoxin B1 impairs mitochondrial functions, activates ROS generation, induces apoptosis and involves Nrf2 signal pathway in primary broiler hepatocytes. Anim. Sci. J. 2016;87:1490–1500. doi: 10.1111/asj.12550. [DOI] [PubMed] [Google Scholar]
- 51.Wogan GN, Paglialunga S. Carcinogenicity of synthetic aflatoxin M1 in rats. Food Cosmet. Toxicol. 1974 doi: 10.1016/0015-6264(74)90012-1. [DOI] [PubMed] [Google Scholar]
- 52.Neal GE, Eaton DL, Judah DJ, Verma A. Metabolism and toxicity of aflatoxins M 1 and B 1 in human-derived in vitro systems. Toxicol. Appl. Pharmacol. 1998;151:152–158. doi: 10.1006/taap.1998.8440. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.