We thank Jehn et al.1 for their interest in our paper2 and their application of our equations to kidney transplant recipients and living donors. Kidney transplant recipients were excluded from our data, and it is plausible that our equations might not be applicable to such patients who, for example, are known to have a high prevalence of tubular proteinuria.3 However, we are uncertain whether the data that they present demonstrate that our equations are invalid in these populations.
First, the authors conclude that the variability between predicted median and measured albumin-to-creatinine ratios (ACRs) is unacceptable by plotting the deviation on a logarithmic scale (supplemental figures 1 and 3 in the work of Jehn et al.1), with negative differences shown as negative logarithms. This method transforms any normal distribution into an apparently bimodal one (Supplemental Figure 1). Preferred methods for estimating the deviation include investigating the distribution of measured ACR for small ranges of predicted ACR values, fitting quantile regression models, or studying a scatterplot of predicted median versus measured ACR on a log scale.
We have now examined Alberta transplant recipients (n=2280 ACR/protein-to-creatinine ratio (PCR) pairs in 627 patients) previously excluded from our analysis. From this, it appears that the variability is similar in transplant recipients and nonrecipients (Figure 1) and for predicted ACR >100 mg/g in the scatterplot in supplemental figure 2 of Jehn et al.1
Figure 1.
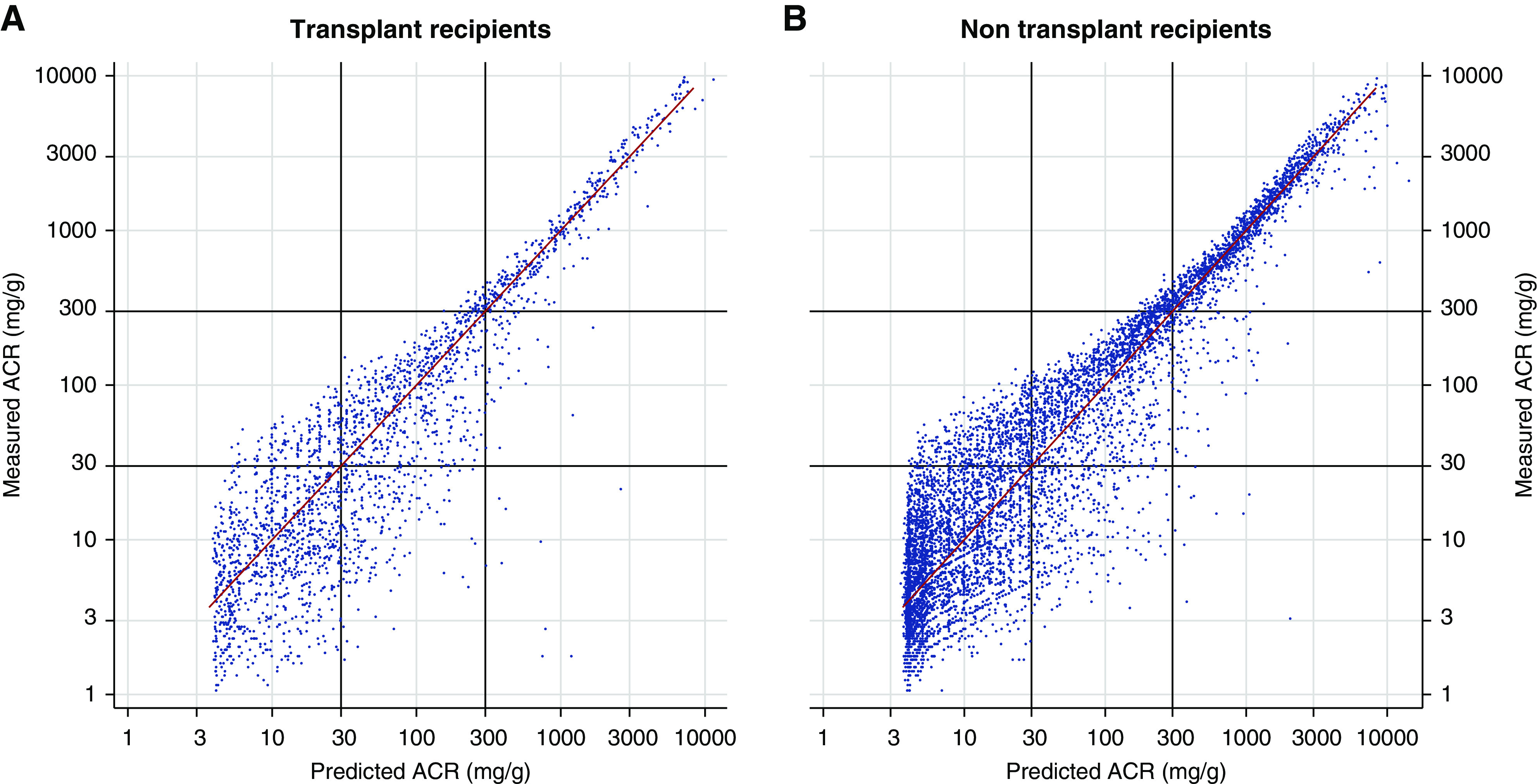
Scatterplots of measured ACR versus predicted median ACR for Alberta (A) kidney transplant recipients and (B) non transplant recipients show similar symmetrical distributions around the red diagonal line of identity, indicating unbiased prediction and similar accuracy. The predicted median ACR was calculated using the equations published in Weaver et al.2 on the basis of a simple linear spline for 2280 ACR/PCR pairs in 627 transplant recipients and 142,049 ACR/PCR pairs in 61,900 non transplant recipients (5% random sample of 7127 pairs shown for clarity).
Second, Jehn et al.1 note departures from the expected values in supplemental figure 2 of their work. We interpret this scatterplot as showing that our equations likely perform adequately in the range of 30–1000 mg/g, with variability as expected, but underestimate the median ACR for predicted ACR <30 and >1000 mg/g. The underestimation (and reduced variability) at lower levels may reflect the paucity of ACR measurements <5 mg/g among their transplant recipients compared with 13% in our transplant recipients. This difference may be related to the use of different lower reporting limits for albumin concentration between laboratories/assays. The underestimation at higher levels may reflect different methods for urinary protein measurement (see discussion and supplemental figure 21 in our study2).
Nevertheless, it is possible that transplantation does affect the ACR/PCR association. Although there is no difference apparent in our data, it would require a larger study to rule out a small difference between the two populations. We do not have data to repeat our analysis in living donors specifically and agree that further research would be helpful in this population as well.
Disclosures
B. Hemmelgarn reports grants from Amgen Canada outside the submitted work. M. Tonelli reports personal fees from B. Braun and grants from Merck outside the submitted work. M. Tonelli also reports that Daichi Sankyo awarded a grant to M. Tonelli’s institution in lieu of a personal honorarium for a lecture at a scientific meeting in 2017 outside the submitted work. All remaining authors have nothing to disclose.
Funding
None.
Supplementary Material
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
Supplemental Material
This article contains the following supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2020050707/-/DCSupplemental.
Supplemental Figure 1. Simulated distribution of differences between predicted and measured values, with positive differences shown on a log scale and negative differences shown on a negative log scale.
References
- 1.Jehn U, Görlich D, Reuter S: The estimation formulae for the urinary albumin-creatinine ratio based on the protein-creatinine ratio are not valid for a kidney transplant and a living donor cohort. J Am Soc Nephrol 31: 1915–1916, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Weaver RG, James MT, Ravani P, Weaver CGW, Lamb EJ, Tonelli M, et al.: Estimating urine albumin-to-creatinine ratio from protein-to-creatinine: Development of equations using same-day measurements. J Am Soc Nephrol 31: 591–601, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Halimi JM, Matthias B, Al-Najjar A, Laouad I, Chatelet V, Marlière JF, et al.: Respective predictive role of urinary albumin excretion and nonalbumin proteinuria on graft loss and death in renal transplant recipients. Am J Transplant 7: 2775–2781, 2007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


