Abstract
Translation control is crucial during virus–host interaction. On one hand, viruses completely rely on the protein synthesis machinery of host cells to propagate and have evolved various mechanisms to redirect the host's ribosomes toward their viral mRNAs. On the other hand, the host rewires its translation program in an attempt to contain and suppress the virus early on during infection; the antiviral program includes specific control on protein synthesis to translate several antiviral mRNAs involved in quenching the infection. As the infection progresses, host translation is in turn inhibited in order to limit viral propagation. We have learnt of very diverse strategies that both parties utilize to gain or retain control over the protein synthesis machinery. Yet novel strategies continue to be discovered, attesting for the importance of mRNA translation in virus–host interaction. This review focuses on recently described translation strategies employed by both hosts and viruses. These discoveries provide additional pieces in the understanding of the complex virus–host translation landscape.
This article is categorized under:
Translation > Translation Mechanisms
Translation > Translation Regulation
Keywords: N6‐methyladenosine, protein synthesis, tRNA fragment, tRNA pool, virus
Translation control of mammalian cells during viral infection can be impacted by tRNA fragments, N6‐methyladenosine (m6A), or modified tRNA pools.
1. INTRODUCTION
1.1. Basis of eukaryotic mRNA translation
A typical mammalian mRNA consists of a 7‐methylguanosine cap (m7G) at the 5′termini, a 5′ untranslated region (5′UTR), a coding sequence (CDS) that starts with a start codon and ends with an in‐frame stop codon, a 3′ untranslated region (3′UTR), and a poly(A) tail (Sonenberg & Hinnebusch, 2009). Viral mRNAs follow the same arrangement principles but with some particularities (described below and reviewed in more details in Jan, Mohr, & Walsh, 2016). Despite the distinctions, translation in mammalian cells for both host and virus mRNAs follows the same three fundamental steps: initiation, elongation, and termination. If translation occurs via the canonical cap‐dependent mechanism, initiation begins with the recruitment of the eIF4F complex, which includes the cap‐binding protein eIF4E, the scaffold protein eIF4G, the RNA helicase eIF4A, to the 5′end of the mRNA via the eIF4E–m7G cap interaction. The eIF4F complex recruits the 43S preinitiation complex, consisting of eIF2‐GTP.Met‐tRNAi.40S ribosomal subunit, to the 5′UTR. If the mRNA is translated via cap‐independent mechanisms, other translation factors can come into play to recruit the ribosome to the mRNA, often involving internal ribosome entry sites (IRES) (Mailliot & Martin, 2018). Either following cap‐dependent or ‐independent initiation, the subsequent steps follow the same principles. The 43S preinitiation complex begins to scan the 5′UTR with the help of eIF4A ATP‐dependent helicase activity to resolve secondary structure. Scanning stops when the 43S complex recognizes a start codon via anticodon‐base pairing with Met‐tRNAi. Although the cognate AUG is the dominant start codon of choice, approximately 60% of mammalian translation initiation occurs at noncognate start codons (which include CUG, GUG, or UUG) (Ingolia, Lareau, & Weissman, 2011), potentially using other initiator tRNAs than Met‐tRNAi (Kearse & Wilusz, 2017). The eIFs are then released and the 60S ribosomal subunit is recruited to the 40S ribosomal subunit, forming an elongation competent 80S monosome (Jackson, Hellen, & Pestova, 2010; Sonenberg & Hinnebusch, 2009). Elongation then begins with a continuous supply of aminoacyl‐tRNA to the A site of the 80S monosome by the eukaryotic elongation factor (eEF) 1A. Peptide bond formation between amino acids in the P site and the A site is catalyzed, while the translocase eEF2 “rachet” the ribosome by one codon in the 3′ direction. The uncharged tRNA is then moved to the E site while the peptidyl‐tRNA transfers to the P site, leaving the A site unoccupied for the next cycle of elongation. When the ribosome reaches a stop codon (UAG, UGA, or UAA), the eukaryotic release factors eRF1.eRF3.GTP is recruited to the A site instead of a charged tRNA to initiate termination. GTP hydrolysis catalyzed by eRF3 causes deacetylation of the peptidyl‐tRNA bond, releasing the polypeptide chain from the last tRNA followed by dissociation of the 80S ribosome (Dever & Green, 2012).
1.2. Translation reprogramming during virus–host interaction
Protein synthesis is a complex cellular process that consumes a large portion of the cell energy expenditure and involves a sophisticated signaling and effector gene network (Buttgereit & Brand, 1995; Jackson et al., 2010; Sonenberg & Hinnebusch, 2009). Unlike for replication and transcription where viruses can encode their own genes to carry out these functions, the synthesis of viral proteins completely relies on the host translation machinery and represents a major interaction interface that is crucial for virus propagation. Viruses deploy a surprisingly diverse set of strategies to redirect the translation machinery on viral RNAs and shutting down host translation. Simultaneously, the antiviral response depends on the same machinery for translating antiviral transcripts while various translation control mechanisms are also engaged by the host to limit viral infection (Bushell & Sarnow, 2002; Hoang, Graber, & Alain, 2018; Jan et al., 2016; Walsh, Mathews, & Mohr, 2013). In this section, we will attempt to briefly summarize the specific translation strategies employed by host cells and viruses during infection and provide the readers with a nonexhaustive, representative literature list for reference. For comprehensive discussions of established knowledge of translation control in virus–host interaction, we invite the readers to peruse previous excellent reviews by Jan et al. (2016) or Walsh et al. (2013).
A number of signaling pathways impact upon translation control during viral infection. For instance, the PI3K‐AKT‐mTORC1 pathway regulates cellular metabolism and is one of the main signaling axes that impact mRNA translation, most notably via its principal downstream effectors S6Ks, 4E‐BPs, and LARP1 (Fonseca, Lahr, Damgaard, Alain, & Berman, 2018; Saxton & Sabatini, 2017). The activation of mTORC1 was reported during the antiviral state induced by type I interferon (Lekmine et al., 2003). Similarly, the early events of virus entry were shown to activate this pathway (Kang et al., 2017; Soares et al., 2009; Zhu et al., 2011). mTORC1 in turn was shown to play an important role in translation of antiviral transcripts, and of crucial antiviral transcription factors (Alain et al., 2010; Cao et al., 2008; Colina et al., 2008; Nehdi et al., 2014; Zakaria et al., 2018). Another translation arm of the antiviral response is the integrated stress response (ISR), a stress‐induced pathway that causes protein synthesis shutdown via phosphorylation of the eIF2α by four kinases: heme‐regulated inhibitor (HRI), general control non‐derepressible 2 (GCN2), protein kinase R (PKR), or PKR‐like endoplasmic reticulum (ER) kinase (PERK). The formation of the ternary complex (TC) requires eIF2‐GTP that is recycled after translation initiation. Phosphorylation of the α subunit of eIF2 (P‐eIF2α) inhibits the recycling step, thus blocking translation initiation by exhausting the cellular pool of active eIF2‐GTP. Stimuli from virus infection are known to activate ISR kinases: structured viral genomic RNA of Sindbis virus activates GCN2 (Berlanga et al., 2006), while the unfolded protein response during virus infection induces PERK (L. Zhang & Wang, 2012). Importantly, the kinase PKR is an interferon stimulated gene (ISG) that is upregulated by type I interferon and is a sensor of double‐stranded RNA or 5′triphosphate RNA, nucleic acid products that characterize all virus replication (Dauber & Wolff, 2009; Nallagatla et al., 2007).
Viruses, in turn, have the capacity to alter the translation environment of the antiviral state. The mTORC1 is a primary target of virus manipulation, inhibited in an attempt to thwart host translation or activated to benefit viral protein production. For example, the matrix protein M of VSV was shown to inhibit AKT phosphorylation (Connor & Lyles, 2002), which in turn dampen host protein synthesis including interferon gene expression (Ahmed et al., 2003). In contrast, various components of the PI3K‐AKT‐mTORC1 signaling have been reported to be activated by viral proteins to benefit virus replication (Chuluunbaatar et al., 2010; Couty, Geras‐Raaka, Weksler, & Gershengorn, 2001; Ehrhardt et al., 2007; Frese et al., 2003; Hirata et al., 2014; Lu et al., 2004; Moorman et al., 2008; Portis & Longnecker, 2004; Werden et al., 2009). The translation initiation complex, the crucial component of cap‐dependent translation initiation, is also targeted by viral proteins using numerous mechanisms, including protease degradation of translation initiation factors, inhibition of complex formation, and redirection of these factors toward viral mRNAs (Hoang et al., 2018). The ISR signaling pathway is no exception and is a frequent subject of virus hijacking to prevent ISR‐mediated translation shutdown. The various strategies range from deploying dsRNA‐binding protein to shield viral dsRNA from detection by PKR (Bierle, Semmens, & Geballe, 2013; Child, Hanson, Brown, Janzen, & Geballe, 2006; Hatada & Fukuda, 1992; Poppers, Mulvey, Perez, Khoo, & Mohr, 2003; Rabouw et al., 2016; Romano et al., 1998; Yue & Shatkin, 1997), expressing proteins that bind directly to ISR kinases to impair their function (Carroll, Elroy‐Stein, Moss, & Jagus, 1993; Gale, Korth, & Katze, 1998; Pavio, Romano, Graczyk, Feinstone, & Taylor, 2003; Van Opdenbosch, Van den Broeke, De Regge, Tabares, & Favoreel, 2012) and enhancing eIF2α dephosphorylation to resolve translation shutdown by P‐eIF2α (B. He, Gross, & Roizman, 1997; Kazemi et al., 2004; F. Zhang, Moon, Childs, Goodbourn, & Dixon, 2010). The activation of certain aspects of the ISR might also be beneficial for virus replication, such as in the case of SARS‐CoV (the strain that caused the 2003 Severe Acute Respiratory Syndrome global outbreak) activating the unfolded protein response via its 8ab protein. When expressing ectopically, 8ab promotes proteolysis of ATF6 to produce the active form that translocates to the nucleus and regulates the transcription program of UPR (Sung, Chao, Jeng, Yang, & Lai, 2009). Additionally, certain viruses can sustain translation in the suboptimal translation condition of the antiviral state via an IRES, structured RNA sequences that are capable of recruiting ribosome to mRNA with the requirement of only a minimal and incomplete set of eIF, mediating a mode of cap‐independent translation initiation (Mailliot & Martin, 2018).
Despite the wealth of knowledge that the field accumulated over the last few decades, the translation landscape of virus–host interaction still contains various uncharted territories that remain to be discovered. With the development of novel techniques to examine previously poorly understood cellular phenomena, emerging aspects of this crucial virus–host interaction have been revealed (Figure 1), many of them prove no less important in defining the fate of the infected cell. In the following sections, we attempt to review the current state of understanding of the emerging aspects of virus–host interactions.
FIGURE 1.
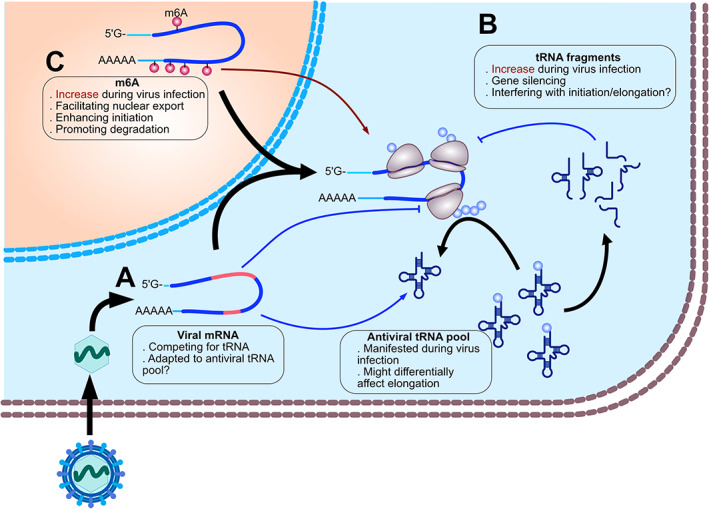
Emerging mechanisms of translation control during viral infection. (A) Transcription of viral mRNAs and alteration of the tRNA pools during the antiviral state impact translation. Viral mRNAs often possess dissimilar codon usage than that of host mRNAs, consequently altering the tRNA pools by their unique consumption of tRNAs. The antiviral state also changes the composition of the tRNA pools. Ultimately, such altered tRNA pools might affect the elongation rate of a defined subset of mRNAs. (B) tRNA fragments impair translation during viral infection. tRNA fragments abundance increases in infected cells. These fragments might in turn inhibit translation via their interaction with translating ribosomes or via the RNA interference machinery. (C) m6A is a layer of translation control in virus–host interaction. m6A machinery is activated during infection. m6A might enhance or disrupt translation depending on the effector proteins
2. EMERGING TRANSLATION STRATEGIES DURING VIRUS–HOST INTERACTION
2.1. Codon usage and the tRNA pool of the antiviral state
During translation elongation, the ribosome incorporates individual amino acid to the elongating peptide chain from the cytoplasmic pool of amino acyl‐tRNA. Although it was generally assumed that the elongation rate in an organism is constant, a ribosome in fact assimilates amino acid at varying rates depending on the codon, the type of amino acid, and the availability of the corresponding amino acyl‐tRNA. Suboptimal codon usage, depletion of certain amino acids, or certain codons can slow the elongation rate via ribosome stalling (Darnell, Subramaniam, & O'Shea, 2018; Gardin et al., 2014; Peil et al., 2013) (Figure 2). Consequently, translation efficiency is a selection pressure that shapes codon usage bias toward the availability of the organism's tRNA pools, such that the codon corresponding to the most abundant tRNAs will be used more frequently (Gingold & Pilpel, 2011) (Figure 1a). Recently, it has been revealed that the tRNA pool is not just unique for each species, but is also dynamically altered between different cellular states (Darnell et al., 2018; Gingold et al., 2014). Virus infection poses a unique challenge for the host tRNA pool: as the virus usurps the host translation machinery, the variety of translating mRNAs degenerates dramatically from tens of thousands of host mRNA species to only a few hundred of virus mRNAs. This environment theoretically imposes a selection pressure for viral genes to conform to the host tRNA pool and thus the host codon usage bias, especially between highly expressed viral genes and the most abundantly expressed tRNAs. Such selection pressure appears to be very strong in shaping bacteriophage codon usage (Carbone, 2008; Lucks, Nelson, Kudla, & Plotkin, 2008; Sharp, Rogers, & McConnell, 1985), while affecting that of avian and mammalian viruses to a lesser extent (Bahir, Fromer, Prat, & Linial, 2009). Intriguingly, it was reported that HIV and HSV genes employ suboptimal codon usage for human cells (Bradel‐Tretheway, Zhen, & Dewhurst, 2003; Ngumbela et al., 2008). Therefore, although it has been clear that viruses have adapted their codon usage to that of their hosts, dissimilarities remain, which raises the following question: are there specific tRNA pools that are manifested during the host's antiviral state, and conversely do specific viruses actively shape the tRNA pools to suit their replication while thwarting host translation?
FIGURE 2.
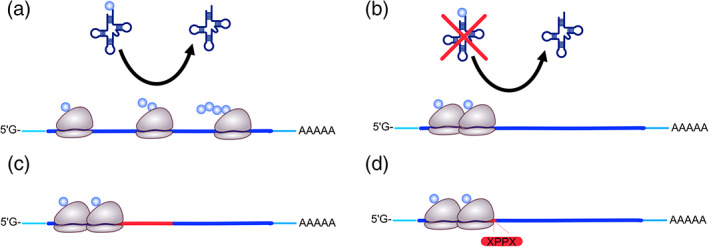
Various scenarios of stalled elongation. (a) Normal elongating ribosomes. (b) Ribosome stalling due to depletion of certain amino acids or tRNAs. (c) Suboptimal codon usage that requires rare tRNAs for decoding may also cause ribosome stalling. (d) Certain amino acid motifs, such as di‐proline (XPPX) depicted here, can also cause ribosome stalling
The altered tRNA pools in infected cells can be initiated by the activation of the antiviral state with interferon. The earliest evidence of antiviral state‐specific tRNA pools came from a study in 1976 that reported inhibition of protein synthesis in cell extracts from interferon‐treated cells, which was overcome by adding crude tRNA preparations (Sen et al., 1976). Additionally, by measuring the incorporation rate of individual amino acid to purified total endogenous tRNA, the authors revealed that interferon treatment impaired amino acylation of a subset of amino acid to tRNAs, most significantly that of leucine, lysine, and serine, proposing that tRNAs corresponding to these amino acids are inactivated. The antiviral state‐specific tRNA pool hypothesis was further corroborated by a study that tracked the codon adaptation of the influenza virus gene PB1 (Smith, Chen, Wilke, & Krug, 2018). Influenza A viruses can exchange gene segments between subtypes; these subtypes in turn are endemic in different host species. The gene fragment encoding the polymerase PB1 was introduced into the human H3N2 influenza viruses in 1968 and since has been proposed to be subjected to selection pressure to conform to human tRNA pools, such adaptation was tracked using PB1 sequence from H3N2 isolates collected over the years. Surprisingly, the author found that as the H3N2 strain continued to circulate seasonally, PB1 codon usage gradually diverged from that of human as evaluated by codon adaptation index (CAI). More astonishingly, H3N2 expressing PB1 gene with codon usage mimicking that of recent strain with low CAI to human replicated better in A549 cell treated with interferon, but not in untreated A549 cells, compared to H3N2 expressing an earlier version of PB1 with a higher CAI to human. To inquire whether interferon treatment induces a different tRNA pool in human cells, the authors performed high‐throughput sequencing of tRNA pools and found that expression of certain tRNAs decreased in response to interferon, and the decrease in PB1 CAI to human was inversely correlated with an increase in PB1 codon usage bias adapting to this specific tRNA pool. It is noteworthy to mention that attempts to optimize PB1 codon usage to the interferon‐specific tRNA pool resulted in interferon resistance but a decrease in overall fitness of the new virus, suggesting that other selection pressures are also at play for codon selection. The mechanisms of how interferon could induce changes in the tRNA pool remain an open question. Of the two main classes of interferon, type I and type II IFN, type I IFN has a more prominent antiviral property and activates various transcription programs, primarily via the JAK/STAT signaling pathway (Ivashkiv & Donlin, 2014; Platanias, 2005). These transcription programs upregulate the expression of hundreds to thousands of antiviral genes, termed ISGs (Schoggins & Rice, 2011). Some of these ISGs are RNAses (Isg20 (Espert et al., 2003), Slfn11 (Valdez et al., 2019)) or related to RNAse (Oas1/2/3 (Kristiansen, Gad, Eskildsen‐Larsen, Despres, & Hartmann, 2011)), of which genes belong to the Schlafen family were shown to be able to degrade tRNAs (M. Li et al., 2018; J. Y. Yang et al., 2018). Thus, it is reasonable to speculate that degrading tRNA via the activation of tRNA‐specific RNAses by interferon is a plausible mechanism that shapes the tRNA pool of interferon‐treated cells.
Simultaneously with the presence of interferon, the tRNA pool of infected cells can also be actively altered by the virus productive infection. Despite the restricted coding capacity owned by their relatively small genomes, some viruses have been reported to encode tRNA genes and this phenomenon has been reported extensively in bacteriophage and cyanophage (Albers & Czech, 2016). In the specific case of the chlorella virus CVK2, viral tRNAs have been reported to be amino‐acylated, strengthening the notion that they contribute to protein synthesis in infected host cells (Nishida, Kawasaki, Fujie, Usami, & Yamada, 1999). In the case of mammalian viruses, tRNA genes were previously reported in the genome of murine gamma herpes virus 68 (Virgin et al., 1997). Alternatively, virus infection can favor the usage of specific tRNAs for protein synthesis without affecting total tRNA pools (Pavon‐Eternod et al., 2013). Although the exact mechanism to select tRNA is not known, certain viruses such as vaccinia virus form virus factories, subcellular entities where viral proteins are translated (David et al., 2011). It is possible that these virus factories can recruit selective tRNAs to its active translation site, potentially skewing cellular tRNA pools in the process. However, very little is known about the role of viral tRNA genes or virus‐mediated modulation of host tRNA pools, and further investigation is required to obtain a clearer picture of their importance in virus–host interaction.
Despite emerging evidence presented above, we should be cautious in formulating the hypothesis that optimizing translation efficiency is the only direction of the virus codon evolution as suboptimal codon bias persist in many viral genes. Codon usage of a gene can also regulate other gene expression in trans, most likely via competing for aa‐tRNAs from the common pools. For instance, it has been reported that codon optimization of the late gene (adenovirus genes can be classified as early or late genes based on the temporal dynamic of their expression in the virus life cycle) encoding the fiber protein of adenovirus impaired expression of the more codon‐optimized hexon structural protein and ultimately hampered virus replication (Villanueva, Martí‐Solano, & Fillat, 2016). Similarly, another study by Chen et al. proposed that virus codon usage bias might differ from that of host to avoid delay in decoding time due to overconsumption and depletion of similar tRNAs (F. Chen et al., 2020). Using ribosome profiling data from various studies to calculate relative codon decoding time of ribosomes (Gardin et al., 2014), the authors found that translation of highly expressed viral genes delayed the decoding time, and such delay correlated with the rarity of host tRNA supply. The authors then ectopically expressed 37 synonymous‐codon altered versions of a reporter gene ranging from poorly to highly codon‐optimized under a strong expression promoter, together with another reporter gene expressed under a weak promoter. This aimed to recapitulate the effect of highly expressed viral genes (the highly transcribed reporter) of various codon adaptation level on host cells general translation (the poorly transcribed reporter). Interestingly, synonymous codon adaptation to host cells resulted in elevated expression of the highly transcribed reporter gene at the expense of the expression of the poorly transcribed reporter gene. Thus, this report supports the model in which high codon adaptation of a few very highly expressed mRNAs might be detrimental for translation of the rest of the mRNAs, probably via depletion of the commonly used tRNAs. Together, these reports propose another direction for adaptation of virus codon usage: the whole genome or certain genes might actively be selected to a suboptimal codon usage to avoid depletion of abundantly used tRNAs and subsequently be exposed to translation constraints. Interestingly, as viruses or host actively shutoff protein synthesis, it would be interesting to investigate if suboptimal codon usage can be a viral strategy toward the goal of overloading host translation machinery with suboptimally adapted mRNAs to quickly deplete rare tRNAs. Overall, the data so far paint a complex picture wherein viral genes are under selection pressure to optimize their codons toward a distinct antiviral‐state tRNA pool, yet at least a subset of viral genes might preserve a certain degree of dissimilarity in codon usage to avoid directly competing for tRNAs with other viral or host genes. Further understanding of this aspect of virus–host interaction might benefit transgene design for viral vector or engineering a better cancer‐specific oncolytic virus that suits a cancer‐optimized codon usage.
2.2. Small tRNA fragments: Stress response element exploited by viruses
Small noncoding RNAs are RNA fragments that are typically <50 nt and inferred to provide a layer of posttranscriptional control. Recently, a group of small noncoding RNA called small tRNA fragments was described, which is the products of cleaved tRNAs in stress condition in various organisms (H. Fu et al., 2009; S. R. Lee & Collins, 2005; Y. S. Lee, Shibata, Malhotra, & Dutta, 2009; Thompson, Lu, Green, & Parker, 2008; Yamasaki, Ivanov, Hu, & Anderson, 2009). Although the nomenclature has yet to be unified, these small tRNA fragments can be classified loosely into two classes depending on the cleavage position on the tRNA. The first class is called tRNA halves (tiRNA), which are 29–50 nt in length, the products of cleavage at the anticodon loop of the tRNA resulting in the 5′ tRNA fragment (5′tiRNA) and the 3′ tRNA fragment (3′tiRNA) (Thompson & Parker, 2009). In mammalian cells, the endonuclease angiogenin (ANG) was found to be responsible for tRNA cleaving to produce tiRNA under various stress condition (H. Fu et al., 2009; Yamasaki et al., 2009). The second class of small tRNA fragments are tRNA‐derived fragments (tRFs), shorter fragments with a length ranging from 14–30 nt. Unbiased short RNA sequencing has discovered this class of RNA fragments that mapped to tRNA loci (Y. S. Lee et al., 2009). Subsequent studies corroborate the existence of this novel small RNA class as a functional cellular biomolecule as evidenced by their importance in cell proliferation (Cole et al., 2009; Couvillion, Sachidanandam, & Collins, 2010; Z. Li et al., 2012). tRFs can be further divided into tRF‐5, tRF‐3, and tRF‐1. tRF‐5 contains the region from 5′end of tRNA to the D‐loop or the region between the D‐loop and the anticodon loop, with three specific size ranges of 14–16 nt, 22–24 nt, and 28–30 nt (Kumar, Anaya, Mudunuri, & Dutta, 2014; Y. S. Lee et al., 2009). tRF‐3 fragment contains the acceptor arms to the TψC loop and are between 18 and 22 nt in length (Y. S. Lee et al., 2009). tRF1 fragments are originated from the 3′end of the primary tRNA transcript, with their 5′end situated after the 3′end of the mature tRNA (Y. S. Lee et al., 2009).
The fact that small tRNA fragments are induced during stress suggests that this class of small RNAs might be involved in some aspects of the cellular ISR, which include alteration of protein synthesis. Indeed, it has been found that the partially cleaved product of tRNA with RNAse A can inhibit protein synthesis in yeast and plant (S. Zhang, Sun, & Kragler, 2009). Similarly, Yamasaki et al. showed that transfection of tiRNAs derived from angiogenin cleavage inhibits translation in mammalian cells (Yamasaki et al., 2009). These observations allow speculations for a role of small tRNA fragments in translation inhibition. Yet only a small fraction of the tRNA pools are cleaved, thus their levels do not seem to be sufficiently altered to affect the rate of translation elongation (Yamasaki et al., 2009). A subsequent study that analyzed the effect of both 5′ and 3′tiRNA on translation using luciferase translation reporter assay revealed that only a subset of tiRNA was capable of inhibiting translation of capped and uncapped mRNA, including 5′tiRNA from Ala‐tRNA(GGC), Gly‐tRNA(GCC), Gly‐tRNA(CCC), and Cys‐tRNA(GCA) as well as 3′tiRNA from Pro‐tRNA (Ivanov, Emara, Villen, Gygi, & Anderson, 2011). Among them, 5′tiRNA‐Ala was the most potent translation inhibitor and was further showed to displace eIF4G and eIF4A from capped and uncapped Renilla luciferase mRNA when added to an in vitro transcription assay in rabbit reticulocyte lysate. This study attributed the translation repression effect of tiRNA to the terminal oligoguanine (TOG) motif found at the 5′end of tRNAAla and tRNACys; mutating this motif ablates the translation inhibition capacity of 5′tiRNA‐Ala and 5′tiRNA‐Cys, while introducing this motif into 5′tiRNA‐Met renders this tiRNA the capacity to inhibit translation. The shorter tRFs were also shown to be capable of inhibiting translation, although in a mechanistically distinct manner than that of tiRNA. Various tRFs have been shown to associate with Argonaut proteins (Haussecker et al., 2010; Maute et al., 2013; Yeung et al., 2009) and were found to repress the expression of transcripts in a sequence‐specific manner (Maute et al., 2013). Thus, tRFs might silence gene expression using the RNAi machinery. Intriguingly, data from virus infection discussed in the following paragraph also argue that the same machinery can be employed by tiRNA to inhibit translation. A study of ribosome‐associated small RNA (RNome) in the archaeon Haloferax volcanii also observed that tRNA fragments can directly bind to ribosomes, which infer another potential mechanism where tRFs directly interfere with ribosome assembly or translation initiation/elongation (Gebetsberger, Zywicki, Künzi, & Polacek, 2012). Whether such an interaction with ribosomes occurs in mammalian cells, as well as the exact outcome of such an interaction on ribosome function and protein synthesis, are exciting questions open for future investigations.
As small tRNA fragments are induced during various stress conditions, their roles in the landscape of virus–host interaction are becoming clearer (Figure 1b). A number of studies have uncovered the involvement of tRNA fragments in virus infection and host response. Wang et al. reported that respiratory syncytial virus (RSV) infection significantly upregulated the level of 5′tiRNA in infected cells (Q. Wang et al., 2013). One of the highly induced 5′tiRNAs, 5′tiRNA‐GluCTC, can silence luciferase reporter expression that contains a reverse complementary sequence of this tiRNA in the 3′UTR. Importantly, treating the cells with an anti‐5′tiRNA‐GluCTC impaired RSV replication, suggesting that the induction of this tRNA fragment is beneficial for RSV infection. In a follow‐up study from the same group, Deng et al. used biotinylated synthetic 5′tiRNA‐GluCTC as a bait to search for enriched mRNAs that interact with this tRNA fragment. Combining with the list of genes with decreased abundance upon RSV infection, the author came up with potential targets of 5′tiRNA‐GluCTC (Deng et al., 2015). One of the candidate genes, APOER2, contains two short regions of 8 and 9 nt that are reverse‐complement with 5′tiRNA‐GluCTC. Importantly, while RSV infection was found to decrease APOER2 mRNAs abundance, treating infected cells with anti‐5′tiRNA‐GluCTC rescued this inhibition. Together, these two studies provide evidence that tRNA fragments are an integrated component of the virus–host interaction. While the tRNA fragments investigated in these studies seem to be proviral, as other cellular biomolecules such as miRNA and protein were found to be capable of both proviral and antiviral functions, it is likely that other tRNA fragments could also have antiviral function. Accordingly, other 5′tiRNA fragments that have been shown elsewhere to inhibit cap‐dependent translation via displacing of components of the eIF4F complex (Ivanov et al., 2011) were also found to be upregulated in RSV‐infected cells (Q. Wang et al., 2013). Thus, the induction of tRNA fragments by RSV or other viruses could also contribute to a global host‐shutoff of protein synthesis, and the implication of this for virus infection is certainly an important subject for further investigations.
As mounting evidence suggests that tRNA fragments can accumulate during viral infection, the mechanisms behind such induction remain currently unaddressed. Although ANG was shown to be the main nuclease that generates tRNA fragments (H. Fu et al., 2009; Yamasaki et al., 2009), ANG expression decreases during interferon treatment, a condition directly linked to virus infection (McNab et al., 2013; Mehraj et al., 2013; Rock et al., 2005), suggesting that other endonucleases might contribute to tRNA fragment genesis during virus infection. Aside from ANG, there are a subset of ISGs that function as RNAses and might produce tRNA fragments from tRNA cleavages. For example, the gene Slfn11 discussed above is an ISG (Valdez et al., 2019) that can also cleave type II tRNA (M. Li et al., 2018). The schlafen family was recently described to be a family of endonuclease that preferably targets tRNA, degrading from the 3′end and producing intact 5′ fragments. Thus, it is reasonable to speculate that the schlafen family, together with other undiscovered tRNA endonucleases, may contribute to the generation of tRNA fragments during specific virus infection (J. Y. Yang et al., 2018). This might also be another mechanism of schlafen‐mediated translation inhibition on top of the reported mechanism of tRNA depletion.
2.3. m6A methylation: An important epi‐transcriptomic modification
Methylation of RNA was first described in 1974 to be a common posttranscriptional modification for ribonucleotide (Desrosiers, Friderici, & Rottman, 1974; Wei, Gershowitz, & Moss, 1975). Methylation was reported to occur on 0.2% of all total nucleotide in poly(A) RNAs, of which methylation of adenosine (m6A) accounts for 50% of all methylation (Wei et al., 1975). Despite its relatively high occurrence, m6A only emerged recently as an important layer of genomic regulation in mammalian cells (C. He, 2010; Nilsen, 2014), corresponding with the development of techniques to survey genome‐wide m6A methylation (Dominissini et al., 2012; Meyer et al., 2012). Since then, this modification has been reported to be involved in various physiological aspects including stress responses (Zhou et al., 2015), stem cell reprogramming (T. Chen et al., 2015), circadian rhythm (Fustin et al., 2013), oocyte maturation (Ivanova et al., 2017), and cancer (Pan, Ma, Liu, Li, & Shu, 2018). Mechanistically, m6A was shown to affect splicing (Dominissini et al., 2012), cap‐dependent translation initiation (Meyer et al., 2015), miRNA processing (Alarcón, Lee, Goodarzi, Halberg, & Tavazoie, 2015), and mRNA stability (Xiao Wang et al., 2014).
The mechanisms that regulate m6A modification and function involve “writers” (m6A methyltransferases), “erasers” (m6A demethyltransferases), and “readers” (effectors that bind to m6A). Methylation of adenine on mRNA is not random, and it was reported that the motif RRACH (R = purine, H = A, C, or U) is the m6A consensus sequence, and methylation preferentially occurs on the CDS and 3′UTR, particularly concentrating near the stop codon region (Dominissini et al., 2012; Meyer et al., 2012). The methyltransferase complex METTL3‐METTL14, together with the accessory proteins WTAP, KIAA1429, RBM15, and ZC3H13 form a protein complex that adds a methyl group to adenosine residues of nuclear pre‐mRNAs (Guo, Tang, Li, Perrimon, & Yan, 2018; Knuckles et al., 2018; Kobayashi et al., 2018; Patil et al., 2016; Schwartz et al., 2014; Wen et al., 2018; Yao et al., 2018). Demethylation of m6A was reported to be carried out by FTO or ALKHB5 (G. Jia et al., 2011; G. Zheng et al., 2013). The function of m6A modification on target mRNAs depends on various “readers,” effector proteins that are recruited by m6A. The YT521‐B homology (YTH) family, its members include YTHDF1, YTHDF2, YTHDF3, YTHDC1, and YTHDC2, is the most well‐studied group of reader proteins that regulates the effect of m6A (Dominissini et al., 2012; Shi et al., 2017; Xiao Wang et al., 2014, 2015). Binding of some reader proteins enhances translation efficiency, such as the case for YTHDF1 and YTHDF3 (Shi et al., 2017; Xiao Wang et al., 2015). More specifically, YTHDF1 was reported to enhance ribosome loading on its target mRNAs via interaction with the translation initiation factors eIF3A and eIF3B (Xiao Wang et al., 2015). In contrast, binding of YTHDF2 to target mRNAs promotes degradation via relocalization to mRNA decay sites such as P‐bodies (Xiao Wang et al., 2014). In another case, nuclear export of target mRNAs was enhanced by YTHDC1 via facilitation of interaction with the splicing factor SRSF3 and nuclear export adaptor NX1 (Roundtree et al., 2017). It is likely that other readers will be discovered in the near future that would have a diverse mode of action on mRNA translation and stability.
m6A is an important process in virus–host interaction (Figure 1c). m6A modification on viral mRNAs was first reported over three decades ago for RNA viruses (Kane & Beemon, 1985; Krug, Morgan, & Shatkin, 1976; Stoltzfus & Dimock, 1976; Thomason, Brian, Velicer, & Rottman, 1976) and more recently for DNA viruses (Hesser, Karijolich, Dominissini, He, & Glaunsinger, 2018; Tsai, Courtney, & Cullen, 2018; Ye, Chen, & Nilsen, 2017). Methylation of adenosine on viral mRNAs was shown to have complex effects on virus replication from positive to negative, and was recently reviewed elsewhere (Dang et al., 2019). Regarding the effect of m6A on translation of viral mRNAs, it was reported that mutation of one methylated adenosine residue on HIV‐1 RNA (A7883) results in impaired nuclear export, suggesting one mechanism by which m6A might affect viral mRNA translation (Lichinchi et al., 2016). However, considering that m6A has a wide range of effect on mRNA translation, it is highly possible that there are multiple mechanisms influencing viral protein synthesis that remain to be discovered. In addition to m6A modification to viral genes, translation control during host antiviral response can also be modulated via m6A. Indeed, increased N6 methylation might be a response to virus infection, as evidenced in HCMV and HIV‐1 infection (Lichinchi et al., 2016; Winkler et al., 2019). Interestingly, this upregulation has so far been shown to be negative for the antiviral response, as interferon‐β mRNA was found to be m6A‐methylated and this modification results in destabilization of this mRNA in an METTL3‐YTHDF2‐dependent manner (Winkler et al., 2019). Similarly, the RNA helicase DDX46 was reported to promote demethylation of MAVS, TRAF3, and TRAF6 mRNAs, which encode important signaling proteins for the antiviral response, and this demethylation inhibits their nuclear exports and impairs translation, ultimately decreasing interferon production (Q. Zheng, Hou, Zhou, Li, & Cao, 2017). In another study, VSV infection was shown to stimulate initially, then to decrease m6A modification in mouse peritoneal macrophage (Y. Liu et al., 2019). Demethylations are important for VSV replication, which is partially attributed to the activity of ALKBH5. This demethyltransferase enzyme was in turn shown to remove m6A modification on α‐ketoglutarate dehydrogenase (OGDH) mRNAs, an important enzyme of the itaconate metabolic pathway that is required for VSV replication, resulted in increased stability and expression of OGDH mRNAs. However, the current knowledge of m6A in antiviral response does not exclude the possibility that this modification may also serve as a positive contributor for translation of antiviral genes, possibly depending on the specific interaction of individual mRNAs with different readers, an area that should be subject of further investigation.
2.4. Other potential mechanisms of translation control during virus–host interaction
Aside from the above discussed mechanisms, separated reports indicate that there are other mechanisms at play in the competition for the translation machinery by both the host and the virus during infection. Although additional studies are required before robust models can be formulated, we feel that it is important to mention these intriguing observations in order to fully capture our current understanding of the translation landscape in virus–host interaction.
2.4.1. Spatial rearrangement of translation activity
mRNA translation can be regulated by spatial distribution of mRNAs (Martin & Ephrussi, 2009). Intriguingly, there is evidence of spatial redistribution of translation activity in infected cells, possibly due to active relocalization of translation factors including those involved in translation initiation and elongation. One line of evidence surrounds the formation of virus factories, active replication sites for viruses that are enriched in host membranes, ribosomes, and proteins, as well as viral nucleic acids and proteins (Netherton & Wileman, 2011). Using the ribopuromycylation method to visualize subcellular translation activity, it has been reported that the virus factory of vaccinia virus and reovirus possess elevated translation activity compared to the surrounding cytoplasm (David et al., 2011; Desmet, Anguish, & Parker, 2014). This advantage of increased translation was attributed to selective enrichment of tRNA‐amino acyl synthetases in the case of vaccinia virus (David et al., 2011) or translation initiation, elongation, termination factor, and ribosomal proteins in the case of reovirus (Desmet et al., 2014).
There are also subcellular locales that house nontranslating mRNAs, namely stress granules (SGs) for stalled mRNAs and processing bodies (PBs) for decaying mRNAs (Anderson & Kedersha, 2006). Of these granules, SG has been reported to be induced during virus infection, suggesting its involvement in virus–host interaction (Ruggieri et al., 2012; White, Cardenas, Marissen, & Lloyd, 2007). The formation of SG begins with stalled translation initiation on mRNA at the 5′UTR that causes ribosome run‐off and exposes the downstream region of the mRNA to various SG‐associated RNA‐binding proteins TIA1, TIAR, and G3BP1 that subsequently aggregate to form RNA granules (Gilks et al., 2004; Matsuki et al., 2013; Tourrière et al., 2003). Such a mechanism of aggregation based on nucleation on naked single‐stranded RNAs, together with the seclusion of various translation initiation factors in the stalled translation initiation complex, proposes that SG formation might inhibit translation and replication of virus when the cytoplasm is overloaded with abnormal amounts of viral RNA. Intriguingly, viruses seem to evolve mechanisms to suppress SG formation. Okonski and Samuel have shown that SG formation was induced by measles virus (MeV) infection in a PKR‐dependent manner (Okonski & Samuel, 2013). However, MeV with a C protein knockout was a more efficient inducer of SG formation than wild‐type MeV. It was determined that the intact C protein would lead to a decrease in PKR expression, which would subsequently allow for SG formation (Jiang, Qin, & Chen, 2016). Similar function has been attributed to the influenza NS1 protein and vaccinia virus E3L protein (Khaperskyy, Hatchette, & McCormick, 2012; Simpson‐Holley et al., 2011). The exact mechanism as well as the implication of SG suppression by viral proteins remains to be elucidated; nevertheless, this regulation might indicate a method to ensure that the host translation remains functional for the virus' replication.
2.4.2. Modification or exploitation of host translation machinery to favor virus translation
Mammalian mRNAs can contain cis‐elements that modulate their translation activity in specific conditions. One example is the 5′ terminal oligo pyrimidine (5′TOP) motif that is situated at the 5′end of mRNAs of genes encoding protein components of the translation machinery (Meyuhas & Kahan, 2015). TOP mRNAs are suppressed during stress conditions, largely regulated via the mTORC1 signaling pathway through its effector LARP1 (Fonseca et al., 2015; Thoreen et al., 2012). Intriguingly, mRNAs of certain viruses also contain cis‐elements that enhance their translations via interaction with host RNA‐binding proteins, in some case in an infected cells‐specific manner. One example of a viral mRNA cis‐element that enhances translation was reported in MeV on its nucleocapsid mRNA. It was observed that during a pathogenic MeV infection, the host cell undergoes global translation shutdown attributed to eIF2α phosphorylation (Inoue, Tsukiyama‐Kohara, Yoneda, Sato, & Kai, 2009; Sato et al., 2007). Intriguingly, the mRNA of the nucleocapsid coding gene of MeV possesses a short stem‐loop structure at its N‐5′ terminus residues 1–15 (Inoue et al., 2011) that recruit the La/SSB (La) autoantigen protein, a well‐described human translation factor (Intine, Tenenbaum, Sakulich, Keene, & Maraia, 2003). This in turn promotes the selective translation of N mRNA during a translational shutdown in MeV‐infected cells, which is crucial for MeV production since it has been previously shown that the accumulation of N protein within an infected cell is required for efficient viral genome replication (Lamb & Parks, 2013). It is noteworthy to mention that LARP1, the RNA‐binding protein that represses TOP mRNA translation, also belongs to the La protein family and similarly interacts with its target mRNAs via the 5′ terminal region of the 5′UTR, albeit conferring an opposite effect on translation (Fonseca et al., 2018). Considering that LARP1 was found to be a positive modulator of extracellular hepatitis C virus infection and dengue virus replication (Plissonnier et al., 2019; Suzuki et al., 2016), the role of the La protein family in virus translation might be an interesting subject for further investigation.
Another curious example of viral cis‐element is the poly(A) leader sequences that are found on mRNAs of vaccinia virus late genes. Vaccinia genes are often described as early or late depending on when they are transcribed during the viral life cycle. Early genes are turned on immediately following entry and include virulence factors that take over the host cell (Assarsson et al., 2008), transcription factors for late genes (Rosales, Sutter, & Moss, 1994), decapping enzymes (S.‐W. Liu, Wyatt, Orandle, Minai, & Moss, 2014), and genes with unknown function. Later in the viral life cycle, expression of late viral gene products is accompanied by their nontemplated 5′ polyadenylation (Z. Yang, Bruno, Martens, Porcella, & Moss, 2011). First reported three decades ago, these nontemplated poly(A) sequences are added to the 5′end of mRNAs of late vaccinia virus genes, likely due to slipping of vaccinia polymerase at a poly‐T motif in the promoter regions (Ahn & Moss, 1989; Ink & Pickup, 1990; Schwer, Visca, Vos, & Stunnenberg, 1987). These poly(A) leaders promote translation in vaccinia‐infected cells in a noncanonical cap‐dependent manner that require most of the translation initiation components, but without the essential eIF3 and eIF4F (Dhungel, Cao, & Yang, 2017; Shirokikh & Spirin, 2008). The virus‐specific enhancement is attributed to the activity of vaccinia virus kinase B1 that phosphorylates the host small ribosomal protein RACK1, which enhances ribosome recruitment to poly(A) leader mRNAs (Jha et al., 2017).
In addition to modifying translation of viral mRNAs, the untranslated regions might also contribute to regulation of host mRNA expression in the antiviral state. It is well known that the 5′ and 3′ untranslated regions of mRNAs are rich in cis‐elements that regulate their translation (Hinnebusch, Ivanov, & Sonenberg, 2016; Mayr, 2017). Recent studies reported that these untranslated regions are dynamically regulated in various contexts via alternative transcription start site (for 5′UTR) or alternative polyadenylation and termination (for 3′UTR) that consequently modulate gene expression via altered translation efficiency (Cheng et al., 2018; Floor & Doudna, 2016; Mayr & Bartel, 2009; Xi Wang, Hou, Quedenau, & Chen, 2016). Curiously, VSV infection has also been reported to cause widespread shortening of host mRNA 3′UTR via alternative polyadenylation (X. Jia et al., 2017). The shortening events occurred more frequently in genes functioning in antiviral and interferon signaling suggest that such 3′UTR shortening is an integrated antiviral response, potentially to enhance the translation efficiency of the antiviral mRNAs via removal of miRNA‐binding site (Y. Fu et al., 2018; Mayr & Bartel, 2009). Our group has also reported that virus infection causes an alternative 5′UTR event that enhances the translation of the ciliopathy gene INPP5E, the expression of which inhibits HSV and VSV infection (Hoang et al., 2019). The canonical 5′UTR of INPP5E mRNA contains multiple uORFs, which strongly inhibits translation of its CDS. Virus infection induces the expression of an alternative, shorter 5′UTR that lacks an intron containing three of the repressive uORFs, thus de‐repress the translation of INPP5E in infected conditions. Curiously, the SARS‐CoV‐2 receptor ACE2 is also an ISG (Ziegler et al., 2020) and gene information from NCBI indicates that ACE2 is translated from two transcript variants NM_001371415.1 and NM_021804.3; both possess the same CDS that encode the same protein but the latter harbors a longer 5′UTR with an intron. Thus, it will be intriguing to investigate if the antiviral state alters the transcript ratio of ACE2 or the splicing of the long 5′UTR. Altogether, these data suggest that the global transcript variants are regulated by the antiviral state, which then affects the translational output of antiviral genes.
3. CONCLUSION AND PERSPECTIVE
The field of translation control has accumulated extensive insight into the mechanisms of host cell‐ and virus‐mediated translation regulation during infection and the antiviral state, as well as the outcomes of these regulations on the antiviral response and virus propagation. Our understanding so far emphasizes on the translation initiation step with various strategies outlined in Section 1 of this review, corroborating the importance of initiation in translation control in mammalian cells. However, emerging evidence suggests that there is much to learn about this interface of virus–host interaction. With the extension of our collective capacity to probe into more aspects of translational control such as tRNA pools, posttranscriptional modification, and translation rate, novel layers of regulation are slowly revealed, providing a more complex picture of translational regulation where almost every component of the host protein synthesis machinery can be altered. The advance of the tRNA fragments field adds one previously poorly understood noncoding RNA species, besides miRNA, as a player in translation control, including during virus–host interaction. Much remains to be explored regarding the biogenesis, mechanism of action, and outcome of tRNA fragments in the cells; yet considering the abundance of these tRNA products, such endeavors guarantee interesting discoveries. Recent studies regarding the tRNA pools also suggest a divergence from the constant tRNA pools model and suggest that modulation of tRNA abundance might play a role in host defense, while some antiviral‐specific tRNA pools might play a role in shaping virus codon usage. Additionally, as tRNA fragments are generated from cleaved tRNAs, it is likely that the product of degradation of certain tRNAs also regulate translation, delivering a one‐two punch to the cell protein synthesis. Last but not least, the revealed effect of posttranscriptional mRNA modification on mRNA translation as well as its widespread occurrence on viral RNAs, of which m6A is the representative phenomenon, warrants further investigation.
As we advance our knowledge on this complex virus–host translation landscape, we are expanding our fundamental understanding of how one of the most ancient self‐replicating organic systems can coexist successfully in a parasitic relationship with the most sophisticated organisms, including us humans. No less important than the fundamental understanding of this interaction, such knowledge will benefit a substantial number of medical applications that are virus‐based, ranging from vaccine development, viral vectors to anticancer oncolytic viruses. For instance, in vaccine development, the method of recombinant viral vector vaccine relies on the expression of an antigen from a safe viral vector backbone, and is used to develop successful vaccines or other vaccines currently in clinical trials against various highly pathogenic viruses including SARS‐CoV‐2. As discussed in Section 2.1 of this review, the synonymous codon usage of the foreign antigen might need to be harmonized with the viral vector or a specific host tRNA pool to maximize the yield/efficacy depending on the specific application. Another example can be made in the engineering of anticancer oncolytic viruses where human transgenes are frequently incorporated into the virus genome to enhance immunotherapeutic efficacy (Seymour & Fisher, 2016). The expression of therapeutic transgene as well as the overall fitness of the engineered oncolytic viruses will be enhanced if various translation regulation aspects, including but not limited to proper codon optimization and incorporation of virus‐specific untranslated regions, are considered during the optimization process. Clearly these examples are just a minuscule representation of the vast number of potential improvements that translation control can contribute toward, and this will only continue to increase with the expansion of knowledge in this field.
CONFLICT OF INTEREST
The authors have declared no conflicts of interest for this article.
AUTHOR CONTRIBUTIONS
Huy‐Dung Hoang: Conceptualization; writing‐original draft; writing‐review and editing. Serge Neault: Writing‐original draft; writing‐review and editing. Adrian Pelin: Writing‐original draft; writing‐review and editing. Tommy Alain: Conceptualization; writing‐original draft; writing‐review and editing.
RELATED WIREs ARTICLES
Translation regulation gets its ‘omics’ moment
Novel viral translation strategies
ACKNOWLEDGMENTS
We apologize for works that are not discussed and cited in our review due to space constraint. This review is supported in part by funding from the Canadian Breast Cancer Foundation/Canadian Cancer Society, the Natural Sciences and Engineering Research Council of Canada, and the Terry Fox Research Institute to T. A.
Hoang H‐D, Neault S, Pelin A, Alain T. Emerging translation strategies during virus–host interaction. WIREs RNA. 2021;12:e1619. 10.1002/wrna.1619
Funding information Canadian Cancer Society Research Institute; Natural Sciences and Engineering Research Council of Canada, Grant/Award Number: RGPIW‐2016‐05228; Terry Fox Research Institute
REFERENCES
- Ahmed, M. , McKenzie, M. O. , Puckett, S. , Hojnacki, M. , Poliquin, L. , & Lyles, D. S. (2003). Ability of the matrix protein of vesicular stomatitis virus to suppress beta interferon gene expression is genetically correlated with the inhibition of host RNA and protein synthesis. Journal of Virology, 77(8), 4646–4657. 10.1128/jvi.77.8.4646-4657.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahn, B. Y. , & Moss, B. (1989). Capped poly(A) leaders of variable lengths at the 5′ ends of vaccinia virus late mRNAs. Journal of Virology, 63(1), 226–232. 10.1128/jvi.63.1.226-232.1989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alain, T. , Lun, X. Q. , Martineau, Y. , Sean, P. , Pulendran, B. , Petroulakis, E. , … Sonenberg, N. (2010). Vesicular stomatitis virus oncolysis is potentiated by impairing mTORC1‐dependent type I IFN production. Proceedings of the National Academy of Sciences of the United States of America, 107(4), 1576–1581. 10.1073/pnas.0912344107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alarcón, C. R. , Lee, H. , Goodarzi, H. , Halberg, N. , & Tavazoie, S. F. (2015). N6‐methyladenosine marks primary microRNAs for processing. Nature, 519(7544), 482–485. 10.1038/nature14281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albers, S. , & Czech, A. (2016). Exploiting tRNAs to boost virulence. Life, 6(1), 4. 10.3390/life6010004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson, P. , & Kedersha, N. (2006). RNA granules. Journal of Cell Biology, 172(6), 803–808. 10.1083/jcb.200512082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assarsson, E. , Greenbaum, J. A. , Sundström, M. , Schaffer, L. , Hammond, J. A. , Pasquetto, V. , … Sette, A. (2008). Kinetic analysis of a complete poxvirus transcriptome reveals an immediate‐early class of genes. Proceedings of the National Academy of Sciences of the United States of America, 105(6), 2140–2145. 10.1073/pnas.0711573105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bahir, I. , Fromer, M. , Prat, Y. , & Linial, M. (2009). Viral adaptation to host: A proteome‐based analysis of codon usage and amino acid preferences. Molecular Systems Biology, 5, 311. 10.1038/msb.2009.71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berlanga, J. J. , Ventoso, I. , Harding, H. P. , Deng, J. , Ron, D. , Sonenberg, N. , … de Haro, C. (2006). Antiviral effect of the mammalian translation initiation factor 2α kinase GCN2 against RNA viruses. The EMBO Journal, 25(8), 1730–1740. 10.1038/sj.emboj.7601073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bierle, C. J. , Semmens, K. M. , & Geballe, A. P. (2013). Double‐stranded RNA binding by the human cytomegalovirus PKR antagonist TRS1. Virology, 442(1), 28–37. 10.1016/j.virol.2013.03.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradel‐Tretheway, B. G. , Zhen, Z. , & Dewhurst, S. (2003). Effects of codon‐optimization on protein expression by the human herpesvirus 6 and 7 U51 open reading frame. Journal of Virological Methods, 111(2), 145–156. 10.1016/s0166-0934(03)00173-3 [DOI] [PubMed] [Google Scholar]
- Bushell, M. , & Sarnow, P. (2002). Hijacking the translation apparatus by RNA viruses. Journal of Cell Biology, 158(3), 395–399. 10.1083/jcb.200205044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buttgereit, F. , & Brand, M. D. (1995). A hierarchy of ATP‐consuming processes in mammalian cells. Biochemical Journal, 312(1), 163–167. 10.1042/bj3120163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao, W. , Manicassamy, S. , Tang, H. , Kasturi, S. P. , Pirani, A. , Murthy, N. , & Pulendran, B. (2008). Toll‐like receptor‐mediated induction of type I interferon in plasmacytoid dendritic cells requires the rapamycin‐sensitive PI(3)K‐mTOR‐p70S6K pathway. Nature Immunology, 9(10), 1157–1164. 10.1038/ni.1645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbone, A. (2008). Codon bias is a major factor explaining phage evolution in translationally biased hosts. Journal of Molecular Evolution, 66(3), 210–223. 10.1007/s00239-008-9068-6 [DOI] [PubMed] [Google Scholar]
- Carroll, K. , Elroy‐Stein, O. , Moss, B. , & Jagus, R. (1993). Recombinant vaccinia virus K3L gene product prevents activation of double‐stranded RNA‐dependent, initiation factor 2α‐specific protein kinase. Journal of Biological Chemistry, 268(17), 12837–12842. [PubMed] [Google Scholar]
- Chen, F. , Wu, P. , Deng, S. , Zhang, H. , Hou, Y. , Hu, Z. , … Yang, J.‐R. (2020). Dissimilation of synonymous codon usage bias in virus–host coevolution due to translational selection. Nature Ecology & Evolution, 4, 1–12. 10.1038/s41559-020-1124-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, T. , Hao, Y. J. , Zhang, Y. , Li, M. M. , Wang, M. , Han, W. , … Zhou, Q. (2015). M6A RNA methylation is regulated by microRNAs and promotes reprogramming to pluripotency. Cell Stem Cell, 16(3), 289–301. 10.1016/j.stem.2015.01.016 [DOI] [PubMed] [Google Scholar]
- Cheng, Z. , Otto, G. M. , Powers, E. N. , Carr, S. A. , Jovanovic, M. , & Brar, G. A. (2018). Pervasive, coordinated protein‐level changes driven by transcript isoform switching during meiosis. Cell, 172, 910–923. 10.1016/j.cell.2018.01.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Child, S. J. , Hanson, L. K. , Brown, C. E. , Janzen, D. M. , & Geballe, A. P. (2006). Double‐stranded RNA binding by a heterodimeric complex of murine cytomegalovirus m142 and m143 proteins. Journal of Virology, 80(20), 10173–10180. 10.1128/JVI.00905-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuluunbaatar, U. , Roller, R. , Feldman, M. E. , Brown, S. , Shokat, K. M. , & Mohr, I. (2010). Constitutive mTORC1 activation by a herpesvirus Akt surrogate stimulates mRNA translation and viral replication. Genes and Development, 24(23), 2627–2639. 10.1101/gad.1978310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole, C. , Sobala, A. , Lu, C. , Thatcher, S. R. , Bowman, A. , Brown, J. W. S. , … Hutvagner, G. (2009). Filtering of deep sequencing data reveals the existence of abundant Dicer‐dependent small RNAs derived from tRNAs. RNA, 15(12), 2147–2160. 10.1261/rna.1738409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colina, R. , Costa‐Mattioli, M. , Dowling, R. J. O. , Jaramillo, M. , Tai, L. H. , Breitbach, C. J. , … Sonenberg, N. (2008). Translational control of the innate immune response through IRF‐7. Nature, 452(7185), 323–328. 10.1038/nature06730 [DOI] [PubMed] [Google Scholar]
- Connor, J. H. , & Lyles, D. S. (2002). Vesicular stomatitis virus infection alters the eIF4F translation initiation complex and causes dephosphorylation of the eIF4E binding protein 4E‐BP1. Journal of Virology, 76(20), 10177–10187. 10.1128/jvi.76.20.10177-10187.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couty, J. P. , Geras‐Raaka, E. , Weksler, B. B. , & Gershengorn, M. C. (2001). Kaposi's sarcoma‐associated herpesvirus G protein‐coupled receptor signals through multiple pathways in endothelial cells. Journal of Biological Chemistry, 276(36), 33805–33811. 10.1074/jbc.M104631200 [DOI] [PubMed] [Google Scholar]
- Couvillion, M. T. , Sachidanandam, R. , & Collins, K. (2010). A growth‐essential Tetrahymena Piwi protein carries tRNA fragment cargo. Genes and Development, 24(24), 2742–2747. 10.1101/gad.1996210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang, W. , Xie, Y. , Cao, P. , Xin, S. , Wang, J. , Li, S. , … Lu, J. (2019). N6‐methyladenosine and viral infection. Frontiers in Microbiology, 10, 417. 10.3389/fmicb.2019.00417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darnell, A. M. , Subramaniam, A. R. , & O'Shea, E. K. (2018). Translational control through differential ribosome pausing during amino acid limitation in mammalian cells. Molecular Cell, 71, 229–243.e11. 10.1016/j.molcel.2018.06.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dauber, B. , & Wolff, T. (2009). Activation of the antiviral kinase PKR and viral countermeasures. Viruses, 1(3), 523–544. 10.3390/v1030523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- David, A. , Netzer, N. , Strader, M. B. , Das, S. R. , Chen, C. Y. , Gibbs, J. , … Yewdell, J. W. (2011). RNA binding targets aminoacyl‐tRNA synthetases to translating ribosomes. Journal of Biological Chemistry, 286(23), 20688–20700. 10.1074/jbc.M110.209452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng, J. , Ptashkin, R. N. , Chen, Y. , Cheng, Z. , Liu, G. , Phan, T. , … Bao, X. (2015). Respiratory syncytial virus utilizes a tRNA fragment to suppress antiviral responses through a novel targeting mechanism. Molecular Therapy, 23(10), 1622–1629. 10.1038/mt.2015.124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desmet, E. A. , Anguish, L. J. , & Parker, J. S. L. (2014). Virus‐mediated compartmentalization of the host translational machinery. MBio, 5(5), e01463‐14. 10.1128/mBio.01463-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desrosiers, R. , Friderici, K. , & Rottman, F. (1974). Identification of methylated nucleosides in messenger RNA from Novikoff hepatoma cells. Proceedings of the National Academy of Sciences of the United States of America, 71(10), 3971–3975. 10.1073/pnas.71.10.3971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dever, T. E. , & Green, R. (2012). The elongation, termination, and recycling phases of translation in eukaryotes. Cold Spring Harbor Perspectives in Biology, 4(7), 1–16. 10.1101/cshperspect.a013706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhungel, P. , Cao, S. , & Yang, Z. (2017). The 5′‐poly(A) leader of poxvirus mRNA confers a translational advantage that can be achieved in cells with impaired cap‐dependent translation. PLoS Pathogens, 13(8), e1006602. 10.1371/journal.ppat.1006602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dominissini, D. , Moshitch‐Moshkovitz, S. , Schwartz, S. , Salmon‐Divon, M. , Ungar, L. , Osenberg, S. , … Rechavi, G. (2012). Topology of the human and mouse m6A RNA methylomes revealed by m6A‐seq. Nature, 485(7397), 201–206. 10.1038/nature11112 [DOI] [PubMed] [Google Scholar]
- Ehrhardt, C. , Wolff, T. , Pleschka, S. , Planz, O. , Beermann, W. , Bode, J. G. , … Ludwig, S. (2007). Influenza A virus NS1 protein activates the PI3K/Akt pathway to mediate antiapoptotic signaling responses. Journal of Virology, 81(7), 3058–3067. 10.1128/jvi.02082-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espert, L. , Degols, G. , Gongora, C. , Blondel, D. , Williams, B. R. , Silverman, R. H. , & Mechti, N. (2003). ISG20, a new interferon‐induced RNase specific for single‐stranded RNA, defines an alternative antiviral pathway against RNA genomic viruses. Journal of Biological Chemistry, 278(18), 16151–16158. 10.1074/jbc.M209628200 [DOI] [PubMed] [Google Scholar]
- Floor, S. N. , & Doudna, J. A. (2016). Tunable protein synthesis by transcript isoforms in human cells. eLife, 5, e10921. 10.7554/eLife.10921 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fonseca, B. D. , Lahr, R. M. , Damgaard, C. K. , Alain, T. , & Berman, A. J. (2018). LARP1 on TOP of ribosome production. Wiley Interdisciplinary Reviews: RNA, 9(5), e1480. 10.1002/wrna.1480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fonseca, B. D. , Zakaria, C. , Jia, J. J. , Graber, T. E. , Svitkin, Y. , Tahmasebi, S. , … Damgaard, C. K. (2015). La‐related protein 1 (LARP1) represses terminal oligopyrimidine (TOP) mRNA translation downstream of mTOR complex 1 (mTORC1). Journal of Biological Chemistry, 290(26), 15996–16020. 10.1074/jbc.M114.621730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frese, K. K. , Lee, S. S. , Thomas, D. L. , Latorre, I. J. , Weiss, R. S. , Glaunsinger, B. A. , & Javier, R. T. (2003). Selective PDZ protein‐dependent stimulation of phosphatidylinositol 3‐kinase by the adenovirus E4‐ORF1 oncoprotein. Oncogene, 22(5), 710–721. 10.1038/sj.onc.1206151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu, H. , Feng, J. , Liu, Q. , Sun, F. , Tie, Y. , Zhu, J. , … Zheng, X. (2009). Stress induces tRNA cleavage by angiogenin in mammalian cells. FEBS Letters, 583(2), 437–442. 10.1016/j.febslet.2008.12.043 [DOI] [PubMed] [Google Scholar]
- Fu, Y. , Chen, L. , Chen, C. , Ge, Y. , Kang, M. , Song, Z. , … Xu, A. (2018). Crosstalk between alternative polyadenylation and miRNAs in the regulation of protein translational efficiency. Genome Research, 28(11), 1656–1663. 10.1101/gr.231506.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fustin, J. M. , Doi, M. , Yamaguchi, Y. , Hida, H. , Nishimura, S. , Yoshida, M. , … Okamura, H. (2013). XRNA‐methylation‐dependent RNA processing controls the speed of the circadian clock. Cell, 155(4), 793–806. 10.1016/j.cell.2013.10.026 [DOI] [PubMed] [Google Scholar]
- Gale, M. J. , Korth, M. J. , & Katze, M. G. (1998). Repression of the PKR protein kinase by the hepatitis C virus NS5A protein: A potential mechanism of interferon resistance. Clinical and Diagnostic Virology, 10(2–3), 157–162. 10.1016/S0928-0197(98)00034-8 [DOI] [PubMed] [Google Scholar]
- Gardin, J. , Yeasmin, R. , Yurovsky, A. , Cai, Y. , Skiena, S. , & Futcher, B. (2014). Measurement of average decoding rates of the 61 sense codons in vivo. eLife, 3, e03735. 10.7554/eLife.03735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebetsberger, J. , Zywicki, M. , Künzi, A. , & Polacek, N. (2012). tRNA‐derived fragments target the ribosome and function as regulatory non‐coding RNA in Haloferax volcanii . Archaea, 2012, 260909. 10.1155/2012/260909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilks, N. , Kedersha, N. , Ayodele, M. , Shen, L. , Stoecklin, G. , Dember, L. M. , & Anderson, P. (2004). Stress granule assembly is mediated by prion‐like aggregation of TIA‐1. Molecular Biology of the Cell, 15(12), 5383–5398. 10.1091/mbc.E04-08-0715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gingold, H. , & Pilpel, Y. (2011). Determinants of translation efficiency and accuracy. Molecular Systems Biology, 7(1), 481. 10.1038/msb.2011.14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gingold, H. , Tehler, D. , Christoffersen, N. R. , Nielsen, M. M. , Asmar, F. , Kooistra, S. M. , … Pilpel, Y. (2014). A dual program for translation regulation in cellular proliferation and differentiation. Cell, 158(6), 1281–1292. 10.1016/j.cell.2014.08.011 [DOI] [PubMed] [Google Scholar]
- Guo, J. , Tang, H. W. , Li, J. , Perrimon, N. , & Yan, D. (2018). Xio is a component of the Drosophila sex determination pathway and RNA N6‐methyladenosine methyltransferase complex. Proceedings of the National Academy of Sciences of the United States of America, 115(14), 3674–3679. 10.1073/pnas.1720945115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatada, E. , & Fukuda, R. (1992). Binding of influenza a virus NS1 protein to dsRNA in vitro. Journal of General Virology, 73(12), 3325–3329. 10.1099/0022-1317-73-12-3325 [DOI] [PubMed] [Google Scholar]
- Haussecker, D. , Huang, Y. , Lau, A. , Parameswaran, P. , Fire, A. Z. , & Kay, M. A. (2010). Human tRNA‐derived small RNAs in the global regulation of RNA silencing. RNA, 16(4), 673–695. 10.1261/rna.2000810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He, B. , Gross, M. , & Roizman, B. (1997). The γ134.5 protein of herpes simplex virus 1 complexes with protein phosphatase 1α to dephosphorylate the α subunit of the eukaryotic translation initiation factor 2 and preclude the shutoff of protein synthesis by double‐stranded RNA‐activated protein kinase. Proceedings of the National Academy of Sciences of the United States of America, 94(3), 843–848. 10.1073/pnas.94.3.843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He, C. (2010). Grand challenge commentary: RNA epigenetics? Nature Chemical Biology, 6(12), 863–865. 10.1038/nchembio.482 [DOI] [PubMed] [Google Scholar]
- Hesser, C. R. , Karijolich, J. , Dominissini, D. , He, C. , & Glaunsinger, B. A. (2018). N6‐methyladenosine modification and the YTHDF2 reader protein play cell type specific roles in lytic viral gene expression during Kaposi's sarcoma‐associated herpesvirus infection. PLoS Pathogens, 14(4), e1006995. 10.1371/journal.ppat.1006995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinnebusch, A. G. , Ivanov, I. P. , & Sonenberg, N. (2016). Translational control by 5′‐untranslated regions of eukaryotic mRNAs. Science, 352(6292), 1413–1416. 10.1126/science.aad9868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirata, N. , Suizu, F. , Matsuda‐Lennikov, M. , Edamura, T. , Bala, J. , & Noguchi, M. (2014). Inhibition of Akt kinase activity suppresses entry and replication of influenza virus. Biochemical and Biophysical Research Communications, 450(1), 891–898. 10.1016/j.bbrc.2014.06.077 [DOI] [PubMed] [Google Scholar]
- Hoang, H. D. , Graber, T. E. , & Alain, T. (2018). Battling for ribosomes: Translational control at the forefront of the antiviral response. Journal of Molecular Biology, 430(14), 1965–1992. 10.1016/j.jmb.2018.04.040 [DOI] [PubMed] [Google Scholar]
- Hoang, H. D. , Graber, T. E. , Jia, J. J. , Vaidya, N. , Gilchrist, V. H. , Xiang, X. , … Alain, T. (2019). Induction of an alternative mRNA 5′ leader enhances translation of the ciliopathy gene Inpp5e and resistance to oncolytic virus infection. Cell Reports, 29(12), 4010–4023.e5. 10.1016/j.celrep.2019.11.072 [DOI] [PubMed] [Google Scholar]
- Ingolia, N. T. , Lareau, L. F. , & Weissman, J. S. (2011). Ribosome profiling of mouse embryonic stem cells reveals the complexity and dynamics of mammalian proteomes. Cell, 147(4), 789–802. 10.1016/j.cell.2011.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ink, B. S. , & Pickup, D. J. (1990). Vaccinia virus directs the synthesis of early mRNAs containing 5′ poly(A) sequences. Proceedings of the National Academy of Sciences of the United States of America, 87(4), 1536–1540. 10.1073/pnas.87.4.1536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue, Y. , Sato, H. , Fujita, K. , Tsukiyama‐Kohara, K. , Yoneda, M. , & Kai, C. (2011). Selective translation of the measles virus nucleocapsid mRNA by La protein. Frontiers in Microbiology, 2, 173. 10.3389/fmicb.2011.00173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue, Y. , Tsukiyama‐Kohara, K. , Yoneda, M. , Sato, H. , & Kai, C. (2009). Inhibition of host protein synthesis in B95a cells infected with the HL strain of measles virus. Comparative Immunology, Microbiology and Infectious Diseases, 32(1), 29–41. 10.1016/j.cimid.2008.08.007 [DOI] [PubMed] [Google Scholar]
- Intine, R. V. , Tenenbaum, S. A. , Sakulich, A. L. , Keene, J. D. , & Maraia, R. J. (2003). Differential phosphorylation and subcellular localization of La RNPs associated with precursor tRNAs and translation‐related mRNAs. Molecular Cell, 12(5), 1301–1307. 10.1016/S1097-2765(03)00429-5 [DOI] [PubMed] [Google Scholar]
- Ivanov, P. , Emara, M. M. , Villen, J. , Gygi, S. P. , & Anderson, P. (2011). Angiogenin‐induced tRNA fragments inhibit translation initiation. Molecular Cell, 43(4), 613–623. 10.1016/j.molcel.2011.06.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanova, I. , Much, C. , Di Giacomo, M. , Azzi, C. , Morgan, M. , Moreira, P. N. , … O'Carroll, D. (2017). The RNA m6A reader YTHDF2 is essential for the post‐transcriptional regulation of the maternal transcriptome and oocyte competence. Molecular Cell, 67(6), 1059–1067.e4. 10.1016/j.molcel.2017.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivashkiv, L. B. , & Donlin, L. T. (2014). Regulation of type I interferon responses. Nature Reviews Immunology, 14(1), 36–49. 10.1038/nri3581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson, R. J. , Hellen, C. U. T. , & Pestova, T. V. (2010). The mechanism of eukaryotic translation initiation and principles of its regulation. Nature Reviews Molecular Cell Biology, 11(2), 113–127. 10.1038/nrm2838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jan, E. , Mohr, I. , & Walsh, D. (2016). A cap‐to‐tail guide to mRNA translation strategies in virus‐infected cells. Annual Review of Virology, 3(1), 283–307. 10.1146/annurev-virology-100114-055014 [DOI] [PubMed] [Google Scholar]
- Jha, S. , Rollins, M. G. , Fuchs, G. , Procter, D. J. , Hall, E. A. , Cozzolino, K. , … Walsh, D. (2017). Trans‐kingdom mimicry underlies ribosome customization by a poxvirus kinase. Nature, 546(7660), 651–655. 10.1038/nature22814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia, G. , Fu, Y. , Zhao, X. , Dai, Q. , Zheng, G. , Yang, Y. , … He, C. (2011). N6‐Methyladenosine in nuclear RNA is a major substrate of the obesity‐associated FTO. Nature Chemical Biology, 7(12), 885–887. 10.1038/nchembio.687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia, X. , Yuan, S. , Wang, Y. , Fu, Y. , Ge, Y. , Ge, Y. , … Xu, A. (2017). The role of alternative polyadenylation in the antiviral innate immune response. Nature Communications, 8(1), 1–12. 10.1038/ncomms14605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang, Y. , Qin, Y. , & Chen, M. (2016). Host‐pathogen interactions in measles virus replication and anti‐viral immunity. Viruses, 8(11), 308. 10.3390/v8110308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kane, S. E. , & Beemon, K. (1985). Precise localization of m6A in Rous sarcoma virus RNA reveals clustering of methylation sites: Implications for RNA processing. Molecular and Cellular Biology, 5(9), 2298–2306. 10.1128/mcb.5.9.2298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang, Y. , Yuan, R. , Zhao, X. , Xiang, B. , Gao, S. , Gao, P. , … Ren, T. (2017). Transient activation of the PI3K/Akt pathway promotes Newcastle disease virus replication and enhances anti‐apoptotic signaling responses. Oncotarget, 8(14), 23551–23563. 10.18632/oncotarget.15796 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Kazemi, S. , Papadopoulou, S. , Li, S. , Su, Q. , Wang, S. , Yoshimura, A. , … Koromilas, A. E. (2004). Control of subunit of eukaryotic translation initiation factor 2 (eIF2) phosphorylation by the human papillomavirus type 18 E6 oncoprotein: Implications for eIF2‐dependent gene expression and cell death. Molecular and Cellular Biology, 24(8), 3415–3429. 10.1128/mcb.24.8.3415-3429.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kearse, M. G. , & Wilusz, J. E. (2017). Non‐AUG translation: A new start for protein synthesis in eukaryotes. Genes and Development, 31(17), 1717–1731. 10.1101/gad.305250.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khaperskyy, D. A. , Hatchette, T. F. , & McCormick, C. (2012). Influenza A virus inhibits cytoplasmic stress granule formation. The FASEB Journal, 26(4), 1629–1639. 10.1096/fj.11-196915 [DOI] [PubMed] [Google Scholar]
- Knuckles, P. , Lence, T. , Haussmann, I. U. , Jacob, D. , Kreim, N. , Carl, S. H. , … Roignant, J. Y. (2018). Zc3h13/Flacc is required for adenosine methylation by bridging the mRNA‐binding factor RbM15/spenito to the m6 a machinery component Wtap/Fl(2)d. Genes and Development, 32(5–6), 415–429. 10.1101/gad.309146.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi, M. , Ohsugi, M. , Sasako, T. , Awazawa, M. , Umehara, T. , Iwane, A. , … Ueki, K. (2018). The RNA methyltransferase complex of WTAP, METTL3, and METTL14 regulates mitotic clonal expansion in adipogenesis. Molecular and Cellular Biology, 38(16), e00116‐18. 10.1128/MCB.00116-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kristiansen, H. , Gad, H. H. , Eskildsen‐Larsen, S. , Despres, P. , & Hartmann, R. (2011). The oligoadenylate synthetase family: An ancient protein family with multiple antiviral activities. Journal of Interferon and Cytokine Research, 31(1), 41–47. 10.1089/jir.2010.0107 [DOI] [PubMed] [Google Scholar]
- Krug, R. M. , Morgan, M. A. , & Shatkin, A. J. (1976). Influenza viral mRNA contains internal N6‐methyladenosine and 5′‐terminal 7‐methylguanosine in cap structures. Journal of Virology, 20(1), 45–53. 10.1128/jvi.20.1.45-53.1976 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar, P. , Anaya, J. , Mudunuri, S. B. , & Dutta, A. (2014). Meta‐analysis of tRNA derived RNA fragments reveals that they are evolutionarily conserved and associate with AGO proteins to recognize specific RNA targets. BMC Medicine, 12(1), 78. 10.1186/s12915-014-0078-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb, R. A. , & Parks, G. D. (2013). Paramyxoviridae: The viruses and their replication. Fields Virology: Sixth Edition, 1, (pp. 957–995). Philadelphia: Lippincott, Williams, and Wilkins. [Google Scholar]
- Lee, S. R. , & Collins, K. (2005). Starvation‐induced cleavage of the tRNA anticodon loop in Tetrahymena thermophila. Journal of Biological Chemistry, 280(52), 42744–42749. 10.1074/jbc.M510356200 [DOI] [PubMed] [Google Scholar]
- Lee, Y. S. , Shibata, Y. , Malhotra, A. , & Dutta, A. (2009). A novel class of small RNAs: tRNA‐derived RNA fragments (tRFs). Genes and Development, 23(22), 2639–2649. 10.1101/gad.1837609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lekmine, F. , Uddin, S. , Sassano, A. , Parmar, S. , Brachmann, S. M. , Majchrzak, B. , … Platanias, L. C. (2003). Activation of the p70 S6 kinase and phosphorylation of the 4E‐BP1 repressor of mRNA translation by type I interferons. Journal of Biological Chemistry, 278(30), 27772–27780. 10.1074/jbc.M301364200 [DOI] [PubMed] [Google Scholar]
- Li, M. , Kao, E. , Malone, D. , Gao, X. , Wang, J. Y. J. , & David, M. (2018). DNA damage‐induced cell death relies on SLFN11‐dependent cleavage of distinct type II tRNAs. Nature Structural and Molecular Biology, 25(11), 1047–1058. 10.1038/s41594-018-0142-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, Z. , Ender, C. , Meister, G. , Moore, P. S. , Chang, Y. , & John, B. (2012). Extensive terminal and asymmetric processing of small RNAs from rRNAs, snoRNAs, snRNAs, and tRNAs. Nucleic Acids Research, 40(14), 6787–6799. 10.1093/nar/gks307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lichinchi, G. , Gao, S. , Saletore, Y. , Gonzalez, G. M. , Bansal, V. , Wang, Y. , … Rana, T. M. (2016). Dynamics of the human and viral m(6)A RNA methylomes during HIV‐1 infection of T cells. Nature Microbiology, 1, 16011. 10.1038/nmicrobiol.2016.11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, S.‐W. , Wyatt, L. S. , Orandle, M. S. , Minai, M. , & Moss, B. (2014). The D10 decapping enzyme of vaccinia virus contributes to decay of cellular and viral mRNAs and to virulence in mice. Journal of Virology, 88(1), 202–211. 10.1128/JVI.02426-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, Y. , You, Y. , Lu, Z. , Yang, J. , Li, P. , Liu, L. , … Cao, X. (2019). N6‐methyladenosine RNA modification–mediated cellular metabolism rewiring inhibits viral replication. Science, 365(6458), 1171–1176. 10.1126/science.aax4468 [DOI] [PubMed] [Google Scholar]
- Lu, Z. , Hu, X. , Li, Y. , Zheng, L. , Zhou, Y. , Jiang, H. , … Ke, Y. (2004). Human papillomavirus 16 E6 oncoprotein interferences with insulin signaling pathway by binding to tuberin. Journal of Biological Chemistry, 279(34), 35664–35670. 10.1074/jbc.M403385200 [DOI] [PubMed] [Google Scholar]
- Lucks, J. B. , Nelson, D. R. , Kudla, G. R. , & Plotkin, J. B. (2008). Genome landscapes and bacteriophage codon usage. PLoS Computational Biology, 4(2), e1000001. 10.1371/journal.pcbi.1000001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mailliot, J. , & Martin, F. (2018). Viral internal ribosomal entry sites: Four classes for one goal. Wiley Interdisciplinary Reviews: RNA, 9(2), e1458. 10.1002/wrna.1458 [DOI] [PubMed] [Google Scholar]
- Martin, K. C. , & Ephrussi, A. (2009). mRNA localization: Gene expression in the spatial dimension. Cell, 136(4), 719–730. 10.1016/j.cell.2009.01.044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuki, H. , Takahashi, M. , Higuchi, M. , Makokha, G. N. , Oie, M. , & Fujii, M. (2013). Both G3BP1 and G3BP2 contribute to stress granule formation. Genes to Cells, 18(2), 135–146. 10.1111/gtc.12023 [DOI] [PubMed] [Google Scholar]
- Maute, R. L. , Schneider, C. , Sumazin, P. , Holmes, A. , Califano, A. , Basso, K. , & Dalla‐Favera, R. (2013). TRNA‐derived microRNA modulates proliferation and the DNA damage response and is down‐regulated in B cell lymphoma. Proceedings of the National Academy of Sciences of the United States of America, 110(4), 1404–1409. 10.1073/pnas.1206761110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayr, C. (2017). Regulation by 3′‐untranslated regions. Annual Review of Genetics, 51(1), 171–194. 10.1146/annurev-genet-120116-024704 [DOI] [PubMed] [Google Scholar]
- Mayr, C. , & Bartel, D. P. (2009). Widespread shortening of 3′UTRs by alternative cleavage and polyadenylation activates oncogenes in cancer cells. Cell, 138(4), 673–684. 10.1016/j.cell.2009.06.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNab, F. W. , Ewbank, J. , Rajsbaum, R. , Stavropoulos, E. , Martirosyan, A. , Redford, P. S. , … O'Garra, A. (2013). TPL‐2–ERK1/2 signaling promotes host resistance against intracellular bacterial infection by negative regulation of type I IFN production. The Journal of Immunology, 191(4), 1732–1743. 10.4049/jimmunol.1300146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehraj, V. , Textoris, J. , Ben Amara, A. , Ghigo, E. , Raoult, D. , Capo, C. , & Mege, J.‐L. (2013). Monocyte responses in the context of Q fever: From a static polarized model to a kinetic model of activation. The Journal of Infectious Diseases, 208(6), 942–951. 10.1093/infdis/jit266 [DOI] [PubMed] [Google Scholar]
- Meyer, K. D. , Patil, D. P. , Zhou, J. , Zinoviev, A. , Skabkin, M. A. , Elemento, O. , … Jaffrey, S. R. (2015). 5′ UTR m6A promotes cap‐independent translation. Cell, 163(4), 999–1010. 10.1016/j.cell.2015.10.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer, K. D. , Saletore, Y. , Zumbo, P. , Elemento, O. , Mason, C. E. , & Jaffrey, S. R. (2012). Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell, 149(7), 1635–1646. 10.1016/j.cell.2012.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyuhas, O. , & Kahan, T. (2015). The race to decipher the top secrets of TOP mRNAs. Biochimica et Biophysica Acta ‐ Gene Regulatory Mechanisms, 1849(7), 801–811. 10.1016/j.bbagrm.2014.08.015 [DOI] [PubMed] [Google Scholar]
- Moorman, N. J. , Cristea, I. M. , Terhune, S. S. , Rout, M. P. , Chait, B. T. , & Shenk, T. (2008). Human cytomegalovirus protein UL38 inhibits host cell stress responses by antagonizing the tuberous sclerosis protein complex. Cell Host and Microbe, 3(4), 253–262. 10.1016/j.chom.2008.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nallagatla, S. R. , Hwang, J. , Toroney, R. , Zheng, X. , Cameron, C. E. , & Bevilacqua, P. C. (2007). 5′‐Triphosphate‐dependent activation of PKR by RNAs with short stem‐loops. Science, 318(5855), 1455–1458. 10.1126/science.1147347 [DOI] [PubMed] [Google Scholar]
- Nehdi, A. , Sean, P. , Linares, I. , Colina, R. , Jaramillo, M. , & Alain, T. (2014). Deficiency in either 4E‐BP1 or 4E‐BP2 augments innate antiviral immune responses. PLoS One, 9(12), e114854. 10.1371/journal.pone.0114854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Netherton, C. L. , & Wileman, T. (2011). Virus factories, double membrane vesicles and viroplasm generated in animal cells. Current Opinion in Virology, 1(5), 381–387. 10.1016/j.coviro.2011.09.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ngumbela, K. C. , Ryan, K. P. , Sivamurthy, R. , Brockman, M. A. , Gandhi, R. T. , Bhardwaj, N. , & Kavanagh, D. G. (2008). Quantitative effect of suboptimal codon usage on translational efficiency of mRNA encoding HIV‐1 Gag in intact T cells. PLoS One, 3(6), e2356. 10.1371/journal.pone.0002356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsen, T. W. (2014). Internal mRNA methylation finally finds functions. Science, 343(6176), 1207–1208. 10.1126/science.1249340 [DOI] [PubMed] [Google Scholar]
- Nishida, K. , Kawasaki, T. , Fujie, M. , Usami, S. , & Yamada, T. (1999). Aminoacylation of tRNAs encoded by Chlorella virus CVK2. Virology, 263(1), 220–229. 10.1006/viro.1999.9949 [DOI] [PubMed] [Google Scholar]
- Okonski, K. M. , & Samuel, C. E. (2013). Stress granule formation induced by measles virus is protein kinase PKR dependent and impaired by RNA adenosine deaminase ADAR1. Journal of Virology, 87(2), 756–766. 10.1128/jvi.02270-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan, Y. , Ma, P. , Liu, Y. , Li, W. , & Shu, Y. (2018). Multiple functions of m6A RNA methylation in cancer. Journal of Hematology and Oncology, 11(1), 48. 10.1186/s13045-018-0590-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patil, D. P. , Chen, C. K. , Pickering, B. F. , Chow, A. , Jackson, C. , Guttman, M. , & Jaffrey, S. R. (2016). M6 A RNA methylation promotes XIST‐mediated transcriptional repression. Nature, 537(7620), 369–373. 10.1038/nature19342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavio, N. , Romano, P. R. , Graczyk, T. M. , Feinstone, S. M. , & Taylor, D. R. (2003). Protein synthesis and endoplasmic reticulum stress can be modulated by the hepatitis C virus envelope protein E2 through the eukaryotic initiation factor 2 kinase PERK. Journal of Virology, 77(6), 3578–3585. 10.1128/jvi.77.6.3578-3585.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavon‐Eternod, M. , David, A. , Dittmar, K. , Berglund, P. , Pan, T. , Bennink, J. R. , & Yewdell, J. W. (2013). Vaccinia and influenza A viruses select rather than adjust tRNAs to optimize translation. Nucleic Acids Research, 41(3), 1914–1921. 10.1093/nar/gks986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peil, L. , Starosta, A. L. , Lassak, J. , Atkinson, G. C. , Virumäe, K. , Spitzer, M. , … Wilson, D. N. (2013). Distinct XPPX sequence motifs induce ribosome stalling, which is rescued by the translation elongation factor EF‐P. Proceedings of the National Academy of Sciences of the United States of America, 110(38), 15265–15270. 10.1073/pnas.1310642110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platanias, L. C. (2005). Mechanisms of type‐I‐ and type‐II‐interferon‐mediated signalling. Nature Reviews Immunology, 5(5), 375–386. 10.1038/nri1604 [DOI] [PubMed] [Google Scholar]
- Plissonnier, M. L. , Cottarel, J. , Piver, E. , Kullolli, M. , Centonze, F. G. , Pitteri, S. , … Parent, R. (2019). LARP1 binding to hepatitis C virus particles is correlated with intracellular retention of viral infectivity. Virus Research, 271, 197679. 10.1016/j.virusres.2019.197679 [DOI] [PubMed] [Google Scholar]
- Poppers, J. , Mulvey, M. , Perez, C. , Khoo, D. , & Mohr, I. (2003). Identification of a lytic‐cycle Epstein‐Barr virus gene product that can regulate PKR activation. Journal of Virology, 77(1), 228–236. 10.1128/jvi.77.1.228-236.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Portis, T. , & Longnecker, R. (2004). Epstein‐Barr virus (EBV) LMP2A mediates B‐lymphocyte survival through constitutive activation of the Ras/PI3K/Akt pathway. Oncogene, 23(53), 8619–8628. 10.1038/sj.onc.1207905 [DOI] [PubMed] [Google Scholar]
- Rabouw, H. H. , Langereis, M. A. , Knaap, R. C. M. , Dalebout, T. J. , Canton, J. , Sola, I. , … van Kuppeveld, F. J. M. (2016). Middle East respiratory coronavirus accessory protein 4a inhibits PKR‐mediated antiviral stress responses. PLoS Pathogens, 12(10), e1005982. 10.1371/journal.ppat.1005982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rock, R. B. , Hu, S. , Deshpande, A. , Munir, S. , May, B. J. , Baker, C. A. , … Kapur, V. (2005). Transcriptional response of human microglial cells to interferon‐γ. Genes and Immunity, 6(8), 712–719. 10.1038/sj.gene.6364246 [DOI] [PubMed] [Google Scholar]
- Romano, P. R. , Zhang, F. , Tan, S.‐L. , Garcia‐Barrio, M. T. , Katze, M. G. , Dever, T. E. , & Hinnebusch, A. G. (1998). Inhibition of double‐stranded RNA‐dependent protein kinase PKR by vaccinia virus E3: Role of complex formation and the E3 N‐terminal domain. Molecular and Cellular Biology, 18(12), 7304–7316. 10.1128/mcb.18.12.7304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosales, R. , Sutter, G. , & Moss, B. (1994). A cellular factor is required for transcription of vaccinia viral intermediate‐stage genes. Proceedings of the National Academy of Sciences of the United States of America, 91(9), 3794–3798. 10.1073/pnas.91.9.3794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roundtree, I. A. , Luo, G. Z. , Zhang, Z. , Wang, X. , Zhou, T. , Cui, Y. , … He, C. (2017). YTHDC1 mediates nuclear export of N6‐methyladenosine methylated mRNAs. eLife, 6, e31311. 10.7554/eLife.31311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruggieri, A. , Dazert, E. , Metz, P. , Hofmann, S. , Bergeest, J. P. , Mazur, J. , … Bartenschlager, R. (2012). Dynamic oscillation of translation and stress granule formation mark the cellular response to virus infection. Cell Host and Microbe, 12(1), 71–85. 10.1016/j.chom.2012.05.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato, H. , Masuda, M. , Kanai, M. , Tsukiyama‐Kohara, K. , Yoneda, M. , & Kai, C. (2007). Measles virus N protein inhibits host translation by binding to eIF3‐p40. Journal of Virology, 81(21), 11569–11576. 10.1128/JVI.00570-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saxton, R. A. , & Sabatini, D. M. (2017). mTOR signaling in growth, metabolism, and disease. Cell, 168(6), 960–976. 10.1016/j.cell.2017.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoggins, J. W. , & Rice, C. M. (2011). Interferon‐stimulated genes and their antiviral effector functions. Current Opinion in Virology, 1(6), 519–525. 10.1016/j.coviro.2011.10.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz, S. , Mumbach, M. R. , Jovanovic, M. , Wang, T. , Maciag, K. , Bushkin, G. G. , … Regev, A. (2014). Perturbation of m6A writers reveals two distinct classes of mRNA methylation at internal and 5′ sites. Cell Reports, 8(1), 284–296. 10.1016/j.celrep.2014.05.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwer, B. , Visca, P. , Vos, J. C. , & Stunnenberg, H. G. (1987). Discontinuous transcription or RNA processing of vaccinia virus late messengers results in a 5′ poly(A) leader. Cell, 50(2), 163–169. 10.1016/0092-8674(87)90212-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen, G. C. , Gupta, S. L. , Brown, G. E. , Lebleu, B. , Rebello, M. A. , & Lengyel, P. (1976). Interferon treatment of Ehrlich ascites tumor cells: Effects on exogenous mRNA translation and tRNA inactivation in the cell extract. Journal of Virology, 17(1), 191–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seymour, L. W. , & Fisher, K. D. (2016). Oncolytic viruses: Finally delivering. British Journal of Cancer, 114(4), 357–361. 10.1038/bjc.2015.481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharp, P. M. , Rogers, M. S. , & McConnell, D. J. (1985). Selection pressures on codon usage in the complete genome of bacteriophage T7. Journal of Molecular Evolution, 21(2), 150–160. 10.1007/BF02100089 [DOI] [PubMed] [Google Scholar]
- Shi, H. , Wang, X. , Lu, Z. , Zhao, B. S. , Ma, H. , Hsu, P. J. , … He, C. (2017). YTHDF3 facilitates translation and decay of N 6‐methyladenosine‐modified RNA. Cell Research, 27(3), 315–328. 10.1038/cr.2017.15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirokikh, N. E. , & Spirin, A. S. (2008). Poly(A) leader of eukaryotic mRNA bypasses the dependence of translation on initiation factors. Proceedings of the National Academy of Sciences of the United States of America, 105(31), 10738–10743. 10.1073/pnas.0804940105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson‐Holley, M. , Kedersha, N. , Dower, K. , Rubins, K. H. , Anderson, P. , Hensley, L. E. , & Connor, J. H. (2011). Formation of antiviral cytoplasmic granules during orthopoxvirus infection. Journal of Virology, 85(4), 1581–1593. 10.1128/JVI.02247-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith, B. L. , Chen, G. , Wilke, C. O. , & Krug, R. M. (2018). Avian influenza virus PB1 gene in H3N2 viruses evolved in humans to reduce interferon inhibition by skewing codon usage toward interferon‐altered tRNA pools. MBio, 9(4), e01222‐18. 10.1128/mBio.01222-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soares, J. A. P. , Leite, F. G. G. , Andrade, L. G. , Torres, A. A. , De Sousa, L. P. , Barcelos, L. S. , … Bonjardim, C. A. (2009). Activation of the PI3K/Akt pathway early during vaccinia and cowpox virus infections is required for both host survival and viral replication. Journal of Virology, 83(13), 6883–6899. 10.1128/jvi.00245-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonenberg, N. , & Hinnebusch, A. G. (2009). Regulation of translation initiation in eukaryotes: Mechanisms and biological targets. Cell, 136(4), 731–745. 10.1016/j.cell.2009.01.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoltzfus, C. M. , & Dimock, K. (1976). Evidence of methylation of B77 avian sarcoma virus genome RNA subunits. Journal of Virology, 18(2), 586–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sung, S. C. , Chao, C. Y. , Jeng, K. S. , Yang, J. Y. , & Lai, M. M. C. (2009). The 8ab protein of SARS‐CoV is a luminal ER membrane‐associated protein and induces the activation of ATF6. Virology, 387(2), 402–413. 10.1016/j.virol.2009.02.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki, Y. , Chin, W.‐X. , Han, Q. , Ichiyama, K. , Lee, C. H. , Eyo, Z. W. , … Yamamoto, N. (2016). Characterization of RyDEN (C19orf66) as an interferon‐stimulated cellular inhibitor against dengue virus replication. PLoS Pathogens, 12(1), e1005357. 10.1371/journal.ppat.1005357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomason, A. R. , Brian, D. A. , Velicer, L. F. , & Rottman, F. M. (1976). Methylation of high‐molecular‐weight subunit RNA of feline leukemia virus. Journal of Virology, 20(1), 123–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson, D. M. , Lu, C. , Green, P. J. , & Parker, R. (2008). tRNA cleavage is a conserved response to oxidative stress in eukaryotes. RNA, 14(10), 2095–2103. 10.1261/rna.1232808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson, D. M. , & Parker, R. (2009). Stressing out over tRNA cleavage. Cell, 138(2), 215–219. 10.1016/j.cell.2009.07.001 [DOI] [PubMed] [Google Scholar]
- Thoreen, C. C. , Chantranupong, L. , Keys, H. R. , Wang, T. , Gray, N. S. , & Sabatini, D. M. (2012). A unifying model for mTORC1‐mediated regulation of mRNA translation. Nature, 485(7396), 109–113. 10.1038/nature11083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tourrière, H. , Chebli, K. , Zekri, L. , Courselaud, B. , Blanchard, J. M. , Bertrand, E. , & Tazi, J. (2003). The RasGAP‐associated endoribonuclease G3BP assembles stress granules. Journal of Cell Biology, 160(6), 823–831. 10.1083/jcb.200212128 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Tsai, K. , Courtney, D. G. , & Cullen, B. R. (2018). Addition of m6A to SV40 late mRNAs enhances viral structural gene expression and replication. PLoS Pathogens, 14(2), e1006919. 10.1371/journal.ppat.1006919 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdez, F. , Salvador, J. , Palermo, P. M. , Mohl, J. E. , Hanley, K. A. , Watts, D. , & Llano, M. (2019). Schlafen 11 restricts flavivirus replication. Journal of Virology, 93(15), e00104‐19. 10.1128/JVI.00104-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Opdenbosch, N. , Van den Broeke, C. , De Regge, N. , Tabares, E. , & Favoreel, H. W. (2012). The IE180 protein of pseudorabies virus suppresses phosphorylation of translation initiation factor eIF2. Journal of Virology, 86(13), 7235–7240. 10.1128/jvi.06929-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villanueva, E. , Martí‐Solano, M. , & Fillat, C. (2016). Codon optimization of the adenoviral fiber negatively impacts structural protein expression and viral fitness. Scientific Reports, 6(1), 1–10. 10.1038/srep27546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virgin, H. W. , Latreille, P. , Wamsley, P. , Hallsworth, K. , Weck, K. E. , Dal Canto, A. J. , & Speck, S. H. (1997). Complete sequence and genomic analysis of murine gammaherpesvirus 68. Journal of Virology, 71(8), 5894–5904. 10.1128/jvi.71.8.5894-5904.1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh, D. , Mathews, M. B. , & Mohr, I. (2013). Tinkering with translation: Protein synthesis in virus‐infected cells. Cold Spring Harbor Perspectives in Biology, 5(1), a012351. 10.1101/cshperspect.a012351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Q. , Lee, I. , Ren, J. , Ajay, S. S. , Lee, Y. S. , & Bao, X. (2013). Identification and functional characterization of tRNA‐derived RNA fragments (tRFs) in respiratory syncytial virus infection. Molecular Therapy, 21(2), 368–379. 10.1038/mt.2012.237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Xi , Hou, J. , Quedenau, C. , & Chen, W. (2016). Pervasive isoform‐specific translational regulation via alternative transcription start sites in mammals. Molecular Systems Biology, 12(7), 875. 10.15252/msb.20166941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Xiao , Lu, Z. , Gomez, A. , Hon, G. C. , Yue, Y. , Han, D. , … He, C. (2014). N 6‐methyladenosine‐dependent regulation of messenger RNA stability. Nature, 505(7481), 117–120. 10.1038/nature12730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Xiao , Zhao, B. S. , Roundtree, I. A. , Lu, Z. , Han, D. , Ma, H. , … He, C. (2015). N6‐methyladenosine modulates messenger RNA translation efficiency. Cell, 161(6), 1388–1399. 10.1016/j.cell.2015.05.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei, C. M. , Gershowitz, A. , & Moss, B. (1975). Methylated nucleotides block 5′ terminus of HeLa cell messenger RNA. Cell, 4(4), 379–386. 10.1016/0092-8674(75)90158-0 [DOI] [PubMed] [Google Scholar]
- Wen, J. , Lv, R. , Ma, H. , Shen, H. , He, C. , Wang, J. , … Diao, J. (2018). Zc3h13 regulates nuclear RNA m6A methylation and mouse embryonic stem cell self‐renewal. Molecular Cell, 69(6), 1028–1038.e6. 10.1016/j.molcel.2018.02.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werden, S. J. , Lanchbury, J. , Shattuck, D. , Neff, C. , Dufford, M. , & McFadden, G. (2009). The myxoma virus M‐T5 ankyrin repeat host range protein is a novel adaptor that coordinately links the cellular signaling pathways mediated by Akt and Skp1 in virus‐infected cells. Journal of Virology, 83(23), 12068–12083. 10.1128/jvi.00963-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White, J. P. , Cardenas, A. M. , Marissen, W. E. , & Lloyd, R. E. (2007). Inhibition of cytoplasmic mRNA stress granule formation by a viral proteinase. Cell Host and Microbe, 2(5), 295–305. 10.1016/j.chom.2007.08.006 [DOI] [PubMed] [Google Scholar]
- Winkler, R. , Gillis, E. , Lasman, L. , Safra, M. , Geula, S. , Soyris, C. , … Stern‐Ginossar, N. (2019). m 6 A modification controls the innate immune response to infection by targeting type I interferons. Nature Immunology, 20(2), 173–182. 10.1038/s41590-018-0275-z [DOI] [PubMed] [Google Scholar]
- Yamasaki, S. , Ivanov, P. , Hu, G. F. , & Anderson, P. (2009). Angiogenin cleaves tRNA and promotes stress‐induced translational repression. Journal of Cell Biology, 185(1), 35–42. 10.1083/jcb.200811106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, J. Y. , Deng, X. Y. , Li, Y. S. , Ma, X. C. , Feng, J. X. , Yu, B. , … Gao, S. (2018). Structure of Schlafen13 reveals a new class of tRNA/rRNA‐ targeting RNase engaged in translational control. Nature Communications, 9(1), 1–13. 10.1038/s41467-018-03544-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, Z. , Bruno, D. P. , Martens, C. A. , Porcella, S. F. , & Moss, B. (2011). Genome‐wide analysis of the 5′ and 3′ ends of vaccinia virus early mRNAs delineates regulatory sequences of annotated and anomalous transcripts. Journal of Virology, 85(12), 5897–5909. 10.1128/JVI.00428-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao, Q. J. , Sang, L. , Lin, M. , Yin, X. , Dong, W. , Gong, Y. , & Zhou, B. O. (2018). Mettl3–Mettl14 methyltransferase complex regulates the quiescence of adult hematopoietic stem cells. Cell Research, 28(9), 952–954. 10.1038/s41422-018-0062-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye, F. , Chen, E. R. , & Nilsen, T. W. (2017). Kaposi's sarcoma‐associated herpesvirus utilizes and manipulates RNA N6‐adenosine methylation to promote lytic replication. Journal of Virology, 91(16), e00466‐17. 10.1128/JVI.00466-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeung, M. L. , Bennasser, Y. , Watashi, K. , Le, S.‐Y. , Houzet, L. , & Jeang, K.‐T. (2009). Pyrosequencing of small non‐coding RNAs in HIV‐1 infected cells: Evidence for the processing of a viral‐cellular double‐stranded RNA hybrid. Nucleic Acids Research, 37(19), 6575–6586. 10.1093/nar/gkp707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yue, Z. , & Shatkin, A. J. (1997). Double‐stranded RNA‐dependent protein kinase (PKR) is regulated by reovirus structural proteins. Virology, 234(2), 364–371. 10.1006/viro.1997.8664 [DOI] [PubMed] [Google Scholar]
- Zakaria, C. , Sean, P. , Hoang, H.‐D. , Leroux, L.‐P. , Watson, M. , Workenhe, S. T. , … Alain, T. (2018). Active‐site mTOR inhibitors augment HSV1‐dICP0 infection in cancer cells via dysregulated eIF4E/4E‐BP axis. PLoS Pathogens, 14(8), e1007264. 10.1371/journal.ppat.1007264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, F. , Moon, A. , Childs, K. , Goodbourn, S. , & Dixon, L. K. (2010). The African swine fever virus DP71L protein recruits the protein phosphatase 1 catalytic subunit to dephosphorylate eIF2α and inhibits CHOP induction but is dispensable for these activities during virus infection. Journal of Virology, 84(20), 10681–10689. 10.1128/jvi.01027-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, L. , & Wang, A. (2012). Virus‐induced ER stress and the unfolded protein respons. Frontiers in Plant Science, 3, 293. 10.3389/fpls.2012.00293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, S. , Sun, L. , & Kragler, F. (2009). The phloem‐delivered RNA pool contains small noncoding RNAs and interferes with translation. Plant Physiology, 150(1), 378–387. 10.1104/pp.108.134767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng, G. , Dahl, J. A. , Niu, Y. , Fedorcsak, P. , Huang, C. M. , Li, C. J. , … He, C. (2013). ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Molecular Cell, 49(1), 18–29. 10.1016/j.molcel.2012.10.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng, Q. , Hou, J. , Zhou, Y. , Li, Z. , & Cao, X. (2017). The RNA helicase DDX46 inhibits innate immunity by entrapping m 6 A‐demethylated antiviral transcripts in the nucleus. Nature Immunology, 18(10), 1094–1103. 10.1038/ni.3830 [DOI] [PubMed] [Google Scholar]
- Zhou, J. , Wan, J. , Gao, X. , Zhang, X. , Jaffrey, S. R. , & Qian, S. B. (2015). Dynamic m6 A mRNA methylation directs translational control of heat shock response. Nature, 526(7574), 591–594. 10.1038/nature15377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, L. , Ding, X. , Zhu, X. , Meng, S. , Wang, J. , Zhou, H. , … Zhu, G. (2011). Biphasic activation of PI3K/Akt and MAPK/Erk1/2 signaling pathways in bovine herpesvirus type 1 infection of MDBK cells. Veterinary Research, 42(1), 57. 10.1186/1297-9716-42-57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler, C. G. K. , Allon, S. J. , Nyquist, S. K. , Mbano, I. M. , Miao, V. N. , Tzouanas, C. N. , … Ordovas‐Montanes, J. (2020). SARS‐CoV‐2 receptor ACE2 is an interferon‐stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell, 181(5), 1016–1035.e19. 10.1016/j.cell.2020.04.035 [DOI] [PMC free article] [PubMed] [Google Scholar]


