Abstract
Background
Perioperative chemotherapy is a standard-of-care treatment for patients with gastric cancer. However, the impact of the postoperative chemotherapy (postCTX) component on overall survival (OS) is not well defined.
Methods
The National Cancer Database (NCDB) 2006–2014 was queried for patients who received preoperative chemotherapy (preCTX) and resection for gastric cancer. Analysis was performed to identify factors influencing receipt of postCTX. The impact of postCTX on OS was evaluated in propensity-matched groups.
Results
Among 3449 patients who received preCTX and resection for gastric cancer, 1091 (31.6%) received postCTX. Independent predictors of receiving postCTX were diagnosis after 2010 (odds ratio [OR] 1.985), distal tumor location (OR 1.348), and 15 or more lymph nodes examined (OR 1.214). Predictors of not receiving postCTX were older age (OR 0.985), comorbidity score higher than 1 (OR 0.592), and black race (OR 0.791). After propensity-matching (1091 per group), the median OS was 56.8 months for those who did receive postCTX versus 52.5 months for those who did not (p = 0.131). Subset analysis according to tumor grade, lymphovascular invasion, number of lymph nodes evaluated, T and N class, and AJCC stage identified an improvement in OS for the patients with N1 disease who received postCTX compared with those who did not (79.6 vs 41.3 months; p = 0.025). However, no other subgroup had a significant survival benefit.
Conclusions
Additional postCTX was administered to a minority of patients who received preCTX and gastrectomy for gastric cancer, and its influence on OS appeared to be limited. Future trials should aim to define patients who will benefit from postCTX.
Electronic supplementary material
The online version of this article (10.1245/s10434-020-09045-w) contains supplementary material, which is available to authorized users.
For patients with locoregional gastric cancer, multimodality therapy offers better progression-free survival (PFS) and overall survival (OS) than surgery alone.1,2 For patients with disease classified as cT2 or higher, the current National Comprehensive Cancer Network (NCCN) guidelines recommend perioperative chemotherapy as the preferred management strategy.3 Improved oncologic outcomes using this approach were first recognized with publication of the MAGIC trial in 2006.4 In that study, patients were randomized to receive three cycles of epirubicin, cisplatin, and fluorouracil (ECF) followed by surgery and then three additional cycles of chemotherapy versus surgery alone. Compared with the surgery-alone cohort, the perioperative chemotherapy group achieved significant improvement in PFS and OS.
Subsequent trials have used a perioperative approach in an effort to define the optimal combination of agents for treatment of patients with gastric cancer.5–9 Most recently, the FLOT4 study, a phase II/III randomized controlled trial, demonstrated higher rates of pathologic complete response and improved OS using a regimen of fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) compared with ECF for patients undergoing curative-intent resection for gastric cancer.8,9 As a result, FLOT has become the preferred perioperative regimen for these patients.
In the aforementioned studies, the rate for initiation and completion of preoperative chemotherapy is 90%.4–9 However, only 50% to 75% of patients who underwent preoperative chemotherapy and resection were able to initiate postoperative therapy, and in most cases, less than 50% completed all the intended therapy. Notably, in each of these studies, OS was analyzed in an intent-to-treat fashion based on the overall planned therapy, and no subset analyses were performed to compare outcomes between patients who initiated or completed postoperative therapy and those who received only preoperative therapy. These findings indicate that preoperative chemotherapy is likely most responsible for the improved outcomes observed.
The potential reasons for failure to receive postoperative therapy are poor performance status, delayed surgical recovery, postoperative complications, early disease progression, and patient preference. Given the observed low rates for initiation and completion of postoperative chemotherapy, its true effect on survival remains unclear. The current study aimed to evaluate factors associated with receipt of postoperative chemotherapy and better define its impact on survival for patients who received preoperative chemotherapy and underwent resection for gastric cancer.
Methods
Data Source
The National Cancer Database (NCDB) 2015 Participant User File (PUF) was the source of all analyzed data. Sponsored jointly by the American College of Surgeons and the American Cancer Society, the NCDB collects de-identified oncology outcomes data from Commission on Cancer facilities across the United States. Data are reported in a standardized fashion by Certified Tumor Registrars at each facility, and more than 70% of patients with new diagnoses are captured annually.10 Because the NCDB contains appropriately de-identified patient data, this study was deemed exempt by the University of Tennessee Health Science Center institutional review board.
Patient Selection
The NCDB was queried for all patients with gastric adenocarcinoma (International Classification of Diseases for Oncology [ICD-0-3] code numbers 8140, 8142, 8144, 8145, 8255, 8480, 8481, 8490) who received preoperative chemotherapy and underwent resection from 2006 to 2014. Patients were excluded if they had distant metastases (M1), were treated with palliative intent, or underwent R2 resection. Patients who received radiation therapy and those that underwent a gastric wedge resection or “local tumor destruction” also were excluded. Finally, patients who experienced 90-day postoperative mortality or had fewer than 90 days of follow-up evaluation and those with missing values for key outcomes or variables used during propensity-matching were excluded (Fig. S1).
Both T and N classifications were based on the American Joint Committee on Cancer (AJCC) eighth edition staging for gastric carcinoma. Pre- and postoperative chemotherapy was defined as the receipt of neoadjuvant and adjuvant systemic chemotherapy, respectively, and determined by the systemic therapy-surgery sequencing variable.
The patients who received postoperative chemotherapy and those who did not after preoperative chemotherapy and gastrectomy were compared with regard to sociodemographic and clinicopathologic factors including age, gender, race, year of diagnosis, insurance status, Charlson-Deyo Comorbidity Index (CDCI), type of surgical procedure performed (partial, total, extended total gastrectomy), primary tumor site (proximal: cardia/fundus; middle: lesser curve/greater curve/body; distal: antrum/pylorus), facility type (academic vs nonacademic), AJCC T and N classification, AJCC stage (ypTNM), number of lymph nodes (LN) examined (< 15 or ≥ 15), tumor grade, lymphovascular invasion (LVI), and resection margin status.
Overall survival (OS) rates for the two groups were compared after propensity score-matching. Finally, a subset analysis was performed based on receipt of postoperative chemotherapy and included tumor grade, LVI, AJCC T and N classification, AJCC stage, margin status, and number of lymph nodes examined.
Statistical Analysis
First, univariable analysis of patient characteristics based on receipt of postoperative chemotherapy was performed. Patient characteristics were reported as medians with interquartile ranges for continuous variables and as frequencies with percentages for categorical variables. Pearson’s Chi square was used to compare categorical variables, and the Wilcoxon rank-sum test was used to compare continuous variables. Any patient characteristic with a p value of 0.2 or lower in the univariable analysis was included in the subsequent multivariable model. A multivariable logistic regression then was performed to identify patient characteristics independently associated with the receipt of postoperative chemotherapy. These data were reported as odds ratios (ORs) with 95% confidence intervals (CIs). Reference variables are indicated as such.
Overall survival was defined as the time from diagnosis to death or censoring. The univariate comparison of OS was performed with a log-rank test using the Kaplan–Meier method and reported in months for median survival and percentages for 5-year survival.
For propensity-matching, 1:1 nearest-neighbor matching was performed using calipers equal to 0.2 of the standard deviation of the logit of the propensity score to create balanced cohorts based on propensity scores generated using a logistic regression model that accounted for key demographic and clinicopathologic covariates (age, gender, race, year of diagnosis, insurance, comorbidities, procedure, tumor location, facility type, facility volume, T and N classifications, AJCC stage [ypTNM], LNs examined, tumor grade, LVI, and resection margin).
Additional comparisons were performed after sub-stratification using univariate methods for OS. Statistical analysis was performed with SPSS statistical software package version 25 (IBM Corp, 2017; Armonk, NY, USA), and significance was defined as a p value lower than 0.05.
Results
Characteristics of the Patients Who Received Preoperative Chemotherapy and Underwent Resection for Gastric Cancer
In the database, 3449 patients with resectable gastric cancer who received preoperative chemotherapy and underwent resection were identified (Table 1). The median age of these patients at diagnosis was 63 years. The patients were more frequently male (66.9%), white (75.4%), likely to have private insurance (46.2%) or Medicare (40.6%), without comorbid disease (72.8%), and treated at an academic facility (57.7%). The majority of the patients had a diagnosis between 2010 and 2014 (73.6%), and the remaining patients had a diagnosis between 2006 and 2009 (26.4%).
Table 1.
(a) Univariable and (b) multivariable comparisons of sociodemographic and clinicopathologic characteristics of patients receiving preoperative chemotherapy and gastrectomy for gastric adenocarcinoma who did and did not receive postoperative chemotherapy
| (a) Characteristic | Overall (n = 3449) n (%) |
No postoperative chemotherapy (n = 2358) n (%) |
Postoperative chemotherapy (n = 1091) n (%) |
p Value | (b) OR | 95% CI | p Value |
|---|---|---|---|---|---|---|---|
| Median age: years (IQR) | 63 (46–79) | 64 (48–80) | 61 (44–78) | <0.001 | 0.985 | (0.975–0.995) | 0.002 |
| Sex | 0.101 | ||||||
| Male | 2308 (66.9) | 1599 (67.8) | 709 (65.0) | Ref | – | – | |
| Female | 1141 (33.1) | 759 (32.2) | 382 (35.0) | 1.021 | (0.867–1.203) | 0.803 | |
| Race | 0.026 | ||||||
| White | 2600 (75.4) | 1787 (75.8) | 813 (74.5) | Ref | – | – | |
| Black | 514 (14.9) | 365 (15.5) | 149 (13.7) | 0.793 | (0.634–0.993) | 0.043 | |
| Asian | 257 (7.5) | 157 (6.7) | 100 (9.2) | 1.192 | (0.898–1.583) | 0.224 | |
| Other | 78 (2.3) | 49 (2.1) | 29 (2.7) | 0.999 | (0.613–1.626) | 0.995 | |
| Year of diagnosis | <0.001 | ||||||
| 2006–2009 | 909 (26.4) | 704 (29.9) | 205 (18.8) | Ref | – | – | |
| 2010–2014 | 2540 (73.6) | 1654 (70.1) | 886 (81.2) | 1.951 | (1.511–2.518) | <0.001 | |
| Insurance status | <0.001 | ||||||
| No insurance | 128 (3.7) | 88 (3.7) | 40 (3.7) | 0.730 | (0.483–1.102) | 0.134 | |
| Private insurance | 1594 (46.2) | 1024 (43.4) | 570 (52.2) | Ref | – | – | |
| Medicare | 1401 (40.6) | 1029 (43.6) | 372 (34.1) | 0.820 | (0.667–1.009) | 0.055 | |
| Medicaid/other government aid | 326 (9.5) | 217 (9.2) | 109 (10.0) | 0.874 | (0.672–1.136) | 0.266 | |
| Charlson-Deyo Comorbidity score | 0.001 | ||||||
| 0 | 2510 (72.8) | 1678 (71.2) | 832 (76.3) | Ref | – | – | |
| 1 | 749 (21.7) | 531 (22.5) | 218 (20.0) | 0.866 | (0.718–1.044) | 0.130 | |
| > 1 | 190 (5.5) | 149 (6.3) | 41 (3.8) | 0.601 | (0.418–0.865) | 0.006 | |
| Surgical procedure | 0.906 | ||||||
| Partial gastrectomy (excluding wedge) | 1221 (35.4) | 829 (35.2) | 392 (35.9) | ||||
| Total gastrectomy | 1736 (50.3) | 1191 (50.5) | 545 (50.0) | ||||
| Extended gastrectomy | 492 (14.3) | 338 (14.3) | 154 (14.1) | ||||
| Primary tumor site | 0.024 | ||||||
| Proximal | 1293 (37.5) | 914 (38.8) | 379 (34.7) | Ref | – | – | |
| Middle | 820 (23.8) | 556 (23.6) | 264 (24.2) | 1.112 | (0.905–1.367) | 0.313 | |
| Distal | 741 (21.5) | 476 (20.2) | 265 (24.3) | 1.355 | (1.096–1.676) | 0.005 | |
| Unspecified | 595 (17.3) | 412 (17.5) | 183 (16.8) | 1.059 | (0.842–1.332) | 0.624 | |
| Facility type | 0.002 | ||||||
| Academic | 1990 (57.7) | 1348 (57.2) | 642 (58.8) | Ref | – | – | |
| Other | 1321 (38.3) | 932 (39.5) | 389 (35.7) | 0.955 | (0.815–1.120) | 0.574 | |
| Unknown | 138 (4.0) | 78 (3.3) | 60 (5.5) | 0.926 | (0.603–1.422) | 0.724 | |
| AJCC tumor classification | 0.006 | ||||||
| T1 | 371 (10.8) | 278 (11.8) | 93 (8.5) | Ref | – | – | |
| T2 | 454 (13.2) | 317 (13.4) | 137 (12.6) | 1.209 | (0.853–1.713) | 0.285 | |
| T3 | 1719 (49.8) | 1136 (48.2) | 583 (53.4) | 1.389 | (0.848–2.276) | 0.192 | |
| T4 | 905 (26.2) | 627 (26.6) | 278 (25.5) | 1.234 | (0.712–2.139) | 0.454 | |
| AJCC nodal classification | 0.061 | ||||||
| NX | 103 (3.0) | 76 (3.2) | 27 (2.5) | ||||
| N0 | 1546 (44.8) | 1091 (46.3) | 455 (41.7) | Ref | – | – | |
| N1 | 678 (19.7) | 456 (19.3) | 222 (20.3) | 1.203 | (0.954–1.518) | 0.118 | |
| N2 | 578 (16.8) | 381 (16.2) | 197 (18.1) | 1.173 | (0.800–1.720) | 0.413 | |
| N3a | 386 (11.2) | 256 (10.9) | 130 (11.9) | 1.035 | (0.680–1.575) | 0.873 | |
| N3b | 158 (4.6) | 98 (4.2) | 60 (5.5) | 1.246 | (0.766–2.028) | 0.375 | |
| AJCC stage (ypTNM)a | 0.008 | ||||||
| I | 590 (17.7) | 431 (19.0) | 159 (15.1) | Ref | – | – | |
| II | 1490 (44.7) | 1020 (44.9) | 470 (44.5) | 0.940 | (0.600–1.472) | 0.786 | |
| III | 1250 (37.5) | 823 (36.2) | 427 (40.4) | 1.003 | (0.513–1.964) | 0.992 | |
| Lymph nodes examined | <0.001 | ||||||
| < 15 | 1172 (34.0) | 852 (36.1) | 320 (29.3) | Ref | – | – | |
| ≥ 15 | 2277 (66.0) | 1506 (63.9) | 771 (70.7) | 1.215 | (1.025–1.440) | 0.025 | |
| Grade | 0.003 | ||||||
| Well-/moderately differentiated | 901 (26.1) | 655 (27.8) | 246 (22.5) | Ref | – | – | |
| Poorly differentiated/undifferentiated | 2272 (65.9) | 1511 (64.1) | 761 (69.8) | 1.165 | (0.969–1.399) | 0.104 | |
| Undetermined/unknown | 276 (8.0) | 192 (8.1) | 84 (7.7) | 1.078 | (0.786–1.478) | 0.641 | |
| Lymphovascular invasion | <0.001 | ||||||
| Not present | 1174 (34.0) | 779 (33.0) | 395 (36.2) | Ref | – | – | |
| Present | 824 (23.9) | 529 (22.4) | 295 (27.0) | 0.956 | (0.774–1.182) | 0.679 | |
| Unknown | 1451 (42.1) | 1050 (44.5) | 401 (36.8) | 1.051 | (0.838–1.318) | 0.666 | |
| Unplanned readmission | 170 (4.9) | 122 (5.2) | 48 (4.4) | 0.329 | |||
| Margin status | 0.608 | ||||||
| R0 | 3194 (92.6) | 2180 (92.5) | 1014 (92.9) | ||||
| R1 | 255 (7.4) | 178 (7.5) | 77 (7.1) | Favors no chemo | ←1→ | Favors chemo |
OR odds ratio, CI confidence interval, IQR interquartile range, AJCC American Joint Committee on Cancer
aPatients labeled as T4 with no subclassification or NX were excluded
The most common resection performed was total gastrectomy (50.3%), and the most common tumor location was the proximal one third of the stomach (37.5%). On pathologic review, T3 tumors (49.8%) and N0 disease (44.8%) were the most common. Additionally, most of the patients had R0 resection (92.6%) and 15 lymph nodes or more examined (66%).
Use of Postoperative Chemotherapy Among the Patients Receiving Preoperative Chemotherapy and Resection for Gastric Cancer
Of the 3449 patients in this cohort, only 1091 (31.6%) received postoperative chemotherapy. In the univariate analysis (Table 1a), the sociodemographic factors significantly associated with the receipt of postoperative chemotherapy were younger age, race, private insurance, comorbidity score of 0, treatment at an academic facility, and diagnosis between 2010 and 2014 (p < 0.05). The clinicopathologic factors significantly associated with receipt of postoperative chemotherapy were tumors located in the distal one third of the stomach, T3 tumors, stage 3 disease, examination of 15 LNs or more, poor tumor differentiation, and presence of LVI (p < 0.05).
In the multivariable analysis (Table 1b), the independent predictors of receiving postCTX were diagnosis after 2010 (OR 1.951; 95% CI 1.511–2.518; p ≤ 0.001), distal tumor location (OR 1.355; 95% CI 1.096–1.676; p = 0.005), and examination of 15 LNs or more (OR 1.215; 95% CI 1.025–1.440; p = 0.025). Independent predictors of not receiving postCTX were older age (OR 0.985; 95% CI 0.975–0.995; p = 0.002), comorbidity score higher than 1 (OR 0.601; 95% CI 0.418–0.865; p = 0.006), and black race (OR 0.793; 95% CI 0.634–0.993; p = 0.043).
Influence of Postoperative Chemotherapy on Survival
Given the clinicopathologic differences between those who did and those who did not receive postoperative chemotherapy, propensity-matching was performed to establish balanced cohorts for survival analysis. After matching for sociodemographic and tumor-related factors (Table 2), the median OS was 56.8 months for those who received additional postoperative chemotherapy and 52.5 months for those who did not (p = 0.131) (Fig. 1). The 5-year OS was 48.9% for the patients who received additional postoperative chemotherapy and 47.3% for those who did not.
Table 2.
Comparison of sociodemographic and clinicopathologic characteristics of patients receiving preoperative chemotherapy and gastrectomy for gastric adenocarcinoma who did and did not receive postoperative chemotherapy after propensity-matching
| Characteristic | No postoperative chemotherapy (n = 1091) n (%) |
Postoperative chemotherapy (n = 1091) n (%) |
p value |
|---|---|---|---|
| Median age: years (IQR) | 61 (44–78) | 61 (44–78) | 0.668 |
| Sex | 0.685 | ||
| Male | 718 (65.8) | 709 (65.0) | |
| Female | 373 (34.2) | 382 (35.0) | |
| Race | 0.823 | ||
| White | 803 (73.6) | 813 (74.5) | |
| Black | 150 (13.7) | 149 (13.7) | |
| Asian | 112 (10.3) | 100 (9.2) | |
| Other | 26 (2.4) | 29 (2.7) | |
| Year of diagnosis | 0.827 | ||
| 2006–2009 | 209 (19.2) | 205 (18.8) | |
| 2010–2014 | 882 (80.8) | 886 (81.2) | |
| Insurance status | 0.674 | ||
| No insurance | 48 (4.4) | 40 (3.7) | |
| Private insurance | 546 (50.0) | 570 (52.2) | |
| Medicare | 382 (35.0) | 372 (34.1) | |
| Medicaid/other government aid | 115 (10.5) | 109 (10.0) | |
| Charlson-Deyo Comorbidity score | 0.747 | ||
| 0 | 817 (74.9) | 832 (76.3) | |
| 1 | 232 (21.3) | 218 (20.0) | |
| > 1 | 42 (3.8) | 41 (3.8) | |
| Surgical procedure performed | 0.888 | ||
| Partial gastrectomy (excluding wedge) | 386 (35.4) | 392 (35.9) | |
| Total gastrectomy | 556 (51.0) | 545 (50.0) | |
| Extended gastrectomy | 149 (13.7) | 154 (14.1) | |
| Primary tumor site | 0.752 | ||
| Proximal | 402 (36.8) | 379 (34.7) | |
| Middle | 250 (22.9) | 264 (24.2) | |
| Distal | 263 (24.1) | 265 (24.3) | |
| Unspecified | 176 (16.1) | 183 (16.8) | |
| Facility type | 0.618 | ||
| Academic | 650 (59.6) | 642 (58.8) | |
| Other | 391 (35.8) | 389 (35.7) | |
| Unknown | 50 (4.6) | 60 (5.5) | |
| AJCC tumor classification | 0.939 | ||
| T1 | 94 (8.6) | 93 (8.5) | |
| T2 | 134 (12.3) | 137 (12.6) | |
| T3 | 596 (54.6) | 583 (53.4) | |
| T4 | 267 (24.5) | 278 (25.5) | |
| AJCC nodal classification | 0.821 | ||
| NX | 20 (1.8) | 27 (2.5) | |
| N0 | 470 (43.1) | 455 (41.7) | |
| N1 | 211 (19.3) | 222 (20.3) | |
| N2 | 209 (19.2) | 197 (18.1) | |
| N3a | 126 (11.5) | 130 (11.9) | |
| N3b | 55 (5.0) | 60 (5.5) | |
| AJCC stage (ypTNM)a | 0.855 | ||
| I | 159 (14.9) | 159 (15.1) | |
| II | 487 (45.7) | 470 (44.5) | |
| III | 420 (39.4) | 427 (40.4) | |
| Lymph nodes examined | 0.851 | ||
| < 15 | 324 (29.7) | 320 (29.3) | |
| ≥ 15 | 767 (70.3) | 771 (70.7) | |
| Grade | 0.873 | ||
| Well-/moderately differentiated | 253 (23.2) | 246 (22.5) | |
| Poorly differentiated/undifferentiated | 750 (68.7) | 761 (69.8) | |
| Undetermined/unknown | 88 (8.1) | 84 (7.7) | |
| Lymphovascular invasion | 0.798 | ||
| Not present | 387 (35.5) | 395 (36.2) | |
| Present | 309 (28.3) | 295 (27.0) | |
| Unknown | 395 (36.2) | 401 (36.8) | |
| Margin status | 0.550 | ||
| R0 | 1021 (93.6) | 1014 (92.9) | |
| R1 | 70 (6.4) | 77 (7.1) |
IQR, interquartile range; AJCC, American Joint Committee on Cancer
aPatients labeled as T4 with no subclassification or NX were excluded
Fig. 1.
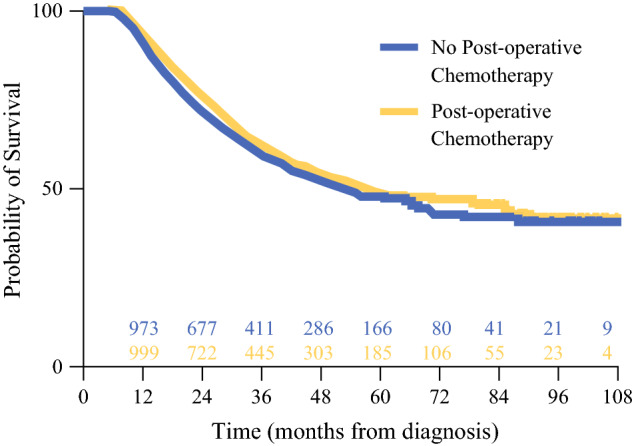
Kaplan-Meier plots for propensity-matched patients who received postoperative therapy and those who did not after preoperative chemotherapy and resection for gastric cancer
To further identify the potential subsets of patients more likely to benefit from postoperative chemotherapy, a sub-stratified survival analysis was performed based on histopathologic features including T and N subclassifications, overall AJCC stage, resection margins, and number of LNs evaluated. As shown in Table 3, the patients with favorable histopathologic features (well-differentiated/moderately differentiated tumors, no LVI, R0 resection) and those with lower T and N classifications (T1/T2 and N0) achieved excellent survival whether they received postoperative chemotherapy or not. On the other hand, the patients with unfavorable histopathology (poorly differentiated/undifferentiated tumors, positive LVI, R1 resection) and those with high T and N classifications (T4 and N2/N3) had equally poor survival regardless of additional therapy. The only subgroup for which a significant difference in OS was detected comprised patients with N1 disease, where those who received postoperative chemotherapy and had a median OS almost double that for those who did not (79.6 vs 41.3 months; p = 0.025). There was a trend toward improved survival for the patients with T3 tumors who received postoperative chemotherapy (54.1 vs 41.3 months; p = 0.61). The analysis by overall AJCC stage (ypTNM) showed that additional postoperative therapy did not significantly benefit patients with stage (I, II, III) disease. Moreover, further stratification by substage groupings did not show any particular category of patients who had a demonstrable survival advantage with additional chemotherapy (Table S1).
Table 3.
Subset survival analysis of tumor-related factors for patients who did not and did receive postoperative chemotherapy
| Characteristic | No postoperative chemotherapy | Postoperative chemotherapy | p value |
|---|---|---|---|
| Median OS Months (95% CI) | |||
| Tumor differentiation | |||
| Well-/moderately differentiated | 111.54 (50.491–172.590) | NR | 0.850 |
| Poorly/undifferentiated | 41.20 (34.978–47.422) | 45.86 (37.871–53.849) | 0.220 |
| Lymphovascular invasion | |||
| Absent | NR | NR | 0.156 |
| Present | 32.72 (26.910–38.530) | 30.62 (24.525–36.715) | 0.969 |
| T class | |||
| T1 | NR | NR | 0.502 |
| T2 | NR | NR | 0.524 |
| T3 | 45.14 (33.465–56.815) | 54.08 (43.188–64.972) | 0.061 |
| T4 | 29.80 (24.257–35.343) | 32.16 (27.608–36.712) | 0.540 |
| N class | |||
| N0 | 111.54 (73.08–149.995) | NR | 0.415 |
| N1 | 41.30 (35.238–47.362) | 79.57 (50.900–108.240) | 0.025 |
| N2 | 31.24 (24.171–38.309) | 31.41 (26.274–36.546) | 0.594 |
| N3a | 25.66 (22.751–28.569) | 21.68 (16.001–27.359) | 0.608 |
| N3b | 16.76 (14.623–18.897) | 19.78 (14.529–25.031) | 0.574 |
| AJCC stage (ypTNM)a | |||
| I | NR | NR | 0.661 |
| II | NR | 85.16 (66.113–104.207) | 0.218 |
| III | 26.32 (23.382–29.258) | 29.63 (25.552–29.708) | 0.194 |
| Resection margin | |||
| R0 | 57.76 (46.778–68.742) | 70.11 (55.230–84.990) | 0.096 |
| R1 | 19.65 (13.180–26.120) | 20.57 (13.001–28.139) | 0.962 |
| Lymph nodes examined | |||
| < 15 | 49.91 (41.457–58.363) | 60.78 (n/a) | 0.057 |
| ≥ 15 | 55.23 (44.726–65.734) | 56.15 (41.312–70.988) | 0.569 |
OS overall survival, CI confidence interval, NR not reached, AJCC American Joint Committee on Cancer
aPatients labeled as T4 with no subclassification or NX were excluded
Discussion
Gastric cancer is an aggressive malignancy, and systemic micrometastases are likely present in most patients with clinical evidence of local or locoregional disease. Thus, in addition to gastrectomy and regional lymphadenectomy, chemotherapy is a fundamental component in the management of these patients.
Given the potential morbidity of gastrectomy as well as the postoperative challenges of physical and nutritional recovery, reliance on adjuvant therapy alone places patients at risk for not receiving any curative-intent chemotherapy. This is evident by the relatively high rates of patients who fail to initiate or complete postoperative treatment in adjuvant therapy trials.11,12
Based on prospective randomized data,4,5 perioperative chemotherapy is the preferred treatment strategy for patients with surgically resectable gastric cancer in the United States3 and other Western countries. The strength of this “sandwich” approach is the assurance that patients receive at least some systemic therapy. However, many patients fail to receive the intended postoperative chemotherapy, calling into question its importance in the overall management of gastric cancer.
In the current analysis, postoperative chemotherapy was administered to a minority of patients who underwent preoperative chemotherapy and gastrectomy and did not experience better OS than a matched cohort of patients who did not receive postoperative chemotherapy.
The factors associated with non-receipt of postoperative chemotherapy were primarily sociodemographic. It was not surprising that older patients and those with higher comorbidity scores were less likely to receive postoperative treatment. This result likely was attributable to greater frailty after preoperative chemotherapy and gastrectomy and is consistent with previous studies.13–16
The current study also identified black patients as less likely to receive postoperative chemotherapy. Although the reasons for this are not entirely clear, previous national studies have demonstrated socioeconomic and racial disparities in the multi-modal treatment of gastric cancer.17–19 Although the NCDB does not capture data regarding surgical complications, it is well established that morbidity associated with gastrectomy limits the receipt of postoperative chemotherapy and can have an adverse impact on prognosis.20–22
The results from prior studies examining the influence of postoperative chemotherapy on patients intended to receive perioperative chemotherapy for gastroesophageal cancers have been variable. Similar to the current analysis, at least two studies from European centers found no significant difference in survival between patients who received postoperative chemotherapy after preoperative chemotherapy and resection and those who did not.16,23
However, other studies have reported improved survival with postoperative chemotherapy. In a single-institution study from the United Kingdom, Mirza et al.24 found that completion of neoadjuvant and adjuvant therapy was independently associated with improved survival. In a French cohort, Luc et al.13 detected an improvement in survival provided at least two cycles of postoperative chemotherapy were administered. In a study from Germany, Glatz et al.14 found that regardless of the number of cycles administered, postoperative chemotherapy significantly improved OS compared with its omission. Finally, in an analysis by Van Putten et al.15 using the Netherlands Cancer Registry, perioperative therapy resulted in a 16% decrease in risk of death (HR 0.84; 95% CI 0.71–0.99) compared with preoperative therapy alone. Notably, some of the patients in that study received postoperative chemoradiation.
Given the absence of improved OS in the entire cohort of the current study, subset analysis was performed to explore for groups of patients that may benefit from postope0rative chemotherapy (Table 3). These findings suggested a potential intersection between the benefit of additional chemotherapy and disease biology. The patients with very good disease biology did well, and the patients with very bad disease biology did poorly irrespective of added chemotherapy, whereas a group of patients with “intermediate” disease biology may have received benefit from further treatment. Admittedly, however, the current data do not permit detailed guidance on who should and who should not receive additional therapy.
Other studies also have investigated to identify subgroups most likely to receive a benefit from additional postoperative chemotherapy, but the findings are variable. For example, in an analysis of 333 patients from Nottingham University Hospitals in the United Kingdom, Saunders et al.25 found that administration of postoperative chemotherapy resulted in improved OS for the patients who had a histopathologic response to preoperative chemotherapy (Manard Tumor Regression grades 1 to 3), whereas those with little to no response (Manard Tumor Regression grades 4 to 5) experienced no OS benefit. However, a study of 134 patients from Germany published the same year reported that the patients with a poor histopathologic response to therapy (> 50% viable tumor cells) and node-positive disease benefited from postoperative chemotherapy, whereas the patients with a good histopathologic response to therapy (< 50% viable tumor cells) and node-negative disease did not.14 A separate and more recent study from Germany found that postoperative chemotherapy resulted in improved recurrence-free survival but not OS for patients with non-intestinal Lauren classification and those receiving FLOT, but no differences were found for patients stratified by age, ypT or ypN classification, or histologic response to preoperative chemotherapy.16
Collectively, the current analysis and the aforementioned studies do not provide conclusive evidence regarding the benefit of postoperative chemotherapy or the lack thereof for patients receiving preoperative therapy and curative-intent resection for gastric cancer. The inconsistencies are likely multifactorial. All these studies were retrospective, and many were small, single-institution investigations. Moreover, the patients treated in these different studies had varying demographics and stages of disease. Some of the studies included patients with lower esophageal cancer, and the chemotherapy regimens were varied. Ultimately, multicenter prospective randomized trials are needed to further define the influence of postoperative chemotherapy for patients who receive preoperative chemotherapy and curative-intent gastrectomy.
With the expectation that many patients will not be able to initiate or complete additional postoperative therapy, the use of a total neoadjuvant approach merits consideration. In a recent phase 2 study, 59 patients with clinical T3/T4 and/or node-positive gastroesophageal adenocarcinoma were treated with a protracted course (6 cycles) of neoadjuvant FLOT with no intent to receive postoperative therapy.26 Of the 50 patients who underwent surgery, 43 (86%) achieved R0 resection and 20 (40%) had either a complete or near complete (< 10% viable tumor) pathologic response. Furthermore, use of this intensified neoadjuvant therapy did not result in greater perioperative morbidity than experienced by patients receiving standard neoadjuvant chemotherapy or upfront resection.27 In a retrospective review of gastric cancer patients treated at Memorial Sloan Kettering Cancer Center, a total neoadjuvant therapy approach was found to result in increased rates of intended chemotherapy delivered and no increase in perioperative complications or length of hospital stay compared with those experienced by patients receiving perioperative chemotherapy.28 Although these results are encouraging, larger prospective comparative studies are needed to further delineate the optimal chemotherapy-surgery sequence for gastric cancer.
The current study had several limitations. It was a retrospective analysis, and the NCDB does not capture several pertinent data points. Notably, the NCDB does not capture information regarding the type or number of chemotherapy cycles or the radiographic or histopathologic response to preoperative therapy. It also contains no information regarding endoscopic or laparoscopic staging, surgeon intent to perform extended lymphadenectomy, the presence or absence of perineural invasion, or the incidence and severity of postoperative complications. Finally, given the period during which the patients in this study were treated, most of them likely received regimens such as ECF, ECX, EOX, and FOLFOX. Because of its superiority over these regimens, FLOT currently is the recommended first-line perioperative chemotherapy, and it is recognized that the current study could not account for how its increasing use would have influenced the results observed. Finally, the NCDB does not contain data on recurrence or disease-free survival.
To our knowledge this is the largest study of a U.S. population to examine the utilization and impact of postoperative chemotherapy after preoperative chemotherapy and resection for gastric cancer. The study showed that less than one third of the patients who underwent neoadjuvant therapy received additional treatment postoperatively. The comparison of propensity-matched groups showed no difference in OS between those who did and those who did not receive postoperative chemotherapy, although the subset analysis found that patients with a low metastatic nodal burden did benefit from additional treatment. Given the significant challenges with the administration of postoperative therapy in controlled trials and clinical practice, future trial design should focus on further delineating which patients will benefit from further treatment as well as consideration of a total neoadjuvant therapy approach.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Selection criteria for the study (PDF 34 kb)
Disclosure
There are no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Al-Batran SE, Lorenzen S. Management of locally advanced gastroesophageal cancer: still a multidisciplinary global challenge? Hematol Oncol Clin North Am. 2017;31:441–452. doi: 10.1016/j.hoc.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 2.Cai Z, Yin Y, Shen C, et al. Comparative effectiveness of preoperative, postoperative, and perioperative treatments for resectable gastric cancer: a network meta-analysis of the literature from the past 20 years. Surg Oncol. 2018;27:563–574. doi: 10.1016/j.suronc.2018.07.011. [DOI] [PubMed] [Google Scholar]
- 3.Network NCC. Gastric Cancer (version 1.2020). http://www.nccn.org/professionals/physician_gls/pdf/gastric_blocks.pdf. Accessed 21 June 2020.
- 4.Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11–20. doi: 10.1056/NEJMoa055531. [DOI] [PubMed] [Google Scholar]
- 5.Ychou M, Boige V, Pignon JP, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol. 2011;29:1715–1721. doi: 10.1200/JCO.2010.33.0597. [DOI] [PubMed] [Google Scholar]
- 6.Cunningham D, Stenning SP, Smyth EC, et al. Perioperative chemotherapy with or without bevacizumab in operable oesophagogastric adenocarcinoma (UK Medical Research Council ST03): primary analysis results of a multicentre, open-label, randomised phase 2–3 trial. Lancet Oncol. 2017;18:357–370. doi: 10.1016/S1470-2045(17)30043-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cats A, Jansen EPM, van Grieken NCT, et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): an international, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:616–628. doi: 10.1016/S1470-2045(18)30132-3. [DOI] [PubMed] [Google Scholar]
- 8.Al-Batran SE, Hofheinz RD, Pauligk C, et al. Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): results from the phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol. 2016;17:1697–1708. doi: 10.1016/S1470-2045(16)30531-9. [DOI] [PubMed] [Google Scholar]
- 9.Al-Batran SE, Homann N, Pauligk C, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet. 2019;393:1948–1957. doi: 10.1016/S0140-6736(18)32557-1. [DOI] [PubMed] [Google Scholar]
- 10.Bilimoria KY, Stewart AK, Winchester DP, Ko CY. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15:683–690. doi: 10.1245/s10434-007-9747-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Macdonald JS, Smalley SR, Benedetti J, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345:725–730. doi: 10.1056/NEJMoa010187. [DOI] [PubMed] [Google Scholar]
- 12.Bang YJ, Kim YW, Yang HK, et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): a phase 3 open-label, randomised controlled trial. Lancet. 2012;379:315–321. doi: 10.1016/S0140-6736(11)61873-4. [DOI] [PubMed] [Google Scholar]
- 13.Luc G, Gersen-Cherdieu H, Degrandi O, Terrebonne E, Chiche L, Collet D. Impact of postoperative chemotherapy in patients with locally advanced gastroesophageal adenocarcinoma treated with perioperative chemotherapy strategy. Am J Surg. 2015;210:15–23. doi: 10.1016/j.amjsurg.2014.12.036. [DOI] [PubMed] [Google Scholar]
- 14.Glatz T, Bronsert P, Schafer M, et al. Perioperative platin-based chemotherapy for locally advanced esophagogastric adenocarcinoma: postoperative chemotherapy has a substantial impact on outcome. Eur J Surg Oncol. 2015;41:1300–1307. doi: 10.1016/j.ejso.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 15.van Putten M, Lemmens V, van Laarhoven HWM, Pruijt HFM, Nieuwenhuijzen GAP, Verhoeven RHA. Poor compliance with perioperative chemotherapy for resectable gastric cancer and its impact on survival. Eur J Surg Oncol. 2019;45:1926–1933. doi: 10.1016/j.ejso.2019.03.040. [DOI] [PubMed] [Google Scholar]
- 16.Sisic L, Blank S, Nienhuser H, et al. The postoperative part of perioperative chemotherapy fails to provide a survival benefit in completely resected esophagogastric adenocarcinoma. Surg Oncol. 2017;33:177–88. [DOI] [PubMed]
- 17.Stessin AM, Sherr DL. Demographic disparities in patterns of care and survival outcomes for patients with resected gastric adenocarcinoma. Cancer Epidemiol Biomarkers Prev. 2011;20:223–233. doi: 10.1158/1055-9965.EPI-10-0158. [DOI] [PubMed] [Google Scholar]
- 18.Al-Refaie WB, Gay G, Virnig BA, et al. Variations in gastric cancer care: a trend beyond racial disparities. Cancer. 2010;116:465–475. doi: 10.1002/cncr.24772. [DOI] [PubMed] [Google Scholar]
- 19.Ikoma N, Cormier JN, Feig B, et al. Racial disparities in preoperative chemotherapy use in gastric cancer patients in the United States: analysis of the National Cancer Data Base, 2006–2014. Cancer. 2018;124:998–1007. doi: 10.1002/cncr.31155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Papenfuss WA, Kukar M, Oxenberg J, et al. Morbidity and mortality associated with gastrectomy for gastric cancer. Ann Surg Oncol. 2014;21:3008–3014. doi: 10.1245/s10434-014-3664-z. [DOI] [PubMed] [Google Scholar]
- 21.Jin LX, Sanford DE, Squires MH III, et al. Interaction of postoperative morbidity and receipt of adjuvant therapy on long-term survival after resection for gastric adenocarcinoma: results from the U.S. Gastric Cancer Collaborative. Ann Surg Oncol. 2016;23:2398–408. [DOI] [PubMed]
- 22.Tokunaga M, Tanizawa Y, Bando E, Kawamura T, Terashima M. Poor survival rate in patients with postoperative intra-abdominal infectious complications following curative gastrectomy for gastric cancer. Ann Surg Oncol. 2013;20:1575–1583. doi: 10.1245/s10434-012-2720-9. [DOI] [PubMed] [Google Scholar]
- 23.Papaxoinis G, Kamposioras K, Weaver JMJ, et al. The role of continuing perioperative chemotherapy post surgery in patients with esophageal or gastroesophageal junction adenocarcinoma: a multicenter cohort study. J Gastrointest Surg. 2019;23:1729–1741. doi: 10.1007/s11605-018-04087-8. [DOI] [PubMed] [Google Scholar]
- 24.Mirza A, Pritchard S, Welch I. The postoperative component of MAGIC chemotherapy is associated with improved prognosis following surgical resection in gastric and gastrooesophageal junction adenocarcinomas. Int J Surg Oncol. 2013;2013:781742. doi: 10.1155/2013/781742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saunders JH, Bowman CR, Reece-Smith AM, et al. The role of adjuvant platinum-based chemotherapy in esophagogastric cancer patients who received neoadjuvant chemotherapy prior to definitive surgery. J Surg Oncol. 2017;115:821–829. doi: 10.1002/jso.24601. [DOI] [PubMed] [Google Scholar]
- 26.Schulz C, Kullmann F, Kunzmann V, et al. NeoFLOT: multicenter phase II study of perioperative chemotherapy in resectable adenocarcinoma of the gastroesophageal junction or gastric adenocarcinoma: very good response predominantly in patients with intestinal type tumors. Int J Cancer. 2015;137:678–685. doi: 10.1002/ijc.29403. [DOI] [PubMed] [Google Scholar]
- 27.Ganschow P, Hofmann L, Stintzing S, et al. Operative results and perioperative morbidity after intensified neoadjuvant chemotherapy with FLOT for gastroesophageal adenocarcinoma: impact of intensified neoadjuvant treatment. J Gastrointest Surg. 2020. 10.1007/s11605-01904511-7. Online ahead of print. [DOI] [PubMed]
- 28.Greally M, Strong VE, Yoon SY, et al. Total neoadjuvant chemo (ctx; TNT) for locally advanced gastric cancer (GC): the Memorial Sloan Kettering Cancer Center experience. J Clin Oncol. 2019;37(15_Suppl):4046.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Selection criteria for the study (PDF 34 kb)


