Abstract
The majority of aquatic toxicity data for petroleum products has been limited to a few intensively studied crude oils and Corexit chemical dispersants, and acute toxicity testing in two standard estuarine test species: mysids (Americamysis bahia) and inland silversides (Menidia beryllina). This study compared the toxicity of two chemical dispersants commonly stock piled for spill response (Corexit EC9500A®, Finasol®OSR 52), three less studied agents (Accell Clean®DWD dispersant; CytoSol® surface washing agent; Gelco200® solidifier), and three crude oils differing in hydrocarbon composition (Dorado, Endicott, Alaska North Slope). Consistent with listings on the U.S. National Contingency Plan Product Schedule, general rank order toxicity was greatest for dispersants and lowest for the solidifier. The results indicate that freshwater species can have similar sensitivity as the conventionally tested mysids and silversides, and that the sea urchin (Arbacia punctulata) appears to be a reasonable addition to increase taxa diversity in standardized oil agent testing.
Introduction
The vast majority of the aquatic toxicity data for oil products has been limited to Corexit chemical dispersants and a few intensively studied crude oils such as Prudhoe Bay/Alaska North Slope (ANSCO), Macondo 252/South Louisiana, and Fuel Oil No. 2 (e.g., Rice et al. 1979; Bejarano 2018; Echols et al. 2019). The diversity of species and endpoints measured in the toxicity testing of oil and spill response agents has been equally narrow, largely focused on acute lethality in two North American estuarine test species: mysids (Americamysis bahia) and inland silversides (Menidia beryllina) (Barron et al. 2013; Bejarano 2018). For example, Bejarano (2018) reported that toxicity records for sublethal endpoints and test durations greater than five days represented only 14% and 1%, respectively, of the available global toxicity data for dispersants. Even agents commonly stockpiled for oil spill response, such as Finasol® OSR52 (Finasol), have limited information beyond the acute toxicity to mysids and silversides (Bejarano 2018). Limited species diversity for assessing chemical hazards is not unique to oil and spill response agents and is a common challenge in ecological risk assessment (Wheeler et al. 2002).
While species diversity is typically low, the ability to benchmark product toxicity using mysids and silversides as the two standard test species continues to be an important aspect of the hazard assessment of oil and spill response agents (e.g., Barron et al. 2013; USEPA 2019). Historically, listing of agents for use in U.S. oil spill response has only required toxicity test data for mysids and silversides, and despite being only North American species, they have been used globally to assess the relative hazards of petroleum products and spill response agents (USEPA 1994; Barron et al. 2013; Bejarano 2018). Attributes include relative low cost, general availability, standardized laboratory culture and toxicity test protocols, sensitivity to a diversity of compounds, and toxicity data for hundreds of petroleum products (e.g., Nimmo and Hamaker 1983; Barron et al. 2013; USEPA 2016; Bejarano 2018).
The objective of the current study is to expand the knowledge base of the toxicity of oils and oil spill response agents using standardized oil mixing and aquatic testing procedures with five aquatic test species, including mysids and silversides as the conventional test organisms. The acute toxicity of spill response agents with only limited data (two chemical dispersants, a solidifier, a surface washing agent), and two infrequently tested crude oils were compared to the extensively studied dispersant Corexit EC9500A® (Corexit 9500) and ANSCO. Sublethal toxicity was also assessed in tests with the three dispersants because of the limited chronic toxicity data for oil spill response agents. The sea urchin Arbacia punctulata was included in the test battery because the species has been proposed in amendments to U.S. dispersant regulations in Subpart J (CFR 2015) of the National Oil and Hazardous Substances Pollution Contingency Plan (NCP). Sea urchins are in the phylum Echinodermata, and their inclusion would expand the diversity of standard aquatic test organisms beyond silversides (Phylum Chordata) and mysids (Phylum Arthropoda).
Materials and Methods
Test Species and Exposure Conditions.
Five aquatic species were used in a total of 29 definitive toxicity tests consisting of acute (48 or 96 h static non-renewal), short-term development (72 h static non-renewal), and 7 d growth and survival (static renewal) exposures with either crude oils or oil spill response agents (Table SI-1). Test species included two crustaceans (mysid shrimp, Americamysis bahia; waterflea, Ceriodaphnia dubia), two fish (inland silverside, Menidia beryllina; fathead minnow, Pimephales promelas) and one echinoderm (purple sea urchin, Arbacia punctulata). Products tested included three chemical dispersants (Corexit 9500, Finasol, Accell Clean® DWD (Accell), the surface washing agent CytoSol® (CytoSol), the solidifier Gelco 200® (Gelco), and three unweathered crude oils (Dorado, Endicott, Alaska North Slope (ANSCO)). Dispersants were only tested with saltwater species (mysid, silverside, sea urchin) because of current restrictions on the use of these agents in freshwater environments. Toxicity data were generated from tests with five concentrations and one control for each product and test species. Standard toxicity test protocols and exposure conditions followed those summarized in Barron et al. (2018), with the addition of the 72 h purple sea urchin larval development test (Table SI-2; USEPA 1995) and chronic mysid and silverside tests (Table SI-3). A fecundity endpoint was not incorporated into the chronic mysid test.
Preparation of Exposure Media.
Stock solutions of the spill response agents (no oil) were prepared separately in glass containers for each of the tested products at ambient laboratory temperatures with dilution water meeting the specific requirements for each test species: reconstituted seawater (20±2 psu for the mysid and the silverside; 30±2 psu for the sea urchin) or reconstituted freshwater (fathead minnow and waterflea). Mixing was first initiated at approximately 600 rpm to achieve a 40% vortex with a stir bar and magnetic stir plate, then an aliquot of each product was added at the center of the vortex. Mixing continued for 15 min, then agent-only stock was then serially diluted for use in toxicity tests without a settling period.
Water accommodated fractions (WAFs) of crude oils were prepared with oil loadings of either 25 g Dorado or Endicott, or 50 g of ANSCO per liter of water; the higher loading of ANSCO was used to achieve sufficient mortality for toxicity analyses. WAFs were prepared in sealed and covered glass jars under slow-stir low energy conditions maintaining a 20% vortex for 18 h and then allowed to settle for 6 h before sampling the aqueous exposure test solutions via slow siphon. The WAF was then serially diluted and used in toxicity tests following the variable dilution method of Barron and Kaaihue (2003).
Analytical Chemistry.
Aliquots of fresh source oils, WAF stock solutions collected immediately after preparation, and exposure media collected from test vessels at the end of the test period were analyzed as described in Barron et al. (2018) for monoaromatic hydrocarbons (BTEX; benzene, toluene, ethylbenzene and xylene), alkanes (C10-C35 normal aliphatics, and branched alkanes [pristine, phytane]), polycyclic aromatic hydrocarbons (PAHs), and total petroleum hydrocarbons (TPH; as total extracted petroleum hydrocarbons). PAH analytes included 2–4 ring compounds and their alkylated homologs (i.e., C0-C4 naphthalenes, C0-C4 phenanthrenes, C0-C3 fluorenes, C0-C4 dibenzothiophenes, C0-C4 napthobenzothiophenes, C0-C4 pyrenes and C0-C4 chrysenes).
Fresh source oils, stock solutions and exposure media were extracted with dichloromethane and analyzed for oil components following SW-846 Method 3500C (USEPA 2007a). BTEX were quantified using an Agilent 7890A Gas Chromatograph (GC) with a 5975C mass selective detector (MSD) with Triple Axis Detector and CombiPal autosampler (CTC Analytics) following USEPA Method 524.3 modified to perform head space analysis instead of purge and trap (USEPA 2009). Alkanes and 45 PAHs (parent and alkyl homolog groups) were quantified using an Agilent 6890N GC with an Agilent 5975 MSD and an Agilent 7683 series autosampler, equipped with a DB-5 capillary column by J&W Scientific (30 m, 0.25 mm I.D., and 0.25 mm film thickness) and a splitless injection port following EPA Method 8270D (USEPA 2014). TPHs were quantified with an Agilent 7890B GC equipped with a flame ionization detector (FID) and 7693 autosampler following EPA Method 8015C (USEPA, 2007b). Concentrations of the detected alkanes and PAHs were summed to compute total alkane and PAH concentrations, respectively. All hydrocarbon measurements met USEPA method quality assurance and quality control requirements, including precision, reproducibility and required detection limits.
Statistical Analyses.
The acute and/or chronic toxicity of each product to standard aquatic test species was estimated using test-specific dose response data. These were generated from the average response across replicates versus nominal concentrations for chemical agents (dispersants, surface washing agents, solidifier) and percent WAFs, or versus hydrocarbon metrics (BTEX, TPH, alkanes, PAH) measured in each WAF. Prior to calculations, corrections for BTEX, TPH and alkanes were made by subtracting any measured concentrations in control water samples from those in WAF samples. Effect concentrations for each metric of hydrocarbon exposure were based on concentrations in the stock WAF solution (initial measured), or on the geometric mean of the concentration in the stock WAF solution and exposure media at the end of the test period (day 2 or 4 of test). All statistical analyses were performed using the R statistical platform (v. 3.3.3) and associated packages (R Development Core Team, 2018; Ritz et al., 2015). Acute endpoints included the - 20th and median lethal concentrations (- LC20 and LC50, respectively), and chronic endpoints included - 20th and median effects concentrations (- EC20, EC50), as well as the no observed effect concentrations (NOEC) and lowest observed effect concentrations (LOEC). Acute to chronic ratios (ACR) for each the three dispersants were computed from the 48 hour (mysid) and 96 hour (silverside) median lethal concentrations divided by the geometric mean of the NOEC and LOEC values from the respective seven-day chronic test.
Comparisons with Global Toxicity Data.
Data generated in toxicity tests were compared with related literature values for the same tested product, hydrocarbon metric of toxicity, and test species. Most data used in comparisons were from the following data sources: NCP Product Schedule (USEPA, 2018) for all tested chemical agents, Bejarano (2018; data compilation) for dispersants, and Barron et al. (2018) for hydrocarbon metrics of toxicity. Toxicity data for dispersants were standardized to a consistent unit (mg/L) by accounting for the specific gravity of each tested product.
Results
Quality Assurance/Quality Control
All 29 definitive toxicity tests of crude oils (ANSCO, Dorado, Endicott), chemical dispersants (Accell, Corexit 9500, Finasol), Cytosol surface washing agent, and Gelco solidifier met quality control and quality assurance requirements, including control survival and water quality conditions (Table SI-2, SI-3; Barron et al. 2018). All analytical chemistry results met quality assurance/quality control protocols as specified in the research specific EPA Quality Assurance Project Plan.
Crude Oil and WAF Chemistry and Toxicity
The three unweathered test oils differed in density and composition of alkanes and PAHs, with ANSCO being unexpectedly more similar to Dorado (Gulf of Mexico) than Endicott oil also from the North Slope of Alaska (Table SI-4; Fig. SI-1; Fig. SI-2). As expected, WAFs of the three oils showed enrichment of napthalenes, and lower levels of three ring and larger PAHs compared to the source oils (Fig. 1). The absence of hopane in WAF indicated a lack of particulate oil in toxicity test media.
Figure 1.
Concentrations of PAHs and alkylated homologs unweathered crude oils and 100% WAF stock solutions prepared with ANSCO (50 g/L oil loading), Dorado (25 g/L) or Endicott (25 g/L oil.
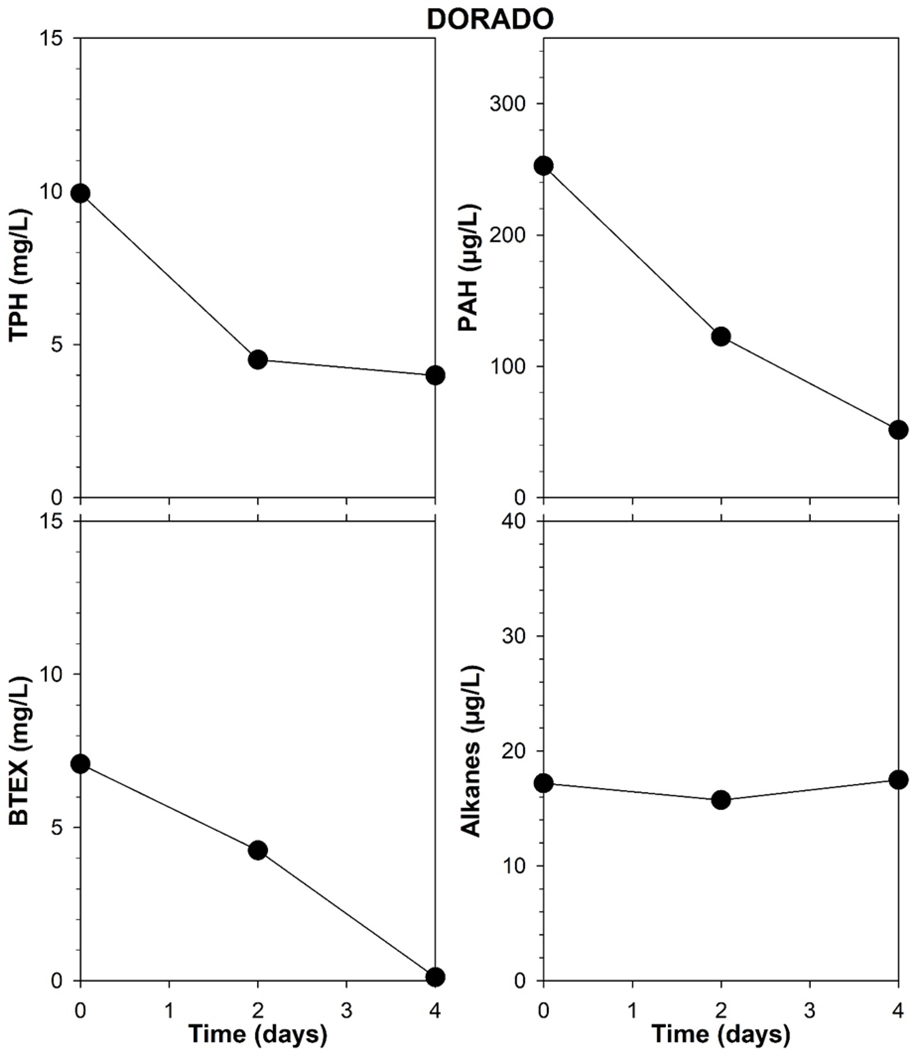
The three oils had generally similar acute toxicity, with LC50 values based on initial measured concentrations ranging from 4.8 to >13 mg TPH/L and 120 to >300 μg total PAHs/L (Table 1). TPH, PAHs, and BTEX in exposure media declined 50 to greater than 90% within the first 48 to 96 hours of toxicity testing, whereas alkane concentrations were relatively more constant (Fig. 2, 3, 4). LC50s based on the mean of concentrations measured at test start and test end were 51 to 90 percent lower, ranging from 2.2 to >5.8 mg TPH/L and 21 to 84 μg total PAHs/L.
Table 1.
Acute toxicity of unweathered Dorado, Endicott and Alaska North Slope (ANSCO) crude oils to the mysid shrimp (Americamysis bahia) and inland silverside (Menidia beryllina). Results are 48-hour (mysid) or 96-hour (silverside) median (LC50) and 20% (LC20) lethal concentrations expressed as percent of water accommodated fraction (WAF) and as either initial measured or mean measured hydrocarbon analytes: TPH (total petroleum hydrocarbons); PAH (sum of detected petrogenic polycyclic aromatic hydrocarbons).
| Oil1 | Test Species | Hydrocarbon | Initial Measured | Mean Measured | Difference | |||
|---|---|---|---|---|---|---|---|---|
| Metric Units | LC20 | LC50 | LC20 | LC50 | %2 | |||
| Mysid | TPH | mg/L | 4.13 | 4.76 | 1.88 | 2.16 | 53 | |
| Dorado | PAH | μg/L | 106 | 121 | 51 | 59 | 51 | |
| Silverside | TPH | mg/L | >9.93 | >9.93 | >3.99 | >3.99 | 60 | |
| PAH | μg/L | >253 | >253 | >52 | >52 | 79 | ||
| Mysid | TPH | mg/L | 6.10 | 7.02 | 3.18 | 3.66 | 48 | |
| Endicott | PAH | μg/L | 139 | 160 | 73 | 84 | 48 | |
| Silverside | TPH | mg/L | >13 | >13 | >5.01 | >5.01 | 61 | |
| PAH | μg/L | >299 | >299 | >74 | >74 | 75 | ||
| Mysid | TPH | mg/L | 9.09 | 11 | 4.48 | 5.23 | 52 | |
| ANSCO | PAH | μg/L | 179 | 209 | 18 | 21 | 90 | |
| Silverside | TPH | mg/L | >11 | >11 | >5.83 | >5.83 | 47 | |
| PAH | μg/L | >219 | >219 | >23 | >23 | 89 | ||
Slow-stir WAF prepared with 50 g ANSCO/L, 25 g Dorado/L, or 25 g Endicott/L.
Percentage difference in LC50 values determined from initial measured and mean measured LC50 values.
Figure 2.
Mean measured concentrations of TPH (total petroleum hydrocarbons), BTEX (benzene, toluene, ethylbenzene, xylenes), total PAHs (polycylic aromatic hydrocarbons), and alkanes in test solutions prepared with unweathered ANSCO (Alaska North Slope crude oil) at day 0, 2 and 4. BTEX value off scale (22.7 mg/L).
Figure 3.
Mean measured concentrations of TPH (total petroleum hydrocarbons), BTEX (benzene, toluene, ethylbenzene, xylenes), total PAHs (polycylic aromatic hydrocarbons), and alkanes in test solutions prepared with unweathered Dorado crude oil at day 0, 2 and 4.
Figure 4.
Mean measured concentrations of TPH (total petroleum hydrocarbons), BTEX (benzene, toluene, ethylbenzene, xylenes), total PAHs (polycylic aromatic hydrocarbons), and alkanes in test solutions prepared with unweathered Endicott crude oil at day 0, 2 and 4.
Spill Response Agent Toxicity
The acute toxicity of the five oil spill agents varied by product type, with LC50s ranging from 5.1 mg/L (Accell dispersant) to greater than 1000 mg/L (Gelco solidifier) (Table 2). Mysids were more sensitive than silversides to dispersants, but had similar sensitivity to the Cytosol surface washing agent and Gelco solidifier as the freshwater minnow and waterflea (Table 2). Lowest observed effect concentrations determined in sublethal tests with the three dispersants ranged from 2.5 to 250 mg/L, with sea urchin development affected at similar concentrations as mysids (Table 3). Acute to chronic ratios of acute lethality and growth effects in mysids and silversides over seven days ranged from 1.1 to 16 for the three dispersants (Table 3). Consistent with the other studies, including listings on the U.S. Product Schedule, the overall general rank order of acute toxicity to mysids and silversides was greatest for dispersants and lowest for solidifiers (Table 2, Table 4). For common spill response chemicals such as Finasol, the diversity of species and sublethal endpoints was very limited except for Corexit dispersants (Fig. 5).
Table 2.
Acute toxicity of five oil spill response agents to the mysid shrimp (Americamysis bahia), inland silverside (Menidia beryllina), waterflea (Ceriodaphnia dubia), and fathead minnow (Pimephales promelas). Data are 96-hour (fish) or 48-hour (invertebrate) median (LC50) and 20% (LC20) lethal concentrations and 95% confidence intervals (95%-CI) in parts per million (μL/L or mg GELCO/L).
| Product Type | Product Tested | Test Species | LC20 (95% CI) | LC501 (95% CI) | NCP2 LC50 |
|---|---|---|---|---|---|
| Dispersant | Corexit | Mysid | 65 (25–105) | 120 (71–169) | 32 |
| Silverside | 166 (129–203) | 201 (195–207) | 25 | ||
| Finasol | Mysid | 19 (18–20) | 28 (27–29) | 9.37 | |
| Silverside | 102 (83–121) | 113 (NC) | 12 | ||
| Accell | Mysid | 4.7 (1–8.4) | 5.1 (3.4–6.9) | 2.07 | |
| Silverside | 7.1 (3.6–11) | 7.9 (5–11) | 5.66 | ||
| Surface washing agent | CytoSol | Mysid | 558 (309–807) | >1,000 | 124 |
| Silverside | 399 (348–449) | 500 (489–514) | 738 | ||
| Waterflea | 159 (81–236) | 322 (226–418) | NA | ||
| Fathead | 130 (129–130) | 143 (141–146) | NA | ||
| Solidifier | Gelco | Mysid | >1,000 | >1,000 | >1,000 |
| Silverside | >1,000 | >1,000 | >10,000 | ||
| Waterflea | >1,000 | >1,000 | NA | ||
| Fathead | >1,000 | >1,000 | NA | ||
NC: not calculated.
NCP LC50: Toxicity value listed in the U.S. National Contingency Plan Product Schedule (NCP; USEPA 2019); NA: no toxicity value listed in NCP.
Table 3.
Chronic toxicity of three chemical dispersants to the mysid shrimp (Americamysis bahia) and inland silverside (Menidia beryllina) in 7-day growth and survival tests, and purple sea urchin (Arbacia punctulata) in 72-hour larval development test. Results are in parts per million (μL/L) as median (EC50) and 20% (EC20) effect concentrations with 95% confidence intervals (95%-CI), and no observed (NOEC) and lowest observed (LOEC) effect concentrations.
| Dispersant | Test Species | Endpoint | EC20 (95%-CI) | EC50 (95%-CI) | NOEC | LOEC | ACR1 |
|---|---|---|---|---|---|---|---|
| Corexit | Mysid | Survival | >100 | >100 | 50 | 100 | 1.7 |
| Biomass | 18 (NC) | >50 | 6.3 | 13 | 13 | ||
| Silverside | Survival | 165 (144–185) | 220 (207–233) | 125 | 250 | 1.1 | |
| Biomass | 162 (131–192) | 224 (203–244) | 125 | 250 | 1.1 | ||
| Urchin | Development | 17 (15–20) | 29 (26–31) | 6.3 | 12.5 | NC2 | |
| Finasol | Mysid | Survival | 10 (9–11) | 11 (6–16) | 5 | 10 | 4.0 |
| Biomass | 5 (1.5–8.4) | 8.4 (5.1–12) | 2.5 | 5 | 7.9 | ||
| Silverside | Survival | 8.9 (NC) | 9.8 (5.6–14) | 5 | 10 | 16 | |
| Biomass | 9.1 (NC) | 10 (9.5–10.5) | 5 | 10 | 16 | ||
| Urchin | Development | 11.4 (11.2–11.6) | 14 (13.7–14.1) | 12.5 | 25 | NC | |
| Accell | Mysid | Survival | >10 | >10 | 1.25 | 2.5 | 2.9 |
| Biomass | 2.1 (NC) | 2.3 (NC) | 1.25 | 2.5 | 2.9 | ||
| Silverside | Survival | 5 (4.5–5.4) | 5.5 (NC) | 5 | 10 | 1.1 | |
| Biomass | 5 (4.3–5.7) | 5.8 (1.2–10) | 1.25 | 2.5 | 4.6 | ||
| Urchin | Development | 6 (1.7–10) | 10.3 (5.9–15) | 6.25 | 12.5 | NC | |
ACR: acute to chronic ratio. acute LC50 divided by geometric mean of NOEC and LOEC.
NC: not calculated because of no acute LC50 value.
Table 4.
Summary of acute toxicity values (ppm;μL/L) from the U.S. National Contingency Plan Product Schedule (USEPA 2019).
| Product | Test Species1 | N2 | mean | Standard Deviation | Minimum | Maximum | Median |
|---|---|---|---|---|---|---|---|
| Bioremediation agents | menidia | 7 | 80539 | 192807 | 40.0 | 552762 | 392 |
| mysid | 7 | 51751 | 123132 | 21.0 | 353302 | 344 | |
| Dispersants | menidia | 19 | 148 | 444 | 1.90 | 1996 | 29.1 |
| mysid | 19 | 76 | 205 | 1.18 | 938 | 21.0 | |
| Miscellaneous agents3 | menidia | 9 | 54999 | 151688 | 5.13 | 484000 | 380 |
| mysid | 11 | 11473 | 29394 | 2.29 | 104000 | 297 | |
| Surface collecting agents (herders) | menidia | 2 | 71 | 67 | 3.33 | 138 | 70.7 |
| mysid | 2 | 146 | 140 | 6.83 | 286 | 146 | |
| Surface washing agents | menidia | 56 | 2025 | 7739 | 3.58 | 52233 | 110 |
| mysid | 57 | 962 | 2703 | 2.32 | 13461 | 91.7 |
menidia: Menidia beryllina; mysid: Americamysis bahia.
Number of agents tested.
Miscellaneous agents include solidifiers and other spill response chemicals, materials, and products.
Figure 5.
Acute and sublethal toxicity of Corexit and Finasol dispersants in standard freshwater and saltwater test species. Data compiled from the current study (black circle), static tests with standard species (Bejarano 2018; open circles), and the U.S. NCP Product Schedule (USEPA 2019; half circle).
Discussion
The purpose of the current study was three fold: (1) add to the knowledge base on the aquatic toxicity of less tested oils and spill response agents, (2) provide relative hazard data and taxa sensitivity in comparison to conventionally tested species (mysid, silverside) and products (Corexit 9500, ANSCO), and (3) provide information on new test methods and species proposed in amendments to U.S. dispersant regulations.
Crude oil toxicity
The majority of oil toxicity testing with aquatic organisms follows one of two general approaches: (1) exposure to rapidly declining exposure concentrations in tests with continuous dilution of spiked oil concentrations; or (2) exposure to serial (variable) dilutions of low energy slow-stir WAFs (Barron and Kaaihue 2003). Concerns for using the slow-stir WAF approach have included environmentally unrealistic constant exposures to oil, and entrainment of particulate oil in test media. The current study showed that hydrocarbon exposures in slow stir static WAF tests with three unweathered crude oils were not constant, with TPH, BTEX, and PAHs concentrations declining 40 to 99% within 48 hours. Hopane has been used as a geochemical biomarker of oil for decades and in oil spill forensics and source attribution (e.g., Wang et al. 2007). In the current study, hopane was used as an indicator of particulate oil in test media because of its recalcitrance and low solubility. The absence of hopane in WAFs prepared from three unweathered crude oils indicated negligible exposure to particulate oil in oil-only toxicity tests.
Reference oils are used to standardize the assessment of effectiveness and hazards of a range of spill response agents by using petroleum of the same physical and chemicals properties and composition. For example, Fuel Oil No. 2 has been used as the reference oil for U.S. listing of oil spill response agents (USEPA 1994). Crude oils have recently been proposed as reference oils in draft revisions to U.S. listing requirements (USEPA 2015). In the current study, three unweathered crude oils differing in aromatic and aliphatic composition were tested as potential reference oils. LC50 values were within a factor of 2.3 using TPH and PAH metrics of hydrocarbon exposure. Assessments of the global toxicity data for petroleum show a narrow range of oil toxicity, with general trends of middle distillates and heavy fuel oils more toxic than crudes and light oils (e.g., Barron et al. 2013). Crude oils typically have narrow ranges of acute toxicity (e.g., within 10-fold) in standardized tests, despite sometimes large ranges in hydrocarbon composition (Barron et al. 2013).
Toxicity of Spill Response Agents
Global toxicity data for comparing and assessing the hazards of oil spill response agents has been largely limited to acute lethality in two standard test species: the estuarine mysid and silverside. These two species have been shown to be generally sensitive to a broad range of chemicals across modes of toxic action and have been used to represent the sensitivity of a diversity of species in aquatic communities (Barron et al. 2013; Barron et al. 2015; Echols et al. 2016; Bejarano 2018). Testing with mysids and silversides have also been recommended for a variety of regulatory assessments including pesticide neurotoxicants, endocrine disruptors, and discharges of effluents into coastal waters (USEPA 2002; OECD 2005; USEPA 2016). The Product Schedule of the U.S. NCP (USEPA 2019) summarizes the toxicity of over 150 oil spill response agents to mysids and silversides determined under prescribed testing protocols. General rank order of acute toxicity to mysids and silversides across Product Schedule categories was greatest for dispersants and lowest for solidifiers. New toxicity data from the current study showed the same rank order, with LC50 values ranging from 5.1 mg/L for Accell dispersant to greater than 1000 mg/L for Gelco solidifier.
In the current study, rapid moderate energy mixing (40% vortex, 15 min) and dosing protocols were used for agent-only tests that differed from the standard low energy WAF methods used in oil-only tests. Mixing methods for spill response agents have received less standardization than oil and dispersant-oil methods, and can include short-term high energy mixing (e.g., USEPA 1994). The mixing protocol for the current study was selected as a compromise between current U.S. requirements for agent-only tests (USEPA 1994) and the desire to use the same mixing method for five agents that spanned the solubility range of fully miscible dispersants to the non-miscible solidifier. Loading concentrations of each of the agents were determined in preliminary studies to achieve a concentration-response series that spanned the median lethal concentration. An a priori decision to test a maximum concentration of 1000 mg/L was based on an initial assessment of agent miscibility.
Chronic toxicity data have been limited to a very few oil spill response agents because of an absence of regulatory requirements and perceived lack of chronic environmental exposures. The prolonged use of dispersants during the 2010 Deepwater Horizon spill in the Gulf of Mexico, and concerns for future spills in deep ocean and polar environments merit consideration of assessing the chronic hazards of selected spill response agents (Bejarano et al. 2017; Barron et al. 2019). In the current study of sublethal toxicity in three taxa of aquatic organisms, three dispersants had EC20s and LOECs ranging from 2 to 250 mg/L in constant exposures of three to seven days. Acute to chronic ratios of the three dispersants ranged from 1.1 to 16, similar to median ACR values for multiple chemicals, taxa groupings, and chronic toxicity test types (Raimondo et al. 2007). In risk assessment, ACRs are used as an index of the potential for adverse effects during longer term sublethal exposures. The ACRs from the current study suggest some potential for sublethal toxicity during prolonged environmental exposures of dispersants to sensitive life stages. Given the paucity of data on the chronic toxicity of most spill response agents, additional research is needed on the sublethal toxicity of those agents that may have prolonged environmental use.
Conclusions
The acute toxicity of oil spill response agents can vary over three orders of magnitude across product type and species, and even within specific categories of agents such as dispersants (Hansen et al. 2014). For example, George-Ares and Clark (2000) summarized effect concentrations for the dispersant Corexit 9500 in a diversity of species ranging from less than 1 to greater than 1000 mg/L. The acute toxicity of Corexit 9500 ranged from 2.3 to greater than a maximum tested concentration of 50 mg/L across eighteen Gulf of Mexico species tested under the same laboratory regime (Echols et al. 2019). In single laboratory tests with mysids and silversides, the acute toxicity of eight dispersants encompassing a range of effectiveness and chemical compositions had acute toxicity values spanning over 1000-fold (2.9 to > 5000 mg/L; Hemmer et al. 2011). The large variance in agent and species-specific toxicity observed in the current study and reported by others (e.g., Hansen et al. 2014) highlights the continued importance of testing mysids and silversides as benchmark species for assessing the relative acute hazards of oil and spill response agents. Additional research is needed to understand relationships between toxicity and the chemical composition and physical properties of spill response agents. Interpreting the benefits and risks of spill response agents requires an understanding of the complex relationships among relative hazards, application rates, efficacy, environmental tradeoffs, and ecological exposures (e.g., Baker 1995; Bejarano 2018).
Substantial species diversity in toxicity data for spill response agents is only available for the two Corexit dispersants 9500 and 9527 (Bejarano 2018). Acute and chronic toxicity values from the current study added substantially to the taxa and endpoint data available for Accell, Finasol, Gelco, and CytoSol, and was generally consistent with previous data for Corexit 9500. Accell and the other two dispersants were only tested with saltwater species (mysid, silverside, sea urchin), consistent with current restrictions on the use of these agents in freshwater environments. Increasing taxa diversity is a common need identified in risk assessments of environmental chemicals and has been recommended as a priority research need for oils and spill response agents (Barron et al. 2013; Bejarano et al. 2017). Sea urchins were tested in the current study to provide additional taxa diversity (Phylum Echinodermata) beyond the conventionally tested silversides (Phylum Chordata) and mysids (Phylum Arthropoda). Reference toxicant tests with sea urchin larvae were within acceptable precision, with coefficient of variation ranging from 21 to 22% in single laboratory tests and 39% across laboratories (USEPA 1995). The limited available information on oil and dispersants indicates that sea urchins may have similar sensitivity as mysids and menidia (Neff et al. 2000; Bejarano 2018). Based on the current assessment of three dispersants, inclusion of the sea urchin development test may be a valuable addition to standardized toxicity testing of marine spill response agents. Testing agents that may be used in freshwater environments with standard freshwater species would also appear to be a reasonable addition to toxicity testing regimes given the availability of organisms, standard culture and testing protocols, and consistent test results.
Supplementary Material
Acknowledgements
We thank Beth Moso and Mark Lampi and three anonymous reviewers for comments on a draft of this manuscript. Pegasus Technical Services, Inc. and Hydrosphere Research were contractors to the U.S. EPA. The views expressed in this article are those of the authors and do not necessarily reflect the opinions or policies of U.S. EPA. This publication does not constitute an endorsement of any commercial product. Data from this study are available through EPA’s Science Hub portal (https://sciencehub.epa.gov/sciencehub/datasets/2137).
References
- Baker JM. 1995. Net environmental benefit analysis for oil spill response. International Oil Spill Conference Proceedings: February-March 1995, Vol. 1995, No. 1, pp. 611–614. 10.7901/2169-3358-1995-1-611 [DOI] [Google Scholar]
- Barron MG Chiasson SC, Bejarano AC. 2019. Ecotoxicology of deep ocean spills In: Deep Oil Spills. Chapter 27, p. 466–479. Springer. [Google Scholar]
- Barron MG Conmy RN, Holder E, Meyer P, Wilson GJ, Principe VE, Wilming MM. 2018. Toxicity of Cold Lake and Western Canadian Select dilbits to standard aquatic test species. Chemosphere 191:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barron MG, Hemmer MJ, Jackson CR. 2013. Development of aquatic toxicity benchmarks for oil products using species sensitivity distributions. Integra Env Assess Manag 9, 610–615. [DOI] [PubMed] [Google Scholar]
- Barron MG, Lilavois CR, Martin TM. 2015. MOAtox: a comprehensive mode of action and acute aquatic toxicity database for predictive model development. Aquat Tox 16, 102–107. [DOI] [PubMed] [Google Scholar]
- Barron MG and Kaaihue L. 2003. Critical evaluation of CROSERF test methods for oil dispersant toxicity testing under subarctic conditions. Mar Poll Bull 46, 1191–1199. [DOI] [PubMed] [Google Scholar]
- Bejarano AC, 2018. Critical Review and Analysis of Aquatic Toxicity Data on Oil Spill Dispersants. Env Tox Chem 37, 2989–3001. [DOI] [PubMed] [Google Scholar]
- Bejarano AC Gardiner WW, Barron MG, Word JQ. 2017. Relative sensitivity of Arctic species to physically and chemically dispersed oil determined from three hydrocarbon measures of aquatic toxicity. Mar Poll Bull 122, 316–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CFR. 2015. National Oil and Hazardous Substances Pollution Contingency Plan; Proposed Rule Part III, 40 CFR Parts 110 and 300. U.S. Code of Federal Regulations; 80, 14 January 22, 2015. [Google Scholar]
- DeLorenzo M, Key P, Chung K, Pisarski E, Shaddrix B, Wirth E, Pennington P, Wade J, Franco M, Fulton M, 2018. Comparative toxicity of two chemical dispersants and dispersed oil in estuarine organisms. Arch Env Contam Tox 74, 414–430. [DOI] [PubMed] [Google Scholar]
- Echols BS, Smith A, Gardinali P, Rand G, 2016. An evaluation of select test variables potentially affecting acute oil toxicity. Arch Env Contam Tox 70, 392–405. [DOI] [PubMed] [Google Scholar]
- Echols BS, Langdon CJ Stubblefield WA, R GM and, Gardinali P. 2019. A comparative assessment of the aquatic toxicity of Corexit 9500 to marine organisms. Arch Env Contam Tox 77, 40–50. [DOI] [PubMed] [Google Scholar]
- Hansen BH Altin D, Bonaunet K, Overjordet IB. 2014. Acute toxicity of eight oil spill response chemicals to temperate, boral, and Arctic species. J Tox Env Health A 77, 9–11. [DOI] [PubMed] [Google Scholar]
- Hemmer MJ, Barron MG and Greene R. 2011. Comparative toxicity of eight oil dispersants, Louisiana sweet crude oil (LSC) and chemically dispersed LSC to two aquatic test species. Env Tox Chem 30, 2244–2252. [DOI] [PubMed] [Google Scholar]
- Neff JM, Ostazeski S, Gardiner W, Stejskal I 2000. Effects of weathering on the toxicity of three offshore Australian crude oils and a diesel fuel to marine animals. Env Tox Chem 19, 1809–1821. [Google Scholar]
- Nimmo DR Hamaker TL. 1982. Mysids in toxicity testing - a review. Hydrobiol 93, 171–178. [Google Scholar]
- OECD. 2005. Detailed review paper on aquatic arthropods in life cycle and two-generation toxicity tests OECD Environment Health and Safety Publication Series on Testing and Assessment No. 50. Organisation for Economic Co-Operation and Development; Paris, France: January 2005. [Google Scholar]
- R Development Core Team, 2018. R: A Language and Environment for Statistical Computing, in: R Foundation for Statistical Computing; (Ed.), Vienna, Austria. [Google Scholar]
- Raimondo S, Montague BJ, Barron MG. 2007. Determinants of variability in acute-to-chronic toxicity ratios (ACRs) in aquatic invertebrates and fish. Env Tox Chem 26, 2019–2023. [DOI] [PubMed] [Google Scholar]
- Rice SD, Moles A, Taylor TL, Karinen JF. 1979. Sensitivity of 39 Alaskan marine species to Cook Inlet crude oil and no. 2 fuel oil. In: Proceedings of the 1979 Joint Conference on Oil Spills (Prevention, Behavior, Control, Cleanup) Washington, DC: American Petroleum Institute; p 549–554 [Google Scholar]
- Ritz C, Baty F, Streibig JC, Gerhard D, 2015. Dose-response analysis using R. PloS one 10, e0146021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- USEPA. 1994. 40 CFR Parts 110 and 300. National Oil and Hazardous Substances Pollution Contingency Plan; Final Rule U.S. Federal Register, Vol. 59, No. 178. Thursday, September 15, 1994. [Google Scholar]
- USEPA. 1995. Short-term methods for estimating the chronic toxicity of effluents and receiving waters to west coast marine and estuarine organisms. EPA-600-R-95–136, Method 1008. Cincinnati, OH: August 1995. 661 pp. [Google Scholar]
- USEPA. 2002. Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. Fifth Edition. Washington, DC: EPA-921-R-02–012. October 2002. [Google Scholar]
- USEPA. 2007a. SW-846, Method 3500C. Organic Extraction and Sample Preparation. U.S. Environmental Protection Agency; February 2007. www.epa.gov/hwsw846/sw-846-test-method-3500c-organic-extraction-and-samplepreparation. [Google Scholar]
- USEPA, 2007b. SW-846, Method 8015C. Nonhalogenated Organics by Gas Chromatography. U.S. Environmental Protection Agency; February 2007. www.epa.gov/hw-sw846/sw-846-test-method-8015c-nonhalogenated-organics-gaschromatography. [Google Scholar]
- USEPA. 2009. Method 524.3. Measurable of Purgeable Organic Compounds in Water by Capillary Column Gas Chromatography/Mass Spectrometry. U.S. Environmental Protection Agency, Cincinnati, OH: EPA 815-B-09e009. June 2009. http://www.accustandard.com/assets/524_3.pdf. [Google Scholar]
- USEPA. 2014. SW-846, Method 8270D. Semivolatile Organic Compounds by Gas Chromatography/Mass Spectrometry. U.S. Environmental Protection Agency; July 2014. www.epa.gov/hw-sw846/sw-846-test-method-8270d-semivolatileorganic-compounds-gas-chromatographymass-spectrometry. [Google Scholar]
- USEPA. 2016. OCSPP 850.1075: Freshwater and saltwater fish acute toxicity test. Ecological Effects Test Guideline. EPA 712-C-16–007. Washington DC. [Google Scholar]
- USEPA. 2006. Swirling Flask Dispersant Effective Test, Revised Standard Dispersant Toxicity Test, and Bioremediation Agent Effective Test. CFR Part 300 Appendix C. July 1, 2006, p. 225–246. [Google Scholar]
- USEPA. 2015. 40 CFR Parts 110 and 300. National Oil and Hazardous Substances Pollution Contingency Plan; Proposed Rule. U.S. Federal Register, Vol. 80, No. 14. January 22, 2015. [Google Scholar]
- USEPA. 2018. Alphabetical List of NCP Product Schedule (Products Available for Use During an Oil Spill), p. 275. [Google Scholar]
- USEPA. 2019. NCP SUBPART J TECHNICAL NOTEBOOK A COMPENDIUM TO THE NCP PRODUCT SCHEDULE. U.S. Environmental Protection Agency; March 2019. [Google Scholar]
- Wang Z Stout SA, Fingas M. 2006. Forensic fingerprinting of biomarkers for oil spill characterization and source identification. Env Foren 7, 105–146. [Google Scholar]
- Wheeler JR Grist EPM, Leung KMY, Morritt D, Crane M. 2002. Species sensitivity distributions: data and model choice. Mar Poll Bull 45, 192–202. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.