Supplemental Digital Content is available in the text.
Keywords: cohort studies, epidemiology, risk assessment, stroke, troponin
Abstract
Background and Purpose:
Stroke is a common cause of death and a leading cause of disability and morbidity. Stroke risk assessment remains a challenge, but circulating biomarkers may improve risk prediction. Controversial evidence is available on the predictive ability of troponin concentrations and the risk of stroke in the community. Furthermore, reports on the predictive value of troponin concentrations for different stroke subtypes are scarce.
Methods:
High-sensitivity cardiac troponin I (hsTnI) concentrations were assessed in 82 881 individuals (median age, 50.7 years; 49.7% men) free of stroke or myocardial infarction at baseline from 9 prospective European community cohorts. We used Cox proportional hazards regression to determine relative risks, followed by measures of discrimination and reclassification using 10-fold cross-validation to control for overoptimism. Follow-up was based upon linkage with national hospitalization registries and causes of death registries.
Results:
Over a median follow-up of 12.7 years, 3033 individuals were diagnosed with incident nonfatal or fatal stroke (n=1654 ischemic strokes, n=612 hemorrhagic strokes, and n=767 indeterminate strokes). In multivariable regression models, hsTnI concentrations were associated with overall stroke (hazard ratio per 1-SD increase, 1.15 [95% CI, 1.10–1.21]), ischemic stroke (hazard ratio, 1.14 [95% CI, 1.09–1.21]), and hemorrhagic stroke (hazard ratio, 1.10 [95% CI, 1.01–1.20]). Adding hsTnI concentrations to classical cardiovascular risk factors (C indices, 0.809, 0.840, and 0.736 for overall, ischemic, and hemorrhagic stroke, respectively) increased the C index significantly but modestly. In individuals with an intermediate 10-year risk (5%–20%), the net reclassification improvement for overall stroke was 0.038 (P=0.021).
Conclusions:
Elevated hsTnI concentrations are associated with an increased risk of incident stroke in the community, irrespective of stroke subtype. Adding hsTnI concentrations to classical risk factors only modestly improved estimation of 10-year risk of stroke in the overall cohort but might be of some value in individuals at an intermediate risk.
Stroke risk assessment in the community remains a challenge. Although many clinical risk factors for stroke have been identified, a substantial proportion of stroke risk remains unexplained.1 Cardiac troponin is a structural protein released into the blood flow as a consequence of myocardial injury.2 Its usefulness as a sensitive marker for myocardial damage has been widely tested and become a routine measurement in clinical practice, in particular, in the diagnosis of acute coronary syndromes.3 However, high-sensitivity assays have led to a significantly enhanced detection of troponin concentrations, considerably broadening its clinical application and interpretation.4 Mild elevations of troponin concentrations are associated with a higher risk for cardiovascular disease and death in the general population.4–6 A recent meta-analysis showed an association between elevated troponin concentrations and incident stroke in the general population but also revealed large heterogeneity among the included studies.7 While the additional benefit of high-sensitivity assayed cardiac troponins to predict overall cardiovascular disease has been repeatedly demonstrated, their value for the prediction of incident stroke in primary prevention is unknown.5,6,8,9 Furthermore, reports on the association and predictive performance of troponin in relation to stroke subtypes are scarce.7,9
Therefore, based on cohorts of the BiomarCaRE (Biomarkers for Cardiovascular Risk Assessment in Europe) consortium,10 our primary goal was to further investigate the relationship of high-sensitivity cardiac troponin I (hsTnI) with incident stroke in Europe and to examine a potential difference in the association of hsTnI with ischemic and hemorrhagic stroke. In a second step, we aimed to determine the additional predictive value of hsTnI beyond classical cardiovascular risk factors.
Methods
Because of the sensitive nature of the data collected for this study, requests to access the dataset from qualified researchers trained in human subject confidentiality protocols may be sent to the corresponding author.
Study Sample
We pooled participant-level data from 9 community cohorts of the BiomarCaRE project with information on hsTnI levels and stroke status at baseline and follow-up (http://www.biomarcare.eu/), the MONICA Brianza Study, the KORA study (Cooperative Health Research in the Region of Augsburg), the DanMONICA study, the FINRISK study, the MATISS Rome study, the Moli-sani study, the Northern Sweden MONICA study, the PRIME (Prospective Epidemiological Study of Myocardial Infarction) from Belfast, and the SHHEC (Scottish Heart Health Extended Cohort), comprising 86 104 unique individuals.10 Each cohort is based on population samples with baseline examinations between 1982 and 2010, comprising in total 21 subcohorts (please see the Data Supplement for details on the enrollment and follow-up procedures of each cohort). The study protocol was approved by the local ethics review boards of all study centers, and all participants provided written informed consent. The protocol adhered to the principles of the Declaration of Helsinki. The data from the cohorts were harmonized in the MONICA Risk, Genetics, Archiving and Monograph (MORGAM) Project.11
Individuals with a positive history of stroke or myocardial infarction based on self-report or prior physician’s diagnosis were excluded from analyses, including individuals in whom baseline data indicated coronary heart disease, but separation between myocardial infarction, angina pectoris, and possible cardiac revascularization was not possible (n=3223). Therefore, 82 881 individuals were included in the analysis.
Follow-Up and Definition of Outcomes
Follow-up was mainly based upon linkage with national hospitalization registries and causes of death registries. In addition, some cohorts obtained information through direct contact to participants or general practitioners and linkage to the national medication registries. Follow-up for mortality was obtained from central death registries. The follow-up for the different cohorts was completed between 2004 and 2012.12
Incident strokes and identification of the stroke subtype were validated according to MORGAM criteria (for details on all outcome classifications, please see https://www.ahajournals.org/journal/str).
Briefly, a stroke was categorized as an ischemic stroke if at least one of the following is present:
Validation of recent brain infarction by necropsy.
Circumscribed hypodensity changes of recent origin in the brain parenchyma on computed tomography.
Typical signs of infarct in the brain parenchyma on magnetic resonance imaging.
An event was also considered as ischemic stroke in the data analysis if there was no validation according to MORGAM criteria or validation was not possible due to insufficient data, but the routine clinical or death certificate diagnoses indicated cerebral infarction (International Classification of Diseases [ICD], Eighth Revision, code of 432, 433, or 434; ICD, Ninth Revision, code of 433 or 434; or ICD, Tenth Revision, code of I63).
Hemorrhagic stroke was defined as first fatal or nonfatal intracerebral or subarachnoid hemorrhage based on MORGAM criteria. To be classified as hemorrhagic stroke, at least one of the following criteria must be present:
Validation of recent intracerebral hemorrhage or subarachnoid hemorrhage by necropsy.
Typical signs of bleeding in the brain parenchyma (intracerebral hemorrhage) or signs of blood in the subarachnoid cisterns or in cerebral ventricles (subarachnoid hemorrhage) on computed tomography or magnetic resonance imaging.
Bloody cerebrospinal fluid (liquor) in the presence of focal neurological signs at onset.
An event was also considered as hemorrhagic stroke in the data analysis if there was no validation according to MORGAM criteria or validation was not possible due to insufficient data but the routine clinical or death certificate diagnoses indicated hemorrhagic stroke (ICD, Eighth Revision, code of 430 or 431; ICD, Ninth Revision, code of 430 or 431; or ICD, Tenth Revision, code of I60 or I61).
Strokes that could be classified neither as ischemic nor hemorrhagic were called indeterminate. An incident coronary event was defined as a composite end point of any cardiac revascularization and acute coronary events including hospitalization for unstable angina pectoris. Incident atrial fibrillation (AF) was defined by date of the first documentation on ECG or assignment of the relevant ICD code (427.4 for ICD, Eighth Revision; 427.3 for ICD, Ninth Revision; and I48 for ICD, Tenth Revision).
Laboratory Methods
All troponin I levels were measured by an hsTnI assay at the central BiomarCaRE laboratory in Hamburg (Abbott Diagnostics; ARCHITECT i2000SR). In line with previous studies, 1.9 pg/mL was considered the limit of detection (LoD).6,8 Observed values below this threshold were included for analyses in this study (assay range, 0–50 000 pg/mL). The assay supported a 10% coefficient of variation at a concentration of 5.2 pg/mL.
Statistical Analyses
Baseline characteristics are expressed as numbers and percentages for categorical variables, medians and interquartile ranges for continuous variables. To avoid possible problems caused by obvious outliers, hsTnI data were winsorized by replacing the 3 highest hsTnI values with the fourth highest value in each subcohort. We used multiple imputation to handle missing data in continuous variables.13 For the dichotomous variables, missing values were recoded to zero (no) to avoid incompleteness (please see https://www.ahajournals.org/journal/str for details on the multiple imputation).
Association Analyses
To examine the association of hsTnI with incident stroke, we performed Cox proportional hazards regression models with overall stroke, ischemic stroke, and hemorrhagic stroke as the outcome. Age was used as the time scale. In these analyses, hsTnI was log-transformed (logarithm of hsTnI+1). We performed incremental adjustment starting with a cohort-adjusted and sex-stratified model (model 1) and then added the variables of the Framingham risk score for estimating the 10-year risk of cardiovascular events in primary care (body mass index, systolic blood pressure, total and high-density lipoprotein cholesterol level, antihypertensive medication, diabetes mellitus, and daily smoking; model 2).14 This set of risk factors was used for all analyses. Adjustment for region within studies was performed where available. In a third step, we calculated a Cox regression analysis with fatal and nonfatal coronary event as time-dependent covariates (model 3). In a further analysis, we also accounted for incident AF as a time-dependent covariate (model 4). In a sensitivity analysis, we excluded individuals with prevalent AF or heart failure at baseline. MONICA Brianza, KORA, MATISS, and PRIME were excluded from the latter 2 analyses since data on AF were not available for these cohorts. Individuals were censored at the end of follow-up, death, or the time of their event in respective analyses.
For categorical analysis, we defined 3 cut points for hsTnI at 1.9 pg/mL (LoD) as the lowest cut point and by further subdividing individuals with observed hsTnI values above the LoD into thirds (resulting in cut points of 2.9 and 4.8 pg/mL, respectively). P trend was calculated for linear increase in log relative hazards with increasing categories. Kaplan-Meier curves for incident overall stroke, ischemic stroke, and hemorrhagic stroke were produced using categorized troponin concentrations. The score log-rank test was used to test for potential survival difference between hsTnI categories. Proportional hazards assumption was tested by plotting scaled Schoenfeld residuals against follow-up time for each covariate separately and using χ2 test to test for a nonzero slope.
Prediction Analyses
In prediction analyses, 10-year absolute risks were estimated by a Weibull curve fitted over age for the baseline hazard and adjusted by the linear predictor of the estimated Cox model with and without hsTnI. Ten-fold cross-validation was used to control for overoptimism. The additional value of hsTnI concentrations to the Framingham risk factors was assessed by calculating C-index improvement, integrated discrimination improvement, as well as categorical and continuous net reclassification improvement (NRI).15 The risk categories used for the categorical NRI analysis were 0% to 5%, 5% to 10%, 10% to 20%, and >20%. We further determined the clinical NRI, which refers to individuals with an intermediate 10-year risk of cardiovascular events (5%–20%). For the calculation of C indices and NRI, the follow-up time was censored at 10 years.
A 2-sided value of P≤0.05 was considered statistically significant. All statistical analyses were conducted with R statistical software, version 3.6.0 (the R project for statistical computing).
Results
Population Characteristics
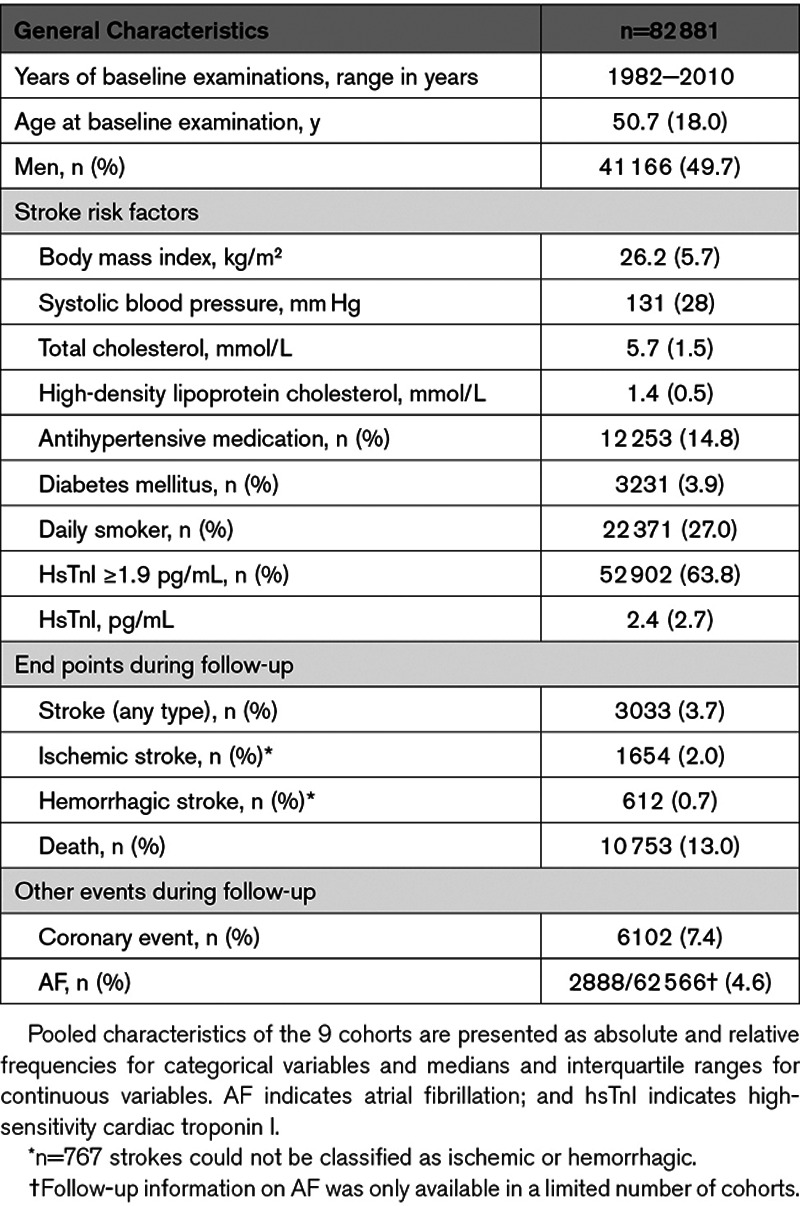
Eighty-two thousand eight hundred eighty-one individuals were included for analyses in this study, 49.7% being men. The median age at baseline was 50.7 (interquartile range, 18.0) years (Table 1). The prevalence of diabetes mellitus at baseline was 3.9%, 14.8% were treated with antihypertensive medication, and about every fourth was a daily smoker. Over a median follow-up of 12.7 years, 3033 (3.7%) individuals were diagnosed with stroke. Of these, 1654 (2.0% of the overall cohort) had an ischemic stroke, 612 (0.7%) had a hemorrhagic stroke, and stroke subtype could not be specified in 767 (0.9%) individuals.
Table 1.
Characteristics of the Study Population
hsTnI Concentrations and Their Association With Incident Stroke
The median hsTnI value in the overall cohort was 2.4 pg/mL (interquartile range, 2.7 pg/mL). Values above the LoD were observed in 52 902 (63.8%) individuals. HsTnI was associated with a significantly increased risk for overall (hazard ratio [HR], 1.15 per 1-SD increase [95% CI, 1.10–1.21]; P<0.001), ischemic (HR per SD, 1.14 [95% CI, 1.09–1.21]; P<0.001), and hemorrhagic stroke (HR per SD, 1.10 [95% CI, 1.01–1.20]; P=0.032). The inclusion of incident coronary events and AF as time-dependent covariates did not markedly alter the results (Table 2). The multivariable adjusted HRs for incident stroke increased with increasing hsTnI categories, and individuals in the highest category had a 42% increased risk for overall stroke compared with individuals with hsTnI levels below the LoD (HR, 1.42 [95% CI, 1.24–1.61]; P<0.001; Figure 1). The HRs for ischemic and hemorrhagic stroke were 1.45 ([95% CI, 1.22–1.74] P<0.001) and 1.39 ([95% CI, 1.07–1.80] P<0.001) for individuals in the highest compared with the lowest category, respectively. Figure 2 displays survival curves according to categories of hsTnI for overall, ischemic, and hemorrhagic stroke. The association of hsTnI with incident overall stroke was observed in all cohorts (Figure 3).
Table 2.
HRs for hsTnI in Relation to Stroke
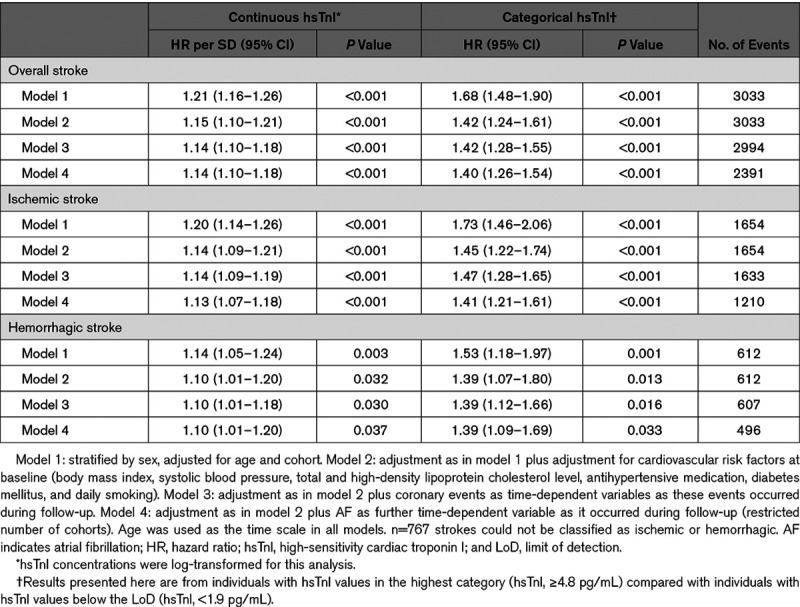
Figure 1.
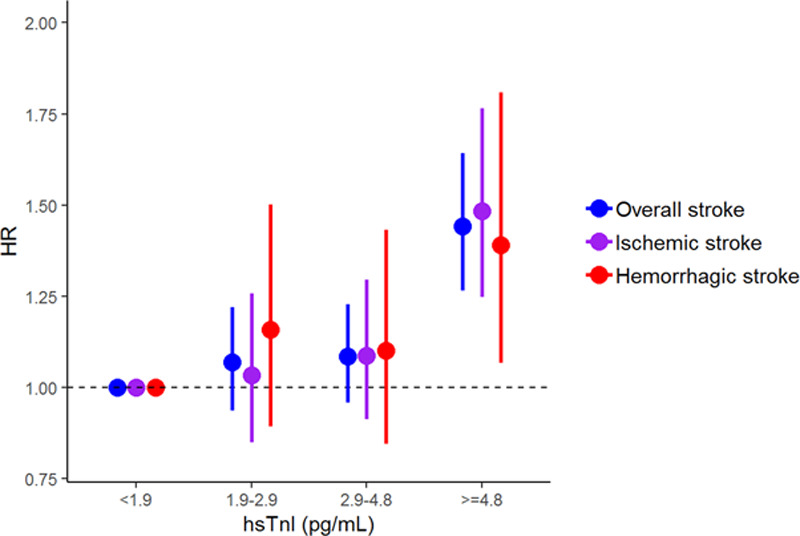
Hazard ratios (HRs) and 95% CIs for high-sensitivity cardiac troponin I (hsTnI) categories. The presented HRs are based on a sex-stratified Cox regression analyses with adjustment for cohort, body mass index, systolic blood pressure, total and high-density lipoprotein cholesterol level, antihypertensive medication, diabetes mellitus, and daily smoking (model 2).
Figure 2.
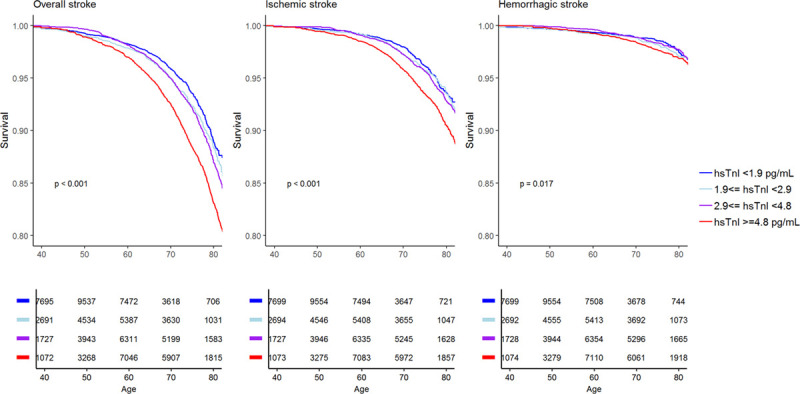
Kaplan-Meier curves of stroke-free survival according to high-sensitivity cardiac troponin I (hsTnI) categories. Numbers below the curves represent individuals at risk for the different hsTnI categories at a certain age. Note that the y axes are truncated at 0.8. Given P values are for the comparison of the highest and the lowest hsTnI categories.
Figure 3.
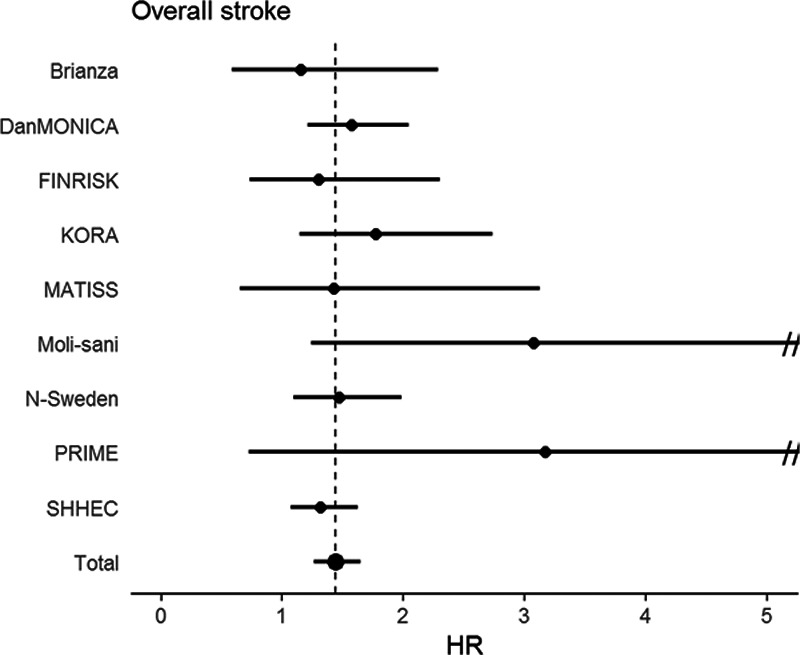
Hazard ratio (HR) for the highest vs the lowest category of high-sensitivity cardiac troponin I (hsTnI) across cohorts. This figure displays the HRs and 95% CIs (adjusted as in model 2; Table 2) for the highest vs the lowest category of hsTnI in the separate and overall cohorts for overall stroke. Note that the x axis is truncated at 5, since the 95% CI was much wider in 2 cohorts (Moli-sani and PRIME [Prospective Epidemiological Study of Myocardial Infarction]). KORA indicates Cooperative Health Research in the Region of Augsburg; and SHHEC, Scottish Heart Health Extended Cohort.
hsTnI Concentrations and Stroke Risk
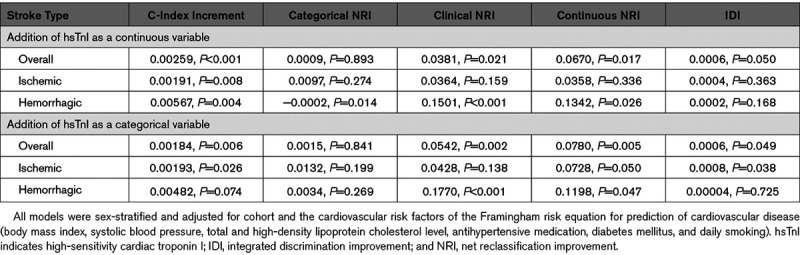
The Framingham risk factors revealed C indices of 0.809, 0.840, and 0.736 for overall, ischemic, and hemorrhagic stroke, respectively. Adding hsTnI as a continuous variable increased the C indices by 0.003 ([95% CI, 0.001–0.004] P<0.001) for overall stroke, by 0.002 ([95% CI, 0.001–0.003] P=0.008) for ischemic stroke, and by 0.006 ([95% CI, 0.002–0.010] P=0.004) for hemorrhagic stroke. The addition of hsTnI as a continuous variable showed a categorical NRI of 0.001 ([95% CI, −0.012 to 0.014] P=0.893) for overall stroke, 0.010 ([95% CI, −0.008 to 0.027] P=0.274) for ischemic stroke, and −0.0002 ([95% CI, −0.0004 to −0.00004] P=0.014) for hemorrhagic stroke. Focusing on individuals with an intermediate 10-year risk (5%–20%), we found a clinical NRI for overall, ischemic, and hemorrhagic stroke of 0.038 ([95% CI, 0.006–0.070] P=0.021), 0.036 ([95% CI, −0.014 to 0.087] P=0.159), and 0.150 ([95% CI, 0.068–0.232] P<0.001), respectively. Details on all risk prediction analyses are provided in Table 3.
Table 3.
Improvement of 10-y Risk Prediction Using Troponin I Measured by High-Sensitivity Assay for Overall, Ischemic, and Hemorrhagic Stroke Over the First 10 y of Follow-Up
Discussion
In our pooled analysis of 9 European community-based cohorts, we demonstrate an association of hsTnI concentrations with incident stroke, independent of stroke subtype. The addition of hsTnI to the variables of the Framingham risk score hardly improved stroke risk prediction among all subjects. Focusing on individuals with an intermediate 10-year stroke risk revealed a moderate improvement in net reclassification for overall stroke, as well as for stroke subtypes.
hsTnI as a Stroke Risk Factor
A recent meta-analysis of 12 studies conducted in the general population demonstrated an association of high-sensitivity assayed troponins and incident stroke.7 However, the authors reported a great heterogeneity in the included studies, and no information on stroke subtype was provided. A report from the ARIC study (Atherosclerosis Risk in Communities) revealed an association of high-sensitivity assayed troponin T with incident ischemic stroke with the highest HRs for cardioembolic stroke but did not show a significant association with hemorrhagic stroke, possibly due to the small numbers of incident events.9 In a more recent analysis, an association of hsTnI with incident ischemic stroke of similar strength compared with the present study was demonstrated using the same hsTnI assay, but they did not report on hemorrhagic stroke.5 In the present study, we found an association of hsTnI with ischemic and hemorrhagic stroke. A possible explanation for the observed association between hsTnI and ischemic stroke is that these events might be related to asymptomatic arrhythmias, in particular, AF.16 Although, in some cases of a (cardio)embolic stroke, a secondary hemorrhagic transformation might have led to a misclassification of stroke subtype, this concept does not sufficiently explain the association of hsTnI with hemorrhagic stroke. Hypertension and increasing age constitute major risk factors for both stroke subtypes, but etiology and further risk factors differ significantly.1,17 Elevated hsTnI values reflect a subclinical manifestation of cardiac injury, likely as a consequence of cardiac stress due to the presence of cardiovascular risk factors, for example, arterial hypertension.18 Therefore, the association of hsTnI with incident stroke might be an indicator of an underlying systemic (vascular) disease, considering the overlap of cardiovascular and cerebrovascular risk factors.
hsTnI as a Predictor of Stroke
HsTnI hardly improved overall risk prediction beyond classical cardiovascular risk factors in this study. Earlier studies demonstrated a potential benefit in global cardiovascular risk prediction by addition of hsTnI, but reports focusing on incident stroke are rare.5,6,8 Two prior studies found no significant improvement in stroke risk prediction by adding high-sensitivity troponin T or hsTnI.8,9 It is difficult to improve discrimination beyond classical risk factors, in particular, when risk indicators such as hsTnI are partially correlated with the other variables.6,15 In the setting of primary prevention, individuals with an estimated intermediate risk for incident disease are most likely to benefit from additional guidance on potential therapy initiation/modification, since guideline recommendations for these individuals are often less clear.1 Considering the observed clinical NRI in our study, the assessment of hsTnI might be of help in selected individuals at intermediate cardiovascular risk in whom standard stroke risk classification yields inconclusive results.
Limitations and Strengths
Several limitations merit consideration. Stroke diagnosis was based on medical records rather than on standard neurological examination or review of brain imaging. Despite the systematic and detailed validation of stroke events, residual misclassification cannot be ruled out. Furthermore, due to the comparatively small number of events, we did not perform separate analyses for subarachnoid and intracerebral hemorrhages, although their pathophysiology differs significantly.
HsTnI was measured only once at the time of inclusion of the individuals in the specific cohort. Therefore, information on possible changes of hsTnI and how these might be related to incident stroke was not available.
Considering that NT-proBNP (N-terminal pro-B-type natriuretic peptide) has been shown to be associated with both ischemic and hemorrhagic stroke,19 it would have been preferable to include NT-proBNP into our analysis and to assess potential interaction of both biomarkers in their association with incident stroke. However, NT-proBNP measurements were not available for all cohorts, and, therefore, the number of incident stroke events would have decreased significantly. Further, earlier studies suggest that the association of troponin with stroke is independent of NT-proBNP levels.4,5,9
Our results are based on European cohorts and cannot be extrapolated to other populations, in particular, since an interaction by race for the association of hsTnI and stroke has been suggested.5
The strength of our study is the large sample from several countries with a long follow-up and carefully harmonized data in which we can provide results with good power, in particular, with regard to the less common outcome of hemorrhagic stroke.
Conclusions
Elevated hsTnI concentrations are associated with an increased risk of incident stroke in the general population, irrespective of stroke subtype. A possible explanation may be underlying systemic cardiovascular disease. However, adjustment for interim cardiovascular disease did not alter the results substantially. Adding hsTnI concentrations to classical cardiovascular risk factors only modestly improved the prediction of 10-year risk of stroke in the overall cohort but might be of some value in intermediate-risk groups. More importantly, the pathophysiological relationship between the cardiac biomarker hsTnI and stroke needs to be further elucidated.
Acknowledgments
We thank the participants and the staff of the cohorts for their continuing dedication and efforts.
Sources of Funding
The BiomarCaRE (Biomarkers for Cardiovascular Risk Assessment in Europe) Project is funded by the European Union Seventh Framework Programme (FP7/2007-2013) under grant agreement No. HEALTH-F2-2011-278913. Dr Schnabel has received funding from the European Research Council under the European Union’s Horizon 2020 research and innovation programme (grant agreement No. 648131), from the European Union’s Horizon 2020 research and innovation programme (grant agreement No. 847770; AFFECT-EU), and the German Center for Cardiovascular Research (DZHK e.V.) (81Z1710103), German Federal Ministry of Research and Education (BMBF; 01ZX1408A) and ERACoSysMed3 (031L0239). The activities of the MONICA Risk, Genetics, Archiving and Monograph (MORGAM) Data Center have been sustained by recent funding from the European Union FP 7 project CHANCES (HEALTH-F3-2010-242244). The MORGAM Biomarker Study (Serum Biomarkers in the MORGAM Populations) has further received funding from the Medical Research Council London (G0601463, No. 80983). The KORA study (Cooperative Health Research in the Region of Augsburg) was initiated and financed by the Helmholtz Zentrum München–German Research Center for Environmental Health, which is funded by the BMBF and by the State of Bavaria. Furthermore, KORA research was supported within the Munich Center of Health Sciences, Ludwig-Maximilians-Universität, as part of LMUinnovativ. The FINRISK surveys were mainly supported by budgetary funds of THL with additional funding from numerous nonprofit foundations. Dr Salomaa (principal investigator) has been supported by the Finnish Foundation for Cardiovascular Research and the Academy of Finland (139635). Dr Niiranen has been supported by the Finnish Medical Foundation, the Emil Aaltonen Foundation, the Paavo Nurmi Foundation, and the Academy of Finland (321351). The DanMONICA cohorts at the Research Center for Prevention and Health were established over a period of 10 years and have been funded by numerous sources, which have been acknowledged, where appropriate, in the original articles. The MATISS Project was partly supported by the National Research Council, by the Istituto Superiore di Sanità-ISS (1984, 1987,1993-1996) and by the Ministry of Health (1998). The Moli-sani Project was partially supported by research grants from Pfizer Foundation (Rome, Italy), the Italian Ministry of University and Research (Rome, Italy)–Programma Triennale di Ricerca, Decreto n.1588, and Instrumentation Laboratory, Milan, Italy. The Northern Sweden MONICA project was supported by the Norrbotten and Västerbotten County councils. Dr Söderberg has been supported by the Swedish Heart-Lung Foundation (20140799, 20120631, and 20100635), the County Council of Västerbotten (ALF, VLL-548791), and Umeå University. The SHHEC (Scottish Heart Health Extended Cohort) received funding from the Scottish Health Department Chief Scientist Organization, the British Heart Foundation, and the FP Fleming Trust.
Disclosures
Dr Di Castelnuovo reports grants from the European Foundation for Alcohol Research as the coapplicant of the ongoing study supported by a research grant (id. EA1767) unrelated to the current study. Dr Kee reports funding from grants of the UK Clinical Research Collaboration and from the Wellcome Trust unrelated to the current work. Dr Schnabel reports personal fees from BMS/Pfizer and lecture and advisory board fees unrelated to the current study. Dr Blankenberg reports research funding from Abbott Diagnostics, Bayer, SIEMENS, Singulex, and Thermo Fisher. He further received honoraria for lectures from Abbott, Abbott Diagnostics, AstraZeneca, Bayer, AMGEN, Medtronic, Pfizer, Roche, SIEMENS Diagnostics, SIEMENS, and Thermo Fisher and as member of Advisory Boards and for consulting for Bayer, Novartis, and Thermo Fisher. Dr Costanzo reports funding from an ERAB grant (id. EA1767) and personal fees as a member of the Organizing Committee and speaker for the Ninth European Beer and Health Symposium (Bruxelles 2019) and for given lecture at the 13th European Nutrition Conference (FENS 2019, Dublin), all unrelated to the current work. Dr Söderberg reports personal fees from Actelion, Ltd. Dr Salomaa has received honoraria from Novo Nordisk and Sanofi for consultations. He also has ongoing research collaboration with Bayer AG (all unrelated to the present study). Dr Koenig reports personal fees from AstraZeneca, Novartis, Pfizer, The Medicines Company, DalCor, Kowa, Amgen, Corvidia, Berlin-Chemie, Sanofi, Bristol-Myers Squibb, and Daichii-Sankyo and nonfinancial support by grants from Abbott, Roche Diagnostics, Beckmann, and Singulex, all unrelated to the current study. The other authors report no conflicts.
Supplementary Material
Footnotes
Nonstandard Abbreviations and Acronyms
- AF
- atrial fibrillation
- BiomarCaRE
- Biomarkers for Cardiovascular Risk Assessment in Europe
- HR
- hazard ratio
- hsTnI
- high-sensitivity cardiac troponin I
- ICD
- International Classification of Diseases
- KORA
- Cooperative Health Research in the Region of Augsburg
- LoD
- limit of detection
- MORGAM
- MONICA Risk, Genetics, Archiving and Monograph
- NRI
- net reclassification improvement
- NT-proBNP
- N-terminal pro-B-type natriuretic peptide
- PRIME
- Prospective Epidemiological Study of Myocardial Infarction
- SHHEC
- Scottish Heart Health Extended Cohort
The Data Supplement is available with this article at https://www.ahajournals.org/doi/suppl/10.1161/STROKEAHA.120.029452.
For Sources of Funding and Disclosures, see page 2776 and 2777.
References
- 1.Meschia JF, Bushnell C, Boden-Albala B, Braun LT, Bravata DM, Chaturvedi S, Creager MA, Eckel RH, Elkind MS, Fornage M, et al. ; American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; Council on Functional Genomics and Translational Biology; Council on Hypertension Guidelines for the primary prevention of stroke: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014453754–3832doi: 10.1161/STR.0000000000000046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Adams JE, III, Bodor GS, Dávila-Román VG, Delmez JA, Apple FS, Ladenson JH, Jaffe AS. Cardiac troponin I. A marker with high specificity for cardiac injury. Circulation 199388101–106doi: 10.1161/01.cir.88.1.101 [DOI] [PubMed] [Google Scholar]
- 3.Keller T, Zeller T, Peetz D, Tzikas S, Roth A, Czyz E, Bickel C, Baldus S, Warnholtz A, Fröhlich M, et al. Sensitive troponin I assay in early diagnosis of acute myocardial infarction. N Engl J Med 2009361868–877doi: 10.1056/NEJMoa0903515 [DOI] [PubMed] [Google Scholar]
- 4.Willeit P, Welsh P, Evans JDW, Tschiderer L, Boachie C, Jukema JW, Ford I, Trompet S, Stott DJ, Kearney PM, et al. High-sensitivity cardiac troponin concentration and risk of first-ever cardiovascular outcomes in 154,052 participants. J Am Coll Cardiol 201770558–568doi: 10.1016/j.jacc.2017.05.062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jia X, Sun W, Hoogeveen RC, Nambi V, Matsushita K, Folsom AR, Heiss G, Couper DJ, Solomon SD, Boerwinkle E, et al. High-sensitivity troponin I and incident coronary events, stroke, heart failure hospitalization, and mortality in the ARIC Study. Circulation 20191392642–2653doi: 10.1161/CIRCULATIONAHA.118.038772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Blankenberg S, Salomaa V, Makarova N, Ojeda F, Wild P, Lackner KJ, Jørgensen T, Thorand B, Peters A, Nauck M, et al. ; BiomarCaRE Investigators Troponin I and cardiovascular risk prediction in the general population: the BiomarCaRE consortium. Eur Heart J 2016372428–2437doi: 10.1093/eurheartj/ehw172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Broersen LHA, Stengl H, Nolte CH, Westermann D, Endres M, Siegerink B, Scheitz JF. Association between high-sensitivity cardiac troponin and risk of stroke in 96 702 individuals: a meta-analysis. Stroke 202011085–1093doi: 10.1161/STROKEAHA.119.028323 [DOI] [PubMed] [Google Scholar]
- 8.Zeller T, Tunstall-Pedoe H, Saarela O, Ojeda F, Schnabel RB, Tuovinen T, Woodward M, Struthers A, Hughes M, Kee F, et al. ; MORGAM Investigators High population prevalence of cardiac troponin I measured by a high-sensitivity assay and cardiovascular risk estimation: the MORGAM Biomarker Project Scottish Cohort. Eur Heart J 201435271–281doi: 10.1093/eurheartj/eht406 [DOI] [PubMed] [Google Scholar]
- 9.Folsom AR, Nambi V, Bell EJ, Oluleye OW, Gottesman RF, Lutsey PL, Huxley RR, Ballantyne CM. Troponin T, N-terminal pro-B-type natriuretic peptide, and incidence of stroke: the atherosclerosis risk in communities study. Stroke 201344961–967doi: 10.1161/STROKEAHA.111.000173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zeller T, Hughes M, Tuovinen T, Schillert A, Conrads-Frank A, Ruijter Hd, Schnabel RB, Kee F, Salomaa V, Siebert U, et al. BiomarCaRE: rationale and design of the European BiomarCaRE project including 300,000 participants from 13 European countries. Eur J Epidemiol 201429777–790doi: 10.1007/s10654-014-9952-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Evans A, Salomaa V, Kulathinal S, Asplund K, Cambien F, Ferrario M, Perola M, Peltonen L, Shields D, Tunstall-Pedoe H, et al. ; MORGAM Project MORGAM (an international pooling of cardiovascular cohorts). Int J Epidemiol 20053421–27doi: 10.1093/ije/dyh327 [DOI] [PubMed] [Google Scholar]
- 12.Kulathinal S, Niemelä M, Niiranen T, Saarela O, Palosaari T, Tapanainen H, Kuulasmaa K; Contributors from Participating Centres, for the MORGAM Project Description of MORGAM Cohorts [Internet]. 2005https://www.thl.fi/publications/morgam/cohorts/index.html. Accessed May 4, 2019
- 13.van Buuren S, Groothuis-Oudshoorn K. Mice: multivariate imputation by chained equations in R. J Stat Softw. 2011;45:67. [Google Scholar]
- 14.D’Agostino RB, Sr, Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM, Kannel WB. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation 2008117743–753doi: 10.1161/CIRCULATIONAHA.107.699579 [DOI] [PubMed] [Google Scholar]
- 15.Pencina MJ, D’Agostino RB, Sr, D’Agostino RB, Jr, Vasan RS. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med 200827157–172discussion 207. doi: 10.1002/sim.2929 [DOI] [PubMed] [Google Scholar]
- 16.McCarthy CP, Yousuf O, Alonso A, Selvin E, Calkins H, McEvoy JW. High-sensitivity troponin as a biomarker in heart rhythm disease. Am J Cardiol 20171191407–1413doi: 10.1016/j.amjcard.2017.01.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.O’Donnell MJ, Xavier D, Liu L, Zhang H, Chin SL, Rao-Melacini P, Rangarajan S, Islam S, Pais P, McQueen MJ, et al. ; INTERSTROKE Investigators Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case-control study. Lancet 2010376112–123doi: 10.1016/S0140-6736(10)60834-3 [DOI] [PubMed] [Google Scholar]
- 18.Seliger SL, Hong SN, Christenson RH, Kronmal R, Daniels LB, Lima JAC, de Lemos JA, Bertoni A, deFilippi CR. High-sensitive cardiac troponin T as an early biochemical signature for clinical and subclinical heart failure: MESA (Multi-Ethnic Study of Atherosclerosis). Circulation 20171351494–1505doi: 10.1161/CIRCULATIONAHA.116.025505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Di Castelnuovo A, Veronesi G, Costanzo S, Zeller T, Schnabel RB, de Curtis A, Salomaa V, Borchini R, Ferrario M, Giampaoli S, et al. ; BiomarCaRE Investigators NT-proBNP (N-terminal pro-B-type natriuretic peptide) and the risk of stroke. Stroke 201950610–617doi: 10.1161/STROKEAHA.118.023218 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


