Abstract
The groups of plant flavonoid metabolites termed anthocyanins and proanthocyanins (PA) are responsible for pigmentation in seeds, flowers and fruits. Anthocyanins and PAs are produced by a pathway of enzymes which are transcriptionally regulated by transcription factors (TFs) that form the MYB-bHLH-WD40 (MBW) complex. In this study, transcriptomic analysis of purple-pigmented kiwifruit skin and flesh tissues identified MYBC1, from subgroup 5 of the R2R3 MYB family, and WRKY44 (highly similar to Arabidopsis TTG2) as candidate activators of the anthocyanin pathway. Transient over-expression of MYBC1 and WRKY44 induced anthocyanin accumulation in tobacco leaves. Dual luciferase promoter activation assays revealed that both MYBC1 and WRKY44 were able to strongly activate the promoters of the kiwifruit F3′H and F3′5′H genes. These enzymes are branch points of the pathway which specifies the type of anthocyanin accumulated. Stable over-expression of MYBC1 and WRKY44 in kiwifruit calli activated the expression of F3′5′H and PA-related biosynthetic genes as well as increasing levels of PAs. These results suggest that while previously characterised anthocyanin activator MYBs regulate the overall anthocyanin biosynthesis pathway, the PA-related TFs, MYBC1 and WRKY44, more specifically regulate key branch points. This adds a layer of regulatory control that potentially balances anthocyanin and PA levels.
Subject terms: Molecular biology, Plant sciences
Introduction
Anthocyanins are a group within the flavonoid family of plant secondary metabolites that determine the colour of flowers and plant organs, as well as indicator for ripeness and quality in fruit1,2. Flavonoids derive from phenylalanine via the general phenylpropanoid pathway which leads to different pathway branches responsible for lignins, stilbenes, condensed tannins (proanthocyanin) and anthocyanins3. Anthocyanin is produced by the biosynthetic pathway consisting of the commonly termed early biosynthetic enzymes, chalcone synthase (CHS), chalcone isomerase (CHI), flavanone 3-hydroxylase (F3H), flavonoid 3′-hydroxylase (F3′H), flavonoid 3′,5′-hydroxylase (F3′5′H) and the late biosynthetic enzymes dihydroflavonol 4-reductase (DFR), leucoanthocyanidin dioxygenase (LDOX), and flavonoid-3-glucosyltransferase (F3GT)2. Within the flavonoid family, proanthocyanins (PAs), also known as condensed tannins, are oligomers of epicatechins and catechins that are usually accumulated in the seed coats of many plants, and are well studied in the model plants Medicago truncatala and Arabidopsis4,5. The enzyme flavonol synthase (FLS) converts dihydroflavonol from the anthocyanin pathway into flavonols. The downstream enzymes leucoanthocyanidin reductase (LAR) and anthocyanidin reductase (ANR) are responsible for the conversion of anthocyanidin to catechin and epicatechin6,7. Parts of the anthocyanin and PA pathways overlap, as enzymes in the respective pathways utilise and potentially compete for the same intermediate substrates. The pathway direction is determined at the F3′H and F3′5′H branch points, two enzymes which hydroxylate the 3′, and 3′ and 5′ position of the B-ring in the carbon backbone, respectively6,8. The roles of F3′H and F3′5′H are critical as they determine the hydroxylation pattern of the anthocyanin aglycone, resulting in the accumulation of cyanidin and delphinidin-based anthocyanins, as well as acting as branch points that provide intermediate substrates for the PA pathway. Dihydroflavonols produced by F3′H and F3′5′H can be converted to flavonols by FLS or leucoanthocyanins by DFR, which can then be converted to the anthocyanidin aglycone by LDOX or flavanols by LAR and ANR. The substrates from the anthocyanin pathway can be intercepted by PA enzymes and diverted into producing PA instead of anthocyanin, made available by the hydroxylation of the substrate by F3′H and F3′5′H.
In addition to the biosynthesis of PAs and anthocyanins being inter-linked, both pathways are regulated by MYB-bHLH-WD40 transcription factor (TF) complexes. MYB TFs can be classified into subgroups and are responsible for many important traits in fruits and plants9. The MYB TFs from subgroup 6 have been implicated in the general activation of the anthocyanin pathway, such as Arabidopsis AtPAP1, apple MdMYB10, Petunia PH4, strawberry FaMYB10, grape VvMYBA1, kiwifruit MYB10/MYB110 and potato StMYBA1 and AN110–19. Arabidopsis MYB123/TT2, apple MYB12, grape VvMYBPA2, peach MYB7, strawberry MYB9/MYB11 and persimmon DkMYB4 from subgroup 5 are involved in the biosynthesis of PA5,20–24. In addition, Arabidopsis MYB111/11/12, VvMYBF1 from grape, apple MdMYB22 and tomato SlMYB12 from subgroup 7 are implicated in elevating flavonol biosynthesis20,25–27. In contrast, MYB TFs from subgroup 4 are repressors of the flavonoid pathway such as Arabidopsis MYB3 and MYB4, which repress PA synthesis; FaMYB1, which represses anthocyanin and flavonol biosynthesis in strawberry; VvMYB4, which represses anthocyanin biosynthesis in grapes; and PpMYB18, which balances anthocyanin and proanthocyanin accumulation in peach21,28–30.
Recent research has indicated that some R2R3 MYBs from subgroup 5 may also contribute to the regulation of both anthocyanin and PA biosynthesis. Grape VvMYB5b encodes for a R2R3 MYB TF that clusters with PA-associated MYBs but was confirmed to activate the promoters of genes that encode enzymes of both the anthocyanin and PA pathways such as VvLAR1, VvANS, VvANR and VvF3′5′H31. Over-expression of VvMYB5b in tobacco induced accumulation of anthocyanin and PA as a result of increased expression of the tobacco versions of these genes. In poplar, MYB6 encodes for a R2R3 MYB TF that is homologous to VvMYB5, and regulates anthocyanin and PA biosynthesis as well as lignin biosynthesis, a side branch of the general phenylpropanoid pathway32. In Freesia hybrida, the R2R3 MYB TF, FhMYB5, clusters with VvMYB5b and MYBs associated with PA regulators and not with the anthocyanin-related subgroup 633. The spatio-temporal expression of FhMYB5 correlates with both anthocyanin and PA accumulation, and its expression significantly up-regulates the expression of F3′H, F3′5′H, DFR, LDOX, in the presence of bHLH partners. In tea, CsMYB5a and CsMYB5e demonstrated the ability to regulate the accumulation of both anthocyanin and PA by activating the associated genes34.
The regulation of anthocyanin and PA also involves the WRKY TF family. Arabidopsis TTG2 encodes for a WRKY TF that participates in trichome development and controls the seed coat tannins by regulating the expression of the vacuolar transporter of glycosylated epicatechin35. A WRKY TF from tea, CsWRKY44, is proposed to be involved in the regulation of catechin production36. In apple, MdWRKY11 promoted the accumulation of flavonoids and anthocyanins by regulating the MdMYB10 and MdHY537,38. In petunia flower petals, the gene PH3 encodes for a WRKY TF that is highly similar to Arabidopsis TTG2, and regulates vacuolar acidification by binding to the MBW complex that transcriptionally activates a vacuolar ATPase39. Further investigation has revealed that the grape VvWRKY26, a homologue to petunia PH3 and Arabidopsis TTG2, is recruited by the VvMYB5a to form a MBW-WRKY complex and enhances the transcription of a set of target genes that are involved in vacuolar hyper-acidification40. The VvMYB5 gene competes with the anthocyanin activator VvMYBA in the MBW complex and reduces its ability to promote anthocyanin accumulation. The recruitment of VvWRKY26 enhances the transcriptional activation of VvMYB5 towards the promoters of genes that are involved in vacuolar hyper-acidification. These studies suggest that WRKY TFs are potentially involved in anthocyanin and PA biosynthesis, via the interaction with the MBW complex.
Kiwifruit (Actinidia sp.) comprises over 70 species with fruits that are distinctive due to differences in colour and texture of both skin and flesh41. In anthocyanin-accumulating kiwifruit, two subgroup 6 MYBs, MYB10 (also called MYBF110 and MYB75, sharing 99.5% identity) and MYB110, activate the core genes of the anthocyanin pathway such as CHS, DFR, F3GT and LDOX16–19. Recently, a kiwifruit MYB, MYB123, which is similar to Arabidopsis TT2 in subgroup 5, demonstrated tissue-specific anthocyanin biosynthesis in the red-centred Actinidia chinensis by activating core genes such as AcANS and AcF3GT142. In addition it was found that the microRNA miR858 targets the RNA of another subgroup 5 gene, MYBC1, in red-coloured A. arguta43. In the purple-skinned and purple-fleshed kiwifruit, MYB110 is responsible for the activation of core kiwifruit anthocyanin pathway but the regulation of the branch points F3′H and F3′5′H, which determine the types of anthocyanins accumulated, has not been elucidated19. Therefore, it was hypothesised that MYB110 may not be the sole regulator for anthocyanin pathway in purple kiwifruit. In order to address this gap in this study, transcriptomic analysis of purple-skinned and purple-fleshed kiwifruit was performed and revealed that MYBC1 and WRKY44 are potential regulators of the anthocyanin pathway, in addition to the previously characterised MYB110. Functional characterisation confirmed the activating roles of MYBC1 and WRKY44 on anthocyanin production, particularly the F3′H and F3′5′H branch points, adding another layer of regulatory control that is shared between anthocyanin and PA biosynthesis.
Results
Transcriptome sequencing of A. purpurea, MaMe Red and MaMe Yellow
The transcriptomic data from purple kiwifruit A. purpurea, MaMe Red and MaMe Yellow at three developmental stages were obtained and approximately 80% of the reads were uniquely mapped to the manually annotated A. chinensis genome44 (Supplementary Table 1). Principal component analysis (PCA) of the transcriptomic data showed that MaMe Red and MaMe Yellow grouped together according to tissue type and developmental stage, and separated from A. purpurea (Supplementary Fig. 1A). The two principal components explained 64% of the total variation from the 54 kiwifruit samples including three biological replicates for skin and flesh tissues sampled at mature green stage, colour change stage, and ripe stage for three kiwifruit species. The skin and flesh tissue were significantly different and separated from each other in A. purpurea, MaMe Red and MaMe Yellow (Supplementary Fig. 1B–D).
Identification of differentially expressed genes (DEGs) involved in flavonoid biosynthesis pathway
Skin tissue of MaMe Red was compared to that of MaMe Yellow at mature green stage, colour stage, and ripe stage to identify DEGs that may be involved in the flavonoid and anthocyanin pathways. A total of 152 genes (Supplementary Table 2) were differentially expressed more than 2 Log2 fold-change (Fig. 1A) between MaMe Red and Yellow, when all three maturity stages were compared. When the same comparisons were made for fruit flesh, there were 141 DEGs (Supplementary Table 3) that showed differential expression greater than 2 Log2 fold-change between the flesh of MaMe Red and MaMe Yellow (Fig. 1B). When overlapping the DEGs that were common between skin and flesh and present in all three maturity stages, there were 49 genes predicted to encode for biosynthetic enzymes that are potentially involved in the flavonoid and anthocyanin biosynthesis (Table 1). In addition, a shortlist of 9 gene models (skin) and 27 gene models (flesh) encoding TFs from the MYB, bHLH, homeobox and NAC families were potential flavonoid biosynthesis regulators (Table 2). The reported bHLH partner of the anthocyanin activating MYBs, termed either bHLH5 or bHLH42 (Acc19563.1)42,45, was not differentially expressed at higher than the 2 Log2 fold-change cut-off.
Figure 1.
Numbers of differentially expressed genes greater than 2 Log2 fold-change from comparing the three developmental stages: mature green, colour change, and ripe. (A) Comparison of gene expression changes in the skin between MaMe Red and MaMe Yellow. (B) Comparison of gene expression changes in the flesh between MaMe Red and Yellow.
Table 1.
Differentially expressed genes (DEGs) encoding biosynthetic enzymes potentially involved in the flavonoid and anthocyanin pathway obtained from the comparison between MaMe Red and MaMe Yellow skin and flesh between mature green (MG), colour change (CC), and ripe (RP) stage.
| MG | CC | RP | Base mean | ||
|---|---|---|---|---|---|
| Skin comparison | |||||
| Acc00260.1 | Chalcone synthase 1 | 2.7 | 5.3 | 5.1 | 3,372 |
| Acc02004.1 | Chalcone synthase 1 | 2.4 | 5.6 | 6.1 | 4,397 |
| Acc08970.1 | Chalcone synthase 1 | 2.9 | 3.6 | 3.1 | 1,990 |
| Acc13879.1 | Anthocyanidin 3-O-glucoside 2″-O-glucosyltransferase | 2.9 | 2.6 | 3.9 | 260 |
| Acc16762.1 | Leucoanthocyanidin dioxygenase (LDOX) | 3 | 7.4 | 9.7 | 2,947 |
| Acc28876.1 | Leucoanthocyanidin dioxygenase (LDOX) | 3.2 | 8.2 | 11.3 | 11,830 |
| Acc03638.1 | Chalcone–flavonone isomerase 3 | 0 | 3.2 | 3.8 | 4,242 |
| Acc03848.1 | Chalcone–flavonone isomerase | 0.2 | 3 | 3.3 | 1,578 |
| Acc11493.1 | Flavonol synthase/flavanone 3-hydroxylase (FLS) | 1.1 | 5.6 | 4.6 | 36 |
| Acc20131.1 | Kaempferol 3-O-beta-d-galactosyltransferase | 1.8 | 8.4 | 9.8 | 4,692 |
| Acc20132.1 | Anthocyanidin 3-O-glucosyltransferase 2 | 0.7 | 8.5 | 11.2 | 77 |
| Acc24966.1 | Chalcone synthase 2 | 1.9 | 6.4 | 5.9 | 3,651 |
| Acc27670.1 | Chalcone–flavonone isomerase | 0 | 2.6 | 2.5 | 214 |
| Acc28896.1 | Flavonoid 3′,5′-hydroxylase 1 (F3′5′H) | − 1 | 2.3 | 3.5 | 65 |
| Acc32899.1 | Leucoanthocyanidin reductase | 0.5 | 3 | 3.3 | 189 |
| Acc20257.1 | Anthocyanidin 5,3-O-glucosyltransferase | 8.9 | 3.2 | 5.2 | 31 |
| Acc19353.1 | Dihydroflavonol 4-reductase (DFR) | 0.6 | 3.6 | 3.2 | 121 |
| Acc26615.1 | Leucoanthocyanidin reductase | − 0.4 | − 2.2 | − 5.6 | 23 |
| Acc26709.1 | Flavonol synthase/flavanone 3-hydroxylase | − 1.9 | − 2.5 | − 3.1 | 416 |
| Acc29470.1 | Isoflavone reductase homolog | 3.7 | 10.7 | 12.4 | 117 |
| Acc32390.1 | Flavonoid 3′,5′-hydroxylase 2 (F3′5′H) | 0.7 | 3.1 | 2.8 | 418 |
| Flesh comparison | |||||
| Acc00260.1 | Chalcone synthase 1 | 4 | 6.3 | 4.1 | 3,372 |
| Acc02004.1 | Chalcone synthase 1 | 3.7 | 5.1 | 5.2 | 4,397 |
| Acc08970.1 | Chalcone synthase 1 | 3.4 | 4.7 | 2.2 | 1,990 |
| Acc16762.1 | Leucoanthocyanidin dioxygenase (LDOX) | 3.8 | 7.7 | 8.9 | 2,947 |
| Acc20131.1 | Kaempferol 3-O-beta-d-galactosyltransferase | 5 | 7.1 | 5.9 | 4,692 |
| Acc20132.1 | Anthocyanidin 3-O-glucosyltransferase 2 | 6.5 | 8.1 | 7.2 | 77 |
| Acc24966.1 | Chalcone synthase 2 | 4.3 | 7.6 | 7.8 | 3,651 |
| Acc28876.1 | Leucoanthocyanidin dioxygenase (LDOX) | 3 | 6.2 | 7.5 | 11,830 |
| Acc29052.1 | Isoflavone reductase homolog | 5.2 | 8.7 | 12 | 1,645 |
| Acc32390.1 | Flavonoid 3′,5′-hydroxylase 2 (F3′5′H) | 4.1 | 6.3 | 2.3 | 417 |
| Acc01005.1 | Dihydroflavonol 4-reductase (DFR) | 0.2 | 4 | 2.5 | 545 |
| Acc03638.1 | Chalcone–flavonone isomerase 3 | 1 | 2.1 | 2.8 | 4,242 |
| Acc03848.1 | Chalcone–flavonone isomerase | 1.7 | 4 | 3.2 | 1,578 |
| Acc14022.1 | Leucoanthocyanidin reductase | 0.4 | 3.7 | 2.9 | 595 |
| Acc18331.1 | Flavonoid 3′-monooxygenase | 1.4 | 4.1 | 5.1 | 408 |
| Acc19353.1 | Dihydroflavonol 4-reductase (DFR) | 0.8 | 5.5 | 2.6 | 121 |
| Acc26709.1 | Flavonol synthase/flavanone 3-hydroxylase | − 1.8 | − 2.4 | − 2.5 | 416 |
| Acc29470.1 | Isoflavone reductase homolog | 2.5 | 7.9 | 10.6 | 117 |
| Acc32899.1 | Leucoanthocyanidin reductase | 0.6 | 4 | 3.9 | 188 |
| Acc28896.1 | Flavonoid 3′,5′-hydroxylase 1 (F3′5′H) | 2.7 | 2.9 | 1.6 | 64 |
Base means are shown as an indication of expression.
Table 2.
Differentially expressed genes (DEGs) encoding transcription factors in the MYB, bHLH, homeobox and NAC families that are possibly involved in the flavonoid and anthocyanin pathway obtained from the comparison between MaMe Red and MaMe Yellow skin and flesh between mature green (MG), colour change (CC), and ripe (RP) stage.
| MG | CC | RP | Base mean | ||
|---|---|---|---|---|---|
| Skin comparison | |||||
| Acc06303.1 | Homeobox-leucine zipper protein HOX16 | − 2 | − 3.7 | − 3.6 | 335 |
| Acc10232.1 | Transcription factor AtMYB114, AcMYB110 | 3 | 9.1 | 10.3 | 1,222 |
| Acc21917.1 | Transcription factor UNE10 (AtbHLH16) | 5.5 | 5.6 | 4.4 | 10 |
| Acc07314.1 | bZIP transcription factor 44 | − 0.3 | 2.3 | 2.1 | 226 |
| Acc11572.1 | Dof zinc finger protein DOF1.2 | 1.4 | 3.2 | 3.3 | 323 |
| Acc22908.1 | Transcription factor MYB108 | 1 | − 2.8 | − 4.4 | 267 |
| Acc12965.1 | Anthocyanin regulatory C1 protein, AcMYBC1 | 2.8 | 6.9 | 6.2 | 63 |
| Acc06281.1 | Transcription factor MYB1R1 | − 0.2 | − 2.7 | − 3.1 | 139 |
| Acc10227.1 | Transcription factor AtMYB75, AcMYB210 | − 0.2 | 5.4 | 4.6 | 5 |
| Acc16887.1 | WRKY transcription factor 44 | − 0.1 | 3.4 | 4.2 | 237 |
| Flesh comparison | |||||
| Acc00493.1 | Transcription factor AtMYB114, AcMYB10 | − 0.2 | 2 | 3.9 | 284 |
| Acc10232.1 | Transcription factor AtMYB114 , AcMYB110 | 4 | 9.3 | 11 | 1,222 |
| Acc12264.1 | Dof zinc finger protein DOF1.2 | 2 | 4.5 | 4.2 | 407 |
| Acc12965.1 | Anthocyanin regulatory C1 protein, AcMYBC1 | 7.2 | 8.3 | 8.4 | 63 |
| Acc21466.1 | Transcription factor bHLH149 | 2 | 3.5 | 2.2 | 41 |
| Acc24307.1 | Transcription factor MYB86 (AtMyb4) | − 5.2 | − 3 | − 2.1 | 19 |
| Acc31401.1 | Myb-related protein B (B-Myb) | − 2.7 | − 2.4 | − 3.2 | 40 |
| Acc02780.1 | WRKY transcription factor 70 | − 0.2 | 2.7 | 4.1 | 101 |
| Acc06319.1 | Myb-related protein B (B-Myb) | − 0.1 | − 2.8 | − 3.3 | 55 |
| Acc06321.1 | Homeobox-leucine zipper protein ATHB-13 | 0.5 | − 3.3 | − 2.8 | 83 |
| Acc07314.1 | bZIP transcription factor 44 | 0.1 | 2 | 2.1 | 226 |
| Acc08333.1 | Transcription factor bHLH14 | − 1.9 | − 2.8 | − 2.1 | 12 |
| Acc10227.1 | Transcription factor AtMYB75, MYB210 | 0.1 | 5.3 | 4.3 | 5 |
| Acc10925.1 | Transcription factor MYB86 (AtMyb4) | 0.5 | − 2.9 | − 3.6 | 14 |
| Acc11572.1 | Dof zinc finger protein DOF1.2 | 0 | 2.1 | 2.6 | 323 |
| Acc13277.1 | Myb-related protein 306 | 0 | − 2.9 | − 4.9 | 10 |
| Acc13287.1 | Cyclic dof factor 3 (AtDOF3.3) | 1.2 | − 2.5 | − 2 | 8 |
| Acc14650.1 | NAC domain-containing protein 21/22 | − 0.1 | − 4.6 | − 3.7 | 11 |
| Acc15830.1 | Transcription factor MYB1R1 | 0.4 | − 2.2 | − 2.6 | 1,320 |
| Acc16026.1 | Anthocyanin regulatory C1 protein, MYB12-like | 1.5 | 4 | 4.1 | 7 |
| Acc16887.1 | WRKY transcription factor 44 | 1.8 | 4.8 | 5.2 | 237 |
| Acc19793.1 | WRKY transcription factor 53 | − 0.4 | − 3.2 | − 2.2 | 330 |
| Acc20254.1 | WRKY transcription factor 27 | 1 | − 3.4 | − 2.3 | 24 |
| Acc23052.1 | Myb-related protein 306 | 0 | − 3.7 | − 3.3 | 115 |
| Acc23779.1 | Transcription factor MYB44 | − 1 | − 2.4 | − 2.9 | 143 |
| Acc28877.1 | Transcription factor bHLH120 | − 0.9 | − 4.4 | − 8.8 | 15 |
| Acc30637.1 | Homeobox-leucine zipper protein HAT5 | − 0.3 | − 2.3 | − 2.6 | 266 |
| Acc06303.1 | Homeobox-leucine zipper protein HOX16 | − 3.4 | − 2.6 | − 0.3 | 335 |
| Acc29560.1 | WRKY transcription factor 40 | 2 | 3.1 | 1.2 | 307 |
| Acc24555.1 | bZIP transcription factor 53 | 2.3 | − 0.6 | − 2.6 | 29 |
Base means are shown as an indication of expression.
To validate these observations against an independent genetic background, a comparison of fruit skin and flesh of the related species A. purpurea was made. Comparison between skin and flesh maturity time points revealed 203 DEGs consistently changing during ripening in skin (Fig. 2A, Supplementary Table 4) and 208 DEGs in flesh (Fig. 2B, Supplementary Table 5). Forty gene models and 26 gene models identified in the skin and flesh comparisons of A. purpurea, respectively, were predicted to encode for biosynthetic enzymes in the flavonoid and anthocyanin pathways (Table 3). In the same comparisons, 24 TFs and 94 TFs were identified from the MYB, bHLH, homeobox and NAC families that may be involved in the flavonoid and anthocyanin biosynthesis in skin and flesh of A. purpurea, respectively (Supplementary Table 6).
Figure 2.
Numbers of differentially expressed genes greater than 2 Log2 fold-change from comparing the three developmental stages: mature green, colour change, and ripe. (A) Comparison of gene expression changes in the skin of A. purpurea. (B) Comparison of gene expression changes in the flesh of Actinidia purpurea.
Table 3.
Differentially expressed genes (DEGs) encoding biosynthetic enzymes potentially involved in the flavonoid and anthocyanin pathway obtained from the comparison between mature green (MG), colour change (CC), and ripe (RP) stage in Actinidia purpurea skin and flesh.
| MG-RP | MG-CC | CC-RP | Base mean | ||
|---|---|---|---|---|---|
| Skin comparison | |||||
| Acc00260.1 | Chalcone synthase 1 | − 1.6 | − 3 | 1.5 | 3,372 |
| Acc02004.1 | Chalcone synthase 1 | − 2.7 | − 3.8 | 1.1 | 4,396 |
| Acc03410.1 | UDP-glycosyltransferase 88B1 | 2.4 | 2.3 | 0.1 | 101 |
| Acc03638.1 | Chalcone–flavonone isomerase 3 | 0.8 | − 1.3 | 2 | 4,241 |
| Acc03848.1 | Chalcone–flavonone isomerase | − 1.2 | − 2.1 | 0.9 | 1,577 |
| Acc05968.1 | Putative glycosyltransferase 7 (AtGT7) | 2.3 | 0.7 | 1.6 | 175 |
| Acc08970.1 | Chalcone synthase 1 | − 1.6 | − 3.1 | 1.5 | 1989 |
| Acc12813.1 | Flavonoid 3′-monooxygenase | 4.1 | 4.3 | − 0.1 | 437 |
| Acc13879.1 | Anthocyanidin3-O-glucoside2″-O-GT | − 0.6 | − 3.1 | 2.5 | 260 |
| Acc14022.1 | Leucoanthocyanidin reductase | − 1.9 | − 3.9 | 2 | 594 |
| Acc16762.1 | Leucoanthocyanidin dioxygenase (LDOX) | − 1.3 | − 3.6 | 2.4 | 2,947 |
| Acc18331.1 | Flavonoid 3′-monooxygenase | 4.3 | 1.7 | 2.6 | 408 |
| Acc19922.1 | Flavonoid 3′,5′-hydroxylase 1 (F3′5′H) | 4.7 | 4.4 | 0.3 | 97 |
| Acc20131.1 | Kaempferol 3-O-beta-d-galactosyltransferase | − 1.4 | − 2.9 | 1.5 | 4,691 |
| Acc20132.1 | Anthocyanidin 3-O-glucosyltransferase 2 | − 1.4 | − 2.8 | 1.4 | 77 |
| Acc20478.1 | UDP-glycosyltransferase 73C3 | − 2.3 | − 2.4 | 0 | 1,379 |
| Acc21858.1 | Leucoanthocyanidin reductase | 4.8 | 3.6 | 1.2 | 100 |
| Acc23638.1 | Isoflavone reductase-like protein | − 3.5 | − 2.8 | − 0.7 | 1,013 |
| Acc23730.1 | Putative dihydroflavonol 4-reductase (DFR) | 2 | 1.3 | 0.7 | 231 |
| Acc24966.1 | Chalcone synthase 2 | − 2.6 | − 3.5 | 0.9 | 3,651 |
| Acc27670.1 | Chalcone–flavonone isomerase | − 1.3 | − 2.1 | 0.8 | 214 |
| Acc28876.1 | Leucoanthocyanidin dioxygenase (LDOX) | − 1.3 | − 3.2 | 1.9 | 11,830 |
| Acc28896.1 | Flavonoid 3′,5′-hydroxylase 1 (F3′5′H) | 2.1 | 0.6 | 1.5 | 64 |
| Acc32899.1 | Leucoanthocyanidin reductase | − 1.4 | − 3.6 | 2.2 | 188 |
| Flesh comparison | |||||
| Acc00260.1 | Chalcone synthase 1 | − 2.6 | − 3.9 | 1.3 | 3,372 |
| Acc02004.1 | Chalcone synthase 1 | − 4.5 | − 4.7 | 0.2 | 4,396 |
| Acc02411.1 | UDP-glycosyltransferase 90A1 | 3.6 | 2 | 1.6 | 111 |
| Acc02866.1 | UDP-glycosyltransferase 76F1 | 4.5 | 3.5 | 1 | 172 |
| Acc03410.1 | UDP-glycosyltransferase 88B1 | 3.4 | 3 | 0.5 | 101 |
| Acc06429.1 | UDP-glycosyltransferase 89A2 | − 4.7 | − 4.8 | 0.1 | 3,379 |
| Acc08970.1 | Chalcone synthase 1 | − 3 | − 4.5 | 1.6 | 1,989 |
| Acc12813.1 | Flavonoid 3′-monooxygenase | 6 | 5.3 | 0.7 | 437 |
| Acc14022.1 | Leucoanthocyanidin reductase | − 2.3 | − 3.3 | 1 | 594 |
| Acc16762.1 | Leucoanthocyanidin dioxygenase (LDOX) | − 3.1 | − 3.7 | 0.7 | 2,947 |
| Acc16896.1 | UDP-glycosyltransferase 73E1 | − 2.7 | − 0.7 | − 2 | 243 |
| Acc18331.1 | Flavonoid 3′-monooxygenase | 4.1 | 2.1 | 2 | 408 |
| Acc21858.1 | Leucoanthocyanidin reductase | 3.8 | 3.2 | 0.6 | 100 |
| Acc23638.1 | Isoflavone reductase-like protein | − 2.4 | − 2.2 | − 0.2 | 1,013 |
| Acc24966.1 | Chalcone synthase 2 | − 5.3 | − 5.6 | 0.3 | 3,651 |
| Acc25835.1 | UDP-glucose iridoid glucosyltransferase | 3.2 | 3.9 | − 0.7 | 865 |
| Acc26709.1 | Flavonol synthase/flavanone 3-hydroxylase | 3.2 | 2.1 | 1.1 | 416 |
| Acc32899.1 | Leucoanthocyanidin reductase | − 2.3 | − 3.4 | 1.1 | 189 |
Base means are shown as an indication of expression.
The common DEGs that appeared on all lists between the skin and flesh comparisons of MaMe Red, MaMe Yellow and A. purpurea are gene models encoding CHS (Acc00260.1, Acc02004.1, Acc08970.1, and Acc24966.1), LDOX (Acc16762.1), and LAR (Acc32889.1). Additionally, the two DEGs that are shared in all four lists encodes for the TFs bZIP44 (Acc07314.1) and MYB110 (Acc10232.1). The lack of CHS and F3GT expression has been associated with the lack of anthocyanin accumulation and the TF MYB110 (Acc10232.1) has been shown to be responsible for anthocyanin biosynthesis in these purple kiwifruit species19. The CHS (Acc08970.1), CHI (Acc03638.1), F3′H (Acc12813.1 and Acc18331.1), F3′5′H (Acc32390.1), LDOX (Acc28876.1), flavonoid 3′ glycosyltransferase F3GT1 (anthocyanin glucosyltransferase Acc20132.1) and the TF MYB10 (Acc00493.1) and MYB110 (Acc10232.1) were previously characterised in purple kiwifruit.
Candidate TFs MYBC1 and WRKY44 identified from DEGs
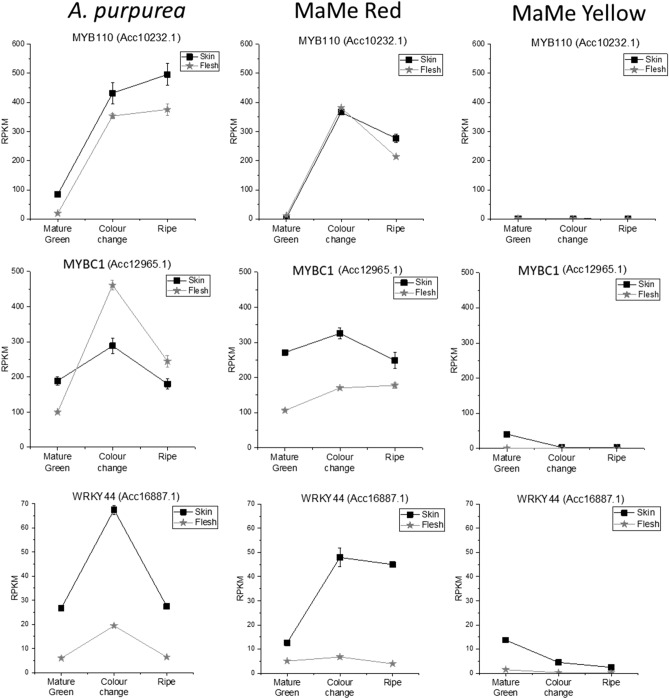
The TF MYB110 (Acc10232.1) was identified in all DEG lists confirming previous studies which have identified its role in elevating anthocyanin biosynthesis16,19,46. MYB10 (Acc00493.1) appears to have a more important role in the kiwifruit species, A. chinensis17, and was identified as a DEG in flesh samples. The MYB TF MYBC1 (Acc12965.1)43 and the WRKY TF termed WRKY44 (Acc16887.1) appeared in all the DEG lists of comparisons between MaMe Red and MaMe Yellow. The expression of MYBC1 exhibited high fold-changes during the colour change and ripe stage compared to the mature green stage in both the skin and flesh (Table 2). MYBC1 was highly expressed during the colour change and ripe stage in the purple coloured A. purpurea and MaMe Red, but barely detected in MaMe Yellow (Fig. 3). Similarly, WRKY44 was highly expressed in the A. purpurea and MaMe Red during the colour change and ripe stage but very lowly expressed in the anthocyanin lacking MaMe Yellow. The expression of WRKY44 exhibited high fold change increases in both the skin and flesh during colour development (Table 2).
Figure 3.
Expression of MYB110 (Acc10232.1), MYBC1 (Acc12965.1) and WRKY44 (Acc16887.1) in the skin and flesh of Actinidia purpurea, MaMe Red, and MaMe Yellow across the three developmental stages. Gene expression was expressed in Reads per Kilobase per Million (RPKM). Data were shown as means ± SEM of three biological replicates.
The expression of MYBC1 and WRKY44 was significantly higher in coloured fruit, as was MYB110, correlating with anthocyanin accumulation. There was some reduction in expression in ripe fruit. The expression of other subgroup 5 MYBs was very low (< 20 RPKM) and the expression patterns did not correlate with the expression of MYB110 or with anthocyanin accumulation (Supplementary Fig. 2). MYBC1 (closest A. chinensis gene model, Acc12965.1) has been implicated in the control of A. arguta anthocyanin levels43. Another TT2-like MYB termed AcMYB123 (closest gene model Acc28234.1) was proposed to regulate the anthocyanin accumulation in the inner pericarp of the red-centred A. chinensis42. In our RNA-seq data, the expression of AcMYB123 (Acc28234.1) and other TT2-like MYBs was barely detected in either skin or flesh (Supplementary Fig. 2). Also, the low expression of other WRKY44-like gene models did not indicate any correlation with anthocyanin accumulation (Supplementary Fig. 3). Therefore, MYBC1 (Acc12965.1) and WRKY44 (Acc16887.1) from purple kiwifruit A. melanandra, A. purpurea, and MaMe Red were selected as candidate genes for functional characterisation.
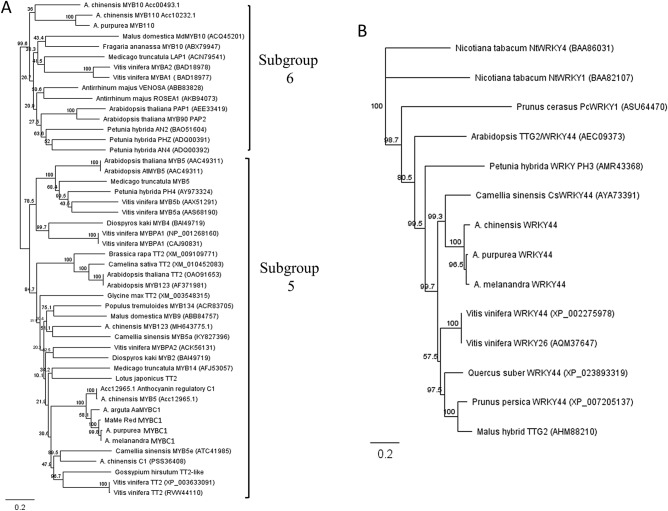
MYBC1 is a subgroup 5 R2R3 MYB and WRKY44 belongs to group I of the WRKY family
Phylogenetic analysis of MYBC1 from A. melanandra (purple-skinned and purple-fleshed), A. purpurea, and MaMe Red suggested a close relationship with TT2 TFs from a variety of plant species (Fig. 4A). Using R2R3 MYB TFs from the anthocyanin-related subgroup 6 and subgroup 5 in the phylogenetic tree revealed that kiwifruit MYBC1 clustered with Gossypium TT2 and Camellia sinensis MYB5a, which belong to subgroup 5 of the R2R3 MYB TF family involved in proanthocyanin biosynthesis34. Deduced amino acid alignments revealed that MYBC1 from A. melanandra, A. purpurea, and MaMe Red shared 98.2% identity and belonged to the R2R3 MYB TF family, as indicated by the highly conserved R2R3 domains (Supplementary Fig. 4). Although grouped with the PA regulators from subgroup 5, the C1 motif was not observed in the kiwifruit MYBC1 sequences, whereas some amino acids in the C3 motif and the VIRTKAx[K/R]C motif common in the PA regulators were observed in kiwifruit MYBC1 deduced amino acid sequences31,34. Similar to previously identified AcMYB123 (Acc28134.1), kiwifruit MYBC1 clustered with the TT2 clade and belongs to the subgroup 5 of the R2R3 MYB TF family in plants.
Figure 4.
Phylogeny trees of deduced amino acid sequences alignments constructed by neighbour-joining method in Geneious 10.0.3. using global alignment with free end gaps and the protein distance was calculated by Jukes-Cantor model. (A) Phylogenetic relationship of MYBC1 from Actinidia melanandra, A. purpurea and MaMe Red with R2R3 MYB TFs from subgroup 5 and subgroup 6 from other plant species. (B) Phylogenetic relationship of WRKY44 from A. melanandra and A. purpurea WRKY TFs from other plant species using Nicotiana tabacum NtWRKY44 (group III) as outgroup.
Phylogenetic analysis of WRKY44 from A. melanandra and A. purpurea revealed similarity with WRKY44/TTG2 TFs from a range of plant species (Fig. 4B). Kiwifruit WRKY44 grouped into the TTG2 clade and close to the Camellia sinensis WRKY44 which is a regulator of PA biosynthesis36. Kiwifruit WRKY44 possesses two WRKY domains composed of the conserved amino acids WRKYGQK toward the N-terminal as well as a zinc-finger ligand and a potential nuclear localisation sequence (Supplementary Fig. 5)47. These results suggested that the kiwifruit WRKY44 belongs to the group I of the WRKY superfamily in plants.
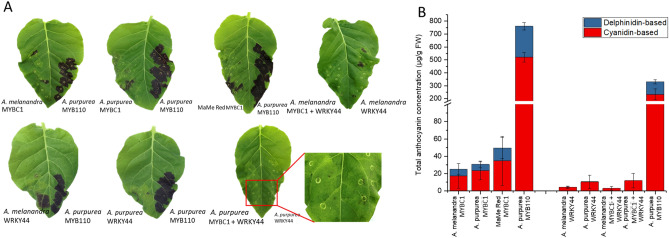
MYBC1 and WRKY44 induced anthocyanin patches on the leaves of Nicotiana tabacum
Transient over-expression of kiwifruit MYBC1 and WRKY44 in the leaves of N. tabacum induced anthocyanin accumulation (Fig. 5A). However, anthocyanin accumulated unevenly around the infiltration sites, as opposed to the complete saturation of anthocyanin at the infiltration site using the positive control, A. purpurea MYB110. The formation of the anthocyanin patches was variable between leaves. In some cases, only small points of anthocyanin accumulation formed near the infiltrated sites. The majority of anthocyanins induced by transient over-expression of MYBC1 are cyanidin-based and the remainder are delphinidin-based anthocyanins, whereas only cyanidin-based anthocyanin was observed from the transient over-expression of WRKY44 (Fig. 5B). These results suggest that both MYBC1 and WRKY44 from the purple kiwifruit species are able to induce anthocyanin accumulation when transiently over-expressed in tobacco leaves. The patchy and variable anthocyanin formation around the infiltration sites may indicate that MYBC1 and WRKY44 may require additional TF partners to fully activate the anthocyanin pathway.
Figure 5.
Transient over-expression of MYBC1 and WRKY44 in Nicotiana tabacum leaves for anthocyanin accumulation. (A) Digital image of N. tabacum leaves 7 days after transient over-expression of MYBC1 and WRKY44 from Actinidia melanandra, A. purpurea and MaMe Red with MYB110 from A. purpurea as a positive control. (B) HPLC measurement of anthocyanin from transient over-expression of MYBC1 and WRKY44 from (A). Data were shown as means ± SEM of three individual experiments.
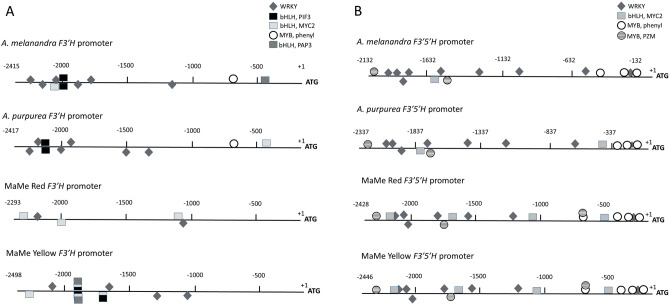
MYB and WRKY binding motifs annotated on kiwifruit F3′H and F3′5′H promoters
Potential binding motifs for the TFs from MYB, bHLH and WRKY families were predicted to be present on the F3′H and F3′5′H promoters previously cloned from A. melanandra, A. purpurea, MaMe Red and MaMe Yellow by the online tool PlantPAN3.019,48. Further screening of TF binding sites and motifs, by only including HIT score of 1, revealed predicted binding sites for MYB, bHLH, and WRKY TFs that are related to the phenylpropanoid pathway and flavonoid biosynthesis (Fig. 6). The common TF binding motifs found on all F3′H promoters were for bHLH TF, AtMYC2 and a WRKY TF, AtTTG2 (Fig. 6A). In addition, within the A. melanandra and A. purpurea F3′H promoters, a binding site for another bHLH TF, AtPIF3, and a binding site for MYB (MYB, phenyl) that binds to the promoters of phenylpropanoid biosynthetic genes were identified. All four F3′5′H promoters harboured the binding motifs for the bHLH TF, AtMYC2, the WRKY TF, AtTTG2, the MYB (MYB, PZM) which is the core consensus for the anthocyanin-related P gene in maize, and the MYB-binding motif found in the promoters of phenylpropanoid biosynthetic genes (Fig. 6B). The presence of these predicted binding sites within the F3′H and F3′5′H promoters suggests regulation by flavonoid-related TFs.
Figure 6.
Predicted binding sites of phenylpropanoid-related transcription factor on Actinidia melanandra, A. purpurea, MaMe Red and MaMe Yellow promoters using PlantPAN3.0. (A) Annotation of WRKY, bHLH, and MYB transcription factors related with phenylpropanoid pathway on F3′H promoters. (B) Annotation of WRKY, bHLH, and MYB transcription factors related with phenylpropanoid pathway on F3′5′H promoters. WRKY: binding site for WRKY, AtTTG2; bHLH, PIF3: binding site for bHLH AtPIF3; bHLH, MYC2: binding site for bHLH AtMYC2; MYB, phenyl: MYB binding motif found in promoters of phenylpropanoid biosynthetic genes. MYB, PZM: core consensus binding site for maize P gene (MYB homologue). The binding sites are displayed on the top (positive strand) and bottom (negative strand) have a similar score to the HIT sequence of 1.
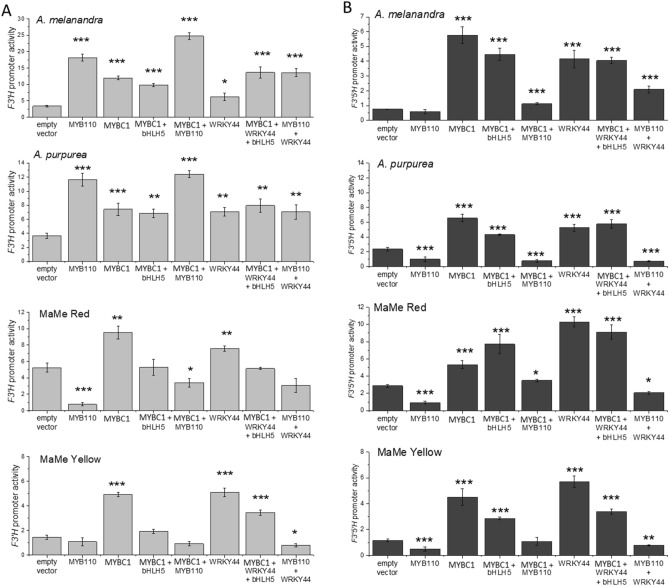
MYBC1 and WRKY44 activated kiwifruit F3′H and F3′5′H promoters
Dual luciferase promoter activation assays in the leaves of N. benthamiana showed that MYBC1 and WRKY44 significantly activated the F3′H promoters from all four species, including MaMe Yellow (Fig. 7A). There were high endogenous promoter readings, again suggesting activation by endogenous tobacco TFs. Previously it has been shown that MYB110 only activated the F3′H promoters from A. melanandra and A. purpurea, but not the MaMe kiwifruits19. The results here suggested that MYBC1 and WRKY44 were able to activate all F3′H promoters including those cloned from MaMe kiwifruits. However, no activation was observed by MYB110 on the MaMe F3′H promoters with or without MYBC1 and/or WRKY44 co-infiltration.
Figure 7.
Dual luciferase promoter activation assays testing the ability of MYBC1 and WRKY44 for promoter activation. (A) F3′H promoters isolated from Actinidia melanandra, A. purpurea, MaMe Red and MaMe Yellow were tested for activation by the MYBC1 and WRKY44 cloned from the respective species. (B) F3′5′H promoters isolated from A. melanandra, A. purpurea, MaMe Red and MaMe Yellow were tested for activation by the MYBC1 and WRKY44 cloned from the respective species. Empty vector and A. purpurea MYB110 were included as control. Data were shown as means ± SEM of four biological replicates. Statistical significance against the empty vector control: p < 0.05*, p < 0.01**, and p < 0.001***.
Infiltrations of MYBC1 and WRKY44 significantly activated F3′5′H promoters from all four species, with and without the co-factor bHLH5 (Acc19563.1) (Fig. 7B). As with F3′H promoters, there were high endogenous promoter readings. Consistent with previous findings, MYB110 was unable to activate F3′5′H promoters from all four kiwifruit species19. However, co-infiltration of MYBC1 or WRKY44 with MYB110 increased the activity of A. melanandra F3′5′H promoter and elevated the MaMe Red F3′5′H promoter when MYBC1 was co-infiltrated with MYB110. In summary, both MYBC1 and WRKY44 were able to significantly activate the F3′H and F3′5′H promoters from all four kiwifruit species, above an already high endogenous activity.
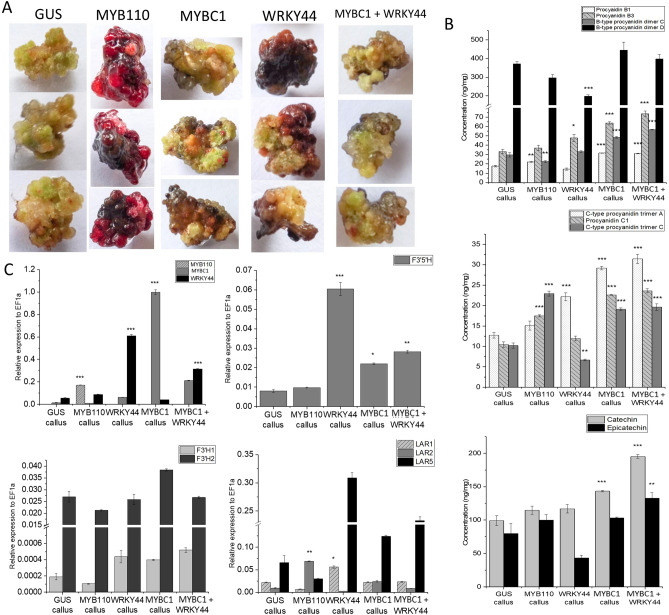
Over-expression of MYBC1 and WRKY44 in A. arguta increases F3′5′H expression and PA content
Transgenic calli were produced which over-expressed GUS, MYB110, MYBC1, WRKY44 and MYBC1/WRKY44 co-expression. These were analysed for gene expression and metabolite composition (Fig. 8A). GUS and MYB110 were transformed in A. arguta ‘K2D4’ as negative and positive controls, respectively. Phenotypically, MYB110 over-expressing calli are intensely purple throughout the tissue due to the high concentration of cyanidin-based anthocyanin, which is the only anthocyanin type detected (Supplementary Fig. 6A). The phenotype of calli over-expressing GUS showed no distinctive difference to the slightly more pigmented calli over-expressing MYBC1 and WRKY44. However, metabolite analysis revealed that MYBC1 and MYBC1/WRKY44 co-expression calli accumulated significantly more procyanidin B1, B3, B-type procyanidin dimer C, procyanidin C1, C-type procyanidin trimer A and C, and catechin (Fig. 8B) compared to the GUS and MYB110 over-expressing calli (see categories of PA49). WRKY44 over-expressing calli accumulated significantly higher levels of procyanidin B3 and C-type procyanidin trimer A than the control calli but a significant decrease in B-type procyanidin dimer D and C-type procyanidin trimer C (Fig. 8B). The accumulation of epicatechin and catechin were highest in calli co-expressing MYBC1/WRKY44. There were also small amounts of cyanidin-based anthocyanin detected in MYBC1 and MYBC1/WRKY44 calli. Plantlets regenerated from MYB110 over-expressing callus showed purple pigmentation throughout the shoots and roots, whereas the plantlets regenerated from MYBC1 and MYBC1/WRKY44 showed purple pigmentation in the roots only, compared to the green plantlets regenerated from the GUS control (Supplementary Fig. 7).
Figure 8.
Stable over-expression of GUS, MYB110, MYBC1, and WRKY44 in Actinidia arguta. (A) Callus formation of GUS, MYB110, MYBC1, WRKY and the co-infiltration of MYBC1/WRKY44 eight weeks after Agrobacteria-mediated over-expression in A. arguta leaf explant. (B) Proanthocyanin and anthocyanin analysis of the calli sampled from (A). (C) Gene expression analysis of the transgenes MYB110, MYBC1, WRKY44 and genes involved in the proanthocyanin and anthocyanin pathway in the callus from stably transformed calli. Data were shown as means ± SEM of three biological replicates. Statistical significance against the GUS control: p < 0.05*, p < 0.01**, and p < 0.001***.
Gene expression analysis revealed MYB110 over-expressing calli had high expression of the F3GT gene (Acc20132.1) in comparison to other calli (Supplementary Fig. 6B). Expression levels of DFR, F3′H1 and F3′H2 (Acc01005.1, Acc12813.1 and Acc18331.1) were similar across all transformed calli. Noticeably, the expression of F3′5′H (Acc32390.1) was significantly elevated in the calli over-expressing WRKY44, MYBC1 and a combination of both, compared to the GUS and MYB110 controls. Expression of the PA-related genes, FLS1 and LAR1 was elevated in the WRKY44 calli, whereas FLS2 was elevated in calli over-expressing GUS and MYB110 controls (Fig. 8C, Supplementary Fig. 6B). LAR5 was elevated in calli transformed with MYBC1 and WRKY44. Expression of both ANR1 and ANR2 was elevated in the calli over-expressing WRKY44, MYBC1 and MYBC1/WRKY44 (Supplementary Fig. 6B).
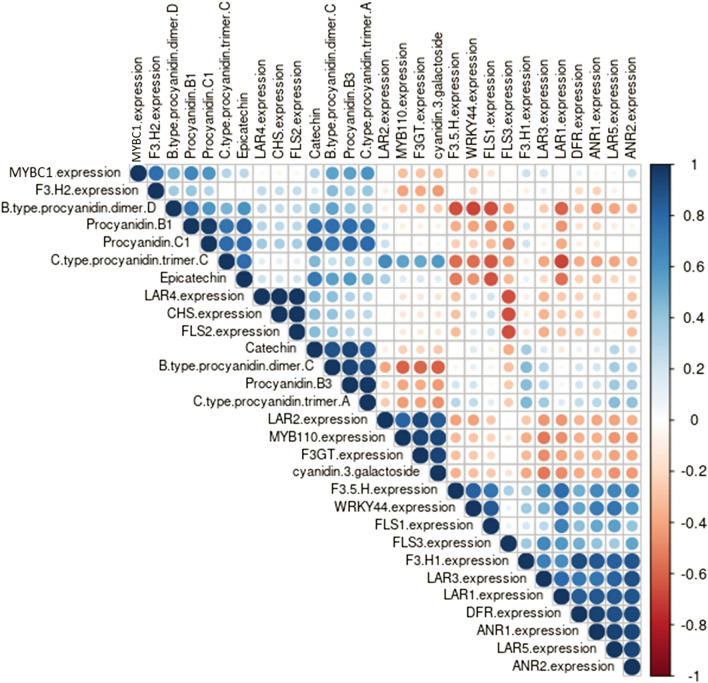
Pearson’s correlation analysis indicated strong positive correlations between MYB110, F3GT, LAR2, cyanidin-3-galactoside and C type procyanidin trimer C (Fig. 9, Supplementary Tables 7, 8). The expression of MYBC1 was strongly correlated with the expression of F3′H2 and the accumulation of B type procyanidin dimer C and D, procyanidin B1, B3, C1 and C type procyanidin trimer A. The expression of WRKY44 was strongly correlated with the expression of F3′5′H, F3′H1, FLS1, LAR1, LAR3, LAR5, DFR, ANR1 and ANR2.
Figure 9.
Correlation matrix of the relationship between gene expression and metabolite concentrations in the stably transformed Actinidia arguta calli. Dark and big blue dots indicate strong positive correlation while small red dots indicate weak negative correlation. Correlation with p value < 0.05 were shown, statistically non-significant correlation were left blank.
Discussion
Kiwifruit MYBC1 and WRKY44 are up-regulated during colour development
Anthocyanin accumulation is regulated at the transcriptional levels by genes encoding biosynthetic enzymes and TFs. Previous studies on red and purple kiwifruit species revealed that the key pathway genes CHS, DFR, F3GT and LDOX are responsible for anthocyanin accumulation19,50,51. The R2R3 MYB TFs, MYB10 (AcMYB75/AcMYBF110) and MYB110, positively regulate the anthocyanin pathway at the transcriptional level16,18,19,52. However, the transcriptional regulation of the cyanidin and delphinidin branch points controlled by the F3′H and F3′5′H has not been fully understood. In this study, transcriptomic analysis of purple kiwifruit at three developmental stages revealed and confirmed genes involved in the anthocyanin biosynthetic pathway (Figs. 1, 2). Two TFs, MYBC1 and WRKY44, exhibited similar expression patterns to the anthocyanin biosynthesis regulator, MYB110 and coincided with anthocyanin accumulation.
Kiwifruit MYBC1 and WRKY44 are PA-related TFs
Kiwifruit MYBC1 belongs to the subgroup 5 of the MYB TF family and shared amino acid residues found in the highly conserved motif VIRTKAx[K/R]C that is characteristic to the PA-regulating TFs, but the C1 and C3 motifs seen in the anthocyanin modulating VvMYB5 were not present in the c-terminus of kiwifruit MYBC1 (Fig. 4A, Supplementary Fig. 4)31,34. Generally, MYB TFs from subgroup 6 contribute to the regulation of the anthocyanin pathway while MYB TFs from subgroup 5 are thought to be involved in PA accumulation. In Arabidopsis, TT2 is responsible for PA accumulation in the endothelium during seed development by regulating flavonoid genes such as DFR and BAN (anthocyanidin reductase) as well as TT8 (bHLH) and TTG1 (WD40)53. In grape berries, VvMYB5a and VvMYB5b are capable of inducing anthocyanin and flavonol accumulation when over-expressed in tobacco31,54. VvMYB5b was also able to activate the promoters of structural genes involved in the anthocyanin and PA pathways such as VvLAR1, VvANS, VvANR and VvF3′5′H, suggesting a regulatory role in different branches of the phenylpropanoid pathway. Tea CsMYB5a and CsMYB5e and freesia FhMYB5 all demonstrated regulatory roles in the anthocyanin and PA biosynthesis pathway33,34. Recently, in red-centred kiwifruit A. chinensis ‘Hongyang’, a TT2 type R2R3 MYB TF, AcMYB123 and a bHLH TF, AcbHLH42 were identified to be involved in the inner pericarp-specific accumulation of anthocyanins by activating the expression of AcANS and AcF3GT42. Although belonging in the same subgroup 5 as the kiwifruit MYBC1 identified in this study, AcMYB123 (Acc28234.1, MH643775.1) only shared 47% sequence identity and the expression was barely detected in sequenced tissues. MYBC1 from A. arguta is highly similar to MYBC1 identified here (98% amino acid sequence identity) and is implicated to be involved in anthocyanin biosynthesis. It is negatively regulated by microRNA85843. Expression of other TT2-like MYBs was barely detected in the purple kiwifruit species studied here, making the possibility of a role in regulation unlikely (Supplementary Figs. 2, 4).
Kiwifruit WRKY44 (Acc16887.1) belongs to group I of the WRKY superfamily and clusters in the TTG2 clade closely with tea CsWRKY44, which is involved in the catechin regulation (Fig. 4B)36. Within the TTG2 clade, Arabidopsis TTG2 encodes for a WRKY TF that participates in trichome formation and tannin production in seed coat endothelium by regulating the vacuolar transport step in PA pathway35,55. Moreover, kiwifruit WRKY44 is homologous to PH3 in petunia, which regulates vacuolar acidification for anthocyanin storage39. In petunia flower petals, PH3 encodes for a WRKY TF that is highly similar to AtTTG2 and the transcription is activated by the MBW complex which then activates the transcription of a vacuolar ATPase to hyper-acidify the vacuole. A close phylogenetic relationship with these WRKY TFs indicates a possible regulatory role for kiwifruit WRKY44 in the anthocyanin and PA pathway.
MYBC1 and WRKY44 regulate F3′H and F3′5′H branch points
Functional characterisation of MYBC1 and WRKY44 revealed that both TFs are able to induce anthocyanin accumulation when transiently over-expressed in tobacco leaves (Fig. 5). The regulatory roles of MYBC1 and WRKY44 were confirmed by the significant activation of all kiwifruit F3′H and F3′5′H promoters, for which the anthocyanin activator, MYB110, showed no regulatory role as previously found (Fig. 7)19. This observation was supported by the presence of multiple phenylpropanoid related MYB and WRKY TF binding sites identified on the promoter sequences (Fig. 6). These findings suggest that MYBC1 and WRKY44 transcriptionally regulate anthocyanin biosynthesis by activating the F3′H and F3′5′H branch points, which determines the hydroxylation patterns of the anthocyanin aglycone.
Kiwifruit calli over-expressing MYBC1 and WRKY44 had no obvious visual phenotype but metabolite and gene expression analysis revealed major differences from calli expressing a GUS-control (Fig. 8). The expression of kiwifruit F3′5′H was significantly up-regulated by the over-expression of WRKY44 and MYBC1. Expression of genes encoding for biosynthetic enzymes in the PA pathway such as FLS1, LAR1, LAR5, ANR1 and ANR2 were elevated in the WRKY44 and MYBC1 calli compared to both GUS-control and MYB110 calli. As a result, the amounts of PA accumulated in those calli increased significantly. Correlation analysis suggested linkage between F3′H and F3′5′H and the B- and C-type procyanidin isomers as a result of the over-expression of WRKY44 and MYBC1. As expected, expression of F3GT was significantly elevated by the over-expression of MYB110 in the anthocyanin accumulating calli, reiterating its critical role in anthocyanin regulation50. Anthocyanin accumulated in the calli over-expressing MYBC1 and MYBC1/WRKY44 at low levels, but with weak correlation data. However, the roots of the plantlets regenerated from the MYBC1 and MYBC1/WRKY44 calli were visibly red, differing from the unpigmented roots of GUS plantlets (Supplementary Fig. 7). Stable lines expressing these genes, as adult vines, will be important research tools.
MYB110, MYBC1 and WRKY44 are in the regulatory network shared between PA and anthocyanin biosynthesis
Core genes encoding enzymes in the anthocyanin pathway, such as CHS, DFR, F3GT, and LDOX, have been shown to be transcriptionally regulated by the subgroup 6 R2R3 MYB TFs, MYB10 and MYB110 in kiwifruit species16–19. However the F3′H and F3′5′H branch points that decide the production of cyanidin and delphinidin were not principally regulated by MYB10 or MYB110 in purple kiwifruit species19. In grape MYBA1 activates F3′5′H promoters whereas two closely related TFs, MYBA6 and MYBA7 from subgroup 6, showed no activation of the F3′5′H promoters56. However, VvMYB5a and VvMYB5b from subgroup 5 activated the F3′5′H promoter by 12 fold as well as other genes involved in the PA and anthocyanin pathways31. In transgenic purple tomatoes, the control of flavonoid biosynthesis showed a specialised regulatory mechanism where the over-expression of TFs Del/Ros1 activated a broader spectrum of the genes including the F3′5′H in the flavonoid pathway but the anthocyanin-related TFs LC/C1 did not57,58. These results suggest different specificities of TFs controlling anthocyanin biosynthesis.
Both MYBC1 and WRKY44 could regulate the anthocyanin pathway at the F3′H and F3′5′H branch points via the MBW complex in kiwifruit (Fig. 10). Functional characterisation showed that MYB110, MYBC1 and WRKY44 regulate different points of the pathway. We propose a model where, when MYB110 is incorporated into the MBW complex, the complex activates core genes such as CHS, DFR, and F3GT (Fig. 10). When MYBC1 forms a MBW complex with bHLH and WD40, it activates the F3′H and F3′5′H branch points which hydroxylate substrates to pass down the anthocyanin pathway. WRKY44 may activate the transcription by engaging with the MBW complex to form MBW-WRKY complex (Fig. 10). Petunia PH3 and Arabidopsis TTG2, homologous to kiwifruit WRKY44, were able to bind to the WD40 protein of the MBW complex and formed a MBW-W complex required to transcriptionally activate the target genes involved in vacuolar acidification (petunia) and hair development (Arabidopsis39). In grapevine, VvWRKY26, a homologue of petunia PH3 and AtTTG2, is recruited specifically by the VvMYB5a driven MBW complex to enhance the expression of target genes involved in vacuolar acidification, probably via the formation of a MBW-WRKY complex40. In kiwifruit, WRKY motifs are located in close proximity to the MYB and bHLH binding motifs on F3′H and F3′5′H promoters, suggesting the possible binding of the MBW-WRKY complex on the promoters for transcriptional activation (Fig. 6). Infiltration of WRKY44 (and MYBC1) was able to significantly increase the promoter activation.
Figure 10.
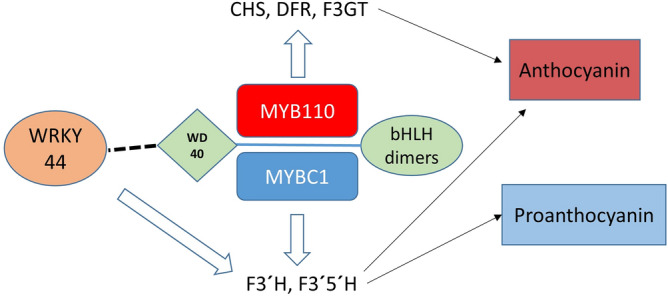
Model of anthocyanin and proanthocyanin pathway regulation by the MYB-BHLH-WD40-WRKY complex. Either the MYB110 or the MYBC1 is the driver of the complex, but not both at the same time. Big white arrows indicate the activation of the MBW-W complex on the biosynthetic genes. Black arrows indicate the impact of the biosynthetic genes on the pathways. Solid line indicates MBW complex partnership and dash line indicates possible interaction with the MBW complex by WRKY44.
The F3′H and F3′5′H enzymes are control points for the accumulation of cyanidin or delphinidin-based anthocyanins. However, they also hydroxylate immediate substrates that feed into the PA biosynthesis by the actions of the downstream enzymes (Supplementary Fig. 8). Activation of the F3′H and F3′5′H will increase the accumulation of dihydroquercetin and dihydromyricetin, which can be converted into anthocyanin precursors by DFR in the anthocyanin pathway or can be converted in quercetin and myricetin by FLS in the proanthocyanin pathway to form flavonols. The anthocyanin precursors can also be intercepted by LAR and ANR enzymes to generate PAs. While MYB110 may increase the general flux through the anthocyanin pathway by activating the core genes, MYBC1 and WRKY44 may increase intermediate substrates via regulation of F3′H and F3′5′H branch points and subsequent competition between enzymes. Hence, MYB110, MYBC1 and WRKY44 are proposed to participate in the same regulatory network shared by anthocyanin and proanthocyanin biosynthesis.
Conclusion
The anthocyanin biosynthesis pathway and the associated transcriptional regulators have been well studied in a variety of plant species. In this study, the kiwifruit MYBC1 and WRKY44 transcriptionally regulate the key F3′H and F3′5′H branch points that have potential for controlling the types of flavonoids accumulated. The involvement of TFs shared by the anthocyanin and PA biosynthesis pathways adds to the potential to fine-tune the balance of the total metabolites in the fruit. These specific hydroxylation steps determine pigmentation as well as increasing nutritional values of fruit. Anthocyanin-enriched extracts from purple kiwifruit showed reduction of key inflammatory signals involved in lung inflammation59. Dietary intake of di- and tri-hydroxylated anthocyanins, flavonols, flavanols and PAs is linked to reductions in markers of cardiovascular disease risk60,61 as well as reducing the incidence of cardiovascular and metabolic diseases and cancers62–64. Understanding the transcriptional regulation of the metabolites will maximise the value of fruit crops as metabolites form a large basis of their nutritional benefits.
Materials and methods
Plant material
Actinidia melanandra, A. purpurea (sometimes referred to as A. arguta var. purpurea) and two progeny lines of a cross between A. macrosperma × A. melanandra (MaMe) were grown at the Plant and Food Research Orchard, Motueka, New Zealand as described previously19. Fruits were harvested at mature green stage and held at 20 °C for ripening. Skin peel and flesh tissues were separated and sampled into liquid nitrogen for mature green stage, colour change stage, and ripe stage.
RNA-sequencing and transcriptomic analysis
RNA was isolated from the finely ground skin peel and flesh tissues of A. purpurea, MaMe Red and MaMe Yellow using the Spectrum Plant Total RNA kit (Sigma-Aldrich, USA) following manufacturer’s protocol. The RNA integrity number (RIN) of the extracted total RNA were above 7 and were sent to The Australian Genome Research Facility Ltd (AGRF) for Illumina RNA library constructions and Illumina HiSeq next generation sequencing. Quality of the RNASeq data was checked using FastQC (version 0.11.2) and using BBMap65 (version 37.93) adapter and quality trimming was carried out, where quality threshold of 20 and 10 bases from 5′ end were trimmed, with a minimum length of sequence after trimming set to 35 bases. Thereafter, quality trimmed reads were mapped to the manually annotated A. chinensis genome44 using STAR66 (version 2.5.2b) splice aware aligner with the ‘quantMode’ turned on. Resulting counts from STAR aligner were used in the differential expression (DE) analysis using DESeq (version 1.18.1) in R (version 3.4.3). The read counts were used to generate Reads per Kilobase Million (RPKM).
Isolation, cloning and sequence alignment of candidate genes
The isolated RNA from A. melanandra, A. purpurea, MaMe Red and MaMe Yellow were reverse transcribed into cDNA using the QuantiTect Reverse Transcription kit (Qiagen, USA) following manufacturer’s protocols. Sequences of candidate genes were PCR amplified from cDNA of A. melanandra, A. purpurea and MaMe Red and cloned into over-expression vector pSAK277 using In-Fusion HD cloning (Takara Bio USA, Inc). Genes were sequenced by Macrogen, Korea. Multiple nucleotide and amino acid sequence alignments and phylogeny trees were created by Geneious 10.0.8.
Transient over-expression of candidate genes in tobacco leaves
Over-expression vector pSAK277 with 35S promoter driving the expression of the candidate gene was transformed into Agrobacterium tumefaciens strain GV3101 by electroporation followed by incubation19. Nicotiana tabacum plants were grown under glasshouse conditions using natural daylight with extension to 16 h. Three leaves of the 6-week-old N. tabacum were infiltrated with Agrobacterium and kept under the same growth conditions. Leaves were photographed and harvested at 7 days after infiltration and stored at − 80 °C until analysis.
Anthocyanin quantification by high performance liquid chromatography (HPLC)
Anthocyanin accumulation from transient over-expression of candidate genes in tobacco leaves was confirmed and measured by high performance liquid chromatography (HPLC–DAD). Approximately 300 mg of freeze-dried tissue powder were used to extract anthocyanin with acidified methanol (0.1% HCl) for two hours at room temperature. The supernatant was spin-dried and resuspended in 20% methanol followed by filtration by syringe filter and diluted with 20% methanol for analysis. Identification was achieved using an Acclaim PA2 C18 column (Dionex, ThermoFisher Scientific) maintained at 35 °C in a Dionex UltiMate 3000 Series HPLC with photodiode array detection at 520 nm (ThermoFisher Scientific, USA). 100% of solvent A (0.1% formic acid) was ramped to 98% at 5 min, then 85% A at 10 min, 80% A at 20 min and 100% B (acetonitrile + 0.1% formic acid) at 34 min at the flow rate of 350 µl/min for 40 min per 5 µl sample run. Standard for cyanidin-3-glucoside was used to quantitate anthocyanin concentrations which are reported as cyanidin-3-glucoside equivalents per gram of fresh weight (FW) or dry weight (DW).
Dual luciferase transient assay in tobacco leaves
Genomic DNA was extracted from skin tissue of A. melanandra, A. purpurea, MaMe Red and MaMe Yellow using the DNeasy Plant Mini kit (Qiagen, USA). The 2.3 kb promoter regions of F3′H and F3′5′H were isolated by PCR and cloned into the vector pGreen 0800-LUC vector using In-Fusion HD cloning and transformed into A. tumefaciens GV3101, as mentioned previously19,67. The promoter sequences were confirmed by sequencing (Macrogen, Korea) and potential binding motifs were screened by the Plant Promoter Analysis Navigator PlantPAN3.0 https://plantpan.itps.ncku.edu.tw/48. The promoter activation dual luciferase assays were performed on leaves of 6-week-old N. benthamiana plants by co-infiltrating the vector carrying the promoter sequence with the over-expression vector containing candidate genes. Four days after infiltration, four leaf discs from each treatment were sampled to assay the firefly luciferase and renilla luciferase assay reagents (Targeting systems, USA). The promoter activities were expressed as a ratio of LUC to REN activity.
Stable over-expression of candidate genes in A. arguta
Newly initiated leaves of A. arguta genotype K2D4 from in vitro grown shoots were excised and inoculated with Agrobacterium tumefaciens strain EHA105 containing the vectors pSAK277 with CaMV 35S:MYB110, 35S:MYBC1, 35S:WRKY44, and 35S:GUS, respectively. The transformation procedure was based on previous reports68,69. Calli formed in the selection medium containing 150 mg/l of kanamycin were excised individually and transferred to fresh regeneration medium containing 150 mg/l of kanamycin for calli growth and bud induction. Approximately 12 weeks after transformation, half of the calli were powdered in liquid nitrogen and stored in − 80 °C until analysis. The rest of the calli were subcultured in fresh regeneration medium. Shoots generated from these calli were excised and subcultured in medium for root elongation before being potted and grown in the containment glasshouse.
Real time quantitative PCR expression analysis
RNA was isolated from the powdered calli using the Spectrum Plant Total RNA kit (Sigma-Aldrich, USA) and reverse transcribed into cDNA using the QuantiTect Reverse Transcription kit (Qiagen, USA) following manufacturer’s protocols. Genes of the anthocyanin and proanthocyanin pathways were identified by BLAST with genes of known function on the Actinidia chinensis genome and the Actinidia arguta RNA-seq data44,70. Gene-specific oligonucleotide primers were designed using Geneious 10.0.8 and are summarised in Supplementary Table 9. RT-qPCR was carried out using the LightCycler 480 instrument with LightCycler 480 SYBR Green I Mastermix (Roche Diagnostics, USA). Each reaction volume was 5 µL and reactions were run in quadruplicate, and non-template control and water control were included in each run. The thermal cycling conditions were 95 °C for 5 min, followed by 50 cycles of 95 °C for 10 s, 60 °C for 10 s and 72 °C for 20 s, then a melting temperature cycle with continuous fluorescence data acquisition from 65 to 95 °C. The data output was analysed by the LightCycler480 software Version 1.5 (https://lifescience.roche.com/en_nz/products/lightcycler14301-480-software-version-15.html) using the Target/Reference ratio to compare the expression level of the target genes normalised to the reference gene, elongation factor 1-α EF1α.
Proanthocyanin and anthocyanin quantification in A. arguta calli
The proanthocyanin and anthocyanin content of the calli was determined by liquid chromatography-mass spectrometry (LC–MS) using an LTQ linear ion trap mass spectrometer fitted with an ESI interface (ThermoFisher Scientific, San Jose, CA, USA) coupled to an Ultimate 3000 UHPLC and PDA detector (Dionex, Sunnyvale, CA, USA) as described previously71. For proanthocyanins, to each sample (~ 40 mg fresh weight) was added 1 ml of ethanol/water 95:5 (v:v) and 0.8 g stainless steel beads 0.9–2 mm (Next Advance Inc., NY, USA). Samples were bead beaten for 4 min (Bullet Blender 24 Gold, Next Advance Inc., NY, USA) and were extracted overnight. After centrifugation at 13,000 × g for 5 min, the supernatant was evaporated to dryness under a stream of nitrogen at 35 °C and reconstituted in 10% methanol (100 µl) for analysis. Compound separation was achieved using a Hypersil GOLD aQ 1.9µ C18 175 Å (Thermo Scientific, Waltham, MA, USA), 150 × 2.1 mm column maintained at 35 °C. The solvents were (A) water + 0.1% formic acid and (B) acetonitrile + 0.1% formic acid (flow rate, 200 µl/min). The initial mobile phase, 95% A/5% B, was held for 5 min, then ramped linearly to 90% A at 10 min, 83% A at 25 min, 77% A at 30 min, 70% A at 40 min, 3% A at 48 min and held for 5 min before resetting to the original conditions. The sample injection volume was 4 μl. The MS data were acquired in the negative mode using a data dependent LC-MS4 method. This method isolates and fragments the most intense parent ion to give MS2 data (daughter ions), then isolates and fragments the most intense daughter ion (MS3 data), then granddaughter ion (MS4 data). The ESI voltage, capillary temperature, sheath gas pressure and sweep gas were set at − 10 V, 275 °C, 35 psi and 5 psi, respectively. Standards for catechin and epicatechin were used to quantitate proanthocyanin concentrations, which, with the exception of epicatechin which is reported as itself, are reported as catechin equivalents per mg of fresh weight (FW).
For anthocyanins, to each sample (~ 10 mg fresh weight) was added 1 ml of methanol/formic acid 95:5 (v:v) and 0.8 g stainless steel beads 0.9–2 mm (Next Advance Inc., NY, USA). Samples were bead beaten for 4 min (Bullet Blender 24 Gold, Next Advance Inc., NY, USA) and were extracted overnight. After centrifugation at 13,000 × g for 5 min, the supernatant was evaporated to dryness under a stream of nitrogen at 35 °C and reconstituted in acetonitrile/formic acid/water (5:3:92 v:v:v; 100 µl) for analysis. Compound separation, identification and quantitation by LC–MS were as described19.
Statistical analysis
The statistical significance of the difference between the empty vector control and the treatment means in the dual luciferase transient assays was tested by two sample t-test. One-way ANOVA was used to determine the statistically significant differences in metabolite and gene expression analysis between the calli transformed with different candidate genes. Correlation matrix using Pearson’s correlation with significance level was calculated and produced in R Studio version 1.2.5033.
Supplementary information
Acknowledgements
We would like to thank Dinum Herath for the advice on A. arguta transformation. Monica Holland for critically reading the manuscript. This research is supported by The New Zealand Government, Ministry of Business, Innovation and Employment Endeavour Fund (MBIE), ‘Turbo Breeding’ (C11X1602) and MBIE ‘Filling the Void’ (C11X1704). YP is supported by the Joint Graduate School in Plant and Food Science, University of Auckland.
Author contributions
Y.P., R.V.E., A.C.A. contributed to project planning and wrote the manuscript. Y.P. conducted experiments, sample collections, data analysis and interpretation. A.T. conducted bioinformatics analysis. J.C. and D.J. performed LCMS experiment and data analysis. All authors contributed to editing the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-70977-0.
References
- 1.Allan AC, Hellens RP, Laing WA. MYB transcription factors that colour our fruit. Trends Plant Sci. 2008;13:99–102. doi: 10.1016/j.tplants.2007.11.012. [DOI] [PubMed] [Google Scholar]
- 2.Winkel-Shirley B. Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 2001;126:485–493. doi: 10.1104/pp.126.2.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hichri I, et al. Recent advances in the transcriptional regulation of the flavonoid biosynthetic pathway. J. Exp. Bot. 2011;62:2465–2483. doi: 10.1093/jxb/erq442. [DOI] [PubMed] [Google Scholar]
- 4.Pang Y, Peel GJ, Wright E, Wang Z, Dixon RA. Early steps in proanthocyanidin biosynthesis in the model legume Medicago truncatula. Plant Physiol. 2007;145:601–615. doi: 10.1104/pp.107.107326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baudry A, et al. TT2, TT8, and TTG1 synergistically specify the expression of BANYULS and proanthocyanidin biosynthesis in Arabidopsis thaliana. Plant J. 2004;39:366–380. doi: 10.1111/j.1365-313X.2004.02138.x. [DOI] [PubMed] [Google Scholar]
- 6.Xie D-Y, Sharma SB, Paiva NL, Ferreira D, Dixon RA. Role of anthocyanidin reductase, encoded by BANYULS in plant flavonoid biosynthesis. Science. 2003;299:396–399. doi: 10.1126/science.1078540. [DOI] [PubMed] [Google Scholar]
- 7.Routaboul J-M, et al. Flavonoid diversity and biosynthesis in seed of Arabidopsis thaliana. Planta. 2006;224:96–107. doi: 10.1007/s00425-005-0197-5. [DOI] [PubMed] [Google Scholar]
- 8.Seitz C, Ameres S, Forkmann G. Identification of the molecular basis for the functional difference between flavonoid 3′-hydroxylase and flavonoid 3′,5′-hydroxylase. FEBS Lett. 2007;581:3429–3434. doi: 10.1016/j.febslet.2007.06.045. [DOI] [PubMed] [Google Scholar]
- 9.Allan AC, Espley RV. MYBs drive novel consumer traits in fruits and vegetables. Trends Plant Sci. 2018;23:693–705. doi: 10.1016/j.tplants.2018.06.001. [DOI] [PubMed] [Google Scholar]
- 10.Borevitz JO, Xia Y, Blount J, Dixon RA, Lamb C. Activation tagging identifies a conserved MYB regulator of phenylpropanoid biosynthesis. Plant Cell. 2000;12:2383–2393. doi: 10.1105/tpc.12.12.2383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Takos AM, et al. Light-induced expression of a MYB gene regulates anthocyanin biosynthesis in red apples. Plant Physiol. 2006;142:1216–1232. doi: 10.1104/pp.106.088104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Espley RV, et al. Red colouration in apple fruit is due to the activity of the MYB transcription factor, MdMYB10. Plant J. 2007;49:414–427. doi: 10.1111/j.1365-313X.2006.02964.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Quattrocchio F, et al. PH4 of Petunia is an R2R3 MYB protein that activates vacuolar acidification through interactions with basic-helix-loop-helix transcription factors of the anthocyanin pathway. Plant Cell. 2006;18:1274–1291. doi: 10.1105/tpc.105.034041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lin-Wang K, et al. An R2R3 MYB transcription factor associated with regulation of the anthocyanin biosynthetic pathway in Rosaceae. BMC Plant Biol. 2010;10:50. doi: 10.1186/1471-2229-10-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.This P, Lacombe T, Cadle-Davidson M, Owens CL. Wine grape (Vitis vinifera L.) color associates with allelic variation in the domestication gene VvmybA1. Theor. Appl. Genet. 2007;114:723–730. doi: 10.1007/s00122-006-0472-2. [DOI] [PubMed] [Google Scholar]
- 16.Fraser LG, et al. An R2R3 MYB transcription factor determines red petal colour in an Actinidia (kiwifruit) hybrid population. BMC Genom. 2013;14:28. doi: 10.1186/1471-2164-14-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li W, et al. Gene expression profiling of development and anthocyanin accumulation in kiwifruit (Actinidia chinensis) based on transcriptome sequencing. PLoS ONE. 2015;10:e0136439. doi: 10.1371/journal.pone.0136439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li W, et al. Kiwifruit R2R3-MYB transcription factors and contribution of the novel AcMYB75 to red kiwifruit anthocyanin biosynthesis. Sci. Rep. 2017;7:1–14. doi: 10.1038/s41598-017-16905-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Peng Y, et al. Differential regulation of the anthocyanin profile in purple kiwifruit (Actinidia species) Hortic. Res. 2019;6:1–16. doi: 10.1038/s41438-018-0076-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang N, et al. MYB12 and MYB22 play essential roles in proanthocyanidin and flavonol synthesis in red-fleshed apple (Malus sieversii f. niedzwetzkyana) Plant J. 2017;90:276–292. doi: 10.1111/tpj.13487. [DOI] [PubMed] [Google Scholar]
- 21.Zhou H, et al. Peach MYB7 activates transcription of the proanthocyanidin pathway gene encoding leucoanthocyanidin reductase, but not anthocyanidin reductase. Front. Plant Sci. 2015;6:908. doi: 10.3389/fpls.2015.00908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schaart JG, et al. Identification and characterization of MYB-b HLH-WD 40 regulatory complexes controlling proanthocyanidin biosynthesis in strawberry (F. ragaria × ananassa) fruits. New Phytol. 2013;197:454–467. doi: 10.1111/nph.12017. [DOI] [PubMed] [Google Scholar]
- 23.Akagi T, et al. DkMyb4 is a Myb transcription factor involved in proanthocyanidin biosynthesis in persimmon fruit. Plant Physiol. 2009;151:2028–2045. doi: 10.1104/pp.109.146985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Terrier N, et al. Ectopic expression of VvMybPA2 promotes proanthocyanidin biosynthesis in grapevine and suggests additional targets in the pathway. Plant Physiol. 2009;149:1028–1041. doi: 10.1104/pp.108.131862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stracke R, et al. Analysis of production of flavonol glycosides-dependent flavonol glycoside accumulation in Arabidopsis thaliana plants reveals MYB11-, MYB12-and MYB111-independent flavonol glycoside accumulation. New Phytol. 2010;188:985–1000. doi: 10.1111/j.1469-8137.2010.03421.x. [DOI] [PubMed] [Google Scholar]
- 26.Czemmel S, et al. The grapevine R2R3-MYB transcription factor VvMYBF1 regulates flavonol synthesis in developing grape berries. Plant Physiol. 2009;151:1513–1530. doi: 10.1104/pp.109.142059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Adato A, et al. Fruit-surface flavonoid accumulation in tomato is controlled by a SlMYB12-regulated transcriptional network. PLoS Genet. 2009;5:e1000777. doi: 10.1371/journal.pgen.1000777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jin H, et al. Transcriptional repression by AtMYB4 controls production of UV-protecting sunscreens in Arabidopsis. EMBO J. 2000;19:6150–6161. doi: 10.1093/emboj/19.22.6150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Aharoni A, et al. The strawberry FaMYB1 transcription factor suppresses anthocyanin and flavonol accumulation in transgenic tobacco. Plant J. 2001;28:319–332. doi: 10.1046/j.1365-313x.2001.01154.x. [DOI] [PubMed] [Google Scholar]
- 30.Pérez-Díaz JR, et al. New member of the R2R3-MYB transcription factors family in grapevine suppresses the anthocyanin accumulation in the flowers of transgenic tobacco. Plant Mol. Biol. 2016;90:63–76. doi: 10.1007/s11103-015-0394-y. [DOI] [PubMed] [Google Scholar]
- 31.Deluc L, et al. The transcription factor VvMYB5b contributes to the regulation of anthocyanin and proanthocyanidin biosynthesis in developing grape berries. Plant Physiol. 2008;147:2041–2053. doi: 10.1104/pp.108.118919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang L, et al. R2R3-MYB transcription factor MYB 6 promotes anthocyanin and proanthocyanidin biosynthesis but inhibits secondary cell wall formation in Populus tomentosa. Plant J. 2019;99:733–751. doi: 10.1111/tpj.14364. [DOI] [PubMed] [Google Scholar]
- 33.Li Y, et al. The R2R3-MYB factor FhMYB5 from Freesia hybrida contributes to the regulation of anthocyanin and proanthocyanidin biosynthesis. Front. Plant Sci. 2019;9:1935. doi: 10.3389/fpls.2018.01935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiang X, et al. CsMYB5a and CsMYB5e from Camellia sinensis differentially regulate anthocyanin and proanthocyanidin biosynthesis. Plant Sci. 2018;270:209–220. doi: 10.1016/j.plantsci.2018.02.009. [DOI] [PubMed] [Google Scholar]
- 35.Gonzalez A, et al. TTG2 controls the developmental regulation of seed coat tannins in Arabidopsis by regulating vacuolar transport steps in the proanthocyanidin pathway. Dev. Biol. 2016;419:54–63. doi: 10.1016/j.ydbio.2016.03.031. [DOI] [PubMed] [Google Scholar]
- 36.Zhang Y, et al. Identification of key genes involved in catechin metabolism in tea seedlings based on transcriptomic and HPLC analysis. Plant Physiol. Biochem. 2018;133:107–115. doi: 10.1016/j.plaphy.2018.10.029. [DOI] [PubMed] [Google Scholar]
- 37.Wang N, et al. Transcriptomic analysis of red-fleshed apples reveals the novel role of MdWRKY11 in flavonoid and anthocyanin biosynthesis. J. Agric. Food Chem. 2018;66:7076–7086. doi: 10.1021/acs.jafc.8b01273. [DOI] [PubMed] [Google Scholar]
- 38.Liu W, et al. MdWRKY11 participates in anthocyanin accumulation in red-fleshed apples by affecting MYB transcription factors and the photoresponse factor MdHY5. J. Agric. Food Chem. 2019;67:8783–8793. doi: 10.1021/acs.jafc.9b02920. [DOI] [PubMed] [Google Scholar]
- 39.Verweij W, et al. Functionally similar WRKY proteins regulate vacuolar acidification in petunia and hair development in Arabidopsis. Plant Cell. 2016;28:786–803. doi: 10.1105/tpc.15.00608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Amato A, et al. The MYB 5-driven MBW complex recruits a WRKY factor to enhance the expression of targets involved in vacuolar hyper-acidification and trafficking in grapevine. Plant J. 2019;99:1220–1241. doi: 10.1111/tpj.14419. [DOI] [PubMed] [Google Scholar]
- 41.Ferguson AA, Seal A. Temperate Fruit Crop Breeding. New York: Springer; 2008. pp. 235–264. [Google Scholar]
- 42.Wang L, et al. A MYB/bHLH complex regulates tissue-specific anthocyanin biosynthesis in the inner pericarp of red-centered kiwifruit Actinidia chinensis cv. Hongyang. Plant J. 2019;99:359–378. doi: 10.1111/tpj.14330. [DOI] [PubMed] [Google Scholar]
- 43.Li Y, et al. MicroRNA858 negatively regulates anthocyanin biosynthesis by repressing AaMYBC1 expression in kiwifruit (Actinidia arguta) Plant Sci. 2020;5:110476. doi: 10.1016/j.plantsci.2020.110476. [DOI] [PubMed] [Google Scholar]
- 44.Pilkington SM, et al. A manually annotated Actinidia chinensis var. chinensis (kiwifruit) genome highlights the challenges associated with draft genomes and gene prediction in plants. BMC Genom. 2018;19:257. doi: 10.1186/s12864-018-4656-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Brendolise C, et al. Multiple copies of a simple MYB-binding site confers trans-regulation by specific flavonoid-related R2R3 MYBs in diverse species. Front. Plant Sci. 2017;8:1864. doi: 10.3389/fpls.2017.01864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Montefiori M, et al. In the Solanaceae, a hierarchy of bHLHs confer distinct target specificity to the anthocyanin regulatory complex. J. Exp. Bot. 2015;66:1427–1436. doi: 10.1093/jxb/eru494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eulgem T, Rushton PJ, Robatzek S, Somssich IE. The WRKY superfamily of plant transcription factors. Trends Plant Sci. 2000;5:199–206. doi: 10.1016/s1360-1385(00)01600-9. [DOI] [PubMed] [Google Scholar]
- 48.Chow C-N, et al. PlantPAN3.0: A new and updated resource for reconstructing transcriptional regulatory networks from ChIP-seq experiments in plants. Nucleic Acids Res. 2019;47:D1155–D1163. doi: 10.1093/nar/gky1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xie D-Y, Dixon RA. Proanthocyanidin biosynthesis-still more questions than answers? Phytochemistry. 2005;66:2127–2144. doi: 10.1016/j.phytochem.2005.01.008. [DOI] [PubMed] [Google Scholar]
- 50.Montefiori M, et al. Identification and characterisation of F3GT1 and F3GGT1, two glycosyltransferases responsible for anthocyanin biosynthesis in red-fleshed kiwifruit (Actinidia chinensis) Plant J. 2011;65:106–118. doi: 10.1111/j.1365-313X.2010.04409.x. [DOI] [PubMed] [Google Scholar]
- 51.Li Y, et al. A key structural gene, AaLDOX, is involved in anthocyanin biosynthesis in all red-fleshed kiwifruit (Actinidia arguta) based on transcriptome analysis. Gene. 2018;648:31–41. doi: 10.1016/j.gene.2018.01.022. [DOI] [PubMed] [Google Scholar]
- 52.Liu Y, et al. Expression differences of pigment structural genes and transcription factors explain flesh coloration in three contrasting kiwifruit cultivars. Front. Plant Sci. 2017;8:1507. doi: 10.3389/fpls.2017.01507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nesi N, Jond C, Debeaujon I, Caboche M, Lepiniec L. The Arabidopsis TT2 gene encodes an R2R3 MYB domain protein that acts as a key determinant for proanthocyanidin accumulation in developing seed. Plant Cell. 2001;13:2099–2114. doi: 10.1105/TPC.010098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Deluc L, et al. Characterization of a grapevine R2R3-MYB transcription factor that regulates the phenylpropanoid pathway. Plant Physiol. 2006;140:499–511. doi: 10.1104/pp.105.067231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Johnson CS, Kolevski B, Smyth DR. TRANSPARENT TESTA GLABRA2, a trichome and seed coat development gene of Arabidopsis, encodes a WRKY transcription factor. Plant Cell. 2002;14:1359–1375. doi: 10.1105/tpc.001404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Matus JT, et al. A group of grapevine MYBA transcription factors located in chromosome 14 control anthocyanin synthesis in vegetative organs with different specificities compared with the berry color locus. Plant J. 2017;91:220–236. doi: 10.1111/tpj.13558. [DOI] [PubMed] [Google Scholar]
- 57.Butelli E, et al. Enrichment of tomato fruit with health-promoting anthocyanins by expression of select transcription factors. Nat. Biotechnol. 2008;26:1301. doi: 10.1038/nbt.1506. [DOI] [PubMed] [Google Scholar]
- 58.Bovy A, et al. High-flavonol tomatoes resulting from the heterologous expression of the maize transcription factor genes LC and C1. Plant Cell. 2002;14:2509–2526. doi: 10.1105/tpc.004218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Peng Y, et al. Kiwifruit with high anthocyanin content modulates NF-κB activation and reduces CCL11 secretion in human alveolar epithelial cells. J. Funct. Foods. 2020;65:103734. [Google Scholar]
- 60.Pojer E, Mattivi F, Johnson D, Stockley CS. The case for anthocyanin consumption to promote human health: A review. Compr. Rev. Food Sci. Food Saf. 2013;12:483–508. doi: 10.1111/1541-4337.12024. [DOI] [PubMed] [Google Scholar]
- 61.Alvarez-Suarez JM, et al. One-month strawberry-rich anthocyanin supplementation ameliorates cardiovascular risk, oxidative stress markers and platelet activation in humans. J. Nutr. Biochem. 2014;25:289–294. doi: 10.1016/j.jnutbio.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 62.Bose M, et al. The major green tea polyphenol,(−)-epigallocatechin-3-gallate, inhibits obesity, metabolic syndrome, and fatty liver disease in high-fat-fed mice. J. Nutr. 2008;138:1677–1683. doi: 10.1093/jn/138.9.1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Caton PW, et al. Regulation of vascular endothelial function by procyanidin-rich foods and beverages. J. Agric. Food Chem. 2010;58:4008–4013. doi: 10.1021/jf9031876. [DOI] [PubMed] [Google Scholar]
- 64.Corder R, et al. Endothelin-1 synthesis reduced by red wine. Nature. 2001;414:863–864. doi: 10.1038/414863a. [DOI] [PubMed] [Google Scholar]
- 65.Bushnell, B. BBMap: A Fast, Accurate, Splice-Aware Aligner. (Lawrence Berkeley National Lab. (LBNL), Berkeley, 2014).
- 66.Dobin A, et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics. 2013;29:15–21. doi: 10.1093/bioinformatics/bts635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hellens RP, et al. Transient expression vectors for functional genomics, quantification of promoter activity and RNA silencing in plants. Plant Methods. 2005;1:13. doi: 10.1186/1746-4811-1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang, T., Atkinson, R. & Janssen, B. Choice of Agrobacterium strain for transformation of kiwifruit. Acta Hortic. (753), 227–232 (2007).
- 69.Herath, D. et al. An improved method for transformation of Actinidia arguta utilized to demonstrate a central role for MYB110 in anthocyanin accumulation in kiwiberry. Plant Cell Tissue Org. Cult.10.1007/s11240-020-01915-1 (2020).
- 70.Nieuwenhuizen NJ, et al. Natural variation in monoterpene synthesis in kiwifruit: transcriptional regulation of terpene synthases by NAC and ETHYLENE-INSENSITIVE3-like transcription factors. Plant Physiol. 2015;167:1243–1258. doi: 10.1104/pp.114.254367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Shaw, O. M., Cooney, J., Sawyer, G. M., Dinnan, H. & Martell, S. Boysenberry and apple juice reduces acute lung inflammation through increased alternatively activated macrophage activity in an acute mouse model of allergic airways disease. bioRxiv10.1101/2020.03.03.974196 (2020). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.