Abstract
Killer meiotic drivers are genetic parasites that destroy ‘sibling’ gametes lacking the driver allele. The fitness costs of drive can lead to selection of unlinked suppressors. This suppression could involve evolutionary tradeoffs that compromise gametogenesis and contribute to infertility. Schizosaccharomyces pombe, an organism containing numerous gamete (spore)-killing wtf drivers, offers a tractable system to test this hypothesis. Here, we demonstrate that in scenarios analogous to outcrossing, wtf drivers generate a fitness landscape in which atypical spores, such as aneuploids and diploids, are advantageous. In this context, wtf drivers can decrease the fitness costs of mutations that disrupt meiotic fidelity and, in some circumstances, can even make such mutations beneficial. Moreover, we find that S. pombe isolates vary greatly in their ability to make haploid spores, with some isolates generating up to 46% aneuploid or diploid spores. This work empirically demonstrates the potential for meiotic drivers to shape the evolution of gametogenesis.
Research organism: S. pombe
Introduction
Parasites are pervasive in biology and can impose extreme fitness costs on their hosts (McLaughlin and Malik, 2017; Sorci and Garnier, 2008). Due to these fitness effects, there can be strong selection for variants of host genes that can subvert parasites (Kutzer and Armitage, 2016; McLaughlin and Malik, 2017; Sorci and Garnier, 2008). However, gene variants that promote host defense may be maladapted for other facets of host physiology, leading to evolutionary tradeoffs. For example, the sickle cell trait has been selected in malaria-endemic human populations as it provides heterozygous individuals some protection against the malaria-causing parasite, Plasmodium falciparum. However, the advantages of this allele come with a high cost as homozygotes develop sickle cell disease (Elguero et al., 2015; Serjeant, 2010).
In addition to external parasites like P. falciparum, organisms are also challenged by a variety of parasitic, or ‘selfish’, DNA sequences within their genomes (Burt and Trivers, 2006). Meiotic drivers are one type of selfish DNA element found throughout eukaryotes. Meiotic drive loci exploit meiosis to increase their chances of being passed on to the next generation. Rather than being transmitted to 50% of the progeny of a heterozygote, these selfish loci use a variety of tactics to promote their own transmission into up to 100% of the gametes (Sandler and Novitski, 1957; Zimmering et al., 1970). This cheating can impose a variety of fitness costs on the host (Zanders and Unckless, 2019). Due to these costs, variants that suppress meiotic drive can be favored by selection (Burt and Trivers, 2006; Crow, 1991; Hartl, 1975). Analogous to the sickle cell trait, this could lead to evolutionary tradeoffs where variants that are suboptimal for some aspect of gametogenesis may be selected due to their ability to mitigate the costs of meiotic drivers.
In this work, we explore the potential selective pressures meiotic drivers can impose on the evolution of gametogenesis. We use the fission yeast S. pombe as it is infested with multiple meiotic drive genes belonging to the wtf (with Tf) gene family (Bravo Núñez et al., 2020; Eickbush et al., 2019; Hu et al., 2017; Nuckolls et al., 2017). Different natural isolates contain between 4 and 14 predicted wtf drivers, almost all of which are found on chromosome 3 (Bowen et al., 2003; Eickbush et al., 2019; Hu et al., 2017). Each wtf meiotic driver encodes two proteins from two largely overlapping transcripts with distinct start sites: a poison (Wtfpoison) and an antidote (Wtfantidote) (Hu et al., 2017; Nuckolls et al., 2017). In wtf+/wtf- heterozygotes, all the developing meiotic products (spores) are exposed to the Wtfpoison, but only those that inherit the wtf+ allele express the corresponding Wtfantidote and neutralize the poison. This allows wtf drivers to gain a transmission advantage into the next generation by killing the spores that do not inherit the wtf+ allele from heterozygous (wtf+/wtf-) diploids (Hu et al., 2017; Nuckolls et al., 2017).
The poison and antidote proteins of a given wtf meiotic driver share a considerable length of amino acid sequence (>200 residues) (Hu et al., 2017; Nuckolls et al., 2017). This shared amino acid sequence may be important for a Wtfantidote to neutralize a given Wtfpoison protein. Strikingly, even a mismatch of two amino acids within the C-terminus can disrupt the ability of a Wtfantidote to neutralize a Wtfpoison (Bravo Núñez et al., 2018). The antidote of a given wtf gene generally does not neutralize poisons produced by other wtf drivers with distinct sequences (Bravo Núñez et al., 2018; Bravo Núñez et al., 2020; Hu et al., 2017). As a result, in diploids heterozygous for different wtf driving alleles, the drivers can be described as ‘competing’ as they exist on separate haplotypes and produce distinct Wtf proteins. When S. pombe isolates outcross, multiple wtf drivers may be in competition during gametogenesis due to rapid evolution of the gene family (Eickbush et al., 2019).
Here, we find that heterozygous, competing wtf drivers provide a selective advantage to atypical spores that inherit more than a haploid complement of wtf drivers. This selective advantage is due to the preferential elimination of haploid spores, as these spores are killed by the wtf driver(s) they do not inherit. The selected atypical spores include aneuploids, diploids, and spores inheriting wtf gene duplications resulting from unequal crossovers between homologous chromosomes. We use a combination of empirical analyses and modeling to demonstrate that competing wtf drivers generate an environment where variants that disrupt meiotic chromosome segregation can increase fitness. Finally, we show that variants that generate high numbers of atypical meiotic products may be common in S. pombe populations. We were unable to determine if meiotic drivers facilitated the evolution of these variants or if the high level of atypical spores produced by S. pombe could have enabled the success of the wtf drivers. Overall, this work demonstrates the capacity of meiotic drivers to impact the evolution of gametogenesis and suggests meiotic drive could have indirectly contributed to the high frequency of atypical spores generated by S. pombe diploids.
Results
The viable spores produced by outcrossed S. pombe diploids are frequently aneuploid or diploid
S. pombe cells generally exist as haploids but will mate to form diploids when starved of nutrients. If this starvation signal is continued, these diploids will proceed into meiosis resulting in the production of spores (Egel, 2004; Figure 1—figure supplement 1). Most S. pombe research is conducted on isogenic strains (derived from 968 h90, 972 h–, and 975 h+) (Fantes and Hoffman, 2016). We refer to the isogenic lab isolate in this work as Sp. Most of our knowledge about S. pombe meiosis thus stems from studying homozygous Sp diploids. Relatively little is known about the meiotic phenotypes of other isolates and of heterozygotes generated by crossing different haploid isolates (referred to as ‘outcrossing’ here) (Avelar et al., 2013; Hu et al., 2017; Jeffares et al., 2015; Zanders et al., 2014).
There are over 100 additional S. pombe isolates that have been sequenced and phenotypically characterized to some extent (Brown et al., 2011; Jeffares et al., 2015; Tusso et al., 2019). All known S. pombe isolates share an average DNA sequence identity of >99% for nonrepetitive regions, and nearly all of these isolates contain three chromosomes. Despite minimal sequence divergence between the strains, outcrossing often yields diploids that exhibit low fertility (i.e. they produce few viable spores) (Avelar et al., 2013; Gutz and Doe, 1975; Hu et al., 2017; Jeffares et al., 2015; Singh and Klar, 2002; Zanders et al., 2014). Differences in karyotype (such as chromosomal rearrangements) between isolates and pervasive meiotic drive are the two demonstrated causes of infertility in heterozygous S. pombe diploids (Avelar et al., 2013; Hu et al., 2017; Nuckolls et al., 2017; Zanders et al., 2014).
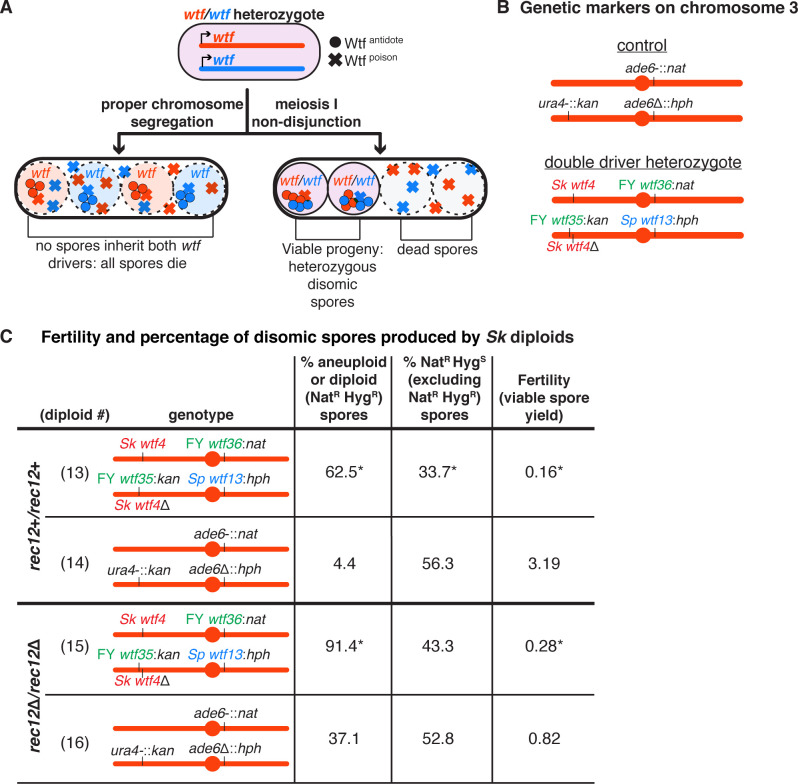
We previously characterized diploids generated by outcrossing Sp to another isolate named S. kambucha (Sk) (Nuckolls et al., 2017; Zanders et al., 2014). Although these Sp/Sk heterozygotes make few viable spores, the majority of the surviving spores are heterozygous aneuploids or diploids, as they contain both the Sp copy and Sk copy of chromosome 3 (Zanders et al., 2014). We refer to this phenotype as ‘disomy’ and refer to spores with this trait as ‘disomic’ (i.e. possessing two copies of a chromosome). Disomy of chromosome 3 is the only tolerable aneuploidy within Sp, as spores disomic for chromosomes 1 or 2, or spores lacking any of the three chromosomes are inviable (Niwa et al., 2006). Additionally, diploid spores, or spores possessing an extra copy of every chromosome are viable. Furthermore, the disomic state of chromosome 3 aneuploids is unstable and the extra chromosome 3 is quickly shed as the spores grow and form colonies (Niwa et al., 2006).
In this study, we first set out to determine if low spore viability and disomy are common amongst the viable spores produced by other outcrossed S. pombe diploids. We analyzed a series of heterozygous diploids made by mating different haploid natural isolates carrying genetic markers (ade6+ or ade6Δ::hphMX6) linked to centromere 3 (Figure 1A). We also measured fertility using the viable spore yield assay. This approach calculates the number of viable spores recovered for each cell placed onto the starvation media that induces spore formation. Ideally, a healthy diploid cell would yield four viable spores. Meiotic mutants can have low viable spore yields due to spore death, but factors like experimental technique, the number of mitotic divisions a diploid completes on the starvation media, and sporulation efficiency also affect viable spore yields. Because of this, viable spore yield values are best considered to have arbitrary units and should be compared to a control experiment carried out by the same researcher.
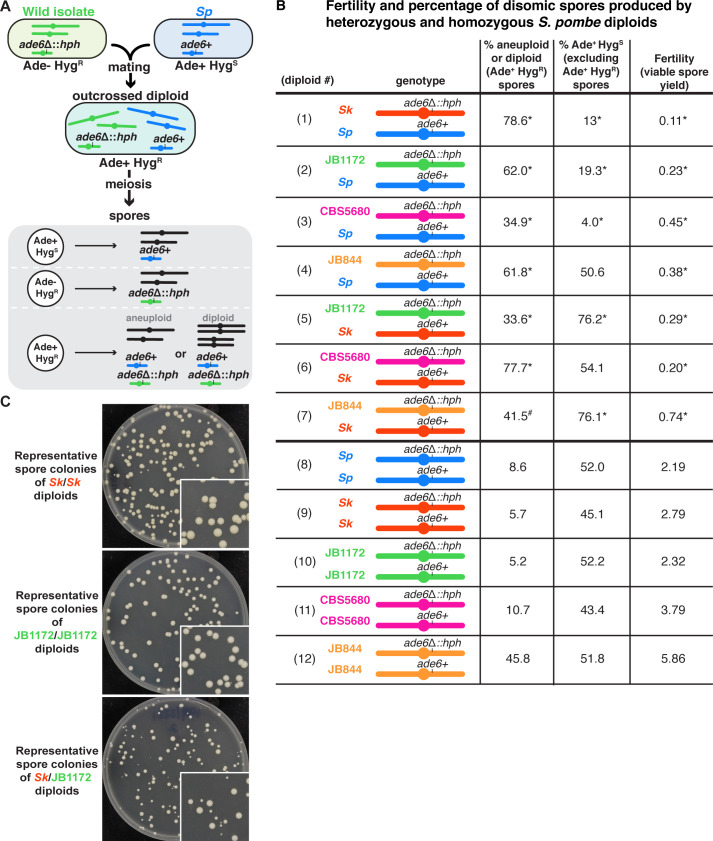
Figure 1. Outcrossed S. pombe diploids frequently produce disomic spores.
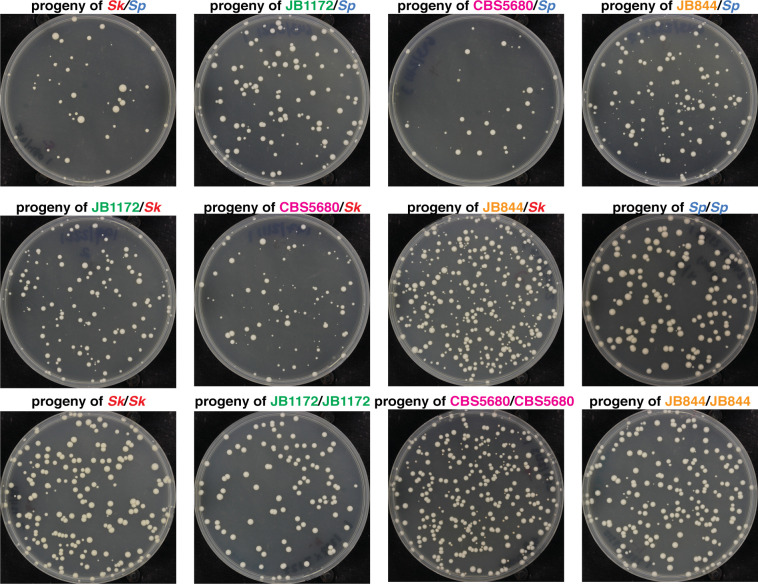
(A) Schematic of the experimental approach. The ade6 gene is linked to centromere 3. The karyotypes of JB1172, CBS5680, and JB844 are unknown, but outside of an inversion on Sp chromosome 1, Sp likely represents the ancestral karyotype (Avelar et al., 2013; Brown et al., 2011). (B) Phenotypes of heterozygous or homozygous S. pombe diploids. Allele transmission of chromosome 3 was assayed using co-dominant markers at ade6 (ade6+ and ade6Δ::hphMX6). The ade6+ allele confers an Ade+ phenotype, while the ade6Δ::hphMX6 provides resistance to Hygromycin B (HygR). Heterozygous aneuploid or diploid spores are Ade+ HygR. The fertility was measured using the viable spore yield assay to determine the number of viable spores per viable diploid. In the absence of drive, we expect 50% of the spores to be Ade+ HygS. A significant departure from 50% indicates drive favoring the overrepresented allele. The phenotypes of each heterozygote were compared to those of homozygous diploids from both parental strain backgrounds. * indicates p-value<0.025 (G-test [Ade+ HygR spores] and Wilcoxon test [fertility]) for the heterozygotes relative to the homozygous diploids from both parental backgrounds. Diploid 7 was only significantly different (p-value<0.025) in the frequency of Ade+ HygR spores when compared to diploid 9, but not when compared to diploid 12. This is indicated with #. To detect biased allele transmission (Ade+ HygS), diploids 2–4 were compared to diploid 8 and diploids 5–7 were compared to diploid 9. * indicates p-value<0.05 (G-test [allele transmission]). More than 200 viable spores were scored for each diploid. Raw data can be found in Figure 1—source data 1 and Figure 1—source data 2. (C) Representative images of the viable spore colonies generated by homozygous Sk and JB1172 diploids and heterozygous Sk/JB1172 diploids. Images of colonies generated by other diploids are shown in Figure 1—figure supplement 2.
Figure 1—figure supplement 1. The Schizosaccharomyces pombe life cycle.
Figure 1—figure supplement 2. Colony phenotypes of spores produced by S. pombe heterozygous and homozygous diploids.
Figure 1—figure supplement 3. Homozygous S. pombe diploids generate variable frequencies of disomic spores.
Similar to the previously characterized Sp/Sk heterozygotes, our genetic analyses revealed that each of the novel heterozygous diploids we tested had low viable spore yields compared to homozygous diploid controls (Figure 1B, compare diploids 2–7 to diploids 8–12). These reduced yields are likely due to increased spore death in the heterozygotes (Hu et al., 2017; Jeffares et al., 2015; Singh and Klar, 2002; Zanders et al., 2014).
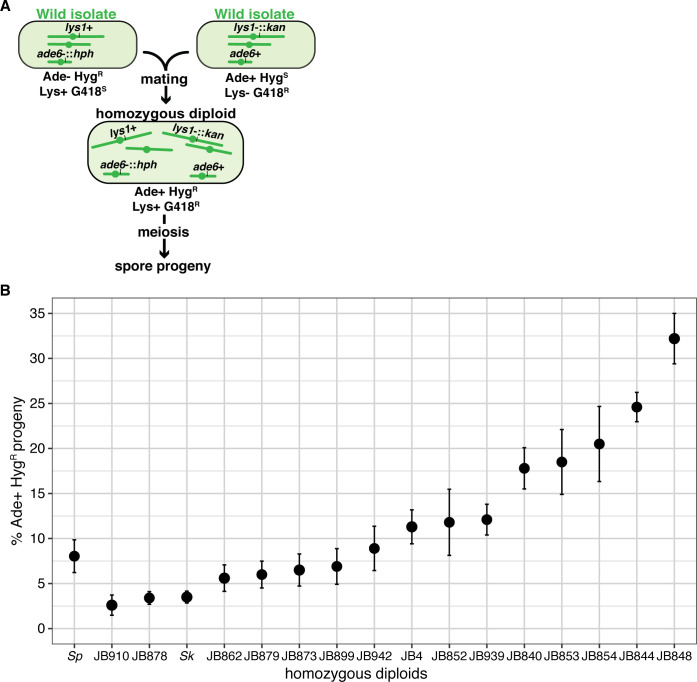
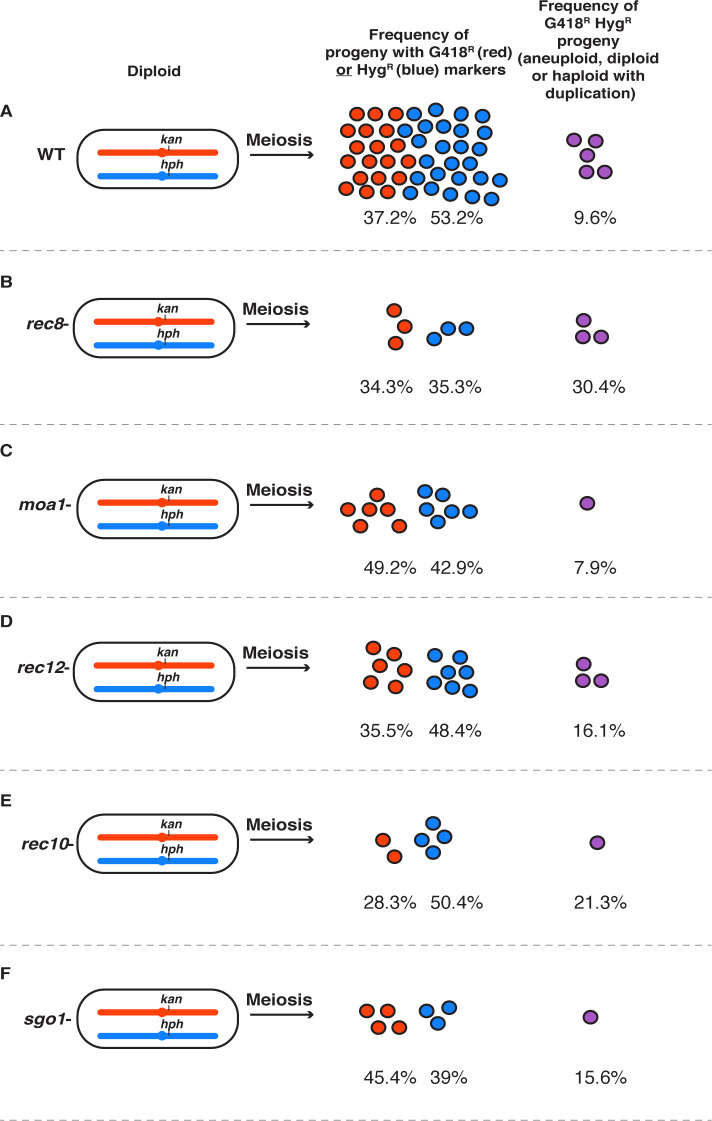
In addition to having low viable spore yields, the heterozygous diploids frequently produced spores that grew into small colonies with irregular shapes, a hallmark of aneuploidy (Figure 1C, Figure 1—figure supplement 2; Niwa et al., 2006). Consistent with this, 34–78% of the viable spores produced by heterozygotes were determined to be disomic for chromosome 3 as they inherited both centromere 3-linked markers (ade6+ and ade6Δ::hphMX6; Figure 1B, diploids 2–7). The frequency of disomic spores was generally higher in heterozygotes than homozygotes. However, there was considerable variation amongst the homozygotes with spore disomy frequencies ranging from 5–46% (Figure 1B, diploids 8–12). In additional experiments testing 14 more homozygous natural isolates, we observed a similar large range of disomy frequencies (Figure 1—figure supplement 3).
Multiple sets of competing meiotic drivers can select for disomic spores by killing haploids
We next wanted to determine why so many of the surviving spores produced by outcrossed S. pombe diploids were heterozygous disomes (aneuploids or diploids) for chromosome 3. We previously proposed a model in which distinct wtf meiotic drivers, which are nearly all on chromosome 3, were killing haploid spores (López Hernández and Zanders, 2018; Zanders et al., 2014). We hypothesized that in the presence of diverged meiotic drivers on opposite haplotypes, haploid spores will inherit only one set of drivers and be killed by the drivers they do not inherit. However, heterozygous disomic spores are more likely to inherit all the competing wtf drive alleles. These disomic spores should thus survive, as they will contain every Wtfantidote necessary to counteract the Wtf poisons (Figure 2A). For example, if two S. pombe isolates containing diverged wtf meiotic drivers were to mate and undergo meiosis, the drivers from one strain would be put in direct competition against the drivers in the other strain. As only spores that inherit both drivers (from both strains) would survive the toxicity of the Wtf poisons, the resulting population would be enriched for these atypical meiotic products. Consistent with this model, Sp/Sk heterozygotes do not make more disomic spores per meiosis than Sp or Sk homozygotes (Zanders et al., 2014).
Figure 2. A high fraction of viable spores are disomic in Sk strains with wtf competition at two loci.
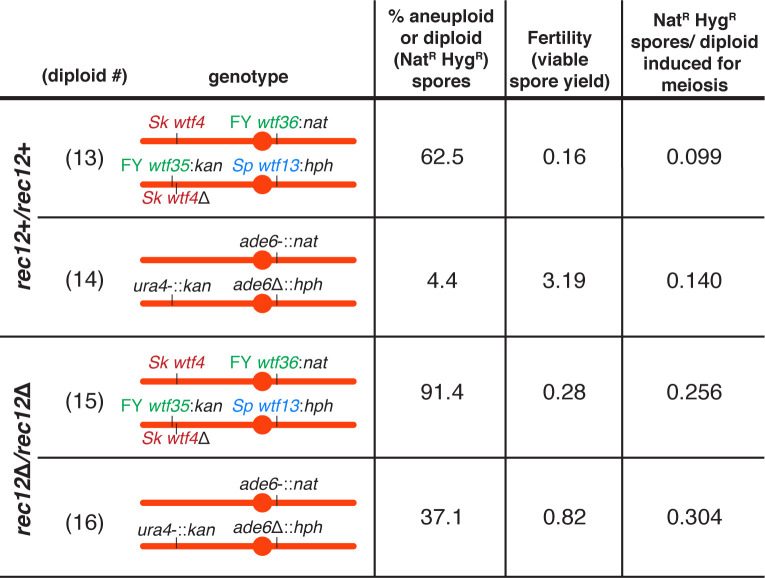
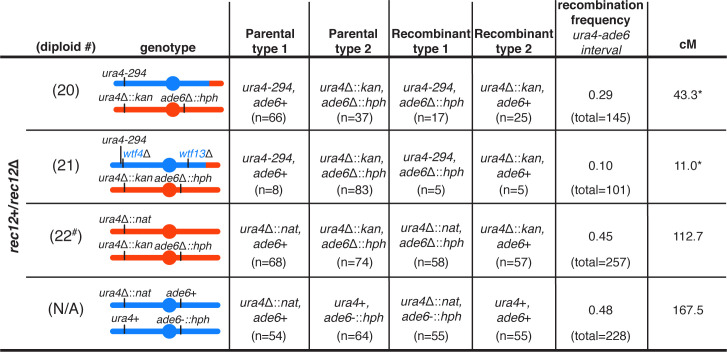
(A) Model for a diploid heterozygous for distinct wtf meiotic drivers. Spores are destroyed by any wtf driver that they do not inherit from the diploid progenitor cell. Meiosis I chromosome missegregation is one mechanism by which spores can inherit wtf alleles on competing haplotypes and survive. (B) Schematic of the genetic markers at ura4 and ade6 in the control diploid and the wtf transgenes inserted at ura4 and ade6 in Sk chromosome 3 in the double driver heterozygote. wtf genes from the Sp, Sk, and FY29033 strains are depicted in blue, red, and green, respectively. The wtf drivers shown here drive when heterozygous and do not counteract the effect of the other drivers (see Figure 2—figure supplement 1). (C) Phenotypes of the double driver heterozygote or control diploid in rec12+ (top) and rec12Δ (bottom) strain backgrounds. We expect NatR HygS spores to be present at 50% in the viable population. A significant departure from the expected 50% indicates drive favoring the overrepresented allele. For statistical analyses, the frequency of disomic spores, allele transmission, and fertility in the double driver heterozygotes was compared to the control diploids. Diploid 13 was compared to control diploid 14, and diploid 15 was compared to control diploid 16. * indicates p-value<0.05 (G-test [allele transmission and NatR HygR spores] and Wilcoxon test [fertility]). The data for diploid 14 were previously published in Bravo Núñez et al., 2020. Raw data can be found in Figure 2—source data 1 and Figure 2—source data 2.
Figure 2—figure supplement 1. Wtfantidote proteins are generally specific for a Wtfpoison and do not provide cross-resistance to other Wtfpoison proteins.
Figure 2—figure supplement 2. Sp diploids with wtf competition at two loci have a high fraction of viable spores that are disomic.
Figure 2—figure supplement 3. wtf poison-only alleles do not enrich for disomes in viable spores.
Figure 2—figure supplement 4. Competing wtf meiotic drivers do not affect the frequency of disomes generated per meiosis.
Figure 2—figure supplement 5. The recombination frequency is altered in Sk diploids with competing wtf meiotic drivers.
To test our model, we engineered an Sk diploid that is heterozygous for two unlinked sets of wtf drivers on chromosome 3. This diploid was generated by crossing two Sk strains that, apart from the different wtf drivers we integrated, are isogenic. We refer to this diploid as the ‘double driver heterozygote’ (Figure 2B). Importantly, all four of the drivers in this strain are functional and cannot fully suppress any of the other three drivers (Figure 2—figure supplement 1; Bravo Núñez et al., 2018; Bravo Núñez et al., 2020; Nuckolls et al., 2017). Consistent with our hypothesis, we found that 63% of the viable spores generated by the double driver heterozygote appeared to be disomic as they inherited both alleles at ade6 (ade6-:FY29033 wtf36:natMX4 and ade6-:Sp wtf13:hphMX6). This was considerably higher than the control diploid (no heterozygous wtf drivers) in which 4% of the viable spores displayed this phenotype (Figure 2C, compare diploid 13 to 14). Additionally, many of the viable spores produced by the double driver heterozygote generated small, misshapen colonies characteristic of aneuploids. Finally, the fertility of the double driver heterozygote was 20-fold lower than the control diploid (Figure 2C, compare diploid 13 to diploid 14). To test if our results were dependent on strain background, we made an analogous double driver heterozygote in the Sp strain background and observed a similar decrease in fertility and increase in disomy amongst the surviving spores (Figure 2—figure supplement 2). These phenotypes are consistent with the destruction of haploid spores that do not inherit every driver.
We also considered an alternative hypothesis that meiosis in the double driver heterozygotes more frequently produces disomic spores. The Wtfpoison proteins can be detected at low levels prior to the meiotic divisions and could thus theoretically alter the fidelity of chromosome segregation (Nuckolls et al., 2017). However, it is important to note that every S. pombe meiosis likely occurs in the presence of multiple distinct Wtfpoison proteins. This makes it difficult to imagine how heterozygosity for competing wtf drivers could specifically interfere with chromosome segregation. Nevertheless, we tested if expressing distinct Wtfpoison proteins from a heterozygous conformation promoted the production of disomic spores. To do this, we used separation-of-function alleles (Sk wtf28poison and Sk wtf4poison) that produce the Wtfpoison proteins, but not the corresponding Wtfantidote proteins. We found that Sk wtf28poison/Sk wtf4poison heterozygotes had very low viable spore yields, but did not exhibit a higher frequency of disomic spores than control diploids (Figure 2—figure supplement 3). This is consistent with our previous analyses of hemizygous (wtfpoison/ade6+) mutants in which we did not observe a high frequency of disomic spores (Bravo Núñez et al., 2018; Nuckolls et al., 2017). These results argue against a role for Wtf proteins in affecting chromosome segregation.
In addition, we used the observed frequency of disomes and the viable spore yield to calculate the number of disomes produced per diploid cell placed on sporulation media. This calculation approximates the number of disomic spores generated per meiosis. We found that the number of disomic spores produced per cell placed on sporulation media did not increase between the double driver heterozygote and the control diploid (Figure 2—figure supplement 4, compare diploid 13 to diploid 14). These results are also inconsistent with the hypothesis that wtf drivers affect the frequency at which meiosis generates disomic spores.
Together, our results are consistent with our hypothesis that disomic spores generated from diploids carrying multiple sets of heterozygous wtf meiotic drivers are enriched in the surviving population due to the destruction of haploid spores, rather than due to Wtf proteins increasing the rate of meiotic chromosome missegregation.
Heterozygosity at wtf loci contributes to the high frequency of disomic spores generated by outcrossed diploids
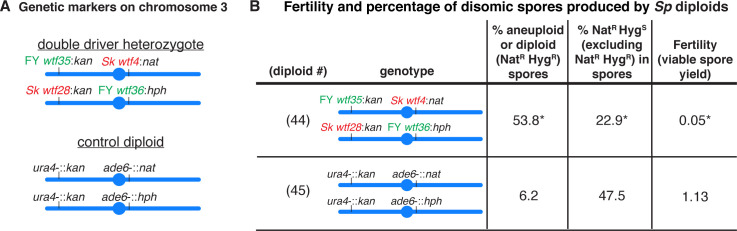
To further determine the contribution of competing wtf drivers to the high level of disomic spores observed in the viable progeny, we decided to test our model in a strain background with more extensive heterozygosity for wtf drivers, like those generated by outcrossing. For these experiments, we started with an Sp/Sk mosaic diploid strain that is heterozygous for eight known or predicted wtf meiotic drivers (Bravo Núñez et al., 2020; Eickbush et al., 2019). This mosaic diploid is homozygous for Sk chromosomes 1 and 2 but is heterozygous for most of chromosome 3 for Sp- and Sk-derived sequences (Figure 3A). These diploids also lack rec12, a gene which encodes the endonuclease that initiates meiotic recombination by generating DNA double-strand breaks (DSBs) (Bergerat et al., 1997; Keeney et al., 1997). The lack of induced recombination in these diploids results in absolute linkage within the Sp and Sk haplotypes on chromosome 3. Because of this, haploid spores will generally inherit either every Sp driver or every Sk driver. We again used markers at the centromere-linked ade6 locus to determine the transmission of Sp- and Sk-haplotypes on chromosome 3.
Figure 3. wtf meiotic driver competition contributes to the high disomy in spores produced by Sp/Sk mosaic diploids.
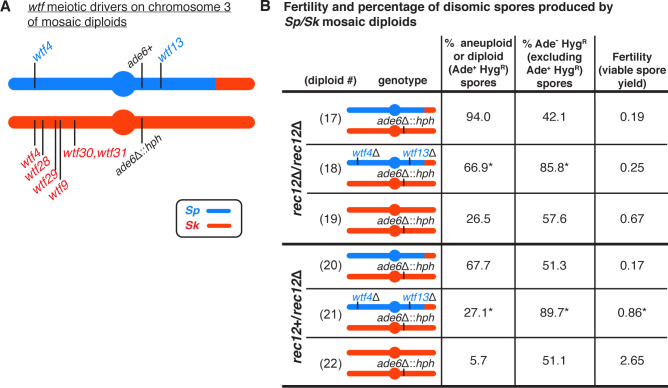
(A) Schematic of the predicted wtf meiotic drivers found on chromosome 3 of the Sp/Sk mosaic diploid. Sp-derived DNA is depicted in blue and Sk-derived DNA in red. (B) Phenotypes of mosaic and control diploids in rec12∆/rec12∆ and rec12+/rec12∆ backgrounds. Allele transmission of chromosome 3 was assayed using markers at ade6 (linked to centromere 3). In the absence of drive, we expect 50% of the spores to be Ade- HygR. Any significant deviation from the expected 50% indicates drive favoring the overrepresented allele. To determine the contribution of wtf meiotic drivers to the frequency of disomic spores and fertility, diploid 18 was compared to diploid 17, and diploid 21 was compared to diploid 20. To determine if there was biased allele transmission, diploids 17 and 18 were compared to control diploid 19, and diploids 20 and 21 were compared to control diploid 22. More than 300 viable spores were scored for each diploid. * indicates p-value<0.05 (G-test [allele transmission and Ade+ HygR spores] and Wilcoxon test [fertility]). Raw data can be found in Figure 3—source data 1 and Figure 3—source data 2.
Figure 3—figure supplement 1. Single deletions of Sp wtf meiotic drivers partially decrease the frequency of disomic spores produced by Sp/Sk mosaic diploids.
Figure 3—figure supplement 2. Observed recombination frequencies are altered by meiotic drive in Sp/Sk mosaic diploids.
Consistent with our previous observations in a similar mosaic diploid (Nuckolls et al., 2017), we saw that the viable spores generated by this diploid were almost exclusively (94%) heterozygous disomes for chromosome 3 (Figure 3B, diploid 17). Recombination promotes faithful segregation of chromosomes, so the lack of recombination in this mosaic diploid likely contributed to the high disomy we observed amongst the viable spores. Lack of recombination is, however, insufficient to explain the majority of the phenotype, as rec12Δ Sk homozygous diploids generate only 27% disomic spores (Figure 3B, diploid 19).
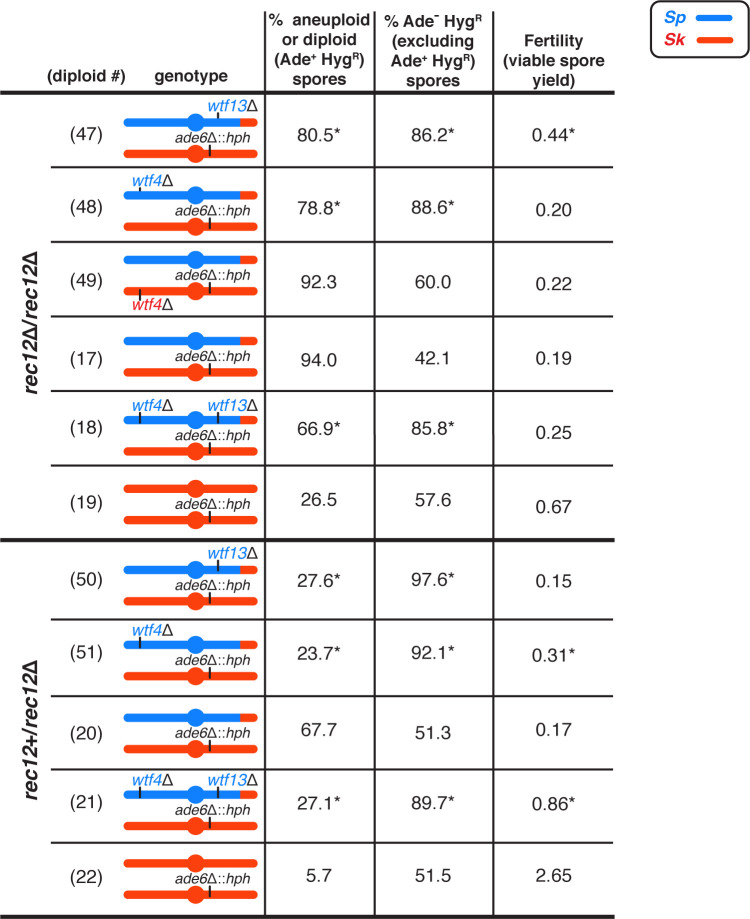
To test if the extremely high frequency of disomic spores generated by the Sp/Sk mosaic diploid was dependent on wtf meiotic drivers, we deleted the predicted Sp drivers, Sp wtf13 and Sp wtf4. This eliminated wtf driver competition as the remaining wtf drivers were either on the same haplotype or homozygous. Consistent with our hypothesis, deleting both Sp drivers significantly decreased the frequency of chromosome 3 heterozygous disomes (from 94% to 67%; Figure 3B, diploids 17 and 18), although it did not significantly increase fertility. Deleting only one of the two Sp drivers was also sufficient to significantly decrease the frequency of disomic spores (Figure 3—figure supplement 1, diploids 47 and 48). However, deleting only one of the six predicted Sk drivers (wtf4) had no effect (Figure 3—figure supplement 1, diploid 49). This was expected as considerable wtf driver competition remains as Sk still has five intact drivers.
We also performed analogous experiments in the presence of meiotic recombination by mating the mosaic haploid strain to a rec12+ Sk strain. Meiotic recombination will produce chromosomes with new combinations of Sp and Sk wtf drivers. Despite these new combinations, our model predicts that heterozygous disomic spores will still have a fitness advantage as they are more likely to inherit every wtf driver. We observed that this rec12+/rec12Δ diploid had low fertility, similar to that of the rec12Δ Sp/Sk mosaic diploid (Figure 3B, compare diploid 17 to diploid 20). To assay disomy amongst the spores produced by the rec12+/rec12Δ mosaic diploid, we again genotyped the ade6 locus (Figure 3A). We found that 68% of the viable spores generated by this mosaic appeared to be disomic for chromosome 3. Deleting both Sp wtf4 and Sp wtf13 in the rec12+/rec12Δ mosaic diploid significantly increased fertility and decreased disomy at ade6 amongst the viable spores from 68% to 27% (Figure 3B, diploids 20 and 21).
We also examined single deletions of Sp wtf4 or Sp wtf13 in a Rec12+ mosaic diploid. Deleting Sp wtf4 or Sp wtf13 individually decreased disomy amongst the viable spores, but only the Sp wtf4 deletion significantly increased fertility (Figure 3—figure supplement 1, diploids 50 and 51). These results demonstrate that wtf driver competition contributes to the extremely high frequency of disomes amongst the surviving spores and can contribute to low spore viability in these mosaic strain backgrounds. However, wtf competition alone was insufficient to explain the total increase in disomy relative to the Sk homozygotes (Figure 3, compare diploid 18 to diploid 19, and diploid 21 to diploid 22). Overall, our results support the model that high disomy observed in the spores generated by outcrossed S. pombe diploids is partially due to competing wtf meiotic drivers.
Driver landscapes affect observed recombination rates on collinear haplotypes
Meiotic drivers are often associated with regions of suppressed recombination, such as chromosomal inversions (Dobzhansky and Sturtevant, 1938; Dyer et al., 2007; Hammer et al., 1989; Larracuente and Presgraves, 2012; Pieper and Dyer, 2016; Stalker, 1961; Svedberg et al., 2018). This state of recombination suppression is thought to be indirectly caused by the driver as linked loci also enjoy a transmission advantage. However, there is little empirical evidence about how the presence or absence of drivers can directly affect recombination landscapes. Fortuitously, our experiments allowed us to address this question by assaying how drivers can affect recombination in otherwise isogenic strains.
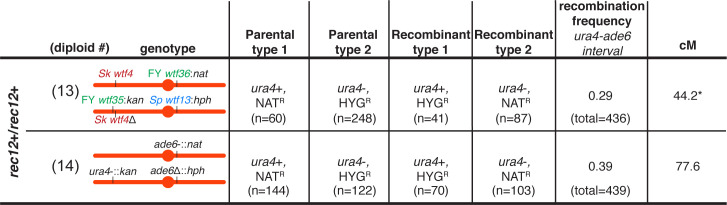
We first assayed recombination between the ade6 and ura4 loci of the double driver heterozygote and control diploid described in Figure 2B. The ade6 and ura4 loci are over 75 cM apart in the Sk control (Figure 2—figure supplement 5). In the Sk double driver heterozygotes, this distance decreased significantly to 44 cM (p-value=0.03, G-test). We hypothesize this is because recombination can uncouple two of the strongest drivers, Sp wtf13 and FY29033 wtf35, which are found on the same haplotype.
We also analyzed the effect of drivers on recombination in the rec12+/rec12Δ mosaic diploids described in Figure 3. In the mosaic diploids with all drivers intact, we observed ade6 and ura4 were 43 cM apart (Figure 3—figure supplement 2), similar to the 62 cM previously observed in Sp/Sk hybrids (Zanders et al., 2014). However, when Sp wtf4 and Sp wtf13 were deleted from the mosaic strain, the observed genetic distance decreased significantly to 11 cM (Figure 3—figure supplement 2; p-value<0.01, G-test). We hypothesize this drop is due to preferential death of recombinants, as recombination would lead to haploids failing to inherit every Sk driver. Overall, our results demonstrate that meiotic drivers can directly affect recombination landscapes.
Meiotic driver competition at a single locus selects for atypical meiotic products
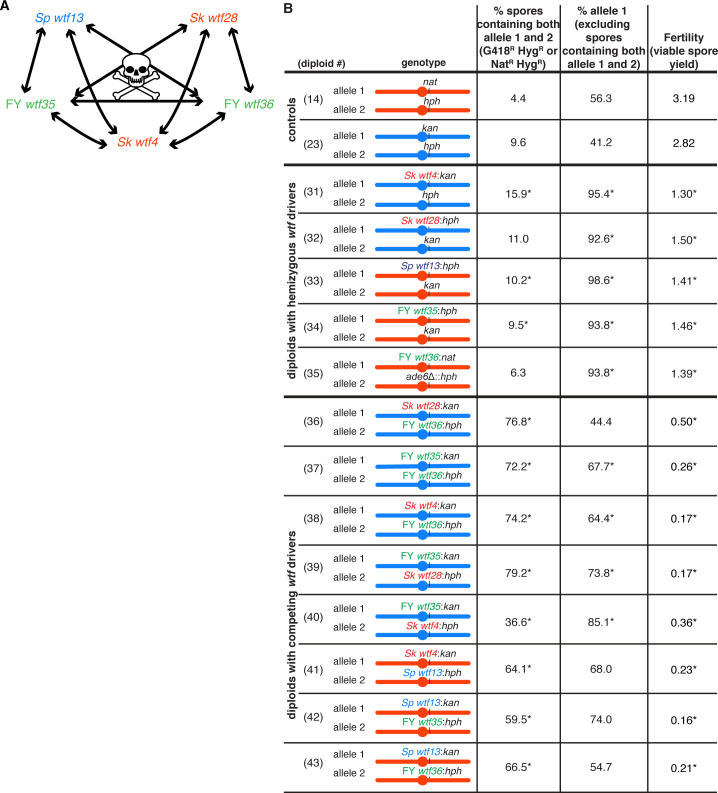
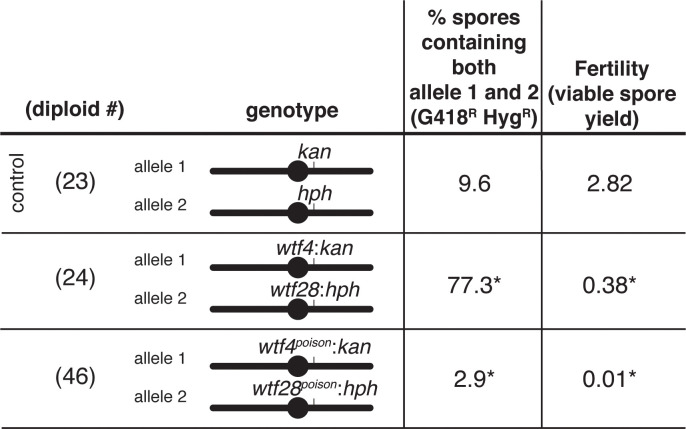
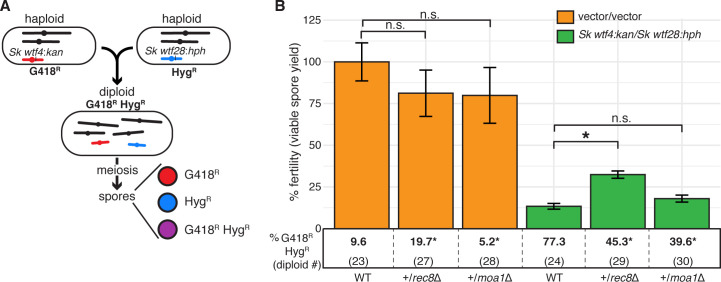
The experiments above test scenarios with at least two sets of competing wtf drive genes. However, when more closely related isolates mate, the number of heterozygous wtf driver loci will be reduced. We analyzed this scenario by measuring the impact of one set of competing meiotic drivers in a diploid. To do this, we tested an Sp diploid heterozygous for Sk wtf4 and Sk wtf28 transgenes integrated at the ade6 locus (Figure 4A). We compared this diploid to a control heterozygote (empty vectors at ade6). We found that Sk wtf4/Sk wtf28 heterozygous diploids had decreased fertility (13% of the control diploid; Figure 4C and Figure 4—figure supplement 1) and 77% of the viable spores appeared to be disomic for chromosome 3 as they inherited both Sk wtf4- and Sk wtf28-linked drug resistance markers (G418R HygR spores, Figure 4C). These phenotypes are not specific to those drivers, the Sp strain background, or the ade6 locus. We also observed similar phenotypes in an Sk background and with drivers at the ura4 locus (Figure 2—figure supplement 1 and Figure 4—figure supplement 2). As in our previous experiments, these phenotypes are consistent with the destruction of spores that do not inherit both wtf drivers leading to the enrichment of spores that inherit both drivers amongst the survivors.
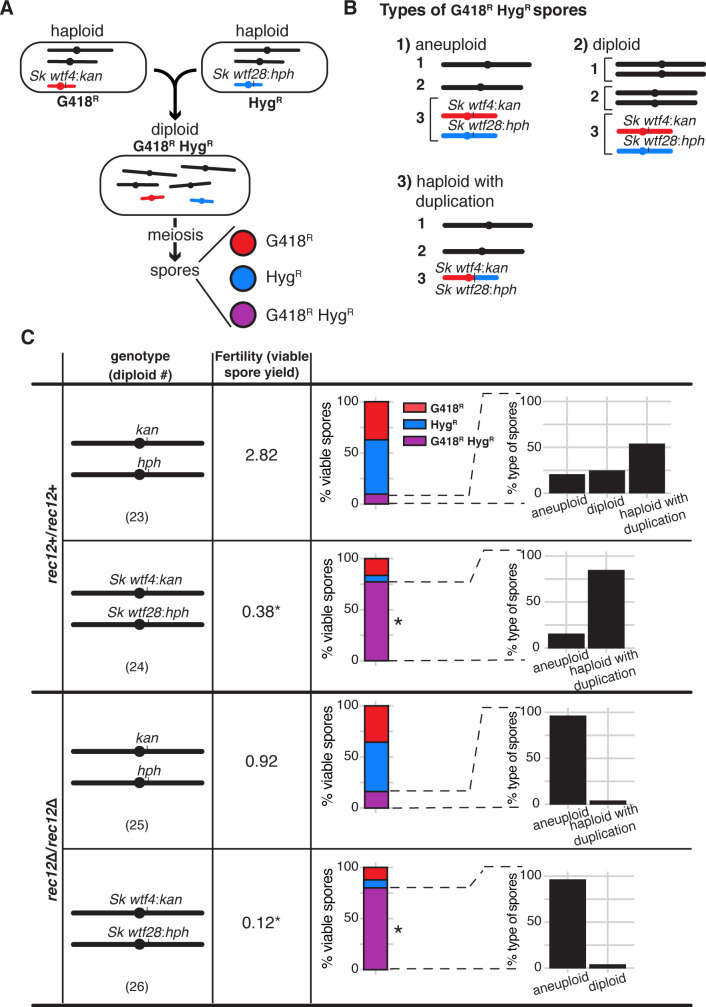
Figure 4. wtf competition at a single locus in meiosis selects for spores that are aneuploid, diploid, or contain a wtf duplication.
(A) Schematic of Sp strains crossed to make an Sk wtf4:kanMX4/Sk wtf28:hphMX6 heterozygote (diploid 24) and the possible spore phenotypes produced by that diploid. A similar control diploid (diploid 23) was created with empty vectors linked to drug resistant markers at the same locus. (B) Types of G418R HygR spores. We distinguished these classes using a series of phenotypic and molecular tests (see Materials and methods, Figure 4—figure supplement 3, Figure 4—figure supplement 4, and Figure 4—figure supplement 5). (C) Viable spores observed in control crosses (vector/vector) or with competing wtf meiotic drivers at the ade6 locus in a rec12+ (top) or rec12Δ (bottom) background. Percentages of G418R HygR (aneuploid, diploid, haploid with a duplication event) spores are shown. For statistical analyses, we compared diploid 24 to control diploid 23, and diploid 26 to control diploid 25. * indicates p-value<0.05 (G-test [G418R HygR] and Wilcoxon test [fertility]). Raw data can be found in Figure 4—figure supplement 5, Figure 4—source data 1, and Figure 4—source data 2. The data for diploid 23 [excluding inset analyses of G418R HygR spores] were previously published in Bravo Núñez et al., 2020.
Figure 4—figure supplement 1. Most spores are destroyed by wtf driver competition.
Figure 4—figure supplement 2. Competing wtf meiotic drivers enrich for spores that contain both wtf alleles in Sk.
Figure 4—figure supplement 3. Methods to distinguish between haploid, diploid, and aneuploid spore colonies.
Figure 4—figure supplement 4. Unequal crossover event at ade6 leads to wtf duplication.
Figure 4—figure supplement 5. Evidence of unequal crossover events between transgenes inserted at ade6.
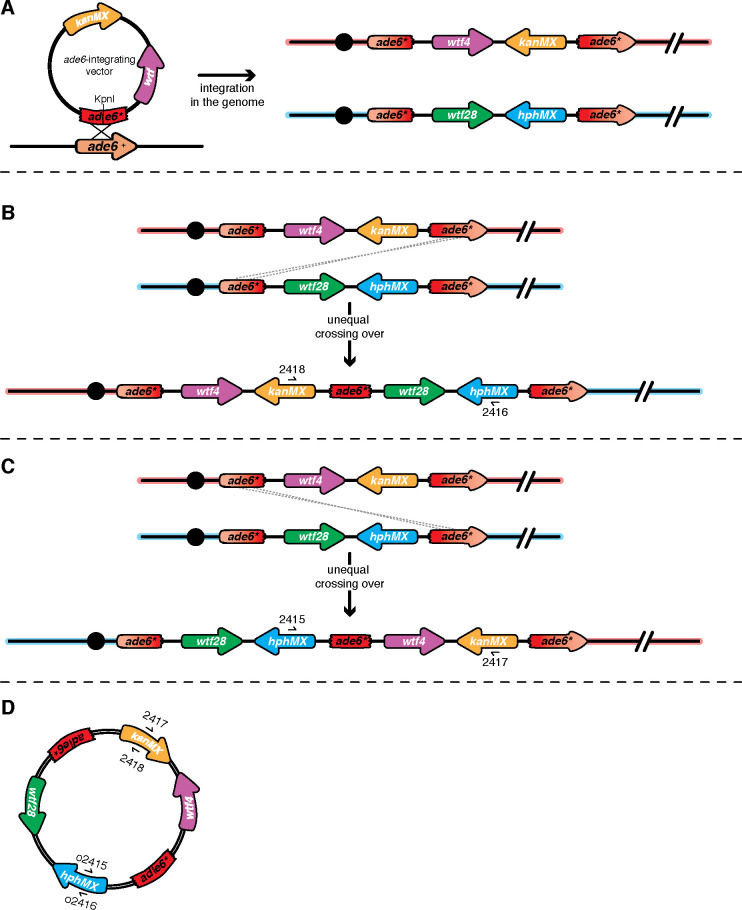
Although the spores produced by diploids with one set of competing meiotic drivers (Sk wtf4/Sk wtf28) often inherited both wtf driver-linked drug resistance alleles, they generally did not exhibit a colony morphology typical of aneuploids, as we observed in our other experiments (Figure 1C, Figure 1—figure supplement 2). We, therefore, investigated the ploidy of the spores of this cross more thoroughly using colony morphology, sporulation phenotypes, phloxin B staining, and a genetic marker loss assay (see Materials and methods, Figure 4—figure supplement 3). We were surprised to discover that amongst the G418R HygR spores generated by the Sk wtf4/Sk wtf28 heterozygote, only 15.4% appeared to be aneuploid and none appeared to be diploid. Instead, the majority (84.6%) of the G418R HygR spores appeared to be haploid (Figure 4C, diploid 24). We reasoned that an unequal interhomolog crossover event at the ade6 locus could have led to duplication of the wtf driver found on the opposite haplotype (Figure 4—figure supplement 4). Such a crossover would allow a haploid spore to inherit both wtf drivers and thus be protected against the toxicity of Wtf poisons. In our previous experiments, a single crossover could not put all the drivers onto one haplotype (Figure 2 and Figure 3).
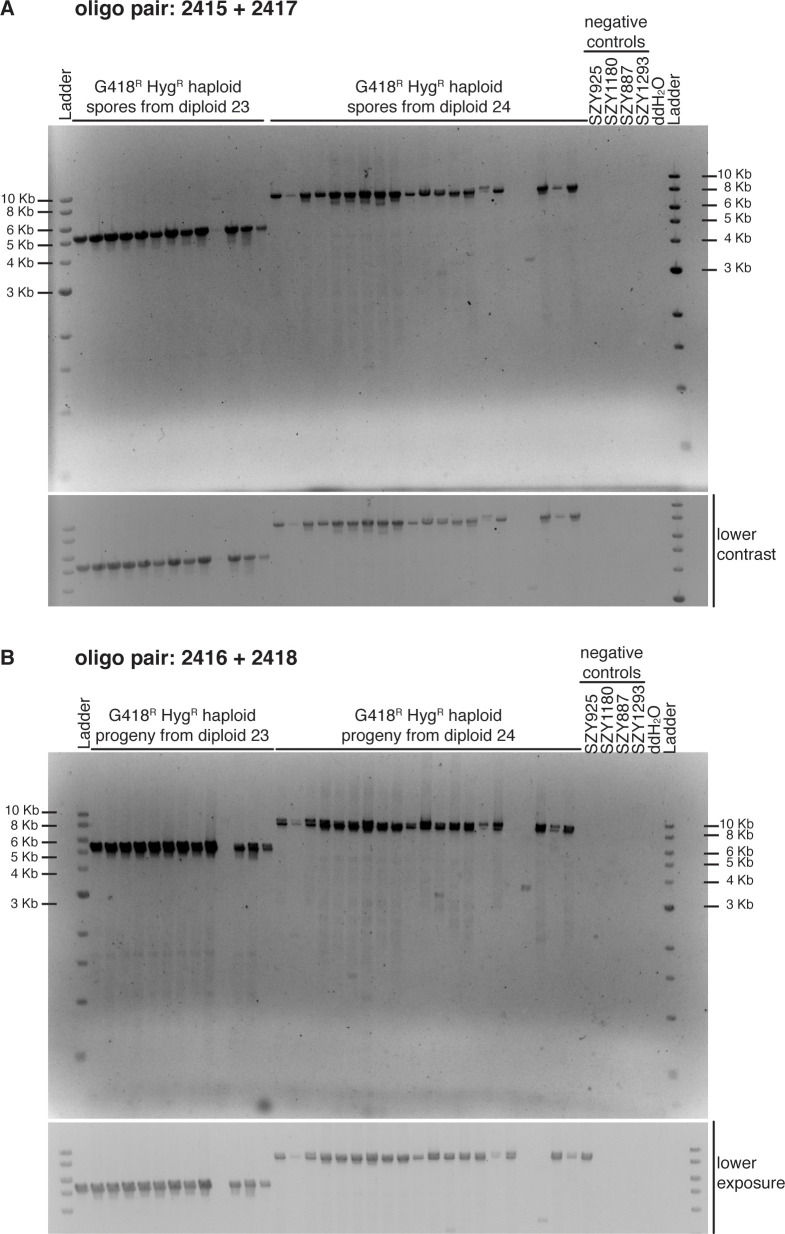
Consistent with the idea that a crossover placed the competing wtf drivers on one haplotype in the Sk wtf4/Sk wtf28 diploid meiosis, the events were Rec12-dependent (Figure 4, diploid 26). In addition, we detected a PCR product consistent with the recombination event in 19 out of the 22 haploid G418R HygR spore colonies tested (Figure 4, diploid 24, Figure 4—figure supplement 4, and Figure 4—figure supplement 5). We detected similar unequal crossover products amongst the G418R HygR haploid spores generated by the control diploid as well, but at much lower frequencies (Figure 4C, diploid 23 and Figure 4—figure supplement 5). Therefore, we concluded that this type of atypical meiotic product (duplication due to an unequal crossover) was enriched amongst the spores of the Sk wtf4/Sk wtf28 heterozygote due to the death of spores that did not inherit both wtf drive genes.
Fitness costs of meiotic mutants can be mitigated or eliminated in diploids with competing wtf drivers
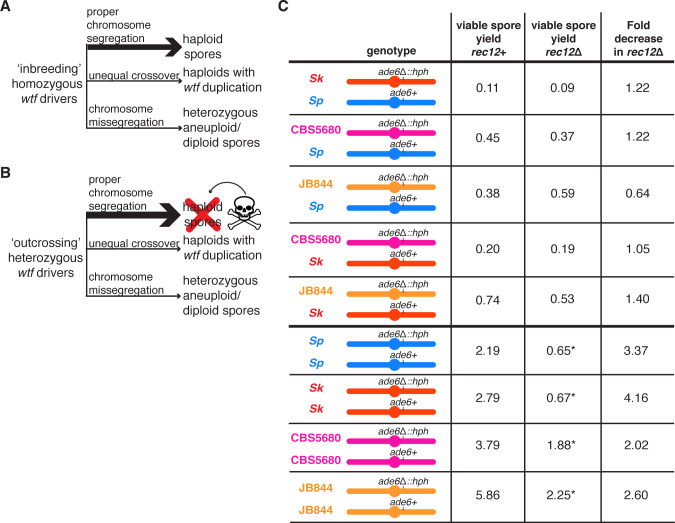
Our results demonstrate that when distinct wtf drivers compete on different haplotypes, such as when S. pombe outcrosses, the atypical spores that inherit more drivers are more fit. Disomic spores that inherit two copies of chromosome 3 most likely inherit the maximal number of wtf drivers, as most wtf drivers are found on that chromosome. Therefore, we hypothesized that the fitness costs of decreasing the fidelity of meiotic chromosome segregation might be offset by the fitness benefits of generating more disomic spores when wtf drivers compete (Figure 5A and B). Consistent with this idea, we previously observed that deleting rec12 imposed no fitness cost on Sp/Sk heterozygotes compared to the Sp/Sp or Sk/Sk homozygotes (Zanders et al., 2014; Figure 5C). We wanted to know if this was specific to Sp/Sk heterozygous diploids or if it might apply more generally to outcrossed S. pombe strains. To address this, we compared fertility in the presence and absence of Rec12 in CBS5680/Sp, JB844/Sp, CBS5680/Sk, and JB844/Sk heterozygotes. We observed that Rec12 did not significantly promote fertility in heterozygotes as it did in homozygotes (Figure 5C). These results demonstrate that the meiosis fitness optimum in inbred strains differs from what is optimal when strains outcross.
Figure 5. The Rec12 protein does not promote fertility in heterozygous S. pombe diploids.
(A) Schematic of viable spores resulting from an ‘inbreeding’ scenario. The three indicated types of spores are viable when wtf drivers are homozygous. (B) Schematic of viable spores resulting from an ‘outcrossing’ scenario where one set of wtf drivers is heterozygous. Haploid spores that only inherit one wtf allele will be killed by the poison of the wtf they did not inherit. Spores that inherit both wtf drivers due to a wtf duplication or disomy (aneuploidy or diploidy) will survive. Other outcomes of meiosis are not represented in this figure. (C) Viable spore yield values of heterozygous and homozygous S. pombe diploids in rec12+ and rec12Δ backgrounds. * indicates p-value<0.05 (Wilcoxon test) when comparing the rec12+ to rec12Δ fertility values. We compared the viable spore yield of each diploid in rec12+ and rec12Δ backgrounds. At least three, but usually more independent diploids were used to calculate viable spore yield. The data for rec12+ diploids is repeated from Figure 1. The data for Sk/Sk rec12Δ diploid (diploid 19) is repeated from Figure 3. The raw data are reported in Figure 5—source data 1.
We reasoned that competing wtf meiotic drivers were contributing to the dispensability of rec12 in the outcrossed diploids. To test that idea, we assayed the fitness costs of deleting rec12 in strains with heterozygous wtf drivers at one or two loci. We found that in a diploid with one set of heterozygous drivers (Sk wtf4/Sk wtf28 at ade6), the cost of deleting rec12 (rec12Δ/rec12Δ) was similar to that observed in the wild-type background (3-fold decrease in fertility) (Figure 6—figure supplement 1). However, we found that deleting rec12 in a genetic background with wtf drivers competing at both ade6 and ura4 had no cost (Figure 2C, compare diploid 15 to diploid 13). These results support our model that the costs of disrupting chromosome segregation can be offset by the fitness benefits of disomic spores in the presence of wtf driver competition.
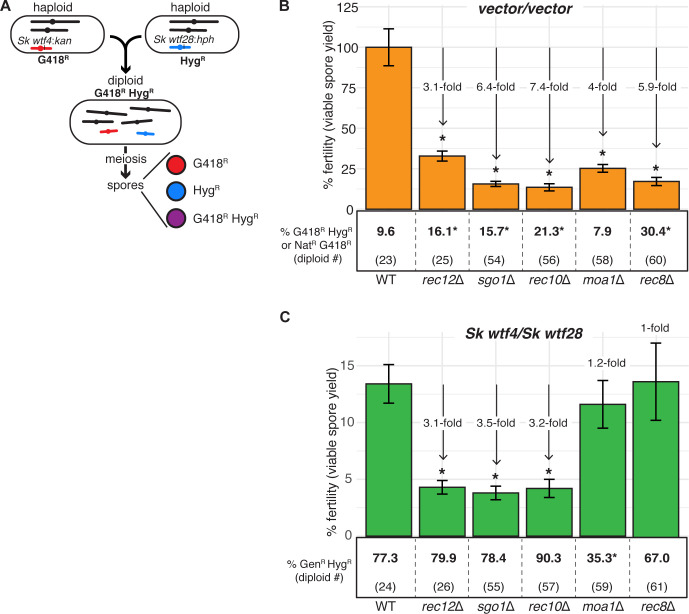
We next tested the fitness costs of deleting other genes that promote accurate meiotic chromosome segregation (rec10, sgo1, moa1, and rec8) in the presence and absence of competing wtf drivers (at ade6). Rec10 is a component of the meiotic chromosome axis (linear elements) that is required for the formation of most meiotic DSBs (Lorenz et al., 2004; Prieler et al., 2005). The Moa1 protein promotes monopolar attachment of sister kinetochores to ensure disjunction of homologs, rather than sister chromatids, in the first meiotic division (Yokobayashi and Watanabe, 2005). Sgo1 protects centromeric cohesion from cleavage during meiosis I and is required for proper segregation of sister chromatids during the second meiotic division (Kitajima et al., 2004). Finally, Rec8 is the meiotic kleisin that plays key roles in recombination and ensuring proper chromosome segregation in both meiotic divisions (Krawchuk et al., 1999; Watanabe and Nurse, 1999; Yoon et al., 2016).
Deleting sgo1 and rec10 had a lower fitness cost in the background with heterozygous wtf drivers than in the background without wtf competition (~3 fold decrease compared to a 6–7-fold decrease) (Figure 6—figure supplement 1 and Figure 6—figure supplement 2). Remarkably, deleting moa1 or rec8 had no effect on fitness in diploids with heterozygous wtf drivers, despite the fact that these mutations decrease fertility by 4- and 6-fold, respectively, in the absence of wtf driver competition (Figure 6—figure supplement 1 and Figure 6—figure supplement 2).
We reasoned that heterozygous moa1 (moa1Δ/moa1+) or rec8 (rec8Δ/rec8+) mutants might slightly increase meiotic chromosome missegregation and thus provide a selective advantage in the presence of wtf competition (Sk wtf4/Sk wtf28 at ade6) by producing disomic spores. Deleting one copy of moa1 did not significantly alter the frequency of disomic spores in the absence of wtf driver competition (Figure 6B, diploid 28). In addition, heterozygosity for moa1 did not suppress the fitness costs of wtf competition (Figure 6B, diploid 30). However, deleting one copy of rec8 significantly increased the production of disomic spores in a background without heterozygous wtf drivers (Figure 6B, diploid 27). This suggests that rec8 exhibits haploinsufficiency and reducing Rec8 protein levels may lead to chromosome segregation errors during meiosis. Consistent with this observation, mutations that reduce the levels of the meiotic cohesin can lead to meiotic defects in mice and flies (Murdoch et al., 2013; Subramanian and Bickel, 2008). As hypothesized, rec8 heterozygosity also increased the fertility of diploids with competing drivers (Figure 6B, diploid 29). Overall, these results suggest that the costs of disrupting chromosome segregation can be partially or totally alleviated by the increased protection against wtf drivers gained by generating more heterozygous disomic spores.
Figure 6. A heterozygous rec8 mutation increases fitness when wtf meiotic drivers are in competition.
(A) Schematic of diploid heterozygous for the HygR and G418R markers at the ade6 locus. (B) Fertility was measured using the viable spore yield assay in diploids with markers linked to competing wtf drivers (Sk wtf4/Sk wtf28) or empty vectors (vector/vector). Error bars represent the standard error of the mean. Underneath each bar graph is the % of G418R HygR (aneuploid, diploid, or haploid with duplication event) spores for each diploid. * indicates p-value<0.05 (G-test [G418R HygR] and Wilcoxon test [fertility]). For statistical analyses, diploids 27 and 28 were compared to diploid 23, and diploids 29 and 30 were compared to diploid 24. Data for diploids 23 and 24 are repeated from Figure 4. Raw data are found in Figure 6—source data 1 and Figure 6—source data 2.
Figure 6—figure supplement 1. Fitness costs of some meiotic mutants are reduced in diploids with competing wtf meiotic drivers relative to a background without wtf competition.
Figure 6—figure supplement 2. Pictorial description of mutant effects on fertility and the frequency of G418R HygR spores.
Driver competition can facilitate the maintenance or spread of alleles that disrupt meiotic chromosome segregation fidelity in a population
Our experiments demonstrate that the effects of meiotic mutants can be quite different in heterozygous S. pombe wherein wtf drivers are competing. To explore this idea further, we turned to population genetic modeling to analyze how drivers affect the evolution of variants that decrease the fidelity of meiotic chromosome segregation.
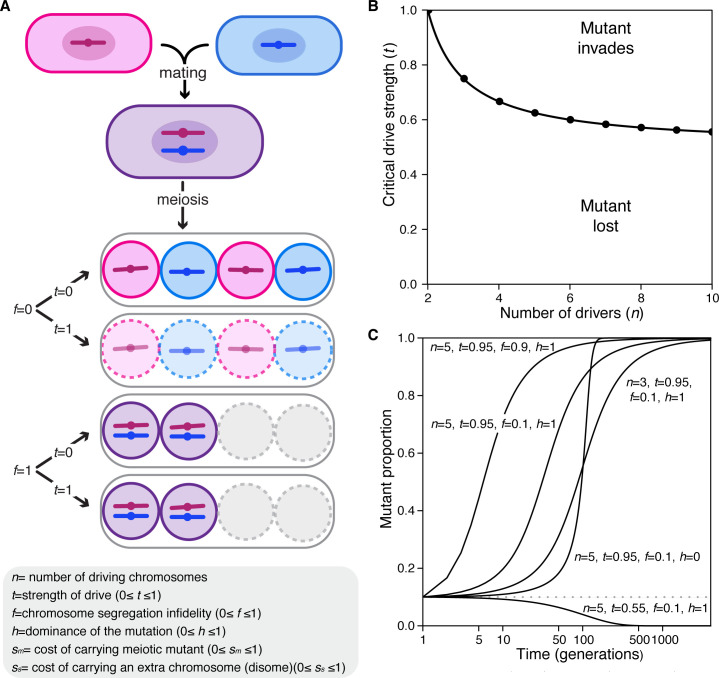
Our model analyzes the evolutionary fate of a hypothetical mutation that disrupts the segregation of chromosome 3, which houses the majority of wtf drivers. For the sake of simplicity, our model assumes that chromosome 3 exhibits whole-chromosome drive. The model also considers six parameters (Figure 7A). The first two parameters relate to the wtf drivers. We varied the number of driving alleles in the population (n) and the strength of their drive (t). Each driving allele was assumed to be at an equal frequency in the population and have the same strength of drive. The next parameters relate to the meiotic mutation. We varied the level of chromosome missegregation caused by the mutation (f) from 0 (no mutant phenotype) to 1 (50% of the resultant spores are heterozygous disomes and the remaining 50% of the spores lack chromosome 3 and are thus inviable). We considered the dominance of the mutation (h) and any additional fitness costs (sm) the mutation may incur, such as potential costs relating to the missegregation of other chromosomes. Finally, we considered additional fitness costs (ss) disomic spores might bear. The full description of the model and additional analyses are presented in Appendix 1.
Figure 7. Population genetics of a meiotic mutant in response to meiotic drive.
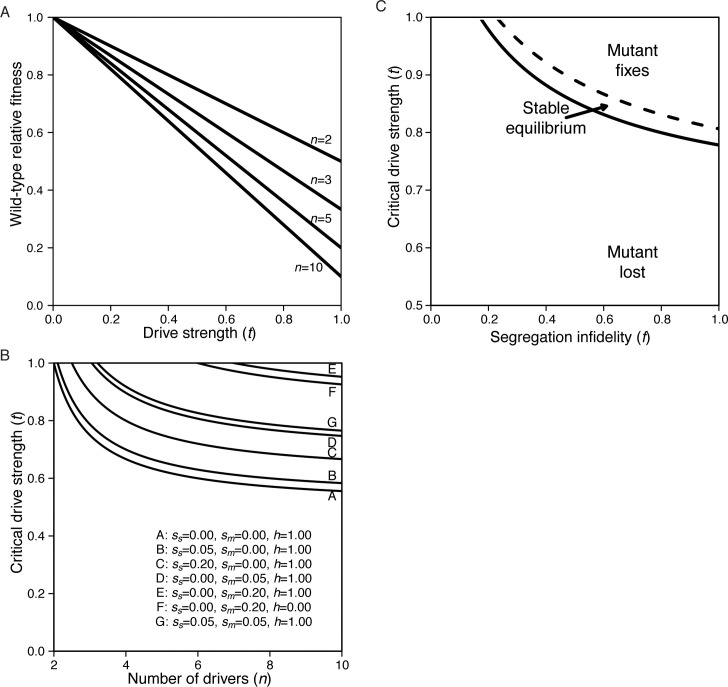
(A) Schematic of the spore progeny generated by S. pombe diploids when the infidelity of chromosome segregation (f) is 0 or 1 and the strength of drive (t) is 0 or 1. (B) Critical drive strength (tcritical) for invasion of the segregation infidelity mutant given n drivers. This assumes that there is no cost of the meiotic mutant. (C) Trajectories of meiotic mutants starting at a frequency of 0.1. Note that the X-axis is a log scale.
We found that a mutation with no additional fitness costs (sm and ss = 0) that disrupts meiotic segregation could invade a population when:
where tcritical is the value of drive strength necessary for such invasion in a population of n drivers. Interestingly, the higher the strength of drive (t), the lower the number of drivers required for mutant invasion (Figure 7B). Importantly, our empirical work demonstrates that drive strength is generally high (t >0.9) and that there are ample distinct wtf driver haplotypes (Eickbush et al., 2019) leaving parameter space for mutants to invade even if they incur considerable costs (sm and ss) (Appendix 1—figure 1; Bravo Núñez et al., 2018; Bravo Núñez et al., 2020; Eickbush et al., 2019; Hu et al., 2017; Nuckolls et al., 2017). With fitness costs applied to the equation above, increasing the number of drivers in the population and decreasing the value of these associated costs both increase the likelihood that the mutation can invade the population (Appendix 1, Appendix 1—figure 1).
To get a broader perspective on the potential evolutionary trajectories of the segregation fidelity mutant, we varied the described parameters and plotted the results. These analyses all started with the mutant at a frequency of 0.1 in the population. We found that changes in some parameters dramatically influenced the trajectories. For example, with all other parameters fixed, at t = 0.55 the mutant is lost, while at t = 0.95, the mutant is fixed in the population (Figure 7C). With the parameters plotted, the dominance of the mutation, h, has little influence on the fate of the mutation but does influence the rate at which that fate is reached. Finally, in our model where the costs are applied, the cost of the mutation, the cost bore by disomic spores, and the degree of segregation infidelity also influence whether or not invasion occurs (Appendix 1—figure 1).
Overall, the results of the model are consistent with our experimental results. Both types of analyses support the idea that meiotic drivers can change the selective landscape of meiosis in outcrossed S. pombe. Instead of meiotic mutants being removed by negative selection due to fitness costs, variants that decrease the fidelity of chromosome segregation can be advantageous due to pervasive meiotic drive.
Discussion
Meiotic drivers shape the evolution of S. pombe meiosis
Most genetic analyses of meiosis are performed in inbred (homozygous) organisms. This approach has been incredibly powerful. It enabled Mendel to establish the founding principles of genetics with his true-breeding peas and facilitated countless discoveries over the last 150 years (Abbott and Fairbanks, 2016). However, studying inbred model systems with very low genetic variability has limitations in that phenotypes that can only be observed in heterozygotes remain hidden. These phenotypes can include those caused by selfish genetic elements, like meiotic drivers, that act primarily in heterozygous or outcrossed organisms. This work highlights the stark differences in fitness optima that can exist between gametogenesis in inbred homozygotes and in heterozygotes generated by outcrossing.
When S. pombe inbreeds to generate homozygotes, the effects of wtf meiotic drivers are largely invisible as every spore inherits the necessary Wtf antidotes required to neutralize the Wtf poisons. Granted these homozygotes faithfully segregate their chromosomes during meiosis and generate four haploid spores, the fitness of these diploids will be relatively high. However, mutations such as rec12∆, rec8∆, or moa1∆ will decrease the relative fitness of such diploids, as these mutations decrease the fidelity of chromosome segregation and spore viability (Davis and Smith, 2003; Martín-Castellanos et al., 2005; Yokobayashi and Watanabe, 2005).
When S. pombe outcrosses, the observed outcomes of meiosis are different from those observed under inbreeding, at least in part due to wtf meiotic drivers. These differences include changes in fertility, spore ploidy, recombination frequencies, and the fitness costs (or benefits) of inaccurate meiotic chromosome segregation. It is important to stress that the known effects wtf drivers exert on meiosis appear to be entirely indirect. There is no evidence that Wtf proteins directly participate in or affect the molecular mechanisms of recombination or chromosome segregation. Rather, the effects of the Wtf proteins (i.e. spore killing) are observed after the completion of meiosis. Despite this, the Wtf proteins have the power to indirectly affect the molecular steps of meiosis as changes in the fidelity of chromosome segregation or other aspects of gametogenesis can alter the number of spores destroyed by drive.
The S. pombe isolates in which wtf genes have been assembled carry between 4–14 intact wtf drive genes (Eickbush et al., 2019; Hu et al., 2017). The wtf genes are amongst the most rapidly evolving genes in S. pombe, which means outcrossing often generates extensive wtf driver heterozygosity. This leads to extensive death of haploid spores in outcrossed strains because 1) Wtfantidote proteins appear to neutralize only the Wtfpoison proteins with highly similar or identical C-termini and 2) it is unlikely that haploid spores will inherit all drivers and thus encode all Wtfantidote proteins (Bravo Núñez et al., 2018; Bravo Núñez et al., 2020). Hence, generating the maximal number of haploid spores does not maximize the fitness of outcrossed S. pombe.
Instead, fitness is maximized in outcrossed S. pombe when the spores inherit as many drivers as possible. If there is just one locus with heterozygous drivers, perhaps due to mating between two closely-related isolates, an unequal crossover event can place both drivers on the same haplotype and allow a haploid spore to survive. It is possible this type of selection may be occurring in nature, as all assayed strains contain multiple loci with 2–3 wtf genes in tandem that could have been produced by unequal crossover events (Eickbush et al., 2019).
When two diverse isolates mate to form heterozygotes, the high number of wtf drivers present in each respective strain will make it unlikely for a haploid spore to inherit every driver. In this scenario, disomic spores that inherit two different copies of chromosome 3, which carries nearly every wtf gene, are most likely to inherit every driver and survive. Importantly, an extra copy of chromosome 3 is the only aneuploidy tolerated in S. pombe (Niwa et al., 2006).
We have previously speculated that the wtf gene family specifically expanded on chromosome 3 and not on chromosomes 1 or 2 as aneuploid spores provide an avenue to mitigate the fitness costs of multiple drivers (López Hernández and Zanders, 2018). The results of this study support and expand on that model. Specifically, we now show that drivers can create a selective landscape wherein variants that decrease the fidelity of chromosome segregation and thus generate more disomic spores can be favorable. In these cases, the fitness costs of mutating genes like rec12, moa1, or rec8 can be offset by the fitness benefits of increased disomy. Our work also adds to previous work demonstrating meiotic drive-independent adaptive potential of aneuploid spores in other fungi (Chuang et al., 2015; Ni et al., 2013).
It is not clear how often S. pombe outcrosses in the wild, and population genetics estimates are confounded by drive and repressed recombination in hybrids (Farlow et al., 2015; Fawcett et al., 2014; Jeffares et al., 2015; Tusso et al., 2019; Zanders et al., 2014). Many S. pombe isolates can switch mating type during clonal growth and thus mate with nearby clonal cells when starved for nutrients (Egel, 1977). This undoubtedly leads to frequent inbreeding in S. pombe and could thus promote selection against mutations that increase the frequency of disomic spores. However, when outcrossing occurs, mutations that increase disomy can have a selective advantage. A mix of inbreeding/outcrossing strategies could lead to the maintenance of variation in the frequency at which meiosis generates disomic spores. Consistent with this, we observed such variation amongst the natural isolates assayed in this study (Figure 1C, diploids 8–12 and Figure 1—figure supplement 3). Strikingly, the strains with the ‘highest’ meiotic fidelity still make ~5% disomic spores, suggesting that chromosome 3 missegregates during the first meiotic division in one out of ten meioses.
Given the results of this study, it is difficult to resist speculating about the potential evolutionary relationships between the frequent disomy of S. pombe spores and the wtf drivers. It is possible that frequent disomy of S. pombe spores was selected to mitigate the cost of wtf drivers. An alternative model is that frequent disomy perhaps predates and/or contributes to the success of the wtf drivers. In this model, the disomy could be beneficial to the wtf genes by preventing them from being too costly to their host and thereby destroying themselves by destroying the host. Unfortunately, at this time we know far too little about fission yeast ecology and evolution to rigourously explore these possibilities (Jeffares, 2018). More knowledge about population structures, outcrossing frequencies, meiotic driver prevalence, and disomy frequencies in other fission yeast species is required to distinguish between the proposed models.
The effects of drive on the evolution of gametogenesis outside of S. pombe
A growing body of evidence indicates that meiotic drive is pervasive in eukaryotes, and more drivers are identified each year. This includes the gamete-killing type of meiotic drivers described in this work (Bauer et al., 2012; Bravo Núñez et al., 2018; Bravo Núñez et al., 2020; Burt and Trivers, 2006; Didion et al., 2015; Grognet et al., 2014; Hammond et al., 2012; Hu et al., 2017; Larracuente and Presgraves, 2012; Long et al., 2008; Nuckolls et al., 2017; Pieper et al., 2018; Rhoades et al., 2019; Vogan et al., 2019; Xie et al., 2019; Yang et al., 2012; Yu et al., 2018), but also extends to other drivers that use completely different methods to gain a transmission advantage. For example, biased gene conversion favoring unbroken DNA during meiotic recombination is a form of meiotic drive tied to the mechanisms of DSB repair (Marais, 2003). This type of drive shapes recombination landscapes and likely promotes the rapid evolution of at least one key recombination protein found in many mammals, including humans (Grey et al., 2018; Úbeda et al., 2019). Other meiotic drivers exploit the asymmetry of female meiosis to promote their transmission into the one viable meiotic product (i.e. the oocyte) (Akera et al., 2017; Akera et al., 2019; Dawe et al., 2018; Kato Yamakake, 1976; Rhoades, 1942). This type of bias has been hypothesized to drive the widespread rapid evolution of karyotypes, centromere sequences, and centromeric proteins (Pardo-Manuel de Villena and Sapienza, 2001; Henikoff et al., 2001; Rosin and Mellone, 2017). In addition, drive during female meiosis in mice can even generate selective pressure to alter the timing of the first meiotic division (Akera et al., 2017; Akera et al., 2019).
It may be tempting to disregard the wtf genes within S. pombe as an anomaly. However, meiotic drivers are ubiquitous, and drive represents an incredibly powerful evolutionary force (Burt and Trivers, 2006; Sandler and Novitski, 1957). Appreciating how wtf genes affect S. pombe will likely provide important insights into how genetic parasites can shape the evolution of meiosis in other eukaryotes.
Materials and methods
Strain construction: S. pombe natural isolates
All yeast strain names and genotypes are described in Supplementary file 1. We made the lys1Δ::kanMX4 and ade6Δ::hphMX6 alleles used in Figure 1 as described in Zanders et al., 2014. Using the standard lithium acetate protocol, we independently transformed the cassettes into seven different S. pombe natural isolates (JB844, JB1172, CBS5680, JB873, JB939, JB929, and NBRC0365). However, we were only successful at transforming both markers into JB844, JB1172, CBS5680, and NBRC0365. We could not find conditions in which to mate and sporulate NBRC0365.
To generate a rec12Δ::ura4+ deletion in the CBS5680 strain background, we first made a ura4-D18 mutation in SZY2111 (ade6Δ::hphMX6 in CBS5680). We amplified the ura4-D18 allele from SZY925 using oligos 35 and 38 and transformed it into SZY2111 to generate SZY3949. We then amplified the rec12Δ::ura4+ cassette from SZY580 using oligos 1194 and 1077 and transformed the cassette into SZY3949 to generate SZY3995. We confirmed the rec12 deletion via PCR using oligos (1120+1108) that bind 730 bases upstream and 224 bases downstream of the deletion cassette. We generated the rec12Δ::ura4+ deletion in the lys1Δ::kanMX4 background of the CBS5680 isolate via crosses. We generated the rec12 deletion in JB844, similarly to how we generated it in the CBS5680 strain.
We found it difficult to make gene deletions in many of the natural isolates used in this study. We had more success, however, making mutations using integrating vectors. Because of this, we used integrating vectors to generate the genetic markers used in Figure 1—figure supplement 3. We used pSZB386 to generate haploid strains with a hphMX6 marker at ade6, without deleting the ade6 gene. We digested this plasmid with KpnI and transformed it into different natural isolates, selecting for transformants that were resistant to Hygromycin B and appeared red on media with low adenine (Bravo Núñez et al., 2018). To generate strains with a kanMX4 marker at lys1, we first ordered a gBlock from IDT (Coralville, IA). This gBlock contained ~1000 bp from the middle of the lys1 gene in which we replaced 50 bp from the center with a KpnI site. We then cloned the gBlock into the BamHI and SalI sites of pFA6 to generate pSZB816. We then digested pSZB816 with KpnI and transformed it into different S. pombe isolates. Finally, we screened for transformants that grew on plates containing G418 and were not able to grow on media lacking lysine.
Plasmid construction: integrating vectors with wtf alleles
Most of the integrating vectors containing wtf alleles were previously described in Bravo Núñez et al., 2018; Bravo Núñez et al., 2020; and Nuckolls et al., 2017. To generate the additional ade6- and ura4-integrating vectors unique to this work, we cloned the wtf genes of interest into the integrating plasmid backbones and confirmed them via sequencing. The DNA templates, oligos, and restriction enzymes used are described in Supplementary file 3.
To generate pSZB923 (Sk wtf4:natMX4), we first digested pSZB189 (which contains Sk wtf4) with SacI to release the Sk wtf4 cassette. We then cloned the cassette into the SacI site of pSZB849.
To make the Sk wtf28 poison-only allele, we mutated the two start sites found in exon one to stop codons (ATG→TAC). Using pSZB254 as a template, we amplified the 5’ fragment (with oligos 651+954) and the 3’ fragment (with oligos 953+733) of Sk wtf28. We used overlap PCR to stitch the fragments together and digested them with SacI. We then cloned this fragment into the SacI site of pSZB386 to generate pSZB414.
Deletions of the moa1, rec10, and sgo1 genes in Sp
We made moa1, rec10, and sgo1 gene deletions using standard deletion cassettes and transformation. To make the moa1Δ::natMX4 cassette, we amplified the upstream region of moa1 with oligos 1673+1187 and the downstream region with oligos 1190+1191 (or 1190+1674) using SZY643 as a template. We also amplified the natMX4 gene (with oligos 1675+1189) using pAG25 as a template (Goldstein and McCusker, 1999). Next, we stitched all the PCR fragments together using overlap PCR and transformed this fragment into SZY44 and SZY643 to make strains SZY2479 and SZY2481, respectively. We confirmed the integration of the deletion cassette at the moa1 locus using oligos AO638+1192, AO1112+1191, and 1701+1702. We also checked that the moa1 gene was not present somewhere else in the genome by using two oligos (1703+1704) within moa1.
To generate a rec10Δ::natMX4 strain, we first amplified the upstream region and the downstream region of rec10 from SZY643 using oligos 1723+1724 and oligos 1727+1728, respectively. We also amplified the natMX4 cassette from pAG25 using oligos 1725+1726 (Goldstein and McCusker, 1999). Using overlap PCR, we stitched the three PCR fragments together and then transformed the final deletion cassette into SZY643 and SZY44 to make strains SZY2517 and SZY2519, respectively. To confirm the integration of the cassette at the correct locus, we used oligo pairs 1731+AO638 and 1732+AO1112. We also confirmed the absence of the wild-type rec10 gene by using internal oligos (1729+1730).
To make the sgo1∆::hphMX6 allele, we amplified the sequences upstream and downstream of sgo1 from SZY643 using oligos 1224+1225 and 1228+1229. We also amplified the hphMX6 cassette from pAG32 using oligos 1226+1227 (Goldstein and McCusker, 1999). We then stitched all the PCR fragments together using overlap PCR and transformed the cassette into yeast to generate strains SZY1735 and SZY1736. We confirmed the sgo1 deletion using oligos AO638+1230, AO1112+1231, and 1230+1231. We then confirmed the absence of the wild-type gene using an internal oligo pair (2088+2089).
Sp wtf4 deletion
To delete Sp wtf4, we utilized the CRISPR/Cas9-based method described in Rodriguez-Lopez et al., 2016. We first cloned a plasmid (pSZB570) encoding Cas9 and a guide RNA targeting Sp wtf4. To do that we first amplified pMZ379 (plasmid containing Cas9) using oligos 1206+1207. These oligos contained the single guide RNA (sgRNA) sequence that targets the Sp wtf4 gene. We then ligated the ends together to generate pSZB570.
We also made a deletion cassette to knockout the Sp wtf3 and Sp wtf4 locus. We used oligos 574+1138 and 1139+471 to amplify the upstream and downstream sequence of the locus using SZY580 as a template. We stitched these two fragments together using overlap PCR. Next, we transformed the deletion fragment and pSZB570 into SZY1595 to generate SZY1699. We used oligos 1069+543 to confirm the Sp wtf4 deletion. We also Sanger sequenced the PCR fragment and found that we had only knocked out the Sp wtf4 gene, not the entire Sp wtf3 and Sp wtf4 locus.
Sp wtf13 deletions
The Sp wtf13 deletions were made similar to the ones described in Bravo Núñez et al., 2018. Using SZY580 as a template, we amplified the upstream (with oligos 1048+1049) and downstream (with oligos 1052+1053) sequence of Sp wtf13. Additionally, we amplified the kanMX4 cassette using oligos 1050+1051, with pFA6 as a template (Wach et al., 1994). We then stitched the upstream region, kanMX4, and the downstream region together using overlap PCR to make an Sp wtf13Δ::kanMX4 cassette. After, we transformed this fragment into SZY580 to generate SZY1391-SZY1394.
To generate the Sp wtf13Δ::kanMX4 Sp wtf4Δ strain, we first digested pAG25 with EcoRV and BamHI to release the natMX4 cassette (Goldstein and McCusker, 1999). We then used this cassette to switch the kanMX4 marker (at the his5 gene) from the SZY1699 strain via transformation to generate SZY1981 and SZY1982. We transformed the Sp wtf13Δ::kanMX4 deletion cassette into SZY1981 and SZY1982 to generate strains SZY2008 and SZY2010, respectively. We confirmed these deletions using a series of PCR reactions. We used two oligo pairs with one oligo outside of the deletion cassette and one oligo internal to the Sp wtf13 gene (1058+1059 and 1060+1061) and two oligo pairs in which one oligo was external to and one oligo was within the deletion cassette (1058+AO638 and 1061+AO112).
Fertility and allele transmission
We assayed fertility and allele transmission as described in Bravo Núñez et al., 2020.
Some of the spore colonies from ade6Δ::hphMX6/ade6+; lys1+/lys1Δ::kanMX4 CBS5680 diploids (SZY2213/SZY2111) were small and red. When we determined their genotype, the colonies were adenine auxotrophs and took five days to grow when replicated to fresh media. We supplemented the plates with more adenine, but the colonies did not grow faster. This slow growth phenotype was curiously not observed in the Ade- parental haploid (SZY2111).
Recombination frequency within the ade6 and ura4 interval
To determine the recombination frequency for diploids 20 and 21, we needed to distinguish the ura4 allele (ura4-294 or ura4Δ::kanMX4) via PCR. We amplified the ura4 locus using two sets of oligos (34+37 and 34+AO638).
For Figure 2—figure supplement 5 and Figure 3—figure supplement 2, we performed power analyses to estimate the total number of spores we needed to assay in order to determine an effect in recombination frequencies (alpha = 0.05 and power = 80%).
Determining ploidy of spore colonies
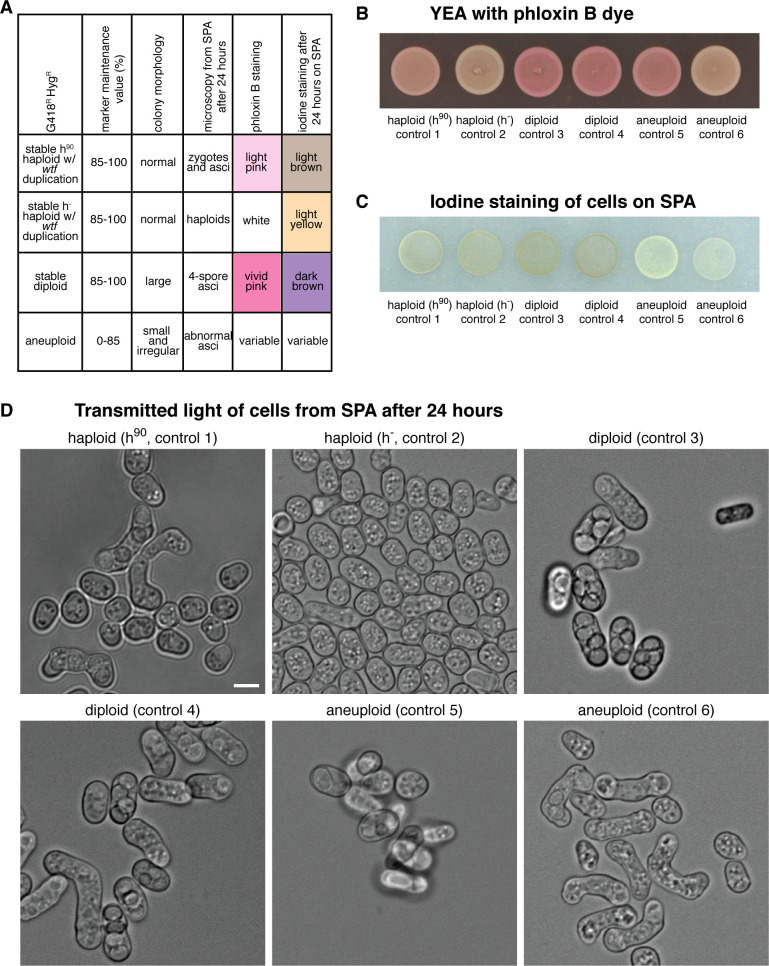
In the various tests to assay the ploidy of the spore colonies for Figure 4, we compared the spore colony phenotypes to the following control strains: a homothallic haploid (SZY925), a heterothallic haploid (SZY1180), a diploid (SZY925/SZY1180), and aneuploid (irregular colonies generated by a cross between SZY1994 and SZY1770) controls. The ploidy of the strains was determined by how closely a test strain resembled one of the controls in the following tests:
Spore colony morphology
To determine the morphology of the spore colonies from different diploids, we diluted the spores to get isolated colonies on YEA+S (0.5% yeast extract, 3% dextrose, agar, and 250 mg/L adenine, histidine, uracil, leucine, and lysine). We then imaged the colonies using the Canon EOS Rebel T3i plate imager. We marked each cell in ImageJ and determined the genotype of each spore colony via replica plating. These images allowed us to correlate the morphology of each spore colony with its genotype. For the spores that had resistance to both G418 and Hygromycin B, we assessed if the colonies were either large, medium, or small, and if the morphology was either round or irregular. Round colonies are typically haploid or diploid. Small and irregular-shaped colonies are characteristic of ‘sick’ colonies or aneuploids (Niwa et al., 2006).
Chromosome loss assay
To determine the ploidy of the spore colonies, we determined the frequency at which each of the G418R HygR strains lost one of the drug markers during vegetative growth. Aneuploids frequently lose their extra chromosome during vegetative growth and it is random which chromosome is lost (Niwa et al., 2006). Diploids and haploids are expected to be more stable. We began by culturing ~26 G418R HygR spore colonies produced by each experimental diploid (diploids 23–26), along with six haploid controls (deemed haploid due to the presence of only one drug marker) in 5 mL of YEL (0.5% yeast extract, 3% dextrose, and 250 mg/L adenine, histidine, uracil, leucine, and lysine) with shaking at 32°C for 24 hr. The next day (day 1), we diluted the cultures and plated the cells on YEA to assay the presence or absence of the drug resistance markers. We also made 1:10 dilutions into 650 μL of YEL in 96-well plates and grew them for 24 hr. We repeated this for five days. Aneuploids readily and randomly lost one marker around day 1 or 2, so that a high fraction (17–100%) of the colonies generated by plating the culture were no longer resistant to both drugs. Haploids and diploids, however, maintained both markers for all 5 days because almost all (~90%) of the colonies generated by plating the culture were still resistant to both drugs. The marker maintenance value was calculated by dividing the number of G418R HygR colonies by the total number of colonies that grew on YEA+S.
Phloxin B staining
Phloxin B was used to differentiate between diploid strains and haploids. Phloxin B is a dye that enters cells with compromised membranes, staining them red (Forsburg and Rhind, 2006). Diploids have a higher concentration of dead cells within a colony than haploids due to their lower stability resulting in a red stain. Haploids are much more stable, which reduces the ability of phloxin B to enter cells, leading haploids to appear white. Haploid strains that are homothallic (h90) look pink (Figure 4—figure supplement 3). We spotted 10 μL of the saturated culture of day 1 (described above) onto YEA+S plates containing 5 mg/L phloxin B and grew the cells at 32°C overnight. We then determined if the spots were red, pink, or white by comparing them to the controls.
Microscopy and iodine staining from SPA plates
Using the cultures from day 1 (described above), we also spotted 10 μL of the saturated culture of each strain and controls onto two SPA plates. We then placed these plates at 25°C for 20 hr. From the first SPA plate, we imaged the cells on a Zeiss (Germany) Observer Z.1 widefield microscope with a 40X (1.2 NA) water-immersion objective and acquired the images using the μManager software. Twenty hours was enough time for diploids to sporulate but not enough for homothallic haploid strains to mate, form diploids, and sporulate, allowing for further distinction between these ploidies. Although this assay did not reliably allow us to distinguish aneuploid cells that could not sporulate (as they would resemble the heterothallic haploid control), we were able to score some homothallic ‘aneuploid’ strains due to the presence of asci with an abnormal shape or number of spores.
The second SPA plate was stained with iodine (Forsburg and Rhind, 2006) after 20 hr at 25°C. Iodine vapors stain the starch present in spore walls a dark brown color, while heterothallic haploid cells that cannot sporulate appear yellow. Diploids stain dark brown, while homothallic haploids that first needed to mate in order to sporulate stained light brown (Figure 4—figure supplement 3).
PCR assay for duplications
Some of the G418R HygR spores we tested appeared to be haploids based on the assays described above. We reasoned they could be the result of an unequal crossover putting both marker genes onto one chromosome. To test this, we used two sets of oligo pairs (2415+2417 and 2416+2418). These PCR reactions only work if the drug cassettes are found in tandem. To confirm that the presence of only one cassette would not lead to band amplification, we used the haploid parental strains (SZY925, SZY1180, SZY887, and SZY1293) as negative controls.
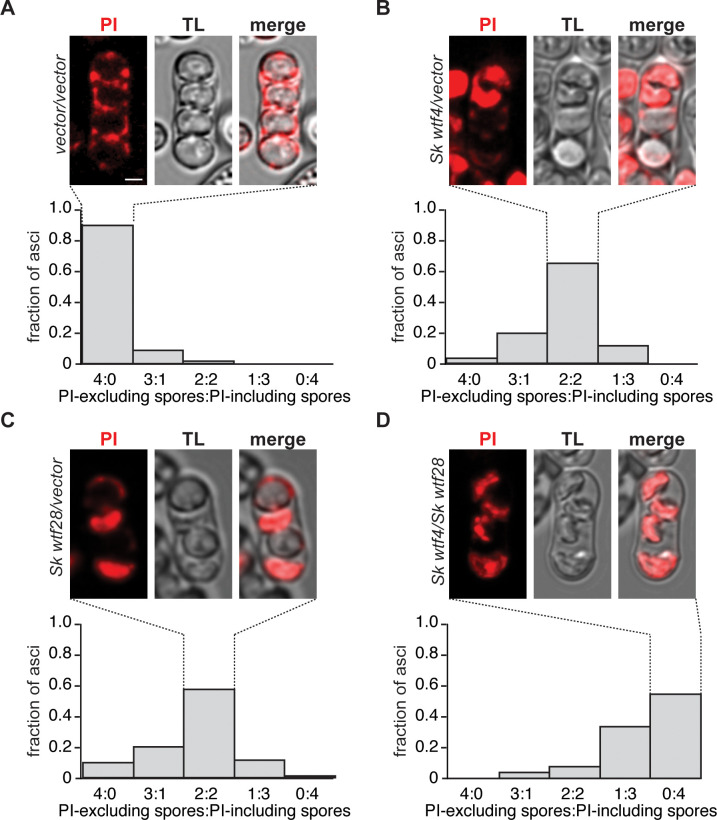
PI staining and confocal microscopy
To determine the number of spores in asci that lost membrane integrity, we stained the spore sacs using the propidium iodide (PI) dye (1 mg/mL) (Nuckolls et al., 2017). These spores were generated by SZY2625/SZY1180, SZY2628/SZY1180, SZY1293/SZY2625, and SZY2628/SZY1293 diploids. We scraped the cells off of the SPA plates and swirled them into 50 µL of ddH20 in an Eppendorf tube. We then incubated the sample at room temperature for 20 min. After the incubation, we mixed the cells with 50 µL lectin (1 mg/mL) and plated them onto a 35 mm glass culture dish (MatTek) pre-coated with lectin to immobilize the cells. We imaged the samples on an LSM-780 (Zeiss) AxioObserver microscope with a 40X C-Apochromat water-immersion objective (NA = 1.2) on photon-counting channel mode with 561 nm excitation. We acquired the PI fluorescence through a 562–642 nm filter. We assayed more than 50 asci for each genotype.
Acknowledgements
We thank Adèle Marston, Thomas Price, and members of the Zanders lab for their helpful comments that improved the manuscript. We are grateful to Gerry Smith for sharing the rec8 mutant strain, and to Jeff Lange and Valeria Eliosa for technical support. This work was performed to fulfill, in part, requirements for MABN’s thesis research in the Graduate School of the Stowers Institute. Original data underlying this manuscript can be accessed from the Stowers Original Data Repository at http://www.stowers.org/research/publications/libpb-1514. This work was supported by the following awards to SEZ: The Stowers Institute for Medical Research (https://www.stowers.org), the March of Dimes Foundation Basil O'Connor Starter Scholar Research Award No. 5-FY18-58 (https://www.marchofdimes.org), the Searle Scholar Award, and the National Institutes of Health (NIH) under the award numbers R00GM114436 and DP2GM132936 (https://www.nih.gov). MABN was also supported by the National Cancer Institute of the NIH under award number F99CA234523. RLU was supported by funding from the University of Kansas. The funders had no role in study design, data collection and analysis, or manuscript preparation.
Appendix 1
Population genetics model
It is counterintuitive that a mutation causing reduced fidelity of chromosome segregation during meiosis could ever be beneficial and spread through a population. To determine whether such a segregation infidelity mutation could invade a population with wtf-like meiotic drivers, we created a simple mathematical model of the scenario.
The model
As described in the main text, we start with a random mating population of infinite size with n distinct drivers – all at the same locus on the same homologous chromosome. Each driving chromosome has the same strength of drive (t) and is completely susceptible to all other driving chromosomes (e.g. the antidote of the 3rd driver does not protect against the poison of the 2nd driver). We assume that each drive chromosome is at equal frequency so the frequency of the ith driver is Pi = 1/n. This means that there are no chromosomes without a driver in the population – a reasonable assumption given observations in isolates of S. pombe (Bravo Núñez et al., 2018; Bravo Núñez et al., 2020; Eickbush et al., 2019; Hu et al., 2017; Nuckolls et al., 2017). With random mating, the proportion of the population that are homozygotes when it comes to the drive chromosome is and the proportion that are heterozygotes is . Since all chromosomes contain distinct wtf drive loci, all heterozygotes are likely to produce spores that will be killed by the Wtfpoison.
The strength of drive (t) ranges from zero (Wtfpoison does not kill any spores) to one (Wtfpoison kills all spores that do not produce the specific Wtfantidote). Therefore, the relative number of spores produced by a heterozygous diploid is 1–t and the average relative fitness of a population with n drivers all at equal frequency (Pi) and strength of drive (t) is:
| (S.1) |
This means that a population with n drivers is, on average, (t-t/n) less fit than a population without drivers (Appendix 1—figure 1A). This ‘drive load’ is quite severe with large t and large n. Based on our findings, various isolates of S. pombe contain wtf drivers that exhibit a drive strength around t = 0.9 and different isolates contain multiple, diverse wtf drivers (this number varies considerably) (Bravo Núñez et al., 2020; Eickbush et al., 2019; Hu et al., 2017). For example, if a given population contained five segregating drivers, the fitness of this population would be 1–0.9 + (0.9/5)=0.28 (or 72% less fit) relative to a population without drive. Such a burden would select for resistance mechanisms that might include positive assortative mating or inbreeding, resistance alleles such as wtf18-2 or aneuploidy (Bravo Núñez et al., 2018). With aneuploidy, some fraction of the spores (f/2, see below) would inherit both drive chromosomes, allowing the spores to produce both antidotes and be protected from the Wtf poisons. However, an equal fraction (f/2) would receive zero copies of chromosome 3 and would therefore be inviable (Niwa et al., 2006).
Appendix 1—figure 1. The drive load due to wtf segregating within populations.
(A) From Equation S.1, n is the number of drivers in the population and t is the strength of drive. Wild-type fitness is relative to a population without any drivers. (B) Meiotic mutant invasion criteria with extrinsic costs. All curves assume f = 0.1. Other parameters are listed in the plot. A mutant is expected to invade when the parameters for t and n fall above the line of interest. (C) Narrow parameter space allows for stable equilibrium. Solid line represents tcritical, below which the segregation mutant cannot invade. Dashed line represents tfixation, above which the mutant fixes. In the space between the lines, the equilibrium ranges from zero (solid line) to one (dashed line). n = 5, sm = 0.2, ss = 0.1, h = 1.
We assume that a single locus controls segregation fidelity and that a mutation at that locus (termed a segregation infidelity mutant) results in an increase in the number of aneuploid spores produced due to a defect in meiosis I. We also assume that this mutation only influences segregation of chromosome 3 (where most wtf drivers reside) but is not on the third chromosome itself (Bowen et al., 2003; Eickbush et al., 2019; Hu et al., 2017). However, reduced segregation fidelity of other chromosomes can be included in the costs discussed below. The parameter f denotes the segregation infidelity and can range from 0 (all spores are haploid) to 1 (all meioses result in 50% of spores being disomic and 50% lacking a third chromosome). Therefore, this segregation infidelity mutant has an intrinsic fitness cost since 50% of the generated spores lack a third chromosome and are therefore inviable. The recursion for the frequency of the infidelity mutant is:
| (S.2A) |
| (S.2B) |
Results
Invasion
For the mutant to invade, we must have (the frequency of the mutation in the next generation must be higher than in the current generation). Solving for in terms of t, we find:
| (S.3) |
Surprisingly, the critical value of t allowing for invasion is independent of the degree of segregation infidelity (f) as long as f > 0 (Figure 7B).
In an attempt to get a more intuitive feel for the path a segregation infidelity mutant might take, we plotted trajectories of such mutants using variable parameters. Figure 7C shows that changes in some parameters influence the trajectories in predictable ways: a) increasing segregation infidelity from f = 0.1 to f = 0.9 allows for a more rapid spread; b) dominant mutations (h = 1) spread more rapidly than recessive ones (h = 0); c) more segregating drivers (n = 5) lead to a more rapid spread than fewer segregating drivers (n = 3); and d) lower strength of drive (t = 0.55 compared to t = 0.95) can lead to the loss of the infidelity mutant as predicted by Equation S.3 and Figure 7B. Note that in this no-cost model, there is no stable internal equilibrium. All infidelity mutants are either lost or fixed. Accounting for additional costs of the infidelity mutant leads to the possibility of a stable equilibrium, though the parameters allowing this equilibrium are narrow (Appendix 1—figure 1C).
Allowing for additional costs of segregation infidelity mutants
Segregation infidelity mutants incur an intrinsic fitness cost since 50% of the spores do not contain a chromosome. However, our model allows us to assess additional costs that this mutant may carry. We imagine two such costs. First, we allow for additional fitness costs (ss) experienced by disomic spores. There is ample evidence that spores disomic for the third chromosome jettison one copy relatively quickly (Niwa et al., 2006), but there may be some initial cost due to gene dosage imbalance or other factors. Second, we can imagine that the meiotic mutant might also bear a cost because it leads to other problems including missegregation of the other chromosomes. We denote this cost as sm with an associated dominance (h). The dominance coefficient (h) is assumed to apply to both the cost of the segregation mutant (sm) and the segregation infidelity (f). The recursion for the frequency of the infidelity mutant is therefore:
| (S.4A) |
| (S.4B) |
Invasion with additional costs
For the mutant to invade, we again must have . Solving for in terms of t, we find:
| (S.5) |
If we assume that there is no cost of being a disomic spore (ss = 0), this becomes:
| (S.5A) |
If we instead assume that there is no cost of carrying the meiotic mutation (sm = 0), this becomes:
| (S.5B) |
Appendix 1—figure 1B shows the effect of these costs on the critical value of drive strength required for a meiotic infidelity mutant to invade. Note that even with high cost (20%), strong drivers select for these meiotic infidelity mutants. In addition to reducing the parameter space allowing for invasion of segregation infidelity mutants, these additional costs also slow the invasion of the mutant. For example, a mutant without extrinsic costs (sm and ss are 0) takes 25 generations to climb from a frequency of 1% to 50% (f = 0.33, t = 0.9, h = 1; n = 5). With a low cost (sm = 0.05) this would take 32 generations, and with a higher cost (sm = 0.20) this would take 755 generations. Note that with higher costs (e.g. sm = 0.3), the mutant does not invade because our t = 0.9 is less than the critical t = 0.978 required for invasion. However, with observed parameters in various isolates of S. pombe (t = 0.9 and 4–14 distinct wtf drivers), the system could tolerate considerable cost and still allow mutants to invade.
Equilibrium frequency of meiotic mutants with costs
Extrinsic costs of segregation infidelity mutants also allow for a narrow range of stable equilibria in some cases. Solving for q provides the equilibrium values. There are three such values: 0, 1 and
| (S.6) |
The value obtained in Equation S.6 is between zero and one for only a narrow range of parameter values. We can think of this in terms of a range of t allowing for a stable equilibrium. Below tcritical, the mutation is lost. However, if t is high enough, the meiotic mutation will fix. This happens when , which occurs when
| (S.7) |
Appendix 1—figure 1C shows the narrow range allowing for stable equilibrium. Note that in the narrow space between the solid line for tcritical and the dashed line for tfixation, the stable equilibrium frequency ranges from zero to one.
Limitations and caveats
Our model is simplistic in several ways but does suggest that a wtf-like poison-antidote system might select for mutations that disrupt ‘normal’ chromosome segregation fidelity. The first caveat is that we assume the population is randomly mating. However, due to spatial proximity of cells, we expect that inbreeding may be common. Such inbreeding would reduce the proportion of matings between haploids with different drive chromosomes and therefore decrease the benefit of segregation infidelity. A second caveat is that we assume all drive strengths are equal and all drivers are at equal frequency in the population. This is a logical starting point but is unlikely to be the case in real populations. Increasing the variance of the frequencies of individual drive chromosomes within a single population will also reduce the proportion of matings between haploids with different drive chromosomes and will decrease the benefit of segregation infidelity. Finally, we do not allow for non-driving third chromosomes. Diploids with a wild-type and a driving chromosome would still benefit from segregation infidelity. This benefit would be due to the disomic spores generated, in which both poison and antidote would be produced, protecting the disomic spores from being killed. However, selection for the infidelity mutation would be weaker because the drive load in the population would be less since fewer spores are killed each generation due to drive.
Funding Statement
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Contributor Information
Sarah E Zanders, Email: sez@stowers.org.
Adèle L Marston, University of Edinburgh, United Kingdom.
Patricia J Wittkopp, University of Michigan, United States.
Funding Information
This paper was supported by the following grants:
Stowers Institute for Medical Research to Sarah E Zanders.
March of Dimes Foundation Basil O'Connor Starter Scholar Research Award No. 5-FY18-58 to Sarah E Zanders.
Kinship Foundation Searle Scholars Award to Sarah E Zanders.
National Institute of General Medical Sciences R00GM114436 to Sarah E Zanders.
National Institute of General Medical Sciences DP2GM132936 to Sarah E Zanders.
National Cancer Institute F99CA234523 to María Angélica Bravo Núñez.
University of Kansas to Robert L Unckless.
Additional information
Competing interests
Inventor on a patent application based on wtf killers (patent application serial 62/491,107).
No competing interests declared.
Inventor on a patent application based on wtf killers (patent application serial 62/491,107).
Author contributions
Conceptualization, Data curation, Formal analysis, Funding acquisition, Validation, Investigation, Visualization, Methodology, Writing - original draft, Writing - review and editing.
Data curation, Formal analysis, Validation, Investigation, Visualization, Methodology, Writing - original draft, Writing - review and editing.
Data curation, Formal analysis, Validation, Investigation.
Conceptualization, Data curation, Formal analysis, Validation, Investigation, Visualization, Methodology, Writing - original draft, Writing - review and editing.
Conceptualization, Data curation, Formal analysis, Supervision, Funding acquisition, Visualization, Methodology, Writing - original draft, Project administration, Writing - review and editing.
Additional files
#Strains SZY1535 and SZY1537 are Ura+ because when the Sk wtf4 allele was originally made (described in Nuckolls et al., 2017), the strains retained the ura4+ cassette. The ura4 cassette is at an unknown location but linked to the wtf4 locus. *Strains SZY3910 and SZY3911 were generated via crossing. One of the parental strains had a wild-type ura4 allele and was thus Ura+. The other parental strain contained the ura4-D18, but had a rec12∆::ura4+, and thus was also Ura+. However, when we crossed the strains to generate SZY3910 and SZY3911, we confirmed the presence of the rec12 allele using PCR, but we did not determine if they contained the ura4-D18 or the ura4+ allele.
Column 1 lists the wtf gene cloned into each vector. Column 2 denotes the isolate from which each wtf was cloned. The DNA templates and oligos used in the PCR reactions to amplify the wtf alleles are shown in columns 3 and 4, respectively. We digested each of the amplified fragments with the enzymes reported in column 5 and then integrated into the target site listed in column 6. The strain number of each of the plasmids that we generated is reported in column 7. The description of each plasmid can be found in Supplementary file 2.
Data availability
All data generated or analysed during this study are included in the manuscript and supporting files.
References
- Abbott S, Fairbanks DJ. Experiments on plant hybrids by Gregor Mendel. Genetics. 2016;204:407–422. doi: 10.1534/genetics.116.195198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akera T, Chmátal L, Trimm E, Yang K, Aonbangkhen C, Chenoweth DM, Janke C, Schultz RM, Lampson MA. Spindle asymmetry drives non-Mendelian chromosome segregation. Science. 2017;358:668–672. doi: 10.1126/science.aan0092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akera T, Trimm E, Lampson MA. Molecular strategies of meiotic cheating by selfish centromeres. Cell. 2019;178:1132–1144. doi: 10.1016/j.cell.2019.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avelar AT, Perfeito L, Gordo I, Ferreira MG. Genome architecture is a selectable trait that can be maintained by antagonistic pleiotropy. Nature Communications. 2013;4:2235. doi: 10.1038/ncomms3235. [DOI] [PubMed] [Google Scholar]
- Bauer H, Schindler S, Charron Y, Willert J, Kusecek B, Herrmann BG. The nucleoside diphosphate kinase gene Nme3 acts as quantitative trait locus promoting non-Mendelian inheritance. PLOS Genetics. 2012;8:e1002567. doi: 10.1371/journal.pgen.1002567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergerat A, de Massy B, Gadelle D, Varoutas PC, Nicolas A, Forterre P. An atypical topoisomerase II from archaea with implications for meiotic recombination. Nature. 1997;386:414–417. doi: 10.1038/386414a0. [DOI] [PubMed] [Google Scholar]
- Bowen NJ, Jordan IK, Epstein JA, Wood V, Levin HL. Retrotransposons and their recognition of pol II promoters: a comprehensive survey of the transposable elements from the complete genome sequence of Schizosaccharomyces pombe. Genome Research. 2003;13:1984–1997. doi: 10.1101/gr.1191603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bravo Núñez MA, Lange JJ, Zanders SE. A suppressor of a wtf poison-antidote meiotic driver acts via mimicry of the driver's antidote. PLOS Genetics. 2018;14:e1007836. doi: 10.1371/journal.pgen.1007836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bravo Núñez MA, Sabbarini IM, Eickbush MT, Liang Y, Lange JJ, Kent AM, Zanders SE. Dramatically diverse Schizosaccharomyces pombe wtf meiotic drivers all display high gamete-killing efficiency. PLOS Genetics. 2020;16:e1008350. doi: 10.1371/journal.pgen.1008350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown WRA, Liti G, Rosa C, James S, Roberts I, Robert V, Jolly N, Tang W, Baumann P, Green C, Schlegel K, Young J, Hirchaud F, Leek S, Thomas G, Blomberg A, Warringer J. A geographically diverse collection of Schizosaccharomyces pombe isolates shows limited phenotypic variation but extensive karyotypic diversity. G3: Genes, Genomes, Genetics. 2011;1:615–626. doi: 10.1534/g3.111.001123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burt A, Trivers R. Genes in conflict: The biology of selfish genetic elements. Cambridge, Mass.: Belknap Press of Harvard University Press; 2006. [Google Scholar]
- Chuang YC, Li WC, Chen CL, Hsu PW, Tung SY, Kuo HC, Schmoll M, Wang TF. Trichoderma reesei meiosis generates segmentally aneuploid progeny with higher xylanase-producing capability. Biotechnology for Biofuels. 2015;8:30. doi: 10.1186/s13068-015-0202-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crow JF. Why is Mendelian segregation so exact? BioEssays. 1991;13:305–312. doi: 10.1002/bies.950130609. [DOI] [PubMed] [Google Scholar]
- Davis L, Smith GR. Nonrandom homolog segregation at meiosis I in Schizosaccharomyces pombe mutants lacking recombination. Genetics. 2003;163:857–874. doi: 10.1093/genetics/163.3.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawe RK, Lowry EG, Gent JI, Stitzer MC, Swentowsky KW, Higgins DM, Ross-Ibarra J, Wallace JG, Kanizay LB, Alabady M, Qiu W, Tseng KF, Wang N, Gao Z, Birchler JA, Harkess AE, Hodges AL, Hiatt EN. A Kinesin-14 motor activates neocentromeres to promote meiotic drive in maize. Cell. 2018;173:839–850. doi: 10.1016/j.cell.2018.03.009. [DOI] [PubMed] [Google Scholar]
- Didion JP, Morgan AP, Clayshulte AM, Mcmullan RC, Yadgary L, Petkov PM, Bell TA, Gatti DM, Crowley JJ, Hua K, Aylor DL, Bai L, Calaway M, Chesler EJ, French JE, Geiger TR, Gooch TJ, Garland T, Harrill AH, Hunter K, McMillan L, Holt M, Miller DR, O'Brien DA, Paigen K, Pan W, Rowe LB, Shaw GD, Simecek P, Sullivan PF, Svenson KL, Weinstock GM, Threadgill DW, Pomp D, Churchill GA, Pardo-Manuel de Villena F. A multi-megabase copy number gain causes maternal transmission ratio distortion on mouse chromosome 2. PLOS Genetics. 2015;11:e1004850. doi: 10.1371/journal.pgen.1004850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobzhansky T, Sturtevant AH. Inversions in the chromosomes of Drosophila pseudoobscura. Genetics. 1938;23:28–64. doi: 10.1093/genetics/23.1.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dyer KA, Charlesworth B, Jaenike J. Chromosome-wide linkage disequilibrium as a consequence of meiotic drive. PNAS. 2007;104:1587–1592. doi: 10.1073/pnas.0605578104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egel R. Frequency of mating-type switching in homothallic fission yeast. Nature. 1977;266:172–174. doi: 10.1038/266172a0. [DOI] [PubMed] [Google Scholar]
- Egel R. Fission yeast in general genetics. In: Egel R, editor. The Molecular Biology of Schizosaccharomyces Pombe: Genetics, Genomics and Beyond. Berlin, Heidelberg: Springer; 2004. pp. 1–12. [DOI] [Google Scholar]
- Eickbush MT, Young JM, Zanders SE. Killer meiotic drive and dynamic evolution of the wtf gene family. Molecular Biology and Evolution. 2019;36:1201–1214. doi: 10.1093/molbev/msz052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elguero E, Délicat-Loembet LM, Rougeron V, Arnathau C, Roche B, Becquart P, Gonzalez JP, Nkoghe D, Sica L, Leroy EM, Durand P, Ayala FJ, Ollomo B, Renaud F, Prugnolle F. Malaria continues to select for sickle cell trait in central africa. PNAS. 2015;112:7051–7054. doi: 10.1073/pnas.1505665112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fantes PA, Hoffman CS. A brief history of Schizosaccharomyces pombe research: a perspective over the past 70 years. Genetics. 2016;203:621–629. doi: 10.1534/genetics.116.189407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farlow A, Long H, Arnoux S, Sung W, Doak TG, Nordborg M, Lynch M. The spontaneous mutation rate in the fission yeast Schizosaccharomyces pombe. Genetics. 2015;201:737–744. doi: 10.1534/genetics.115.177329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fawcett JA, Iida T, Takuno S, Sugino RP, Kado T, Kugou K, Mura S, Kobayashi T, Ohta K, Nakayama J, Innan H. Population genomics of the fission yeast Schizosaccharomyces pombe. PLOS ONE. 2014;9:e104241. doi: 10.1371/journal.pone.0104241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forsburg SL, Rhind N. Basic methods for fission yeast. Yeast. 2006;23:173–183. doi: 10.1002/yea.1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein AL, McCusker JH. Three new dominant drug resistance cassettes for gene disruption in Saccharomyces cerevisiae. Yeast. 1999;15:1541–1553. doi: 10.1002/(SICI)1097-0061(199910)15:14<1541::AID-YEA476>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- Grey C, Baudat F, de Massy B. PRDM9, a driver of the genetic map. PLOS Genetics. 2018;14:e1007479. doi: 10.1371/journal.pgen.1007479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grognet P, Lalucque H, Malagnac F, Silar P. Genes that bias Mendelian segregation. PLOS Genetics. 2014;10:e1004387. doi: 10.1371/journal.pgen.1004387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutz H, Doe FJ. On homo- and heterothallism in Schizosaccharomyces pombe. Mycologia. 1975;67:748–759. doi: 10.1080/00275514.1975.12019805. [DOI] [PubMed] [Google Scholar]
- Haldane JBS. The combination of linkage values and the calculation of distance between the loci fo linked factors. Journal of Genetics. 1919;8:299–309. [Google Scholar]
- Hammer MF, Schimenti J, Silver LM. Evolution of mouse chromosome 17 and the origin of inversions associated with t haplotypes. PNAS. 1989;86:3261–3265. doi: 10.1073/pnas.86.9.3261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammond TM, Rehard DG, Xiao H, Shiu PK. Molecular dissection of Neurospora spore killer meiotic drive elements. PNAS. 2012;109:12093–12098. doi: 10.1073/pnas.1203267109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartl DL. Modifier theory and meiotic drive. Theoretical Population Biology. 1975;7:168–174. doi: 10.1016/0040-5809(75)90012-X. [DOI] [PubMed] [Google Scholar]
- Henikoff S, Ahmad K, Malik HS. The centromere paradox: stable inheritance with rapidly evolving DNA. Science. 2001;293:1098–1102. doi: 10.1126/science.1062939. [DOI] [PubMed] [Google Scholar]
- Hu W, Jiang ZD, Suo F, Zheng JX, He WZ, Du LL. A large gene family in fission yeast encodes spore killers that subvert Mendel's law. eLife. 2017;6:e26057. doi: 10.7554/eLife.26057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeffares DC, Rallis C, Rieux A, Speed D, Převorovský M, Mourier T, Marsellach FX, Iqbal Z, Lau W, Cheng TM, Pracana R, Mülleder M, Lawson JL, Chessel A, Bala S, Hellenthal G, O'Fallon B, Keane T, Simpson JT, Bischof L, Tomiczek B, Bitton DA, Sideri T, Codlin S, Hellberg JE, van Trigt L, Jeffery L, Li JJ, Atkinson S, Thodberg M, Febrer M, McLay K, Drou N, Brown W, Hayles J, Carazo Salas RE, Ralser M, Maniatis N, Balding DJ, Balloux F, Durbin R, Bähler J. The genomic and phenotypic diversity of Schizosaccharomyces pombe. Nature Genetics. 2015;47:235–241. doi: 10.1038/ng.3215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeffares DC. The natural diversity and ecology of fission yeast. Yeast. 2018;35:253–260. doi: 10.1002/yea.3293. [DOI] [PubMed] [Google Scholar]
- Kato Yamakake TA. Cytological Studies of Maize [Zea Mays L.] and Teosinte [Zea Mexicana Schrader Kuntze] in Relation to Their Origin and Evolution. Research Bulletin / Massachusetts Agricultural Experiment Station. Amherst, Mass: Massachusetts Agricultural Experiment Station; 1976. [Google Scholar]
- Keeney S, Giroux CN, Kleckner N. Meiosis-specific DNA double-strand breaks are catalyzed by Spo11, a member of a widely conserved protein family. Cell. 1997;88:375–384. doi: 10.1016/S0092-8674(00)81876-0. [DOI] [PubMed] [Google Scholar]
- Kitajima TS, Kawashima SA, Watanabe Y. The conserved kinetochore protein shugoshin protects centromeric cohesion during meiosis. Nature. 2004;427:510–517. doi: 10.1038/nature02312. [DOI] [PubMed] [Google Scholar]
- Krawchuk MD, DeVeaux LC, Wahls WP. Meiotic chromosome dynamics dependent upon the rec8+, rec10+ and rec11+ genes of the fission yeast Schizosaccharomyces pombe. Genetics. 1999;153:57–68. doi: 10.1093/genetics/153.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kutzer MAM, Armitage SAO. Maximising fitness in the face of parasites: a review of host tolerance. Zoology. 2016;119:281–289. doi: 10.1016/j.zool.2016.05.011. [DOI] [PubMed] [Google Scholar]
- Larracuente AM, Presgraves DC. The selfish segregation distorter gene complex of Drosophila melanogaster. Genetics. 2012;192:33–53. doi: 10.1534/genetics.112.141390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long Y, Zhao L, Niu B, Su J, Wu H, Chen Y, Zhang Q, Guo J, Zhuang C, Mei M, Xia J, Wang L, Wu H, Liu YG. Hybrid male sterility in rice controlled by interaction between divergent alleles of two adjacent genes. PNAS. 2008;105:18871–18876. doi: 10.1073/pnas.0810108105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López Hernández JF, Zanders SE. Veni, vidi, vici: the success of wtf meiotic drivers in fission yeast. Yeast. 2018;35:447–453. doi: 10.1002/yea.3305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenz A, Wells JL, Pryce DW, Novatchkova M, Eisenhaber F, McFarlane RJ, Loidl J. S. pombe meiotic linear elements contain proteins related to synaptonemal complex components. Journal of Cell Science. 2004;117:3343–3351. doi: 10.1242/jcs.01203. [DOI] [PubMed] [Google Scholar]
- Marais G. Biased gene conversion: implications for genome and sex evolution. Trends in Genetics. 2003;19:330–338. doi: 10.1016/S0168-9525(03)00116-1. [DOI] [PubMed] [Google Scholar]
- Martín-Castellanos C, Blanco M, Rozalén AE, Pérez-Hidalgo L, García AI, Conde F, Mata J, Ellermeier C, Davis L, San-Segundo P, Smith GR, Moreno S. A large-scale screen in S. pombe identifies seven novel genes required for critical meiotic events. Current Biology. 2005;15:2056–2062. doi: 10.1016/j.cub.2005.10.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin RN, Malik HS. Genetic conflicts: the usual suspects and beyond. The Journal of Experimental Biology. 2017;220:6–17. doi: 10.1242/jeb.148148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murdoch B, Owen N, Stevense M, Smith H, Nagaoka S, Hassold T, McKay M, Xu H, Fu J, Revenkova E, Jessberger R, Hunt P. Altered cohesin gene dosage affects mammalian meiotic chromosome structure and behavior. PLOS Genetics. 2013;9:e1003241. doi: 10.1371/journal.pgen.1003241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni M, Feretzaki M, Li W, Floyd-Averette A, Mieczkowski P, Dietrich FS, Heitman J. Unisexual and heterosexual meiotic reproduction generate aneuploidy and phenotypic diversity de novo in the yeast cryptococcus neoformans. PLOS Biology. 2013;11:e1001653. doi: 10.1371/journal.pbio.1001653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niwa O, Tange Y, Kurabayashi A. Growth arrest and chromosome instability in aneuploid yeast. Yeast. 2006;23:937–950. doi: 10.1002/yea.1411. [DOI] [PubMed] [Google Scholar]
- Nuckolls NL, Bravo Núñez MA, Eickbush MT, Young JM, Lange JJ, Yu JS, Smith GR, Jaspersen SL, Malik HS, Zanders SE. wtf genes are prolific dual poison-antidote meiotic drivers. eLife. 2017;6:e26033. doi: 10.7554/eLife.26033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardo-Manuel de Villena F, Sapienza C. Female meiosis drives karyotypic evolution in mammals. Genetics. 2001;159:1179–1189. doi: 10.1093/genetics/159.3.1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieper KE, Unckless RL, Dyer KA. A fast-evolving X-linked duplicate of importin-α2 is overexpressed in sex-ratio drive in Drosophila neotestacea. Molecular Ecology. 2018;27:5165–5179. doi: 10.1111/mec.14928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieper KE, Dyer KA. Occasional recombination of a selfish X-chromosome may permit its persistence at high frequencies in the wild. Journal of Evolutionary Biology. 2016;29:2229–2241. doi: 10.1111/jeb.12948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prieler S, Penkner A, Borde V, Klein F. The control of Spo11's interaction with meiotic recombination hotspots. Genes & Development. 2005;19:255–269. doi: 10.1101/gad.321105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhoades MM. Preferential segregation in maize. Genetics. 1942;27:395–407. doi: 10.1093/genetics/27.4.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhoades NA, Harvey AM, Samarajeewa DA, Svedberg J, Yusifov A, Abusharekh A, Manitchotpisit P, Brown DW, Sharp KJ, Rehard DG, Peters J, Ostolaza-Maldonado X, Stephenson J, Shiu PKT, Johannesson H, Hammond TM. Identification of rfk-1, a meiotic driver undergoing RNA editing in Neurospora. Genetics. 2019;212:93–110. doi: 10.1534/genetics.119.302122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Lopez M, Cotobal C, Fernandez-Sanchez O, Borbaran Bravo N, Oktriani R, Abendroth H, Uka D, Hoti M, Wang J, Zaratiegui M, Bahler J. A CRISPR/Cas9-based method and primer design tool for seamless genome editing in fission yeast. Wellcome Open Res 1:19. 10.12688/wellcomeopenres. 2016;10038:3. doi: 10.12688/wellcomeopenres.10038.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosin LF, Mellone BG. Centromeres drive a hard bargain. Trends in Genetics. 2017;33:101–117. doi: 10.1016/j.tig.2016.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandler L, Novitski E. Meiotic drive as an evolutionary force. The American Naturalist. 1957;91:105–110. doi: 10.1086/281969. [DOI] [Google Scholar]
- Serjeant GR. One hundred years of sickle cell disease. British Journal of Haematology. 2010;151:425–429. doi: 10.1111/j.1365-2141.2010.08419.x. [DOI] [PubMed] [Google Scholar]
- Singh G, Klar AJ. The 2.1-kb inverted repeat DNA sequences flank the mat2,3 silent region in two species of Schizosaccharomyces and are involved in epigenetic silencing in Schizosaccharomyces pombe. Genetics. 2002;162:591–602. doi: 10.1093/genetics/162.2.591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith GR. Genetic analysis of meiotic recombination in Schizosaccharomyces pombe. Methods in Molecular Biology. 2009;557:65–76. doi: 10.1007/978-1-59745-527-5_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorci G, Garnier S. Parasitism. In: Jørgensen S. E, Fath B. D, editors. Encyclopedia of Ecology. Oxford: Academic Press; 2008. pp. 2645–2650. [DOI] [Google Scholar]
- Stalker HD. The genetic systems modifying meiotic drive in Drosophila paramelanica. Genetics. 1961;46:177–202. doi: 10.1093/genetics/46.2.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subramanian VV, Bickel SE. Aging predisposes oocytes to meiotic nondisjunction when the cohesin subunit SMC1 is reduced. PLOS Genetics. 2008;4:e1000263. doi: 10.1371/journal.pgen.1000263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svedberg J, Hosseini S, Chen J, Vogan AA, Mozgova I, Hennig L, Manitchotpisit P, Abusharekh A, Hammond TM, Lascoux M, Johannesson H. Convergent evolution of complex genomic rearrangements in two fungal meiotic drive elements. Nature Communications. 2018;9:4242. doi: 10.1038/s41467-018-06562-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tusso S, Nieuwenhuis BPS, Sedlazeck FJ, Davey JW, Jeffares DC, Wolf JBW. Ancestral admixture is the main determinant of global biodiversity in fission yeast. Molecular Biology and Evolution. 2019;36:1975–1989. doi: 10.1093/molbev/msz126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Úbeda F, Russell TW, Jansen VAA. PRDM9 and the evolution of recombination hotspots. Theoretical Population Biology. 2019;126:19–32. doi: 10.1016/j.tpb.2018.12.005. [DOI] [PubMed] [Google Scholar]
- Vogan AA, Ament-Velásquez SL, Granger-Farbos A, Svedberg J, Bastiaans E, Debets AJ, Coustou V, Yvanne H, Clavé C, Saupe SJ, Johannesson H. Combinations of spok genes create multiple meiotic drivers in Podospora. eLife. 2019;8:e46454. doi: 10.7554/eLife.46454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wach A, Brachat A, Pöhlmann R, Philippsen P. New heterologous modules for classical or PCR-based gene disruptions in Saccharomyces cerevisiae. Yeast. 1994;10:1793–1808. doi: 10.1002/yea.320101310. [DOI] [PubMed] [Google Scholar]
- Watanabe Y, Nurse P. Cohesin Rec8 is required for reductional chromosome segregation at meiosis. Nature. 1999;400:461–464. doi: 10.1038/22774. [DOI] [PubMed] [Google Scholar]
- Xie Y, Tang J, Xie X, Li X, Huang J, Fei Y, Han J, Chen S, Tang H, Zhao X, Tao D, Xu P, Liu YG, Chen L. An asymmetric allelic interaction drives allele transmission bias in interspecific rice hybrids. Nature Communications. 2019;10:2501. doi: 10.1038/s41467-019-10488-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, Zhao X, Cheng K, Du H, Ouyang Y, Chen J, Qiu S, Huang J, Jiang Y, Jiang L, Ding J, Wang J, Xu C, Li X, Zhang Q. A killer-protector system regulates both hybrid sterility and segregation distortion in rice. Science. 2012;337:1336–1340. doi: 10.1126/science.1223702. [DOI] [PubMed] [Google Scholar]
- Yokobayashi S, Watanabe Y. The kinetochore protein Moa1 enables cohesion-mediated monopolar attachment at meiosis I. Cell. 2005;123:803–817. doi: 10.1016/j.cell.2005.09.013. [DOI] [PubMed] [Google Scholar]
- Yoon S-W, Lee M-S, Xaver M, Zhang L, Hong S-G, Kong Y-J, Cho H-R, Kleckner N, Kim KP. Meiotic prophase roles of Rec8 in crossover recombination and chromosome structure. Nucleic Acids Research. 2016;5:gkw682. doi: 10.1093/nar/gkw682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Zhao Z, Zheng X, Zhou J, Kong W, Wang P, Bai W, Zheng H, Zhang H, Li J, Liu J, Wang Q, Zhang L, Liu K, Yu Y, Guo X, Wang J, Lin Q, Wu F, Ren Y, Zhu S, Zhang X, Cheng Z, Lei C, Liu S, Liu X, Tian Y, Jiang L, Ge S, Wu C, Tao D, Wang H, Wan J. A selfish genetic element confers non-Mendelian inheritance in rice. Science. 2018;360:1130–1132. doi: 10.1126/science.aar4279. [DOI] [PubMed] [Google Scholar]
- Zanders SE, Eickbush MT, Yu JS, Kang JW, Fowler KR, Smith GR, Malik HS. Genome rearrangements and pervasive meiotic drive cause hybrid infertility in fission yeast. eLife. 2014;3:e02630. doi: 10.7554/eLife.02630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanders SE, Unckless RL. Fertility costs of meiotic drivers. Current Biology. 2019;29:R512–R520. doi: 10.1016/j.cub.2019.03.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmering S, Sandler L, Nicoletti B. Mechanisms of meiotic drive. Annual Review of Genetics. 1970;4:409–436. doi: 10.1146/annurev.ge.04.120170.002205. [DOI] [PubMed] [Google Scholar]