Abstract
During one-lung ventilation (OLV), titrating the positive end-expiratory pressure (PEEP) to target a low driving pressure (∆P) could reduce postoperative pulmonary complications. However, it is unclear how to conduct PEEP titration: by stepwise increase starting from zero PEEP (PEEPINCREMENTAL) or by stepwise decrease after a lung recruiting manoeuvre (PEEPDECREMENTAL). In this randomized trial, we compared the physiological effects of these two PEEP titration strategies on respiratory mechanics, ventilation/perfusion mismatch and gas exchange. Patients undergoing video-assisted thoracoscopic surgery in OLV were randomly assigned to a PEEPINCREMENTAL or PEEPDECREMENTAL strategy to match the lowest ∆P. In the PEEPINCREMENTAL group, PEEP was stepwise titrated from ZEEP up to 16 cm H2O, whereas in the PEEPDECREMENTAL group PEEP was decrementally titrated, starting from 16 cm H2O, immediately after a lung recruiting manoeuvre. Respiratory mechanics, ventilation/perfusion mismatch and blood gas analyses were recorded at baseline, after PEEP titration and at the end of surgery. Sixty patients were included in the study. After PEEP titration, shunt decreased similarly in both groups, from 50 [39–55]% to 35 [28–42]% in the PEEPINCREMENTAL and from 45 [37–58]% to 33 [25–45]% in the PEEPDECREMENTAL group (both p < 0.001 vs baseline). The resulting ∆P, however, was lower in the PEEPDECREMENTAL than in the PEEPINCREMENTAL group (8 [7–11] vs 10 [9–11] cm H2O; p = 0.03). In the PEEPDECREMENTAL group the PaO2/ FIO2 ratio increased significantly after intervention (from 140 [99–176] to 186 [152–243], p < 0.001). Both the PEEPINCREMENTAL and the PEEPDECREMENTAL strategies were able to decrease intraoperative shunt, but only PEEPDECREMENTAL improved oxygenation and lowered intraoperative ΔP.
Clinical trial number NCT03635281; August 2018; “retrospectively registered”
Electronic supplementary material
The online version of this article (10.1007/s10877-020-00582-z) contains supplementary material, which is available to authorized users.
Keywords: Positive end-expiratory pressure, One-lung ventilation, Shunt, Driving pressure, Oxygenation
Introduction
During thoracic surgery with one lung ventilation (OLV), application of positive end-expiratory pressure (PEEP) improves gas exchange and lung mechanics [1–6]. However, the approach to PEEP titration remains controversial. Recently, we documented the highly variable impact of different PEEP levels on alveolar recruitment and gas exchange [2]; our results confirmed the expert’s opinions which suggest to personalize PEEP level to balance alveolar recruitment and hyperinflation [3]. Recently, PEEP titration to the “lowest” driving pressure (ΔP), i.e. the difference between inspiratory plateau pressure and total PEEP, has gained a central role in the scientific debate [3–6]. Park et al. found a lower rate of postoperative pulmonary complications (PPC) in patients submitted to an incremental PEEP titration to the lowest ΔP [4]. Conversely, other studies suggest an open lung approach based on a decremental PEEP trial subsequent a lung recruitment manoeuvre (LRM) [7–10].
To our knowledge, physiological trials comparing the incremental versus the decremental ΔP-oriented PEEP titration during OLV are lacking.
In this study, we randomly assigned patients scheduled for video-assisted thoracic surgery in OLV to an incremental PEEP (PEEPINCREMENTAL) versus an open lung approach with decremental PEEP titration (PEEPDECREMENTAL). Our hypothesis was that the PEEPDECREMENTAL approach would result in more improvement in the respiratory mechanics, ventilation/perfusion mismatch and gas exchange compared to the PEEPINCREMENTAL technique.
Methods
The trial was approved by the Ethics Committee of our institution (protocol N.11072017) and written informed consent was obtained from each patient before surgery. The trial was registered in Clinicaltrial.gov (NCT03635281). The study was performed in the Department of Anaesthesia and Intensive Care at the University Hospital of Ferrara (Italy) from August 2017 to October 2018. Results are reported according to the Consolidated Standards of Reporting Trials (CONSORT) checklist for randomized trials. The CONSORT Checklist is reported in Supplement file 1.
Population
We screened all patients scheduled for video-assisted thoracoscopic surgery (i.e. lobectomy or wedge resection) requiring lateral position and OLV for at least 2 h. Exclusion criteria were: ASA (American Society of Anesthesiologists Physical Status Classification) score ≥ 4, severe chronic respiratory failure (chronic obstructive pulmonary disease patients with Global Initiative for Chronic Obstructive Lung Disease stage 3 or 4), preoperative haemoglobin less than 10 g dl−1, hemodynamic instability during LRM defined as a decrease in systolic arterial pressure of more than 20% from baseline, and unplanned thoracotomy conversion.
The day before the procedure, all patients underwent spirometry in sitting position according to the American Thoracic Society’s standards (SpiroPro; Jaeger, Germany). Spirometry measurements included vital capacity, forced expiratory volume in the 1st second (FEV1), forced vital capacity (FVC), expiratory reserve volume (ERV), and transfer coefficient (KCO).
Anesthesia
Anaesthesia was induced with propofol (1.5 to 2 mg kg−1), fentanyl (3 μg kg−1), and rocuronium (0.6 mg kg−1). Target-controlled propofol infusion was performed with estimated effect-site concentration of 2–4 μg ml−1, targeting a bispectral index (Aspect A-2000; Aspect Medical System, USA) of 40–60. Neuromuscular blockade was provided with continuous infusion of rocuronium based on train-of-four neuromuscular monitoring. All patients were breathing 80% oxygen during induction of general anaesthesia. The trachea was intubated with an appropriately sized double lumen tube whose correct positioning was bronchoscopy confirmed. Ultrasound-guided thoracic paravertebral blocks were performed in lateral decubitus with two injections of 8 ml Ropivacaine 0.75% [11]. Appropriate spread of local anaesthetic was confirmed with the movement of the pleura. Patients were ventilated in volume-controlled mode with constant flow using a Dräger Perseus ventilator (Drägerwerk AG and Co. KGaA, Germany). During two-lung (bilateral) ventilation, TV was set to 7 ml kg−1 predicted body weight (PBW) and PEEP was set to zero. These settings were maintained for approximately 10 min before shifting to OLV. When OLV started, TV was reduced to 5 ml kg−1 (PBW) and PEEP was initially maintained unchanged. PEEP was then adjusted according to the study group (see below). PEEP was applied according to the study group (see below). FiO2 was set to maintain peripheral oxygen saturation (SpO2) equal to or greater than 92% while respiratory rate was adjusted to keep arterial PaCO2 between 40 and 60 mmHg.
Randomization and study intervention
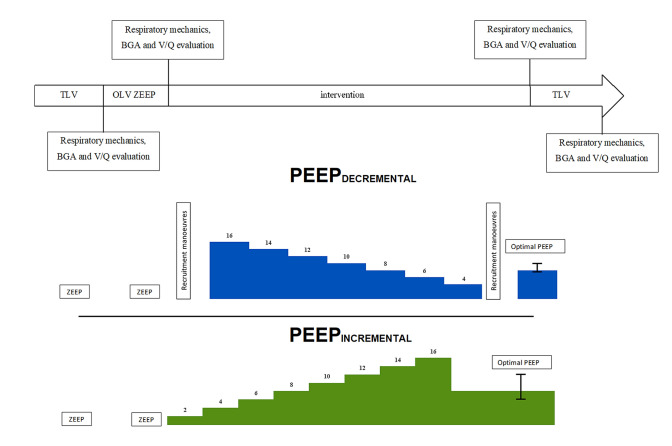
After inclusion in the study, patients were randomized to the PEEPINCREMENTAL or a PEEPDECREMENTAL protocol. Randomization was performed 1:1 using randomization in block sizes of 2 and 4. The time-course of the study is summarized in Fig. 1.
Fig. 1.
Resume of study protocol
The PEEPINCREMENTAL protocol followed the strategy proposed by Park and coworkers [4]; whereas the PEEPDECREMENTAL protocol followed the strategy proposed by the ongoing “protective ventilation with high versus low positive end-expiratory pressure during one-lung ventilation for thoracic surgery” (PROTHOR) trial [12].
In the PEEPINCREMENTAL group, PEEP was increased stepwise by 2 cm H2O steps, from ZEEP up to 16 cm H2O, while maintaining TV and RR constant. Each PEEP level was kept for 1 min before measuring ∆P. At the end of the PEEP titration trial, the “best” PEEP defined as the level associated with lowest ∆P, was set and maintained until extubation. (see Supplement File 1 for details).
Patients in PEEPDECREMENTAL group were submitted to a LRM immediately followed by a decremental PEEP trial. As part of the LRM, respiratory rate was set to 6 min−1 with an inspiratory–expiratory ratio of 1:1. TV was then increased in steps of 2 mL kg−1 PBW until reaching a target plateau pressure of 30 cm H2O. The step increase in TV was performed over a period of 20–30 s. Three breaths were allowed at the target plateau pressure (see Supplement File 2 for details). After performing the LRM, TV and respiratory rate were returned to the starting values, while PEEP was set to 16 cm H2O. Then PEEP was decreased in steps of 2 cm H2O, down to 4 cm H2O, in order to identify the “best” (i.e. the lowest) ΔP. Each level of PEEP was maintained for 1 min before measuring ∆P. Subsequently, another LRM, analogous to the first one, was performed and PEEP was set to the “best” PEEP level identified during the decremental PEEP trial and maintained until extubation. Safety-endpoints for interruption of the LRM were a ± 20% variation in heart rate or a decrease of more than 20% of mean arterial pressure [13].
Lung mechanics and ventilation/perfusion assessment
Respiratory mechanics were assessed by the constant V′/rapid occlusion method previously described in details [2]. The end-inspiratory plateau pressure was measured as the airway pressure (Paw) at the end of an end-inspiratory occlusion performed by increasing end-inspiratory pause to 40% of the inspiratory time. Driving pressure (ΔP) was calculated as plateau pressure—PEEP; static respiratory system compliance was calculated as TV/(end-inspiratory plateau pressure—PEEP). Mechanical power (J/min) at each time-point was calculated as described by Gattinoni et al. with the following formula [14]:
where RR is respiratory rate, ΔV is tidal volume, ELrs is respiratory system elastance and Raw is airway resistance [14, 15].
Total energy load (J) was calculated as the product of power and ventilation time.
Shunt and V/Q matching were assessed by the Beacon Caresystem (Mermaid Care A/S, Denmark) in two-lung ventilation (TLV), OLV before intervention, OLV after intervention and in TLV at the end of the surgery. Briefly, to assess V/Q matching, the automatic lung parameter estimator (ALPE) approach [16, 17] used in the Beacon Caresystem, requires modification of FIO2 in three or four steps in a process taking 5–10 min. At each FIO2 level, steady state is identified and measurements are automatically taken of ventilation, SpO2, O2 consumption, CO2 production, and inspiratory and expiratory fractions of O2 and CO2. Oxygenation at the various FIO2 levels are used to estimate shunt and low V/Q mismatch whereas end-tidal to arterial CO2 gradient is used to calculate high V/Q mismatch. Low V/Q mismatch is reported as the O2 partial pressure difference between alveolar air and lung capillary blood prior to mixing with shunted venous blood, thus quantifying the primary effect of low V/Q on O2 exchange. High V/Q mismatch is reported as the CO2 partial pressure difference between alveolar air and lung capillary blood prior to mixing with shunted venous blood quantifying the primary effect of high V/Q on CO2 exchange. The ALPE approach has been validated and applied in varied patient populations [1, 18–20] including patients undergoing OLV [2].
Statistical analysis
All analyses were pre-planned, unless specified as post-hoc. Normal distribution was tested by the Shapiro–Wilk normality test. Data are reported as mean ± SD or median [interquartile range] as appropriate. Differences between measurements were analysed using repeated measures ANOVA or Friedman’s rank analysis for data with normal or not normal distribution, respectively. When multiple comparisons were made, p-values were adjusted by the Bonferroni post hoc procedure. Two-tailed statistical hypothesis testing was performed with a p value of ≤ 0.05 considered statistically significant. Statistical analysis was performed using SPSS Statistics for Windows, version 20.0 (IBM, USA).
Several post-hoc analyses were performed. Firstly, we investigated whether the effect of PEEPINCREMENTAL and PEEPDECREMENTAL strategies on the ∆P and PaO2/FIO2 could be influenced by baseline ∆P values. To perform this analysis, we divided the population according to a baseline ∆P, either ≤ 14 cm H2O or > 14 cm H2O; this cut-off was derived from previous studies performed both in critically ill patients [21] and in one lung ventilation [2]. Furthermore, we investigated whether baseline comorbidities of the patients could influence the response to the two ventilation strategies. With this purpose, we analysed patients stratified for age, body mass index (BMI) or history of COPD.
Sample size
The sample size was based on the impact of PEEP titration on the shunt fraction (compared to OLV at ZEEP). Based on previous data, we assumed a decrease in shunt of at least 7% in the PEEPDECREMENTAL group [22] and at of least 2% in PEEPINCREMENTAL group [2], with a 6% pooled standard deviation. According to this analysis, including 31 patients in each group was deemed enough to demonstrate a significant difference between the two groups, with an 90% power and a 5% alfa error. Assuming a 5% loss to follow up, for example for unplanned conversion to thoracotomy surgery or intraoperative hemodynamic instability, we planned to enrol 65 patients.
Results
Population
During the study period, 66 patients were screened for eligibility. Of those, 63 met the inclusion criteria and were included in the study; three (3) patients underwent unplanned thoracotomy conversion, leaving 60 patients for final analysis. There were no missing data or protocol deviation during the study. None of the patients analysed were excluded due to intraoperative hemodynamic instability. Preoperative clinical and demographical characteristics of the patients are presented in Table 1. The flowchart of the study is shown in Figure S1.
Table 1.
Characteristic’s patients
| Variable | All patients n = 60 |
PEEPINCREMENTAL n = 30 |
PEEPDECREMENTAL n = 30 |
p value |
|---|---|---|---|---|
| Age | 68 ± 9 | 66 ± 8 | 67 ± 9 | 0.571 |
| BMI | 27.5 ± 5.5 | 27.0 ± 6.2 | 27.8 ± 5.5 | 0.576 |
| ASA score | 0.506 | |||
| II | 11 | 4 | 7 | |
| III | 49 | 26 | 23 | |
| Sex (M/F), n | 40/20 | 18/12 | 22/8 | 0.411 |
| Surgery side (L/R) | 29/31 | 13/17 | 15/15 | 0.312 |
| Type of surgery | 0.061 | |||
| Lobectomy | 55 | 30 | 25 | |
| Wedge resection | 5 | – | 5 | |
| Duration of MV (min) | 212 [175–255] | 218 [195–255] | 205 [175–255] | 0.57 |
| Duration of OLV (min) | 192 [166–240] | 195 [180–240] | 190 [145–240] | 0.49 |
| Comorbidities | ||||
| Diabetes, n (%) | 8 (13) | 4 (13) | 4 (13) | 0.999 |
| Hypertension, n (%) | 35 (50) | 10 (33) | 16 (53) | 0.192 |
| Vascular disease, n (%) | 20 (33) | 10 (33) | 10 (33) | 0.999 |
| COPD, n (%) | 10 (16) | 6 (20) | 4 (13) | 0.731 |
| Preoperative spirometry | ||||
| FVC (% predicted) | 104 [93–117] | 99 [79–117] | 107 [96–118] | 0.204 |
| FEV1 (%) | 96 [79–109] | 90 [70–105] | 105 [86–116] | 0.061 |
| FEV1/FVC | 75 ± 12 | 77 ± 16 | 74 ± 9 | 0.75 |
| KCO (% predicted) | 74 [62–93] | 69 [58–80] | 86 [67–98] | 0.086 |
| ERV (% predicted) |
BMI body mass index, MRC Medical Research Council Scale, MV mechanical ventilation, OLV one lung ventilation, VC vital capacity, FEV1 forced expiratory volume in the 1st second, FVC forced vital capacity, MEF maximal expiratory flow, ERV expiratory reserve volume
p values are referred to comparison between PEEPINCREMENTAL and PEEPDECREMENTAL group
V/Q mismatch
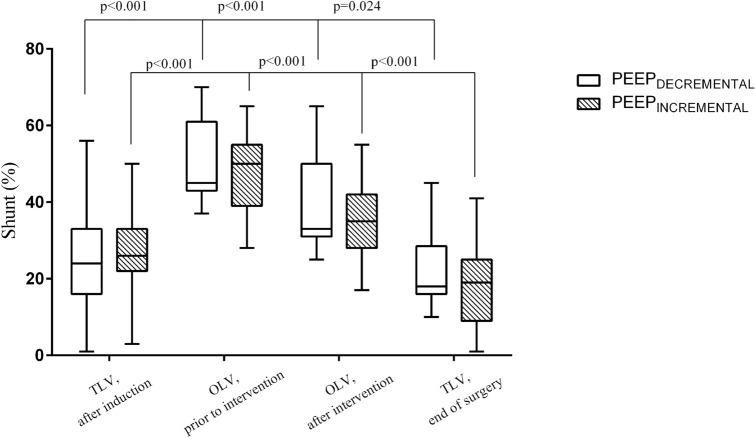
After anaesthesia induction, shunt was 25 [19–33] % in TLV and increased to 47 [39–56] % (p < 0.001) when patients were posed in lateral decubitus and ventilated in OLV at ZEEP. After PEEP titration, shunt decreased similarly in both groups (Table 2, Fig. 2), from 50 [39–55]% to 35 [28–42]% in the PEEPINCREMENTAL group (mean difference 20 [95% CI 16–25]%; p < 0.001 vs baseline) and from 45 [37–58] % to 33 [25–45]% in the PEEPDECREMENTAL group, (mean difference 22 [95% CI 18–26]%; p < 0.001 vs baseline). There was a reduction in shunt after intervention in 77% (23/30) of the patients in the PEEPINCREMENTAL versus 90% (27/30) in the PEEPDECREMENTAL group (p = 0.17 for comparison among groups).
Table 2.
Mechanical ventilation variables and V̇/Q̇ measurement during the study period
| Variable | TLV, after induction | OLV, prior to intervention | OLV, after intervention | TLV, End of surgery | ||||
|---|---|---|---|---|---|---|---|---|
| PEEPINCREMENTAL | PEEPDECREMENTAL | PEEPINCREMENTAL | PEEPDECREMENTAL | PEEPINCREMENTAL | PEEPDECREMENTAL | PEEPINCREMENTAL | PEEPDECREMENTAL | |
| Mechanical ventilation | ||||||||
| Paw (cm H2O) | 20 [15–23] | 18 [14–20.5] | 23 [19.5–27.5] | 22 [20–26] | 25.5 [22.2–30] | 24 [23–29] | 24.5 [20.7–28.2] | 18 [14–20.5] |
| Plateau pressure (cm H2O) | 13 [10–15] | 13 [10–15] | 15 [14–19] | 15 [14–19] | 19 [17–22] | 17.5 [15–21] | 19 [16–22] | 18 [15–20] |
| Driving Pressure (cm H2O) | 13 [10–15] | 12 [10–16] | 15 [14–19] | 14 [13–19] | 10 [9–11] | 8 [7–11]#* | 10 [8–12] | 9 [7–12] |
| PEEP (cm H2O) | 0 | 0 | 0 | 0 | 8 [6–12] | 8 [8–10] | 8 [6–12] | 8 [8–10] |
| Tidal volume | 440 [385–500] | 440 [400–490] | 315 [270–350] | 310 [290–350] | 315 [270–350] | 310 [290–350] | 440 [390–500] | 440 [400–490] |
| Respiratory rate | 14 [14–15] | 14 [13–15] | 16 [14–16] | 16 [14–16] | 16 [14.2–17.5] | 16 [16–18] | 16 [14–16] | 16 [14–17] |
| Mechanical power (J/m) | 10.8 [8.6–12.4] | 9.5 [8.1–12.7] | 5.6 [4.5–6.8] | 5.8 [4.4–6.6] | 6.8 [5.4–8.8]* | 7.0 [5.9–8.6]* | 12.8 [10.0–16.1] | 13.3 [11.4–15.0] |
| V/Q̇ variables | ||||||||
| Shunt (%) | 26.2 [21.8–32.9] | 24.0 [16.1–33.1] | 49.8 [39.0–55.0] | 45.2 [37.5–58.1] | 34.9 [28.0–42.1]* | 33.1 [24.7–45.1]* | 18.6 [9.0–25.2] | 18.5 [9.8–22.6] |
| Low V̇/Q̇ (mmHg) | 39 [17–65] | 35 [23–71] | 86 [37–150] | 69 [23–144] | 55 [38–75] | 57 [34–101] | 28 [22–70] | 35 [24–72] |
| High V̇/Q̇ (mmHg) | 11 [6–15] | 11 [6–15] | 13 [10–16] | 11 [7–15] | 13 [11–18] | 13 [6.7–17] | 12 [10–16] | 12 [7–17] |
Paw Peak airway pressure, PEEP positive end-expiratory pressure; V̇/Q̇ = Ventilation/perfusion ratio
#p < 0.05 vs the other group, *p < 0.05 vs prior to intervention
Fig. 2.
Shunt evaluation during the study in the PEEPINCREMENTAL and PEEPDECREMENTAL group
Low V/Q decreased from 86 [37–150] mmHg to 55 [38–75] mmHg in the PEEPINCREMENTAL group (p = 0.14 vs baseline) and from 69 [23–144] mmHg to 57 [34–101] mmHg in the PEEPDECREMENTAL group (p = 0.58 vs baseline).
High V/Q mismatch was similar among the two groups. (Table 2).
Respiratory mechanics and gas exchange
During the OLV pre-intervention (at ZEEP), the ∆P did not differ between the two groups, with observations of 15 [14–19] cm H2O for the PEEPINCREMENTAL and 14 [13–19] cm H2O for the PEEPDECREMENTAL group. The PEEP titration procedure resulted in a similar “optimal” median level in the two groups (PEEPINCREMENTAL: 8 [6–12] cm H2O, range 4–16 cm H2O; PEEPDECREMENTAL: 8 [8–10] cm H2O, range 6–12 cm H2O; p = 0.74 for comparison) (Table 2, Fig. 3). Nevertheless, the ∆P was 10 [9–11] cm H2O in the PEEPINCREMENTAL and 8 [7–11] cm H2O in the PEEPDECREMENTAL group (p = 0.03; Table 2).
Fig. 3.
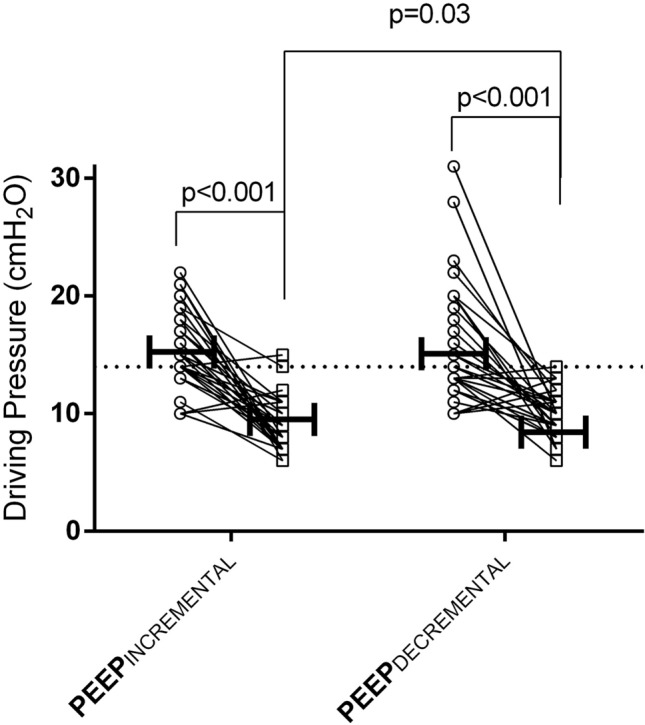
Individual changes in driving pressure before and after the study intervention in the PEEPINCREMENTAL and PEEPDECREMENTAL group
The energy load applied during the LRM in the PEEPDECREMENTAL group was 5.8 [4.7–7.1] J. The total energy load in the OLV post-intervention step was 1.175 [0.920–1.791] J in the PEEPINCREMENTAL and 1.484 [1.091–2.054] J in PEEPDECREMENTAL group (p = 0.25)].
Only the patients in the PEEPDECREMENTAL group experienced a significant increase in the PaO2/FIO2 ratio after intervention (from 140 [99–176] to 186 [152–243], p < 0.001). In the PEEPDECREMENTAL group the PaO2/FIO2 ratio increased significantly after intervention. There were no significant changes in PaCO2 before or after intervention in both groups (Table 3).
Table 3.
Blood gas analysis during the study period
| Variable | TLV, after induction | OLV, prior to intervention | OLV, after intervention | TLV, End of surgery | ||||
|---|---|---|---|---|---|---|---|---|
| PEEPINCREMENTAL | PEEPDECREMENTAL | PEEPINCREMENTAL | PEEPDECREMENTAL | PEEPINCREMENTAL | PEEPDECREMENTAL | PEEPINCREMENTAL | PEEPDECREMENTAL | |
| PaO2/FIO2 ratio (mmHg) | 350 [238–438] | 298 [219–447] | 139 [103–202] | 140 [99–176] | 153 [103–192] | 186 [152–243] #* | 423 [241–492] | 402 [262–463] |
| FIO2 | 50 [40–55] | 40 [40–50] | 58 [49–66] | 55 [49–60] | 65 [56–73]* | 50 [40–52]#* | 57 [50–65] | 50 [44–60] |
| PaCO2 (mmHg) | 47 [42–51] | 38 [42–52] | 55 [50–59] | 53 [49–60] | 59 [54–62] | 54 [51–64] | 49 [45–52] | 50 [43–53] |
| pH | 7.36 ± 0.05 | 7.36 ± 0.05 | 7.31 ± 0.05 | 7.31 ± 0.07 | 7.27 ± 0.05* | 7.28 ± 0.06* | 7.31 ± 0.04 | 7.31 ± 0.06 |
| HCO3− | 26.9 ± 2.6 | 26.5 ± 2.5 | 27.0 ± 1.8 | 26.7 ± 2.3 | 26.4 ± 1.7 | 26.3 ± 2.5 | 24.7 ± 1.9 | 24.4 ± 2.3 |
| Lactate (mmol/L) | 1 [0.7–1.2] | 0.8 [0.7–1.2] | 0.8 [0.7–1.2] | 0.8 [0.7–1.1] | 0.8 [0.6–1] | 0.8 [0.7–1.1] | 0.9 [0.7–1.0] | 0.8 [0.7–1.0] |
| Hb (g/dL) | 12.6 ± 1.8 | 12.4 ± 1.6 | 12.1 ± 1.6 | 12.0 ± 1.6 | 12.1 ± 1.7 | 11.9 ± 1.6 | 12.0 ± 1.9 | 11.5 ± 1.7 |
PaCO2 arterial partial pressure of carbon dioxide, PaO2 arterial partial pressure of oxygen, FIO2 fraction of inspired oxygen, Hb hemoglobin
#p < 0.05 vs the other group, *p < 0.05 vs prior to intervention
Perioperative assessment
Intraoperative hemodynamic variables, as well as the number and kind of postoperative complications, are shown in Supplement Table 3. Patients in the two groups did not differ in terms of intraoperative management.
Post-hoc subgroups analysis
We performed various sub-group analyses to investigate whether different responses to the two PEEP titration strategies could be influenced by different clinical characteristics. No difference was found in PEEPINCREMENTAL or PEEPDECREMENTAL strategies when stratifying patients for age, BMI or history of COPD (Supplemental Fig. 2).
Among patients with ∆P > 14 cm H2O prior to intervention (n = 34), the reduction in ∆P was more pronounced in the PEEPDECREMENTAL group (from 19 [17–22] cm H2O to 10 [8–11] cm H2O) than in the PEEPINCREMENTAL one (from 18 [15–20] cm H2O to 10 [9–12] cm H2O), p = 0.02 for group comparison in favour of PEEPDECREMENTAL group).
Discussion
The main result of our study is that, among patients undergoing video-assisted thoracic surgery in OLV, an open lung approach strategy based on low TV combined with a ΔP-oriented decremental PEEP titration resulted in lower values of driving pressure and increase in oxygenation when compared to a PEEPINCREMENTAL strategy. However, both strategies were equally able to reduce shunt and driving pressure at the end of surgery.
Individualized PEEP has been shown to improve regional ventilation distribution and oxygenation and to decrease the incidence of postoperative atelectasis and PPCs in both abdominal [23] and thoracic surgery [4]. On the other hand, high intraoperative driving pressure has been found to be an independent predictor of PPCs [5, 6]. Combining these concepts, a ∆P-oriented PEEP setting has been recently proposed for patient undergoing OLV during thoracic surgery. However, there are at least two different approaches to PEEP setting in this context: the incremental or the decremental approach. We found that both approaches were able to reduce ∆P to “safe” levels [6, 21, 24], and that, surprisingly the PEEP levels needed to minimize the ∆P were similar in both groups (Table 2). However, the PEEPDECREMENTAL strategy resulted in the lowest intraoperative ∆P. These results gain clinical relevance due to the described relationship between intraoperative ∆P and postoperative outcomes [5, 6]. Our data confirm the results of recent studies on the effects of the open lung approach applied during OLV [8]. Rauseo and coworkers found that a PEEPDECREMENTAL strategy was able to decrease transpulmonary driving pressure and to improve oxygenation [8] and Ferrando and co-workers showed that a PEEPDECREMENTAL was able to preserve the improvement in static compliance obtained through a LRM [7]. This could explain our findings of a lower ∆P and of an improvement in oxygenation in the PEEPDECREMENTAL group. We hope that our physiological data could help to interpret the results of the clinical studies on PEEP setting during OLV. Indeed, in our study we reproduced the protocols of two recent randomized controlled trials, the Park study [4] and the ongoing PROTHOR trial [12].
One major concern is that the PEEPDECREMENTAL approach could critically decrease patient’s cardiac output through both a preload and an afterload effect of the LRM needed to recruit the lungs before PEEP titration. However, the hemodynamic impact of the LRM during OLV has been previously shown to be mostly negligible [7, 8, 25, 26]. Some authors advocated the risk that the high amount of energy delivered to the lungs could result in alveolar hyperinflation and thus in a sort of unconscious “harmful” strategy [27, 28]. Nonetheless, in our patients we found that the mechanical power applied during the LRM was 5.8 [4.7–7.1] J, considerably lower than the “harmful” threshold of 25 J/min Joules suggested in patients with ARDS [29]. Additionally, the high V/Q fraction, a suitable surrogate of hyperinflation, was similar between the two groups (Table 2),
We also assessed the differential effects of the two strategies on the cohort of patients with higher baseline ΔP (i.e. higher than 14 cm H2O) during OLV. This sub-group analysis showed that these patients had a greater decrease in ∆P and increase in PaO2/FIO2 ratio when randomized to the PEEPDECREMENTAL group as compared to the PEEPINCREMENTAL one. Thus, despite our data should be extrapolated to the clinical context with caution, we speculate that patients with more compromised oxygenation and lung mechanics could be the best candidate to the PEEPDECREMENTAL strategy.
Our study has some limitations. First of all, our results could have been influenced by the effects of the LRM, which was performed only in the PEEPDECREMENTAL group. However, we would like to point out that the LRM is part of the open lung approach [30] and thus it is impossible to differentiate the role of PEEP and LRM in our PEEPDECREMENTAL group. On the other hand, since we were interested in reproducing the PEEPINCREMENTAL strategy proposed in the Park’s clinical trial [4], we did not apply any LRM in the PEEPINCREMENTAL group. Secondly, our study was not powered to investigate clinical outcomes of such PPCs. Thirdly, we did not record advanced hemodynamic parameters and, thus, we cannot report on the differential impact of the two strategies. However, previous studies have shown that both PEEP and LRM have slight and transient effects on cardiac output during OLV [25, 26]. Lastly, we used the PaO2/FIO2 ratio as index of oxygenation but applied a fixed FIO2 in the two groups, and this could have partially influenced our results [20].
In conclusion, we have shown the beneficial physiological effects of two ∆P-oriented PEEP titration strategies during OLV. According to our data, as compared with an incremental PEEP titration approach, decremental PEEP titration immediately after a LRM was more effective in decreasing ∆P and improving oxygenation, particularly in patients with higher (i.e. > than 14 cm H2O) intraoperative ∆P.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file1 (JPG 173 kb)
Supplemental Figure S1. Flowchart of the study
Supplementary file2 (TIF 105 kb)
Supplemental Figure S2. Forest plot comparing the effect of PEEPINCREMENTAL or PEEPDECREMENTAL strategies on driving pressure when stratifying patients for age, BMI or history of COPD
Author contributions
SS: This author helped to the design of the study, analyzed the data and wrote the paper. GS: This author helped to the design of the study, analyzed the data and wrote the paper. KDS: This author helped to revise and wrote the paper. SG: This author helped to collected the data. CG: This author helped to revise and wrote the paper. MP: This author helped to collect data and revise the paper. MF: This author helped to revise the paper. RDM: This author helped to revise the paper. RR: This author helped to collect data and revise the paper. RSE: This author helped to revise and wrote the paper. VCA: This author helped to the design of the study, analyzed the data and wrote the paper.
Funding
Support was provided solely from institutional and/or departmental sources.
Compliance with ethical standards
Conflict of interest
None of the authors received compensation to perform this study. Dr. Rees is a board member and minor shareholder of Mermaid Care A/S (Nørresundby, Denmark), who commercially produces the ALPE system. Dr. Karbing has performed consultancy work for Mermaid Care A/S. The remaining authors declare no competing interests.
Ethical approval
The trial was approved by the Ethics Committee of our institution (protocol No. 11072017).
Informed consent
Informed consent was obtained from each patient before surgery.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Spadaro Savino and Grasso Salvatore have contributed equally to this work.
References
- 1.Spadaro S, Karbing DS, Mauri T, et al. Effect of positive end-expiratory pressure on pulmonary shunt and dynamic compliance during abdominal surgery. Br J Anaesth. 2016;116(6):855–861. doi: 10.1093/bja/aew123. [DOI] [PubMed] [Google Scholar]
- 2.Spadaro S, Grasso S, Karbing DS, et al. Physiologic evaluation of ventilation perfusion mismatch and respiratory mechanics at different positive end-expiratory pressure in patients undergoing protective one-lung ventilation. Anesthesiology. 2018;128(3):531–538. doi: 10.1097/ALN.0000000000002011. [DOI] [PubMed] [Google Scholar]
- 3.Young CC, Harris EM, Vacchiano C, et al. Lung-protective ventilation for the surgical patient: international expert panel-based consensus recommendations. Br J Anaesth. 2019;123(6):898–913. doi: 10.1016/j.bja.2019.08.017. [DOI] [PubMed] [Google Scholar]
- 4.Park M, Ahn HJ, Kim JA, et al. Driving pressure during thoracic surgery: a randomized clinical trial. Anesthesiology. 2019;130(3):385–393. doi: 10.1097/ALN.0000000000002600. [DOI] [PubMed] [Google Scholar]
- 5.Blank RS, Colquhoun DA, Durieux ME, et al. Management of one-lung ventilation: impact of tidal volume on complications after thoracic surgery. Anesthesiology. 2016;124(6):1286–1295. doi: 10.1097/ALN.0000000000001100. [DOI] [PubMed] [Google Scholar]
- 6.Neto AS, Hemmes SN, Barbas CS, et al. Association between driving pressure and development of postoperative pulmonary complications in patients undergoing mechanical ventilation for general anaesthesia: a meta-analysis of individual patient data. Lancet Respir Med. 2016;4(4):272–280. doi: 10.1016/S2213-2600(16)00057-6. [DOI] [PubMed] [Google Scholar]
- 7.Ferrando C, Mugarra A, Gutierrez A, et al. Setting individualized positive end-expiratory pressure level with a positive end-expiratory pressure decrement trial after a recruitment maneuver improves oxygenation and lung mechanics during one-lung ventilation. Anesth Analg. 2014;118(3):657–665. doi: 10.1213/ANE.0000000000000105. [DOI] [PubMed] [Google Scholar]
- 8.Rauseo M, Mirabella L, Grasso S, et al. Peep titration based on the open lung approach during one lung ventilation in thoracic surgery: a physiological study. BMC Anesthesiol. 2018;18(1):156. doi: 10.1186/s12871-018-0624-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Girgis K, Hamed H, Khater Y, Kacmarek RA. decremental PEEP trial identifies the PEEP level that maintains oxygenation after lung recruitment. Respir Care. 2006;51(10):1132–1139. [PubMed] [Google Scholar]
- 10.Gernoth C, Wagner G, Pelosi P, Luecke T. Respiratory and haemodynamic changes during decremental open lung positive end-expiratory pressure titration in patients with acute respiratory distress syndrome. Crit Care. 2009;13(2):R59. doi: 10.1186/cc7786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Batchelor TJP, Rasburn NJ, Abdelnour-Berchtold E, et al. Guidelines for enhanced recovery after lung surgery: recommendations of the Enhanced Recovery After Surgery (ERAS®) Soc Eur Soc Thorac Surg (ESTS). 2019;55(1):91–115. doi: 10.1093/ejcts/ezy301. [DOI] [PubMed] [Google Scholar]
- 12.Kiss T, Wittenstein J, Becker C, et al. Protective ventilation with high versus low positive end-expiratory pressure during one-lung ventilation for thoracic surgery (PROTHOR): study protocol for a randomized controlled trial. Trials. 2019;20(1):213. doi: 10.1186/s13063-019-3208-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Villagra A, Ochagavia A, Vatua S, et al. Recruitment maneuvers during lung protective ventilation in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2002;165:165–170. doi: 10.1164/ajrccm.165.2.2104092. [DOI] [PubMed] [Google Scholar]
- 14.Gattinoni L, Tonetti T, Cressoni M, et al. Ventilator-related causes of lung injury: the mechanical power. Intensive Care Med. 2016;42(10):1567–1575. doi: 10.1007/s00134-016-4505-2. [DOI] [PubMed] [Google Scholar]
- 15.Spadaro S, Caramori G, Rizzuto C, et al. Expiratory flow limitation as a risk factor for pulmonary complications after major abdominal surgery. Anesth Analg. 2017;124(2):524–530. doi: 10.1213/ANE.0000000000001424. [DOI] [PubMed] [Google Scholar]
- 16.Rees SE, Kjærgaard S, Thorgaard P, Malczynski J, Toft E, Andreassen S. The Automatic Lung Parameter Estimator (ALPE) system: non-invasive estimation of pulmonary gas exchange parameters in 10–15 minutes. J Clin Monit Comput. 2002;17:43–52. doi: 10.1023/A:1015456818195. [DOI] [PubMed] [Google Scholar]
- 17.Karbing DS, Kjærgaard S, Andreassen S, Espersen K, Rees SE. Minimal model quantification of pulmonary gas exchange in intensive care patients. Med Eng Phys. 2011;33:240–248. doi: 10.1016/j.medengphy.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 18.Kjaergaard S, Rees S, Malczynski J, Nielsen JA, Thorgaard P, Toft E, Andreassen S. Non-invasive estimation of shunt and ventilation-perfusion mismatch. Intensive Care Med. 2003;29(5):727–734. doi: 10.1007/s00134-003-1708-0. [DOI] [PubMed] [Google Scholar]
- 19.Kjaergaard S, Rees SE, Grønlund J, et al. Hypoxaemia after cardiac surgery: clinical application of a model of pulmonary gas exchange. Eur J Anaesthesiol. 2004;21(4):296–301. doi: 10.1097/00003643-200404000-00008. [DOI] [PubMed] [Google Scholar]
- 20.Karbing DS, Kjaergaard S, Smith BW, et al. Variation in the PaO2/ FIO2 ratio with FIO2: mathematical and experimental description, and clinical relevance. Crit Care. 2007;11(6):R118. doi: 10.1186/cc6174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in Intensive Care Units in 50 countries. JAMA. 2016;315(8):788–800. doi: 10.1001/jama.2016.0291. [DOI] [PubMed] [Google Scholar]
- 22.Tusman G, Böhm SH, Sipmann FS, Maisch S. Lung recruitment improves the efficiency of ventilation and gas exchange during one-lung ventilation anesthesia. Anesth Analg. 2004;98(6):1604–1609. doi: 10.1213/01.ANE.0000068484.67655.1A. [DOI] [PubMed] [Google Scholar]
- 23.Pereira SM, Tucci MR, Morais CCA, et al. Individual positive end-expiratory pressure settings optimize intraoperative mechanical ventilation and reduce postoperative atelectasis. Anesthesiology. 2018;129(6):1070–1081. doi: 10.1097/ALN.0000000000002435. [DOI] [PubMed] [Google Scholar]
- 24.Amato MB, Meade MO, Slutsky AS, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372(8):747–755. doi: 10.1056/NEJMsa1410639. [DOI] [PubMed] [Google Scholar]
- 25.Cinnella G, Grasso S, Natale C, et al. Physiological effects of a lung-recruiting strategy applied during one-lung ventilation. Acta Anaesthesiol Scand. 2008;52:766–775. doi: 10.1111/j.1399-6576.2008.01652.x. [DOI] [PubMed] [Google Scholar]
- 26.Garutti I, Martinez G, Cruz P, Piñeiro P, Olmedilla L, de la Gala F. The impact of lung recruitment on hemodynamics during one-lung ventilation. J Cardiothorac Vasc Anesth. 2009;23(4):506–508. doi: 10.1053/j.jvca.2008.12.023. [DOI] [PubMed] [Google Scholar]
- 27.Cipulli F, Vasques F, Duscio E, Romitti F, Quintel M, Gattinoni L. Atelectrauma or volutrauma: the dilemma. J Thorac Dis. 2018;10(3):1258–1264. doi: 10.21037/jtd.2018.02.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kidane B, Choi S, Fortin D, et al. Use of lung-protective strategies during one-lung ventilation surgery: a multi-institutional survey. Ann Transl Med. 2018;6(13):269. doi: 10.21037/atm.2018.06.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cressoni M, Gotti M, Chiurazzi C, et al. Mechanical power and development of ventilator-induced lung injury. Anesthesiology. 2016;124(5):1100–1108. doi: 10.1097/ALN.0000000000001056. [DOI] [PubMed] [Google Scholar]
- 30.Carramiñana A, Ferrando C, Unzueta M, et al. Rationale and study design for an individualized perioperative open lung ventilatory strategy in patients on one-lung ventilation (iPROVE-OLV) J Cardiothorac Vasc Anesth. 2019;33(9):2492–2502. doi: 10.1053/j.jvca.2019.01.056. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary file1 (JPG 173 kb)
Supplemental Figure S1. Flowchart of the study
Supplementary file2 (TIF 105 kb)
Supplemental Figure S2. Forest plot comparing the effect of PEEPINCREMENTAL or PEEPDECREMENTAL strategies on driving pressure when stratifying patients for age, BMI or history of COPD