Abstract
Introduction
Plasma levels of eight combined proteins have shown value as biomarkers for detection of colorectal cancer (CRC). However, their value in identifying colorectal adenoma needs further evaluation. The aim was to evaluate the eight proteins (AFP, CA19-9, CEA, CyFra21-1, Ferritin, Galectin-3, hs-CRP and TIMP-1) in detection of high-risk adenoma (HRA) and in prediction of recurrence of adenoma. Furthermore, the discrimination between HRA and low-risk adenoma (LRA) or CRC lesions was evaluated.
Methods
The study included 4698 individuals undergoing diagnostic colonoscopy. Automated ELISA platforms were used in the determination of protein levels in samples collected just before colonoscopy.
Results
Univariably, five proteins (AFP, CEA, CyFra21-1, hs-CRP and TIMP-1), respectively, significantly discriminated individuals with HRA from individuals with non-malignant findings. Multivariably, the combination of CEA and hs-CRP improved performance; AUC= 0.63 (sensitivity=0.19 at specificity=0.90). CyFra21-1, Ferritin and TIMP-1 demonstrated significant discrimination between individuals with HRA and LRA in univariable analyses, respectively. Performance was improved in multivariable analysis; AUC=0.61 (sensitivity=0.13 at specificity=0.90). Discrimination between individuals with colorectal adenomas and healthy individuals was significant for CA19-9, CEA, hs-CRP and TIMP-1, respectively, in univariable analyses. Multivariable analysis improved performance; AUC=0.63 (sensitivity=0.17 at specificity=0.90). All proteins except AFP demonstrated significant discrimination between individuals with HRA and CRC. Combination of CEA, CyFra21-1, Ferritin, hs-CRP and TIMP-1 in multivariable analysis improved discrimination; AUC=0.78 (sensitivity=0.34 at specificity=0.90). Association between plasma levels of any of the eight proteins and recurrence of colorectal adenomas after endoscopic removal could not be demonstrated.
Discussion
The protein panel shows a promising potential in detection of colorectal adenomas in general, but specifically of HRA. However, improvements are needed for the panel to be valuable as a screening test. Finally, plasma levels of the eight proteins were not predictive of recurrence of colorectal adenomas.
Keywords: biomarkers, tumor, neoplasm recurrence, local, colorectal adenomas, colorectal neoplasms, alpha-feto protein, cancer antigen 19-9, carcino embryogenic antigen, cytokeratin fragment 21-1, Ferritin, Galectin-3, high sensitivity C-reactive protein, tissue inhibitor of metalloproteinases-1
Introduction
Colorectal cancer (CRC) is a leading cause of cancer-related deaths.1 Diagnostics and treatments of CRC have improved substantially during the last decade, but the disease continues to pose a major health challenge.2 Early diagnosis of CRC increases overall survival,3 and population-based screening programs have been implemented in many countries worldwide.4 In addition, detection and removal of pre-cancerous lesions (colorectal adenomas) identified via screening has led to a decrease of CRC incidence,5 which makes colorectal adenomas an important diagnostic target.
The natural history of CRC occurs over years and involves progression from low risk to high risk precancerous adenomas and, eventually, to carcinoma.6 The lifetime-incidence of colorectal adenomas is approximately 35%,7 and the majority of CRC cases develop through malignant transformation of high-risk adenoma (HRA).8 It has been estimated that 10% of all adenomas and approximately 25% of HRA will progress to CRC;9 diagnosis and removal of precancerous adenomas prevent progression to cancer.10 Therefore, adenoma detection has a high priority in screening and early detection of colorectal neoplasia.
The predominant design of current screening programs includes an initial screening test to select a high-risk group for subsequent diagnostic colonoscopy.4 Available clinical-approved screening tests for CRC include feces based tests (guaiac fecal occult blood test (gFOBT), fecal immunochemical test (FIT), multitarget stool DNA test (Cologuard)) and blood-based test for methylated SEPT9 DNA (mSEPT9) (Epi proColon). However, the performance of the screening tests in identification of advanced, high-risk adenomas (HRA) are not impressive; sensitivity=20-34% at specificity=91-97% for FIT,11,12 sensitivity=9-14% at specificity=92-95% for gFOBT,11,12 sensitivity=42-54% at specificity=90% for multitarget stool DNA test13,14 and sensitivity=11-21% for mSEPT9 test (specificity not reported).15,16 The inadequate performance of screening tests for detection of HRA combined with the fact that almost all adenomas are asymptomatic could lead to individuals that are not identified with HRA and thereby are not offered colonoscopy (false-negative result), and indeed colonoscopy examinations may be offered to individuals without adenomas by a false-positive result of the screening test. Furthermore, the determination of the malignant potential of adenomas by colonoscopy is challenging.17,18 The ability to identify individuals with HRA and to differentiate between individuals with low-risk adenoma (LRA) and potentially malignantly transformable HRA, as well as to identify individuals at risk of recurrence of adenomas after polypectomy, would provide an opportunity to prioritize the diagnostic and follow-up colonoscopies offered to individuals in current and future screening programs.
The need for developing new screening options for early detection of CRC and HRA is urgent, and attention has been focused on identifying new blood-based biomarkers as the compliance associated with stool-based tests is estimated at 60–65%.19–21
A key element in the process of oncogenesis is genomic and transcriptional alterations, which consequently changes the expression of various proteins in tumor tissue and in the circulation. These proteins represent a major entity of blood-based biomarkers, and determination of proteins in the circulation, individually or in combination, may be used in detection of individuals with colorectal adenomas or CRC.22–27 Various blood-based biomarkers have shown value although limited in discrimination between LRA and HRA28,29 as well as in prediction of recurrence following polypectomy.30 Of particular interest is a previous evaluation of a panel of eight blood-based proteins associated with CRC indicating a possible future role as biomarkers for early detection of CRC.31 The eight proteins of the panel were chosen based on characteristics and current knowledge of their role in the carcinogenesis of CRC (Table 1). However, the accuracy of this specific protein panel in HRA detection and prediction of recurrence needs further elucidation.
Table 1.
Characteristics and Current Knowledge in Association with CRC and HRA of AFP, CA19-9, CEA, CyFra21-1, Ferritin, Galectin-3, Hs-CRP and TIMP-1
| Marker | References | |
|---|---|---|
| AFP |
|
[32,33] |
| CA19-9 |
|
[33–37] |
| CEA |
|
[34–38] |
| CyFra21-1 |
|
[27,39,40] |
| Ferritin |
|
[26,27,37,41,42] |
| Galectin-3 |
|
[43] |
| hs-CRP |
|
[27] |
| TIMP-1 |
|
[44–46] |
Abbreviations: AFP, alpha-feto protein; CA19-9, cancer antigen 19–9; CEA, carcino embryogenic antigen; CRC, colorectal cancer; CyFra21-1, cytokeratin fragment 21–1; HRA, high risk adenoma; hs-CRP, high sensitivity C-reactive protein; TIMP-1, tissue inhibitor of metalloproteinases-1.
The aim of the present study was to evaluate the value of these eight proteins (Table 1) in plasma, individually or combined, in detection of primary HRA and subsequently in prediction of adenoma recurrence. In addition, the aims included whether the proteins could be used to differentiate HRA lesions from LRA lesions, HRA lesions from CRC lesions and whether the proteins could differentiate between individuals with adenoma lesions from individuals with non-malignant findings or no findings (clean colorectum) at colonoscopy.
Materials and Methods
The study was a part of the Endoscopy II protocol.31,47 In brief, blood samples were collected prospectively from individuals referred to diagnostic colonoscopy due to symptoms attributable to CRC. Blood for EDTA-plasma samples were collected before colonoscopy and handled and stored at −80°C within two hours according to a validated Standard Operating Procedure. The Endoscopy II protocol was initiated in 2010 and terminated in 2012 with inclusion of 4698 individuals with valid clinical and biomarker data. A previous publication based on the Endoscopy II protocol evaluated the value of plasma levels of the eight serological proteins in discrimination of individuals with CRC and HRA from the entire group of included individuals.31 The present study was designed as a spin-off of the previous publication with the use of the same analyses methodologies and measurements of the eight proteins. Table 2 presents an overview of findings at colonoscopy in the study.
Table 2.
Inclusion and Findings at Colonoscopy in the Endoscopy II Study
| Men | Women | Total | Median Age | |
|---|---|---|---|---|
| Included individuals in Endoscopy II | 2243 | 2455 | 4698 | 64 (18–96) |
| Individuals with CRC | 306 | 206 | 512 | |
| Stage I–II CRC | 156 | 108 | 264 | |
| Stage III–IV CRC | 150 | 97 | 247 | |
| No stage available | – | 1 | 1 | |
| Individuals with colorectal adenoma | 384 | 305 | 689 | |
| LRA | 168 | 122 | 290 | |
| ● No recurrence at follow-up | 249 | |||
| ● Recurrence at follow-up | 41 | |||
| ● CRC at follow-upa | 2 | |||
| HRA | 216 | 183 | 399 | |
| ● No recurrence at follow-up | 300 | |||
| ● Recurrence at follow-up | 99 | |||
| ● CRC at follow-upa | 9 |
Notes: aNumbers of individuals with CRC at follow-up are included in numbers of individuals with recurrence at follow-up.
Abbreviations: CRC, colorectal cancer, LRA, low-risk colorectal adenoma, HRA, high-risk colorectal adenoma.
Data registered in the Endoscopy II database comprised age, gender, comorbidity and findings at colonoscopy including pathological and histological classification of colorectal adenomas (LRA/HRA) and CRC. The definition of LRA was one lesion ˂1cm, ˂3 lesions, tubular histology or low-grade dysplasia. The definition of HRA was one lesion ≥1 cm, ≥3 lesions, villous histology or high-grade dysplasia.
In the present study, recurrence of colorectal adenoma was recorded in individuals with a primary diagnosis of colorectal adenoma in a period from 12 months after the primary diagnosis and until the end of December 2017 (median follow-up period of 75 (35–94) months).
For further statistical analysis, three endpoints were defined based on outcome at colonoscopy:
Endpoint 1: The discrimination of individuals with HRA from all other individuals with non-malignant findings (including LRA) or clean colorectum.
Endpoint 2: The discrimination of individuals with HRA from individuals with LRA.
Endpoint 3: The discrimination of individuals with HRA from individuals with CRC.
Endpoint 3 was subdivided into
Endpoint 3a: The discrimination of individuals with HRA from individuals with early-stage CRC (Stage I and II).
Endpoint 3b: The discrimination of individuals with HRA from individuals with late-stage CRC (Stage III and IV).
A secondary Endpoint was defined as the discrimination of individuals with HRA and LRA from all other individuals excluding individuals with CRC.
Plasma protein levels were determined by using the Abbott ARCHITECT® i2000 automated immunoassay platform (Abbott laboratory inc., Abbott Park, IL, USA) utilizing a two-step dual monoclonal immunoassay. All the laboratory analyses were performed by Abbott Center of Excellence at VU Medical Center (Amsterdam, The Netherlands), and Abbott in-house research prototype and on-market reagents were used for determinations. All achieved results from the analysis were included in the database.
Statistics
Initial univariable logistic regression analysis with the binary endpoints as the dependent variable and plasma levels of AFP, CA19-9, CEA, CyFra21-1, Ferritin, Galectin-3, hs-CRP and TIMP-1 as explanatory variables (log transformed (base2)) were performed. Individual biomarkers found to have significant univariable discrimination (p-value<0.05), as well as age and gender, were then used as explanatory covariates in multivariable logistic regression analysis reducing the number of explanatory covariates using a stepwise 10-fold cross-validation selection method. For each endpoint, a predictor (a linear combination of the significant explanatory covariates) has been established. The results are presented by the odds ratios (OR) with 95% confidence intervals (CI), sensitivity at 70, 80 and 90% specificity and receiver operating characteristic (ROC) curves with area under the ROC curve (AUCROC) as a measure of discrimination.
Analysis of time to adenoma recurrence has been done using the Cox proportional hazards model with the mean plasma values of the eight protein biomarkers as continuous covariates on the log scale (base 2). The resulting models were assessed using martingale residuals.
P-values less than 5% were considered significant. Database management and statistics were performed using SAS (v9.4, SAS Institute, Cary, N.C., USA). In addition, R was used for statistical calculations (R Development Core Team, Vienna, Austria, http://www.R-project.org).
Results
Descriptive statistics are presented in Table 2.
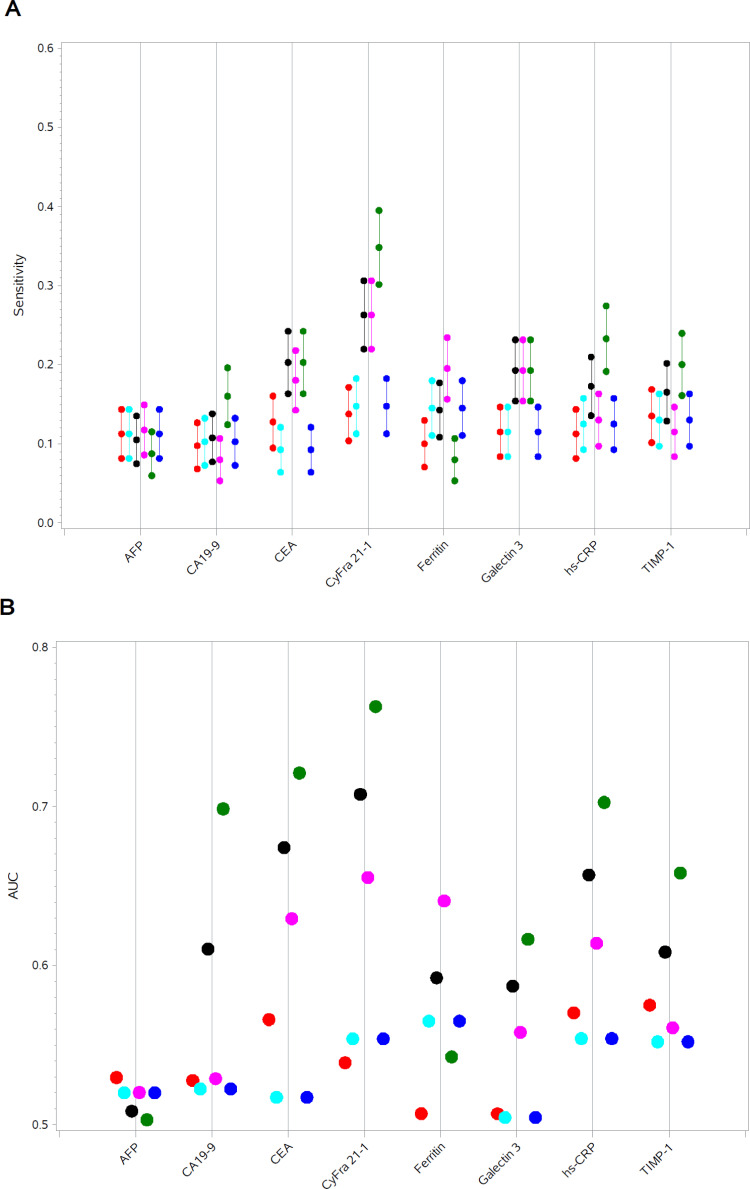
Data from the univariate analyses of Endpoint 1–3 and the secondary Endpoint are illustrated in Figure 1A and B.
Figure 1.
(A) Sensitivities at specificity=0.90 in univariate analyses of Endpoint 1–3 (including 3a and b) and secondary Endpoint. (B) AUCROC in univariate analyses of Endpoint 1–3 (including 3a and 3b) and secondary Endpoint.
Notes: Red: Endpoint 1, Turquise: Endpoint 2, Black: Endpoint 3, Magenta: Endpoint 3a, Green: Endpoint 3b, Blue: Secondary Endpoint.
Abbreviations: AFP, alpha-feto protein; CA19-9, cancer antigen 19–9; CEA, carcino embryogenic antigen; CyFra21-1, cytokeratin fragment 21–1; Hs-CRP, high sensitivity C-reactive protein; TIMP-1, tissue inhibitor of metalloproteinases-1.
In discrimination between individuals with HRA and all other individuals with non-malignant findings or clean colorectum (Endpoint 1), plasma levels of AFP (AUCROC=0.53, p-value=0.033, sensitivity=0.11 at specificity=0.90), CEA (AUCROC=0.57, p-value˂0.001, sensitivity=0.13 at specificity=0.90), CyFra21-1 (AUCROC=0.54, p-value=0.010, sensitivity=0.14 at specificity=0.90), hs-CRP (AUCROC=0.57, p-value˂0.001, sensitivity=0.11 at specificity=0.90) and TIMP-1 (AUCROC=0.58, p-value˂0.001, sensitivity=0.14 at specificity=0.90), respectively, demonstrated significance. Plasma levels of all five proteins were increased in individuals with HRA compared to individuals with non-malignant findings or clean colon (OR=1.11–1.70).
Regarding Endpoint 2, univariable analyses showed that plasma levels of CyFra21-1 (AUCROC=0.55, p-value=0.018, sensitivity=0.15 at specificity=0.90), Ferritin (AUCROC=0.57, p-value=0.010, sensitivity=0.13 at specificity=0.90) and TIMP-1 (AUCROC=0.55, p-value=0.006, sensitivity=0.16 at specificity=0.90) were significantly different among individuals with HRA compared to individuals with LRA, respectively; Plasma levels of CyFra21-1 and TIMP-1 were decreased (OR=0.61–0.81), and plasma levels of Ferritin were increased (OR=1.15).
The results of the univariable analyses for the eight individual proteins in the discrimination of individuals with HRA from individuals with CRC (Endpoint 3) are presented for individuals with all stages of CRC (Endpoint 3), early-stage CRC (stage I+II) (Endpoint 3a) and late-stage CRC (stage III+IV) (Endpoint 3b).
A significant discrimination between individuals with HRA and individuals with CRC (all stages) was demonstrated for plasma levels of all proteins, except AFP; CA19-9 (AUCROC=0.61, p-value˂0.001, sensitivity=0.11 at specificity=0.90), CEA (AUCROC=0.67, p-value˂0.001, sensitivity=0.20 at specificity=0.90), CyFra21-1 (AUCROC=0.71, p-value˂0.001, sensitivity=0.26 at specificity=0.90), Ferritin (AUCROC=0.59, p-value˂0.001, sensitivity=0.14 at specificity=0.90), Galectin-3 (AUCROC=0.59, p-value˂0.001, sensitivity=0.19 at specificity=0.90), hs-CRP (AUCROC=0.66, p-value˂0.001, sensitivity=0.17 at specificity=0.90) and TIMP-1 (AUCROC=0.61, p-value˂0.001, sensitivity=0.17 at specificity=0.90). Plasma levels of CA19-9, CEA, CyFra21-1, Galectin-3, hs-CRP and TIMP-1 were decreased in individuals with HRA compared to individuals with CRC (all stages), (OR=0.48–0.76), and plasma levels of Ferritin were increased in individuals with HRA (OR=1.21).
Similar results were achieved when the analyses were restricted to discriminate individuals with HRA from individuals with late stage CRC (Endpoint 3b). Plasma levels of CA19-9 (AUCROC=0.70, p-value<0.001, sensitivity=0.16 at specificity=0.90), CEA (AUCROC=0.72, p-value<0.001, sensitivity=0.20 at specificity=0.90), CyFra21-1 (AUCROC=0.76, p-value<0.001, sensitivity=0.35 at specificity=0.90), Galectin-3 (AUCROC=0.62, p-value<0.001, sensitivity=0.19 at specificity=0.90), hs-CRP (AUCROC=0.70, p-value<0.001, sensitivity=0.23 at specificity=0.90) and TIMP-1 (AUCROC=0.66, p-value<0.001, sensitivity=0.20 at specificity=0.90) were significantly decreased in individuals with HRA (OR=0.37–0.68). Plasma levels of Ferritin were significantly increased (OR=1.09); AUCROC=0.54 with sensitivity=0.08 at specificity=0.90. Plasma levels of AFP did not reach significance.
Restricting the analyses to discriminate individuals with HRA from individuals with early stage CRC (Endpoint 3a), plasma levels of CA19-9 (AUCROC=0.53, p-value=0.021, sensitivity=0.08 at specificity=0.90), CEA (AUCROC=0.63, p-value<0.001, sensitivity=0.18 at specificity=0.90), CyFra21-1 (AUCROC=0.66, p-value<0.001, sensitivity=0.26 at specificity=0.90), Ferritin (AUCROC=0.64, p-value<0.001, sensitivity=0.20 at specificity=0.90), Galectin-3 (AUCROC=0.56, p-value=0.020, sensitivity=0.19 at specificity=0.90) and hs-CRP (AUCROC=0.61, p-value<0.001, sensitivity=0.13 at specificity=0.90) were persistently significant, but plasma levels of TIMP-1 lost significance. Plasma levels of CA19-9, CEA, CyFra21-1, Galectin-3 and hs-CRP were decreased (OR=0.54–0.87) and plasma levels of Ferritin were increased (OR=1.36) in individuals with HRA compared to individuals with early stage CRC.
For the secondary Endpoint (discrimination of individuals with HRA and LRA from all other individuals excluding individuals with CRC), the plasma levels of CA19-9 (AUCROC=0.54, p-value=0.004, sensitivity=0.13 at specificity=0.90), CEA (AUCROC=0.56, p-value<0.001, sensitivity=0.14 at specificity=0.90), hs-CRP (AUCROC=0.55, p-value<0.001, sensitivity=0.15 at specificity=0.90) and TIMP-1 (AUCROC=0.56, p-value<0.001, sensitivity=0.14 at specificity=0.90) were significantly decreased in individuals (OR=0.69–0.92) with adenomas compared to all other individuals.
Results of the multivariable analyses for Endpoint I, Endpoint 2, Endpoint 3 and the secondary Endpoint are presented in Table 3.
Table 3.
Results of Multivariate Analyses of Endpoint 1–3 and Secondary Endpoint
| Odds Ratio | Lower CI | Upper CI | p-value | AUCROC | Sensitivity at 70% Specificity | Sensitivity at 80% Specificity | Sensitivity at 90% Specificity | |
|---|---|---|---|---|---|---|---|---|
| Endpoint 1 | ||||||||
| CEA | 1.17 | 1.05 | 1.31 | 0.003 | 0.63 | 0.49 | 0.33 | 0.19 |
| Hs-CRP | 1.07 | 1.01 | 1.13 | 0.020 | ||||
| Endpoint 2 | ||||||||
| Ferritin | 0.85 | 0.76 | 0.94 | 0.003 | 0.61 | 0.41 | 0.33 | 0.13 |
| TIMP-1 | 1.60 | 1.11 | 2.32 | 0.012 | ||||
| Endpoint 3 | ||||||||
| CA19-9 | 0.89 | 0.80 | 1.00 | 0.048 | 0.78 | 0.70 | 0.56 | 0.34 |
| CEA | 0.70 | 0.61 | 0.80 | ˂0.001 | ||||
| CyFra21-1 | 0.60 | 0.50 | 0.73 | ˂0.001 | ||||
| Ferritin | 1.34 | 1.22 | 1.48 | ˂0.001 | ||||
| Hs-CRP | 0.82 | 0.75 | 0.89 | ˂0.001 | ||||
| TIMP-1 | 1.94 | 1.30 | 2.90 | 0.001 | ||||
| Endpoint 3a | ||||||||
| CEA | 0.71 | 0.61 | 0.83 | ˂0.001 | 0.74 | 0.64 | 0.48 | 0.28 |
| CyFra21-1 | 0.73 | 0.59 | 0.90 | 0.003 | ||||
| Ferritin | 1.43 | 1.28 | 1.59 | ˂0.001 | ||||
| Hs-CRP | 0.86 | 0.78 | 0.95 | 0.002 | ||||
| Endpoint 3b | ||||||||
| CA19-9 | 0.78 | 0.68 | 0.89 | ˂0.001 | 0.80 | 0.67 | 0.58 | 0.39 |
| CEA | 0.71 | 0.60 | 0.83 | ˂0.001 | ||||
| CyFra21-1 | 0.55 | 0.44 | 0.67 | ˂0.001 | ||||
| Secondary Endpoint | ||||||||
| AFP | 1.10 | 1.00 | 1.21 | 0.048 | 0.63 | 0.50 | 0.37 | 0.17 |
| CEA | 1.20 | 1.10 | 1.31 | ˂0.001 | ||||
| CyFra21-1 | 0.88 | 0.80 | 0.98 | 0.018 | ||||
| Hs-CRP | 1.05 | 1.01 | 1.10 | 0.024 |
Abbreviations: AFP, alpha-feto protein; CA19-9, cancer antigen 19–9; CEA, carcino embryogenic antigen; CyFra21-1, cytokeratin fragment 21–1; Hs-CRP, high sensitivity C-reactive Protein; TIMP-1, tissue inhibitor of metalloproteinases-1.
For Endpoint 1, the multivariable analysis included CEA and hs-CRP; AUCROC=0.63 with sensitivity=0.19 at specificity=0.90.
The multivariable analysis for Endpoint 2 included Ferritin and TIMP-1; AUCROC=0.61 with sensitivity=0.13 at specificity=0.90.
For Endpoint 3, the multivariable analysis included CA19-9, CEA, CyFra21-1, Ferritin, hs-CRP and TIMP-1 and showed AUCROC=0.78 with sensitivity=0.34 at specificity=0.90. Similar results were achieved when the discrimination was restricted to individuals with HRA from individuals with early-stage CRC (stage I+II) (Endpoint 3a); the analysis included CEA, CyFra21-1, Ferritin and hs-CRP; AUCROC=0.74 at sensitivity=0.28 at specificity=0.90. Restricting the control group to late-stage CRC (Endpoint 3b), CA19-9, CEA and CyFra21-1 were included in the multivariable analysis; AUCROC=0.80 at sensitivity=0.39 at specificity=0.90.
The multivariable analysis for the secondary Endpoint included AFP, CEA, CyFra21-1 and hs-CRP; AUCROC=0.63 with sensitivity=0.17 at specificity=0.90.
The potential use of plasma levels of the eight proteins in prediction of recurrence of colorectal adenomas after endoscopic removal was assessed. The hazard ratios (HR) calculated for individual plasma levels of all eight proteins and adenoma recurrence by multivariate analyses with 10-fold cross-validation were not significant for any of the proteins as shown in Figure 2. Adenoma pathology (LRA/HRA) as a predictor of recurrence was also included, and the presence of HRA showed significant increased HR (HR=3.01–3.11) compared to the presence of LRA (data not shown). In individuals with colorectal adenoma at baseline (inclusion), nine individuals with HRA and two individuals with LRA were diagnosed with CRC during the follow-up period (Table 2). Among these individuals, plasma levels of the eight proteins investigated in this study were not found significantly altered compared to levels in individuals with colorectal adenomas at baseline without CRC during the follow-up period (data not shown).
Figure 2.
Hazard ratios for individual plasma levels of AFP, CA19-9, CEA, CyFra21-1, Ferritin, Galectin-3, hs-CRP and TIMP-1 and adenoma recurrence.
Abbreviations: AFP, alpha-feto protein; CA19-9, cancer antigen 19–9; CEA, carcino embryogenic antigen; CyFra21-1, cytokeratin fragment 21–1; Hs-CRP, high sensitivity C-reactive protein; TIMP-1, tissue inhibitor of metalloproteinases-1.
Discussion
In the present study, the value of eight plasma proteins was evaluated as individual or combined biomarkers for detection of HRA. Furthermore, plasma levels of the proteins determined at the time of primary diagnosis of colorectal adenoma were evaluated as predictors of adenoma recurrence. The study was spin-off from a previous publication based on the Endoscopy II protocol31 with the primary aim to evaluate the value of the identical protein panel as biomarkers for early detection of CRC.
In the discrimination of individuals with HRA from individuals with non-malignant findings (including LRA) or clean colorectum, plasma levels of five proteins (AFP, CEA, CyFra21-1, hs-CRP and TIMP-1) were significant with increased plasma levels in individuals with HRA, but the discrimination was weak. The combination of two proteins (CEA and hs-CRP) in a multivariate analysis improved the performance, but the discrimination was still only moderate.
Accuracies of approved tests for CRC screening in discrimination of individuals with advanced adenoma from individuals with non-malignant findings or clean colorectum are reported as sensitivities=20-34% at specificities=91-97% for FIT,11,12 sensitivities=9-14% at specificities=92-95% for gFOBT,11,12 sensitivities=42-54% at specificity=90% for multitarget stool DNA test13, 14 and sensitivities=11-21% for mSEPT9 test (specificity not reported).15,16 A direct comparison between these tests and the results of the present study is not possible due to differences in design and methods. However, it is indicated that the accuracy of the multivariable analysis of the current study in discrimination of individuals with HRA from individuals with non-malignant findings or clean colorectum are comparable to the accuracy of gFOBT and mSEPT9 tests, but are inferior to the accuracy of FIT and multi-target stool DNA test. Overall, it is concluded that the accuracy of the eight proteins evaluated in the study is too moderate to discriminate individuals with HRA from healthy individuals to be valuable in daily routine of screening for HRA.
The three proteins CyFra21-1, Ferritin and TIMP-1, respectively, were shown to statistically discriminate between individuals with HRA and individuals with LRA. However, the discrimination was weak. Plasma levels of Ferritin were increased in individuals with HRA, and levels of CyFra21-1 and TIMP-1 were decreased. Combination of two of the proteins (Ferritin and TIMP-1) in a multivariable analysis improved the discrimination slightly. In conclusion, plasma levels of the investigated proteins (individually or in combination) are not valuable as biomarkers for identification of individuals with HRA in a population with colorectal adenoma.
Furthermore, the performance in discrimination of individuals with colorectal adenomas from individuals with other non-malignant findings or clean colorectum at colonoscopy, whilst statistically significant, was not strong enough to be valuable as biomarkers, and the use in daily routines is therefore limited.
When evaluated individually, all proteins except AFP were found to significantly discriminate individuals with HRA from individuals with CRC (all stages) with a moderate discrimination. Similar results were obtained when the discrimination was restricted to individuals with HRA from individuals with late-stage CRC, but the discrimination was improved. In the discrimination of individuals with HRA from individuals with early-stage CRC, TIMP-1 and AFP were not shown as valuable biomarkers. The discrimination using the remaining proteins was moderate and similar to the discrimination of individuals with HRA from individuals with CRC (all stages).
Plasma levels of six out the seven significant proteins (CA19-9, CEA, CyFra21-1, Galectin-3, hs-CRP, TIMP-1) were increased in individuals with CRC compared to individuals with HRA with the exception of Ferritin where plasma levels were decreased in individuals with CRC. The increased plasma levels could be explained by the positive correlation of the six proteins with colorectal neoplasia previously described in the literature where increasing levels during disease progression have been observed (Table 1). The interaction between plasma levels of Ferritin and colorectal neoplasia is, however, more complex. Even though possible positive correlations between levels of Ferritin and cancer-specific processes (including CRC) have been shown, iron deficiency anemia caused by chronic gastrointestinal bleeding might antagonize this effect.41,42 The progression in colorectal neoplasia from HRA to CRC might cause increased gastrointestinal bleeding, which could explain why levels of Ferritin are decreased in individuals with CRC compared to individuals with HRA in the present study.
The combination of six significant proteins (CA19-9, CEA, CyFra21-1, Ferritin, hs-CRP and TIMP-1) in a multivariable analysis improved the discrimination of individuals with HRA from individuals with CRC (all stages). Restricting the control group to early-stage CRC, two proteins lost significance (CA19-9 and TIMP-1), and four proteins were included in the model (CEA, CyFra21-1, Ferritin and hs-CRP). Restricting the control group to late-stage CRC, three proteins lost significance compared to the discrimination from all stages (Ferritin, hs-CRP and TIMP-1), and three proteins (CA19-9, CEA and CyFra21-1) were included in the model.
The discrimination of individuals with HRA by a single significant protein or proteins in combination improved when the control group was restricted to individuals with late-stage CRC compared to individuals with early-stage CRC or all stages of CRC. This might also be explained by the positive correlation between the proteins and CRC as previously described (Table 1). Consequently, a potential difference in plasma levels of the proteins would be more pronounced as the carcinogenesis of CRC progresses which would improve the discrimination as observed in the present study.
However, the overall performance of the multivariable analyses in discriminating individuals with HRA from individuals with CRC in the present study, although the performance is promising, still needs improvements to be valuable as a screening test in daily routines.
The assessment of identifying individuals at risk of recurrence of colorectal adenoma based on plasma levels of the eight proteins at the time of diagnosis of the primary adenoma did not reach significance for any of the proteins. HRs for all eight proteins of this study are shown in Figure 2. The power of this analysis was sufficient to detect any clinically relevant differences, suggesting that there is no association between the plasma levels of the eight proteins and the probability of recurrence of colorectal adenoma. Similar evaluation of plasma levels of the specific proteins as predictors of adenoma recurrence has not yet been published.
The plasma proteins investigated in this study are not specific for colorectal adenoma or CRC, and altered expression in the circulation of the eight proteins can be observed in various biological processes including inflammation or carcinogenesis in other organs. The differential panel of the plasma proteins discriminating HRA from CRC and HRA from the remaining subjects suggests the possibility of developing a reflex algorithm, improving the identification of these outcomes. There is however a need for better and more specific biomarkers, in particular, for the separation of HRA and LRA.
Conclusion
In conclusion, combination of plasma levels of two (CEA and hs-CRP) of the eight proteins investigated in the present study shows a promising potential in detection of primary HRA (discrimination of individuals with HRA versus healthy individuals plus individuals with LRA). However, to be recommended and implemented as a screening test for HRA in daily routines, the achieved results by combination of biomarkers need improvements. Similarly, the detection of colorectal adenomas in general demonstrated potential, but the results are too limited to be implemented in clinical practice. Finally, plasma levels of the eight proteins could not significantly predict recurrence of colorectal adenomas.
Future perspectives to improve performance in detection colorectal adenomas, specifically HRA, may include combination of the protein panel of the present study with other experimental or established biomarkers (proteins or genetic/epigenetic markers) which may result in a high-performing model for a blood-based screening test.
Acknowledgments
The research nurses, secretaries and technicians at the participating hospital departments and laboratories are thanked for their skillful work.
Abbott Laboratories Inc. (Abbott Park, Il, USA) is thanked for the protein analyses at their Center of Excellence (VUMC, Amsterdam, The Netherlands).
Funding Statement
The study received financial support from The Andersen Isted Fund, The Augustinus Foundation, The Beckett Fund, The Inger Bonnén Fund, The Hans & Nora Buchard Fund, CEO Jens Bærentsen (private donation), The Walter Christensen Family Fund, The P.M. Christiansen Family Fund, The Aase & Ejnar Danielsen Fund, The Erichsen Family Fund, The Knud & Edith Eriksen Fund, The Svend Espersen Fund, The Elna and Jørgen Fagerholt Fund, The Sofus Carl Emil Friis Fund, The Torben & Alice Frimodt Fund, The Eva & Henry Frænkel Fund, The Gangsted Fund, The Thora & Viggo Grove Fund, The H Foundation, The Erna Hamilton Fund, The Sven & Ina Hansen Fund, The Søren & Helene Hempel Fund, The Henrik Henriksen Fund, The Jørgen Holm Family Fund, Foundation Jochum, The KID Fund, The Kornerup Fund, The Linex Fund, The Dagmar Marshall Fund, The “Midtjyske Bladfund”, The Axel Muusfeldt Fund, The Børge Nielsen Family Fund, The Michael Hermann Nielsen Fund, The Arvid Nilsson Fund, The Obel Family Fund, The Krista & Viggo Petersen Fund, The Willy & Ingeborg Reinhard Fund, The Kathrine & Vigo Skovgaard Fund, The Toyota Fund, The Vissing Fund, The Wedell-Wedellsborg Fund and Hvidovre University Hospital (The Capital Region of Denmark).
Abbreviations
AFP, alpha-feto protein; AUC, area under the ROC curve; CA19-9, cancer antigen 19-9; CEA, carcino embryogenic antigen; CRC, colorectal cancer; CyFra21-1, cytokeratin Fragment 21-1; ELISA, enzyme-linked immunosorbent assay; HR, hazard ratio; HRA, high-risk adenoma; Hs-CRP, high sensitivity C-reactive protein; LRA, low-risk adenoma; OR, odds ratio; ROC, receiver operating characteristic; TIMP-1, tissue inhibitor of metalloproteinases-1.
Ethics Approval and Informed Consent
The Ethics Committee of the Capital Region of Denmark (H‐3‐2009‐110) and the Danish Data Protection Agency (2007‐58‐0015) approved the study, which was performed according to the Declaration of Helsinki II. Informed consent was signed by all participating individuals on the day of the colonoscopy.
Data Sharing Statement
All data can be presented by contacting corresponding author.
Disclosure
Hans Jørgen Nielsen reports grants and non-financial support from Abbott Laboratories Inc., Chicago, USA, outside the submitted work. The authors report no other potential conflicts of interest for this work.
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi: 10.3322/caac.21262 [DOI] [PubMed] [Google Scholar]
- 2.Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66(4):683–691. doi: 10.1136/gutjnl-2015-310912 [DOI] [PubMed] [Google Scholar]
- 3.Wolf A, Fontham ETH, Church TR, et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J Clin. 2018;68(4):250–281. doi: 10.3322/caac.21457 [DOI] [PubMed] [Google Scholar]
- 4.Schreuders EH, Ruco A, Rabeneck L, et al. Colorectal cancer screening: a global overview of existing programmes. Gut. 2015;64(10):1637–1649. doi: 10.1136/gutjnl-2014-309086 [DOI] [PubMed] [Google Scholar]
- 5.Shaukat A, Mongin SJ, Geisser MS, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013;369(12):1106–1114. doi: 10.1056/NEJMoa1300720 [DOI] [PubMed] [Google Scholar]
- 6.Morson BC. The evolution of colorectal carcinoma. Clin Radiol. 1984;35(6):425–431. doi: 10.1016/S0009-9260(84)80033-1 [DOI] [PubMed] [Google Scholar]
- 7.Lieberman DA, Weiss DG, Bond JH, et al. Use of colonoscopy to screen asymptomatic adults for colorectal cancer. N Engl J Med. 2000;343(3):162–168. doi: 10.1056/NEJM200007203430301 [DOI] [PubMed] [Google Scholar]
- 8.Calderwood AH, Lasser KE, Roy HK. Colon adenoma features and their impact on risk of future advanced adenomas and colorectal cancer. World J Gastrointest Oncol. 2016;8(12):826–834. doi: 10.4251/wjgo.v8.i12.826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hisabe T, Hirai F, Matsui T. Development and progression of colorectal cancer based on follow-up analysis. Dig Endosc. 2014;26:73–77. doi: 10.1111/den.12276 [DOI] [PubMed] [Google Scholar]
- 10.Edwards BK, Ward E, Kohler BA, et al. Annual report to the nation on the status of cancer, 1975-2006, featuring colorectal cancer trends and impact of interventions (risk factors, screening, and treatment) to reduce future rates. Cancer. 2010;116(3):544–573. doi: 10.1002/cncr.24760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Park DI, Ryu S, Kim Y-H, et al. Comparison of guaiac-based and quantitative immunochemical fecal occult blood testing in a population at average risk undergoing colorectal cancer screening. Am J Gastroenterol. 2010;105(9):2017–2025. doi: 10.1038/ajg.2010.179 [DOI] [PubMed] [Google Scholar]
- 12.Brenner H, Tao S. Superior diagnostic performance of faecal immunochemical tests for haemoglobin in a head-to-head comparison with guaiac based faecal occult blood test among 2235 participants of screening colonoscopy. Eur J Cancer. 2013;49(14):3049–3054. doi: 10.1016/j.ejca.2013.04.023 [DOI] [PubMed] [Google Scholar]
- 13.Ahlquist DA, Zou H, Domanico M, et al. Next-generation stool DNA test accurately detects colorectal cancer and large adenomas. Gastroenterology. 2012;142(2):248–e26. doi: 10.1053/j.gastro.2011.10.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. N Eng J Med. 2014;370(14):1287–1297. doi: 10.1056/NEJMoa1311194 [DOI] [PubMed] [Google Scholar]
- 15.Church TR, Wandell M, Lofton-Day C, et al. Prospective evaluation of methylated SEPT9 in plasma for detection of asymptomatic colorectal cancer. Gut. 2014;63(2):317–325. doi: 10.1136/gutjnl-2012-304149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jin P, Kang Q, Wang X, et al. Performance of a second-generation methylated SEPT9 test in detecting colorectal neoplasm. J Gastroenterol Hepatol. 2015;30(5):830–833. doi: 10.1111/jgh.12855 [DOI] [PubMed] [Google Scholar]
- 17.Ma MX, Bourke MJ. Sessile serrated adenomas: how to detect, characterize and resect. Gut Liver. 2017;11(6):747–760. doi: 10.5009/gnl16523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Loughrey MB, Shepherd NA. Problematic colorectal polyps: is it cancer and what do I need to do about it? Surg Pathol Clin. 2017;10(4):947–960. doi: 10.1016/j.path.2017.07.009 [DOI] [PubMed] [Google Scholar]
- 19.Parente F, Boemo C, Ardizzoia A, et al. Outcomes and cost evaluation of the first two rounds of a colorectal cancer screening program based on immunochemical fecal occult blood test in northern Italy. Endoscopy. 2013;45(01):27–34. doi: 10.1055/s-0032-1325800 [DOI] [PubMed] [Google Scholar]
- 20.Klabunde C, Blom J, Bulliard J-L, et al. Participation rates for organized colorectal cancer screening programmes: an international comparison. J Med Screen. 2015;22(3):119–126. doi: 10.1177/0969141315584694 [DOI] [PubMed] [Google Scholar]
- 21.van der Vlugt M, Grobbee EJ, Bossuyt PM, et al. Adherence to colorectal cancer screening: four rounds of faecal immunochemical test-based screening. Br J Cancer. 2017;116(1):44–49. doi: 10.1038/bjc.2016.399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Song -L-L, Li Y-M. Current noninvasive tests for colorectal cancer screening: an overview of colorectal cancer screening tests. World J Gastrointest Oncol. 2016;8(11):793–800. doi: 10.4251/wjgo.v8.i11.793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Erben V, Bhardwaj M, Schrotz-King P, Brenner H. Metabolomics biomarkers for detection of colorectal neoplasms: a systematic review. Cancers. 2018;10(8):246. doi: 10.3390/cancers10080246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nikolaou S, Qiu S, Fiorentino F, Rasheed S, Tekkis P, Kontovounisios C. Systematic review of blood diagnostic markers in colorectal cancer. Tech Coloproctology. 2018;22(7):481–498. doi: 10.1007/s10151-018-1820-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen H, Qian J, Werner S, Cuk K, Knebel P, Brenner H. Development and validation of a panel of five proteins as blood biomarkers for early detection of colorectal cancer. Clin Epidemiol. 2017;9:517–526. doi: 10.2147/CLEP.S144171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Werner S, Krause F, Rolny V, et al. Evaluation of a 5-marker blood test for colorectal cancer early detection in a colorectal cancer screening setting. Clin Cancer Res. 2016;22(7):1725–1733. doi: 10.1158/1078-0432.CCR-15-1268 [DOI] [PubMed] [Google Scholar]
- 27.Wild N, Andres H, Rollinger W, et al. A combination of serum markers for the early detection of colorectal cancer. Clin Cancer Res. 2010;16(24):6111–6121. doi: 10.1158/1078-0432.CCR-10-0119 [DOI] [PubMed] [Google Scholar]
- 28.Semaan A, van Ellen A, Meller S, et al. SEPT9 and SHOX2 DNA methylation status and its utility in the diagnosis of colonic adenomas and colorectal adenocarcinomas. Clin Epigenetics. 2016;8:100. doi: 10.1186/s13148-016-0267-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Uratani R, Toiyama Y, Kitajima T, et al. Diagnostic potential of cell-free and exosomal MicroRNAs in the identification of patients with high-risk colorectal adenomas. PLoS One. 2016;11(10):10. doi: 10.1371/journal.pone.0160722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang Z-H, Ren -L-L, Zheng P, et al. miR-194 as a predictor for adenoma recurrence in patients with advanced colorectal adenoma after polypectomy. Cancer Prev Res. 2014;7(6):607–616. doi: 10.1158/1940-6207.CAPR-13-0426 [DOI] [PubMed] [Google Scholar]
- 31.Wilhelmsen M, Christensen IJ, Rasmussen L, et al. Detection of colorectal neoplasia: combination of eight blood-based, cancer-associated protein biomarkers. Int J Cancer. 2017;140(6):1436–1446. doi: 10.1002/ijc.30558 [DOI] [PubMed] [Google Scholar]
- 32.Feng Y, Li Y, Dai W, Mo S, Li Q, Cai S. Clinicopathologic features and prognostic factors in alpha-fetoprotein-producing colorectal cancer: analysis of 78 cases. Cell Physiol Biochem. 2018;51(5):2052–2064. doi: 10.1159/000495824 [DOI] [PubMed] [Google Scholar]
- 33.Wang YR, Yan JX, Wang LN. The diagnostic value of serum carcino-embryonic antigen, alpha fetoprotein and carbohydrate antigen 19-9 for colorectal cancer. J Can Res Ther. 2014;10(8):307–309. doi: 10.4103/0973-1482.151538 [DOI] [PubMed] [Google Scholar]
- 34.Kim NH, Lee MY, Park JH, et al. Serum CEA and CA 19-9 levels are associated with the presence and severity of colorectal neoplasia. Yonsei Med J. 2017;58(5):918–924. doi: 10.3349/ymj.2017.58.5.918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Thomsen M, Skovlund E, Sorbye H, et al. Prognostic role of carcinoembryonic antigen and carbohydrate antigen 19-9 in metastatic colorectal cancer: a BRAF-mutant subset with high CA 19-9 level and poor outcome. Br J Cancer. 2018;118(12):1609–1616. doi: 10.1038/s41416-018-0115-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Giessen-jung C, Nagel D, Glas M, et al. Preoperative serum markers for individual patient prognosis in stage I-III colon cancer. Tumor Biol. 2015;36(10):7897–7906. doi: 10.1007/s13277-015-3522-z [DOI] [PubMed] [Google Scholar]
- 37.Gao Y, Wang J, Zhou Y, Sheng S, Qian SY, Huo X. Evaluation of serum CEA, CA19-9, CA72-4, CA125 and ferritin as diagnostic markers and factors of clinical parameters for colorectal cancer. Sci Rep. 2018;8:245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Duffy M, Lamerz R, Haglund C, et al. Tumor markers in colorectal cancer, gastric cancer and gastrointestinal stromal cancers: European group on tumor markers 2014 guidelines update. Int J Cancer. 2014;134(11):2513–2522. doi: 10.1002/ijc.28384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lim DH, Lee JH, Kim JW. Feasibility of CYFRA 21-1 as a serum biomarker for the detection of colorectal adenoma and advanced colorectal adenoma in people over the age of 45. J Clin Lab Anal. 2018;32(1):e22163. doi: 10.1002/jcla.22163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lee JH. Clinical usefulness of serum CYFRA 21–1 in patients with colorectal cancer. Nucl Med Mol Imaging. 2013;47(3):181–187. doi: 10.1007/s13139-013-0218-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alkhateeb AA, Connor JR. The significance of ferritin in cancer: anti-oxidation, inflammation and tumorigenesis. Biochim Biophys Acta. 2013;1836(2):245–254. doi: 10.1016/j.bbcan.2013.07.002 [DOI] [PubMed] [Google Scholar]
- 42.Schneider C, Bodmer M, Jick SS, Meier CR. Colorectal cancer and markers of anemia. Eur J Cancer Prev. 2018;27(6):530–538. doi: 10.1097/CEJ.0000000000000397 [DOI] [PubMed] [Google Scholar]
- 43.Tao L, Jin L, Dechun L, Hongqiang Y, Changhua K, Guijun L. Galectin-3 expression in colorectal cancer and its correlation with clinical pathological characteristics and prognosis. Open Med. 2017;12(1):226–230. doi: 10.1515/med-2017-0032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Meng C, Yin X, Liu J, Tang K, Tang H, Liao J. TIMP-1 is a novel serum biomarker for the diagnosis of colorectal cancer: a meta-analysis. PLoS One. 2018;13(11):11. doi: 10.1371/journal.pone.0207039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nielsen HJ, Brünner N, Jorgensen LN, et al. Plasma TIMP-1 and CEA in detection of primary colorectal cancer: a prospective, population based study of 4509 high-risk individuals. Scand J Gastroenterol. 2011;46(1):60–69. doi: 10.3109/00365521.2010.513060 [DOI] [PubMed] [Google Scholar]
- 46.Christensen IJ, Brünner N, Dowell B, et al. Plasma TIMP-1 and CEA as markers for detection of primary colorectal cancer: a prospective validation study including symptomatic and non-symptomatic individuals. Anticancer Res. 2015;35(9):4935–4941. [PubMed] [Google Scholar]
- 47.Rasmussen L, Christensen IJ, Herzog M, Micallef J, Nielsen HJ. Circulating cell-free nucleosomes as biomarkers for early detection of colorectal cancer. Oncotarget. 2018;9(12):10247–10258. doi: 10.18632/oncotarget.21908 [DOI] [PMC free article] [PubMed] [Google Scholar]