Abstract
In 2017 the World Health Organization revised the criteria for classification of pancreatic neuroendocrine neoplasms (pNENs) after a consensus conference at the International Agency for Research on Cancer. The major change in the new classification was to subclassify the original G3 group into well-differentiated pancreatic neuroendocrine tumors G3 (pNETs G3) and poorly differentiated pancreatic neuroendocrine carcinomas (pNECs), which have been gradually proven to be completely different in biological behavior and clinical manifestations in recent years. In 2019 this major change subsequently extended to NENs involving the entire digestive tract. The updated version of the pNENs grading system marks a growing awareness of these heterogeneous tumors. This review discusses the clinicopathological, genetic and therapeutic features of poorly differentiated pNECs and compare them to those of well-differentiated pNETs G3. For pNETs G3 and pNECs (due to their lower incidence), there are still many problems to be investigated. Previous studies under the new grading classification also need to be reinterpreted. This review summarizes the relevant literature from the perspective of the differences between pNETs G3 and pNECs in order to deepen understanding of these diseases and discuss future research directions.
Keywords: Neuroendocrine neoplasms, Pancreatic neuroendocrine tumors G3, Pancreatic neuroendocrine carcinomas, Gene sequencing, Clinical management, Histopathology
Core tip: The major change in the 2017 World Health Organization (WHO) classification of pancreatic neuroendocrine neoplasms was to further subclassified the original G3 group into pancreatic neuroendocrine tumors G3 (pNETs G3) and pancreatic neuroendocrine carcinoma (pNEC). In 2019 this major change subsequently extended to neuroendocrine neoplasms involving the entire digestive tract. This review comprehensively summarizes the differences between pNET G3 and pNEC, which is the major update in latest WHO grading classification for pancreatic neuroendocrine neoplasms by the aspects of histology, gene mutation, and clinical management.
INTRODUCTION
Neuroendocrine neoplasms (NENs) are a heterogeneous group of tumors characterized by neural antigens such as chromogranin A or synaptophysin (Figure 1A and B). Chromogranin A is a member of the granin family of acidic secretory glycoproteins that occur in secretory granules of a wide variety of endocrine cells and neurons, and synaptophysin is an integral membrane glycoprotein that occurs in presynaptic vesicles of neurons. Chromogranin A and synaptophysin are considered to be the most valuable marker of NENs. Neuroendocrine tumors (NETs) are generated from specialized cells called neuroendocrine cells whereas neuroendocrine carcinomas (NECs) may be derived from more primitive progenitor cells, but the details remain unclear. NENs occur throughout the body, with most in the digestive tract, pancreas or lungs. Pancreatic NENs (pNENs) are rare in clinical practice and account for < 3% of primary pancreatic tumors[1]. The incidence and prevalence of NENs have been rising recently due to increased awareness and sophisticated diagnostic techniques. The prevalence of pancreatic neuroendocrine tumors (pNETs) in Japan was 2.69 per 100000 people in 2010, representing a 1.2-fold increase in the number of patients in 2005. The new-onset functioning pNETs in 2010 was similar to that in 2005; however, the number of new-onset nonfunctioning pNETs in 2010 was 1.7-fold greater than that in 2005[2]. In the United States, the incidence rate of pNETs increased approximately fourfold from 1973 (0.2 per 100000) to 2012 (0.8 per 100000), and the incidence in Canada also grew steadily during this period[3,4]. pNETs G3 and pancreatic neuroendocrine carcinomas (pNECs) are considered to be rarer than pNETs G1/G2, which may account for 7.5% of all pNENs and have a higher rate in nonfunctioning tumors[2]. In 2010, World Health Organization (WHO) released a grading system for gastroenteropancreatic (GEP) NENs, which classifies the degree of malignancy according to Ki-67 proliferation index and mitotic count. The pNENs were divided into three groups (Table 1): (1) Well-differentiated pNETs G1 [mitoses < 2/10 high-power field (HPF) and Ki67 index < 2%]; (2) well-differentiated pNETs G2 (mitoses 2-20/10 HPF or Ki67 index 2%-20%); and (3) pNECs G3 (mitoses > 20/10 HPF or Ki67 index > 20%). With advances in clinical practice and basic research, especially gene sequencing and analysis, in recent years, it has been gradually recognized that the G3 group contains at least two completely different subtypes, namely the well-differentiated pNETs G3 and poorly differentiated pNECs. Some researchers believe that G3 contains three subtypes[5]. In 2017, WHO revised the grading classification of pNENs (Table 2). The most important change in the new grading classification was to divide the original G3 group into pNETs G3 and pNECs, and other changes, such as a 3% cutoff level of Ki67 index instead of the original 2%, were used in the new classification to distinguish G1 and G2. A significantly higher risk of progression was observed using a 5% cutoff level of Ki67 index in G1 and G2[6]. For the above reasons, WHO uses 3% as the cutoff level of Ki67. The update of the 2017 WHO pNENs grading system was based on the consensus that pNETs G3 and pNECs are completely different in many respects such as biological behavior and treatment options[7]. The newest WHO classification and fifth edition classification of digestive system tumors clarify that the G3 group is divided into NETs G3 and NECs in all NENs of the digestive tract[8].
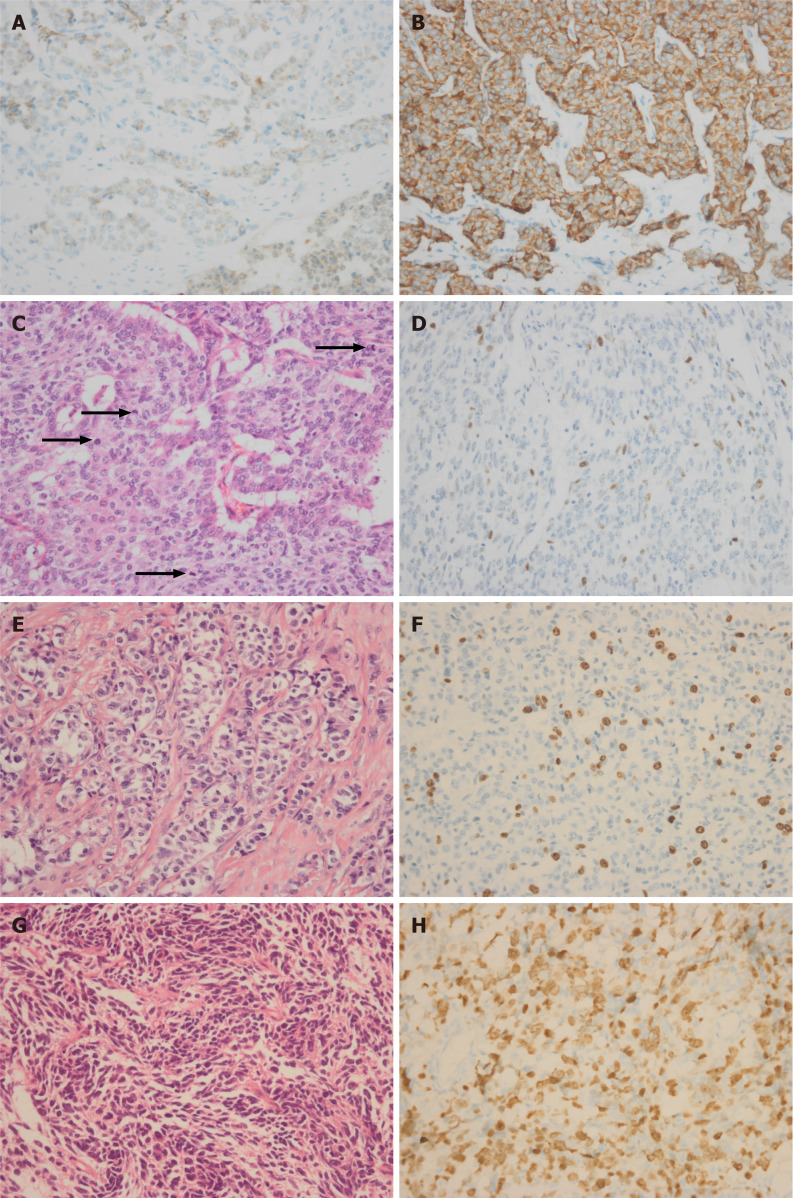
Figure 1.
Immunohistochemical staining (400 ×). A and B: Immunohistochemical positivity for chromogranin A (A) or synaptophysin (B) is considered to be a marker for neuroendocrine neoplasms. Mitotic figures rating [G3 26/10 high-power field (HPF)] is higher than the Ki67 index rating (G2, 16%); C and D: Four mitoses in one HPF is shown by black arrows (C) and related Ki67 immunohistochemical staining is shown in D; E and F: Hemtoxylin and eosin (H&E) staining of well-differentiated pancreatic neuroendocrine tumor G3 (E) and related Ki67 immunohistochemical staining (F); G and H: H&E staining of poorly differentiated pancreatic neuroendocrine carcinoma (G) and related Ki-67 immunohistochemical staining (H).
Table 1.
2010 World Health Organization grading classification of gastroenteropancreatic neuroendocrine neoplasms
| Classification/grade | Mitotic index (per 10 HPF) | Ki67 index (%) |
| G1 (NET G1) | < 2 | < 2 |
| G2 (NET G2) | 2-20 | 2-20 |
| G3 (NEC) | > 20 | > 20 |
HPF: High-power field; NET: Neuroendocrine tumor; NEC: Neuroendocrine carcinoma.
Table 2.
2017 World Health Organization grading classification of pancreatic neuroendocrine neoplasms
| Classification/grade | Mitotic index (per 10 HPF) | Ki67 index (%) |
| pNET | ||
| G1 | < 3 | < 3 |
| G2 | 3–20 | 3–20 |
| G3 | > 20 | > 20 |
| pNEC | ||
| Small cell type | > 20 | > 20 |
| Large cell type | > 20 | > 20 |
HPF: High-power field; pNET: Pancreatic neuroendocrine neoplasm; pNEC: Pancreatic neuroendocrine carcinoma.
Mitotic index is based on evaluation of mitoses in 50 HPF in areas of higher density and is expressed as mitoses per 10 HPF; the Ki-67 index value is determined by counting at least 500 cells in the regions of highest labeling.
HISTOLOGICAL DIFFERENCES
In the 2017 WHO classification, well-differentiated pNENs with mitotic rate > 20/10 HPF or Ki67 index > 20% are defined as pNETs G3. When the Ki67 index rating is inconsistent with the mitosis rate, the higher is considered as a determinant factor. Ki67 is a nuclear protein involved in cell cycle regulation and expressed in all phases of cell duplication. Using the Ki67 antibody, it was possible to perform large-scale studies of pathological biopsy tissues to assess the proliferative activity in different neoplasms[9]. Ki67 index has become one of the most reliable prognostic factors to classify pNENs. It is recommended to count between 500 and 1000 tumor cells in randomly selected fields and the “hot spot” is chosen as the corresponding index. Manual count of a camera-captured, printed image appears to be the most reliable method to determine the Ki-67 index while the “eye-ball” estimation is the method with the poorest reliability and reproducibility. In most cases, the Ki67 index exceeds 20% but the mitotic figures do not exceed 20/10 HPF. Basturk et al[10] showed a group of pNETs G3 with discordance between the mitotic rate and Ki67 index; usually having a Ki67 index in the G3 range but a mitotic rate suggesting G2. The patients with mitotic count G2 and Ki67 index G3 had longer survival time compared to the patients with poorly differentiated NECs (median survival of 54.1 mo vs 11 mo and 5-year survival of 29.1% vs 16.1%) and had shorter survival time than patients with G2 had[10]. There are still a number of cases in which the mitotic figures rate is higher than the Ki67 index rate (Figure 1C and D). Komaç et al[11] reported the Ki67 levels in 29 patients and the mitotic count in only one patient was in the higher grade. The difference between pNETs G3 with mitotic count G2 and pNETs G3 with Ki67 index G2 is not clear at present. The histological features of pNETs G3 are similar to those of well-differentiated pNETs G1/G2, including “salt and pepper” nuclei, cellular uniformity, central ovoid nuclei, variety of architectures such as ribbons or trabeculae, nesting, gyriform, or pseudorosettes (Figure 1E and F). pNETs G3 is considered to be a continuation of pNETs G1 and G2, but completely different from pNECs. The evidence supporting this comes from studies in which it was found that some pNETs G3 patients were first diagnosed as G1 or G2 but rediagnosed as pNETs G3 after the second pathological biopsy after disease progression. In some pNETs, the pathological diagnosis of the primary pancreatic lesion was G1 or G2 but liver metastasis was diagnosed as G3. Panzuto et al[12] retrospectively analyzed 43 patients with sporadic enteropancreatic NENs including 19 NETs G1 patients (44.2%) and 24 NETs G2 patients (55.8%) at the initial histological evaluation. When receiving histological verification after disease progression, 13 patients were reclassified as G1 (30.2%), 26 as G2 (60.5%), and four as G3 (9.3%)[12]. Tang et al[13] reported that the high-grade component occurred either within the primary tumor (48%) or metastatic sites (52%). The radiographic features, clinical presentation, and the genotype of these well-differentiated NETs with high-grade component remained similar to G1/G2[13].
Poorly differentiated pNECs can be histologically divided into small and large cell types. The main histological features of the small cell variant include small cells with large nuclei (high N/C ratio), nuclear molding, dark chromatin with inconspicuous nucleoli and immature or finely speckled chromatin (as seen in pulmonary small cell carcinoma or acute myeloid leukemia). The large cell variant includes large undifferentiated cells with bizarre forms or syncytial aggregates, irregular overlapping nuclei with prominent nucleoli, variable chromatin (fine or coarse) and abundant cytoplasm (delicate, dense or granular)[14] (Figure 1G and H). It is worth noting that pNECs may be combined with other malignant non-neuroendocrine components such as pancreatic adenocarcinoma. If the pancreatic adenocarcinoma component exceeds 30% of neoplastic cells, a diagnosis of composite NECs with adenocarcinoma is made.
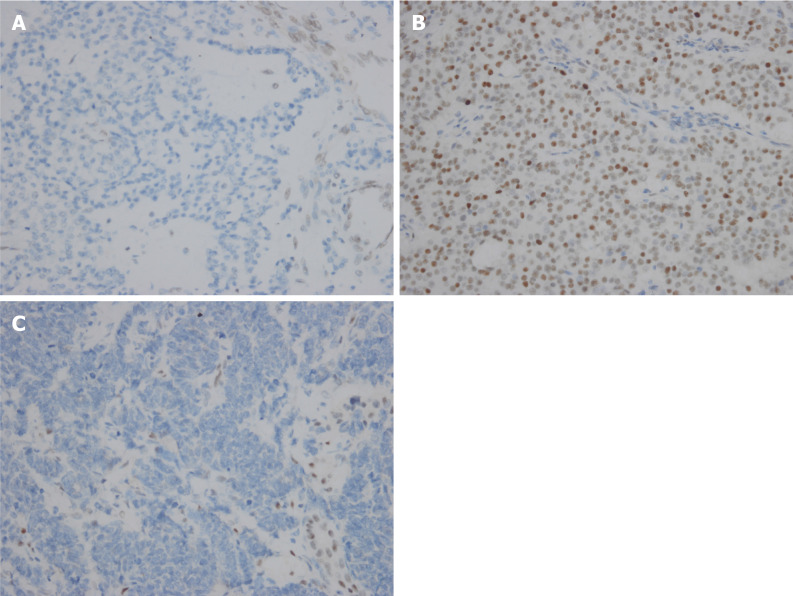
In summary, the histomorphological differences between pNETs G3 and pNECs are obvious. However, in practice some pNETs G3 and pNECs cannot be effectively distinguished. Tang et al[15] reassessed pathological diagnosis of 33 cases of pNETs G3 using the 2010 WHO criteria to classify them into well-differentiated NETs, small cell poorly differentiated NECs, and large cell poorly differentiated NECs. As a result, 20 of 33 cases (61%) had an uncertain diagnosis rendered by any of three pathologists or there was disagreement in classification among the three pathologists[15]. Other supplementary information may help to solve the problem[15,16]. Coexisting lower grade pNETs components in prior specimens tend to diagnose pNETs G3, while coexisting conventional carcinoma components tend to be diagnosed as pNECs. Loss of nuclear expression for DAXX and ATRX and preserved expression for Rb and p53 tend to be diagnosed as pNETs G3 (Figure 2A) and pNECs (Figure 2B and C), respectively. Briest et al[17] reported that nuclear forkhead box (FOX) M1 expression was absent in all 26 NETs G3, whereas 40% (2/5) of NECs specimens displayed high nuclear FOXM1 staining. The cell adhesion molecule L1, which has an important function for development of the nervous system[18], also may be a potential marker for poorly differentiated pNECs[19]. Somatostatin receptors are commonly expressed in pNETs[20,21]. Somatostatin receptors might be considered as a feature of well-differentiated NETs, although somatostatin receptor 2A was also positive in 16% of poorly differentiated NECs and a few poorly differentiated NECs expressed somatostatin receptor 5[22]. Clinical presentation due to excess hormone secretion and elevated plasma neuroendocrine markers (e.g., chromogranin A) help diagnose pNETs G3, and abdominal pain, jaundice, weight loss and elevated plasma carcinoma markers [e.g., carcinoembryonic antigen, carbohydrate antigen (CA)19-9 and CA125] tend to diagnose pNECs. Kang et al[23] reported that low enhancement in the portal venous phase, nonuniform enhancement, and combined tumor vasculature in the hepatic NECs group were larger than those in the hepatic NETs group. In addition, cancer treatment has made significant progress recently with immunotherapy and immune checkpoint inhibitor treatment such as PD-1/PD-L1, which has been approved by the US FDA in recent years. The expression level of PD-L1 in tumor tissues may be related to the efficacy of immune checkpoint inhibitor therapy. Cavalcanti et al[24] showed for the first time that PD-L1 is expressed in GEP NENs, and PD-L1 positivity rate and signal intensity are directly correlated with grade increase from G1 to G3. Whether pNETs G3 differs from pNECs in PD-L1 expression remains to be confirmed.
Figure 2.
Immunohistochemical staining (400 ×). A: Loss of nuclear expression for ATRX in pancreatic neuroendocrine tumor G3; B and C: Aberrant expression for p53 (B) and loss of expression of Rb1 (C) in pancreatic neuroendocrine carcinoma.
DIFFERENCES IN GENETIC AND EPIGENETIC CHARACTERISTICS
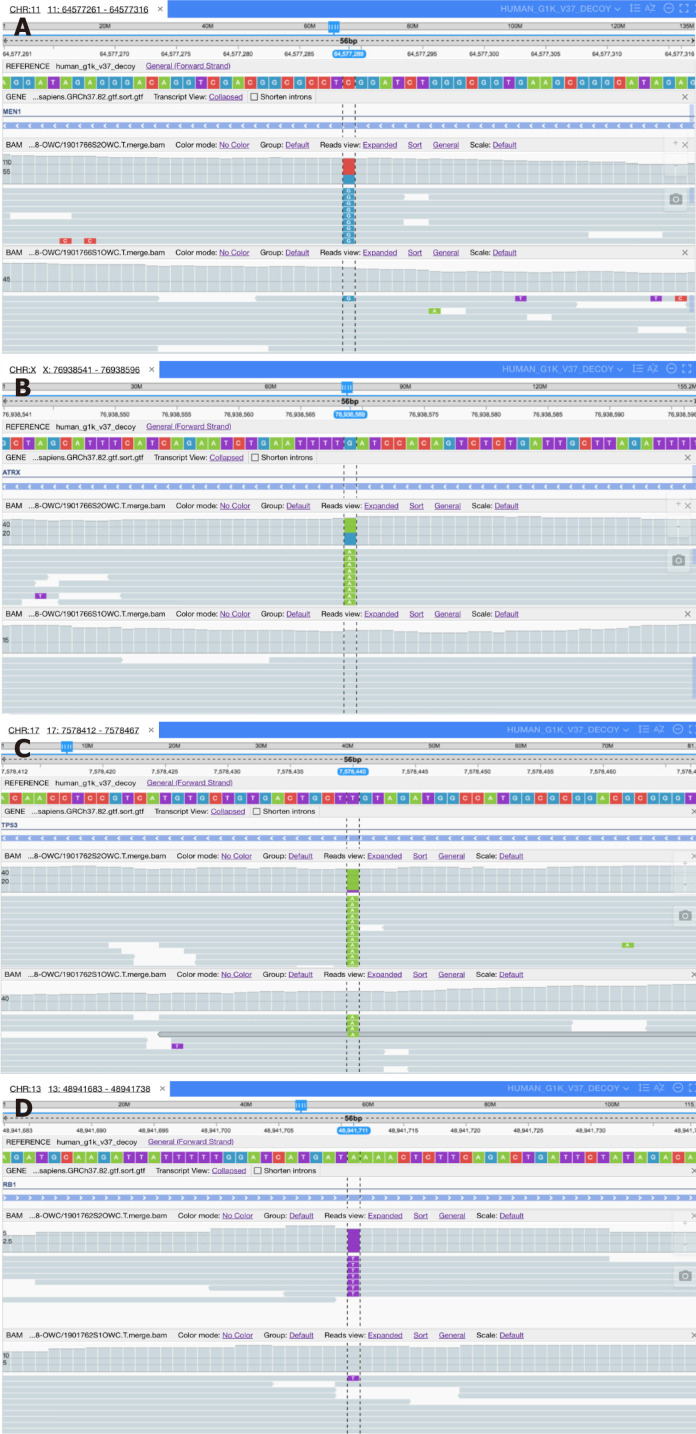
A series of gene sequencing studies in recent years has confirmed that the genetic characteristics of the two groups are different. The data for pNETs G3 gene sequencing is mainly derived from sequencing of pNETs, and the pNETs G3 genome landscape is considered to be similar to that of pNETs G1/G2. In 2010, Jiao et al[25] determined the exomic sequences of 10 nonfamilial pNETs and verified some mutated genes in 58 additional pNETs. Forty-four percent of the tumors had somatic inactivating mutations in MEN1, which encodes menin which is the main cause of multiple endocrine neoplasia type 1 syndrome; 43% had mutations in genes encoding either of the two subunits of a transcription/chromatin remodeling complex, including DAXX and ATRX; and 14% had mutations in genes involved in the mammalian target of rapamycin (mTOR) pathway. For successful identification of genes and pathways, they excluded poorly differentiated small cell and large cell NECs. Heaphy et al[26] evaluated telomere status in pNETs with ATRX or DAXX gene mutations and reported that the mutations were associated with alternative lengthening of telomeres (telomerase independent telomere maintenance mechanism). The DAXX/ATRX complex can negatively regulate the alternative lengthening of telomeres by repressing the HR DNA repair complex[27]. Scarpa et al[28] performed whole-genome sequencing of 102 primary pNETs (mostly pNETs G1/G2) and classified gene mutations into four main pathways: Chromatin remodeling, DNA damage repair, increased mTOR signaling (including undescribed EWSR1 gene fusions), and telomere maintenance. Other research confirmed and expanded their findings[29-31] (Figure 3). In epigenetic epidemiological studies, aberrant DNA methylation of the promoter in RASSF1A and CDKN2A were revealed[32,33]. Guo et al[34] examined the methylation changes of specific gene promoter regions observed in 18 pNETs and showed that the promoters of APC/p73/RAR (33%), p16 INK4a/p15 INK4b/hMLH1/p14 ARF (39%) and BRCA1 (50%) were commonly hypermethylated. The difference in genetic or epigenetic characteristics between pNETs G3 and pNETs G1/G2 deserves further investigation and may help us better understand how some G1/G2 could evolve into pNETs G3.
Figure 3.
High-throughput sequencing. A: Base C (red) mutates to G (blue) on the Men1 gene of chromosome 11 in pancreatic neuroendocrine tumor G3 (pNET G3); B: Base G (blue) mutates to A (green) on the ARTX gene of chromosome X in pNET G3; C: Base T (purple) mutates to A (green) on the Tp53 gene of chromosome 17 in pancreatic neuroendocrine carcinoma (pNEC); D: Base A (green) mutates to T (purple) on the Rb1 gene of chromosome 13 in pNEC.
Compared with pNETs, pNECs is relatively lacking in gene sequencing data. The reasons for this scarcity include that pNECs is rarer than pNETs and it is difficult to obtain surgically resected specimens for sequencing due to the rapid progress at the time of diagnosis. By using targeted exomic sequencing, Yachida et al[35] investigated the genetic features of nine small cell pNECs, 10 large cell pNECs and 11 well-differentiated pNETs. Inactivating mutations in the TP53 and RB1 genes and activating KRAS gene mutation were identified only in pNECs, whereas none of the 11 well-differentiated pNETs showed those mutations[35]. Seventy patients, 21 pNETs G3 and 49 pNECs, were analyzed by Hijioka et al[36] No KRAS mutation was found in pNETs G3, whereas KRAS mutations were detected by PCR gene mutation analysis in 48.7% of pNECs. Vijayvergia et al[37] reported TP53, PIK3CA, RB1 and KRAS gene mutations in four patients with pNECs by next-generation sequencing of 50 cancer-related mutant genes. At the same time, the data from pNECs gene sequencing were used to compare with other NECs in different locations, such as lung, or different pathological types in the same location, such as pancreatic adenocarcinoma. The gene alterations of pNECs are similar to those of pulmonary neuroendocrine cancer, with a few differences[38,39]. Kimura presented a case of pNECs that was genetically similar to invasive ductal adenocarcinoma. Altered KRAS, TP53, and SMAD4/DPC4 suggested that poorly differentiated invasive ductal adenocarcinomas may transform into NECs[40]. Konukiewitz et al[41] conducted a genetic study in 12 pNECs and 11 pNETs G3 using massive parallel sequencing and compared their findings with known data for pancreatic ductal adenocarcinoma. They indicated that pNETs G3 shared singular mutations in five different genes with pNECs, namely, TP53, CDKN2A, ARID1A, LRP1B and APC. Almost half of the pNECs are genetically and phenotypically related to pancreatic ductal adenocarcinoma[41].
DIFFERENCES IN MANAGEMENT
pNECs has a high degree of malignancy and poor prognosis. Although pNETs is better in biological behavior and clinical manifestations, the pNETs G3 and G1/G2 also have the potential for distant metastasis; thus, they should be given adequate attention in clinical management. Other than the WHO 2017 classification of pNENs, TNM staging is important for clinical management. TNM staging has undergone several major improvements too. There once existed two parallel systems. One was administered by European Neuroendocrine Tumor Society (ENETS) in 2006/2007[42,43], and the other was published by American Joint Commission on Cancer (AJCC)/International Union Against Cancer (UICC) in 2009[44]. The definition of the T stages for pNENs is different between the AJCC/UICC and ENETS classifications, and AJCC/UICC applied the same TNM classification for the exocrine pancreatic tumors[45]. Luo et al[46] evaluated the application of these two staging systems using the Surveillance, Epidemiology, and End Results registry and proposed a modified system based on analysis of the two existing classification systems. In 2016, the new AJCC/UICC classification (eighth edition) largely adopted the ENETS classification and was therefore included in the WHO 2017 staging recommendations.
When G3 contained pNECs in the 2010 WHO grading classification, many clinicians raised a lot of questions[47-49]. In fact, pNETs G3 and pNECs should attract completely different management strategies due to the inherent differences mentioned above. For patients with pNETs with early TNM staging, surgery should be recommended. Despite the lack of definitive evidence, pNETs G3 patients without metastatic lesions should undergo curative surgery first. ENETS guidelines suggest that the wait and see approach might be preferred for asymptomatic pNETs G1, with strict patient selection[50]. Compared with pNETs G1/G2, pNETs G3 has a higher risk of distant metastasis, and the wait and see approach may be not well suited. Radical surgery of pNETs comprises typical pancreatic resection (pancreaticoduodenectomy, distal pancreatectomy and total pancreatectomy) and atypical pancreatic resection (middle pancreatectomy, middle-preserving pancreatectomy)[51]. The data on neoadjuvant therapy for patients with pNETs, especially for those radical surgery is difficult to implement at the beginning, is still not available[52]. Neoadjuvant peptide receptor radionuclide therapy for pNENs with high risk of recurrence may be useful[53]. pNETs patients with distant metastasis are generally considered unsuitable for surgical treatment. For pNETs and pNECs, the most likely site for metastasis is the liver, although metastases to other sites have also been reported[54]. Similar to patients with colorectal cancer with resectable liver metastases, pNETs patients with only liver metastases can still undergo partial liver resection[55,56]. Patients with pNETs G3, which is more malignant and more likely to have liver metastases than G1/2, may also benefit from surgical resection of primary lesions and liver metastases[57]. There are still not enough data showing whether pNETs G3 patients with complete surgical resection benefit from postoperative adjuvant treatment, and platinum-based therapy is less effective for pNETs G3 compared with pNECs[36]. For patients with pNETs with metastasis, systemic therapy is the main method. The major methods of systemic treatment include the following: (1) Chemotherapy: Streptozocin- or temozolomide-based therapies have been widely used in first-line chemotherapy before other new systemic treatments appeared[58-60]. Welin et al[61] studied the treatment response in poorly differentiated endocrine carcinomas patients who failed first-line platinum-based chemotherapy to temozolomide alone or in combination with capecitabine and bevacizumab. The results indicated that patients with a Ki67 index < 60%, positive immunohistochemistry for CgA, positive somatostatin receptor scintigraphy, and lack of response to first-line therapy seemed to respond more often to temozolomide-based chemotherapy[61]. In line with this, a randomized trial of NETs G3 is now recruiting to evaluate two regimens: Temozolomide plus capecitabine versus cisplatin plus etoposide; (2) Targeted therapy: With progress of basic research, small-molecule drugs such as mTOR signaling pathway inhibitors and tyrosine kinase inhibitors are considered to be potentially effective for advanced pNETs[62]. Well-differentiated pNETs always have a rich vascular network, which can be targeted by antiangiogenic drugs[63]. mTOR signaling inhibitor everolimus and multi-targeted tyrosine kinase inhibitor sunitinib have been approved for pNENs in the US and Europe[64]. However, the best targeted therapy for pNETs G3 still needs further investigation; (3) Somatostatin analogs are effective in controlling excess hormone secretion in patients with functioning pNETs and they also have antitumor effects[65]. Nevertheless, because of the lack of data for pNETs G3, the use of somatostatin analogs might be restricted to patients with low hepatic tumor involvement and stable disease. The use of somatostatin analogs in pNETs G3 should be undertaken with close monitoring and confirmed high somatostatin receptor expression on related imaging; and (4) Peptide receptor radionuclide therapy: For patients with advanced and well-differentiated pNETs, peptide receptor radiation therapy (PRRT) has evolved from the development of nuclide imaging technology. PRRT has been evaluated in recent years in patients with pNETs G3. Carlsen et al[66] assessed the benefits and adverse effects of PRRT in patients with GEP NENs G3 and showed that PRRT may be beneficial. Zhang et al[67] analyzed the long-term outcome, efficacy, and safety of PRRT in patients with somatostatin-receptor-expressing G3 NENs and reported that PRRT was tolerated well and efficacious, especially in patients with a Ki67 index < 55%, and even in patients for whom chemotherapy had failed.
pNECs has more rapid disease progress and worse prognosis than pNETs G3. Patients with NECs treated with palliative chemotherapy had a median OS of 11.2 mo, compared with 1.7 mo for untreated patients[68]. In contrast to pNETs, the effect of surgical resection in pNECs is not clear. Even in patients with pNECs at an early stage, it is usually difficult to achieve satisfactory results with surgery alone. Most pNECs already have distant metastases when diagnosed and some have recurrence and metastasis < 1 year after surgery. This reflects that the biological behavior of pNECs is highly malignant and existing diagnostic methods fail to identify early metastases, but there are still some patients who achieve long-term survival through aggressive surgery[69]. Based on the treatment paradigm for limited-stage small-cell lung cancer, neoadjuvant chemotherapy and radiation can be considered in many GEP NECs patients, particularly when radical resection is difficult. However, pNECs often progresses rapidly, and neoadjuvant therapy may cause these patients to completely lose their surgical opportunities during treatment. No studies have examined adjuvant postoperative treatment for pNECs. Nevertheless, the aggressive behavior and high rate of recurrence of pNECs have led to consideration of adjuvant treatment, using platinum-based chemotherapy, and also in SCLC[70].
Chemotherapy has become the main treatment for advanced pNECs. GEP NECs currently has no standard chemotherapy regimen. Based on SCLC treatment, platinum-based combination regimens are widely used as first-line treatment[71]. In pNECs, the association of cisplatin and etoposide is the first chemotherapeutic option[72]. However, as mentioned above, the SCLC genetic characteristics are different from those of pNECs, therefor the optimal first-line chemotherapy regimen for pNECs is still uncertain. Whole brain prophylactic radiotherapy is not needed due to rare brain metastases in patients with pNECs[73]. Second-line chemotherapy options are also being explored[74-76]. PRRT is often thought to be an effective, well-tolerated treatment for well-differentiated NETs, but some patients with poorly differentiated pNECs may also benefit[77,78]. Although the application of new molecular targeted drugs has brought success to treatment of various types of tumors, no new targeted drugs have been approved for pNECs. The reasons for that might be due to the underlying genetic changes or pathway of these diseases and too few patients with pNECs, which has prevented effective prospective clinical trials. New clinical trials of targeted drugs for SCLC are underway, which provides some hope for pNECs. In recent years, the rise of immunotherapy and other treatments combined with immunotherapy have brought new expectations to pNECs. As mentioned before, pNECs or pNETs G3 is characterized by strong PD-L1 expression in both the tumor and infiltrating immune cells; therefore, immunotherapy may provide longer overall survival and better quality of life[79,80].
CONCLUSION
This review summarizes the differences between pNETs G3 and pNECs, which is the major update in latest WHO grading classification for pNENs, in relation to histology, gene mutations, and clinical management. pNENs are rare and heterogeneous, and previously ignored tumors, but have lately attracted increasing interest by researchers. More information and ideas are constantly emerging with a focus on these specific tumors. In 2017, WHO updated the classification of pNENs, and the G3 group in the 2010 version was divided into pNETs G3 and pNECs, which differ from each other, according to evidence from basic research and clinical practice. Therefore, a lot of past research data deserve re-examination and new prospective research using the new classification is urgently needed. In addition, many questions remain about pNETs G3 and pNECs. For example, some gene sequencing data from pNETs G3 and pNECs are available but not sufficient to identify biomarkers that can guide clinical management and development of new drugs. New sequencing technologies and analysis methods may help, such as single-cell sequencing, which provides better understanding of the function of individual cells and circulating tumor DNA that can reflect the entire tumor genome and monitor tumor progression at various time points. The management options for pNETs G3 and pNECs are still limited. The existing clinical evidence about these tumors, especially pNECs, is basically derived from individual cases or retrospective studies. This may be related to the rare incidence of pNETs G3 and pNECs. To solve this problem, cooperation should be strengthened, and resources should be integrated. Establishment of the models in vitro and in vivo may also be of help[81,82].
Footnotes
Conflict-of-interest statement: Authors declare no conflict of interests for this article.
Manuscript source: Invited manuscript
Peer-review started: December 31, 2019
First decision: May 5, 2020
Article in press: June 17, 2020
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Casadei R S-Editor: Dou Y L-Editor: A E-Editor: Liu MY
Contributor Information
Ming-Yi Zhang, Department of Biotherapy, State Key Laboratory of Biotherapy and Cancer Center, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China.
Du He, Department of Pathology, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China.
Shuang Zhang, Department of Biotherapy, State Key Laboratory of Biotherapy and Cancer Center, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China. shuang.zhang@scu.edu.cn.
References
- 1.Fesinmeyer MD, Austin MA, Li CI, De Roos AJ, Bowen DJ. Differences in survival by histologic type of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2005;14:1766–1773. doi: 10.1158/1055-9965.EPI-05-0120. [DOI] [PubMed] [Google Scholar]
- 2.Ito T, Igarashi H, Nakamura K, Sasano H, Okusaka T, Takano K, Komoto I, Tanaka M, Imamura M, Jensen RT, Takayanagi R, Shimatsu A. Epidemiological trends of pancreatic and gastrointestinal neuroendocrine tumors in Japan: a nationwide survey analysis. J Gastroenterol. 2015;50:58–64. doi: 10.1007/s00535-014-0934-2. [DOI] [PubMed] [Google Scholar]
- 3.Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, Shih T, Yao JC. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients With Neuroendocrine Tumors in the United States. JAMA Oncol. 2017;3:1335–1342. doi: 10.1001/jamaoncol.2017.0589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hallet J, Law CH, Cukier M, Saskin R, Liu N, Singh S. Exploring the rising incidence of neuroendocrine tumors: a population-based analysis of epidemiology, metastatic presentation, and outcomes. Cancer. 2015;121:589–597. doi: 10.1002/cncr.29099. [DOI] [PubMed] [Google Scholar]
- 5.Milione M, Maisonneuve P, Spada F, Pellegrinelli A, Spaggiari P, Albarello L, Pisa E, Barberis M, Vanoli A, Buzzoni R, Pusceddu S, Concas L, Sessa F, Solcia E, Capella C, Fazio N, La Rosa S. The Clinicopathologic Heterogeneity of Grade 3 Gastroenteropancreatic Neuroendocrine Neoplasms: Morphological Differentiation and Proliferation Identify Different Prognostic Categories. Neuroendocrinology. 2017;104:85–93. doi: 10.1159/000445165. [DOI] [PubMed] [Google Scholar]
- 6.Rindi G, Falconi M, Klersy C, Albarello L, Boninsegna L, Buchler MW, Capella C, Caplin M, Couvelard A, Doglioni C, Delle Fave G, Fischer L, Fusai G, de Herder WW, Jann H, Komminoth P, de Krijger RR, La Rosa S, Luong TV, Pape U, Perren A, Ruszniewski P, Scarpa A, Schmitt A, Solcia E, Wiedenmann B. TNM staging of neoplasms of the endocrine pancreas: results from a large international cohort study. J Natl Cancer Inst. 2012;104:764–777. doi: 10.1093/jnci/djs208. [DOI] [PubMed] [Google Scholar]
- 7.Rindi G, Klimstra DS, Abedi-Ardekani B, Asa SL, Bosman FT, Brambilla E, Busam KJ, de Krijger RR, Dietel M, El-Naggar AK, Fernandez-Cuesta L, Klöppel G, McCluggage WG, Moch H, Ohgaki H, Rakha EA, Reed NS, Rous BA, Sasano H, Scarpa A, Scoazec JY, Travis WD, Tallini G, Trouillas J, van Krieken JH, Cree IA. A common classification framework for neuroendocrine neoplasms: an International Agency for Research on Cancer (IARC) and World Health Organization (WHO) expert consensus proposal. Mod Pathol. 2018;31:1770–1786. doi: 10.1038/s41379-018-0110-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, Washington KM, Carneiro F, Cree IA WHO Classification of Tumours Editorial Board. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182–188. doi: 10.1111/his.13975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gerdes J, Schwab U, Lemke H, Stein H. Production of a mouse monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer. 1983;31:13–20. doi: 10.1002/ijc.2910310104. [DOI] [PubMed] [Google Scholar]
- 10.Basturk O, Yang Z, Tang LH, Hruban RH, Adsay V, McCall CM, Krasinskas AM, Jang KT, Frankel WL, Balci S, Sigel C, Klimstra DS. The high-grade (WHO G3) pancreatic neuroendocrine tumor category is morphologically and biologically heterogenous and includes both well differentiated and poorly differentiated neoplasms. Am J Surg Pathol. 2015;39:683–690. doi: 10.1097/PAS.0000000000000408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Komaç Ö, Bengi G, Sağol Ö, Akarsu M. C-reactive protein may be a prognostic factor for the whole gastroenteropancreatic neuroendocrine tumor group. World J Gastrointest Oncol. 2019;11:139–152. doi: 10.4251/wjgo.v11.i2.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Panzuto F, Cicchese N, Partelli S, Rinzivillo M, Capurso G, Merola E, Manzoni M, Pucci E, Iannicelli E, Pilozzi E, Rossi M, Doglioni C, Falconi M, Delle Fave G. Impact of Ki67 re-assessment at time of disease progression in patients with pancreatic neuroendocrine neoplasms. PLoS One. 2017;12:e0179445. doi: 10.1371/journal.pone.0179445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tang LH, Untch BR, Reidy DL, O'Reilly E, Dhall D, Jih L, Basturk O, Allen PJ, Klimstra DS. Well-Differentiated Neuroendocrine Tumors with a Morphologically Apparent High-Grade Component: A Pathway Distinct from Poorly Differentiated Neuroendocrine Carcinomas. Clin Cancer Res. 2016;22:1011–1017. doi: 10.1158/1078-0432.CCR-15-0548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Basturk O, Tang L, Hruban RH, Adsay V, Yang Z, Krasinskas AM, Vakiani E, La Rosa S, Jang KT, Frankel WL, Liu X, Zhang L, Giordano TJ, Bellizzi AM, Chen JH, Shi C, Allen P, Reidy DL, Wolfgang CL, Saka B, Rezaee N, Deshpande V, Klimstra DS. Poorly differentiated neuroendocrine carcinomas of the pancreas: a clinicopathologic analysis of 44 cases. Am J Surg Pathol. 2014;38:437–447. doi: 10.1097/PAS.0000000000000169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tang LH, Basturk O, Sue JJ, Klimstra DS. A Practical Approach to the Classification of WHO Grade 3 (G3) Well-differentiated Neuroendocrine Tumor (WD-NET) and Poorly Differentiated Neuroendocrine Carcinoma (PD-NEC) of the Pancreas. Am J Surg Pathol. 2016;40:1192–1202. doi: 10.1097/PAS.0000000000000662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Singhi AD, Klimstra DS. Well-differentiated pancreatic neuroendocrine tumours (PanNETs) and poorly differentiated pancreatic neuroendocrine carcinomas (PanNECs): concepts, issues and a practical diagnostic approach to high-grade (G3) cases. Histopathology. 2018;72:168–177. doi: 10.1111/his.13408. [DOI] [PubMed] [Google Scholar]
- 17.Briest F, Konukiewitz M, Anlauf M, Klöppel G, Grabowski P, Sipos B. Expression of FOXM1 in G3 neuroendocrine tumors (NET G3) and G3 neuroendocrine carcinomas (NEC G3) of the pancreas and the intestine; 15th Annual ENETS conference; 2018 Mar 7-9; Barcelona, Spain. [Google Scholar]
- 18.Schachner M. Neural recognition molecules and synaptic plasticity. Curr Opin Cell Biol. 1997;9:627–634. doi: 10.1016/s0955-0674(97)80115-9. [DOI] [PubMed] [Google Scholar]
- 19.Kaifi JT, Zinnkann U, Yekebas EF, Schurr PG, Reichelt U, Wachowiak R, Fiegel HC, Petri S, Schachner M, Izbicki JR. L1 is a potential marker for poorly-differentiated pancreatic neuroendocrine carcinoma. World J Gastroenterol. 2006;12:94–98. doi: 10.3748/wjg.v12.i1.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kajtazi Y, Kaemmerer D, Sänger J, Schulz S, Lupp A. Somatostatin and chemokine CXCR4 receptor expression in pancreatic adenocarcinoma relative to pancreatic neuroendocrine tumours. J Cancer Res Clin Oncol. 2019;145:2481–2493. doi: 10.1007/s00432-019-03011-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Herrera-Martínez AD, Gahete MD, Pedraza-Arevalo S, Sánchez-Sánchez R, Ortega-Salas R, Serrano-Blanch R, Luque RM, Gálvez-Moreno MA, Castaño JP. Clinical and functional implication of the components of somatostatin system in gastroenteropancreatic neuroendocrine tumors. Endocrine. 2018;59:426–437. doi: 10.1007/s12020-017-1482-3. [DOI] [PubMed] [Google Scholar]
- 22.Konukiewitz B, Schlitter AM, Jesinghaus M, Pfister D, Steiger K, Segler A, Agaimy A, Sipos B, Zamboni G, Weichert W, Esposito I, Pfarr N, Klöppel G. Somatostatin receptor expression related to TP53 and RB1 alterations in pancreatic and extrapancreatic neuroendocrine neoplasms with a Ki67-index above 20. Mod Pathol. 2017;30:587–598. doi: 10.1038/modpathol.2016.217. [DOI] [PubMed] [Google Scholar]
- 23.Kang XN, Zhang XY, Bai J, Wang ZY, Yin WJ, Li L. Analysis of B-ultrasound and contrast-enhanced ultrasound characteristics of different hepatic neuroendocrine neoplasm. World J Gastrointest Oncol. 2019;11:436–448. doi: 10.4251/wjgo.v11.i5.436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cavalcanti E, Armentano R, Valentini AM, Chieppa M, Caruso ML. Role of PD-L1 expression as a biomarker for GEP neuroendocrine neoplasm grading. Cell Death Dis. 2017;8:e3004. doi: 10.1038/cddis.2017.401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jiao Y, Shi C, Edil BH, de Wilde RF, Klimstra DS, Maitra A, Schulick RD, Tang LH, Wolfgang CL, Choti MA, Velculescu VE, Diaz LA, Jr, Vogelstein B, Kinzler KW, Hruban RH, Papadopoulos N. DAXX/ATRX, MEN1, and mTOR pathway genes are frequently altered in pancreatic neuroendocrine tumors. Science. 2011;331:1199–1203. doi: 10.1126/science.1200609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Heaphy CM, de Wilde RF, Jiao Y, Klein AP, Edil BH, Shi C, Bettegowda C, Rodriguez FJ, Eberhart CG, Hebbar S, Offerhaus GJ, McLendon R, Rasheed BA, He Y, Yan H, Bigner DD, Oba-Shinjo SM, Marie SK, Riggins GJ, Kinzler KW, Vogelstein B, Hruban RH, Maitra A, Papadopoulos N, Meeker AK. Altered telomeres in tumors with ATRX and DAXX mutations. Science. 2011;333:425. doi: 10.1126/science.1207313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pickett HA, Reddel RR Australian Pancreatic Cancer Genome Initiative, Khanna KK, Gill AJ, Wheeler DA, Gibbs RA, Musgrove EA, Bassi C, Tortora G, Pederzoli P, Pearson JV, Waddell N, Biankin AV, Grimmond SM. Molecular mechanisms of activity and derepression of alternative lengthening of telomeres. Nat Struct Mol Biol. 2015;22:875–880. doi: 10.1038/nsmb.3106. [DOI] [PubMed] [Google Scholar]
- 28.Scarpa A, Chang DK, Nones K, Corbo V, Patch AM, Bailey P, Lawlor RT, Johns AL, Miller DK, Mafficini A, Rusev B, Scardoni M, Antonello D, Barbi S, Sikora KO, Cingarlini S, Vicentini C, McKay S, Quinn MC, Bruxner TJ, Christ AN, Harliwong I, Idrisoglu S, McLean S, Nourse C, Nourbakhsh E, Wilson PJ, Anderson MJ, Fink JL, Newell F, Waddell N, Holmes O, Kazakoff SH, Leonard C, Wood S, Xu Q, Nagaraj SH, Amato E, Dalai I, Bersani S, Cataldo I, Dei Tos AP, Capelli P, Davì MV, Landoni L, Malpaga A, Miotto M, Whitehall VL, Leggett BA, Harris JL, Harris J, Jones MD, Humphris J, Chantrill LA, Chin V, Nagrial AM, Pajic M, Scarlett CJ, Pinho A, Rooman I, Toon C, Wu J, Pinese M, Cowley M, Barbour A, Mawson A, Humphrey ES, Colvin EK, Chou A, Lovell JA, Jamieson NB, Duthie F, Gingras MC, Fisher WE, Dagg RA, Lau LM, Lee M, Pickett HA, Reddel RR, Samra JS, Kench JG, Merrett ND, Epari K, Nguyen NQ, Zeps N, Falconi M, Simbolo M, Butturini G, Van Buren G, Partelli S, Fassan M. Whole-genome landscape of pancreatic neuroendocrine tumours. Nature. 2017;543:65–71. doi: 10.1038/nature21063. [DOI] [PubMed] [Google Scholar]
- 29.Ji S, Yang W, Liu J, Zhao J, Chen L, Ni Q, Long J, Yu X. High throughput gene sequencing reveals altered landscape in DNA damage responses and chromatin remodeling in sporadic pancreatic neuroendocrine tumors. Pancreatology. 2018;18:318–327. doi: 10.1016/j.pan.2018.01.006. [DOI] [PubMed] [Google Scholar]
- 30.Corbo V, Beghelli S, Bersani S, Antonello D, Talamini G, Brunelli M, Capelli P, Falconi M, Scarpa A. Pancreatic endocrine tumours: mutational and immunohistochemical survey of protein kinases reveals alterations in targetable kinases in cancer cell lines and rare primaries. Ann Oncol. 2012;23:127–134. doi: 10.1093/annonc/mdr048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vandamme T, Beyens M, Boons G, Schepers A, Kamp K, Biermann K, Pauwels P, De Herder WW, Hofland LJ, Peeters M, Van Camp G, Op de Beeck K. Hotspot DAXX, PTCH2 and CYFIP2 mutations in pancreatic neuroendocrine neoplasms. Endocr Relat Cancer. 2019;26:1–12. doi: 10.1530/ERC-18-0120. [DOI] [PubMed] [Google Scholar]
- 32.Dammann R, Schagdarsurengin U, Liu L, Otto N, Gimm O, Dralle H, Boehm BO, Pfeifer GP, Hoang-Vu C. Frequent RASSF1A promoter hypermethylation and K-ras mutations in pancreatic carcinoma. Oncogene. 2003;22:3806–3812. doi: 10.1038/sj.onc.1206582. [DOI] [PubMed] [Google Scholar]
- 33.Karpathakis A, Dibra H, Thirlwell C. Neuroendocrine tumours: cracking the epigenetic code. Endocr Relat Cancer. 2013;20:R65–R82. doi: 10.1530/ERC-12-0338. [DOI] [PubMed] [Google Scholar]
- 34.Guo M, Jia Y, Yu Z, House MG, Esteller M, Brock MV, Herman JG. Epigenetic changes associated with neoplasms of the exocrine and endocrine pancreas. Discov Med. 2014;17:67–73. [PMC free article] [PubMed] [Google Scholar]
- 35.Yachida S, Vakiani E, White CM, Zhong Y, Saunders T, Morgan R, de Wilde RF, Maitra A, Hicks J, Demarzo AM, Shi C, Sharma R, Laheru D, Edil BH, Wolfgang CL, Schulick RD, Hruban RH, Tang LH, Klimstra DS, Iacobuzio-Donahue CA. Small cell and large cell neuroendocrine carcinomas of the pancreas are genetically similar and distinct from well-differentiated pancreatic neuroendocrine tumors. Am J Surg Pathol. 2012;36:173–184. doi: 10.1097/PAS.0b013e3182417d36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hijioka S, Hosoda W, Matsuo K, Ueno M, Furukawa M, Yoshitomi H, Kobayashi N, Ikeda M, Ito T, Nakamori S, Ishii H, Kodama Y, Morizane C, Okusaka T, Yanagimoto H, Notohara K, Taguchi H, Kitano M, Yane K, Maguchi H, Tsuchiya Y, Komoto I, Tanaka H, Tsuji A, Hashigo S, Kawaguchi Y, Mine T, Kanno A, Murohisa G, Miyabe K, Takagi T, Matayoshi N, Yoshida T, Hara K, Imamura M, Furuse J, Yatabe Y, Mizuno N. Rb Loss and KRAS Mutation Are Predictors of the Response to Platinum-Based Chemotherapy in Pancreatic Neuroendocrine Neoplasm with Grade 3: A Japanese Multicenter Pancreatic NEN-G3 Study. Clin Cancer Res. 2017;23:4625–4632. doi: 10.1158/1078-0432.CCR-16-3135. [DOI] [PubMed] [Google Scholar]
- 37.Vijayvergia N, Boland PM, Handorf E, Gustafson KS, Gong Y, Cooper HS, Sheriff F, Astsaturov I, Cohen SJ, Engstrom PF. Molecular profiling of neuroendocrine malignancies to identify prognostic and therapeutic markers: a Fox Chase Cancer Center Pilot Study. Br J Cancer. 2016;115:564–570. doi: 10.1038/bjc.2016.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dreijerink KM, Derks JL, Cataldo I, Scarpa A, Valk GD, Speel EJ. Genetics and Epigenetics of Pancreatic Neuroendocrine Tumors and Pulmonary Carcinoids. Front Horm Res. 2015;44:115–138. doi: 10.1159/000382138. [DOI] [PubMed] [Google Scholar]
- 39.George J, Lim JS, Jang SJ, Cun Y, Ozretić L, Kong G, Leenders F, Lu X, Fernández-Cuesta L, Bosco G, Müller C, Dahmen I, Jahchan NS, Park KS, Yang D, Karnezis AN, Vaka D, Torres A, Wang MS, Korbel JO, Menon R, Chun SM, Kim D, Wilkerson M, Hayes N, Engelmann D, Pützer B, Bos M, Michels S, Vlasic I, Seidel D, Pinther B, Schaub P, Becker C, Altmüller J, Yokota J, Kohno T, Iwakawa R, Tsuta K, Noguchi M, Muley T, Hoffmann H, Schnabel PA, Petersen I, Chen Y, Soltermann A, Tischler V, Choi CM, Kim YH, Massion PP, Zou Y, Jovanovic D, Kontic M, Wright GM, Russell PA, Solomon B, Koch I, Lindner M, Muscarella LA, la Torre A, Field JK, Jakopovic M, Knezevic J, Castaños-Vélez E, Roz L, Pastorino U, Brustugun OT, Lund-Iversen M, Thunnissen E, Köhler J, Schuler M, Botling J, Sandelin M, Sanchez-Cespedes M, Salvesen HB, Achter V, Lang U, Bogus M, Schneider PM, Zander T, Ansén S, Hallek M, Wolf J, Vingron M, Yatabe Y, Travis WD, Nürnberg P, Reinhardt C, Perner S, Heukamp L, Büttner R, Haas SA, Brambilla E, Peifer M, Sage J, Thomas RK. Comprehensive genomic profiles of small cell lung cancer. Nature. 2015;524:47–53. doi: 10.1038/nature14664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kimura T, Miyamoto H, Fukuya A, Kitamura S, Okamoto K, Kimura M, Muguruma N, Ikemoto T, Shimada M, Yoneda A, Bando Y, Takishita M, Takayama T. Neuroendocrine carcinoma of the pancreas with similar genetic alterations to invasive ductal adenocarcinoma. Clin J Gastroenterol. 2016;9:261–265. doi: 10.1007/s12328-016-0655-6. [DOI] [PubMed] [Google Scholar]
- 41.Konukiewitz B, Jesinghaus M, Steiger K, Schlitter AM, Kasajima A, Sipos B, Zamboni G, Weichert W, Pfarr N, Klöppel G. Pancreatic neuroendocrine carcinomas reveal a closer relationship to ductal adenocarcinomas than to neuroendocrine tumors G3. Hum Pathol. 2018;77:70–79. doi: 10.1016/j.humpath.2018.03.018. [DOI] [PubMed] [Google Scholar]
- 42.Rindi G, Klöppel G, Alhman H, Caplin M, Couvelard A, de Herder WW, Erikssson B, Falchetti A, Falconi M, Komminoth P, Körner M, Lopes JM, McNicol AM, Nilsson O, Perren A, Scarpa A, Scoazec JY, Wiedenmann B all other Frascati Consensus Conference participants; European Neuroendocrine Tumor Society (ENETS) TNM staging of foregut (neuro)endocrine tumors: a consensus proposal including a grading system. Virchows Arch. 2006;449:395–401. doi: 10.1007/s00428-006-0250-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rindi G, Klöppel G, Couvelard A, Komminoth P, Körner M, Lopes JM, McNicol AM, Nilsson O, Perren A, Scarpa A, Scoazec JY, Wiedenmann B. TNM staging of midgut and hindgut (neuro) endocrine tumors: a consensus proposal including a grading system. Virchows Arch. 2007;451:757–762. doi: 10.1007/s00428-007-0452-1. [DOI] [PubMed] [Google Scholar]
- 44.Sobin LH, Gospodarowicz MK, Wittekind C. UICC: TNM classification of malignant tumours, 7th ed. Wiley-Blackwell: Oxford, 2009. [Google Scholar]
- 45.Klöppel G, Rindi G, Perren A, Komminoth P, Klimstra DS. The ENETS and AJCC/UICC TNM classifications of the neuroendocrine tumors of the gastrointestinal tract and the pancreas: a statement. Virchows Arch. 2010;456:595–597. doi: 10.1007/s00428-010-0924-6. [DOI] [PubMed] [Google Scholar]
- 46.Luo G, Javed A, Strosberg JR, Jin K, Zhang Y, Liu C, Xu J, Soares K, Weiss MJ, Zheng L, Wolfgang CL, Cives M, Wong J, Wang W, Sun J, Shao C, Wang W, Tan H, Li J, Ni Q, Shen L, Chen M, He J, Chen J, Yu X. Modified Staging Classification for Pancreatic Neuroendocrine Tumors on the Basis of the American Joint Committee on Cancer and European Neuroendocrine Tumor Society Systems. J Clin Oncol. 2017;35:274–280. doi: 10.1200/JCO.2016.67.8193. [DOI] [PubMed] [Google Scholar]
- 47.Hijioka S, Hosoda W, Mizuno N, Hara K, Imaoka H, Bhatia V, Mekky MA, Tajika M, Tanaka T, Ishihara M, Yogi T, Tsutumi H, Fujiyoshi T, Sato T, Hieda N, Yoshida T, Okuno N, Shimizu Y, Yatabe Y, Niwa Y, Yamao K. Does the WHO 2010 classification of pancreatic neuroendocrine neoplasms accurately characterize pancreatic neuroendocrine carcinomas? J Gastroenterol. 2015;50:564–572. doi: 10.1007/s00535-014-0987-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Raj N, Valentino E, Capanu M, Tang LH, Basturk O, Untch BR, Allen PJ, Klimstra DS, Reidy-Lagunes D. Treatment Response and Outcomes of Grade 3 Pancreatic Neuroendocrine Neoplasms Based on Morphology: Well Differentiated Versus Poorly Differentiated. Pancreas. 2017;46:296–301. doi: 10.1097/MPA.0000000000000735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Vélayoudom-Céphise FL, Duvillard P, Foucan L, Hadoux J, Chougnet CN, Leboulleux S, Malka D, Guigay J, Goere D, Debaere T, Caramella C, Schlumberger M, Planchard D, Elias D, Ducreux M, Scoazec JY, Baudin E. Are G3 ENETS neuroendocrine neoplasms heterogeneous? Endocr Relat Cancer. 2013;20:649–657. doi: 10.1530/ERC-13-0027. [DOI] [PubMed] [Google Scholar]
- 50.Falconi M, Zerbi A, Crippa S, Balzano G, Boninsegna L, Capitanio V, Bassi C, Di Carlo V, Pederzoli P. Parenchyma-preserving resections for small nonfunctioning pancreatic endocrine tumors. Ann Surg Oncol. 2010;17:1621–1627. doi: 10.1245/s10434-010-0949-8. [DOI] [PubMed] [Google Scholar]
- 51.Maurizi A, Partelli S, Falconi M. Pancreatic Surgery. Front Horm Res. 2015;44:139–148. doi: 10.1159/000382143. [DOI] [PubMed] [Google Scholar]
- 52.Ambe CM, Nguyen P, Centeno BA, Choi J, Strosberg J, Kvols L, Hodul P, Hoffe S, Malafa MP. Multimodality Management of "Borderline Resectable" Pancreatic Neuroendocrine Tumors: Report of a Single-Institution Experience. Cancer Control. 2017;24:1073274817729076. doi: 10.1177/1073274817729076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Partelli S, Bertani E, Bartolomei M, Perali C, Muffatti F, Grana CM, Schiavo Lena M, Doglioni C, Crippa S, Fazio N, Zamboni G, Falconi M. Peptide receptor radionuclide therapy as neoadjuvant therapy for resectable or potentially resectable pancreatic neuroendocrine neoplasms. Surgery. 2018;163:761–767. doi: 10.1016/j.surg.2017.11.007. [DOI] [PubMed] [Google Scholar]
- 54.Ghetie C, Cornfeld D, Ramfidis VS, Syrigos KN, Saif MW. Bone lesions in recurrent glucagonoma: A case report and review of literature. World J Gastrointest Oncol. 2012;4:152–155. doi: 10.4251/wjgo.v4.i6.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Touzios JG, Kiely JM, Pitt SC, Rilling WS, Quebbeman EJ, Wilson SD, Pitt HA. Neuroendocrine hepatic metastases: does aggressive management improve survival? Ann Surg. 2005;241:776–783; discussion 783-785. doi: 10.1097/01.sla.0000161981.58631.ab. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fendrich V, Langer P, Celik I, Bartsch DK, Zielke A, Ramaswamy A, Rothmund M. An aggressive surgical approach leads to long-term survival in patients with pancreatic endocrine tumors. Ann Surg. 2006;244:845–851; discussion 852-853. doi: 10.1097/01.sla.0000246951.21252.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Birnbaum DJ, Turrini O, Vigano L, Russolillo N, Autret A, Moutardier V, Capussotti L, Le Treut YP, Delpero JR, Hardwigsen J. Surgical management of advanced pancreatic neuroendocrine tumors: short-term and long-term results from an international multi-institutional study. Ann Surg Oncol. 2015;22:1000–1007. doi: 10.1245/s10434-014-4016-8. [DOI] [PubMed] [Google Scholar]
- 58.Kouvaraki MA, Ajani JA, Hoff P, Wolff R, Evans DB, Lozano R, Yao JC. Fluorouracil, doxorubicin, and streptozocin in the treatment of patients with locally advanced and metastatic pancreatic endocrine carcinomas. J Clin Oncol. 2004;22:4762–4771. doi: 10.1200/JCO.2004.04.024. [DOI] [PubMed] [Google Scholar]
- 59.Delaunoit T, Ducreux M, Boige V, Dromain C, Sabourin JC, Duvillard P, Schlumberger M, de Baere T, Rougier P, Ruffie P, Elias D, Lasser P, Baudin E. The doxorubicin-streptozotocin combination for the treatment of advanced well-differentiated pancreatic endocrine carcinoma; a judicious option? Eur J Cancer. 2004;40:515–520. doi: 10.1016/j.ejca.2003.09.035. [DOI] [PubMed] [Google Scholar]
- 60.Kulke MH, Stuart K, Enzinger PC, Ryan DP, Clark JW, Muzikansky A, Vincitore M, Michelini A, Fuchs CS. Phase II study of temozolomide and thalidomide in patients with metastatic neuroendocrine tumors. J Clin Oncol. 2006;24:401–406. doi: 10.1200/JCO.2005.03.6046. [DOI] [PubMed] [Google Scholar]
- 61.Welin S, Sorbye H, Sebjornsen S, Knappskog S, Busch C, Oberg K. Clinical effect of temozolomide-based chemotherapy in poorly differentiated endocrine carcinoma after progression on first-line chemotherapy. Cancer. 2011;117:4617–4622. doi: 10.1002/cncr.26124. [DOI] [PubMed] [Google Scholar]
- 62.Yao JC, Shah MH, Ito T, Bohas CL, Wolin EM, Van Cutsem E, Hobday TJ, Okusaka T, Capdevila J, de Vries EG, Tomassetti P, Pavel ME, Hoosen S, Haas T, Lincy J, Lebwohl D, Öberg K RAD001 in Advanced Neuroendocrine Tumors, Third Trial (RADIANT-3) Study Group. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:514–523. doi: 10.1056/NEJMoa1009290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhang J, Francois R, Iyer R, Seshadri M, Zajac-Kaye M, Hochwald SN. Current understanding of the molecular biology of pancreatic neuroendocrine tumors. J Natl Cancer Inst. 2013;105:1005–1017. doi: 10.1093/jnci/djt135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas C, Valle J, Metrakos P, Smith D, Vinik A, Chen JS, Hörsch D, Hammel P, Wiedenmann B, Van Cutsem E, Patyna S, Lu DR, Blanckmeister C, Chao R, Ruszniewski P. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:501–513. doi: 10.1056/NEJMoa1003825. [DOI] [PubMed] [Google Scholar]
- 65.Baudin E, Planchard D, Scoazec JY, Guigay J, Dromain C, Hadoux J, Debaere T, Elias D, Ducreux M. Intervention in gastro-enteropancreatic neuroendocrine tumours. Best Pract Res Clin Gastroenterol. 2012;26:855–865. doi: 10.1016/j.bpg.2013.01.008. [DOI] [PubMed] [Google Scholar]
- 66.Carlsen EA, Fazio N, Granberg D, Grozinsky-Glasberg S, Ahmadzadehfar H, Grana CM, Zandee WT, Cwikla J, Walter MA, Oturai PS, Rinke A, Weaver A, Frilling A, Gritti S, Arveschoug AK, Meirovitz A, Knigge U, Sorbye H. Peptide receptor radionuclide therapy in gastroenteropancreatic NEN G3: a multicenter cohort study. Endocr Relat Cancer. 2019;26:227–239. doi: 10.1530/ERC-18-0424. [DOI] [PubMed] [Google Scholar]
- 67.Zhang J, Kulkarni HR, Singh A, Niepsch K, Müller D, Baum RP. Peptide Receptor Radionuclide Therapy in Grade 3 Neuroendocrine Neoplasms: Safety and Survival Analysis in 69 Patients. J Nucl Med. 2019;60:377–385. doi: 10.2967/jnumed.118.215848. [DOI] [PubMed] [Google Scholar]
- 68.Alese OB, Jiang R, Shaib W, Wu C, Akce M, Behera M, El-Rayes BF. High-Grade Gastrointestinal Neuroendocrine Carcinoma Management and Outcomes: A National Cancer Database Study. Oncologist. 2019;24:911–920. doi: 10.1634/theoncologist.2018-0382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Haugvik SP, Janson ET, Österlund P, Langer SW, Falk RS, Labori KJ, Vestermark LW, Grønbæk H, Gladhaug IP, Sorbye H. Surgical Treatment as a Principle for Patients with High-Grade Pancreatic Neuroendocrine Carcinoma: A Nordic Multicenter Comparative Study. Ann Surg Oncol. 2016;23:1721–1728. doi: 10.1245/s10434-015-5013-2. [DOI] [PubMed] [Google Scholar]
- 70.Strosberg JR, Coppola D, Klimstra DS, Phan AT, Kulke MH, Wiseman GA, Kvols LK North American Neuroendocrine Tumor Society (NANETS) The NANETS consensus guidelines for the diagnosis and management of poorly differentiated (high-grade) extrapulmonary neuroendocrine carcinomas. Pancreas. 2010;39:799–800. doi: 10.1097/MPA.0b013e3181ebb56f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Rinke A, Gress TM. Neuroendocrine Cancer, Therapeutic Strategies in G3 Cancers. Digestion. 2017;95:109–114. doi: 10.1159/000454761. [DOI] [PubMed] [Google Scholar]
- 72.Nakano K, Takahashi S, Yuasa T, Nishimura N, Mishima Y, Sakajiri S, Yokoyama M, Tsuyama N, Ishikawa Y, Hatake K. Feasibility and efficacy of combined cisplatin and irinotecan chemotherapy for poorly differentiated neuroendocrine carcinomas. Jpn J Clin Oncol. 2012;42:697–703. doi: 10.1093/jjco/hys085. [DOI] [PubMed] [Google Scholar]
- 73.Riihimäki M, Hemminki A, Sundquist K, Sundquist J, Hemminki K. The epidemiology of metastases in neuroendocrine tumors. Int J Cancer. 2016;139:2679–2686. doi: 10.1002/ijc.30400. [DOI] [PubMed] [Google Scholar]
- 74.Hadoux J, Malka D, Planchard D, Scoazec JY, Caramella C, Guigay J, Boige V, Leboulleux S, Burtin P, Berdelou A, Loriot Y, Duvillard P, Chougnet CN, Déandréis D, Schlumberger M, Borget I, Ducreux M, Baudin E. Post-first-line FOLFOX chemotherapy for grade 3 neuroendocrine carcinoma. Endocr Relat Cancer. 2015;22:289–298. doi: 10.1530/ERC-15-0075. [DOI] [PubMed] [Google Scholar]
- 75.Zhu J, Strosberg JR, Dropkin E, Strickler JH. Treatment of High-Grade Metastatic Pancreatic Neuroendocrine Carcinoma with FOLFIRINOX. J Gastrointest Cancer. 2015;46:166–169. doi: 10.1007/s12029-015-9689-0. [DOI] [PubMed] [Google Scholar]
- 76.Hentic O, Hammel P, Couvelard A, Rebours V, Zappa M, Palazzo M, Maire F, Goujon G, Gillet A, Lévy P, Ruszniewski P. FOLFIRI regimen: an effective second-line chemotherapy after failure of etoposide-platinum combination in patients with neuroendocrine carcinomas grade 3. Endocr Relat Cancer. 2012;19:751–757. doi: 10.1530/ERC-12-0002. [DOI] [PubMed] [Google Scholar]
- 77.Lorenzoni A, Capozza A, Artale S, Maccauro M, Seregni EC. Impressive Response to Tandem Treatment With [90Y]DOTATOC and [177Lu]DOTATOC in Grade 3 Pancreatic Neuroendocrine Carcinoma. Clin Nucl Med. 2018;43:506–508. doi: 10.1097/RLU.0000000000002116. [DOI] [PubMed] [Google Scholar]
- 78.Demirci E, Kabasakal L, Toklu T, Ocak M, Şahin OE, Alan-Selcuk N, Araman A. 177Lu-DOTATATE therapy in patients with neuroendocrine tumours including high-grade (WHO G3) neuroendocrine tumours: response to treatment and long-term survival update. Nucl Med Commun. 2018;39:789–796. doi: 10.1097/MNM.0000000000000874. [DOI] [PubMed] [Google Scholar]
- 79.Vijayvergia N, Dasari A, Deng M, Litwin S, Al-Toubah T, Alpaugh RK, Dotan E, Hall MJ, Ross NM, Runyen MM, Denlinger CS, Halperin DM, Cohen SJ, Engstrom PF, Strosberg JR. Pembrolizumab monotherapy in patients with previously treated metastatic high-grade neuroendocrine neoplasms: joint analysis of two prospective, non-randomised trials. Br J Cancer. 2020;122:1309–1314. doi: 10.1038/s41416-020-0775-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Goodman A. Dual-Immunotherapy Approach Shows Efficacy in High-Grade Neuroendocrine Tumors. [published 10 May 2019] Available from: https://ascopost.com/issues/may-10-2019/dual-immunotherapy-in-high-grade-neuroendocrine-tumors/
- 81.Swidnicka-Siergiejko AK, Gomez-Chou SB, Cruz-Monserrate Z, Deng D, Liu Y, Huang H, Ji B, Azizian N, Daniluk J, Lu W, Wang H, Maitra A, Logsdon CD. Chronic inflammation initiates multiple forms of K-Ras-independent mouse pancreatic cancer in the absence of TP53. Oncogene. 2017;36:3149–3158. doi: 10.1038/onc.2016.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Benten D, Behrang Y, Unrau L, Weissmann V, Wolters-Eisfeld G, Burdak-Rothkamm S, Stahl FR, Anlauf M, Grabowski P, Möbs M, Dieckhoff J, Sipos B, Fahl M, Eggers C, Perez D, Bockhorn M, Izbicki JR, Lohse AW, Schrader J. Establishment of the First Well-differentiated Human Pancreatic Neuroendocrine Tumor Model. Mol Cancer Res. 2018;16:496–507. doi: 10.1158/1541-7786.MCR-17-0163. [DOI] [PubMed] [Google Scholar]