Abstract
Background and Aim
Hepatic steatosis (HS) is associated with diabetes, hypertension, and obesity, comorbidities recently related to COVID‐19 severity. Here, we assessed if tomographic HS is also a risk factor for severe COVID‐19 pneumonia.
Methods
We included 213 patients with a positive real time polymerase chain reaction (RT‐PCR) test and chest computed tomography (CT) from an out‐hospital facility and a hospital. We obtained information on demographics; weight; height; smoking history; diabetes; hypertension; and cardiovascular, lung, and renal disease. Two radiologists scored the CO‐RADs system (COVID‐19 Reporting and Data System) (1 = normal, 2 = inconsistent, 3–4 = indeterminate, and 5 = typical findings) and the chest CT severity index (≥20 of 40 was considered severe disease). They evaluated the liver‐to‐spleen ratio (CTL/S) and defined tomographic steatosis as a CTL/S index ≤0.9. We used descriptive statistics, χ2 and t student tests, logistic regression, and reported odds ratio (OR) with 95% confidence interval (CI).
Results
Of the patients, 61% were men, with a mean age of 51.2 years, 48.3% were CO‐RADs 1 and 51.7% CO‐RADs 2–5. Severe tomographic disease was present in 103 patients (48.4%), all CO‐RADs 5. This group was older; mostly men; and with a higher prevalence of obesity, hypertension, diabetes, and HS (69.9 vs 29%). On multivariate analysis, age (OR 1.058, 95% CI 1.03–1.086, P < 0.0001), male gender (OR 1.9, 95% CI 1.03–3.8, P = 0.04), and HS (OR 4.9, 95% CI 2.4–9.7, P < 0.0001) remained associated.
Conclusion
HS was independently associated with severe COVID pneumonia. The physiopathological explanation of this finding remains to be elucidated. CTL/S should be routinely measured in thoracic CT scans in patients with COVID‐19 pneumonia.
Keywords: CO‐RADs, computed tomography severity index, COVID‐19 pneumonia, hepatic steatosis
Hepatic steatosis is an independent risk factor for chest computed tomography ?(CT)?severe ?COVID‐19? infection (odds ratio 4.9, 95% confidence interval 2.4–?9.7, P < 0.0001). Liver CT density should be measured and reported in all chest CT scans in patients with ?COVID‐19? pneumonia.
Introduction
Ever since the first cases of the COVID‐19 pandemic began to surface, it was recognized that certain groups were at an increased risk of complications from the viral infection. Risk factors associated with greater incidence of severe/critical illness and mortality are age, diabetes, obesity, hypertension, and metabolic syndrome, among others. 1 , 2 , 3 On the other hand, it is also well recognized that patients with some of these comorbidities, mainly type 2 diabetes and obesity, are at higher risk for non‐alcoholic fatty liver disease (NAFLD), recently renamed metabolic fatty liver disease (MAFLD), which at the same time may worsen these disorders 4 , 5 , 6 , 7 and which has been implicated in the inflammatory response (increased neutrophil‐to‐lymphocyte ratio [NLR]) with poorer outcomes in patients with COVID‐19. 8
Global prevalence of NAFLD has increased in the past two decades, and it has been calculated to be present in up to 24%. 7 , 9 Similarly, the estimated prevalence of NAFLD in Mexico and other regions of Latin America is around 20–29.9% and 30.4% respectively. 5 NAFLD is a complex entity with hepatic and extrahepatic pathophysiological and clinical manifestations, which finally result in ectopic deposits of fatty substrates in the liver, with a spectrum ranging from simple hepatic steatosis (HS) without inflammation to steatohepatitis, advanced fibrosis, and cirrhosis. 7 Imaging plays a role in its assessment, and nonenhanced computed tomography (CT) has proven to be a useful and accurate noninvasive method for diagnosing HS and quantifying the severity of liver fat. 10 , 11 This is because HS reduces attenuation of the liver, manifesting as a less dense liver parenchyma than expected, which is quantified in Hounsfield Units (HU). In contrast, as the spleen is devoid of fat, it can be used as an internal control, and several methods have shown good correlation to macrovesicular steatosis on biopsy, such as liver attenuation index and liver‐to‐spleen ratio (CTL/S), and have been used as markers of HS. 12 Although CT is not accurate in assessing milder forms of HS (≤5% of fat content), it is a reliable method with high specificity in the diagnosis of moderate to severe HS (≥20% of fat infiltration). 9 , 13
On the other hand, chest CT scans are useful in different scenarios of COVID‐19 (i.e. diagnosis and prognosis). 14 Pulmonary involvement is assessed by the chest CT severity score. This score showed a correlation with other paraclinical and clinical scores, including a respiratory rate ≥30/min, resting blood oxygen saturation ≤93%, and partial pressure of arterial blood oxygen (PaO2)/oxygen concentration (FiO2) ≤300 mmHg. Indeed, with a cut‐off value of ≥19.5/40, it has a sensitivity of 83.3% and a specificity of 94% in the identification of severe infection and an NPV of 96.3% to rule out severe forms. 15 A chest CT severity score above this threshold correlates with parameters of ventilatory impairment (mechanical ventilation risk, respiratory rate, and hypoxemia) and adverse inflammatory response (neutrophilia, lymphopenia, hypersensitive CRP, and procalcitonin). 15
During the CT evaluation of patients with COVID‐19 pneumonia, the CT scan volume routinely includes the upper abdomen, and thus, most of the liver and spleen are visualized. Therefore, HS can be assessed in the same dataset. Due to the high worldwide prevalence of HS and its relationship with many disorders associated with the risk of COVID‐19 infection, we hypothesize that tomographic liver steatosis could represent an additional independent risk factor for severe SARS‐CoV‐2 infection according to the CT severity score definition.
Methods
From 16 March 2020 to 26 May 2020, we conducted a transversal study including 213 consecutive patients from two different institutions, an out‐hospital facility (n = 143) and a hospital (n = 70). Inclusion criteria were patients with a positive real time polymerase chain reaction (RT‐PCR) test (Extraction kit PureDireX, Bio‐Helix, Keelung City, Taiwan) for COVID‐19 obtained from a nasopharyngeal and oropharyngeal swab and from whom a chest CT scan was obtained. Clinical records from patients with COVID‐19 were reviewed by two third‐year radiology residents (Melissa Echeverri‐Bolaños and Robert Escarria‐Panesso). Data collection included age, gender, weight and height, body mass index (BMI), smoking history (including current and past smokers), and comorbidities (diabetes, hypertension, cardiovascular disease, lung disease, and renal disease). Local Medical Ethics Committee Approval was obtained prior to the study, and informed consent was waived.
CT technique and postprocessing
Patients were scanned in two different commercial CT systems of 16‐slices (Somatom Sensation 16, Siemens Healthinneers, Erlangen, Germany) and 512‐slices (Revolution CT, GE Medical Systems, Milwaukee, WI, USA) with a kV of 100–120, 200–300 mA with automated current modulation, a pitch factor of 1.375, and X‐ray rotation speed of 0.5 s in a single breathold at full inspiration. The tube parameters were selected at radiologist discretion (Eric Kimura‐Hayama and Julieta Galicia‐Alba). Images were reconstructed with a matrix of 512 × 512, a slice thickness of 0.6–1.25 mm with an increment of 0.6–1.25 mm, and a Lung or B46f kernel. The dataset was analyzed in a dedicated workstation (AW 4.7 GE Medical Systems or AW Server GE Medical Systems) by two different body‐imaging radiologists with 7 years of experience (Julieta Galicia‐Alba and Talia Nila Chargoy‐Loustaunau), and final scores were determined by consensus when discrepancy was noted.
CT interpretation and analyses included identification of areas of ground‐glass opacities, crazy‐paving pattern (i.e. ground‐glass opacities with interlobular septal thickening), consolidations, and cicatricial bands according to the standard nomenclature of Fleischner Society, 16 and outcomes were classified according to the time course into four categories as previously described by Pan et al. 17 In addition, the presence of air bronchogram, inverse halo or atoll sign, cardiac enlargement, dilatation of main pulmonary artery, mediastinal lymph node enlargement, tree‐in‐bud nodules, coronary artery calcification, and pleural effusion was also recorded.
CT findings were classified into five categories according to the RSNA Expert Consensus criteria, 18 and the CO‐RADs (COVID‐19 Reporting and Data System) system from the COVID‐19 Working Group of the Dutch Radiological Society. 19 In these scoring systems, CT findings are classified from normal or inconsistent findings to typical findings of COVID‐19 pneumonia. In addition, the CO‐RADs scoring system includes a sixth category for patients with a positive RT‐PCR test. According to this, all patients in our study group were categorized as CO‐RADs 6.
Estimation of pulmonary involvement was obtained through calculation of the chest CT severity index according to the original description of Yang et al. 15 In summary, lungs are divided into 20 segments according to the Boyden bronchial segmentation: 10 segments in the right lung and 10 in the left. If the segment was normal, it was scored with 0, 1 if the abnormal finding represented less than 50% of the segment, or 2 if it involved more than 50% of the segmental area. The total CT score was the sum of the individual segmental scores and ranged from 0 (no involvement) to 40 (i.e. all 20 segments are involved in more than 50% of their extension). A CT score of ≥20/40 was considered to demonstrate severe disease and has been shown to correlate with worse outcomes. 15
Liver density was measured by averaging the HU of a 1.5 cm2 circular region of interest (ROI) in four different areas of both lobes, each separated by the hepatic veins. Spleen density was obtained from a 1.5‐cm2 ROI placed in the parenchyma. In both cases, the ROIs avoided areas of high density (e.g. calcifications) or low density due to vascular structures, cysts, or vessel entry points at hila. The liver‐to‐spleen ratio (CTL/S) was calculated, and steatosis was defined if the CTL/S index was ≤0.9. 20 , 21
Statistical analysis
Quantitative data are presented as means and standard deviations, and qualitative data are presented as percentages. We used t‐student test and Chi‐square test according to the variable distribution. We performed logistic regression analysis and reported odds ratios (OR) with 95% confidence interval (CI). P ≤ 0.05 was indicative of a statistically significant difference. Statistical analyses were performed using software SPSS, version 24 (IBM, New York, NY, USA).
Results
Table 1 depicts baseline characteristics of the 213 patients included. There were 131 men and 82 women, with an average age of 51.2 years (±14.8 years).
Table 1.
Baseline characteristics of patients
| Parameter | Value |
|---|---|
| Age (years) | 51.2 ± 14.8 |
| Gender | |
| Male | 131 (61.5%) |
| Female | 82 (28.5%) |
| Comorbidities | |
| Obesity (BMI ≥30 kg/m2) | 61 (28.6%) |
| Overweight (BMI 25–29.9 kg/m2) | 78 (36.6%) |
| Diabetes mellitus | 44 (20.6%) |
| Hypertension | 57 (26.7%) |
| Smoking history | 49 (23%) |
| Cardiac disease | 13 (6.1%) |
| Lung disease | 6 (2.8%) |
| Renal disease | 1 (0.4%) |
| Any comorbidity† | 108 (50.7%) |
| Hepatic steatosis by CTL/S | 104 (48.8%) |
| Main CT findings/tomographic phase | |
| Ground‐glass opacities/phase 1 | 61 (34.2%) |
| Crazy‐paving pattern/phase 2 | 31 (17.4%) |
| Consolidation/phase 3 | 54 (30.3%) |
| Cicatricial changes/phase 4 | 32 (17.9%) |
| Additional CT findings | |
| Air bronchogram | 73 (34.2%) |
| Atoll sign | 27 (12.6%) |
| Tree‐in‐bud nodules | 4 (1.8%) |
| Lymph node enlargement | 41 (19.2%) |
| Cardiac enlargement | 27 (12.6%) |
| Main pulmonary artery dilatation | 50 (23.4%) |
| Coronary artery calcification | 67 (31.4%) |
| Pleural effusion | 4 (1.8%) |
| CO‐RADs 13 /RSNA Expert Consensus 14 | |
| 1 (normal)/no pneumonia | 103 (48.3%) |
| 2 (low‐risk)/atypical findings | 2 (0.8%) |
| 3 (indeterminate)/indeterminate findings | 4 (1.8%) |
| 4 (high risk)/indeterminate findings | 9 (4.2%) |
| 5 (very high risk)/typical findings | 95 (44.6%) |
| 6 (positive RT‐PCR)/NA | 213 (100%) |
| Chest CT severity score | |
| ≤19 | 110 (51.6%) |
| ≥20 | 103 (48.4%) |
Includes obesity; overweight; diabetes mellitus; hypertension; smoking history; and cardiac, lung, and renal diseases.
BMI, body mass index; CT, computed tomography; NA, not applicable; RT‐PCR, real time polymerase chain reaction.
A total of 108 patients (50.7%) had at least one of the studied comorbidities, with overweight being the most prevalent in 36.6%, followed by obesity in 28.6% and hypertension (26.7%). We observed tomographic steatosis in 104 patients (48.8%).
Table 1 also shows all the tomographic findings. All patients had the CTL/S ratio available.
CT showed normal findings in 48.3% of cases (i.e. CO‐RADs 1) and was abnormal in 51.7% of patients (i.e. CO‐RADs 2–5). CO‐RADs categories 4 and 5 with high and very high probability of viral pneumonia comprised 48.8% of all abnormal scans (4.2 and 44.6%, respectively), and nearly half of all patients had severe disease (i.e. CT score ≥ 20) (48.4%), all of them CO‐RADs 5. Nearly two thirds of patients were in tomographic phases 1 and 3, where ground‐glass opacities and consolidation are predominant, respectively.
When comparing the group with severe tomographic disease (CT score ≥ 20) versus the group with nonsevere pneumonia, the former had a higher prevalence of male gender (71.6 vs 52.3%), obesity (39.8 vs 18.1%), hypertension (40.7 vs 13.6%), diabetes (29.1 vs 12.7%), and HS (69.9 vs 29%); they were also older (Table 2 and Fig. 1). The remaining comorbidities were similar between the groups.
Table 2.
Comparison of groups with and without chest computed tomography (CT) severity ≥20
| Variable | Chest CT severity score | P | |
|---|---|---|---|
| ≤19, n = 110 | ≥20, n = 103 | ||
| Age, years ± SD | 46.5 ± 14.5 | 56.3 ± 13.5 | 0.0001 |
| Male gender, n (%) | 58 (52.3) | 73 (71.6) | 0.004 |
| Obesity, n (%) | 20 (18.1) | 41 (39.8) | <0.0005 |
| Overweight, n (%) | 36 (32.7) | 42 (40.7) | 0.0007 |
| Hypertension, n (%) | 15 (13.6) | 42 (40.7) | <0.00001 |
| Diabetes mellitus, n (%) | 14 (12.7) | 30 (29.1) | <0.003 |
| Smoking history, n (%) | 26 (23.6) | 23 (22.3) | 0.82 |
| Cardiac disease, n (%) | 5 (4.5) | 8 (7.8) | 0.2 |
| Lung disease, n (%) | 3 (2.7) | 3 (2.5) | 0.91 |
| Renal disease, n (%) | 0 (0) | 1 (1) | 0.29 |
| Any comorbidity, n (%) | 40 (36.3) | 68 (66) | 0.000015 |
| Hepatic steatosis, n (%) | 32 (29) | 72 (69.9) | <0.00001 |
Figure 1.
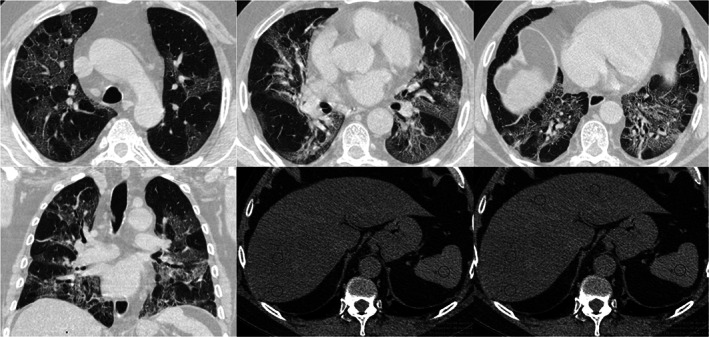
Chest CT severity score and CTL/S. Non‐enhanced CT images in a 57 years‐old male with COVID‐19 pneumonia. Axial images in upper, mid and basal portions of the thorax (figs. a‐c) and coronal reformation (fig. d) demonstrate multiple and bilateral ground‐glass opacities that involve more than 50% of some segments. These infiltrates are typical of COVID‐19 infection, categorized as CO‐RADS 5. The chest CT severity score was 36. The upper abdominal axial images from the same data set (figs. e‐f) showed decrease liver attenuation (ROIs average of 29 UH) when compare to spleen (45 HU). The CTL/S ratio was 0.65
At the logistic regression analysis, the only variables that remained associated with CT lung severity ≥20/40 were age (OR 1.058, 95% CI 1.03–1.086, P < 0.0001), male gender (OR 1.9, 95% CI 1.03–3.8, P = 0.04), and HS (OR 4.9, 95% CI 2.4–9.7, P < 0.0001). In another logistic regression model that considered only the variables age, male gender, and a combined variable of obesity/HS, we found similar results for age and gender, whereas the combination of both obesity and HS showed an OR of 5.9 (95% CI 2.6–13.7, P < 0.0001).
Discussion
Here, we evaluated the presence of tomographic HS and its association with the chest CT severity score, a tool that correlates with ventilatory impairment and an intense adverse inflammatory response among patients with SARs‐COVID19 pneumonia. 15
We found that HS, defined as the CTL/S index ≤0.9, 20 , 21 was significantly associated with severe pneumonia with an OR of 4.9.
Interestingly, both NAFLD and COVID‐19 infection are more prevalent in men and are linked to worst clinical outcomes (case fatality and cirrhosis progression). 22 , 23 Furthermore, HS has been also associated with some recognized COVID‐19 risk factors such as hypertension, obesity, and diabetes. Despite these facts, our results remained significant after adjusting for likely comorbidities, including obesity and diabetes, as well as for age and gender.
This is the first report that considers CT data analysis for both detection of HS and CT severity of COVID‐19 pneumonia. In a previous report in Asian populations by Zheng et al., authors suggested that the risk of obesity to COVID‐19 severity is significantly greater in those with fatty liver disease. 24 Unlike those results, after adjustment was performed, obesity did not showed a significant association with severe disease, and only age and HS remained significant. This might be the result of a different definition of obesity (BMI ≥25 kg/m2 in Zheng et al. vs ≥30 kg/m2 in our study) due to different ethnicities. According to a study of overweight and obesity in Asian American men, this population may develop health complications at lower BMIs than other racial/ethnic groups and tend to have higher percentages of body fat at lower BMIs. 25 Indeed, BMI cut‐off points for overweight and obesity in Asians are lowered to 23–27.4 kg/m2 and ≥27.5 kg/m2, respectively, compared to standard BMI cut‐off points for overweight (25–29.9 kg/m2) and obesity (≥30 kg/m2) in other races. 26 However, according to our results, combined obesity/HS resulted in a significant association with severe disease with an OR 6.49, which suggests a synergic interaction between them. In accordance with these findings, Ji et al. also reported a higher risk of progression to severe COVID‐19 and longer viral shedding time in patients with NAFLD. 27
The underlying pathophysiologic mechanisms that explain this association are unknown and warrant future research. However, there is evidence of increased hepatic and circulating interleukin‐6 levels in patients with NAFLD, 28 , 29 which might have an additive/synergistic role in promoting greater severity of COVID‐19. Obesity and NAFLD have been associated with increased production of pro‐inflammatory cytokines such as TNF‐α by adipose cells and Kupffer cells. 27 In a recent meta‐analysis, a higher NLR, a well‐known marker of systemic inflammation, was significantly associated with poorer outcomes in patients with COVID‐19 infection. 30 Also in patients with NAFLD, there is a significant association between this ratio and liver fibrosis severity. Moreover, it has been recently demonstrated that this association contributes to the COVID‐19‐induced inflammatory storm, carrying a greater risk for severe SARS‐CoV‐2 illness. 8
In addition, in patients with COVID‐19 infection, liver injury ranks second after lung injury. 31 This damage might be associated with overactivation of Kupffer cells, a virally induced cytotoxic T cell response and induction of a dysregulated innate immune response. 32 Furthermore, postmortem liver biopsies in these patients have shown microvesicular steatosis. 27
Our study has some limitations. First, the decision to perform a chest CT relied on the referring physician. Thus, a selection bias might be present as milder cases might not be included, hampering the validity of our results in this subgroup. Second, no histological or liver function tests are available in order to rule out the presence of other forms of NAFLD (i.e. cirrhosis and steatohepatitis), which have been implicated in poorer outcomes in this group of patients, 33 and it is well recognized that the accuracy of CT for detecting liver steatosis depends on the degree of fatty infiltration. The area under the curve (AUCs) of HS defined by CTL/S in diagnosis of ≥5% and ≥30% HS are 0.732 and 0.925, respectively. 20 Thus, according to our results, the presence of HS as a marker of increased risk of CT severity score may be only applied to patients with moderate to severe HS (≥30%). However, further studies are needed to know whether or not milder forms of fatty infiltration leads to an increased risk of severe forms of COVID‐19 in this population.
In conclusion, our data demonstrated that the risk of COVID‐19 pneumonia severity measured with the chest CT severity score is greater in those with HS. CTL/S should be routinely measured using thoracic CT scans in patients with COVID‐19 pneumonia.
Acknowledgments
We acknowledge all health‐care providers involved in the care of patients with COVID‐19 in our institutions.
Andres Palomar‐Lever and Gustavo Barraza contributed equally to this work.
Declaration of conflict of interest: None.
References
- 1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID‐19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020; 323: 1239. [DOI] [PubMed] [Google Scholar]
- 2. Mantovani A, Byrne CD, Zheng MH, Targher G. Diabetes as a risk factor for greater COVID‐19 severity and in‐hospital death: a meta‐analysis of observational studies. Nutr. Metab. Cardiovasc. Dis. 2020; 30: 1236–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Targher G, Mantovani A, Wang XB et al Patients with diabetes are at higher risk for severe illness from COVID‐19. Diabetes Metab. 2020; In press. 10.1016/j.diabet.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Yki‐Järvinen H. Non‐alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014; 2: 901–10. [DOI] [PubMed] [Google Scholar]
- 5. Chalasani N, Younossi Z, Lavine JE et al The diagnosis and management of non‐alcoholic fatty liver disease: practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology. 2012; 55: 2005–23. [DOI] [PubMed] [Google Scholar]
- 6. Friedman SL, Neuschwander‐Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 2018; 24: 908–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Angulo P. Nonalcoholic fatty liver disease. N. Engl. J. Med. 2002; 346: 1221–31. [DOI] [PubMed] [Google Scholar]
- 8. Targher G, Mantovani A, Byrne CD et al Detrimental effects of metabolic dysfunction‐associated fatty liver disease and increased neutrophil‐to‐lymphocyte ratio on severity of COVID‐19. Diabetes Metab. 2020; In press. 10.1016/j.diabet.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Younossi Z, Anstee QM, Marietti M et al Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2018; 15: 11–20. [DOI] [PubMed] [Google Scholar]
- 10. Zhang YN, Fowler KJ, Hamilton G et al Liver fat imaging‐a clinical overview of ultrasound, CT, and MR imaging. Br. J. Radiol. 2018; 91: 20170959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bohte AE, van Werven JR, Bipat S, Stoker J. The diagnostic accuracy of US, CT, MRI and 1H‐MRS for the evaluation of hepatic steatosis compared with liver biopsy: a meta‐ analysis. Eur. Radiol. 2011; 21: 87–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Limanond P, Raman SS, Lassman C et al Macrovesicular hepatic steatosis in living related liver donors: correlation between CT and histologic findings. Radiology. 2004; 230: 276–80. [DOI] [PubMed] [Google Scholar]
- 13. Wang XJ, Malhi H. Nonalcoholic fatty liver disease. Ann. Intern. Med. 2018; 169: ITC65–80. [DOI] [PubMed] [Google Scholar]
- 14. Rubin GD, Ryerson CJ, Haramati LB et al The role of chest imaging in patient management during the COVID‐19 pandemic: a multinational consensus statement from the Fleischner Society. Radiology. 2020; 7: 201365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Yang R, Li X, Liu H et al Chest CT severity score: an imaging tool for assessing severe COVID‐19. Radiology. 2020; 2: 2 10.1148/ryct.2020200047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hansell DM, Bankier AA, MacMahon H, McLoud TC, Müller NL, Remy J. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008; 246: 697–722. [DOI] [PubMed] [Google Scholar]
- 17. Pan F, Ye T, Sun P et al Time course of lung changes at chest CT during recovery from coronavirus disease 2019 (COVID‐19). Radiology. 2020; 295: 715–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Simpson S, Kay FU, Abbara S, Bhalla S, Chung JH, Chung M. Radiological Society of North America Expert Consensus Statement on reporting chest CT findings related to COVID‐19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA. Radiology. 2020; 2: 2 10.1148/ryct.2020200152. [DOI] [Google Scholar]
- 19. Prokop M, van Everdingen W, van Rees Vellinga T, Jet Quarles van Ufford JT, Stöger L et al CO‐RADS ‐ a categorical CT assessment scheme for patients with suspected COVID‐19: definition and evaluation. Radiology. 2020. 10.1148/radiol.2020201473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Byun J, Lee SS, Sung YS et al CT indices for the diagnosis of hepatic steatosis using non‐enhanced CT images: development and validation of diagnostic cut‐off values in a large cohort with pathological reference standard. Eur. Radiol. 2019; 29: 4427–35. [DOI] [PubMed] [Google Scholar]
- 21. Zhang Y, Wang C, Duanmu Y et al Comparison of CT and magnetic resonance mDIXON‐Quant sequence in the diagnosis of mild hepatic steatosis. Br. J. Radiol. 2018; 91: 20170587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lonardo A, Suzuki A. Sexual dimorphism of NAFLD in adults. Focus on clinical aspects and implications for practice and translational research. J. Clin. Med. 2020; 9: 1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Gebhard C, Regitz‐Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID‐19 outcomes in Europe. Biol. Sex Differ. 2020; 11: 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Zheng K, Gao F, Wang XB et al Obesity as a risk factor for greater severity of COVID‐19 in patients with metabolic associated fatty liver disease. Metabolism. 2020; 108: 154244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Mui P, Hill SE, Thorpe RJ. Overweight and obesity differences across ethnically diverse subgroups of Asian American men. Am. J. Mens Health. 2018; 12: 1958–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Jih J, Mukherjea A, Vittinghoff E et al Using appropriate body mass index cut points for overweight and obesity among Asian Americans. Prev. Med. 2014; 65: 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Ji D, Qin E, Xu J et al Non‐alcoholic fatty liver diseases in patients with COVID‐19: A retrospective study. J. Hepatol. 2020; 8: 451–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. van der Poorten D, Milner KL, Hui J et al Visceral fat: a key mediator of steatohepatitis in metabolic liver disease. Hepatology. 2008; 48: 449–57. [DOI] [PubMed] [Google Scholar]
- 29. Wieckowska A, Papouchado BG, Li Z, Lopez R, Zein NN, Feldstein AE. Increased hepatic and circulating interleukin‐6 levels in human nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2008; 103: 1372–9. [DOI] [PubMed] [Google Scholar]
- 30. Lagunas‐Rangel FA. Neutrophil‐to‐lymphocyte ratio and lymphocyte‐to‐C‐reactive protein ratio in patients with severe coronavirus disease 2019 (COVID‐19): a meta‐analysis. J. Med. Virol. 2020; In press. 10.1002/jmv.25819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ji D, Qin E, Xu J et al Implications of non‐alcoholic fatty liver diseases (NAFLD) in patients with COVID‐19. A preliminary analysis. J. Hepatol. 2020; 73: 451–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Bangash MN, Patel J, Parekh D. COVID‐19 and the liver: little cause for concern. Lancet Gastroenterol. Hepatol. 2020; 5: 529–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Targher G, Mantovani A, Byrne CD et al Risk of severe illness from COVID‐19 in patients with metabolic dysfunction‐associated fatty liver disease and increased fibrosis scores. Gut. 2020; 69: 1–2. [DOI] [PubMed] [Google Scholar]


