Abstract
Background
To find economical and clinically available immune-related prognostic markers that could predict the overall survival (OS) of newly diagnosed multiple myeloma (NDMM) in the new drug era.
Material/Methods
Absolute lymphocyte count (ALC) and absolute monocyte count (AMC) were measured in routine blood samples from 102 patients with NDMM, and the lymphocyte-monocyte ratio (LMR) was derived. All the patients were receiving bortezomib-based chemotherapy as induction treatment. Log-rank testing was used for comparing the differences between groups. Univariate and multivariate tests were used to identify prognostic markers.
Results
The median ALC and LMR values at diagnosis were 1.43×109/L and 3.7, respectively, and served as the cutoff point. As prognostic factors, ALC, LMR, and a new staging system combining ALC and the ISS staging system (L-ISS) were expected to have a significant impact on predicting OS. Furthermore, multivariate analysis showed that ALC ≥1.43×109/L (hazard ratio [HR]: 0.223; 95% confidence interval [CI]: 0.071–0.705; P=0.011), LMR ≥3.7 (HR: 0.363; 95% CI: 0.139–0.947; P=0.038), and L-ISS late stage (HR: 1.619; 95% CI: 1.065–2.743; P=0.027) were independent predictors for OS.
Conclusions
ALC and LMR can serve as surrogate markers for patients’ antitumor immunity at the initial diagnosis of multiple myeloma. A new immune-related staging system, L-ISS, which combines ALC and the ISS staging system, can predict clinical outcomes in patients who are receiving bortezomib-based chemotherapy.
MeSH Keywords: Lymphocyte Count, Multiple Myeloma, Prognosis
Background
Multiple myeloma (MM) is a common hematological malignant disease characterized by abnormal plasma cell proliferation and monoclonal immunoglobulin production. It is more common among the elderly, and it takes second place among hematological malignancies [1,2]. Early identification of high-risk groups and appropriate treatment regimens may increase the rate of remission and extend overall survival (OS) [3]. With the introduction of new drugs (protease inhibitor and immunomodulatory agents), disease prognosis has improved significantly in the last decade [4,5]. However, MM is still incurable, with a 52.2% 5-year OS rate based on data from the Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database 2009–2015. Currently, the international staging system (ISS) [6] and the Durie-Salmon (DS) staging system [7] are the main prognostic assessment systems for MM, but they do not account for host immunity or new drugs’ effect. In recent years, antitumor immune status has been recognized as affecting patients’ prognosis in many malignant diseases including hematological malignancy. During the same time period, it was found that absolute lymphocyte count (ALC) of peripheral blood could reflect host immune function and predict patients’ prognosis in several diseases [8–11]. A reduced lymphocyte count is considered a poor prognostic factor in malignancy, suggesting an important role for immunosurveillance and clearance of lymphocytes [12]. The survival of MM cells depends on their interaction with the microenvironment [13]. A new prognostic evaluation system that incorporates immune-related biomarkers is urgently needed. Studies have shown that tumor-associated macrophages (TAMs) play a role in supporting MM cell growth, survival, and drug resistance in the pathogenesis of MM [14]. In several tumors, an elevation in the absolute monocyte count (AMC) of peripheral blood has been reported to be associated with a poor prognosis and could be considered a prognostic factor [15,16]. In addition, an elevation in the lymphocyte–monocyte ratio (LMR) is considered a good prognostic indicator for newly diagnosed MM (NDMM) patients [17]. Suriu et al. [18] showed that the level of ALC had no significant effects on the outcome of MM patients treated with new drugs, although other research yielded the opposite conclusion. So, it is debatable whether ALC can be used as one of the criteria for evaluating the prognosis of patients taking new drugs. At present, there are few reports about the effect of ALC, AMC, and LMR on the treatment and prognosis of MM. In the current study, the effects of these 3 factors on OS in Chinese NDMM patients and the relationship between ALC and clinical parameters were analyzed retrospectively. We also explored whether ALC could function as an antitumor immune-related marker that has prognostic value for NDMM patients in the era of novel agents.
Material and Methods
General information
This study included 102 NDMM patients from January 1, 2014, to December 31, 2018, in Shengjing Hospital of China Medical University who were treated with bortezomib-based chemotherapy. The diagnosis was made according to the International Myeloma Working Group guidelines for symptomatic MM. All the patients were classified according to the staging criteria [6]. The patients had no immune deficiency disease and no history of transplantation, other malignant tumor, or prior immunosuppressive therapy. The following baseline data were collected: sex, age, body mass index (BMI), clinical stage, typing, hemoglobin (HGB) at first diagnosis, ALC, AMC, LMR, platelet count (PLT), lactate dehydrogenase (LDH), β2-microglobulin (β2-MG), serum albumin (ALB), serum calcium concentration, and immunoglobulin heavy chain (IgH) quantity. Univariate and multivariate analyses were done to find the clinical parameters that might affect OS.
Statistical analysis
OS was defined as the time from diagnosis to death (regardless of cause). Univariate analysis of survival was done by Kaplan-Meier curve, and multivariate Cox regression models were used to verify the association between multiple variables and prognosis. Log-rank test was used for making comparisons between groups. P<0.05 was considered statistically significant. SPSS 26.0 software was used for data analysis.
Results
Patients’ characteristics
Patients’ baseline clinical and laboratory characteristics are listed in Table 1. There were 287 patients who received a diagnosis of MM during this period, and 102 patients were enrolled in the study. We excluded 185 patients as follows: 2 patients had a history of solid tumor, 13 patients received immunosuppressive therapy, 31 patients refused chemotherapy, and 139 patients received the traditional regimen without bortezomib. The median follow-up time was 427 (range: 5–1812) days. By the end of follow-up (February 10, 2019), 65 of the 102 enrolled patients (63.7%) were still alive.
Table 1.
The relationship between ALC and prognostic factors (P<0.05).
| Characteristics | Low ALC group ALC <1.43×109/L |
High ALC group ALC ≥1.43×109/L |
P-value |
|---|---|---|---|
| Sex | 0.297 | ||
| Male | 36 | 31 | |
| Female | 15 | 20 | |
| Age, y | 0.003 | ||
| <60 | 17 | 32 | |
| ≥60 | 34 | 19 | |
| BMI, kg/m2 | 0.992 | ||
| <22.85 | 20 | 22 | |
| ≥22.85 | 21 | 23 | |
| DS staging | 0.13 | ||
| Stage I | 1 | 6 | |
| Stage II | 7 | 8 | |
| Stage III | 43 | 37 | |
| Group A | 31 | 43 | 0.008 |
| Group B | 20 | 8 | |
| ISS staging | 0.007 | ||
| Stage I | 1 | 4 | |
| Stage II | 12 | 24 | |
| Stage III | 38 | 23 | |
| ISS dichotomy | 0.002 | ||
| Early stage | 13 | 28 | |
| Late stage | 38 | 23 | |
| L-ISS staging | <0.001 | ||
| Stage I | 0 | 5 | |
| Stage II | 12 | 44 | |
| Stage III | 39 | 2 | |
| L-ISS dichotomy | <0.001 | ||
| Early stage | 12 | 49 | |
| Late stage | 39 | 2 | |
| M-fraction type | 0.644 | ||
| IgG | 24 | 25 | |
| IgA | 8 | 12 | |
| Light chain | 14 | 12 | |
| Light chain type | 0.231 | ||
| κ | 24 | 31 | |
| lambda; | 24 | 19 | |
| HGB, g/L | <0.001 | ||
| <100 | 44 | 27 | |
| ≥100 | 7 | 24 | |
| PLT | 0.038 | ||
| <135×109/L | 23 | 13 | |
| ≥135×109/L | 28 | 38 | |
| LDH, U/L | 0.971 | ||
| <243 | 29 | 31 | |
| ≥243 | 20 | 22 | |
| β2-MG, mg/L | 0.004 | ||
| <5.5 | 11 | 25 | |
| ≥5.5 | 37 | 24 | |
| ALB, g/L | 0.19 | ||
| <35 | 35 | 29 | |
| ≥35 | 16 | 22 | |
| Ca, mmol/L | 0.065 | ||
| <2.75 | 48 | 42 | |
| ≥2.75 | 3 | 9 | |
| IgG dichotomy, g/L | 0.26 | ||
| <70 | 17 | 14 | |
| ≥70 | 6 | 10 | |
| IgA dichotomy, g/L | 0.848 | ||
| <50 | 5 | 8 | |
| ≥50 | 3 | 4 |
The median peripheral blood ALC was 1.43 (range: 0.48–5.32)×109/L at the time of initial diagnosis, which was considered as the cutoff point in our study. In order to determine whether ALC can enhance the prognostic effect of ISS, we divided 102 patients into 3 groups by combining ALC and ISS (L-ISS). The L-ISS staging system was defined as follows: L-ISS stage I (good prognostic group): ALC ≥1.43×109/L plus ISS stage I; L-ISS stage II (general prognostic group): neither I or III; and L-ISS stage III (poor prognostic group): ALC <1.43×109/L plus ISS stage III. In order to identify the effect of ALC among high-stage patients, we divided the patients into dichotomous groups. For both ISS and L-ISS, early stage included stage I and II and late stage was defined by stage III. Median values of AMC, LMR, and BMI were set as cutoff points as follows: AMC, 0.4×109/L; LMR, 3.7; and BMI, 22.85 kg/m2.
The relationships between the prognostic factors and ALC are shown in Table 1. The low ALC group was significantly associated with older age, DS stage group B, stage III of ISS and L-ISS staging system, late ISS stage, late L-ISS stage, HGB <100 g/L, low PLT group, and high β2-MG group (P<0.05). No significant differences were found between the high and low ALC group with regard to sex, BMI, DS staging, M-fraction type, light chain type, LDH, ALB, serum calcium, and IgH quantity (P>0.05) (Table 1).
Analysis the effects of clinical parameters on OS by univariate analysis
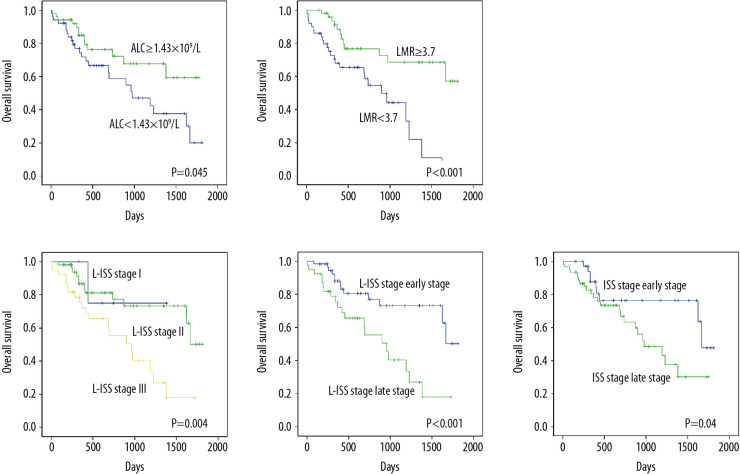
The results showed that patients with low ALC (Figure 1A) and LMR (Figure 1B), L-ISS stage III (Figure 1C), and late stage of L-ISS (Figure 1D) and ISS (Figure 1E) had significantly shorter OS than others (P<0.05, Table 1). Meanwhile, patients with low IgG and ALB had longer OS than others (P<0.05, Table 2). There were no significant difference in OS between sex, age, BMI, DS staging, ISS staging, light chain type, AMC, HGB, PLT, LDH, β2-MG, calcium, and the quantity of immunoglobin (P>0.05) (Table 2).
Figure 1.
(A) ALC <1.43×109/L predicted shorter OS. (B) LMR <3.7 predicted shorter OS. (C) Patients in L-ISS stage III were predicted to have shorter OS. (D) Patients in late stage of L-ISS were predicted to have shorter OS. (E) Patients in late stage of ISS predicted to have shorter OS.
Table 2.
Univariate analysis of the influence of clinical parameters on OS in MM patients (P<0.05).
| Characteristics | n | OS, % | P-value | |
|---|---|---|---|---|
| Sex | Male | 67 | 62.7 | 0.805 |
| Female | 35 | 65.7 | ||
| Age, y | <60 | 49 | 71.4 | 0.083 |
| ≥60 | 53 | 56.6 | ||
| BMI, kg/m2 | <22.85 | 42 | 66.7 | 0.335 |
| ≥22.85 | 44 | 75.0 | ||
| DS staging | Stage I | 7 | 85.7 | 0.138 |
| Stage II | 15 | 66.7 | ||
| Stage III | 80 | 61.3 | ||
| Group A | 74 | 67.6 | 0.054 | |
| Group B | 28 | 53.6 | ||
| ISS staging | Stage I | 5 | 100 | 0.088 |
| Stage II | 36 | 71.9 | ||
| Stage III | 61 | 60.7 | ||
| ISS dichotomy | Early stage | 41 | 75.7 | 0.04 |
| Late stage | 61 | 60.7 | ||
| L-ISS staging | Stage I | 5 | 80 | 0.004 |
| Stage II | 56 | 75 | ||
| Stage III | 41 | 46.3 | ||
| L-ISS dichotomy | Early stage | 61 | 78 | <0.001 |
| Late stage | 41 | 48.7 | ||
| M-fraction type | IgG | 49 | 75.5 | 0.004 |
| IgA | 20 | 65 | ||
| Light chain | 26 | 46.2 | ||
| Light chain type | κ | 55 | 60 | 0.519 |
| λ | 43 | 72.1 | ||
| ALC | <1.43×109/L | 51 | 52.9 | 0.045 |
| ≥1.43×109/L | 51 | 74.5 | ||
| AMC | <0.4×109/L | 52 | 67.3 | 0.284 |
| ≥0.4×109/L | 50 | 60.0 | ||
| LMR | <3.7 | 50 | 50.0 | <0.001 |
| ≥3.7 | 52 | 76.9 | ||
| HGB, g/L | <100 | 71 | 60.6 | 0.142 |
| ≥100 | 31 | 71.0 | ||
| PLT | <135×109/L | 36 | 61.1 | 0.381 |
| ≥135×109/L | 66 | 65.2 | ||
| LDH, U/L | <243 | 60 | 58.3 | 0.914 |
| ≥243 | 42 | 60.0 | ||
| β2-MG dichotomy, mg/L | <5.5 | 36 | 77.8 | 0.053 |
| ≥5.5 | 61 | 60.7 | ||
| ALB, g/L | <35 | 64 | 69.8 | 0.039 |
| ≥35 | 38 | 55.3 | ||
| Ca, mmol/L | <2.75 | 90 | 64.4 | 0.545 |
| ≥2.75 | 12 | 58.3 | ||
| IgG dichotomy, g/L | <70 | 31 | 80.6 | 0.362 |
| ≥70 | 16 | 68.8 | ||
| IgA dichotomy, g/L | <50 | 13 | 76.9 | 0.109 |
| ≥50 | 7 | 42.9 |
Relationship between OS and related factors in multivariate analysis
The results of the multivariate analysis of factors that might influence OS are shown in Table 3. The prognostic value of ALC, LMR, ALB, L-ISS dichotomy, and ISS dichotomy was assessed. The results showed that ALC ≥1.43×109/L (HR: 0.223; 95% CI: 0.071–0.705; P=0.011), LMR ≥3.7 (HR: 0.363; 95% CI: 0.139–0.947; P=0.038), and L-ISS staging system (HR: 1.619; 95% CI: 1.065–2.743; P=0.027) were still independent prognostic factors for predicting OS for the NDMM treated with a bortezomib-based regime (Table 3).
Table 3.
Relationship between OS and related factors in the multivariate analysis (P<0.05).
| P-value | HR (95% CI) | |
|---|---|---|
| ALC ≥1.43×109/L | 0.011 | 0.223 (0.071–0.705) |
| LMR ≥3.7 | 0.038 | 0.363 (0.139–0.947) |
| ALB ≥35 g/L | 0.311 | 0.564 (0.186–1.709) |
| L-ISS late stage | 0.027 | 1.619 (1.065–2.743) |
| ISS late stage | 0.624 | 0.699 (0.167–2.928) |
Discussion
Prediction of the prognosis in MM remains a challenge, despite commonly used prognostic tools such as the DS staging, ISS stage, and revised ISS (R-ISS) staging systems. These staging systems are based on tumor burden and tumor cytogenetics and thus neglect markers of immune dysfunction [19]. Under normal conditions, when tumor cells are present in the body, the immune system recognizes them due to their heterogeneity and produces an immune response [20]. Individual differences in immune condition could affect patients’ prognosis. Currently, immune function tests are costly and cannot be performed in some hospitals. A cost-effective and widely available clinical test that can predict progression is needed.
Lymphocytes represent an important cellular component of the body’s immune response, and they include several subgroups, each of which plays a different role; consequently, the antitumor immune response associated with lymphocytes is very complex [21,22]. Changes in immunologic and inflammatory biomarkers, such as the ratio of peripheral blood neutrophils to lymphocytes, have been shown to affect clinical outcomes [23–25]. Although ALC does not represent the whole picture of the immunologic anti-tumor mechanism and the function and quantity of subsets, it can be used as an important index of immune function [26]. Monocytes can reduce the antitumor ability of the host and promote the survival and proliferation of tumor cells, which is regarded as an indicator of poor prognosis. The underlying mechanism is still being investigated, but one of the observations is that tumor-derived chemotactic factors recruit circulating monocytes to the tumor site. These monocytes are identified as TAMs [27], and they have been shown to play an important role in tumor cell growth, angiogenesis, and metastasis [28]. Previous research found that the peripheral blood AMC can serve as a surrogate marker for TAMs [29,30]. ALC is also an important marker of immune remodeling after autologous stem cell transplant, and the level of ALC recovery after transplant is an independent prognostic factor [8]. In vitro studies showed that B and T lymphocytes were inhibited by MM cells, indicating that ALC is associated with disease severity. In our study, patients with a low ALC were elderly, had advanced stage disease, and had severe anemia, which aligned with the previous study. Ege et al. [31] analyzed 537 MM patients retrospectively and found a significantly longer OS for MM patients with ALC ≥1.4×109/L compared with those with ALC <1.4×109/L (65 vs. 26 months, P<0.0001), which indicated that ALC was an important prognostic factor in NDMM at initial diagnosis. Other studies indicated that low ALC (<1.6×109/L) at initial diagnosis of MM patients predicted shorter OS and LMR > 4 predicted better survival tendency [18].
Taken together, the levels of ALC, AMC, and LMR might be considered as being representative of the host immune system. Therefore, our study was performed to identify whether peripheral blood ALC, which represents the patient’s immunity, and AMC, which represents the tumor microenvironment, combined with LMR had prognostic significance for MM.
The prognostic impact of ALC, AMC, and LMR in NDMM patients treated with bortezomib-based chemotherapy was retrospectively evaluated in the present study. The results showed that both ALC ≥1.43×109/L and LMR ≥3.7 were associated with longer OS and were also independent predictors for OS in a multivariate analysis. The state of the immune system should therefore be carefully monitored as a potential variable influencing outcome in MM patients. AMC was not significantly associated with OS, suggesting that AMC alone does not represent a prognostic factor for MM patients. Overall, ALC and LMR were independent predictors, whereas AMC was not, indicating ALC could predict prognosis for MM patients. In addition, ALC is a surrogate marker of host immune function. So, we explored whether a new staging system incorporating patients’ initial immune function could identify high-risk MM patients. We combined the level of ALC and the ISS staging system into the L-ISS staging system, and we found that it further defined MM patients’ prognosis. The L-ISS staging system potentially improved the predictive value for NDMM patients being treated with bortezomib-based chemotherapy, which verified that the initial immune function was important.
However, some limitations were present in our study. Our results were based on a single-center analysis in China and the number of enrolled patients was not large. Moreover, these patients did not have test results for the lymphocyte subsets, including levels of CD8+ and CD4+ T cells and NK cells, to clearly define their immune status. In future studies, lymphocyte subsets should be quantified to clarify details about immune function (e.g., T cells, B cells, or NK cells) and their relationship with prognosis of MM patients.
Although these weaknesses existed, our study demonstrated that the ALC and LMR at diagnosis could be reveal the MM patients’ initial immune status and be related with prognosis. A multicenter study is still required to verify if this finding applies to more patients.
Conclusions
The treatment of MM has gone through the traditional treatment era and the new drugs era, and it is currently in the era of immunotherapy. Focusing on MM patients’ host immune function is quite important. ALC at initial diagnosis, an economical and clinically available index reflecting host systemic immunity, could predict NDMM patients’ prognosis in the bortezomib era. Owing to the significant prognostic value of ALC in NDMM, further studies on ALC, LMR, and even lymphocyte subsets could be conducted to better understand the immune-related role in individuals with MM.
Footnotes
Conflict of interest
None.
Source of support: This work was supported by The Natural Science Foundation of China (No. 81600115)
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. Cancer J Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 2.Kumar SK, Rajkumar V, Kyle RA, et al. Multiple myeloma. Nat Rev Dis Primer. 2017;3:17046. doi: 10.1038/nrdp.2017.46. [DOI] [PubMed] [Google Scholar]
- 3.Sonneveld P, Avet-Loiseau H, Lonial S, et al. Treatment of multiple myeloma with high-risk cytogenetics: A consensus of the International Myeloma Working Group. Blood. 2016;127:2955–62. doi: 10.1182/blood-2016-01-631200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rosiñol L, Oriol A, Rios R, et al. Bortezomib, lenalidomide, and dexamethasone as induction therapy prior to autologous transplant in multiple myeloma. Blood. 2019;134:1337–45. doi: 10.1182/blood.2019000241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Touzeau C, Moreau P. Ixazomib in the management of relapsed multiple myeloma. Future Oncol Lond Engl. 2018;14:2013–20. doi: 10.2217/fon-2017-0710. [DOI] [PubMed] [Google Scholar]
- 6.Greipp PR, San Miguel J, Durie BGM, et al. International staging system for multiple myeloma. J Clin Oncol. 2005;23:3412–20. doi: 10.1200/JCO.2005.04.242. [DOI] [PubMed] [Google Scholar]
- 7.Deng S, Zhang B, Zhou Y, et al. The role of 18F-FDG PET/CT in multiple myeloma staging according to IMPeTUs: Comparison of the Durie-Salmon Plus and other staging systems. Contrast Media Mol Imaging. 2018;2018 doi: 10.1155/2018/4198673. 4198673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sweiss K, Lee J, Mahmud N, et al. Combined immune score of lymphocyte to monocyte ratio and immunoglobulin levels predicts treatment-free survival of multiple myeloma patients after autologous stem cell transplant. Bone Marrow Transplant. 2020;55:199–206. doi: 10.1038/s41409-019-0681-3. [DOI] [PubMed] [Google Scholar]
- 9.Jimenez-Zepeda VH, Reece DE, Trudel S, et al. Absolute lymphocyte count as predictor of overall survival for patients with multiple myeloma treated with single autologous stem cell transplant. Leuk Lymphoma. 2015;56:2668–73. doi: 10.3109/10428194.2014.1003057. [DOI] [PubMed] [Google Scholar]
- 10.Jia T, Zhang R, Zhu H-Y, et al. Prognostic significance of peripheral blood absolute monocyte count and lymphocyte to monocyte ratio in anaplastic large cell lymphoma. Cancer Biomark Sect Dis Markers. 2018;22:807–13. doi: 10.3233/CBM-181505. [DOI] [PubMed] [Google Scholar]
- 11.Pushpam D, Rajput N, Chopra A, et al. Association of absolute lymphocyte count and peripheral blood lymphocyte subsets percentage with minimal residual disease at the end of induction in pediatric B cell acute lymphoblastic leukemia. Pediatr Hematol Oncol. 2019;36:138–50. doi: 10.1080/08880018.2019.1594469. [DOI] [PubMed] [Google Scholar]
- 12.Romano A, Parrinello NL, Simeon V, et al. High-density neutrophils in MGUS and multiple myeloma are dysfunctional and immune-suppressive due to increased STAT3 downstream signaling. Sci Rep. 2020;10:1983. doi: 10.1038/s41598-020-58859-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Alameda D, Saez B, Lara-Astiaso D, et al. Characterization of freshly isolated mesenchymal stromal cells from healthy and multiple myeloma bone marrow: transcriptional modulation of the microenvironment. Haematologica. 2020 doi: 10.3324/haematol.2019.235135. [Online ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Petty AJ, Yang Y. Tumor-associated macrophages in hematologic malignancies: new insights and targeted therapies. Cells. 2019;8(12):1526. doi: 10.3390/cells8121526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li Z, Xu Z, Huang Y, et al. The predictive value and the correlation of peripheral absolute monocyte count, tumor-associated macrophage and microvessel density in patients with colon cancer. Medicine (Baltimore) 2018;97:e10759. doi: 10.1097/MD.0000000000010759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Binder M, Rajkumar SV, Lacy MQ, et al. Peripheral blood biomarkers of early immune reconstitution in newly diagnosed multiple myeloma. Am J Hematol. 2019;94:306–11. doi: 10.1002/ajh.25365. [DOI] [PubMed] [Google Scholar]
- 17.Dosani T, Covut F, Beck R, et al. Significance of the absolute lymphocyte/monocyte ratio as a prognostic immune biomarker in newly diagnosed multiple myeloma. Blood Cancer J. 2017;7:e579. doi: 10.1038/bcj.2017.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Suriu C, Akria L, Azoulay D, et al. Absolute lymphocyte count as a prognostic marker in newly diagnosed multiple myeloma patients. Int J Lab Hematol. 2016;38:e56–59. doi: 10.1111/ijlh.12482. [DOI] [PubMed] [Google Scholar]
- 19.Jimenez-Zepeda VH, Duggan P, Neri P, et al. Revised International Staging System applied to real world multiple myeloma patients. Clin Lymphoma Myeloma Leuk. 2016;16:511–18. doi: 10.1016/j.clml.2016.06.001. [DOI] [PubMed] [Google Scholar]
- 20.Costa F, Das R, Kini Bailur J, et al. Checkpoint inhibition in myeloma: Opportunities and challenges. Front Immunol. 2018;9:2204. doi: 10.3389/fimmu.2018.02204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Reading JL, Gálvez-Cancino F, Swanton C, et al. The function and dysfunction of memory CD8+ T cells in tumor immunity. Immunol Rev. 2018;283:194–212. doi: 10.1111/imr.12657. [DOI] [PubMed] [Google Scholar]
- 22.Fang F, Xiao W, Tian Z. NK cell-based immunotherapy for cancer. Semin Immunol. 2017;31:37–54. doi: 10.1016/j.smim.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 23.Kelkitli E, Atay H, Cilingir F, et al. Predicting survival for multiple myeloma patients using baseline neutrophil/lymphocyte ratio. Ann Hematol. 2014;93:841–46. doi: 10.1007/s00277-013-1978-8. [DOI] [PubMed] [Google Scholar]
- 24.Onec B, Okutan H, Albayrak M, et al. The predictive role of the neutrophil/lymphocyte ratio in survival with multiple myeloma: A single center experience. J Clin Lab Anal. 2017;31(2):e22032. doi: 10.1002/jcla.22032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jang JE, Kim YR, Kim S-J, et al. A new prognostic model using absolute lymphocyte count in patients with primary central nervous system lymphoma. Eur J Cancer. 2016;57:127–35. doi: 10.1016/j.ejca.2016.01.016. [DOI] [PubMed] [Google Scholar]
- 26.Joshua D, Suen H, Brown R, et al. The T cell in myeloma. Clin Lymphoma Myeloma Leuk. 2016;16:537–42. doi: 10.1016/j.clml.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 27.Noy R, Pollard JW. Tumor-associated macrophages: From mechanisms to therapy. Immunity. 2014;41:49–61. doi: 10.1016/j.immuni.2014.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer. 2004;4:71–78. doi: 10.1038/nrc1256. [DOI] [PubMed] [Google Scholar]
- 29.Li Y-L, Shi Z-H, Wang X, et al. Tumor-associated macrophages predict prognosis in diffuse large B-cell lymphoma and correlation with peripheral absolute monocyte count. BMC Cancer. 2019;19:1049. doi: 10.1186/s12885-019-6208-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huang J-J, Li Y-J, Xia Y, et al. Prognostic significance of peripheral monocyte count in patients with extranodal natural killer/T-cell lymphoma. BMC Cancer. 2013;13:222. doi: 10.1186/1471-2407-13-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ege H, Gertz MA, Markovic SN, et al. Prediction of survival using absolute lymphocyte count for newly diagnosed patients with multiple myeloma: A retrospective study. Br J Haematol. 2008;141:792–98. doi: 10.1111/j.1365-2141.2008.07123.x. [DOI] [PubMed] [Google Scholar]