Abstract
BK virus reactivation as a result of therapeutic immunosuppression following renal transplant can result in BK polyomavirus nephropathy and renal allograft loss. This is a complex and challenging clinical problem with a range of management options and practices reported in literature. The current standard for early diagnosis and treatment is surveillance by measuring viral DNA in blood using qPCR. Immunosuppression reduction is the cornerstone of effective management but is associated with a risk of acute rejection following treatment.
Keywords: BK polyomavirus nephropathy, kidney transplant, immune monitoring, treatment, surveillance
Prevalence
BK polyoma virus (BKV) is a non-enveloped DNA virus first discovered in the urine of a kidney transplant recipient in 1971.1 Its genome has an early region which codes for the large and small T antigens, a late region which codes for the capsid proteins VP1-3, and agnoprotein, and a non-coding control region (NCCR). BKV strains have six genotypes based on polymorphisms in VP1 and NCCR.2
BKV is widely prevalent in general population with over 80% individuals having antibodies against BK virus.3,4 The most common mode of transmission is through respiratory secretions, resulting in a mild self-limited respiratory infection.5 Viral spread to other organs is believed to be via bloodstream and in immunocompetent individuals, it remains clinically silent in renal tubular epithelium.
“Presumptive” BK Polyoma virus nephropathy (PVN) is defined as persistently high BK viral load in plasma >10,000 copies/mL for four weeks. Renal allograft biopsy remains the gold standard for diagnosing “definite” PVN.6–12 Since the allograft involvement is focal, and the possibility of sampling error is high, two cores containing medulla are required for an adequate biopsy sample.8,9 Intragraft polyomavirus gene expression on renal biopsy has recently been reported as a useful adjunct to the diagnosis of PVN with the potential to differentiate from T-cell-mediated rejection.13 Biopsy proven “definite” PVN has an incidence of 5–6%, with a higher incidence in ABO-incompatible donors and following desensitization in highly sensitized recipients.14–16
The Banff Working Group on Polyomavirus Nephropathy recently published a morphologic classification of definite PVN into three groups, Class I, II, and III, based on polyomavirus load and Banff ci score (interstitial fibrosis) for ease of diagnostic communication and comparative data analysis.17 However, this was a retrospective observational analysis which has not been validated in a mixed population.
Impact
BK-virus-related disease is commonly seen in kidney transplant and hematopoietic stem cell transplant recipients. The cause for reactivation is therapeutic immunosuppression (IS) following transplant.18 BK viruria can be seen in 60% of kidney transplant recipients, while BK viremia is seen in up to 13% kidney transplant recipients, and nephropathy in 10%.19–21 The actual reported incidence varies; however, with the choice of induction IS, maintenance IS, and screening modality used, hence the wide variations in literature. In US, 5.7%– 7.5% of renal allografts are lost to PVN.22
PVN is therefore a serious clinical problem in kidney transplantation. PVN is difficult to treat since there is no BKV-specific anti-viral therapy. Any anti-virals currently in use work poorly and suffer from substantial host toxicity. PVN is treated by stimulating host immune response by IS reduction; however, there is a risk of acute rejection following virus clearance,23 further complicating treatment options since rejection treatment requires escalation of IS which often results in BKV recurrence.
The current standard for management is monitoring for viral DNA using qPCR. Other investigational surveillance tools include monitoring BKV-specific CMIR,24 and donor-derived cell-free DNA (dd-cfDNA). dd-cfDNA is a non-specific marker of injury. Since BKV causes interstitial inflammation and tubulitis, elevated levels of dd-cfDNA have been reported in a study of allograft rejection in kidney transplant in the setting of PVN.25 Since BKV is also known to be associated with development of de novo donor-specific antibodies (DSA),26 elevated dd-cfDNA levels in this infection could actually represent alloantibody-mediated microcirculation injury. Persistent viremia (lasting >140 days) was found to be strongly associated with development of Class II DSAs. The association of Class II DSA with antibody-mediated rejection (ABMR) and graft loss is well known.27
Most studies have found that humoral immune response does not play a significant role in preventing development of PVN.28 Despite the presence of a high level of antibodies, patients with PVN can have high levels of viral load and low CD8+ T cells.29 BKV-specific cell-mediated immune response (CMIR) was demonstrated in normal individuals to be the mechanism responsible for prevention of BKV reactivation in immunocompetent individuals.30 Low levels of BKV-specific interferon-gamma (IFNγ) producing T cells correlate with progression to PVN, while reconstitution of these cells correlates with resolution of nephropathy.31–34 Immune monitoring could help in identifying patients at risk of PVN;34–38 however, this knowledge is still evolving and has not been used in guiding treatment recommendations.
Management Strategies
Risk Factors
The most common factor associated with risk of developing PVN is the intensity of immunosuppression. Donor factors associated with a higher risk include transplanting kidney from BKV seropositive donor to seronegative donor,39,40 number of HLA mismatches, ABO-incompatibility, and ischemia reperfusion injury.6,14,41,42 Recipient factors include old age, male sex, desensitization, and prior kidney transplant with PVN.16,43
Surveillance
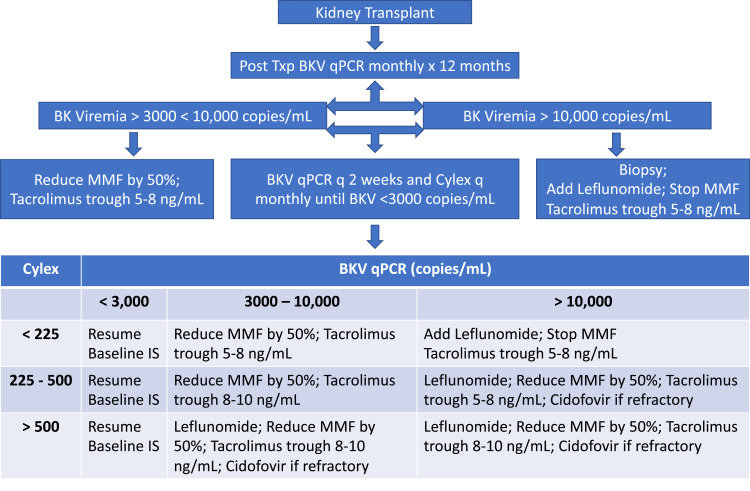
The mainstay of treatment of PVN is immunosuppression reduction. A wide variation in treatment practices is observed based on individual clinician experience. Most centers monitor BKV post-transplant at 3, 6, 9, and 12 months.44 However, with more intense induction regimen or in those with risk factors, it is prudent to perform routine surveillance at monthly intervals in the first 12 months following transplant. This is standard in our center. In addition to following viral loads with qPCR, we also follow ImmuKnow Immune Cell Function Assay (Cylex Inc., Columbia, MD, USA) as an indirect measure of CMIR. Our approach is outlined in Figure 1.
Figure 1.
Monitoring and treatment protocol for BK viremia at our center.
Immunosuppression Reduction and Antiviral Therapy
For BKV viral load <10,000 copies/mL, IS dose reduction should be considered. For viral loads >10,000 copies/mL, a common initial approach involves calcineurin inhibitor dose reduction by 25–50%. Switching to Cyclosporine A (CsA) has been shown to have some benefit as well.45 Switching from Tacrolimus to CsA is a common approach used in our center in patients with persistent viremia; However, a higher incidence of biopsy-proven acute rejection is seen with this approach.46 Failure of reduction in viral load should prompt reduction of mycophenolate mofetil (MMF) by 50%, or discontinuation of MMF or switching to an mTOR inhibitor.47,48 Switching from MMF to Leflunomide is another option associated with favorable outcomes.49–52 We routinely switch from MMF to Leflunomide in our center; however, the practices vary by center and physician experience. In refractory cases, most common therapeutic option is Cidofovir, use of which is limited by its nephrotoxicity.53–55 Brincidofovir is a prodrug of cidofovir and has also been used with limited success.56,57 IVIG preparations have high titers of neutralizing antibodies to BK virus and can help expedite virus clearance and have been used as a useful adjunctive therapy.58–61 Fluoroquinolones have been tried but failed to show therapeutic benefit.62–64 There is no strong evidence supporting antiviral treatment for PVN;46 however, for patients with persistent BK viremia despite adequate immunosuppression reduction, therapeutic options are outlined in Table 1.
Table 1.
Anti-Virals for PVN
| Anti-Virals | |||
|---|---|---|---|
| Name | Class/Mechanism | Dose | Comments |
| Leflunomide49–52 | Anti-Inflammatory; Anti-Viral; Immunosuppressive | PO: Loading- 100 mg daily for 3–5 days; maintenance- 20-60 mg qD; Trough Level −50-100 μg/mL | Can be used following discontinuation of MMF. |
| Cidofovir53–55 | Nucleoside analog | IV: 0.25–1.0 mg/Kg at 1–3 weeks | Used in refractory cases; Nephrotoxicity is the most serious adverse effect. |
| Brincidofovir56,57 | Investigational Prodrug of Cidofovir; Anti-viral activity | PO: 2 mg/Kg twice weekly | Reasonably well tolerated; Investigational. |
| Intravenous immunoglobulin (IVIG)58–61 | Immunoglobulin preparation with high titers of neutralizing antibodies to BK virus | IV: 0.25–2.0 g/Kg | Can be used as an adjunct to other measures in refractory cases. |
| Levofloxacin62–64 | Fluoroquinolones; Antiviral, inhibit helicase activity of large T antigen | PO: 500 mg qD (renally adjusted) | Levofloxacin failed to show benefit in randomized controlled trials. |
| Everolimus47,48 | Inhibits mammalian target of rapamycin (mTOR) kinase activity, inhibiting T and B lymphocyte activation and proliferation. | PO 0.75 mg twice daily adjusted to trough levels of 3–8 ng/mL. | Can be used following discontinuation of MMF. Limited literature supporting its use. |
Conclusion
Due to lack of strong evidence, no strong treatment recommendations can be made; however, it is prudent to start with immunosuppression reduction and add anti-virals for persistent viremia not responding to immunosuppression reduction based on physician experience. Regular monitoring of qPCR remains the cornerstone of early diagnosis and treatment. Novel monitoring strategies being investigated include immune monitoring and ddcf DNA.
Abbreviations
BKV, BK virus; NCCR, non-coding control region; PVN, BK polyoma virus nephropathy; qPCR, quantitative polymerase chain reaction; ddcfDNA, donor-derived cell-free DNA; DSA, donor-specific antibodies; ABMR, antibody-mediated rejection; CMIR, cell-mediated immune response; JCV, JC virus; IFNγ, interferon-gamma; MMF, mycophenolate mofetil; CsA, cyclosporine A; ATP, adenosine triphosphate; ELISPOT, enzyme-linked immunoSpot; IS, Immunosuppression; PML, progressive multifocal leukoencephalopathy.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Gardner SD, Field AM, Coleman DV, et al. New human papovavirus (B.K.) isolated from urine after renal transplantation. Lancet. 1971;1(7712):1253–1257. doi: 10.1016/S0140-6736(71)91776-4 [DOI] [PubMed] [Google Scholar]
- 2.Chatterjee M, Weyandt TB, Frisque RJ. Identification of archetype and rearranged forms of BK virus in leukocytes from healthy individuals. J Med Virol. 2000;60(3):353–362. doi: [DOI] [PubMed] [Google Scholar]
- 3.Flaegstad T, Ronne K, Filipe AR, et al. Prevalence of anti BK virus antibody in Portugal and Norway. Scan J Infectious Dis. 1989;21(2):145–147. doi: 10.3109/00365548909039961 [DOI] [PubMed] [Google Scholar]
- 4.Stolt A, Sasnauskas K, Koskela P, et al. Seroepidemiology of the human polyomaviruses. J Gen Virol. 2003;84(Pt 6):1499–1504. doi: 10.1099/vir.0.18842-0 [DOI] [PubMed] [Google Scholar]
- 5.Goudsmit J, Wertheim-van Dillen P, van Strien A, et al. The role of BK virus in acute respiratory tract disease and the presence of BKV DNA in tonsils. J Med Virol. 1982;10(2):91–99. doi: 10.1002/jmv.1890100203 [DOI] [PubMed] [Google Scholar]
- 6.Hirsch HH, Brennan DC, Drachenberg CB, et al. Polyomavirus-associated nephropathy in renal transplantation: interdisciplinary analyses and recommendations. Transplantation. 2005;79(10):1277–1286. doi: 10.1097/01.TP.0000156165.83160.09 [DOI] [PubMed] [Google Scholar]
- 7.Hirsch HH, Steiger J. Polyomavirus BK. Lancet Infect Dis. 2003;3(10):611–623. doi: 10.1016/S1473-3099(03)00770-9 [DOI] [PubMed] [Google Scholar]
- 8.Drachenberg CB, Papadimitriou JC, Hirsch HH, et al. Histological patterns of polyomavirus nephropathy: correlation with graft outcome and viral load. Am J Transplant. 2004;4(12):2082–2092. doi: 10.1046/j.1600-6143.2004.00603.x [DOI] [PubMed] [Google Scholar]
- 9.Hirsch HH, Randhawa P. AST infectious diseases community of practice. BK virus in solid organ transplant recipients. Am J Transplant. 2009;9(Suppl 4):S136–S146. [DOI] [PubMed] [Google Scholar]
- 10.Sar A, Worawichawong S, Benediktsson H, et al. Interobserver agreement for polyomavirus nephropathy grading in renal allografts using the working proposal from the 10th Banff Conference on Allograft Pathology. Hum Pathol. 2011;42(12):2018–2024. doi: 10.1016/j.humpath.2011.03.008 [DOI] [PubMed] [Google Scholar]
- 11.Haas M, Sis B, Racusen LC, et al. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant. 2014;14(2):272–283. doi: 10.1111/ajt.12590 [DOI] [PubMed] [Google Scholar]
- 12.Masutani K, Shapiro R, Basu A, et al. The Banff 2009 working proposal for polyomavirus nephropathy: a critical evaluation of its utility as a determinant of clinical outcome. Am J Transplant. 2012;12(4):907–918. doi: 10.1111/j.1600-6143.2012.03993.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Adam BA, Kikic Z, Wagner S, et al. Intragraft gene expression in native kidney BK virus nephropathy versus T cell-mediated rejection: prospects for molecular diagnosis and risk prediction [published online ahead of print, 2020 May 5]. Am J Transplant. 2020. doi: 10.1111/ajt.15980 [DOI] [PubMed] [Google Scholar]
- 14.Sharif A, Alachkar N, Bagnasco S, et al. Incidence and outcomes of BK virus allograft nephropathy among ABO- and HLA-incompatible kidney transplant recipients. Clin J Am Soc Nephrol. 2012;7:1320–1327. doi: 10.2215/CJN.00770112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boan P, Hewison C, Swaminathan R, et al. Optimal use of plasma and urine BK viral loads for screening and predicting BK nephropathy. BMC Infect Dis. 2016;16:342. doi: 10.1186/s12879-016-1652-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gabardi S, Townsend K, Martin ST, et al. Evaluating the impact of pre-transplant desensitization utilizing a plasmapheresis and low-dose intravenous immunoglobulin protocol on BK viremia in renal transplant recipients. Transpl Infect Dis. 2013;15:361–368. doi: 10.1111/tid.12087 [DOI] [PubMed] [Google Scholar]
- 17.Nickeleit V, Singh HK, Randhawa P, et al. The Banff working group classification of definitive polyomavirus nephropathy: morphologic definitions and clinical correlations. J Am Soc Nephrol. 2018;29:680–693. doi: 10.1681/ASN.2017050477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Leung AY, Chan M, Tang SC, et al. Real-time quantitative analysis of polyoma BK viremia and viruria in renal allograft recipients. J Virol Meth. 2002;103(1):51–56. doi: 10.1016/S0166-0934(01)00447-5 [DOI] [PubMed] [Google Scholar]
- 19.Hirsch HH, Knowles W, Dickenmann M, et al. Prospective study of polyomavirus type BK replication and nephropathy in renal-transplant recipients. N Engl J Med. 2002;347(7):488–496. doi: 10.1056/NEJMoa020439 [DOI] [PubMed] [Google Scholar]
- 20.Brennan DC, Agha I, Bohl DL, et al. Incidence of BK with tacrolimus versus cyclosporine and impact of preemptive immuno-suppression reduction. Am J Transplant. 2005;5(3):582–594. doi: 10.1111/j.1600-6143.2005.00742.x [DOI] [PubMed] [Google Scholar]
- 21.Hirsch HH. BK virus: opportunity makes a pathogen. Clin Infect Dis. 2005;41(3):354–360. doi: 10.1086/431488 [DOI] [PubMed] [Google Scholar]
- 22.Kuypers DR. Management of polyomavirus-associated nephro-pathy in renal transplant recipients. Nat Rev Nephrol. 2012;8(7):390–402. doi: 10.1038/nrneph.2012.64 [DOI] [PubMed] [Google Scholar]
- 23.Van Aalderen MC, Heutinck KM, Huisman C, et al. BK virus infection in transplant recipients: clinical manifestations, treatment options and the immune response. Neth J Med. 2012;70(4):172–183. [PubMed] [Google Scholar]
- 24.Sharma R, Tzetzo S, Patel S, et al. BK virus in kidney transplant: current concepts, recent advances, and future directions. Exp Clin Transplant. 2016;14(4):377–384. doi: 10.6002/ect.2016.0030 [DOI] [PubMed] [Google Scholar]
- 25.Bloom RD, Bromberg JS, Poggio ED, et al. Cell-free DNA and active rejection in kidney allografts. JASN Jul. 2017;28(7):2221–2232. doi: 10.1681/ASN.2016091034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sawinski D, Forde KA, Trofe-Clark J, et al. Persistent BK viremia does not increase intermediate-term graft loss but is associated with de novo donor-specific antibodies. J Am Soc Nephrol. 2015;26:966–975. doi: 10.1681/ASN.2014010119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Haas M, Loupy A, Lefaucheur C, et al. The Banff 2017 Kidney Meeting Report: revised diagnostic criteria for chronic active T cell-mediated rejection, antibody-mediated rejection, and prospects for integrative endpoints for next-generation clinical trials. Am J Transplant. 2018;18(2):293–307. doi: 10.1111/ajt.14625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hariharan S, Cohen EP, Vasudev B, et al. BK virus-specific antibodies and BKV DNA in renal transplant recipients with BKV nephritis. Am J Transplant. 2005;5(11):2719–2724. doi: 10.1111/j.1600-6143.2005.01080.x [DOI] [PubMed] [Google Scholar]
- 29.Chen Y, Trofe J, Gordon J, et al. Interplay of cellular and humoral immune responses against BK virus in kidney transplant recipients with polyomavirus nephropathy. J Virol. 2006;80(7):3495–3505. doi: 10.1128/JVI.80.7.3495-3505.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Drummond JE, Shah KV, Donnenberg AD. Cell-mediated immune responses to BK virus in normal individuals. J Med Virol. 1985;17(3):237–247. doi: 10.1002/jmv.1890170305 [DOI] [PubMed] [Google Scholar]
- 31.Batal I, Zeevi A, Heider A, et al. Measurements of global cell-mediated immunity in renal transplant recipients with BK virus reactivation. Am J Clin Pathol. 2008;129(4):587–591. doi: 10.1309/23YGPB1E758ECCFP [DOI] [PubMed] [Google Scholar]
- 32.Binggeli S, Egli A, Dickenmann M, et al. BKV replication and cellular immune responses in renal transplant recipients. Am J Transplant. 2006;6(9):2218–9;author reply 20. doi: 10.1111/j.1600-6143.2006.01460.x [DOI] [PubMed] [Google Scholar]
- 33.Prosser SE, Orentas RJ, Jurgens L, et al. Recovery of BK virus large T-antigen-specific cellular immune response correlates with resolution of BK virus nephritis. Transplantation. 2008;85(2):185–192. doi: 10.1097/TP.0b013e31815fef56 [DOI] [PubMed] [Google Scholar]
- 34.Comoli P, Azzi A, Maccario R, et al. Polyomavirus BK-specific immunity after kidney transplantation. Transplantation. 2004;78(8):1229–1232. doi: 10.1097/01.TP.0000137932.44791.D3 [DOI] [PubMed] [Google Scholar]
- 35.Comoli P, Binggeli S, Ginevri F, et al. Polyomavirus-associated nephropathy: update on BK virus-specific immunity. Transpl Infect Dis. 2006;8(2):86–94. doi: 10.1111/j.1399-3062.2006.00167.x [DOI] [PubMed] [Google Scholar]
- 36.Comoli P, Cioni M, Basso S, et al. Immunity to polyomavirus BK infection: immune monitoring to regulate the balance between risk of BKV nephropathy and induction of alloimmunity. Clin Dev Immunol. 2013;2013:256923. doi: 10.1155/2013/256923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schachtner T, Muller K, Stein M, et al. BK virus-specific immunity kinetics: a predictor of recovery from polyomavirus BK-associated nephropathy. Am J Transplant. 2011;11(11):2443–2452. doi: 10.1111/j.1600-6143.2011.03693.x [DOI] [PubMed] [Google Scholar]
- 38.Schachtner T, Stein M, Sefrin A, et al. Inflammatory activation and recovering BKV-specific immunity correlate with self-limited BKV replication after renal transplantation. Transplant Int. 2014;27(3):290–301. doi: 10.1111/tri.12251 [DOI] [PubMed] [Google Scholar]
- 39.Smith JM, McDonald RA, Finn LS, et al. Polyomavirus nephropathy in pediatric kidney transplant recipients. Am J Transplant. 2004;4(12):2109–2117. doi: 10.1111/j.1600-6143.2004.00629.x [DOI] [PubMed] [Google Scholar]
- 40.Sood P, Senanayake S, Sujeet K, et al. Donor and recipient BKV- specific IgG antibody and post transplantation BKV infection: a prospective single-center study. Transplantation. 2013;95(6):896–902. doi: 10.1097/TP.0b013e318282ba83 [DOI] [PubMed] [Google Scholar]
- 41.Awadalla Y, Randhawa P, Ruppert K, et al. HLA mismatching increases the risk of BK virus nephropathy in renal transplant recipients. Am J Transplant. 2004;4(10):1691–1696. doi: 10.1111/j.1600-6143.2004.00563.x [DOI] [PubMed] [Google Scholar]
- 42.Prince O, Savic S, Dickenmann M, et al. Risk factors for polyoma virus nephropathy. Nephrol Dial Transplant. 2009;24(3):1024–1033. doi: 10.1093/ndt/gfn671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ramos E, Drachenberg CB, Portocarrero M, et al. BK virus nephropathy diagnosis and treatment: experience at the University of Maryland Renal Transplant Program. Clin Transplant. 2002;143–153. [PubMed] [Google Scholar]
- 44.Dalianis T, Eriksson B-M, Felldin M, et al. Management of BK-virus infection – Swedish recommendations. Infect Dis. 2019;51(7):479–484. doi: 10.1080/23744235.2019.1595130 [DOI] [PubMed] [Google Scholar]
- 45.Chen XT, Li J, Deng RH, et al. The therapeutic effect of switching from tacrolimus to low-dose cyclosporine A in renal transplant recipients with BK virus nephropathy. Biosci Rep. 2019;39(2):BSR20182058. doi: 10.1042/BSR20182058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gard L, van Doesum W, Niesters HGM, et al. A delicate balance between rejection and BK polyomavirus associated nephropathy; a retrospective cohort study in renal transplant recipients. PLoS One. 2017;12(6):e0178801. doi: 10.1371/journal.pone.0178801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bowman LJ, Brueckner AJ, Doligalski CT. The role of mTOR inhibitors in the management of viral infections: a review of current literature. Transplantation. 2018;102:S50–S59. doi: 10.1097/TP.0000000000001777 [DOI] [PubMed] [Google Scholar]
- 48.Bussalino E, Marsano L, Parodi A, et al. Everolimus for BKV nephropathy in kidney transplant recipients: a prospective, controlled study. J Nephrol. 2020. doi: 10.1007/s40620-020-00777-2 [DOI] [PubMed] [Google Scholar]
- 49.Josephson MA, Gillen D, Javaid B, et al. Treatment of renal allograft polyoma BK virus infection with leflunomide. Transplantation. 2006;81(5):704–710. doi: 10.1097/01.tp.0000181149.76113.50 [DOI] [PubMed] [Google Scholar]
- 50.Williams JW, Javaid B, Kadambi PV, et al. Leflunomide for polyomavirus type BK nephropathy. N Engl J Med. 2005;352(11):1157–1158. doi: 10.1056/NEJM200503173521125 [DOI] [PubMed] [Google Scholar]
- 51.Williams JW, Mital D, Chong A, et al. Experiences with leflunomide in solid organ transplantation. Transplantation. 2002;73(3):358–366. doi: 10.1097/00007890-200202150-00008 [DOI] [PubMed] [Google Scholar]
- 52.Hirsch HH, Randhawa P. AST infectious diseases community of practice. BK polyomavirus in solid organ transplantation. Am J Transplant. 2013;13(Suppl 4):179–188. [DOI] [PubMed] [Google Scholar]
- 53.Kadambi PV, Josephson MA, Williams J, et al. Treatment of refractory BK virus-associated nephropathy with cidofovir. Am J Transplant. 2003;3(2):186–191. doi: 10.1034/j.1600-6143.2003.30202.x [DOI] [PubMed] [Google Scholar]
- 54.Keller LS, Peh CA, Nolan J, et al. BK transplant nephropathy successfully treated with cidofovir. Nephrol Dial Transplant. 2003;18(5):1013–1014. doi: 10.1093/ndt/gfg061 [DOI] [PubMed] [Google Scholar]
- 55.Kuypers DR, Vandooren AK, Lerut E, et al. Adjuvant low-dose cidofovir therapy for BK polyomavirus interstitial nephritis in renal transplant recipients. Am J Transplant. 2005;5(8):1997–2004. doi: 10.1111/j.1600-6143.2005.00980.x [DOI] [PubMed] [Google Scholar]
- 56.Reisman L, Habib S, McClure GB, et al. Treatment of BK virus-associated nephropathy with CMX001 after kidney transplantation in a young child. Pediatr Transplant. 2014;18:E227–E231. doi: 10.1111/petr.12340 [DOI] [PubMed] [Google Scholar]
- 57.Papanicolaou GA, Lee YJ, Young JW, et al. Brincidofovir for polyomavirus-associated nephropathy after allogeneic hema-topoietic stem cell transplantation. Am J Kidney Dis. 2015;65(5):780–784. doi: 10.1053/j.ajkd.2014.11.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Randhawa P, Pastrana DV, Zeng G, et al. Commercially available immunoglobulins contain virus neutralizing antibodies against all major genotypes of polyomavirus BK. Am J Transplant. 2015;15(4):1014‐1020. doi: 10.1111/ajt.13083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sener A, House AA, Jevnikar AM, et al. Intravenous immu-noglobulin as a treatment for BK virus associated nephropathy: one-year follow-up of renal allograft recipients. Transplantation. 2006;81(1):117–120. doi: 10.1097/01.tp.0000181096.14257.c2 [DOI] [PubMed] [Google Scholar]
- 60.Vu D, Shah T, Ansari J, et al. Efficacy of intravenous immunoglobulin in the treatment of persistent BK viremia and BK virus nephropathy in renal transplant recipients. Transplant Proc. 2015;47(2):394‐398. doi: 10.1016/j.transproceed.2015.01.012 [DOI] [PubMed] [Google Scholar]
- 61.Moon J, Chang Y, Shah T, et al. Effects of intravenous immunoglobulin therapy and Fc gamma receptor polymorphisms on BK virus nephropathy in kidney transplant recipients [published online ahead of print, 2020 Apr 23]. Transpl Infect Dis. 2020:e13300. doi: 10.1111/tid.13300. [DOI] [PubMed] [Google Scholar]
- 62.Gabardi S, Waikar SS, Martin S, et al. Evaluation of fluoroquinolones for the prevention of BK viremia after renal transplantation. Clin J Am Soc Nephrol. 2010;5(7):1298–1304. doi: 10.2215/CJN.08261109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sharma BN, Li R, Bernhoff E, et al. Fluoroquinolones inhibit human polyomavirus BK (BKV) replication in primary human kidney cells. Antiviral Res. 2011;92(1):115–123. doi: 10.1016/j.antiviral.2011.07.012 [DOI] [PubMed] [Google Scholar]
- 64.Knoll GA, Humar A, Fergusson D, et al. Levofloxacin for BK virus prophylaxis following kidney transplantation: a randomized clinical trial. JAMA. 2014;312(20):2106–2114. doi: 10.1001/jama.2014.14721 [DOI] [PubMed] [Google Scholar]