In December of 2019, an outbreak of pneumonia of unknown cause was reported in Wuhan, Hubei Province, China. By January 2020, a novel coronavirus—that was named severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2)—was isolated from patients in Wuhan and was identified as the causative pathogen of the disease, which was named Coronavirus disease of 2019 (COVID‐19). In the middle of March, the World Health Organization (WHO) announced COVID‐19 outbreak a pandemic. 1 According to the daily report of the WHO, as of 18 July 2020, COVID‐19 has spread rapidly to infect more than 14 000 000 people and has caused roughly 597 000 deaths globally. 2
The typical clinical presentation of SARS‐CoV‐2 infection is that of viral pneumonia, while the most severe of cases are marked by the development of respiratory failure and multi‐organ dysfunction, 1 occasionally in the frame of a 'cytokine‐storm' syndrome driven by a dysregulated immune response. 3
So far, several patient‐related features that associate with greater disease severity and mortality have been recognized. Age is a principal factor driving disease course and mortality in reports from China, Italy and the United Kingdom. 4 , 5 , 6 Mortality is relatively low across a broad age range from 0 to 50 years, followed by an exponential increase with progressing age. In a summary report from the Chinese Center Disease Control and Prevention among 72 314 cases records of COVID‐19, the overall case‐fatality rate (CFR) was 2.3%, but in the age‐group 70‐79 and >80 years, the CFR increased to 8.0% and 14.8%, respectively. 7 The reported CFR for these age groups in Italy was 12.8% and 20.8%, respectively. 4 The risk of acquisition of SARS‐CoV‐2 infection for children is roughly similar to that of adults, although they present a substantially lower likelihood for severe disease. 8 Apart from advancing age, severe infection is more commonly observed among males, 5 , 6 in patients with obesity, 5 , 6 , 9 diabetes mellitus, 6 , 10 hypertension, 5 coronary artery disease, 11 chronic kidney disease, 6 among smokers, 5 , 10 and among individuals with chronic obstructive pulmonary disease (COPD). 12 , 13 Trends towards a more severe course have been observed for certain racial/ethnic backgrounds, with Black, 5 , 6 Asian or mixed‐race individuals 6 more prone to severe disease and death compared to those of Caucasian origin. 13
Interestingly, these factors that foretell a severe course of SARS‐CoV‐2 and mortality considerably overlap with conditions known to associate with decreased telomere length. This allows for a hypothesis of a potential pathogenetic link between telomere shortening and severe SARS‐CoV‐2 infection to be made.
Telomeres are regions of repetitive sequences at the end of eukaryote chromosomes that ensure genome integrity and prevent fusion between adjacent chromosomes. 14 With each cell cycle, telomeres become gradually shorter, so that decreases in telomere length following consecutive cellular divisions correspond to a constantly diminishing replicative capacity. 15 In rapidly replicating cells such as germline and hematopoietic stem cells, their length is maintained relatively stable by action of the enzyme telomerase and its catalytical subunit telomerase reverse transcriptase (hTERT). 14
A shorter telomere length and/or telomere dysfunction has been ascertained with advancing age, 16 in males versus females, in obese versus to lean adults, in smokers versus non‐smokers and in people with type 2 diabetes mellitus, hypertension, chronic kidney disease, 17 COPD 12 and coronary artery disease versus those without these diseases. 18 The findings regarding trends according to ethnic background are equivocal, but reports converge towards a steeper decline of telomere length with increasing age among Black and Hispanic individuals compared with Caucasians. 19 , 20 , 21
Of note, these observations are typically made by measuring telomere length in peripheral white blood cells, principally peripheral blood mononuclear cells (PBMCs), a population comprised of monocytes and T, B and NK lymphocytes or in their sub‐populations. The telomere length in these differentiated circulating cell types essentially reflects that of the whole hematopoietic lineage. In a simplified model approach, the rate of replenishment and hence absolute number of vastly recruited and/or lost peripheral lymphocytes in the setting of an acute viral (such as SARS‐CoV‐2) infection would be an increasing function of telomere length (and hence replicative capacity) of their precursor cells 15 ; of note, an absolute lymphopenia has been identified as a predictor of severe disease and adverse outcomes from early on in the course of COVID‐19 pandemic. 22
The conceptual idea of susceptibility to acute viral infection with respect to telomere length is not new. Cohen et al reported that in a relatively selected population of healthy adults aged between 18 and 55 years, following experimental exposure to Rhinovirus 39 (a single‐stranded RNA virus), a shorter telomere length in PBMCs, total lymphocytes, and CD4+ and CD8+ T‐lymphocyte subsets was associated with an increased probability of upper respiratory infection. 23
It could be argued that the presented overlap between the epidemiological features of COVID‐19 and telomere length may be confounded by advanced age itself, since short telomeres can be considered as an index of ageing and physical frailty 24 , 25 as well as by the presence of comorbidities that limit cardiovascular reserves. Nonetheless, the telomere shortening‐related functional changes in cellular components of the immune system could, at least partly, be explanatory to these observations. A diminishing telomere length in human lymphocytes is related to the process of their replicative senescence (or biological “ageing”). 26 The hypothesis that cellular senescence may be a key factor that drives susceptibility to severe SARS‐CoV‐2 infection has been recently postulated in a review article by Malavolta et al 27 The authors additionally reviewed putative therapeutic strategies utilizing agents that target senescent cells or aspects of their secretory physiology. As regards lymphocyte senescence in particular, this is marked by loss of telomerase activity and telomere shortening which may lead to differential effects based on lymphocyte type and functional setting. 26 Although the process of immune cell senescence and its relationship to telomere shortening has been fairly well characterized in the setting of chronic viral infections such as HIV and chronic hepatitis B and C, 28 its relevance in the setting of acute viral exposure is less well defined. Disruption of telomere integrity by anti‐telomerase agent KLM‐001 inhibits proliferation and cellular dysfunction and induces apoptosis in human CD4+ lymphocytes in vitro. 29 Conversely, CD8+ lymphocyte senescence associated with critical telomere shortening induces a state of 'hyperfunction' with evasion of apoptosis, increased secretion of pro‐inflammatory cytokines such as tumour necrosis factor‐alpha and interleukin‐6 and loss of surface CD28, a co‐stimulatory receptor necessary for the mobilization of targeted T‐cell immune responses. 30 In the previously mentioned study by Cohen et al, the risk of development of clinical illness after viral exposure was negatively associated with telomere length in CD8+ CD28‐ lymphocytes. 23 It is possible that this distinctive mode of cytokine secretory hyperfunction of functionally aged lymphocytes may establish an increased cytokine in vivo environment, which may accelerate telomere shortening and loss of CD28 in co‐existing non‐senescent lymphocytes. 26 Hence, in the setting of an acute infection this vicious circle may present a hindrance to effective immune responses in on one hand and predispose to exaggerated and uncontrolled inflammatory responses, much the same as the case of SARS‐CoV‐2 'cytokine storm'. 3
Furthermore, in vitro studies have highlighted the feasibility of partial reversal of the senescent functional phenotype through manipulation of components of the telomere/telomerase system. Preservation of telomere length through constitutive expression of telomerase (hTERT) gene in CD8 + lymphocytes from HIV‐infected human donors can restore their antiviral activity. 31 Likewise, exposure of CD8 + lymphocytes to telomerase activator TAT2 enhances their antiviral immune function. 32
Besides, the complex interplay between telomere‐related pathways and other ageing‐associated physiological changes such as decreased activity of proteins of the sirtuin family, 33 , 34 intracellular nicotinamide adenine dinucleotide (NAD+) depletion 33 and mitochondrial dysfunction 25 , 34 may play an complementary role as regards the aforementioned observations (Figure 1).
FIGURE 1.
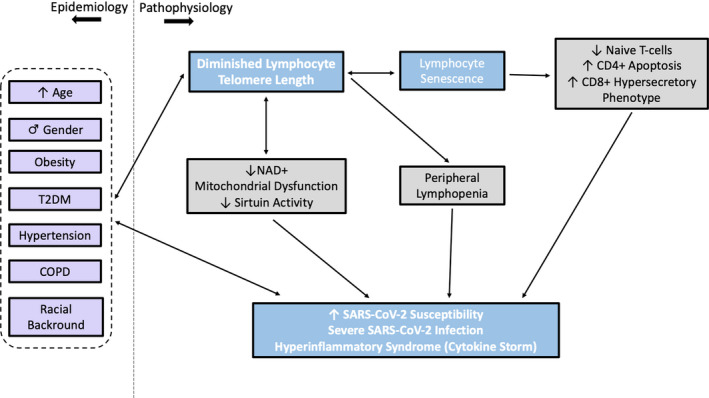
An explanatory scheme illustrating the epidemiological and pathophysiological aspects of the proposed relationship between diminishing lymphocyte telomere length and severe coronavirus disease of 2019 (COVID‐19). T2DM: type 2 diabetes mellitus; COPD: chronic obstructive pulmonary disease; NAD+: nicotinamide adenine dinucleotide; SARS‐CoV‐2: acute respiratory syndrome coronavirus 2.
Based on the above evidence, we hypothesize that individuals who exhibit lymphocyte telomere shortening may be more prone to severe and potentially lethal SARS‐CoV‐2 infection. We further speculate that this observation is driven by a complex immune dysregulation tracing back to immune cell senescence associated with telomere shortening, leading to increased susceptibility to infection and clinical disease (particularly pneumonia) by SARS‐CoV‐2, as well as unfavourable disease progression potentially marked by cytokine‐storm syndrome. This hypothesis would explain certain unique epidemiological aspects of the current pandemic, namely the continuous relationship of ascending age with disease severity and the susceptibility of certain patient groups to severe infection. Already, several therapeutic agents currently undergoing clinical testing for SARS‐CoV‐2 infection target components of the senescence‐associated pro‐inflammatory cellular phenotype. 27 Taking into consideration that manipulation of the telomere/telomerase system can ameliorate the deleterious effects of senescence on lymphocyte function in vitro, 31 , 32 the presented theory could also harbour implications for identifying high‐risk individuals and potentially guiding future therapeutic research against SARS‐CoV‐2 and other viral pathogens.
CONFLICT OF INTEREST
The authors declare no conflicts of interest.
REFERENCES
- 1. He F, Deng Y, Li W. Coronavirus Disease 2019 (COVID‐19): What we know? J Med Virol. 2020;92:719–725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO . Coronavirus disease (COVID‐19) Pandemic. https://www.who.int/emergencies/diseases/novel-coronavirus-2019. Accessed May 20, 2020
- 3. Francesca Coperchini LC, Croce L, Magri F, Rotondi M. The cytokine storm in COVID‐19: An overview of the involvement of the chemokine/chemokine‐receptor system. Cytokine Growth Factor Rev. 2020;53:25–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Onder G, Rezza G, Brusaferro S. Case‐fatality rate and characteristics of patients dying in relation to COVID‐19 in Italy. JAMA. 2020;323. [DOI] [PubMed] [Google Scholar]
- 5. Frederick K, Ho CAC‐M, Stuart R, et al. Modifiable and non‐modifiable risk factors for COVID‐19: results from UK Biobank. https://www.medrxiv.org/content/10.1101/2020.04.28.20083295v1.full.pdf. Accessed Accessed 24 July, 2020.
- 6. The OpenSAFELY Collaborative EW , Alex J, Krishnan J. OpenSAFELY: factors associated with COVID‐19‐related hospital death in the linked electronic health records of 17 million adult NHS patients. https://www.medrxiv.org/content/10.1101/2020.05.06.20092999v1.full.pdf+html. Accessed Accessed 24 July, 2020.
- 7. Wu JT, Leung K, Bushman M, et al. Estimating clinical severity of COVID‐19 from the transmission dynamics in Wuhan, China. Nat Med. 2020;26:506–510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Bi Q, Wu Y, Mei S, et al. Epidemiology and Transmission of COVID‐19 in Shenzhen China: analysis of 391 cases and 1,286 of their close contacts. medRxiv. 2003;2020(2020):2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Jian‐Min PB, He W, Fei WU, et al. Gender differences in patients with COVID‐19: focus on severity and mortality. medRxiv. 2020;2020(02):20026864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. CDC . People Who Are at Increased Risk for Severe Illness. ttps://www.cdc.gov/coronavirus/2019-ncov/specific-groups/people-at-higher-risk.html. Accessed 19 July 2020.
- 11. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Maremanda KP, Sundar IK, Li D, Rahman I. Age‐dependent assessment of genes involved in cellular senescence, telomere and mitochondrial pathways in human lung tissue of smokers, COPD and IPF: Associations with SARS‐CoV‐2 COVID‐19 ACE2‐TMPRSS2‐Furin‐DPP4 axis. medRxiv. 2020. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Williamson EJ, Walker AJ, Bhaskaran K, et al. OpenSAFELY: factors associated with COVID‐19 death in 17 million patients. Nature. 2020. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Shay JW, Wright WE. Telomeres and telomerase: three decades of progress. Nat Rev Genet. 2019;20(5):299‐309. [DOI] [PubMed] [Google Scholar]
- 15. Aviv A. Telomeres and COVID‐19. FASEB J. 2020;34(6):7247‐7252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Shammas MA. Telomeres, lifestyle, cancer, and aging. Curr Opin Clin Nutr Metab Care. 2011;14(1):28‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ameh OI, Okpechi IG, Dandara C, Kengne AP. Association between telomere length, chronic kidney disease, and renal traits: a systematic review. OMICS. 2017;21(3):143‐155. [DOI] [PubMed] [Google Scholar]
- 18. Turner KJ, Vasu V, Griffin DK. Telomere biology and human phenotype. Cells. 2019;8(1):73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Brown L, Needham B, Ailshire J. Telomere length among older U.S. adults: differences by race/ethnicity, gender, and age. J Aging Health. 2017;29(8):1350‐1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hunt SC, Chen W, Gardner JP, et al. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell. 2008;7(4):451‐458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Diez Roux AV, Ranjit N, Jenny NS, et al. Race/ethnicity and telomere length in the Multi‐Ethnic Study of Atherosclerosis. Aging Cell. 2009;8(3):251‐257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Zhao Q, Meng M, Kumar R, et al. Lymphopenia is associated with severe coronavirus disease 2019 (COVID‐19) infections: A systemic review and meta‐analysis. Int J Infect Dis. 2020;96:131‐135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Cohen S, Janicki‐Deverts D, Turner RB, et al. Association between telomere length and experimentally induced upper respiratory viral infection in healthy adults. JAMA. 2013;309(7):699‐705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Haapanen MJ, Perala MM, Salonen MK, et al. Telomere length and frailty: the helsinki birth cohort study. J Am Med Dir Assoc. 2018;19(8):658‐662. [DOI] [PubMed] [Google Scholar]
- 25. Salimi S, Hamlyn JM. COVID‐19 and crosstalk with the hallmarks of aging. J Gerontol. 2020. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chou JP, Effros RB. T cell replicative senescence in human aging. Curr Pharm Des. 2013;19(9):1680‐1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Malavolta M, Giacconi R, Brunetti D, Provinciali M, Maggi F. Exploring the relevance of senotherapeutics for the current SARS‐CoV‐2 emergency and similar future global health threats. Cells. 2020;9(4):909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Bellon M, Nicot C. Telomere dynamics in immune senescence and exhaustion triggered by chronic viral infection. Viruses. 2017;9(10):289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Cao D, Zhao J, Nguyan LN, et al. Disruption of telomere integrity and DNA repair machineries by KML001 induces T cell senescence, apoptosis, and cellular dysfunctions. Front Immunol. 2019;10:1152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Effros RB. The role of CD8 T cell replicative senescence in human aging. Discov Med. 2005;5(27):293‐297. [PubMed] [Google Scholar]
- 31. Dagarag M, Evazyan T, Rao N, Effros RB. Genetic manipulation of telomerase in HIV‐specific CD8+ T cells: enhanced antiviral functions accompany the increased proliferative potential and telomere length stabilization. J Immunol. 2004;173(10):6303‐6311. [DOI] [PubMed] [Google Scholar]
- 32. Fauce SR, Jamieson BD, Chin AC, et al. Telomerase‐based pharmacologic enhancement of antiviral function of human CD8+ T lymphocytes. J Immunol. 2008;181(10):7400‐7406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Omran HM, Almaliki MS. Influence of NAD+ as an ageing‐related immunomodulator on COVID 19 infection: a hypothesis. J Infect Public Health. 2020. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Amano H, Sahin E. Telomeres and sirtuins: at the end we meet again. Mol Cell Oncol. 2019;6(5):e1632613. [DOI] [PMC free article] [PubMed] [Google Scholar]


