Abstract
Objective
The SARS-CoV-2 pathogen has established endemicity in humans. This necessitates the development of rapid genetic surveillance methodologies to serve as an adjunct with existing comprehensive, albeit though slower, genome sequencing-driven approaches.
Methods
A total of 21,789 complete genomes were downloaded from GISAID on May 28, 2020 for analyses. We have defined the major clades and subclades of circulating SARS-CoV-2 genomes. A rapid sequencing-based genotyping protocol was developed and tested on SARS-CoV-2-positive RNA samples by next-generation sequencing.
Results
We describe 11 major mutations which defined five major clades (G614, S84, V251, I378 and D392) of globally circulating viral populations. The clades can specifically identify using an 11-nucleotide genetic barcode. An analysis of amino acid variation in SARS-CoV-2 proteins provided evidence of substitution events in the viral proteins involved in both host entry and genome replication.
Conclusion
Globally circulating SARS-CoV-2 genomes could be classified into 5 major clades based on mutational profiles defined by an 11-nucleotide barcode. We have successfully developed a multiplexed sequencing-based, rapid genotyping protocol for high-throughput classification of major clade types of SARS-CoV-2 in clinical samples. This barcoding strategy will be required to monitor decreases in genetic diversity as treatment and vaccine approaches become widely available.
Keywords: SARS-CoV-2, genetic surveillance, barcoding, genome variation
Introduction
The SARS-CoV-2 infection has now reached 188 countries across all continents, except Antarctica. A pandemic was declared by the World Health Organization (WHO) on March 11, 2020 and as of August 11, 2020 more than 20 million cases have been confirmed globally (Dong et al. 2020). SARS-CoV-2 belongs to the Coronaviridae family, genus Betacoronavirus, which are enveloped, positive-sense, single-stranded RNA viruses, of zoonotic origin. Among human RNA viruses, coronaviruses have the largest known genome (∼30 kb), which encodes the structural proteins (spike, envelope, membrane and nucleocapsid), nonstructural proteins (nsp1-16), and accessory proteins (ORF3a, ORF6, ORF7a and b, ORF8, ORF10). Structural proteins are required for host cell entry, viral assembly and exit (Bárcena et al., 2009, Hoffmann et al., 2020). Nonstructural proteins are involved in genome replication-transcription and formation of vesicles (Hagemeijer et al. 2014), whereas accessory proteins interfere with host innate defense mechanisms (Wu et al., 2020a, Wu et al., 2020b, Liu et al., 2014). During genome replication within the host, the virus acquires genome mutations, which can be passed on to virus progeny in subsequently infected individuals.
More than 52,600 complete and high coverage genomes are available on GISAID as of August 11. Systematically tracking of mutations in SARS-CoV-2 genomes is therefore important as it allows monitoring of the molecular epidemiology of circulating viral sequences nationally and internationally. Here, we have investigated the nucleotide variation landscape of a large set of globally derived SARS-CoV-2 genomes and defined major mutation events. This analysis allowed us to produce a first-generation genetic classifier, or ‘barcode’, defining the major clades of the virus circulating up to May 28, 2020. Notably, this barcode allowed reliable tracking of the spatial distribution and prevalence of these viral clades over time. While most of the nonsynonymous mutation events appear neutral with respect to protein function and stability, we also found evidence of mutations in the spike protein that may modulate interactions between SARS-CoV-2 and the human host.
Materials and Methods
Phylogenomic Analysis
1,427 coronavirus genomes were downloaded from Virus Pathogen Database and Analysis Resource (ViPR)(Pickett et al. 2012) on February 14, 2020, including 329 SARS, 35 SARS-CoV-2, 61 HCoV-NL63, 521 MERS, 52 HCoV-HKU1, 170 HCoV-OC43, 97 bovine coronaviruses and 61 mouse hepatitis viruses. Sequence alignment was performed using MAFFT (version 7.407) (Katoh and Standley 2013) software and then trimmed by trimAL (version 1.4.1) (Capella-Gutiérrez et al. 2009). Phylogenetic analyses of the complete genomes were done with FastTree (version 2.1.10) (Price et al. 2009) software with default parameters, and iTOL (version 5) (Letunic and Bork 2007) was used for phylogenetic tree visualisation.
21,789 complete SARS-CoV2 genomes were downloaded from GISAID (Shu and McCauley 2017) (May 28, 2020). Sequence alignment was performed with MAFFT (version 7.407) (Katoh and Standley 2013) and trimmed by trimAL (version 1.4.1) (Capella-Gutiérrez et al. 2009). Phylogenetic analyses of the complete genomes were performed with RAxML (version 8.2.12) (Stamatakis 2014) with 10,000 bootstrap replicates, employing the GTRCAT model. A random set of genomes from each defined subclade was used to generate a simplified phylogenetic tree using the same method. iTOL (version 5) (Letunic and Bork 2007) was used for the phylogenetic tree visualisation. Single nucleotide polymorphisms (SNPs) from each of the genomes were called by Parsnp (version 1.2) from the Harvest suite (Treangen et al. 2014) using MN908947.3 as the reference genome, and the SNPs were further annotated by SnpEff (version 4.3 m) (Cingolani et al. 2012). To compare the lineages recently defined by Rambaut and colleagues (Rambaut et al. 2020), Pangolin software (version 1.1.2) was applied to assign the assemblies belonging to each clade in our study.
Sequencing-based multiplexed genotyping
Sequencing-based multiplexed genotyping primers (Table 1 ) were designed and tested on SARS-CoV-2-positive RNA samples collected from nasopharyngeal swabs of critical patients in Sulaiman Alhabib Hospital from Riyadh, Saudi Arabia. The swab was placed in 1 mL of TRIzol (Ambion, USA) and transported to King Abdullah University of Science and Technology (KAUST) for further downstream applications. RNA was extracted using the Direct-zol RNA Miniprep kit (Zymo Research, USA) following the manufacturer’s instructions. RT-PCR was done using the one-step SuperScript III with Platinum Taq DNA Polymerase (Thermo Fisher, USA) on the 7900 H T Fast Real-Time PCR instrument (Applied Biosystems, USA). We chose 24 SARS-CoV-2 positive samples and generated cDNA using the VILOTM SuperscriptTM III kit (ThermoFisher, USA) and amplicons encompassing clade-specific SNPs were produced with the Q5 Hot Start High-Fidelity Master Mix (NEB, USA). The thermocycling conditions employed in this study were as follows: 1 cycle of 98 °C for 30 sec followed by 35 cycles of 98 °C for 10 sec,64 °C for 30 sec and 72 °C for 30 sec, followed by the final extension at 72 °C for 2 min. The PCR products were elecctrophoesed on a 2% (w/v) agarose gel. The Illumina library preparation was performed using the ARTIC V3 PCR tiling protocol (https://artic.network/ncov-2019) with 20% PhiX control and a loading concentration of 8 pmol. The prepared library were sequenced using on a MiSeq platform using a 600 cycle V3 kit with paired-end sequencing. To facilities the virus clade typing from the data generated by MiSeq, a Python script was developed using the mapped. bam file as the input. The barcode and clade information can be obtained using the script from the. vcf files generated by bcftools outputs (https://github.com/raeece/sarscov2barcode).
Table 1.
Primer sets for targeted multiplex PCR
| Clade | Position in the genome | Primer sequence | Amplicon size (bp) | Melting temperature Tm (°C) |
|---|---|---|---|---|
| G614 | 241 | GF-1: 5’- TGTCGTTGACAGGACACGAG-3’ GR-1: 5’- TCCTCCACGGAGTCTCCAAA-3’ |
228 | 60.94 59.33 |
| G614 | 3037 | GF-2: 5’- ATGAGTTCGCCTGTGTTGTG-3’ GR-2: 5’- TGTCTGATTGTCCTCACTGCC-3’ |
392 | 58.77 60.00 |
| G614 | 14408 | GF-3: 5’ - TGGGATCAGACATACCACCCA - 3’ GR-3: 5’ - GTGCAGCTACTGAAAAGCACG - 3’ |
334 | 60.27 60.40 |
| G614 | 23403 | GF-4: 5’ -CTGATGCTGTCCGTGATCCA - 3’ GR-4: 5’ - ACTAGCGCATATACCTGCACC - 3’ |
302 | 59.82 60.00 |
| S84 | 8782 | SF-1: 5’- GCGTCATATTAATGCGCAGGT-3’ SR-1: 5’- GCAGCCAAAACACAAGCTGA-3’ |
663 | 59.47 59.90 |
| S84 | 28144 | SF-2: 5’ - CGTGGATGAGGCTGGTTCTA - 3’ SR-2: 5’ - CCCACTGCGTTCTCCATTCT - 3’ |
300 | 59.18 60.04 |
| V251 | 26144 | VF-1: 5’- TCAGGTGATGGCACAACAAGT-3’ VR-1: 5’- GTACGCACACAATCGAAGCG-3’ |
468 | 60.13 60.25 |
| I378 | 1397 | IF-1: 5’ – GAAACTTCATGGCAGACGGG-3’ IR-1: 5’ – GCTAGCACGTGGAACCCAAT-3’ |
303 | 59.20 60.68 |
| I378 | 28688 | IF-2: 5’ - ACCGCTCTCACTCAACATGG - 3’ IR-2: 5’ - GCAGTACGTTTTTGCCGAGG - 3’ |
632 | 60.04 60.11 |
| D392 | 1440 | DF-1: 5’ - AGGTGCCACTACTTGTGGTT - 3’ DR-1: 5’ - AGTTTCAAGAGTGCGGGAGA - 3’ |
607 | 59.16 58.95 |
| D392 | 2891 | DF-2: 5’ - CGGTGCACCAACAAAGGTTAC - 3’ DR-2: 5’ - GCAGAAGTGGCACCAAATTCC - 3’ |
450 | 60.27 60.34 |
We calculated the maximum throughput of samples for genotyping on the MiSeq run based on the following assumptions: (i) targeted sequencing depth of at least 100X, pair-end reads across 10 positions per sample except the position 28688 for which the targeted depth was 30X, (ii) MiSeq V3 kit generates 44-50 million reads and 70% of the total reads are higher than Q30 as specified by Illumina (https://www.illumina.com/systems/sequencing-platforms/miseq/specifications.html).
Protein Structure Analysis
Experimentally determined protein structures were obtained from the Protein Data Bank (PDB). SwissModel (Arnold et al. 2006), I-Tasser (Yang et al. 2014), RaptorX (Källberg et al. 2014) and an in-house modelling pipeline was used to produce protein structure homology models. Phobius (Käll et al. 2007) was used for prediction of trans-membrane regions. RaptorX was also used for predicting secondary structure, protein disorder and solvent exposure of amino acids. Pymol (version 1.8.6.2) was used for visualisation.
Results
Five clades of SARS-CoV-2 are characterised by 11 major mutations across the globe
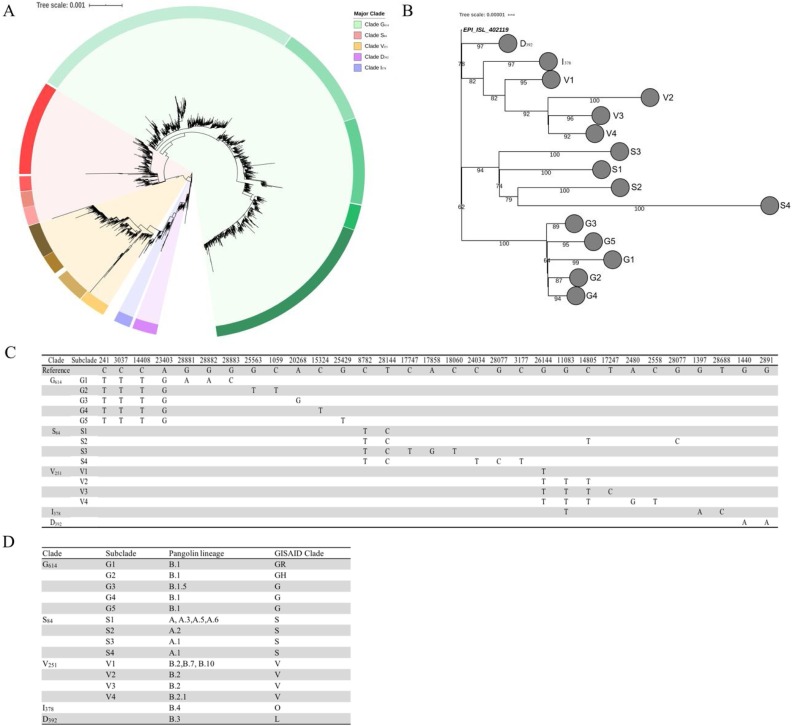
The SARS-CoV-2 genome is genetically most closely related to horseshoe bat SARS-related coronaviruses (Lu et al., 2020, Zhu et al., 2019) (SARSr-CoV), 96% to RaTG13, 93% to RmYN02(Zhou et al. 2020) and 88% to bat-SL-CoVZC45 and bat-SL-CoVZXC21 and also to SARSr-CoVs from pangolins (Tang et al. 2020) (Fig. S1). We studied their chronological occurrences of SARS-CoV-2 genomes during the early stages of the pandemic and global spread of the SARS-CoV-2 across human populations, which allowed us to define several clades of the virus that share unique SNPs. We observed 8,427 SNPs in the current dataset (May 28, 2020) and we defined five major clades which contains 15 subclades (Fig.1 , Fig.S2) compared to the prototype (MN908947.3). The clades were named by the respective amino acid mutations: S84 (Orf8, L84S), V251 (Orf3a, G251V), I378 (Orf1ab, V378I), D392 (Orf1ab, G392D) and G614 (S, D614G). A collapsed simplified phylogenetic tree to illustrate the correspondence of the subclades is shown in Fig.1B along with the clade-defining mutations (Fig.1C). Recent work has defined (Rambaut et al. 2020) 2 viral lineages - A and B (analogous to clades L and S described in (Tang et al. 2020)). The major clades proposed here correspond to lineage A (Clade S84), lineage B.2, B.7 and B.10 (Clade V251), lineage B.1 (Clade G614), lineage B.4 (Clade I378), B.3 (Clade D392) (Fig.1D). Due to intrinsic virus mutation rates, further refinements in nomenclature will likely need be defined in the future; however, a robust, high throughput barcoding scheme will be required to breach the surveillance gap and lag time between molecular diagnostics and full-genome sequencing to provide actionable genetic surveillance data.
Fig. 1.
Clades of SARS-CoV-2. (A) A global SNP-based radial phylogeny of SARS-CoV-2 genomes defining five major clades (G614, S84, V251, I378 and D392) and several subclades based on nucleotide substitution events. (B) A simplified phylogenetic tree to illustrate the evolutionary relationship of the clades/subclades based on random sampling of complete genomes from each subclade. (C) The clade and subclade-defining SNPs for each clade and subclade. *These SNPs are developed independently in more than one clade hence are not clade-defining SNPs (refer to Fig. S2). (D) A comparative guide to clades defined by our study and the lineages recently defined by Rambaut et al. (Rambaut et al. 2020) and GISAID (Shu and McCauley 2017).
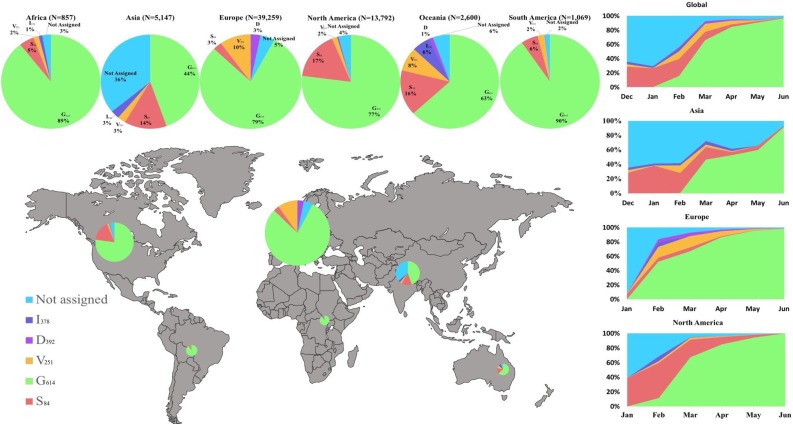
To obtain a global picture of the regional distribution of the clades over time during 6 month period (December 24, 2019 to June 30, 2020), we plotted the relative proportions of the major clades and their cumulative trends (Fig.2 ). We observed major differences in the apparent spread for individual clades: Clade G614 represents 71.14% of all the sequenced viral sequences, followed by S84 (10.85%), V251 (7.66%), D392 (1.03%) and I378 (1.70%). The remaining 7.6% were not assigned to a major clade. Clade G614 is widely distributed globally; whereas Clade S84 represents 17% of North American sampled genomes, and 16% of those from Oceania. Asia has the greatest number of unassigned genomes (36%). The global and regional cumulative trends were plotted over time, with the majority of the trends revealing the increasing dominance of one or two clades in each geographic region in the sampled genomes (Fig.2). This is likely attributable to founder effects during the early phases of the seeding of the local epidemics from imported cases and subsequent dissemination in the regions.
Fig. 2.
Global distribution of various major and minor clades of SARS-CoV2 genomes and their relative prevalence over a 6 month period from December 24, 2019 to June 30, 2020 from the outbreak and early stages of the pandemic. The size of each pie chart is proportional to the numbers within each respective clade. The cumulative trend of the clades is shown on the right and the span of time indicates the first and last observed case in each particular clade.
Newly sequenced SARS-CoV-2 genomes could be assigned to clades using a rapid NGS-based genotyping protocol
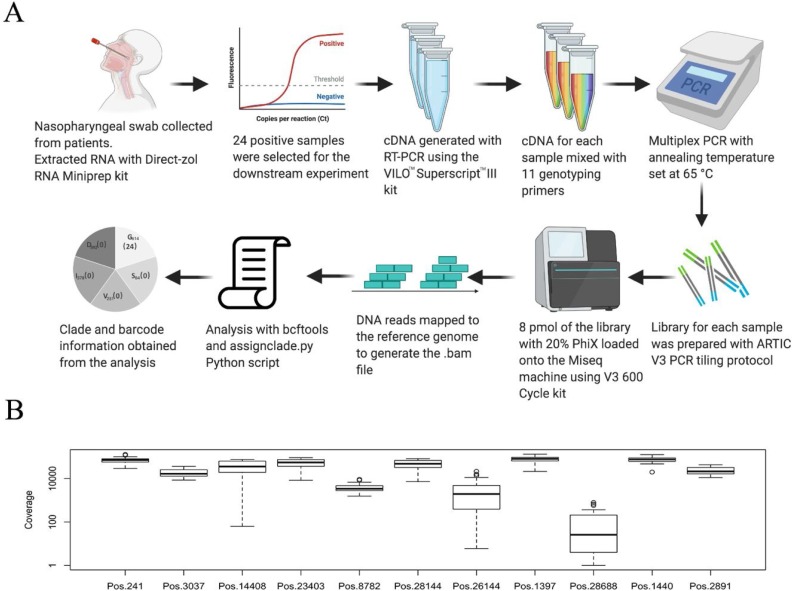
We have validated a set of oligonucleotide primers (Fig.S3) that can be utilised for rapid NGS sequencing-based genotyping of the major clades of SARS-CoV-2 using an Illumina MiSeq 600 V3 kit (Fig.3 A). We have tested this protocol on 24 RNA samples from COVID-19 patients, and generated the barcode information from the publicly accessible Python script (https://github.com/raeece/sarscov2barcode). The sequencing results show a high coverage of reads in the amplified regions across all 24 samples except for the position 28688, which accompany a G1397A mutation (Fig.3B). We observed the C241 T, C3037 T, C14408 T and G23403A substitutions in all 24 tested samples, indicating that they all belong to Clade G614.
Fig. 3.
Workflow from clinical sample collection, next-generation sequencing and SARS-CoV-2 clade assignment. (A) Schematic representation of the genotyping method described in this study. Positive samples were subjected to RNA extraction and multiplex RT-PCR. The amplicons were purified and prepared for the Illumina library. The sequencing was performed using MiSeq 600 cycles V3 kit and results were analyzed using our clade-defining script. (B) Boxplot of the coverage showing the log fold depth of the 11 clade-defining positions across the 24 SARS-CoV-2 genomes in multiplex sequencing-based genotyping. The primer sequences and PCR products of each pair of the primer are shown in Table 1 and Fig.S3.
Based on our calculation, roughly 78 K reads are needed for a given sample to obtain >800X genome coverage in 10 clade-defining sites (Table S1) except position 28688 (30X coverage). Based on the commercially-available multiplexing solutions available for Illumina library preparations, up to 384 samples can be processed with a MiSeq V3 600 Cycles kit in a single run to obtain reliable genotype information on the major clades. A higher number of samples could be genotyped with higher levels of multiplexing with custom-made index sequences for significant cost reduction in the future.
The robustness of our 11-site genomic barcoding method was verified by assigning ∼93.68% of the 79,486 SARS-CoV-2 genomes that became available in GISAID until August 3, 2020 to one of the 5 major clades. This barcode represents a snapshot of the early phases of the genetic diversity of the virus during the first 6 months of its global spread and is expected to change over time, however, a barcoding strategy to monitor the progress of virus elimination after vaccines become widely available will be strategically useful to monitor decreases in viral genetic diversity over time. In addition, our barcode could serve as a reference for setting the baseline for global genomic diversity analysis at the beginning of the pandemic.
Mutations are not equally distributed across the SARS-CoV-2 genome
Based on our analysis, we observed that the genes Orf3a, Orf6, Orf7, Orf8, N and Orf10 accumulated markedly more mutations than expected solely by random drift (Fig.S4) (e.g., Real/Expected ratio: N: 1.43; ORF3a: 1.52; E:1.17). This mutation rate may indicate adaptation to the human host following recent spill over from an, as yet, unknown animal reservoir. Conversely, several nonstructural proteins showed a lower-than-expected mutation rate (e.g., Real/Expected ratio: nsp5: 0.81; nsp10: 0.74). These proteins are predicted to be involved in evading host immune defenses, in enhancing viral expression and in cleavage of the replicase polyprotein, based on prior studies of related betacoronaviruses (Báez-Santos et al., 2015, Posthuma et al., 2017). Hence, this lower mutation rate may indicate purifying selection to maintain these functions essential for efficient immune evasion and subsequent viral dissemination. Indeed, structural proteins in coronaviruses undergo a greater degree of antigenic variation which increases the fitness of the virus by means of adaptation to the host and by facilitating immune escape (Walls et al. 2020).
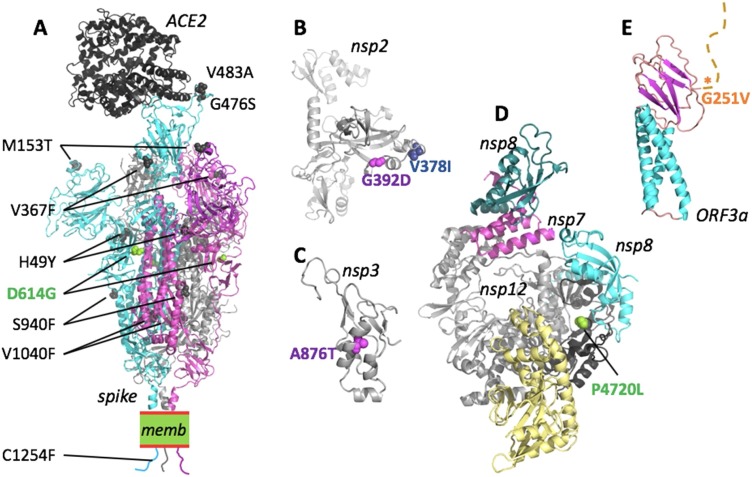
Structural protein modeling confirmed that most of the nonsynonymous mutations in the nonstructural proteins were neutral (Figs.S5-S11). Conversely, several nonsynonymous mutations in the spike protein might have functional consequences: notably, the G614 clade–defining mutation D614 G is located in subdomain 1 (SD1; Fig.4 , Fig.S5). In the trimeric S, D614 engages stabilising interactions within SD1 (R646 or the backbone of F592, depending on the chain) and with the S1 of the adjacent chain (T859 and K854). Replacement of D614 with a glycine would entail losing these stabilising electrostatic interactions and increase the dynamics in this region. Recent studies have shown D614 G disrupts a critical interprotomer contact and that this dramatically shifts the S proteintrimer conformation toward an ACE2-binding and fusion-competent state (Pascal et al. 2020). Hospitalised patients with G614 had higher viral titres in upper respiratory tract specimens but no discernible differences in disease outcome (Korber et al., 2020). Notably, V483A, V367 F and G476S are localised in the receptor-binding domain (RBD) of the spike protein which mediates binding to the host receptor angiotensin-converting enzyme 2 (ACE2) (Fig. 4) (Hoffmann et al. 2020). All of the viral genomes harbouring V483A and G476S mutations belong to Clade S84. Interestingly, the V367 F mutation has appeared independently in Clade V251 and Clade S84, suggesting that this mutation is under positive selection and contributes to viral fitness.
Fig. 4.
Mapping of SARS-CoV-2 clade-defining mutations onto the proteins. Nonsynonymous mutations for proteins where the 3D structure was experimentally determined (spike, nsp12/7/8) or can be inferred with reasonable confidence. Mutations are colour-coded as for the corresponding clades in Fig.3(D: magenta; G: light green; I: blue; V: orange). For a detailed analysis, see Fig.S5-11. (A) The structure of the SARS-CoV-2 spike trimer in its open conformation (chains are cyan, magenta and grey) bound to the human receptor ACE2 (black) modeled based on PDB accessions 6m17 and 6vyb. Identified nonsynonymous mutations are shown as spheres in the model. For reasons of visibility only mutations of two of the three spike chains are labeled. memb. indicates the plasma membrane. (B) Fragment comprising residues 180-534 of nsp2, modelled by AlphaFold35. Both clade-defining mutations are located in solvent-exposed regions and would not lead to steric clashes. (C) The substitution A876 T (corresponding to residue A58 in the nsp3 cleavage product numbering) is situated in the N-terminal ubiquitin-like domain of nsp3. The structure of this domain can be inferred based on the 79% identical structure of residues 1-112 from SARS-CoV (PDB id 2idy). The substitution A876 T can be accommodated with only minor structural adjustments and is not expected to have a substantial influence on the protein stability or function. (D) The structure shows the nsp12 in complex with nsp7 (magenta) and nsp8 (cyan and teal), based on PDB 7btf. P4720 (P323 in nsp12 numbering) is located in the ‘interface domain’ (black). In this position, the P323 L substitution is not predicted to disrupt the folding or protein interactions and hence is not expected to have strong effects. (E) A theoretical model for the Orf3a monomer has been proposed by AlphaFold36. The structure-function relationship of this protein remains to be clarified. The mutation G251 V is located C-terminal to the β-sandwich domain and the tail (marked by an asterisk).
We found the V483A substitution has an equivalent amino acid substitution located in a similar position within the RBD in the MERS-CoV spike protein which reduces its binding to its cognate receptor DPP4/CD26 (Kleine-Weber et al. 2018). However, V483 is more than 10 Å away from ACE2 and could affect receptor binding by SARS-CoV-2 only indirectly by altering the structural dynamics of the RBD loop it is a part of. The V367 F mutation is located an even greater distance from ACE2 (Fig.4, Fig.S5). The exchange of the small hydrophobic residue valine with a bulky hydrophobic phenylalanine might influence the efficiency of glycosylation of the nearby N343, or the positioning of the sugars. The substitution G476S would lead to possible clashes with predicted interacting ACE2 residues and with the RBD residue N487. However, minor reorientation of the side chains might allow an additional hydrogen bond to be formed between S476 and ACE2 Q24 and E23, thus enhancing the affinity to ACE2 (Fig.S5). An equivalent amino acid substitution located in an analogous position within the RBD in the SARS-CoV spike protein was associated with neutralisation escape from monoclonal antibodies, together with other mutations observed previously (Rockx et al. 2010). Given that V483A and V367 F are solvent exposed and markedly alter the surface characteristics of the RBD, they might also facilitate antibody escape. Indeed, V483A mutation was shown to give a selective advantage to the virus by escaping several neutralising antibodies (Li et al. 2020). Escape mutations in the RBD of the SARS-CoV S protein (T332I, F460C and L443R) were identified previously (Rockx et al. 2010). These mutations negatively impact viral fitness through reducing the affinity to the host receptor (Tang et al. 2014). The nonsynonymous mutations in the N protein, which have key roles in viral assembly, might also have functional implications. The hotspot mutations S202 N, R203 K and G204R all cluster in a linker region where they might potentially enhance RNA binding and alter the response to serine phosphorylation events (Fig.S5). The clade I378–defining mutation (T28688C) in the nucleocapsid protein, which has key roles in viral assembly, is synonymous. However, we observed nonsynonymous mutations in the nucleocapsid protein that are predicted to have functional implications. The hotspot mutations S202 N, R203 K and G204R all cluster in a linker region where they might potentially enhance RNA binding and alter the response to serine phosphorylation events (Fig. S6).
Discussion
In this study, we have defined 5 major clades (G614, S84, V251, I378 and D392) of fully-sequenced SARS-CoV-2 genomes available until May 28, 2020 in the GISAID database. The clustering of these genomes revealed the spread of clades to diverse geographical regions (Fig. 1, Fig. 2). This pattern contrasts with those observed for other epidemic coronaviruses, such as MERS-CoV, which display distinct geographical clustering (Kim et al. 2016). For example, clade G614, which was first detected on January is now dominant in the sampled genomes globally and its percentage is increasing globally from Europe, to North America, Oceania, and then to Asia (Korber et al. 2020). This pattern demonstrates efficient viral transmission through frequent intercontinental travel during the period when international travel restrictions were only present sporadically and which has enabled the virus to spread to multiple distant locations within a short period of time. This observation reinforces the importance of curtailing international travel and imposing restrictions early in pandemics and imposing social distancing in order to contain the global spread of viruses.
We have observed a distinct distribution of the major clades in different parts of the world (Fig.2). Most of the viral genomes which have not been assigned to a major clade are found in Asia and have earlier detection dates in January and February around the start of the epidemic in China. We observed a decrease in the genetic diversity of the virus over time following dissemination from China, especially in Europe and North America that each notably now has clade G614 as the predominant clade type, which is likely due the increased fitness advantage that this mutation brings to the virus as was reported recently (Korber et al. 2020). An important caveat of the present study is that the current sampling of available public genomes does likely not represent the extant genetic diversity of virus populations in circulation. This is due to biases of genome data deposits from the sequencing laboratories based mainly in the Northern hemisphere and new datasets may define new clades or subclades in the near future from other regions, including Africa, the Indian subcontinent and Latin America from where there are comparably few genomes available at present. In this case, additional identifiers within an evolving barcode scheme can be added to track and monitor future emerging clades with higher resolution. On the other hand, the genetic stability of SARS-CoV-2 (Jia et al. 2020) may result in the continuing circulation of a limited number of clades until such time as mitigation measures including the isolation of vulnerable populations and the availability of efficacious antivirals and vaccines might reduce the genetic diversity in circulation. This molecular genotyping approach has been demonstrated for other viruses (e.g. measles, poliovirus, rotavirus and human papillomaviruses) with herd-immunity vaccination programmes working to eliminate pathogens from endemic circulation in humans (Brown et al., 2019, Mankertz et al., 2011, Grassly, 2013, Soares-Weiser et al., 2012). The availability of a barcoding scheme that rapidly generates SNPs allowing clade assignment will be critical in this elimination phase when widespread availability of vaccines permits eradication of SARS-CoV-2 from endemicity in humans.
In this study, we have designed and tested the first iteration of a multiplexed sequencing-based SARS-CoV-2 genotyping protocol for major clade assignment (Fig.3). This will facilitate genetic surveillance to be performed at high-throughput level from SARS-CoV-2 detection to clade assignment. Future versions of the multiplexed primer sets can be optimised to cover the relatively low coverage regions (position 26144 and 28688). We concluded that the relatively low coverage at these two positions was a result of the position of mutation relative to the amplicon sites and not related to the PCR conditions used (Fig.S12). Our approach is also compatible with genotyping by other platforms, such as the MinION nanopore sequencer from Oxford Nanopore Technologies (ONT). Different sets of primers can also be designed to meet the requirements of different sequencing platforms. For example, the amplicon sizes could be optimised for the Illumina iSeq100 for rapid genotyping of a large number of samples without compromising the robustness of the genotyping calls at a fraction of current costs using a MiSeq. Moving forward, further studies may also include subclade-defining sites to have a higher resolution of each sequenced sample to assist in tracking and surveillance of the virus. The U.S. Food and Drug Administration (USFDA) has authorised Illumina COVIDSeq test which is intended for the detection of SARS-CoV-2 virus RNA for research use (https://www.illumina.com/products/by-type/ivd-products/covidseq.html). According to the COVIDseq instructions, up to 384 results per lane can be processed on the NovaSeq 6000 System for whole-genome sequecing. Our methodology is complementary to the COVIDSeq test and aimed at revealing the major clades of the virus in a given sample.
Our work provides a baseline genomic epidemiology of SARS-CoV-2 prior to the introduction of therapeutic and prophylactic approaches. The mutational landscape of global populations of over 21,789 SARS-CoV-2 genomes provides an evidence-based framework for tracking the clades that comprise the pandemic on different continents. However, due to the biases in the representation of countries depositing SARS-CoV-2 genomes, with over-representation of North American and European genomes, the available genome data represents only a minute proportion of the total COVID-19 positive cases from each of these regions. Therefore, this may require that the genetic barcode described here may need to be updated in order to be globally representative, once sufficient numbers of genomes covering less represented parts of the world are eventually sequenced and deposited to publicly-available databases. As viral genetic diversity is anticipated to decrease with the advent of widespread vaccination, the barcoding approach will allow more rapid discrimination of autochthonous and imported cases to monitor interruption of viral transmission.
Conclusion
This study provides a baseline reference of genomic diversity at the early stages of the COVID-19 pandeic and will prove useful for the monitoring of changes in circulating clades of SARS-CoV-2 in different geographic regions over time.. An 11-nucleotide genetic barcode of SARS-CoV-2 is presented which identifies the 5 major clades of circulating viral genomes. The robustness of our 11-site genomic barcoding approach was validated by coreectly assigning ∼94% of the 79,486 SARS-CoV-2 genomes available in GISAID until August 3, 2020 to one of the 5 major clades. We have designed the first version of a high-throughput and robust genotyping protocol that can be readily applied for SARS-CoV-2 detection and clade assignment.This barcoding strategy will be important to target genomic sequencing efforts and to monitor decreases in viral genetic diversity as intervention approaches become widely available.
Funding
Work in AP’s laboratory is supported by the KAUST faculty baseline fund (BAS/1/1020-01- 01) and research grants from the Office for Sponsored Research (OSR-2015-CRG4-2610, OCRF-2014-CRG3-2267). This work was also supported by funding from King Abdullah University of Science and Technology (KAUST), Office of Sponsored Research (OSR), under award number FCC/1/1976-25-01 to STA.
Ethical approval
This study is covered by IRB approval 20IBEC14 and HAP-01-R-082 to work on the SARS-CoV-2 positive RNA samples derived from COVID-19 positive patients at the Dr.Suliman Al-Habib Medical group in KSA.
Author Contributions
A.P. conceived the study and supervised the work; A.P., W.W.H., S.T.A. and Q.G. designed the research; Q.G., M.S., R.Na and S.T.A. analysed data; Q.G., M.S., S.T.A. and M.J.C. wrote the initial draft of the manuscript, followed by edits from A.P, M.J.C, W.W.H and R.Nu; R.Nu. and S.M. contributed new reagents; Q.G., S.H., S.M., R.Nu., A.A.O., S.S and A.A.M provided samples, designed and preformed the genotyping experiment. All authors have commented and edited various sections of the draft manuscript.
Conflict of interest
The authors declare no conflict of interest.
.
Acknowledgments
We are deeply grateful to all laboratories contributing genomic data and metadata to GISAID and nextstrain.org databases. We thank KAUST Rapid Research Response Team (R3T) for supporting our research. We thank Olga Douvropoulou for her support during the work. We also thank Richard Culleton (Nagasaki University, Japan) and Gabo Gonzalez (UCD, Ireland) for their critical comments on the manuscript draft.
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.ijid.2020.08.052.
Appendix A. Supplementary data
The following is Supplementary data to this article:
References
- Arnold K., Bordoli L., Kopp J., Schwede T. The SWISS-MODEL workspace: A web-based environment for protein structure homology modelling. Bioinformatics. 2006 doi: 10.1093/bioinformatics/bti770. [DOI] [PubMed] [Google Scholar]
- Báez-Santos Y.M., St. John SE, Mesecar AD. The SARS-coronavirus papain-like protease: Structure, function and inhibition by designed antiviral compounds. Antiviral Research. 2015 doi: 10.1016/j.antiviral.2014.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bárcena M., Oostergetel G.T., Bartelink W., Faas F.G.A., Verkleij A., Rottier P.J.M. Cryo-electron tomography of mouse hepatitis virus: Insights into the structure of the coronavirion. Proc Natl Acad Sci U S A. 2009 doi: 10.1073/pnas.0805270106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown K.E., Rota P.A., Goodson J.L., Williams D., Abernathy E., Takeda M. Genetic characterization of measles and rubella viruses detected through global measles and rubella elimination surveillance, 2016-2018. Morb Mortal Wkly Rep. 2019 doi: 10.15585/mmwr.mm6826a3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capella-Gutiérrez S., Silla-Martínez J.M. Gabaldón T. trimAl: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics. 2009;25(15):1972–1973. doi: 10.1093/bioinformatics/btp348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cingolani P., Platts A., Wang L.L., Coon M., Nguyen T., Wang L.L. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin). 2012 doi: 10.4161/fly.19695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong E., Du H., Gardner L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis [Internet] 2020;3099(20):19–20. doi: 10.1016/S1473-3099(20)30120-1. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassly NC. The final stages of the global eradication of poliomyelitis. Philosophical Transactions of the Royal Society B: Biological Sciences. 2013 doi: 10.1098/rstb.2012.0140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagemeijer M.C., Monastyrska I., Griffith J, van der Sluijs P, Voortman J, van Bergen en Henegouwen PM. Membrane rearrangements mediated by coronavirus nonstructural proteins 3 and 4. Virology. 2014 doi: 10.1016/j.virol.2014.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Mü MA, Drosten C, Pö S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor Article SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell [Internet] 2020;181:1–10. doi: 10.1016/j.cell.2020.02.052. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia Y., Shen G., Zhang Y., Huang K., Ho H., Hor W. Analysis of the mutation dynamics of SARS-CoV-2 reveals the spread history and emergence of RBD mutant with lower ACE2 binding affinity. bioRxiv. 2020 [Google Scholar]
- Käll L., Krogh A., Sonnhammer E.L.L. Advantages of combined transmembrane topology and signal peptide prediction-the Phobius web server. Nucleic Acids Res. 2007;35(SUPPL.2) doi: 10.1093/nar/gkm256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Källberg M., Margaryan G., Wang S., Ma J., Xu J. Raptorx server: A resource for template-based protein structure modeling. Methods Mol Biol. 2014 doi: 10.1007/978-1-4939-0366-5_2. [DOI] [PubMed] [Google Scholar]
- Katoh K, Standley DM. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol Biol Evol. 2013 doi: 10.1093/molbev/mst010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim Y.J., Il, Kim Y.J., Lemey P., Lee I., Park S., Bae J.Y. The recent ancestry of Middle East respiratory syndrome coronavirus in Korea has been shaped by recombination. Sci Rep. 2016 doi: 10.1038/srep18825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleine-Weber H., Elzayat M.T., Wang L., Graham B.S., Müller M.A., Drosten C. Mutations in the Spike Protein of Middle East Respiratory Syndrome Coronavirus Transmitted in Korea Increase Resistance to Antibody-Mediated Neutralization. J Virol. 2018 doi: 10.1128/JVI.01381-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korber B., Fischer W.M., Gnanakaran S., Yoon H., Theiler J., Abfalterer W. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell. 2020 doi: 10.1016/j.cell.2020.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letunic I., Bork P. Interactive Tree Of Life (iTOL): An online tool for phylogenetic tree display and annotation. Bioinformatics. 2007 doi: 10.1093/bioinformatics/btl529. [DOI] [PubMed] [Google Scholar]
- Li Q., Wu J., Nie J., Zhang L., Hao H., Liu S. The impact of mutations in SARS-CoV-2 spike on viral infectivity and antigenicity. Cell. 2020 doi: 10.1016/j.cell.2020.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu D.X., Fung T.S., Chong K.K.L., Shukla A., Hilgenfeld R. Accessory proteins of SARS-CoV and other coronaviruses. Antiviral Research. 2014 doi: 10.1016/j.antiviral.2014.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu R., Zhao X., Li J., Niu P., Yang B., Wu H. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020 doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mankertz A., Mihneva Z.R., Gold H., Baumgarte S., Baillot A., Helble R. Spread of measles virus D4-Hamburg, Europe, 2008-2011. Emerg Infect Dis. 2011 doi: 10.3201/eid1708.101994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pascal K.E., Veinotte K., Egri S.B., Schaffner S.F., Jacob E. Structural and Functional Analysis of the D614G SARS-CoV-2 Spike Protein Variant. bioRxiv. 2020 doi: 10.1016/j.cell.2020.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pickett B.E., Sadat E.L., Zhang Y., Noronha J.M., Squires R.B., Hunt V. ViPR: An open bioinformatics database and analysis resource for virology research. Nucleic Acids Res. 2012 doi: 10.1093/nar/gkr859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Posthuma C.C., te Velthuis A.J.W., Snijder E.J. Nidovirus RNA polymerases: Complex enzymes handling exceptional RNA genomes. Virus Research. 2017 doi: 10.1016/j.virusres.2017.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price MN, Dehal PS, Arkin AP. Fasttree: Computing large minimum evolution trees with profiles instead of a distance matrix. Mol Biol Evol. 2009 doi: 10.1093/molbev/msp077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rambaut A., Holmes E.C., Toole ÁO, Hill V., Mccrone J.T., Ruis C. A dynamic nomenclature proposal for SARS-CoV-2 lineages to assist genomic epidemiology. Nat Microbiol. 2020 doi: 10.1038/s41564-020-0770-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rockx B., Donaldson E., Frieman M., Sheahan T., Corti D., Lanzavecchia A. Escape from Human Monoclonal Antibody Neutralization Affects In Vitro and In Vivo Fitness of Severe Acute Respiratory Syndrome Coronavirus. J Infect Dis. 2010 doi: 10.1086/651022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shu Y., McCauley J. GISAID: Global initiative on sharing all influenza data – from vision to reality. Eurosurveillance. 2017 doi: 10.2807/1560-7917.ES.2017.22.13.30494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soares-Weiser K., MacLehose H., Bergman H., Ben-Aharon I., Nagpal S., Goldberg E. 2012. Vaccines for preventing rotavirus diarrhoea: vaccines in use. In: Cochrane Database of Systematic Reviews. [DOI] [PubMed] [Google Scholar]
- Stamatakis A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;30(9):1312–1313. doi: 10.1093/bioinformatics/btu033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang X., Wu C., Li X., Song Y., Yao X., Wu X. On the origin and continuing evolution of SARS-CoV-2. Natl Sci Rev. 2020;6(3) doi: 10.1093/nsr/nwaa036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang X.C., Agnihothram S.S., Jiao Y., Stanhope J., Graham R.L., Peterson E.C. Identification of human neutralizing antibodies against MERS-CoV and their role in virus adaptive evolution. Proc Natl Acad Sci U S A. 2014 doi: 10.1073/pnas.1402074111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treangen T.J., Ondov B.D., Koren S., Phillippy A.M. The harvest suite for rapid core-genome alignment and visualization of thousands of intraspecific microbial genomes. Genome Biol. 2014;15(11) doi: 10.1186/s13059-014-0524-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walls A.C., Park Y.J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020 doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu A., Peng Y., Huang B., Ding X., Wang X., Niu P. Genome Composition and Divergence of the Novel Coronavirus (2019-nCoV) Originating in China. Cell Host Microbe. 2020 doi: 10.1016/j.chom.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu F., Zhao S., Yu B., Chen Y.M., Wang W., Song Z.G. A new coronavirus associated with human respiratory disease in China. Nature. 2020 doi: 10.1038/s41586-020-2008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J., Yan R., Roy A., Xu D., Poisson J., Zhang Y. The I-TASSER suite: Protein structure and function prediction. Nature Methods. 2014 doi: 10.1038/nmeth.3213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H., Chen X., Hu T., Li J., Song H., Liu Y. A Novel Bat Coronavirus Closely Related to SARS-CoV-2 Contains Natural Insertions at the S1/S2 Cleavage Site of the Spike Protein. Curr Biol. 2020 doi: 10.1016/j.cub.2020.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu N., Zhang D., Wang W., Li X., Yang B., Song J. A novel coronavirus from patients with pneumonia in China. N Engl J Med. 2019;2020 doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.