Abstract
Perturbations in fatty acid (FA) metabolism as well as thinning of the cerebral cortex have been associated with cognitive decline in the elderly. Predominant FAs in the brain are docosahexaenoic acid (DHA) and arachidonic acid (ARA). Approximately 2–8% of esterified DHA and 3–5% of esterified ARA in the brain are replaced daily. DHA and ARA are derivatives of 18-carbon essential FAs, α-linolenic acid and linoleic acid, that must be imported into the brain from the circulation. In blood, FAs are primarily transported in triacylglycerols (TAGs) from which they can be released at the blood–brain-barrier and transported inside the brain. We show that circulating levels of TAGs carrying 18-carbon FAs are positively associated with cortical thickness in middle-aged adults. These associations are stronger in cortical regions with higher expression of genes regulating long-chain FA metabolism and cellular membranes, and cortical thickness in the same regions may be related to cognitive performance.
Subject terms: Metabolomics, Neuroscience
Eeva Sliz et al. show an association between triacylglycerols species carrying 18-carbon unsaturated fatty acids and cortical thickness by combining serum lipidomics and brain imaging data. This association is more pronounced in cortical regions with higher expression of genes regulating fatty acid metabolism and cellular membranes.
Introduction
Fatty acids (FAs) are essential building blocks of other lipids, such as triacylglycerols (TAGs), phospholipids, and cholesteryl esters. The brain requires high quantities of FAs for normal structure and function. Docosahexaenoic acid (DHA; FA22:6ω3) and arachidonic acid (ARA; FA20:4ω6) are the predominant FAs in the brain, constituting ~20% of the dry weight of the human brain1. It has been estimated that 2–8% of esterified DHA and 3–5% of esterified ARA in the brain are replaced daily2. DHA and ARA are derivatives of α-linolenic acid (ALA, FA18:3ω3) and linoleic acid (LA, FA18:2ω6), respectively. These essential FAs, along with other polyunsaturated FAs (PUFAs), must be imported into the brain from the circulation, whereas saturated and monounsaturated FAs can be synthesized within the brain. In blood many of the circulating FAs are transported within TAGs that are synthesized in the liver and transported within lipoprotein particles. Lipoprotein particles can enter endothelial cells of the blood–brain-barrier (BBB) by endocytosis, and, subsequently, FAs can be released from TAGs3. Alternatively, lipoprotein particles can interact with membrane-bound lipoprotein lipase to release unesterified FAs that can enter BBB by transporter proteins or passive diffusion3. Eventually, TAG-derived FAs within endothelial cells can be transported into neuronal and non-neuronal cells of the brain to be used for phospholipid synthesis or other functions3.
FAs vary in the degree of saturation and length of their acyl-carbon chain. Generally, shorter-chain, saturated FAs are associated with higher risk of cardiovascular diseases, whereas longer-chain PUFAs have beneficial cardiovascular effects4,5. Higher dietary intake and circulating concentrations of PUFAs have been associated with better cognitive functioning and lower risk of neurodegenerative diseases6–8. The brain uses FAs mostly for synthesis of phospholipids, which are essential components of cell membranes9. PUFA derivatives have additional functions in the brain—they act as drivers of multiple signaling processes, such as inflammatory or endocannabinoid signaling3. Lack of PUFAs has a detrimental effect on normal brain physiology, arising from, among others, altered structural features of cellular membranes and defects in preventing neuroinflammation6.
In the present study, we show that circulating concentrations of particular TAG species carrying 18-carbon FAs, including the essential ALA and LA, associate with thicker cerebral cortex in middle-aged adults. We further demonstrate that these TAG species show larger effect sizes in cortical regions with higher expression of genes regulating long-chain FA metabolism and cellular membranes, and that cortical thickness in these regions may be related to cognitive performance.
Results
Recent developments in lipidomics technologies have enabled identification of lipids with improved specificity and sensitivity. Here, we study the associations between cortical thickness and circulating concentrations of 518 TAG species (Supplementary Fig. 1 and Supplementary Data 1), each of which was characterized by the total number of acyl-chain carbons and double bonds and by the identity of one of the three esterified FAs. After excluding individuals on lipid-lowering medication, our study sample consisted of 441 middle-aged adults from the Saguenay Youth Study (SYS)10 in whom brain magnetic resonance images of the brain (1.5T Avanto, Siemens) were acquired and TAG species concentrations were quantified in fasted serum samples (TrueMass Complex Lipid PanelTM, Metabolon) were available. The basic characteristics of the studied participants are in Table 1.
Table 1.
Study population characteristics.
| Characteristic | Valuea |
|---|---|
| Number | 441 |
| Males (%) | 189 (43) |
| Age, years | 49.1 ± 4.9 |
| BMI, kg/m2 | 27.6 ± 5.2 |
| Total TAG, mmol/L | 1.21 ± 0.7 |
| Mean cortical thickness, mm | 2.43 ± 0.09 |
| Total cortical surface area, m2 | 0.17 ± 0.016 |
BMI body mass index, TAG triacylglycerol.
aValues are mean ± standard deviation.
TAG associations with cortical thickness
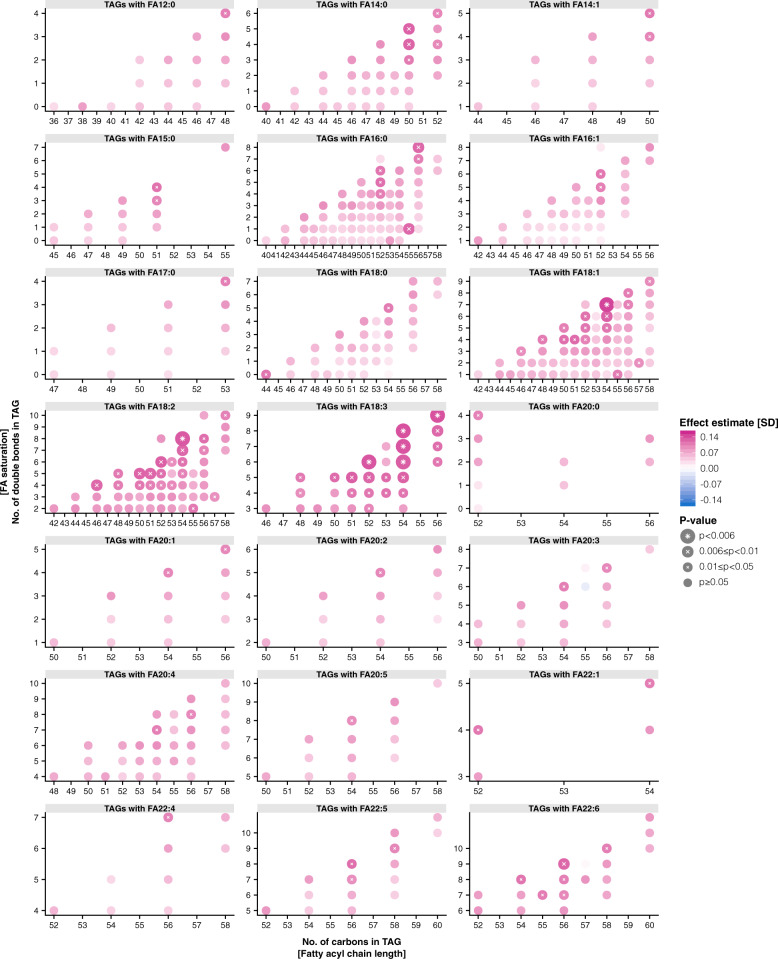
In linear regression models adjusted for age and sex, we found positive associations (p < 0.006) between mean cortical thickness and concentrations of 7 of 518 tested TAG species (TAG52:6FA18:3, TAG54:6FA18:3, TAG54:7FA18:1, TAG54:7FA18:3, TAG54:8FA18:2, TAG54:8FA18:3, TAG56:9FA18:3; Fig. 1 and Supplementary Data 1). Statistical significance was considered at p < 0.006 (0.05/9) to correct for the nine principal components explaining 95% of variation in serum concentrations of 518 TAG species11–13.
Fig. 1. Associations between mean thickness of the cerebral cortex and serum concentrations of 518 triacylglycerols.
We tested associations between mean thickness of the cerebral cortex and serum concentrations of 518 triacylglycerol species in middle-aged adults (n = 441) using linear regression models. Prior to model fitting, each variable was adjusted for age and sex, and transformed using rank-based inverse normal transformation. Statistical significance was considered at p < 0.006 (0.05/9) to correct for the first nine principal components explaining 95% of variation in serum concentrations of 518 triacylglycerol species. Each plot shows the results of the association tests by sets of triacylglycerol species carrying the same fatty acyl-chain; individual points indicate effect estimates (color) and p-values (size) for each of the triacylglycerol species. Triacylglycerol species are characterized by the total number of acyl-chain carbons (x-axes) and double bonds (y-axes) per triacylglycerol molecule. FA fatty acid, TAG,triacylglycerol; SD standard deviation.
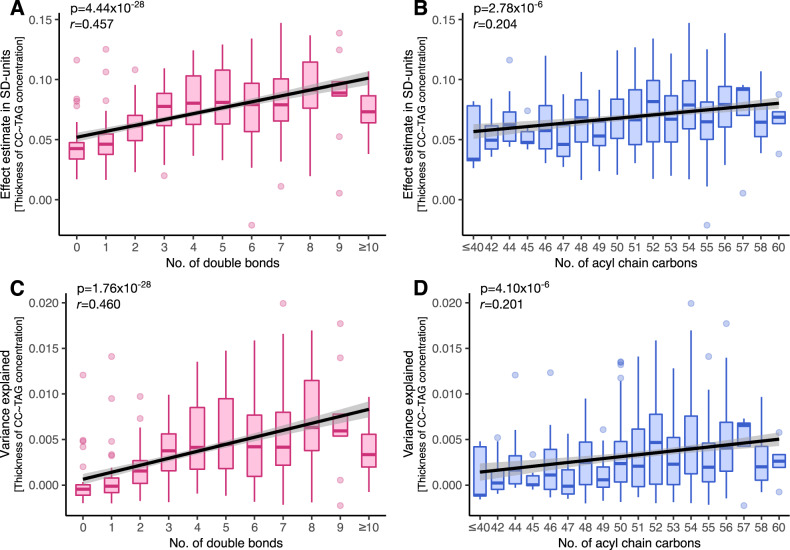
Further, we found that the number of double bonds and the number of acyl-chain carbons per TAG species correlated positively with both the effect sizes obtained from the 518 regression models (p = 4.4 × 10–28 and p = 2.8 × 10–6, respectively) and the proportions of the variance in mean cortical thickness explained by these models (p = 1.8 × 10–28 and p = 4.1 × 10–6, respectively; Fig. 2). These findings indicate that the longer and more unsaturated the FAs carried by TAG molecules are, the stronger the positive associations between the concentration of those TAG species and cortical thickness are.
Fig. 2. Correlations of fatty acyl unsaturation and chain length with the test statistics of the associations between mean thickness of the cerebral cortex and concentrations of 518 triacylglycerols.
We studied if the association statistics of the mean thickness of the cerebral cortex with concentrations of 518 triacylglycerol species vary with fatty acyl unsaturation or chain length. To do this, we extracted effect estimates (y-axes in a and b) and coefficients of determination (adjusted R2; y-axes in c and d) from each of the 518 linear models, and studied their correlation (Pearson’s r) with the number of double bonds (x-axes in a and c) or the number of acyl-chain carbons (x-axes in b and d) per triacylglycerol species. CC cerebral cortex, SD standard deviation, TAG triacylglycerol.
To evaluate if FAs per se drive these associations, we tested associations between mean cortical thickness and 21 FA species extracted from TAG fraction of lipids (TAG-FAs). Only TAG-FA18:3 (p = 0.0071), TAG-FA22:1 (p = 0.034), and TAG-FA22:6 (p = 0.036) showed nominally significant associations (Supplementary Fig. 2). We also tested associations between mean cortical thickness and total concentrations of 28 FA species (total-FA; from all circulating protein or lipid carriers, including TAGs). Only total-FA22:6 (p = 0.009) and total-FA24:0 (p = 0.043) showed nominally significant associations (Supplementary Fig. 3).
The results of supplemental analyses further adjusted for apolipoprotein E (APOE) alleles were comparable to the original findings (Supplementary Data 1 and Supplementary Fig. 4). We also tested the associations between total cortical surface area and TAGs, but we did not observe statistically significant associations (Supplementary Fig. 5).
In silico analyses of TAG effect sizes across cortex
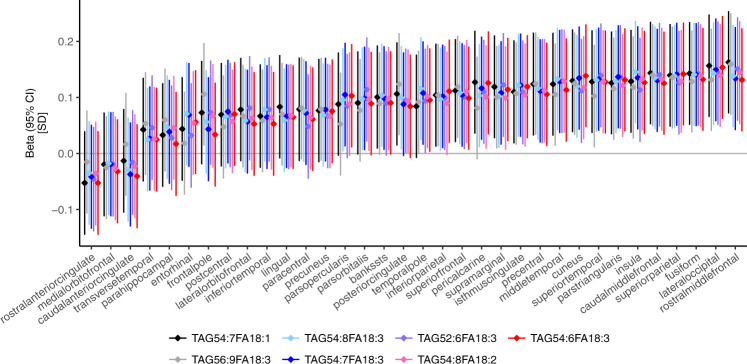
We observed that the seven TAGs significantly associated with mean cortical thickness were associated similarly with thickness at each of the 34 regions of the cerebral cortex, as segmented by FreeSurfer using the Desikan-Killiany atlas14 (Fig. 3). The associations between seven TAGs and cortical thickness varied across the 34 regions, being most pronounced in the rostral middle frontal cortex and least pronounced in the anterior cingulate cortex (Fig. 3). From these region-specific association tests, we averaged effect estimates (betas) of the seven TAGs in each of the 34 regions, deriving an “interregional average-beta profile” (Supplementary Data 2). We utilized this “interregional average-beta profile” in downstream analyses to estimate (i) relative contributions of different cell types (virtual histology) and (ii) the cognitive implications.
Fig. 3. Associations of seven triacylglycerol species with cortical thickness in 34 regions of the cerebral cortex.
Cortical thicknesses in each of the 34 regions (average over two hemispheres) were regressed on serum concentrations of each of the seven triacylglycerol species. Regression model fitting was done in a sample of 441 middle-aged adults. Before model fitting, all variables were adjusted for age and sex and inverse-rank normal transformed. TAG triacylglycerol, FA fatty acid, SD standard deviation, CI confidence interval.
In virtual histology analyses15, we found that the “interregional average-beta profile” was associated with interregional profiles of messenger RNA (mRNA) expression of genes specific to astrocytes (p = 1.9 × 10–3), CA1 pyramidal neurons (p = 1.9 × 10–3) and S1 pyramidal neurons (p = 3.0 × 10–3; Supplementary Fig. 6). These results suggest that the TAG associations with cortical thickness may involve these cell types. In functional element enrichment analyses, we found that astrocyte-specific genes were enriched in biological functions relevant to long-chain FA metabolic processes (13 genes with r > 0; Supplementary Data 3). Neuronal cell-specific genes were enriched in biological functions relevant to cell membrane, ion transport and actin remodeling (33 and 45 genes for CA1 and S1, respectively, with r > 0; Supplementary Data 4 and 5).
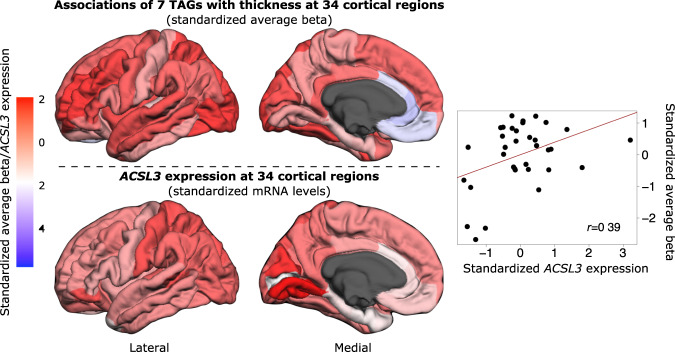
Of note, the astrocyte-specific genes relevant to long-chain FA metabolic processes included ACSL3, encoding acyl-CoA synthetase long-chain family member 3, which is an enzyme required for the incorporation of long-chain FAs into cellular lipids16. ACSL3 is one of two predominant isoforms of this enzyme in the brain17. In the present study, the “interregional average-beta profile”, that was derived from the associations between seven TAGs and cortical thickness, correlated positively with the interregional profile of ACSL3 expression (Fig. 4), indicating that the associations of seven TAGs with cortical thickness are more pronounced in cortical regions with higher expression of this gene.
Fig. 4. Interregional variations in the associations of seven triacylglycerols with cortical thickness and expression of ACSL3.
Thirty-four cortical regions segmented by FreeSurfer are shown with color-coded values of the associations between seven triacylglycerols and cortical thickness (standardized average-beta) and ACSL3 expression (standardized mRNA levels). Correlation between interregional variations in the average-beta of the seven triacylglycerols on cortical thickness and ACSL3 expression is shown on the right. TAG triacylglycerol, ACSL3 acyl-CoA synthetase long-chain family member 3.
Correlations with cognitive functioning
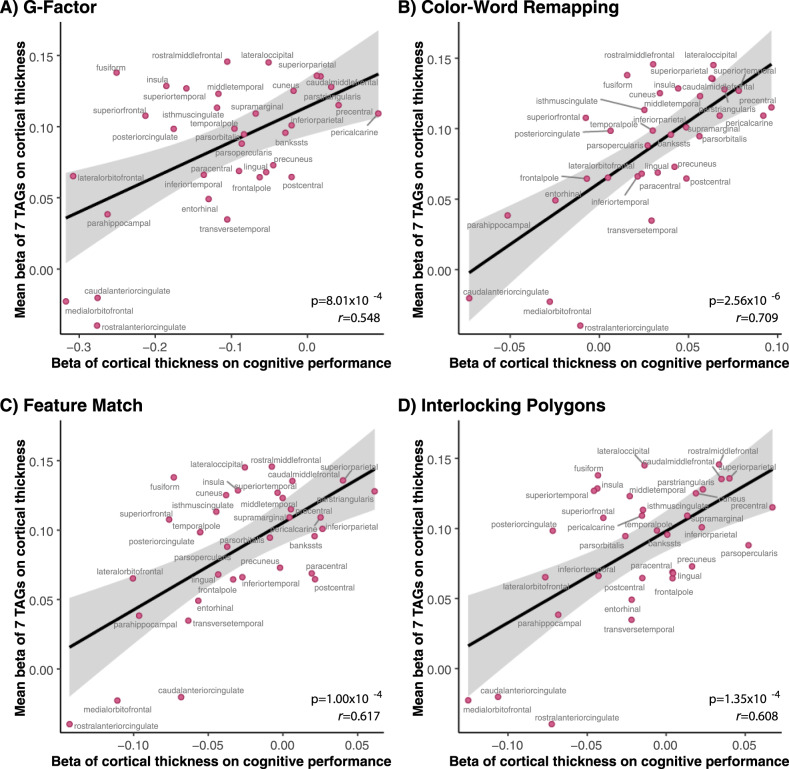
To examine if our findings have cognitive implications, we tested whether interregional variations in the associations between seven TAGs and cortical thickness (i.e., the “interregional average-beta profile”) correlate with interregional variations in the associations between cortical thickness and measures of cognitive functioning. These analyses showed positive associations for several cognitive measures, as well as for the general cognition “G-Factor”18 (p = 8.01 × 10–4, Fig. 5 and Supplementary Fig. 7). This finding indicates that, in cortical regions where thickness is most closely associated with the seven TAGs, the thickness is also most closely associated with cognitive functioning.
Fig. 5. Correlations between interregional variations in associations of seven triacylglycerols with cortical thickness and of cortical thickness with cognitive functioning.
To assess these correlations, we used Pearson’s r. The figure shows the results for the general cognition “G-Factor”8 (a) and three most significant individual measures, namely color-word remapping (b), feature match (c), and interlocking polygons (d). The results for the remaining tests are presented in Supplementary Fig. 8.
Discussion
Thickness of the cerebral cortex is related to cognitive functioning in aging adults19–21, but the molecular factors modulating this relationship are incompletely understood. We studied associations between 518 circulating TAG species and mean thickness of the cerebral cortex in a general population of middle-aged adults. We identified seven circulating TAG species carrying 18-carbon unsaturated FAs to be positively associated with mean cortical thickness. We also demonstrated that these seven TAG species show larger effect estimates on cortical regions with higher expression of astrocyte- and neuronal cell-specific genes involved in the regulation of long-chain FA metabolism and cellular membranes. Finally, we demonstrate that thickness of cortical regions most closely associated with these seven TAG species is also most closely associated with better cognitive functioning.
Currently, there are no published studies examining associations between thickness of the cerebral cortex and a comprehensive set of circulating TAG species in a general population. A recent study reported associations between mild cognitive impairment (MCI) and Alzheimer’s disease (AD) with serum levels of 84 TAG species in 689 participants from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) cohort (average age 75 years)22. The TAG species in that study were defined by the total number of acyl-chain carbons and double bonds, but unlike in the present study, they were not defined by the identity of any of the three esterified FAs. Also unlike in the present study, the primary analyses of that work focused on studying component scores from principal component analysis instead of studying individual TAG species. Supportive of our findings, the authors found that lower component scores of PC5 loaded by PUFA-containing TAGs (PUTGs) were observed in both MCI and AD patients than in healthy controls, and that lower levels of these PUTGs were associated with lower thickness of the entorhinal cortex and hippocampus22. The results in our cohort (average age 49 years) indicate that the positive associations between PUTGs and cortical thickness are also seen in generally healthy middle-aged adults, and that the particular PUTG species carrying 18-carbon FAs may play a key role in PUTG associations with thicker cortex.
Circulating total-TAG concentration has been reported as being associated with both thicker23 and thinner24 cerebral cortex. The former study was completed in a community-based sample of middle-aged adults (average age 51 years), whereas the latter study was carried out in a population (average age 60 years) involving patients with the metabolic syndrome. In the metabolic syndrome, blood levels of saturated FAs are high, whereas levels of PUFAs are low25. The contradictory results of the above-described studies could arise from different FA composition of TAGs in the community-based sample of individuals versus patients with the metabolic syndrome. We did not find an association between cortical thickness and total TAG concentration (beta = 0.078; p = 0.10). Our main findings, however, are supportive of the possibility that differing FA composition may contribute to the reported variability in the associations of total TAGs with cortical thickness: TAGs carrying long-chain PUFAs rather than TAGs carrying shorter-chain saturated FAs are associated with thicker cerebral cortex.
The seven TAGs we identified as associated with mean cortical thickness carry 18-carbon FAs, but the total circulating concentrations of these FAs were not associated with mean cortical thickness (Supplementary Fig. 3). This finding suggests that brain uptake of essential FAs may be preferred in a form of TAG. A study that reported omega-3 PUFAs esterified to TAGs having superior bioavailability compared with free FAs or FA ethyl esters26 provides evidence, at least partially, supporting this hypothesis. Current understanding is that the uptake of FAs to the brain happens via FA-transport proteins or passive diffusion after hydrolysis of TAGs at the BBB:3 FA uptake requires concomitant FA activation to acyl-coenzyme A (CoA) by long-chain-fatty-acid-CoA synthases3, such as ACSL327, mRNA expression of which was found to be correlated with the TAG-associated effect estimates across the cerebral cortex in the present study. There is, however, evidence that intact TAGs may cross the BBB in mice28. It will be an interesting topic for future studies to investigate if intact TAGs can cross the BBB in humans, and if TAG species with a particular FA composition are preferred over others in delivering FAs to the brain.
Five out of the seven TAGs (TAG52:6FA18:3, TAG54:6FA18:3, TAG54:7FA18:3, TAG54:8FA18:3, and TAG56:9FA18:3) we identified as associated with thicker cortex carry FA18:3 that includes the essential ALA. Dietary supplementation of ALA is crucial for normal neural development29, and, therefore, ALA is a fundamental constituent of baby formulas29. Lack of dietary ALA decreases phospholipid synthesis and induces changes in FA composition of membrane phospholipids in the rat brain30. ALA is a precursor for DHA, the key structural FA in the cerebral cortex3. The brain’s ability to synthesize DHA from ALA is limited, and thus it is thought that most of the brain DHA is derived from the circulation31. Unexpectedly, we did not find significant associations between DHA-containing lipids and cortical thickness. Out of the nominally significant TAG species carrying DHA, TAG56:9FA22:6 showed the strongest association (beta = 0.125, p = 0.0074); this association may have been significant in a larger sample. Circulating total-FA22:6 showed a nominally significant (beta = 0.123, p = 0.009) association with cortical thickness. In our sample; however, the associations between cortical thickness and FA18:3-containing TAGs were more robust than the associations of total-FA22:6 or FA22:6-containing TAGs. This could imply that, in middle-aged adults, maintenance of higher circulating concentration of ALA-containing TAGs may have more beneficial effect on cortical thickness than maintenance of higher circulating concentration of DHA or DHA-containing TAGs.
The remaining two of the seven TAGs (TAG54:7FA18:1 and TAG54:8FA18:2) associating with thicker cortex in the present study carry FA18:1 and FA18:2. The latter includes essential LA that is used for synthesis of ARA, the other of the predominant FAs in the brain along with DHA1. The former includes oleic acid (OA; FA18:1ω9), which is the main monounsaturated FA within the brain3 and constitutes ~40% of the FAs in the phospholipid fraction of adult human brain32. Considering the essential role of phospholipids in the formation of cellular membranes, it is not surprising that, in the functional element enrichment analyses, we found positive correlations between the seven TAG-associated effect estimates on cortical thickness and expression of neuronal cell-specific genes with function related to cell membranes. Similarly to PUFAs, also OA plays a role in anti-inflammatory signaling in the brain33 in addition to its essential role as a structural component in phospholipid bilayers. Overall, a considerable body of literature underscores the multiple functions of FAs in the normal brain physiology. In line with this, recent evidence suggests that perturbations of FA metabolism are closely related to development of neurodegenerative diseases6,8,34—the results of the present study may have implications for understanding the role of circulating TAGs in FA delivery into the brain.
This study elucidates the associations between circulating TAGs and the thickness of the human cerebral cortex. Our key finding is that TAG species carrying 18-carbon FAs associate with thicker cortex in middle-aged adults. All significant TAGs had a total of 6 or more double bonds and 52 or more of acyl-chain carbons; overall, the effect sizes of TAGs on cortical thickness were positively correlated with both the number of double bonds and acyl-chain length in TAGs. We found that the interregional variation in TAG-associated effects on cortical thickness was associated with interregional variations of expression of genes specific to astrocytes and pyramidal neurons. Our results further suggest that the seven circulating TAGs may have stronger associations with cortical thickness in regions with higher expression of genes with functions related to cellular membranes and long-chain FA metabolism—here, one of the most intriguing findings was that the effect estimates of the seven TAGs tended to be larger in cortical regions with higher expression of ACSL3, a gene encoding an enzyme participating in FA uptake by the brain. Lastly, our findings may have cognitive implications, as thickness of cortical regions most closely associated with the seven TAGs is also most closely associated with better cognitive functioning. Further studies should be designed to determine if dietary interventions can increase circulating TAG-FA18:3 concentrations and if this could help to prevent cognitive impairment related to cortical thinning.
Methods
Study population
The Saguenay Youth Study (SYS) is a population-based study conducted in the Saguenay-Lac-St.-Jean region of Quebec, Canada; it is aimed at investigating the etiology and early stages of common cardiometabolic and brain diseases10. The SYS includes 486 families with a total of 1028 adolescents and 962 parents10. In the present work, we studied data from 441 SYS adults in whom brain magnetic resonance imaging (MRI) and lipidomics data were available, and who were not using lipid-lowering medications. Written consent was obtained from all participants, and the study was approved by local research ethics committees.
Cortical thickness estimation
T1-weighted (T1W) magnetic resonance imaging (MRI) of the brain was conducted using with a 1.5 Tesla Avanto (Siemens) scanner. The T1W images were acquired using the three-dimensional Magnetization Prepared Rapid Gradient Echo sequence with 176 sagittal slices (1-mm isotropic resolution, TR = 2.400 ms, TE = 2.65 ms, TI = 1000 ms, and flip angle = 8°). FreeSurfer (v 5.3.) cortical reconstruction was used to derive measures of cortical thickness and surface area in 34 regions (per hemisphere), as segmented using the Desikan-Killiany atlas14. The mean cortical thickness was calculated as average thickness over the 34 regions and two hemispheres, weighting for cortical area per region.
Serum lipidomics
All lipidomic measurements were performed using the TrueMass Complex Lipid PanelTM by Metabolon (Morrisville, USA). Lipids were extracted from fasted serum samples and their concentrations (μM) were determined in mass spectrometry analyses.
Majority (78%; N = 342) of the study participants had blood withdrawal for lipidomic quantifications and brain MRI scan done on the same day. The average time between blood withdrawal and MRI scan was 0.76 days ± 0.86 (SEM).
Statistics and reproducibility
Linear regression modeling
In the association analyses, mean cortical thickness was modeled as a function of the concentration values of each TAG species in linear regression models. Prior to these analyses, each variable was adjusted for age and sex, and inverse-rank-transformed to normality11. The transformed TAG concentrations have mean = 0 and standard deviation (SD) = 1, and thus the effect estimates presented in this paper are in SD-units. Owing to the high correlation of the TAG species concentrations (Supplementary Fig. 8), the number of the independent tests performed is lower than the number of TAGs tested. To estimate the number of independent tests performed, we performed principal component analysis of the TAG data35,36. We found that the first nine principal components explained 95% of variation in the TAG data and accordingly we considered statistical significance at p < 0.006 (0.05/9), as done previously11–13,37.
To study if the TAG associations with mean cortical thickness differ by FA chain length or degree of saturation, we determined the linear fit of the beta estimates derived from each of the 518 linear models and the number of carbons or the number of double bonds in the TAGs.
We performed further supplementary analyses as follows. For insight on whether FAs per se rather than TAGs drive these associations, we determined the associations between mean cortical thickness and (i) circulating concentrations of 21 FA species extracted from TAG fraction of lipids (TAG-FA), and (ii) circulating total concentrations of 28 FA species (total-FA) extracted from all protein or lipid carriers, including TAGs. To evaluate the impact of APOE ε4-carrier status on associations between mean cortical thickness and TAG concentrations, we additionally adjusted the linear regression models for APOE alleles as determined by rs429358 and rs7412 genotypes. In addition, we tested the associations between total cortical surface area and TAG species in linear models, where the surface area was modeled as a function of the concentration values of each TAG species. In all the supplementary analyses, adjustments and data transformations were the same as those described above.
For the seven TAG species that showed significant associations with mean cortical thickness, we used linear regression models to determine their associations with cortical thickness at each of the 34 cortical regions defined by FreeSurfer (mean of the two hemispheres). As the association profiles across the 34 regions were similar for all seven TAG species (Fig. 3), we derived the beta estimates from these models and calculated their averages in order to determine the “interregional average-beta profile” of the seven TAGs with the thickness at each of the 34 cortical regions (Supplementary Data 2).
Virtual histology and functional element enrichment analyses
“Virtual histology” is a method to study relative contributions of nine cell types (CA1 and S1 pyramidal neurons, interneuron, astrocyte, microglia, oligodendrocyte, as well as ependymal, endothelial, and mural cells) to observed differences in cortical thickness across the 34 regions defined by FreeSurfer15. To perform virtual histology analyses, the “interregional average-beta profile” of the seven TAGs across the 34 regions was tested for association with interregional profiles of the expression of genes specific to the nine cell types; this was done by comparing distributions of correlation coefficients (r) of the interregional average-beta profile with cell-specific gene-expression profiles to the null distribution obtained by shuffling the “cell-type” membership15,38.
Within each significant cell type, we tested the correlation coefficients for the “interregional average-beta profile” with interregional profiles of gene expression for all the genes in the cell type. The genes whose expression profiles were positively (r > 0) or negatively (r < 0) correlated with the “interregional average-beta profile” were tested for enrichment of biological processes or pathways using STRING v11.039.
Tests on cognitive performance
The study participants completed a computer-based battery of 12 tests designed to assess executive functioning, reasoning, working memory, and visual-spatial skills (Cambridge Brain Sciences platform, www.cambridgebrainscience.com)18. The 12 tests were (1) color-word remapping, (2) spatial planning, (3) self-ordered search, (4) paired associates learning, (5) digit span, (6) spatial span, (7) visuospatial working memory, (8) interlocking polygons, (9) feature match, (10) odd one out, (11) grammatical reasoning, and (12) spatial rotation. In addition, we determined the general factor “g” (G-Factor) representing the correlations in performance scores between all 12 cognitive tasks18.
We studied the associations of the performance in each test with thickness of the 34 cortical regions in linear regression models, where each test result or the G-Factor served as outcome and cortical thickness of each region as an explanatory variable. Prior to linear model fitting, all the variables were adjusted for age and sex and transformed using inverse-rank-based normal transformation. We sought to test if this interregional profile of the associations between cortical thickness and cognitive performance is correlated with the interregional profile of the associations between cortical thickness and the “interregional average-beta profile.” To do this, we determined correlation between the effect estimates derived from the two sets of association tests (i.e., the betas of thickness of the 34 cortical regions on cognitive performance vs. the betas of the seven TAGs on thickness of the 34 cortical regions).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Supplementary information
Description of Additional Supplementary Files
Acknowledgements
We thank the following individuals for their contributions in data acquisition and storage in the SYS: Manon Bernard (database architect, The Hospital for Sick Children) and Hélène Simard (CÉGEP de Jonquière). We thank all participants who took part in the Saguenay Youth Study. The Saguenay Youth Study has been funded by the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, Canadian Foundation for Innovation, and National Institutes for Health.
Author contributions
E.S., J.S., and Z.P. conceptualized the study. E.S. and J.S. analyzed the data and generated results. T.P. and Z.P. supervised the study. J.S., C.S., T.P., and Z.P. managed the SYS cohort and data. S.B., S.S., T.P., and Z.P. obtained funding and provided additional study resources. E.S. and Z.P. wrote the original manuscript. All authors contributed to revising the content and approved the final version.
Data availability
The summary statistics are available within the article and its data supplement. The individual level participant data analyzed in this study are available by application from the Saguenay Youth Study.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information is available for this paper at 10.1038/s42003-020-01189-5.
References
- 1.Bentsen H. Dietary polyunsaturated fatty acids, brain function and mental health. Microb. Ecol. Health Dis. 2017;28:1281916. [Google Scholar]
- 2.Rapoport SI, Chang MCJ, Spector AA. Delivery and turnover of plasma-derived essential PUFAs in mammalian brain. J. Lipid Res. 2001;42:678–685. [PubMed] [Google Scholar]
- 3.Bazinet RP, Layé S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014;15:771–785. doi: 10.1038/nrn3820. [DOI] [PubMed] [Google Scholar]
- 4.Kris-Etherton PM, Hecker KD, Binkoski AE. Polyunsaturated fatty acids and cardiovascular health. Nutr. Rev. 2004;62:414–426. doi: 10.1111/j.1753-4887.2004.tb00013.x. [DOI] [PubMed] [Google Scholar]
- 5.Tortosa-Caparrós E, Navas-Carrillo D, Marín F, Orenes-Piñero E. Anti-inflammatory effects of omega 3 and omega 6 polyunsaturated fatty acids in cardiovascular disease and metabolic syndrome. Crit. Rev. Food Sci. Nutr. 2017;57:3421–3429. doi: 10.1080/10408398.2015.1126549. [DOI] [PubMed] [Google Scholar]
- 6.Cardoso C, Afonso C, Bandarra NM. Dietary DHA and health: cognitive function ageing. Nutr. Res. Rev. 2016;29:281–294. doi: 10.1017/S0954422416000184. [DOI] [PubMed] [Google Scholar]
- 7.Schaefer EJ, et al. Plasma phosphatidylcholine docosahexaenoic acid content and risk of dementia and alzheimer disease: the framingham heart study. Arch. Neurol. 2006;63:1545–1550. doi: 10.1001/archneur.63.11.1545. [DOI] [PubMed] [Google Scholar]
- 8.Zhang Y, et al. Intakes of fish and polyunsaturated fatty acids and mild-to-severe cognitive impairment risks: a dose-response meta-analysis of 21 cohort studies. Am. J. Clin. Nutr. 2016;103:330–340. doi: 10.3945/ajcn.115.124081. [DOI] [PubMed] [Google Scholar]
- 9.Tracey TJ, Steyn FJ, Wolvetang EJ, Ngo ST. Neuronal lipid metabolism: multiple pathways driving functional outcomes in health and disease. Front. Mol. Neurosci. 2018;11:1–25. doi: 10.3389/fnmol.2018.00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pausova Z, et al. Cohort profile: The Saguenay Youth Study (SYS) Int. J. Epidemiol. 2017;46:e19. doi: 10.1093/ije/dyw023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sliz, E. et al. A variant near DHCR24 associates with microstructural properties of white matter and peripheral lipid metabolism in adolescents. Mol. Psychiatry10.1038/s41380-019-0640-9 (2020). [DOI] [PMC free article] [PubMed]
- 12.Kettunen J, et al. Genome-wide study for circulating metabolites identifies 62 loci and reveals novel systemic effects of LPA. Nat. Commun. 2016;7:11122. doi: 10.1038/ncomms11122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Millwood IY, et al. Association of CETP gene variants with risk for vascular and nonvascular diseases among Chinese adults. JAMA Cardiol. 2018;3:34–43. doi: 10.1001/jamacardio.2017.4177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.French L, Paus T. A FreeSurfer view of the cortical transcriptome generated from the Allen Human Brain Atlas. Front. Neurosci. 2015;9:1–5. doi: 10.3389/fnins.2015.00323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shin J, et al. Cell-specific gene-expression profiles and cortical thickness in the human brain. Cereb. Cortex. 2018;28:3267–3277. doi: 10.1093/cercor/bhx197. [DOI] [PubMed] [Google Scholar]
- 16.Ohkuni A, Ohno Y, Kihara A. Identification of acyl-CoA synthetases involved in the mammalian sphingosine 1-phosphate metabolic pathway. Biochem. Biophys. Res. Commun. 2013;442:195–201. doi: 10.1016/j.bbrc.2013.11.036. [DOI] [PubMed] [Google Scholar]
- 17.Mashek DG, Li LO, Coleman RA. Rat long-chain acyl-CoA synthetase mRNA, protein, and activity vary in tissue distribution and in response to diet. J. Lipid Res. 2006;47:2004–2010. doi: 10.1194/jlr.M600150-JLR200. [DOI] [PubMed] [Google Scholar]
- 18.Hampshire A, Highfield RR, Parkin BL, Owen AM. Fractionating human intelligence. Neuron. 2012;76:1225–1237. doi: 10.1016/j.neuron.2012.06.022. [DOI] [PubMed] [Google Scholar]
- 19.Yuan P, Raz N. Prefrontal cortex and executive functions in healthy adults: a meta-analysis of structural neuroimaging studies. Neurosci. Biobehav. Rev. 2014;42:180–192. doi: 10.1016/j.neubiorev.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dickerson BC, et al. Detection of cortical thickness correlates of cognitive performance: reliability across MRI scan sessions, scanners, and field strengths. Neuroimage. 2008;39:10–18. doi: 10.1016/j.neuroimage.2007.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Singh V, et al. Spatial patterns of cortical thinning in mild cognitive impairment and Alzheimer’s disease. Brain. 2006;129:2885–2893. doi: 10.1093/brain/awl256. [DOI] [PubMed] [Google Scholar]
- 22.Bernath, M. M. et al. Serum triglycerides in Alzheimer disease. Neurology94, e2088–e2098 (2020). [DOI] [PMC free article] [PubMed]
- 23.Krishnadas R, et al. Cardio-metabolic risk factors and cortical thickness in a neurologically healthy male population: Results from the psychological, social and biological determinants of ill health (pSoBid) study. NeuroImage Clin. 2013;2:646–657. doi: 10.1016/j.nicl.2013.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schwarz NF, et al. Differential associations of metabolic risk factors on cortical thickness in metabolic syndrome. NeuroImage Clin. 2018;17:98–108. doi: 10.1016/j.nicl.2017.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vessby B. Dietary fat, fatty acid composition in plasma and the metabolic syndrome. Curr. Opin. Lipidol. 2003;14:15–19. doi: 10.1097/00041433-200302000-00004. [DOI] [PubMed] [Google Scholar]
- 26.Dyerberg J, Madsen P, Møller JM, Aardestrup I, Schmidt EB. Bioavailability of marine n-3 fatty acid formulations. Prostaglandins Leukot. Essent. Fat. Acids. 2010;83:137–141. doi: 10.1016/j.plefa.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 27.Pélerin H, et al. Gene expression of fatty acid transport and binding proteins in the blood-brain barrier and the cerebral cortex of the rat: Differences across development and with different DHA brain status. Prostaglandins Leukot. Essent. Fat. Acids. 2014;91:213–220. doi: 10.1016/j.plefa.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 28.Banks WA, et al. Triglycerides cross the blood-brain barrier and induce central leptin and insulin receptor resistance. Int. J. Obes. 2018;42:391–397. doi: 10.1038/ijo.2017.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Clark KJ, Makrides M, Neumann MA, Gibson RA. Determination of the optimal ratio of linoleic acid to alpha-Iinolenic acid in infant formulas. J. Pediatr. 1992;120:S151–S158. doi: 10.1016/S0022-3476(05)81250-8. [DOI] [PubMed] [Google Scholar]
- 30.Gazzah N, et al. Decrease of brain phospholipid synthesis in free‐moving n‐3 fatty acid deficient rats. J. Neurochem. 1995;64:908–918. doi: 10.1046/j.1471-4159.1995.64020908.x. [DOI] [PubMed] [Google Scholar]
- 31.Igarashi M, et al. Docosahexaenoic acid synthesis from α-linolenic acid by rat brain is unaffected by dietary n-3 PUFA deprivation. J. Lipid Res. 2007;48:1150–1158. doi: 10.1194/jlr.M600549-JLR200. [DOI] [PubMed] [Google Scholar]
- 32.Naudí A, et al. Lipidomics of human brain aging and alzheimer’s disease pathology. Int. Rev. Neurobiol. 2015;122:133–189. doi: 10.1016/bs.irn.2015.05.008. [DOI] [PubMed] [Google Scholar]
- 33.Kim H, et al. Oleic acid ameliorates Aβ-induced inflammation by downregulation of COX-2 and iNOS via NFκB signaling pathway. J. Funct. Foods. 2015;14:1–11. doi: 10.1016/j.jff.2015.01.027. [DOI] [Google Scholar]
- 34.Snowden SG, et al. Association between fatty acid metabolism in the brain and Alzheimer disease neuropathology and cognitive performance: a nontargeted metabolomic study. PLoS Med. 2017;14:1–19. doi: 10.1371/journal.pmed.1002266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gao X, Starmer J, Martin ER. A multiple testing correction method for genetic association studies using correlated single nucleotide polymorphisms. Genet. Epidemiol. 2008;32:361–369. doi: 10.1002/gepi.20310. [DOI] [PubMed] [Google Scholar]
- 36.Johnson RC, et al. Accounting for multiple comparisons in a genome-wide association study (GWAS) BMC Genomics. 2010;11:724. doi: 10.1186/1471-2164-11-724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sliz E, et al. NAFLD risk alleles in PNPLA3, TM6SF2, GCKR and LYPLAL1 show divergent metabolic effects. Hum. Mol. Genet. 2018;27:2214–2223. doi: 10.1093/hmg/ddy124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Patel Y, Shin J, Gowland PA, Pausova Z, Paus T. Maturation of the human cerebral cortex during adolescence: myelin or dendritic arbor? Cereb. Cortex. 2019;29:3351–3362. doi: 10.1093/cercor/bhy204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Szklarczyk, D. et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 47, D607–D613 (2019). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
The summary statistics are available within the article and its data supplement. The individual level participant data analyzed in this study are available by application from the Saguenay Youth Study.