Abstract
The Coronavirus disease 2019 (COVID-19) pandemic has been especially devastating among nursing home residents, with both the health circumstances of individual residents as well as communal living settings contributing to increased morbidity and mortality. Preventing the spread of COVID-19 infection requires a multipronged approach that includes early identification of infected residents and health care personnel, compliance with infection prevention and control measures, cohorting infected residents, and furlough of infected staff. Strategies to address COVID-19 infections among nursing home residents vary based on the availability for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) tests, the incorporation of tests into broader surveillance efforts, and using results to help mitigate the spread of COVID-19 by identifying asymptomatic and presymptomatic infections. We review the tests available to diagnose COVID-19 infections, the implications of universal testing for nursing home staff and residents, interpretation of test results, issues around repeat testing, and incorporation of test results as part of a long-term response to the COVID-19 pandemic. We propose a structured approach for facility-wide testing of residents and staff and provide alternatives if testing capacity is limited, emphasizing contact tracing. Nursing homes with strong screening protocols for residents and staff, that engage in contact tracing for new cases, and that continue to remain vigilant about infection prevent and control practices, may better serve their residents and staff by thoughtful use of symptom- and risk-based testing strategies.
Keywords: Sentinel surveillance, infection prevention, SARS-CoV-2, skilled nursing facility, long-term care
Although there is general agreement that increased access to testing is important for personal and public health, the selection and use of diagnostic tests to mitigate Coronavirus Disease 2019 (COVID-19) infections in post-acute and long-term care settings is complex and should be tailored to individual sites.
Case
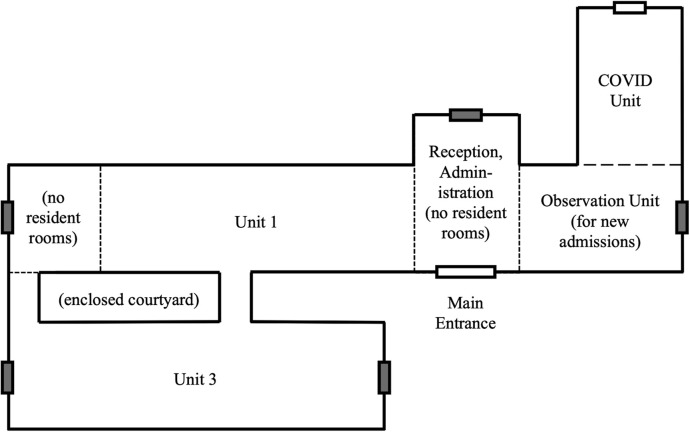
A 134-bed nursing home in northeast Georgia implemented several measures to prevent introducing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) into the building: limited entry, screening of staff for potential exposures and symptoms of COVID-19 infection, staff temperature checks, universal masking, and visitor restriction. The nursing home designated one area of their building to serve as a COVID Unit and another to serve as on observation unit in preparation for the eventual arrival of new admissions (Figure 1 ). Occupancy was approximately 75%.
Fig. 1.
General floorplan for the nursing home described in the case. White rectangles indicate entry points with screening stations for staff. Gray rectangles indicate exit point. The small dashed lines indicate distinct sections of the building connected by a common hallway. The large dashed line indicates the physical barrier put in place to separate the COVID-19 unit from remainder of the building.
On March 31, 2020, a 76-year-old man developed a catheter-associated urinary tract infection. He responded to antibiotics and his fever resolved within 12 hours. Because he met the nursing home's enhanced screening criteria for COVID-19 (Table 1 ),1 he was placed on transmission-based precautions and a laboratory test for SARS-CoV-2 was ordered.
Table 1.
Enhanced Screening Criteria for COVID-19 Infection in Nursing Home Residents∗
| Fever ≥ 37.5°C (99.5°F) |
| Cough |
| Shortness of breath |
| Increase in oxygen requirements or >3% decrease in usual pulse oximetry |
| Confusion or change in mental status |
| Exacerbations of congestive heart failure or chronic obstructive pulmonary disease |
| Muscle aches, headache |
| Sore throat, runny nose |
| Chest pain |
| Diarrhea, nausea or vomiting |
| Falls |
| Change in condition |
Adapted from Gaur et al.1
What Tests Should be Used to Diagnose COVID-19 Infection in Nursing Home Residents?
The Food and Drug Administration (FDA) has authorized several diagnostic and serologic (blood-based) tests as part of the Emergency Use Authorization (EUA) process (Table 2 ). Importantly, SARS-CoV-2 tests authorized under the EUA process have not undergone the comprehensive review normally conducted.
Table 2.
Overview of Tests for SARS-CoV-2
| Type of Test | What It Measures | Specimen Type | CPT Codes/Approximate Cost∗ | Advantages | Disadvantages |
|---|---|---|---|---|---|
| RT-PCR (molecular) | Nucleic acid from SARS-CoV-2 (RNA) | Nasopharyngeal swab | CPT code 87635/$51.31–$100 |
|
|
| Antigen test (molecular) | SARS-CoV-2 antigens | Nasal swab | CPT code 87426/(payment rates not available). |
|
|
| Antibody test (serology) | SARS-CoV-2 antibodies in the blood | Blood | CPT codes 86328 and 86769/$42.13–$45.23 |
|
|
CPT, current procedural terminology; Medicare payment rates as of May 19, 2020.2
Molecular Tests
The most common molecular tests used to diagnose current COVID-19 infection use reverse-transcriptase polymerase chain reaction (RT-PCR) to detect SARS-CoV-2 viral RNA. When an RT-PCR test is performed, RNA from the virus is extracted and amplified for detection. Because of the need to extract the viral RNA, RT-PCR tests usually require more time to complete than antigen tests. A number of different RT-PCR tests are available, most of which are laboratory based with a few point-of-care options as well.3 Although there are reports assessing the relative sensitivity and specificity of the different tests,4 there is not yet a gold standard for diagnosing COVID-19 infection. A widely quoted study from Wuhan, China, reported that RT-PCR was 70% sensitive when compared with a computed tomography (CT) scan of the chest.5 This study was conducted in the early stages of the pandemic on 1014 patients. Since then, the number of RT-PCR–based tests and availability of those tests, as well as the prevalence and clinical recognition of signs and symptoms of COVID-19 infection, has changed dramatically. All of these factors influence the sensitivity and specificity of diagnostic RT-PCR tests. Even if the sensitivity of RT-PCR–based testing has increased to an optimistic 90% to 95%, it still means that 1 of every 10 to 20 negative results will be a false negative and miss a COVID-19 infection. A lower level of sensitivity may not be an issue in settings or regions where the disease prevalence is low. Where the disease is prevalent, however, such as during a facility outbreak, negative results must be viewed with caution.
The sensitivity of the test also depends on when testing is done during the course of illness, with the peak of viral shedding occurring around the time of symptom onset.6 A Bayesian hierarchical model, fit to previously published studies describing the performance of SARS-CoV-2 RT-PCR tests, reported the probability of a false negative RT-PCR test over time.7 From exposure (day 0) to the typical time of symptom manifestation (day 5), the probability of a false negative results changes from 100% on day 1, to 67% on day 4, reaching a nadir of 20% 3 days after symptom onset on day 8. In some individuals, RT-PCR tests will remain positive for weeks after symptom resolution.8 Evaluations of these individuals for virus able to cause in vitro infection in cell culture9 as well as epidemiological investigations10 indicate that after 8 days following symptom onset, individuals who no longer have symptoms of COVID-19 infection do not appear to transmit infection. These findings led to policy recommendations from the Centers for Disease Control and Prevention (CDC) to consider discontinuation of isolation at 10 days after illness onset and at least 1 day after fever resolution and improvement of symptoms.11
The sample site and collection technique can also influence the sensitivity of the test. Currently, the most common and accepted method for sample collection is a thin swab used to collect a sample from the posterior nasopharynx. The test is uncomfortable and may be difficult to collect from uncooperative residents, especially those with cognitive dysfunction. Because nasopharyngeal swabs may induce sneezing or coughing, health care workers collecting these samples must wear personal protective equipment (PPE). A study from China collected samples from 205 individuals who had a symptomatic COVID-19 infection. The proportion of positive tests from sputum was 72% (75/104), followed by anterior nares swabs (63%; 5/8) and oropharyngeal swabs (32%; 126/398).12 A study from Washington State compared results from nasopharyngeal swabs collected by health care workers with self-collected samples from 530 individuals who presented to ambulatory care clinics with respiratory tract symptoms. Compared with nasopharyngeal swabs collected by health care workers, self-collected anterior nares and mid-turbinate samples had a sensitivity of 94% and 96%, respectively.13 Many long-term care residents, however, may not be able to collect their own sample and in general, represent a different population compared with symptomatic, ambulatory adults. A study of 190 individuals compared test results for nasopharyngeal swabs with those from combined oral and anterior nares swabs, reporting similar sensitivities of 91.7% and 94.4%, respectively.10 This may be a more feasible and comfortable approach for sample collection from long-term care residents as well as staff, particularly in areas with mandates for serial testing of health care workers. This also offers the possibility that staff may effectively collect their own samples. Self-collected samples have the advantage of decreasing staff exposure and their use of PPE.11
Antigen Tests
On May 9th, 2020, the FDA provided the first EUA for a test designed to detect an antigen specific to SARS-CoV-2; a second antigen-based test received an EUA on July 2, 2020.12 , 13 Antigen tests target markers on the outer surface of the viral capsule and do not require extraction of RNA. Viral structures that represent good candidates for antigen-based testing include nucleocapsid phosphoproteins and spike glycopeptide proteins.14 Advantages to antigen-based tests are the relatively lower cost compared with RT-PCR tests, the ease of collection samples from the anterior nares, the potential to scale the number of tests, and the rapid turn-around time, providing answers within minutes.13 A disadvantage to antigen-based tests, however, is lower sensitivity, which can lead to false negative results. This is a particular issue in the setting of outbreaks or in regions with increased prevalence rates of SARS-CoV-2. The FDA recommends that a second PCR-based test should be used to confirm negative results for individuals likely to have COVID-19.12 Given that only 2 of the 164 diagnostic tests given EUAs are antigen-based,13 the role for these tests is still evolving and may be best suited for diagnosing symptomatic individuals seeking medical care, rather than screening asymptomatic individuals or health care workers. Regardless, the Centers for Medicare and Medicaid Services (CMS) recently announced a plan to deploy the 2 available antigen test kits to all US nursing homes to assist in universal screening efforts,15 although noting caution in interpreting negative results.16 Facilities using antigen tests need to make sure they have obtained the required federal and/or state Clinical Laboratory Improvement Amendments (CLIA) waivers, competency trained staff, a recording system to document and track results, and are able to report results to state and/or federal agencies as required.16 , 17
Antibody Tests
The purpose of serologic tests for SARS-CoV-2 is to ascertain previous COVID-19 infection. These tests may also help identify individuals who can donate convalescent plasma as well as inform epidemiologic studies.18 Serologic tests are not authorized and should not be used to diagnose current SARS-CoV-2 infection.
Antibody tests assess for the presence of immunoglobulin (Ig)M, IgG, and IgA antibodies to nucleocapsid phosphoproteins and spike glycopeptide proteins specific to SARS-CoV-2, which are the same groups of proteins targeted for molecular antigen tests.19 The performance of these tests is subject to the prevalence of COVID-19 infections among the population tested, which varies across states, cities, and care settings. The positive predictive value (PPV) and negative predictive value (NPV) of serologic tests is lower in areas with a low prevalence of COVID-19 infections.
Both IgM and IgG antibodies typically develop 1 to 3 weeks following symptom onset, with IgM levels declining over the next several weeks. The role of IgA in immunity is still unclear. The longevity of IgG antibodies specific to SARS-CoV-2 is not yet known. Studies of convalescent serum obtained from 176 individuals following infection with SARS-CoV2 indicates a decline in IgG levels by year 3.20 These findings, and similar results describing long-lasting immune protection following infection with Middle East respiratory syndrome (MERS) coronavirus,21 suggest that most individuals who have recovered from a COVID-19 infection are not at risk for reinfection.
Case, Continued
Staff wearing appropriate PPE (disposable surgical mask, face shield, gown, and gloves) collected a nasopharyngeal swab from the resident. On April 1, the laboratory notified the nursing home that the test was positive for SARS-CoV-2.
After Identifying a Case of COVID-19, Should the Nursing Home Proceed With Universal Testing of Their Residents and Staff?
Following the diagnosis of their first case of COVID-19 infection, whether the index patient is a resident or staff member, nursing homes with sufficient supplies, personnel, and resources should proceed with facility-wide testing of residents and staff for SARS-CoV-2 (Table 3 ).23
Table 3.
Preparedness Checklist for Facility-wide Testing∗
| Category | Checklist | Comments |
|---|---|---|
| Develop a testing plan |
|
Each state may have differing guidelines pertaining to testing. Facilities should consider both staff and residents when forming a testing program. Facilities should consider whether to test all residents or a targeted group, such as a single unit. Facilities should decide whether and when to repeat testing based on results, prevalence in the community, and anticipated actions that will be guided by results. Facilities should consider who is responsible for testing costs, being particularly mindful that some staff and residents may not have health insurance. |
| Testing of residents |
|
Residents with recent COVID-19 might not need to be retested. Use of separate teams when conducting PCR testing, such as one to prepare each resident and the resident's rooms, and a second to perform the specimen collection can improve testing efficiency and reduce frontline staff burden. Ideally, PPE worn during sample collection includes a respirator, eye protection, a gown, and gloves. In settings without respirators, a surgical mask and face shield may be used instead. |
| Testing of staff |
|
Some states do not need a provider order for the staff testing but this is requested by the laboratory. Some insurance providers are not covering testing of asymptomatic nursing home staff. When possible, sample collection should be in a well-ventilated area, which includes outdoors and, weather permitting, may be feasible to accomplish with staff members. |
| Dealing with SARS-Cov-2 testing results from residents |
|
One option is to communicate with resident's family that they will only be informed of positive test results. Plan for use of multiple means of communication to residents, family members, and/or responsible parties. This may include use of recorded phone systems, email, website updates, town-hall updates, and direct phone communication. Communication about resident results should ensure individual privacy. Prepare for the additional works of moving residents and their belongings to a COVID-19 cohorting area. |
| Dealing with SARS-Cov-2 results from staff |
|
Plan for use of multiple means of communication to residents, family members, and/or responsible parties. This may include use of recorded phone systems, email, website updates, town-hall updates, and direct phone communication. Communication about employee results should ensure individual privacy. CMS has a temporary waiver for nurse aides' certification.22 |
Adapted from CDC guidance.23
Universal testing helps identify people with asymptomatic or presymptomatic COVID-19 infection. The incubation period, the time between exposure and symptom onset, for COVID-19 infections is 4 to 5 days, and may be as long as 14 days.24 , 25 Early reports of COVID-19 outbreaks in nursing homes reported the median time for presymptomatic infection, the time during which individuals may spread infections before developing symptoms, was 4 days (interquartile range of 3 to 5).26 To put this in perspective, the incubation period and presymptomatic phase for seasonal influenza are 2 days and 1 day, respectively.27 Evidence-based model studies suggest that people with asymptomatic, presymptomatic, or mildly symptomatic COVID-19 infections cause 50% to 80% of SARS-CoV-2 transmission in the community.28 These observations, coupled with the high attack rate of COVID-19 infections among nursing home residents and staff, underscore the importance of widespread testing with a rapid turn-around time that can help guide infection prevention and control measures to reduce the risk of further SARS-CoV-2 transmission.
Unfortunately, many nursing homes may not be able to implement universal testing for a multitude of reasons, including lack of access to testing, inadequate supplies of PPE, limited personnel, and insufficient resources to pay for testing.29, 30, 31, 32 As they attempt to respond to federal33 and state34 recommendations and requirements, many nursing homes will need to consider other approaches to testing, such as unit-based testing and/or risk-based testing. Regardless of how they proceed, it is essential that nursing homes work with their laboratory provider to prioritize residents and staff tests and ensure rapid turnaround times (48 hours or less).35
Unit-based strategies, although not desirable, would be most suitable for situations in which testing capacity is limited. For this approach, the building focuses testing on residents and staff members who had close contact with the index case. Testing can also be extended to include high-risk individuals, such as those leaving the facility frequently (eg, for hemodialysis, radiation therapy, or off-site chemotherapy) and those recently admitted. Determination of who to test first is inherently complex and may be best approached by a multidisciplinary team that includes the infection preventionist, medical director, director of nursing, and administrator.
Given that nursing homes have only recently started to contemplate reopening for visitation,33 , 36 the most likely source of new infections is from staff. The risk of staff introducing SARS-CoV-2 into the building is related to the prevalence of SARS-CoV-2 in the community. In addition to nurses and nurse assistants, personnel working in many other roles may also transmit infection, including respiratory therapists, physical and occupational therapists, radiology technicians, environmental services, social workers, physicians, advanced practice providers, and food and nutrition staff. Less commonly, residents may become exposed to people with asymptomatic or presymptomatic COVID-19 infections in other health care venues as well as by the personnel who assist with transportation to and from other settings.
Case, Continued
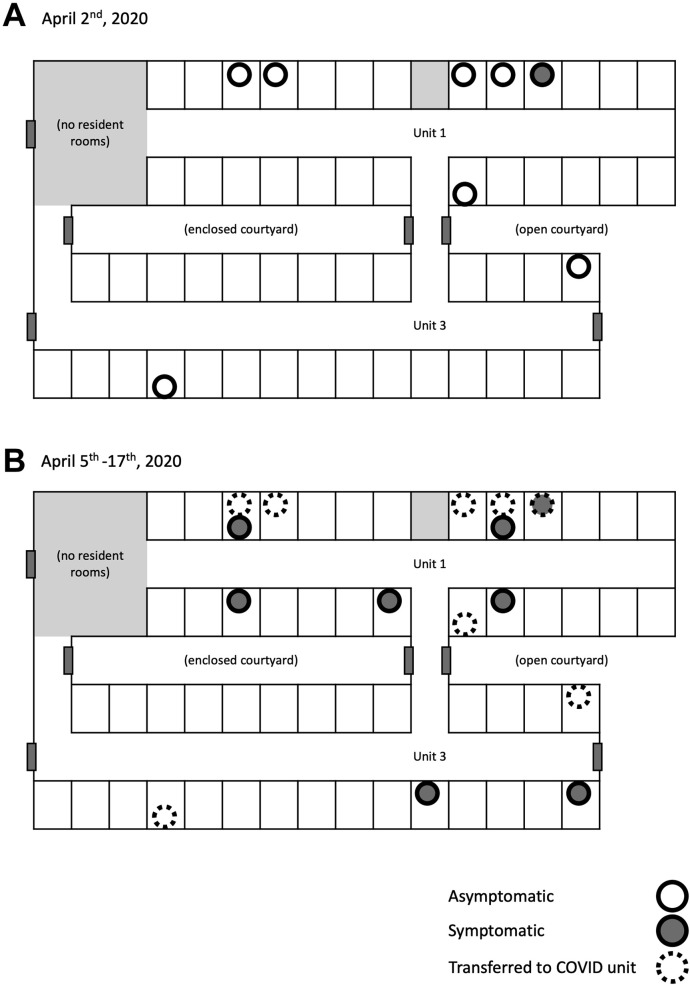
On April 2, all 97 residents were restricted to their rooms and tested for SARS-CoV-2. On April 3, laboratory results indicated that an additional 7 residents had COVID-19 infection; all 7 were asymptomatic at the time of testing (Figure 2 A). The 8 residents with COVID-19 infection were moved to the designated COVID Unit. On April 3 and 4, 119 of 121 staff members were tested for SARS-CoV-2. Of those, 15 were positive and were placed on home quarantine.
Fig. 2.
More detailed floor plan of Units 1 and 3 of the nursing home described in the case. (A) On April 1, the index resident (gray circle) developed symptoms that trigged testing for COVID-19 infection. Among the SARS-CoV-2 tests collected on April 2, 7 more residents tested positive for SARS-CoV-2 (white circle). All of these individuals were transferred to the COVID Unit. (B) From April 5 to 17, an additional 7 residents developed signs and symptoms of COVID-19 infection. All of the residents had negative tests from April 2 with subsequent positive SARS-CoV-2 tests when they became symptomatic.
What are the Next Steps the Nursing Home Should Take in Response to Positive Test Results?
Responding to positive tests is a dynamic process that must be tailored to each site. Any resident with signs and symptoms suggestive of possible COVID-19 infection should be placed in transmission-based precautions in the resident's current room. For SARS-CoV-2, available evidence supports transmission via short-range aerosols (ie, respiratory droplets) that are inhaled or acquired onto skin and subsequently introduced into eyes, nose, or mouth. Accordingly, in health care settings, both contact and droplet precautions are indicated to reduce the risk of transmission to healthcare workers. Because SARS-CoV-2 is an emerging pathogen that may cause severe disease and for which there is not yet an effective vaccine or treatment, the CDC continues to recommend respirators during the care of people with known or suspected COVID-19 infection, while also recognizing that these remain in short supply.37 Given the risk of transmission posed by respiratory droplets and fomites, the roommate may be best served by moving into a private room on the same unit. These decisions are easier in buildings operating below full occupancy.
If the test is positive, the resident should be moved to a unit (or area of a unit) specifically designated for those COVID-19 infections. Ideally, this unit is both physically and operationally separated from other areas of the building. Physical separation may be achieved through engineering controls such as temporary plastic barriers and separate entrances. Achieving distinct operations involves staff amenable to working only on that unit as well as dedicating equipment, including medication carts, oxygen concentrators, emergency supplies, and handling of clean and dirty linens. Clinical staff who agree to work in a unit dedicated to caring for residents with COVID-19 should not have contact with personnel assigned to other areas; this includes separate areas for changing clothes as well as for meals and breaks. Furthermore, staff-to-resident ratios should be increased to account for the increased monitoring, clinical assessment, and supportive measures required in the care of residents with COVID-19 infections.
In addition to implementing transmission-based precautions for all residents on the unit after a COVID-19 case is identified, contact tracing of the infected residents and especially staff is important to prevent further spread of infection. A first step is to note the physical location of the new cases as these patterns help to determine the extent to which the building should engage in contact tracing. In addition to identifying the location of ill residents, contact tracing of nurses and nurse aides can start by reviewing the work assignments for at least 48 hours before either symptom onset or, for asymptomatic cases, the positive test result. While a cluster of cases at one unit may be readily traced to staff assigned to those rooms, detection of cases among several units suggest the possibility of more than 1 infected staff member or of a single staff member, such as a social worker or physician, who interacts with residents throughout the building. For widespread infections across the facility, representatives from other services within the building, such as environmental services; food and nutrition; and physical, occupational, or recreation therapy, may also offer important perspectives, particularly around which of their staff members may have recently called off sick or who work at more than one building.
Case, Continued
Between April 5 and April 10, 6 residents who had previously tested negative developed signs and symptoms of COVID-19 infection. All were positive for SARS-CoV-2 and were transferred to the COVID unit (Figure 2B). On April 17, a seventh resident tested positive for SARS-CoV-2 after developing backpain, chest pain, and headache. The facility implemented face shields in addition to wearing a mask during interactions with residents and during mutual staff interactions.
How Should Nursing Homes Incorporate Test Results into Their Long-term Response to the COVID-19 Pandemic?
Although the initial point prevalence of SARS-Cov-2 testing of all the residents helped to identify asymptomatic or presymptomatic residents with COVID-19, the test only indicates each resident's status at that point in time. A single test might not identify exposed residents who are very early in the incubation period.7 As illustrated in this case, several residents developed symptoms a few days after the facility-wide testing. With the increasing availability of testing, nursing homes experiencing an outbreak may repeat tests for SARS-CoV-2 every 3 to 7 days, which may help with early identification and cohorting of infected residents.35
Additional measures to prevent the introduction of SARS-CoV-2 into the facility are to address the management of new admissions to the facility. For new admissions with a known COVID-19 infection, the most recent guidance from the CDC about time-based discontinuation of transmission-based precautions38 should help nursing homes stratify new admissions, perhaps choosing to delay a resident's arrival until the resident has reached day 10 following symptoms onset with ≥24 hours without a fever and improvement in symptoms. Those who have not yet reached these goals would be best served by admission to a designated COVID unit. For new admissions without current or previously documented COVID-19 infection, we recommend both careful screening and testing those individuals on arrival. Further, as space permits, these people are appropriate for an observation unit composed primarily of single beds, with a transfer to the general population only after 2 weeks on transmission-based precautions and room restriction.
Evidence of continued transmission of SARS-CoV-2 based on testing results should prompt efforts to step up ongoing infection prevention and control measures. They should also determine the need and frequency of staff and resident testing in that particular facility. As mentioned previously, however, insufficient access to rapid diagnostic testing, PPE, staff, and resources may render seemingly insurmountable challenges. Even under these dire circumstances, nursing homes will continue to strive to provide the best care they can offer for residents entrusted to their care (Table 3).
Nursing homes should continue to train their staff members in proper use of PPE. Buildings can prioritize N-95 and similar respiratory protective equipment to staff working in dedicated COVID units and encourage proper use of masks among all staff members.39 , 40 Staff may also have to change some of their behaviors to reduce the risk of staff-to-staff transmission. Specifically, staff may need proscriptive guidance from leadership about staggering breaks and mealtimes, maintaining social distancing when not wearing PPE, particularly during mealtimes, and wearing masks during staff-to-staff interactions. Leadership should also foster a culture in which all staff encourage peers on the proper use of PPE, including donning and doffing techniques. Rather than pointing our flaws or lapses, vigilance regarding all staff using strong infection prevention and control practices can be framed as supportive by using messages such as “I care about your health” or “I've got your back.”
Implications for Practice and Policy
The prospect of reopening nursing homes, which includes permitting visitors, presents additional complexities. Efforts by states to relax stay-at-home orders, reopen businesses, and even encourage tourism40 contribute to increased spread of SARS-CoV-2 in the community, which in turn means increased risk for nursing home staff to acquire and transmit the virus to their residents. As part of plans to reopen nursing homes, CMS recommended “baseline” testing for all nursing residents and staff, along with the capacity to continue weekly tests for staff.33 After the initial response to the CMS recommendation, most, if not all, nursing homes should continue surveillance testing. Older adults often manifest atypical signs and symptoms of infection. Although it may be tempting to attribute a change in condition to exacerbation of known heart failure or sinus congestion to allergies or a “summer cold,” the severity of the illness experienced by some of those infected with SARS-CoV-2, coupled with the lack of effective treatment and a vaccine, means that all health care settings have a responsibility to actively surveil their population for COVID-19 infections. Even in counties without known COVID-19 infections, residents unfortunate enough to reside in a nursing home that does not engage in surveillance testing may become “canaries in the coal mine” for that region, with their risk for infection exacerbated by living in a congregate setting.
Surveillance for COVID-19 infections is not a simple prospect and may generate new problems that nursing homes must address. In regions with high rates of COVID-19 infections in the community, surveillance of asymptomatic staff members may lead to a significant proportion of individuals on furlough. The discomfort of nasopharyngeal swabs may cause both residents and staff to decline repeat surveillance tests. Further, issues around employee health privacy as well as costs involved with paying for surveillance of residents and staff are open questions that federal, state, and local health authorities are still addressing. Nursing homes should carry out contact tracing within their building, using information about shift assignments and residents' illnesses to understand potential transmission among staff. Local health departments should conduct contact tracing for staff members as it pertains to their activities outside of work.
With regard to paying for tests, nursing homes should submit claims for residents' tests to their primary insurer. For staff, although some states may assist with collecting and processing samples, many nursing homes struggle to acquire testing kits and most will likely have to absorb those costs, which can be exorbitant. For the nursing home described here, weekly RT-PCR surveillance tests would cost on the order of at least $25,000 per month. When coupled with staff shortages, insufficient supplies of PPE, increased reporting requirements, rapidly evolving policy changes, and specter of increased inspections focused on infection prevention and control,41 this becomes a bitter pill for nursing homes to swallow.
Conclusion
Universal testing may have a role in helping to guide nursing homes' responses to the COVID-19 pandemic. Testing, however, is only one of many measures needed to prevent the spread of SARS-CoV-2 among the frail older adults living in congregate settings. Nursing homes with strong screening protocols for residents and staff, that engage in contact tracing for new cases, and that continue to remain vigilant about infection prevent and control practices, may better serve their residents and staff by thoughtful use of symptom- and risk-based testing strategies.
Acknowledgments
Dr. Jump reports support for this work in part through the Cleveland Geriatric Research Education and Clinical Center (GRECC) at the VA Northeast Ohio Healthcare System. The contents do not represent the views of the US Department of Veterans Affairs or the US government.
Footnotes
Dr. Jump discloses research funding from Pfizer and Merck; she has served on advisory boards for Pfizer and Roche. Dr. Dumyati discloses serving on an advisory board for Roche. The other authors declare no conflicts of interest.
References
- 1.Gaur S., Dumyati G., Nace D.A., Jump R.L.P. Unprecedented solutions for extraordinary times: Helping long-term care settings deal with the COVID-19 pandemic. Infect Control Hosp Epidemiol. 2020;41:729–730. doi: 10.1017/ice.2020.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Centers for Medicare & Medicaid Services Medicare Administrative Contractor (MAC) COVID-19 Test Pricing. 2020. 2020. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwjt6_esnN3rAhWyc98KHVL2DeMQFjAAegQIARAB&url=https%3A%2F%2Fwww.cms.gov%2Ffiles%2Fdocument%2Fmac-covid-19-test-pricing.pdf&usg=AOvVaw0H7cRnSxE23c70BjQVX6u0 Available at:
- 3.Food and Drug Administration In vitro diagnostics EUAs. FDA. 2020. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/vitro-diagnostics-euas Available at:
- 4.Zhen W., Manji R., Smith E., Berry G.J. Comparison of four molecular in vitro diagnostic assays for the detection of SARS-CoV-2 in nasopharyngeal specimens. J Clin Microbiol. 2020;58:e00743–e00820. doi: 10.1128/JCM.00743-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ai T., Yang Z., Hou H. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Radiology. 2020;296:E32–E40. doi: 10.1148/radiol.2020200642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sethuraman N., Jeremiah S.S., Ryo A. Interpreting diagnostic tests for SARS-CoV-2. JAMA. 2020;323:2249–2251. doi: 10.1001/jama.2020.8259. [DOI] [PubMed] [Google Scholar]
- 7.Kucirka L.M., Lauer S.A., Laeyendecker O. Variation in false-negative rate of reverse transcriptase polymerase chain reaction–based SARS-CoV-2 tests by time since exposure. Ann Intern Med. 2020;173:262–267. doi: 10.7326/M20-1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tu Y.-P., Jennings R., Hart B. Swabs collected by patients or health care workers for SARS-CoV-2 testing. N Engl J Med. 2020;383:494–496. doi: 10.1056/NEJMc2016321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Food and Drug Administration Coronavirus (COVID-19) Update: FDA authorizes first antigen test to help in the rapid detection of the virus that causes COVID-19 in patients. FDA. 2020. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-first-antigen-test-help-rapid-detection-virus-causes Available at:
- 10.Food and Drug Administration Coronavirus (COVID-19) Update: Daily Roundup July 6, 2020. FDA. 2020. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-daily-roundup-july-6–2020 Available at:
- 11.Centers for Disease Control and Prevention Duration of isolation and precautions for adults with COVID-19. 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html Available at:
- 12.Wang W., Xu Y., Gao R. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843–1844. doi: 10.1001/jama.2020.3786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.LeBlanc J.J., Heinstein C., MacDonald J. A combined oropharyngeal/nares swab is a suitable alternative to nasopharyngeal swabs for the detection of SARS-CoV-2. J Clin Virol. 2020;128:104442. doi: 10.1016/j.jcv.2020.104442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yong S.K., Su P.-C., Yang Y.-S. Molecular targets for the testing of COVID-19. Biotechnol J. 2020;15:2000152. doi: 10.1002/biot.202000152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Division N. Trump administration announces initiative for more and faster COVID-19 testing in nursing homes. HHS.gov. 2020. https://www.hhs.gov/about/news/2020/07/14/trump-administration-announces-initiative-more-faster-covid-19-testing-nursing-homes.html Available at:
- 16.Centers for Medicare and Medicaid Services Frequently asked questions: COVID-19 testing at skilled nursing facilities/nursing homes. https://www.cms.gov/files/document/covid-faqs-snf-testing.pdf Available at:
- 17.American Health Care Association Point-of-Care Antigen Test Devices - AHCA. 2020. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwjCqrvrnpTrAhVwxFkKHdflDNsQFjAAegQIARAB&url=https%3A%2F%2Fwww.ahcancal.org%2Ffacility_operations%2Fdisaster_planning%2FDocuments%2FPOC-Antigen-Tests.pdf&usg=AOvVaw1mgTkmedUYywCx9CLPEEZp Available at:
- 18.Centers for Disease Control and Prevention Information for laboratories about coronavirus (COVID-19) 2020. https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antibody-tests-guidelines.html Available at:
- 19.The Food and Drug Administration EUA Authorized Serology Test Performance. FDA. 2020. https://www.fda.gov/medical-devices/emergency-situations-medical-devices/eua-authorized-serology-test-performance Available at:
- 20.Wu L.-P., Wang N.-C., Chang Y.-H. Duration of antibody responses after severe acute respiratory syndrome. Emerg Infect Dis. 2007;13:1562–1564. doi: 10.3201/eid1310.070576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Payne D.C., Iblan I., Rha B. Persistence of antibodies against Middle East respiratory syndrome coronavirus. Emerg Infect Dis. 2016;22:1824–1826. doi: 10.3201/eid2210.160706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Centers for Medicare & Medicaid Services R14SOM.pdf. https://www.cms.gov/Regulations-and-Guidance/Guidance/Transmittals/downloads/R14SOM.pdf Available at: [PubMed]
- 23.Centers for Disease Control and Prevention Performing facility-wide SARS-CoV-2 testing in nursing homes. 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/nursing-homes-facility-wide-testing.html Available at:
- 24.Centers for Disease Control and Prevention Coronavirus disease 2019 (COVID-19): Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19) 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html Available at:
- 25.Lauer S.A., Grantz K.H., Bi Q. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: Estimation and application. Ann Intern Med. 2020;172:577–582. doi: 10.7326/M20-0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kimball A., Hatfield K.M., Arons M. Asymptomatic and presymptomatic SARS-CoV-2 infections in residents of a long-term care skilled nursing facility - King County, Washington, March 2020. MMWR Morb Mortal Wkly Rep. 2020;69:377–381. doi: 10.15585/mmwr.mm6913e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Centers for Disease Control and Prevention How flu spreads. 2018. https://www.cdc.gov/flu/about/disease/spread.htm Available at:
- 28.Furukawa N.W., Brooks J.T., Sobel J. Evidence supporting transmission of severe acute respiratory syndrome coronavirus 2 while presymptomatic or asymptomatic. Emerg Infect Dis. 2020;26:E201595. doi: 10.3201/eid2607.201595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Abbasi J. “Abandoned” nursing homes continue to face critical supply and staff shortages as COVID-19 toll has mounted. JAMA. 2020 doi: 10.1001/jama.2020.10419. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 30.Grabowski D.C., Mor V. Nursing home care in crisis in the wake of COVID-19. JAMA. 2020 doi: 10.1001/jama.2020.8524. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 31.Centers for Medicare and Medicaid Services Toolkit on state actions to mitigate COVID-19 prevalence in nursing homes. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwjo263Olo7qAhVvoXIEHcYLAnkQFjACegQIBRAB&url=https%3A%2F%2Fskillednursingnews.com%2F2020%2F06%2Ffor-covid-19-testing-to-succeed-in-nursing-homes-lab-access-is-critical-but-capacity-hard-to-define%2F&usg=AOvVaw0HIMUq8euObAQhsVCAkwcM Available at:
- 32.American Health Care Association/National Center for Assisted Living New state by state breakdown: COVID-19 testing for nursing homes and assisted living communities. https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwi9o5_CmY7qAhUHlXIEHVFmA1oQFjAAegQIAhAB&url=https%3A%2F%2Fwww.ahcancal.org%2FNews%2Fnews_releases%2FDocuments%2FState-Breakdown-COVID-Testing-LTC.pdf&usg=AOvVaw0pLAJ0P_g_oTle7HJs8Vne Available at:
- 33.The Centers for Medicare and Medicaid Services Nursing home reopening recommendations for state and local officials | CMS. 2020. https://www.cms.gov/medicareprovider-enrollment-and-certificationsurveycertificationgeninfopolicy-and-memos-states-and/nursing-home-reopening-recommendations-state-and-local-officials Available at:
- 34.No. 202.30 Continuing Temporary Suspension and Modification of Laws Relating to the Disaster Emergency. Governor Andrew M. Cuomo. 2020. https://www.governor.ny.gov/news/no-20230-continuing-temporary-suspension-and-modification-laws-relating-disaster-emergency Available at:
- 35.Centers for Disease Control and Prevention Coronavirus Disease 2019 (COVID-19) testing guidelines for nursing homes. 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/nursing-homes-testing.html Available at:
- 36.CMS Guidance for infection control and prevention of Coronavirus Disease 2019 (COVID-19) in nursing homes (REVISED) | CMS. https://www.cms.gov/medicareprovider-enrollment-and-certificationsurveycertificationgeninfopolicy-and/guidance-infection-control-and-prevention-coronavirus-disease-2019-covid-19-nursing-homes-revised Available at:
- 37.Centers for Disease Control and Prevention Clinical questions about COVID-19: Questions and answers. 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/faq.html Available at:
- 38.Centers for Disease Control and Prevention Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings (interim guidance) 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/disposition-hospitalized-patients.html Available at:
- 39.CMS Guidance for use of certain industrial respirators by health care personnel | CMS. https://www.cms.gov/medicareprovider-enrollment-and-certificationsurveycertificationgeninfopolicy-and/guidance-use-certain-industrial-respirators-health-care-personnel Available at:
- 40.Parker-Pope T. How NOT to wear a mask. The New York Times. 2020. https://www.nytimes.com/2020/04/08/well/live/coronavirus-face-mask-mistakes.html Available at:
- 41.Verma S. Verma: Taking action to protect America's nursing home residents against COVID-19. Your Valley. https://yourvalley.net/stories/verma-taking-action-to-protect-americas-nursing-home-residents-against-covid-19,171220 Available at: