ABSTRACT
Objective:
To describe the incidence of active tuberculosis and the occurrence of adverse events after isoniazid treatment in patients with latent tuberculosis infection (LTBI) who also had chronic inflammatory diseases and were treated with immunobiologic agents in an endemic area in Brazil.
Methods:
The diagnosis of LTBI was based on anamnesis, clinical examination, chest X-ray, and a tuberculin skin test (TST). Patients received prophylactic treatment (isoniazid for six months) in accordance with the Brazilian guidelines.
Results:
A total of 101 patients were evaluated between July of 2011 and July of 2015. Of those, 55 (54.46%) were women (mean age, 53.16 ± 1.76 years) and 46 (45.54%) were men (mean age, 45.39 ± 2.13 years). A total of 79 patients (78.22%) were being treated with immunobiologic agents and 22 (21.78%) were being treated with immunomodulatory or immunosuppressive agents. In the screening for LTBI, 53 patients (52.48%) had a TST induration ≥ 10 mm. Chest X-ray findings consistent with LTBI were observed in 36 patients (35.64%). Isoniazid preventive therapy was effective in 96 (95.05%) of the 101 patients evaluated. It is of note that 84 (83.17%) of the patients experienced no adverse effects from the use of isoniazid and that 83 (98.81%) of those patients completed the prophylactic treatment (p = 0.002). Active tuberculosis was diagnosed in 5 (6.33%) of the 79 patients treated with immunobiologic agents and in 1 (4.55%) of the 22 patients treated with other immunomodulators/immunosuppressants.
Conclusions:
A six-month course of isoniazid proved to be safe and effective in the treatment of LTBI, which is essential to reducing the risk of developing active tuberculosis.
Keywords: Latent tuberculosis, Tuberculin test, Tumor necrosis factors/antagonists & inhibitors, Isoniazid
RESUMO
Objetivo:
Descrever a incidência de tuberculose ativa e a ocorrência de eventos adversos do tratamento com isoniazida em pacientes diagnosticados com tuberculose latente (TBL), portadores de doenças inflamatórias crônicas e tratados com agentes imunobiológicos em uma área endêmica no Brasil.
Métodos:
O diagnóstico de TBL foi feito com base em anamnese, exame clínico, radiografia de tórax e teste tuberculínico (TT). O tratamento profilático foi realizado segundo diretrizes brasileiras com isoniazida por seis meses.
Resultados:
Foram estudados 101 pacientes entre julho de 2011 e julho de 2015. Desses, 55 (54,46%) eram mulheres (média de idade = 53,16 ± 1,76 anos) e 46 (45,54%) eram homens (média de idade = 45,39 ± 2,13 anos), sendo que 79 (78,22%) foram tratados com agentes imunobiológicos e 22 (21,78%) com outros agentes imunomoduladores ou imunossupressores. Na triagem para TBL, 53 pacientes (52,48%) apresentaram TT ≥ 10 mm. A radiografia de tórax alterada por imagens compatíveis com TBL foi observada em 36 pacientes (35,64%). O tratamento profilático com isoniazida mostrou uma eficácia de 95,05% (96/101). É relevante mencionar que 84 (83,17%) dos pacientes não apresentaram nenhum efeito adverso à isoniazida e, desses, 83 (98,81%) completaram o tratamento profilático (p = 0,002). Tuberculose ativa foi diagnosticada em 5 (6,33%) dos 79 pacientes tratados com agentes imunobiológicos e em 1 (4,55%) dos 22 pacientes tratados com outros imunomoduladores/imunossupressores.
Conclusões:
O uso de isoniazida por seis meses mostrou-se seguro e eficaz no tratamento da TBL nesses pacientes, o que é essencial para reduzir o risco de desenvolvimento de tuberculose ativa.
Descritores: Tuberculose latente, Teste tuberculínico, Fatores de necrose tumoral/antagonistas & inibidores, Isoniazida
INTRODUCTION
Tuberculosis is an airborne infectious disease caused by Mycobacterium tuberculosis; because tuberculosis incidence and mortality are high, surveillance and treatment are a priority according to the World Health Organization. 1
Approximately 2 billion people worldwide are infected with M. tuberculosis, and approximately 1 in 10 individuals will develop tuberculosis at some point in their lives. Although mortality from tuberculosis has significantly decreased, the World Health Organization estimated that there were 10.4 million new cases of tuberculosis worldwide in 2016, i.e., 140 cases per 100,000 population. 2
Brazil is one of the 22 countries that collectively account for 80% of all cases of tuberculosis worldwide, ranking 16th in terms of absolute numbers of cases (66,796 in 2016) and 22nd in terms of tuberculosis incidence rate (32.4/100,000 population in 2016). 3
Some groups are more susceptible to tuberculosis infection than the general population, including people living with HIV/AIDS and patients with immune-mediated chronic inflammatory diseases such as rheumatoid arthritis, ankylosing spondylitis, psoriasis, and Crohn’s disease, as well as patients with immunosuppressive conditions such as diabetes, chronic kidney disease, and solid organ transplantation. Patients with chronic inflammatory diseases are currently treated with immunosuppressants or immunomodulators; they are increasingly being treated with anti-TNF biologic agents and are at an increased risk of developing opportunistic infections, including tuberculosis. 1 , 4
TNF inhibitors revolutionized the clinical treatment of chronic inflammatory diseases, improving the quality of life of patients. However, biologic agents increase the risk of developing tuberculosis. TNF plays an essential role in maintaining the immune response, particularly the integrity of granulomas in tuberculosis infection. In addition, TNF directly activates macrophages, which are responsible for phagocytosis of pathogens. In nearly 90% of individuals infected with M. tuberculosis, the bacilli remain contained within granulomas, resulting in latent tuberculosis infection (LTBI). Approximately 5-10% will develop active tuberculosis disease, the risk being highest in the first 5 years following infection. 5
Given that TNF-α is a mediator of inflammatory pathways and has bactericidal properties, the adverse consequences of TNF-α inhibition raise major concerns for treatment with biologic agents. TNF-α inhibitors can cause severe immunosuppression, which leads to failure to control LTBI and, consequently, tuberculosis disease, reinfection with M. tuberculosis having been reported in several studies of patients treated with biologic agents. 4
The primary objective of the present study was to describe the incidence of active tuberculosis and the occurrence of adverse events after isoniazid treatment in patients with LTBI and chronic inflammatory disease treated with biologic agents in an endemic area in Brazil.
METHODS
This was a prospective cohort study involving 101 patients with immune-mediated chronic inflammatory diseases. All of the patients were candidates for treatment with biologic agents and tested positive for LTBI, isoniazid preventive therapy (IPT) therefore being indicated. The selected patients were followed for 4 years. The study was conducted at a university hospital between June of 2011 and June of 2015, being approved by the local research ethics committee (Protocol no. 058.05.11).
The diagnosis of LTBI was based on anamnesis, clinical examination, chest X-ray, and a Mantoux tuberculin skin test (TST), being made after active tuberculosis disease had been excluded. The TST was performed by a trained technician in the clinical analysis laboratory of the hospital and consisted of delivering PPD RT23 tuberculin intradermally in the middle third of the volar aspect of the left forearm at a dose of 0.1 mL, which is equivalent to two tuberculin units. The results of the TST were read 72 h after PPD RT23 administration. A TST induration ≥ 5 mm was considered positive. Posteroanterior and lateral chest X-rays were performed in the radiology department of the hospital and were evaluated by two independent physicians (a radiologist in the radiology department of the hospital and a pulmonologist). Chest X-ray findings were classified as normal, abnormal because of the presence of granuloma or a small calcified nodule, or abnormal because of the presence of other, minimal, residual changes (striae, nodular fibrosis, pleural thickening, or any combination of the three). The history of risk factors for tuberculosis, including respiratory symptoms, current or previous tuberculosis treatment, and a history of exposure to tuberculosis, was also evaluated.
IPT was performed in accordance with the Third Brazilian Thoracic Association Guidelines on Tuberculosis, 6 i.e., at a dose of 5-10 mg/kg of body weight (maximum dose, 300 mg/day) for 6 months. Treatment with biologic agents was initiated no sooner than 1 month after initiation of IPT. Adverse reactions to isoniazid and clinical outcomes of IPT were evaluated. Figure 1 shows a flow chart of LTBI screening outcomes among candidates for treatment with TNF-α inhibitors.
Figure 1. Flow chart of latent tuberculosis infection (LTBI) screening outcomes among candidates for treatment with TNF-α inhibitors. TST: tuberculin skin test; CXR: chest X-ray; TB: tuberculosis; and IPT: isoniazid preventive therapy.
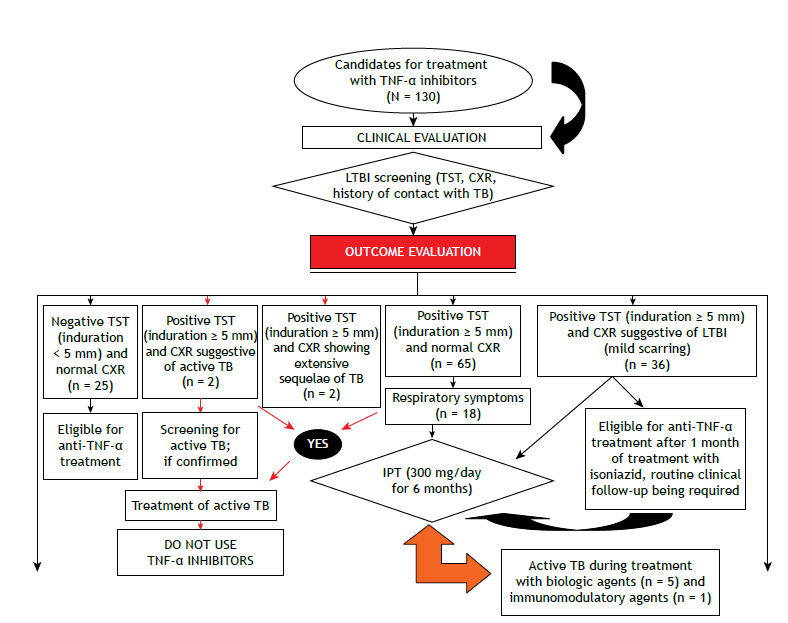
Isoniazid toxicity was measured during IPT by determining serum levels of alanine aminotransferase and aspartate aminotransferase. Hepatotoxicity was defined as elevated liver transaminase levels (≥ three- to fivefold above the upper limit of normal). 7
All statistical analyses were performed with the Statistical Package for the Social Sciences, version 16.0 (SPSS Inc., Chicago, IL, USA). The two-tailed Pearson’s chi-square test with Yates’ correction was used in order to determine significant associations. The two-tailed Fisher’s exact test was used for contingency tables with fewer than five cells (p < 0.05), and the Student’s t-test was used in order to compare the means. A multivariate analysis was performed in order to assess the strength of association among variables of interest; ORs, prevalence ratios (relative risk), and their respective 95% CIs were calculated. Values of p < 0.05 were considered significant.
RESULTS
A total of 101 patients were included in the analysis. Of those, 55 (54.46%) were women (mean age, 53.16 ± 1.76 years) and 46 (45.54%) were men (mean age, 45.39 ± 2.13 years). Patient age ranged from 21 years to 76 years (Table 1). Of the 101 participants, 79 (78.22%) were being treated with biologic agents and 22 (21.78%) were being treated with immunomodulatory or immunosuppressive agents. Most (n = 84) of the patients (83.17%) had rheumatic diseases, including rheumatoid arthritis (in 42) and ankylosing spondylitis (in 42). Of the 55 women in the sample, 34 (61.82%) had rheumatoid arthritis. Of the 46 men, 31 (67.39%) had ankylosing spondylitis. In addition, 9 patients (7.92%) had psoriasis and 8 (8.91%) had Crohn’s disease (Table 1). With regard to LTBI screening, 53 patients (52.48%) had a TST induration > 10 mm. TST induration ranged from 5 mm to 40 mm, with a mean TST induration of 12.06 ± 0.60 mm. The mean TST induration was 7.31 ± 0.28 mm in the patients with a TST induration of 5-10 mm and 16.36 ± 0.70 mm in those with a TST induration ≥ 10 mm (Table 1).
Table 1. Demographic, epidemiological, and clinical characteristics of the study patients.a .
| Variable | Total | Biologic agents | Immunosuppressants | p* |
|---|---|---|---|---|
| (N = 101) | (n = 79) | (n = 22) | ||
| Sex | ||||
| Male | 46 (45.54) | 40 (86.96) | 6 (13.04) | 0.051 |
| Female | 55 (54.46) | 39 (70.91) | 16 (29.09) | |
| Age, years | ||||
| Male | 45.40 ± 2.13 | 0.002 | ||
| Female | 53.16 ± 1.76 | |||
| Age group, years | ||||
| < 40 | 25 (24.75) | 21 (84.00) | 4 (16.00) | |
| 40-59 | 52 (51.49) | 40 (76.92) | 12 (23.08) | |
| ≥ 60 | 24 (23.76) | 18 (75.00) | 6 (25.00) | |
| BCG vaccination | ||||
| Yes | 83 (82.18) | 64 (81.01) | 19 (86.36) | 0.075 |
| No | 18 (17.82) | 15 (18.99) | 3 (13.64) | |
| History of contact with a tuberculosis case | ||||
| Yes | 39 (38.61) | 30 (76.92) | 9 (23.08) | 0.803 |
| No | 62 (61.39) | 49 (79.03) | 13 (20.97) | |
| History of tuberculosis treatment | ||||
| Yes | 10 (9.90) | 9 (90.00) | 1 (10.00) | 0.068 |
| No | 91 (90.10) | 70 (76.92) | 21 (23.08) | |
| Respiratory symptoms | ||||
| Yes | 18 (17.82) | 15 (83.33) | 3 (16.67) | 0.075 |
| No | 83 (82.18) | 64 (77.11) | 19 (22.89) | |
| Chest X-ray | ||||
| Abnormal | 36 (35. 64) | 27 (75.0) | 9 (25.0) | 0.560 |
| Normal | 65 (64.36) | 52 (80.0) | 13 (20.0) | |
| Tuberculin skin test induration, mm | ||||
| 5-10 | 48 (47.52) | 38 (48.10) | 10 (45.45) | |
| >10 | 53 (52.48) | 41 (51.90) | 12 (54.55) | |
| Tuberculin skin test induration, mm | ||||
| 5-10 | 7.31 ± 0.28 | 0.0001 | ||
| > 10 | 16.36 ± 0.70 | |||
| Clinical diagnosis | ||||
| Rheumatic disease | 84 (83.17) | 64 (76.19) | 20 (23.81) | |
| Skin disease | 9 (7.92) | 9 (100.0) | 0 (0.00) | |
| Gastrointestinal disease | 8 (8.91) | 6 (75.00) | 2 (25.0) | |
Values expressed as n (%) or mean ± SD. *Two-tailed Pearson’s chi-square test with Yates’ correction. The two-tailed Fisher’s exact test was used for contingency tables with fewer than five cells. The Student’s t-test was used for means and SDs.
With regard to epidemiological risk factors, 39 patients (38.61%) reported a history of contact with a tuberculosis case. In addition, 10 (9.90%) reported having previously received tuberculosis treatment and 18 (17.82%) reported respiratory symptoms. Furthermore, 83 (82.18%) had been vaccinated with BCG. Moreover, 36 (35.64%) had chest X-ray findings consistent with LTBI (Table 1).
With regard to the number of patients receiving treatment with immunomodulators, immunosuppressants, or a combination of the two when the TST was performed, 46 (45.54%) were being treated with ≥ 10 mg of weekly methotrexate (mean weekly dose, 12.837 ± 0.614 mg) and 21 (20.79%) were being treated with ≥ 15 mg of daily prednisone (mean daily dose, 12.540 ± 0.949 mg).
Most (n = 91; 90.09%) of the patients received IPT for 6 months. Of those 91 patients, 86 (94.50%) completed the treatment. Ten patients (9.90%) received IPT for 9 months. Therefore, 96 patients (95.05%) completed the prophylactic treatment. Of the remaining 5 patients, 4 (3.96%) discontinued IPT because of adverse reactions to isoniazid and 1 (0.99%) abandoned IPT (Table 2). Of the patients who received IPT, 84 (83.17%) had no adverse reactions to isoniazid. Of those, 83 (98.81%) completed the prophylactic treatment (p = 0.002). Only 1 patient (1.19%) abandoned IPT, at 2 months of treatment, because the decision to use biologic agents in that patient had to be reconsidered.
Table 2. Variables associated with isoniazid preventive therapy (N = 101).a .
| Variable | Total | Completed the treatment | Did not complete the treatment | p |
|---|---|---|---|---|
| Duration of IPT | ||||
| 6 months | 91 (90.09%) | 86 (94.50) | 5 (5.49) | |
| 9 months | 10 (9.90 %) | 10 (100.0) | 0 (0.00) | |
| ≥ 1 month of IPT before treatment with | ||||
| Biologic agents | 79 (78.21) | 78 (98.73) | 1 (2.27) | 0.001 |
| Other immunosuppressants | 22 (21.78) | 18 (81.82) | 4 (18.28) | |
| Adverse reactions to isoniazid | ||||
| Yes | 17 (16.83) | 13 (76.47) | 4 (23.53) | 0.002 |
| No | 84 (83.17) | 83 (98.81) | 1 (1.19) | |
| ALT levels at 30 days of IPT | ||||
| Normal | 96 (95.05) | 93 (96.88) | 3 (3.12) | 0.001 |
| Abnormal | 5 (4.95) | 3 (60.0) | 2 (40.0) | |
| AST levels at 30 days of IPT | ||||
| Normal | 96 (95.05) | 93 (96.88) | 3 (3.12) | 0.001 |
| Abnormal | 5 (4.95) | 3 (60.0) | 2 (40.0) | |
IPT: isoniazid preventive therapy; ALT: alanine aminotransferase; and AST: aspartate aminotransferase. aValues expressed as n (%). *Fisher’s exact test.
In 96 patients (95.05%), liver transaminase levels were found to be normal at 30 days of IPT. Of those 96 patients, 93 (96.88%) completed the treatment. Transient elevation of liver transaminases was observed in 5 patients (4.95%). Hepatotoxicity was observed in only 3 patients (2.97%). Of those, only 2 did not complete the treatment (p = 0.001; Table 2). Table 3 shows the results of our multivariate analysis of factors associated with completion of IPT.
Table 3. Multivariate analysis of factors associated with completion of isoniazid preventive therapy.
| Variable | OR (95% CI) | p |
|---|---|---|
| Drug treatment | ||
| Biologic agents | 17.3 (1.82-164.5) | 0.0130 |
| Other immunosuppressants (prednisone) | ||
| Treatment with methotrexate | ||
| Yes | 1.63 (0.26-10.1) | 0.6013 |
| No | ||
| Adverse reactions | ||
| Yes | 25.53 (2.64-246.7) | 0.0051 |
| No | ||
| ALT levels | ||
| Normal | 20.60 (2.46-173.3) | 0.0053 |
| Abnormal | ||
| ALT: alanine aminotransferase. | ||
Infliximab was the most widely used biologic agent, in 46 patients (58.23%), followed by etanercept, in 15 (18.99%), adalimumab, in 12 (15.19%), golimumab, in 1 (1.27%), and bevacizumab, in 1 (1.27%). Four patients (5.05%) used biologic agents having different immune targets, including rituximab, tocilizumab, abatacept, and ustekinumab. The mean follow-up period was 3.08 ± 0.13 years (range, 6 months to 4 years). At the end of the study period, 68 patients (86.07%) had been receiving treatment with biologic agents for more than 1 year, and 46 (58.23%) received treatment for 4 years.
By the end of the study, active tuberculosis had been diagnosed in 5 patients receiving treatment with biologic agents and in 1 patient receiving treatment with immunomodulatory or immunosuppressant agents. Of those 6 patients, 5 were diagnosed with pulmonary tuberculosis and 1 was diagnosed with extrapulmonary tuberculosis. All 6 were cured. The mean duration of treatment with anti-TNF biologic agents before the onset of symptoms of active tuberculosis was 24.0 ± 8.09 months (range, 3-16 weeks). Of the 5 patients receiving treatment with biologic agents, 4 had rheumatic disease, 4 were using infliximab, 1 was using etanercept, and all tested positive for LTBI (Figure 2).
Figure 2. Cases of active tuberculosis (TB) during treatment with biologic agents.

The relative risk of developing active tuberculosis was 1.39 times higher (95% CI: 0.17-11.3) in patients treated with biologic agents (78.22%) than in those not treated with biologic agents (21.78%). In addition, it was 3.15 times higher (95% CI: 0.38-25.9) in patients using prednisone (61.38%) than in those not using prednisone (38.61%). Furthermore, it was 7.95 times higher (95% CI: 0.96-65.50) in patients with a history of contact with a tuberculosis case (38.61%) than in those without such a history (61.39%; Table 4).
Table 4. Relative risk of developing tuberculosis.
| Variable | n (%) | Cases of active tuberculosis, n | RR | 95% CI | p |
|---|---|---|---|---|---|
| Biologic agent use | 79 (78.22) | 5 | 1.39 | 0.17-11.3 | 0.7567 |
| Immunosuppressant use | 22 (21.78) | 1 | |||
| Methotrexate use | 52 (51.48) | 2 | 0.47 | 0.09-2.46 | 0.3719 |
| No methotrexate use | 49 (48.51) | 4 | |||
| Prednisone use | 62 (61.38) | 5 | 3.15 | 0.38-25.9 | 0.2870 |
| No prednisone use | 39 (38.61) | 1 | |||
| History of contact with tuberculosis | 39 (38.61) | 5 | 7.95 | 0.96- 65.5 | 0.0541 |
| No history of contact with tuberculosis | 62 (61.39) | 1 | |||
| Abnormal chest X-ray | 36 (35.64) | 2 | 0.90 | 0.17-4.69 | 0.9032 |
| Normal chest X-ray | 65 (64.36) | 4 | |||
| TST induration = 5-10 mm | 48 (47.52) | 2 | 0.55 | 0.11-2.88 | 0.4809 |
| TST induration ≥ 10 mm | 53 (52.48) | 4 |
RR: relative risk; and TST: tuberculin skin test.
DISCUSSION
Treatment with TNF inhibitors has been reported to increase the risk of active tuberculosis by 1.6- to 25.1-fold. 8 In our study, the risk of developing active tuberculosis was 1.39 times higher in patients who received treatment with biologic agents than in those who did not. Because of this risk, special emphasis has been given to the importance of LTBI treatment in endemic countries, such as Brazil. However, there is insufficient evidence for the efficacy of different prophylactic treatment regimens for LTBI. Most developing countries recommend 9 months of IPT (the 9H regimen). However, alternative regimens such as 3 months of treatment with rifampin and isoniazid (the 3RH regimen) and 4 months of treatment with rifampin result in less hepatotoxicity and better adherence to treatment than does the 9H regimen. Nevertheless, there is little evidence supporting their clinical efficacy. The 3RH regimen has been shown to be equivalent to 6 months of isoniazid treatment (the 6H regimen) and the 9H regimen in terms of efficacy and safety, its use being recommended by the World Health Organization. 9
Lee et al. 8 reported that, despite prophylactic treatment, 5 patients developed active tuberculosis during follow-up; however, all 5 were treated with the 9H regimen, which was related to isoniazid resistance. In our study, 5 patients developed active tuberculosis despite IPT with the 6H regimen. Coskunol et al. 10 found that 5 patients receiving anti-TNF-α therapy developed active tuberculosis; of those, 3 had ulcerative colitis and 2 had ankylosing spondylitis. All 5 were using infliximab. Of the 5 patients, 4 were diagnosed with pulmonary tuberculosis and 1 was diagnosed with extrapulmonary tuberculosis. The time from initiation of anti-TNF-α therapy to tuberculosis infection was 6 months, in 3 patients; 15 months, in 1; and 24 months, in 1. In our study, the time from initiation of anti-TNF-α therapy to tuberculosis infection ranged from 10 months to 48 months. These results show that tuberculosis cannot be completely prevented during treatment with TNF inhibitors, even in patients receiving preventive treatment for LTBI. Therefore, the possibility of tuberculosis should always be considered. 4
In a classic study published in 2001, Keane et al. 11 examined the role of treatment with biologic agents in the occurrence of opportunistic infections by analyzing cases of tuberculosis after treatment with infliximab. During the study period, there were 70 reported cases of tuberculosis after treatment with infliximab, for a median of 12 weeks. In a study of data from a Brazilian national registry of biologic agent use in patients with rheumatoid arthritis, 750 patients treated with biologic agents were compared with 287 controls receiving treatment with immunosuppressive agents. 12 There were 3 confirmed cases of tuberculosis in the group of patients treated with biologic agents, a finding that underscores the importance of continuously monitoring biologic agent use. Infliximab was the most commonly used TNF inhibitor, 12 a finding that is consistent with ours.
According to Lee et al., 13 prophylactic treatment significantly reduces the risk of tuberculosis, with an incidence rate of 0.33. Therefore, LTBI treatment significantly reduces the risk of developing active tuberculosis and tuberculosis transmission in the community. This means that LTBI diagnosis and treatment constitute a strategy to eliminate active tuberculosis, preventing new cases. Long-term studies have shown that IPT for 3, 6, and 12 months reduces the risk of developing active tuberculosis by 21%, 65%, and 75%, respectively. 14 Treatment adherence has been recognized as a critical parameter, treatment efficacy being highest when at least 80% of doses are administered. 14
According to Tost et al., 15 a tuberculosis treatment regimen is useful when more than 95% of patients are cured and less than 5% develop severe intolerance. In our study, patients received IPT for 6 months, with a high treatment completion rate (> 90.0%) and a low treatment abandonment rate (0.99%). With regard to the safety of isoniazid, adverse effects were uncommon; transient adverse effects were reported in only 16.83% of the sample, a finding that is consistent with those of another study, 16 and hepatotoxicity was observed in only 2.97%. These data are promising because the study patients had immune-mediated chronic inflammatory diseases and were receiving treatment with immunosuppressive or immunomodulatory agents, as well as having associated comorbidities.
Footnotes
Financial support: None.
Study carried out at the Universidade Federal do Ceará - UFC - Fortaleza (CE) Brasil.
REFERENCES
- 1.De Oliveira Uehara SN, Emori CT, Perez RM, Mendes-Correa MC, de Souza Paiva Ferreira A, de Castro Amaral Feldner AC, et al. High incidence of tuberculosis in patients treated for hepatitis C chronic infection. Braz J infect Dis. 2016;20(2):205–209. doi: 10.1016/j.bjid.2015.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization . Geneva: World Health Organization; 2017. http://www.who.int/tb/publications/global_report/en [Google Scholar]
- 3.Brasil Indicadores prioritários para o monitoramento do Plano Nacional pelo Fim da Tuberculose como Problema de Saúde Pública no Brasil. Bol Epidemiol. 2017;48(8):1–10. [Google Scholar]
- 4.Shim TS. Diagnosis and Treatment of Latent Tuberculosis Infection due to Initiation of Anti-TNF Therapy. Tuberc Respir Dis (Seoul) 2014;76(6):261–268. doi: 10.4046/trd.2014.76.6.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bonfiglioli KR, Ribeiro CM, Moraes JC, Saad CG, Souza FH, Calich AL. Screening in rheumatoid arthritis patients prior to anti-TNF treatment in an endemic area. Int J Tuberc Lung Dis. 2014;18(8):905–911. doi: 10.5588/ijtld.13.0755. [DOI] [PubMed] [Google Scholar]
- 6.Conde MB, Melo FA, Marques AM, Cardoso NC, Pinheiro VG, Dalcin Pde T. III Brazilian Thoracic Association Guidelines on tuberculosis. J Bras Pneumol. 2009;35(10):1018–1048. doi: 10.1590/S1806-37132009001000011. [DOI] [PubMed] [Google Scholar]
- 7.Brasil . Manual de Recomendações para o Controle da Tuberculose no Brasil. Série A. Normas e Manuais Técnicos. Brasília: Ministério da Saúde; 2011. [Google Scholar]
- 8.Lee EH, Kang YA, Leem AY, Park MS, Kim YS, Kim SK. Active Tuberculosis Incidence and Characteristics in Patients Treated with Tumor Necrosis Factor Antagonists According to Latent Tuberculosis Infection. Sci Rep. 2017;7(1):6473–6473. doi: 10.1038/s41598-017-06899-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim HW, Kim JS. Treatment of Latent Tuberculosis Infection and Its Clinical Efficacy. Tuberc Respir Dis (Seoul) 2018;81(1):6–12. doi: 10.4046/trd.2017.0052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.l Coskunol, Baysak A, Dalli A, Uluorman F, Can G. Anti-TNF-alpha therapy in patients with latent tuberculosis incidence. Eur Respir J. 2015;46:PA2969–PA2969. doi: 10.1183/13993003.congress-2015.PA2969. [DOI] [Google Scholar]
- 11.Keane J, Gershon S, Wise RP, Mirabile-Levens E, Kasznica J, Schwieterman WD. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med. 2001;345(15):1098–1104. doi: 10.1056/NEJMoa011110. [DOI] [PubMed] [Google Scholar]
- 12.Titton DC, Silveira IG, Louzada-Junior P, Hayata AL, Carvalho HM, Ranza R. Brazilian biologic registry BiobadaBrasil implementation process and preliminary results. Rev Bras Reumatol. 2011;51(2):152–160. doi: 10.1590/S0482-50042011000200005. [DOI] [PubMed] [Google Scholar]
- 13.Lee J, Kim E, Jang EJ, Lee CH, Lee EY, Im JP. Efficacy of Treatment for Latent Tuberculosis in Patients Undergoing Treatment with a Tumor Necrosis Factor Antagonist. Ann Am Thorac Soc. 2017;14(5):690–697. doi: 10.1513/AnnalsATS.201608-647OC. [DOI] [PubMed] [Google Scholar]
- 14.Duarte R, Villar M, Carvalho A. Latent tuberculosis infection treatment Current recommendations. Rev Port Pneumol. 2010;16(5):809–814. doi: 10.1016/S0873-2159(15)30073-8. [DOI] [PubMed] [Google Scholar]
- 15.Tost JR, Vidal R, Maldonado J, Caylà J. Effectiveness and tolerance of antituberculosis treatment regimens without isoniazid and rifampicin analysis of 85 cases. Bronconeumol. 2008;44(9):478–483. doi: 10.1016/S1579-2129(08)60086-5. [DOI] [PubMed] [Google Scholar]
- 16.Souza CT, Hökerberg YH, Pacheco SJ, Rolla VC, Passos SR. Effectiveness and safety of isoniazid chemoprophylaxis for HIV-1 infected patients from Rio de Janeiro. Mem Inst Oswaldo Cruz. 2009;104(3):462–467. doi: 10.1590/S0074-02762009000300011. [DOI] [PubMed] [Google Scholar]


