Abstract
Objective:
As a fractionated course of radiotherapy proceeds tumour shrinkage leads to resolution of hypoxia and the initiation of accelerated proliferation of radioresistant cancer cells with better repair capacity. We hypothesise that, in tumours with significant hypoxia, improved tumour control could be achieved with biphasic fractionation schedules that either use acceleration after 3–4 weeks of conventional radiotherapy or deliver a higher proportional dose towards the end of a course of treatment. We conducted a modelling study based on the concept of biological effective dose (BED) comparing such novel regimens with conventional fractionation.
Methods:
The comparator conventional fractionation schedule 70 Gy in 35 fractions delivered over 7 weeks was tested against the following novel regimens, both of which were designed to be isoeffective in terms of late normal tissue toxicity.
40 Gy in 20 fractions over 4 weeks followed by 22.32 Gy in 6 consecutive daily fractions (delayed acceleration)
30.4 Gy in 27 fractions over 4 weeks followed by 40 Gy in 15 fractions over 3 weeks (temporal dose redistribution)
The delayed acceleration regimen is exactly identical to that of the comparator schedule over the first 28 days and the BED gains with the novel schedule are achieved during the second phase of treatment when reoxygenation is complete. For the temporal redistribution regimen, it was assumed that the reoxygenation fraction progressively increases during the first 4 weeks of treatment and an iterative approach was used to calculate the final tumour BED for varying hypoxic fractions.
Results:
Novel fractionation with delayed acceleration or temporal fractionation results in tumour BED gains equivalent to 3.5–8 Gy when delivered in 2 Gy fractions.
Conclusion:
In hypoxic tumours, novel fractionation strategies result in significantly higher tumour BED in comparison to conventional fractionation.
Advances in knowledge:
We demonstrate that novel biphasic fractionation regimens could overcome the effects of tumour hypoxia resulting in biological dose escalation.
Introduction
Advances in technical radiotherapy have enabled superior spatial distribution of dose, resulting in the delivery of treatment with greater anatomic precision. However, there has been little effort focused on varying the temporal distribution of dose and most current schedules of fractionated radiotherapy use a uniform dose per fraction for the whole course of treatment.1 This is despite several studies providing significant evidence of temporal variations within radiobiological processes during treatment.
Hypoxia is one of the main causative factors of radioresistance and there have been a number of studies on the dynamics of hypoxia during a course of treatment. A small number of studies made direct measurements of oxygen tension before and during radiotherapy with some showing an improvement in oxygenation during radiotherapy.2,3 However, these findings were equivocal and likely influenced by the timing of taking measurements in relation to irradiation. Hypoxia imaging studies are easier to perform and have demonstrated a reduction in hypoxic volume as a course of radiotherapy progresses.4–7 In a18F-FAZA positron emission tomography (PET) study in patients with squamous cell carcinoma of the head and neck, a decrease in the hypoxic fraction from 21.7% at baseline to 3.6% during treatment was demonstrated.4 Similar findings have been reported in a number of other hypoxia PET imaging studies.5–7 These results suggest that tumour shrinkage improves the ability of the tumour vasculature to supply oxygen and nutrients, resulting in an increase in the reoxygenation rate which in turn leads to a significant reduction in the hypoxic fraction, as treatment proceeds.
Tumour cell repopulation is more prominent after around 3–4 weeks of radiotherapy with increased proliferation of clonogens at a rate faster than before treatment.8–11 This could be due to increases in growth fraction and shortened cell cycle time resulting from tumour shrinkage and improved blood supply.8–11 However, other studies suggest that release of cytokines by the inflammatory response to radiation may also contribute to this phenomenon.12 The “kick-off time” of accelerated repopulation is independent of dose delivered and is generally held to be around 21–28 days after the start of a course of radiotherapy.8,13–15 Accelerated repopulation is well recognised in certain tumour types such as squamous cell carcinoma of the head and neck, urothelial bladder cancer, and cervical cancer.14–17 However, in some tumours such as prostate cancer, evaluation of radiobiological data remains equivocal and some investigations do not show any evidence for this phenomenon.18
Several studies have demonstrated that tumour cells which survive to proliferate as a course of radiotherapy progresses are more radioresistant and have better DNA repair capacity (i.e. reduced α/β ratio) than at the onset of treatment, both due to the repopulation of intrinsically radioresistant cells as well as adaptive changes induced by exposure to fractionated radiotherapy.19–22 In vitro studies of prostate cancer and neuroblastoma cell lines have shown that fractionated radiotherapy leads to the selective proliferation of radioresistant tumour cells.19,21 Furthermore, a study of glioma stem cells showed that fractionated radiotherapy results in activation of checkpoint proteins leading to cell cycle arrest and synthesis of DNA repair proteins.23 It has also been shown that certain lung cancer cell lines surviving fractionated radiotherapy induce expression of genes associated with DNA repair.24
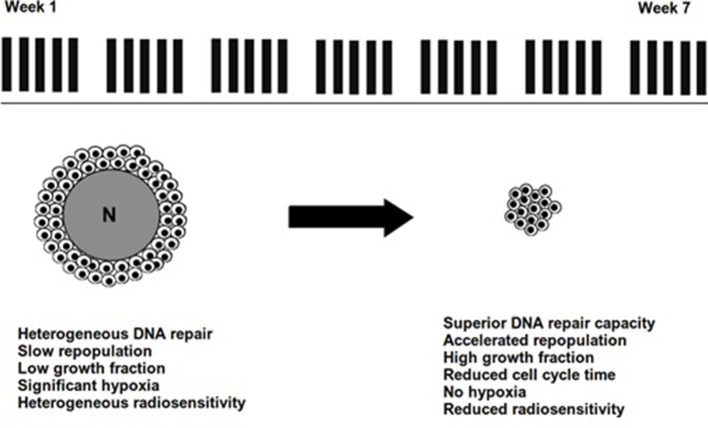
The therapeutic implications of the hypothesied changes in radiobiological phenomena during a course of radiotherapy are listed in Table 1. As summarised in Figure 1, as a treatment course proceeds, surviving clonogens are likely to be better oxygenated, proliferating faster and more radioresistant with superior repair capacity than at the outset.
Table 1.
The therapeutic implications of changes in tumour-specific radiobiological phenomena during a course of radiotherapy
| Phenomenon | Changes as the radiotherapy course progresses | Therapeutic implication |
|---|---|---|
| Repair | Selective survival and proliferation of cells with better DNA repair capacity | Higher doses per fraction will be more beneficial |
| Reoxygenation | Decrease in hypoxic fraction | OER* returns to unity resulting in the delivery of higher biological dose to tumour towards the end of the treatment course |
| Reassortment | Reduction in cell cycle time and increase in the proportion of proliferating cells | Diminished role for reassortment and fractionation |
| Repopulation | Accelerated repopulation commences 2–4 weeks after starting radiotherapy | Overall treatment time may need to be reduced to counter accelerated repopulation |
| Radiosensitivity | Selective survival and proliferation of radioresistant cells | Higher doses per fraction will be more beneficial as α/β is likely to be reduced. |
OER, oxygen enhancement ratio.
Figure 1.
Dynamic changes of radiobiological phenomena during radiotherapy. Tumour shrinkage results in decreased demand for oxygenation as well as better perfusion resulting in resolution of hypoxia. In addition, fractionated radiotherapy results in the selective proliferation of radioresistant cancer stem cells with better DNA repair capacities. (N denotes the central necrotic mass of a hypoxic tumour which is surrounded by viable tumour cells, some of which have a reduced supply of oxygen. The black bars indicate fractions of radiotherapy.)
Resolution of hypoxia should reduce the cumulative oxygen enhancement ratio (OER) for the whole tumour during treatment. Therefore, for tumours in which hypoxia and delayed repopulation are known factors of treatment failure, the use of accelerated fractionation after 3–4 weeks of conventional fractionation could lead to better outcomes than if acceleration was initiated from the outset. Alternatively, greater effect could be achieved by delivering a higher proportion of dose during the latter part of a course of radiotherapy when the tumour is relatively better oxygenated.
We conducted a radiobiological modelling study to test the hypothesis that regimens based on the above premise may result in a superior biological effective dose (BED) to tumour compared with conventional schedules.25
Methods
Objective
The objective is to evaluate alternative fractionation schedules that intend to deliver a higher BED to significantly hypoxic tumours whilst respecting a specified normal tissue dose constraint. For the purposes of this study, we assume that the hypoxic fraction reduces significantly during the first 28 days of a course of conventional fractionated radiotherapy.
Derivation of alternative fractionation schedules
We compare a conventional schedule of 70 Gy in 35 fractions delivered over 7 weeks with 2 alternative fractionation regimens derived from the methodology summarised below and described more fully in the Appendix. Both alternatives are designed to match thelate-responding normal tissue BED of the conventional schedule.
Accelerated radiotherapy after 4 weeks of conventional radiotherapy
Acceleration delivered over a whole course of radiotherapy could be counterproductive, since the highest proportion of dose might be given before the tumour has fully reoxygenated. We seek to overcome this by using hypofractionation and/or accelerated hyperfractionation only after 3–4 weeks of conventional radiotherapy, thereby ensuring that reoxygenation is probablycomplete before fractionation is altered.
The methodology involves calculating the optimum dose per fraction required to maximize the cell kill during the final (fully oxic) phase, as described in section A1 of the Supplementary Material.26–28 Once the optimum dose per fraction is derived then the corresponding number of fractions during the final phase is calculated based on consideration of the normal tissue tolerance. It is assumed that the critical normal tissue receives the same dose and dose–fractionation as the tumour. The optimum fraction size is dependent on the values of the BED equivalent repopulation rate (K), which could range from 0.6 to 0.9 Gy/day, for tumours with a significant repopulation rate.
Delivery of a proportionally higher total dose using increased dose per fraction after 4 weeks
Dose redistribution by delivering a proportionally higher Phase-2 dose forms the basis of this approach. It requires hyperfractionation over the first 28 days followed by hypofractionation, when reoxygenation is assumed to be nearly complete. In this scheme, the total dose, treatment duration, BED10Gy (for acute reactions) and BED3Gy (for late reactions) are designed to be identical to those in a conventional schedule of 70 Gy in 35 fractions.26 Since the treatment durations are identical the repopulation effect is assumed to be the same in both the novel and comparator schedule.
The objective is to derive a regimen that would deliver a proportionally higher BED during the latter part of treatment, i.e. during weeks 5–7 of radiotherapy. We chose a value of 60% during the second phase of treatment to ensure a balanced redistribution of dose. This is achieved by using larger fraction sizes in the second phase with the same number of fractions (15) as in the comparator schedule. The method described by Joiner is then used to determine the fractionation regimen for the first 4 weeks of radiotherapy.26
Calculation of improvement in tumour BEDs
Accelerated radiotherapy after 4 weeks of conventional radiotherapy
In both the comparator and novel schedule, the hypoxic and reoxygenation conditions during the first 28 days are identical since the fractionation during this period is the same in both cases. Therefore, the tumour BEDs at 28 days, although unknown, are identical and all the BED gains in the novel schedule are achieved in the final (accelerated) phase. Thus, the numerical difference between this Phase-2 BED and that of the post-28 day BED of the comparator schedule yields the absolute BED improvement for the entire novel treatment.
The methodology is described in more detail in section A1 of the Appendix. As mentioned previously, the derivation of the optimal fractionation regimen is based on an assumed value for the BED equivalent repopulation rate (K). However, in clinical practice this value is likely to vary between individual tumours. Therefore, tumour BED is computed for each fractionation regimen for a range of K values, in addition to the one on which the regimen was derived. As mentioned previously, it is further assumed that repopulation is fully underway at week 4 (kick-off time is less than 28 days). While this is likely to be the case for many tumours, there may be instances where the kick-off time is in excess of 28 days and therefore an additional analysis is performed where tumour BED is calculated assuming the kick-off time is 35 days. Finally, we consider the impact on tumour BED if repopulation does not occur, even though this approach is recommended only for tumours in which accelerated repopulation is a well-recognised phenomenon.
Delivery of a proportionally higher total dose with a high dose per fraction after 4 weeks
Assessment of the improvement in tumour BED is more involved in this case. This is because, unlike the above, the fractionation pattern is now altered in both phases, meaning that the Phase-1 BED of the novel schedule will not be the same as that of the comparator schedule and must therefore be calculated separately. This requires assumptions to be made about the pattern of reoxygenation during the first 28 days.
It is assumed that the reoxygenation rate increases progressively with time independent of the Phase-1 dose delivered and that hypoxic cells which have not already been sterilised by radiation move directly to an oxic state according to either a linear, sigmoidal reoxygenation vs time model. In the sigmoid model, it is assumed that a reoxygenation rate of 50% (t0) is reached by Day 21. However, we performed further analysis when the increase in rate is faster (t0 = 14 days) and slower (t0 = 21 days). In the linear model, it was assumed that the reoxygenation rate increases uniformly to reach completion (100%) by Day 28. We performed further analysis assuming completion of reoxygenation is reached by days 21 and 35 to represent fast and slow rises in reoxygenation rate. Since there could be tumours in which time-dependent increases in reoxygenation rate do not happen, we also tested our regimen in a fixed reoxygenation model where the rate is assumed to be 5% for all fractions.
The OER for hypoxic cells is assumed to be 2 and the modelling considers only discrete cellular jumps from fully hypoxic to fully oxic status. Following conventional practice, and as recommended by Chapman and Nahum,29 the oxic α and β values are respectively changed to α/OER and β/OER2 in hypoxic conditions. This step inherently assumes that the OER exerts simply a dose modifying influence and is therefore fraction-size independent. There is some evidence that effective OERs become smaller with reduced fraction sizes, especially at those significantly less than about 2 Gy although to incorporate that possibility requires the assumption of separate OER factors for α and β.30–33 The methodology for achieving this has been disused in more depth by Jones et al, but the required factors usually need to be derived from in-vitro studies, in-vivo-based data being rarely available and/or difficult to derive.34 Furthermore, some more recent in-vitro studies suggest that OERs may increase, decrease or remain the same as fraction size is altered, and this adds to the overall uncertainty.35 Since OER calculations are required only in Phase 1 of the temporal dose redistribution exercise, for which fraction sizes are either 2 Gy (for the reference schedule) or 1.12 Gy (for the novel schedule), the selection here of a dose-independent OER value of 2, being somewhat lower than the more usually assumed range of 2.5–3, is considered to be realistic. If it is the case that the effective OER is lower in Phase 1 of the novel regimen than in the comparator schedule then that would mean that the true BED improvements are likely to be better than those calculated here.
Repopulation is not considered as it is assumed to be the same in the reference and novel schedules, overall treatment times being the same in all cases.
Modelling the effects of reoxygenation on a tumour with oxic and anoxic components could be achieved by an iterative approach as shown by the work of Denekemp et al and Jones et al.34,36
A hypothetical two-compartment tumour initially made up of anoxic fractions ranging from 0 to 1 in increments of 0.1 is considered. To account for differences in intrinsic radiosensitivity (i.e. in α), we consider tumours with an α/β of 3 Gy as well as 10 Gy. The BED for the novel and conventional schedule is computed using an iterative approach for each hypoxic fraction, α/β ratio and reoxygenation model using the methodology described more fully in section A2 of the appendix and briefly outlined below.
For each fraction the following steps are followed:
For each dose fraction the reoxygenated fraction (RF) is calculated after deriving the reoxygenation rate using either the linear or sigmoid model.
After each dose the reoxygenated cell fraction is subtracted from the hypoxic fraction and added to the oxic fraction.
Steps (a) and (b) are repeated iteratively for each dose delivery. The surviving fraction of cells is calculated for both the oxic and hypoxic components after each dose and progressively summed until reoxygenation is complete.
Tumour BED is then calculated for the novel and conventional regimen based on the finalsurviving fraction of cells at the end of treatment using an assumed radiosensitivity (α) value of 0.3 Gy-1. The increment in the novel BED over the comparator BED being the measure of treatmentimprovement.
Calculation of normal tissue BED
BED10Gy and BED3Gy for normal tissues were computed for the conventional and novel regimens to compare acute and late normal tissue toxicity respectively. Since some of the novel regimens have accelerated fractionation, weekly BED10Gy is also calculated as a measure of dose intensity.
Results
Tumour BED
Accelerated radiotherapy after 4 weeks of conventional radiotherapy
Table 2 lists the derived optimal fraction sizes, adjusted fraction sizes and number of fractions for each value of K. The BED gains for each alternative regimen and the conventional regimen for the corresponding K value is listed in Table 3. The derived BED gains are also expressed as equivalent dose in 2 Gy fractions (EQD2), the associated physical dose when delivered in 2 Gy fractions. As shown in this table, the highest gains are seen with high repopulation rates (large K), with a marginally superior gain with a tumour α/β ratio of 3 Gy. These results suggest that 40 Gy in 20 fractions during days 1–26 followed by 22.32 Gy in 6 fractions during days 27–32 is the regimen which stands to achieve the highest gains in BED from this approach.
Table 2.
Optimal fraction sizes for use in phase-2 of a delayed acceleration regimen for various BED- equivalent repopulation rates (K values)
| K (Gy/day) | Optimal fraction size (Gy) | Adjusted fraction size (Gy) | Number of fractions |
|---|---|---|---|
| 0.6 | 2.68 | 2.65 | 10 |
| 0.7 | 3 | 3.08 | 8 |
| 0.8 | 3.32 | 3.37 | 7 |
| 0.9 | 3.63 | 3.72 | 6 |
BED, biological effective dose.K = BED equivalent repopulation rate;
BED, biological effective dose.K = BED equivalent repopulation rate.
The phase-1 fractionation is 40 Gy in 20 fractions, i.e. unchanged from that of the comparator schedule.
Table 3.
Comparison of overall tumour BED gains for conventional and delayed acceleration regimen for each BED-equivalent repopulation rate (K)
| K (Gy/day) | Phase-2 Dose per fraction (Gy) | Phase-2 Total dose (Gy) | Total BED10Gy novel regimen | Total BED10Gy conventional regimen | Absolute BED gain in 2 Gy equivalent dose | Total BED3Gy novel regimen | Total BED3Gy conventional regimen | Absolute BED gain in 2 Gy equivalent dose |
|---|---|---|---|---|---|---|---|---|
| 0.6 | 2.65 | 26.5 | 77.3 | 73.2 | 3.4 | 112.4 | 105.9 | 3.9 |
| 0.7 | 3.08 | 24.64 | 76.7 | 71.4 | 4.4 | 113.1 | 104.1 | 5.4 |
| 0.8 | 3.37 | 23.59 | 76.3 | 69.6 | 5.6 | 113.6 | 102.3 | 6.8 |
| 0.9 | 3.72 | 22.32 | 75.9 | 67.8 | 6.8 | 114 | 100.5 | 8.1 |
BED, biological effective dose.
The phase-1 fractionation is 40Gy in 20 fractions and the corresponding phase-1 and phase-2 BEDs are added to obtain the total BED.
supplementary tables shows the corresponding BED values for each regimen for a wide range of K values (0.3–0.9 Gy/day), kick-off times of 28 and 35 days and if repopulation does not occur (K = 0).
Predictably, the best gains with the novel regimen are seen when the K-value is high, there being only marginal differences between each of the associated accelerated fractionation schedules. If repopulation does not occur or when the K-value is low (0.3 Gy per day) the accelerated regimen results in more modest BED gains. However, and as indicated before, this regimen should only be applied in tumours where the phenomenon of accelerated repopulation is well recognised as a cause of treatment failure. In such cases, the new results show that the novel regimes will almost always out-perform conventional fractionation.
Delivery of a proportionally higher total dose with a high dose per fraction after 4 weeks
As shown in section A.2 of the Appendix, the final treatment regimen derived from this method would be 30.24 Gy in 27 fractions over 4 weeks followed by 40 Gy in 15 fractions over 3 weeks. To achieve the delivery of 27 fractions within the first 4 weeks of radiotherapy, and to avoid any sequential use of closely spaced fractions, weekend and/or bidaily treatments would be required.
The BED10Gy and BED3Gy for a range of initial hypoxic fractions with linear, sigmoidal and fixed reoxygenation models are listed in Table 4, along with the associated EQD2s. The BED of models with slow and fast rise in reoxygenation rate are shown in Supplementary Tables for the sigmoid and linear models respectively.
Table 4.
Comparison of total tumour BED10Gyand BED3Gyfor each hypoxic fraction of tumour and model of reoxygenation between conventional and redistributed dose regimens
| Initial Hypoxic fraction | Linear model of increase in reoxygenation rate | Sigmoid model of increase in reoxygenation rate | Fixed reoxygenation rate model | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| α / β ratio = 10 Gya | α / β ratio = 3 Gy** | α / β ratio = 10 Gya | α / β ratio = 3 Gy** | α / β ratio = 10 Gya | α / β ratio = 3 Gy** | |||||||||||||
| Conv BED | Novel BED | BED gain (EQD2) | Conv BED |
Novel BED | BED gain (EQD2) | Conv BED |
Novel BED | BED gain (EQD2) | Conv BED | Novel BED | BED gain (EQD2) | Conv BED | Novel BED | BED gain (EQD2) | Conv BED |
Novel BED | BED gain (EQD2) | |
| 0 | 84 | 84 | 0 | 116.7 | 116.7 | 0 | 84 | 84 | 0 | 116.7 | 116.7 | 0 | 84 | 84 | 0 | 116.7 | 116.7 | 0 |
| 0.1 | 81.2 | 83.1 | 1.6 | 114.7 | 115.9 | 0.7 | 71.7 | 78.1 | 5.3 | 106.7 | 113.1 | 3.8 | 51.6 | 52.9 | 1.1 | 87.5 | 92.3 | 2.9 |
| 0.2 | 79.7 | 82.4 | 2.3 | 113.1 | 115.3 | 1.3 | 69.4 | 76 | 5.5 | 101.6 | 110.4 | 5.3 | 49.3 | 50.6 | 1.1 | 80.2 | 85.2 | 3 |
| 0.3 | 78.7 | 81.8 | 2.6 | 111.6 | 114.6 | 1.8 | 68.1 | 74.8 | 5.6 | 98 | 108.3 | 6.2 | 47.9 | 49.3 | 1.2 | 76.8 | 80.9 | 2.5 |
| 0.4 | 77.9 | 81.3 | 2.8 | 110.4 | 114 | 2.2 | 67.1 | 73.9 | 5.7 | 95.4 | 106.5 | 6.7 | 47 | 48.3 | 1.1 | 72.7 | 77.8 | 3.1 |
| 0.5 | 77.3 | 80.9 | 3 | 109.2 | 113.4 | 2.5 | 66.4 | 73.2 | 5.7 | 93.3 | 104.9 | 7 | 46.2 | 47.6 | 1.2 | 70.2 | 75.4 | 3.1 |
| 0.6 | 76.7 | 80.5 | 3.2 | 108.2 | 112.9 | 2.8 | 65.8 | 72.6 | 5.7 | 91.5 | 103.6 | 7.3 | 45.6 | 47 | 1.2 | 68.3 | 73.4 | 3.1 |
| 0.7 | 76.3 | 80.2 | 3.3 | 107.2 | 112.4 | 3.1 | 65.3 | 72.1 | 5.7 | 90 | 102.3 | 7.4 | 45.1 | 46.5 | 1.2 | 66.6 | 71.7 | 3.1 |
| 0.8 | 75.9 | 79.9 | 3.3 | 106.4 | 111.9 | 3.3 | 64.8 | 71.7 | 5.8 | 88.6 | 101.2 | 7.6 | 44.7 | 46 | 1.1 | 65.1 | 70.3 | 3.1 |
| 0.9 | 75.5 | 79.6 | 3.4 | 105.5 | 111.4 | 3.5 | 64.4 | 71.3 | 5.7 | 87.4 | 100.2 | 7.7 | 44.3 | 45.6 | 1.1 | 63.8 | 69 | 3.1 |
| 1 | 75.2 | 79.3 | 3.4 | 104.8 | 111 | 3.7 | 64.1 | 70.9 | 5.7 | 86.3 | 99.3 | 7.8 | 43.9 | 45.3 | 1.2 | 62.6 | 67.8 | 3.1 |
70 Gy in 35 fractions over 7 weeksBED, biological effective doseConv, conventional regimen.
EQD2, Equivalent dose in 2 Gy fractions.
Novel Regimen: 30.24 Gy in 27 fractions over 4 weeks followed by 40 Gy in 15 fractions over 3 weeks.
α = 0.3 Gy−1 **α = 0.09 Gy−1
It can be seen that the novel regimen continues to show significant BED gains even with the fixed reoxygenation rate model although, in this case the absolute BEDs are lower for both conventional and novel regimens because fixed-rate reoxygenation results in a more selective survival of hypoxic (and radioresistant) cells. Fixed-rate reoxygenation is unlikely but, even if this were the case, the novel regimen still has superior BED to conventional fractionation.
The highest gains are achieved in tumours with larger hypoxic fractions, lower α/β ratio and with the sigmoid model of reoxygenation. In the sigmoid model of time-dependent increase in reoxygenation rate, greater gains in BED are observed when the increase in rate is slower than if there is rapid rise in reoxygenation rate.
Normal tissue toxicity
Table 5 lists the BED for acute and late responding normal tissues for the conventional and novel regimen, which indicate that the novel regimens are virtually isoeffective in terms of late toxicity. In the case of central nervous system (CNS), the calculations (using an α/β of 2 Gy) have been based on the assumption that a physical dose sparing of at least 20% can be achieved in all the regimens. For acute toxicity, with the strategy of conventional radiotherapy followed by acceleration, the total BED10Gy is less than the conventional regimen, although it is delivered with a slightly higher intensity due to shortening of treatment duration.
Table 5.
Comparison of normal tissue toxicity of the novel and conventional regimen
| Regimen | Acute toxicity (BED10Gy) | Acute toxicity (BED10Gy) per week | Late toxicity (BED3Gy) | CNS toxicity (BED2Gy)a |
|---|---|---|---|---|
| Conventional fractionation | ||||
| 70 Gy in 35 fractions over 7 weeks | 84 Gy | 12 Gy | 116.7 Gy | 100.8 Gy |
|
Conventional followed by hypofractionated accelerated radiotherapy 40 Gy in 20 fractions over 4 weeks followed by the second phase as follows: |
||||
| 26.5 Gy in 10 fractions | 81.5 Gy | 15.8 Gy | 116.7 Gy | 101.3 Gy |
| 24.64 Gy in eight fractions | 80.2 Gy | 16 Gy | 116.7 Gy | 101.6 Gy |
| 23.59 Gy in seven fractions | 79.5 Gy | 16.8 Gy | 116.9 Gy | 101.9 Gy |
| 22.32 Gy in six fractions | 78.6 Gy | 17.2 Gy | 116.7 Gy | 102.8 Gy |
| Hyperfractionation followed by hypofractionation | ||||
| 30.24 in 27 fractions over 4 weeks followed by 40 Gy in 15 fractions over 3 weeks | 84.3 Gy | 12.0 Gy | 117.0 Gy | 101.2 Gy |
BED, biological effective dose. CNS, central nervous system.
This column shows the potential CNS toxicity in terms of BED calculated with α/β = 2 Gy and on the assumption that a physical dose sparing of 20% is achievable. (For reference, a CNS dose of 50 Gy in 25 fractions over 5 weeks is associated with a BED2Gy of 100.0 Gy).
Discussion
The objective of our modeling work was to investigate whether in principle a BED gain could be achieved by the described novel strategies in fractionation. Our findings show that, in tumours with significant hypoxia, an improved tumour BED could be achieved by using fractionation strategies that either use acceleration in the latter part of treatment or use initial hyperfractionation followed by hypofractionation. In both cases, the altered fractionation is designed to cause no increase in normal tissue toxicity.
The first alternative strategy discussed here, of acceleration after 4 weeks of conventional radiotherapy, is independent of the dynamics of reoxygenation and of any assumption on its relationship with absolute dose. It depends on achieving an improvement by manipulating fractionation only after the majority of cells are fully oxygenated, making the modelling results more robust. However, since accelerated fractionation is used, acute toxicity may be increased even though the regimens are designed to be isotoxic in terms of late toxicity. As shown in Table 3 and Supplementary Tables, these regimen outperform conventional fractionation except when the repopulation rate is very low or when accelerated repopulation does not occur. In terms of improved tumour control, the absolute increases in tumour BEDs indicate that the novel schedules are capable of delivering overall EQD2s (equivalent doses when delivered in 2 Gy fractions) of between 3.5 and 8 Gy. These figures may seem relatively modest, but it should be recalled that the reference schedule delivers a “true” tumour BED which is substantially less than the conventionally calculated value, i.e. without any allowance for the effects of hypoxia. The achievable BED gains at the higher end of the EQD2 range are therefore not insignificant.
The DAHANCA trials in head and neck cancer achieved significant improvements in survival by shortening the overall treatment duration by 1 week.37 As such, we feel our accelerated regimen does have the potential to achieve clinically significant improvement in outcome.
Most current schedules of accelerated radiotherapy, involve treating multiple fractions per day or additional fractions on weekends.9,10,37 These schedules involve the delivery of a higher weekly dose from the outset, leading to most of the dose being delivered when hypoxia is still present, resulting in a reduced biological effect. Therefore, any gain resulting from accelerating is offset by losses due to hypoxia. Restricting the treatment acceleration until after reoxygenation of hypoxic cells is more complete overcomes this problem.
The meta-analysis of hyperfractionated and accelerated Radiotherapy in Carcinomas of Head and neck (MARCH) Collaborative Group report a significant improvement in local control with accelerated radiotherapy with a hazard ratio (HR) of 0.74 [confidence interval (CI) 0.67–0.83].10 However, regional control is not significantly improved by accelerated radiotherapy (HR 0.9; CI 0.77–1.04).10 Since hypoxic tumours have a more aggressive phenotype they are more likely to have nodal metastases. The divergence of outcome in local and regional control with use of accelerated radiotherapy could be due to tumour hypoxia reducing any therapeutic gain from shortening overall treatment duration.
Similarly with hypofractionated schedules, the potential reduction in biological effect due to hypoxia is even greater than in conventional regimens because the entire duration of treatment is virtually completed before improvements in tumour oxygenation can take effect. Indeed, the meta-analysis of concurrent hypoxia modification and radiotherapy in head and neck cancer showed a larger numerical impact in patients treated with hypofractionation versus conventional fractionation.38 The hazard ratio of hypoxia modification was 0.56 (95% CI 0.40–0.77) in hypofractionated schedules while it was 0.77 (0.67–0.89) with conventional fractionation.38
The second alternative strategy, of proportional dose escalation after 4 weeks, has the advantage that it is designed to be isotoxic with the conventional regimen in terms of both acute and late toxicity. In tumours with significant hypoxia, this strategy results in substantial gains (with EQD2s again of up to 8 Gy) and, since overall treatment time is not altered, these gains are independent of repopulation rate. However, the methodology needs assumptions to be made on the model of reoxygenation independent of the dose delivered during the first 4 weeks of radiotherapy. We have tested three models of reoxygenation and in all instances the novel regimen is superior to the conventional regimen. In addition, for the models of time-dependent increase in reoxygenation rate we considered a wide range of parameters to simulate slow and fast rises to test the robustness of our regimen. The largest gains are achieved in tumours with high hypoxic fractions, with slow rise in reoxygenation rate using the sigmoid model. Even under more modest conditions of a hypoxic fraction of 10%, BED gains of 3–8 Gy (EQD2) are achieved with the novel regimen. Furthermore, the gains are much higher if the tumour α/β ratio is lower.
Both the strategies considered here are designed to be isoeffective to the comparator schedule in terms of normal tissue effects, i.e. the normal tissue BEDs are unchanged in the alternative schedules. Except in the case of CNS, for which a physical dose sparing of 20% is assumed to be achjevable, the normal tissues are assumed to receive 100% of the prescribed tumour dose. As most normal tissue volumes will receive a lesser dose in practice than the tumour the assumption made here provides an inherent degree of additional safety.
Even though our models required several assumptions on radiobiological processes, we tested our regimen using a wide range of values for these parameters. Overall, our results strongly suggest a likely superiority for the novel regimens in a wide variety of circumstances. However, both strategies require treatment during weekends or additional fractions per day which may result in practical difficulties for radiotherapy departments.
The choice of an optimised fractionation regimen should ideally be based on the extent of hypoxia, reoxygenation rate, repopulation rate and the intrinsic radiosensitivity of the tumour. The failure of previous experimentation with modified fractionation schedules to make significant improvements in outcome could be attributed to the empirical basis on which they were used. The potential for personalised selection of fractionation regimen based on predictive biomarkers to improve outcomes over empirical application has already been shown by Jones and Dale.39 Although a number of biomarkers of hypoxia, tumour radiosensitivity and repopulation have shown considerable promise none have been validated prospectively.40–43
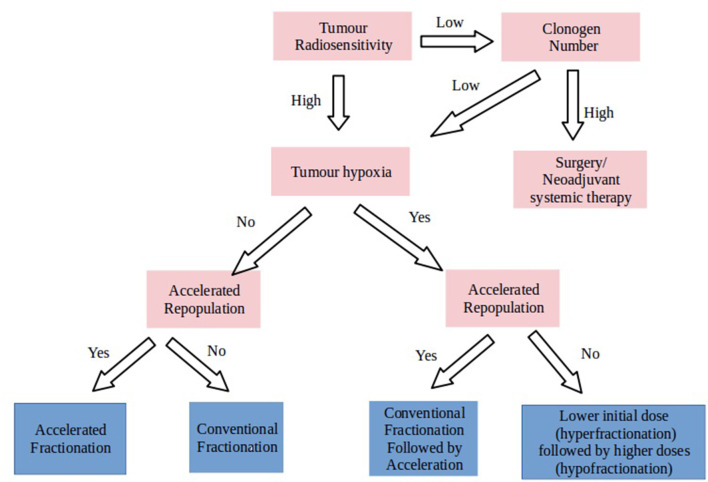
As shown in Figure 2, well-oxygenated tumours with high radiosensitivity could be treated with conventional schedules. If the clonogen number is low they might even be suitable for a dose de-escalation approach to reduce toxicity of treatment. For well-oxygenated tumours with a high repopulation rate, hypofractionated regimens are more suited. However, if hypoxia is present, the best approach would be 3–4 weeks of conventional radiotherapy followed by acceleration. For hypoxic tumours with low repopulation rates, fractionation regimens that deliver a potentially higher total dose after 3–4 weeks of radiotherapy would be more beneficial. For radioresistant tumours with a high clonogen population, alternative treatment modalities such as neoadjuvant chemotherapy or surgery used as the initial method of treatment would be required as these tumours are unlikely to be cured by radiotherapy alone.
Figure 2.
Schema for biomarker driven personalised fractionation.
Conclusion
This work provides a basis for further modeling studies that could potentially lead to clinical trials of fractionation schedules that either deliver a higher proportional dose towards the end of a course of treatment or use acceleration after 4 weeks of conventional radiotherapy. Such approaches are indicated in tumours with significant hypoxia and accelerated repopulation such as squamous cell carcinoma of the head and neck and muscle-invasive urothelial bladder cancer.
Footnotes
Acknowledgement: Professors Peter Hoskin, Catharine West and Ananya Choudhury are supported by the NIHR Manchester Biomedical Research Centre.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
The authors Ananya Choudhury and Roger G. Dale contributed equally to the work.
Contributor Information
Nuradh Joseph, Email: nuradh@gmail.com.
Norman F. Kirkby, Email: norman.kirkby@manchester.ac.uk.
Peter J Hoskin, Email: peterhoskin@nhs.net.
Catharine M L West, Email: catharine.west@manchester.ac.uk.
Ananya Choudhury, Email: ananya.choudhury@christie.nhs.uk.
Roger G. Dale, Email: r.dale@imperial.ac.uk.
REFERENCES
- 1.Withers HR. The 4Rs of radiotherapy : edsLett J. T, Adler H, Zelle M, Advances in Radiation Biology: New York, Academic Press; 1975. 241–71. [Google Scholar]
- 2.Cooper RA, West CM, Logue JP, Davidson SE, Miller A, Roberts S, et al. Changes in oxygenation during radiotherapy in carcinoma of the cervix. Int J Radiat Oncol Biol Phys 1999; 45: 119–26. doi: 10.1016/s0360-3016(99)00093-0 [DOI] [PubMed] [Google Scholar]
- 3.Sheridan MT, West CM, Cooper RA, Stratford IJ, Logue JP, Davidson SE, et al. Pretreatment apoptosis in carcinoma of the cervix correlates with changes in tumour oxygenation during radiotherapy. Br J Cancer 2000; 82: 1177–82. doi: 10.1054/bjoc.1999.1059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zegers CML, Hoebers FJP, van Elmpt W, Bons JA, Öllers MC, Troost EGC, et al. Evaluation of tumour hypoxia during radiotherapy using [18F]HX4 PET imaging and blood biomarkers in patients with head and neck cancer. Eur J Nucl Med Mol Imaging 2016; 43: 2139–46. doi: 10.1007/s00259-016-3429-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee N, Nehmeh S, Schöder H, Fury M, Chan K, Ling CC, et al. Prospective trial incorporating pre-/mid-treatment [18F]-misonidazole positron emission tomography for head-and-neck cancer patients undergoing concurrent chemoradiotherapy. Int J Radiat Oncol Biol Phys 2009; 75: 101–8. doi: 10.1016/j.ijrobp.2008.10.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Servagi-Vernat S, Differding S, Hanin F-X, Labar D, Bol A, Lee JA, et al. A prospective clinical study of ¹⁸F-FAZA PET-CT hypoxia imaging in head and neck squamous cell carcinoma before and during radiation therapy. Eur J Nucl Med Mol Imaging 2014; 41: 1544–52. doi: 10.1007/s00259-014-2730-x [DOI] [PubMed] [Google Scholar]
- 7.Bollineni VR, Koole MJB, Pruim J, Brouwer CL, Wiegman EM, Groen HJM, et al. Dynamics of tumor hypoxia assessed by 18F-FAZA PET/CT in head and neck and lung cancer patients during chemoradiation: possible implications for radiotherapy treatment planning strategies. Radiother Oncol 2014; 113: 198–203. doi: 10.1016/j.radonc.2014.10.010 [DOI] [PubMed] [Google Scholar]
- 8.Withers HR, Taylor JM, Maciejewski B. The hazard of accelerated tumor clonogen repopulation during radiotherapy. ActaOncol 1998; 27: 131–46. [DOI] [PubMed] [Google Scholar]
- 9.Brenner DJ. Accelerated repopulation during radiotherapy: quantitative evidence for delayed onset. Radiat. Oncol. Investig 1993; 1: 167–72. [Google Scholar]
- 10.Bourhis J, Overgaard J, Audry H, Ang KK, Saunders M, Bernier J, et al. Hyperfractionated or accelerated radiotherapy in head and neck cancer: a meta-analysis. Lancet 2006; 368: 843–54. doi: 10.1016/S0140-6736(06)69121-6 [DOI] [PubMed] [Google Scholar]
- 11.Marcu LG, Bezak E. Influence of stem-cell cycle time on accelerated re-population during radiotherapy in head and neck cancer. Cell Prolif 2012; 45: 404–12. doi: 10.1111/j.1365-2184.2012.00832.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Trott KR, Kummermehr J. Accelerated repopulation in tumours and normal tissues. RadiotherOncol 1991; 22: 159–60. [DOI] [PubMed] [Google Scholar]
- 13.Dörr W, Three DW. Three a's of repopulation during fractionated irradiation of squamous epithelia: asymmetry loss, acceleration of stem-cell divisions and abortive divisions. Int J Radiat Biol 1997; 72: 635–43. doi: 10.1080/095530097142780 [DOI] [PubMed] [Google Scholar]
- 14.Blanchard P, Hill C, Guihenneuc-Jouyaux C, Baey C, Bourhis J, Pignon JP, et al. Mixed treatment comparison meta-analysis of altered fractionated radiotherapy and chemotherapy in head and neck cancer. J Clin Epidemiol 2011; 64: 985–92. doi: 10.1016/j.jclinepi.2010.10.016 [DOI] [PubMed] [Google Scholar]
- 15.Dale RG, Hendry JH, Jones B, Robertson AG, Deehan C, Sinclair JA, et al. Practical methods for compensating for missed treatment days in radiotherapy, with particular reference to head and neck schedules. Clin Oncol 2002; 14: 382–93. doi: 10.1053/clon.2002.0111 [DOI] [PubMed] [Google Scholar]
- 16.Maciejewski B, Majewski S. Dose fractionation and tumour repopulation in radiotherapy for bladder cancer. Radiother Oncol 1991; 21: 163–70. doi: 10.1016/0167-8140(91)90033-d [DOI] [PubMed] [Google Scholar]
- 17.Petereit DG, Sarkaria JN, Chappell R, Fowler JF, Hartmann TJ, Kinsella TJ, et al. The adverse effect of treatment prolongation in cervical carcinoma. Int J Radiat Oncol Biol Phys 1995; 32: 1301–7. doi: 10.1016/0360-3016(94)00635-X [DOI] [PubMed] [Google Scholar]
- 18.Tamponi M, Gabriele D, Maggio A, Stasi M, Meloni GB, Conti M, et al. Prostate cancer dose-response, fractionation sensitivity and repopulation parameters evaluation from 25 international radiotherapy outcome data sets. Br J Radiol 2019; 92: 20180823. doi: 10.1259/bjr.20180823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McDermott N, Meunier A, Mooney B, Nortey G, Hernandez C, Hurley S, et al. Fractionated radiation exposure amplifies the radioresistant nature of prostate cancer cells. Sci Rep 2016; 6: 34796. doi: 10.1038/srep34796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ogawa K, Utsunomiya T, Mimori K, Tanaka F, Haraguchi N, Inoue H, et al. Differential gene expression profiles of radioresistant pancreatic cancer cell lines established by fractionated irradiation. Int J Oncol 2006; 28: 705–13. [PubMed] [Google Scholar]
- 21.Russell J, Wheldon TE, Stanton P. A radioresistant variant derived from a human neuroblastoma cell line is less prone to radiation-induced apoptosis. Cancer Res 1995; 55: 4915–21. [PubMed] [Google Scholar]
- 22.Shimura T, Kakuda S, Ochiai Y, Kuwahara Y, Takai Y, Fukumoto M, et al. Targeting the AKT/GSK3β/cyclin D1/Cdk4 survival signaling pathway for eradication of tumor radioresistance acquired by fractionated radiotherapy. Int J Radiat Oncol Biol Phys 2011; 80: 540–8. doi: 10.1016/j.ijrobp.2010.12.065 [DOI] [PubMed] [Google Scholar]
- 23.Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006; 444: 756–60. doi: 10.1038/nature05236 [DOI] [PubMed] [Google Scholar]
- 24.Desai A, Webb B, Gerson SL. Cd133+ cells contribute to radioresistance via altered regulation of DNA repair genes in human lung cancer cells. Radiother Oncol 2014; 110: 538–45. doi: 10.1016/j.radonc.2013.10.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fowler JF. 21 years of biologically effective dose. Br J Radiol 2010; 83: 554–68. doi: 10.1259/bjr/31372149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Joiner MC. A simple alpha/beta-independent method to derive fully isoeffective schedules following changes in dose per fraction. Int J Radiat Oncol Biol Phys 2004; 58: 871–5. doi: 10.1016/j.ijrobp.2003.10.036 [DOI] [PubMed] [Google Scholar]
- 27.Jones B, Tan LT, Dale RG. Derivation of the optimum dose per fraction from the linear quadratic model. Br J Radiol 1995; 68: 894–902. doi: 10.1259/0007-1285-68-812-894 [DOI] [PubMed] [Google Scholar]
- 28.Dale RG, Jones B. High-LET radiotherapy : Dale R. G, Jones B, Radiobiogical Modelling in Radiation Oncology. London: British Institute of Radiology; 2007. 272. [Google Scholar]
- 29.Chapman JD, Nahum AE (). Radiotherapy treatment planning – linear quadratic radiobiology. . 13 Pub: CRC Press; 2015. 978–1-4398-6259-9authorsISBN. [Google Scholar]
- 30.Skarsgard LD, Harrison I. Dose dependence of the oxygen enhancement ratio (OER) in radiation inactivation of Chinese hamster V79-171 cells. Radiat Res 1991; 127: 243–7. [PubMed] [Google Scholar]
- 31.Resvesz I, Palcic B. Radiation dose dependence of the sensitisation of oxygen and oxygen mimic sensitisers. Acta. Radiol 1985; 24: 209–17. [DOI] [PubMed] [Google Scholar]
- 32.Marples B, Joiner MC, Skov KA. The effect of oxygen on low-dose hypersensitivity and increased radioresistance in Chinese hamster V79-379A cells. Radiat Res 1994; 138(1 Suppl): S17–20. [PubMed] [Google Scholar]
- 33.Harriss-Phillips WM, Bezak E, Yeoh EK. Altered fractionation outcomes for hypoxic head and neck cancer using the HYP-RT Monte Carlo model. Br J Radiol 2013; 86: 20120443. doi: 10.1259/bjr.20120443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jones B, Carabe-Fernandez A, Dale RG. The Oxygen Effect : Dale R. G, Jones B, Radiobiogical Modelling in Radiation Oncology. London: British Institute of Radiology; 2007. 138–57. [Google Scholar]
- 35.Wenzl T, Wilkens JJ. Theoretical analysis of the dose dependence of the oxygen enhancement ratio and its relevance for clinical applications. Radiat Oncol 2011; 6: 171–7. doi: 10.1186/1748-717X-6-171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Denekamp J, McNally NJ, Fowler JF, Joiner MC. Misonidazole in fractionated radiotherapy: are many small fractions best? Br J Radiol 1980; 53: 981–90. doi: 10.1259/0007-1285-53-634-981 [DOI] [PubMed] [Google Scholar]
- 37.Overgaard J, Hansen HS, Specht L, Overgaard M, Grau C, Andersen E, et al. Five compared with six fractions per week of conventional radiotherapy of squamous-cell carcinoma of head and neck: DAHANCA 6 and 7 randomised controlled trial. Lancet 2003; 362: 933–40. doi: 10.1016/s0140-6736(03)14361-9 [DOI] [PubMed] [Google Scholar]
- 38.Overgaard J. Hypoxic modification of radiotherapy in squamous cell carcinoma of the head and neck--a systematic review and meta-analysis. Radiother Oncol 2011; 100: 22–32. doi: 10.1016/j.radonc.2011.03.004 [DOI] [PubMed] [Google Scholar]
- 39.Jones B, Dale RG. Radiobiological modeling and clinical trials. Int J Radiat Oncol Biol Phys 2000; 48: 259–65. doi: 10.1016/s0360-3016(00)00542-3 [DOI] [PubMed] [Google Scholar]
- 40.Forker LJ, Choudhury A, Kiltie AE. Biomarkers of tumour radiosensitivity and predicting benefit from radiotherapy. Clin Oncol 2015; 27: 561–9. doi: 10.1016/j.clon.2015.06.002 [DOI] [PubMed] [Google Scholar]
- 41.Yang L, West CM. Hypoxia gene expression signatures as predictive biomarkers for personalising radiotherapy. Br J Radiol 2019; 92: 0036. doi: 10.1259/bjr.20180036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lara PC, Rey A, Santana C, Afonso JL, Diaz JM, González GJ, et al. The role of Ki67 proliferation assessment in predicting local control in bladder cancer patients treated by radical radiation therapy. Radiother Oncol 1998; 49: 163–7. doi: 10.1016/s0167-8140(98)00033-4 [DOI] [PubMed] [Google Scholar]
- 43.Yang J, Yue J-B, Liu J, Yu J-M. Repopulation of tumor cells during fractionated radiotherapy and detection methods (review. Oncol Lett 2014; 7: 1755–60. doi: 10.3892/ol.2014.1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.