Abstract
OBJECTIVE
The FreeStyle Libre (FSL) flash glucose-monitoring device was made available on the U.K. National Health Service (NHS) drug tariff in 2017. This study aims to explore the U.K. real-world experience of FSL and the impact on glycemic control, hypoglycemia, diabetes-related distress, and hospital admissions.
RESEARCH DESIGN AND METHODS
Clinicians from 102 NHS hospitals in the U.K. submitted FSL user data, collected during routine clinical care, to a secure web-based tool held within the NHS N3 network. The t and Mann-Whitney U tests were used to compare the baseline and follow-up HbA1c and other baseline demographic characteristics. Linear regression analysis was used to identify predictors of change in HbA1c following the use of FSL. Within-person variations of HbA1c were calculated using 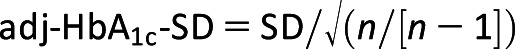 .
.
RESULTS
Data were available for 10,370 FSL users (97% with type 1 diabetes), age 38.0 (±18.8) years, 51% female, diabetes duration 16.0 (±49.9) years, and BMI of 25.2 (±16.5) kg/m2 (mean [±SD]). FSL users demonstrated a −5.2 mmol/mol change in HbA1c, reducing from 67.5 (±20.9) mmol/mol (8.3%) at baseline to 62.3 (±18.5) mmol/mol (7.8%) after 7.5 (interquartile range 3.4–7.8) months of follow-up (n = 3,182) (P < 0.0001). HbA1c reduction was greater in those with initial HbA1c ≥69.5 mmol/mol (>8.5%), reducing from 85.5 (±16.1) mmol/mol (10%) to 73.1 (±15.8) mmol/mol (8.8%) (P < 0.0001). The baseline Gold score (score for hypoglycemic unawareness) was 2.7 (±1.8) and reduced to 2.4 (±1.7) (P < 0.0001) at follow-up. A total of 53% of those with a Gold score of ≥4 at baseline had a score <4 at follow-up. FSL use was also associated with a reduction in diabetes distress (P < 0.0001). FSL use was associated with a significant reduction in paramedic callouts and hospital admissions due to hypoglycemia and hyperglycemia/diabetic ketoacidosis.
CONCLUSIONS
We show that the use of FSL was associated with significantly improved glycemic control and hypoglycemia awareness and a reduction in hospital admissions.
Introduction
Continuous glucose monitoring (CGM) is an established method of monitoring interstitial glucose levels to improve metabolic control in diabetes. The benefits include improvements in glycemic control and hypoglycemia (1–4). Another form of interstitial glucose monitoring known as “flash” glucose monitoring (FreeStyle Libre [FSL]; Abbott Diabetes Care) became available on the U.K. National Health Service (NHS) drug tariff in 2017. In contrast to CGM devices, the FSL does not have alarms to alert the user to hypo/hyperglycemia. However, the advantages of FSL include lower costs and factory calibration, removing the need for frequent painful fingerstick calibrations during the 14-day wear period (5). FSL is also known as intermittent CGM, as data from the FSL sensor are only transmitted when the sensor is scanned with a reading device (reader or mobile phone app).
Randomized controlled trials (RCTs) have demonstrated that FSL use is associated with a significant reduction in the incidence of hypoglycemia in people with type 1 and type 2 diabetes, but to date, a reduction in HbA1c has not been reported (6–8). However, several observational studies have reported improvements in glycemic control (9–14). There are no comprehensive, real-world, large population-based data sets looking at the impact of FSL on multiple aspects of diabetes care. In this study, we use data from the nationwide audit for FSL conducted by the Association of British Clinical Diabetologists (ABCD) to assess the patterns of use of FSL and to study its effect on glycemic control, hypoglycemia, diabetes-related distress, and hospital admissions due to hypoglycemia and hyperglycemia/diabetic ketoacidosis (DKA).
Research Design and Methods
Patient Recruitment and Data Collection
Data for this study were obtained from the nationwide audit of FSL conducted by ABCD (http://www.diabetologists-abcd.org.uk/n3/FreeStyle_Libre_Audit.htm). This nationwide audit commenced in November 2017 using paper forms to collect data, which were then entered onto a secure online tool on the NHS IT network. This network provides maximum security and allows analysis of anonymized national audit data. The tool has the facility to detect data from the same patient entered in two sites (e.g., hospital and primary care) and to merge the data when exported. Data were collected at baseline and follow-up during routine clinical care. Baseline pre-FSL data included demographics, source of FSL funding, previous structured diabetes education completion, HbA1c values from the previous 12 months, Gold score (15) (to assess hypoglycemia awareness), severe hypoglycemia, paramedics callouts, and hospital admissions due to hypoglycemia, hyperglycemia, and DKA over the previous 12 months. The Gold score is a seven-point questionnaire validated for identifying impaired awareness of hypoglycemia (IAH); Gold score ≥4 determines IAH.
We also collected diabetes-related distress scores at baseline and follow-up using the two-item diabetes distress screening instrument (DDS2) (16). The DDS2 asks respondents to rate on a six-point scale the degree to which the following items caused distress: 1) “feeling overwhelmed by the demands of living with diabetes,” and 2) “feeling that I am often failing with my diabetes regimen.” At follow-up, we collected data on all of the above along with FSL-specific measures, such as the number of scans per day and time in range (TIR). At follow-up, we also collected data on adverse effects and reasons for discontinuation due to FSL.
Ethical Approval
The ABCD nationwide audit program has Caldicott Guardian approval. The program is an audit, not research. The NHS encourages audit of clinical practice, and there are guidelines, which were followed, in particular, that contributing centers only collect data from routine clinical practice, and all data collected are anonymized at the point of submission to the central database.
Statistical Methods
For reporting all of the study outcomes, including HbA1c, Gold score, paramedic outcalls, and hospital admissions, we restricted the statistical analysis to those with at least one follow-up. The χ2 test of association was used to compare categorical variables, and the Mann-Whitney U test or t tests were used to compare continuous variables before and after the use of FSL. An analysis stratified by various strata of age, baseline BMI, duration of diabetes, baseline HbA1c, and sex looking at pre- and post-FSL HbA1c and Gold score (15) and diabetes distress screening (DDS) score (16) was performed to understand the usefulness of FSL across these subgroups.
To identify independent predictors of HbA1c reduction in response to use, change in the post-FSL HbA1c was modeled as an independent variable with an average of the pre-FSL HbA1c, age, sex, BMI, duration of diabetes, baseline BMI, number of FSL scans, and structured diabetes education as independent predictors. The follow-up period was defined as the difference between the time of FSL initiation and the date of the most recent HbA1c measurement. The comparison of hospital admissions and paramedic callouts was also restricted only to patients with at least one follow-up. To investigate the effects of intraindividual variations of HbA1c with FSL use, we calculated the intraindividual mean (HbA1c-Mean) and SD (HbA1c-SD), respectively. HbA1c values obtained prior to FSL initiation and follow-up values post-FSL were used. The interindividual difference in the number of HbA1c assessments was adjusted according to the formula: 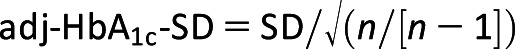 as previously described (16). All of the statistical analyses were done in R 3.6.3 (http://www.r-project.org/).
as previously described (16). All of the statistical analyses were done in R 3.6.3 (http://www.r-project.org/).
Results
Demographic Characteristics of the Study Population
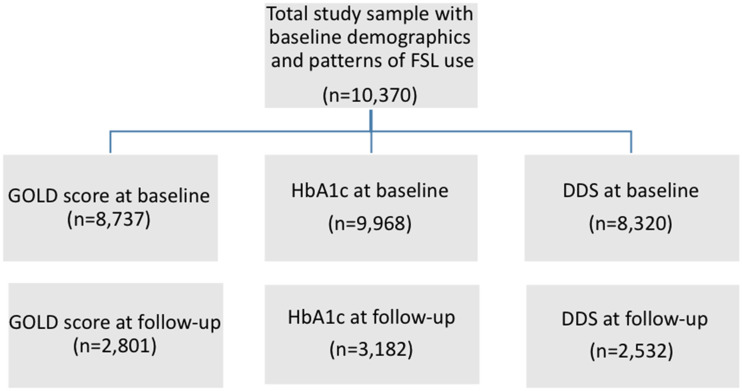
The available data from the study participants started on FSL are shown in Fig. 1. Baseline demographics, indications for starting FSL, structured diabetes education completion, and funding for FSL were available for 10,370 study participants from 102 NHS hospitals across the U.K. Baseline HbA1c, Gold score, and DDS score were recorded for 9,968, 8,737, and 8,320 patients, respectively, while follow-up data were available for 3,182, 2,801, and 2,532 patients, respectively. Table 1 shows the baseline characteristics of the whole study population in comparison with those with at least one follow-up. The mean age of the study participants was 38.0 (±18.0) years with 51% females with a mean duration of diabetes of 16.0 (±49.9) years, a mean baseline HbA1c of 69.8 (±18.2) mmol/mol (8.5%), and baseline BMI of 25.2 (±16.5) kg/m2 (mean [±SD]). The majority of those in the study, 10,058 (97%), had type 1 diabetes, while the remaining had type 2 diabetes or other forms of diabetes. Structured diabetes education had been completed by 6,764 (65%) of the study participants; the majority of FSL users were NHS funded (7,602 [73%]). The baseline demographic characteristics in those with at least one follow-up were similar to the entire study cohort.
Figure 1.
Study schematic showing data for HbA1c, Gold score, and DDS score in the ABCD nationwide audit of FSL. Study outline shows the number of patients recruited in the study and sample size of those with follow-up for HbA1c, Gold score, and DDS score.
Table 1.
Baseline demographic characteristics of study participants with and without follow-up
| Baseline data in all study participants (n = 10,370) | Baseline data in patients with follow-up (n = 3,182) | |
|---|---|---|
| Age (years) | 38.0 (±18.8) | 39.5 (±19.6) |
| Sex, females | 5,322 (51) | 1,688 (53) |
| Baseline BMI (kg/m2) | 25.2 (±6.4) | 25.1 (±6.2) |
| Duration of diabetes (years) | 16.0 (±49.9) | 17 0.1 (±15.5) |
| Type 1 diabetes | 10,058 (97) | 3,126 (98) |
| Insulin pump | 2,428 (23) | 862 (27) |
| White Caucasians | 8,524 (82) | 2,713 (88) |
| NHS funded | 7,602 (73) | 2,354 (74) |
| Number of test strips used per day | 7.7 (±9.8) | 8.1 (±10.1) |
| Mean pre-FSL HbA1c (mmol/mol) | 69.8 (±18.2) | 67.5 (±20.9) |
| Baseline Gold score | 2.7 (±1.8) | 2.7 (±1.9) |
| Completion of structured education | 6,764 (65) | 2,002 (65) |
Data are mean (±SD) or n (%).
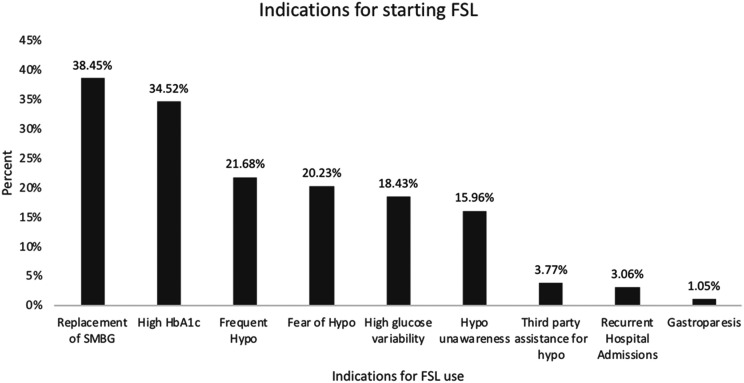
Indications for Starting FSL
There were multiple indications for FSL initiation in the study population (Fig. 2). The most common indication for starting FSL was the replacement of self-monitoring of blood glucose (38.5%) followed by high baseline HbA1c (34.5%), frequent hypoglycemia (21.7%), and fear of hypoglycemia (20.2%).
Figure 2.
Indications for starting FSL in the ABCD nationwide audit of FSL. Multiple indications for FSL initiation in the study population are shown. Hypo, hypoglycemia; SMBG, self-monitoring of blood glucose.
Effect on Glycemic Control and HbA1c Variability
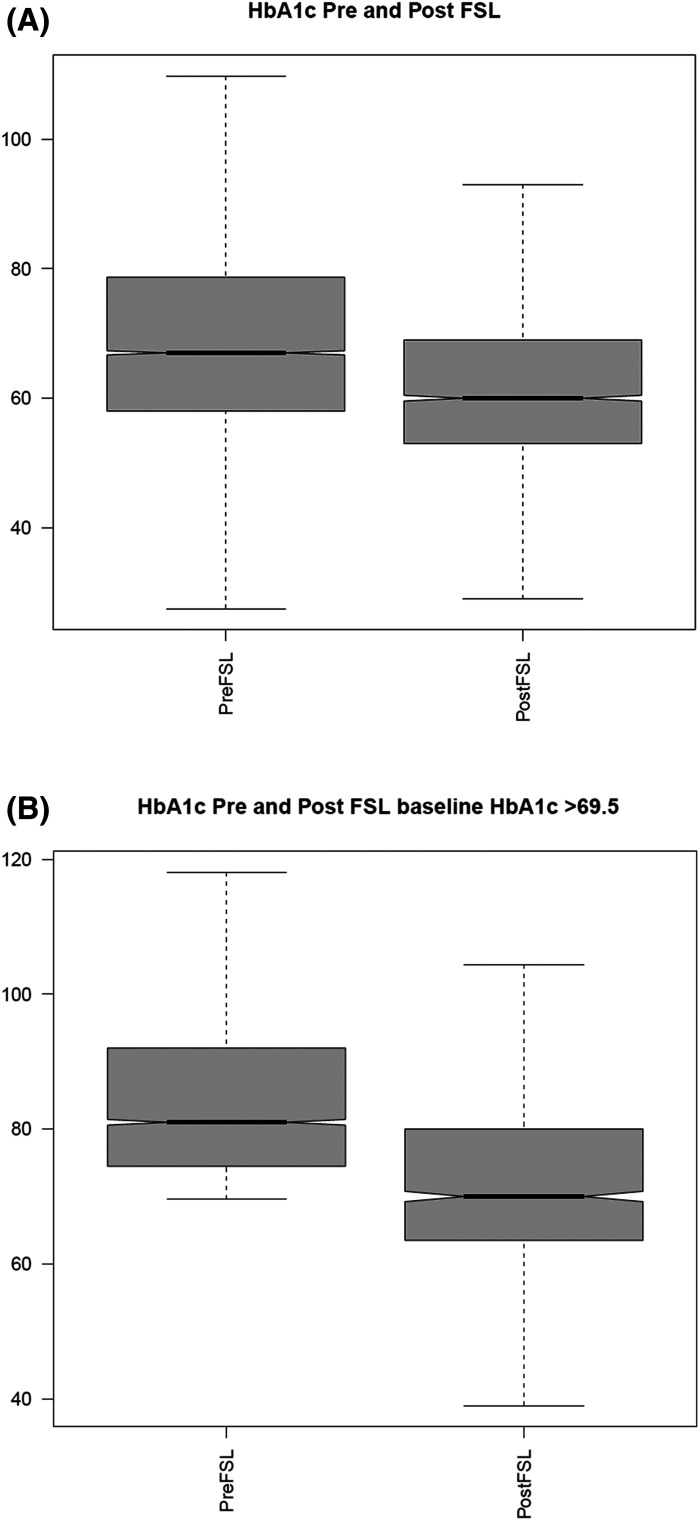
Across the entire study population, the mean HbA1c reduced from 67.5 (±20.9) mmol/mol (8.3%) to 62.3 (±18.5) mmol/mol (7.9%), and in those with baseline HbA1c >69.5 mmol/mol (8.5%), it reduced from 85.5 (±16.1) mmol/mol (10%) to 73.2 (±15.8) mmol/mol (8.8%) (Fig. 3A and B). Table 2 shows baseline and follow-up HbA1c in various strata of age, duration of diabetes, baseline BMI, and baseline HbA1c. The greatest reduction in HbA1c was seen in those with baseline HbA1c >69.5 (−12.3 mmol/mol) followed by females (−10 mmol/mol), the age range of 19–60 years (−8.6 mmol/mol), and duration of diabetes <5 years (−8.4 mmol/mol).
Figure 3.
Distribution of HbA1c change pre- and post-FSL use in the ABCD nationwide audit of FSL in the study population (A) and in those with a baseline HbA1c of ≥69.5 mmol/mol (B).
Table 2.
Baseline and post-FSL HbA1c and Gold score in various strata of age, duration of diabetes, baseline BMI, and baseline HbA1c
| Pre-FSL HbA1c | Post-FSL HbA1c | P value | Pre-FSL Gold score | Post-FSL Gold score | P value | |
|---|---|---|---|---|---|---|
| All | 69.8 (±18.2) | 62.3 (±18.5) | <0.0001 | 2.7 (±1.8) | 2.4 (±1.7) | <0.0001 |
| Age (years) | ||||||
| ≤18 | 63.3 (±19.02) | 58 (±14.9) | <0.0001 | NA | NA | NA |
| 19–60 | 71.3 (±17.5) | 62.7 (±31) | <0.0001 | 2.5 (±1.7) | 2.2 (±1.5) | <0.0001 |
| >60 | 65.3 (±13.5) | 60.4 (±11.4) | <0.0001 | 3.1 (±1.9) | 2.6 (±1.8) | <0.0001 |
| Sex | ||||||
| Male | 69.1 (±18.5) | 61.9 (±22.4) | <0.0001 | 2.70 (±1.7) | 2.3 (±1.6) | <0.0001 |
| Female | 70.4 (±17.8) | 60.0 (±14.7) | <0.0001 | 2.7 (±1.7) | 2.4 (±1.6) | <0.0001 |
| Baseline BMI (kg/m2) | ||||||
| ≤25 | 69.7 (±19.9) | 62.6 (±23.5) | <0.0001 | 2.8 (±1.6) | 2.4 (±1.7) | <0.0001 |
| 25–30 | 69.3 (±13.8) | 61.8 (±16.9) | <0.0001 | 2.6 (±1.7) | 2.3 (±1.6) | <0.0001 |
| >30 | 70.6 (±15.3) | 63.4 (±13.7) | <0.0001 | 2.6 (±1.7) | 2.4 (±1.7) | <0.0001 |
| Duration of diabetes (years) | ||||||
| ≤5 | 68.8 (±19.7) | 60.4 (±15.0) | <0.0001 | 2.69 (±1.7) | 2.55 (±1.6) | 0.10 |
| 5–15 | 73.1 (±19.3) | 66.9 (±28.4) | <0.0001 | 2.44 (±1.6) | 2.15 (±1.4) | <0.0001 |
| >15 | 68.4 (±16.6) | 61.2 (±12.7) | <0.0001 | 2.89 (±1.8) | 2.4 (±1.7) | <0.0001 |
| Baseline HbA1c (mmol/mol) | ||||||
| ≤69.5 (8.5%) | 57.7 (±7.7) | 56.2 (±17.4) | <0.0001 | 2.8 (±1.7) | 2.4 (±1.6) | <0.0001 |
| >69.5 (8.5%) | 85.5 (±16.0) | 73.1 (±15.8) | <0.0001 | 2.5 (±1.7) | 2.3 (±1.6) | 0.0005 |
| Diabetes education | ||||||
| Yes | 68.3 (±16.2) | 61.7 (±19.2) | <0.0001 | 2.7 (±1.7) | 2.4 (±1.6) | <0.0001 |
| No | 72.6 (±21.2) | 63.8 (±16.3) | <0.0001 | 2.8 (±1.7) | 2.5 (±1.6) | 0.0007 |
Data are mean (±SD) unless otherwise indicated. NA, not applicable.
Predictors of HbA1c reduction (Table 3) were higher baseline HbA1c (β = 0.37 [±0.1]) (P < 0.0001) and greater number of FSL scans per day (β = 0.10 [±0.1]) (P < 0.0001). Age, sex, BMI, structured diabetes education completion, and duration of diabetes did not predict a change in HbA1c following FSL initiation. This model explained 29% variability (adjusted R2 = 0.29) in the change in HbA1c following FSL initiation.
Table 3.
Linear regression model showing predictors of decline in HbA1c following the use of FSL
| β | SE | P value | |
|---|---|---|---|
| Pre-FSL HbA1c | 0.37 | 0.01 | <0.0001 |
| Number of FSL scans | 0.10 | 0.01 | <0.0001 |
| Completion of structured education | 0.82 | 0.48 | 0.090 |
| Age | −0.02 | 0.01 | 0.153 |
| Baseline BMI | 0.04 | 0.04 | 0.237 |
| Sex | −0.30 | 0.42 | 0.483 |
| Duration of diabetes | −0.02 | 0.02 | 0.382 |
We did a subset analysis in people with type 1 diabetes using insulin pumps (n = 862) with both baseline and follow-up HbA1c data. In this subgroup, the mean HbA1c reduced from 65.3 (±13) mmol/mol (8.1%) to 60.2 (±25) mmol/mol (7.7%). When the analysis was restricted to those using an insulin pump and with a baseline HbA1c of ≥69.5 mmol (8.5%), HbA1c fell from 80.8 (±11) mmol/mol (9.5%) to 70.1 (±13) mmol/mol (8.6%).
To understand the effect of the number of FSL scans on the change in glycemic control, we stratified the patients into two groups: group 1, those with ≥10 scans per day, and group 2, those with <10 scans per day. The baseline HbA1c reduced from 71.8 (±17) mmol/mol (8.7%) to 66.5 (±15) mmol/mol (8.2%) in group 1, while it reduced from 63.5 (±14) mmol/mol (8%) to 57.9 (±21) mmol/mol (7.4%) in group 2. The absolute drop in HbA1c was more significant in those with higher baseline HbA1c of ≥69.5 mmol/mol (8.5%), with a reduction in HbA1c from 82.1 (±11) mmol/mol (9.7%) to 66.9 (±12) mmol/mol (8.3%) in group 1 and reduction in HbA1c from 85.2 (±16) mmol/mol (9.9%) to 75.8 (±15) mmol/mol (9.1%) in group 2.
The median number of HbA1c readings in the year pre-FSL was 2 (interquartile range [IQR] 2–4), and post-FSL HbA1c was 1 (IQR 1–3). The HbA1c variability, calculated as the adjusted SD for HbA1c, reduced significantly from pre-FSL use, 24 (±14), to post-FSL, 23 (±12) (P = 0.01).
Effect on Self-Reported Hypoglycemia Awareness
In the entire study population, the baseline Gold score was 2.7 (±1.8), which reduced to 2.4 (±1.7) (P < 0.0001) at follow-up. Table 2 shows baseline and follow-up Gold score in various strata of age, duration of diabetes, baseline BMI, and baseline HbA1c. The greatest improvement in Gold score following FSL was seen in those aged >60 years and those with a longer duration of diabetes, lower BMI, and lower HbA1c. In those with paired baseline and follow-up data, 53% of those with baseline Gold score of ≥4 reported a score of <4 at follow-up (regaining hypoglycemia awareness), while 5% of those with baseline Gold score of <4 reported a follow-up score of ≥4 (IAH). We did an analysis in people with type 1 diabetes using insulin pumps (n = 862) with both baseline and follow-up Gold score (n = 1,145). In this subgroup, the Gold score reduced from 2.75 (±1.6) to 2.49 (±1.6).
DDS Score
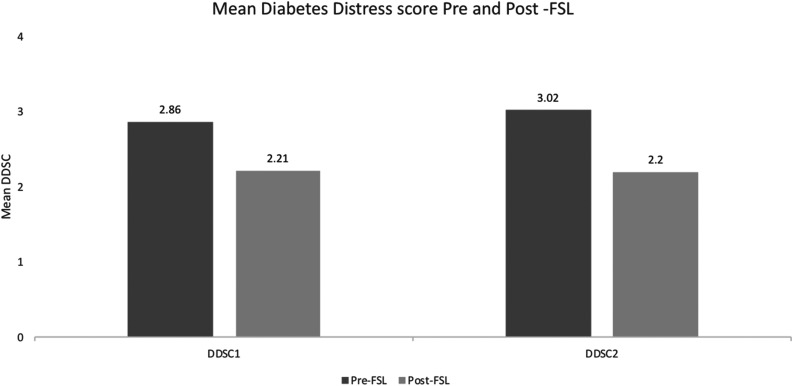
The mean DDS2 component 1 (feeling overwhelmed with demands of living with diabetes) significantly improved from 2.9 at baseline to 2.2 at follow-up (P < 0.0001), and the mean DDS2 component 2 (feeling that I am often failing with my diabetes routine) improved significantly from 3.0 to 2.2 at follow-up (P < 0.0001) (Fig. 4).
Figure 4.
DDS score (DDSC) before and after use of FSL in the ABCD nationwide audit of FSL. Shown is the change in the two components of the DDS score before and after FSL initiation. The DDS2 asks respondents to rate on a six-point scale the degree to which the following items caused distress: 1) “feeling overwhelmed by the demands of living with diabetes,” and 2) “feeling that I am often failing with my diabetes regimen.”
FSL Use, TIR, User Experience, and Side Effects
At follow-up, 89% reported FSL use >70% of the time with a mean of 12.9 (±14.1) scans per day and mean captured sensor data of 87% (±16).
Of those with both follow-up HbA1c and TIR data (n = 2,191), in only 343 (15%) of cases did clinicians report on the internationally accepted TIR (3.9–10 mmol/L [70–180 mg/dL]), with a median TIR of 43% (27–56%).
With the use of FSL, 68% of patients said that they detected a greater proportion of time in hypoglycemia, while 80% said that they were able to reduce the proportion of time in hypoglycemia. Concerning the rate of hypoglycemia, 85% reported that they were able to reduce the rate of hypoglycemia (56% said “a little less,” and 29% said “a lot less”), and 75% reported a reduction in the rate of nocturnal hypoglycemia (45% said “a little less,” and 30% said “a lot less”). Of the 3,182 patients with follow-up, 358 patients (11%) reported problems with FSL; of these, 224 (7%) had technical problems concerning the sensor or the device. A total of 101 patients (3%) reported itching, redness, rash, or allergic reaction, while 33 patients (1%) reported bleeding at the site of the device.
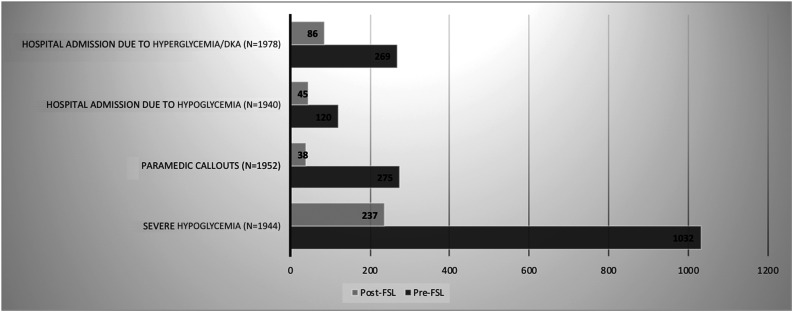
Severe Hypoglycemia, Paramedic Callouts, and Hospital Admissions
These analyses were restricted to those who had both baseline and follow-up events recorded. Comparing the 12-month pre-FSL with 7.5 (IQR 3.4–7.8) months (range 0.3–64 months) of follow-up in this cohort, the total number of paramedic callouts (n = 1,940) decreased from 275 to 38, while the total number of hospital admissions due to hyperglycemia/DKA (n = 1,978) decreased from 269 to 86 following FSL, and the number of admissions due to hypoglycemia (n = 1,952) decreased from 120 to 45 following FSL initiation. In the adult cohort, the total number of episodes of severe hypoglycemia (n = 1,944), defined as those requiring third-party assistance, reduced from 1,032 to 237; the total number of people with at least one episode of severe hypoglycemia at baseline was 357, which reduced to 104 at follow-up (Fig. 5).
Figure 5.
Total number of paramedic callouts, severe hypoglycemia, and hospital admissions during the 12 months before and the 7.5 months of follow-up using FSL in the ABCD nationwide audit. This image shows the change in type 1 diabetes–related resource utilization following FSL initiation.
In a prorated analysis by month, with the use of FSL, the number of hyperglycemia and DKA admissions reduced from 22 per month to 11 per month, the number of hypoglycemia-related admissions reduced from 10 per month to 6 per month, paramedic callouts reduced from 22 per month to 5 per month, and episodes of severe hypoglycemia reduced from 86 per month to 31 per month.
In a sensitivity analysis restricted to those with 12 months’ follow-up (n = 409), the number of paramedic callouts for hypoglycemia decreased from 83 to 4 following FSL, the number of hospital admissions due to hyperglycemia/DKA decreased from 38 to 30, and the number of hospital admissions due to hypoglycemia decreased by 27 to 2 following FSL initiation.
Conclusions
We present the analysis of the largest real-world data set from the nationwide study of flash glucose monitoring (FSL) in people with type 1 diabetes in the U.K. We show that FSL use is associated with improved glycemic control, hypoglycemic awareness, reduced diabetes-related distress, and reduced hospital admissions. In this large observational study, FSL use was associated with significant improvements in glycemic control, especially in those with a higher baseline HbA1c and in those performing a greater number of scans per day. While several RCTs for CGM have shown improved glycemic control in those with type 1 diabetes, to date, there are no RCT data that demonstrate a reduction in HbA1c through FSL use. The SELFY study (An Evaluation of Self-Management of Diabetes Using FreeStyle Libre Flash Glucose Monitoring System in Young People), a single-arm pediatric study, showed enhanced glucose TIR and a 4.4 mmol/mol reduction in HbA1c compared with self-monitoring of blood glucose after an 8-week follow-up period (6). The IMPACT trial (8), primarily designed to assess the effect of FSL in those with well-controlled type 1 diabetes, demonstrated a significant reduction in hypoglycemia but no significant change in HbA1c, a likely reflection of the low baseline HbA1c (50 mmol/mol) (6.7%). The findings of our study are in keeping with the IMPACT study in terms of reported reductions in hypoglycemia. We also found a less substantial change in HbA1c in those with a lower baseline HbA1c that is in agreement with previous studies that have reported a more beneficial effect of FSL in those with higher baseline HbA1c (12,14).
The findings of our study are also in agreement with a recent meta-analysis (9) of 1,723 participants with type 1 or type 2 diabetes that showed similar reductions in HbA1c following FSL use. This meta-analysis also demonstrated that the change in HbA1c with FSL use is highly correlated with baseline HbA1c. A real-world study of 900 FSL users from Edinburgh by Tyndall et al. (12), demonstrated a 4 mmol/mol reduction in HbA1c overall, and, similar to our findings, they observed a more substantial reduction in HbA1c in those with a higher baseline HbA1c and also those with a higher number of scans per day at follow-up. Overall, these results confirm the findings from clinical trials showing that the degree of engagement with the FSL device is an independent predictor of HbA1c response in people with diabetes.
In this study, FSL use was associated with a significant reduction in HbA1c variability during the follow-up period of 7.5 months, as seen in RCTs with CGM. Because HbA1c variability is associated with both micro- (17,18) and macrovascular complications, at least in people with type 2 diabetes (19), if reduced HbA1c variability is sustained, it is possible that FSL may be associated with reduced complication rates in due course, beyond the benefits from the described reduction in HbA1c.
The FSL has been shown to reduce the amount of time spent in hypoglycemia in people with type 1 and type 2 diabetes in RCTs and observational data. In this study, we used the Gold score to assess hypoglycemia awareness. Following the use of the FSL, the Gold score reduced significantly; almost half who had a Gold score of ≥4 at baseline had restoration of hypoglycemia awareness at follow-up, which may be a reflection of the significant reductions in self-reported hypoglycemia. However, our findings are in contrast with a previous observational study (12), which showed no improvement in Gold score or the proportion with impaired awareness of hypoglycemia as assessed by the Gold score. This may reflect the higher proportion of individuals with impaired awareness of hypoglycemia (25% vs. 13%) and higher baseline Gold score (2.7 vs. 2) in our cohort. We observed significant improvements in both components of the DDS score (DDS2 instrument) in those who started on FSL. A recent study (12) described improvements in diabetes-related distress but a paradoxical increase in anxiety and depression on the Hospital Anxiety and Depression Scale in those using FSL. This could potentially reflect the demands that access to continuous glucose data place on an individual; although not assessed in our study, this is an area that would benefit from future qualitative research.
We report significant reductions in paramedic callouts and hospital admissions with the use of FSL in the 7.6-month follow-up period. The most significant reductions were seen in paramedic callouts followed by admissions due to hyperglycemia and DKA and those due to hypoglycemia. These findings are consistent with the data reported from the Edinburgh cohort (12) and Belgian cohort (14); however, a long-term follow-up and cost-effectiveness analysis are needed to evaluate the long-term clinical and economic benefits.
Our study has several limitations. The data for this study were obtained from a nationwide audit of FSL in routine clinical care and, as such, lacked a comparator arm and the methodically controlled data collection in RCTs. Nonetheless, these data represent the largest nationwide, real-world experience with FSL in all aspects of diabetes care. Most of our study participants consisted of people with type 1 diabetes who fulfilled the criteria for funding by the NHS in the U.K. The majority received NHS funding for their FSL device. The access criteria have resulted in approximately one-third of people living with diabetes being reimbursed for the FSL, which gives an indication of our representative selection criterion. The mean HbA1c at baseline was 69.8 mmol/mol (8.5%), not dissimilar to national audit data from England and Wales, which report a mean HbA1c of 64 mmol/mol (8%) for pump users and 71 mmol/mol (8.6%) for those on multiple-dose injection therapy. The average baseline HbA1c in our real-world study was higher as compared with the IMPACT trial (8) and the FUTURE study (14). However, this reflects the real-world nature of the study.
Our study may also be affected by regression to mean in HbA1c measures (20), a tendency for HbA1c to fall on repeat testing. However, we have minimized this effect by taking an average of available HbA1c measures 1 year prior to FSL use and including all HbA1c measures available during the follow-up period. We compared the paramedic callouts and hospital admissions 1 year before starting FSL with the paramedic callouts and hospital admissions in the seven-and-a-half–month follow-up period. However, we have also done a sensitivity analysis in a subset of patients with a 12-month follow-up period and show that the beneficial effects of FSL persist for key outcomes. Given the significant reduction in the episodes of severe hypoglycemia and paramedic callouts, these findings will have implications for morbidity, mortality, and costs related to diabetes, and further studies are needed to confirm these.
In summary, we report an analysis of the largest real-world data set observing FSL use in type 1 diabetes and show that its use is associated with significant improvements in glycemic control, hypoglycemia awareness, severe hypoglycemia, and a reduction in hospital admissions. Long-term follow-up and cost-effectiveness analysis are needed to assess if these benefits from FSL are sustained and affordable to health care systems.
Article Information
Acknowledgments. The authors would like to thank all the clinicians and support staff who participated in the nationwide study, listed at https://abcd.care/Resource/ABCD-Freestyle-Libre-Audit-Contributors.
Duality of Interest. The ABCD nationwide FSL audit is supported by a grant from Abbott Laboratories. E.G.W. serves on the advisory panel for Abbott Diabetes Care, Dexcom, and Eli Lilly and Company; has received research support from Diabetes UK; and is on the speakers bureau for Abbott Diabetes Care, Dexcom, Eli Lilly and Company, Insulet Corporation, Novo Nordisk, and Sanofi. C.W. has a spouse/partner serving on the advisory panel for Celgene and on the speakers bureau for LEO Pharma and Novartis. R.E.J.R. serves on the advisory panel for Novo Nordisk A/S and on the speakers bureau for BioQuest. T.S. is on the speakers bureau for Novo Nordisk Foundation and reports a relationship with Bristol-Myers Squibb, Eli Lilly and Company, and Sanofi. No other potential conflicts of interest relevant to this article were reported.
The FSL audit was independently initiated and performed by ABCD, and the authors remain independent in the analysis and preparation of this report.
Author Contributions. E.G.W., C.W., R.E.J.R., and T.S. conceived the presented idea. H.D., E.G.W., C.W., R.E.J.R., and T.S. contributed to the data analysis. H.D. wrote the first draft of the manuscript. All of the authors contributed to the writing of the manuscript and made extensive comments, criticism, and changes in the final draft of the paper. All of the authors saw the final version of the manuscript. H.D. is the guarantor of this work and, as such, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
H.D. and E.G.W. are joint first authors.
R.E.J.R. and T.S. are joint senior authors.
This article is featured in a podcast available at https://www.diabetesjournals.org/content/diabetes-core-update-podcasts.
References
- 1.Battelino T, Danne T, Bergenstal RM, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the International Consensus on Time in Range. Diabetes Care 2019;42:1593–1603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beck RW, Riddlesworth T, Ruedy K, et al.; DIAMOND Study Group . Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: the DIAMOND Randomized Clinical Trial. JAMA 2017;317:371–378 [DOI] [PubMed] [Google Scholar]
- 3.Heinemann L, Deiss D, Hermanns N, et al. HypoDE: research design and methods of a randomized controlled study evaluating the impact of real-time CGM usage on the frequency of CGM glucose values <55 mg/dl in patients with type 1 diabetes and problematic hypoglycemia treated with multiple daily injections. J Diabetes Sci Technol 2015;9:651–662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Heinemann L, Freckmann G, Ehrmann D, et al. Real-time continuous glucose monitoring in adults with type 1 diabetes and impaired hypoglycaemia awareness or severe hypoglycaemia treated with multiple daily insulin injections (HypoDE): a multicentre, randomised controlled trial. Lancet 2018;391:1367–1377 [DOI] [PubMed] [Google Scholar]
- 5.Bilir SP, Hellmund R, Wehler B, Li H, Munakata J, Lamotte M. Cost-effectiveness analysis of a flash glucose monitoring system for patients with type 1 diabetes receiving intensive insulin treatment in Sweden. Eur Endocrinol 2018;14:73–79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Campbell FM, Murphy NP, Stewart C, Biester T, Kordonouri O. Outcomes of using flash glucose monitoring technology by children and young people with type 1 diabetes in a single arm study. Pediatr Diabetes 2018;19:1294–1301 [DOI] [PubMed] [Google Scholar]
- 7.Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Flash glucose-sensing technology as a replacement for blood glucose monitoring for the management of insulin-treated type 2 diabetes: a multicenter, open-label randomized controlled trial. Diabetes Ther 2017;8:55–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bolinder J, Antuna R, Geelhoed-Duijvestijn P, Kröger J, Weitgasser R. Novel glucose-sensing technology and hypoglycaemia in type 1 diabetes: a multicentre, non-masked, randomised controlled trial. Lancet 2016;388:2254–2263 [DOI] [PubMed] [Google Scholar]
- 9.Evans M, Welsh Z, Ells S, Seibold A. The impact of flash glucose monitoring on glycaemic control as measured by HbA1c: a meta-analysis of clinical trials and real-world observational studies. Diabetes Ther 2020;11:83–95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fokkert MJ, Damman A, van Dijk PR, et al. Use of FreeStyle Libre Flash Monitor Register in the Netherlands (FLARE-NL1): patient experiences, satisfaction, and cost analysis. Int J Endocrinol 2019;2019:4649303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Alawadi F, Rashid F, Bashier A, et al. The use of Free Style Libre Continues Glucose Monitoring (FSL-CGM) to monitor the impact of Ramadan fasting on glycemic changes and kidney function in high-risk patients with diabetes and chronic kidney disease stage 3 under optimal diabetes care. Diabetes Res Clin Pract 2019;151:305–312 [DOI] [PubMed] [Google Scholar]
- 12.Tyndall V, Stimson RH, Zammitt NN, et al. Marked improvement in HbA1c following commencement of flash glucose monitoring in people with type 1 diabetes. Diabetologia 2019;62:1349–1356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Moreno-Fernandez J, Pazos-Couselo M, González-Rodriguez M, et al. Clinical value of flash glucose monitoring in patients with type 1 diabetes treated with continuous subcutaneous insulin infusion. Endocrinol Diabetes Nutr 2018;65:556–563 [DOI] [PubMed] [Google Scholar]
- 14.Charleer S, De Block C, Van Huffel L, et al. Quality of life and glucose control after 1 year of nationwide reimbursement of intermittently scanned continuous glucose monitoring in adults living with type 1 diabetes (FUTURE): a prospective observational real-world cohort study. Diabetes Care 2020;43:389–397 [DOI] [PubMed] [Google Scholar]
- 15.Gold AE, MacLeod KM, Frier BM. Frequency of severe hypoglycemia in patients with type I diabetes with impaired awareness of hypoglycemia. Diabetes Care 1994;17:697–703 [DOI] [PubMed] [Google Scholar]
- 16.Fisher L, Glasgow RE, Mullan JT, Skaff MM, Polonsky WH. Development of a brief diabetes distress screening instrument. Ann Fam Med 2008;6:246–252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Virk SA, Donaghue KC, Cho YH, et al. Association between HbA1c variability and risk of microvascular complications in adolescents with type 1 diabetes. J Clin Endocrinol Metab 2016;101:3257–3263 [DOI] [PubMed] [Google Scholar]
- 18.Penno G, Solini A, Zoppini G, et al.; Renal Insufficiency and Cardiovascular Events (RIACE) Study Group . Hemoglobin A1c variability as an independent correlate of cardiovascular disease in patients with type 2 diabetes: a cross-sectional analysis of the renal insufficiency and cardiovascular events (RIACE) Italian multicenter study. Cardiovasc Diabetol 2013;12:98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Parry HM, Deshmukh H, Levin D, et al. Both high and low HbA1c predict incident heart failure in type 2 diabetes mellitus. Circ Heart Fail 2015;8:236–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McDonald TJ, Warren R. Diagnostic confusion? Repeat HbA1c for the diagnosis of diabetes. Diabetes Care 2014;37:e135–e136 [DOI] [PubMed] [Google Scholar]