Case Presentation
A 27-year-old woman initially presented in January 2005 to an outside clinician with a history of a hyperpigmented exophitic tumor located on the right major labia 5 mm from the clitoris. This lesion was biopsied and reported by the outside institution as a vulvar melanoma with no further details. The patient was then referred to the National Cancer Institute (INCan) in July 2005. At the time of her presentation, the patient was noted to have no significant past medical or surgical history. On physical examination, she had a hyperpigmented lesion in the vulva measuring 2 cm, adjacent to a recent scar approximately 5 mm to the right of the clitoris. Palpation of the inguinal lymph nodes did not reveal any disease. The outside pathology was reviewed and was confirmed as an ulcerated melanoma with vertical growth, Breslow 7 mm. A CT scan from outside of the institution reported uterine myoma and negative inguinal lymph nodes.
Dr. Leitao
Based on this information, what would be your approach in further management?
There are two general points that must mentioned. The first is that there are limited data specific to mucosal melanomas (which includes vulvovaginal) and the management of mucosal melanomas is often extrapolated from the management of cutaneous melanomas. The other point is that the management of melanoma has dramatically changed since 2005. For this case, a CT scan is sufficient. However, there are certainly many who would consider PET/CT scans in the workup of patients with vulvar melanoma. Routine imaging is actually not recommended for early stage cutaneous melanomas in the NCCN guidelines. [1] Either CT or PET/CT are allowed if imaging is performed. Mucosal melanomas tend to have a worse outcome than cutaneous ones and performing a scan to rule out metastatic disease is certainly worth considering.
It is now standard to test melanomas for alterations in KIT, NRAS, and BRAF. This can be done using various available methodologies. Mucosal melanomas, as compared to cutaneous melanomas, more often have KIT alterations and less NRAS and BRAF alterations. The results of this testing does not alter the primary treatment of resectable, locally confined lesions, but may have implications in cases such lesions may not be resectable or in the setting of recurrent disease. More recently, the decision for adjuvant therapy may also be impacted by these results.
The primary treatment for resectable lesions without known metastasis is for wide local excision, as well as, surgical nodal assessment for lesions with a Breslow thickness greater than 0.75 mm. The goal of the wide local excision is to obtain a 2 cm clinically negative margin. Sentinel lymph node mapping is considered standard nodal assessment for melanoma. One should decide whether to perform unilateral or bilateral groin sentinel lymph node mapping based on the laterality of the primary tumor. If within 2 cm of the midline, a bilateral sentinel lymph node mapping is recommended. For this case, I would perform a wide local excision obtaining a 2 cm clinically negative margin and sentinel lymph node mapping of both groins.
In August 2005, a wide local excision of the vulvar lesion with unilateral sentinel lymph node mapping was performed.
Dr. Perez-Montiel
The specimen of the vulvar lesion measured 5×3.6×1.8 cm, containing a portion of labia major and minora. A dark and ulcerated lesion of 0.8×0.5 cm with peripheral borders elevated was identified. The sentinel lymph node measured 2.5×2 cm, and showed a neoplastic dark area of 1.3 cm. Microscopically in the vulva, a neoplastic lesion was located in superficial and deep dermis. This lesion was formed by nests of polygonal cells with broad cytoplasm, nuclei with pleomorphic and large nucleoli. Three atypical mitosis/mm2 were seen. The neoplastic lesion had 5 mm of invasion in the deepest area (Figure 1A). The epidermis had ulceration and adjacent epidermis showed atypical melanocytic nests in the dermoepidermal junction and individual cells in the epidermis (Figure 1B). Neither lymphovascular invasion nor tumor regression was identified. The surgical margins were at least 2 mm from the tumor. Sentinel lymph node had a metastatic melanoma of 1.3 cm, without extracapsular extension. (Figure 2)
Figure 1.
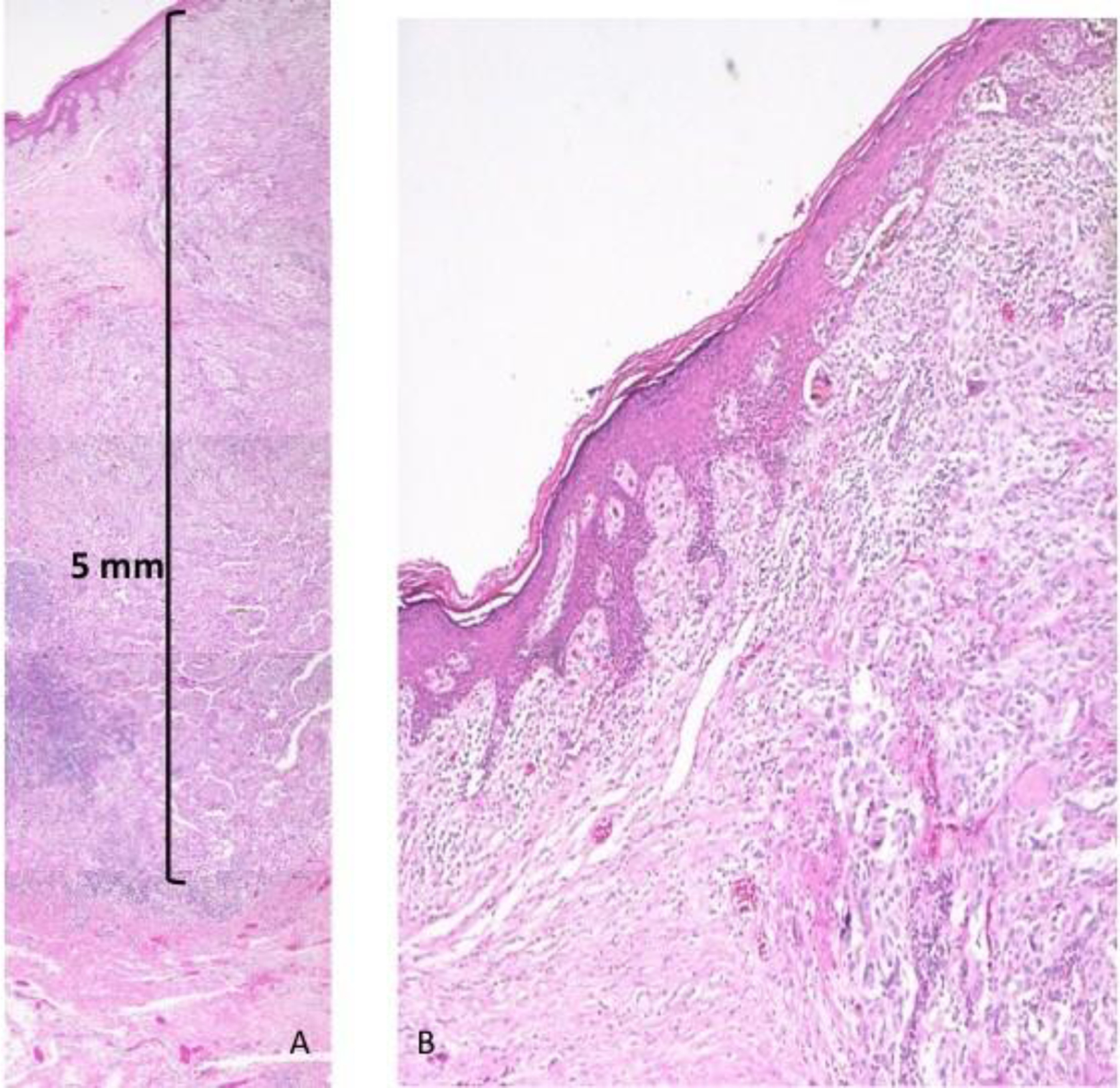
A. Hematoxylin & eosin staining reveling deepest level of invasion in the superficial and deep dermis (Breslow 5 mm).
B. Hematoxylin & eosin staining 4X reveling transition between normal skin and melanoma. The epidermis has ulceration and adjacent epidermis shows atypical melanocytic nests in dermoepidermal junction and individual cells in the epidermis.
Figure 2.
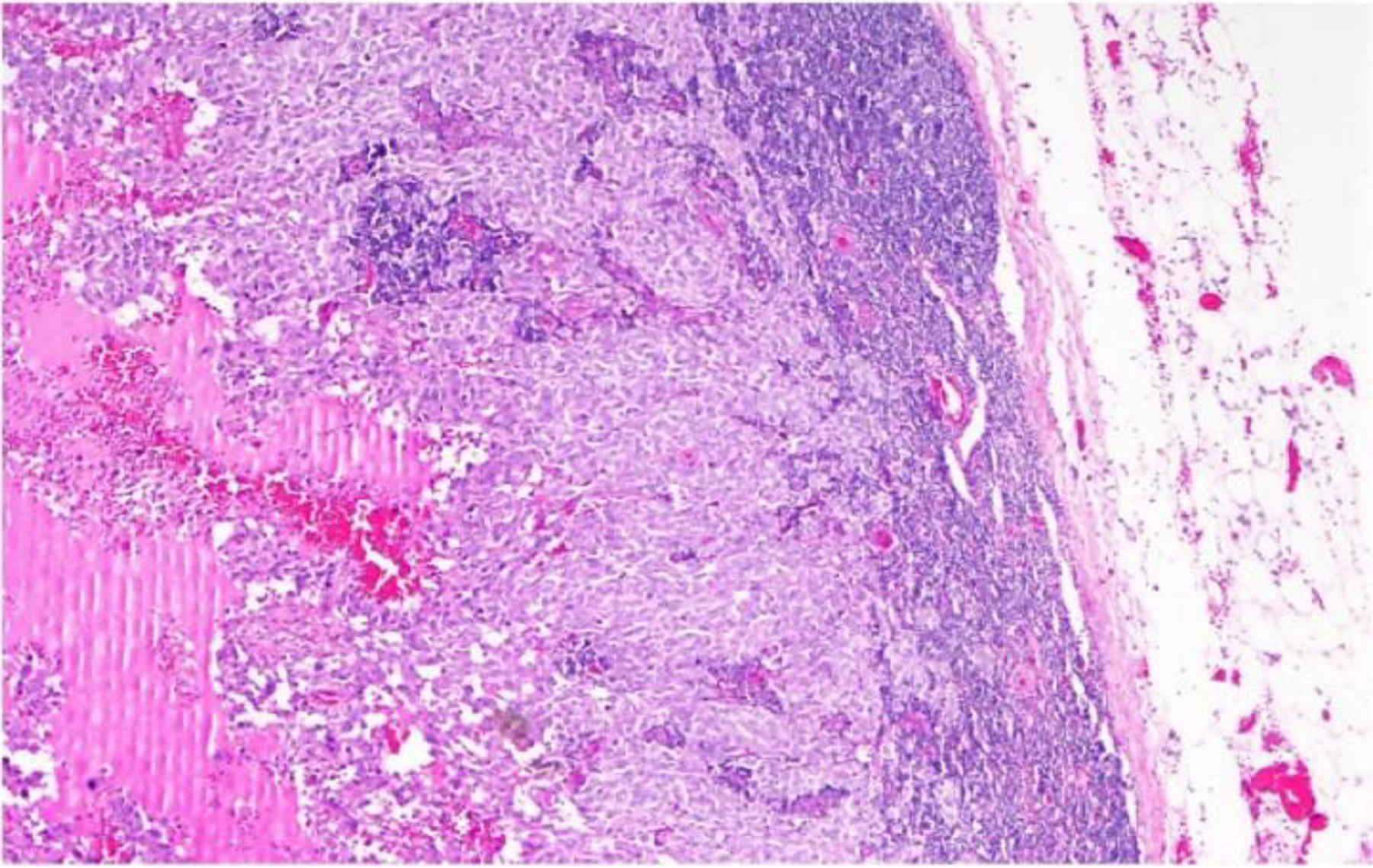
Hematoxylin & eosin staining 4X reveling a lymph node with melanoma metastasis of 1.3 cm, without extracapsular extension.
A definitive diagnosis of vulvar melanoma with Breslow of 5 mm was made, with negative surgical margins and sentinel lymph node with metastasis. At this time, the decision was made to proceed with an ipsilateral inguinopelvic dissection. A total of nine lymph nodes were removed and all were negative for disease. The patient did not receive radiotherapy or chemotherapy as adjuvant treatment.
Dr. Leitao
What is your approach in the setting of a positive sentinel lymph node in a patient with vulvar melanoma?
The preferred current approach in patients with melanoma and a positive sentinel lymph node is very different from what it was in 2005. Completion lymphadenectomy was the “standard” at that time. However, based on the results of the Multicenter Selective Lymphadenectomy Trials I and II (MSLT-I, MSLT-II), completion lymphadenectomy does not confer a melanoma-specific survival advantage. Therefore, many do not perform a completion lymphadenectomy but rather perform close groin surveillance every 3 months with a delayed completion lymphadenectomy in cases that develop any suspicious findings during this close groin surveillance. Groin surveillance can be done using ultrasound. The sentinel lymph node was only site of nodal disease in 70% of patients enrolled in MSLT-I. [2] Completion lymphadenectomy in these 70% of cases to remove normal nodes with its’ associated morbidity does not seem justified. The concept then became to closely follow patients with sentinel lymph node positive disease and only intervene at the time the disease presented clinically in these 30% of cases with additional nodal disease. MSLT-II confirmed that this was a safe and less morbid alternative. [3] These findings were also seen in another randomized trial (DeCOG-SLT) evaluating immediate versus delayed completion lymphadenectomy in patients with SLN metastasis. [4] My current practice is to obtain imaging every 3 months. We alternate with groin US and CT chest, abdomen and pelvis, as surveillance for distant recurrence is also needed.
Adjuvant therapy in patients with node positive melanoma (AJCC stage III) was not associated with improved outcomes and, therefore, these patients were merely observed. However, adjuvant CTLA and PD1 inhibition has been recently shown, in randomized trials, to significantly improve progression-free and overall survival in patients with node-positive melanoma. Ipilimumab significantly improved recurrence-free (median 27.6 months vs. 17.1 months) and overall survival (5-yr overall survival 65.4% vs. 54.4%) compared to placebo. [5] Nivolumab had a statistically significant improved recurrence-free survival as compared to ipilimumab with the median not reached in those receiving nivolumab (HR 0.65 (97.56%CI: 0.51–0.83). [6] Additionally, nivolumab therapy had much less toxicities. Pembrolizumab also resulted in a significant improvement in recurrence-free survival as compared to placebo with the median not reached (HR 0.57 (98.4% CI:0.43–0.74). [7] Adjuvant combined BRAF and MEK inhibition in patients with BRAF V600E or V600K mutations resulted in a significant improvement in relapse-free survival as compared to placebo (HR 0.47 (95%CI:0.39–0.58)). [8] The combined use of dabrafenib and trametinib led to a 3-yr recurrence-free survival of 58% compared to 39% in those receiving the placebo.
The above are truly impressive and promising. We now can offer beneficial adjuvant therapy in patients with node-positive melanoma. We offer adjuvant pembrolizumab or nivolumab to all patients with node-positive vulvovaginal melanoma. If they have a V600E or V600K mutation, we offer dabrafenib plus trametinib. The question remains though as to how long to continue adjuvant immunotherapy. The length of therapy has varied among the studies from up to one year, up to 18 doses, up to 3 years, and up to progression or toxicity. Practically speaking, I would consider continuing these therapies until progression and/or development of toxicities, although this may be subject to debate.
In February 2006, a routine chest x-ray was performed and this revealed a suspicious mediastinal nodule. A CT scan of the thorax, abdomen and pelvis was performed. The findings on the chest x-ray were nonspecific, as confirmed by the CT scan of the chest; however, the CT scan of the abdomen and pelvis showed an isodense-dependent lesion in both adnexae measuring 3.5- and 6.5 cm; respectively. A diagnostic laparoscopy was performed and the findings revealed a subserosal uterine pediculated myoma measuring 4×3 cm and a subserosal sessile myoma measuring 7×6 cm. A myomectomy was performed without complications. The recovery from this surgery was uneventful. The patient continued on routine surveillance.
The patient was without evidence of disease for four years. In December 2010, while on routine examination, a hyperpigmented lesion with irregular edges was identified on the vulva, at the lower aspect of the clitoris, midline above the urethra, measuring 0.4 cms. The hyperpigmented vulvar lesion was excised, and the findings revealed melanoma-in-situ with all margins negative for disease.
Dr. Perez-Montiel
The vulvar lesion measured 0.8×.6×.3 cm with a 0.2×0.2 flat dark lesion. Microscopically a small lesion measuring 0.2 cm formed by atypical melanocityc nest with epidermal transmigration located in dermoepidermal junction was identified. The diagnosis of melanoma-in-situ with free surgical margins was made.
The patient was then without evidence of disease for five years. Subsequently, in January 2015, a hyperpigmented lesion was noted in the left lower labia measuring 7 mm. An incisional biopsy of the vulvar region was performed and findings revealed malignant melanoma, Breslow 0.9 mm, with abundant inflammatory reaction associated with melanoma-in-situ, positive for melan-a. One month later, a wide local excision of the vulva (right lower lip) and sentinel lymph node was performed for a lesion measuring 7 mm.
Dr. Perez-Montiel
The specimen measured 3.2×2×1.3 cms. Epidermal surface showed multifocal flat dark lesions in the entire specimen. Cut surface was homogenous. Microscopically, this tumor had no ulceration noted, Breslow was 1.1 cms, without evidence of tumor regression and abundant lymphocytic perilesional infiltrate was present. Surgical margins were at least 3 mm from the tumor. A negative sentinel lymph node was identified.
Dr. Perez-Montiel
What clinical significance does tumor regression provide in the setting of vulvar melanoma?
Histologic characteristics as Breslow (defined as a tumor thickness measured from the top of the epidermal granular layer to the deepest point of invasion), ulcer (defined as absence of an intact epidermis overlying of the primary melanoma), growth pattern (superficial spreading, nodular malignant and acral lentigous melanoma), mitotic index, tumor regression and lymph node status are considered prognostic factors. Unlike skin melanoma, in vulvar melanoma these parameters are controversial. Breslow is the most important prognostic factor in skin melanomas, but in vulvar melanoma some studies have failed to correlate Breslow with prognosis. [9] However in a recent study, the tumor thickness (>2.0 mm) and increased dermal mitotic rate (>2 per mm2) showed good correlation as prognostic factor. [10] The prognostic significance of presence of tumor regression is controversial in cutaneous melanoma, but, a large study of vulvar melanomas showed regression in 25% of the cases. This finding was associated with improved disease specific survival and overall survival. A difference between the first tumor and the recurrence was the peritumoral lymphoid infiltrate, this feature has been informed in cutaneous melanoma as a prognostic factor, when is present, metastasis are less frequent and survival is better, but in vulvar melanoma these features have not demonstrated significance for prognosis. [10,11]
In October 2018, the patient reported palpating a nodule in her genital area. The physical examination revealed an ulcerated nodular lesion of 1.5×1.0 cm at the right labia majora. A PET/CT was ordered and showed an enlarged uterus with heterogeneous density, without fatty separation from adnexa, measuring approximately 16×17 cm and with a focal zone of metabolism at the level of the right labia majora with SUVmax of 7.7.
Dr. Santiago Concha
Imaging was performed on an integrated 16 slice PET/CT scanner, with scanning from the skull base to the toes. CT scanning was performed without oral or intravenous contrast material. There is a normal biodistribution in skull, neck, thorax and abdomino-pelvic organs, with physiological uptake in the spleen, liver, kidneys and urinary bladder. There is no adenopathy or nodal hypermetabolism in the abdomen or pelvis. In the coronal view there is a focal zone of hypermetabolism at the subcutaneous vulvar lesión with a SUVmax of 7.7 (arrows in Figures 3A&B). An axial slice at this level demonstrates a zone of hypermetabolism at the level of the right labia majora with SUVmax of 7.7 (arrows in Figure 3C&D). In the CT scan, an enlarged uterus of 16×17 cm with heterogeneous density, without fatty separation from annexes, and without metabolism was shown, which may correspond to a uterine myomatosis.
Figure 3.
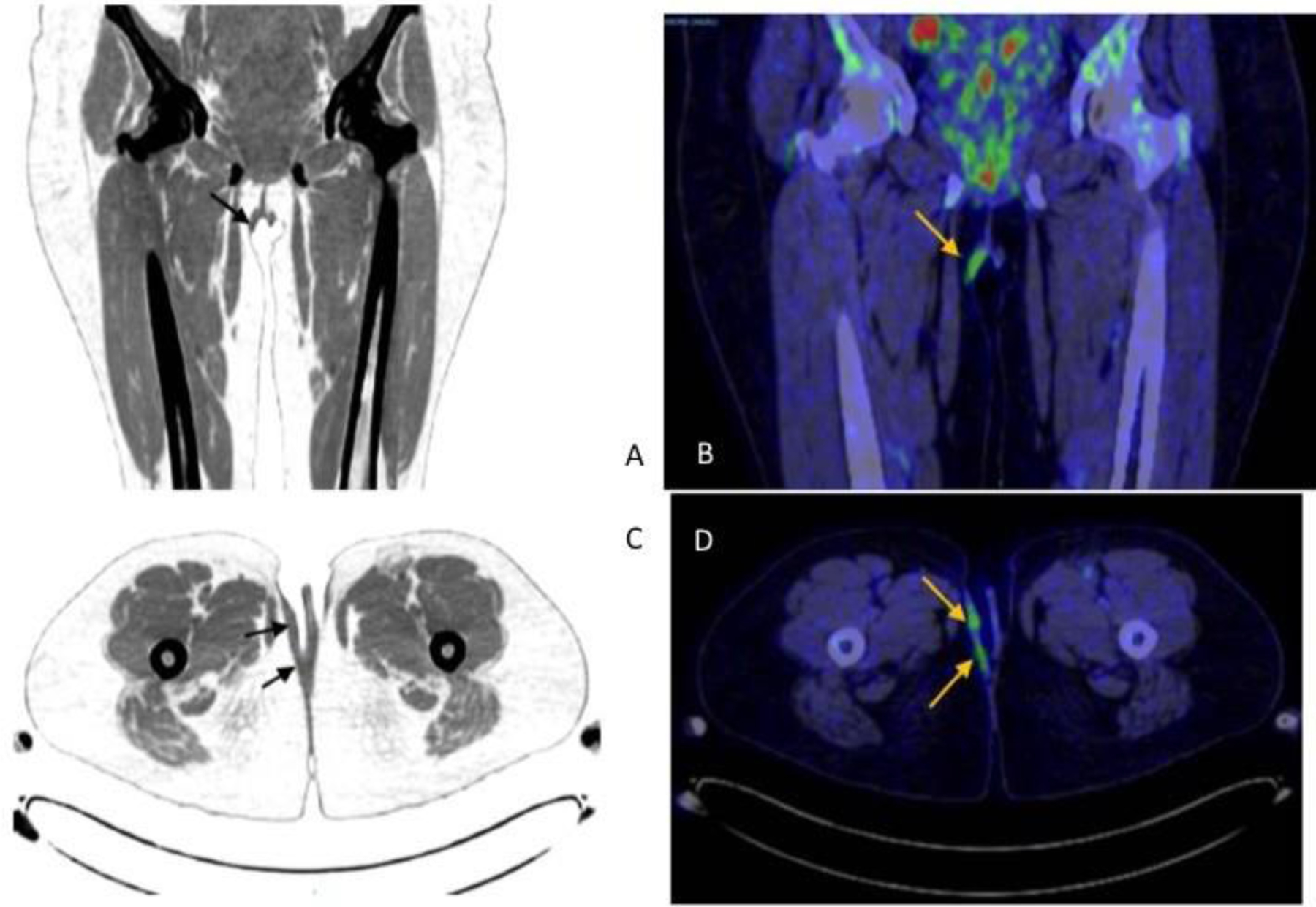
A and B. A coronal slice revealing a focal zone of hypermetabolism (demostrated with arrows) at the subcutaneoous vulvar lesión with a SUVmax of 7.7. In the CT scan, this hypermetabolic zone is demonstrated with arrows.
C and D. An axial slice revealing the same zone of hipermetabolism at the level of the right labia majora, demonstrated with arrows.
A wide local resection of the vulvar lesion and total hysterectomy with bilateral salpingectomy by laparotomy was performed. The pathology showed a melanoma in vertical growth phase associated with melanoma in situ, pigmented ulcerated, with perilesional lymphocytic response, without evidence of tumor regression, Breslow 3 mm and mitotic index of 5 per mm2. There was evidence of melanoma-in-situ at the medial and upper margins. The rest of the margins were negative for disease. In a separate procedure, the margins were resected, which were reported negative for disease. There was no evidence of disease in the hysterectomy and bilateral salpingo-oophorectomy specimen. She had an uncomplicated post-operative course and was discharge the day after surgery. Her last visit was on April 17, 2019, without any clinical evidence of recurrence.
Dr. Leitao
Closing Summary:
The management of melanoma has undergone significant clinically meaningful changes in recent years. Unfortunately, there are limited data specific to mucosal (including vulvovaginal) melanomas. Wide local excision with sentinel lymph node mapping is the current preferred approach as long as it does not require an exenterative procedure. I would recommend against exenterative procedures in patients with locally extensive disease, as these patients unfortunately have a significant risk of distant recurrence. In our institution, we favor use of immunotherapy with concurrent hypofractionated radiation therapy in such cases. Observation in patients with negative margin excisions and negative sentinel lymph nodes is preferred. In patients with positive sentinel lymph nodes, completion lymphadenectomy is not likely to offer a benefit over close groin surveillance. Adjuvant immunotherapy or targeted therapy should be offered to patients with node-positive vulvar melanoma. We can now offer truly beneficial adjuvant therapy while at the same time avoiding the morbidity of groin lymphadenectomy in the majority of cases. Close surveillance is important. The management of patients who develop recurrence must be individualized. Surgical excision of local recurrence and select other recurrences, such as isolated peritoneal or lung metastases, is a consideration. Extensive surgical resections; however, need to be very carefully considered. In patients who are not surgical candidates, immunotherapy is often the first choice of therapy. There is a real need for continued and dedicated research specific to mucosal melanomas and a multidisciplinary approach is always encouraged. The management of these rare tumors requires a multidisciplinary approach. At our institution, we arrange for all patients with vulvovaginal melanoma to also be seen by a member of the medical oncology team who specialize in mucosal melanoma. If needed, we also arrange for them to be seen by our dedicated radiation oncologist. All members of this comprehensive team see patients on the same day and this has helped in providing patients with the best possible treatment plan.
Contributor Information
Salim Abraham Barquet-Muñoz, Division of Surgery. Department of Gynecology, Instituto Nacional de Cancerología, México City.
Mario M Leitao, Jr, Division of Gynecology, Department of Surgery, Memorial Sloan-Kettering Cancer Center.
Bernardino Gabriel Santiago Concha, Department of Radiotherapy, Instituto Nacional de Cancerología, Mexico City.
María Delia Pérez-Montiel, Department of Pathology, Instituto Nacional de Cancerología, México City.
References
- 1.National Comprehensive Cancer Network. Cutaneous Melanoma (version 2.2019). https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf. Accessed May 17, 2019. [DOI] [PubMed]
- 2.Morton DL, Thompson JF, Cochran AJ, Mozzillo N, Elashoff R, Essner R, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med 2006;355:1307–1317 [DOI] [PubMed] [Google Scholar]
- 3.Faries MB, Thompson JF, Cochran AJ, Andtbacka RH, Mozzillo N, Zager JS, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med 2017;376:2211–2222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Leiter U, Stadler R, Mauch C, Hohenberger W, Brockmeyer N, Berking C, et al. Complete lymph node dissection versus no dissection in patients with sentinel lymph node biopsy positive melanoma (DeCOG-SLT): a multicenter, randomized, phase 3 trial. Lancet Oncol 2016;17:757–767 [DOI] [PubMed] [Google Scholar]
- 5.Eggermont AM, Chiarion-Sileni V, Grob JJ, Dummer R, Wolchok JD, Schmidt H, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med 2016;375:1845–1855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Weber J, Mandala M, Del Vecchio M, Gogas HJ, Arance AM, Cowey CL, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med 2017;377:1824–1835 [DOI] [PubMed] [Google Scholar]
- 7.Eggermont AMM, Blank CU, Mandala M, Long CV, Atkinson V, Dalle S, et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma. N Engl J Med 2018;378:1789–1801 [DOI] [PubMed] [Google Scholar]
- 8.Long GV, Hauschild A, Santinami M, Atkinson V, Mandala M, Chiarion-Sileni V, et al. Adjuvant dabrafenib plus trametinib in stage III BRAF-mutated melanoma N Engl J Med 2017;377:1813–1823 [DOI] [PubMed] [Google Scholar]
- 9.Boer FL, Ten Eikelder MLG, Kapiteijn EH, Creutzberg CL, Galaal K, van Poelgeest MIE. Vulvar malignant melanoma: Pathogenesis, clinical behavior and management: Review of the literature. Cancer Treat Rev. 2019. 73:91–103. [DOI] [PubMed] [Google Scholar]
- 10.Nagarajan P, Curry JL, Ning J, Piao J, Torres-Cabala CA, Aung PP, et al. Tumor thickness and mitotic rate robustly predict melanoma-specific survival in patients with primary vulvar melanoma: a retrospective review of 100 Clin Cancer Res. 2017,15;23:2093–2104 [DOI] [PubMed] [Google Scholar]
- 11.Prognostic Ladanvi A. and predictive significance of immune cells infiltrating cutaneous melanoma. Pigment Cell Melanoma 2015,28:490–500 [DOI] [PubMed] [Google Scholar]


