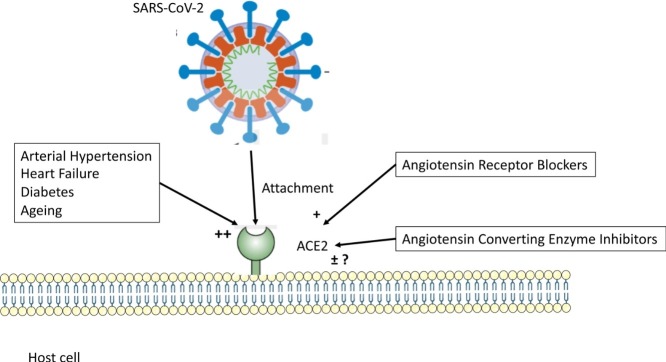
Graphical abstract
Keywords: Angiotensin-converting enzyme inhibitors, Angiotensin receptor blockers, Arterial hypertension, Heart failure
Abstract
Severe acute respiratory syndrome coronavirus 2, which is responsible for the current coronavirus disease 2019 pandemic, uses angiotensin-converting enzyme 2 as a gateway into host cells. In this review, we summarise the biology of this enzyme, which plays a key role in cardiovascular homeostasis. Blockers of the renin–angiotensin system modify the expression and activity of angiotensin-converting enzyme 2 in different ways. The effects of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers on the expression and enzyme activity of angiotensin-converting enzyme 2 are reviewed, and the consequences of these treatments for the severity of coronavirus disease 2019 infection are discussed.
Résumé
Le coronavirus 2 du syndrome respiratoire aigu sévère (SARS-CoV-2), responsable de la pandémie actuelle de COVID-2019, utilise l’enzyme de conversion de l’angiotensine de type 2 (ACE2) comme porte d’entrée dans les cellules hôtes. Dans cette revue, nous résumons la biologie de cette enzyme qui joue un rôle clé dans l’homéostasie cardiovasculaire. Les différents bloqueurs du système rénine–angiotensine modifient différemment l’expression et l’activité de ACE2. Les effets des inhibiteurs de l’enzyme de conversion de l’angiotensine et des bloqueurs des récepteurs AT1 de l’angiotensine sur l’expression et l’activité de ACE2 sont passés en revue; nous discutons également des conséquences des traitements par les bloqueurs du système rénine–angiotensine sur la gravité de l’infection par le coronavirus 2019.
Mots clés: Inhibiteurs de l’enzyme de conversion de l’angiotensine, Bloqueur des récepteurs de l’angiotensine, Hypertension artérielle, Insuffisance cardiaque
Background
The vector of the coronavirus disease 2019 (COVID-19) pandemic is a new coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the “little brother” of SARS-CoV-1, which was responsible for the SARS outbreak that began in China in 2002 and killed 800 people worldwide. The two viruses share a common access into human host cells: angiotensin-converting enzyme 2 (ACE2), which is particularly abundant in the lungs [1]. Given that the virus affects multiple organs, including the kidneys [2], brain [3] and heart [4], dissemination probably occurs as it passes from the respiratory epithelium to the vascular endothelium of the circulatory system [5]. The cases of Kawasaki-like disease reported in children infected by SARS-CoV-2 [6] support the view that it causes systemic vasculitis.
The remarkable work published in Cell on 16 April 2020 [7] established the relationship between SARS-CoV-2 and ACE2, and immediately raised at least three questions concerning the renin-angiotensin system (RAS):
-
•
whether and how pharmacological RAS blockers might alter the expression of ACE2 and affect susceptibility to disease or the severity of SARS-CoV-2 infection in patients with previous cardiovascular diseases and/or diabetes;
-
•
whether treatment with RAS blockers should be discontinued in patients with hypertension or heart failure to avoid exacerbating the risk in the event of SARS-CoV-2 infection;
-
•
whether a RAS blocker could be useful in the treatment of the “cytokine storm” that occurs in the severe forms of COVID-19.
The fact that there was no evidence to support any aspect of this speculation mattered little as the hypothesis gained weight, initially via social media and subsequently via the medical press [8]. For a brief overview of these questions, it is worthwhile recalling a few basic facts.
Infection with SARS-CoV-2
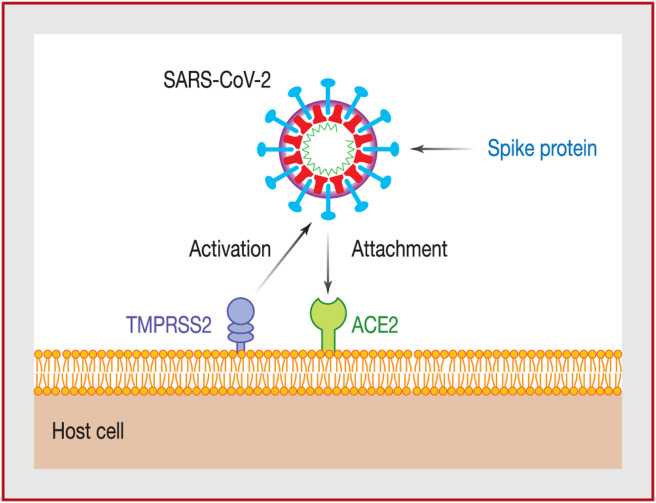
In the majority of cases, the SARS-CoV-2 coronavirus enters the body through the respiratory system, sometimes through the digestive tract and, in rare cases, through the ocular conjunctiva (Fig. 1 ). Upon contact with epithelial cells in the lungs, the virus binds to transmembrane ACE2 molecules, following activation of the spike protein by transmembrane protease serine 2 (TMPRSS2). Infection with SARS-CoV-2 is caused when the viral surface spike protein binds to the human ACE2 receptor; thus ACE2 appears to function as a gateway into the cell for SARS-CoV-2.
Figure 1.
Mode of entry of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) into an epithelial cell. Adapted from Clerkin et al. [45]. ACE2: angiotensin-converting enzyme 2; TMPRSS2: transmembrane protease serine 2.
Binding between the virus and ACE2 has been shown to “consume” ACE2, and viral infection may therefore reduce local tissue and plasma concentrations of ACE2. Once inside the cell, the virus releases ribonucleic acid, which uses the molecular machinery of the infected cell – specifically its Golgi apparatus – to replicate and form new viruses that exit the host cell and spread throughout the body.
Expression and enzyme functions of ACE2
ACE2 was identified in 2000, and is a transmembrane zinc metalloprotease with 42% homology with ACE. It is an 805-amino acid-long carboxypeptidase, with a 17-amino acid N-terminal signal peptide and a C-terminal membrane anchor [9]. ACE2 is mainly expressed in the lungs (alveolar epithelial cells and pulmonary circulation), the heart (fibroblasts, cardiomyocytes, endothelial cells, pericytes and fat cells), the blood vessels (endothelial cells and smooth muscle cells), the kidneys (glomerular and tubular cells and podocytes), the intestine (enterocytes), the liver (hepatocytes and cholangiocytes) and the testes.
ACE2 catalyses the cleavage of angiotensin II (Ang II) into Ang-(1–7), which in turn activates mitochondrial assembly (MAS) receptors. MAS activation exerts vasodilatory, antifibrotic and anti-inflammatory effects, facilitating the associated cardioprotective actions of nitric oxide release, vasodilation and blunting of inflammation [10].
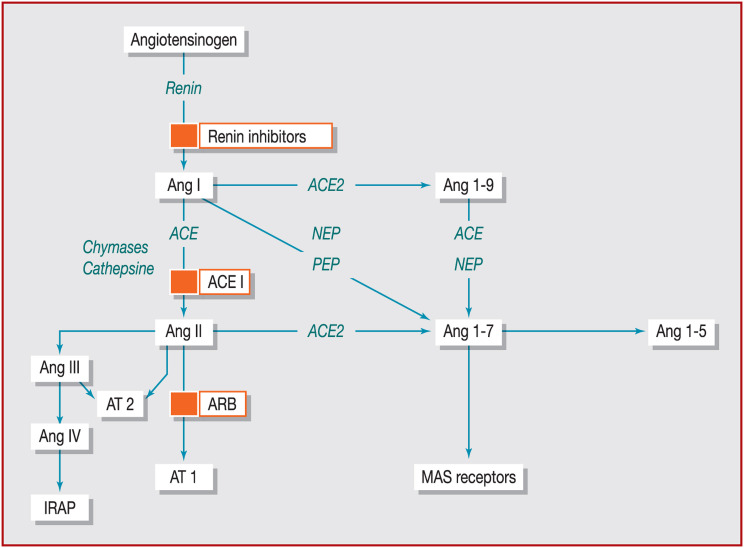
ACE2 plays a key role in the homeostasis of systemic blood pressure, counterbalancing the vasoconstrictive action of Ang II, which itself is generated by cleavage of Ang I catalysed by ACE [11]. It also catalyses the formation of Ang-(1–9) from Ang I, but with 400-fold less efficacy than the Ang II to Ang-(1–7) pathway [9]. Fig. 2 shows this often-ignored part of the RAS, and the levels at which Ang II receptor type 1 (AT1 receptor) blockers (ARBs) and ACE inhibitors act.
Figure 2.
Multiple effector peptides of the renin–angiotensin system. ACE: angiotensin-converting enzyme; ACE2: angiotensin-converting enzyme 2; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin II receptor blocker; AT1: angiotensin II receptor type 1; AT2: angiotensin II receptor type 2; IRAP: insulin-regulated aminopeptidase; MAS: mitochondrial assembly; NEP: neutral endopeptidase; PEP: prolyl endopeptidase.
It has been speculated, but not fully substantiated, that binding of SARS-CoV-2 to ACE2 alters tissue concentrations of the latter, and that massive losses of ACE2 may play a role in the pathophysiology of cardiorespiratory failure. In animal models, loss of ACE2 resulted in increased production of reactive oxygen species, and administration of recombinant ACE2 was shown to inhibit Ang II effects on transforming growth factor-beta 1 activation and collagen production [12]. Importantly, lower quantities of ACE2 cannot counterbalance the harmful effects of Ang II in promoting degradation of cardiac and lung structure and function [13].
In contrast to Ang II, which, via its AT1 receptors, has vasoconstrictive, proinflammatory, profibrotic and hypertrophic effects on cardiac and vascular cells, ACE2 has a doubly protective effect on cardiovascular target organs, as it reduces the amount of Ang II available and synthesises Ang-(1–7)–an antifibrotic anti-inflammatory vasodilator – via its MAS receptors. ACE2 therefore has two parallel functions with additive effects: it reduces concentrations of Ang II (clearance); and it has cellular effects that counteract those of the AT1 receptors of Ang II (antagonist).
The activity of ACE2 is generally considered to be increased in all diseases affecting the cardiovascular system, probably via activation of compensatory mechanisms that counteract the effects of Ang II, and which are elevated in these pathological conditions. In these patients, the increase in plasma and tissue concentrations of Ang II leads to overexpression of ACE2, although in amounts that are insufficient to normalise Ang II concentrations and restore a favourable balance between the two pathways.
In patients with a high cardiovascular risk, there is an imbalance between the ACE and ACE2 pathways. ACE2 enzyme activity has been measured in patients with various cardiovascular diseases [14], and is significantly increased in patients with hypertension (+53%) and in patients with heart failure with reduced ejection fraction (+261%), but not in patients with heart failure with preserved ejection fraction. Plasma ACE2 activity is also increased in patients with diabetes, especially those with albuminuria [15].
Hisatake et al. [16] reported that patients with acute heart failure had lower serum ACE activity and plasma aldosterone concentrations than healthy volunteers, whereas there were no differences between the two groups in terms of plasma renin activity or plasma Ang II concentrations. Patients with acute heart failure had higher serum ACE2 concentrations than healthy volunteers (+64%; P = 0.002), but their serum Ang-(1–7) concentrations were significantly lower (–23%; P = 0.005). However, these results should be interpreted with caution because, in this study, patients with acute heart failure were significantly older than the healthy volunteers (62 vs. 47 years); moreover, the sex ratio was not the same in both groups (male/female ratio: 36/13 in the acute heart failure group and 35/3 in the control group). Interestingly, in two larger independent cohorts of patients with heart failure, Sama et al. [17] recently reported that plasma concentrations of ACE2 were higher in men than in women. This study found no correlation between higher plasma ACE2 concentrations and use of either ACE inhibitors or ARBs.
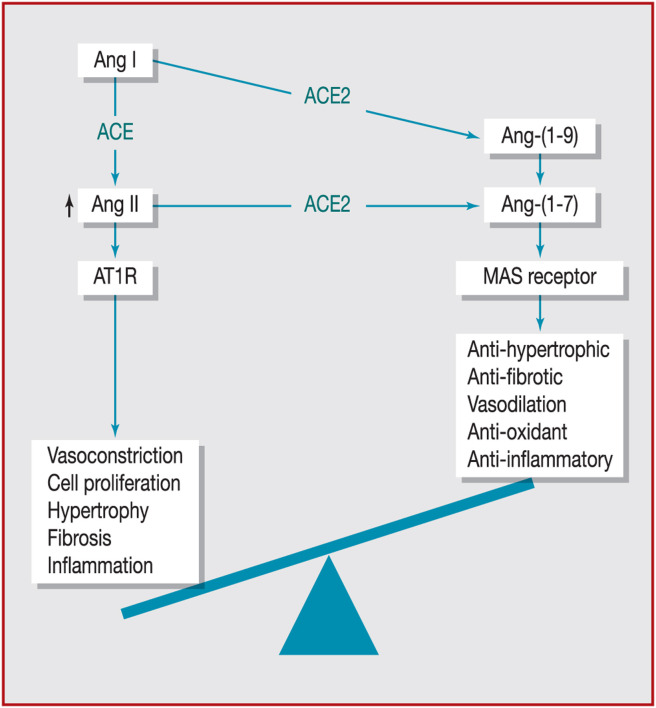
Fig. 3 summarises the imbalance between the synthesis of Ang II by ACE and that of Ang-(1–7) by ACE2 in heart failure.
Figure 3.
The imbalance between the synthesis of angiotensin (Ang) II by angiotensin-converting enzyme (ACE) and that of Ang-(1–7) by angiotensin-converting enzyme 2 (ACE2) in heart failure. Adapted from Patel et al. [13]. AT1: angiotensin II receptor type 1; MAS: mitochondrial assembly.
How ACE inhibitors and ARBs change the expression of ACE2
This is a key point that generated questions – and sometimes panic – in the medical and patient communities after word came out that ACE2 is the receptor for SARS-CoV-2. Part of the misunderstanding stems from the fact that ACE inhibitors are mistaken for ACE2 inhibitors. Any possible effect of ACE inhibitors on ACE2 expression or activity must be indirect, via their respective substrates. Another source of confusion in understanding the effects of RAS blockers on ACE2 is that the effects on transcription and on ACE2 activity can be different; messenger ribonucleic acid and protein levels in experimental data are often found to change in opposite directions. The European Society of Hypertension Task Force [18] produced an excellent review summarising virtually all the experimental and clinical results available to date.
ACE inhibitors and ARBs can have different consequences on the components of the RAS, in particular ACE2, either directly or by downregulation. Once again, there is no direct biochemical effect of ACE inhibitors on ACE2, characterised during its identification as captopril insensitive [11]. Most pharmacological studies have been conducted in animal models and, while they are highly convergent with regard to the effects of ARBs, they are far less so for those of ACE inhibitors.
In the majority of experimental studies, RAS blockade by ARBs increased ACE2 transcription, expression and activity [19], [20], [21]. Ang II acutely induced ACE2 internalisation via its AT1 receptor in ACE2-transfected cultured cells [22]; thus, if ARBs as a drug class upregulate membrane-bound ACE2, it is reasonable to assume that this is because of AT1 receptor blockade.
The effects of ACE inhibitors on ACE2 are not so clear. In animal models, ACE inhibitors exert different effects on ACE2 concentrations; a review of the literature would appear to be in favour of the absence of any clear effect of ACE inhibitors on ACE2 [19]. There are very few published results of human studies showing changes in the expression and activity of ACE2 during ACE inhibition. Esler et al. [23] suggested that the difference arises from an increase in Ang II levels after ARB treatment (but not after ACE inhibition): high Ang II levels would appear to impose an increased substrate load on the enzyme, thus requiring it to be upregulated. We proposed this hypothesis in a letter to Nature Reviews Cardiology [24], and it was confirmed a few weeks later in the same journal by Ferrario et al. – Carlos Ferrario being one of the few research scientists who has been working on the biology of ACE2 for 20 years [25]. Ferrario concluded his letter by saying that ACE inhibitors and ARBs have different effects on ACE2 because of their opposing effects on the concentration of Ang II.
In line with the hypothesis that Ang II levels counter-regulate ACE2, the effects of RAS blockers on ACE2 concentrations could be explained as follows: ACE inhibitors slightly lower the production of Ang II and increase the formation of Ang-(1–7) by increasing the level of Ang I, but also by inhibiting the metabolism of Ang-(1–7) by ACE – the enzyme activity of ACE2 is therefore not altered by ACE inhibitors; blockade of Ang II AT1 receptors results in a sustained increase in plasma and tissue concentrations of Ang II and Ang I – increased levels of Ang II could thus increase ACE2 activity.
Logically, mineralocorticoid receptor blockers reduce the expression of ACE2 in animals [26] and in patients with heart failure [27]. Furthermore, preliminary – but sometimes conflicting – data suggest that aliskiren (a direct renin inhibitor) may alter the expression and activity of ACE2 [28], [29].
In severe forms of COVID-19, ARBs could exert a potentially beneficial pulmonary effect, thus limiting the consequences of the cytokine storm. Decreased alveolar concentrations of ACE2 during acute lung injury result in decreased Ang II metabolism, leading to higher local levels of the peptide, which increase alveolar permeability and worsen lung injury. Thus, ARBs could potentially exert a protective action in the late phases of COVID-19 by counteracting the negative effects of Ang II. Several clinical trials are currently underway to test this hypothesis.
How do RAS blockers affect COVID-19?
In the early days of the COVID-19 epidemic, it was questioned whether patients with increased ACE2 expression and elevated enzyme activity were at higher risk of infection [5]. Indeed, concurrent cerebrovascular disease, diabetes, high blood pressure or coronary heart disease were frequently reported early in the epidemic [30], [31]. Rapidly, the link between the increase in ACE2 in these diseases and the penetration of SARS-CoV-2 led to the question of whether chronic treatment with renin–angiotensin antagonists was likely to increase the risk or severity of COVID-19.
The hypothesis was that SARS-CoV-2 not only uses ACE2 to enter cells, but also reduces ACE2 concentrations, thus reducing the protective effects of ACE2 [32]. In addition, it was suggested that high concentrations of Ang II during SARS-CoV-2 infection may be partly responsible for inflammatory complications [33]. In one study involving only a few patients, high plasma concentrations of Ang II were correlated with viral load and severe lung damage [34].
As RAS blockers may have a potentially harmful effect in COVID-19, it was initially recommended that these agents should be replaced by calcium channel blockers, which have no known effect on ACE2 [20]. However, learned societies in the USA, Europe and France all rightly recommended that treatment with an ACE inhibitor or an ARB should not be discontinued. Although the risks associated with stopping these treatments are very well known, their effect on viral diseases was only assumed.
We now have observational studies that have provided simple coherent answers to the questions. The results of these clinical studies were published in record time, are consistent, and were the subject of an editorial in the New England Journal of Medicine in early May [35].
Mancia et al. [36] conducted a case-control study, matching 6272 patients with COVID-19 to 30,759 controls based on age, sex and place of residence. Neither ACE inhibitors nor ARBs were associated with an increased likelihood of infection by SARS-CoV-2. The conclusion that RAS blockers do not modify susceptibility to COVID-19 applies to both sexes, and is irrespective of age. Reassuringly, additional analyses showed that RAS blockers were not associated with adverse disease progression in patients with COVID-19.
Reynold et al. [37] studied computerised medical records from New York University. Of the 12,594 patients tested for suspected COVID-19, 5894 were positive, 1002 of whom presented with severe forms, defined by admission to intensive care or the need for mechanical ventilation or death of the patient. The relationship between previous pharmacological treatments and the likelihood and severity of infection were tested in the entire population and then in those with hypertension. After propensity score analysis, no association was found between treatment with ACE inhibitors, ARBs or any other antihypertensive class.
An European review [16] has shown that there is no clear evidence that hypertension per se is a risk factor for infection with SARS-CoV-2 or for severity of COVID-19. The study even appears to suggest that ACE inhibitors and ARBs could be associated with a relative protective effect against SARS-CoV-2 infection and, in affected patients, with a better prognosis. A recent clinical study [38] suggested that among patients with COVID-19 who had hypertension, those receiving a RAS blocker had significantly lower concentrations of C-reactive protein (P = 0.049) and procalcitonin (P = 0.008) compared with patients receiving another class of antihypertensive drugs. Of note, the two groups of patients had similar blood pressure measurements. Furthermore, a lower proportion of critical patients (9.3% vs. 22.9%; P = 0.061) and a lower death rate (4.7% vs. 13.3%; P = 0.216) were observed in the RAS blocker group, although these differences failed to reach statistical significance.
Knowledge of the pathophysiology of SARS-CoV-2 is growing rapidly, and a number of potential therapeutic approaches are being considered. Injection of recombinant ACE2 is one therapeutic strategy currently being tested [39]. This would enable SARS-CoV-2 to be captured in the blood, thus reducing its dispersal to the organs expressing ACE2 (heart, kidney, liver and vessels), and thereby limiting the consequences of the sepsis that occurs in severe forms of COVID-19. Another therapeutic approach is based on the major role played by Ang II in the immune storm manifestations of severe stages of COVID-19 in addition to inflammatory cytokines [40], [41]. ACE inhibitors are being used to reduce Ang II production, whereas ARBs are being used to curb its effects in an interesting attempt to limit the adverse course of COVID-19. Thus losartan, the first drug in the ARB class, is currently being tested in both outpatients [42] and hospitalised patients [43]. Lastly, Italian teams are conducting a new multicentre study (NCT04318418) [44] that will include 3000 hospitalised patients with COVID-19 with pulmonary symptoms. Patients will be classed according to the degree of severity of lung disease, and compared with control patients who have been infected by SARS-CoV-2, but have no pulmonary symptoms or a minor form of pulmonary impairment. The relationship between ACE inhibitor or ARB therapy and the severity of COVID-19 will be assessed by multivariable analysis.
Conclusions
ACE is often confused with ACE2, whereas these two different enzymes have different sites of activity, different substrates and different biological effects. This has caused a certain amount of confusion and concern among the public, and even among physicians. Many patients have felt the need to call their physicians to enquire about the risk related to hypertension, and whether they should discontinue treatment with RAS blockers. Faced with uncertainty, many physicians in both general practice surgeries and emergency and infectious disease departments have stopped or modified RAS blocker therapy. The effects of ACE inhibitors on ACE2 activity are indirect, and there is very little clinical evidence of positive regulation of ACE2 by ACE inhibitors or ARBs in humans. In addition, there is no clear clinical evidence that RAS blockers (ACE inhibitors or ARBs) are associated with an increased risk of SARS-CoV-2 infection or an increased risk of adverse COVID-19 progression. Thus, learned societies in the USA, Europe and France all unanimously and strongly recommend that patients taking ACE inhibitors or ARBs for hypertension, heart failure or other medical indications should continue their current therapy unless specifically advised otherwise by their physician or healthcare provider. Currently available data on COVID-19 do not support the differential use of ACE inhibitors or ARBs in these patients.
Disclosure of interest
The authors declare that they have no competing interest.
References
- 1.Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xia H., Lazartigues E. Angiotensin-converting enzyme 2 in the brain: properties and future directions. J Neurochem. 2008;107:1482–1494. doi: 10.1111/j.1471-4159.2008.05723.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Harris R.C. Podocyte ACE2 protects against diabetic nephropathy. Kidney Int. 2012;82:255–256. doi: 10.1038/ki.2012.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hamming I., Cooper M.E., Haagmans B.L., et al. The emerging role of ACE2 in physiology and disease. J Pathol. 2007;212:1–11. doi: 10.1002/path.2162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gustafson D., Raju S., Wu R., et al. Overcoming barriers: the endothelium as a linchpin of coronavirus disease 2019 pathogenesis? Arterioscler Thromb Vasc Biol. 2020;40:1818–1829. doi: 10.1161/ATVBAHA.120.314558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Viner R.M., Whittaker E. Kawasaki-like disease: emerging complication during the COVID-19 pandemic. Lancet. 2020;395:1741–1743. doi: 10.1016/S0140-6736(20)31129-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hoffmann M., Kleine-Weber H., Schroeder S., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271e8–280e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fang L., Karakiulakis G., Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020;8:e21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tipnis S.R., Hooper N.M., Hyde R., Karran E., Christie G., Turner A.J. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J Biol Chem. 2000;275:33238–33243. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- 10.Sampaio W.O., Souza dos Santos R.A., Faria-Silva R., da Mata Machado L.T., Schiffrin E.L., Touyz R.M. Angiotensin-(1-7) through receptor MAS mediates endothelial nitric oxide synthase activation via Akt-dependent pathways. Hypertension. 2007;49:185–192. doi: 10.1161/01.HYP.0000251865.35728.2f. [DOI] [PubMed] [Google Scholar]
- 11.Guang C., Phillips R.D., Jiang B., Milani F. Three key proteases–angiotensin-I-converting enzyme (ACE), ACE2 and renin–within and beyond the renin–angiotensin system. Arch Cardiovasc Dis. 2012;105:373–385. doi: 10.1016/j.acvd.2012.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Patel V.B., Zhong J.C., Grant M.B., Oudit G.Y. Role of the ACE2/Angiotensin 1-7 Axis of the Renin-Angiotensin System in Heart Failure. Circ Res. 2016;118:1313–1326. doi: 10.1161/CIRCRESAHA.116.307708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gheblawi M., Wang K., Viveiros A., et al. Angiotensin-Converting Enzyme 2: SARS-CoV-2 Receptor and Regulator of the Renin-Angiotensin System: celebrating the 20th anniversary of the discovery of ACE2. Circ Res. 2020;126:1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Uri K., Fagyas M., Kertesz A., et al. Circulating ACE2 activity correlates with cardiovascular disease development. J Renin Angiotensin Aldosterone Syst. 2016:17. doi: 10.1177/1470320316668435. 1470320316668435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Soro-Paavonen A., Gordin D., Forsblom C., et al. Circulating ACE2 activity is increased in patients with type 1 diabetes and vascular complications. J Hypertens. 2012;30:375–383. doi: 10.1097/HJH.0b013e32834f04b6. [DOI] [PubMed] [Google Scholar]
- 16.Hisatake S., Kiuchi S., Kabuki T., Oka T., Dobashi S., Ikeda T. Serum angiotensin-converting enzyme 2 concentration and angiotensin-(1-7) concentration in patients with acute heart failure patients requiring emergency hospitalisation. Heart Vessels. 2017;32:303–308. doi: 10.1007/s00380-016-0877-z. [DOI] [PubMed] [Google Scholar]
- 17.Sama I.E., Ravera A., Santema B.T., et al. Circulating plasma concentrations of angiotensin-converting enzyme 2 in men and women with heart failure and effects of renin-angiotensin-aldosterone inhibitors. Eur Heart J. 2020;41:1810–1817. doi: 10.1093/eurheartj/ehaa373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kreutz R., Algharably E.A.E., Azizi M., et al. Hypertension, the renin–angiotensin system, and the risk of lower respiratory tract infections and lung injury: implications for COVID-19. Cardiovasc Res. 2020;116:1688–1699. doi: 10.1093/cvr/cvaa097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ferrario C.M., Jessup J., Chappell M.C., et al. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 20.Soler M.J., Ye M., Wysocki J., William J., Lloveras J., Batlle D. Localisation of ACE2 in the renal vasculature: amplification by angiotensin II type 1 receptor blockade using telmisartan. Am J Physiol Renal Physiol. 2009;296:F398–F405. doi: 10.1152/ajprenal.90488.2008. [DOI] [PubMed] [Google Scholar]
- 21.Wang X., Ye Y., Gong H., et al. The effects of different angiotensin II type 1 receptor blockers on the regulation of the ACE-AngII-AT1 and ACE2-Ang(1-7)-MAS axes in pressure overload-induced cardiac remodelling in male mice. J Mol Cell Cardiol. 2016;97:180–190. doi: 10.1016/j.yjmcc.2016.05.012. [DOI] [PubMed] [Google Scholar]
- 22.Deshotels M.R., Xia H., Sriramula S., Lazartigues E., Filipeanu C.M. Angiotensin II mediates angiotensin converting enzyme type 2 internalisation and degradation through an angiotensin II type I receptor-dependent mechanism. Hypertension. 2014;64:1368–1375. doi: 10.1161/HYPERTENSIONAHA.114.03743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Esler M., Esler D. Can angiotensin receptor-blocking drugs perhaps be harmful in the COVID-19 pandemic? J Hypertens. 2020;38:781–782. doi: 10.1097/HJH.0000000000002450. [DOI] [PubMed] [Google Scholar]
- 24.Mourad J.J., Levy B.I. Interaction between RAAS inhibitors and ACE2 in the context of COVID-19. Nat Rev Cardiol. 2020;17:313. doi: 10.1038/s41569-020-0368-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ferrario C.M., Ahmad S., Groban L. Mechanisms by which angiotensin-receptor blockers increase ACE2 levels. Nat Rev Cardiol. 2020;17:378. doi: 10.1038/s41569-020-0387-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Karram T., Abbasi A., Keidar S., et al. Effects of spironolactone and eprosartan on cardiac remodelling and angiotensin-converting enzyme isoforms in rats with experimental heart failure. Am J Physiol Heart Circ Physiol. 2005;289:H1351–H1358. doi: 10.1152/ajpheart.01186.2004. [DOI] [PubMed] [Google Scholar]
- 27.Keidar S., Gamliel-Lazarovich A., Kaplan M., et al. Mineralocorticoid receptor blocker increases angiotensin-converting enzyme 2 activity in congestive heart failure patients. Circ Res. 2005;97:946–953. doi: 10.1161/01.RES.0000187500.24964.7A. [DOI] [PubMed] [Google Scholar]
- 28.Ding W., Li X., Wu W., et al. Aliskiren inhibits angiotensin II/angiotensin 1-7(Ang II/Ang1-7) signal pathway in rats with diabetic nephropathy. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2018;34:891–895. [PubMed] [Google Scholar]
- 29.Hsu C.N., Wu K.L., Lee W.C., Leu S., Chan J.Y., Tain Y.L. Aliskiren administration during early postnatal life sex-specifically alleviates hypertension programmed by maternal high fructose consumption. Front Physiol. 2016;7:299. doi: 10.3389/fphys.2016.00299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Guan W.J., Ni Z.Y., Hu Y., et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang X., Yu Y., Xu J., et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centred, retrospective, observational study. Lancet Respir Med. 2020;8:475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lambert D.W., Yarski M., Warner F.J., et al. Tumour necrosis factor-alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe-acute respiratory syndrome-coronavirus (SARS-CoV) receptor, angiotensin-converting enzyme-2 (ACE2) J Biol Chem. 2005;280:30113–30119. doi: 10.1074/jbc.M505111200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gurwitz D. Angiotensin receptor blockers as tentative SARS-CoV-2 therapeutics. Drug Dev Res. 2020;81:537–540. doi: 10.1002/ddr.21656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu Y., Yang Y., Zhang C., et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63:364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jarcho J.A., Ingelfinger J.R., Hamel M.B., D’Agostino R.B., Sr., Harrington D.P. Inhibitors of the Renin-Angiotensin-Aldosterone System and COVID-19. N Engl J Med. 2020;382:2462–2464. doi: 10.1056/NEJMe2012924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mancia G., Rea F., Ludergnani M., Apolone G., Corrao G. Renin–Angiotensin–Aldosterone system blockers and the risk of COVID-19. N Engl J Med. 2020;382:2431–2440. doi: 10.1056/NEJMoa2006923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Reynolds H.R., Adhikari S., Pulgarin C., et al. Renin-Angiotensin-Aldosterone system inhibitors and risk of COVID-19. N Engl J Med. 2020;382:2441–2448. doi: 10.1056/NEJMoa2008975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yang G., Tan Z., Zhou L., et al. Effects of Angiotensin II Receptor Blockers and ACE (Angiotensin-Converting Enzyme) Inhibitors on Virus Infection, Inflammatory Status, and Clinical Outcomes in Patients With COVID-19 and Hypertension: a single-centre retrospective study. Hypertension. 2020;76:51–58. doi: 10.1161/HYPERTENSIONAHA.120.15143. [DOI] [PubMed] [Google Scholar]
- 39.2020. Recombinant Human Angiotensin-converting Enzyme 2 (rhACE2) as a Treatment for patients with COVID-19 (APN01-COVID-19)https://clinicaltrials.gov/ct2/show/NCT04335136.com [ClinicalTrials.gov Identifier: NCT04335136] [Google Scholar]
- 40.Gu H., Xie Z., Li T., et al. Angiotensin-converting enzyme 2 inhibits lung injury induced by respiratory syncytial virus. Sci Rep. 2016;6:19840. doi: 10.1038/srep19840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zou Z., Yan Y., Shu Y., et al. Angiotensin-converting enzyme 2 protects from lethal avian influenza A H5N1 infections. Nat Commun. 2014;5:3594. doi: 10.1038/ncomms4594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.2020. Losartan for patients with COVID-19 not requiring hospitalisation. https://clinicaltrials.gov/ct2/show/NCT04311177?term=NCT04311177&draw=2&rank=1.com [ClinicalTrials.gov Identifier: NCT04311177] [Google Scholar]
- 43.2020. Losartan for patients with COVID-19 requiring hospitalisation. https://clinicaltrials.gov/ct2/show/NCT04312009?term=NCT04312009?term=NCT04312009&draw=2&rank=1.com [ClinicalTrials.gov Identifier: NCT04312009] [Google Scholar]
- 44.Di Castelnuovo A., De Caterina R., de Gaetano G., Iacoviello L. Controversial relationship between renin-angiotensin system inhibitors and severity of COVID-19: announcing a large multicentre case-control study in Italy. Hypertension. 2020 doi: 10.1161/HYPERTENSIONAHA.120.15370. [DOI] [PubMed] [Google Scholar]
- 45.Clerkin K.J., Fried J.A., Raikhelkar J., et al. COVID-19 and cardiovascular disease. Circulation. 2020;141:1648–1655. doi: 10.1161/CIRCULATIONAHA.120.046941. [DOI] [PubMed] [Google Scholar]