Abstract
Acute kidney injury (AKI) is an important complication of COVID-19 encompassing a wide range of presentations. SARS-CoV-2 is proposed to cause AKI in the patients through various mechanisms. We are, nevertheless, far from a comprehensive understanding of the underlying pathophysiological mechanisms of the kidney injury in this infection. AKI has been shown to be a marker of disease severity and also a negative prognostic factor for survival. Unfortunately, no effective preventive strategy to decrease the risk of kidney damage in these patients has yet been identified. In this hypothesis, we highlight the potential protective effects of acetazolamide, a carbonic anhydrase inhibitor, in preventing the proximal tubular damage caused by the virus through disrupting the virus-endosome fusion and also interfering with the lysosomal proteases. Our proposed mechanisms could pave the way for further in vitro studies and subsequent clinical trials.
Keywords: COVID-19, SARS-CoV-2, Acute kidney injury, Acetazolamide, Carbonic anhydrases
The novel coronavirus, which had initially led to pneumonia of unknown etiology in a series of patients in China's Hubei province in December 2019, has now precipitated a global health crisis (Habibzadeh and Stoneman, 2020). It has been shown that SARS-CoV-2 uses the same SARS-CoV receptor ACE2 for cell entry (Hoffmann et al., 2020; Shojaei et al., 2020; Sureda et al., 2020). This receptor is not only present in alveolar cells but also has a high expression level in other organs including kidneys (Hamming et al., 2004). Although further investigations on the prevalence of kidney injury in COVID-19 infection and the potential mechanisms of injury are needed, investigations on different cohorts of patients with COVID-19 infection have highlighted the importance of acute kidney injury (AKI) as an important complication of this infection and also a critical prognostic factor (Cheng et al., 2020). A recent meta-analysis has shown that the odds of death in patients with COVID-19 infection with AKI is more than 16 times higher than those without AKI (Mou and Zhang, 2020).
Prior studies on SARS-CoV have shown that the virus enters its target cells via a pH-sensitive endocytosis pathway (Yang et al., 2004). On the grounds of preliminary findings in patients with COVID-19 suggestive of the effectiveness of treatments such as Chloroquine, which is known to cause elevation of endosomal pH and therefore disrupt virus-endosome fusion in other viral infections, it is tempting to suggest that this therapeutic strategy might be effective here as well (Drożdżal et al., 2020; Gao et al., 2020; Khan et al., 2010; Shojaei et al., 2020). Furthermore, it has been shown that SARS-CoV-2 uses TMPRSS2 and CatB/L proteases for cell invasion, the latter which are blocked by elevated endosomal pH (Hoffmann et al., 2020).
Although the exact mechanism of kidney injury in patients with COVID-19 infection remains elusive, many potential mechanisms could be responsible for kidney damage in these patients. Cytokine storm and elevated levels of pro-inflammatory cytokines (e.g. IL-6) have been observed in patients with COVID-19 infection, particularly those in critical conditions (Mahmudpour et al., 2020). Therefore, the inflammatory state in the kidney and increased vascular permeability might be responsible for the development of AKI. In addition, direct viral damage might be an important factor in the pathophysiology of AKI in this infection. In a postmortem histopathological examination of six patients who had developed AKI during the course of COVID-19 infection, varying degrees of acute tubular necrosis, and presence of virus-like particles in the kidneys were observed (Diao et al., 2020). Electron microscopic examination of 26 patients with COVID-19 infection revealed coronavirus-like particles with distinctive spikes in renal podocytes and tubular epithelial cells providing further supporting evidence for direct virus invasion to the kidneys in this infection (Su et al., 2020). Extensive tubular vacuolization suggestive of acute tubular injury and presence of rare ill-defined round particles in the podocytes are among other histopathologic findings in this infection (Bradley et al., 2020). Kidney injury caused by direct SARS-CoV-2 invasion corroborated by postmortem histopathological studies can potentially be prevented through targeting virus-endosome fusion.
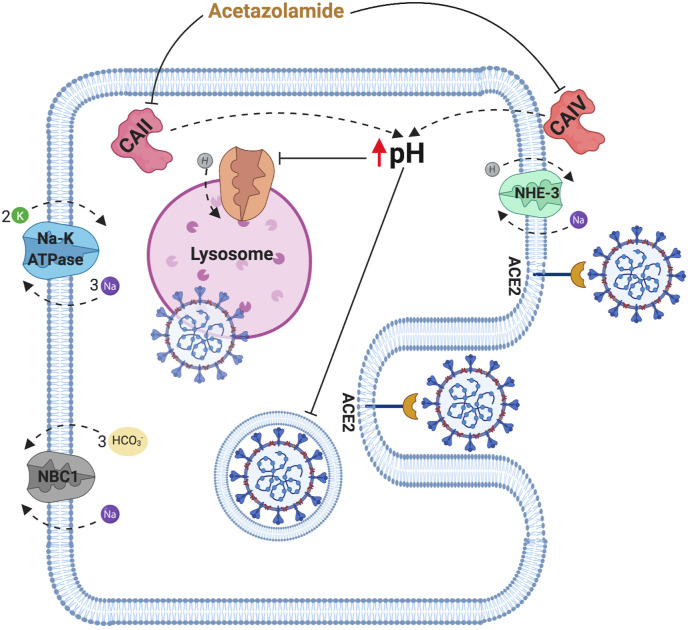
Proximal tubular cells have the highest expression of ACE2 receptors on their apical membranes and are therefore most susceptible to direct virus damage (Soleimani, 2020; Ye et al., 2006). Carbonic anhydrases are a group of metalloenzymes catalyzing the hydration of CO2 forming carbonic acid. There are two pools of carbonic anhydrase enzymes, namely carbonic anhydrase II and carbonic anhydrase IV. carbonic anhydrase II, the predominant type in the proximal tubule, is expressed in the cytoplasm while carbonic anhydrase IV is located on the plasma membrane with an extracellular active site (Zhu and Sly, 1990). Acetazolamide (Diamox) can inhibit both pools of carbonic anhydrases and has been shown to cause an increase in the cytosolic pH (Henderson et al., 1986; Skorecki et al., 2016). Despite the fact that endosomal pH might not necessarily be identical to the intracellular pH, the alkaline cytosolic pH in these cells can possibly disrupt virus-endosome fusion and the ensuing cytopathic effects. Furthermore, the decreased intracellular H+ can decrease the ability of the lysosomal transporters responsible for maintaining the acidity of the lysosome (Mindell, 2012). This decrease in lysosomal acidity can subsequently inactivate the pH-dependent proteases which are responsible for the cleavage of viral glycoproteins enabling viral replication (Fig. 1 ) (Burkard et al., 2014; Soleimani, 2020).
Fig. 1.
Schematic diagram depicting the potential mechanism of action of acetazolamide in the prevention of acute kidney injury in COVID-19 infection. CAII: carbonic anhydrase enzyme type 2, CAIV: carbonic anhydrase enzyme type 4, NBC1: Sodium bicarbonate cotransporter 1, NHE-3: Sodium hydrogen antiporter 3.
It is also notable that proximal tubular cells are proposed to be uniquely more alkaline in patients with hereditary proximal renal tubular acidosis (RTA) (Kamel and Halperin, 2017). Although the prevalence of AKI in patients with proximal RTA who have had COVID-19 infection has not yet been studied, these patients might be more resistant to the kidney injury caused by the SARS-CoV-2 due to mechanisms we highlighted.
Although some studies have investigated the potential effectiveness of acetazolamide in other clinical settings associated with AKI through different mechanisms, the possible protective effect of this drug in AKI in patients with COVID-19 has not yet been studied (Davidov et al., 2006; El Hamamsy et al., 2018; Pochedly, 1973).
We, therefore, hypothesize that prophylactic administration of low dose acetazolamide in those considered to be at high risk of developing severe COVID-19 infection might exert a protective effect against kidney injury in these patients by acting through two inter-related mechanisms inhibiting virus invasion and replication. Although long term administration of acetazolamide could potentially have less proximal tubular alkalization effect once the serum bicarbonate levels have dropped, to the best of our knowledge this effect has not yet been investigated. Further experimental studies to shed light on the efficacy of this preventive strategy are needed.
CRediT authorship contribution statement
Parham Habibzadeh: Writing - original draft. Mohammad Mofatteh: Writing - review & editing. Saeid Ghavami: Writing - review & editing. Jamshid Roozbeh: Supervision, Writing - review & editing.
Declaration of competing interest
None to Declare.
References
- Bradley B.T., Maioli H., Johnston R., Chaudhry I., Fink S.L., Xu H., Najafian B., Deutsch G., Lacy J.M., Williams T., Yarid N., Marshall D.A. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet. 2020;396 doi: 10.1016/S0140-6736(20)31305-2. 320–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burkard C., Verheije M.H., Wicht O., van Kasteren S.I., van Kuppeveld F.J., Haagmans B.L., Pelkmans L., Rottier P.J., Bosch B.J., de Haan C.A. Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner. PLoS Pathog. 2014;10 doi: 10.1371/journal.ppat.1004502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y., Luo R., Wang K., Zhang M., Wang Z., Dong L., Li J., Yao Y., Ge S., Xu G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97(5):829–838. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidov T., Hong J.J., Malcynski J.T. Novel use of acetazolamide in the treatment of rhabdomyolysis-induced myoglobinuric renal failure. J. Trauma. 2006;61:213–215. doi: 10.1097/01.ta.0000224136.77198.7e. [DOI] [PubMed] [Google Scholar]
- Diao B., Wang C., Wang R., Feng Z., Tan Y., Wang H., Wang C., Liu L., Liu Y., Liu Y., Wang G., Yuan Z., Ren L., Wu Y., Chen Y. medRxiv; 2020. Human Kidney Is a Target for Novel Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection. 2020.2003.2004.20031120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drożdżal S., Rosik J., Lechowicz K., Machaj F., Kotfis K., Ghavami S., Łos M.J. FDA approved drugs with pharmacotherapeutic potential for SARS-CoV-2 (COVID-19) therapy. Drug Resist. Updates. 2020;53:100719. doi: 10.1016/j.drup.2020.100719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El Hamamsy M., Kamal N., Bazan N.S., El Haddad M. Evaluation of the effect of acetazolamide versus mannitol on cisplatin-induced nephrotoxicity, a pilot study. Int. J. Clin. Pharm. 2018;40:1539–1547. doi: 10.1007/s11096-018-0677-x. [DOI] [PubMed] [Google Scholar]
- Gao J., Tian Z., Yang X. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020;14:72–73. doi: 10.5582/bst.2020.01047. [DOI] [PubMed] [Google Scholar]
- Habibzadeh P., Stoneman E.K. The novel coronavirus: a bird's eye view. Int. J. Occup. Environ. Med. 2020;11:65–71. doi: 10.15171/ijoem.2020.1921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson R.M., Bell P.B., Cohen R.D., Browning C., Iles R.A. Measurement of intracellular pH with microelectrodes in rat kidney in vivo. Am. J. Physiol. Ren. Physiol. 1986;250:F203–F209. doi: 10.1152/ajprenal.1986.250.2.F203. [DOI] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Muller M.A., Drosten C., Pohlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2) doi: 10.1016/j.cell.2020.02.052. 271-280.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamel K.S., Halperin M.L. Elsevier Health Sciences; 2017. Fluid, Electrolyte and Acid-Base Physiology E-Book: A Problem-Based Approach. [Google Scholar]
- Khan M., Santhosh S.R., Tiwari M., Lakshmana Rao P.V., Parida M. Assessment of in vitro prophylactic and therapeutic efficacy of chloroquine against Chikungunya virus in vero cells. J. Med. Virol. 2010;82:817–824. doi: 10.1002/jmv.21663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahmudpour M., Roozbeh J., Keshavarz M., Farrokhi S., Nabipour I. COVID-19 cytokine storm: the anger of inflammation. Cytokine. 2020:155151. doi: 10.1016/j.cyto.2020.155151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mindell J.A. Lysosomal acidification mechanisms. Annu. Rev. Physiol. 2012;74:69–86. doi: 10.1146/annurev-physiol-012110-142317. [DOI] [PubMed] [Google Scholar]
- Mou Z., Zhang X. medRxiv; 2020. The Dangers of Acute Kidney Injury in COVID-19 Patients: A Meta-Analysis of First Reports. 2020.2004.2029.20079038. [Google Scholar]
- Pochedly C. Hyperuricemia in leukemia and lymphoma. I. Clinical findings and pathophysiology. N. Y. State J. Med. 1973;73:1085–1092. [PubMed] [Google Scholar]
- Shojaei S., Suresh M., Klionsky D.J., Labouta H.I., Ghavami S. Autophagy and SARS-CoV-2 infection: apossible smart targeting of the autophagy pathway. Virulence. 2020;11:805–810. doi: 10.1080/21505594.2020.1780088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skorecki K., Chertow G.M., Marsden P.A., Taal M.W., Yu A.S.L. 10 ed. Elsevier; 2016. Brenner & Rector's the Kidney. [Google Scholar]
- Soleimani M. Acute kidney injury in SARS-CoV-2 infection: direct effect of virus on kidney proximal tubule cells. Int. J. Mol. Sci. 2020;21 doi: 10.3390/ijms21093275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su H., Yang M., Wan C., Yi L.-X., Tang F., Zhu H.-Y., Yi F., Yang H.-C., Fogo A.B., Nie X. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020;98(1):219–227. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sureda A., Alizadeh J., Nabavi S.F., Berindan-Neagoe I., Cismaru C.A., Jeandet P., Los M.J., Clementi E., Nabavi S.M., Ghavami S. Endoplasmic reticulum as a potential therapeutic target for covid-19 infection management? Eur. J. Pharmacol. 2020;882:173288. doi: 10.1016/j.ejphar.2020.173288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z.Y., Huang Y., Ganesh L., Leung K., Kong W.P., Schwartz O., Subbarao K., Nabel G.J. pH-dependent entry of severe acute respiratory syndrome coronavirus is mediated by the spike glycoprotein and enhanced by dendritic cell transfer through DC-SIGN. J. Virol. 2004;78:5642–5650. doi: 10.1128/JVI.78.11.5642-5650.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye M., Wysocki J., William J., Soler M.J., Cokic I., Batlle D. Glomerular localization and expression of Angiotensin-converting enzyme 2 and Angiotensin-converting enzyme: implications for albuminuria in diabetes. J. Am. Soc. Nephrol. 2006;17:3067–3075. doi: 10.1681/ASN.2006050423. [DOI] [PubMed] [Google Scholar]
- Zhu X.L., Sly W.S. Carbonic anhydrase IV from human lung. Purification, characterization, and comparison with membrane carbonic anhydrase from human kidney. J. Biol. Chem. 1990;265:8795–8801. [PubMed] [Google Scholar]