Abstract
Degradation of bio-based (polylactic acid [PLA] cups, Mater-Bi® [MB] bags) and biodegradable plastics (biodegradable extruded polystyrene [bioPS] plates, biodegradable high density polyethylene [bioHDPE] bags) were compared to conventional plastics (recycled polyethylene terephthalate [rPET] cups, HDPE bags, extruded PS plates) in a salt marsh over a 32-week period. Following 4 weeks, biofilm developed on all plastics, resulting in an increased weight and concomitant decrease in UV transmission for most plastics. All plastics produced microplastic particles beginning at 4 weeks, with single-use bags producing the most microplastics over the 32-week period. At 32 weeks, SEM revealed microcracks and delamination for all plastics except PLA and MB, the latter of which degraded through embrittlement. IR spectral analysis indicated degradation for all plastics except PLA. Results suggest that degradation rates of bio-based and biodegradable plastics vary widely, with MB bags and bioPS plates demonstrating the greatest degradation, while PLA cups demonstrated the least degradation.
Keywords: Microplastics, High density polyethylene, Polystyrene, Polyethylene terephthalate, Polylactic acid
Graphical abstract
Highlights
-
•
Degradation of bio-based and biodegradable plastics varied widely.
-
•
Microplastic particles were produced from all tested products in as little as 4 weeks.
-
•
Single-use bags produced the greatest amount of microplastics.
-
•
Mater-Bi® bags and polystyrene plates made with green2technology™ exhibited the most degradation.
-
•
PLA cups exhibited the least degradation.
1. Introduction
As a result of the growing awareness of the global pollution problem resulting from the mismanaged disposal of conventional oil-based plastics and the demand for more sustainable products, environmentally friendly, or eco-friendly plastics are becoming increasingly popular among consumers. One type of eco-friendly plastic is the bio-based plastics, also called bioplastics, which are derived from renewable resources, such as plant materials or methane. Currently, bio-based plastics, the most popular type of eco-friendly plastic, represent about 1% of the 335 million tonnes of plastic produced annually (European Bioplastics, 2018). With rising consumer demand, global bio-based plastic production is expected to increase about 24% from 2.11 million tonnes in 2018 to 2.62 million tonnes in 2023 (European Bioplastics, 2018). Much of this growth is expected to be driven by the increased production of polylactic acid (PLA), the most commonly used bio-based plastic (Iwata, 2015). PLA is made by breaking down the starch in corn and other plants to produce glucose that is fermented to produce lactic acid, which is then polymerized into PLA (Khare and Deshmukh, 2006).
Another type of eco-friendly plastic are the biodegradable plastics, which can be composed of either biomass-based or oil-based polymers, and are engineered to decompose more quickly than conventional plastics primarily through the enzymatic action of microorganisms (Song et al., 2009). Oil-based polymers can be made biodegradable by blending the synthetic polymeric material with natural compounds, such as algae and other plant material (e.g. starch, cellulose, lignin, and chitin), or by the addition of organic catalysts.
Degradation for all types of plastics, whether considered eco-friendly or not, is ultimately the result of chemical changes in the structure of the material reducing its molecular weight, invariably weakening its structural integrity (reviewed by Singh and Sharma, 2008). For most oil-based plastics, degradation is the result of oxidative reactions, beginning with photooxidation from UV radiation exposure followed by temperature-dependent thermooxidative reactions (Andrady, 2011). Collectively, these reactions result in a decrease in the molecular weight of the polymers and an increase in the presence of oxygen-rich functional groups. Other degradative mechanisms for oil-based plastics, such as hydrolysis and biodegradation, occur at rates several orders of magnitude slower than oxidative mechanisms (Andrady, 2011). By contrast, most eco-friendly plastics have physical and chemical properties to either enhance biodegradation or, as in the case of PLA, facilitate degradation under commercial composting conditions.
Because plastic degradation is dependent upon factors such as UV radiation, oxygen, and temperature, the rate at which any plastic degrades will largely be dependent upon environmental conditions. For example, polyethylene films and thermoplastic materials were found to degrade at slower rates while submerged in seawater compared to those in the air, probably as a result of lower sunlight exposure, lower temperatures, and lower oxygen levels (Pegram and Andrady, 1989; Andrady et al., 1993). Mechanical forces, such as those associated with turbulence and abrasion could increase the susceptibility of plastics to fragmentation (Pegram and Andrady, 1989; Browne et al., 2010). Surface biofouling, such as that associated with biofilm, may reduce the rate of plastic degradation by reducing the amount of UV radiation reaching the surface of the plastic, while simultaneously increasing the density of the object causing it to sink (Kershaw et al., 2011).
Although it is generally assumed that bio-based and biodegradable plastics will degrade faster than oil-based plastics, only a limited number of studies have been conducted, and their results have been mixed. For example, Napper and Thompson (2019) exposed several different types of carrier bags, including biodegradable, oxo-biodegradable, and compostable, to open-air, soil, and seawater for up to 3 years. They found that none of the bags could be relied upon to substantially deteriorate more than conventional HDPE plastic bags in all three environments; however, it is of interest that the compostable bag completely disappeared in seawater after only 3 months (Napper and Thompson, 2019). These results corroborate those of O'Brine and Thompson (2010), who found that a compostable bag had 100% surface area loss following 24 weeks of exposure in seawater. These limited studies highlight the need to test a wider range of polymer types and product types in various environments.
Regardless of the rate of degradation, as the plastic material ages in the environment, microscopic particles, known as microplastics, would be expected to be produced. A previous study conducted in a salt marsh habitat indicated that several oil-based plastics, including those composed of high density polyethylene, polypropylene, and extruded polystyrene, began releasing microplastic particles through the process of surface delamination in as little as 8 weeks in (Weinstein et al., 2016). In subtidal conditions (10 m depth) in the Clyde Sea (Scotland), rope made from polyethylene, polypropylene, and nylon exhibited a reduction of weight averaging between 0.39% and 1.02% per month, presumably due to the loss of material in the form of microplastics (Welden and Cowie, 2017). In coastal waters, the degradation of plastic litter is believed to be a major source of microplastics (Gray et al., 2018), and their presence has received much attention due to their potential to adversely impact a variety of organisms (reviewed by Avio et al., 2017). Ingested microplastic particles can increase gut residency time among invertebrates, producing fatal injuries associated with gut blockage, as well as getting caught in respiratory structures (Watts et al., 2014; Gray and Weinstein, 2017; Leads et al., 2019). In addition, microplastic ingestion could lead to reduced energetics, adversely impacting both growth and fitness (reviewed by Auta et al., 2017).
Increased consumer demand for bio-based and biodegradable plastics is predicated, in part, upon the assumption that they pose less of a threat to the environment because of their faster degradation rates. However, these assumptions are largely based on assessments performed under optimal conditions in laboratory settings or in industrial composting facilities (Song et al., 2009). Given the uncertainty regarding just how long these plastics may persist as litter in coastal waterways (Barnes et al., 2009; Tosin et al., 2012), and to the extent to which they may produce microplastic particles as they degrade, it is necessary to conduct studies in situ to understand how they will degrade under environmental conditions.
One common habitat associated with southeastern U.S. estuarine systems are salt marshes. Salt marshes are physically dynamic intertidal habitats, characterized by cyclical fluctuations in dissolved oxygen, UV radiation, temperature, and salinity largely driven by diurnal tides having a 1.5–3.0 m range (Wenner et al., 1998). Salt marshes and their associated tidal creeks serve as the hydrographic link between anthropogenic activities in the upland portion of the watershed and the adjacent estuary (Holland et al., 2004), and as such, higher levels of plastic debris have been associated with salt marshes occurring near population centers (Viehman et al., 2011). Although plastic debris can be found in all habitats associated with estuaries, the highest abundances can be found in areas associated with natural debris in salt marshes known as tidal wrack (Viehman et al., 2011). In Charleston Harbor, SC (USA), Wertz (2015) estimated that there were 6.9 metric tonnes of plastic debris in the salt marshes surrounding the harbor.
The objective of this study was to investigate the degradation of several different types of bio-based and biodegradable plastics relative to that of conventional, oil-derived plastics, in a salt marsh habitat. Our hypothesis was that bio-based and biodegradable plastics, in general, would exhibit more degradation than conventional plastics.
2. Materials and methods
Seven different types of plastic strips (15.2 × 2.5 cm) were compared: high density polyethylene (HDPE), extruded polystyrene (PS), recycled polyethylene terephthalate (rPET), polylactic acid (PLA), Mater-Bi® (MB), biodegradable extruded polystyrene (bioPS), and biodegradable HDPE (bioHDPE) (Table 1 ). HDPE, PS, and rPET were categorized as conventional oil-based plastics; bioHDPE and bioPS were categorized as biodegradable plastics; and PLA and MB were categorized as bio-based plastics. Biodegradable extruded polystyrene strips (bioPS) were cut from wheat-colored D&W Fine Pack® Enviroware® plates. According to the manufacturer, these single use plates are manufactured using green2technology™ to ensure maximum recycled content, are produced with CO2 rather than pentane, and have an active organic catalyst added to the polymer during manufacturing to accelerate the degradation rate (D&W Fine Pack, 2016). Material attributes, relative to conventional PS include smaller structural cells and an increased density. Biodegradable HDPE (bioHDPE) strips were cut from green single-use grocery bags manufactured by Green Herc™. Green Herc™ biodegradable bags contain an algae-based additive that the manufacturer claims allow them to fully degrade within 1–15 years with 28.4% degradation in the first 105 days (WebstaurantStore, 2019). Polylactic acid (PLA) strips were cut from GreenStripe® Cold Cups made from Ingeo™, a plant-based plastic polymer (Eco-Products, 2019). These cups meet ASTM standards for compostability (Eco-Products, 2019). Mater-Bi® (MB) strips were cut from green-colored BioBag® regular shopper bags (BioBag USA, 2016). Mater-Bi® is a proprietary plant-based bio-based plastic derived from vegetable oils, starches, and cellulose (Novamont, 2019). According to the manufacturer, this product meets several European and international standards for compostability. Beyond those additives mentioned above, we have no knowledge regarding the use of other additives, such as plasticizers, in any of these products.
Table 1.
Characteristics of plastics examined in this study.
| Plastic type | Abbreviation | Product | Color | Thickness | Classification |
|---|---|---|---|---|---|
| High density polyethylene | HDPE | WebstaurantStore Single use grocery bag | Green | 13.0 μm | Conventional |
| Extruded polystyrene | PS | Food Lion® foam plate (22.5 cm diameter) | White | 2.0 mm | Conventional |
| Recycled polyethylene terephthalate (25% post-consumer plastic) | rPET | BlueStripe™ Cold cups (710 mL) | Clear | 0.3 mm | Conventional |
| Biodegradable high density polyethylene | bioHDPE | Green Herc™ Single use grocery bags with algal based additive | Green | 12.7 μm | Biodegradable |
| Biodegradable extruded polystyrene | bioPS | D&W Fine Pack® Enviroware® biodegradable active organic catalyst-infused plates (22.5 cm diameter) | Wheat | 2.0 mm | Biodegradable |
| Polylactic acid | PLA | GreenStripe® Cold cups (710 mL) | Clear | 0.2 mm | Bio-based |
| Mater-Bi® | MB | BioBag® Regular shopper bags | Green | 20.3 μm | Bio-based |
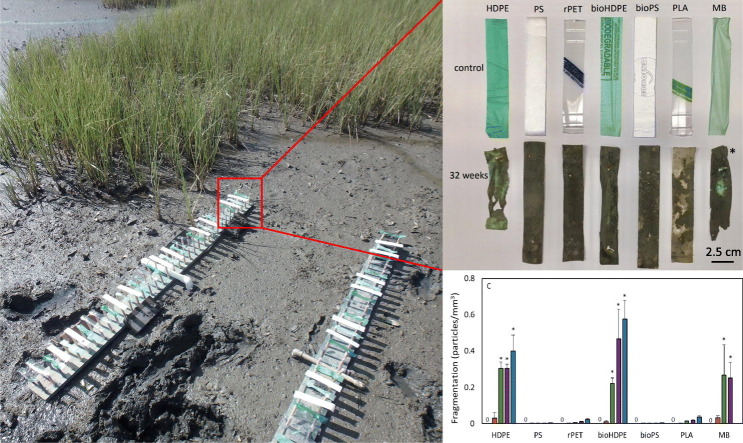
Plastic strips were field-deployed in an intertidal salt marsh in the Ashley River adjacent to The Citadel campus (32°47′45.44″N, 79°57′50.65″W) using the apparatus previously described in Weinstein et al. (2016) (Fig. 1 ). Briefly, each apparatus consisted of a wooden board (152.4 × 15.2 × 2.5 cm) (l × w × h) with between 60 and 80 perpendicularly oriented wooden dowels (1.1 cm diameter, 12.7 cm height), arranged in two rows of between 30 and 40 dowels each. Each plastic strip (30–40 total strips per apparatus; 4–6 of each polymer type per apparatus) was attached at either end to a wooden dowel (one from each row) with a finishing nail. In this manner, a total of 203 plastic strips (29 of each polymer) were deployed.
Fig. 1.

Photograph of field-deployed apparatus with plastic strips in salt marsh.
The apparatus were deployed on 25 May 2017 in an area of the marsh devoid of shade (i.e. overhanging trees or smooth cordgrass [Sporobolus alterniflorus]). During each tidal cycle, the plastic strips were submerged at high tide for approximately 6 h and exposed at low tide for approximately 6 h. Plastic strips (n = 7 for each polymer type) were randomly removed from the apparatus and brought back to the laboratory on 22 June 2017, 20 July 2017, 14 September 2017, and 11 January 2018, which represented 4, 8, 16, and 32-week exposure periods, respectively. One extra set of strips (n = 1 for each polymer) were deployed in the event that strips were lost during the 32-week exposure. Control strips (0 weeks) were not field deployed.
During the 32-week exposure, the plastic strips were subject to the local environmental and climatic conditions of the Charleston Harbor estuary, located on the southeastern coast of the United States. Charleston Harbor is a partially mixed, ebb-dominant estuary with semidiurnal tides and a tidal range of 1.4 m. The Charleston Harbor estuary is dominated by tidal salt marshes and is characterized as a low-energy system with limited wave action (Ellis et al., 2016). During the exposure period, salinity ranged from 17.3 to 26.9 ppt. Total monthly precipitation during the exposure period ranged from 4.1 cm (November 2017) to 21.9 cm (July 2017) (National Weather Service, 2018). Average monthly atmospheric temperature ranged from 7.4 °C (January 2018) to 27.8 °C (July 2017) (National Weather Service, 2018), and water temperature ranged from 10.0 °C (January 2018) to 31.4 °C (July 2017) (National Oceanic and Atmospheric Administration, 2018a). The number of daylight hours ranged from 10 h (December 2017) to 14 h and 19 min (June 2017) (National Oceanic and Atmospheric Administration, 2018b).
Following removal from the field, four replicate strips for each plastic type at each sampling time (except for MB at 32 weeks, which none remained) were examined for biofilm development and weighed. These same four replicate strips were then used to quantify surface area, fragmentation, and UV transmittance. One replicate strip per exposure period was used for Fourier Transformed Infrared Spectroscopy (FTIR) analysis and, at 0 and 32-weeks, scanning electron microscopy (SEM). The two remaining strips were archived. To measure weight, dried plastic strips (with biofilm) were weighed using a Mettler-Toledo PB153-S Balance. To measure surface area, plastic strips were mounted to a camera, photographed, and the digital images were examined using ImageJ software (Rasband, 2015). Following these measurements, the biofilm was removed from one side of these strips, and the strips were analyzed for UV transmittance.
To determine the transmittance of UV radiation and visible light through the plastic strips together with any biofilm that had developed on the upper side of the strip, a Macam Photometrics Model UV-203 IP-67 Radiometer was used to measure UV-A (332–406 nm), UV-B (292–330 nm) and visible light (400–710 nm). Plastic strips were individually placed directly on top of the appropriate UV or visible light sensor with the biofilm cleaned side of the strip facing toward the sensor. Measurements were made outdoors on The Citadel campus in downtown Charleston, SC, USA using ambient light conditions on 3 April 2018 between 1300 and 1500 EDT. Conditions were mostly sunny with <10% cloud cover. During this period, ambient irradiance levels were as follows: UV-A = 54.8 ± 3.0 W/m2, UV-B = 2555.6 ± 225.8 mW/m2, and visible = 444.0 ± 31.7 W/m2.
To determine fragmentation, plastic strips (prior to being cleaned of biofilm) were subject to a laboratory fragmentation test. Strips were individually placed in a 4 L amber jug filled with 1 L filtered brackish seawater (salinity = 22.6 ± 4.6‰) and gently rolled on a Wheaton Roller Culture Apparatus at 3 rpm for 6 h. Following 6 h, the plastic strip was removed, and the seawater poured through 500, 150, and 63 μm sieves. Particles retained on sieves were enumerated using a dissecting scope. These data were then normalized to the number of particles produced per mm3 for each plastic strip.
A number of measures were taken to reduce microplastic contamination for the laboratory fragmentation test. For example, stainless steel sieves and glass jars were used throughout the procedure. In addition, white cotton laboratory coats and nitrile gloves were worn in the laboratory when processing samples. Microplastic contamination within the laboratory was also quantified using procedural blanks. Procedural blanks were processed in the same manner as samples except they did not contain plastic strips. Laboratory procedural blanks (n = 16) contained an average of 2.1 ± 2.4 microplastic particles/blank. Reported data were not blank-corrected.
To determine the extent of changes in surface functional groups over time, attenuated total reflection FTIR was used on one strip per plastic type at each sampling time. The spectra were collected using a Thermo Scientific Nicolet 6700 FTIR with a Pike Technologies GladiATR™ accessory, with the measurement range set to 400–4000 cm−1.
For SEM, preparations from control plastic strips and those collected at 32 weeks post-deployment were coated with approximately 1.5 nm of platinum using a Denton Sputter-Edge Coater. Samples were then examined on a JEOL 5600LV SEM. For MB, samples collected at 16 weeks post-deployment were analyzed.
Data on effects of exposure period on surface area, weight, % UV or visible light transmittance, and the amount of microplastic particles emitted for each plastic type were compared using a repeated measures ANOVA using the statistical software R version 3.3.3. Differences between plastic types and exposure times for weight, surface area, UV and visible light transmittance, and particle amount were determined using least square mean comparisons with a Bonferroni adjustment. Percent UV/Visible light transmission data were arcsine square root transformed, surface area was log10 transformed, and weight and particle abundance were log10 (x + 1) transformed to meet the assumptions of normality and homogeneity of variance.
3. Results
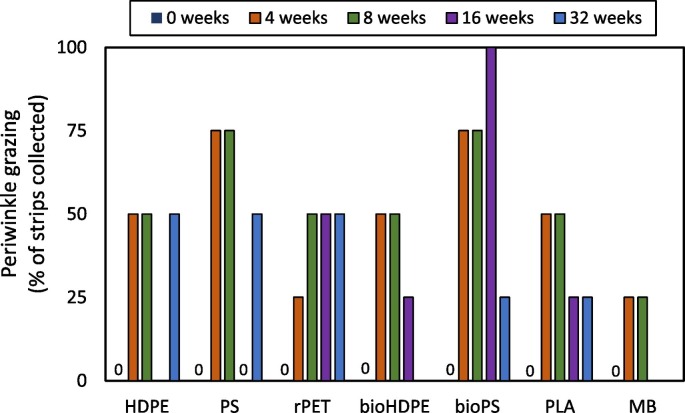
Of the 203 plastic strips initially deployed, 183 were successfully collected. The loss of 20 strips resulted in a reduction in the number of strips available for collection at 32 weeks. The number of strips available for each plastic type at 32 weeks was as follows: HDPE n = 6, PS n = 4, rPET n = 7, bioPS n = 6, bioHDPE n = 6, PLA n = 7, MB n = 0. Following one week of deployment, biofilm was observed covering the upper side of all strips, and marsh periwinkles (Littoraria irrorata) were observed grazing on strips of all plastic types. Both observations continued throughout the duration of the study. Periwinkle trails were observed on 41.7% of the collected strips throughout the study, although this ranged from 0 to 100% depending on the plastic type and collection time (Fig. 2 ).
Fig. 2.
Percentage of collected plastic strips showing evidence of periwinkle grazing over the 32-week exposure in a salt marsh habitat. HDPE = high density polyethylene. PS = extruded polystyrene. rPET = recycled polyethylene terephthalate. bioHDPE = biodegradable HDPE. bioPS = biodegradable extruded PS. PLA = polylactic acid. MB = Mater-Bi®. No strips were available at 32 weeks for MB.
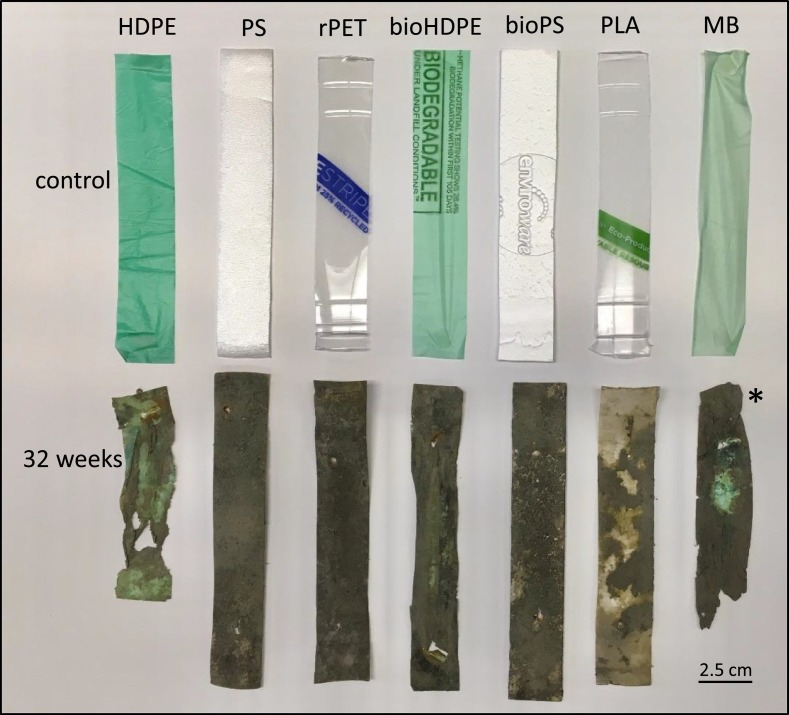
During the 32-week exposure, visual inspection revealed that the MB and HDPE strips, both from single use bags, began to shrivel and became increasingly brittle beginning at 8 and 16 weeks, respectively. At 16 and 32 weeks for the MB and HDPE strips, respectively, several of the strips had broken apart (Fig. 3 ). For all of the other plastic types, there was no visible evidence of shriveling or large-scale fragmentation during the 32-week exposure.
Fig. 3.
Photograph of control and post-deployment plastic strips. Post-deployment strips were collected after 32 weeks of exposure in salt marsh except for MB, which was collected following 16 weeks of exposure. HDPE = high density polyethylene. PS = extruded polystyrene. rPET = recycled polyethylene terephthalate. bioHDPE = biodegradable HDPE. bioPS = biodegradable extruded PS. PLA = polylactic acid. MB = Mater-Bi®. * indicates 16-week MB sample.
A repeated measures ANOVA comparing surface area produced a significant model for exposure time (p < 0.0001) but not for plastic type (p = 0.44). Surface area of the plastic strips was significantly decreased relative to control strips at 16 and 32 weeks (Fig. 4A). The largest decreases were evident in the HDPE and MB strips. For HDPE, surface area decreased 34.1% and 66.5% at 16 and 32 weeks, respectively. For MB, significant decreases in surface area were first evident at 8 weeks. Relative to control strips, MB strips at 8 and 16 weeks had decreased 52.6% and 56.3%.
Fig. 4.
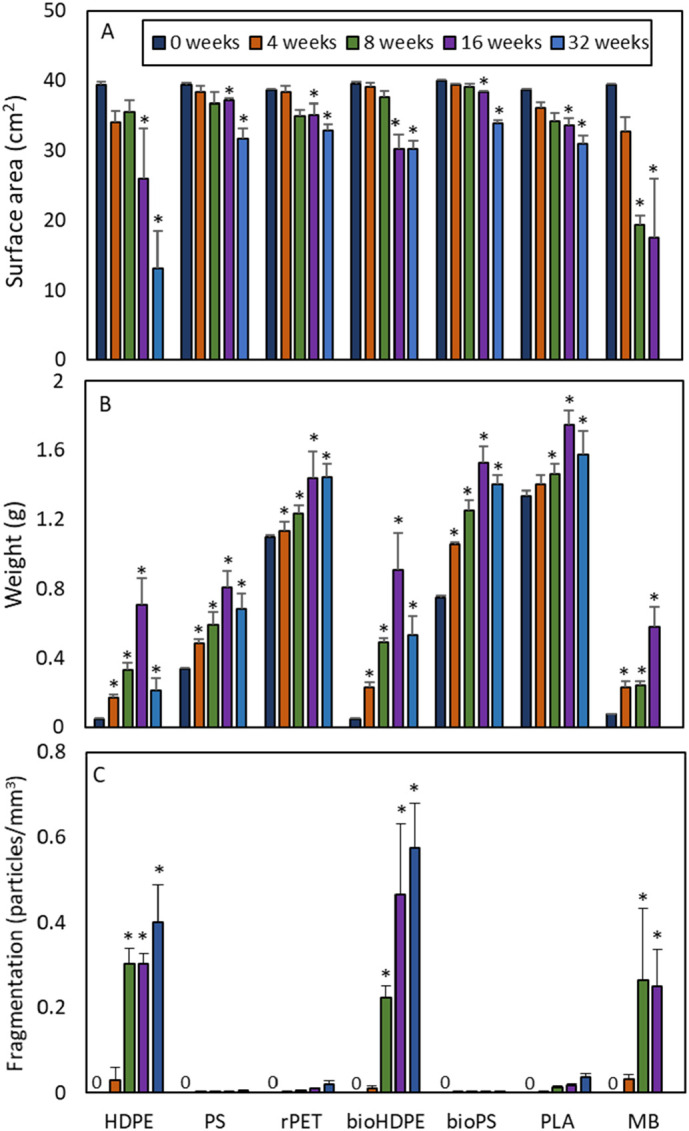
Surface area of strips (A), weight of strips with biofilm (B), and number of microplastic particles produced in the laboratory fragmentation test (C) over a 32-week exposure in a salt marsh habitat. For each plastic type, significant differences (p ≤ 0.05) relative to the week 0 strips (control) are denoted by an *. No microplastic particles were produced by the week 0 (control) strips for all plastic types. Mean ± SD. HDPE = high density polyethylene. PS = extruded polystyrene. rPET = recycled polyethylene terephthalate. bioHDPE = biodegradable HDPE. bioPS = biodegradable extruded PS. PLA = polylactic acid. MB = Mater-Bi®. Sample sizes between 0 and 16 weeks were as follows: surface area (n = 4), weight (n = 7), and fragmentation (n = 4). At 32 weeks, samples sizes were reduced due to loss of some strips as follows: surface area and fragmentation (n = 3 for PS), weight (n = 6 for HDPE, bioPS, and bioHDPE). No strips were available to test at 32 weeks for MB.
In general, the average weight of the strips from all plastic types progressively increased over time as a result of the accretion of biofilm (Fig. 4B). A repeated measures ANOVA comparing weight across all exposure times and plastic types was significant (p < 0.0001 for both). Significantly increased weight relative to the control strips was first evident at 4 weeks for all plastic types except PLA, which had a significantly increased weight beginning at 8 weeks (Fig. 4B). The weight of HDPE and bioHDPE strips, both of which were from single use plastic bags, significantly decreased between 16 and 32 weeks. The decreases in weight between these two exposure periods were 69.7% and 41.1% for HDPE and bioHDPE, respectively.
The production of microplastic particles from field-collected strips in the laboratory fragmentation test was first evident following 4 weeks of exposure for all plastic types and increased throughout the experiment (Fig. 4C). A repeated measures ANOVA comparing particle production across all exposure times and plastic types was significant (p < 0.0001 for each). These data were driven by the significant increase in particle production at 8, 16, and 32 weeks by the HDPE, bioHDPE, and MB (8 and 16 weeks only) strips, relative to controls. At 4 weeks, the average number of particles produced per mm3 by each strip ranged from 6 × 10−5 particles/mm3 for PS to 0.03 particles/mm3 for HDPE and MB. At 32 weeks, the average number of particles ranged from 0.003 particles/mm3 for bioPS to 0.6 particles/mm3 for bioHDPE.
Transmittance of UV-A, UV-B, and visible light through the strips of all polymers was negatively related to exposure period as a result of the accreting biofilm. The repeated measures ANOVA for all three types of transmittance across all exposure times and plastic types was significant (p < 0.0001 for each) (Fig. 5A–C). For UV-A and visible light, PLA and rPET allowed for the highest transmission across all exposure periods. For UV-B, PLA allowed for the highest transmission. In all three cases, transmission was significantly lowest in bioPS. After only 4 weeks of exposure, the relative decrease in transmittance of UV-A, UV-B, and visible light across all plastic types compared to controls ranged between 40.0% to 95.4%, 48.4% to 85.7%, and 31.0% to 54.2%, respectively. After 32 weeks, transmittance of UV-A, UV-B, and visible light across all plastic types was reduced from between 88.3% to 99.4%, 65.6% to 97.0%, and 69.1% to 97.8%, respectively, relative to that of the controls.
Fig. 5.
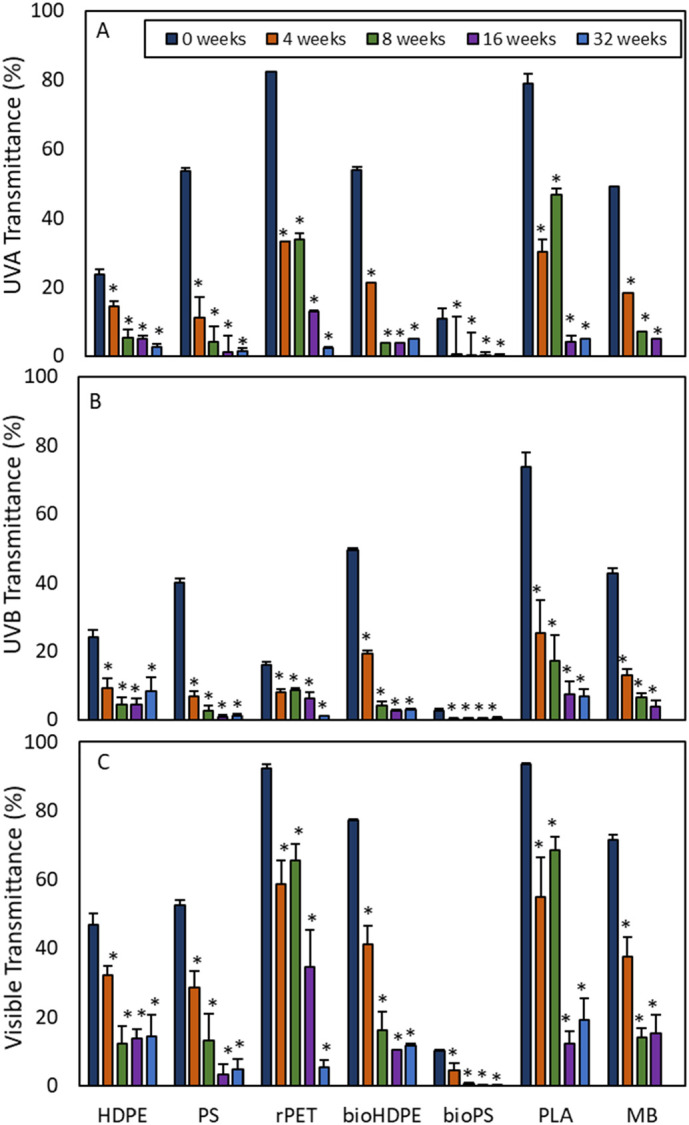
Percent transmittance of UV-A (A), UV-B (B) and visible light (C) through plastic strips over a 32-week exposure in a salt marsh habitat. Percent transmittance was significantly reduced for all plastic strips at weeks 4, 8, 16, and 32 relative to week 0 strips (control). Mean ± SD. HDPE = high density polyethylene. PS = extruded polystyrene. rPET = recycled polyethylene terephthalate. bioHDPE = biodegradable HDPE. bioPS = biodegradable extruded PS. PLA = polylactic acid. MB = Mater-Bi®. Sample sizes between 0 and 16 weeks were n = 4. At 32 weeks, samples sizes were reduced to n = 3 for PS due to loss of some strips. No strips were available to test at 32 weeks for MB.
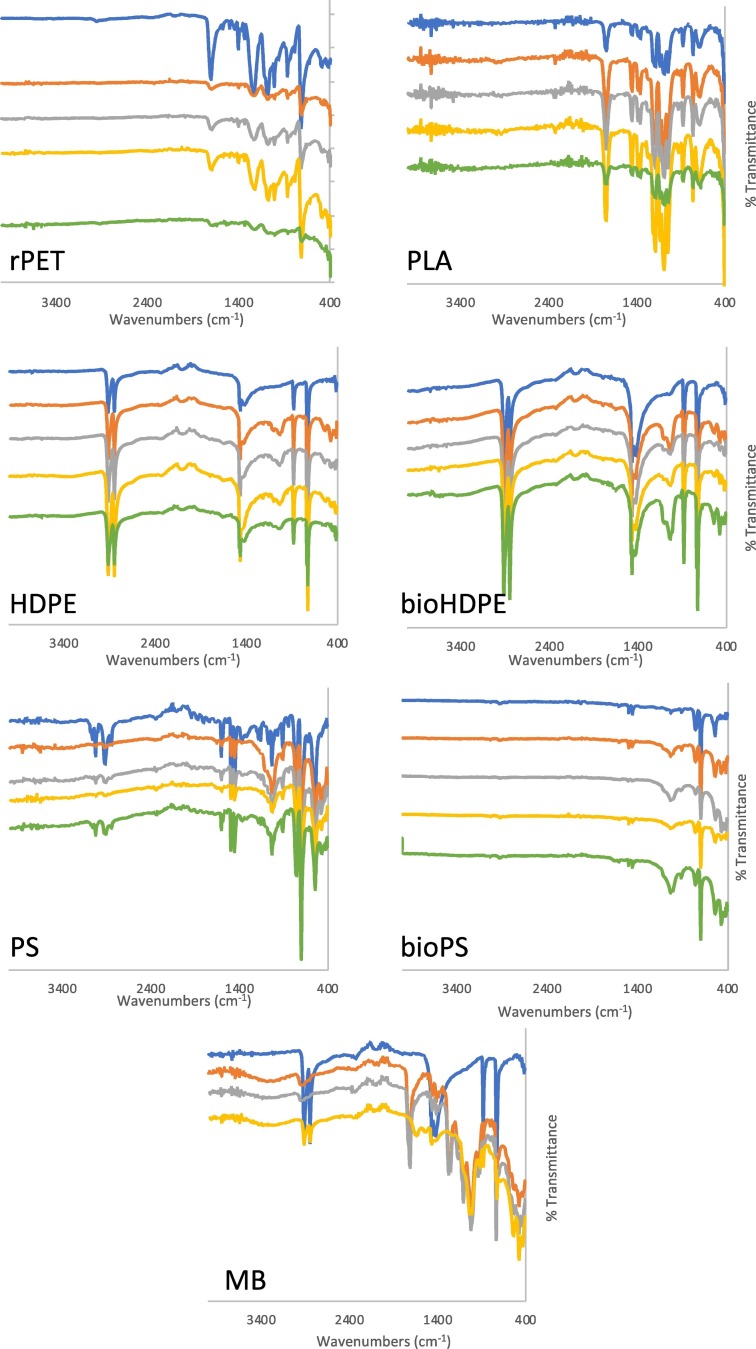
Infrared spectra were obtained for each polymer at 0 (control), 4, 8, 16, and 32 weeks (Fig. 6 ). For the rPET sample, no new peaks appeared in the IR spectra over the course of environmental exposure, but transmittance across all peaks decreased, indicating degradation. No differences in degradation were observed between HDPE and bioHDPE samples. For both HDPE and bioHDPE, small peaks at 1030 cm−1 (C—O stretch) and 1650 cm−1 (C O stretch) emerged in the IR spectra over the exposure period, indicating the presence of oxygen functional groups that formed as a result of minor surface degradation. For PS and bioPS samples, the expected IR peaks for aromatic groups (~3000 and 1450 cm−1) were observed and remained mostly unchanged, except for a slight decrease in intensity throughout the 32-week exposure period. The C—O stretch peak (1027 cm−1) became more prominent with increasing environmental exposure, particularly for the bioPS sample. Of note, a ~ 1500% increase in peak area was observed for bioPS, while this peak increased by only ~200% for PS.
Fig. 6.
FT-IR spectra of each plastic sample at 4 weeks (orange), 8 weeks (gray), 16 weeks (yellow), and 32 weeks (green) compared to controls (blue). (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
Surprisingly, IR spectra indicated the PLA samples underwent the least amount of degradation during exposure, with almost no change observed between the control and 32-week sample. The MB sample experienced the most significant amount of degradation, with complete deterioration and sample loss occurring between the 16-and 32-week time points. Significant changes were also noted in the IR spectra. While strong C—H peaks (2913, 2845, 1460 cm−1) were present initially in the control sample, the intensity of these peaks decreased by 16 weeks. Additionally, similar to other samples, a large C—O peak (1037 cm−1) emerged in the spectra.
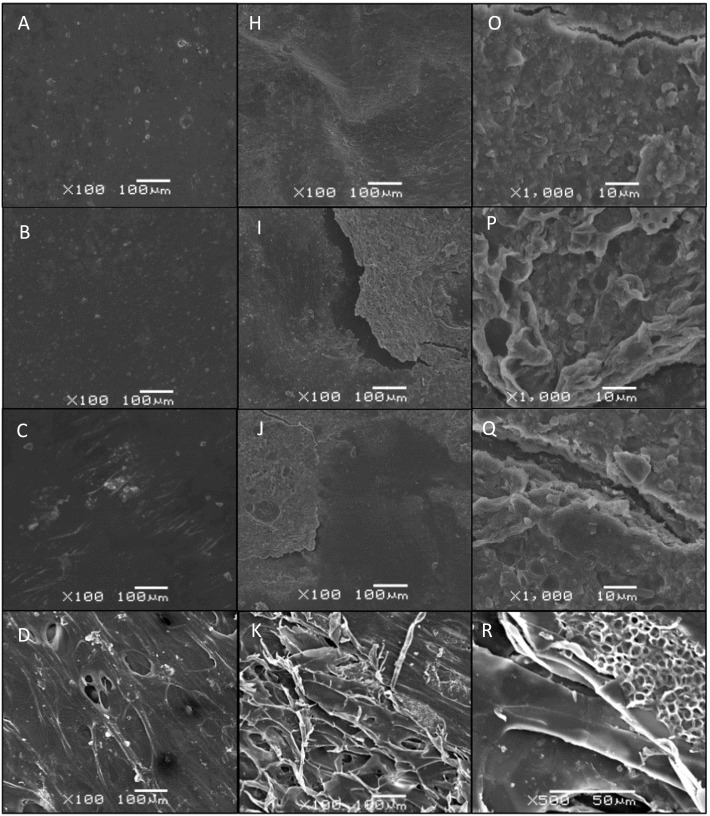
SEM examination of the surface of the control strips revealed that, in general, the plastics chosen for this study had a largely featureless and smooth surface (Fig. 7A–G). The only exception was the control bioPS strips which exhibited a sponge-like surface with numerous cavities and pores (Fig. 7D). Following 32 weeks in the salt marsh, the surface of the HDPE and bioHDPE strips exhibited moderate degradation characterized by pitting, microcracks, and occasional areas where it appeared that the overlying surface layers had been removed (Fig. 7H and O, and J and Q, respectively). Qualitatively, there appeared to be no difference in the extent of degradation between HDPE and bioHDPE (Fig. 7O and Q, respectively). The surface of the 32-week PS strips similarly exhibited moderate surface degradation characterized by cracking, pitting and erosion (Fig. 7I and P). By contrast, the surface of the 32-week bioPS strips exhibited extensive surface degradation as evidenced by the presence of numerous holes across the surface extending deep into the material along with widespread areas where overlying surface layers had been lost (Fig. 7K and R). The surface of the 32-week rPET strips exhibited areas where extensive pitting and surface delamination were evident; however, unlike the HDPE and PS strips, there were no microcracks or large holes on the surface (Fig. 7L and S). The surface of the 32-week PLA and the 16-week MB strips exhibited limited areas of minor degradation, and there was no evidence of microcracks, pitting, or surface delamination (Fig. 7M and T, and Fig. 7N and U, respectively).
Fig. 7.
SEM images of the surface topography of 0-week (control) and post-deployment plastic strips. Post-deployment strips for SEM were collected after 32 weeks of exposure in salt marsh except for MB, which was collected following 16 weeks of exposure. Surface topography for the 0-week (control) strips was nearly featureless for most plastic types, including HDPE (A), PS (B), bioHDPE (C), rPET (E), PLA (F), and MB (G). The only exception was bioPS (D), which exhibited numerous ovoid depressions. Surface topography of post-deployment plastic strips following removal of biofilm exhibited minor to moderate degraded surfaces, including HDPE (H and O), bioHDPE (J and Q), PS (I and P), bioPS (K and R), and rPET (L and S). Post-deployed PLA (M and T) and MB (N and U) strips both exhibited limited surface erosion. HDPE = high density polyethylene. PS = extruded polystyrene. rPET = recycled polyethylene terephthalate. bioHDPE = biodegradable HPDE. bioPS = biodegradable extruded PS. PLA = polylactic acid. MB = Mater-Bi®.
4. Discussion
While the growing popularity of bio-based and biodegradable plastics is based, in part, upon the assumption by consumers that they degrade faster than conventional oil-based plastics, there is currently little scientific data available to support or refute this notion. Here, we report that, at least in a salt marsh habitat, the degradation rates of these plastics vary widely. Evidence suggests that the bag composed of MB and the plate composed of bioPS degraded more than their conventional counterparts, while the cup composed of PLA degraded slower than any of the tested products. Of note was the observation that all tested plastics produced microplastic particles beginning in as little as 4 weeks post-deployment.
Direct comparison between biodegradable and bio-based plastics and their conventional counterparts among the various products proved useful in interpreting the results of the current study. For single use plastic bags, the most degradation was exhibited by strips from the MB bags, which began disintegrating by 16 weeks and none remained attached to the experimental apparatus at 32 weeks. Relative to the other single use bags, the MB bags were >60% thicker, but had similar levels of UV transmittance. MB bags were produced from a bio-based plastic called Mater-Bi®, which is manufactured completely from renewable raw materials of agricultural origin and from non-genetically modified starch (Novamont, 2019). In the current study, the MB strips were extensively fragmented and degraded following 16 weeks (113 days) in a salt marsh. Other studies conducted under controlled laboratory conditions using seawater have found considerably slower degradation rates for Mater-Bi®. For example, Müller et al. (2012) found negligible degradation (4.5% mass loss) of Mater-Bi® bags in seawater following 49 days, and only 50% degradation following 389 days. Tosin et al. (2012) found that Mater-Bi® degraded 68.9% following 236 days when exposed to seawater in the laboratory, but when buried in wet sand in aquaria, total degradation was achieved within 9 months. Collectively, these results suggest that laboratory studies may underestimate the degradation rates of Mater-Bi® in coastal waters, and they underscore the value of field studies, such as that performed here, since plastics in these studies are exposed to the full range of environmental factors that enhance degradation (e.g. sunlight, heat, physical abrasion, wave action, diverse microbial communities, etc.).
The degradation of strips from the MB bags in the current study was fundamentally different than that of the other plastics. These strips significantly shriveled over time, an observation borne out by the surface area analyses. Furthermore, SEM analysis suggested that, unlike many of the other plastics in the current study, as well as conventional plastics in a previous study (Weinstein et al., 2016), degradation did not involve surface delamination. Instead, evidence suggested that degradation was the result of embrittlement. Degradation-induced embrittlement as a result of linear polymers undergoing random chain scission resulting in a transition of the material from a ductile to a brittle state has been previously described for polyethylene films at temperatures up to 90 °C (Fayolle et al., 2007). Andrady (2011) has also suggested that embrittlement is a common pathway by which extensively degraded plastics emit microplastic particles. Embrittlement of extensively eroded plastics, especially those made from polyethylene, has been documented on the beaches in Kauai (Hawaii) (Cooper and Corcoran, 2010). Certainly, further research on the underlying mechanisms by which MB degrades in coastal habitats is warranted.
Strips from the HDPE plastic bags were more degraded at 32 weeks than those from the bioHDPE bags. Similar to the MB strips, the HDPE strips increasingly shriveled over time becoming more brittle, and by 32 weeks, most had visibly fragmented. Similar changes were not evident for the bioHDPE strips, which generally maintained their shape and integrity throughout the study. At their surface, strips of both HDPE and bioHDPE exhibited minor degradation as evidenced by SEM and FT-IR, suggesting that the observed differences between these two plastics may not have been restricted to processes occurring at the surface. Previous studies have shown that the addition of plant material to synthetic plastics during the manufacturing process can dramatically alter the physical properties of a plastic (reviewed by Mir et al., 2018). For example, the addition of grapefruit seed extract into a composite plastic film increased the moisture content from 16.43 to 35.75% (Kanmani and Rhim, 2014). The authors attribute the increase in moisture content to the hydrophilic character of the extract which may have increased the availability of hydroxyl groups within the plastic polymer to bind to water molecules. Functionally, moisture can also influence polymer degradation by providing water for the growth and multiplication of microbes, thus accelerating polymer degradation through microbial action (Ho et al., 1999). Increased moisture content of plastic films has also been noted following the addition of betel leaf extract (Nouri and Nafchi, 2014). Although the nature, concentration, and mechanism of action of the algal additive in the bioHDPE bags used in this study is proprietary, the evidence provided here suggests that this additive may have increased the moisture content of the plastic thereby preventing shrinkage, while concurrently not necessarily imparting any enhanced degradability to the material over the course of this 32-week study. It is possible that if this experiment had been conducted for a longer period, the degradation of the bioHDPE may have occurred at a faster rate than that of HDPE.
For the disposable plates, more degradation was exhibited by strips from the bioPS plates as evidenced by both SEM and FT-IR. Degradation of these bioPS strips occurred despite the significantly lower transmittance of UV radiation. Transmittance of UV-A and UV-B were 80.2% and 93.0% lower, respectively, in control bioPS strips relative to control PS strips. The bioPS strips also had a significantly greater weight increase (105.4% greater) at 32-weeks relative to control PS strips. According to the manufacturer for the bioPS plates, these single use plates are manufactured using green2technology™ to ensure maximum recycled content, are produced with CO2 rather than pentane, and have an active organic catalyst added to the polymer during manufacturing to accelerate the degradation rate (D&W Fine Pack, 2016).
Considerable research has been performed on enhancing the degradation of PS through the addition of catalysts (reviewed by Singh and Sharma, 2008). The challenge in making PS eco-friendlier has been the decreased responsiveness that virgin PS has to degradation mediated by UV radiation due to the presence of phenyl rings in its repeating units (Bandyopadhyay and Basak, 2007). The addition of compounds that act as photo-catalysts by transferring absorbed energy from UV radiation to different bonds in the PS polymer matrix have been found to be effective in enhancing degradation (Bandyopadhyay and Basak, 2007). For example, laboratory studies have demonstrated that the addition of ZnO to PS under UV radiation exposure resulted in a net weight loss of 16% in only 2 h, and with the addition of the photoreactive dye, Eosin Y, further increased weight loss by 18% (Bandyopadhyay and Basak, 2007). The addition of acid catalysts, such as zeolites, has also been reported in the literature for use in transforming polymers into hydrocarbons by favoring hydrogen transfer reactions (Singh and Sharma, 2008). They have also been found to lower the apparent activation reaction for decomposition reactions (Garforth et al., 1997). The relative weight increase of the bioPS strips may have been the result of an increased water content associated with the addition of this organic catalyst, as happens with additives derived from plants (as described above). Although the nature of the active organic catalyst in the bioPS bags is proprietary, the evidence provided here suggests that its presence, along with the other features of green2technology™, were effective at enhancing the degradability to the material over the course of this 32-week study. Also contributing to the higher rate of degradation of the bioPS strips in this study was their darker color (wheat) relative to that of the PS strips (white), which may have produced higher temperatures at their surface. The results from this study support the claim by the manufacturer that the degradation rate of this type of eco-friendly PS is accelerated relative to that of conventional PS.
For the two plastic cups, the strips of rPET demonstrated greater degradation than those composed of PLA as evidenced by both SEM and FT-IR. However, relative to the other plastic products examined in this study, rPET strips degraded slowly. Studies have indicated that the degradation of PET in the marine environment is relatively slow. For example, Ioakeimidis et al. (2016) found PET remains structurally intact in the submarine environment for approximately fifteen years with decreases in native functional groups occurring thereafter. Although one of the limitations of the present study is that we did not directly compare recycled and virgin PET strips, evidence from the literature suggests that recycled PET may degrade faster than virgin PET. Oromiehie and Mamizadeh (2004) reported that the recycling process for PET results in a decrease in average molecular weight and mechanical properties of the end-product due to the hydrolytic chain excision and thermomechanical degradation that occurs during processing. Likewise, Torres et al. (2000) found that recycled PET was more brittle than virgin PET and attributed it to the presence of contaminants and residual moisture associated with the recycling melt process. Collectively, these observations suggest that the decreased mechanical properties associated with recycled PET would result in a faster degradation rate in the environment than virgin PET. Future studies directly comparing the degradation rates of recycled versus virgin PET are warranted.
The least amount of degradation in this study was exhibited by the strips from the PLA cups, which following 32 weeks of exposure, only exhibited limited surface degradation and almost no changes to spectra produced by FT-IR. The relatively slow degradation of PLA has previously been observed in both home compost, where negligible degradation was observed after 180 days (<5% mass loss) (Song et al., 2009), and in soil burial tests, where no degradation was observed after 6 weeks (Ohkita and Lee, 2006). However, PLA can be degraded relatively quickly in an industrial composter where it is hydrolyzed into smaller molecules after 45–60 days at 50–60 °C (Tokiwa and Calabia, 2006). This is consistent with the notion that PLA degradation is the result of a two-step process: temperature-and humidity-dependent hydrolysis followed by biodegradation (Lunt, 1998; Ho et al., 1999; Rujnić-Sokele and Pilipović, 2017). Salinity may have also contributed to the slow rate of degradation observed in the current study. Deroiné et al., 2014 found decreased rates of degradation of PLA in seawater compared to distilled water. Of note in the current study was the relatively slow development of a biofilm on the PLA strips, as indicated by both weight and the relatively high transmittance of UV radiation through the strips. This suggests that, unlike other plastic polymers, UV radiation does not play an important role in PLA degradation.
It is worth noting that the weathering of plastics in intertidal habitats, such as salt marshes, would be expected to occur at faster rates than in other habitats. This is a consequence of exposure to UV radiation, temperature, and oxygen while the plastic is surficial at low tide, and exposure to mechanical forces associated with scouring particulate matter and turbulence while the plastic is submerged at high tide. In fact, evidence for the weathering of most plastics examined in this study was found in as little as 4 weeks of exposure. These first few weeks of exposure were marked by the least amount of biofilm, and consequently, the greatest amount of UV transmittance, which would have facilitated photo-oxidation reactions. The role of photo-oxidation in the degradation of plastics has been previously noted in a study by Biber et al. (2019), who found that the deterioration of polyethylene, PET, PS, and Biothene® was faster in direct sunlight compared to shade, and in air compared to water. These results corroborate those of an earlier study by Pegram and Andrady (1989), who found little evidence of weathering of polyethylene samples floating in seawater over an 8-week period, whereas those exposed to air nearly embrittled over the same period. In situ studies, such as the current study and those described above, are important in filling critical gaps in our knowledge regarding how plastic litter weathers under different environmental conditions, as well as understanding which habitats and conditions are conducive to microplastic formation.
In the current study, all tested plastics produced microplastic particles in as little as 4 weeks post-deployment. In contrast, microplastic particles were not observed until 8 weeks in a previous plastic degradation study conducted at this same site (Weinstein et al., 2016). Since both studies were initiated at about the same time of year (25 May 2017 vs. 13 June 2013), these results suggest that the rate at which plastic degradation begins could be influenced by year-to-year variability in environmental factors (e.g. cloud cover, temperature, tidal heights, storms, etc.). Further research on how variability in these factors, including those associated with climate change, influence plastic degradation is certainly warranted. It should also be noted that although we did not confirm that the microplastic particles generated in the fragmentation test matched the plastic composition of the strips in this study, in a previous study (Weinstein et al., 2016), we found the average correlation in FTIR spectra between particles and strips was >90%.
The highest amounts of microplastic particles were produced by plastic strips from the single use bags, suggesting that these bags may be producing disproportionately more microplastics in the first 6 months as litter than other types of plastic. Plastic bags account for a large proportion of plastic debris in coastal regions. The Ocean Conservancy reported that single use bags were the seventh most common type of debris collected during their annual International Coastal Cleanup in 2018 (Ocean Conservancy, 2019). In the intertidal areas of Charleston Harbor, Wertz (2015) estimated that there were nearly 22,000 single use grocery bags weighing close to 1.1 metric tonnes, representing 15.7% of the total plastic debris. Apart from the well documented effects that single use bags have on marine life, including sea turtles, this research suggests that the early stages of degradation of these bags present additional risks associated with the production of microplastic particles. Further research on this topic is warranted, especially considering the recent easing of single use plastic bag bans associated with the COVID-19 pandemic.
5. Conclusion
The results of this study demonstrated that bio-based and biodegradable plastics exhibit a wide range of degradation rates in a salt marsh. Based on those tested, it appears that the shopper bags composed of Mater-Bi® and PS plates made with green2technology™ degraded at rates faster than conventional, petroleum-derived plastics. In contrast, cups made with the plant-based polymer PLA had the slowest rate of degradation of any plastic tested. Increased consumer use of this plastic, in combination with the waste disposal issues that plague all single use plastics, could potentially contribute to an increased presence of plastic litter in the environment. Microplastic particles were produced by eco-friendly and conventional plastics alike in this study in as little as 4 weeks. The potential adverse impacts resulting from the ingestion of microplastic particles by marine organisms, especially invertebrates, has been documented in numerous laboratory studies. One question that remains to be established is whether microplastic particles from biodegradable or bio-based plastics persist as long as those from conventional plastics. From a broader perspective, we agree with Napper and Thompson (2019) in that it also remains to be established whether the fragmentation of any plastic litter into microplastic particles represents an even greater environmental risk than the original intact plastic item itself.
CRediT authorship contribution statement
John E. Weinstein: Conceptualization, Funding acquisition, Writing - original draft. Jack L. Dekle: Methodology, Investigation. Rachel R. Leads: Supervision, Investigation, Formal analysis. Rebecca A. Hunter: Formal analysis, Investigation.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgment
The present study was supported in part by the South Carolina Sea Grant Consortium with National Oceanic and Atmospheric Administration Award N468 (R/ER-46) and the state of South Carolina. The authors acknowledge the participation of S. Kell and J. Upchurch in various aspects of this study. The authors would also like to acknowledge S. Morton with the NOAA/NOS Marine Biotoxins Program in Charleston, SC for providing time and assistance in the scanning electron microscopy portion of this study.
Disclaimer
The statements, findings, conclusions, and recommendations are those of the authors and do not necessarily reflect the views of the South Carolina Sea Grant Consortium or the National Oceanic and Atmospheric Administration.
References
- Andrady A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011;62:1596–1605. doi: 10.1016/j.marpolbul.2011.05.030. [DOI] [PubMed] [Google Scholar]
- Andrady A.L., Pegram J.E., Song Y. Studies on enhanced degradable plastics. II. Weathering of enhanced photodegradable polyethylenes under marine and freshwater floating exposure. J. Environ. Polym. Degrad. 1993;1:117–126. [Google Scholar]
- Auta H.S., Emenike C.U., Fauziah S.H. Distribution and importance of microplastics in the marine environment: a review of the sources, fate, effects, and potential solutions. Environ. Int. 2017;102:165–176. doi: 10.1016/j.envint.2017.02.013. [DOI] [PubMed] [Google Scholar]
- Avio C.G., Gorbi S., Regoli F. Plastics and microplastics in the oceans: from emerging pollutants to emerged threat. Mar. Environ. Res. 2017;128:2–11. doi: 10.1016/j.marenvres.2016.05.012. [DOI] [PubMed] [Google Scholar]
- Bandyopadhyay A., Basak G.C. Studies on photocatalytic degradation of polystyrene. Mater. Sci. Technol. 2007;23(3):307–314. [Google Scholar]
- Barnes D.K., Galgani F., Thompson R.C., Barlaz M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009;364(1526):1985–1998. doi: 10.1098/rstb.2008.0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biber N.F.A., Foggo A., Thompson R.C. Characterising the deterioration of different plastics in air and seawater. Mar. Pollut. Bull. 2019;141:595–602. doi: 10.1016/j.marpolbul.2019.02.068. [DOI] [PubMed] [Google Scholar]
- BioBag USA BioBag product information. 2016. http://biobagusa.com/products/commercial/shopping-produce-bags/ Retrieved from BioBag USA website.
- Browne M.A., Galloway T.S., Thompson R.C. Spatial patterns of plastic debris along estuarine shorelines. Environ. Sci. Technol. 2010;44:3404–3409. doi: 10.1021/es903784e. [DOI] [PubMed] [Google Scholar]
- Cooper D.A., Corcoran P.L. Effects of mechanical and chemical processes on the degradation of plastic beach debris on the island of Kauai, Hawaii. Mar. Pollut. Bull. 2010;60(5):650–654. doi: 10.1016/j.marpolbul.2009.12.026. [DOI] [PubMed] [Google Scholar]
- D&W Fine Pack Enviroware® product information. 2016. https://www.dwfinepack.com/wp-content/uploads/Web-Enviroware-8-28-2018.pdf Retrieved from D&W Fine Pack website.
- Deroiné M., Le Duigou A., Corre Y.-M., Le Gac P.-Y., Davies P., César G., Bruzaud S. Accelerated ageing of polylactide in aqueous environments: comparative study between distilled water and seawater. Polym. Degrad. Stab. 2014;108:319–329. [Google Scholar]
- Eco-Products Product information for Ingeo™. 2019. https://www.ecoproducts.com/ingeo_brand_pla.html Retrieved from Eco-Products website.
- Ellis K.K., Callahan T., Greenfield D., Sanger D., Robinson J. 2016. Hydrologic Assessments of Tidal Creeks to Inform Nutrient Management Recommendations. [Google Scholar]
- European Bioplastics Bioplastics market data 2018. 2018. https://www.european-bioplastics.org/wp-content/uploads/2016/02/Report_Bioplastics-Market-Data_2018.pdf Retrieved from European Bioplastics website.
- Fayolle B.C., Colin X., Audouin L., Verdu J. Mechanism of degradation induced embrittlement in polyethylene. Polym. Degrad. Stab. 2007;92(2):231–238. [Google Scholar]
- Garforth A., Fiddy S., Lin Y.H., Ghanbari-Siakhali A., Sharratt P.N., Dwyer J. Catalytic degradation of high density polyethylene: an evaluation of mesoporous and microporous catalysts using thermal analysis. Thermochim. Acta. 1997;294(1):65–69. [Google Scholar]
- Gray A.D., Weinstein J.E. Size-and shape-dependent effects of microplastic particles on adult daggerblade grass shrimp (Palaemonetes pugio) Environ. Toxicol. Chem. 2017;36(11):3074–3080. doi: 10.1002/etc.3881. [DOI] [PubMed] [Google Scholar]
- Gray A.D., Wertz H., Leads R.R., Weinstein J.E. Microplastic in two South Carolina Estuaries: occurrence, distribution, and composition. Mar. Pollut. Bull. 2018;128:223–233. doi: 10.1016/j.marpolbul.2018.01.030. [DOI] [PubMed] [Google Scholar]
- Ho K.L.G., Pometto A.L., Hinz P.N. Effects of temperature and relative humidity on polylactic acid plastic degradation. J. Environ. Polym. Degrad. 1999;7(2):83–92. [Google Scholar]
- Holland A.F., Sanger D.M., Gawle C.P., Lerberg S.B., Santiago M.S., Riekerk G.H.M., Zimmerman L.E., Scott G.I. Linkages between tidal creek ecosystems and the landscape and demographic attributes of their watersheds. J. Exp. Mar. Biol. Ecol. 2004;298:151–178. [Google Scholar]
- Ioakeimidis C., Fotopoulou K.N., Karapanagioti H.K., Geraga M., Zeri C., Papathanassiou E., Papatheodorou G. The degradation potential of PET bottles in the marine environment: an ATR-FTIR based approach. Sci. Rep. 2016;6 doi: 10.1038/srep23501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwata T. Biodegradable and bio-based polymers: future prospects of eco-friendly plastics. Angew. Chem. Int. Ed. 2015;54(11):3210–3215. doi: 10.1002/anie.201410770. [DOI] [PubMed] [Google Scholar]
- Kanmani P., Rhim J.W. Development and characterization of carrageenan/grapefruit seed extract composite films for active packaging. Int. J. Biol. Macromol. 2014;68:258–266. doi: 10.1016/j.ijbiomac.2014.05.011. [DOI] [PubMed] [Google Scholar]
- Kershaw P., Katsuhiko S., Lee S., Samseth J., Woodring D., Smith J. UNEP Year Book. 2011. Plastic debris in the ocean; pp. 20–33. [Google Scholar]
- Khare A., Deshmukh S. Studies toward producing eco-friendly plastics. J. Plast. Film Sheeting. 2006;22(3):193–211. [Google Scholar]
- Leads R.R., Burnett K.G., Weinstein J.E. The effect of microplastic ingestion on survival of the grass shrimp Palaemonetes pugio (Holthuis, 1949) challenged with Vibrio campbellii. Environ. Toxicol. Chem. 2019;38(10):2233–2242. doi: 10.1002/etc.4545. [DOI] [PubMed] [Google Scholar]
- Lunt J. Large-scale production, properties and commercial applications of polylactic acid polymers. Polym. Degrad. Stab. 1998;59(1–3):145–152. [Google Scholar]
- Mir S.A., Dar B.N., Wani A.A., Shah M.A. Effect of plant extracts on the techno-functional properties of biodegradable packaging films. Trends Food Sci. Technol. 2018;80:141–154. [Google Scholar]
- Müller C., Townsend K., Matschullat J. Experimental degradation of polymer shopping bags (standard and degradable plastic, and biodegradable) in the gastrointestinal fluids of sea turtles. Sci. Total Environ. 2012;416:464–467. doi: 10.1016/j.scitotenv.2011.10.069. [DOI] [PubMed] [Google Scholar]
- Napper I.E., Thompson R.C. Environmental deterioration of biodegradable, oxo-biodegradable, compostable, and conventional plastic carrier bags in the sea, soil, and open-air over a 3-year period. Environ. Sci. Technol. 2019;53:4775–4783. doi: 10.1021/acs.est.8b06984. [DOI] [PubMed] [Google Scholar]
- National Oceanic and Atmospheric Administration National Data Buoy Center. 2018. www.ndbc.noaa.gov/station_history.php?station=chts1 Retrieved from the National Oceanic and Atmospheric Administration website.
- National Oceanic and Atmospheric Administration Global Monitoring Laboratory. 2018. www.esrl.noaa.gov/gmd/grad/solcalc/ Retrieved from the National Oceanic and Atmospheric Administration website.
- National Weather Service Forecast Office NOAA online weather data. 2018. www.weather.gov/climate/xmacis.php?wfo=chs Retrieved from the National Weather Service Forecast Office website.
- Nouri L., Nafchi A.M. Antibacterial, mechanical, and barrier properties of sago starch film incorporated with betel leaves extract. Int. J. Biol. Macromol. 2014;66:254–259. doi: 10.1016/j.ijbiomac.2014.02.044. [DOI] [PubMed] [Google Scholar]
- Novamont Mater-Bi®. 2019. https://www.novamont.com/eng/mater-bi Retrieved from Novamont website:
- O’Brine T., Thompson R.C. Degradation of plastic carrier bags in the marine environment. Mar. Pollut. Bull. 2010;60(12):2279–2283. doi: 10.1016/j.marpolbul.2010.08.005. [DOI] [PubMed] [Google Scholar]
- Ocean Conservancy . The Ocean Conservancy; 2019. The Beach and Beyond, International Coastal Cleanup 2019 Report. [Google Scholar]
- Ohkita T., Lee S.H. Thermal degradation and biodegradability of poly (lactic acid)/corn starch biocomposites. J. Appl. Polym. Sci. 2006;100(4):3009–3017. [Google Scholar]
- Oromiehie A., Mamizadeh A. Recycling PET beverage bottles and improving properties. Polym. Int. 2004;53(6):728–732. [Google Scholar]
- Pegram J.E., Andrady A.L. Outdoor weathering of selected polymeric materials under marine exposure conditions. Polym. Degrad. Stab. 1989;26:333–345. [Google Scholar]
- Rasband W.S. U.S. National Institutes of Health; Bethesda, Maryland, USA: 2015. ImageJ.http://imagej.nih.gov/ij/ [Google Scholar]
- Rujnić-Sokele M., Pilipović A. Challenges and opportunities of biodegradable plastics: a mini review. Waste Manag. Res. 2017;35(2):132–140. doi: 10.1177/0734242X16683272. [DOI] [PubMed] [Google Scholar]
- Singh B., Sharma N. Mechanistic implications of plastic degradation. Polym. Degrad. Stab. 2008;93:561–584. [Google Scholar]
- Song J.H., Murphy R.J., Narayan R., Davies G.B.H. Biodegradable and compostable alternatives to conventional plastics. Philos. Trans. R. Soc. B Biol. Sci. 2009;364(1526):2127–2139. doi: 10.1098/rstb.2008.0289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokiwa Y., Calabia B.P. Biodegradability and biodegradation of poly (lactide) Appl. Microbiol. Biotechnol. 2006;72(2):244–251. doi: 10.1007/s00253-006-0488-1. [DOI] [PubMed] [Google Scholar]
- Torres N., Robin J.J., Boutevin B. Study of thermal and mechanical properties of virgin and recycled poly (ethylene terephthalate) before and after injection molding. Eur. Polym. J. 2000;36(10):2075–2080. [Google Scholar]
- Tosin M., Weber M., Siotto M., Lott C., Degli-Innocenti F. Laboratory test methods to determine the degradation of plastics in marine environmental conditions. Front. Microbiol. 2012;3:225. doi: 10.3389/fmicb.2012.00225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viehman S., Vander Pluym J.L., Schellinger J. Characterization of marine debris in North Carolina salt marshes. Mar. Pollut. Bull. 2011;62:2771–2779. doi: 10.1016/j.marpolbul.2011.09.010. [DOI] [PubMed] [Google Scholar]
- Watts A.J., Lewis C., Goodhead R.M., Beckett S.J., Moger J., Tyler C.R., Galloway T.S. Uptake and retention of microplastics by the shore crab Carcinus maenas. Environ. Sci. Technol. 2014;48(15):8823–8830. doi: 10.1021/es501090e. [DOI] [PubMed] [Google Scholar]
- WebstaurantStore Product information for Green Herc™ 1/6 size biodegradable plastic t-shirt bag. 2019. https://www.webstaurantstore.com/green-herc-1-6-size-biodegradable-plastic-t-shirt-bag-case/433NHTBIO.html Retrieved from WebstaurantStore website.
- Weinstein J.E., Crocker B.K., Gray A.D. From macroplastic to microplastic: degradation of high-density polyethylene, polypropylene, and polystyrene in a salt marsh habitat. Environ. Toxicol. Chem. 2016;35(7):1632–1640. doi: 10.1002/etc.3432. [DOI] [PubMed] [Google Scholar]
- Welden N.A., Cowie P.R. Degradation of common polymer ropes in a sublittoral marine environment. Mar. Pollut. Bull. 2017;118(1–2):248–253. doi: 10.1016/j.marpolbul.2017.02.072. [DOI] [PubMed] [Google Scholar]
- Wenner E.L., Holland A.F., Sanger D.M. Proceedings, Ocean Community Conference '98. vol. 2. 1998. Assessing short-term variability in dissolved oxygen and other water quality variables in shallow estuarine habitats; pp. 802–806. [Google Scholar]
- Wertz H. College of Charleston; 2015. Marine Debris in Charleston Harbor: Characterizing Plastic Particles in the Field and Assessing Their Effects on Juvenile Clams (Mercenaria mercenaria) [Google Scholar]