After the recognition early in 2020 that angiotensin-converting enzyme 2 (ACE2) is the main receptor for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),1 concerns were rapidly raised about the use of renin-angiotensin system (RAS) blockers in patients with coronavirus disease 2019 (COVID-19).2–4 The concerns were largely based on previous studies showing that angiotensin II type 1 (AT1) receptor blockers (ARBs) and ACE inhibitors can upregulate ACE2 in certain experimental conditions, although results have not always been consistent.3–6 Nevertheless, the question is important because certain groups of patients at risk of severe COVID-19—including those with hypertension, heart disease, diabetes mellitus, and the elderly—are often treated with RAS blockers.2
In polarized epithelia, like the lungs, kidneys and intestine, ACE2 in its full-length form is anchored to the apical plasma membrane.7,8 Although kidneys have abundant ACE2 in the proximal tubule, lungs have a low level of ACE2 expression.9,10 However, type 2 pneumocytes express ACE2 and, moreover, possess Transmembrane protease, serine 2 (TMPRSS2), a protease critical for priming of the ACE2–SARS-CoV-2 complex, a step needed for cell entry.1,10 The effect of ACE inhibitors and ARBs on ACE2 protein expression in the lungs, however, has not been previously reported. To gain insight into this question, we used kidney and lung lysates to examine the effect of captopril and telmisartan, administered for 2 weeks, to pharmacologically inhibit ACE activity and the AT1 receptor, respectively.
We also studied the effect of kidney ACE deficiency on kidney ACE2 expression in two genetic models of kidney ACE ablation to investigate the effect of kidney ACE deficiency on kidney ACE2 expression. Our findings in the two models of kidney ACE genetic ablation, global in the ACE.4 mice (Supplemental Figure 1) and restricted to the kidney in the ACE8/8 mice (Supplemental Figure 2), revealed that lack of ACE protein was associated with a significant reduction of kidney ACE2 protein that was not accompanied by a reduction in kidney ACE2 mRNA.
Kidney cortex of mice treated with captopril or telmisartan for 2 weeks and respective vehicle-treated controls were used to evaluate ACE2 mRNA, protein, and activity (see methods in Supplemental Appendix). No significant changes in mRNA levels were found between captopril-treated and control mice (99%±21% of control). ACE2 activity and protein were lower in lysates from captopril-treated mice (81%±8% and 71%±5% of control mice, respectively) but the difference did not reach statistical significance. In isolated membranes, however, the decrease in kidney ACE2 protein was profound and statistically significant (37%±4% of the vehicle-treated mice; P=0.0004) (Figure 1A). This decrease in membrane-bound ACE2 was associated with a significant increase in cytosolic ACE2 protein (Figure 1D) suggesting internalization of the protein. By confocal microscopy (Figure 1C), ACE2 staining was mostly apical but could also be seen in the cytoplasm of tubular cells from captopril-treated mice which was less evident in control mice. ACE staining, by contrast, remained restricted to the apical membrane (Figure 1C).
Figure 1.
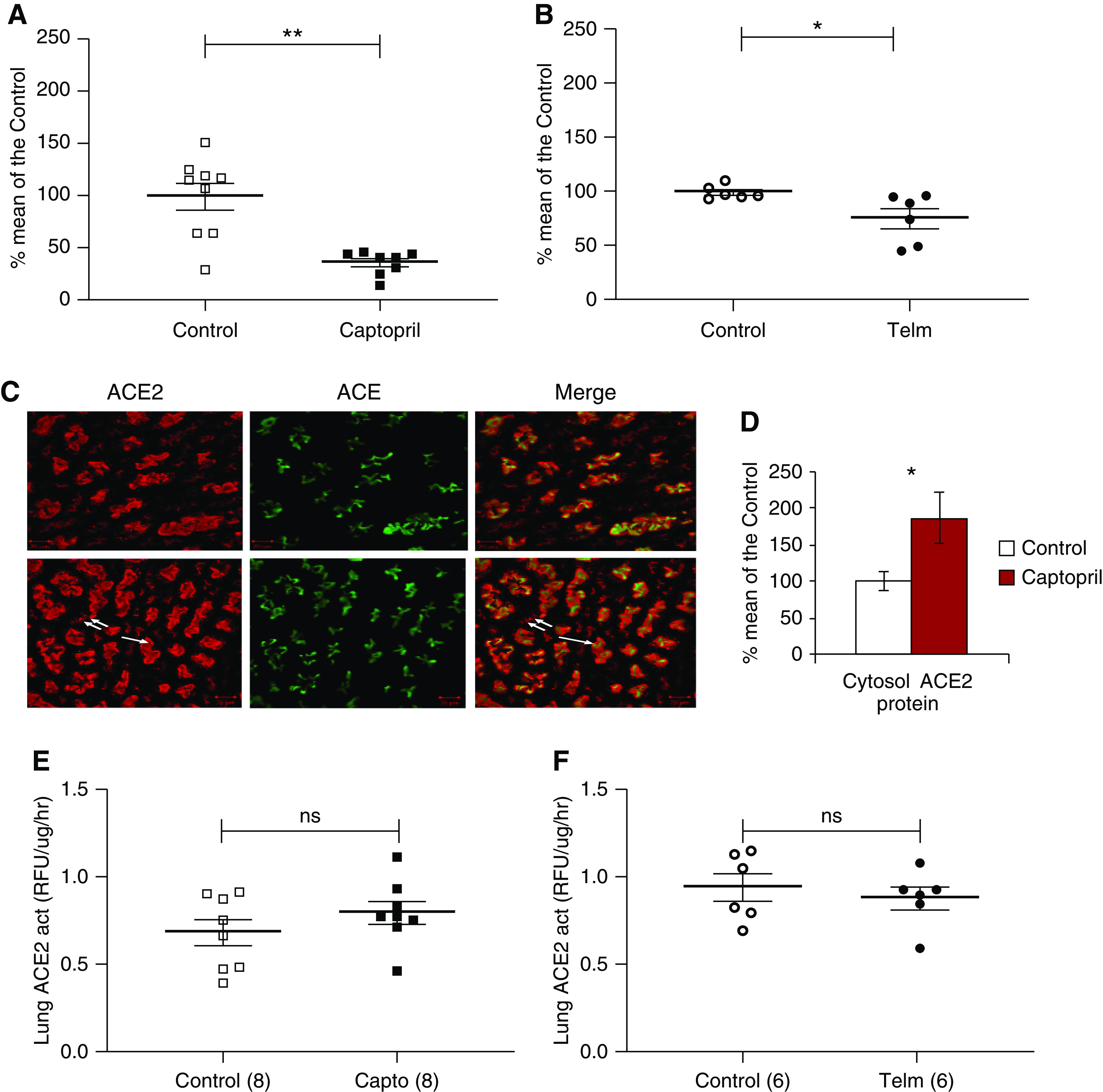
ACE2 expression in kidney and lung membranes from captopril and telmisartan treated mice. (A and B) ACE2 protein in kidney membranes from (A) captopril- and (B) telmisartan-treated mice. *P<0.05; **P<0.01. (C) Confocal microscopy of kidney proximal tubule showing ACE2 (red), ACE (green), and merged image (yellow). ACE2 and ACE are mainly in the apical site in both control (upper panel) and captopril treated mice (lower panel). ACE2 staining is also seen in the cytoplasm (double arrow) of a captopril-treated mouse which is not as evident in control mice. The inset (D) (bar graphs) shows a significant increase in cytosolic ACE2 protein in cytosolic membranes from captopril treated as compared with vehicle-treated mice, *P<0.05. Data are expressed as percentage of control. (E and F) ACE2 enzymatic activity in isolated membrane preparations from lungs from (E) captopril- and (F) telmisartan-treated mice. In lungs, no significant differences were detected as compared to their respective controls. Act, activity; Telm, telmisartan.
ACE2 protein in total kidney lysates from telmisartan-treated mice was also not significantly different from vehicle-treated mice (114%±16%). However, in isolated kidney membranes, there was a significant decrease in ACE2 protein (76%±9% of the vehicle-treated mice; P=0.03) (Figure 1B).
In lung tissue, ACE2 protein is low.9,10 Consistent with this, attempts to perform Western blots with either total lysates or isolated membranes did not yield a signal. Therefore, our results are limited to ACE2 activity that, although low, was consistently detected and can be considered a surrogate for relative protein abundance. Neither captopril nor telmisartan had a significant effect on ACE2 activity in lung total lysates (Supplemental Figure 3).
Likewise, in lung membranes, ACE2 activity was unaffected by either captopril (Figure 1E) or telmisartan (Figure 1F).
In assessing the relative quantities of ACE2 that can act as the SARS-CoV-2 receptor, what matters most is the abundance of full-length, membrane-bound ACE2 protein. Within lung tissue, ACE2 protein is detectable only in type 2 pneumocytes. In this cell type, Transmembrane protease, serine 2 (TMPRSS2), a protease critical for activation and fusion of the SARS-CoV-2 and ACE2 complex, is also present.1,10
In polarized epithelia like in the lungs, kidneys, and intestine, ACE2 in its full-length form is anchored to the apical plasma membrane.7,8,10 Here we show that captopril and telmisartan both decrease kidney ACE2 protein in kidney membranes without significantly affecting protein abundance in total kidney lysates (Figure 1). Captopril, in particular, produced a marked decline in ACE2 protein in kidney membranes while increasing cytosolic ACE2 (Figure 1).
In conclusion, genetic ablation and inhibition of kidney ACE are both accompanied by a reduction of kidney ACE2 expression. This suggests changes in the expression in one enzyme may elicit similarly directional changes in the other homolog such that formation and degradation of angiotensin II can be regulated. The administration of an ACE inhibitor, captopril, and an ARB, telmisartan, decreased ACE2 protein expression in kidney-isolated membranes. Each type of RAS blocker had no detectable effect on ACE2 activity in lung-isolated membranes. These findings altogether show that ACE2 is not increased in two organs that are potential target sites for SARS-CoV-2 infection. In fact, in kidney apical membranes, ACE2 protein is decreased after both captopril and telmisartan administration. Therefore, we conclude that RAS blockers do not increase ACE2 in either lung or kidney epithelia. If changes in full-length ACE2 are indeed sufficient to affect SARS-CoV-2 infectivity, the risk cannot be increased by ACE inhibitors or ARBs. This experimental finding in mouse organs support the position of many medical societies and recent publications expressing the view that the use of RAS blockers should be continued in patients at risk of contracting COVID-19. Ongoing clinical trials may or may not show benefit of using RAS blockers in patients with COVID-19, but what it is clear is that there is no increased risk for infectivity by using RAS blockers.
Disclosures
D. Batlle is a coinventor of the patent “Active Low Molecular Weight Variants of Angiotensin Converting Enzyme 2,” founder of “Angiotensin Therapeutics Inc.”J. Wysocki is a coinventor of the patent “Active Low Molecular Weight Variants of Angiotensin Converting Enzyme 2.” All remaining authors have nothing to disclose.
Funding
D. Batlle was funded by National Institute of Diabetes and Digestive Kidney Diseases grant RO1DK104785.
Supplementary Material
Acknowledgments
This work was supported by the Joseph and Bessie Feinberg Foundation (Dr. Daniel Batlle). We thank Dr. Hong D. Xiao (Providence Portland Medical Center, Portland, OR) and Dr. Kenneth E. Bernstein (Cedars Sinai, Los Angeles, CA) for generously providing kidneys from ACE deficiency models. Dr. Daniel Batlle reports nonfinancial support from Angiotensin Therapeutics Inc., outside the submitted work. Dr. Maria Jose Soler reports personal fees from AstraZeneca, nonfinancial support from Boehringer Ingelheim, nonfinancial support from Eli Lilly, personal fees and nonfinancial support from Esteve, personal fees from FMC, personal fees from Janssen, personal fees from Mundipharma, and personal fees from NovoNordisk, outside the submitted work.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
Supplemental Material
This article contains the following supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2020050667/-/DCSupplemental.
Supplemental Figure 1. ACE2 protein, enzymatic activity and mRNA in kidneys from ACE.4 mice.
Supplemental Figure 2. ACE2 protein, activity and mRNA in kidneys from ACE 8/8 mice and wild type controls.
Supplemental Figure 3. ACE2 activity in total cell lysates from lungs of captopril (A) and telmisartan (B) treated mice.
References
- 1.Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al.: SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181: 271–280.e8, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fang L, Karakiulakis G, Roth M: Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? [published correction appears in Lancet Respir Med 8: e54, 2020]. Lancet Respir Med 8: e21, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Danser AJ, Epstein M, Batlle D: Renin-angiotensin system blockers and the COVID-19 pandemic: At present there is no evidence to abandon renin-angiotensin system blockers. Hypertension 75: 1382–1385, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vaduganathan M, Vardeny O, Michel T, McMurray JJV, Pfeffer MA, Solomon SD: Renin–angiotensin–aldosterone system inhibitors in patients with Covid-19. N Engl J Med 382: 1653–1659, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soler MJ, Ye M, Wysocki J, William J, Lloveras J, Batlle D: Localization of ACE2 in the renal vasculature: Amplification by angiotensin II type 1 receptor blockade using telmisartan. Am J Physiol Renal Physiol 296: F398–F405, 2009. [DOI] [PubMed] [Google Scholar]
- 6.Ferrario CM, Jessup J, Chappell MC, Averill DB, Brosnihan KB, Tallant EA, et al.: Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation 111: 2605–2610, 2005. [DOI] [PubMed] [Google Scholar]
- 7.Ye M, Wysocki J, William J, Soler MJ, Cokic I, Batlle D: Glomerular localization and expression of angiotensin-converting enzyme 2 and angiotensin-converting enzyme: Implications for albuminuria in diabetes. J Am Soc Nephrol 17: 3067–3075, 2006. [DOI] [PubMed] [Google Scholar]
- 8.Hamming I, Timens W, Bulthuis MLC, Lely AT, Navis GJ, et al. H: Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 203: 631–637, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Serfozo P, Wysocki J, Gulua G, Schulze A, Ye M, Liu P, et al.: Ang II (angiotensin II) conversion to angiotensin-(1-7) in the circulation is POP (prolyloligopeptidase)-dependent and ACE2 (angiotensin-converting enzyme 2)-independent. Hypertension 75: 173–182, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ziegler CGK, Allon SJ, Nyquist SK, Mbano IM, Miao VN, Tzouanas CN, et al.: HCA Lung Biological Network : SARS-CoV-2 receptor ACE2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell 181: 1016–1035.e19, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


