1. INTRODUCTION
Various cutaneous eruptions in COVID‐19 patients have been described. Although contrasting in morphology, all have overwhelmingly been benign in nature and resolve over days/weeks. Here, we report three patients with severe COVID‐19 and coagulopathies who developed large sacral/buttocks ulcerations arising during their disease course.
2. RESULTS
Patient 1
A 68‐year‐old man with hypertension and obesity presented to the emergency department (ED) complaining of fever, chills, cough, and shortness of breath (SOB). A chest radiograph revealed bilateral pulmonary infiltrates, leading to hospitalisation. COVID‐19 testing by reverse transcription‐polymerase chain reaction (RT‐PCR) was positive on admission day 2, and intravenous (IV) antibiotics, hydroxychloroquine, and azithromycin were initiated. He was intubated and mechanically ventilated on day 4 for hypoxic respiratory failure and acute respiratory distress syndrome (ARDS). Vasopressors were initiated for circulatory shock. He developed atrial fibrillation with rapid ventricular response (RVR) and was started on IV heparin. On day 12, a 5 × 11 cm sacral wound was noted, with suspicion of “deeper ulceration.”
He was transferred to our hospital on day 17. Laboratory studies at admission revealed elevated D‐dimers (11 360 ng/mL FEU), ferritin (1121 ng/mL), fibrinogen (681 mg/dL), and normocytic anaemia (Hb 9.2 g/dL). On day 19, dermatology consultation was conducted. Examination revealed a large black eschar on his sacrum/buttocks with surrounding violaceous induration and retiform purpuric edges (Figure 1A). A punch biopsy at the plaque edge revealed a thrombotic vasculopathy (Figure 1B). A hypercoagulation panel revealed elevated cardiolipin Immunoglobulin M (IgM) (62IgM Phospholipid Units) and Immunoglobulin G (IgG) (23IgG Phospholipid Units).
FIGURE 1.
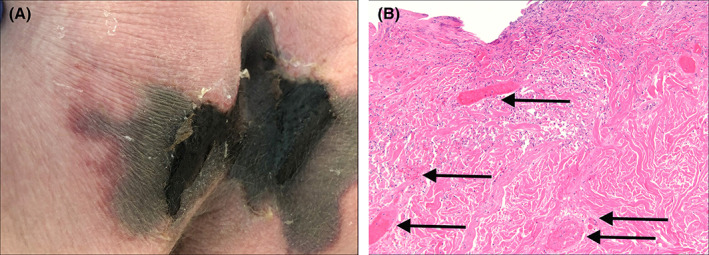
A, A livedoid plaque involving the sacrum and buttocks of patient 1. Note the central black eschar with a jagged superior border in the sacrum and lateral retiform purpuric borders. B, A photomicrograph (100×) of a punch biopsy taken from the border of patient 1's livedoid plaque displaying fibrin thrombi (arrows) in numerous blood vessels, consistent with a thrombotic vasculopathy
He became unresponsive, and an magnetic resonance imaging of the brain on day 32 revealed small subacute and remote haemorrhages in his bilateral frontal and parietal lobes, suggestive of haemorrhagic leukoencephalopathy. Because of non‐purposeful movements and loss of several brainstem reflexes, comfort care was initiated, and he expired on day 37.
Patient 2
A 56‐year‐old man with IgG kappa multiple myeloma with large granular lymphocytic leukaemia, hypertension, and obesity presented to the ED with fever, SOB, and cough. A CT scan of the chest revealed bilateral ground‐glass opacities, leading to hospitalisation. He was intubated on day 4 for hypoxemic respiratory failure and ARDS. COVID‐19 testing came back positive on day 5, and IV antibiotics, hydroxychloroquine, and azithromycin were initiated.
He developed melanotic stools secondary to a bleeding duodenal ulcer on day 7, requiring multiple blood transfusions and vasopressor support. On day 10, he was noted to have elevated D‐dimers (5250 ng/mL FEU) and other serologic abnormalities (Table 1). He was treated with tocilizumab and underwent sclerotherapy/clipping to control duodenal bleeding. A sacro‐coccygeal skin ulceration was documented on day 16.
TABLE 1.
Characteristics of three patients with severe COVID‐19 and sacral/buttocks ulcerations
| Patient 1 | Patient 2 | Patient 3 | ||
|---|---|---|---|---|
| Demographics | Gender | Male | Male | Male |
| Age | 69 | 56 | 73 | |
| Ethnicity | Caucasian | Caucasian | Caucasian | |
| Infection course | Route of infection | Community | Community | Community |
| Length of hospital stay | 39 | 39 | 27 | |
| Discharge disposition | Deceased | Acute rehab | Long term acute care | |
| Medical history | Comorbidities | HTN, gout, obesity (BMI 36) | Multiple myeloma with large granular lymphocytic leukaemia, pre‐diabetes, HTN, obesity (BMI 47) | HTN, COPD, CHF, CAD, obesity (BMI 32.7) |
| Charlson comorbidity index | 4 | 5 | 5 | |
| Labs | D‐dimer (ng/mL FEU) | 11 360 | 5250 | 1680, 6730 |
| Fibrinogen (mg/dL) | 681 | 393 | — | |
| Haemoglobin (g/dL) | 9.2 | |||
| Platelets (k/μL) | 389 | 49 | 320 | |
| Medications for COVID‐19 | HCQ, Azithromycin | HCQ, Azithromycin, Tocilizumab | HCQ, Azithromycin | |
| Hospitalisation day ulceration first noted (size of ulcer) | 12 (5 cm × 11 cm) | 19 (6 cm × 4 cm) | 7 (not documented) | |
Abbreviations: BMI, body mass index; CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; HCQ, hydroxychloroquine; HTN, hypertension.
On day 17, he was transferred to our hospital, by then having received ~44 U of packed red blood cells, 13 U of fresh frozen plasma, and 3 U of platelets. A wound care consultation for a sacro‐coccygeal ulceration revealed a 6 × 4 cm black eschar with surrounding induration and erythema (not shown). Biopsy was not performed. On day 22, he developed a fever and was reinitiated on IV antibiotics. A CT scan ruled out an underlying abscess, but because of suspicion, his fever was related to bacteraemia via the ulcer; it was debrided on day 24, leaving a deep wound (Figure 2A). He remained afebrile, his respiratory status improved, and he was discharged on day32.
FIGURE 2.
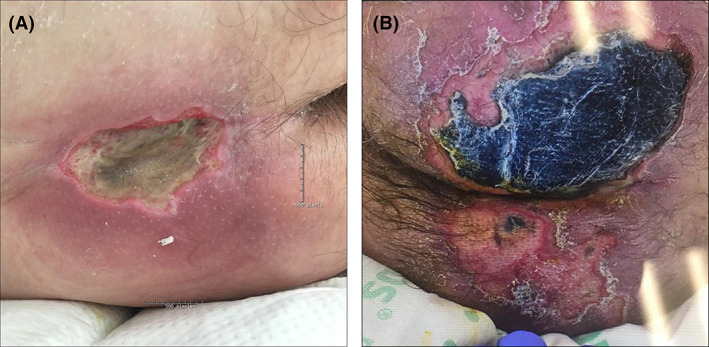
A, Large sacral ulcer in patient 2 following surgical debridement prompted by fever. Prior to debridement, the ulceration was covered by a black eschar (not shown). Surrounding erythema and focal violaceous discoloration is still noted at this later stage. B, Patient 3's ulceration is mostly confined to the left gluteal area and is covered by a black eschar. His right gluteal area has full‐thickness epidermal sloughing and focal eschar formation
Patient 3
A 73 year‐old man with chronic obstructive pulmonary disease, congestive heart failure, hypertension, and obesity presented to the ED with fever, chills, cough, and SOB. A CT chest revealed bilateral ground‐glass opacities, leading to hospitalisation. COVID‐19 testing came back positive later that day, and he was initiated on hydroxychloroquine and azithromycin. He was intubated on day 3 because of hypoxemic respiratory failure and ARDS. He did not require vasopressor therapy. Laboratory studies at this time included elevated D‐dimers (1680 ng/mL FEU) and other serologic abnormalities (Table 1). On day 6, gluteal erythema was noted, and the wound care team recommended topical medication, attributing erythema to occlusion and moisture. On day 14, he was extubated.
On day 15, he developed atrial fibrillation with RVR and was started on IV heparin, but this was discontinued because of epistaxis and haematuria. On day 18, he was febrile, reintubated because of respiratory failure, and initiated on vancomycin and meropenem. At that time, the buttocks plaque was noted to be violaceous in colour. Consultations for his ulcer were undertaken on day 24, at which time he was noted to have a large black eschar on his left gluteal region (Figure 2B). D‐dimers were elevated to 6730 ng/mL FEU. He was extubated and discharged to a long‐term acute care centre on day 31.
Sixteen days after discharge, he was rehospitalised for a gluteal abscess associated with his ulceration and started on IV antibiotics. Wound cultures were positive for Staphylococcus epidermidis and Staphylococcus lugdunensis susceptible to current antibiotics, and blood cultures were negative. His infected ulceration was debrided in the operating room and showed improvement following this and with IV antibiotics. He was discharged after a 7‐day hospitalisation.
3. DISCUSSION
A variety of cutaneous eruptions occurring in COVID‐19 patients have been described. The most common include morbilliform rashes, urticaria, vesiculo‐papular (varicella‐like) eruptions, acral lesions (“COVID toes”), and livedoid eruptions. 1 Both transient livedo reticularis and fixed livedoid plaques on the sacrum/buttocks have been described. 1 , 2 Although rashes described thus far may eventually prove to have diagnostic/prognostic value, none result in significant morbidity. Prominent acro‐cyanosis has been described in severely ill patients but not sequelae secondary to skin necrosis. 3 , 4 In a recently published photographic atlas of COVID‐19 patients from Spain, sizeable cutaneous ulceration was not observed. 1
Here, we describe three patients who developed large sacral/buttocks ulcers during their severe COVID‐19 disease courses. Location on sacral/buttocks areas correlates with areas where both COVID‐19‐associated livedoid plaques and pressure ulcers occur. 1 , 5 As all three of our patients developed ARDS requiring mechanical ventilation and had laboratory evidence of substantial systemic coagulopathy, it may be that this degree of cutaneous damage is limited to, or more likely in, patients with severe COVID‐19. Given the characteristics of our three patients, we believe these large ulcerations are likely caused by cutaneous ischaemia related to a combination of pressure, COVID‐19‐associated coagulopathy, and possibly other factors directly or indirectly related to COVID‐19.
Livedoid plaques seem to be a unique cutaneous feature in COVID‐19 patients. A prominent livedoid plaque was noted in patient 1. Both retiform purpuric edges of patient 1's plaque and thrombotic vasculopathy on histology are consistent with a livedoid plaque as the predominant cause of ulceration. This patient also had the most significant D‐dimer elevation of our three patients and was positive for IgM cardiolipin antibodies. To our knowledge, only one additional report of a severe livedoid plaque on the sacrum/buttocks concerning impending ulceration has been reported. This occurred in a 32‐year‐old man who also required intubation for severe COVID‐19. 5 We were unable to examine patients 2 or 3 early enough in their disease courses to determine if they had livedoid plaques, and unfortunately, neither had biopsies to evaluate for thrombotic vasculopathy.
Our patients also share many features placing them at increased risk of sacral/buttocks ulcerations because of hospital‐acquired pressure injuries (HAPIs). Risk factors directly attributed to severe COVID‐19 include hypoperfusion and systemic coagulopathy. Other indirect risk factors that our patients variably displayed include immobility and recumbency for prolonged periods, mechanical ventilation (making it difficult to turn/examine patients), poor nutrition, and faecal contamination/irritation. 6 , 7 In one study, average time to HAPI development after admission was 11.4 days. 6 Although it is unclear exactly when ulcerations in our patients first occurred, patient 3's ulcer was first documented significantly earlier than this average time, and patient 1's ulcer was first documented at approximately this average time but was already quite large (Table 1). Both observations support the likelihood that factors directly related to COVID‐19 are involved in their pathogenesis. The risk of traditional HAPIs in COVID‐19 patients are recognised, and challenges to prevent/treat these have been proposed. 7
Sacral/buttocks ulcerations in COVID‐19 patients have not yet been reported but are important, as they can serve as portals of entry for bacteria, leading to bacteraemia or sepsis. This is underscored by the ulcer‐related complications of patients 2 and 3, requiring medical intervention. Thus, even if patients are successfully treated for COVID‐19‐associated ARDS, these ulcers can lead to additional problems prolonging hospitalisation, leading to hospital readmission or even death. Furthermore, long‐term sequelae such as chronic pain and/or difficulty ambulating may result.
In summary, our observations in three patients establish the skin as a potential target organ of damage because of COVID‐19 that can add significant morbidity and mortality risk for patients both during and after their viral disease course. These observations support that wound care specialists consulted to see severe COVID‐19 patients should recognise the particular importance of monitoring for and preventing sacral/buttocks ulcers. Sacral/buttocks cutaneous abnormalities should be closely monitored, and supportive care should be implemented to avoid added pressure that may amplify cutaneous ischaemia. Observation of more patients examined early and followed prospectively during their disease course is needed to determine if the presence of livedoid plaques is a prerequisite or important risk factor for significant ulceration in COVID‐19 patients. Because thrombotic events have emerged as important causes of morbidity and mortality in COVID‐19 patients, more aggressive anticoagulation therapy may eventually help prevent significant skin breakdown in affected patients when sacral/buttocks erythema and/or livedoid plaques are identified. 8 , 9 , 10
CONFLICT OF INTEREST
Anthony P. Fernandez is an investigator for Pfizer, Corbus, Mallinckrodt, Novartis, and Roche pharmaceuticals. He receives personal research support from Mallinckrodt and Novartis; honorarium from AbbVie, UCB, Novartis, Mallinckrodt, and Celgene for consulting and advisory board participation; and honorarium from AbbVie, Novartis, and Mallinckrodt for teaching and speaking. Other authors have no conflicts of interests to report.
REFERENCES
- 1. Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID‐19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183(1):71–77. 10.1111/bjd.19163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Manalo IF, Smith MK, Cheeley J, Jacobs R. A dermatologic manifestation of COVID‐19: transient livedo reticularis. J Am Acad Dermatol. 2020. 10.1016/j.jaad.2020.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Zhang Y, Cao W, Xiao M, et al. Clinical and coagulation characteristics of 7 patients with critical COVID‐2019 pneumonia and acro‐ischemia. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E006. 10.3760/cma.j.issn.0253-2727.2020.0006. [DOI] [PubMed] [Google Scholar]
- 4. Llamas‐Velasco M, Muñoz‐Hernández P, Lázaro‐González J, et al. Thrombotic occlusive vasculopathy in skin biopsy from a livedoid lesion of a COVID‐19 patient. Br J Dermatol. 2020. 10.1111/bjd.19222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID‐19 infection: a report of 5 cases. Transl Res. 2020; epub ahead of print;220:1‐13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Rondinelli J, Zuniga S, Kipnis P, et al. Hospital‐acquired pressure injury: risk‐adjusted comparisons in an integrated healthcare delivery system. Nurs Res. 2018;67(1):16‐25. 10.1097/NNR.0000000000000258 PubMed PMID: 29240656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Tang J, Li B, Gong J, Li W, Yang J. Challenges in the management of critically ill COVID‐19 patients with pressure ulcer. Int Wound J. 2020. 10.1111/iwj.13399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Connors JM, Levy JH. COVID‐19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033–2040. 10.1182/blood.2020006000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Helms J, Tacquard C, Severac F, et al. High risk of thrombosis in patients with severe SARS‐CoV‐2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46(6):1089–1098. 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Paranjpe I, Fuster V, Lala A, et al. Association of Treatment Dose Anticoagulation with in‐hospital survival among hospitalized patients with COVID‐19. J Am Coll Cardiol. 2020;76(1):122–124. 10.1016/j.jacc.2020.05.001 pii: S0735‐1097(20)35218–9. [Epub ahead of print] PubMed PMID: 32387623. [DOI] [PMC free article] [PubMed] [Google Scholar]
ACKNOWLEDGEMENTS
The authors thank Janine Sot, MBA, for her expertise in preparing Figures 1 and 2 for this manuscript.


