Abstract
In the present study, we explored phytochemical constituents of Tinospora cordifolia in terms of its binding affinity targeting the active site pocket of the main protease (3CL pro) of SARS-CoV-2 using molecular docking study and assessed the stability of top docking complex of tinosponone and 3CL pro using molecular dynamics simulations with GROMACS 2020.2 version. Out of 11 curated screened compounds, we found the significant docking score for tinosponone, xanosporic acid, cardiofolioside B, tembetarine and berberine in Tinospora cordifolia. Based on the findings of the docking study, it was confirmed that tinosponone is the potent inhibitor of main protease of SARS-CoV-2 with the best binding affinity of −7.7 kcal/mol. Further, ADME along with toxicity analysis was studied to predict the pharmacokinetics and drug-likeness properties of five top hits compounds. The molecular dynamics simulation analysis confirmed the stability of tinosponone and 3CL pro complex with a random mean square deviation (RMSD) value of 0.1 nm. The computer-aided drug design approach proved that the compound tinosponone from T. cordifolia is a potent inhibitor of 3CL main protease of SARS-CoV-2. Further, the in vitro and in vivo-based testing will be required to confirm its inhibitory effect on SARS-CoV-2.
Communicated by Ramaswamy H. Sarma
Keywords: Main protease, docking, Tinospora cordifolia, tinosponone, xanosporic acid
Introduction
Coronaviruses are positive-sense RNA viruses with non-segmented genomes and overlapping Open reading frame (ORF 1a and 1b) with a size genome structures ranging from 28 to 32 KB. The polyproteins are processed into non-structural proteins (NSP) and structural proteins. Coronaviruses regularly undergo recombination after infecting host cells (Lauber et al., 2013). The proteases of novel coronavirus share high percentage of sequence and structure similarity with SARS coronavirus which has the main role in the processing of viral polyprotein. These proteins perform stripping ubiquitin to help in evading immune response of the host (Shanker et al., 2020). Small molecules and drug inhibitors can be designed to block the cascade signaling pathways to protect the host system. Viral proteins have multiple functional domains such as a largest NSP-3 protein and proteases like protease and papain-like protease (3CLpro and PLpro) are involved in polyprotein processing (Báez-Santos et al., 2015). Novel coronavirus infection leads to over activation of CD4 effector T-cells resulting in excessive production of inflammatory cytokines termed as cytokine storm that recruits excess immune cells at the site of infection and reduces tissue capacity which leads to organ failure (Felsenstein et al., 2020). Plant-based immuno-modulatory agents would reduce the severity of hyperactivation of inflammatory response and reduce cytokine storm. Several medicinal plant extracts and their constituents with immuno-modulatory activities proved their ability to reduce the over inflammatory response of cytokines (Liu et al., 2016). Tinospora cordifolia which is a medicinal plant native to India commonly called as Guduchi, heart-leaved moonseed and used in Ayurvedic formulations as a medicine to treat several diseases. Tinospora cordifolia is found to have antineoplastic, antidiabetic, hypolipidemic, antioxidant and anti-inflammatory effects. Phytochemical studies of T. cordifolia showed the presence of numerous components such as tinosponone, tinosporin, berberine, palmitine, choline, tembetarine, isocolumbin and tetrahydropalmatine and other alkaloids, steroids, lactones, glycosides and sesquiterpenoids. Tinospora cordifolia has an effect on the expression of cyclooxygenase-1, tumor necrosis factor-α, iNOS genes, cytokines (IL-6, IL-1b and PGE2) and NF-kB activation and has the potential to reduce the expression of the COX-2 enzyme and thereby reducing the expression of TNF-α, synthesis of IL-1B and IL-6 and other pro-inflammatory interleukins (Tiwari et al., 2018). The phytosterols such as stigmasterol and β-sitosterol found in T. cordifolia are found to be responsible for the inhibition of cyclooxygenase-2 synthesis and adhesion molecules in TNF-α stimulated by cells (Philip et al., 2018). Stigmasterol inhibits prostaglandin E2 and other matrix degradation mediators (Joshi & Kaur, 2016; Sharma et al., 2019; Singh & Chaudhuri, 2017; Tiwari et al., 2018; Upadhyay et al., 2010; Yanez et al., 2017). The phytochemicals of medicinal plants have anti-viral properties and hence, in the present study, we explored the inhibitory effect of phytochemicals of T. cordifolia against the SARS-CoV-2 by computational analysis.
Methodology
Protein preparation for docking
The main protease (3CLpro) in complex with an inhibitor N3 was selected from PDB for the present study. The ligands bound to the protein were removed using Chimera. Structure-based virtual screening was performed using PyRx, Main protease (Mpro, aka 3CLpro, 3-chymotrypsin-like protease, (PDB ID: 6lu7) was used as the macromolecule (receptor). The amino acids interacting with the old ligand were considered as the active site in the generated protein and receptor grid (Peele et al., 2020).
Ligand preparation and virtual screening
The phytochemicals, namely phytosterols, berberine, cardiofolioside B, chasmanthin, columbin, cordifolide A, palmarin, tembetarine, tinosponone, tinosporinone, xanosporic acid of T. cordifolia was downloaded from Indian Medicinal Plants, Phytochemistry and Therapeutics (IMPPAT) database (https://cb.imsc.res.in/imppat/home) in 3D-SDF format. IMPPAT is a curated database of Indian medicinal plants, phytochemical and therapeutic associations (Mohanraj et al., 2018). Energy minimized was performed using Chimera (http://www.cgl.ucsf.edu/chimera). Hydrogen atoms were added before docking and the top-ranked two ligand poses of T. cordifolia phytochemical compounds bound to the active site pocket of main protease were used for further analysis. The top hits were analyzed for finding ADME properties using the pkCSM online server (http://biosig.unimelb.edu.au/pkcsm/prediction).
Molecular dynamics simulations
The complex of main protease and the top-ranking phytochemical drug candidate with best docking score was prepared for MD simulations and run for 10 ns using GROMACS 2020.2 using CHARMM 36 force field. Topology generation and energy minimization of ligand were performed using the CGenFF server. The trajectories files were generated and saved. The random mean square deviation (RMSD) of the complex with respect to time frame was plotted in the graph (Abraham Peele et al., 2020).
Results and discussion
Molecular docking for phytochemicals of T. cordifolia
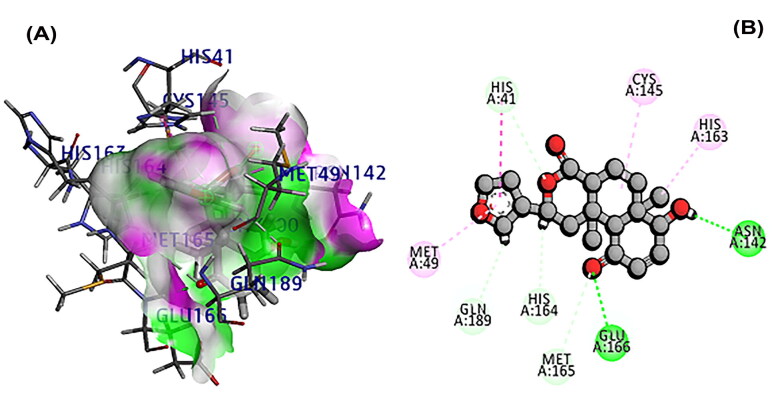
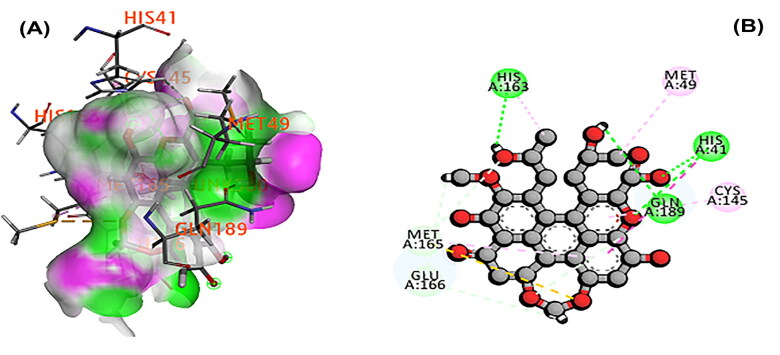
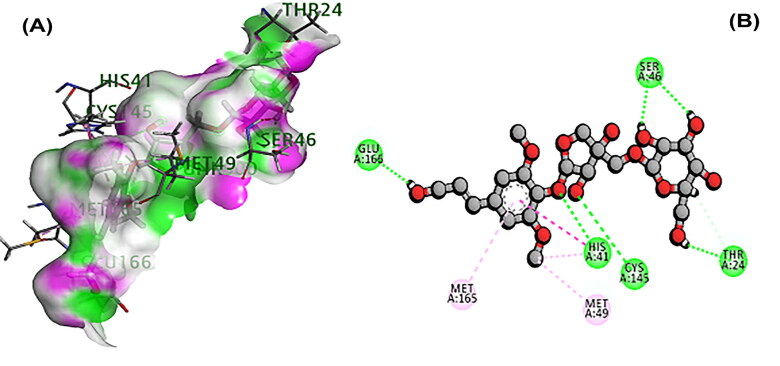
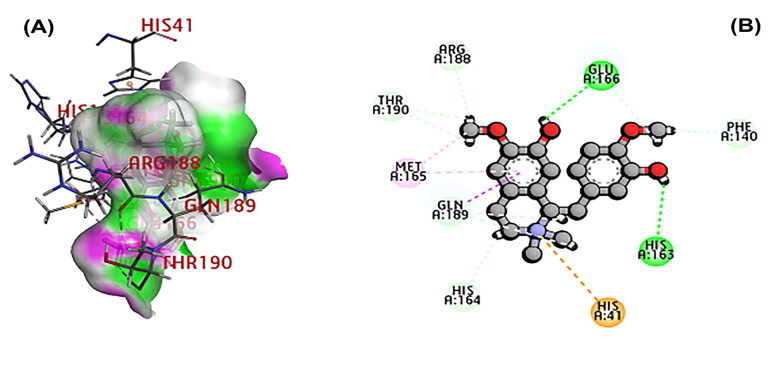
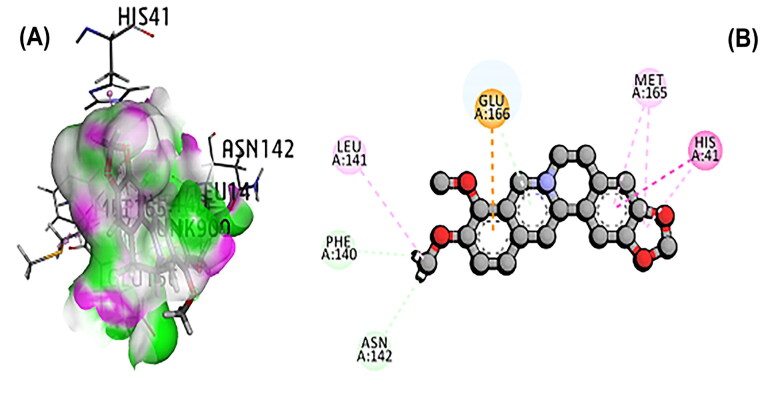
The 3CL main protease is essential for SARS-CoV-2 replication and considered as crucial target for developing the drug (Peele et al., 2020; Wu et al., 2020; Zhang et al., 2020). A total of 11 phytochemicals of T. cordifolia was screened by in silico analysis. Docking results proved that the top 5 potential phytochemical molecules of T. cordifolia could be considered as 3CLpro inhibitors. The stable docking poses of tinosponone, xanosporic acid cardiofolioside B, tembetarine and berberine with main protease showed the best docking scores, namely −7.7, −7.5, −7.3, −6.6 and −6.5 kcal/mol, respectively and the interactions were analyzed using 2D diagram. Based on the lowest docking energy value of docked complex, tinosponone was selected as potent inhibitor molecule (Table 1). The compound tinosponone showed hydrogen bond interactions with GLU166 and ASN 142 (Figure 1(a,b)). Xanosporic acid showed hydrogen bond interactions with HIS 41, HIS 163 and GLN 189 (Figure 2(a,b)). Cardiofolioside B showed interactions of hydrogen bond with SER 46, HIS 41, CYS 145, THR 24 (Figure 3(a,b)). Tembetarine showed hydrogen bond interactions with GLU166, HIS 163 (Figure 4(a,b)). Berberine showed hydrogen bond interactions with PHE 140 and ASN 142 (Figure 5(a,b)).
Table 1.
ADME properties of phytochemicals of T. cordifolia along with their binding energies.
| S. No. | Phytochemicals | Molecular weight (g/mol) | No. of hydrogen bond donors | No. of hydrogen bond acceptors | Topological polar surface area (Å2) | Partition coefficient (Log P) | Docking score (Kcal/mol) |
|---|---|---|---|---|---|---|---|
| 1 | Tinosponone | 330.38 | 1 | 5 | 76.74 | 2.81 | −7.7 |
| 2 | Xanosporic acid | 536.49 | 5 | 10 | 176.12 | 3.38 | −7.5 |
| 3 | Cardiofolioside B | 504.48 | 7 | 13 | 196.99 | −2.65 | −7.3 |
| 4 | Tembetarine | 344.43 | 2 | 4 | 58.92 | 3.03 | −6.6 |
| 5 | Berberine | 336.37 | 0 | 1 | 40.80 | 3.10 | −6.5 |
Figure 1.
(A) Docked pose of tinosponone with 3CL main protease. (B) Ligand interaction of tinosponone with 3CL main protease.
Figure 2.
(A) Docked pose of xanosporic acid with 3CL main protease. (B) Ligand interaction of xanosporic acid with 3CL main protease.
Figure 3.
(A) Docked poses of cardiofolioside B with 3CL main protease. (B) Ligand interaction of cardiofolioside B with 3CL main protease.
Figure 4.
(A) Docked poses of tembetarine molecule with 3CL main protease. (B) Ligand interaction of tembetarine with 3CL main protease.
Figure 5.
(A) Docked pose of berberine with 3CL main protease. (B) Ligand interaction of berberine with 3CL main protease.
ADME prediction
The top 5 phytochemicals of T. cordifolia shown better binding affinity with 3CL main protease was subjected to ADME analysis. The compound, namely tinasponone has satisfied Lipinski’s rule of five conditions and the data were represented in Table 1. Tinasponone possessed good pharmacokinetic properties of druggability with better permeability through membranes and does not interfere with the sites of CYP450 metabolism of other drugs. The physicochemical properties of tinosponone have revealed that the molecular weight 330.38 g/mol with five hydrogen bond acceptors and one hydrogen bond donor, topological polar surface area (TPSA), 76.74Å2 and lipophilicity, LogP 2.81. AMES test for toxicity prediction of tinasponone by pkCSM server has shown that the tinasponone has no toxicity.
Molecular dynamics simulations
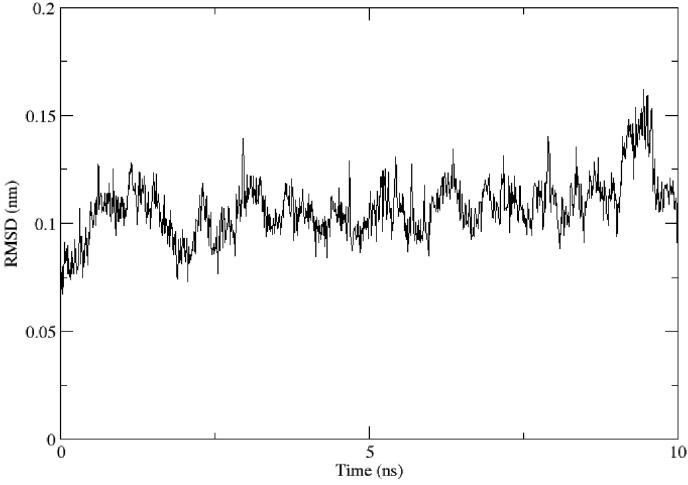
The nature of binding was studied using molecular dynamics simulation study. The top-ranked drug, tinosponone with main protease complex has shown the stability conformation throughout the running of simulation for 10 ns and the RMSD value was obtained below 0.2 nm. The average deviation was found to be negligible, i.e. 0.12 nm (Figure 6). Hence, from the present study on in silico approach, it is suggested that the best docked phytochemical compounds could be tested biologically to develop potent drugs against novel coronavirus.
Figure 6.
RMSD analysis for top-ranked lead tinosponone and main protease complex.
Conclusion
Based on virtual screening and molecular docking analysis, the phytochemical compounds, namely tinosponone, xanosporic acid, cardiofolioside B, tembetarine and berberine of T. cordifolia were identified as possible lead molecules to fight against SARS-CoV-2. The present in silico study proved that tinosponone as potent, selective and nontoxic inhibitor of 3CL protease of SARS-CoV-2.
Acknowledgment
The authors acknowledge the facilities rendered by Vignan’s Foundation for Science, Technology and Research (Deemed to be University), Vadlamudi-522213 and DST-FIST networking facility.
Correction Statement
This article has been republished with minor changes. These changes do not impact the academic content of the article.
Disclosure statement
No potential conflict of interest is reported by the authors.
References
- Abraham Peele, K., Srihansa, T., Krupanidhi, S., Vijaya Sai, A., & Venkateswarulu, T. C. (2020). Design of multi-epitope vaccine candidate against SARS-CoV-2: A in-silico study. Journal of Biomolecular Structure & Dynamics, 1–10. 10.1080/07391102.2020.1770127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Báez-Santos, Y. M., John, S. E. S., & Mesecar, A. D. (2015). The SARS-coronavirus papain-like protease: Structure, function and inhibition by designed antiviral compounds. Antiviral Research, 115, 21–38. 10.1016/j.antiviral.2014.12.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Felsenstein, S., Herbert, J. A., McNamara, P. S., & Hedrich, C. M. (2020). COVID-19: Immunology and treatment options. Clinical Immunology, 215, 108448. 10.1016/j.clim.2020.108448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi, G., & Kaur, R. (2016). Tinospora cordifolia: A phytopharmacological review. International Journal of Pharmaceutical Sciences and Research, 7(3), 890–897. 10.13040/IJPSR.0975-8232 [DOI] [Google Scholar]
- Lauber, C., Goeman, J. J., del Carmen Parquet, M., Nga, P. T., Snijder, E. J., Morita, K., & Gorbalenya, A. E. (2013). The footprint of genome architecture in the largest genome expansion in RNA viruses. PLoS Pathogens, 9(7), e1003500. 10.1371/journal.ppat.1003500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, Q., Zhou, Y. H., & Yang, Z. Q. (2016). The cytokine storm of severe influenza and development of immunomodulatory therapy. Cellular & Molecular Immunology, 13(1), 3–10. 10.1038/cmi.2015.74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohanraj, K., Karthikeyan, B. S., Vivek-Ananth, R. P., Chand, R. B., Aparna, S. R., Mangalapandi, P., & Samal, A. (2018). IMPPAT: A curated database of Indian Medicinal Plants, Phytochemistry and Therapeutics. Scientific Reports, 8(1), 1–17. 10.1038/s41598-018-22631-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peele, K. A., Potla Durthi, C., Srihansa, T., Krupanidhi, S., Sai, A. V., Babu, D. J., Indira, M., Reddy, A. R., & Venkateswarulu, T. C. (2020). Molecular docking and dynamic simulations for antiviral compounds against SARS-CoV-2: A computational study. Informatics in Medicine Unlocked, 19, 100345–100346. 10.1016/j.imu.2020.100345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philip, S., Tom, G., & Vasumathi, A. V. (2018). Evaluation of the anti-inflammatory activity of Tinospora cordifolia (Willd.) Miers chloroform extract – A preclinical study. The Journal of Pharmacy and Pharmacology, 70(8), 1113–1125. 10.1111/jphp.12932 [DOI] [PubMed] [Google Scholar]
- Shanker, A. K., Bhanu, D., Alluri, A., & Gupta, S. (2020). Whole-genome sequence analysis and homology modelling of the main protease and non-structural protein 3 of SARS-CoV-2 reveal an aza-peptide and a lead inhibitor with possible antiviral properties. New Journal of Chemistry, 44(22), 9202–9212. 10.1039/D0NJ00974A [DOI] [Google Scholar]
- Sharma, P., Dwivedee, B. P., Bisht, D., Dash, A. K., & Kumar, D. (2019). The chemical constituents and diverse pharmacological importance of Tinospora cordifolia. Heliyon, 5(9), e02437. 10.1016/j.heliyon.2019.e02437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh, D., & Chaudhuri, P. K. (2017). Chemistry and pharmacology of Tinospora cordifolia. Natural Product Communications, 12(2), 299–308. 10.1177/1934578X1701200240 [DOI] [PubMed] [Google Scholar]
- Tiwari, P., Nayak, P., Prusty, S. K., & Sahu, P. K. (2018). Phytochemistry and pharmacology of Tinospora cordifolia: A review. Systematic Reviews in Pharmacy, 9(1), 70–78. 10.5530/srp.2018.1.14 [DOI] [Google Scholar]
- Upadhyay, A. K., Kumar, K., Kumar, A., & Mishra, H. S. (2010). Tinospora cordifolia (Willd.) Hook. f. and Thoms. (Guduchi) – Validation of the Ayurvedic pharmacology through experimental and clinical studies. International Journal of Ayurveda Research, 1(2), 112–121. 10.4103/0974-7788.64405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, C., Liu, Y., Yang, Y., Zhang, P., Zhong, W., Wang, Y., Wang, Q., Xu, Y., Li, M., Li, X., Zheng, M., Chen, L., & Li, H. (2020). Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharmaceutica Sinica B, 10(5), 766–788. 10.1016/j.apsb.2020.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanez, M., Blanchette, J., & Jabbarzadeh, E. (2017). Modulation of inflammatory response to implanted biomaterials using natural compounds. Current Pharmaceutical Design, 23(41), 6347–6357. 10.2174/1381612823666170510124348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, L., Lin, D., Sun, X., Curth, U., Drosten, C., Sauerhering, L., Becker, S., Rox, K., & Hilgenfeld, R. (2020). Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved α-ketoamide inhibitors. Science, 368(6489), 409–412. 10.1126/science.abb3405 [DOI] [PMC free article] [PubMed] [Google Scholar]