Abstract
Alzheimer's disease (AD) and Parkinson's disease (PD) are, respectively, the most prevalent and fastest growing neurodegenerative diseases worldwide. The former is primarily characterized by memory loss and the latter by the motor symptoms of tremor and bradykinesia. Both AD and PD are progressive diseases that share several key underlying mitochondrial, inflammatory, and other metabolic pathologies. This review will detail how these pathologies intersect with ketone body metabolism and signaling, and how ketone bodies, particularly d-β-hydroxybutyrate (βHB), may serve as a potential adjunctive nutritional therapy for two of the world's most devastating conditions.
Keywords: Alzheimer's disease, Parkinson's disease, Inflammation, Microglia, Insulin, Mitochondria, Oxidative stress, Ketone bodies, d-β-hydroxybutyrate, Ketone ester
Abbreviations
- Aβ
amyloid β
- AD
Alzheimer's disease
- AKT
protein kinase B
- APP
amyloid precursor protein
- A2ATP13A2
ATPase cation transporting 13A2
- βHB
d-β-hydroxybutyrate
- BDNF
brain-derived neurotrophic factor
- CD36
cluster of differentiation 36
- cGM
cerebral glucose metabolism
- CHCHD2
coiled-coil-helix-coiled-coil-helix domain containing 2
- CSF
cerebrospinal fluid
- DJ-1
PARK7
- FBXO7
F-box only protein 7
- FOXO3A
Forkhead box O 3A
- GBA
glucocerebrosidase
- GSK3β
glycogen synthase kinase 3β
- HCAR2
hydroxycarboxylic acid receptor 2
- HDACs
class I/II histone deacetylases
- HLA-DR
human leukocyte antigen-DR isotype
- IDE
insulin degrading enzyme
- IR
insulin receptor
- IRS1
insulin receptor substrate 1
- LRRK2/PARK8
leucine-rich repeat kinase 2
- MCI
mild cognitive impairment
- MHC
major histocompatibility complex
- MnSOD
manganese superoxide dismutase
- MPP+
1-methyl-4-phenylpyridinium
- MPTP
1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- mTOR
mechanistic target of rapamycin
- NAD+/NADH
nicotinamide adenine dinucleotide
- NADPH
nicotinamide adenine dinucleotide phosphate
- NFκB
nuclear factor κ-light-chain-enhancer of activated B cells
- NLRP3
NOD-, LRR- and pyrin domain-containing protein 3
- NO
nitric oxide
- PD
Parkinson's disease
- PGC1-α
PPARG coactivator 1 α
- PINK1
PTEN-induced kinase 1
- PLA2G6
phospholipase A2 group VI
- Q/QH2
coenzyme Q
- Q
ubiquinone
- QH2
ubiquinol
- ROS
reactive oxygen species
- SIRT3
mitochondrial sirtuin 3
- SNCA
α-synuclein
- TLR
toll-like receptor
- TNF-α
tumor necrosis factor α
- VPS35
vacuolar protein sorting-associated protein 35
- ΔG
Gibb's free energy
1. Mitochondrial dysfunction
1.1. Definition
“Mitochondrial dysfunction” is a blanket term used to describe all manner of maladaptive mitochondrial phenotypes, including impairment in electron transport chain components, fission and fusion dynamics, intracellular trafficking, mitophagy, and so on. However, the concept can be simplified by defining mitochondrial dysfunction in terms of its two ultimate consequences:
-
(1)
Underproduction of ATP.
-
(2)
Overproduction of reactive oxygen species (ROS).
For the purposes of this review, we will define mitochondrial dysfunction by these two consequences. In this section, we first present data that suggest mitochondrial dysfunction is a common, and perhaps causal, factor in the pathogenesis of AD and PD, before moving into a mechanistic discussion about how the ketone body, d-β-hydroxybutyrate (βHB), may correct or compensate for mitochondrial dysfunction.
1.2. Mitochondrial dysfunction in the pathogenesis of Alzheimer's disease and Parkinson's disease
Studies on cognitively normal individuals with a maternal family history of AD, carriers of the AD-risk allele, ApoE4, and animal models suggest that one of the earliest recognizable features of AD is a decrease in cerebral glucose metabolism (cGM) (Blass et al., 2000; Cunnane et al., 2011; Mosconi et al., 2008; Reiman et al., 1996). Although the exact cascade of pathological events remains to be determined, and may vary among individuals, evidence suggests that a decrease in cGM may coincide with, or even precede, the preclinical deposition of amyloid β (Aβ) (Andersen et al., 2017; Vlassenko et al., 2010). The AD-associated deficiency in cGM is attributable to impaired mitochondrial oxidative phosphorylation and is consistent with the observation that electron transport chain complex IV is less active than normal, both in diagnosed AD patients and in individuals at high risk for developing AD (Maurer et al., 2000; Mosconi et al., 2007).
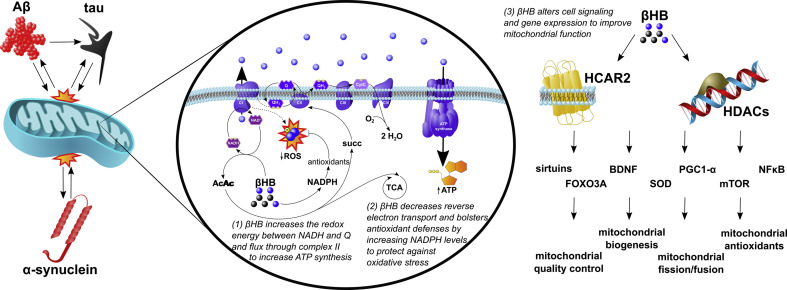
Mitochondrial dysfunction can contribute to the classical pathological hallmark of AD, Aβ plaques, by promoting the amyloidogenic processing of amyloid precursor protein (APP) (Wilkins and Swerdlow, 2017). Several lines of evidence support the hypothesis that mitochondrial dysfunction precedes Aβ pathology, including that complex IV inhibition induces the amyloidogenic pathway and that cell lines created by transferring mitochondrial DNA from AD patients into healthy cells exhibited a decrease in complex IV activity and ATP production with an increase in ROS (Cardoso et al., 2004; Gabuzda et al., 1994). However, since Aβ oligomers (and also tau oligomers) can reciprocally induce mitochondrial dysfunction (by mechanisms that include directly impairing respiratory chain protein function, increasing mitochondrial membrane permeabilization, inducing mitochondrial fission, disrupting mitophagy, and impairing axonal transport (Hu et al., 2017)), whether the amyloid and tau pathologies, or mitochondrial dysfunction, occur first to initiate a vicious cycle remains an open question (Fig. 1 ).
Fig. 1.
Neurodegenerative pathology, mitochondrial dysfunction, and βHB: The amyloid (Aβ) and tau pathologies of Alzheimer's disease and the α-synuclein pathology of Parkinson's disease are in positive feedback with mitochondrial dysfunction. Mitochondrial dysfunction is characterized by (i) the underproduction of ATP and (ii) overproduction of reactive oxygen species (ROS). βHB can improve mitochondrial function through three mechanisms: (1) βHB catabolism decreases the NAD+/NADH ratio and increases the Q/QH2 ratio to increase the redox span of the electron transport chain and, thereby, increase the generation of ATP. In addition, the rate limiting step of βHB catabolism generates succinate, an oxidative fuel for complex II that bypasses the complex I blockade present in Parkinson's disease to further increase ATP production in this condition. (2) By increasing the Q/QH2 ratio and levels of NADPH, βHB catabolism decreases the generation of ROS by reverse electron transport (dashed arrow) and bolsters antioxidant defenses. (3) βHB is also a signaling molecule that binds to its own G-protein coupled hydroxycarboxylic acid receptor 2 (HCAR2) and inhibits histone deacetylases (HDACs), both widely expressed throughout the brain, to regulate a wide variety of critical enzymes, transcription factors, and cofactors and, thereby, improve mitochondrial function.
Swerdlow and Khan (2004) have proposed that, while dysfunctional amyloid processing may be the primary insult in the 5% of cases caused by deterministic genetic mutations, in the remaining 95% of sporadic AD cases, mitochondrial dysfunction represents the primary insult (Swerdlow and Khan, 2004). While it is important to note that very low levels of intracellular Aβ, even in the absence of extracellular Aβ deposition, can impair mitochondrial function (Du et al., 2010), Swerdlow's “mitochondrial cascade hypothesis” has gained traction in the field. Perhaps the most human-relevant line of evidence supporting the hypothesis regards the maternal heritability of AD. Recall, complex IV is underactive in AD (Maurer et al., 2000) and that mitochondria and mitochondrial DNA, which codes for electron transport chain components, including all three catalytic components of complex IV (Kadenbach and Hüttemann, 2015), are inherited by the embryo from the mother's egg. In this context, it is interesting that maternal inheritance impacts a person's risk of having a decreased cGM and developing AD far more than paternal inheritance (Cunnane et al., 2011; Mosconi et al., 2007, Mosconi et al., 2010). Strikingly, cognitively normal adults who have mothers with AD exhibit a 50% decrease in complex IV activity compared to adults who have fathers with AD (Mosconi et al., 2011). Such data are consistent with the notion that, at least in some cases, mitochondrial dysfunction may affect a person's chances of developing neurodegenerative disease as early as conception.
The PD brain is similarly characterized by a decrease in ATP levels (Hu et al., 2000) and a corresponding decrease in electron transport chain protein function; although, in PD, complex I activity is impaired (Shapira et al., 1990). In fact, animal models of PD often rely on toxins that inhibit complex I to induce mitochondrial dysfunction and parkinsonism (MPTP and rotenone). Furthermore, not only are the environmental risk factors of PD associated with mitochondrial dysfunction, but all the major known risk genes for PD affect mitochondrial function, including SNCA, LRRK2, VPS35, GBA, CHCHD2, PINK1, Parkin, DJ-1, PLA2G6, ATP13A2, and FBXO7 (Helley et al., 2017; Park et al., 2018).
Of particular interest is the protein product of SNCA, α-synuclein. As is the case for Aβ and tau in AD, α-synuclein in PD may induce, and be induced, by mitochondrial dysfunction (Rocha et al., 2018) (Fig. 1). Evidence suggests that this positive feedback loop may involve complex I inhibition and mitochondrial dysfunction contributing to a decrease in autophagy, the ATP-dependent cellular recycling process known to promote α-synuclein disposal (Thomas et al., 2018; Xilouri et al., 2016). α-Synuclein, which itself contains a mitochondrial targeting sequence, can, in turn, impair mitochondrial protein import, alter mitochondrial morphology, induce oxidative stress, and even further inhibit complex I in order to establish a vicious cycle that culminates progressive neurodegeneration (Devi et al., 2008; Rocha et al., 2018).
1.3. d-β-hydroxybutyrate may correct or compensate for mitochondrial dysfunction in Alzheimer's disease and Parkinson's disease
The first study investigating whether βHB can protect cell models of AD and PD was performed in by Kashiwaya et al. (2000). In this study, the investigators treated hippocampal neurons with neurotoxic Aβ and dopaminergic neurons with 1-methyl-4-phenylpyridinium (MPP+) to model AD and PD, respectively. As expected, these treatments decreased neuron viability. However, when the cells were pretreated with a clinically achievable and safe concentration of βHB (4 mM), they were resistant to Aβ and MPP+-induced cell death (Kashiwaya et al., 2000). In this seminal publication, the authors postulated that the protective effects of βHB were due to improved mitochondrial function, a prediction that has been supported by other studies that have shown exogenously administered βHB can boost ATP production and/or prevent Aβ- or MPP+-induced superoxide generation in neurons in vitro (Maalouf et al., 2011; Marosi et al., 2016; Tieu et al., 2003).
In vivo studies have also begun to explore the potential benefits of exogenous βHB on mitochondrial function in animal models of AD and PD. In one such study, a ketone monoester, an orally ingestible compound that is metabolized directly into βHB, was fed to 3xTgAD mice starting at 8 months of age for a duration of 8 months. When the brains of the 16-month old mice were examined, the ketone monoester supplemented mice exhibited an increase in the Gibb's free energy (ΔG) of ATP hydrolysis and a decrease in lipid and protein oxidation in their hippocampi relative to controls (Pawlosky et al., 2017). The same research group also showed that the ketone diet decreased hippocampal Aβ and p-tau load and improved anxiety and context-dependent memory in AD mice (Kashiwaya et al., 2013). Complementary results have been obtained from a mouse model of PD in which subcutaneously administered exogenous βHB appeared to help circumvent the PD-associated complex I blockade (by increasing electron entry at complex II of the electron transport chain), increase ATP production, and improve symptoms of parkinsonism (Tieu et al., 2003).
These exciting preliminary findings beg the question, how might βHB improve mitochondrial function in AD and PD? Based on existing literature, the mechanisms by which βHB, either exogenously administered or endogenously produced, may increase mitochondrial ATP production and decrease mitochondrial ROS in the brain can be divided into three categories, each of which is elaborated upon in the following three paragraphs: (1) effects on redox ratios and electron transport chain function, (2) effects on ROS production, nicotinamide adenine dinucleotide phosphate (NADPH), and antioxidant status, and (3) effects on cell signaling and gene expression.
The effects on redox ratios and electron transport chain function refers, first and foremost, to the positive effect of βHB catabolism on the redox span between the mitochondrial nicotinamide adenine dinucleotide (NAD+/NADH) and coenzyme Q (Q/QH2) couples. In oxidative metabolism, mitochondrial NADH passes its electron pair through complex I to ubiquinone (Q) to generate NAD+ and ubiquinol (QH2). Since the NAD+/NADH couple has a more negative redox potential (i.e., holds electrons in a higher energy state) than the Q/QH2 couple, this process of passing electrons from NADH to Q liberates potential energy that is used to pump protons from the matrix into the intermembrane space. Interestingly, βHB catabolism decreases the matrix NAD+/NADH ratio while increasing the Q/QH2 ratio (at least in βHB-perfused rat hearts), increasing the difference in redox potentials between these two couples (Sato et al., 1995). The effect of increasing the “redox span” between electron carriers is analogous to increasing the height span from which a bowling ball is dropped to the ground. In both cases, more energy is available to do work. In the case of the bowling ball height span, more kinetic energy is available to break your toes. In the case of the NAD+/NADH-Q/QH2 redox span, more electrons can be pumped across the inner mitochondrial membrane to fuel ATP production by chemiosmosis. Moreover, βHB catabolism may be able to increase ATP production in the PD brain by circumventing the PD-associated blockade of complex I (Benecke et al., 1993; Devi et al., 2008; Mann et al., 1992). This mechanism makes biochemical sense because the rate limiting step of βHB catabolism generates succinate, an oxidative fuel that feeds into complex II, and, thereby, should bypass the complex I blockade. This more PD-specific mechanism is supported by in vivo data showing that βHB protected PD mice from neurodegeneration, but not when flux through complex II was blocked (Tieu et al., 2003). In summary, by increasing the redox span between NAD+/NADH and Q/QH2, and by increasing flux through complex II in PD, βHB catabolism may increase the production of ATP, alleviating one of the two ultimate consequences of mitochondrial dysfunction (Fig. 1).
The other consequence of mitochondrial dysfunction, oxidative stress, may be addressed by the effects of βHB on ROS production, NADPH, and antioxidant status. As mentioned in the previous paragraph, βHB catabolism increases the Q/QH2 ratio. In addition to increasing the redox span within the electron transport chain to increase proton pumping and ATP production, a higher Q/QH2 ratio also carries the benefit of a decrease in “reverse electron transport,” the process by which most ROS are generated by mitochondria. In reverse electron transport, QH2, rather than passing electrons forward to complex III, passes electrons backward at complex I to oxygen to generate superoxide radicals. Thus, by increasing the Q/QH2 ratio, βHB catabolism decreases the generation of ROS. Complimentarily, βHB catabolism can also bolster antioxidant defenses by decreasing the NADP+/NADPH ratio (Norwitz et al., 2019; Veech et al., 2019). Unlike NADH, which functions to support oxidative metabolism, NADPH is used for the reductive biosynthesis of antioxidants. In fact, NADPH is required to support all known intracellular antioxidant species, including glutathione, thioredoxins, and vitamins C and E (Veech et al., 2019). The multiple mechanisms by which βHB catabolism can increase NADPH levels and antioxidant defenses in neurodegenerative diseases has been reviewed elsewhere (Norwitz et al., 2019) (Fig. 1).
The final mechanism by which βHB could alleviate mitochondrial dysfunction in AD and PD is the most expansive. d-β-hydroxybutyrate is not only a nutritional macromolecule, but also a signaling molecule. It has several G-protein coupled receptors, including hydroxycarboxylic acid receptor 2 (HCAR2), and inhibits class I/II histone deacetylases (HDACs) to alter gene expression (Lang et al., 2019; Newman and Verdin, 2014a, Newman and Verdin, 2014b; Veech et al., 2017). A detailed discussion of the multitudinous effects of βHB signaling on cell metabolism is beyond the scope of this chapter, but may include induction of the pro-longevity sirtuin proteins and Forkhead box O 3A (FOXO3A) transcription factor, the neurotropic factor brain-derived neurotrophic factor (BDNF), the antioxidant enzyme manganese superoxide dismutase (MnSOD), the master regulator of mitochondrial biogenesis PPARG coactivator 1 alpha (PGC1-α), and several autophagy proteins, as well as inhibition of the anti-longevity mechanistic target of rapamycin (mTOR) and proinflammatory transcription factor nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) (Norwitz et al., 2019). Either directly or indirectly, any or all of such cell signaling effects could improve mitochondrial quality, increase mitochondrial ATP production, and decrease oxidative stress (Fig. 1).
2. Inflammation
2.1. Central role in neurodegenerative conditions
It is generally accepted that neuroinflammation contributes to neurodegenerative diseases. This neuroinflammation is mediated, in large part, by hyperactive microglia (Gabandé-Rodríguez et al., 2019) and astrocytes (Li et al., 2019), which together contribute to chronic low-grade release of cytokines, such as the interleukins, IL-1β and IL-6, and tumor necrosis factor α (TNF-α), pathological levels of phagocytosis, disease-specific toxin production, and neurological damage in general (Alam et al., 2016; Bachiller et al., 2018; Krasemann et al., 2017).
2.2. Microglia activation
Microglia activation leads to the release of inflammatory factors, such as nitric oxide (NO) and prostaglandins (Bachiller et al., 2018). These responses are highly controlled and are accompanied by a metabolic shift that remains poorly understood. In neurodegenerative diseases, the increased inflammatory signaling caused by neuronal damage cause dysregulation of microglia homeostasis, which in turn release proinflammatory cytokines. As the observant reader has, or will, notice, such vicious cycles are characteristic of neurodegenerative diseases (Hopkins and Rothwell, 1995).
In AD, the accumulation of Aβ activates microglia via cluster of differentiation 36 (CD36) and the Toll-like receptor (TLR) heterodimer TLR2-TLR6. In turn, microglial activation and secretion of NO may contribute to the formation of senile Aβ plaques (Stewart et al., 2010), establishing a positive feedback loop. Moreover, the contribution of hyperreactive microglia to the formation of tau tangles may be a missing link in the amyloid cascade model of AD in which Aβ pathology leads to downstream tau pathology (Bachiller et al., 2018).
In PD, higher expression of human leukocyte antigen-DR isotype (HLA-DR), a major histocompatibility complex (MHC) class II receptor, has been found in the post-mortem substantia nigra (Imamura et al., 2003; McGeer et al., 1988). This, together with the fact that α-synuclein activates TLRs (Béraud et al., 2011; Theodore et al., 2008), is consistent with a critical role for inflammation in the death of dopaminergic neurons (Schröder et al., 2018).
2.3. Astrocyte activation
Astrocytes have a broad set of functions, including regulating blood-brain barrier permeability and maintaining synaptic integrity. Importantly, astrocytes have a metabolic role that includes quenching inflammatory factors (Phillips et al., 2014).
Activated astrocytes are divided into two subgroups: A1 astrocytes are, to generalize, neurotoxic (Liddelow et al., 2017), whereas A2 astrocytes are neuroprotective (Christopherson et al., 2005; Giordano et al., 2009; Li et al., 2019). Although an imbalance in the equilibrium toward the harmful A1 fate is presumed in cases of neurodegenerative disease, it is important to treat this matter with nuance and give credit to activated astrocytes as more than the “bad guys.” For example, transplantation of astrocytes helps to clear Aβ plaques (Pihlaja et al., 2011). However, astrocytes can also produce neurotoxic Aβ oligomers (Rossi and Volterra, 2009). Furthermore, Aβ might disrupt astrocyte metabolism, possibly contributing to an increase in the (neurotoxic) A1 over the (neuroprotective) A2 fate, inducing yet another positive feedback loop (Vincent et al., 2010). In PD, the situation is similar: early accumulation of α-synuclein in astrocytes causes an increase in microglia hyperactivation (Halliday and Stevens, 2011), blood-brain barrier permeability, and energy imbalances (Li et al., 2019), all of which are associated with disease progression.
2.4. d-β-hydroxybutyrate dampens neuroinflammation via histone deacetylase and NLRP3 inflammasome inhibition
Ketogenic diets have proven to be successful in the treatment of multiple neurodegenerative diseases (Vanitallie et al., 2005, Włodarek, 2019), although it is not entirely clear whether the benefits are derived from the ketones themselves or other aspects of the diet. Still, by acting as potent signaling molecules to alter intracellular signaling cascades and gene expression, there is a high probability that at least some of the benefits derive from βHB itself (Gano et al., 2014; Maalouf et al., 2009).
Among other signaling functions, βHB inhibits HDACs (Pinto et al., 2018), and it is possible that, in this way, βHB calms microglia and astrocyte hyperactivation. For example, βHB suppresses HDAC-induced oxidative stress (Shimazu et al., 2013) and induces microglia to adopt the anti-inflammatory M2 morphology (Huang et al., 2018).
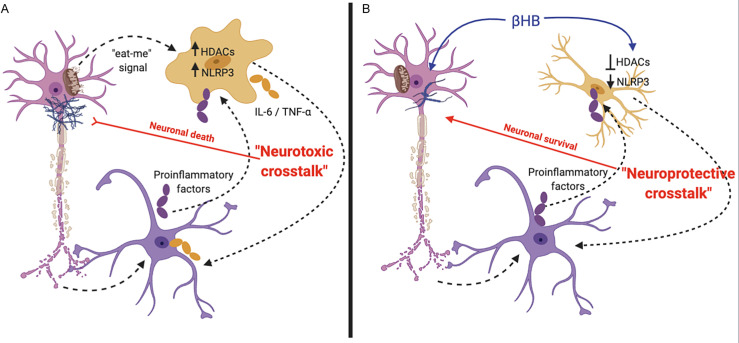
Additionally, one of the key regulators of inflammation is the NOD-, LRR- and pyrin domain-containing protein 3 (NLRP3) inflammasome (Shao et al., 2018). Interestingly, while βHB has been shown to inhibit the NLRP3 inflammasome, this effect seems to be independent of HDAC inhibition and other better-known βHB signaling mechanisms; rather, βHB inhibits the inflammasome by altering potassium flux (Youm et al., 2015) (Fig. 2 ).
Fig. 2.
Anti-inflammatory role of βHB in neurodegenerative diseases: (A) Neurodegeneration induces the A1 proinflammatory astrocyte (purple cell) response and triggers microglia-mediated (orange cell) phagocytosis. Transcription and release of proinflammatory factors increase the HDAC- and NLRP3-mediated inflammatory responses in glia, promoting neuronal (pink cell) death by neurotoxic cross-talk between proinflammatory microglia and astrocytes. (B) βHB may decrease inflammation signaling, in part, by inhibiting HDACs and impairing NLRP3 inflammasome formation.
Evidently, there is much more clinical and basic science work to be done to elucidate the putative cell-signaling-dependent anti-inflammatory effects of βHB.
3. Glucose and insulin
3.1. Lack of energy substrates in neurodegeneration
For its size, the brain is our most energetically demanding organ (Pontzer et al., 2016); however, it is metabolically inflexible. Under non-ketotic conditions, human brains, which cannot metabolize fat as fuel, are dependent on glucose (Balasse, 1979). Correspondingly, deficiencies in the brain's ability to metabolize glucose are thought to contribute to neurodegeneration.
Even in the preclinical stages, patients with AD (Willette et al., 2015) and PD (Hu et al., 2001) exhibit impaired cGM, but not impaired cerebral uptake of ketones (Cunnane et al., 2016). As it is believed that our species developed its superior capacity for ketogenesis specifically to fuel the brain in times of glucose scarcity, in our perspective, there are no apparent compelling reasons that ketones would not prove as beneficial in the context of pathological metabolic scarcity as they evidently were in supporting our species' evolutionary exposure to glucose dietary scarcity.
3.2. Insulin resistance: A hallmark of Alzheimer's disease
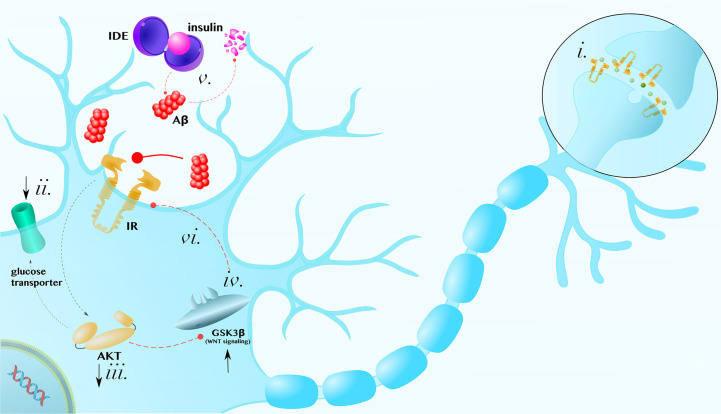
Insulin is a crucial regulator of many neuronal processes (Arnold et al., 2018). Both the presynaptic axon terminal and postsynaptic density are highly enriched with insulin receptors. In these compartments, insulin modulates catecholamine release and uptake, the trafficking of ion-gated channels, and the expression and localization of neurotransmitter receptors, such as GABA and NMDA receptors (Chiu et al., 2008) (Fig. 3i).
Fig. 3.
Insulin in the brain: (i) Insulin receptors (IR) are enriched at synapses, where they regulate neurotransmitter release and the localization of receptors. (ii) Insulin resistance decreases glucose transporters in the membrane, (iii) leads to the inhibition of downstream proteins, like AKT, (iv) and induces neurotoxic anti-WNT GSK3β activity. (v) Hyperinsulinemia prevents Aβ degradation by insulin degrading enzyme (IDE), establishing a positive feedback loop. (vi) Furthermore, GSK3β activity, which is antagonistic to neuroprotective WNT signaling, also inhibits insulin signaling. The figure is meant to be representative, not comprehensive, regarding the mechanisms by which insulin resistance can contribute to the degeneration of neurons.
Figure adapted, with permission, from Norwitz, N. G., et al. (2019). Multi-loop model of Alzheimer disease: An integrated perspective on the Wnt/GSK3β, α-synuclein, and type 3 diabetes hypothesis, Frontiers in Aging Neuroscience, 11, 184. doi: 10.3389/fnagi.2019.00184.
Interestingly, brain insulin resistance can occur without systemic insulin resistance and, because insulin reaches the cerebrospinal fluid (CSF) via the capillary endothelial cells of the blood-brain barrier and its transport and elimination are regulated separately (Banks et al., 2012), brain cells can be exposed to different insulin levels than those of peripheral tissues.
While it is true that insulin resistance increases with age (Yaffe et al., 2012), it is worth observing that inducing localized insulin resistance mimics many of the biochemical and clinical features of AD (Lannert and Hoyer, 1998) and that selective insulin transport through the blood-brain barrier is upregulated in many conditions that are prone to neurodegeneration, including obesity, diabetes, hypertriglyceridemia, and chronic inflammation (Heni et al., 2014).
At a cellular level, insulin resistance can result from a decrease in glucose transporters in the membrane (Niccoli et al., 2016; Nishida et al., 2017; Willette et al., 2015) and inhibition of insulin signaling proteins, such as protein kinase B (AKT), or their regulatory partners, such as the canonical WNT-β-catenin-glycogen synthase kinase 3β (GSK3β) pathway (Su et al., 2019; Tanokashira et al., 2019) (Fig. 3ii–iv). Correspondingly, direct pharmacological AKT activation rescues AD-like memory impairment and aberrant synaptic plasticity (Yi et al., 2018).
Insulin resistance and associated hyperglycemia, enhance other pathological hallmarks in many neurodegenerative diseases, including AD. The glycation of Aβ oligomers increase their pathogenicity (Li et al., 2013). In addition, insulin degrading enzyme (IDE) functions to degrade both insulin and Aβ. Therefore, hyperinsulinemia prevents Aβ degradation and, reciprocally, Aβ can further exacerbate insulin resistance by preventing insulin degradation (Fig. 3v) (Farris et al., 2003; O'Neill, 2013; Pérez et al., 2000; Zhao et al., 2017). What's more, such vicious cycles can compound: For example, insulin-AKT pathway dysfunction can contribute to an increase in GSK3β activity, which is antagonistic to neuroprotective WNT signaling (Fig. 3iv) (Lee et al., 2009; Magrané et al., 2005), whereas GSK3β can contribute to insulin resistance by phosphorylating and inhibiting insulin receptor substrate 1 (IRS1) (Lee and Kim, 2007) (Fig. 3vi). Thus, insulin resistance can exacerbate AD via a complex network of pathological positive feedback interactions (Norwitz et al., 2019).
In PD, insulin resistance impairs nigrostriatal dopamine function (Morris et al., 2011), α-synuclein negatively regulates insulin signaling (Gao et al., 2015), and insulin receptors in the substantia nigra are significantly decreased (Takahashi et al., 1996).
4. Ketotherapeutics
Because brain cells can oxidize ketone bodies and ketones regulate fuel metabolism (Edmond et al., 1987), interventions that induce ketosis offer an exciting opportunity to prevent, slow, halt, or reverse the progression of neurodegenerative diseases (Norwitz et al., 2019). Additionally, most ketogenic interventions are safe (Cicero et al., 2015; Murray et al., 2016; Soto-Mota et al., 2019), well tolerated, and improve other exacerbating comorbidities, such as type II diabetes (Hallberg et al., 2018; Lennerz et al., 2018) and inflammation (Sherrier and Li, 2019).
4.1. Endogenous nutritional ketosis
In monitored patients, fasting has proven to be safe (Stewart and Laura Fleming, 1973), a statement also supported by the fact that, for centuries, billions of people have fasted safely for religious reasons. The hormonal profile of the fasting state is perhaps the largest difference between endogenous and exogenous ketosis: endogenous ketosis is marked by a low-insulin, high-cortisol and glucagon environment that heavily promotes lipolysis (Gomez-Arbelaez et al., 2016).
High-fat low-carbohydrate ketogenic diets also result in a low-insulin, high-cortisol and glucagon environment, but differ from fasting in that ketones are derived partly from dietary fat, as well as from body fat. Foods high in medium chain triglycerides are particularly ketogenic compared with other fat sources (Kesl et al., 2016).
4.2. Exogenous nutritional ketosis
Many ketone salts are commercially available, their main limitation being that unhealthy intake of salt is required to achieve therapeutic levels of βHB. In addition, most salts provide a racemixture, rather than the pure bio-relevant d-βHB isoform (Stubbs et al., 2017).
By contrast ketone esters, specifically the best-studied βHB monoester, induces deep ketosis (levels observed after several days of fasting or following a strict ketogenic diet; > 3.0 mM) because it yields only d-βHB and does not carry the restriction of the accompanying salt bolus. Furthermore, ketone esters allow for the accurate titration of blood ketone levels within 30 min (Clarke et al., 2012). It is important to note that ketone salts and esters have different metabolic (Stubbs et al., 2017) and tolerability profiles (Stubbs et al., 2019).
To date, there is anecdotal support for the use of ketone esters in human AD (Newport et al., 2015) and clinical studies are in progress for their use in PD. For example, our group, Norwitz et al., is currently investigating whether this form of exogenous ketone can increase cerebral energy production (as measured by magnetic resonance spectroscopy) improve physical performance, and improve broad-spectrum symptomology and quality of life in persons with PD (ISRCTN10531043, completed; ISRCTN16599164, completed; ISRCTN64294760, temporarily suspended because of SARS-CoV-2 pandemic).
4.3. Endogenous vs exogenous nutritional ketosis
Endogenous and exogenous ketosis each comes with its own potential benefits and drawbacks. Endogenous ketosis, by requiring a more comprehensive metabolic shift, has the advantage of activating a larger array of metabolic pathways. Furthermore, fasting and ketogenic diets may help the body adapt to better utilize ketones as a fuel and signaling molecule, as opposed to exogenous ketogenic strategies in which the body can choose to continue to metabolize glucose. Finally, whereas it is well established that fasting is safe in the long-term, and the same can also probably be said of well-formulated ketogenic diets, little is known about the long-term impact of the high-glucose, high-insulin, high-ketone condition that results from addition of a ketone supplement to a typical carbohydrate-rich Western diet. The latter is not a metabolic state to which our species evolved and it is possible that long-term exposure to simultaneously high glucose and ketones could have unforeseen consequences.
On the other hand, endogenous ketosis does not permit the specific targeting of ketone levels, whereas exogenous ketosis does. In particular, ketone esters permit the induction of deep ketosis (> 3.0 mM), which may have particular therapeutic benefits by activating particular genetic and/or metabolic pathways. Furthermore, given the current state of food culture, social climate, and nutritional guidelines/common knowledge, fasting and ketogenic diets can come with compliance difficulties for many patients. This will, hopefully, change as culture and nutrition science evolve, but, at this time, imposes a serious practical limitation on endogenous ketosis interventions. Exogenous ketone supplements, by contrast, are easy to consume on a long-term basis. Finally, it is worth noting that ketone supplements can be stacked on top of fasting or ketogenic diets to induce deeper “therapeutic ketosis” (~3.0 mM) without incurring the risk of long-term exposure to high glucose and ketones.
4.4. Ketogenic interventions in patients with mild cognitive impairment
Recently, there have been several ketotherapeutic interventions conducted in patients with mild cognitive impairment (MCI), a precursor to Alzheimer's disease. For example, in a randomized crossover pilot study of a Mediterranean-style ketogenic diet for MCI, a 6-week ketogenic diet improved Alzheimer's biomarkers, including CSF levels of Aβ and tau, as compared to a 6-week American Heart Association diet control (Neth et al., 2019). Furthermore, medium chain triglycerides, which induce mild ketosis (Kesl et al., 2016) have demonstrated clinical efficacy in several trials. In a 6-month study of 52 MCI patients, medium chain triglyceride consumption at 2 Tbsp/day improved episodic memory, executive function, and processing speed compared to baseline and compared to a placebo control (Fortier et al., 2019). A recent meta-analysis confirmed that medium chain triglyceride interventions in Alzheimer's patients tend to improve functional cognitive measures, in part, by inducing ketosis (Avgerinos et al., 2019). The general efficacy of these early studies may be due to the fact that, while neurodegenerating brains appear to lose their ability to metabolize glucose, ketones remain a viable fuel and cerebral ketone uptake tends to parallel blood ketone levels (Croteau et al., 2018). Therefore, it will be important for future studies implementing other interventions to investigate whether deeper levels of ketosis, such as those induced by ketone esters, are even more effective.
4.5. Ketogenic interventions, mitochondria, and SIRT3
Since this review placed particular emphasis on mitochondrial dysfunction as the basis for neurodegenerative disease, it is important to remark on the recent evidence implicating activation of mitochondrial sirtuin 3 (SIRT3), a protein whose activity is reduced in AD patients in association with Aβ pathology (Cheng et al., 2019), as one mechanism by which ketogenic interventions may protect against AD. In AD mice, intermittent fasting to induce ketosis increased SIRT3 activity and protected against hyperexcitability and hippocampal synaptic dysfunction (Liu et al., 2019). While one could reasonably postulate that mechanisms related to intermittent fasting, other than ketosis, are responsible for the increase in SIRT3 activity, recent evidence suggests the neuroprotective effect is, indeed, due to βHB itself. In particular, supplementation with a ketone ester increased SIRT3 expression in a SIRT3 haploinsufficient mouse model of AD, preventing GABA neuron degeneration and protecting against excitotoxicity (Cheng et al., 2019). Therefore, the SIRT3 promoting anti-excitotoxicity effects ketones is one mechanism by which both endogenous and exogenous ketogenic interventions may prove therapeutic in neurodegenerative disease patients.
4.6. Intermittent fasting, metabolic switching, brain network stability, and disease prevention
It would be inappropriate to extrapolate from current clinical data that particular ketogenic interventions, while helping improve symptomology and disease markers in individuals already afflicted with neurodegenerative disease, would also help to prevent disease onset in individuals at risk for AD and PD. Given the growing popularity of ketogenic diets for brain health, even among individuals as young as their twenties, it is worth noting that no long-term trials on ketogenic interventions for complete neurocognitive disease prevention have yet been performed and, therefore, worth considering that promoting “metabolic flexibility” (in brains that still retain the ability to adequately utilize glucose) may be ideal for disease prevention. For example, intermittent fasting strategies (applied in metabolically healthy individuals) that induce ketosis but also permit the body to switch on glucose metabolism in a cyclic manner can both activate ketosis-associated cellular repair and defense pathways, while also optimally promoting healthy anabolic processes, such as the growth of synapses, during periods of carbohydrate feeding (de Cabo et al., 2019; Mattson et al., 2018). In the literature, this is commonly referred to as activating the “metabolic switch.” While there is no data yet comparing the safety or efficacy of chronic ketosis vs “metabolic switching” for neurodegenerative disease progression, it is logical to assume that the latter may come with certain to-be-discovered advantages for the simple reason that our species evolved to intermittent fast, rather than to chronically eat ketogenic diets or consume ketone supplements (Mattson et al., 2018).
Again, no long-term preventative studies for ketogenic interventions in neurodegenerative diseases have been conducted. However, it’s worth mentioning that a new fMRI-based whole-brain-scale biomarker of brain aging termed “network stability” (defined as the brain’s ability to communicate among regions) has recently been developed. A collaboration between Stony Brook and Oxford Universities showed that both ketogenic diets and exogenous ketones (ketone monoester) improve network stability in young healthy individuals. By contrast, standard Western diets and glucose decrease network stability (Mujica-Parodi et al., 2020). These data are consistent with the hypothesis that ketogenic interventions could prevent neurodegenerative disease and cognitive decline, as has been demonstrated in mice (Roberts et al., 2017). Whether neuroketotherapeutics actually do prevent, rather than treat, neurodegenerative disease currently remains in the realm of informed speculation.
5. Summary and relevance statement
Most neurodegenerative diseases, including AD and PD, are associated with the key pathologies of mitochondrial dysfunction, neuroinflammation, and glucose hypometabolism and/or insulin resistance. Currently, there are no effective therapies for slowing the progression of either AD or PD. By addressing these core pathologies (and likely others), endogenously or exogenously induced ketosis might prove to be a novel and useful adjunctive therapeutic for these neurodegenerative conditions.
References
- Alam Q., Alam M.Z., Mushtaq G., Damanhouri G.A., Rasool M., Kamal M.A. Inflammatory process in Alzheimer's and Parkinson's diseases: Central role of cytokines. Current Pharmaceutical Design. 2016;22(5):541–548. doi: 10.2174/1381612822666151125000300. [DOI] [PubMed] [Google Scholar]
- Andersen J.V., Christensen S.K., Aldana B.I., Nissen J.D., Tanila K., Waagepetersen H.S. Alterations in cerebral cortical glucose and glutamine metabolism precedes amyloid plaques in the APPswe/PSEN1dE9 mouse model of Alzheimer's disease. Neurochemical Research. 2017;42:1589–1598. doi: 10.1007/s11064-016-2070-2. [DOI] [PubMed] [Google Scholar]
- Arnold S.E. Brain insulin resistance in type 2 diabetes and Alzheimer disease: Concepts and conundrums. Nature Reviews Neurology. 2018;14:168–181. doi: 10.1038/nrneurol.2017.185. Nature Publishing Group. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avgerinos K.I. Medium chain triglycerides induce mild ketosis and may improve cognition in Alzheimer's disease. A systematic review and meta-analysis of human studies. Ageing Research Reviews. 2019;58:101001. doi: 10.1016/j.arr.2019.101001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachiller S., Jiménez-Ferrer I., Paulus A., Yang Y., Swanberg M., Deierborg T. Microglia in neurological diseases: A road map to brain-disease dependent-inflammatory response. Frontiers in Cellular Neuroscience. 2018;12:488. doi: 10.3389/fncel.2018.00488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balasse E.O. Kinetics of ketone body metabolism in fasting humans. Metabolism, Clinical and Experimental. 1979;28(1):41–50. doi: 10.1016/0026-0495(79)90166-5. [DOI] [PubMed] [Google Scholar]
- Banks W.A., Owen J.B., Erickson M.A. Insulin in the brain: There and back again. Pharmacology and Therapeutics. 2012;136:82–93. doi: 10.1016/j.pharmthera.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benecke R., Strümper P., Weiss H. Electron transfer complexes I and IV of platelets are abnormal in Parkinson's disease but normal in Parkinson-plus syndromes. Brain. 1993;116:1451–1463. doi: 10.1093/brain/116.6.1451. [DOI] [PubMed] [Google Scholar]
- Béraud D., Twomey M., Bloom B., Mittereder A., Ton V., Neitzke K. α-Synuclein alters toll-like receptor expression. Frontiers in Neuroscience. 2011;5:80. doi: 10.3389/fnins.2011.00080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blass J.P., Sheu R.K.-F., Gibson G.E. Inherent abnormalities in energy metabolism in Alzheimer disease interaction with cerebrovascular compromise. Annals of the New York Academy of Sciences. 2000;903:204–221. doi: 10.1111/j.1749-6632.2000.tb06370.x. [DOI] [PubMed] [Google Scholar]
- Cardoso S.M., Santana I., Swerdlow R.H., Oliveira C.R. Mitochondria dysfunction of Alzheimer's disease cybrids enhances Aβ toxicity. Journal of Neurochemistry. 2004;89(6):1417–1426. doi: 10.1111/j.1471-4159.2004.02438.x. [DOI] [PubMed] [Google Scholar]
- Cheng A. SIRT3 haploinsufficiency aggravates loss of GABAergic interneurons and neuronal network hyperexcitability in an Alzheimer's disease model. Journal of Neuroscience. 2019;40(3):694–709. doi: 10.1523/jneorusci.1446-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu S.L., Chen C.M., Cline H.T. Insulin receptor signaling regulates synapse number, dendritic plasticity, and circuit function in vivo. Neuron. 2008;58(5):708–719. doi: 10.1016/j.neuron.2008.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christopherson K.S., Ullian E.M., Stokes C.C.A., Mullowney C.E., Hell J.W., Agah A. Thrombospondins are astrocyte-secreted proteins that promote CNS synaptogenesis. Cell. 2005;120(3):421–433. doi: 10.1016/j.cell.2004.12.020. [DOI] [PubMed] [Google Scholar]
- Cicero A.F.G. Middle and long-term impact of a very low-carbohydrate ketogenic diet on cardiometabolic factors: A multi-center, cross-sectional, clinical study. High Blood Pressure & Cardiovascular Prevention. 2015;22(4):389–394. doi: 10.1007/s40292-015-0096-1. Springer International Publishing. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke K., Tchabanenko K., Pawlosky R., Carter E., Todd King M., Musa-Veloso K. Kinetics, safety and tolerability of (R)-3-hydroxybutyl (R)-3-hydroxybutyrate in health subjects. Regulatory Toxicology and Pharmacology. 2012;63(3):401–408. doi: 10.1016/j.yrtph.2012.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croteau E. Ketogenic medium chain triglycerides increase brain energy metabolism in Alzheimer's disease. Journal of Alzheimer's Disease. 2018;64(2):551–561. doi: 10.3233/JAD-180202. [DOI] [PubMed] [Google Scholar]
- Cunnane S., Nugent S., Roy M., Courchesne-Loyer A., Croteau E., Tremblay S. Brain fuel metabolism, aging, and Alzheimer's disease. Nutrition. 2011;27:3–20. doi: 10.1016/j.nut.2010.07.021. Elsevier Inc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunnane S. Can ketones help rescue brain fuel supply in later life? Implications for cognitive health during aging and the treatment of Alzheimer's disease. Frontiers in Molecular Neuroscience. 2016;9:53. doi: 10.3389/fnmol.2016.00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Cabo R. Effects of intermittent fasting on health, aging, and disease. New England Journal of Medicine. 2019;381(26):2541–2551. doi: 10.1056/NEJMra1905136. [DOI] [PubMed] [Google Scholar]
- Devi L., Raghavendran V., Prabhu B.M., Avadhani N.G., Anandatheerthavarada H.K. Mitochondrial import and accumulation of α-synuclein impair complex I in human dopaminergic neuronal cultures and Parkinson disease brain. Journal of Biological Chemistry. 2008;283:9089–9100. doi: 10.1074/jbc.M710012200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du H., Guo L., Yan S., Sosunov A.A., McKhann G.M., ShiDu Yan S. Early deficits in synaptic mitochondria in an Alzheimer's disease mouse model. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(43):18670–18675. doi: 10.1073/pnas.1006586107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edmond J. Capacity for substrate utilization in oxidative metabolism by neurons, astrocytes, and oligodendrocytes from developing brain in primary culture. Journal of Neuroscience Research. 1987;18(4):551–561. doi: 10.1002/jnr.490180407. [DOI] [PubMed] [Google Scholar]
- Farris W. Insulin-degrading enzyme regulates the levels of insulin, amyloid β-protein, and the β-amyloid precursor protein intracellular domain in vivo. Proceedings of the National Academy of Sciences of the United States of America. 2003;100(7):4162–4167. doi: 10.1073/pnas.0230450100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fortier M. A ketogenic drink improves brain energy and some measures of cognition in mild cognitive impairment. Alzheimer's & Dementia. 2019;15(5):625–634. doi: 10.1016/j.jalz.2018.12.017. [DOI] [PubMed] [Google Scholar]
- Gabandé-Rodríguez E., Keane L., Capasso M. Microglial phagocytosis in aging and Alzheimer's disease. Journal of Neuroscience Research. 2019;98:284–298. doi: 10.1002/jnr.24419. [DOI] [PubMed] [Google Scholar]
- Gabuzda D., Busciglio J., Chen L.B., Mabudaira P., Yankner B.A. Inhibition of energy metabolism alters the processing of amyloid precursor protein and induces a potentially amyloidogenic derivative. The Journal of Biological Chemistry. 1994;269(18):13623–13628. [PubMed] [Google Scholar]
- Gano L.B., Patel M., Rho J.M. Ketogenic diets, mitochondria, and neurological diseases. Journal of Lipid Research. 2014;55(11):2211–2228. doi: 10.1194/jlr.R048975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao S. Alpha-synuclein overexpression negatively regulates insulin receptor substrate 1 by activating mTORC1/S6K1 signaling. International Journal of Biochemistry and Cell Biology. 2015;64:25–33. doi: 10.1016/j.biocel.2015.03.006. Elsevier Ltd. [DOI] [PubMed] [Google Scholar]
- Giordano G., Kavanagh T.J., Costa L.G. Mouse cerebellar astrocytes protect cerebellar granule neurons against toxicity of the polybrominated diphenyl ether (PBDE) mixture DE-71. Neurotoxicology. 2009;30(2):326–329. doi: 10.1016/j.neuro.2008.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Arbelaez D. Body composition changes after very low-calorie-ketogenic diet in obesity evaluated by three standardized methods. The Journal of Clinical Endocrinology & Metabolism. 2016;102(2):488–498. doi: 10.1210/jc.2016-2385. Oxford University Press. [DOI] [PubMed] [Google Scholar]
- Hallberg S.J. Effectiveness and safety of a novel care model for the management of type 2 diabetes at 1 year: An open-label, non-randomized, controlled study. Diabetes Therapy: Research, Treatment and Education of Diabetes and Related Disorders. 2018;9(2):583–612. doi: 10.1007/s13300-018-0373-9. Springer Healthcare. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halliday G.M., Stevens C.H. Glia: Initiators and progressors of pathology in Parkinson's disease. Movement Disorders: Official Journal of the Movement Disorder Society. 2011;26(1):6–17. doi: 10.1002/mds.23455. [DOI] [PubMed] [Google Scholar]
- Helley M.P., Pinnell J., Sportelli C., Tieu K. Mitochondria: A common target for genetic mutations and environmental toxicants in Parkinson's disease. Frontiers in Genetics. 2017;8:177. doi: 10.3389/fgene.2017.00177. Frontiers Media S.A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heni M. Evidence for altered transport of insulin across the blood-brain barrier in insulin-resistant humans. Acta Diabetologica. 2014;51(4):679–681. doi: 10.1007/s00592-013-0546-y. [DOI] [PubMed] [Google Scholar]
- Hopkins S.J., Rothwell N.J. Cytokines and the nervous system. I: Expression and recognition. Trends in Neurosciences. 1995;18(2):83–88. [PubMed] [Google Scholar]
- Hu H., Tan C.C., Tan L., Yu J.T. A mitocentric view of Alzheimer's disease. Molecular Neurobiology. 2017;54:6046–6060. doi: 10.1007/s12035-016-0117-7. Humana Press Inc. [DOI] [PubMed] [Google Scholar]
- Hu M.T.M., Taylor-Robinson S.D., Chaudhuri K.R., Bell J.D., Labbé C., Cunningham V.J. Cortical dysfunction in non-demented Parkinson's disease patients—A combined 31 P-MRS and 18 FDG-PET study. Brain. 2000;123:340–352. doi: 10.1093/brain/123.2.340. [DOI] [PubMed] [Google Scholar]
- Hu M.T.M. A comparison of 18F-dopa PET and inversion recovery MRI in the diagnosis of Parkinson's disease. Neurology. 2001;56(9):1195–1200. doi: 10.1212/WNL.56.9.1195. [DOI] [PubMed] [Google Scholar]
- Huang C., Wang P., Xu X., Zhang Y., Gong Y., Hu W. The ketone body metabolite β-hydroxybutyrate induces an antidepression-associated ramification of microglia via HDACs inhibition-triggered Akt-small RhoGTPase activation. Glia. 2018;66(2):256–278. doi: 10.1002/glia.23241. [DOI] [PubMed] [Google Scholar]
- Imamura K., Hishikawa N., Sawada M., Nagatsu T., Yoshida M., Hashizume Y. Distribution of major histocompatibility complex class II-positive microglia and cytokine profile of Parkinson's disease brains. Acta Neuropathologica. 2003;106(6):518–526. doi: 10.1007/s00401-003-0766-2. [DOI] [PubMed] [Google Scholar]
- Kadenbach B., Hüttemann M. The subunit composition and function of mammalian cytochrome c oxidase. Mitochondrion. 2015;24:64–76. doi: 10.1016/j.mito.2015.07.002. [DOI] [PubMed] [Google Scholar]
- Kashiwaya Y., Bergman C., Lee J.H., Wan R., King M.T., Mughal M.R. A ketone ester diet exhibits anxiolytic and cognition-sparing properties, and lessens amyloid and tau pathologies in a mouse model of Alzheimer's disease. Neurobiology of Aging. 2013;34(6):1530–1539. doi: 10.1016/j.neurobiolaging.2012.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashiwaya Y., Takeshima T., Mori N., Nakashima K., Clarke K., Veech R.L. d-hydroxybutyrate protects neurons in models of Alzheimer's and Parkinson's disease. Proceedings of the National Academy of Sciences of the United States of America. 2000;97:5440–5444. doi: 10.1073/pnas.97.10.5440. www.pnas.org Retrieved from. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kesl S.L. Effects of exogenous ketone supplementation on blood ketone, glucose, triglyceride, and lipoprotein levels in Sprague–Dawley rats. Nutrition and Metabolism. 2016;13(1):9. doi: 10.1186/s12986-016-0069-y. BioMed Central. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krasemann S., Madore C., Cialic R., Baufeld C., Calcagno N., El Fatimy R. The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity. 2017;47(3):566–581.e9. doi: 10.1016/j.immuni.2017.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang C., Campbell K.R., Ryan B.J., Carling P., Attar M., Vowles J. Single-cell sequencing of iPSC-dopamine neurons reconstructs disease progression and identifies HDAC4 as a regulator of Parkinson cell phenotypes. Cell Stem Cell. 2019;24:1–14. doi: 10.1016/j.stem.2018.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lannert H., Hoyer S. Intracerebroventricular administration of streptozotocin causes long-term diminutions in learning and memory abilities and in cerebral energy metabolism in adult rats. Behavioral Neuroscience. 1998;112(5):1199–1208. doi: 10.1037//0735-7044.112.5.1199. [DOI] [PubMed] [Google Scholar]
- Lee J., Kim M.S. The role of GSK3 in glucose homeostasis and the development of insulin resistance. Diabetes Research and Clinical Practice. 2007;77(3 Suppl):S49–S57. doi: 10.1016/j.diabres.2007.01.033. [DOI] [PubMed] [Google Scholar]
- Lee H.K. The insulin/AKT signaling pathway is targeted by intracellular β-amyloid. Molecular Biology of the Cell. 2009;20(5):1533–1544. doi: 10.1091/mbc.E08-07-0777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lennerz B.S. Management of type 1 diabetes with a very low–carbohydrate diet. Pediatrics. 2018;141:e20173349. doi: 10.1542/peds.2017-3349. American Academy of Pediatrics. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li K., Li J., Zheng J., Qin S. Reactive astrocytes in neurodegenerative diseases. Aging and Disease. 2019;10(3):664–675. doi: 10.14336/AD.2018.0720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X.-H. Glycation exacerbates the neuronal toxicity of β-amyloid. Cell Death & Disease. 2013;4(6):e673. doi: 10.1038/cddis.2013.180. Nature Publishing Group. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liddelow S.A., Guttenplan K.A., Clarke L.E., Bennett F.C., Bohlen C.J., Schirmer L. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541(7638):481–487. doi: 10.1038/nature21029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. SIRT3 mediates hippocampal synaptic adaptations to intermittent fasting and ameliorates deficits in APP mutant mice. Nature Communications. 2019;10(1):1886. doi: 10.1038/s41467-019-09897-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maalouf M., Rho J.M., Mattson M.P. The neuroprotective properties of calorie restriction, the ketogenic diet, and ketone bodies. Brain Research Reviews. 2009;59(2):293–315. doi: 10.1016/j.brainresrev.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maalouf M., Schweizer F., Zhao M., Eisenberg D. Enhancing mitochondrial function with the ketones acetoacetate and β-hydroxybutyrate alleviates soluble Aβ42 toxicity. Alzheimer's & Dementia. 2011;7(4):S798. doi: 10.1016/j.jalz.2011.05.2301. [DOI] [Google Scholar]
- Magrané J. Intraneuronal β-amyloid expression downregulates the AKT survival pathway and blunts the stress response. Journal of Neuroscience. 2005;25(47):10960–10969. doi: 10.1523/JNEUROSCI.1723-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann V.M., Cooper J.M., Krige D., Daniel S.E., Schapira A.H.V., Marsden C.D. Brain, skeletal muscle and platelet homogenate mitochondrial function in Parkinson's disease. Brain. 1992;115:333–342. doi: 10.1093/brain/115.2.333. https://www.ncbi.nlm.nih.gov/pubmed/1606472 Retrieved from. [DOI] [PubMed] [Google Scholar]
- Marosi K., Kim S.W., Moehl K., Scheibye-Knudsen M., Cheng A., Cutler R. 3-Hydroxybutyrate regulates energy metabolism and induces BDNF expression in cerebral cortical neurons. Journal of Neurochemistry. 2016;139:769–781. doi: 10.1111/jnc.13868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattson M.P. Intermittent metabolic switching, neuroplasticity and brain health. Nature Reviews Neuroscience. 2018;19(2):63–80. doi: 10.1038/nrn.2017.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurer I., Zierz S., Möller H.-J. A selective defect of cytochrome c oxidase is present in brain of Alzheimer disease patients. Neurobiology of Aging. 2000;21:455–462. doi: 10.1016/s0197-4580(00)00112-3. www.elsevier.com/locate/neuaging Retrieved from. [DOI] [PubMed] [Google Scholar]
- McGeer P.L., Itagaki S., Boyes B.E., McGeer E.G. Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson's and Alzheimer's disease brains. Neurology. 1988;38(8):1285–1291. doi: 10.1212/wnl.38.8.1285. [DOI] [PubMed] [Google Scholar]
- Morris J.K. Insulin resistance impairs nigrostriatal dopamine function. Experimental Neurology. 2011;231(1):171–180. doi: 10.1016/j.expneurol.2011.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosconi L., Pupi A., De Leon M.J. Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer's disease. Annals of the New York Academy of Sciences. 2008;1147:180–195. doi: 10.1196/annals.1427.007. Blackwell Publishing Inc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosconi L., Brys M., Switalski R., Mistur R., Glodzik L., Pirraglia E. Maternal family history of Alzheimer's disease predisposes to reduced brain glucose metabolism. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(48):19067–19072. doi: 10.1073/pnas.0705036104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosconi L., De Leon M., Murray J., L E., Lu J., Javier E. Reduced mitochondria cytochrome oxidase activity in adult children of mothers with Alzheimer's disease. Journal of Alzheimer's Disease. 2011;27(3):483–490. doi: 10.3233/JAD-2011-110866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosconi L., Glodzik L., Mistur R., McHugh P., Rich K.E., Javier E. Oxidative stress and amyloid-beta pathology in normal individuals with a maternal history of Alzheimer's. Biological Psychiatry. 2010;68(10):913–921. doi: 10.1016/j.biopsych.2010.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mujica-Parodi L.R., Amgalan A., Sultan S.F., Antal B., Sun X., Skiena S. Diet modulates brain network stability, a biomarker for brain aging, in young adults. Proceedings of the National Academy of Sciences of the United States of America. 2020;117(11):6170–6177. doi: 10.1073/pnas.1913042117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray A.J. Novel ketone diet enhances physical and cognitive performance. The FASEB Journal. 2016;30(12):4021–4032. doi: 10.1096/fj.201600773R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neth B.J. Modified ketogenic diet is associated with improved cerebrospinal fluid biomarker profile, cerebral perfusion, and cerebral ketone body uptake in older adults at risk for Alzheimer's disease: A pilot study. Neurobiology of Aging. 2019;86:54–63. doi: 10.1016/j.neurobiolaging.2019.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman J.C., Verdin E. β-Hydroxybutyrate: Much more than a metabolite. Diabetes Research and Clinical Practice. 2014;106:173–181. doi: 10.1016/j.diabres.2014.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman J., Verdin E. Ketone bodies as signaling metabolites. Trends in Endocrinology and Metabolism. 2014;25:42–52. doi: 10.1016/j.tem.2013.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newport M.T. A new way to produce hyperketonemia: Use of ketone ester in a case of Alzheimer's disease. Alzheimer's & Dementia. 2015;11(1):99–103. doi: 10.1016/j.jalz.2014.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niccoli T. Increased glucose transport into neurons rescues Aβ toxicity in Drosophila. Current Biology. 2016;26:2291–2300. doi: 10.1016/j.cub.2016.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishida Y. Decreased glucose transporter 1 expression at the blood-brain barrier exacerbates Alzheimer disease-like phenotypes in mouse models. Journal of the Neurological Sciences. 2017;381:768. doi: 10.1016/j.jns.2017.08.2167. [DOI] [Google Scholar]
- Norwitz N.G., Hu M.T., Clarke K. The mechanisms by which the ketone body D-β-hydroxybutyrate may improve the multiple cellular pathologies of Parkinson's disease. Frontiers in Nutrition. 2019;6:63. doi: 10.3389/fnut.2019.00063. Frontiers. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norwitz N.G. Multi-loop model of Alzheimer disease: An integrated perspective on the Wnt/GSK3β, α-synuclein, and type 3 diabetes hypothesis. Frontiers in Aging Neuroscience. 2019;11:184. doi: 10.3389/fnagi.2019.00184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Neill C. PI3-kinase/AKT/mTOR signaling: Impaired on/off switches in aging, cognitive decline and Alzheimer's disease. Experimental Gerontology. 2013;48:647–653. doi: 10.1016/j.exger.2013.02.025. [DOI] [PubMed] [Google Scholar]
- Park J.S., Davis R.L., Sue C.M. Mitochondrial dysfunction in Parkinson's disease: New mechanistic insights and therapeutic perspectives. Current Neurology and Neuroscience Reports. 2018;18:21. doi: 10.1007/s11910-018-0829-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pawlosky R.J., Kemper M.F., Kashiwaya Y., King M.T., Mattson M.P., Veech R.L. Effects of a dietary ketone ester on hippocampal glycolytic and tricarboxylic acid cycle intermediates and amino acids in a 3xTgAD mouse model of Alzheimer's disease. Journal of Neurochemistry. 2017;141(2):195–207. doi: 10.1111/jnc.13958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pérez A. Degradation of soluble amyloid β-peptides 1-40, 1-42, and the Dutch variant 1-40Q by insulin degrading enzyme from Alzheimer disease and control brains. Neurochemical Research. 2000;25(2):247–255. doi: 10.1023/A:1007527721160. [DOI] [PubMed] [Google Scholar]
- Phillips E.C., Croft C.L., Kurbatskaya K., O'Neill M.J., Hutton M.L., Hanger D.P. Astrocytes and neuroinflammation in Alzheimer's disease. Biochemical Society Transactions. 2014;42(5):1321–1325. doi: 10.1042/BST20140155. [DOI] [PubMed] [Google Scholar]
- Pihlaja R., Koistinaho J., Kauppinen R., Sandholm J., Tanila H., Koistinaho M. Multiple cellular and molecular mechanisms are involved in human Aβ clearance by transplanted adult astrocytes. Glia. 2011;59(11):1643–1657. doi: 10.1002/glia.21212. [DOI] [PubMed] [Google Scholar]
- Pinto A., Bonucci A., Maggi E., Corsi M., Businaro R. Anti-oxidant and anti-inflammatory activity of ketogenic diet: New perspectives for neuroprotection in Alzheimer's disease. Antioxidants. 2018;7(5):63. doi: 10.3390/antiox7050063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pontzer H. Metabolic acceleration and the evolution of human brain size and life history. Nature. 2016;533(7603):390–392. doi: 10.1038/nature17654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiman E.M., Caselli R.J., Yun L.S., Chen K., Bandy D., Minoshima S. Preclinical evidence of Alzheimer's disease in persons homozygous for the ɛ4 allele for apolipoprotein E. New England Journal of Medicine. 1996;334(12):752–758. doi: 10.1056/nejm199603213341202. [DOI] [PubMed] [Google Scholar]
- Roberts M.N., Wallace M.A., Tomilov A.A., Zhou Z., Marcotte G.R., Tran D. A ketogenic diet extends longevity and healthspan in adult mice. Cell Metabolism. 2017;26(3):539–546. doi: 10.1016/j.cmet.2017.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocha E.M., De Miranda B., Sanders L.H. Alpha-synuclein: Pathology, mitochondrial dysfunction and neuroinflammation in Parkinson's disease. Neurobiology of Disease. 2018;109:249–257. doi: 10.1016/j.nbd.2017.04.004. [DOI] [PubMed] [Google Scholar]
- Rossi D., Volterra A. Astrocytic dysfunction: Insights on the role in neurodegeneration. Brain Research Bulletin. 2009;80(4–5):224–232. doi: 10.1016/j.brainresbull.2009.07.012. [DOI] [PubMed] [Google Scholar]
- Sato K., Kashiwaya Y., Keon C.A., Tsuchiya N., King M.T., Radda G.K. Insulin, ketone bodies, and mitochondrial energy transduction. The FASEB Journal. 1995;9:651–658. doi: 10.1096/fasebj.9.8.7768357. [DOI] [PubMed] [Google Scholar]
- Schröder J.B., Pawlowski M., Meyer zu Hörste G., Gross C.C., Wiendl H., Meuth S.G. Immune cell activation in the cerebrospinal fluid of patients with Parkinson's disease. Frontiers in Neurology. 2018;9:1081. doi: 10.3389/fneur.2018.01081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shao B.-Z., Cao Q., Liu C. Targeting NLRP3 inflammasome in the treatment of CNS diseases. Frontiers in Molecular Neuroscience. 2018;11:320. doi: 10.3389/fnmol.2018.00320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapira A.H. Mitochondrial complex I deficiency in Parkinson's disease. Journal of Neurochemistry. 1990;54:823–827. doi: 10.1111/j.1471-4159.1990.tb02325.x. [DOI] [PubMed] [Google Scholar]
- Sherrier M., Li H. The impact of keto-adaptation on exercise performance and the role of metabolic-regulating cytokines. The American Journal of Clinical Nutrition. 2019;110:562–573. doi: 10.1093/ajcn/nqz145. [DOI] [PubMed] [Google Scholar]
- Shimazu T., Hirschey M.D., Newman J., He W., Shirakawa K., Le Moan N. Suppression of oxidative stress by β-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science (New York, N.Y.) 2013;339(6116):211–214. doi: 10.1126/science.1227166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto-Mota A. Safety and tolerability of sustained exogenous ketosis using ketone monoester drinks for 28 days in healthy adults. Regulatory Toxicology and Pharmacology. 2019;109:104506. doi: 10.1016/j.yrtph.2019.104506. [DOI] [PubMed] [Google Scholar]
- Stewart W.K., Laura Fleming W. Features of a successful therapeutic fast of 382 days’ duration. Postgraduate Medical Journal. 1973;49:203–209. doi: 10.1136/pgmj.49.569.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart C.R., Stuart L.M., Wilkinson K., van Gils J.M., Deng J., Halle A. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nature Immunology. 2010;11(2):155–161. doi: 10.1038/ni.1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stubbs B.J. On the metabolism of exogenous ketones in humans. Frontiers in Physiology. 2017;8(Oct):1–13. doi: 10.3389/fphys.2017.00848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stubbs B.J. Gastrointestinal effects of exogenous ketone drinks are infrequent, mild and vary according to ketone compound and dose. International Journal of Sport Nutrition and Exercise Metabolism. 2019:1–23. doi: 10.1123/ijsnem.2019-0014. (Figure 1) [DOI] [PubMed] [Google Scholar]
- Su M. Mechanisms associated with type 2 diabetes as a risk factor for Alzheimer-related pathology. Molecular Neurobiology. 2019;56:5815–5834. doi: 10.1007/s12035-019-1475-8. [DOI] [PubMed] [Google Scholar]
- Swerdlow R.H., Khan S.M. A “mitochondrial cascade hypothesis” for sporadic Alzheimer's disease. Medical Hypotheses. 2004;63(1):8–20. doi: 10.1016/j.mehy.2003.12.045. [DOI] [PubMed] [Google Scholar]
- Takahashi M. Insulin receptor mRNA in the substantia nigra in Parkinson's disease. Neuroscience Letters. 1996;204(3):201–204. doi: 10.1016/0304-3940(96)12357-0. [DOI] [PubMed] [Google Scholar]
- Tanokashira D., Fukuokaya W., Taguchi A. Involvement of insulin receptor substrates in cognitive impairment and Alzheimer's disease. Neural Regeneration Research. 2019;14:1330–1334. doi: 10.4103/1673-5374.253535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theodore S., Cao S., McLean P.J., Standaert D.G. Targeted overexpression of human alpha-synuclein triggers microglial activation and an adaptive immune response in a mouse model of Parkinson disease. Journal of Neuropathology and Experimental Neurology. 2008;67(12):1149–1158. doi: 10.1097/NEN.0b013e31818e5e99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas H.E., Zhang Y., Stefely J.A., Veiga S.R., Thomas G., Kozma S.C. Mitochondrial complex I activity is required for maximal autophagy. Cell Reports. 2018;24(9):2404–2417.e8. doi: 10.1016/j.celrep.2018.07.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tieu K., Perier C., Caspersen C., Teismann P., Wu D.C., Du Yan S. d-β-hydroxybutyrate rescues mitochondrial respiration and mitigates features of Parkinson disease. Journal of Clinical Investigation. 2003;112:892–901. doi: 10.1172/JCI200318797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanitallie T.B., Nonas R.D., Di Ricco A., Boyar N.P., Hyams K., Heymsfield S.B. Treatment of Parkinson disease with diet-induced hyperketonemia: A feasibility study. Neurology. 2005;64:728–730. doi: 10.1212/01.WNL.0000152046.11390.45. [DOI] [PubMed] [Google Scholar]
- Veech R.L., Bradshaw P.C., Clarke K., Curtis W., Pawlosky R., King M.T. Ketone bodies mimic the life span extending properties of caloric restriction. IUBMB Life. 2017;69:305–314. doi: 10.1002/iub.1627. [DOI] [PubMed] [Google Scholar]
- Veech R.L., Todd King M., Pawlosky R., Kashiwaya Y., Bradshaw P.C., Curtis W. The “great” controlling nucleotide coenzymes. IUBMB Life. 2019;71:1–15. doi: 10.1002/iub.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent A.J., Gasperini R., Foa L., Small D.H. Astrocytes in Alzheimer's disease: Emerging roles in calcium dysregulation and synaptic plasticity. Journal of Alzheimer's Disease: JAD. 2010;22(3):699–714. doi: 10.3233/JAD-2010-101089. [DOI] [PubMed] [Google Scholar]
- Vlassenko A.G., Vaishnavi S.N., Couture L., Sacco D., Shannon B.J., Mach R.H. Spatial correlation between brain aerobic glycolysis and amyloid-β (Aβ) deposition. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(41):17763–17767. doi: 10.1073/pnas.1010461107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkins H.M., Swerdlow R.H. Amyloid precursor protein processing and bioenergetics. Brain Research Bulletin. 2017;133:71–79. doi: 10.1016/j.brainresbull.2016.08.009. Elsevier Inc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willette A.A. Association of insulin resistance with cerebral glucose uptake in late middle-aged adults at risk for Alzheimer disease. JAMA Neurology. 2015;72(9):1013–1020. doi: 10.1001/jamaneurol.2015.0613. American Medical Association. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Włodarek D. Role of ketogenic diets in neurodegenerative diseases (Alzheimer's disease and Parkinson's disease) Nutrients. 2019;11(1):169. doi: 10.3390/nu11010169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xilouri M., Brekk O.R., Stefanis L. Autophagy and alpha-synuclein: Relevance to Parkinson's disease and related synucleopathies. Movement Disorders. 2016;31:178–192. doi: 10.1002/mds.26477. John Wiley and Sons Inc. [DOI] [PubMed] [Google Scholar]
- Yaffe K. Diabetes, glucose control, and 9-year cognitive decline among older adults without dementia. Archives of Neurology. 2012;69(9):1170–1175. doi: 10.1001/archneurol.2012.1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi J.H. Direct pharmacological AKT activation rescues Alzheimer's disease like memory impairments and aberrant synaptic plasticity. Neuropharmacology. 2018;128:282–292. doi: 10.1016/j.neuropharm.2017.10.028. Elsevier Ltd. [DOI] [PubMed] [Google Scholar]
- Youm Y.-H., Nguyen K.Y., Grant R.W., Goldberg E.L., Bodogai M., Kim D. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nature Medicine. 2015;21(3):263–269. doi: 10.1038/nm.3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao N. Apolipoprotein E4 impairs neuronal insulin signaling by trapping insulin receptor in the endosomes. Neuron. 2017;96(1):115–129.e5. doi: 10.1016/j.neuron.2017.09.003. Cell Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
Further reading
- Arcuri C., Mecca C., Bianchi R., Giambanco I., Donato R. The pathophysiological role of microglia in dynamic surveillance, phagocytosis and structural remodeling of the developing CNS. Frontiers in Molecular Neuroscience. 2017;10:191. doi: 10.3389/fnmol.2017.00191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chriett S., Dąbek A., Wojtala M., Vidal H., Balcerczyk A., Pirola L. Prominent action of butyrate over β-hydroxybutyrate as histone deacetylase inhibitor, transcriptional modulator and anti-inflammatory molecule. Scientific Reports. 2019;9(1):1–14. doi: 10.1038/s41598-018-36941-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das T., Hoarau J.J., Jaffar Bandjee M.C., Maquart M., Gasque P. Multifaceted innate immune responses engaged by astrocytes, microglia and resident dendritic cells against Chikungunya neuroinfection. The Journal of General Virology. 2015;96(Pt. 2):294–310. doi: 10.1099/vir.0.071175-0. [DOI] [PubMed] [Google Scholar]
- Deora V., Albornoz E.A., Zhu K., Woodruff T.M., Gordon R. The ketone body β-hydroxybutyrate does not inhibit synuclein mediated inflammasome activation in microglia. Journal of Neuroimmune Pharmacology: The Official Journal of the Society on NeuroImmune Pharmacology. 2017;12(4):568–574. doi: 10.1007/s11481-017-9754-5. [DOI] [PubMed] [Google Scholar]
- Lenz K.M., Nelson L.H. Microglia and beyond: Innate immune cells as regulators of brain development and behavioral function. Frontiers in Immunology. 2018;9:698. doi: 10.3389/fimmu.2018.00698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepeta K., Lourenco M.V., Schweitzer B.C., Martino Adami P.V., Banerjee P., Catuara-Solarz S. Synaptopathies: Synaptic dysfunction in neurological disorders—A review from students to students. Journal of Neurochemistry. 2016;138(6):785–805. doi: 10.1111/jnc.13713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liddelow S.A., Barres B.A. Reactive astrocytes: Production, function, and therapeutic potential. Immunity. 2017;46(6):957–967. doi: 10.1016/j.immuni.2017.06.006. [DOI] [PubMed] [Google Scholar]
- Lohman R.-J., Iyer A., Fairlie T.J., Cotterell A., Gupta P., Reid R.C. Differential anti-inflammatory activity of HDAC inhibitors in human macrophages and rat arthritis. Journal of Pharmacology and Experimental Therapeutics. 2016;356(2):387–396. doi: 10.1124/jpet.115.229328. [DOI] [PubMed] [Google Scholar]
- Longo V.D., Mattson M.P. Fasting: Molecular mechanisms and clinical applications. Cell Metabolism. 2014;19(2):181–192. doi: 10.1016/j.cmet.2013.12.008. Elsevier Inc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monahan A.J., Warren M., Carvey P.M. Neuroinflammation and peripheral immune infiltration in Parkinson's disease: An autoimmune hypothesis. Cell Transplantation. 2008;17(4):363–372. [PubMed] [Google Scholar]
- Norwitz N.G., Hu M.T., Clarke K. 2020. Can a ketone drink improve exercise performance in patients with Parkinson's disease? ISRCTN16599164. [Google Scholar]
- Norwitz N.G., Hu M.T., Clarke K. 2020. Can a ketone drink increase ATP in the brains of patients with Parkinson's disease? ISRCTN10531043. [Google Scholar]
- Norwitz N.G., Hu M.T., Clarke K. 2020. Can a ketone drink reduce the severity of symptoms of Parkinson's disease? ISRCTN64294760. [Google Scholar]
- Robinson A.M., Williamson D.H. Physiological roles of ketone bodies as substrates and signals in mammalian tissues. Physiological Reviews. 1980;60(1):143–187. doi: 10.1152/physrev.1980.60.1.143. [DOI] [PubMed] [Google Scholar]
- Sarkar S., Malovic E., Harishchandra D.S., Ghaisas S., Panicker N., Charli A. Mitochondrial impairment in microglia amplifies NLRP3 inflammasome proinflammatory signaling in cell culture and animal models of Parkinson's disease. NPJ Parkinson's Disease. 2017;3:30. doi: 10.1038/s41531-017-0032-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinhauser M.L. The circulating metabolome of human starvation. JCI Insight. 2018;3(16) doi: 10.1172/jci.insight.121434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taggart A.K.P. D-β-hydroxybutyrate inhibits adipocyte lipolysis via the nicotinic acid receptor PUMA-G. The Journal of Biological Chemistry. 2005;280(29):26649–26652. doi: 10.1074/jbc.c500213200. [DOI] [PubMed] [Google Scholar]
- Troncoso-Escudero P., Parra A., Nassif M., Vidal R.L. Outside in: Unraveling the role of neuroinflammation in the progression of Parkinson's disease. Frontiers in Neurology. 2018;9:860. doi: 10.3389/fneur.2018.00860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueno M., Fujita Y., Tanaka T., Nakamura Y., Kikuta J., Ishii M. Layer V cortical neurons require microglial support for survival during postnatal development. Nature Neuroscience. 2013;16(5):543–551. doi: 10.1038/nn.3358. [DOI] [PubMed] [Google Scholar]
- Ulland T.K., Song W.M., Huang S.C.-C., Ulrich J.D., Sergushichev A., Beatty W.L. TREM2 maintains microglial metabolic fitness in Alzheimer's disease. Cell. 2017;170(4):649–663.e13. doi: 10.1016/j.cell.2017.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veech R.L. The therapeutic implications of ketone bodies: The effects of ketone bodies in pathological conditions: Ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins, Leukotrienes and Essential Fatty Acids. 2004;70(3):309–319. doi: 10.1016/j.plefa.2003.09.007. [DOI] [PubMed] [Google Scholar]
- Volmar C.-H., Wahlestedt C. Histone deacetylases (HDACs) and brain function. Neuroepigenetics. 2015;1:20–27. doi: 10.1016/j.nepig.2014.10.002. [DOI] [Google Scholar]