Abstract
Background
Early risk stratification applying cardiac biomarkers may prove useful in sudden cardiac arrest patients. We investigated the prognostic utility of early-on levels of high sensitivity cardiac troponin-T (hs-cTnT), copeptin and N-terminal pro-B-type natriuretic peptide (NT-proBNP) in patients with out-of-hospital cardiac arrest (OHCA).
Methods
We conducted a prospective observational unicenter study, including patients with OHCA of assumed cardiac origin from the southwestern part of Norway from 2007 until 2010. Blood samples for later measurements were drawn during cardiopulmonary resuscitation or at hospital admission.
Results
A total of 114 patients were included, 37 patients with asystole and 77 patients with VF as first recorded heart rhythm. Forty-four patients (38.6%) survived 30-day follow-up. Neither hs-cTnT (p = 0.49), nor copeptin (p = 0.39) differed between non-survivors and survivors, whereas NT-proBNP was higher in non-survivors (p < 0.001) and significantly associated with 30-days all-cause mortality in univariate analysis, with a hazard ratio (HR) for patients in the highest compared to the lowest quartile of 4.6 (95% confidence interval (CI), 2.1–10.1), p < 0.001. This association was no longer significant in multivariable analysis applying continuous values, [HR 0.96, (95% CI, 0.64–1.43), p = 0.84]. Similar results were obtained by dividing the population by survival at hospital admission, excluding non-return of spontaneous circulation (ROSC) patients on scene [HR 0.93 (95% CI, 0.50–1.73), P = 0.83]. We also noted that NT-proBNP was significantly higher in asystole- as compared to VF-patients, p < 0.001.
Conclusions
Early-on levels of hs-cTnT, copeptin and NT-proBNP did not provide independent prognostic information following OHCA. Prediction was unaffected by excluding on-scene non-ROSC patients in the multivariable analysis.
Trial registration
ClinicalTrials. gov, NCT02886273.
Keywords: Out-of-hospital cardiac arrest, Prognosis, High-sensitivity cardiac troponin T, Copeptin, N-terminal pro-B-type natriuretic peptide
Background
Out-of-hospital cardiac arrest (OHCA) is a leading cause of death and represents a major health problem in western countries. In Europe, the annual incidence of Emergency Medical Services (EMS) attended OHCA is estimated to 84 per 100,000 inhabitants [1]. The mortality rate is high, with an overall rate of survival to hospital discharge of only 10% [1, 2]. Early and precise risk stratification of cardiac arrest patients is important to guide further treatment decisions. On-scene observations [3] and 24-h intensive care follow-up recordings [4, 5] are currently being used to predict survival from OHCA.
Heart disease is a major risk factor for sudden cardiac arrest [6], with coronary artery disease and cardiomyopathies as the two most common causes of sudden cardiac death [6, 7]. Cardiac arrest with global ischemia and subsequent resuscitation may contribute to myocardial injury and dysfunction [8, 9]. Cardiac troponin (cTn) and copeptin are biomarkers used for early diagnosis of acute myocardial infarction (AMI) [10, 11] and may also serve as prognostic indicators following an acute coronary syndrome (ACS) [10, 12]. N-terminal pro-B-type natriuretic peptide (NT-proBNP) is a well-established diagnostic marker of heart failure [13] and an important predictor of mortality in patients with heart failure [13] or ACS [14]. Both NT-proBNP [15, 16] and copeptin [17, 18] have been found to predict mortality in critically ill patients admitted to a medical intensive care unit (ICU).
Although cardiac biomarkers may be useful for prognostication in OHCA-patients, available data regarding the predictive value of high-sensitivity cardiac troponin-T (hs-cTnT) [9, 19–21], copeptin [21–23] and NT-proBNP [19, 21, 24, 25] diverge. The inconsistency in previously reported results may be related to heterogeneity in patient population regarding cause of cardiac arrest and primary heart rhythm, timing of blood sampling and follow-up time. Previous studies have also mainly focused on resuscitated patients admitted to the ICU.
In our study, we included patients with OHCA of assumed cardiac origin. In a previous publication [21], we have assessed the prognostic utility of hs-cTnT, copeptin and NT-proBNP in OHCA patients with ventricular fibrillation (VF). We also compared ischemic- with non-ischemic cardiac arrest. The primary aim of the present study was to evaluate whether these three biomarkers may serve as independent prognostic indicators in OHCA patients with shockable- and non-shockable heart rhythm, adjusting for clinical variables related to cardiac arrest. As a second aim, this study was performed to improve our understanding of pathophysiology related to OHCA.
Methods
Study subjects and design
From February 2007 until November 2010 we collected blood samples from patients ≥18 years of age with OHCA of assumed cardiac origin, defined according to the Utstein definitions [26], in the southwestern part of Norway. All patients recruited in this study received out-of-hospital advanced cardiac life support according to the 2005 European Resuscitation Council guidelines with Norwegian modifications [27]. Blood sampling was performed by the EMS paramedics, 20 ml of blood was collected into ethylenediamine tetra acetic acid (EDTA) -tubes, using a venous cannula, during or immediately after termination of cardiopulmonary resuscitation (CPR). In patients with return of spontaneous circulation (ROSC) without a prehospital blood sample, blood was collected immediately after hospital admission.
We applied clinical information from hospital records and collected additional information from electrocardiograms, echocardiography and coronary angiography, to confirm an assumed cardiac cause of OHCA and to categorize patients [21]. OHCA-patients were divided into two groups according to first recorded heart rhythm, asystole or VF. VF-patients were further categorized according to an acute ischemic or non-ischemic mechanism for sudden cardiac arrest, and whether or not they had previously known heart disease.
Informed consent was collected retrospectively. All survivors gave written, informed consent. If the patient did not regain consciousness before death, the next-of-kin were asked for consent on the patient’s behalf. This study was approved by the Regional Board of Research Ethics and the Norwegian Health Authorities, conducted in accordance with the Helsinki Declaration of 1975, as revised in 1989, and registered in ClinicalTrials.gov, identifier: NCT02886273.
Laboratory methods
After collection, blood samples were centrifuged at 2500 rpm for 10 min. Within 24 h if stored at room-temperature, or within 48 h if stored at + 4 °C. EDTA-plasma was stored in aliquots at − 70 °C. Measurements of hs-cTnT, copeptin and NT-proBNP were performed at the Department of Multidisciplinary Laboratory Medicine and Medical Biochemistry, Akershus University Hospital, applying standardized methods, as previous reported [21]. Hs-cTnT (according to local stability analyses) and NT-proBNP [28] are found to be stable in EDTA-plasma for up to 3 days stored at room-temperature, and even longer when stored at + 4 °C. Ex vivo copeptin stability in EDTA-plasma is shown for at least 7 days at room temperature and 14 days at + 4 °C [29].
Statistical methods
Descriptive statistics are presented as medians with interquartile range (IQR; 25th to 75th percentile) for continuous data and as numbers and percentages for categorical data. Differences in baseline characteristics were assessed by the Kruskal-Wallis Test for continuous data and Fisher’s exact test for categorical data. Due to a right-skewed distribution, hs-cTnT, copeptin and NT-proBNP levels were logarithmically transformed to the base-e (loge) prior to analysis. Student’s independent two-sample t-test was used after loge-transformation to assess between-group differences, comparing biomarker-levels in non-survivors with survivors, between the asystole- and VF-group, and between patients who died on scene and those surviving until hospital admission. Spearman’s rank correlation coefficient was calculated to identify a possible relation between biomarker levels and time of blood sampling.
Patients were divided into quartiles (Q1–4) according to the hs-cTnT, copeptin and NT-proBNP concentrations. The Kaplan-Meier product limits were used for plotting the times to event and the log-rank test was used to test for the equality of the survival curves. Cox regression models, applying both quartiles and continuous loge-transformed values, were fitted for each of the biomarkers for the analysis of all-cause mortality within 30-days. We employed two different models for the multivariable analysis, by including age, loge-creatinine, VF as first recorded heart rhythm, and duration of resuscitation for Model 1, and adding gender, witnessed cardiac arrest and bystander-initiated CPR for Model 2. Hazard ratios (HR) with 95% confidence intervals were calculated for each of the higher quartiles as compared to quartile 1. For continuous loge-transformed values, we employed HR and 95% CI per standard deviation (SD) increase of the biomarkers. Subgroup analyses were performed in survivors at hospital admission and for VF-patients.
Statistical analysis was performed using the statistical package SPSS version 25 (IBM Corp. Armonk, NY). All tests were 2 -sided with a significance level of 5% without multiplicity adjustment.
Results
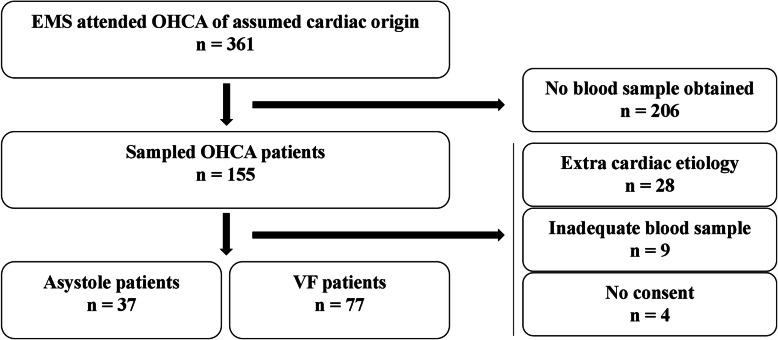
During the study period, 361 patients suffered cardiac arrest of assumed cardiac origin, and were eligible for inclusion in this study [21]. Out of these, 155 patients were sampled. Retrospectively, a total of 41 patients had to be excluded for different reasons (Fig. 1). The final population of 114 OHCA-patients was further divided into two groups according to the first recorded heart rhythm; 37 patients with asystole and 77 patients with VF. The group of VF-patients was further classified according to the presence of an acute ischemic event, by which 53 patients had signs of an AMI [21], and 10 of these had previously been diagnosed with heart disease. Twenty-one patients suffered sudden cardiac arrest without signs of an AMI [21], of whom 18 had evidence of prior heart disease, including coronary artery disease and/or chronic heart failure. Due to lack of clinical information, three patients could not be classified as having an ischemic- or non-ischemic cardiac arrest.
Fig. 1.
Flow chart displaying selection and classification of patients with out-of-hospital cardiac arrest recruited between February 2007 and November 2010
The baseline characteristics of the patients stratified according to 30-day mortality are presented in Table 1. Out of 114 patients, 70 (61.4%) died on scene or before hospital discharge. All patients (38.6%) surviving to hospital discharge were still alive at 30-day follow-up. Compared to 30-day survivors, non-survivors were older, had increased prevalence of heart failure and diabetes and worse kidney function. Asystole as first recorded heart rhythm was more frequent in non-survivors (53%), they also had worse cardiac arrest conditions with a significant lower proportion of witnessed cardiac arrest and bystander-initiated CPR, and longer duration of resuscitation.
Table 1.
Baseline characteristics and laboratory values of patients suffering out-of-hospital cardiac arrest
| Total population (n = 114) | Survivors (n = 44) | Non-survivors (n = 70) | P-value | |
|---|---|---|---|---|
| Age, y | 67 (56–78) | 61 (51–68) | 72 (62–83) | < 0.001 |
| Male gender | 95 (83) | 36 (82) | 59 (84) | 0.80 |
| Previous history | ||||
| Angina pectoris | 17 (18) | 6 (14) | 11 (22) | 0.42 |
| Myocardial infarction | 33 (31) | 10 (23) | 23 (36) | 0.20 |
| Previous PCI | 12 (11) | 5 (11) | 7 (11) | 1.00 |
| Previous CABG | 12 (11) | 2 (5) | 10 (16) | 0.12 |
| Heart failure | 28 (26) | 7 (16) | 21 (34) | 0.046 |
| Hypertension | 53 (52) | 19 (43) | 34 (58) | 0.17 |
| Diabetes mellitus | 17 (15) | 2 (5) | 15 (25) | 0.007 |
| Hypercholesterolemia | 44 (42) | 22 (51) | 22 (36) | 0.16 |
| Smoking | 0.81 | |||
| Current smoker | 27 (32) | 13 (33) | 14 (31) | |
| Ex-smoker | 40 (47) | 20 (50) | 20 (44) | |
| Cardiac arrest conditions | ||||
| Witnessed cardiac arrest | 87 (77) | 39 (89) | 48 (70) | 0.022 |
| Bystander-initiated CPR | 93 (82) | 40 (91) | 53 (76) | 0.049 |
| Duration of resuscitation, min | 23 (10–38) | 9 (5–15) | 32.5 (23–45) | < 0.001 |
| Initial rhythm | < 0.001 | |||
| VF | 77 (68) | 44 (100) | 33 (47) | |
| Asystole | 37 (33) | 0 (0) | 37 (53) | |
| Baseline blood samples | ||||
| Creatinine (μmol/L) | 102 (87–122) | 93 (82–116) | 110 (92–125) | 0.013 |
| Total cholesterol (mmol/L) | 4.2 (3.6–5.3) | 4.9 (4.1–6.5) | 3.9 (3.4–5.0) | 0.002 |
| CRP (mg/L) | 2.5 (1.1–9.9) | 2.1 (1.2–4.6) | 3.9 (1.0–18.0) | 0.15 |
| Glucose (mmol/L) | 12.6 (8.0–16.9) | 12.6 (8.8–14.7) | 12.6 (6.6–17.8) | 0.94 |
| Copeptin (pmol/L)a | 436 (216–825) | 388 (195–825) | 445 (244–879) | 0.39 |
| hs-cTnT (ng/L)b | 71 (26–231) | 100 (21–289) | 66 (26–207) | 0.49 |
| NT-proBNP (pmol/L)c | 61 (25–234) | 30 (12–98) | 105 (35–495) | < 0.001 |
Data are presented as median (interquartile range) or numbers (%)
a n = 110 (96%), b n = 111 (97%), c n = 112 (98%)
Abbreviations: CPR cardiopulmonary resuscitation, PCI percutaneous coronary intervention, CABG coronary artery bypass graft, CRP C-reactive protein, hs-cTnT high-sensitivity cardiac troponin T, NT-proBNP N-terminal pro-B-type natriuretic peptide
Blood samples were drawn from 75 patients during resuscitation and from 39 patients at hospital admission, with a median time from cardiac arrest until blood sampling of 31 and 73.5 min, respectively. We found no correlation between biomarker levels and time of blood sampling, except for hs-cTnT (r = 0.31, p = 0.001). Median hs-cTnT concentration was 71 (IQR; 26–231) ng/L, ranging from 6.0 to 8333 ng/L. Hs-cTnT levels did not differ significantly between survivors and non-survivors [median 100 IQR: (21–289) ng/L vs. 66 (26–207) ng/L, p = 0.49]. Kaplan-Meier analysis revealed no significant association between quartiles of hs-cTnT and all-cause mortality at 30-days follow-up (log-rank p = 0.57). In multivariable analysis, there were no significant associations between hs-cTnT and 30-days all-cause mortality, neither in the total population, nor for survivors at hospital admission or in VF-patients (Table 2).
Table 2.
Univariate- and multivariable Cox regression models applying continuous loge-transformed values of hs-cTnT, copeptin and NT-proBNP
| Univariate | Multivariable | |||||
|---|---|---|---|---|---|---|
| Model 1 | Model 2 | |||||
| HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Total population | ||||||
| loge Hs-cTnT | 0.99 (0.78–1.29) | 0.99 | 0.87 (0.66–1.15) | 0.34 | 0.90 (0.68–1.19) | 0.46 |
| loge Copeptin | 1.08 (0.84–1.39) | 0.54 | 0.88 (0.65–1.20) | 0.42 | 0.92 (0.66–1.28) | 0.62 |
| loge NT-proBNP | 1.86 (1.44–2.41) | < 0.001 | 0.96 (0.64–1.43) | 0.84 | 0.88 (0.57–1.36) | 0.57 |
| Patients surviving to hospital admission | ||||||
| loge Hs-cTnT | 0.96 (0.67–1.38) | 0.83 | 0.85 (0.58–1.25) | 0.41 | 0.87 (0.61–1.26) | 0.48 |
| loge Copeptin | 1.40 (0.95–2.06) | 0.093 | 1.20 (0.75–1.93) | 0.45 | 1.07 (0.62–1.83) | 0.81 |
| loge NT-proBNP | 1.86 (1.24–2.78) | 0.003 | 0.93 (0.50–1.73) | 0.83 | 0.79 (0.41–1.49) | 0.46 |
| VF-patients | ||||||
| loge Hs-cTnT | 0.85 (0.59–1.24) | 0.41 | 0.78 (0.52–1.17) | 0.24 | 0.85 (0.56–1.28) | 0.43 |
| loge Copeptin | 1.15 (0.81–1.63) | 0.44 | 1.01 (0.67–1.51) | 0.98 | 0.95 (0.60–1.52) | 0.85 |
| loge NT-proBNP | 1.67 (1.07–2.60) | 0.023 | 0.84 (0.43–1.64) | 0.61 | 0.51 (0.22–1.19) | 0.12 |
Abbreviations: loge hs-cTnT loge-transformed value of hs-cTnT, loge Copeptin loge-transformed value of copeptin, loge NT-proBNP loge-transformed value of N-terminal pro B-type natriuretic peptide, VF ventricular fibrillation, HR hazard ratio, 95% CI 95% confidence interval. Model 1; multivariable model adjusting for age, loge-creatinine, first recorded heart rhythm and duration of resuscitation. Model 2; multivariable model adjusting for age, loge-creatinine, first recorded heart rhythm, duration of resuscitation, witnessed cardiac arrest and bystander-initiated cardiopulmonary resuscitation
Copeptin levels were elevated in all patients [436 (216–825) pmol/L], and did not differ between the two outcomes, p = 0.38. In survival analysis, copeptin quartiles did not separate survivors and non-survivors after 30 days, log-rank test p = 0.67. There were no statistically significant associations between copeptin levels and all-cause mortality at 30-days follow-up, neither in univariate analysis, nor in multivariable models (Table 2).
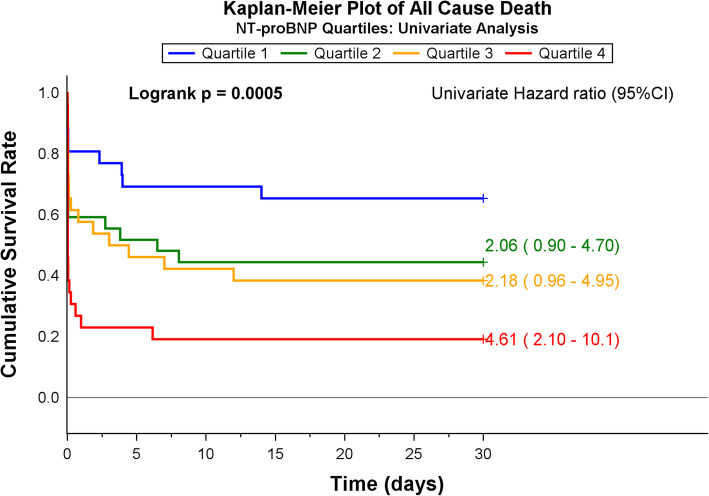
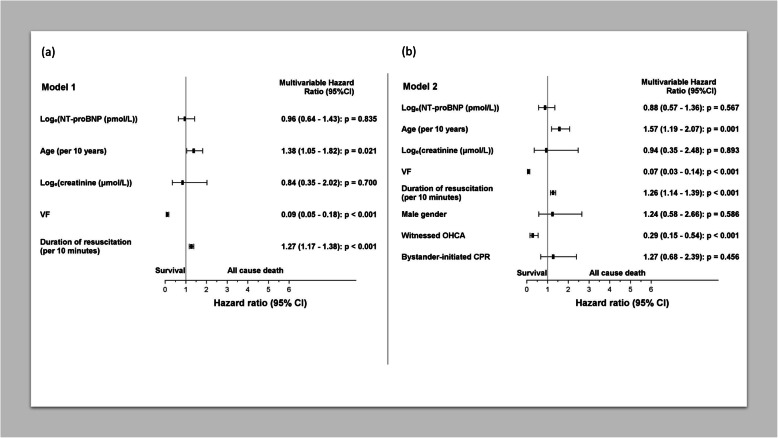
NT-proBNP was the only biomarker demonstrating a significant difference between non-survivors and survivors [105 (35–495) pmol/L vs. 30 (12–98) pmol/L, p < 0.001]. Patients were grouped into quartiles according to NT-proBNP levels; Quartile 1 (Q1): < 24 pmol/L, Q2: 25–60 pmol/L, Q3: 62–218 pmol/L, and Q4: > 250 pmol/L. The median NT-proBNP level in Q4 was 720 (IQR; 371–2506). Q4-patients were significantly older with increased prevalence of heart failure and reduced kidney function, and there was a larger proportion with asystole as first recorded heart rhythm, Supplemental Table 1. Kaplan-Meier analysis revealed that the upper NT-proBNP quartile was significantly associated with all-cause mortality at 30-days (Fig. 2). Assessed as a continuous variable, this gave a univariate HR of 1.86, p < 0.001 for the total population, with similar findings in survivors at hospital admission [HR 1.86, p = 0.003] and in VF-patients [HR 1.67, p = 0.023]. Adding age, loge-creatinine value, VF as primary heart rhythm and duration of resuscitation in a multivariable Cox regression model (Model 1), resulted in loss of the significant association between NT-proBNP and outcome, both in the total population [HR 0.96, p = 0.84] (Fig. 3) and in the former two subgroups (Table 2). Similar results were obtained when adding gender, witnessed cardiac arrest and bystander-initiated CPR in multivariable model 2 (Table 2).
Fig. 2.
Survival curves up to 30-days in OHCA-patients stratified by NT-proBNP
quartiles
Fig. 3.
Adjusted hazard ratio and 95% confidence intervals for 30-day all-cause mortality according to selected risk factors from two different multivariable models. a Model 1, b Model 2. Hazard ratio (squares), 95% confidence interval (lines). Abbreviations: OHCA, out-of-hospital cardiac arrest; CPR, cardiopulmonary resuscitation; 95% CI, 95% confidence interval
Among the clinical variables, increasing age was associated with death in both models (Model 2; p = 0.001). Witnessed cardiac arrest, VF as first recorded heart rhythm and duration of resuscitation were also significantly associated with outcome (Fig. 3).
NT-proBNP was significantly higher in patients who died on-scene (non-ROSC patients) compared to survivors at hospital admission [106.2 (46.5–706.2) pmol/L vs. 46.8 (17.2–164.4), p = 0.003], but there was no significant difference in NT-proBNP levels when comparing on-scene non-ROSC patients with those who died in hospital (p = 0.17). Hs-cTnT and copeptin did not differ significantly between on-scene non-ROSC patients and survivors at hospital admission.
At 30-day follow-up, all patients in the asystole-group had died, as compared to 43% in the VF-group. Baseline characteristics of patients with asystole versus VF as first recorded heart rhythm are presented in Table 3. NT-proBNP was the only biomarker that differed between these two groups, with a significantly higher median value of 250 (42–1305) pmol/L in the asystole group compared to 51 (18–111) pmol/L in the VF-group, p < 0.001. This difference is driven by VF-patients presenting with ischemic cardiac arrest (68.8% of the VF-population), p < 0.001, and was not evident when comparing non-ischemic cardiac patients with the asystole group, p = 0.74. Hs-cTnT and copeptin were elevated in both asystole- and VF patients, but there was no significant difference between the two groups. These findings are illustrated by applying loge-transformed values of the biomarkers as shown in Suppl. Fig. 1.
Table 3.
Baseline characteristics and laboratory values of patients suffering out-of-hospital cardiac arrest, arranged according to first recorded heart rhythm
| Asystole-patients (n = 37) | VF-patients (n = 77) | P-value | |
|---|---|---|---|
| Age, y | 75 (66–86) | 63 (54–75) | < 0.001 |
| Male sex | 26 (70) | 69 (90) | 0.015 |
| Death at 30 days | 37 (100) | 33 (43) | < 0.001 |
| Died on scene | 26 (70) | 9 (12) | |
| Died in hospital | 11 (30) | 24 (31) | |
| Cardiac arrest conditions | |||
| Witnessed cardiac arrest | 26 (70) | 61 (80) | 0.24 |
| Bystander-initiated CPR | 26 (70) | 67 (87) | 0.040 |
| Duration of resuscitation, min | 28.5 (20–35) | 15 (6–43) | 0.082 |
| Previous history | |||
| Angina pectoris | 9 (32) | 8 (12) | 0.037 |
| Myocardial infarction | 11 (32) | 22 (30) | 0.82 |
| Previous PCI | 4 (12) | 8 (11) | 1.00 |
| Previous CABG | 5 (15) | 7 (10) | 0.51 |
| Heart failure | 11 (33) | 17 (23) | 0.34 |
| Hypertension | 21 (64) | 32 (46) | 0.097 |
| Diabetes mellitus | 7 (21) | 10 (14) | 0.40 |
| Hypercholesterolemia | 12 (36) | 32 (44) | 0.53 |
| Smoking | 0.56 | ||
| Current smoker | 10 (39) | 17 (29) | |
| Ex-smoker | 10 (39) | 30 (51) | |
| Baseline blood samples | |||
| Creatinine (μmol/L) | 101 (92–130) | 102 (87–121) | 0.42 |
| Total cholesterol (mmol/L) | 4.1 (3.5–5.0) | 4.3 (3.7–5.4) | 0.16 |
| CRP (mg/L) | 12.0 (2.0–50.0) | 1.9 (1.1–4.1) | < 0.001 |
| Glucose (mmol/L) | 10.7 (3.7–15.7) | 13.7 (9.6–17.2) | 0.034 |
| Copeptin (pmol/L)a | 402 (244–939) | 453 (210–765) | 0.82 |
| hs-cTnT (ng/L)b | 67 (28–226) | 73 (26–231) | 0.77 |
| NT-proBNP (pmol/L)c | 250 (42–1305) | 51 (18–111) | < 0.001 |
Data are presented as median (interquartile range) or numbers (%)
a n = 110 (96%), b n = 111 (97%), c n = 112 (98%)
Abbreviations: VF ventricular fibrillation, OHCA out-of-hospital cardiac arrest, CPR cardiopulmonary resuscitation, PCI percutaneous coronary intervention, CABG coronary artery bypass graft, CRP C-reactive protein, hs-cTnT high-sensitivity cardiac troponin T
NT-proBNP N-terminal pro-B-type natriuretic peptide
Discussion
We evaluated the prognostic utility of early-on levels of hs-cTnT, copeptin and NT-proBNP for all-cause mortality at 30-days follow-up in clinically characterized patients with OHCA of assumed cardiac origin, presenting with asystole or VF.
NT-proBNP was found to be significantly related to death in univariate analysis, whereas hs-cTnT and copeptin were not associated with outcome. Adjusting for demographic- and clinical variables, the prognostic value of NT-proBNP was attenuated and no longer statistically significant.
These findings are consistent with two previous studies [24, 25], reporting a similar association between admission levels of NT-proBNP and outcome in resuscitated OHCA patients. There are several possible mechanisms for increased NT-proBNP/BNP secretion during cardiac arrest and CPR, including hypoxemia [30], ischemia [31], ischemia-reperfusion induced inflammation [32], therapeutic interventions with administration of fluids [33] and vasopressors [34], and mechanical ventilation with supplementary oxygen administration [35]. Myhre et al. 2016 [24] demonstrated an increase in NT-proBNP levels from admission up to 96 h after hospitalization in OHCA-VT/VF patients. Longer time to ROSC and higher admission levels of hs-cTnT were found to be associated with high NT-proBNP concentrations after 24 h in multivariable analysis. Furthermore, 24-h NT-proBNP levels provided additional prognostic information for the prediction of 1-year mortality. These associations may reflect myocardial changes brought about by the cardiac arrest, whereas earlier on-site levels of NT-proBNP in our study most likely will reflect the pre-cardiac arrest condition, as patients in Q4 as compared to lower quartiles were more prone to heart failure, were older and presented with worse renal function. Accordingly, NT-proBNP levels did not differ between on-scene non-ROSC patients and those who died in hospital. The prognostic value of comorbidity has previously been claimed to be of less importance in OHCA patients [36, 37], and this may explain the lack of prognostic utility for early-on NT-proBNP in our study. However, early measurements of NT-proBNP may help to identify OHCA-patients with clinically silent heart insufficiency or coronary artery disease, in need of special medical attention.
Demographic- and clinical factors, such as age, witnessed cardiac arrest, bystander-initiated CPR, primary heart rhythm and time to ROSC, are previously found to be prognostically important in cardiac arrest [3, 38–42]. Furthermore, plasma concentrations of NT-proBNP are known to be associated with age and kidney function [13]. Multivariable analysis, adjusting for these established risk-variables, cancelled the significant univariate association between early-on levels of NT-proBNP and outcome. However, in accordance with previous observations [3, 39–42], we also noted that increasing age and longer duration of CPR was significantly associated with increased risk of death, while witnessed cardiac arrest and VF as primary heart rhythm was associated with a more favourable outcome (Fig. 3).
In contrast to previous reports [3, 39], bystander-initiated CPR was not found to be an independent outcome-predictor in our study, probably cancelled by a larger proportion of VF-patients and a short EMS response-time between 8 to 15 min in our recruitment area.
Furthermore, we found that NT-proBNP differentiated between ischemic VF patients and those presenting with asystole. This could be explained by a difference in baseline risk variables, as previously reported [21]. Patients in the asystole group were older and a higher proportion suffered from prior cardiac morbidity, including established coronary artery disease and heart failure. However, no difference in NT-proBNP was observed when comparing non-ischemic VF patients with those presenting with asystole. These groups had similar baseline characteristics and differed mainly by the presenting arrhythmia, which may relate to prognosis. Accordingly, all patients in the asystole group died as compared to 50% in the non-ischemic VF group.
The two other biomarkers, hs-cTnT and copeptin, were not related to outcome, neither in univariate, nor in multivariable analysis. As previously demonstrated [9], we found that essentially all resuscitated OHCA-patients had elevated levels of hs-cTnT. Gilje et al. [20] found that hs-cTnT peaked 24 h after admission following OHCA, but only hs-cTnT at 48 and 72 h, respectively, was independently associated with all-cause mortality. In our study, we analysed only one blood sample very close to the cardiac arrest, which may not reflect the peak values of hs-cTnT, and the prognostic utility may have been missed. This assumption is supported by the FINNRESUSCI substudy by Røsjø et.al [9], where admission levels of hs-cTnT failed to differ between hospital non-survivors and survivors and did not yield independent prognostic information at 1-year follow up in OHCA-VF/VT patients.
Copeptin levels were markedly increased in all patients in our study and did not differ between 30-days non-survivors and survivors. Admission levels of copeptin have previously been demonstrated to independently predict organ dysfunction and death in the ICU following OHCA [22]. As a predictor of long-term mortality, copeptin levels at day 3 were shown to perform better than copeptin measured at ICU-admission [23]. In our early-on sample, elevated copeptin may largely reflect the stress response during cardiac arrest, rather than outcome related hemodynamic instability following ROSC. The prognostic utility of copeptin claimed in the FINNRESUSCI population [22], is based on samples harvested up to 6 h after admission and did not include on-scene non-ROSC patients.
Strengths
Blood was collected very early after OHCA and includes non-admitted patients without ROSC, a patient category usually missed out in previous studies. Also, the initial cardiac rhythm was recorded in all patients. Pre-hospital data were collected in accordance with the Utstein guidelines [26], and advanced cardiac life support was performed by the EMS paramedics according to current guidelines [27].
Limitations
The small study population is one of the limitations. Further studies with larger patient populations should be performed to assess the prognostic value of these three biomarkers. Inclusion of patients was restricted to the largest ambulance centres in the area located closest to the hospital and to the medical support helicopter to ensure timely delivery of blood samples, limiting the potential recruitment area. Furthermore, patient recruitment could only be performed when there was enough EMS crew present at the OHCA-scene. Samples were obtained during resuscitation and not after death was declared. Unfortunately, there was a selection bias due to unbalanced blood sampling in the ROSC and non-ROSC group of patients. A few patients lacked detailed information regarding the OHCA. Our study is limited to 30-days observations of outcome.
Conclusions
Early-on levels of hs-cTnT, copeptin and NT-proBNP did not provide prognostic information following OHCA. Early measurement of NT-proBNP seems to reflect the pre-cardiac arrest condition, and may be useful to identify OHCA-patients with heart insufficiency and coronary artery disease, also including subjects with clinically silent conditions.
Supplementary information
Additional file 2: Table S1. Baseline characteristics and laboratory values of patients suffering out-of-hospital cardiac arrest, arranged according to Quartiles of NT-proBNP. Data are presented as median (interquartile range) or numbers (%). a n = 110 (96%), b n = 111 (97%), c n = 112 (98%). Abbreviations: OHCA, out-of-hospital cardiac arrest; CPR, cardiopulmonary resuscitation; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; CRP, C- reactive protein; hs-cTnT, high-sensitivity cardiac troponin T; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
Acknowledgements
Not applicable.
Abbreviations
- ACS
Acute coronary syndrome
- AMI
Acute myocardial infarction
- CI
Confidence interval
- CPR
Cardiopulmonary resuscitation
- cTn
Cardiac troponin
- EDTA
Ethylenediamine tetra acetic acid
- EMS
Emergency medical services
- HR
Hazard ratio
- Hs-cTnT
High-sensitivity cardiac troponin-T
- ICU
Intensive care unit
- IQR
Interquartile range
- NT-proBNP
N-terminal pro-B-type natriuretic peptide
- OHCA
Out-of-hospital cardiac arrest
- ROSC
Return of spontaneous circulation
- VF
Ventricular fibrillation
Authors’ contributions
R.A: investigation, formal analysis, writing – original draft. T.O: resources, writing – review and editing. H.R: resources, writing – review and editing. He.S: resources, writing – review and editing. T.L: investigation, resources, writing – review and editing. HA: methodology, investigation, writing – review and editing. Ha.S: formal analysis, writing – review and editing. D.N: conceptualization, methodology, writing – review and editing. All authors read and approved the final manuscript.
Funding
This study was supported with grants from the Regional Health Authorities in Western Norway and the Laerdal Foundation for Acute Medicine. The funding body did not participate in the design of the study, collection, analysis, and interpretation of data or in writing the manuscript.
Availability of data and materials
Local database. The datasets analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Informed consent was collected retrospectively. All survivors gave written, informed consent. If the patient did not regain consciousness before death, the next-of-kin were asked for consent on the patient’s behalf. This study was approved by the Regional Board of Research Ethics and the Norwegian Health Authorities, conducted in accordance with the Helsinki Declaration of 1975, as revised in 1989.
Consent for publication
Not applicable.
Competing interests
T.O. and H.R. are partners in a patent application filed by the University of Oslo regarding the use of secretoneurin as a biomarker in cardiovascular disease and in patients with critical illness. H. R and T.O. have financial interests in CardiNor AS, which holds the license to commercialize secretoneurin. T.O. and H.R. have also received personal payments from CardiNor AS. H. R have also received personal fees from Novartis and Thermo Fisher BRAHMS. T. O has received honoraria and research support from Roche Diagnostics via Akershus University Hospital.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12872-020-01630-x.
References
- 1.Gräsner JT, Lefering R, Koster RW, et al. EuReCa ONE – 27 nations, ONE Europe, ONE registry; a prospective one-month analysis of out-of-hospital cardiac arrest outcomes in 27 countries in Europe. Resuscitation. 2016;105:188–195. doi: 10.1016/j.resuscitation.2016.06.004. [DOI] [PubMed] [Google Scholar]
- 2.Berdowski J, Berg RA, Tijssen JG, et al. Global incidences of out-of-hospital cardiac arrest and survival rates: systematic review of 67 prospective studies. Resuscitation. 2010;81:1479–1487. doi: 10.1016/j.resuscitation.2010.08.006. [DOI] [PubMed] [Google Scholar]
- 3.Sasson C, Rogers MAM, Dahl J, et al. Predictors of survival from out-of-hospital cardiac arrest; a systematic review and meta-analysis. Circ Cardiovasc Qual Outcomes. 2010;3:63–81. doi: 10.1161/CIRCOUTCOMES.109.889576. [DOI] [PubMed] [Google Scholar]
- 4.Le Gall JR, Lemeshow S, Saulnier F. A new simplified acute physiology score (SAPSII) based on a European/north American multicentre study. JAMA. 1993;270:2957–2963. doi: 10.1001/jama.1993.03510240069035. [DOI] [PubMed] [Google Scholar]
- 5.Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related organ failure assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European society of intensive care medicine. Intensive Care Med. 1996;22:707–710. doi: 10.1007/BF01709751. [DOI] [PubMed] [Google Scholar]
- 6.Mozaffarian D, Benjamin EJ, Go AS, et al. American heart Assosciation statistics committee and stroke statistics subcommittee. Heart disease and stroke statistics-2015 update: a report from the American Heart Association. Circulation. 2015;131:e29–322. doi: 10.1161/CIR.0000000000000152. [DOI] [PubMed] [Google Scholar]
- 7.Zipes DP, Wellens HJ. Sudden cardiac death. Circulation. 1998;98:2334–2351. doi: 10.1161/01.CIR.98.21.2334. [DOI] [PubMed] [Google Scholar]
- 8.Nolan JP, Neumar RW, Adrie C, et al. Post-cardiac arrest syndrome: epidemiology, pathophysiology, treatment and prognostication. Resuscitation. 2008;79:350–379. doi: 10.1016/j.resuscitation.2008.09.017. [DOI] [PubMed] [Google Scholar]
- 9.Røsjø H, Vaahersalo J, Hagve T-A, et al. Prognostic value of high-sensitivity troponin T levels in patients with ventricular arrhythmias and out-of-hospital cardiac arrest: data from the prospective FINNRESUSCI study. Crit Care. 2014;18:605. doi: 10.1186/s13054-014-0605-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roffi M, Patrono C, Collet JP, et al. 2015 ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2016;37(3):267–315. doi: 10.1093/eurheartj/ehv320. [DOI] [PubMed] [Google Scholar]
- 11.Slagman A, Searle J, Müller C, et al. Temporal release pattern of Copeptin and troponin T in patients with suspected acute coronary syndrome and spontaneous acute myocardial infarction. Clin Chem. 2015;61:1273–1282. doi: 10.1373/clinchem.2015.240580. [DOI] [PubMed] [Google Scholar]
- 12.Narayan H, Dhillon OS, Quinn PA, et al. C-terminal provasopressin (copeptin) as a prognostic marker after acute non-ST elevation myocardial infarction: Leicester acute myocardial infarction peptide II (LAMP II) study. Clin Sci. 2011;121:79–89. doi: 10.1042/CS20100564. [DOI] [PubMed] [Google Scholar]
- 13.Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2016;37:2129–2200. doi: 10.1093/eurheartj/ehw128. [DOI] [PubMed] [Google Scholar]
- 14.Galvani M, Ottani F, Oltrona L, et al. N-terminal pro-brain natriuretic peptide on admission has prognostic value across the whole spectrum of acute coronary syndrome. Circulation. 2004;110:128–134. doi: 10.1161/01.CIR.0000134480.06723.D8. [DOI] [PubMed] [Google Scholar]
- 15.Varpula M, Pulkki K, Karlsson S, et al. Predictive value of N-terminal pro-brain natriuretic peptide in severe sepsis and septic shock. Crit Care Med. 2007;35:1277–1283. doi: 10.1097/01.CCM.0000261893.72811.0F. [DOI] [PubMed] [Google Scholar]
- 16.Okkonen M, Varpula M, Linko R, et al. N-terminal-pro-BNP in critically ill patients with acute respiratory failure: a prospective cohort study. Acta Anaesthesiol Scand. 2011;55:749–757. doi: 10.1111/j.1399-6576.2011.02439.x. [DOI] [PubMed] [Google Scholar]
- 17.Koch A, Yagmur E, Hoss A, et al. Clinical relevance of copeptin plasma levels as a biomarker of disease severity and mortality in critically ill patients. J Clin Lab Anal. 2018;32:e22614. doi: 10.1002/jcla.22614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Krychtiuk KA, Honeder MC, Lenz M, et al. Copeptin predicts mortality in critically ill patients. PLoS One. 2017;12:e0170436. doi: 10.1371/journal.pone.0170436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Isenschmid C, Kalt J, Gamp M, et al. Routine blood markers from different biological pathways improve early risk stratification in cardiac arrest patients: results from the prospective, observational COMMUNICATE study. Resuscitation. 2018;130:138–145. doi: 10.1016/j.resuscitation.2018.07.021. [DOI] [PubMed] [Google Scholar]
- 20.Gilje P, Kouls S, Thomsen JH, et al. High-sensitivity troponin T as a prognostic marker after out-of-hospital cardiac arrest – a targeted temperature management (TTM) trial substudy. Resuscitation. 2016;107:156–161. doi: 10.1016/j.resuscitation.2016.06.024. [DOI] [PubMed] [Google Scholar]
- 21.Aarsetøy R, Aarsetøy H, Hagve TA, et al. Initial phase NT-proBNP, but not copeptin and high-sensitivity cardiac troponin T yielded diagnostic and prognostic information in addition to clinical assessment of out-of-hospital cardiac arrest patients with documented ventricular fibrillation. Front Cardiovasc Med. 2018;5:44. doi: 10.3389/fcvm.2018.00044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ristagno G, Latini R, Plebani M, et.al. Copeptin levels are associated with organ dysfunction and death in the intensive care unit after out-of-hospital cardiac arrest. Crit Care 2015;19:132. [DOI] [PMC free article] [PubMed]
- 23.Geri G, Dumas F, Chenvier-Gobeaux C, et al. Is copeptin level associated with 1-year mortality after out-of-hospital cardiac arrest? Insights from the Paris registry. Crit Care Med. 2015;43:422–429. doi: 10.1097/CCM.0000000000000716. [DOI] [PubMed] [Google Scholar]
- 24.Myhre PL, Tiainen M, Pettilä V, et al. NT-proBNP in patients with out-of-hospital cardiac arrest: results from the FINNRESUSCI study. Resuscitation. 2016;104:12–18. doi: 10.1016/j.resuscitation.2016.04.007. [DOI] [PubMed] [Google Scholar]
- 25.Smit B, Spelstra-de Man AM, Girbes AR, et al. NT-proBNP in cardiopulmonary resuscitated patients treated with mild therapeutic hypothermia is not independently associated with mortality: a retrospective observational study. BMC Anesthesiol. 2015;15:48. doi: 10.1186/s12871-015-0023-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jacobs I, Nadkarni V, Bahr J, et al. Cardiac arrest and cardiopulmonary resuscitation outcome reports: update and simplification of the Utstein templates for resuscitation registries: a statement for healthcare professionals from a task force of the international liaison committee on resuscitation. Circulation. 2004;110:3385–3397. doi: 10.1161/01.CIR.0000147236.85306.15. [DOI] [PubMed] [Google Scholar]
- 27.Lexow K, Sunde K. Why Norwegian 2005 guidelines differs slightly from the ERC guidelines. Resuscitation. 2007;72:490–492. doi: 10.1016/j.resuscitation.2006.07.018. [DOI] [PubMed] [Google Scholar]
- 28.Yeo K-T, Wu AHB, Apple FS, et al. Multicenter evaluation of the Roche NT-proBNP assay and comparison to the biosite triage BNP assay. Clin Chim Acta. 2003;338:107–115. doi: 10.1016/j.cccn.2003.08.016. [DOI] [PubMed] [Google Scholar]
- 29.Morgenthaler NG, Struck J, Alonso C, Bergmann A. Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin Chem. 2006;52:112–119. doi: 10.1373/clinchem.2005.060038. [DOI] [PubMed] [Google Scholar]
- 30.Casals G, Ros J, Sionis A, et al. Hypoxia induces B-type natriuretic peptide release in cell lines derived from human cardiomyocytes. Am J Physiol Heart Circ Physiol. 2009;297:550–555. doi: 10.1152/ajpheart.00250.2009. [DOI] [PubMed] [Google Scholar]
- 31.Sabatine MS, Morrow DA, de Lemos JA, et al. Acute changes in circulating natriuretic peptide levels in relation to myocardial ischemia. J Am Coll Cardiol. 2004;44:1988–1995. doi: 10.1016/j.jacc.2004.07.057. [DOI] [PubMed] [Google Scholar]
- 32.Ogawa T, de Bold AJ. Brain natriuretic peptide production and secretion in inflammation. J Transp Secur. 2012;962647. [DOI] [PMC free article] [PubMed]
- 33.Heringlake M, Heide C, Bahlmann L, et al. Effects of tilting and volume loading on plasma levels and urinary excretion of relaxin, NT-proANP and NT-proBNP in male volunteers. J Appl Phsiol. 2004;97:173–179. doi: 10.1152/japplphysiol.01196.2003. [DOI] [PubMed] [Google Scholar]
- 34.Valette X, Lemoine S, Allouche S, et al. Effect of lipopolysaccharide, cytokines and cathecholamines on brain natriuretic peptide release from human myocardium. Acta Anaesthesiol Scand. 2012;56:860–865. doi: 10.1111/j.1399-6576.2012.02683.x. [DOI] [PubMed] [Google Scholar]
- 35.Yildiz S, Uzun G, Uz O, et al. N-terminal pro-B-type natriuretic peptide levels increase after hyperbaric oxygen therapy in diabetic patients. Clin Invest Med. 2008;31:E231–E235. doi: 10.25011/cim.v31i5.4868. [DOI] [PubMed] [Google Scholar]
- 36.Winther-Jensen M, Kjaergaard J, Nielsen N, et al. Comorbidity burden is not associated with higher mortality after out-of-hospital cardiac arrest. Scand Cardiovasc J. 2016;50:305–310. doi: 10.1080/14017431.2016.1210212. [DOI] [PubMed] [Google Scholar]
- 37.Hirlekar G, Jonsson M, Karlsson T, et al. Comorbidity and survival in out-of-hospital cardiac arrest. Resuscitation. 2018;133:118–123. doi: 10.1016/j.resuscitation.2018.10.006. [DOI] [PubMed] [Google Scholar]
- 38.Sulzgruber P, Sterz F, Poppe M, et al. Age-spesific prognostication after out-of-hospital cardiac arrest – the ethical dilemma between ‘life-sustaining treatment’ and ‘the right to die’ in the elderly. Eur Heart J Acute Cardiovasc Care. 2017;6:112–120. doi: 10.1177/2048872616672076. [DOI] [PubMed] [Google Scholar]
- 39.Herlitz J, Engdahl J, Svensson L, et al. Factors associated with an increased chance of survival among patients suffering from an out-of-hospital cardiac arrest in a national perspective in Sweden. Am Heart J. 2005;149:61–66. doi: 10.1016/j.ahj.2004.07.014. [DOI] [PubMed] [Google Scholar]
- 40.Lindner TW, Langørgen J, Sunde K, et al. Factors predicting the use of therapeutic hypothermia and survival in unconscious out-of-hospital cardiac arrest patients admitted to the ICU. Crit Care. 2013;17:R147. doi: 10.1186/cc12826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wilbrandt I, Norsted K, Schmidt H, et al. Predictors of outcome among cardiac arrest patients: the importance of initial cardiac arrest rhythm versus time to return of spontaneous circulation, a retrospective cohort study. BMC Emerg Med. 2015;15:3. doi: 10.1186/s12873-015-0028-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Reynolds JC, Frisch A, Rittenberger JC, et al. Duration of resuscitation efforts and functional outcome after out-of-hospital cardiac arrest: when should we change to novel therapies? Circulation. 2013;128:2488–2494. doi: 10.1161/CIRCULATIONAHA.113.002408. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 2: Table S1. Baseline characteristics and laboratory values of patients suffering out-of-hospital cardiac arrest, arranged according to Quartiles of NT-proBNP. Data are presented as median (interquartile range) or numbers (%). a n = 110 (96%), b n = 111 (97%), c n = 112 (98%). Abbreviations: OHCA, out-of-hospital cardiac arrest; CPR, cardiopulmonary resuscitation; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; CRP, C- reactive protein; hs-cTnT, high-sensitivity cardiac troponin T; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
Data Availability Statement
Local database. The datasets analysed during the current study are available from the corresponding author on reasonable request.