The digestive tract can be targeted by SARS-CoV-2 and its interactions with the immune system, and the lungs may have meaningful implications in the pathogenesis of COVID-19. This gastrointestinal involvement needs to be considered in clinical practice.
Keywords: immune-mediated disease, lung-gut axis, microbiota, immunomodulators
Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has a direct impact on the gastrointestinal system, as up to 50% of fecal samples from coronavirus disease 2019 (COVID-19) patients contain detectable viral RNA despite a negative rhino-pharyngeal swab. This finding, together with an intestinal expression of angiotensin conversion enzyme 2 protein, suggests a possible fecal-oral transmission for SARS-CoV-2. Furthermore, gastrointestinal (GI) symptoms are common in COVID-19 patients including watery diarrhea, vomiting—particularly in children—nausea, and abdominal pain.
Pathogenesis of SARS-CoV-2 infection presents significant similarities to those of some immune-mediated diseases, such as inflammatory bowel diseases or rheumatoid arthritis, leading to the hypothesis that targeted therapies used for the treatment of immune-mediated disease could be effective to treat (and possibly prevent) the main complications of COVID-19.
In this review, we synthesize the present and future impact of SARS-CoV-2 infection on the gastrointestinal system and on gastroenterology practice, hypothesizing a potential role of the “gut-lung axis” and perhaps of the gut and lung microbiota into the interindividual differential susceptibility to COVID-19 19 disease.
Finally, we speculate on the reorganization of outpatient gastroenterology services, which need to consider, among other factors, the major psychological impact of strict lockdown measures on the whole population.
INTRODUCTION
The new pathogenic virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was recently identified as the causative agent of the novel acute respiratory disease named coronavirus disease 2019 (COVID-19) that started in the Hubei Province, China, and rapidly spread around the world.1, 2 SARS-CoV-2 belongs to the β-CoV family and is enveloped by the spike glycoprotein trimmer (S) and the hemagglutinin-esterase (HE); it is characterized by a high infectivity: respiratory droplets and direct contacts represent the main form of transmission.3
Despite being considered mainly a respiratory disease, it has now become clear that COVID-19 can have a direct impact on the gastrointestinal system which, in turn, might play a role in the pathogenesis of the disease.4 Furthermore, the pandemic itself and the lockdown measures to slow it could drive major changes in social behaviors and health care assistance that might affect patients with digestive diseases.
In this review, we focus on the current evidence of the relationship between SARS-CoV-2 and the gastrointestinal system; we outline the main implications on the management of patients with digestive diseases; and we try to prefigure the possible “gastrointestinal scenarios” we are about to face in the upcoming months.
COVID-19 AND GASTROINTESTINAL SYSTEM: CLINICAL ASPECTS
The pathognomonic symptoms of COVID-19 are cough, fever, and dyspnea. However, the disease can present a wider spectrum of clinical manifestations.5 Siddiqi and colleagues proposed a 3-phase model based on the observed symptoms to classify COVID-19. When the infection occurs (first stage), it typically presents with mild symptoms (malaise, low-grade fever, and dry cough). Some patients (up to 20%) develop atypical pneumonia (second stage), with high-grade fever, cough, dyspnea, and possibly hypoxia. Finally, a minority of patients progress to a third stage, characterized by a systemic hyperinflammatory state, that can lead to acute respiratory distress syndrome (ARDS), multi-organ failure, and death.6
Besides typical respiratory symptoms, other clinical manifestations have also been described, including gastrointestinal ones.4 Indeed, gastrointestinal symptoms had been associated with other coronavirus diseases, such as the 2002 SARS and the 2012 Middle East Respiratory Syndrome (MERS) epidemics,7 and diarrhea had been proposed as a possible prognostic factor for these diseases, even though no firm conclusions could be drawn.8, 9
In a recent systematic review, gastrointestinal symptoms were found in a broad range of SARS-CoV-2 patients. Anorexia was the most common symptom, reported in up to 40% of the patients, followed by other, more gastrointestinal-specific symptoms, including watery diarrhea (9.6%–16%), nausea and vomiting (6.6%–15.3%), and abdominal pain (5.7%x–14.5%).10 Gastrointestinal bleeding has also been reported, but it is not clear whether it was directly caused by the infection or should be considered a secondary manifestation of systemic damage in critically ill patients.11
Recent reports from Wuhan, China, demonstrated that 23% of COVID-19 patients presented with only digestive symptoms, 33% presented with both digestive and respiratory symptoms, and 44% presented with only respiratory symptoms.12 Furthermore, it has been shown that gastrointestinal symptoms can sometimes precede respiratory involvement and be the first manifestation of the infection.13 These data suggest the possible need to include SARS-CoV-2 infection among the differential diagnosis of acute diarrhea and/or vomiting, at least for the duration of this pandemic.
Notably, a recent review speculated about the possible role of diarrhea as a prognostic factor in COVID-19. No conclusive correlation was found: in some studies, gastrointestinal symptoms correlated with worse outcomes, whereas other findings suggested that no association was present.7
PATHOGENESIS OF SARS-COV-2 INFECTION: AN EMERGING ROLE FOR THE GASTROINTESTINAL TRACT
Angiotensin conversion enzyme 2 (ACE2) serves as the receptor used by the virus to enter human cells: the spike protein S binds to the receptor and mediates the fusion of the membranes. The process is regulated by the cell surface–associated transmembrane protease serine protease 2 (TMPRSS2), which operates in protein S cleavage and priming.14
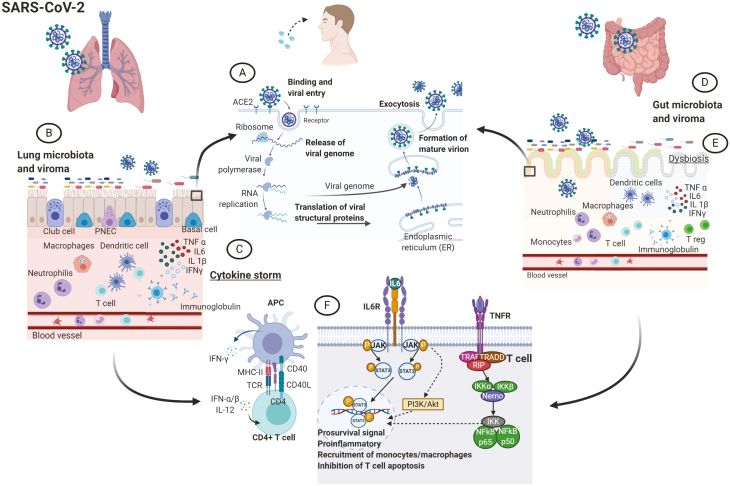
ACE and TMPRSS2 are mainly expressed by type 2 pneumocytes15 but have also been found on intestinal cells.16, 17 SARS-CoV-2 is believed to infect ACE2-expressing cells, triggering them to produce inflammatory mediators which lead to immune cells activation. Consequently, these cells release cytokines (such as interleukin [IL]-2, IL-6, IL-17, and tumor necrosis factor [TNF]), which mediate local and systemic manifestations of COVID-1918 (Fig. 1).
Figure 1.
The lung-gut axis in COVID-19. Both lung and gut epithelium expresses the ACE2 receptor, recognized by SARS-CoV-2 to entry in the host cells where the virus starts to replicate (A). In the lung (B), once the virus has entered the cells, it prompts immune system activation characterized by pro-inflammatory cytokines and immune cell recruitment. If viral clearance is not sufficient, the immune response further progresses toward a cytokine storm syndrome (C). In the gut (D), the virus targets the cells and elicits the immune response in a similar fashion, although it is not yet clear whether the magnitude of the inflammatory response reaches the same level of the lung. Possibly, the immune response could lead to a dysbiosis (E) with a propagation of the pro-inflammatory state. Antigen presenting cells (APCs) are involved in the activation of the immune system in response to various exogenous triggers (F) from virus and bacteria: COVID-19 can activate inflammatory pathways that present some similarities with some immune-mediated diseases, such as IBD or rheumatoid arthritis. Abbreviations: IFN, interferon; IKK, inhibitory kappa B kinases; NF-kB: nuclear factor kappa-light-chain-enhancer of activated B cells; PNEC, pulmonary neuroendocrine cells; RNA, ribonucleic acid; STAT, signal transducer and activator of transcription; TNFR, tumor-necrosis factor receptor; TRADD, TNFR1-associated death domain protein; TRAF, tumor necrosis factor receptor-associated factors; T reg, T regulatory cell; NEMO, NF-κB essential modulator.
Viral RNA has been detected in fecal samples from up to 50% of COVID-19 patients, thus the possibility of a fecal-oral transmission for SARS-CoV-2 has been advanced.19 In the study from Xiao et al, SARS-CoV-2 RNA duration time in the stool ranged from 1 to 12 days, and about 25% of patients still had positive fecal samples after oral and nasopharyngeal swab negativization16; similar results were also found in pediatric cohorts.20, 21 These studies suggest that fecal-oral route is at least possible for SARS-CoV-2 and might even last longer than “conventional” salivary droplet transmission. However, in a study conducted by Wolfel et al, although viral RNA was found in the stool samples of the patients, the presence of infectious SARS-CoV-2 was not detected, thus questioning the actual capability of the virus to be transmitted via a fecal-oral route.22
Various etiopathogenetic hypotheses have been advanced to explain the occurrence of diarrhea in COVID-19 patients including loss in enterocytes absorption capability,23 microscopic inflammatory gastrointestinal mucosal damage, and an impaired function of ACE2, which has a recognized role in maintaining gut homeostasis.24 Endoscopy performed on a COVID-19 patient with diarrhea did not show macroscopic mucosal alterations, but microscopy examination revealed the presence of numerous lymphocytes infiltrating the lamina propria.16 Furthermore, in a recent letter, Effenberg et al reported higher levels of fecal calprotectin levels in hospitalized COVID-19 patients with diarrhea compared with patients who never reported it.25 However, although these 2 reports might suggest that SARS-CoV-2 can directly target the gastrointestinal system, no definitive conclusion can be drawn based on these results.
Finally, alterations in liver function tests during COVID-19 have been reported.5 Indeed, ACE2 was found on hepatocytes and cholangiocytes, although to a lesser extent compared with lungs and intestine. Thus, SARS-CoV-2 could theoretically target liver cells.26 Nevertheless, a causative role for SARS-CoV-2 cannot be inferred based on these findings because fluctuations of aminotransferases are often recorded in patients with systemic viral infections and actually reflect systemic inflammatory activation rather than direct viral damage (a phenomenon referred to as “bystander hepatitis”).27 A recent study conducted on 115 hospitalized COVID-19 patients did not find any significant difference in liver function tests when comparing COVID-19 with other forms of pneumonia, thus suggesting that liver impairment might not be a prominent feature in COVID-19.28
SIMILARITIES AND DIFFERENCES WITH OTHER IMMUNE-MEDIATED DISORDERS
Immune-mediated diseases are conceived as multifactorial diseases whose pathogenesis relies on a complex interplay between genetic predisposition and environmental factors; in this context, infections have been widely advocated as potential triggers for the activation of immune-mediated diseases.29 After the removal of the initial trigger, activated immune cells still continue to actively participate in damage production and disease progression; this establishes a vicious circle of abnormal inflammation and tissue damage that perpetuates disease activity30, 31 (Fig. 1).
COVID-19, especially in its later and more severe stages, is characterized by a significant dysregulation of immune cells.32 Neutrophilia has been associated with a higher risk for ARDS and death.33 Furthermore, studies on peripheral blood mononuclear cells have revealed some intriguing results: though all T-cell subtypes were numerically reduced, such a decrease was more prominent in memory and regulatory T cells. Interestingly, a correlation between disease severity and the levels of inflammatory biomarkers was observed, with a sharp rise in IL-1, IL-2, IL-6, IL-8, IL-13, IL-17, and TNF,1, 34 when comparing mild and severe patients. These findings are consistent with the notion that the severe forms of COVID-19 depend upon an immune-mediated process characterized by a systemic hyperinflammatory state.
During SARS-CoV-2 infection, an escalating transition from mild to severe immune activation can be observed, with a progressive rise of proinflammatory cytokines.6 Neutrophils accumulate at the site of infection, where they wield antiviral functions but, at the same time, contribute to the propagation of the immune response. If viral clearance is inadequate, their unceasing activation sets off the progression towards a hyperinflammatory state. As a matter of fact, unfavorable outcomes in COVID-19 have been associated with an exaggerated immune response to SARS-CoV-2: a cytokine storm syndrome can develop, leading to ARDS and multi-organ failure.35, 36 Novel pathogenic hypotheses can be formed in regard to the prevention and treatment of the immune-mediated disease caused by SARS-CoV-2 infection. Notably, the cytokines overexpressed in the most advanced stages of COVID-19 are not directly involved in virus clearance.37 This led to the hypothesis that targeted therapies used for the treatment of immune-mediated diseases could prove effective to treat and—possibly—prevent the main complications of COVID-19. Interleukin-6 inhibitors have been proposed and used with encouraging results.38 Furthermore, baricitinib, a Janus kinases (JAK) 1 and 2 inhibitor, has been conceived as another viable treatment. The role of the different JAKs in the response to viral infections is not fully understood. Even though JAK 2 inhibition could potentially reduce viral entry within the host cells, JAK 1 inhibition might lead to a further reduction in viral clearance.39, 40
The cytokine repertoire induced by SARS-CoV-2 infection in complex COVID-19 presents significant similarities with those of some immune-mediated diseases, characterized by an aberrant activation of innate and adaptive immunity, possibly sustained by genetic or microbial predisposition31, 41 (Fig. 1). A fascinating consideration comes from the recent report of isolated cases of Kawasaki disease (KD) with concurrent COVID-19 pediatric infection.42 The cause of KD remains unknown, but various studies have described an association between viral respiratory infections and KD, ranging from 9% to 42% of patients with KD testing positive for a respiratory viral infection in the 30-days leading up to diagnosis of KD.43 The immune system plays a pivotal role in the pathogenesis of KD,44 and severe refractory cases of KD can benefit from treatment with immunomodulator agents.45
The putative link between SARS-CoV-2 infection and the onset of Kawasaki disease gives rise to the question about the actual extent of the mutual influences between the virus and the immune system. In this regard, the case of IBD is particularly interesting, given that the virus can exert a tropism on intestinal cells. ACE2 and TMPRSS2 were found to be upregulated in inflamed intestine of IBD patients.46, 47 These findings might hint at an augmented susceptibility to SARS-CoV-2 in IBD patients. On the other hand, it has been suggested that the soluble form of ACE2, whose serum concentrations are elevated in IBD,48 might exert a protective role against the infection by acting as a competitive receptor for the virus; soluble ACE2 is obtained through proteolytic cleavage, mediated by a protease (ADAM17) upregulated in active IBD.49
Despite some remarkable similarities, COVID-19 remains a very distinct entity from immune-mediated diseases (Table 1). First of all, COVID-19 has a definite etiologic agent, whose opportune and timely clearance is associated with disease resolution. Furthermore, the cytokine overproduction is generally abrupt and restricted in duration. Finally, limited information is available regarding immune cells involvement in COVID-19, whereas the major role of these cell types has been widely described in immune-mediated disorders.31, 41
Table 1.
Similarities and Difference Between COVID19 and Other Immune-Mediated Disorders
| COVID-19 | IMMUNE-MEDIATED DISEASES (eg, IBD, RA) | |
|---|---|---|
| Virus infection and clearance |

|

|
| Genetic, immune, and environmental predisposition |

|

|
| Severe immune activation |

|

|
| Rise of proinflammatory cytokines |

|

|
| Endothelial, epithelial, and other nonimmune cells |

|

|
| Gut microbiota involvement |

|

|
| ARDS and multi-organ Failure |

|

|
| IL-6 inhibitors and JAK inhibitors |

|

|
| Acute mortality and morbidity |

|

|
THE LUNG-GUT AXIS AND THE POSSIBLE ROLE OF GUT MICROBIOTA
Gut microbiota seems to play a fundamental role in the maturation and regulation of the immune system, with intestinal bacteria and their metabolites providing local and systemic signals to immune cells; vice versa, the immune system exerts a major influence on microbiota composition and functions.50
Despite being numerically smaller compared with gut microbiota, lung microbiota share the same main microbial phyla and relative abundance.51 Bacterial colonization of the respiratory tract has been implicated in the maturation and homeostasis of the lung immune system,52 and an association between lung microbiome dysbiosis and clinical manifestations of pulmonary disorders has been reported.53
Recent evidences have postulated a bidirectional relationship between the 2 microbiota.53 Fecal microbiota transplantation (FMT) was proved to have an impact on lung microbiota composition.54 Furthermore, it has been observed that gut-resident innate lymphoid cells, implicated in repair functions, can move to the lung under inflammatory stimulus.55 Lastly, a 2-way communication through the lymphatic system, involving both bacteria and their metabolites, has been documented.56–58 The ensemble of the relevant connections between the intestine and the lung, including gut and lung microbiota, their intercompartment crosstalk, and gut and lung immune system crosstalk with local or long-reaching interactions, has been called the “gut-lung axis.” 53
This axis has already been implicated in several diseases, with some interesting findings regarding viral respiratory infections reported in murine models, where a Th17-mediated modulation of gut microbiota composition has been observed.59 Furthermore, impairment of gut microbiota was found to be associated with more severe forms of respiratory influenza in mice, in which microbiota-derived signals were able to exert their effects on lung stromal cells and keep epithelial cells in an IFN-primed state, inducing an antiviral state in nonimmune cells.60 It could be argued that the composition of gut microbiota can have an impact on the immune response against some viral respiratory agents and, thus, on the outcome of the disease. Therefore, a putative role for the gut-lung axis in the modulation of the pathophysiology of SARS-CoV-2 infection and the subsequent immune activation can be hypothesized (Fig. 1).
ACE2 on the enterocytes serves as regulator of dietary aminoacids uptake and has been implicated in gut immune and microbial homeostasis; it has been shown that its activation, under the regulation of tryptophan, leads to the production of antimicrobial peptides.24 Even though we do not know the exact implications of SARS-CoV-2 on the intestine, given that ACE2 is recognized by the virus, it is possible to hypothesize that its occupancy might inhibit its regulatory activity and produce intestinal dysbiosis; furthermore, it has been shown that ACE2 expression is downregulated by SARS-CoV-2,61 which could further contribute to limit ACE2 functions. Regardless of the exact mechanism, if SARS-CoV-2 is able to produce intestinal damage, the possibility of gut dysbiosis and augmented intestinal permeability needs to be taken into account. Such conditions might have an impact on lung diseases through 2 possible mechanisms: the translocation of bacteria and their metabolites or the migration of activated immune cells.57, 62, 63 Indeed, a study suggested a correlation between the composition of gut microbiota and the outcome of critically ill patients with ARDS: intestinal bacteria seemed to have an influence on the evolution of these patients, due to a gut-to-lung bacterial translocation secondary to increased gut and alveolo-capillary permeability.64 Whether these findings are valid for COVID-induced ARDS remains unknown.
To date, there is no direct evidence supporting the notion that acting on the gut-lung axis might exert an effect on the course of SARS-CoV-2 infection. However, this remains a fascinating hypothesis due to the possible implications in clinical practice. Of note, the only report linking the gut-lung axis to COVID-19 have been Chinese guidelines recommending the use of probiotics in severe COVID-19 patients to preserve intestinal homeostasis and avoid bacterial infection.65
PRESENT CLINICAL IMPLICATIONS FOR GASTROENTEROLOGISTS
Outpatient Services and Endoscopy
The outbreak of the COVID-19 pandemic had a dramatic impact on the organization and execution of every clinical activity. Given their close contact with patients, health care professionals are at great risk of being infected, with an estimated prevalence up to 10% in the most affected regions.66 The significant prevalence of gastrointestinal symptoms10 and the possibility of a fecal-oral transmission16 entail that gastroenterologists may be deeply involved; furthermore, non-COVID-19 patients with preexisting digestive diseases still need appropriate and timely care during the pandemic. Therefore, the utmost effort has been made since the beginning of the emergency to provide essential care for both COVID-19 and non-COVID-19 patients while keeping health care professionals safe.
Health care professionals’ workflows had to be entirely reconsidered: outpatient services, especially for patients on chronic immunosuppressive therapy, have been redesigned by providing waiting rooms that can guarantee social distancing, strictly scheduled accesses, preventive nasal swab in case of suggestive symptoms, and prioritizing access to nondeferrable patients. Additionally, telemedicine tools have been massively implemented.67
Endoscopists are at high risk from inhalation of airborne droplets, mucosal contact, and potential fecal-oral transmission. The European Society of Gastrointestinal Endoscopy and the European Society of Gastroenterology and Endoscopy Nurses Associated (ESGE/ESGENA) produced a detailed position statement for the COVID-19 pandemic, listing urgent (nondeferrable) and nonurgent (requiring a case-by-case evaluation) procedures. Patients risk stratification of being infected by SARS-CoV-2 is needed for scheduled GI endoscopy activities: in cases of high risk or confirmed infection, adequate PPE is required.68 Reprocessing of flexible endoscopes and accessories should be performed according to published guidelines, with use of viricidal agents recommended.69
In spite of expert opinions and society recommendations, deciding whether or not to postpone an endoscopy is not always unambiguous, particularly in IBD, where endoscopic findings can guide patients’ management.70 Nondeferrable colonoscopies should be performed in cases of new diagnosis confirmation, severe flares, subacute obstructions, and worsening of cholangitis and jaundice in a patient with known primary sclerosing cholangitis.71 However, no clear indication is available for those endoscopies that cannot be considered urgent but may be associated with unfavorable consequences if rescheduled in the long term, such as colorectal cancer surveillance, efficacy assessment of therapies, and postoperative recurrence evaluation in Crohn’s disease.
Clinical Management of Gastrointestinal Diseases and Fecal Microbiota Transplant
Immune-mediated diseases, patients undergoing chemotherapy, and transplant patients raise some concerns for a theoretically higher risk of infectious diseases. Given the paucity of evidences currently available, a cautious attitude in prescribing immunomodulators is generally advisable; however, a protective role for these patients has been theorized. Interleukin-6 inhibitors are currently used to treat COVID-19 complications38; furthermore, observational studies reporting no severe cases in IBD patients living in epidemic areas might suggest that this population is actually at lower risk,72, 73 and immunomodulators may have played a role in these observations. The preliminary data from the Surveillance Epidemiology of Coronavirus Under Research Exclusion for Inflammatory Bowel Disease (SECURE-IBD) registry have shown that increasing age, ≥2 comorbidities, systemic corticosteroids, and sulfasalazine or mesalamine represent risk factors in IBD patients for severe COVID-19. The results on aminosalicylates are indeed surprising: they could depend on some unrecognized confounding factors or the biological activity of the drug, but no clear interpretation is possible to date.74
Corticosteroids, the mainstay for the treatment of many immune-mediated diseases, have been associated with worse outcomes in influenza pneumonia75 and with a reduction of viral clearance in patients with SARS76 and MERS.77 Current evidences seem to point against their use in COVID-19, as their administration was associated with higher mortality, longer hospitalizations, and a higher risk for secondary bacterial infections.78 Furthermore, the SECURE-IBD study showed that systemic corticosteroids significantly increased the risk for hospitalization, need for intensive care, and death.74 Consequently, steroid prescription should be cautiously weighted in non-COVID-19 patients, and prednisone ≥20 mg/day should be avoided.79
Thiopurines reduce the number and function of activated T cells32 and have been associated with an increased risk for viral infections.80 To date, there is insufficient evidence to conclude that thiopurines might actually increase the risk of SARS-CoV-2 infection. Because they still continue to exert their immunosuppressive effect for weeks after cessation,81 dose reduction and therapy discontinuation are not currently recommended.79
Calcineurin and mammalian target of rapamycin inhibitors are not associated with major concerns of infections.82 Indeed, mammalian target of rapamycin inhibition has been associated with possible antiviral activity,83 and calcineurin inhibition was reported to suppress coronaviruses replication.84 Treatment withdrawal is not recommended; however, due to the possible interactions with some antiviral agents, a dose adjustment or suspension could be required in the case of SARS-CoV-2 patients beingtreated with specific drugs.27
Anti-TNF agents are associated with an increase in infectious risk85; however, TNF might exert a pathogenetic role in severe cases of COVID-19 because it can cause increased expression of ACE2 and lymphocyte death.86 Tursi et al reported the case of a young man with IBD treated with adalimumab who developed COVID-19 and quickly recovered.87 In the SECURE-IBD registry, anti-TNF agents did not seem to affect the course of COVID-19, whereas combination therapy (anti-TNF and thiopurines) was associated with worse outcomes.74 Continuation of anti-TNF treatment is currently recommended79; furthermore, adalimumab is under study as a treatment for cytokine storm syndrome in COVID-19.88 Conversely, the use of combination therapy should probably be discouraged, at least in those patients with other risk factors for severe COVID-19.
Vedolizumab and ustekinumab are not associated with an increased risk for viral infections,89, 90 and their continuation is currently recommended.79 Vedolizumab is a gut-selective agent that prevents lymphocytes homing; in a phase 1 clinical trial, its use in human immunodeficiency virus (HIV)–positive patients did not increase the viral load.91 Ustekinumab targets the p40 subunit common to IL-12 and IL-23, thus blocking Th1 cells priming and Th17 cells activation. The suppression of the Th17 response might theoretically contribute to mitigate COVID-19 cytokine storm, but this approach has never been tested.32
The blockade of the JAK pathways leads to a wider suppression of cytokine-mediated responses. JAK 1 and 3 inhibition downregulates IFNγ, which has a role in viral clearance37 and has been associated with an increased risk for herpes-zoster infection.92 Conversely, JAK 2 inhibition has been proposed as a therapeutic strategy for COVID-19.93 No definitive conclusions on tofacitinib (JAK 1 and 3 inhibitor) can be drawn in respect to SARS-CoV-2: discontinuation is not currently recommended,79 but initiation in epidemic areas should be avoided, unless appropriate alternatives are not available.94
Another interesting case could be made for fecal microbiota transplant (FMT), a clearly known effective treatment for recurrent Clostridium difficile infections.95, 96 In December 2019, the consensus report on stool banking for microbiota transplantation was released, where specific recommendations about stool donor screening were made.97 Given the increasing evidence of a potential gastrointestinal involvement in SARS-CoV-2 infection and the possible existence of a fecal-oral route of transmission, several measures have been updated to assure the best safety possible against COVID-19 transmission through FMT.
EMERGING NEEDS AND FUTURE IMPLICATIONS
The COVID-19 pandemic has changed priorities and forced a reorganization of several outpatient services and inpatients management. Furthermore, the strict lockdown measures have major psychological impact on the whole population, including children and adolescents, as they are abruptly experiencing a dramatic change in life habits, increased social isolation and subjective feelings of loneliness, and changed management of deaths and comfort.98 Moreover, diet habits are changing as well, as most subjects have gained weight during the lockdown. All these things will have an unpredictable impact on patients with gastrointestinal diseases, on health personnel involved in gastrointestinal services, and also on organizational schemes for the “post-COVID” era.
In an attempt to reduce hospital visits and minimize the contagious risk, considerable numbers of visits and procedures have been postponed. Prioritizing urgent patients is undoubtedly imperative, but the compelling focus on the strategies to reduce contagions could lead to an improper neglection of “stable” patients, thus exposing them to otherwise preventable risks in the long term. Some concerns about the possible long-lasting impact of this situation on cirrhotic patients have been recently raised. Early diagnosis and close monitoring of cirrhosis complications are crucial for their management; the consequences of missed identifications of early hepatocellular carcinoma, delayed screening endoscopies, and unperformed follow-up visits are expected in the following months.99 Similar considerations could be made for IBD. Deep remission and treat-to-target strategies correlate with better long-term outcomes,70 and tight control management is associated with reduced rates of disease progression and complications.100 To what extent the current shift in the management of IBD can impact the natural history of the disease is unpredictable. We can reasonably expect that diagnostic and therapeutic delays have occurred since the beginning of the pandemic, and we will have to cope with their consequences in the near future.
We do expect an increase in the requests for gastrointestinal clinic visits and endoscopic procedures to make up for patients postponed during the pandemic. However, the risk of new cancer diagnosis because of delay of screening procedures (eg, colonoscopies) is not predictable. Additionally, intestinal infections by SARS-CoV-2 or pandemic-associated environmental changes may also lead to stress-related functional gastrointestinal disorders (eg, reactivation of IBS and postinfectious IBS).
Health personnel had to face the pandemic by reorganizing shift and schedules, patient-physician relationships, and education tasks. This will induce health personnel to participate more actively in the organization of the post-COVID-19 era. Hospital and services, in fact, will not be allowed to come back to former schedules as the post-COVID-19 era maintains the risk of a second peak of the pandemic. Furthermore, “space restrictions” to avoid assembling and the consequent reduction of the total number of patients entering the outpatient clinic will outstand the organization, web-based telemedicine, and the suitability of clinical procedures.
Conflicts of Interest: FS has served on the advisory boards for Abbvie, Janssen, MSD, Sanofi, and Takeda. GP received consultancy fees from Alphasigma. VP is on the scientific advisory board of Actial Farmaceutica Srl, Italy. DP received speaker fees from AbbVie, MSD, Takeda, Janssen, and Pfizer. GC is a consultant for and has received food and beverage compensation from Cook Medical, Boston Scientific, and Olympus. AG reports personal fees for consultancy for Eisai S.r.l., 3PSolutions, Real Time Meeting, Fondazione Istituto Danone, Sinergie S.r.l. Board MRGE, and Sanofi S.p.A, personal fees for acting as a speaker for Takeda S.p.A, AbbVie, and Sandoz S.p.A, and personal fees for acting on advisory boards for VSL3 and Eisai. IB is a consultant for Apollo Endosurgery, Cook Medical, and Boston Scientific; is a board member for Endo Tools; research grant recipient from Apollo Endosurgery; and has received food and beverage compensation from Apollo Endosurgery, Cook Medical, Boston Scientific, and Endo Tools. AA has received consulting and/or advisory board fees from AbbVie, Allergan, Amgen, Biogen, Bristol-Myers Squibb, Celgene, Celltrion, Ferring, Gilead, Janssen, Lilly, MSD, Mylan, Pfizer, Samsung Bioepis, Sandoz, and Takeda; lecture and/or speaker bureau fees from AbbVie, Amgen, Biogen, Ferring, Giliead, Janssen, MSD, Mitsubishi-Tanabe, Nikkiso, Pfizer, Sandoz, Samsung Bioepis, Takeda; and research grants from MSD, Pfizer, Takeda. All the other authors have no conflicts of interest to declare.
ACKNOWLEDGMENTS
The authors wish to thank Alessia Leonetti for her support in proofreading the manuscript.
Specific author contribution: FS, GI, and AA designed the project. FS, GI, GP, LRL, LMV, VP, YR, and IB performed the literature reseach and wrote the manuscript. DP, MN, GC, YR, GC, AG, and AA also contributed to literature research and to revise critically the manuscript. All authors reviewed and approved the final draft of the article before submission. AA oversaw the project and guarantees for the integrity of the work.
Conflicts of interest: FS: advisory board for Abbvie, Janssen, MSD, Sanofi, Takeda. GP received consultancy fees from Alphasigma. VP is in scientific advisory board of Actial Farmaceutica Srl, Italy. DP received speaker fees from AbbVie, MSD, Takeda and Janssen, Pfizer. GC: consultant for and food and beverage compensation from Cook Medical, Boston Scientific, and Olympus. AG reports personal fees for consultancy for Eisai S.r.l., 3PSolutions, Real Time Meeting, Fondazione Istituto Danone, Sinergie S.r.l. Board MRGE, and Sanofi S.p.A, personal fees for acting as a speaker for Takeda S.p.A, AbbVie, and Sandoz S.p.A, and personal fees for acting on advisory boards for VSL3 and Eisai. IB: consultant for Apollo Endosurgery, Cook Medical, and Boston Scientific; board member for Endo Tools; research grant recipient from Apollo Endosurgery; food and beverage compensation from Apollo Endosurgery, Cook Medical, Boston Scientific, and Endo Tools. AA: consulting and/or advisory board fees from AbbVie, Allergan, Amgen, Biogen, Bristol-Myers Squibb, Celgene, Celltrion, Ferring, Gilead, Janssen, Lilly, MSD, Mylan, Pfizer, Samsung Bioepis, Sandoz, Takeda; lecture and/or speaker bureau fees from AbbVie, Amgen, Biogen, Ferring, Giliead, Janssen, MSD, Mitsubishi-Tanabe, Nikkiso, Pfizer, Sandoz, Samsung Bioepis, Takeda; and research grants from MSD, Pfizer, Takeda. All the other authors have no potential conflicts of interest to disclose.
REFERENCES
- 1. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhu N, Zhang D, Wang W, et al. ; China Novel Coronavirus Investigating and Research Team A novel Coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Jin Y, Yang H, Ji W, et al. Virology, epidemiology, pathogenesis, and control of covid-19. Viruses. 2020;12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wan Y, Li J, Shen L, et al. Enteric involvement in hospitalised patients with COVID-19 outside Wuhan. Lancet Gastroenterol Hepatol. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Guan WJ, Ni ZY, Hu YHY, et al. Clinical characteristics of Coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Siddiqi HK, Mehra MR. COVID-19 illness in native and immunosuppressed states: a clinical-therapeutic staging proposal. J Heart Lung Transplant. 2020;39:405–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. D’Amico F, Baumgart DC, Danese S, et al. Diarrhea during COVID-19 infection: pathogenesis, epidemiology, prevention and management. Clin Gastroenterol Hepatol. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Leung WK, To KF, Chan PK, et al. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterology. 2003;125:1011–1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Choi WS, Kang CI, Kim Y, et al. ; Korean Society of Infectious Diseases Clinical presentation and outcomes of Middle East respiratory syndrome in the Republic of Korea. Infect Chemother. 2016;48:118–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cheung KS, Hung IF, Chan PP, et al. Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from the Hong Kong cohort and systematic review and meta-analysis. Gastroenterology. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tian Y, Rong L, Nian W, et al. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51:843–851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Han C, Duan C, Zhang S, et al. Digestive symptoms in COVID-19 patients with mild disease severity. Am J Gastroenterol. 2020:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Buscarini E, Manfredi G, Brambilla G, et al. GI symptoms as early signs of COVID-19 in hospitalised Italian patients. Gut. 2020. doi: 10.1136/gutjnl-2020-321434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Brake SJ, Barnsley K, Lu W, et al. Smoking upregulates angiotensin-converting enzyme-2 receptor: a potential adhesion site for novel Coronavirus SARS-CoV-2 (Covid-19). J Clin Med. 2020;9:841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Xiao F, Tang M, Zheng X, et al. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Zhang H, Li HB, Lyu JR, et al. Specific ACE2 expression in small intestinal enterocytes may cause gastrointestinal symptoms and injury after 2019-nCoV infection. Int J Infect Dis. 2020;96:19–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Lin L, Lu L, Cao W, et al. Hypothesis for potential pathogenesis of SARS-CoV-2 infection–a review of immune changes in patients with viral pneumonia. Emerg Microbes Infect. 2020;9:727–732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Xing YH, Ni W, Wu Q, et al. Prolonged viral shedding in feces of pediatric patients with coronavirus disease 2019. J Microbiol Immunol Infect. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Xu Y, Li X, Zhu B, et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat Med. 2020;26:502–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wölfel R, Corman VM, Guggemos W, et al. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020:1–5. [DOI] [PubMed] [Google Scholar]
- 23. Gu J, Han B, Wang J. COVID-19: gastrointestinal manifestations and potential fecal-oral transmission. Gastroenterology. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Hashimoto T, Perlot T, Rehman A, et al. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature. 2012;487:477–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Effenberger M, Grabherr F, Mayr L, et al. Faecal calprotectin indicates intestinal inflammation in COVID-19. Gut. 2020. doi: 10.1136/gutjnl-2020-321388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chai X, Hu L, Zhang Y, et al. Specific ACE2 expression in cholangiocytes may cause liver damage after 2019-nCoV infection. bioRxiv. 2020. https://www.biorxiv.org/content/biorxiv/early/2020/02/04/2020.02.03.931766.full.pdf. Accessed May 2, 2020. [Google Scholar]
- 27. Boettler T, Newsome PN, Mondelli MU, et al. Care of patients with liver disease during the COVID-19 pandemic: EASL-ESCMID position paper. JHEP Rep. 2020;2:100113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5:428–430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ercolini AM, Miller SD. The role of infections in autoimmune disease. Clin Exp Immunol. 2009;155:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Ye Q, Wang B, Mao J. The pathogenesis and treatment of the “Cytokine Storm’’ in COVID-19. J Infect. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Fragoulis GE, Liava C, Daoussis D, et al. Inflammatory bowel diseases and spondyloarthropathies: from pathogenesis to treatment. World J Gastroenterol. 2019;25:2162–2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Neurath MF. COVID-19 and immunomodulation in IBD. Gut. 2020;69:1335–1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with Coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2763184. Accessed May 2, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Qin C, Zhou L, Hu Z, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020;2019:4–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Pedersen SF, Ho YC. SARS-CoV-2: a storm is raging. J Clin Invest. 2020;130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Chen G, Wu D, Guo W, et al. Clinical and immunologic features in severe and moderate Coronavirus Disease 2019. J Clin Invest. 2020;130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Schett G, Sticherling M, Neurath MF. COVID-19: risk for cytokine targeting in chronic inflammatory diseases? Nat Rev Immunol. 2020;20:271–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Ortiz-Martinez Y. Tocilizumab: a new opportunity in the possible therapeutic arsenal against COVID-19. Travel Med Infect Dis. 2020:101678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Richardson P, Griffin I, Tucker C, et al. Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. Lancet. 2020;395:e30–e31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Favalli EG, Biggioggero M, Maioli G, et al. Baricitinib for COVID-19: a suitable treatment? Lancet Infect Dis. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Lee KH, Ahn BS, Cha D, et al. Understanding the immunopathogenesis of autoimmune diseases by animal studies using gene modulation: a comprehensive review. Autoimmun Rev. 2020;19:102469. [DOI] [PubMed] [Google Scholar]
- 42. Jones VG, Mills M, Suarez D, et al. COVID-19 and Kawasaki disease: novel virus and novel case. Hosp Pediatr. 2020;10:537–540. [DOI] [PubMed] [Google Scholar]
- 43. Turnier JL, Anderson MS, Heizer HR, et al. Concurrent respiratory viruses and Kawasaki disease. Pediatrics. 2015;136:e609–e614. [DOI] [PubMed] [Google Scholar]
- 44. Rowley AH, Shulman ST. Pathogenesis and management of Kawasaki disease. Expert Rev Anti Infect Ther. 2010;8:197–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Agarwal S, Agrawal DK. Kawasaki disease: etiopathogenesis and novel treatment strategies. Expert Rev Clin Immunol. 2017;13:247–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Garg M, Royce SG, Tikellis C, et al. Imbalance of the renin-angiotensin system may contribute to inflammation and fibrosis in IBD: a novel therapeutic target? Gut. 2020;69:841–851. [DOI] [PubMed] [Google Scholar]
- 47. Jablaoui A, Kriaa A, Mkaouar H, et al. Fecal serine protease profiling in inflammatory bowel diseases. Front Cell Infect Microbiol. 2020;10:21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Garg M, Burrell LM, Velkoska E, et al. Upregulation of circulating components of the alternative renin-angiotensin system in inflammatory bowel disease: a pilot study. J Renin Angiotensin Aldosterone Syst. 2015;16:559–569. [DOI] [PubMed] [Google Scholar]
- 49. Wong E, Cohen T, Romi E, et al. Harnessing the natural inhibitory domain to control TNFα converting enzyme (TACE) activity in vivo. Sci Rep. 2016;6:35598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157:121–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Sze MA, Dimitriu PA, Hayashi S, et al. The lung tissue microbiome in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2012;185:1073–1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Gollwitzer ES, Saglani S, Trompette A, et al. Lung microbiota promotes tolerance to allergens in neonates via PD-L1. Nat Med. 2014;20:642–647. [DOI] [PubMed] [Google Scholar]
- 53. Enaud R, Prevel R, Ciarlo E, et al. The gut-lung axis in health and respiratory diseases: a place for inter-organ and inter-kingdom crosstalks. Front Cell Infect Microbiol. 2020;10:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Madan JC, Koestle DC, Stanton BA, et al. Serial analysis of the gut and respiratory microbiome in cystic fibrosis in infancy: interaction between intestinal and respiratory tracts and impact of nutritional exposures. MBio. 2012;3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Huang Y, Mao K, Chen X, et al. S1P-dependent interorgan trafficking of group 2 innate lymphoid cells supports host defense. Science. 2018;359:114–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Trompette A, Gollwitzer ES, Yadava K, et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med. 2014;20:159–166. [DOI] [PubMed] [Google Scholar]
- 57. Bingula R, Filaire M, Radosevic-Robin N, et al. Desired turbulence? Gut-lung axis, immunity, and lung cancer. J Oncol. 2017;2017:5035371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. McAleer JP, Kolls JK. Contributions of the intestinal microbiome in lung immunity. Eur J Immunol. 2018;48:39–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Wang J, Li F, Wei H, et al. Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell-dependent inflammation. J Exp Med. 2014;211:2397–2410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Bradley KC, Finsterbusch K, Schnepf D, et al. Microbiota-driven tonic interferon signals in lung stromal cells protect from influenza virus infection. Cell Rep. 2019;28:245–256.e4. [DOI] [PubMed] [Google Scholar]
- 61. Verdecchia P, Cavallini C, Spanevello A, et al. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Intern Med. 2020:1–0. www.elsevier.com/locate/ejim. Accessed May 7, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Samuelson DR, Welsh DA, Shellito JE. Regulation of lung immunity and host defense by the intestinal microbiota. Front Microbiol. 2015;6:1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Huffnagle GB. The Microbiota and Allergies/Asthma What Is the Evidence Linking Changes in the Microbiota to the Development of Allergic Disease?www.plospathogens.org.
- 64. Mukherjee S, Hanidziar D. More of the gut in the lung: how two microbiomes meet in ARDS. Yale J Biol Med. 2018;91:143–149. [PMC free article] [PubMed] [Google Scholar]
- 65. Gao QY, Chen YX, Fang JY. 2019 Novel coronavirus infection and gastrointestinal tract. J Dig Dis. 2020;21:125–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Grasselli G, Pesenti A, Cecconi M. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy. JAMA. 2020;323:1545. [DOI] [PubMed] [Google Scholar]
- 67. Scaldaferri F, Pugliese D, Privitera G, et al. Impact of COVID-19 pandemic on the daily management of biotechnological therapy in inflammatory bowel disease patients: reorganisational response in a high-volume Italian inflammatory bowel disease centre. United European Gastroenterol J. 2020. doi: 10.1177/2050640620929133. Accessed May 7, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Gralnek IM, Hassan C, Beilenhoff U, et al. ESGE and ESGENA Position Statement on gastrointestinal endoscopy and the COVID-19 pandemic. Endoscopy. 2020;52:483–490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Beilenhoff U, Biering H, Blum R, et al. Reprocessing of flexible endoscopes and endoscopic accessories used in gastrointestinal endoscopy: position statement of the European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Gastroenterology Nurses and Associates (ESGENA) - update 2018. Endoscopy. 2018;50:1205–1234. [DOI] [PubMed] [Google Scholar]
- 70. Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015;110:1324–1338. [DOI] [PubMed] [Google Scholar]
- 71. Iacucci M, Cannatelli R, Labarile N, et al. Endoscopy in inflammatory bowel diseases during the COVID-19 pandemic and post-pandemic period. Lancet Gastroenterol Hepatol. 2020;5:598–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Norsa L, Indriolo A, Sansotta N, et al. Uneventful course in IBD patients during SARS-CoV-2 outbreak in northern Italy. Gastroenterology. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. An P, Ji M, Ren H, et al. Protection of 318 inflammatory bowel disease patients from the outbreak and rapid spread of COVID-19 infection in Wuhan, China. SSRN Electron J. 2020. [Google Scholar]
- 74. Brenner EJ, Ungaro RC, Gearry RB, et al. Corticosteroids, but not TNF antagonists, are associated with adverse COVID-19 outcomes in patients with inflammatory bowel diseases: results from an international registry. Gastroenterology. 2020. doi: 10.1053/j.gastro.2020.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Ni YN, Chen G, Sun J, et al. The effect of corticosteroids on mortality of patients with influenza pneumonia: a systematic review and meta-analysis. Crit Care. 2019;23:99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Lee N, Allen Chan KC, Hui DS, et al. Effects of early corticosteroid treatment on plasma SARS-associated coronavirus RNA concentrations in adult patients. J Clin Virol. 2004;31:304–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Arabi YM, Mandourah Y, Al-Hameed F, et al. ; Saudi Critical Care Trial Group Corticosteroid therapy for critically ill patients with middle east respiratory syndrome. Am J Respir Crit Care Med. 2018;197:757–767. [DOI] [PubMed] [Google Scholar]
- 78. Yang Z, Liu J, Zhou Y, et al. The effect of corticosteroid treatment on patients with coronavirus infection: a systematic review and meta-analysis. J Infect. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Rubin DT, Abreu MT, Rai V, et al. Management of patients with Crohn’s disease and ulcerative colitis during the COVID-19 pandemic: results of an International Meeting. Gastroenterology. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. O’Connor A, Qasim A, O’Moráin CA. The long-term risk of continuous immunosuppression using thioguanides in inflammatory bowel disease. Ther Adv Chronic Dis. 2010;1:7–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Danese S, Cecconi M, Spinelli A. Management of IBD during the COVID-19 outbreak: resetting clinical priorities. Nat Rev Gastroenterol Hepatol. 2020;17:253–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Anon. Infections Associated With Specific Immunosuppressive Agents in Transplant Recipients http://www.antimicrobe.org/new/t34_dw.html#r61. Accessed May 9, 2020.
- 83. Bowman LJ, Brueckner AJ, Doligalski CT. The role of mTOR inhibitors in the management of viral infections: a review of current literature. Transplantation. 2018;102:S50–S59. [DOI] [PubMed] [Google Scholar]
- 84. Tanaka Y, Sato Y, Sasaki T. Suppression of Coronavirus replication by cyclophilin inhibitors. Viruses. 2013;5:1250–1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Bonovas S, Fiorino G, Allocca M, et al. Biologic therapies and risk of infection and malignancy in patients with inflammatory bowel disease: a systematic review and network meta-analysis. Clin Gastroenterol Hepatol. 2016;14:1385–1397.e10. [DOI] [PubMed] [Google Scholar]
- 86. Mehta AK, Gracias DT, Croft M. TNF activity and T cells. Cytokine. 2018;101:14–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Tursi A, Angarano G, Monno L, et al. COVID-19 infection in Crohn’s disease under treatment with adalimumab. Gut. 2020;69:1364–1365. [DOI] [PubMed] [Google Scholar]
- 88. Anon. Chinese Clinical Trial Register (ChiCTR) - The World Health Organization International Clinical Trials Registered Organization Registered Platform http://www.chictr.org.cn/showprojen.aspx?proj=49889. Accessed May 9, 2020.
- 89. Ng SC, Hilmi IN, Blake A, et al. Low frequency of opportunistic infections in patients receiving vedolizumab in clinical trials and post-marketing setting. Inflamm Bowel Dis. 2018;24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Ghosh S, Gensler LS, Yang Z, et al. Correction to: ustekinumab safety in psoriasis, psoriatic arthritis, and Crohn’s disease: an integrated analysis of phase II/III clinical development programs. Drug Saf. 2019;42:809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Sneller MC, Clarridge KE, Seamon C, et al. An open-label phase 1 clinical trial of the anti-α4β7 monoclonal antibody vedolizumab in HIV-infected individuals. Sci Transl Med. 2019;11. [DOI] [PubMed] [Google Scholar]
- 92. Sandborn WJ, Ghosh S, Panes J, et al. ; Study A3921063 Investigators Tofacitinib, an oral Janus kinase inhibitor, in active ulcerative colitis. N Engl J Med. 2012;367:616–624. [DOI] [PubMed] [Google Scholar]
- 93. Wu D, Yang XO. TH17 responses in cytokine storm of COVID-19: an emerging target of JAK2 inhibitor fedratinib. J Microbiol Immunol Infect. 2020;53:368–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Mao R, Liang J, Shen J, et al. ; Chinese Society of IBD, Chinese Elite IBD Union; Chinese IBD Quality Care Evaluation Center Committee Implications of COVID-19 for patients with pre-existing digestive diseases. Lancet Gastroenterol Hepatol. 2020;5:425–427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Ianiro G, Maida M, Burisch J, et al. Efficacy of different faecal microbiota transplantation protocols for Clostridium difficile infection: a systematic review and meta-analysis. United European Gastroenterol J. 2018;6:1232–1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Ianiro G, Murri R, Sciumè GD, et al. Incidence of bloodstream infections, length of hospital stay, and survival in patients with recurrent clostridioides difficile infection treated with fecal microbiota transplantation or antibiotics: a prospective cohort study. Ann Intern Med. 2019;171:695–702. [DOI] [PubMed] [Google Scholar]
- 97. Cammarota G, Ianiro G, Kelly CR, et al. International consensus conference on stool banking for faecal microbiota transplantation in clinical practice. Gut. 2019;68:2111–2121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Sani G, Janiri D, Di Nicola M, et al. Mental health during and after the COVID-19 emergency in Italy. Psychiatry Clin Neurosci. 2020;74:372. [DOI] [PubMed] [Google Scholar]
- 99. Tapper EB, Asrani SK. COVID-19 pandemic will have a long-lasting impact on the quality of cirrhosis care. J Hepatol. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Ungaro RC, Yzet C, Bossuyt P, et al. Deep remission at 1 year prevents progression of early Crohn’s disease. Gastroenterology. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]