Abstract
The outbreak of the 2019 novel coronavirus disease (SARS‐CoV‐2) has resulted in a major epidemic threat worldwide. However, the effects of neoviruses on infected pregnant women and especially on their fetuses and newborns are not well understood. Most up‐to‐date evidences about how SARS‐CoV‐2 affected patients especially in pregnancy were collected by conducting a comprehensive search of medical literature electronic databases. Immune‐related data of pregnant women, fetuses and newborns were further analysis. According to the limited literature, SARS‐CoV‐2 utilizes angiotensin converting enzyme 2 as its receptor and causes severe hypoxemia. Insufficiency of angiotensin converting enzyme 2 in pregnant women and the effects of hypoxia on the placental oxygen supply will cause severe perinatal complications. In addition, SARS‐CoV‐2 infection may disrupt maternal‐fetal immune tolerance and cause immunological damage to embryos. Because of these reasons, pregnancy complications such as fetal demise or premature birth, preeclampsia, intrauterine growth restriction, respiratory dyspnea, nervous system dysplasia and immune system defects are likely to occur in pregnant women with COVID‐19 or their newborns. Pregnant women infected with SARS‐CoV‐2 should be treated as a special group and given special attention. Fetuses and newborns of SARS‐CoV‐2‐infected pregnant women should be given more protection to reduce the occurrence of adverse events. In this review, we intend to provide an overview of the physiological and immunological changes that induce the pregnancy complications. This article will benefit the treatment and prognosis of fetuses and newborns of SARS‐CoV‐2‐infected pregnant women.
Keywords: angiotensin converting enzyme 2, COVID‐19, immunity, pregnancy, SARS‐CoV‐2
Introduction
Since the first case of the 2019 novel coronavirus disease (COVID‐19, previously known as 2019‐nCoV) was reported in Wuhan, China, in December 2019, neovirus infection has spread throughout China and the world. 1 This outbreak has been caused by SARS‐CoV‐2 that is a new coronavirus discovered in humans for the first time. It belongs to the coronavirus‐β genus and is similar to Middle East Respiratory Syndrome coronavirus (MERS‐CoV) and Severe Acute Respiratory Syndrome coronavirus (SARS‐CoV). 2 SARS‐CoV‐2 infection mainly causes interstitial pneumonia, hypoxemia and respiratory distress syndrome but is more infectious than SARS‐CoV and MERS‐CoV. Pregnant women, as a special group, can be infected with SARS‐CoV‐2, which often affects the fetuses and newborns of these women.
Since early February, Zhu et al. 3 first reported the clinical characteristics of 10 neonates born to mothers with confirmed SARS‐CoV‐2 infection, clinical data of pregnant women infected with SARS‐CoV‐2 are limited, and controversy exists over the prevalence and severity of pregnant women. Although some studies support the opinion that the COVID‐19 in pregnant women are similar to those in non‐pregnant ones, others show that the severity of infection in pregnant women is more serious. A systematic review on outcomes of SARS, MERS and COVID‐19 during pregnancy supported that miscarriage, preeclampsia, cesarean and perinatal death were more common than in the general population. 4 Consistent conclusion is that the incidence of premature delivery and cesarean section after infection in pregnant women is higher. The latest systematic review including eighteen articles with 108 pregnancies and 75 neonates reported an extremely high rate of cesarean section (92%) partly due to fetal distress and suspected a response in neonate to maternal SARS‐CoV‐2 infection. Severe maternal morbidity and perinatal death were reported in some studies. 5 Up to now, Zhu et al. reported the highest postnatal morbidity in 10 neonates, included 6 cases of respiratory dyspnea, 4 cases with digestive symptoms and 1 case of death. 3 Therefore, maternal SARS‐CoV‐2 infection deserves more attention. Analysis of physiological and immunological changes during pregnancy can explain the causes of poor prognosis and further provide theory basis for clinical guidelines on follow‐up of infected pregnant women and their newborns. 6
In this review article, we will reveal the causes of potential adverse pregnancy outcomes and poor prognosis of newborns by explaining the mechanism underlying hypoxemia decreases in coronavirus functional receptors and immunopathogenesis after SARS‐CoV‐2 infection (as showed in Figure 1). We are calling for obstetricians and neonatologists to establish a reasonable monitoring program as soon as possible to reduce the incidence of adverse pregnancy outcomes and neonatal complications.
Figure 1.
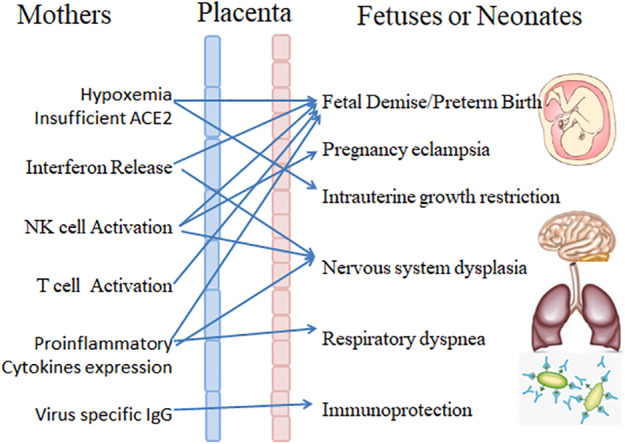
When SARS‐CoV‐2 infects pregnant women, hypoxemia, a decrease in coronavirus functional receptors and immunopathogenesis (interferon release, natural killer (NK) cell activation, T cell activation, and cytokine expression) occur. Adverse events include fetal demise/preterm birth, preeclampsia, intrauterine growth restriction, nervous system dysplasia, respiratory dyspnea and immune system defects.
Hypoxia and a decrease in ACE2 content in pregnant women with SARS‐CoV‐2 infection affect fetal development
Similar to SARS‐CoV, SARS‐CoV‐2 uses angiotensin converting enzyme 2 (ACE2) as its functional receptor for infecting human cells. 7 , 8 Studies have shown that ACE2 is mainly expressed in the respiratory, cardiovascular and digestive systems, which makes these organs susceptible to neoviruses. After the lung is infected by SARS‐CoV‐2, the pathological manifestations are pulmonary edema, hyaline membrane formation and pulmonary interstitial thickening, 9 while the main clinical manifestations are respiratory dyspnea and even acute respiratory failure. Since pregnant women are a special group of women with a fetus in their uterus, SARS‐CoV‐2 infection not only affects the pregnant women themselves but also affects the fetuses, which should be paid more attention when considering treatment.
Effects of maternal hypoxemia caused by SARS‐CoV‐2 infection on fetuses
Any change in a pregnant woman's body will affect the growth and development of the fetus. Studies have found that under physiological conditions, the cardiopulmonary load of pregnant women is already significantly increased in the second and third trimesters. If virus infection causes lung injury in pregnant women, this will accelerate the occurrence of respiratory distress. Once maternal respiratory failure occurs, hypoxemia can lead to an inadequate supply of blood and oxygen to the placenta, resulting in fetal distress, abortion, premature delivery and/or other adverse pregnancy events. During the epidemic of SARS‐CoV, the placental pathology of pregnant women infected with SARS‐CoV confirmed that virus infection led to an insufficient oxygen supply and various placental lesions, such as increases in intervillous and subvillous fibrin in the placenta in 3 cases of acute delivery due to SARS and extensive fetal thromboangiopathy, which was found in 2 placentas delivered in the late stage of the convalescence of SARS that were characterized by well‐defined nonvascular fibrotic villi and accompanied by intrauterine growth retardation and oligohydramnios. 10 Pathological study of COVID‐19 suggested that there were no villitis and chorioamnionitis in three placentas; however, there were some changes with one case of placenta, which showed the concomitant morphology of chorionic hemangioma and another one with massive placental infarction. What needs to be pointed out is that all three placenta were from patients in their third trimester with emergency cesarean section and lack of data in early pregnancy. 11
The influence of SARS‐CoV‐2 infection on pregnant women is still being investigated via clinical observations and data collection. Considering that SARS‐CoV‐2 shares the same infection route and receptor as SARS‐CoV and is more infectious than SARS‐CoV, its influence on pregnant women and their fetuses cannot be ignored. As a consequence, it is highly recommended that pregnant women infected with SARS‐CoV‐2 should undergo pathological examination of the placenta to assess the effect of SARS‐CoV‐2 infection at the time of delivery, and long‐term follow‐up should be conducted in neonates born under these conditions over time.
Effects of insufficient ACE2 in pregnant women with SARS‐CoV‐2 infection on fetuses
ACE2, as a key enzyme of the RAS system, is a functional receptor in SARS‐CoV‐2‐infected cells and is mainly expressed in the respiratory, digestive, cardiovascular and urinary systems. As a carboxypeptidase, ACE2 can effectively degrade angiotensin II into angiotensin 1–7, thus ensuring the blood perfusion of important organs. When cells expressing ACE2 were infected by SARS‐CoV‐2, the virus replicated and caused an immune response. In addition, it caused the destruction of ACE2‐positive cells, which reduced the level of ACE2 and caused the organs with high expression of ACE2 to appear hypoperfused. The decrease in ACE2 in pregnant women not only caused poor perfusion of their organs but also decreased placental blood perfusion, which affected the development of the fetus. Both human and animal studies have shown that the use of ACE inhibitors in early pregnancy can cause maternal weight loss and decreased blood pressure, uterine blood flow and amniotic fluid production, followed by fetal hypotension or death. The most common adverse effects of ACE inhibitors in the second or third trimester of pregnancy included intrauterine growth retardation, neonatal hypotension, renal failure, oligohydramnios, patent ductus arteriosus and pulmonary hypoplasia 12 Other studies have found that an ACE2 gene polymorphism in pregnant women is associated with recurrent abortion and fetal growth restriction. 13 , 14 Thus, SARS‐CoV‐2 infection in women is likely to cause adverse reactions similar to those caused by ACE2 inhibitors or ACE2 gene polymorphism in pregnant women and their fetuses. There are reports that ACE2 levels are higher in pregnant women than in normal nonpregnant women, 15 which suggests that pregnant women may be more susceptible to SARS‐CoV‐2 and shows the importance of ACE2 in pregnancy. Therefore, special attention should be paid to the effects of SARS‐CoV‐2 infection on pregnant women and on fetuses and neonates born to them.
The immune response in pregnant women infected with SARS‐CoV‐2 infection affects fetal and neonatal development
SARS‐CoV‐2 infection is similar to SARS‐CoV in that it can cause immune overreaction, which is manifested by excessive activation of immune cells and production of a large amount of interferon or a cytokine storm, thus causing immune damage while eliminating the virus. Due to the particular circumstances of pregnant women, once these changes occur during pregnancy, the maternal immune response to SARS‐CoV‐2 will have some adverse effects on fetuses. Whether this affects the normal development of fetuses and neonates including the development of the immune and nervous systems, is worthy of further study.
Effects of interferon levels in pregnant women with SARS‐CoV‐2 infection on fetuses and neonates
Elevated levels of interferon after maternal infection by the virus may affect fetal development and increase the risk of neurological diseases in newborns. Type I interferon (IFN‐I) is the most important immune factor involved in the early stage of antivirus infection. IFN‐I mediates placental dysplasia in mice infected with ZIKV virus (a single‐stranded, positive‐stranded RNA virus different from coronavirus) because IFN‐I receptor (IFNAR) signaling in the embryo inhibits the development of the placental labyrinth, leading to the abnormal constriction of the maternal‐fetal barrier. 16 Placental morphological changes due to the exposure of human chorionic explants to IFN‐I in the second trimester induced cytoskeletal rearrangement in the chorionic core, which finally increased the occurrence of fetal demise and intrauterine growth restriction. The overexpression of IFN‐I resulting from intrauterine TORCH infection is associated with clinical microcephaly, intracranial calcification and ventricular broadening, suggesting that IFN‐I is a mediator of abnormal brain development. 17 In patients with viral infection, IFN‐I is secreted by infected epithelial cells, fibroblasts and plasma dendritic cells and then binds to IFNAR to initiate downstream signaling to induce an antiviral status. Previous studies on SARS‐CoV have shown that the viral membrane protein may be a potential pathogen‐associated molecular pattern, which induces the upregulation of interferon‐β (IFN‐β). Although there has been no report on the existence of a SARS‐CoV‐2 ‘membrane protein’ similar to SARS‐CoV, in view of the high homology between SARS‐CoV‐2 and SARS‐CoV, great attention should be paid to pregnant women infected with SARS‐CoV‐2 and their newborns. 18 In particular, IFN‐I as a first‐line drug may cause adverse effects on the placenta or fetus.
Animal experiments have shown that exposure to IFN‐γ can inhibit embryo implantation and cause placental defects. Overexpression of IFN‐γ can lead to abnormal brain development in mice, which is also an internal factor potentially contributing to abortion and fetal intrauterine growth restriction. 19 Furthermore, IFN activate protective mechanisms aimed at both virus control and elimination, and some data have shown that the interferon‐gamma (IFN‐γ) level is elevated and finally decreased in severe patients with SARS‐CoV‐2 infection. 20 , 21
Based on the above theory, the antiviral response of pregnant women infected with SARS‐CoV‐2 may have an adverse effect on fetal development mediated by interferon, which should be given more attention.
Effects of elevated levels of proinflammatory cytokines in pregnant women with SARS‐CoV‐2 infection on fetuses and neonates
Increased levels of cytokines such as interleukin‐1β (IL‐1β), interleukin (IL‐6) and tumor necrosis factor‐α (TNF‐α) in pregnant women infected with SARS‐CoV‐2 can increase the risk of fetal preterm delivery and affect the development of the fetal respiratory system and nervous system. As was the case for SARS‐CoV, inflammatory cytokine storms are an important cause of severe manifestations of COVID‐19. 22 After infection with the virus, infected cells in response to SARS‐CoV‐2 infection release cytokines, recruiting leukemia and lymphocytes. The monocyte–macrophage system and lymphocytes cells are quickly activated and produce cytokines and chemokines, which are potential mediators of coronavirus immunopathology accompanying the clearance of the virus. Increased levels of IL‐1β, IL‐6, TNF‐α and other cytokines detected in pregnant women can induce labor and lead to abortion in the first and second trimesters and preterm birth in the third trimester. 23 , 24 In addition, IL‐1β and IL‐6 also mediate fetal defects related to maternal immune activation, including developmental abnormalities associated with bronchopulmonary dysplasia and brain injury. 25 This explains the high incidence of respiratory dyspnea among neonates born to SARS‐CoV‐2‐infected mothers reported by Zhu. 3 In addition, during pregnancy, the increasing in the pro‐inflammatory factor IL‐6 will upregulate the expression of IL‐17 by maternal T helper 17 (Th17) cells. Overexpression of IL‐17 may activate the IL‐17 receptor in fetal neurons, leading to cortical dysplasia and behavioral abnormalities in fetuses 26 , 27 and increasing the possibility that offspring will develop mental illness in adulthood. 28 Meanwhile, exposure to TNF‐α can lead to embryonic development disruption and fetal neural tube defects. 29 Therefore, obstetricians should be alert to the problems of early labor caused by the expression of inflammatory factors in SARS‐CoV‐2‐infected pregnant women and carry out post‐partum evaluations and long‐term follow‐up to monitor the respiratory and neurological development of newborns.
Potential effects of NK cell activation in pregnant women with SARS‐CoV‐2 infection on fetuses and neonates
Decreased numbers of natural killer (NK) cells in pregnant women may potentially cause intrauterine fetal demise, fetal growth restriction and preeclampsia. NK cells are the most important immune cells in the decidua in early pregnancy. Unlike the composition of peripheral blood cells, decidual NK cells account for the highest proportion of decidual leukocytes. Instead of playing a role as natural killer cells, these cells cooperate with trophoblast cells to participate in uterine vascular remodeling with the purpose of maximizing uterine blood flow into the placenta, which is necessary to shape placental formation and the fetal blood supply. 30 Peripheral blood NK accumulate in the decidua to form a decidual NK population in the first trimester. 31 , 32 However, the activation and decrease in the number of NK cells in peripheral blood will lead to the failure of decidual NK cell accumulation in the decidua, which impedes the reconstruction of uterine vessels. Failure of uterine vascular reconstruction leads to an insufficient placental blood supply, which is the cause of preeclampsia, fetal growth restriction, abortion and severe birth defects. Furthermore, viral infection in mice may lead to activation of decidual NK cells and expression of the activating ligand, NKG2D, resulting in abnormal migration of trophoblast cells, increased apoptosis of placental cells and fetal absorption. 33 In addition, NK cells may also directly participate in fetal development by secreting growth‐promoting factors, including gonadotropin, osteocalcin and osteopontin. 34 It has been reported that the number of NK cells is decreased in patients with SARS‐CoV‐2 infection. 35 Collectively, the decreased number and function of NK cells caused by COVID‐19 may potentially induce fetal growth restriction, abortion and preeclampsia during pregnancy, indicating that more attention should be paid to pregnant women with COVID‐19. Measures should be taken to evaluate placental and cord blood supply, blood pressure and fetal development.
Effects of T cell activation in pregnant women with SARS‐CoV‐2 infection on fetuses
The activation of maternal T cells due to viral infection may lead to the disruption of fetal‐maternal tolerance and cause fetal death. Pregnancy bias towards T‐helper 2 (Th2) system dominance will protect the fetus, while activation of both T‐helper 1 (Th1) and Th2 immunity occurred during SARS‐CoV‐2 infection. 1 Predominantly Th1 responses in severe patient will be very harmful to fetus. Besides, viral infection can activate CD8+ T cells through cytotoxic T lymphocyte (CTL) epitopes in the virus protein, thus changing the responses of maternal CD8+ T cells and disrupting maternal‐fetal immune tolerance. 36 Indian researchers have identified 5 CTL epitopes in the surface glycoprotein of the SARS‐CoV‐2, suggesting that SARS‐CoV‐2 can activate CD8+ T cells. 37 This conjecture has been confirmed by the immune status of patients with SARS‐CoV‐2 infection, whose CD8+ T cells in peripheral blood are extremely reduced in number but are over activated, with high concentrations of cytotoxic particles. 9 Thus, the levels of activated T cells in pregnant women should be detected, and more measures should be taken to monitor fetal status than usual.
Maternal specific IgG may protect neonates against SARS‐CoV‐2
Virus‐specific IgG may protect newborns from infection while inhibiting the development of the neonatal immune system. The presence of SARS‐CoV antibodies in cord blood and breast milk suggests that infection with SARS‐CoV during pregnancy can lead to passive immunization. 38 Pathogen‐specific IgG can pass through the placenta as early as 12 weeks of gestation and exist in neonates for several months. In three neonates delivered by COVID‐19 mother, SARS‐CoV‐2 IgG levels were elevated and the RT‐PCR test was negative noting that antibody transmission through placental. 39 Convalescent plasma as a potential therapy for COVID‐19 suggesting the maternal antibodies may provide protective effects on newborns.
Summary
Because SARS‐CoV‐2 is a novel coronavirus, the potential impact of COVID‐19 on pregnant women and their newborns is still being determined based on the accumulated data. Based on the limited studies on COVID‐19 literature, we reveal the potential adverse effects after maternal SARS‐CoV‐2 infection on fetuses and newborns, including preterm delivery, respiratory dyspnea, intrauterine growth restriction, abnormal development of the respiratory and nervous systems and poor development of the immune system. The underlying mechanisms of these effects are hypoxemia, a decrease in coronavirus functional receptor content and immune‐related injuries caused by SARS‐CoV‐2 infection. We can conclude that SARS‐CoV‐2 infection not only affects pregnant women but also affects the development of fetuses and newborns, resulting in a variety of pregnancy complications and endangering the normal development of newborns until they reach adulthood. At present, the number of SARS‐CoV‐2 cases diagnosed worldwide continues to increase, and a large number of pregnant women are suffering from the disease and are in urgent need of more comprehensive and accurate treatment. This review will provide theoretical support and therapeutic direction.
Disclosure
None declared.
References
- 1. Huang C, Wang Y, Li X et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395: 497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhu N, Zhang D, Wang W et al. A novel coronavirus from patients with pneumonia in China 2019. N Engl J Med 2020; 382: 727–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Zhu H, Wang L, Fang C et al. Clinical analysis of 10 neonates born to mothers with SARS‐CoV‐2 pneumonia. Transl Pediatr 2020; 9: 51–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Di Mascio D, Khalil A, Saccone G et al. Outcome of coronavirus spectrum infections (SARS, MERS, COVID 1‐19) during pregnancy: A systematic review and meta‐analysis. Am J Obstet Gynecol MFM 2020; 2020: 100107. 10.1016/j.ajogmf.2020.100107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zaigham M, Andersson O. Maternal and perinatal outcomes with COVID‐19: A systematic review of 108 pregnancies. Acta Obstet Gynecol Scand 2020; 99: 823–829. 10.1111/aogs.13867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Dashraath P, Wong JLJ, Lim MXK et al. Coronavirus disease 2019 (COVID‐19) pandemic and pregnancy. Am J Obstet Gynecol 2020; 222: 521–531. 10.1016/j.ajog.2020.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Zhou P, Yang XL, Wang XG et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020; 579: 270–273. 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Hoffmann M, Kleine‐Weber H, Schroeder S et al. SARS‐CoV‐2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020; 181: 271–280.e8. 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xu Z, Shi L, Wang YJ, Zhang JY, Huang L, Zhang C, et al. Pathological findings of SARS‐CoV‐2 associated with acute respiratory distress syndrome. Lancet Respir Med 2020; 8: 420–422. 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Ng WF, Wong SF, Lam A et al. The placentas of patients with severe acute respiratory syndrome a pathophysiological evaluation. Pathology 2006; 38: 210–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chen S, Huang B, Luo DJ et al. Pregnant women with new coronavirus infection: A clinical characteristics and placental pathological analysis of three cases. Zhonghua Bing Li Xue Za Zhi 2020; 49: E005. 10.3760/cma.j.cn112151-20200225-00138. [DOI] [PubMed] [Google Scholar]
- 12. Bullo M, Tschumi S, Bucher BS, Bianchetti MG, Simonetti GD. Pregnancy outcome following exposure to angiotensin‐converting enzyme inhibitors or angiotensin receptor antagonists. Hypertension 2012; 60: 444–450. [DOI] [PubMed] [Google Scholar]
- 13. He J, Lu YP, Li J et al. Fetal but not maternal angiotensin converting enzyme (ACE)‐2 gene Rs2074192 polymorphism is associated with increased risk of being a small for gestational age (SGA) newborn. Kidney Blood Press Res 2018; 43: 1596–1606. [DOI] [PubMed] [Google Scholar]
- 14. Wang Z, Wang P, Wang X et al. Significant association between angiotensin‐converting enzyme gene insertion/deletion polymorphism and risk of recurrent miscarriage: A systematic review and meta‐analysis. Metabolism 2013; 62: 1227–1238. [DOI] [PubMed] [Google Scholar]
- 15. Li W, Moore MJ, Vasilieva N et al. Angiotensin‐converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003; 426: 450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Yockey LJ, Jurado KA, Arora N et al. Type I interferons instigate fetal demise after Zika virus infection. Sci Immunol 2018; 3(19): eaao1680. http://doi:10.1126/sciimmunol.aao1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Adams Waldorf KM, McAdams RM. Influence of infection during pregnancy on fetal development. Reproduction 2013; 146: R151–R162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wang Y, Liu L. The membrane protein of severe acute respiratory syndrome coronavirus functions as a novel cytosolic pathogen‐associated molecular pattern to promote Beta interferon induction via a toll‐like‐receptor‐related TRAF3‐independent mechanism. MBio 2016; 7: e01872–e01815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Yockey LJ, Iwasaki A. Role of interferons and cytokines in pregnancy and fetal development. Immunity 2018; 49: 397–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Sun D, Li H, Lu XX et al. Clinical features of severe pediatric patients with coronavirus disease 2019 in Wuhan: A single center's observational study. World J Pediatr 2020; 16: 251–259. 10.1007/s12519-020-00354-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Chen G, Wu D, Guo W et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest 2020; 130(5): 2620–2629. 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Channappanavar R, Perlman S. Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Semin Immunopathol 2017; 39: 529–539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Sadowsky DW, Adams KM, Gravett MG, Witkin SS, Novy MJ. Preterm labor is induced by intraamniotic infusions of interleukin‐1 b and tumor necrosis factor‐α but not by interleukin‐6 or interleukin‐8 in a nonhuman primate model. Am J Obstet Gynecol 2006; 195: 1578–1589. [DOI] [PubMed] [Google Scholar]
- 24. Robertson SA, Christiaens I, Dorian CL et al. Interleukin‐6 is an essential determinant of on‐time parturition in the mouse. Endocrinology 2010; 151: 3996–4006. [DOI] [PubMed] [Google Scholar]
- 25. Boyle AK, Rinaldi SF, Norman JE, Stock SJ. Preterm birth: Inflammation, fetal injury and treatment strategies. J Reprod Immunol 2017; 119: 62–66. [DOI] [PubMed] [Google Scholar]
- 26. Kim S, Kim H, Yim YS et al. Maternal gut bacteria promote neurodevelopmental abnormalities in mouse offspring. Nature 2017; 549: 528–832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Jones KL, Croen LA, Yoshida CK et al. Autism with intellectual disability is associated with increased levels of maternal cytokines and chemokines during gestation. Mol Psychiatry 2017; 22: 273–279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Estes ML, McAllister AK. Maternal immune activation: Implications for neuropsychiatric disorders. Science 2016; 353: 772–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Taubeneck MW, Daston GP, Rogers JM, Gershwin ME, Ansari A, Keen CL. Tumor necrosis factor‐alpha alters embryonic zinc metabolism and is developmentally toxic in mice. J Nutr 1995; 125: 908–919. [DOI] [PubMed] [Google Scholar]
- 30. Liu S, Diao L, Huang C, Li Y, Zeng Y, Kwak‐Kim JYH. The role of decidual immune cells on human pregnancy. J Reprod Immunol 2017; 124: 44–53. [DOI] [PubMed] [Google Scholar]
- 31. Carlino C, Stabile H, Morrone S et al. Recruitment of circulating NK cells through decidual tissues: A possible mechanism controlling NK cell accumulation in the uterus during early pregnancy. Blood 2008; 111: 3108–3115. [DOI] [PubMed] [Google Scholar]
- 32. Yang F, Zheng Q, Jin L. Dynamic function and composition changes of immune cells during normal and pathological pregnancy at the maternal‐fetal interface. Front Immunol 2019; 10: 2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Thaxton JE, Nevers T, Lippe EO, Blois SM, Saito S, Sharma S. NKG2D blockade inhibits poly (I:C)‐triggered fetal loss in wild type but not IL‐10−/− mice. J Immunol 2013; 190: 3639–3647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Fu B, Zhou Y, Ni X et al. Natural killer cells promote fetal development through the secretion of growth‐promoting factors article natural killer cells promote fetal development through the secretion of growth‐promoting factors. Immunity 2017; 47: 1100–1113. [DOI] [PubMed] [Google Scholar]
- 35. Wang F, Nie J, Wang H et al. Characteristics of peripheral lymphocyte subset alteration in COVID‐19 pneumonia. J Infect Dis 2020; 221: 1762–1769. 10.1093/infdis/jiaa150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Tilburgs T, Strominger JL. CD8+ effector T cells at the fetal‐maternal interface, balancing fetal tolerance and antiviral immunity. Am J Reprod Immunol 2013; 69: 395–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Baruah V, Bose S. Immunoinformatics‐aided identification of T cell and B cell epitopes in the surface glycoprotein of SARS‐CoV‐2. J Med Virol 2020; 92: 495–500. 10.1002/jmv.25698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Robertson CA, Lowther SA, Birch T, Coy K, Saw J, Parashar UD. SARS and pregnancy: A case report. Emerg Infect Dis 2004; 10: 345–348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Parazzini F, Bortolus R, Mauri PA et al. Delivery in pregnant women infected with SARS‐CoV‐2: A fast review. Int J Gynaecol Obstet 2020; 150: 41–46. 10.1002/ijgo.13166. [DOI] [PMC free article] [PubMed] [Google Scholar]


