Abstract
Background
Despite a lack of clinical evidence, hydroxychloroquine and azithromycin are being administered widely to patients with verified or suspected coronavirus disease 2019 (COVID‐19). Both drugs may increase risk of lethal arrhythmias associated with QT interval prolongation.
Methods and Results
We analyzed a case series of COVID‐19–positive/suspected patients admitted between February 1, 2020, and April 4, 2020, who were treated with azithromycin, hydroxychloroquine, or a combination of both drugs. We evaluated baseline and postmedication QT interval (corrected QT interval [QTc]; Bazett) using 12‐lead ECGs. Critical QTc prolongation was defined as follows: (1) maximum QTc ≥500 ms (if QRS <120 ms) or QTc ≥550 ms (if QRS ≥120 ms) and (2) QTc increase of ≥60 ms. Tisdale score and Elixhauser comorbidity index were calculated. Of 490 COVID‐19–positive/suspected patients, 314 (64%) received either/both drugs and 98 (73 COVID‐19 positive and 25 suspected) met study criteria (age, 62±17 years; 61% men). Azithromycin was prescribed in 28%, hydroxychloroquine in 10%, and both in 62%. Baseline mean QTc was 448±29 ms and increased to 459±36 ms (P=0.005) with medications. Significant prolongation was observed only in men (18±43 ms versus −0.2±28 ms in women; P=0.02). A total of 12% of patients reached critical QTc prolongation. Changes in QTc were highest with the combination compared with either drug, with much greater prolongation with combination versus azithromycin (17±39 ms versus 0.5±40 ms; P=0.07). No patients manifested torsades de pointes.
Conclusions
Overall, 12% of patients manifested critical QTc prolongation, and the combination caused greater prolongation than either drug alone. The balance between uncertain benefit and potential risk when treating COVID‐19 patients should be carefully assessed.
Keywords: azithromycin, COVID‐19, hydroxychloroquine, monitoring, QT interval
Subject Categories: Arrhythmias
Nonstandard Abbreviations and Acronyms
- COVID‐19
coronavirus disease 2019
- QTc
corrected QT interval
Clinical Perspective
What Is New?
Among coronavirus disease 2019 patients treated with hydroxychloroquine, azithromycin, or the combination, 12% manifested with critical corrected QT interval prolongation.
The combination resulted in greater corrected QT interval prolongation than either drug alone.
What Are the Clinical Implications?
While we await results of definitive clinical trials, it is best to carefully assess risk/benefit of therapy and corrected QT interval monitoring may be a safer approach, especially for patients taking the drug combination.
The ongoing coronavirus disease 2019 (COVID‐19) pandemic is an unprecedented public health challenge at a global level.1 As of May 26, 2020, 1 618 757 Americans have tested positive and 96 909 of those patients have succumbed to this illness.2 In the absence of a vaccine or any proven therapeutic agent, hydroxychloroquine and azithromycin, often used in combination, have emerged as a potential therapy on the basis of extremely limited clinical evidence.3 These drugs are currently being prescribed in COVID‐19–positive or COVID‐19–suspected patients in growing numbers across the United States and around the world.4
Hydroxychloroquine is an antimalarial drug that has also been used to treat arthritis and systemic lupus erythematosus. Azithromycin is a macrolide antibiotic used to treat a wide variety of bacterial infections. Both drugs prolong the QT interval by blocking the KCNH2‐encoded hERG/Kv11.1 cardiac potassium channel, thereby increasing the risk of torsades de pointes.5, 6, 7 In general, hydroxychloroquine is reasonably well tolerated and used chronically in arthritis and systemic lupus erythematosus patients without heart rhythm monitoring.8 In 2013, the Food and Drug Administration issued a black box warning for azithromycin following multiple reported cases of corrected QT interval (QTc) prolongation followed by torsades de pointes.9 Because the use of these drugs in COVID‐19 patients is so recent and based largely on in vitro studies10 and anecdotal observations,11 randomized clinical trial data are lacking, and community prescribing practices are as yet unclear. More important, there is no information available about the extent of QTc prolongation with a combination of hydroxychloroquine and azithromycin. From first principles, simultaneous block of the hERG/Kv11.1 cardiac potassium channel with both agents may cause critical QTc prolongation and an elevated risk of torsades de pointes. We therefore evaluated prescribing practices and monitored the QTc in COVID‐19–positive or COVID‐19–suspected patients who received these medications.
Methods
The authors will make the data and methods used to conduct the research available to any researcher for purposes of reproducing the results. This study was approved by the Cedars‐Sinai Institutional Review Board, and the requirement for informed consent was waived for this deidentified analysis.
Patient Population
We identified all patients with confirmed COVID‐19 infection as well as patients under investigation admitted at Cedars‐Sinai Medical Center (Los Angeles, CA) between February 1, 2020, and April 4, 2020, who received azithromycin, hydroxychloroquine, or both as part of their medical treatment. A detailed, retrospective chart review was then performed via the electronic health record (Epic, Verona, WI) at our institution. COVID‐19 testing was performed in all individuals with the live virus polymerase chain reaction test. Of the patients under investigation and confirmed COVID‐19 cases, we only included patients who had at least 2 12‐lead ECGs performed in our MUSE system between January 1, 2020, and April 5, 2020. Patients with paced ventricular rhythms, atrial fibrillation, atrial flutter, supraventricular tachycardia, or ECGs otherwise unsuitable for accurate QT interval measurement were excluded. In addition, patients without ECGs performed on day 2 of medication administration or later were excluded (Figure 1). During the time that these patients were treated, our hospital policy required that daily ECGs be performed in all patients started on hydroxychloroquine just before and for the duration of therapy. If a patient had QTc ≥470 ms at baseline or reached critical QTc prolongation (defined below in Outcome), then continuous telemetry monitoring was recommended.
Figure 1. Study design.
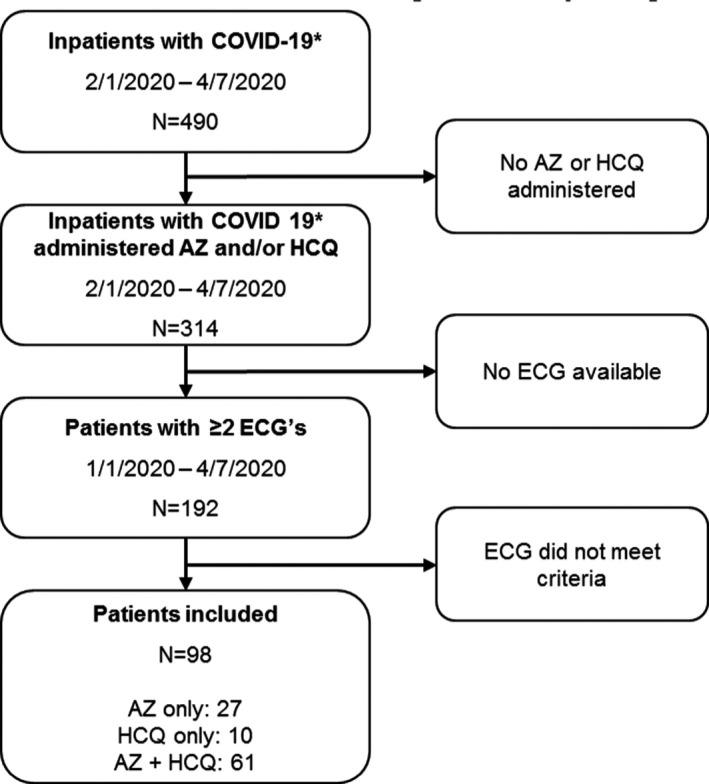
*Coronavirus disease 2019 (COVID‐19)–positive patients and patients under investigation. AZ indicates azithromycin; and HCQ, hydroxychloroquine.
Data Collection and Definitions
We compared standard 12‐lead ECG parameters, including the QTc (Bazett formula) from the baseline ECG closest to and before medication administration, with the longest QTc observed postmedication administration, up to 24 hours after the last medication dose. Given variability in heart rates, QTc prolongation was also evaluated using the Fridericia formula for correction of the QT interval. QT measurements were independently reviewed and validated by a cardiologist (A.R.) to ensure accuracy of the automated measurement and manually remeasured if required. Patients were assigned to 1 of 3 groups, depending on which medication they received: azithromycin, hydroxychloroquine, or azithromycin+hydroxychloroquine. Patient demographics, clinical history, laboratory findings, and medications were obtained from detailed chart review, including risk factors to calculate the Tisdale score12 and the Elixhauser comorbidity index13 for each patient. The Tisdale score consists of age ≥68 years, female sex, use of loop diuretics, potassium level ≤3.5 mEq/L, baseline QTc ≥450 ms, acute myocardial infarction, number of QT‐prolonging medications, sepsis, and heart failure and has been developed as a risk score for QTc prolongation. The Elixhauser comorbidity index incorporates 21 different variables, including obesity, diabetes mellitus, peripheral artery disease, chronic kidney disease, HIV, and cancer, and is used to predict in‐hospital mortality.
Outcome
The outcome of interest was postmedication critical QTc prolongation, defined as follows: (1) maximum postmedication QTc ≥500 ms (if QRS <120 ms) or QTc ≥550 ms (if QRS ≥120 ms) and (2) mean increase from baseline QTc to maximum postmedication QTc of ≥60 ms.14, 15
Statistical Analysis
We evaluated the mean change from baseline QTc to longest QTc overall and by medication type using repeated‐measures ANOVA. We also evaluated the frequency of critical ECG changes overall, by medication type and by patient characteristics using the χ2 test. Given the relatively low number of patients who received hydroxychloroquine alone, the decision was made to only compare azithromycin with the combination of azithromycin+hydroxychloroquine. We used logistic regression to evaluate whether age and sex (model 1) or Tisdale score and Elixhauser score (model 2) were associated with critical QTc prolongation. Analyses were performed using SAS version 9.4 (SAS Institute Inc, Cary, NC). A 2‐sided P<0.05 was considered statistically significant.
Results
A total of 490 patients were admitted to our medical center with a verified or suspected diagnosis of COVID‐19 during the study time period, and 314 (64%) were treated with azithromycin, hydroxychloroquine, or the combination (Figure 1). Of these, 192 patients had a 12‐lead ECG performed during treatment, and a subgroup of 98 confirmed COVID infection or patients under investigation (age, 62±17 years; 61% men) met criteria for inclusion in the study (Table 1). Ninety‐two of the excluded patients had either ECGs with indeterminate QT intervals (see Methods) or no ECG on day 2 of medication administration or later, and 2 patients had extreme variation in heart rate, with near doubling of heart rate between baseline and maximal QTc prolongation. Given the limitations of QT interval correction at significantly elevated heart rates, we excluded both patients from the final analysis. Only 1 patient included in the analysis required manual QT measurement using the MUSE electronic calipers and the tangent method of calculation. Of the 98 patients, 73 were COVID‐19 positive and 25 were patients under investigation. Azithromycin was prescribed in 28%, hydroxychloroquine in 10%, and the combination in 62%.
Table 1.
Patient Characteristics
| Characteristics | Confirmed COVID‐19/PUI (n=98) |
|---|---|
| Age, mean±SD, y | 62.3±17.0 |
| Male sex, n (%) | 60 (61) |
| Body mass index, mean±SD, kg/m2 | 27.8±6.6 |
| Ethnicity, n (%) | |
| Hispanic | 15 (15) |
| Non‐Hispanic | 79 (81) |
| Unknown | 4 (4) |
| Race, n (%) | |
| Asian | 5 (5) |
| Black/African American | 17 (17) |
| White | 69 (70) |
| Other | 5 (5) |
| Unknown | 2 (2) |
| Heart failure, n (%) | 20 (20) |
| Hypertension, n (%) | 59 (60) |
| Diabetes mellitus, n (%) | 22 (22) |
| Chronic kidney disease, n (%) | 14 (14) |
| Chronic obstructive pulmonary disease, n (%) | 25 (26) |
| ICU patients, n (%) | 48 (49) |
| Non‐ICU patients, n (%) | 50 (51) |
| Medications, n (%) | |
| Azithromycin | 27 (28) |
| Hydroxychloroquine | 10 (10) |
| Azithromycin+hydroxychloroquine | 61 (62) |
| Tisdale score, n (%) | |
| Low risk (<7) | 1 (1) |
| Moderate risk (7–10) | 36 (37) |
| High risk (≥11) | 61 (62) |
| Elixhauser comorbidity index, mean±SD | 15.2±13.2 |
COVID‐19 indicates coronavirus disease 2019; ICU, intensive care unit; and PUI, patients under investigation.
At baseline, the mean QTc (Bazett) was 448±29 ms, and 20% of patients had QTc ≥470 ms (Table 2). With drug administration, overall QTc (Bazett) increased to 459±36 ms (P=0.005). Since a similar increase in QTc was observed using correction of the QT interval with the Fridericia formula (420±32–438±36 ms; P<0.0001), the Bazett formula was used for the rest of the analysis. Subgroup analysis by sex revealed baseline and postdrug QTc of 449.1±31.4 and 467.3±37.8 ms in men, respectively (P=0.002). In women, baseline and postdrug QTc values were 444.9±24.1 and 444.7±28.6 ms, respectively (P=0.97). The difference in mean QTc prolongation in men versus women was significant (18±43 ms versus −0.2±28 ms; P=0.02). Further analysis by sex showed no significant difference in Tisdale score or Elixhauser comorbidity index. High Tisdale scores were similar in men and women (62% versus 63%; P=0.88). The average Elixhauser comorbidity index in men was lower and nonsignificant when compared with women. A trend toward significance was noted when comparing overall body mass index in men versus women: 42.4% of men were obese versus 26.3% of women (P=0.11).
Table 2.
ECG Characteristics
| Characteristics | Confirmed COVID‐19/PUI (n=98) |
|---|---|
| Baseline ECG intervals | |
| Ventricular rate, bpm | 92±19 |
| RR, ms | 681±148 |
| QRS, ms | 92±19 |
| QT, ms | 368±44 |
| QTc (Bazett), ms | 448±29 |
| Postdrug ECG | |
| Ventricular rate, bpm | 82±19 |
| RR, ms | 768±180 |
| QRS, ms | 94±19 |
| QT, ms | 400±48 |
| QTc (Bazett), ms | 459±36 |
| Mean change in QTc, ms | 11.1±38.5 |
| Patients with baseline QTc ≥470 ms | 20 (20) |
| Patients meeting critical QTc threshold | |
| ≥500 ms (QRS <120 ms) | 7 (7) |
| ≥550 ms (QRS ≥120 ms) | 1 (1) |
| Patients with critical absolute change in QTc | |
| ΔQTc ≥60 ms and QTc <500/550‐ms threshold | 4 (4) |
| ΔQTc ≥60 ms and QTC ≥500/550‐ms threshold | 7 (7) |
| Total patients with critical QTc prolongation | 12 (12) |
Data are given as mean±SD or number (percentage). Bpm indicates beats per minute; COVID‐19, coronavirus disease 2019; PUI, patients under investigation; and QTc, corrected QT interval.
Of note, multiple patterns were observed for use, duration, and dosage of both medications. Although most patients (81.6%) received at least a 5‐day course of each medication, there were many patients who received a higher dose on day 1 compared with the dosage on days 2 to 5. For example, azithromycin was given either orally or intravenously, and the dosage was either 500 mg daily or 500 mg on day 1 followed by 250 mg daily on days 2 to 5. For hydroxychloroquine, most patients (87.0%) received 400 mg orally twice daily before receiving a dosage of 200 mg orally twice daily on days 2 to 5. The medications were started together (50.8%), staggered by 1 day (29.5%), or administered >1 day after the other (19.7%). Patients with lower values of QTc at baseline were more likely to be started on the combination of the 2 medications. In contrast, patients with a higher baseline QTc were more likely to receive either hydroxychloroquine or azithromycin rather than the combination.
The mean change in QTc values was greatest in the combined hydroxychloroquine and azithromycin group, and when compared with the azithromycin group, QTc prolongation caused by the combination was observed to be several‐fold higher than prolongation caused by azithromycin alone (17.2±39.0 ms versus 0.5±40.3 ms; P=0.07) (Figure 2). A subgroup of 12% of patients was observed to reach a critical level of QTc prolongation: QTc ≥500 ms (if QRS <120 ms), QTc ≥550 ms (if QRS ≥120 ms), or change in QTc ≥60 ms (Figure 3). Nine out of 60 men (15.0%), and 3 of 38 women (7.9%) reached critical QTc prolongation (P=0.30). Among the 12 patients with critical QTc prolongation, 5 were on azithromycin and 7 were on the combination of hydroxychloroquine and azithromycin (Table 3).There were no significant differences with regard to age, sex, sepsis, diabetes mellitus, baseline QTc ≥450 ms, or number of QT‐prolonging medications. Patients with critical QTc prolongation had significantly greater use of loop diuretics (P=0.007) and acute myocardial infarction (P=0.01) (Table 4). A total of 75% of patients with critical QTc prolongation had a high Tisdale score, whereas 60.5% of patients without critical QTc changes had a high Tisdale score (P=0.33). Similarly, there was no significant difference in the Elixhauser comorbidity indexes of the 2 groups (P=0.86). In logistic regression models, age and sex were not significantly associated with critical QTc prolongation (P>0.35), nor were Tisdale score and Elixhauser score (P>0.20). No patients had syncope, torsades de pointes, or other lethal arrhythmias during or after drug administration.
Figure 2. Boxplot of corrected QT interval (QTc) changes by medication group.

The QTc change is greatest in the combined hydroxychloroquine (HCQ) and azithromycin (AZ) group, and when compared with the AZ group alone, the change in QTc is borderline significant (P=0.07).
Figure 3. Change in corrected QT interval (QTc) from baseline to postmedication administration.
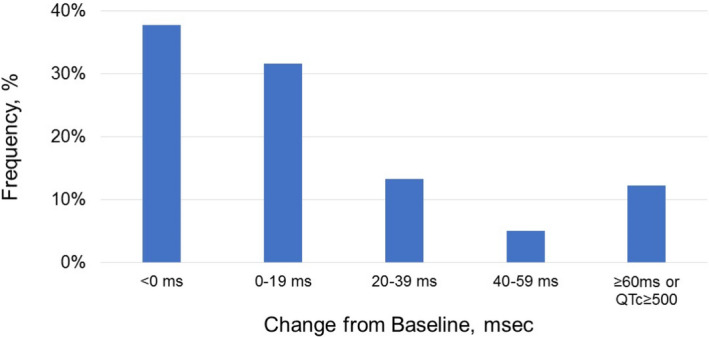
Critical QTc prolongation (Δ ≥60 ms or QTc ≥500 ms [≥550 ms in patients with QRS ≥120 ms]) was observed in 12% of patients.
Table 3.
Baseline and Postdrug ECG Characteristics by Medication Administered
| ECG Characteristics | Azithromycin (n=27) | Azithromycin+Hydroxychloroquine (n=61) | P Value |
|---|---|---|---|
| Baseline ECG intervals | |||
| Ventricular rate, bpm | 102±23 | 89±17 | 0.01a |
| RR, ms | 622±168 | 701±135 | 0.02a |
| QRS, ms | 94±24 | 93±17 | 0.84 |
| QT, ms | 364±61 | 367±37 | 0.84 |
| QTc (Bazett), ms | 463±39 | 439±20 | 0.005a |
| Postdrug ECG | |||
| Ventricular rate, bpm | 92±23 | 78±15 | 0.006a |
| RR, ms | 686±168 | 794±148 | 0.003a |
| QRS, ms | 93±24 | 94±18 | 0.76 |
| QT, ms | 383±62 | 405±43 | 0.10a |
| QTc (Bazett), ms | 464±38 | 457±38 | 0.41 |
| Change in QTc, ms | 0.5±40.3 | 17.2±39.0 | 0.07 |
| Patients meeting critical QTc thresholdb | 3 (11) | 5 (8) | 0.66 |
| Patients with absolute ΔQTc ≥60 ms | 4 (15) | 7 (12) | 0.66 |
| Patients with critical QTc prolongation overallc | 5 (19) | 7 (12) | 0.37 |
Data are given as mean±SD or number (percentage). Bpm indicates beats per minute; and QTc, corrected QT interval.
P<0.05.
For ≥500 ms (QRS interval <120 ms) or ≥550 ms (QRS interval ≥120 ms).
By QTc threshold or absolute change criteria.
Table 4.
Characteristics of Patients With Critical QTc Prolongation
| Variable | Critical QTc Prolongation (n=12) | Noncritical Change in QTc (n=86) | P Value |
|---|---|---|---|
| Tisdale score | |||
| Aged ≥68 y | 3 (25) | 39 (45) | 0.18 |
| Female sex | 3 (25) | 35 (41) | 0.29 |
| Loop diuretic | 6 (50) | 14 (16) | 0.007a |
| Serum potassium ≤3.5 mEq/L | 1 (8) | 18 (21) | 0.30 |
| Baseline QTc ≥450 ms | 3 (25) | 38 (44) | 0.21 |
| Acute MI | 5 (42) | 11 (13) | 0.01a |
| ≥2 QT‐prolonging drugs | 11 (92) | 63 (73) | 0.16 |
| Sepsis | 9 (75) | 62 (72) | 0.83 |
| Heart failure | 3 (25) | 17 (20) | 0.67 |
| Elixhauser index | |||
| COPD | 2 (17) | 23 (27) | 0.45 |
| Diabetes mellitus | 3 (25) | 19 (22) | 0.82 |
| Hypertension | 7 (58) | 52 (61) | 0.89 |
| CKD | 3 (25) | 11 (13) | 0.26 |
| Male sex | 9 (75) | 51 (59) | 0.29 |
| Low/moderate Tisdale score | 3 (25) | 34 (40) | 0.33 |
| High Tisdale score | 9 (75) | 52 (61) | 0.33 |
| Elixhauser index | 14.6±9.2 | 15.3±13.7 | 0.86 |
Data are given as number (percentage) or mean±SD. CKD indicates chronic kidney disease; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; and QTc, corrected QT interval.
P<0.05.
Discussion
At a single large community medical center, we studied 98 COVID‐19–positive or COVID‐19–suspected patients treated with hydroxychloroquine and azithromycin, administered either individually or in combination. Although lethal arrhythmias were not observed during the limited period of observation, 12% of patients manifested critical QTc prolongation. QTc prolongation was many‐fold higher when hydroxychloroquine and azithromycin were administered in combination compared with azithromycin alone. Moreover, patients who achieved critical QTc prolongation were not distinguished by significantly higher Tisdale scores, nor by higher Elixhauser comorbidity indexes.
Our finding of greatest QTc prolongation in the combined hydroxychloroquine+azithromycin group is important. In our study, patients with lower values of QTc at baseline were more likely to be started on the combination of the 2 medications. In contrast, patients with a higher baseline QTc were more likely to receive either hydroxychloroquine or azithromycin rather than the combination. This suggests that prescription practice may have been driven by a concern about excessive QTc prolongation with the use of both medications in combination. Further review of our data showed that some practitioners decided on a staggered dosing regimen of one drug followed by the other drug later in the hospital stay. However, given the prolonged half‐lives of these medications, such practice still results in a combined drug effect on the QTc.
Another interesting finding in our study was that men had significantly higher mean QTc prolongation than women. The incidence of torsades de pointes is known to be higher in women, and female sex is listed as a risk factor in the Tisdale score for QT prolongation. The reasons for this discrepancy still need to be explained, but this could be related to the majority of patients in our case series being men. Because rates of high Tisdale scores and Elixhauser comorbidity indexes were also not significantly varied between the sexes, this finding cannot be explained by differences in overall morbidity burden, and would benefit from further study.
Overall, our findings support the need to monitor the QTc during the period that these 2 medications are administered, especially when used in combination. We were not able to distinguish patients more likely to have critical QTc prolongation by the Tisdale score or Elixhauser index, making monitoring during medication administration likely to be a safer approach.14 In addition to QTc prolongation as a result of KCNH2‐encoded hERG/Kv11.1 cardiac potassium channel blockage, both drugs can provoke potentially lethal arrhythmias by additional mechanisms.16, 17 In fact, polymorphic ventricular tachycardia with azithromycin has been reported in the absence of QTc prolongation.18 Given that critical QTc prolongation was observed with both administration of azithromycin (n=5) and the combination of hydroxychloroquine+azithromycin (n=7), monitoring of the QTc should not be restricted to the combination of these drugs alone. This is consistent with the 2013 black box warning issued by the Food and Drug Administration on the increased risk of torsades de pointes with azithromycin.
Limitations
The variation in dosing patterns and duration of treatment for each medication are important to note as they can potentially influence when the ECG with the longest QTc may be observed. We are reporting findings from a case series of patients, and the lack of a control group is a limitation. In addition, because of inconsistency in obtaining daily ECGs during medication administration, we believe that the 12% of patients who experienced critical QTc prolongation was likely an underestimation. Furthermore, variability in heart rates, especially in the setting of sepsis, can affect QT interval correction. We reported the QTc using Bazett correction, which is consistent with most of the published literature as well as ECG recording machines in typical use. Along these lines, cytokine storm associated with COVID‐19 infection may also prolong QTc, and although we do not have an accurate estimate of the number of patients in our study with cytokine storm, this is another variable that should be considered when starting additional QTc‐prolonging therapy. The relatively small sample size does not allow for feasible assessment of torsades de pointes risk with these agents.
Conclusions
Prescribing practices for azithromycin and hydroxychloroquine amid the COVID‐19 pandemic were found to be diverse, but most patients in our study received a combination of the 2 drugs. Among patients prescribed azithromycin, hydroxychloroquine, or a combination of both, 12% achieved a critical level of QTc prolongation. QTc values were highest with the combination of hydroxychloroquine and azithromycin, with QTc prolongation many‐fold higher in the combination group than with azithromycin alone. Especially if ongoing clinical trials indicate benefit from use of these medications, these findings could inform clinical therapeutics and ECG monitoring in COVID‐19 patients.
Sources of Funding
Cedars‐Sinai Health System. Dr Chugh holds the Pauline and Harold Price Chair in Cardiac Electrophysiology Research.
Disclosures
None.
(J Am Heart Assoc. 2020;9:e017144 DOI: 10.1161/JAHA.120.017144.)
Preprint posted on MedRxiv, April 25, 2020. doi: https://doi.org/10.1101/2020.04.22.20075671.
For Sources of Funding and Disclosures, see page 7.
References
- 1. Park M, Cook AR, Lim JT, Sun Y, Dickens BL. A systematic review of COVID‐19 epidemiology based on current evidence. J Clin Med. 2020;9:E967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO Health Emergency Coronavirus 19 Dashboard. Available at: https://covid19.who.int/region/amro/country/us. Accessed May 26, 2020.
- 3. Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, Doudier B, Courjon J, Giordanengo V, Vieira VE, et al. Hydroxychloroquine and azithromycin as a treatment of COVID‐19: results of an open‐label non‐randomized clinical trial. International Journal of Antimicrobial Agents. 2020. doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 4. Ohe M, Shida H, Jodo S, Kusunoki Y, Seki M, Furuya K, Goudarzi H. Macrolide treatment for COVID‐19: will this be the way forward? Biosci Trends. 2020;14:159–160. [epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 5. Hancox JC, Hasnain M, Vieweg WV, Crouse EL, Baranchuk A. Azithromycin, cardiovascular risks, QTc interval prolongation, torsade de pointes, and regulatory issues: a narrative review based on the study of case reports. Ther Adv Infect Dis. 2013;1:155–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. McGhie TK, Harvey P, Su J, Anderson N, Tomlinson G, Touma Z. Electrocardiogram abnormalities related to anti‐malarials in systemic lupus erythematosus. Clin Exp Rheumatol. 2018;36:545–551. [PubMed] [Google Scholar]
- 7. Traebert M, Dumotier B, Meister L, Hoffmann P, Dominguez‐Estevez M, Suter W. Inhibition of hERG K+ currents by antimalarial drugs in stably transfected HEK293 cells. Eur J Pharmacol. 2004;484:41–48. [DOI] [PubMed] [Google Scholar]
- 8. Tang C, Godfrey T, Stawell R, Nikpour M. Hydroxychloroquine in lupus: emerging evidence supporting multiple beneficial effects. Intern Med J. 2012;42:968–978. [DOI] [PubMed] [Google Scholar]
- 9. Ray WA, Murray KT, Hall K, Arbogast PG, Stein CM. Azithromycin and the risk of cardiovascular death. N Engl J Med. 2012;366:1881–1890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, Liu X, Zhao L, Dong E, Song C, et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2). Clin Infect Dis. 2020;ciaa237. Available at: 10.1093/cid/ciaa237. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Lakkireddy DR, Chung MK, Gopinathannair R, Patton KK, Gluckman TJ, Turagam M, Cheung J, Patel P, Sotomonte J, Lampert R, et al. Guidance for cardiac electrophysiology during the coronavirus (COVID‐19) pandemic from the Heart Rhythm Society COVID‐19 Task Force; electrophysiology section of the American College of Cardiology; and the Electrocardiography and Arrhythmias Committee of the Council on Clinical Cardiology, American Heart Association. Heart Rhythm. 2020. Available at: http://heartrhythmjournal.com/retrieve/pii/S1547527120302897. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tisdale JE, Jaynes HA, Kingery JR, Mourad NA, Trujillo TN, Overholser BR, Kovacs RJ. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013;6:479–487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. van Walraven C, Austin PC, Jennings A, Quan H, Forster AJ. A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care. 2009;47:626–633. [DOI] [PubMed] [Google Scholar]
- 14. Giudicessi JR, Noseworthy PA, Friedman PA, Ackerman MJ. Urgent guidance for navigating and circumventing the QTc prolonging and torsadogenic potential of possible pharmacotherapies for COVID‐19. Mayo Clin Proc. 2020;S0025‐6196(20)30313‐X. DOI: 10.1016/j.mayocp.2020.03.024. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Roden DM, Harrington RA, Poppas A, Russo AM. Considerations for drug interactions on QTc in exploratory COVID‐19 (coronavirus disease 2019) treatment. Circulation. 2020; doi: 10.1161/CIRCULATIONAHA.120.047521. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 16. Capel RA, Herring N, Kalla M, Yavari A, Mirams GR, Douglas G, Bub G, Channon K, Paterson DJ, Terrar DA, et al. Hydroxychloroquine reduces heart rate by modulating the hyperpolarization‐activated current If: novel electrophysiological insights and therapeutic potential. Heart Rhythm. 2015;12:2186–2194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Zhang M, Xie M, Li S, Gao Y, Xue S, Huang H, Chen K, Liu F, Chen L. Electrophysiologic studies on the risks and potential mechanism underlying the proarrhythmic nature of azithromycin. Cardiovasc Toxicol. 2017;17:434–440. [DOI] [PubMed] [Google Scholar]
- 18. Kim MH, Berkowitz C, Trohman RG. Polymorphic ventricular tachycardia with a normal QT interval following azithromycin. Pacing Clin Electrophysiol. 2005;28:1221–1222. [DOI] [PubMed] [Google Scholar]


