Abstract
The coronavirus disease 2019 (COVID-19) pandemic, originating from Wuhan, China, is known to cause severe acute respiratory symptoms. The occurrence of a cytokine storm in the lungs is a critical step in the disease pathogenesis, as it causes pathological lesions, pulmonary edema, and acute respiratory distress syndrome, potentially resulting in death. Currently, there is no effective treatment that targets the cytokine storm and helps regenerate the damaged tissue. Mesenchymal stem cells (MSCs) are known to act as anti-inflammatory/immunomodulatory candidates and activate endogenous regeneration. As a result, MSC therapy is a potential treatment approach for COVID-19. Intravenous injection of clinical-grade MSCs into COVID-19 patients can induce an immunomodulatory response along with improved lung function. Dental pulp stem cells (DPSCs) are considered a potential source of MSCs for immunomodulation, tissue regeneration, and clinical application. Although some current clinical trials have treated COVID-19 patients with DPSCs, this therapy has not been approved. Here, we review the potential use of DPSCs and their significance in the development of a therapy for COVID-19.
Keywords: dental pulp stem cells, COVID-19, coronavirus, cytokines storm, immunomodulation, regeneration
Introduction
A new infection emerged in Wuhan, China toward the end of 2019. This infection induced pneumonia and had clinical manifestations that were shortly recognized to be caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)1. This novel disease was named coronavirus disease 2019 (COVID-19) by the World Health Organization2. Over 13,500,000 cases and 580,000 deaths (https://www.worldometers.info/coronavirus/) have been reported worldwide, as of July 17, 2020. COVID-19 can be divided into three stages corresponding to different clinical manifestations, based on the pathological findings of infected respiratory tract data3. Under asymptomatic conditions, the virus binds to epithelial cells in the nasal cavity via the main receptor expressed in the respiratory epithelium, angiotensin-converting enzyme II (ACE2)4,5. At this stage, the infection rate is diagnosed using the polymerase chain reaction from nasal swab samples. By conducting the airway response stage, the virus spreads downward the respiratory tract, along the conducting airways, eliciting a vigorous innate immune response and exhibiting clinical symptoms. During stage 3, the virus infects the alveolar cells, which continue to undergo apoptosis, and the patient subsequently develops pulmonary infiltration, hypoxia, and very severe disease.
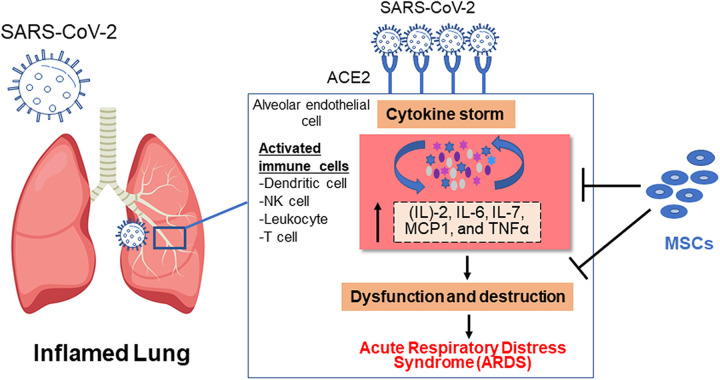
As reported, the immune-mediated inflammation markers, interleukin (IL)-2, -6, -7, monocyte chemoattractant protein-1, and tumor necrosis factor alpha (TNF-α), play a significant role in the pathogenesis of COVID-196,7. Recruitment of these inflammatory cytokines into the lung tissues causes edema, lung dysfunction, and acute respiratory distress syndrome (ARDS) (Fig. 1), which may lead to death6. The development of COVID-19 is accompanied by a decline in lymphocytes and a significant increase in neutrophil numbers8. The number of B cells, T cells, and natural killer (NK) cells decreases in patients with severe infection8. To date, hygiene measures and alleviation strategies have been directed at minimizing the transmission of the infection. Symptomatic and supportive treatments, oxygen supplementation, and mechanical ventilation are currently used to combat the disease. Mesenchymal stem cells (MSCs) are promising in cell-based therapies for many diseases9 including infectious diseases such as avian influenza H9N2, infections by hepatitis viruses B and C, and human immunodeficiency virus (HIV) infection10,11. MSCs are thought to aid COVID-19 treatment through two strategies: (1) immunomodulation of immune cells, decreasing the inflammation12, and (2) regeneration of the damaged lung tissues13 (Fig. 1). A preliminary study described that the transplantation of MSCs was reliable and efficient in treating patients with COVID-19 with severe conditions14. MSCs can be isolated from different sources9,15. Dental pulp stem cells (DPSCs) are known for their unique immunomodulatory and regenerative effects16. In this article, we highlight DPSCs as a potential therapeutic option for patients with COVID-19.
Figure 1.
Release of a cytokine storm in response to SARS-CoV-2 and a potential mechanism of MSCs in the treatment of COVID-19 patients. Data are obtained from Refs6,7,17,18. COVID-19: coronavirus disease 2019; MSC: mesenchymal stem cell; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2.
MSCs Versus Non-cellular Treatments for COVID-19
Scientists are attempting to develop new potential therapeutic approaches to combat COVID-19. Some of these treatments involve the use of vaccines, monoclonal antibodies, peptides, small-molecule drugs, interferon-based therapies, protease inhibitors, and adjunctive medications19. However, some medications such as hydroxychloroquine and azithromycin are not recommended for use in the clinic, owing to reported toxicity and cardiovascular complications20. Remdesivir, an antiviral drug, was approved by the Food and Drug Administration for the treatment of COVID-19. However, a recent study showed that it only reduces the time of clinical improvement and that there was no significant difference when compared with a placebo group19,21. Lopinavir/Ritonavir or other protease inhibitors, used for HIV treatment, were also not suggested because of harmful pharmacodynamics and insignificant clinical trial outcomes19. Though interferons are beneficial in the treatment of SARS and Middle East Respiratory Syndrome, they lack efficacy against COVID-1922. Monoclonal antibodies were suggested as a therapeutic tool; however, they only provide protection against the early stage of the disease23. Adjunctive medications such as antimicrobial agents, corticosteroids, and ACE inhibitors are reported as a potential treatment; however, more clinical studies are required to clarify their mechanisms in COVID-1924. Hence, no pharmacological treatments efficiently stop disease progression.
Studies on the pathogenesis of COVID-19 suggest that a dysregulated immune response occurs, resulting in extreme inflammation and deadly ARDS25 (Fig. 1). Thus, immunomodulation can be an efficient therapy. The safety and efficacy of MSCs have been presented in preclinical models of ARDS26, making them promising candidates for COVID-19. Moreover, the use of MSCs to treat patients with COVID-19 was safe and had no harmful effects14. In brief, MSCs represent a potential agent for treating COVID-19 because they can modulate the immune response and uphold the host immune system.
MSCs and Infectious Diseases
MSCs are multipotent stromal cells that originate from stromal tissues. They are fibroblast-like cells; adhere to plastic substrates; express different clusters of differentiation (cell surface markers) CD90, CD105, CD29, CD44, CD54, CD166, Stro-1, and MHC-I; and express the absence of hematopoietic markers CD45, CD31, CD11b, CD19, CD34, CD14, and HLA-DR27,28. Owing to their attractive cell biology and roles in the field of tissue engineering, MSCs have been extensively explored over the past 30 years9. MSCs have a pioneering characteristic to differentiate intrinsically and release several beneficial growth factors and cytokines. MSCs possess effective anti-inflammatory and immunomodulatory properties that can treat inflammatory and immune-mediated conditions29. They have been reported to be safe and efficient for clinical application and their mechanism of action is well established30,31. MSC preconditioning with proinflammatory cytokines licenses the immunosuppressive activity by inducing the expression of anti-inflammatory factors32–34, reducing inflammation.
Owing to the above-mentioned potential, MSCs are promising agents for the treatment of different tissue disorders9 and for the control of infectious diseases11. MSCs have antimicrobial properties that reduce acute lung injury (ALI) induced by bacterial infections35. MSCs improve the phagocytic function of host macrophages, leading to more effective eradication of bacteria35. Other reports also validate MSC therapy for malaria, a parasitic disease that causes severe destruction of red blood cells and anemia36. Gupta et al. demonstrated that transplantation of allogeneic hematopoietic stem cells that did not express CCR5, the receptor to enter target immunological cells, could effectively cure HIV37. Earlier studies further showed that hematopoietic stem cells could be engineered to target HIV-infected cells38. Additionally, bone marrow–derived MSCs (BMMSCs) recover liver function in hepatitis B by regulating the Treg/Th17 cell balance39. In the lung, MSCs and their derived extracellular vesicles were shown to reduce the levels of chemokines and proinflammatory cytokines, decreasing the migration of inflammatory cells into the lungs. This can be a promising approach to cure ALI, which is induced by influenza viruses10,40. Improved oxygenation and decreased pulmonary edema were shown following the transplantation of human MSCs in ARDS in a sheep model41. MSCs have also been suggested for the treatment of the H7N9 viral infection that has similar clinical effects on the lung as COVID-1942. As noted, there are various sources from which MSCs can be isolated. However, bone marrow43 and adipose tissue44 are the most common adult tissue sources for MSCs. As a result, the infusion of adipose tissue–derived MSCs into patients with COVID-19 could improve the clinical outcomes and immunomodulatory functions in severe cases14. Human umbilical cord Wharton’s jelly-derived MSCs were used to treat patients with severe COVID-1945. Dental tissue–derived MSCs have also shown promise in tissue regeneration because of their potent capacities to differentiate into multiple lineages, immunomodulate, and secrete trophic factors. This collectively supports the use of MSCs as a potential treatment for COVID-19.
Immunomodulation and Regenerative Properties of DPSCs
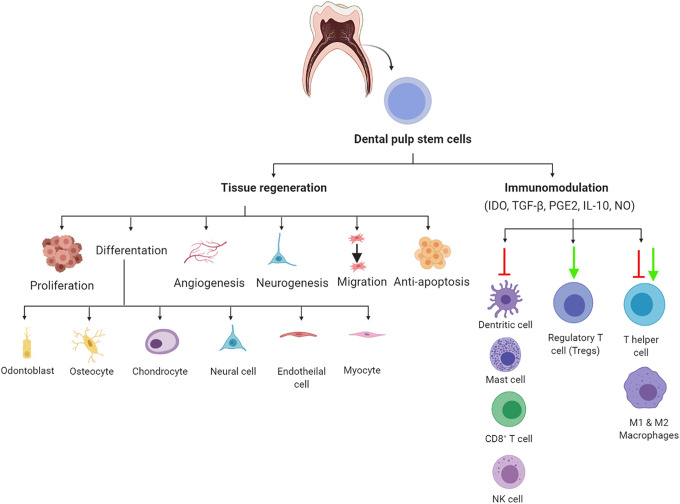
DPSCs, isolated from the pulp tissue, are the most common form of dental tissue–derived MSCs. Other stem cells from exfoliated deciduous teeth (SHED), stem cells from apical papilla, periodontal ligament stem cells, and dental follicle precursor cells have also been reported46. DPSCs self-renew, are multipotent, and have unique biological properties that make them promising agents, capable of improving the endurance of affected tissues16 (Fig. 2). DPSCs have an effective immunomodulatory function that is able to address a diverse set of autoimmune and inflammation-related diseases47. The immunomodulatory mechanisms associated with the release of soluble factors, such as indoleamine 2, 3-dioxygenase (IDO), prostaglandin E2 (PGE2), transforming growth factor-β (TGF-β), and human leukocyte antigen G5 (HLA-G5), and interfaces with immune cells such as B cells, T cells, dendritic cells, and macrophages are hallmarks of their function48. DPSCs promote high expression of trophic factors such as PGE2, IL-6, and IDO49–51. A mixed lymphocyte reaction assay further revealed the strong enhancement of the immunosuppression of the DPSC-conditioned medium (CM)51. DPSCs inhibit the function of proinflammatory M1 macrophages by inhibiting the secretion of TNF-α via the IDO-mediated pathway52 (Fig. 2). They also polarize macrophages toward the anti-inflammatory M2 phenotype through increased secretion of IL-10, PGE2, IL-6, and granulocyte-macrophage colony-stimulating factor (GM-CSF)53. Furthermore, they have the capability to inhibit the activation of Th17 cells54 and stimulate the growth of CD25+, CD4+, and FoxP3+ regulatory T cells55. DPSCs also induce T cell apoptosis and inhibit the survival rate of NK cells and Th156, resulting in the reduced production of IFN-γ and IL-17 by Th1. At a level of humoral immunity, DPSCs hamper the proliferation, antibody production, and differentiation potential of B cells and inhibit the proliferation of allogeneic T and B cells by the release of TGF-β156. This secretion of TGF-β can suppress the activation of human peripheral blood mononuclear cells57.
Figure 2.
Schematic diagram of a potential mechanism of DPSCs in the treatment of COVID-19. Data are obtained from Refs16,48,58. Figure is made with biorender: https://biorender.com/. COVID-19: coronavirus disease 2019; DPSCs: dental pulp stem cells.
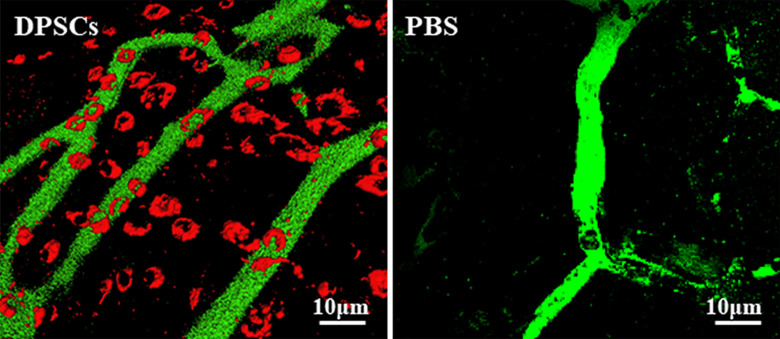
DPSCs play an important role in tissue regeneration58 and have the capability to repair different tissue disorders16. While BMMSCs are representative MSCs due to their high regenerative potential, DPSCs have higher angiogenic, neurogenic, and regenerative potential59, exhibiting an alternative multipurpose stem cell source for cellular therapies (Fig. 2). Neovascularization in the ischemic hindlimb has been demonstrated after DPSC transplantation60 (Fig. 3). The DPSC regeneration mechanisms via migration activity are achieved through modulating the response to granulocyte colony-stimulating factor (G-CSF), improving their regenerative potential51. Transplanted DPSCs produce a broad spectrum of cytokines and growth factors that modify neighboring cells. The success in restoring damaged tissues is in part related to the disclosure of paracrine factors in the host tissues. These paracrine effects promote the recruitment of progenitor cells, improve angiogenesis/neurogenesis, and modify the immune response61. The following paracrine factors are highly expressed in DPSCs, hepatocyte growth factor, vascular endothelial growth factor, insulin-like growth factor, fibroblast growth factor, macrophage colony-stimulating factor, stromal cell-derived factor 1, GM-CSF, G-CSF, and a small number of cytokines (IL-6, -8, -10)61. The release of trophic factors following DPSC transplantation induces pulp regeneration, but the DPSCs themselves are not incorporated into the new tissue50. DPSCs differentiate into different cell types such as cardiomyocytes, melanocytes, myocytes, neurons, and hepatocyte-like cells16. BMMSCs and adipose-derived stem cells (ADSCs) induce regenerated pulp tissue similar to DPSCs. The transplantation of DPSCs into pulpectomized teeth compared with BMMSCs or ADSCs resulted in the generation of a larger amount of pulp59. This can be explained by the increased release of trophic factors that promote angiogenesis, neurogenesis, anti-apoptosis, and chemotactic factors49. For these reasons, DPSCs are now considered as one of the best future sources of MSCs for use in regenerative medicine16. Thus, they are used to treat various tissue disorders, including dental, neurological, corneal, cardiovascular, hepatic, muscular dystrophy, pancreatic, and renal tissue diseases16,62,63. Moreover, the transplantation of allogeneic DPSCs (with matched and mismatched dog leukocyte antigen) into pulpectomized teeth of dogs regenerated pulp tissue in both matched and mismatched transplanted teeth. There was no toxicity or adverse event associated with the transplantation of DPSCs64. These results showed that DPSCs have high immunosuppressive, immunomodulatory, and tissue regeneration activities. Therefore, it is of interest in the development of COVID-19 treatment options (Fig. 2).
Figure 3.
Neovascularization in ischemic hindlimb after transplantation of DPSCs. Three-dimensional confocal laser micrograph. Red: 1,1′-dioetadeeyl-3,3,3′.3′- tetramethylindocarboeyanine dioetadeeyl (DiI)-labeled DPSCs. Green: capillaries with labeling with fluorescein isothiocyanate dextran. Data are obtained from Iohara et al60. DPSCs: dental pulp stem cells.
DPSCs and Lung Injury
Loss of alveolar structures and accumulation of inflammatory cells, followed by fibrosis, are the main characteristics of ARDS65. The activation of macrophages plays a role in the pathophysiology of ARDS66, where M1 macrophages release proinflammatory cytokines and increase tissue fibrosis67. Therefore, managing macrophages is a valuable strategy to treat ARDS. Wakayama et al. showed that intravenous infusion of SHED and their CM were assessed in chemically induced ALI in a mouse model. The results showed that the survival rate and regeneration improved after the administration of either SHED or CM. Moreover, this treatment upregulated the anti-inflammatory effect by activating M2 macrophages68. In COVID-19, the reported clinical studies used the intravenous injection of MSCs as a satisfactory and less invasive method. Fortunately, MSCs injected intravenously are trapped mostly in the lungs, the most affected organ in COVID-19, and to a smaller extent in other tissues69. This suggests that intravenous administration of DPSCs will be localized to the lung tissues. Compared with other sources of MSCs, DPSCs exhibit higher potential for treating COVID-19 for the following reasons:
DPSCs can be easily isolated in a less invasive manner from discarded teeth and comply with ethical considerations70.
DPSCs are abundantly available, are easy to harvest, and have effective therapeutic abilities49,71,72.
In vitro, they show a high proliferative ability73, provide sufficient cell numbers in a short period. Besides, demonstrated a multi-differentiation potential46.
DPSCs and SHED have immunomodulatory functions similar to those of BMMSCs. Consequently, they are considered good candidates for cell-based approaches for immune- and inflammation-related diseases48.
Even though the above-mentioned facts highlight the potential of DPSCs in combating COVID-19, all MSC sources have a restricted lifespan in vitro and lose their original characteristics with serial passaging. To overcome these challenges, immortalized DPSCs have been developed74. The piggyBac system, composed of mutant baculovirus strains derived from the cabbage looper moth Trichoplusia ni, was used to immortalize DPSCs75. Orimoto et al. transduced a mutant cyclin-dependent kinase 4 (CDK4R24C), cyclin D1, and telomerase reverse transcriptase into DPSCs to establish an immortalized DPSC cell line with a high proliferative rate74. In addition, immortalized DPSCs using human telomerase reverse transcriptase were reported76.
Clinical Trials of DPSCs in COVID-19 Patients
DPSCs are thought to fight against COVID-19 by preventing the cytokine storm through immunomodulation. Moreover, DPSCs can regenerate and reconstruct damaged tissues via reparative properties and effects of trophic factors (Fig. 2). Currently, there are many clinical trials using MSCs from different sources to treat COVID-19 (https://clinicaltrials.gov/). Two of the early recorded trials used DPSCs (Table 1). A protocol for a clinical trial by Ye et al. to assess the safety and efficiency of DPSCs from allogeneic donors in severe cases of COVID-19 was recently published77. The study reported on the safety and efficacy of DPSCs for the treatment of COVID-19. The time consumed for clinical improvement and the improvement of laboratory tests such as blood tests, liver and kidney functions, inflammatory markers, and immunological tests influenced the outcomes of the study77. However, the specific underlying mechanisms and outcomes of these trials are still not clear. Going forward, the appropriate cell dosage and concentration should be optimized to increase the efficacy and safety of DPSC-based COVID-19 therapies.
Table 1.
The Clinical Trials of DPSCs on COVID-19 Patients.
| Name of the study | Number of the study | Status | Reference |
|---|---|---|---|
| Safety and efficacy study of human allogeneic dental pulp mesenchymal stem cells to treat severe COVID-19 patients77. | NCT04336254 | Recruiting | https://clinicaltrials.gov/ |
| Novel Coronavirus induced severe pneumonia treated by dental pulp mesenchymal stem cells. | NCT04302519 | Not yet recruiting | https://clinicaltrials.gov/ |
COVID-19: coronavirus disease 2019; DPSCs: dental pulp stem cells.
Conclusion
COVID-19 is a global pandemic and requires the simultaneous development of an effective therapy and vaccine. One of the most prominent findings in COVID-19 has been the presence of a cytokine storm in the lungs in severely impacted patients. MSCs are ACE2-negative at the gene level (improbable of getting infected) and have been extensively used in other immune-related diseases. MSCs, specifically DPSCs, can be great candidates for the treatment of this novel disease, owing to their ability to inhibit the release of cytokines through their immunomodulation and regenerative capacity.
Acknowledgments
We would like to thank Editage (www.editage.com) for English language editing.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Mohammed Zayed  https://orcid.org/0000-0002-3361-0943
https://orcid.org/0000-0002-3361-0943
References
- 1. Gorbalenya AE, Baker SC, Baric RS, de Groot RJ, Drosten C, Gulyaeva AA, Haagmans BL, Lauber C, Leontovich AM, Neuman BW, Penzar D, et al. The species severe acute respiratory syndrome-related coronavirus: classifying 2019- nCOV and naming it SARS-CoV-2. Nat Microbiol. 2020;5(4):536–544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mason RJ. Pathogenesis of COVID-19 from a cell biology perspective. Eur Respir J. 2020;55(4):2000607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu N, Bi Y, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395(10224):565–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Metcalfe SM. Mesenchymal stem cells and management of COVID-19 pneumonia. Med Drug Discov. 2020;5:100019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020;71(15):762–768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pittenger MF, Discher DE, Péault BM, Phinney DG, Hare JM, Caplan AI. Mesenchymal stem cell perspective: cell biology to clinical progress. NPJ Regen Med. 2019;4:22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Li Y, Xu J, Shi W, Chen C, Shao Y, Zhu L, Lu W, Han X. Mesenchymal stromal cell treatment prevents H9N2 avian influenza virus-induced acute lung injury in mice. Stem Cell Res Ther. 2016;7(1):159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Mezey E, Nemeth K. Mesenchymal stem cells and infectious diseases: smarter than drugs. Immunol Lett. 2015;168(2):208–214. [DOI] [PubMed] [Google Scholar]
- 12. Prockop DJ. The exciting prospects of new therapies with mesenchymal stromal cells. Cytotherapy. 2017;19(1):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wang S, Wu J, Liu G-H. First stem cell transplantation to regenerate human lung. Protein Cell. 2018;9(3):244–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Leng Z, Zhu R, Hou W, Feng Y, Yang Y, Han Q, Shan G, Meng F, Du D, Wang S, Fan J, et al. Transplantation of ACE2 (-) mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging Dis. 2020;11(2):216–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Zayed M, Caniglia C, Misk N, Dhar MS. Donor-matched comparison of chondrogenic potential of equine bone marrow- and synovial fluid-derived mesenchymal stem cells: implications for cartilage tissue regeneration. Front in Vet Sci. 2017;3:121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Yamada Y, Nakamura-Yamada S, Kusano K, Baba S. Clinical potential and current progress of dental pulp stem cells for various systemic diseases in regenerative medicine: a concise review. Int J Mol Sci. 2019;20(5):1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. de Castro LL, Lopes-Pacheco M, Weiss DJ, Cruz FF, Rocco PRM. Current understanding of the immunosuppressive properties of mesenchymal stromal cells. J Mol Med (Berl). 2019;97(5):605–618. [DOI] [PubMed] [Google Scholar]
- 18. Weiss ARR, Dahlke MH. Immunomodulation by mesenchymal stem cells (MSCs): mechanisms of action of living, apoptotic, and dead MSCs. Front Immunol. 2019;10:1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jean S-S, Lee P-I, Hsueh P-R. Treatment options for COVID-19: the reality and challenges. J Microbiol Immunol Infect. 2020;53(3):436–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Nguyen LS, Dolladille C, Drici MD, Fenioux C, Alexandre J, Mira JP, Moslehi JJ, Roden DM, Funck-Brentano C, Salem JE. Cardiovascular toxicities associated with hydroxychloroquine and azithromycin: an analysis of the world health organization pharmacovigilance database. Circulation. 2020;142(3):303–330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, Fu S, Gao L, Cheng Z, Lu Q, Hu Y, et al. Remdesivir in adults with severe covid-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395(10236):1569–1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Zumla A, Chan JFW, Azhar EI, Hui DSC, Yuen K-Y. Coronaviruses — drug discovery and therapeutic options. Nat Rev Drug Discov. 2016;15(5):327–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Martinez MA. Compounds with therapeutic potential against novel respiratory 2019 coronavirus. Antimicrob Agents Chemother. 2020;64(5):e00399–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kabir MT, Uddin MS, Hossain MF, Abdulhakim JA, Alam MA, Ashraf GM, Bungau SG, Bin-Jumah MN, Abdel-Daim MM, Aleya L. nCOVID-19 pandemic: from molecular pathogenesis to potential investigational therapeutics. Front Cell Dev Biol. 2020;8:616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, et al. Pathological findings of COVID-19 pandemic: From molecular pathogenesis to potential investigational therapeutics associated with acute respiratory distress syndrome. Lancet Resp Med. 2020;8(4):420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Horie S, Gonzalez HE, Laffey JG, Masterson CH. Cell therapy in acute respiratory distress syndrome. J Thorac Dis. 2018;10(9):5607–5620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Young HE, Steele TA, Bray RA, Detmer K, Blake LW, Lucas PW, Black AC., Jr Human pluripotent and progenitor cells display cell surface cluster differentiation markers CD10, CD13, CD56, and MHC class-I. Proc Soc Exp Biol Med. 1999;221(1):63–71. [DOI] [PubMed] [Google Scholar]
- 28. Trusler O, Huang Z, Goodwin J, Laslett AL. Cell surface markers for the identification and study of human naive pluripotent stem cells. Stem Cell Res. 2018;26:36–43. [DOI] [PubMed] [Google Scholar]
- 29. Gomez-Salazar M, Gonzalez-Galofre ZN, Casamitjana J, Crisan M, James AW, Péault B. Five decades later, are mesenchymal stem cells still relevant? Front Bioeng Biotechnol. 2020;8:148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Attico E, Sceberras V, Pellegrini G. Approaches for effective clinical application of stem cell transplantation. Curr Transplant Rep. 2018;5(3):244–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Moll G, Hoogduijn MJ, Ankrum JA. Editorial: safety, efficacy and mechanisms of action of mesenchymal stem cell therapies. Front Immunol. 2020;11:243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ryan JM, Barry F, Murphy JM, Mahon BP. Interferon-gamma does not break, but promotes the immunosuppressive capacity of adult human mesenchymal stem cells. Clin Exp Immunol. 2007;149(2):353–363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-κB signaling in resident macrophages. Blood. 2011;118(2):330–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Jin P, Zhao Y, Liu H, Chen J, Ren J, Jin J, Bedognetti D, Liu S, Wang E, Marincola F, Stroncek D. Interferon-γ and tumor necrosis factor-α polarize bone marrow stromal cells uniformly to a Th1 phenotype. Sci Rep. 2016;6:26345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Lee JW, Krasnodembskaya A, McKenna DH, Song Y, Abbott J, Matthay MA. Therapeutic effects of human mesenchymal stem cells in ex vivo human lungs injured with live bacteria. Am J Respir Crit Care Med. 2013;187(7):751–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Saei AA, Ahmadian S. Stem cell engineering might be protective against severe malaria. Biosci Hypotheses. 2009;2(1):48–49. [Google Scholar]
- 37. Gupta RK, Peppa D, Hill AL, Galvez C, Salgado M, Pace M, McCoy LE, Griffith SA, Thornhill J, Alrubayyi A, Huyveneers LEP, et al. Evidence for HIV-1 cure after CCR5Δ32/Δ32 allogeneic haemopoietic stem-cell transplantation 30 months post analytical treatment interruption: a case report. The Lancet HIV 2020;7(5):e340–e347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Holt N, Wang J, Kim K, Friedman G, Wang X, Taupin V, Crooks GM, Kohn DB, Gregory PD, Holmes MC, Cannon PM. Human hematopoietic stem/progenitor cells modified by zinc-finger nucleases targeted to CCR5 control HIV-1 in vivo. Nat Biotechnol. 2010;28(8):839–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Xu L, Gong Y, Wang B, Shi K, Hou Y, Wang L, Lin Z, Han Y, Lu L, Chen D, Lin X, et al. Randomized trial of autologous bone marrow mesenchymal stem cells transplantation for hepatitis B virus cirrhosis: Regulation of Treg/Th17 cells. J Gastroenterol. Hepatol. 2014;29(8):1620–1628. [DOI] [PubMed] [Google Scholar]
- 40. Khatri M, Richardson LA, Meulia T. Mesenchymal stem cell-derived extracellular vesicles attenuate influenza virus-induced acute lung injury in a pig model. Stem Cell Res Ther. 2018;9(1):17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Asmussen S, Ito H, Traber DL, Lee JW, Cox RA, Hawkins HK, McAuley DF, McKenna DH, Traber LD, Zhuo H, Wilson J, et al. Human mesenchymal stem cells reduce the severity of acute lung injury in a sheep model of bacterial pneumonia. Thorax. 2014;69(9):819–825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Chen J, Hu C, Chen L, Tang L, Zhu Y, Xu X, Chen L, Gao H, Lu X, Yu L, Dai X, et al. Clinical study of mesenchymal stem cell treating acute respiratory distress syndrome induced by epidemic influenza A (H7N9) infection, a hint for COVID-19 treatment. Engineering (Beijing, China). Epub ahead of print 2020 DOI:10.1016/j.eng.2020.02.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143–147. [DOI] [PubMed] [Google Scholar]
- 44. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211–228. [DOI] [PubMed] [Google Scholar]
- 45. Zhang Y, Ding J, Ren S, Wang W, Yang Y, Li S, Meng M, Wu T, Liu D, Tian S, Tian H, et al. Intravenous infusion of human umbilical cord Wharton’s jelly-derived mesenchymal stem cells as a potential treatment for patients with COVID-19 pneumonia. Stem Cell Res Ther. 2020;11(1):207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Huang GTJ, Gronthos S, Shi S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res. 2009;88(9):792–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Andrukhov O, Behm C, Blufstein A, Rausch-Fan X. Immunomodulatory properties of dental tissue-derived mesenchymal stem cells: implication in disease and tissue regeneration. World J Stem Cells. 2019;11(9):604–617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Li Z, Jiang CM, An S, Cheng Q, Huang YF, Wang YT, Gou YC, Xiao L, Yu WJ, Wang J. Immunomodulatory properties of dental tissue-derived mesenchymal stem cells. Oral Dis. 2014;20(1):25–34. [DOI] [PubMed] [Google Scholar]
- 49. Ishizaka R, Hayashi Y, Iohara K, Sugiyama M, Murakami M, Yamamoto T, Fukuta O, Nakashima M. Stimulation of angiogenesis, neurogenesis and regeneration by side population cells from dental pulp. Biomaterials. 2013;34(8):1888–1897. [DOI] [PubMed] [Google Scholar]
- 50. Iohara K, Murakami M, Takeuchi N, Osako Y, Ito M, Ishizaka R, Utunomiya S, Nakamura H, Matsushita K, Nakashima M. A novel combinatorial therapy with pulp stem cells and granulocyte colony-stimulating factor for total pulp regeneration. Stem Cells Transl Med. 2013;2(7):521–533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Murakami M, Horibe H, Iohara K, Hayashi Y, Osako Y, Takei Y, Nakata K, Motoyama N, Kurita K, Nakashima M. The use of granulocyte-colony stimulating factor induced mobilization for isolation of dental pulp stem cells with high regenerative potential. Biomaterials. 2013;34(36):9036–9047. [DOI] [PubMed] [Google Scholar]
- 52. Lee S, Zhang QZ, Karabucak B, Le AD. DPSCs from inflamed pulp modulate macrophage function via the TNF-α/IDO axis. J Dent Res. 2016;95(11):1274–1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Zhang QZ, Nguyen AL, Yu WH, Le AD. Human oral mucosa and gingiva: a unique reservoir for mesenchymal stem cells. J Dent Res. 2012;91(11):1011–1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Ding G, Niu J, Liu Y. Dental pulp stem cells suppress the proliferation of lymphocytes via transforming growth factor-β1. Hum Cell. 2015;28(2):81–90. [DOI] [PubMed] [Google Scholar]
- 55. Lan Q, Fan H, Quesniaux V, Ryffel B, Liu Z, Zheng SG. Induced Foxp3(+) regulatory t cells: a potential new weapon to treat autoimmune and inflammatory diseases? J Mol Cell Biol. 2012;4(1):22–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Zhao Y, Wang L, Jin Y, Shi S. Fas ligand regulates the immunomodulatory properties of dental pulp stem cells. J Dent Res. 2012;91(10):948–954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Morsczeck C, Völlner F, Saugspier M, Brandl C, Reichert TE, Driemel O, Schmalz G. Comparison of human dental follicle cells (DFCS) and stem cells from human exfoliated deciduous teeth (SHED) after neural differentiation in vitro. Clin Oral Investig. 2010;14(4):433–440. [DOI] [PubMed] [Google Scholar]
- 58. Nakashima M, Iohara K, Murakami M. Dental pulp stem cells and regeneration. Endod Topics. 2013;28(1):38–50. [Google Scholar]
- 59. Ishizaka R, Iohara K, Murakami M, Fukuta O, Nakashima M. Regeneration of dental pulp following pulpectomy by fractionated stem/progenitor cells from bone marrow and adipose tissue. Biomaterials. 2012;33(7):2109–2118. [DOI] [PubMed] [Google Scholar]
- 60. Iohara K, Zheng L, Wake H, Ito M, Nabekura J, Wakita H, Nakamura H, Into T, Matsushita K, Nakashima M. A novel stem cell source for vasculogenesis in ischemia: subfraction of side population cells from dental pulp. Stem Cells. 2008;26(9):2408–2418. [DOI] [PubMed] [Google Scholar]
- 61. Shi X, Mao J, Liu Y. Pulp stem cells derived from human permanent and deciduous teeth: biological characteristics and therapeutic applications. Stem Cells Transl Med. 2020;9(4):445–464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Botelho J, Cavacas MA, Machado V, Mendes JJ. Dental stem cells: recent progresses in tissue engineering and regenerative medicine. Ann Med. 2017;49(8):644–651. [DOI] [PubMed] [Google Scholar]
- 63. Lan X, Sun Z, Chu C, Boltze J, Li S. Dental pulp stem cells: an attractive alternative for cell therapy in ischemic stroke. Front Neurol. 2019;10:824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Nakashima M, Iohara K, Murakami M, Nakamura H, Sato Y, Ariji Y, Matsushita K. Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: a pilot clinical study. Stem Cell Res Ther. 2017;8(1):61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Matthay MA, Ware LB, Zimmerman GA. The acute respiratory distress syndrome. J Clin Invest. 2012;122(8):2731–2740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Herold S, Gabrielli NM, Vadász I. Novel concepts of acute lung injury and alveolar-capillary barrier dysfunction. Am J Physiol Lung Cell Mol Physiol. 2013;305(10):L665–681. [DOI] [PubMed] [Google Scholar]
- 67. Wynn TA, Ramalingam TR. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012;18(7):1028–1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Wakayama H, Hashimoto N, Matsushita Y, Matsubara K, Yamamoto N, Hasegawa Y, Ueda M, Yamamoto A. Factors secreted from dental pulp stem cells show multifaceted benefits for treating acute lung injury in mice. Cytotherapy. 2015;17(8):1119–1129. [DOI] [PubMed] [Google Scholar]
- 69. Barbash IM, Chouraqui P, Baron J, Feinberg MS, Etzion S, Tessone A, Miller L, Guetta E, Zipori D, Kedes LH, Kloner RA, et al. Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: Feasibility, cell migration, and body distribution. Circulation. 2003;108(7):863–868. [DOI] [PubMed] [Google Scholar]
- 70. Egusa H, Sonoyama W, Nishimura M, Atsuta I, Akiyama K. Stem cells in dentistry-part I: stem cell sources. J Prosthodont Res. 2012;56(3):151–165. [DOI] [PubMed] [Google Scholar]
- 71. Iohara K, Zheng L, Ito M, Tomokiyo A, Matsushita K, Nakashima M. Side population cells isolated from porcine dental pulp tissue with self-renewal and multipotency for dentinogenesis, chondrogenesis, adipogenesis, and neurogenesis. Stem Cells. 2006;24(11):2493–2503. [DOI] [PubMed] [Google Scholar]
- 72. Tsutsui TW. Dental pulp stem cells: advances to applications. Stem Cells Cloning. 2020;13:33–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG, Shi S. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci. U S A. 2003;100(10):5807–5812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Orimoto A, Kyakumoto S, Eitsuka T, Nakagawa K, Kiyono T, Fukuda T. Efficient immortalization of human dental pulp stem cells with expression of cell cycle regulators with the intact chromosomal condition. PLoS One. 2020;15(3):e0229996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Li X, Wang L, Su Q, Ye L, Zhou X, Song D, Huang D. Highly proliferative immortalized human dental pulp cells retain the odontogenic phenotype when combined with a beta-tricalcium phosphate scaffold and BMP2. Stem Cells Int. 2020;2020:4534128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Wilson R, Urraca N, Skobowiat C, Hope KA, Miravalle L, Chamberlin R, Donaldson M, Seagroves TN, Reiter LT. Assessment of the tumorigenic potential of spontaneously immortalized and htert-immortalized cultured dental pulp stem cells. Stem Cells Transl Med. 2015;4(8):905–912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Ye Q, Wang H, Xia X, Zhou C, Liu Z, Xia Z-e, Zhang Z, Zhao Y, Yehenala J, Wang S, Zhou G, et al. Safety and efficacy assessment of allogeneic human dental pulp stem cells to treat patients with severe COVID-19: structured summary of a study protocol for a randomized controlled trial (phase I / II). Trials. 2020;21(1):520. [DOI] [PMC free article] [PubMed] [Google Scholar]