Abstract
Sarcoidosis and tuberculosis share several similar clinical and pathogenic characteristics that make some researchers consider a common pathogenesis for these diseases. Human leukocyte antigen (HLA) genotypes are studied both in sarcoidosis and tuberculosis patients, but to our knowledge, there are no comparative studies of genetic predisposition for sarcoidosis and tuberculosis development.
The aim of this review was to analyse the relationship between HLA genotypes and the development of sarcoidosis and tuberculosis. Original and review articles published in various online databases from 1960 to 2019 were studied.
The search results showed opposite effects of the HLA genotypes on predisposition to sarcoidosis or tuberculosis. It was revealed that the genotypes predisposing to the development of sarcoidosis (HLA-DRB1*03/07/15) have protective properties against the development of tuberculosis. Moreover, genotypes causing the development of tuberculosis (HLA-DRB1*04) have a protective effect on the development of sarcoidosis.
The results of this narrative review of the literature may allude to the existence of genetic predispositions that lead to the development of an antibacterial or autoimmune response to mycobacteria.
Short abstract
The HLA-DRB1*03/07/15 genotypes predispose to the development of sarcoidosis and have protective properties against the development of tuberculosis, while the HLA-DRB1*04 genotype has an opposite effect on the development of these diseases https://bit.ly/2Tl2rj1
Introduction
Sarcoidosis and tuberculosis (TB) are granulomatous diseases that affect various organs and share some clinical and pathogenic similarities, and because of this, doctors and scientists have long suggested the possibility of a common pathogenic mechanism for these diseases [1].
It is recognised that sarcoidosis is an autoimmune disease triggered by different inorganic (dust, paint, vaccinations) and infection factors (viruses, bacteria, fungi), among which the greatest attention is paid to mycobacterial infections [2]. The signs of mycobacterial infection, such as structural cell wall elements and nucleic acids, have been repeatedly described in sarcoid granulomas [3, 4]. Also, antibodies against bacterial proteins and mononuclear cell activation after incubation with bacterial antigens have been observed in patients with sarcoidosis [5, 6]. Because of the absence of active TB infection in these patients, these findings can indicate a possible previous mycobacterial infection, which was suggested by Scadding [7] in 1960. The ability of bacteria to cause autoimmune inflammation is explained by a possible mimicry of bacterial proteins (p36 proteins, heat shock proteins HSP65, and HSP7, ESAT-6 and KatG enzymes) with human autoantigens (tubulin, desmin, vimentin) [8]. The cross-reactivity of anti-TB antibodies and anti-DNA autoantibodies obtained in patients with systemic lupus erythematosus was also shown by Shoenfeld et al. [9]. Anti-TB antibodies were found to react with ssDNA, dsDNA, and other polynucleotides, whereas anti-DNA autoantibodies bound to three glycolipids shared among all mycobacteria and derived from the mycobacterial cell wall.
A similar clinical, radiological picture, as well as similar pathogenic pathways, can indicate a relationship between sarcoidosis and TB. Mycoplasma tuberculosis initiates the processes of productive inflammation in TB, which causes the formation of granulomatous inflammation in affected organs [1]. In sarcoidosis, granuloma formation is also described, induced by the influence of trigger factors and various microorganisms, including M. tuberculosis. However, sarcoid granulomas are lacking severe caseous necrosis, which is a specific criterion for the differential diagnosis of diseases [10]. Unfortunately, this difference in the histological picture leads to a large number of errors in the differentiation of diagnoses [11, 12]. A variety of immunological studies that have determined the activity of M. tuberculosis have also provided estimations of the diagnosis. The significant difference in the results of immunological tests (ELISPOT, QuantiFERON TB test, Diaskintest) has been described, with 80–94% negative results in sarcoidosis and predominantly positive results in TB, even without bacterial excretion [13]. It can be assumed, that M. tuberculosis, being an aetiological factor of TB, might also be one of the infectious triggers in sarcoidosis. The severity of inflammation depends on the immunogenetic characteristics of the macro-organism and the human leukocyte antigen (HLA) system is the main coordinator of the development of both autoimmune and infectious types of inflammation [14]. Gene polymorphisms of the HLA system are the most studied risk factors [15]. These genes encode major histocompatibility complex (MHC) molecules presenting antigens on the cell surface to T-lymphocytes. The HLA system is one of the first to come into contact with foreign antigens, which explains its influence on the development of the subsequent immune response, particularly in autoimmune processes [16].
This review aims to summarise recent genetic and immunological literature, describing the relationship between HLA genotypes and the development of sarcoidosis and TB.
Methods
Original and review articles indexed in the online databases Medline/PubMed, Scopus and ResearchGate from 1970 to 2019 were studied.
The initial selection of articles was based on the keywords: sarcoidosis, Löfgren's syndrome, TB, pulmonary TB, HLA genes, genetic predisposition. The inclusion criteria for the original articles were publications describing the study design and the results of HLA genotyping of adults and children aged 0–18 years without HIV infection, with an active/chronic form of sarcoidosis and pulmonary TB.
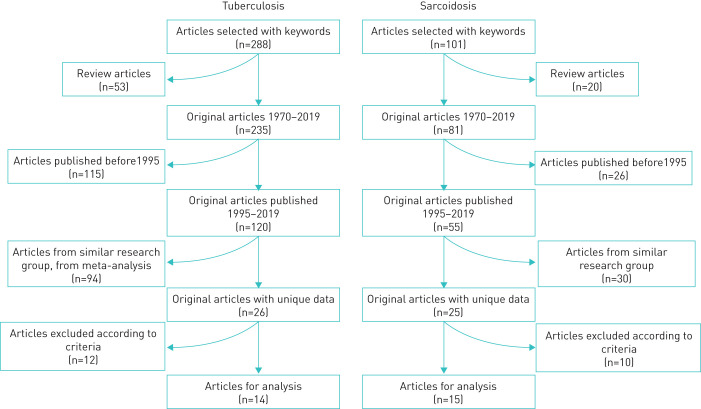
In total, 388 publications were selected by keywords, of which 288 described the immunogenetic studies of TB and 100 publications described sarcoidosis (figure 1).
FIGURE 1.
Study design diagram.
Literature reviews (n=73) and publications prior to 1995 (n=141) were excluded from the analysis because of the serological methods of HLA-DR/DQ genotyping. Among several publications from one group of authors, articles with the most complete information and a large number of analysed parameters were selected. Articles used in meta-analyses were excluded. In total, data were subsequently analysed in 28 publications (14 for TB and 15 for sarcoidosis).
According to the data presented in the publications, the examination of TB patients included: analysis of clinical symptoms, radiography, laboratory, and bacteriological examination. If necessary, in order to verify the diagnosis and conduct differential diagnosis, a histological method for examining diagnostic material was used.
Statistical analysis of the data was not performed due to the small number of studies.
The results of the analysis
According to the studied sources and several meta-analyses, the presence of the HLA-DRB1*04 and HLA-DRB1*08 genotypes is most typical for TB, while a protective function was particularly noted with the HLA-DRB1*03 and HLA-DRB1*07 genotypes [17, 18]. It is important to stress the point that genotype distribution strongly varies among different ethnic groups. Thus, the HLA-DRB1*16 genotype is a predisposing factor for TB development in Polish people, but a protective effect was found in people from Central India [19].
Several studies have established the relationship of different HLA genotypes with the development of active or chronic forms of TB. For the active form of TB, the presence of the HLA-DRB1*01.04 genotypes are more typical, whereas for the chronic form, the combinations of HLA-DRB1*16 with HLA-DQB1*05/03 and HLA-DRB1*04 with HLA-DQB1*02 are more prevalent [20].
The results of HLA-DRB1, DQB1-genotyping in studied publications are presented in table 1.
TABLE 1.
HLA-DRB1, DQB1 genotypes in various forms of tuberculosis
| First author, year [ref.] | Patients with lung TB | Method of genotyping | The statistically approved HLA genotypes | Country | |
| Of predisposition | Protective | ||||
| Archakova, 2008 [20] | Adults (307) | PCR-SSP | DRB1*04/ 16 | DRB1*03/07/15 | Russia |
| Pavlova, 2003 [21] | Adults (114) | PCR-SSP | DRB1*04/16 | DQB1*03 | Russia |
| Duarte, 2011 [22] | Adults (92) | PCR-SSP | DRB1*14 | - | Portugal |
| Amirzargar, 2004 [19] | Adults (40) | PCR-SSP | DRB1*07 | DRB1*03/05/15 | Iran |
| Dubaniewicz, 2005 [23] | Adults (61) | PCR-SSP | DRB1*14/16 | DQB1*02 | Poland |
| Kim, 2005 [24] | Adults (160) | PCR-SSO, PCR-SSP | DRB1*08.03 | DRB1*10 | Korea |
| Terán-Escandón, 1999 [25] | Adults (50) | PCR-SSP | DRB1*15/01 | DRB1*08/44 | Mexico |
| Vejbaesya, 2002 [26] | Adults (82) | PCR-SSO | DRB1*16.02 | DRB1*07.01 | Thailand |
| Starshinova, 2018 [27] | Children (n=98) | PCR-SSP | DRB1*04 | DRB1*07 | Russia |
| Wamala, 2016 [28] | Adults (n=43) | PCR-SSP | - | DQB1*03.03 | Uganda |
| Toyo-Oka, 2017 [29] | Adults (n=682) | PCR-SSP | DRB1*09.01 | - | Thailand |
| Wu, 2013 [31] | Adults (n=231) | PCR-SSP | DRB1*04 | - | China |
| de Lima, 2016 [31] | Adults (n=316) | PCR-SSP | DRB1*04 | - | Brazil |
| Wang, 2001 [32] | Adults (n=74) | PCR-SSP | DRB1*15 | DRB1*11 | China |
TB: tuberculosis; HLA: human leukocyte antigen; PCR-SSP: single specific primer PCR; PCR-SSO: sequence-specific oligonucleotide PCR.
In sarcoidosis, a relationship between the development of the disease and HLA genotypes of classes I, II, and III was detected. The carrying of the HLA-DRB1 and HLA-DQB1 genotypes also turned out to be most significant (table 2). In addition, an association between a genotype and the form of the disease was found. For Löfgren's syndrome, the presence of the HLA-DRB1*01.03 or HLA-DQB1*02.01 genotypes was more typical, whereas for the chronic form, HLA-DRB1*07/14/15 or HLA-DQB1*06.02 was found. A protective effect has been stated for the HLA-DRB1*01/04 genotypes.
TABLE 2.
HLA genotypes in sarcoidosis
| HLA genes | ||
| HLA I | HLA-B*07/08, HLA-A3 |
|
| HLA II | Löfgren's syndrome | HLA-DRB1*01/03, HLA-DQB1*02.01 |
| Chronic form | HLA-DQB1*06.02, HLA-DRB1*07/14/15 |
|
| Protective effect | HLA-DRB1*01/04 | |
| HLA III | BTNL2, C4, C6orf10, HSPA1L, LTA, NOTCH4, TAP2, TNF, VEGF | |
HLA: human leukocyte antigen.
The results of HLA-DRB1, DQ1 genotyping in studied publications are presented in table 3.
TABLE 3.
HLA-DRB1, DQB1 genotypes in various forms of sarcoidosis
| First author, year [ref.] | Patients with sarcoidosis and Löfgren's syndrome | Method of genotyping | The statistically approved HLA genotypes | Country | |
| Of predisposition | Protective | ||||
| Berlin, 1997 [33] | Sarcoidosis (n=122) | PCR-SSP | DRB1*03/17 | - | Scandinavia |
| Löfgren's syndrome (n=34) | DRB1*17 | - | |||
| Chronic form (n=57) | DR B1*14/15 | - | |||
| Bogunia-Kubik, 2001 [32] | Sarcoidosis (n=53) | PCR-SSP | DRB1*03 | DRB1*11 | Poland |
| Planck, 2002 [35] | Löfgren's syndrome (n=19) | PCR-SSP | DR B1*17 | - | Scandinavia |
| Foley, 2001 [36] | Sarcoidosis (n=345) | PCR-SSP | DRB1*03 | DRB1*01/04 | UK, Poland, Czech Republic |
| Grunewald, 2010 [37] | Sarcoidosis (n=724) | PCR-SSP | DRB1*03/14/15 | DRB1*01/03/07 | Scandinavia |
| Rossman, 2003 [38] | Sarcoidosis (n= 948) | PCR-SSP | DRB1*11.01/15.01/12.01 | DRB1*04.01/04.04/04.07/15.03 | USA |
| black | DRB1*11.01/1201 | DRB1*15.03 | |||
| white | DRB1*11.01/04.02/15.01 | DRB1*04.01 | |||
| Darlington, 2011 [39] | Patients with sarcoidosis (n=1000) out of whom had symptoms associated with HS (Heerfordt's syndrome) (n=83) | PCR-SSP | DRB1*04 - uveitis | DRB1*04 - system sarcoidosis | Sweden |
| Levin, 2015 [40] | Sarcoidosis (n=1277) | Illumina HumanOmni1-Quad | DRB1*12/11 | DRB1*03 | African Americans |
| da Costa, 2013 [41] | Sarcoidosis (n=63) | QuantiGene Plex assay | DRB1*14 | DRB1*15/16 | Brazil |
| Mortaz, 2015 [42] | Pulmonary sarcoidosis (n=51), Extrapulmonary sarcoidosis (n=39) | PCR-SSP | DRB1 *7 – pulmonary, DRB1 *12 - Extrapulmonary |
- | Iran |
| Ozyilmaz, 2014 [43] | Pulmonary sarcoidosis (n=86), Extrapulmonary sarcoidosis (n=46) | PCR-SSP | DRB1*15 | DRB1*11 - extrapulmonary | |
| Sharma, 2003 [44] | Sarcoidosis (n=56) | PCR-SSP | DRB1*11/14 (chronic form) | DRB1*07 | India |
| Voorter, 2005 [45] | Sarcoidosis (n=149) | PCR-SSP | DRB1*15.01/01 – severe form | - | Netherlands |
| Yanardag, 2017 [46] | Sarcoidosis (n=74) | PCR-SSP | DRB1*07/14/15 | - | Turkey |
| Papadopoulos, 2006 [47] | Sarcoidosis (n=66) | PCR-SSP | DRB1*02/14 | - | Germany |
HLA: human leukocyte antigen; PCR-SSP: single specific primer PCR.
There are several studies revealing genetic predisposition for sarcoid granuloma formation in various organs. Thus, HLA-DRB1*04/15 alleles carriers are more prone to developing sarcoidosis of the cardiovascular system, and the HLA-DRB1*04 alleles are more likely to cause uveitis [49].
Also, the association between ethnicity and sarcoidosis is worth mentioning (among ethnic groups, such as African-American and European people with a high prevalence of sarcoidosis, various HLA genotypes were found) [48, 49].
The presence of the HLA-DRB1*11.01 genotype increased the disease risk in both races, whereas the HLA-DRB1*12.01/15.03 genotypes were more typical for African-American people, and HLA-DRB1*15.01/04.01 for white people. Moreover, the HLA-DRB1*03.01 genotype is a predisposing factor for the development of sarcoidosis for European people; for African-American people, this genotype is of protective value [38].
Discussion
The study on the HLA genotypes distribution in patients with TB and sarcoidosis showed that the acute form of TB most often occurs with the HLA-DRB1*04 genotypes; the chronic form is associated with a combination of HLA-DRB1*16 with HLA-DQB1*05/03, or HLA -DRB1*04 with HLA-DQB1*02. A protective effect was characteristic for the HLA-DRB1*3/7/15 genotypes. The most significant genotypes affecting the development of sarcoidosis were HLA-DRB1 and HLA-DQB1. Many studies have shown that the development of Löfgren's syndrome is associated with the HLA-DRB1*01.03 or HLA-DQB1*02.01 genotypes, and the development of a chronic form is associated with genotypes HLA-DRB1*07/14/15 or HLA-DQB1*06.02. The HLA-DRB1*01/04 genotype has a protective effect.
Similar results were obtained by a group of researchers from Poland. According to their data, HLA-DRB1*15 is statistically more common in sarcoidosis than in TB, whereas relatively healthy individuals among patients with sarcoidosis were more often found to have HLA-DRB1*3/11 genotypes. The HLA-DRB1*04/08 and 16 genotypes were determined much less frequently in comparison to the control group and the HLA-DRB1*14 genotype, in comparison with TB patients. Analysis of the genotypes associated with a predisposition to TB showed a positive association with HLA-DRB1*14 and 16 alleles and a negative association with HLA-DRB1*04 genotype in comparison with healthy individuals. A comprehensive analysis of the results revealed that for the Polish population, the HLA-DRB1*11 genotype predisposes to the development of sarcoidosis and is protective for the development of TB, whereas the HLA-DRB1*14 genotype has the opposite effect [50, 51].
Despite conflicting and ambiguous results derived from the studies on the genetic predisposition to TB and sarcoidosis, our data analysis revealed another genotype distribution pattern for these diseases. The HLA-DRB1*03/07/15 genotypes are predisposing risk factors for the development of sarcoidosis, whereas the protective effect of these genotypes is revealed in TB. As for the HLA-DRB1*04 genotype, this was associated with the development of TB, whereas a protective effect was revealed for sarcoidosis (table 4).
TABLE 4.
HLA-DRB1 genotypes in sarcoidosis and tuberculosis
| Disease | Susceptibility | Protective effect |
| Sarcoidosis | HLA-DRB1*03/07/15 | HLA-DRB1*04 |
| Tuberculosis | HLA-DRB1*04 | HLA-DRB1*03/07/15 |
The differences observed when comparing the data shown in table 4 may be associated with the molecular structure of the HLA proteins. According to some studies of binding activity based on the silicate analysis in the HLA-DRB1*04 genotype, MHC molecules have a weak affinity for mycobacterial antigens, whereas in the HLA-DRB1*03.01 genotype, there was a high affinity of MHC proteins for bacterial antigens [31]. A previous in silico analysis found that patients with Löfgren's syndrome express HLA-DR alleles capable of binding a significantly higher number of bacterial epitopes than other HLA-DR alleles [52].
Interestingly, DRB1*03.01, shows the highest predicted binding to M. tuberculosis epitopes [53]. According to silicate binding analysis, The HLA-DRB1*03.01 allele consistently demonstrated the highest binding affinities for all six peptides, while HLA-DRB1*03.02 had relatively strong binding affinities for the proteins SodA (M. tuberculosis). The authors suggested that this difference in binding may be due to the difference in amino acid residues 28 and 86, which determined the ability to bind HSP 3–13 and HSP 4–15 [54, 55].
Contini et al. [56] revealed a relationship between the affinity for mycobacterial proteins and the type HLA-DRB1. It turned out that with genotypes characteristic of the development of TB (HLA-DRB1*08.01, *08.02, *14.01, *15.01 and *15.02), the affinity for proteins is lower than with other genotypes HLA-DRB1 *03.01, *07.01, *11.01, *11.02, *13.01 and *13.02, among which one can distinguish genotype characteristics for the development of sarcoidosis.
Moreover, studies of the activation of T-lymphocytes by mycobacterial proteins showed that the culture filtrate protein of 10 mycobacteria induces the release of interferon (IFN) and cytotoxic reactions in CD4+ T-lymphocytes with DRB1*04 molecules [57].
Interesting results were obtained by Selvaraj et al. [58]. In response to live M. tuberculosis and culture filtrate antigen in patients with TB, the level of pro-inflammatory cytokine interleukin (IL)-6 increased with the DRB1*04 genotype, the level of IFN-γ decreased with the HLA-DRB1*15 genotype, whereas among the group in HLA-DRB1*03 representatives, IFN-γ levels increased. However, researchers find it difficult to explain the results.
Perhaps the HLA-DRB1*03 molecule, having a greater affinity for proteins, triggers a stronger immune response, which leads to auto-inflammatory processes, while the HLA-DRB1*04 molecule with moderate affinity properties activates the antimicrobial immune response. Such hyperactivation can be seen in the pathogenesis of coeliac disease, a chronic autoimmune disease that affects the small intestine. In representatives of the HLA-DQ2/8 genotypes, the molecules have high affinity for gliadin, which leads to hyperactivation of the immune response and the development of autoimmune reactions [59].
Thus, it can be assumed that depending on the HLA-DRB1 genotypes the antibacterial inflammation and/or autoimmune processes may characterise the immune response in mycobacterial infection.
However, it is worth noting that autoimmune reactions occur in TB patients. For example, there are autoimmune reactions, such as granulomatosis with polyangiitis, arthritis, and uveitis, which are also detected in TB [60]. The presence of an autoimmune process in TB may be confirmed by studies showing the presence of autoantibodies in patients with TB. Antibodies to anti-cardiolipin IgG, anti-b2 glycoprotein IgG, anti-prothrombin, anti-proteinase 3 and anti-neutrophil cytoplasmatic antibodies decreased after anti-TB treatment [61]. Unfortunately, there are no data on the immunogenetic characteristics of TB patients who showed signs of autoimmune inflammation, which could help to determine the role of various genotypes in the development of the immune response in contact with the M. tuberculosis.
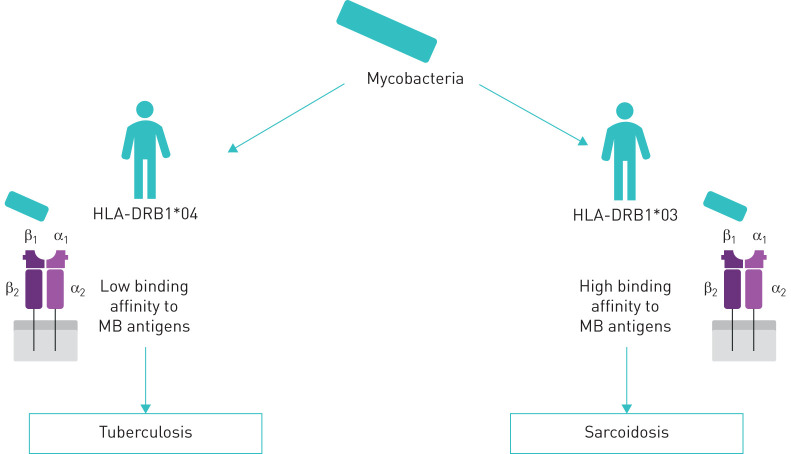
According to our point of view, mycobacterial antigens of the HLA-DRB1*03/07/15 genotypes carriers are triggers developing sarcoid reactions with an autoimmune component, while the HLA-DRB1*04 genotype is associated with a high risk of developing TB, as presented in schematic form in figure 2.
FIGURE 2.
Predisposition to sarcoidosis or tuberculosis development according to HLA-DRB1 genotypes. MB: mycobacterial.
Similar results were found when studying the role of the Epstein–Barr virus (EBV) in the development of autoimmune diseases in individuals with different alleles of the HLA-DRB1 genotype. It is considered that antigens of the EBV can cause the development of autoimmune reactions by the mechanism of molecular mimicry. For example, an autoimmune reaction in multiple sclerosis was detected in HLA-DRB1*15 genotype carriers against myelin basic protein, which has a structure similar to the EBNA-1 virus protein [62]. For carriers of HLA-DRB1*04 an increased risk of developing rheumatoid arthritis with EBV infection was found, which is associated with the molecular similarity of the virus glycoprotein-110 and the QKRAA sequence located in DRB1*04.01 [63] The HLA-DRB1*04.05 genotype has a protective effect on the development of typhoid fever caused by Salmonella enterica, but it is a predisposing factor for the development of rheumatoid arthritis in the Asian population [64].
Conclusion
Thus, the HLA system and specific properties of external factors, determine the development of the immune response when a person comes in contact with this factor. Determining HLA genotypes may result in assessing a risk degree for developing TB or sarcoidosis in the foci of mycobacterial infection and forecasting a possible course of the disease, thus providing more effective preventive measures and future prospects for differential diagnosis.
The study of immunogenetic characteristics, with respect to the development of TB or sarcoidosis, is a very promising area and needs further investigation to obtain more accurate data necessary for making practical recommendations.
Footnotes
Conflict of interest: A. Malkova has nothing to disclose.
Conflict of interest: A. Starshinova has nothing to disclose.
Conflict of interest: Y. Zinchenko has nothing to disclose.
Conflict of interest: N. Basantsova has nothing to disclose.
Conflict of interest: V.A. Mayevskaya has nothing to disclose.
Conflict of interest: P. Yablonskiy has nothing to disclose.
Conflict of interest: Y. Shoenfeld has nothing to disclose.
Support statement: This work was supported by a grant from the Government of the Russian Federation (contract No. 14.W03.31.0009 dated February 13, 2017) on the allocation of grants for the state support of scientific researches conducted under the guidance of leading scientists. Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Mortaz E, Masjedi MR, Abedini A, et al. Common features of tuberculosis and sarcoidosis. Int J Mycobacteriol 2016; 5: S240–S241. doi: 10.1016/j.ijmyco.2016.09.031 [DOI] [PubMed] [Google Scholar]
- 2.Bindoli S, Dagan A, Torres-Ruiz JJ, et al. Sarcoidosis and autoimmunity: from genetic background to environmental factors. Isr Med Assoc J 2016; 18: 197–202. [PubMed] [Google Scholar]
- 3.Moscovic EA. Sarcoidosis and mycobacterial l-forms: a critical reappraisal of pleomorphic chromogenic bodies (Hamazaki corpuscles) in lymph nodes. Pathol Annu 1978; 13: 69–164. [PubMed] [Google Scholar]
- 4.Gupta D, Agarwal R, Aggarwal AN, et al. Molecular evidence for the role of mycobacteria in sarcoidosis: a meta-analysis. Eur Respir J 2007; 30: 508–516. doi: 10.1183/09031936.00002607 [DOI] [PubMed] [Google Scholar]
- 5.Dubaniewicz A, Dubaniewicz-Wybieralska M, Sternau A, et al. Mycobacterium tuberculosis complex and mycobacterial heat shock proteins in lymph node tissue from patients with pulmonary sarcoidosis. J Clin Microbiol 2006; 44: 3448–3451. doi: 10.1128/JCM.01433-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Drake WP, Dhason MS, Nadaf M, et al. Cellular recognition of Mycobacterium tuberculosis ESAT-6 and KATg peptides in systemic sarcoidosis. Infect Immun 2007; 75: 527–530. doi: 10.1128/IAI.00732-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Scadding JG. Mycobacterium tuberculosis in the aetiology of sarcoidosis. BMJ 1960; 2: 1617–1623. doi: 10.1136/bmj.2.5213.1617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Elkington P, Tebruegge M, Mansour S. Tuberculosis: an infection-initiated autoimmune disease? Trends Immunol 2016; 37: 815–818. doi: 10.1016/j.it.2016.09.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shoenfeld Y, Vilner Joyce C, Lavie G, et al. Monoclonal anti-tuberculosis antibodies react with DNA, and monoclonal anti-DNA autoantibodies react with Mycobacterium tuberculosis. Clin Exp Immunol 1986; 66: 255–261. [PMC free article] [PubMed] [Google Scholar]
- 10.Lewinsohn DM, Leonard MK, LoBue PA, et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention clinical practice guidelines: diagnosis of tuberculosis in adults and children. Clin Infect Dis 2017; 64: 111–115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wiesel AA, Guryleva ME. Sarcoidosis: an introduction to the problem and the history of learning. Pract Pulmonol 2001; 2: 10–11. [Google Scholar]
- 12.Pavlova MV, Belokurov MA, Chernokhaeva IV, et al. Difficulties in differential diagnostics of respiratory tuberculosis and sarcoidosis. Tuberc Lung Dis 2016; 94: 57–61 (in Russian). [Google Scholar]
- 13.Belokurov MА, Starshinova AА, Zhuravlev VY, et al. Immunological methods in diagnosis of sarcoidosis end tuberculosis of lung. J Infectol 2015; 7: 98–104 (in Russian). doi: 10.22625/2072-6732-2015-7-2-98-104 [DOI] [Google Scholar]
- 14.Dubaniewicz A, Zimmermann A, Smigielska M, et al. Sarcoidosis and tuberculosis: a connection to the human leukocyte antigen system. Adv Exp Med Biol 2013; 756: 229–237. doi: 10.1007/978-94-007-4549-0_29. [DOI] [PubMed] [Google Scholar]
- 15.Arango M, Perricone C, Kivity S, et al. HLA-DRB1 the notorious gene in the mosaic of autoimmunity. J Immunol Res 2017; 65: 82–98. doi: 10.1007/s12026-016-8817-7 [DOI] [PubMed] [Google Scholar]
- 16.Sung YS. The HLA system: genetics, immunology, clinical testing, and clinical implications. Yonsei Med J 2007; 48: 11–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen BF, Wang R, Chen YJ, et al. Association between HLA-DRB1 alleles and tuberculosis: a meta-analysis. Genet Mol Res 2015; 14: 15859–15868. doi: 10.4238/2015.December.1.37 [DOI] [PubMed] [Google Scholar]
- 18.Oliveira-Cortez A, Melo AC, Chaves VE, et al. Do HLA class II genes protect against pulmonary tuberculosis? A systematic review and mete-analysis. Eur J Clin Microbiol Infect Dis 2016; 35: 1567–1580. doi: 10.1007/s10096-016-2713-x [DOI] [PubMed] [Google Scholar]
- 19.Amirzargar AA, Yalda A, Hajabolbaghi M, et al. The association of HLA-DRB, DQA1, DQB1 alleles and haplotype frequency in Iranian patients with pulmonary tuberculosis. Int J Tuberc Lung Dis 2004; 8: 1017–1021. [PubMed] [Google Scholar]
- 20.Archakova LI. Improving therapy based on the study of immunogenetic factors in the formation of pulmonary tuberculosis. Rus Immune J 2008; 11: 188–189. [Google Scholar]
- 21.Pavlova MV, Archakova LI, Sapozhnikova NV, et al. The value of the immunogenetic characteristics of the patient in the clinical course pulmonary tuberculosis. Med Alliance 2003; 2: 27–41. [Google Scholar]
- 22.Duarte R, Carvalho C, Pereira C, et al. HLA class II alleles as markers of tuberculosis susceptibility and resistance. Rev Port Pneumol 2011; 17: 15–19. doi: 10.1016/S0873-2159(11)70005-8 [DOI] [PubMed] [Google Scholar]
- 23.Dubaniewicz A, Moszkowska G, Szczerkowska Z. Frequency of DRB1-DQB1 two-locus haplotypes in tuberculosis: preliminary report. Tuberculosis (Edinb) 2005; 85: 259–267. doi: 10.1016/j.tube.2004.12.003 [DOI] [PubMed] [Google Scholar]
- 24.Kim HS, Park MH, Song EY, et al. Association of HLA-DR and HLA-DQ genes with susceptibility to pulmonary tuberculosis in Koreans: preliminary evidence of associations with drug resistance, disease severity, and disease recurrence. Hum Immunol 2005; 66: 1074–1081. doi: 10.1016/j.humimm.2005.08.242 [DOI] [PubMed] [Google Scholar]
- 25.Terán-Escandón D, Terán-Ortiz L, Camarena-Olvera A, et al. Human leukocyte antigen-associated susceptibility to pulmonary tuberculosis: molecular analysis of class II alleles by DNA amplification and oligonucleotide hybridization in Mexican patients. Chest 1999; 115: 428–433. doi: 10.1378/chest.115.2.428 [DOI] [PubMed] [Google Scholar]
- 26.Vejbaesya S, Chierakul N, Luangtrakool K, et al. Associations of HLA class II alleles with pulmonary tuberculosis in Thais. Eur J Immunogenet 2002; 29: 431–434. 10.1046/j.1365-2370.2002.00352.x. [DOI] [PubMed] [Google Scholar]
- 27.Starshinova A, Dovgalyuk I, Berkos A, et al. The effect of human leukocyte antigens DRB1 alleles on development of different Tuberculosis forms in children. Int J Mycobacteriol 2018; 7: 117–121. doi: 10.4103/ijmy.ijmy_41_18 [DOI] [PubMed] [Google Scholar]
- 28.Wamala D, Buteme HK, Kirimunda S, et al. Association between human leukocyte antigen class II and pulmonary tuberculosis due to mycobacterium tuberculosis in Uganda. BMC Infect Dis 2016; 16: 23. doi: 10.1186/s12879-016-1346-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Toyo-Oka L, Mahasirimongkol S, Yanai H, et al. Strain-based HLA association analysis identified HLA-DRB1*09:01 associated with modern strain tuberculosis. HLA 2017; 90: 149–156. doi: 10.1111/tan.13070. [DOI] [PubMed] [Google Scholar]
- 30.Wu F, Zhang W, Zhang L, et al. NRAMP1, VDR, HLA-DRB1, and HLA-DQB1 gene polymorphisms in susceptibility to tuberculosis among the Chinese Kazakh population: a case–control study. BioMed Res Int 2013; 2013: 1–8. doi: 10.1155/2013/484535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.de Lima DS, Ogusku MM, Santos MPD, et al. Alleles of HLA-DRB1*04 associated with pulmonary tuberculosis in Amazon Brazilian population. PLoS ONE 2016; 11: e0147543. doi: 10.1371/journal.pone.0147543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang J, Song C, Wang S. Association of HLA-DRB1 genes with pulmonary tuberculosis. Zhonghua Jie He He Hu Xi Za Zhi 2001; 24: 302–305 (article in Chinese). [PubMed] [Google Scholar]
- 33.Berlin M, Fogdell-Hahn A, Olerup O, et al. HLA-DR predicts the prognosis in Scandinavian patients with pulmonary sarcoidosis. Am J Respir Crit Care Med 1997; 156: 1601–1605. doi: 10.1164/ajrccm.156.5.9704069 [DOI] [PubMed] [Google Scholar]
- 34.Bogunia-Kubik K, Tomeczko J, Suchnicki K, et al. HLA-DRB1*03, DRB1*11 or DRB*12 and their respective DRB3 specificities in clinical variants of sarcoidosis. Tissue Antigens 2001; 57: 87–90. doi: 10.1034/j.1399-0039.2001.057001087.x [DOI] [PubMed] [Google Scholar]
- 35.Planck A, Eklund A, Yamaguchi E, et al. Angiotensin-converting enzyme gene polymorphism in relation to HLA-DR in sarcoidosis. J Intern Med 2002; 251: 217–222. doi: 10.1046/j.1365-2796.2002.00946.x [DOI] [PubMed] [Google Scholar]
- 36.Foley PJ, McGrath DS, Puscinska E, et al. Human leukocyte antigen-DRB1 position 11 residues are a common protective marker for sarcoidosis. Am J Respir Cell Mol Biol 2001; 25: 272–277. doi: 10.1165/ajrcmb.25.3.4261 [DOI] [PubMed] [Google Scholar]
- 37.Grunewald J, Brynedal B, Darlington P, et al. Different HLA-DRB1 allele distributions in distinct clinical subgroups of sarcoidosis patients. Respir Res 2010; 11: 25. doi: 10.1186/1465-9921-11-25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rossman MD, Thompson B, Frederick M, et al. HLA-DRB1*1101: a significant risk factor for sarcoidosis in blacks and whites. J Hum Genet 2003; 73: 720–735. doi: 10.1086/378097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Darlington P, Tallstedt L, Padyukov L, et al. HLA-DRB1 alleles and symptoms associated with Рeerfordt's syndrome in sarcoidosis. J Eur Respire 2011; 38: 1151–1157. doi: 10.1183/09031936.00025011 [DOI] [PubMed] [Google Scholar]
- 40.Levin AM, Adrianto I, Datta I, et al. Association of HLA-DRB1 with sarcoidosis susceptibility and progression in African Americans. Am J Respir Cell Mol Biol 2015; 53: 206–216. doi: 10.1165/rcmb.2014-0227OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.da Costa CH, Silva VL, Fabricio-Silva GM, et al. HLA in a cohort of Brazilian patients with sarcoidosis. Hum Immunol 2013; 74: 1326–1332. doi: 10.1016/j.humimm.2013.07.009. [DOI] [PubMed] [Google Scholar]
- 42.Mortaz E, Sereshki HA, Abedini A, et al. Association of serum TNF-α, IL-8 and free light chain with HLA-DR B alleles expression in pulmonary and extra-pulmonary sarcoidosis. J Inflamm (Lond) 2015; 12: 21. doi: 10.1186/s12950-015-0066-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ozyilmaz E, Goruroglu Ozturk O, Yunsel D, et al. Could HLA-DR B1*11 allele be a clue for predicting extra-pulmonary sarcoidosis? Sarcoidosis Vasc Diffuse Lung Dis 2014; 31: 154–162. [PubMed] [Google Scholar]
- 44.Sharma SK, Balamurugan A, Pandey RM, et al. Human leukocyte antigen-DR alleles influence the clinical course of pulmonary sarcoidosis in Asian Indians. Am J Respir Cell Mol Biol 2003; 29: 225–231. doi: 10.1165/rcmb.2003-0007OC [DOI] [PubMed] [Google Scholar]
- 45.Voorter CE, Drent M, van den Berg-Loonen EM. Severe pulmonary sarcoidosis is strongly associated with the haplotype HLA-DQB1*0602-DRB1*150101. Hum Immunol 2005; 66: 826–835. doi: 10.1016/j.humimm.2005.04.003 [DOI] [PubMed] [Google Scholar]
- 46.Yanardag H, Tetikkurt C, Bilir M, et al. Association of HLA antigens with the clinical course of sarcoidosis and familial disease. Monaldi Arch Chest Dis 2017; 87: 835. doi: 10.4081/monaldi.2017.835. [DOI] [PubMed] [Google Scholar]
- 47.Papadopoulos KI, Wassmuth R, Sponsel T, et al. Sarcoidosis and autoimmunity: evidence of differential associations with HLA class II markers. Int J Endocrinol Metab 2006; 4: 13–18. [Google Scholar]
- 48.Fischer A, Ellinghaus D, Nutsua M, et al. Identification of immune-relevant factors conferring sarcoidosis genetic risk. J Respir Crit Care Med 2015; 192: 727–736. doi: 10.1164/rccm.201503-0418OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kishore A, Petrek M. Immunogenetics of sarcoidosis. J Int Trends Immun 2013; 1: 43–53. [Google Scholar]
- 50.Dubaniewicz A, Dubaniewicz-Wybieralska M, Moszkowska G, et al. Comparative analysis of DR and DQ alleles occurrence in sarcoidosis and tuberculosis in the same ethnic group: preliminary study. Sarcoidosis Vasc Diffuse Lung Dis 2006; 23: 180–189. [PubMed] [Google Scholar]
- 51.Dubaniewicz A, Moszkowska G. Analysis of occurrence of DRB and DQ alleles in sarcoidosis and tuberculosis from Northern Poland. Pneumonol Alergol Pol 2007; 75: 13–21 (Polish). [PubMed] [Google Scholar]
- 52.Saltini C, Pallante M, Puxeddu E, et al. Avium binding to HLA-Dr expressed alleles in silico: a model of phenotypic susceptibility to sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis 2008; 25: 100–116. [PubMed] [Google Scholar]
- 53.Amicosante M, Puxeddu E, Saltini C. Reactivity to mycobacterial antigens by patients with Lofgren's syndrome as a model of phenotypic susceptibility to disease and disease progression. Am J Respir Crit Care Med 2009; 180: 685–686. doi: 10.1164/ajrccm.180.7.685 [DOI] [PubMed] [Google Scholar]
- 54.Posch PE, Hurley CK, Geluk A, et al. The impact of DR3 microvariation on peptide binding: The combinations of specific DR-beta residues critical to binding differ for different peptides. Hum Immunol 1996; 49: 96–105. doi: 10.1016/0198-8859(96)00061-4 [DOI] [PubMed] [Google Scholar]
- 55.Posch PE, Araujo HA, Creswell K, et al. Microvariation creates significant functional differences in the DR3 molecules. Hum Immunol 1995; 42: 61–71. doi: 10.1016/0198-8859(94)00074-Z [DOI] [PubMed] [Google Scholar]
- 56.Contini S, Pallante M, Vejbaesya S, et al. A model of phenotypic susceptibility to tuberculosis: deficient in silico selection of Mycobacterium tuberculosis epitopes by HLA alleles. Sarcoidosis Vasc Diffuse Lung Dis 2008; 25: 21–28. [PubMed] [Google Scholar]
- 57.Shams H, Klucar P, Weis SE, et al. Characterization of a Mycobacterium tuberculosis peptide that is recognized by human CD4+ and CD8+ T cells in the context of multiple HLA alleles. J Immunol 2004; 173: 1966–1977. doi: 10.4049/jimmunol.173.3.1966 [DOI] [PubMed] [Google Scholar]
- 58.Selvaraj P, Rajeswari DN, Jawahar MS, et al. Influence of HLA-DRB1 alleles on Th1 and Th2 cytokine response to Mycobacterium tuberculosis antigens in pulmonary tuberculosis. Tuberculosis 2007; 87: 544–550. doi: 10.1016/j.tube.2007.08.001 [DOI] [PubMed] [Google Scholar]
- 59.Parzanese I, Qehajaj D, Patrinicola F, et al. Celiac disease: from pathophysiology to treatment. World J Gastrointest Pathophysiol 2017; 8: 27–38. doi: 10.4291/wjgp.v8.i2.27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ribeiro FM, Goldenberg T. Mycobacteria and autoimmunity. Lupus 2015; 24: 374–381. doi: 10.1177/0961203314559634 [DOI] [PubMed] [Google Scholar]
- 61.Elkayam O, Bendayan D, Segal R, et al. The effect of anti-tuberculosis treatment on levels of anti-phospholipid and anti-neutrophil cytoplasmatic antibodies in patients with active tuberculosis. Rheumatol Int 2013; 33: 949–953. doi: 10.1007/s00296-012-2487-0 [DOI] [PubMed] [Google Scholar]
- 62.Sundqvist E, Sundström P, Lindén M, et al. Epstein–Barr virus and multiple sclerosis: interaction with HLA. Genes Immun 2012; 13: 14–20. doi: 10.1038/gene.2011.42 [DOI] [PubMed] [Google Scholar]
- 63.Toussirot E, Roudier J. Epstein–Barr virus in autoimmune diseases. Best Pract Res Clin Rheumatol 2008; 22: 883–896. doi: 10.1016/j.berh.2008.09.007 [DOI] [PubMed] [Google Scholar]
- 64.Dunstan SJ, Hue NT, Han B, et al. Variation at HLA-DRB1 is associated with resistance to enteric fever. Nat Genet 2014; 46: 1333–1336. doi: 10.1038/ng.3143 [DOI] [PMC free article] [PubMed] [Google Scholar]