Abstract
The present study aimed to clarify the influence of long non-coding RNA small nuclear host gene 16 (lncRNA SNHG16) on cardiomyocyte proliferation following ischemia/reperfusion injury (IRI) and the potential mechanism. An IRI model in mice was established by performing ligation of the anterior descending coronary artery (LAD). Primary cardiomyocytes were isolated from newborn mice and subjected to H2O2 treatment to mimic in vitro IRI. Relative levels of SNHG16 and miRNA-770-5p in both in vivo and in vitro IRI models were examined. The regulatory effects of SNHG16 and miRNA-770-5p on the proliferative ability of H2O2-treated cardiomyocytes were assessed by Cell Counting Kit-8 (CCK-8) and 5-ethynyl-2'-deoxyuridine (EdU) assay. The binding relationship between SNHG16 and miRNA-770-5p was verified through dual-luciferase reporter gene assay. It is found that SNHG16 was time-dependently downregulated in the IRI models. Overexpression of SNHG16 enhanced the proliferative ability of the cardiomyocytes. miRNA-770-5p was found to be a direct target of SNHG16. Moreover, SNHG16 was able to negatively regulate the miRNA-770-5p level. Overexpression of miRNA-770-5p partially reversed the role of SNHG16 on accelerating cardiomyocyte proliferation. Collectively, SNHG16 accelerates the proliferative ability of cardiomyocytes following IRI by negatively regulating miRNA-770-5p.
Keywords: SNHG16, miRNA-770-5p, ischemia/reperfusion injury
Introduction
Globally, coronary heart disease is one of the most common, high-risk diseases and the major reason for death and disability. Acute myocardial ischemia/reperfusion injury (IRI) is the leading cause of coronary heart disease worldwide (1). After restoring blood supply to ischemic myocardium, dysfunction of cell metabolism and structural damage are further aggravated, which eventually lead to IRI (2). IRI is commonly seen after cardiac surgery. Oxidative stress is believed to be the major mechanism underlying the pathogenesis of IRI (3,4). With the enhanced incidence of coronary heart diseases, myocardial infarction and cardiac surgery, IRI is a common focus of research. It is necessary to uncover the pathogenesis of IRI to effectively prevent its occurrence.
Long non-coding RNA (lncRNA) is a transcript with 200 nt in length and it cannot directly encode proteins (5). Several lncRNAs have been discovered to be dysregulated during the progression of IRI, which provides a promising direction for uncovering the pathogenesis of IRI (6-10). For example, it has previously been reported that lncRNA nuclear enriched abundant transcript 1 (NEAT1) can protect cardiomyocytes from apoptosis via targeting miR-520a by modulating expression levels of apoptosis-related proteins (11). Gong et al indicated that upregulation of MALAT1 exerts a critical role in promoting cardiomyocyte apoptosis by acting as a competing endogenous RNAs (ceRNA) to sponge miR-144(12). In addition, knockdown of NEAT1 can protect against IRI by inhibiting troponin levels and cardiocytes apoptosis through regulation of the mitogen-activated protein kinase (MAPK) signaling pathway (13). Therefore, clarification of lncRNAs involved in the occurrence and development of IRI contributes to improve the prognosis of affected individuals.
lncRNA small nuclear host gene 16 (SNHG16) is an important member of the SNHG family, serving as an oncogene in osteosarcoma (14), bladder cancer (15), lung cancer (16), glioma (17), ovarian cancer (18) and breast cancer (19). Moreover, a previous study showed that CTCFI-induced upregulation of SNHG16 accelerated cardiac hypertrophy by targeting the miR-182-5p/IGF1 axis (20). Furthermore, SNHG16 was found to promote proliferation and inflammatory response of macrophages through the miR-17-5p/NF-κB signaling pathway in patients with atherosclerosis (21). However, it is unclear whether SNHG16 is involved in the proliferative change in cardiomyocytes following IRI. In the present study, we constructed both in vivo and in vitro IRI models by performing ligation of the anterior descending coronary artery (LAD) in mice and by exposing primary cardiomyocytes to H2O2, respectively. We aimed to discover the biological role of SNHG16 in affecting the proliferative ability of cardiomyocytes.
Materials and methods
Establishment of I/R mouse model
A total of 50 male mice (180-220 g, 6-8 weeks of age) obtained from the Experimental Animal Center, Qingdao University were randomly divided into 5 groups (sham group and 4 time-point groups, 10 mice for each group). Mice were housed in a temperature controlled room (21±2˚C) with a 12:12-h light/dark cycle (lights on at 06:00). All mice had free access to water and food. Animal experiments were repeated in triplicate, and a total of 150 mice were used in this study. The mice were anesthetized with 10% chloral hydrate at a dose of 200-250 mg/kg, intubated, and mechanically ventilated. No mice exhibited signs of peritonitis after the administration of 10% chloral hydrate. After thoracotomy, the left anterior descending coronary artery was inserted using 4.0 suture at the beginning 2-3 cm. A small soft film was placed between the suture and the artery. Ligation was performed after observation of 15-min stabilization of breath and blood pressure. Thirty minutes later, the ligature was removed and reperfusion was allowed for 120 min. For the sham group, mice were subjected to thoracotomy without ligation. During the experiments, mouse electrocardiogram was monitored. ST segment elevation and upright T wave were observed after ligation, which were relieved after reperfusion. All animal protocols were approved by the Animal Ethics Committee of Qingdao University (Qingdao, Shandong, China).
Cell culture
According to previously described methods (22,23), newborn mice (1-2 days of age) were sacrificed by cervical dislocation (after being anesthetized using 10% chloral hydrate at a dose of 250 mg/kg). No mice exhibited signs of peritonitis after the administration of 10% chloral hydrate. Mouse heart was immediately collected, washed in ice-cold HBSS and cut into 1-mm3 pieces. Heart tissues were digested in trypsin containing 0.125% EDTA (ethylenediaminetetraacetic acid) overnight at 4˚C in the dark. On the following day, heart homogenate was incubated in collagenase II solution in a water bath shaker (37˚C, 100 x g). Dulbecco's modified Eagle's medium (DMEM) (Gibco; Thermo Fisher Scientific, Inc.) containing 10% fetal bovine serum (FBS) (Gibco; Thermo Fisher Scientific, Inc.) was utilized to maintain the activity of cardiomyocytes. In order to construct the IRI model in vitro, the cells were cultured in the medium containing 200 µmol/l H2O2 for 0, 6, 12, 24 h, respectively.
Cell transfection
Cardiomyocytes were inoculated in a 96-well plate at 6x106 cells/ml. Twenty-four hours later, the cells were transfected with plasmid using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.) following the manufacturer's instructions. After transfection for 24 h, the cells were then used for the experiments. The final concentration of the pRL-CMV plasmids was 0.1 µg/µl. All the mimics, inhibitors and plasmids were purchased from GenePharma (Shanghai, China). The sequences were: miR-770-5p mimic, 5'-UCCAGUACCACGUGUCAGGGCCA-3'; miRNA-770-5p-control, 5'-UCGCUUGGUGCAGGUCGGGAA-3 and miR-770-5p inhibitor, 5'-UGGCCCUGACACGUGGUACUGGA-3'.
RNA extraction and quantitative real-time polymerase chain reaction (qPCR)
Extraction of total RNA in cells was performed using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.). RNA was reversely transcribed into complementary deoxyribose nucleic acid (cDNA), and the latter was applied for PCR using SYBR Green method (Takara) for 5 min at 94˚C, and 40 cycles for 30 sec at 94˚C, 30 sec at 55˚C and 90 sec at 72˚C with Roche LightCycler 480 (Roche, USA). U6 and GAPDH were enrolled as internal controls. Primers were as below: SNHG16 forward, 5'-CAGAATGCCATGGTTTCCCC-3' and reverse, 5'-TGGCAAGAGACTTCCTGAGG-3'; GAPDH forward, 5'-CGGAGTCAACGGATTTGGTCGTAT-3' and reverse, 5'-AGCCTTCTCCATGGTGGTGAAGAC-3'; U6 forward, 5'-CTCGCTTCGGCAGCACA-3' and reverse, 5'-AACGCTTCACGAATTTGCGT-3'.
Dual-luciferase reporter gene assay
Cardiomyocytes were co-transfected with SNHG16-MT (mutant)/SNHG16-WT (wild-type) and miRNA-770-5p mimics/NC, respectively. After transfection of 48 h, cells were lysed for determining relative luciferase activity with a Promega device (Promega Corp.). The 3'-UTR of wild-type EGFR and the containing mutations in the putative binding site were inserted downstream of the firefly luciferase reporter into the psiCHECK-2 vector (Promega Corp.). The corresponding mutant construct was created through mutating the seed regions of the miR-133b binding sites and was named as 3'-UTR mut EGFR. Cells were seeded into a 12-well plate at 80% confluence and cotransfected with 0.5 µg reporter plasmid, 40 nM miR-133b mimics or negative control with Lipofectamine 2000. For the reason of transfection efficiency, all samples were cotransfected with 0.05 µg pRL-CMV plasmid expressing Renilla Luciferase (Promega Corp.) as an internal control. Luciferase assay was conducted 48 h after transfection by Dual Luciferase Reporter Assay System (Promega Corp.). Firefly luciferase activity was standardized to Renilla luciferase activity. There experiments were conducted three times for each assay.
Cell Counting Kit-8 (CCK-8)
Cardiomyocytes were seeded in the 96-well plate at 2x103 cells per well and cultured overnight. Absorbance (A) at 450 nm was recorded at the appointed time points using the CCK-8 Kit (Dojindo Laboratories) by the TECAN infinite M200 Multimode microplate reader (Tecan).
5-Ethynyl-2'-deoxyuridine (EdU)
Cardiomyocytes were seeded in a 96-well plate with 300 cells per well. Cardiomyocytes were labeled with 50 µmol/l EdU at 37˚C for 2 h. After 30-min fixation in 4% paraformaldehyde, cells were incubated with phosphate-buffered saline (PBS) containing 0.5% Triton-100 for 20 min. After washing with PBS containing 3% bovine serum albumin (BSA), 100 µl of dying solution was applied per well for a 1-h incubation in dark. Subsequently, the cells were counterstained with 100 µl of Hoechst for 10 min. EdU-positive cells, Hoechst-labeled cells and merged images were captured under a fluorescent microscope (IX70; Olympus).
Statistical analyses
Statistical Product and Service Solutions (SPSS) 18.0 (SPSS Inc.) was used for data analyses. Data are expressed as mean ± standard deviation. Differences between groups were analyzed by the t-test. Comparisons among multiple groups were performed by one-way analysis of variance (ANOVA) followed by least significant difference (LSD) analysis. Comparisons among four groups were analyzed by Tukey's test. Each experiment was repeated at least three times. P<0.05 was considered to indicate a statistically significant difference.
Results
SNHG16 is downregulated in IRI models
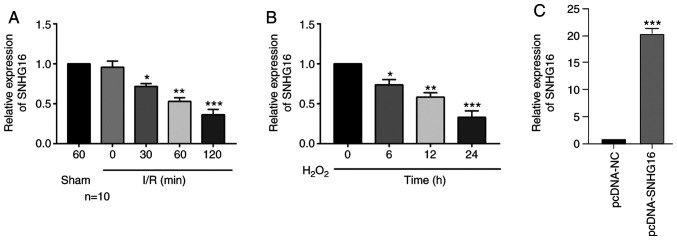
The in vivo IRI model in mice was first constructed by performing LAD. With the prolongation of reperfusion, the relative level of SNHG16 was significantly downregulated (Fig. 1A). Meanwhile, primary cardiomyocytes were isolated from newborn mice and subjected to H2O2 treatment. SNHG16 was significantly downregulated as well with the prolongation of H2O2 treatment in cardiomyocytes (Fig. 1B).
Figure 1.
SNHG16 is downregulated in the IRI models. (A) Relative level of SNHG16 in mice undergoing sham operations for 60 min, and IRI for 0, 30, 60 and 120 min. *P<0.05, **P<0.01, ***P<0.001, compared to the sham group. (B) Relative level of SNHG16 in primary cardiomyocytes exposed to H2O2 for 0, 6, 12 and 24 h. *P<0.05, **P<0.01, ***P<0.001, compared to the 0 h group. (C) Relative level of SNHG16 in primary cardiomyocytes transfected with pcDNA-NC and pcDNA-SNHG16. ***P<0.001, compared to the pcDNA-NC group. SNHG16, small nuclear host gene 16; IRI, ischemia/reperfusion injury.
SNHG16 accelerates the proliferation of cardiomyocytes
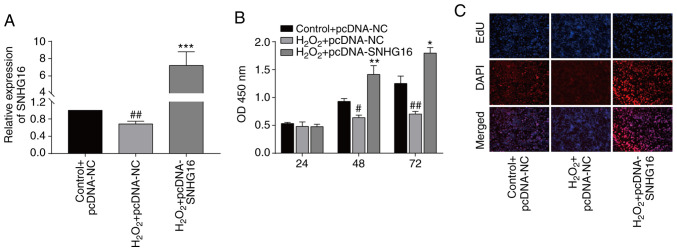
To uncover the biological function of SNHG16 in IRI, we constructed pcDNA-SNHG16. Transfection of pcDNA-SNHG16 in untreated and H2O2-treated cardiomyocytes markedly upregulated the SNHG16 level, indicating a high transfection efficacy (Figs. 1C and 2A). At 48 and 72 h, transfection of pcDNA-SNHG16 in H2O2-treated cardiomyocytes enhanced the viability relative to the H2O2+pcDNA-NC group (Fig. 2B). EdU assay identically showed an elevation in the EdU-positive rate after overexpression of SNHG16 in cardiomyocytes treated with H2O2 compared to H2O2+pcDNA-NC group (Fig. 2C).
Figure 2.
SNHG16 accelerates the proliferative ability of cardiomyocytes. (A) Relative level of SNHG16 in the primary cardiomyocytes following transfection and/or exposure to H2O2 in the following groups, control+pcDNA-NC, H2O2+pcDNA-NC or H2O2+pcDNA-SNHG16. ##P<0.01, H2O2+pcDNA-SNHG16 group vs. H2O2+pcDNA-NC group, ***P<0.001, H2O2+pcDNA-SNHG16 vs. control+pcDNA-NC group. (B) CCK-8 assay demonstrates the viability in the primary cardiomyocytes in the groups: Control+pcDNA-NC, H2O2+pcDNA-NC or H2O2+pcDNA-SNHG16. #P<0.05, ##P<0.01 H2O2+pcDNA-SNHG16 group vs. H2O2+pcDNA-NC group, *P<0.05, **P<0.01, H2O2+pcDNA-SNHG16 group vs. control+pcDNA-NC group. (C) DAPI-labeled, EdU-labeled and merged images of primary cardiomyocytes in the group: Control+pcDNA-NC, H2O2+pcDNA-NC or H2O2+pcDNA-SNHG16 (magnification, x100). SNHG16, small nuclear host gene 16.
SNHG16 was able to bind to miRNA-770-5p
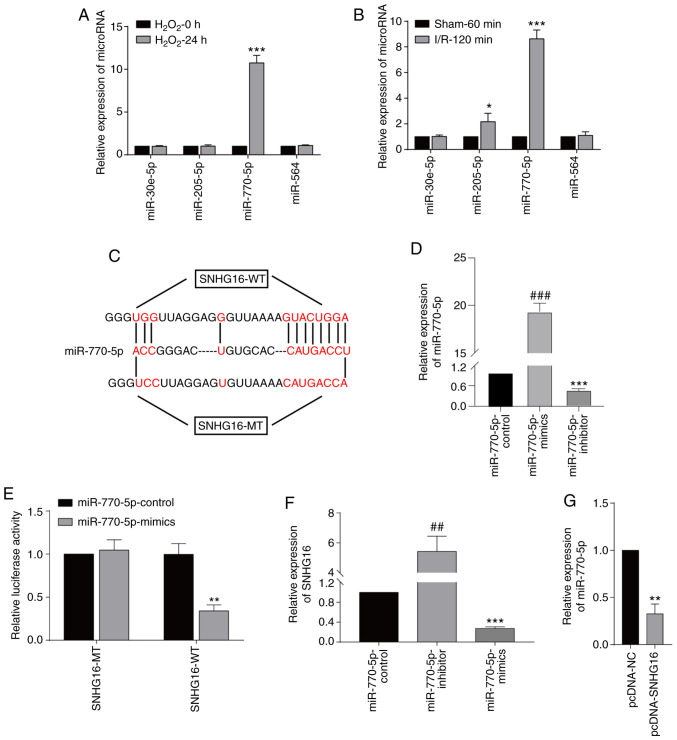
In order to predict the targets of SNHG16, the sequence of SNHG16 was obtained from NCBI (https://www.ncbi.nlm.nih.gov), and then the sequence was uploaded to the online tool miRDB (http://mirdb.org). The results showed that miRNA-30e-5p, miRNA-205-5p, miRNA-770-5p and miRNA-564 were predicted to be the targets of SNHG16. miRNA-770-5p was the only miRNA to be significantly upregulated after 24-h H2O2 treatment in the cardiomyocytes (Fig. 3A). In mice undergoing 120-min I/R injury, only the miRNA-770-5p level was highly significantly upregulated (Fig. 3B). Potential binding sequences between SNHG16 and miRNA-770-5p were identified (Fig. 3C). Transfection of the miR-770-5p-mimics and miR-770-5p-inhibitor significantly upregulated and downregulated miR-770-5p, respectively, in the cardiomyocytes when compared to the miR-770-5p-control (Fig. 3D). Furthermore, luciferase activity was markedly reduced after co-transfection of SNHG16-WT and miRNA-770-5p mimics, verifying their binding relationship (Fig. 3E). Transfection of the miRNA-770-5p inhibitor significantly upregulated the SNHG16 level, and in contrast, transfection of the miRNA-770-5p mimics significantly elevated the SNHG16 level (Fig. 3F). Moreover, transfection of pcDNA-SNHG16 significantly downregulated the miRNA-770-5p level, confirming their negative correlation (Fig. 3G).
Figure 3.
SNHG16 binds to miR-770-5p. (A) Relative levels of miRNA-30e-5p, miRNA-205-5p, miRNA-770-5p and miRNA-564 in primary cardiomyocytes undergoing 0 or 24-h H2O2 exposure. ***P<0.001 H2O2-24 h group vs. H2O2-0 h group (B) Relative levels of miRNA-30e-5p, miRNA-205-5p, miRNA-770-5p and miRNA-564 in mice undergoing 60-min sham operation or 120-min IRI. *P<0.05, ***P<0.001, I/R-120 min vs. sham 60 group (C) Potential binding sequences between SNHG16 and miR-770-5p. (D) Relative level of miR-770-5p in primary cardiomyocytes transfected with miR-770-5p-control, miR-770-5p-mimics and miR-770-5p-inhibitor. ###P<0.001, miR-770-5p-inhibitor group vs. miR-770-5p-mimics group, ***P<0.001, miR-770-5p-inhibitor group vs. miR-770-5p-control group. (E) Dual-luciferase reporter gene assay showed the luciferase activity in cardiomyocytes co-transfected with SNHG16-MT/SNHG16-WT and miRNA-770-5p mimics/NC. **P<0.01, miR-770-5p-mimics group vs. miR-770-5p-control group. (F) SNHG16 expression following miRNA-770-5p mimics or miRNA-770-5p inhibitor transfection. ##P<0.01, miR-770-5p-inhibitor group vs. miR-770-5p-mimics group, ***P<0.001, miR-770-5p-mimics group vs. miR-770-5p-control group. (G) Relative levels of miR-770-5p in cardiomyocytes transfected with pcDNA-NC or pcDNA-SNHG16. **P<0.01, pcDNA-NC group vs. pcDNA-SNHG16 group. SNHG16, small nuclear host gene 16; IRI, ischemia/reperfusion injury.
Overexpression of miRNA-770-5p reversed the role of SNHG16 in cardiomyocytes
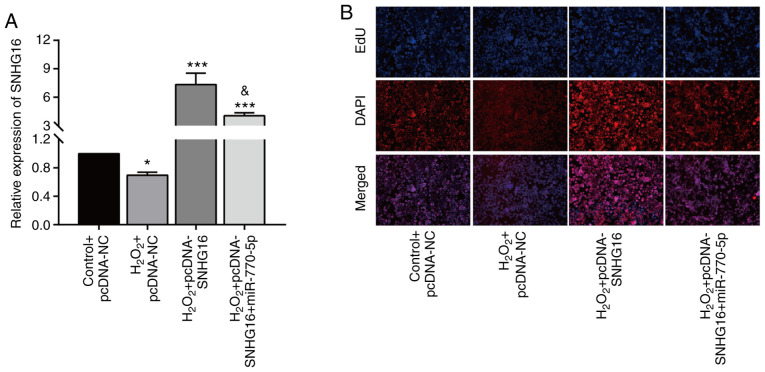
Rescue experiments were carried out to explore the involvement of miRNA-770-5p in SNHG16-mediated cardiomyocyte cytoactivation. After H2O2 treatment, the upregulation of SNHG16 in cardiomyocytes transfected with pcDNA-SNHG16 was partially downregulated by co-transfection of miRNA-770-5p mimics (Fig. 4A). Moreover, the enhanced EdU-positive rate in cardiomyocytes overexpressing SNHG16 was reduced to some extent by miRNA-770-5p overexpression (Fig. 4B). Collectively, SNHG16 promotes the cytoactivation of cardiomyocytes in H2O2-treated cardiomyocytes by negatively regulating miRNA-770-5p.
Figure 4.
Overexpression of miR-770-5p reverses the effect of SNHG16 on cardiomyocytes. (A) Relative level of SNHG16 in primary cardiomyocytes in the control+pcDNA-NC, H2O2+pcDNA-NC, H2O2+pcDNA-SNHG16 or H2O2+pcDNA-SNHG16+miR-770-5p mimic groups. *P<0.05, ***P<0.001, compared to the control+pcDNA-NC group; &P<0.05, compared to the H2O2+pcDNA-SNHG16 group. (B) DAPI-labeled, EdU-labeled and merged images of primary cardiomyocytes transfected in the control+pcDNA-NC, H2O2+pcDNA-NC, H2O2+pcDNA-SNHG16 or H2O2+pcDNA-SNHG16+miR-770-5p mimic groups (magnification, x100). SNHG16, small nuclear host gene 16.
Discussion
Ischemic heart disease following myocardial infarction and congestive heart failure is the leading cause of global death (24). With the improvement of therapeutic strategies, thrombolytic therapy is widely applied in the treatment of acute myocardial infarction. It is able to quickly restore the blood flow of the coronary artery in the ischemic area, causing myocardial reperfusion, and decline of necrotic myocardial area (25). However, studies have shown that reperfusion after ischemia can aggravate cardiomyocyte damage, mainly manifested by accelerated myocardial necrosis, myocardial infarct size expansion, no-reflow phenomenon, arrhythmia and other symptoms (25,26). Therefore, how to minimize the occurrence of reperfusion injury has become the focus of research.
Recent studies have shown the involvement of lncRNAs in the regulation of cardiovascular disease (27,28). Wang et al (29) reported that lncRNA NRF regulates programmed necrosis and myocardial injury during ischemia/reperfusion injury (IRI) by targeting miR-873, providing a potential target for the treatment of myocardial ischemic diseases. In focal myocardial IRI, lncRNA UCA1 was found to negatively regulate p27 expression and to exert a pro-apoptotic role in primary cardiomyocytes (30). In the present paper, long non-coding RNA small nuclear host gene 16 (lncRNA SNHG16) was time-dependently downregulated in mice undergoing LAD and in primary cardiomyocytes treated with H2O2. Both CCK-8 and EdU assays demonstrated the promotive effect of SNHG16 on the proliferative ability of cardiomyocytes. The findings indicate that SNHG16 may be a key regulator involved in IRI.
A growing body of evidence has confirmed that lncRNAs regulate miRNA expression and functions as competitive endogenous (ce)RNAs (31,32). The ceRNA theory proposes a complex regulatory network involving miRNAs, lncRNAs and circular (circ)RNAs. They usually contain mRNA binding sites and influence tumor progression by competitively binding microRNA response elements (MREs) (33-35). For example, SNHG16 was found to promote the proliferative ability of osteosarcoma by competitively binding miR-205 and further upregulating ZEB1 (14). SNHG16 was found to mediate HMGB1 expression by absorbing miR-218-5p, thus exerting a carcinogenic effect on pancreatic cancer (36). The present study found many miRNA-binding sites in the sequences of SNHG16 through bioinformatics analysis. Furthermore, dual-luciferase reporter gene assay confirmed the binding between SNHG16 and miRNA-770-5p. In primary cardiomyocytes, SNHG16 was negatively correlated with miRNA-770-5p. In addition, overexpression of miRNA-770-5p partially reversed the role of SNHG16 in accelerating cardiomyocyte proliferation.
In conclusion, SNHG16 was downregulated in the IRI mouse model and in the H2O2-exposed primary cardiomyocytes. Overexpression of SNHG16 accelerated the proliferative ability of cardiomyocytes following IRI by negatively regulating miRNA-770-5p.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
LY and TW designed the study and performed the experiments. LY and YL established the animal models. JM and YP collected the data, RY and YW analyzed the data, and LY and TW prepared the manuscript. All authors read and approved the manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics approval and consent to participate
This study was approved by the Animal Ethics Committee of Qingdao University Animal Center.
Patient consent for publication
No patients participated in this study.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Hausenloy DJ, Yellon DM. Myocardial ischemia-reperfusion injury: A neglected therapeutic target. J Clin Invest. 2013;123:92–100. doi: 10.1172/JCI62874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Song XD, Feng JP, Yang RX. Alamandine protects rat from myocardial ischemia-reperfusion injury by activating JNK and inhibiting NF-KB. Eur Rev Med Pharmacol Sci. 2019;23:6718–6726. doi: 10.26355/eurrev_201908_18563. [DOI] [PubMed] [Google Scholar]
- 3.Ibanez B, Heusch G, Ovize M, Van de Werf F. Evolving therapies for myocardial ischemia/reperfusion injury. J Am Coll Cardiol. 2015;65:1454–1471. doi: 10.1016/j.jacc.2015.02.032. [DOI] [PubMed] [Google Scholar]
- 4.Martindale JJ, Metzger JM. Uncoupling of increased cellular oxidative stress and myocardial ischemia reperfusion injury by directed sarcolemma stabilization. J Mol Cell Cardiol. 2014;67:26–37. doi: 10.1016/j.yjmcc.2013.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li X, Wu Z, Fu X, Han W. Long noncoding RNAs: Insights from biological features and functions to diseases. Med Res Rev. 2013;33:517–553. doi: 10.1002/med.21254. [DOI] [PubMed] [Google Scholar]
- 6.Schmitz SU, Grote P, Herrmann BG. Mechanisms of long noncoding RNA function in development and disease. Cell Mol Life Sci. 2016;73:2491–2509. doi: 10.1007/s00018-016-2174-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dabek J, Owczarek A, Gasior Z, Ulczok R, Skowerski M, Kulach A, Mazurek U, Bochenek A. Oligonucleotide microarray analysis of genes regulating apoptosis in chronically ischemic and postinfarction myocardium. Biochem Genet. 2008;46:241–247. doi: 10.1007/s10528-007-9137-3. [DOI] [PubMed] [Google Scholar]
- 8.Pitts KR, Derry JM, Kerkof K, Lawrence WA, Toombs CF. Differentially regulated functional gene clusters identified during ischemia and reperfusion in isolated cardiac myocytes using coverslip hypoxia. J Pharmacol Toxicol Methods. 2008;57:42–51. doi: 10.1016/j.vascn.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 9.Mercer TR, Mattick JS. Structure and function of long noncoding RNAs in epigenetic regulation. Nat Struct Mol Biol. 2013;20:300–307. doi: 10.1038/nsmb.2480. [DOI] [PubMed] [Google Scholar]
- 10.Small EM, Olson EN. Pervasive roles of microRNAs in cardiovascular biology. Nature. 2011;469:336–342. doi: 10.1038/nature09783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu HJ, Tang GM, Shao PY, Zou HX, Shen WF, Huang MD, Pan HH, Zhai CL, Qian G. Long non-coding RNA NEAT1 modulates hypoxia/reoxygenation-induced cardiomyocyte injury via targeting microRNA-520a. Exp Ther Med. 2019;18:2199–2206. doi: 10.3892/etm.2019.7788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gong X, Zhu Y, Chang H, Li Y, Ma F. Long noncoding RNA MALAT1 promotes cardiomyocyte apoptosis after myocardial infarction via targeting miR-144-3p. Biosci Rep. 2019;39(BSR20191103) doi: 10.1042/BSR20191103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Du XJ, Wei J, Tian D, Yan C, Hu P, Wu X, Yang W, Hu X. NEAT1 promotes myocardial ischemia-reperfusion injury via activating the MAPK signaling pathway. J Cell Physiol. 2019;234:18773–18780. doi: 10.1002/jcp.28516. [DOI] [PubMed] [Google Scholar]
- 14.Zhu C, Cheng D, Qiu X, Zhuang M, Liu Z. Long noncoding RNA SNHG16 promotes cell proliferation by sponging MicroRNA-205 and upregulating ZEB1 expression in osteosarcoma. Cell Physiol Biochem. 2018;51:429–440. doi: 10.1159/000495239. [DOI] [PubMed] [Google Scholar]
- 15.Feng F, Chen A, Huang J, Xia Q, Chen Y, Jin X. Long noncoding RNA SNHG16 contributes to the development of bladder cancer via regulating miR-98/STAT3/Wnt/β-catenin pathway axis. J Cell Biochem. 2018;119:9408–9418. doi: 10.1002/jcb.27257. [DOI] [PubMed] [Google Scholar]
- 16.Han W, Du X, Liu M, Wang J, Sun L, Li Y. Increased expression of long non-coding RNA SNHG16 correlates with tumor progression and poor prognosis in non-small cell lung cancer. Int J Biol Macromol. 2019;121:270–278. doi: 10.1016/j.ijbiomac.2018.10.004. [DOI] [PubMed] [Google Scholar]
- 17.Yang BY, Meng Q, Sun Y, Gao L, Yang JX. Long non-coding RNA SNHG16 contributes to glioma malignancy by competitively binding miR-20a-5p with E2F1. J Biol Regul Homeost Agents. 2018;32:251–261. [PubMed] [Google Scholar]
- 18.Yang XS, Wang GX, Luo L. Long non-coding RNA SNHG16 promotes cell growth and metastasis in ovarian cancer. Eur Rev Med Pharmacol Sci. 2018;22:616–622. doi: 10.26355/eurrev_201802_14284. [DOI] [PubMed] [Google Scholar]
- 19.Cai C, Huo Q, Wang X, Chen B, Yang Q. SNHG16 contributes to breast cancer cell migration by competitively binding miR-98 with E2F5. Biochem Biophys Res Commun. 2017;485:272–278. doi: 10.1016/j.bbrc.2017.02.094. [DOI] [PubMed] [Google Scholar]
- 20.Wang D, Lin B, Zhang W, Wang X. doi: 10.1002/cbin.11333. Up-regulation of SNHG16 induced by CTCF accelerates cardiac hypertrophy by targeting miR-182-5p/IGF1 axis. Call Biol Int, Mar 3, 2020 (Epub ahead of print). [DOI] [PubMed] [Google Scholar]
- 21.An JH, Chen ZY, Ma QL, Wang HJ, Zhang JQ, Shi FW. LncRNA SNHG16 promoted proliferation and inflammatory response of macrophages through miR-17-5p/NF-κB signaling pathway in patients with atherosclerosis. Eur Rev Med Pharmacol Sci. 2019;23:8665–8677. doi: 10.26355/eurrev_201910_19184. [DOI] [PubMed] [Google Scholar]
- 22.Goldenberg I, Shainberg A, Jacobson KA, Shneyvays V, Grossman E. Adenosine protects against angiotensin II-induced apoptosis in rat cardiocyte cultures. Mol Cell Biochem. 2003;252:133–139. doi: 10.1023/a:1025551229566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen J, Larsson L, Haugen E, Fedorkova O, Angwald E, Waagstein F, Fu M. Effects of autoantibodies removed by immunoadsorption from patients with dilated cardiomyopathy on neonatal rat cardiomyocytes. Eur J Heart Fail. 2006;8:460–467. doi: 10.1016/j.ejheart.2005.10.019. [DOI] [PubMed] [Google Scholar]
- 24.Pagidipati NJ, Gaziano TA. Estimating deaths from cardiovascular disease: A review of global methodologies of mortality measurement. Circulation. 2013;127:749–756. doi: 10.1161/CIRCULATIONAHA.112.128413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dong J, Zhao Y, He XK. Down-regulation of miR-192 protects against rat ischemia-reperfusion injury after myocardial infarction. Eur Rev Med Pharmacol Sci. 2018;22:6109–6118. doi: 10.26355/eurrev_201809_15950. [DOI] [PubMed] [Google Scholar]
- 26.Wu H, Ye M, Yang J, Ding J. Modulating endoplasmic reticulum stress to alleviate myocardial ischemia and reperfusion injury from basic research to clinical practice: A long way to go. Int J Cardiol. 2016;223:630–631. doi: 10.1016/j.ijcard.2016.08.266. [DOI] [PubMed] [Google Scholar]
- 27.Wang K, Long B, Zhou LY, Liu F, Zhou QY, Liu CY, Fan YY, Li PF. CARL lncRNA inhibits anoxia-induced mitochondrial fission and apoptosis in cardiomyocytes by impairing miR-539-dependent PHB2 downregulation. Nat Commun. 2014;5(3596) doi: 10.1038/ncomms4596. [DOI] [PubMed] [Google Scholar]
- 28.Viereck J, Kumarswamy R, Foinquinos A, Xiao K, Avramopoulos P, Kunz M, Dittrich M, Maetzig T, Zimmer K, Remke J, et al. Long noncoding RNA Chast promotes cardiac remodeling. Sci Transl Med. 2016;8(326ra22) doi: 10.1126/scitranslmed.aaf1475. [DOI] [PubMed] [Google Scholar]
- 29.Wang K, Liu F, Liu CY, An T, Zhang J, Zhou LY, Wang M, Dong YH, Li N, Gao JN, et al. The long noncoding RNA NRF regulates programmed necrosis and myocardial injury during ischemia and reperfusion by targeting miR-873. Cell Death Differ. 2016;23:1394–1405. doi: 10.1038/cdd.2016.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu Y, Zhou D, Li G, Ming X, Tu YF, Tian J, Lu H, Yu B. Long non coding RNA-UCA1 contributes to cardiomyocyte apoptosis by suppression of p27 expression. Cell Physiol Biochem. 2015;35:1986–1998. doi: 10.1159/000374006. [DOI] [PubMed] [Google Scholar]
- 31.Ulitsky I. Interactions between short and long noncoding RNAs. FEBS Lett. 2018;592:2874–2883. doi: 10.1002/1873-3468.13085. [DOI] [PubMed] [Google Scholar]
- 32.Chan JJ, Tay Y. Noncoding RNA: RNA regulatory networks in cancer. Int J Mol Sci. 2018;19(1310) doi: 10.3390/ijms19051310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tay Y, Rinn J, Pandolfi PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014;505:344–352. doi: 10.1038/nature12986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kartha RV, Subramanian S. Competing endogenous RNAs (ceRNAs): New entrants to the intricacies of gene regulation. Front Genet. 2014;5(8) doi: 10.3389/fgene.2014.00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Karreth FA, Pandolfi PP. ceRNA cross-talk in cancer: When ce-bling rivalries go awry. Cancer Discov. 2013;3:1113–1121. doi: 10.1158/2159-8290.CD-13-0202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu S, Zhang W, Liu K, Liu Y. LncRNA SNHG16 promotes tumor growth of pancreatic cancer by targeting miR-218-5p. Biomed Pharmacother. 2019;114(108862) doi: 10.1016/j.biopha.2019.108862. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.