Abstract
The prevalence of non-alcoholic fatty liver disease (NAFLD) is increasing worldwide and parallels comorbidities such as obesity, metabolic syndrome, dyslipidemia, and diabetes. Recent studies describe the presence of NAFLD in non-obese individuals, with mechanisms partially independent from excessive caloric intake. Increasing evidences, in particular, point towards a close interaction between dietary and environmental factors (including food contaminants), gut, blood flow, and liver metabolism, with pathways involving intestinal permeability, the composition of gut microbiota, bacterial products, immunity, local, and systemic inflammation. These factors play a critical role in the maintenance of intestinal, liver, and metabolic homeostasis. An anomalous or imbalanced gut microbial composition may favor an increased intestinal permeability, predisposing to portal translocation of microorganisms, microbial products, and cell wall components. These components form microbial-associated molecular patterns (MAMPs) or pathogen-associated molecular patterns (PAMPs), with potentials to interact in the intestine lamina propria enriched in immune cells, and in the liver at the level of the immune cells, i.e., Kupffer cells and stellate cells. The resulting inflammatory environment ultimately leads to liver fibrosis with potentials to progression towards necrotic and fibrotic changes, cirrhosis. and hepatocellular carcinoma. By contrast, measures able to modulate the composition of gut microbiota and to preserve gut vascular barrier might prevent or reverse NAFLD.
Keywords: body weight, fat, fatty liver, genetics, intestinal permeability, leaky gut, NAFLD
1. Introduction
Non-alcoholic fatty liver disease (NAFLD) is the most common cause of chronic liver disease worldwide [1].
The acronym NAFLD refers to the presence of hepatic steatosis without other causes for secondary hepatic fat accumulation. By definition, NAFLD subjects have no signs of acute or chronic liver disease other than fatty liver and consume no or little alcohol [2] (Table 1).
Table 1.
Most frequent causes of liver steatosis.
| Metabolic: Nonalcoholic fatty liver (NAFLD) or metabolic-dysfunction-associated fatty liver disease (MAFLD) |
| Alcoholic liver disease (ALD) |
| Hepatitis C (in particular genotype 3) |
| Lipodystrophy |
| Parenteral nutrition |
| Starvation |
| Wilson disease |
| Abetalipoproteinemia |
| Drugs (e.g., methotrexate, tamoxifen, glucocorticoids, amiodarone, valproate, anti-retroviral agents for HIV) |
| Acute fatty liver of pregnancy |
| HELLP (hemolytic anemia, elevated liver enzymes, low platelet count) syndrome |
| Reye syndrome |
| Inborn errors of metabolism (LCAT deficiency, cholesterol ester storage disease, Wolman disease) |
| Drug-induced liver disease |
| Food contaminants of environmental origin (e.g., endocrine disrupting chemicals, persistent organic pollutants, metals) |
In the USA, NAFLD occurs in more than 65 million residents and causes enormous economic burden, i.e., >100 billion USD annually [3]. NAFLD worldwide has a prevalence of almost 30–40% in the adult population and increases in type 2 diabetes mellitus (50%) [1,4,5], obesity (30–76%) [6], and morbid obesity (up to 90%) [7,8].
Early identification and targeted treatment of patients with NAFLD can prevent consequences related to complications (including management of end-stage disease and HCC) and rising health cost. Further beneficial effects can derive the reduction of risk factors for extrahepatic complications, which include cardiovascular disease and malignancy [9,10].
This review discusses the most recent updates on the gut–liver axis and the aspects involving the gut barrier, intestinal permeability, the microbioma, and the role of local molecules and environmental factors with respect to emerging problem of NAFLD worldwide (Figure 1).
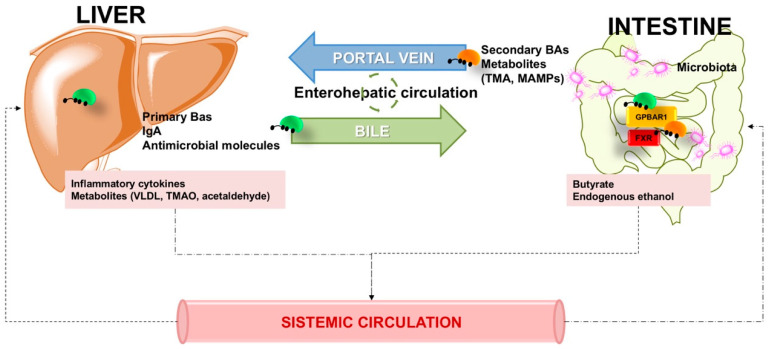
Figure 1.
The communication between the liver and the gut is bidirectional. The liver secretes primary bile acids and antimicrobial molecules (immunoglobulin A (IgA) and angiogenin) into the biliary tract. Molecules reach the intestinal lumen and contribute to maintenance of gut eubiosis (while inhibiting intestinal bacterial overgrowth). During the enterohepatic circulation of bile, BAs act as signalling molecules by interacting with the nuclear receptor farnesoid X receptor (FXR) and the G protein-coupled bile acid receptor (GPBAR1). This interaction contributes to modulate hepatic bile acid synthesis, glucose metabolism, lipid metabolism and dietary energy utilization. Gut products include host and/or microbial metabolites and microbial-associated molecular patterns (MAMPs). The systemic circulation also plays a role, since products are translocated to the liver via the portal vein and can influence liver functions. The systemic circulation extends the gut–liver axis since liver metabolites from dietary, endogenous or xenobiotic substances (free fatty acids, choline metabolites and ethanol metabolites) are transported to the intestine through the capillary system. This mechanism of continuous recirculation of molecules across blood capillaries can distinctively affect the intestinal barrier. Butyrate, the short chain fatty acids, is protective (i.e. improves the colonic defensive border), while acetaldehyde can activate a barrier damage. Abbreviations: BAs, bile acids; MAMPs, microbial-associated molecular patters; TMA, trimethylamine; TMAO, trimethylamine N-oxide; VLDL, very-low density lipoprotein. Adapted from Tripathi et al. [11] and Di Ciaula et al. [12].
The liver secretes primary bile acids and antimicrobial molecules (immunoglobulin A (IgA) and angiogenin) into the biliary tract. Molecules reach the intestinal lumen and contribute to maintenance of gut eubiosis (while inhibiting intestinal bacterial overgrowth). During the enterohepatic circulation of bile, bile acids (BAs) act as signaling molecules by interacting with the nuclear receptor farnesoid X receptor (FXR) and the G protein-coupled bile acid receptor (GPBAR1). This interaction contributes to modulate hepatic bile acid synthesis, glucose metabolism, lipid metabolism, and dietary energy utilization. Gut products include host and/or microbial metabolites and microbial-associated molecular patterns (MAMPs). The systemic circulation also plays a role, since products are translocated to the liver via the portal vein and can influence liver functions. The systemic circulation extends the gut–liver axis since liver metabolites from dietary, endogenous, or xenobiotic substances (i.e., free fatty acids, choline metabolites, and ethanol metabolites) are transported to the intestine through the capillary system. This mechanism of continuous recirculation of molecules across blood capillaries can distinctively affect the intestinal barrier. Butyrate, the short chain fatty acid, is protective (i.e., improves the colonic defensive border), while acetaldehyde can activate a barrier damage.
2. Risk Factors, Clinical Features, and Pathophysiology of NAFLD
2.1. Definition of NAFLD
The term NAFLD comprises a wide spectrum of conditions [13,14], ranging from simple non-alcoholic fatty liver (NAFL) without significant inflammation (the majority of cases), to non-alcoholic steatohepatitis (NASH) (affecting about 5% of the population). In NASH, hepatic steatosis is associated with pericellular fibrosis, ballooning degeneration of hepatocytes, lobular inflammation, and apoptosis [15]. NASH has potential to progress to (cryptogenic) cirrhosis in 20% of cases. Furthermore, hepatocellular carcinoma (HCC) can originate from cirrhosis [6,16,17,18] but also from NASH [19].
2.2. Risk Factors
NAFLD can be considered as the hepatic expression of gluco-lipid metabolic disturbances that includes insulin resistance and type 2 diabetes mellitus, expansion of visceral fat/frank obesity, dyslipidemia (involving hypertriglyceridemia and low HDL-cholesterol), and ultimately metabolic syndrome (MetS). In particular, triglyceride/HDL-cholesterol ratio [20] and total cholesterol/HDL-cholesterol ratio [21] are independently linked with the presence of NAFLD, being considered reliable predictors of liver disease.
The term NAFLD has recently received a different acronym, i.e., MAFLD (metabolic-dysfunction associated fatty liver disease). According to this novel definition, MAFLD consists of “inclusive” (not only exclusive) criteria relying on evidence of hepatic steatosis, in addition to one of the following three criteria, namely overweight/obesity, presence of type 2 diabetes mellitus, or evidence of metabolic dysregulation [22]. Indeed, body mass index (BMI) correlates with the increasing prevalence of both NAFLD and NASH [23,24]. The cut-off values for overweight and obesity, however, differ with ethnicity. In the Caucasian race, overweight and obesity occur at a BMI 25.0–29.9 kg/m2 and ≥30 kg/m2, respectively. In the Asian population, overweight and obesity occur at a BMI 22.5–24.9 kg/m2 and ≥25 kg/m2, respectively. Furthermore, Asia has the most worrisome trends with BMI when stratified by ages [25]. NAFLD can be a feature in obese subjects without metabolic abnormalities or even nonobese subjects (body mass index (BMI) < 25 kg/m2) with insulin resistance [26,27].
Thus, NAFLD can also develop in lean (likely metabolically-impaired) subjects as well [28] and this possibility is frequent in Asia [29]. Besides genetic factors, the onset and progression of NAFLD in lean subjects is related with dietary and environmental factors, and show metabolic characteristics (i.e., insulin-resistance, dyslipidemia) similar to those observed in overweight and obese subjects [30].
Several lean individuals in the United States suffer from NAFLD, possibly due to the coexistence of diabetes and/or hypertension [31]. In a large cross-sectional Asian study, the presence of NAFLD or AFLD was associated in non-obese subjects (BMI < 25 kg/m2) with the score of coronary artery calcification, an expression of subclinical atherosclerosis [32]. Additionally, NAFLD prevalence and liver fibrosis has been found to increase with age [33].
2.3. Clinical Aspects
While most subjects with NAFLD remain asymptomatic, some patients, especially those developing NASH, may complain of vague symptoms (i.e., fatigue, malaise, and right upper abdominal discomfort). NAFLD subjects could ask for medical advice because of mildly elevated liver alanine aminotransferases or incidental increased echogenicity (“bright liver”) at ultrasound (or eventually decreased hepatic attenuation by computed tomography, as well as increased fat signal by magnetic resonance imaging). NAFLD, however, may develop with normal aminotransferase levels. The ultimate diagnosis of NAFLD requires the confirmation of steatosis by liver histology or imaging, no relevant alcohol consumption. or other causes of liver steatosis, and exclusion of a coexisting other chronic liver disease.
The early fat storage in the liver could drive subclinical liver abnormalities, leading to NAFLD development and progression, that can increase risk for advanced liver disease and liver-related mortality [33]. NAFLD also increases the risk of non-liver-related complications, such as cardiovascular disease and malignancy [2,9,10,34,35,36]. In addition, patients with NAFLD suffer from poor quality of life as compared to healthy individuals. A marked worsening occurs with advanced liver diseases, such as liver cirrhosis [37,38]. No specific therapy exists for NAFLD, apart from healthy lifestyles [28,39]. The utility of systematic screening of the population is still debated because of unclear cost-effectiveness [40]. Specific research protocols can target subgroups of patients with symptomatic or asymptomatic NAFLD [41].
2.4. Pathophysiology of NAFLD
The pathophysiology of NAFLD is complex, multifactorial, and partly unknown [28,42,43].
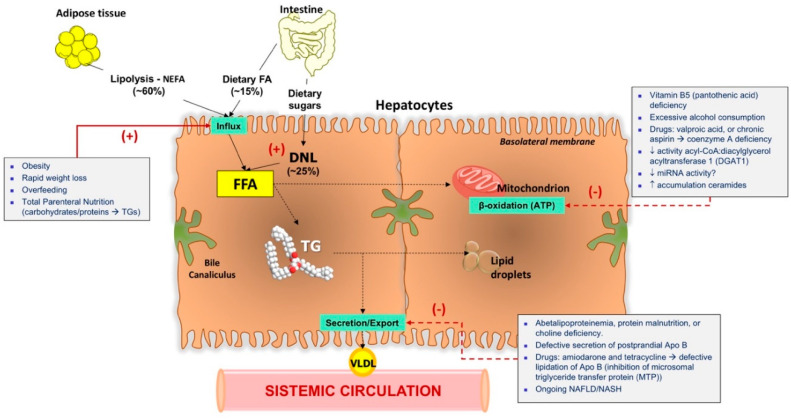
NAFLD is due to excessive (>5%) storage of hepatic triglycerides (TG) as micro- and macro-vesicular deposits, associated with an accumulation of free fatty acids (FFAs), ceramides, and cholesterol [44,45]. In health, as shown in Figure 2, there are three essential steps of FFA accumulation in the liver:
-
(a)Influx of non-esterified fatty acids (NEFA): This step accounts for ~60% of total FFAs in the liver and brings to enrichment of the FFA pool in hepatocytes. NEFA originates from lipolysis of triglycerides in adipocyte under the control of insulin [46]. Once in the liver, FFAs have different fates:
- As fatty acyl-CoA, FFAs can enter the mitochondria under the control of carnitine palmitoyltransferase (CPT)-1 and undergo β-oxidation to acetyl-CoA joining the tricarbossilic acid cycle with production of ATP;
- FFAs are also esterified to TG (no more than 5% in the hepatocyte) via the key enzymes diaglyceride acyltransferase (DGAT)1 and DGAT2. This step is also controlled by insulin and excess TG can be stored as lipid droplets or exported to blood as very-low-density lipoprotein [47].
-
(b)
De novo lipogenesis (DNL) of FAs: This step accounts for ~25% of total FFAs in the liver and originate from dietary sugars. With DNL, hepatocytes converts excess glucose and fructose to FAs. Insulin mediates the transport of absorbed dietary carbohydrates to its target tissues (skeletal muscle and liver for storage as glycogen). Glucose in hepatocytes is partly metabolized to pyruvic acid via glycolysis and then to acetyl-CoA, to generate ATP in the tricarboxylic acid cycle and promote gluconeogenesis during hypoglycemia. The first step of DNL is the synthesis of malonyl-CoA from cytosolic acetyl-CoA by acetyl-CoA carboxylase (ACC). Malonyl-CoA serves as a substrate to form saturated FA via FA synthase (FAS). Stearoyl CoA desaturase (SCD)1 is an endoplasmic reticulum (ER) enzyme responsible for the formation of monounsaturated FAs from saturated FAs [41].
-
(c)
Influx of dietary FAs: Influx of FAs from diet is ~15% of all amount of FFAs in the liver [48]. Bile acids (BA) hydrolyze intestinal dietary TGs to form nascent chylomicrons. The FAs made from TG hydrolysis are taken up by adipose tissue and liver [49]. At the same time, BA acts as a potent metabolic regulator in the terminal ileum by activating the farnesoid X receptor (FXR) plus the pregnane X receptor (PXR), as well as the G-protein-coupled bile acid receptor-1 (GPBAR-1). These interactions produce effects in the liver and in the muscle, in adipocytes and brown adipose tissue for energy expenditure [12,50].
Figure 2.
Mechanisms of lipid accumulation in the liver. In health, free fatty acids (FFA) originate from lipolysis of triglycerides (TG) in adipose tissue leading to circulating non-esterified fatty acids, (NEFA, ~60%), from dietary fatty acids (~15%), and from dietary sugars undergoing de novo lipogenesis (DNL, ~25%) mediated by several proteins (sterol regulatory element-binding protein 1, stearoyl CoA-desaturase, and fatty acid synthase). FA in hepatocytes undergo mitochondrial β-oxidation or re-esterification with glycerol to form TG. TG can be stored in lipid droplets or exported to blood as very-low density lipoproteins (VLDL). NAFLD is the consequence of excess flow of FFA and accumulation of intrahepatic TG due to several abnormalities depicted in the boxes, and associated with increased influx of FFA, increased DNL, decreased β-oxidation, and decreased secretion/export of TG as very-low density lipoproteins (VLDL). Abbreviations: DNL, de novo lipogenesis; FFA, free fatty acids; NEFA, non-esterified fatty acids; TG, triglycerides; VLDL, very-low density lipoproteins. (+), increased; (−), decreased.
Metabolic abnormalities can markedly disrupt the pathways governing FFA/TG/lipid metabolism in the liver, leading to NAFLD and damage of hepatocyte (Figure 3 and Figure 4). The progressive development of insulin resistance, expansion of visceral obesity, sedentary behaviors, and high-calorie diet can all affect FFA homeostasis. This ongoing inflammation induced by metabolic stress is associated with an increase of lipolysis and influx of NEFA, de-novo lipogenesis, influx of dietary FFA (especially fructose), TG deposition in droplets, and the decrease of FFA in mitochondrial oxidation, or secretion/export of FFA or TG into VLDL [51]. Ingested glucose is re-directed to the liver during insulin resistant status [47] and glucose is primarily processed to FA via DNL [46,52]. If FFAs synthesis increases, they are converted to fatty acyl-CoA, which is further esterified into TGs and stored in hepatocytes. DNL is also enhanced in NAFLD patients [47,53]. In general, the oversupply or disrupted disposal free fatty acids (FFAs) in the liver activates mechanisms that leads to the assembly of lipotoxic species, i.e., FFAs, TGs, Lysophosphatidylcholine (LPC), ceramides, and free cholesterol (Table 2).
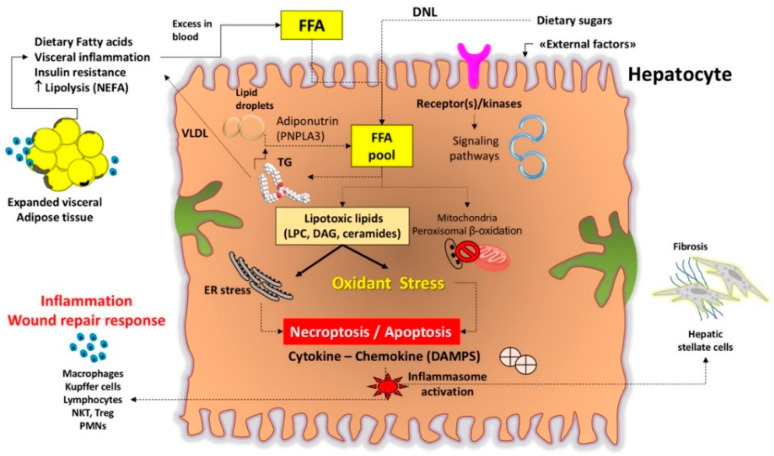
Figure 3.
Mechanisms of lipotoxicity in the liver. Excessive accumulation of free fatty acids (FFA) is the result of increased influx, increased synthesis, decreased mitochondrial oxidation of FFA, or decreased secretion/export of FFA. Adiponutrin (also known as patatin-like phospholipase domain-containing protein 3, PNLPA3) governs the lipolysis of lipid droplets with FA flowing back to the hepatocyte FFA pool. With excess FFA, their disposal through beta-oxidation or formation of triglyceride is insufficient and lipotoxic species form (Lysophosphatidylcholine, LPC; diacylglycerol, DAG; ceramides) that mediate endoplasmic reticulum (ER) stress, oxidant stress and activation of the inflammasome (the multiprotein cytoplasmic complex that responds to damage-associated molecular patterns (DAMPs), as part of the innate immunity response). External factors, poorly understood so far, may include dysregulation of cytokines and adipokines, ATP depletion, toxic uric acid, sleep apnea leading to periodic hypoxia, and products of the gut microbiome (tumor necrosis factor (TNFa), endogenous ethanol, and endotoxins like lipopolysaccharides (LPS)). Such events promote the phenotype of NASH, hepatocellular injury, inflammation, stellate cell activation, and progressive accumulation of excess extracellular matrix. Further target structures of damage are other intracellular organelles, the nucleus, receptors and signaling pathways. See also [10,41,72,73].
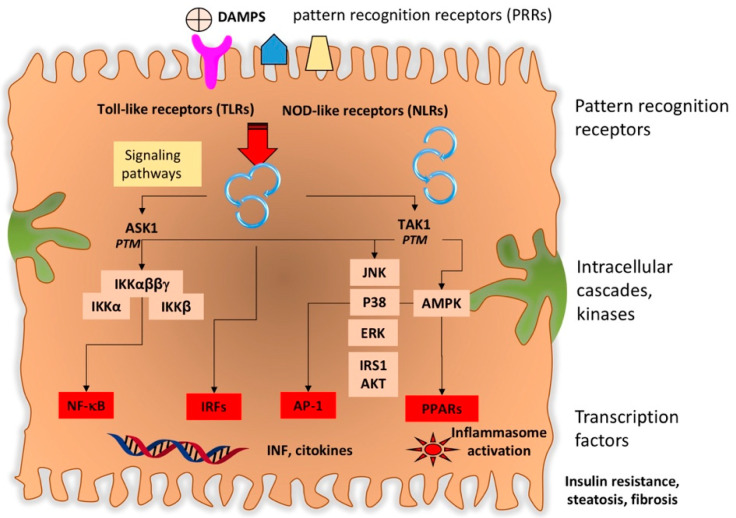
Figure 4.
Detailed picture representing the ultimate consequence of circulating damage-associated molecular patterns (DAMs) on hepatocytes. DAMs represent the continuous stimulus for at least three intracellular events in the hepatocyte: (1) response of pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs) and NOD-like receptors (NLRs). (2) Activation of intracellular cascades, kinases representing the downstream signaling pathways, such as the apoptosis signal-regulating kinase 1 (ASK1) and TGF-b-activated kinase 1 (TAK1). Both kinases are activated through post-transcriptional modification (PTM) and activate other key kinases (C-Jun N-terminal kinase, JNK), AMP-activated kinase, AMPK, and IkB). (3) Activation of transcription factors such as interferon regulatory factors (IRFs), nuclear factor (NF)-kB, activator protein 1 (AP-1), and peroxisome proliferator-activated receptors (PPARs). This ultimate event leads to the production of inflammatory cytokines and chemokines, with all metabolic consequences observed in NAFLD (insulin resistance, steatohepatitis, fibrogenesis, etc.). Additional endogenous targets regulating the innate immune elements in NASH include CASP8 and FADD-like apoptosis regulator (CFLAR), tumor necrosis factor (TNF) a-induced protein 3 (TNFAIP3), cylindromatosis (CYLD), transmembrane BAX inhibitor motif-containing 1 (TMBIM1), TNF receptor-associated factor 6 (TRAF6), TRAF1, TRAF3, dual-specificity phosphatase 14 (DUSP14), tripartite motif 8 (TRIM8), dickkopf-3 (DKK3), and TRAF5 (See also [41]).
Table 2.
Lipid species accumulating in NAFLD.
| Free Fatty Acids (FFA) |
|
| Triglycerides (TG) |
| Lysophosphatidylcholine (LPC) |
| Ceramides |
|
| Free Cholesterol |
|
Notably, the onset and progression of NAFLD does not seem to be exclusively secondary to an exaggerated caloric intake. In particular, loaded cholesterol in the hepatocyte can impair mitochondrial and lysosomal function, with direct toxic effects (i.e., oxidative stress and lipotoxicity) in both nonobese and obese patients [60]. Free cholesterol and oxLDL can also accumulate in the portal vein wall and this step can predispose to portal venous NLRP3 inflammasome-mediated inflammation and fibrosis leading to NAFLD [61].
Furthermore, the onset and progression of NAFLD can also be linked with exposure to environmental chemicals (e.g., volatile organic chemicals, persistent organic pollutants, metals, particulate matter, pesticides) through several mechanisms disrupting biochemical, metabolic, signaling, and endocrine pathways [62].
In general, the mechanisms of lipotoxicity involved in NAFLD onset and progression are multiple and involve receptor/kinases-mediated interactions, the endoplasmic reticulum/other intracellular organelles, mitochondria [63], the nucleus, and several signaling pathways. Endoplasmic reticulum stress, inflammatory changes (metabolic inflammation or meta-inflammation) [64], production of reactive oxygen species, and cell death [65] represent further events associated with liver damage. The innate immune system contributes to metabolic inflammation with the recruitment of Kupffer cells, dendritic cells, lymphocytes, as well as hepatocytes and endothelial cells [66,67].
Ultimately, the activation of several transcription factors promotes the release of inflammatory cytokines and chemokines, and stimulates hepatic stellate cells with propensity to collagen deposition, and further aggravation of the insulin resistance status, both potential harmful features in the progression of NASH [68]. In addition, the adipose tissue, skeletal muscle, the heart, the pancreatic islets, certain areas of the brain (mainly hippocampus, cerebellum, hypothalamus), and the intestinal microbiota represent additional organs potential targets of lipotoxicity [41,69,70,71].
3. Beyond NAFLD: The Gut Liver-Axis, the Gut Barrier, and the Liver Barrier
NAFLD develops because of the interaction of genes (epistasis) and environmental factors (exposome) [74,75]. The environmental factors act through intestinal, microbial, and dietary modifications [11,76], and can be linked with exposure to food contaminants, contaminated consumer products or air pollution [77,78,79,80,81,82,83,84].
Changes in this subtle equilibrium involving networking, bidirectional cross-talk, and control of inflammation paves the way to initiation, perpetuation, and aggravation of metabolic liver damage [85]. For example, a diet enriched in fat could easily change the intestinal mucus [86] and the microbiome, leading to alterations in the intestinal barrier and gut vascular barrier, therefore promoting the influx of bacterial products in the portal tract towards the liver [87] with consequences on local and systemic inflammation controlling metabolic abnormalities.
The term gut-liver axis refers to the bidirectional relationship between the intestinal microbiome, the gut, and the liver [88]. There is a close communication between the liver and the intestine, which also involves the biliary tract, the portal vein, the systemic circulation, and a series of systemic mediators [11]. The interaction is bidirectional since gut-derived products permeate the intestine going through the portal vein to the liver, while liver-secreted bile and antibodies are flowing from the liver through the intestine. Moreover, BAs are markedly influenced by the intestinal microbiome but also control the intestinal microbiome, besides their classical physiologic functions that allow food digestion and absorption, as well as stimulation of nuclear receptors and specific receptors at the intestinal level [12,89,90]. The gut barrier integrity results from a balanced cross-talk between microbiome, mucus, intestinal cells, immunological system and gut-vascular barrier. The stability of this equilibrium is essential in maintaining intestinal homeostasis [91,92,93] (Figure 5).
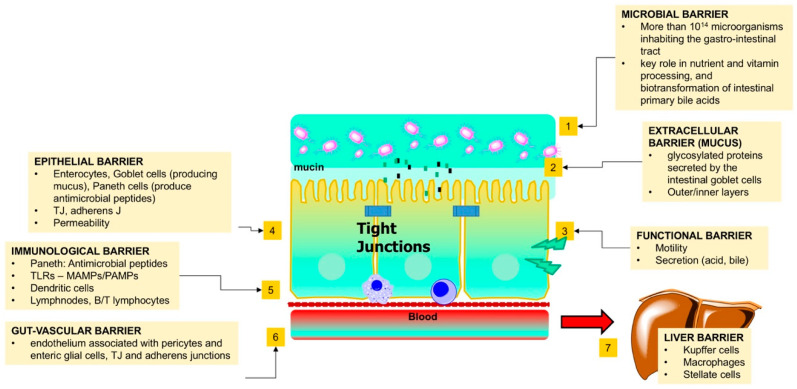
Figure 5.
Components of the physiological gut barrier. The number of microorganisms inhabiting the gastro-intestinal tract has been estimated to exceed 1014. Starting from the gut lumen, we observe microbes (1) in the most superficial layer of the mucous (2). The functional barrier (3) includes the role of gastrointestinal motility ad secretion of gastric acid and bile the epithelial barrier (4) is composed by the collection of gut cells and tight junctions. The immunological barrier (5) includes antimicrobial peptides, toll-like receptors, microbial- (MAMPs) and pathogen- (PAMPs) associated molecular patterns, and B/T lymphocytes. The gut vascular barrier (6) and the ultimate liver barrier (7) are depicted, as well. Adapted from Nicoletti et al. [91].
3.1. Intestinal Microbiome
A first level of the gut barrier is the resident microbiota, which is composed by hundred trillions of microorganisms [94], with more than 10 times the genes of the human genome and a weight of about 1–2 kg [95]. The gut microbiota plays a key role in nutrient and vitamin processing, and biotransformation of intestinal primary BAs to secondary BAs [12,89,96].
3.2. Intestinal Mucus
A second level of gut barrier is the intestinal mucus, i.e., heavily glycosylated proteins secreted by the intestinal goblet cells [97]. This barrier represents an extracellular layer. The thickness of the mucus increases from the stomach towards the colon [98,99]. An important function of the mucus is the prevention of direct contact between harmful/toxic substances/microbiome with the enterocyte. The microbiota in health is physically separated from the intestinal epithelium by the mucus. The microbiota colonizes the outer layer of the mucus and microorganisms use nutrients from the mucus itself.
In general, the microbiota is able to cross-talk with the host via several metabolic products. Secondary metabolites include short chain fatty acids (SCFAs) like propionate, butyrate, acetate (produced during dietary fiber degradation), vitamins, and immunomodulatory peptides [100,101,102,103,104].
The outer layer is populated by bacteria attached directly or via mucin-IgA interaction (IgA produced by plasma cells and transported transcellularly in the enterocytes) and prevents the washing effects of peristalsis [105]. Paneth cells also dismiss antibacterial lectins, such as the regenerating islet-derived protein III (REG3G) that inhibits bacterial adhesion to the mucosa. While intestinal mucin controls the layering of the microbiota in the lumen, the microbiota itself contributes to shape the mucin [106], likely via signaling to goblet cells and the pathway involving the inflammasome NLRP6 [107]. Bacteria also stimulates cell-mediated immunity via Toll-like receptor (TLR)-mediated signaling [108]. In addition, some bacteria use mucus for nutrition and modulate inflammatory changes. In particular, Akkermansia muciniphila is a mucus degrading bacteria and its abundance is higher close to the mucus layer [109]. This anaerobe, Gram negative, mucus degrading specialist populates the intestinal lumen [110,111] and its reduced abundance is associated with inflammation, impaired barrier integrity, and non-alcoholic liver damage [112,113]. Mucin-degrading bacteria increase and mucus thickness decreases in the absence of dietary fiber [114]. Mucin glycosylation is also under the control of the ratio Bacteroides:Firmicutes [115]. Secondary metabolites are able to modulate other function, namely the differentiation of immune cells, i.e., T regulatory cells [116], macrophages, and microbicidal activity [117]. Effects of secondary metabolites are possible upon fiber metabolization, which involve white and brown adipose ratio [118].
Notably, the inner mucus layer is sterile because it does not harvest bacteria due to enrichment in in antimicrobial peptides and in proteins excluding bacteria (lypd8 and zymogen granulae protein 16, ZG16) [119] is rather static (unstirred), and is in contact with epithelial cells. This level contributes to the absorption of water and nutrients [98].
In summary, the mucus is a dynamic structure conferring protection to the host [98,120]. Changes of mucus and diet have consequences on microbiota distribution and composition. In ulcerative colitis microorganism come in contact with the epithelium and can perpetuate the local inflammation [121]. If the mucus function fails and qualitative/quantitative changes of mucus occur, inflammation is possible with absorption of toxic substances, as seen in cystic inflammatory bowel disease (IBD) and cystic fibrosis. Mice models show that a high MUC2 mucin production increases the susceptibility of goblet cells to apoptosis and endoplasmic reticulum stress, while alcohol intake and cirrhosis is associated with increased mucus thickness. In mice, abnormal MUC2 inside the epithelial cells leads to inflammatory changes, which resemble the ulcerative colitis. In addition, high-fat diets can disrupt the intrinsic structure of colonic mucin, as evident in mice developing liver steatosis [86,122].
3.3. Gastrointestinal Motility, Secretions, and Enterohepatic Circulation of BAs
A third level of the gut barrier is a dynamic assembly. It depends on the kinetics of gastrointestinal motility and secretions, with both events influencing the outer part of the mucus layer. This situation prevents the proliferations of microorganism and provides clearance of luminal debris, contributing to protection against pathogens. Fundamental fluids are the gastric acid and bile containing BAs as one of the three species of biliary lipids (together with cholesterol and phospholipids) [12]. Both fluids have antimicrobial properties [91]. In the stomach and small intestine, only Helicobacter pylori and Lactobacilli respectively survive in the acidic environment [123]. Change of these conditions might lead to both qualitative and quantitative modifications of the gut microbiota composition, abnormal intestinal homeostasis, and disease [91].
The enterohepatic circulation of bile and BAs plays a key role at the level of the gut-liver axis and the intestinal microbiota is in close, bidirectional contact with BAs [124,125,126,127,128]. The maintenance of the body BA pool depends on hepatic BA synthesis, biliary secretion, gallbladder concentration and contraction, intestinal transit, microbial biotransformation, intestinal re-absorption, and fecal excretion. In the liver the primary BAs (cholic acid (Ca) and chenodeoxycholic acid (CDCA)) are synthesized from cholesterol within the “classical pathway” by the rate-limiting microsomal enzyme cholesterol 7α-hydroxylase (CYP7A1) and CYP8B1 at a later step. Within the “alternative pathway”, the CYP27A1 enzyme is involved in BAs synthesis. BAs are conjugated to the amino acids glycine or taurine by the enzymes BA CoA synthase (BACS) and BA-CoA-amino acid N-acetyltransferase (BAAT). This process increases the solubility of BAs for secretion into bile by the bile salt export pump (BSEP) [129]. During fasting, most of the bile flows from the liver and is concentrated into the gallbladder, which acts as a reservoir. Gallbladder contraction in the fasting state secretes bile periodically into the duodenum where the intestinal circulation starts. About 20% emptying takes place in the fasting state at the end of phase II of the migrating myoelectric complex [130,131] under the control of the vagus and enterohormone motilin [131]. More than 50–60% gallbladder emptying occurs after a meal due to the enterohormone cholecystokinin (CCK) [132]. Periodic episodes of gallbladder relaxation/refilling several times during the day, accounting for the rhythmic activity of the gallbladder and pulsatile secretion of BAs in the intestine. This function requires the inhibitory effect of mediators on the gallbladder like the vasointestinal peptide (VIP, released in the duodenum by gastric acid) [133], BAs (via the gallbladder receptor GPBAR-1) [12,127], and the intestinal FGF15/19 (following the BA/FXR interaction in the ileum) acting on the FGF4/β-Klotho receptor also expressed in the gallbladder [132,134,135].
Most of the BAs (>95%) undergo terminal ileal active reabsorption into the portal vein and back to the liver [136], while the remaining BAs enter the colon, undergo biotransformation (deconjugation, dehydrogenation, and dehydroxylated) to the secondary BAs (deoxycholic acid, DCA and litocholic acid, LCA), and tertiary bile acid deoxycholic acid (DCA) by the resident gut microbiota. Secondary/tertiary BAs undergo passive diffusion and reabsorption to the liver through the portal vein [49]. In the ileocyte, BA uptake, intracellular transport and secretion into the portal vein require the apical sodium dependent bile acid transporter (ASBT), the cellular intestinal BA binding protein (I-BABP), and the basolateral heterodimeric organic solute transporter (OSTα/β), respectively. BAs are minimally (<5%) lost in the feces and this amount equals the amount synthetized daily and secreted in the liver. About 10–50% of re-absorbed BAs undergo peripheral spillover into systemic circulation [137]. The liver recycles absorbed BAs and secretes them back to the biliary tract. This is the so-called enterohepatic circulation which, several times every day, is the exchanging mechanism between the gut and the liver.
In the terminal ileum, primary BAs entering the enterocyte, act on the orphan farnesoid-X receptor (FXR = NR1H4) leading to increased enterokine fibroblast growth factor 19 (FGF19) that enters the portal circulation with effects on the gallbladder and liver. In the liver, FGF19 binds to FGF receptor 4 (FGFR4)/β-Klotho, a step activating c-Jun N-terminal kinase/extracellular signal-regulated kinase (JNK/ERK) signaling, which inhibits expression of CYP7A1 and CYP8B1 and hepatic BA synthesis, in synergy with the FXR-SHP inhibitory pathway [124,138]. BAs enter the liver via sodium taurocholate cotransporting polypeptide (NTCP) and organic anion transporting polypeptide (OATP), acting act as physiological nuclear ligands for FXR, which regulates target gene transcription by binding to RXRs as a heterodimer [139]. This results in increased transcription of the small heterodimer partner (SHP) expression. SHP, in turn, inhibits LRH-1, preventing the activation of target genes that participate in BA and fatty acid synthesis. In the absence of BAs, LRH-1 acts together with LXR to stimulate BA synthesis [140,141,142]. FXR also regulates the enzymatic activity that is involved in BA conjugation to glycine or taurine and hepatic BA secretion by of BSEP, and hepatic phospholipid secretion by ABCB4. BAs re-entering the liver also interact with the liver GPBAR-1 expressed in Kupffer cells, in concert with the pathway activated by the FGFR4/β-Klotho. FXR activation also coordinates BA detoxification enzymes (i.e., cytosolic sulfotransferase 2A1 (SULT2A1), aldol-keto reductase 1 B7 (AKR1B7), cytochrome P450 3A4/3a11 (CYP3A4/Cyp3a11), and UDP-glycosyltransferase 2B4 (UTG2B4)) [143].
In the ileum, BAs also activate the ileal G protein-coupled receptor (GPBAR-1 = TGR5), leading to secretion of three hormones, peptide YY (PYY), glucagon-like peptide 1 (GLP-1), and glucagon-like peptide 2 (GLP-2). This interaction has metabolic effects on glucose metabolism [144], insulin metabolism [145], energy expenditure [146], anti-inflammatory immune response [147], and appetite via GPBAR-1 receptors found in brown adipose tissue and muscle [144,148]. BAs are also excreted from the hepatocyte into the portal vein via specific transporters, i.e., the resistance protein 3 and 4 (MRP3, MRP4) and OSTα/β. From the peripheral circulation, BAs also undergo renal uptake by the apical sodium/dependent bile acid transporter (ASBT) in the proximal tubule. MRP 2, 3, 4 transporters regulate the glomerular filtration of BAs [149].
In the intestine, micellization of BAs contribute to the digestion and absorption of cholesterol, triglycerides, and fat-soluble vitamins. BAs also are signaling molecules in modulating epithelial cell proliferation, gene expression, and lipid and glucose metabolism. Thus, the enterohepatic circulation of BAs as signaling molecules produces profound effects on lipid and glucose metabolism.
3.4. Intestinal Monocellular Layer
A fourth level of the gut barrier is the monocellular layer [150], which include enterocytes, Goblet cells (which produce mucus), Tufts cells (with chemosensory function) [151], and Paneth cells (which produce antimicrobial peptides) [152]. This cellular barrier has physical, electrical, and chemical properties. The layer is impermeable to most solutes that need a specific transporter to cross the barrier, a mechanism involving transcellular pathway. Intercellular spaces are closed by the presence of specific apical junctional complex, i.e., a tight junctions (TJs) and the adherens junctions. Over 40 proteins contribute to a TJ (i.e., claudins, peripheral membrane proteins, zonula occludens (ZO) 1 and 2, occluding, E-cadherins, and desmosomes) [153,154,155]. The cytoskeleton connects TJs and adherens junctions, and TJs contribute to active and passive transport through the gut barrier [156]. TJs regulate the passive flow of the solutes and water through the paracellular pathway, and operate as a size- and charge-selective filter [157]. The two different routes involve the leak pathway (transport of larger substances namely proteins and bacterial components) and a second claudin-mediated pathway, which limits the flow to small (<4 Å) molecules. Due to the active transport across cells sealed with TJs, the human body prevents the uncontrolled translocation of substances and allows an active transcellular transport through the enterocytes [158]. Moreover, cytokines, tumor necrosis factor-alfa (TNFα), interferon gamma (IFNγ) signaling kinases, and cytoskeleton (myosin light chain kinases (MLCK)) contribute to regulate TJ [159,160,161,162]. Such mechanisms might be impaired during liver disease [163,164,165]. The negative charge of brush border microvilli (depending on polar carbohydrates and charged transmembrane proteins) opposes the negative charge of bacterial cell wall and inhibits bacterial translocation [166].
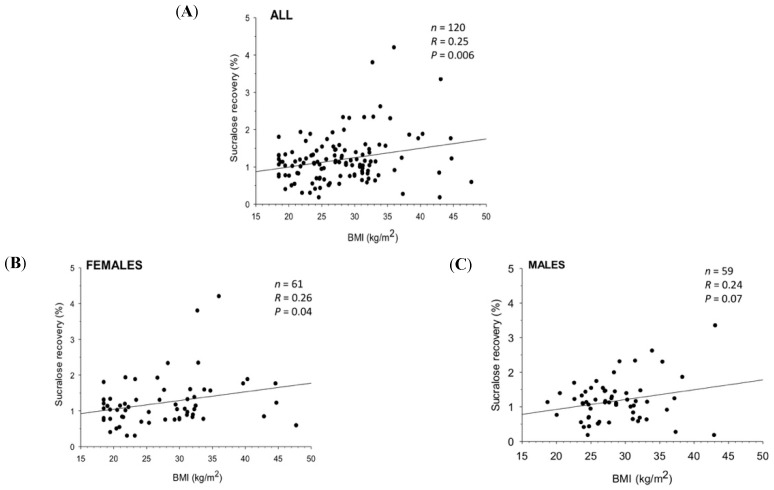
We assessed urine recovery of orally administered sucrose, lactulose/mannitol, and sucralose by using triple quadrupole mass-spectrometry and high-performance liquid chromatography. We found that increased colonic (but not stomach and small intestinal) permeability was linked to obesity and liver steatosis, regardless dietary habits, age, and physical activity [167] (Figure 6).
Figure 6.
Colonic permeability (leakage) in a group of 120 subjects (61 females and 59 males; mean age 45 ± SEM1.2 years, range 18–75). Graphs show the correlation between urinary sucralose recovery (marker of colonic permeability) and body mass index (BMI) in: (A) all subjects; (B) females; (C) males. The prevalence of abnormal colonic permeability was higher in both overweight and obese than in normal weight subjects, and sucralose recovery increased significantly with BMI overall, in females (p = 0.04), and tended to increase also in males (p = 0.07). To measure overall intestinal permeability, four saccharide probes are administered orally and simultaneously. Stomach permeability is assessed by administering 20 g of sucrose; small intestine permeability is assessed by administering lactulose (5 g) and mannitol (1 g); and colonic permeability is assessed with 1 g of sucralose. A urinary sample is collected in the morning of the test, after an overnight fast. Sugars are dissolved in 250 mL of tap water and orally administered. Urines are collected every hour during six hours in a sterile container. Urinary samples are measured by chromatography/mass spectrometry (UPLC-MS/MS, AB Analitica, Padua, Italy). The fraction of the excreted sugars was calculated based on the quantity of sugar ingested by the patients and expressed as a percentage value, according to the manufacturer (AB analitica, Padua, Italy). Their presence in urine, in different amounts compared to the expected, indicated impaired permeability of the stomach, small intestine, or colon. Normal values for permeability test are expressed as a percentage) adapted from Di Palo et al. [167]).
3.5. Immunological Gut Barrier
A fifth level of the gut barrier is the immunological barrier, starting from the secretory ability of the Paneth cells to secrete several antimicrobial peptides (i.e., defensins, cathelicidines, resistin-like molecules, bactericidal-permeability-inducing proteins and lectins, and IgA immunoglobulins) into the gut lumen at the inner face of the mucin layer hosts [93,168]. A further barrier is the population of intraepithelial cells (conventional and unconventional αβ and δχT cells and mononuclear phagocytes [169]) and lamina propria cells.
Intraepithelial lymphocytes express type I cytokines and act as first-line cytolytic defense. These lymphocytes release antimicrobial peptides in response to cytokines released by intestinal epithelial cells or by engaging in the activation of NK cell receptors expressed by intestinal intraepithelial lymphocytes lineages [169]. Mononuclear phagocytes have protrusions that act as direct sensors of the intestinal lumen [170,171] while developing oral tolerance after delivering food antigenic peptides to lamina propria dendritic cells [172].
Immune cells act as second line of defense and contribute to tissue regeneration when the lamina propria is damaged. The cells are highly specialized to recognize microbial antigens or metabolites. Cells consist of CD4+ T cells lymphocytes, innate-like cells (iNKT cells), and mucosa associated invariant T cells.
Lipids presented on CD1 molecules are recognized by NKT cells [173]. Mucosa-associated invariant T cells will interact with raboflavin metabolites presented by MR1 molecules [174,175]. CD4+ T cells include two populations of cells.
Th17 cells release IL17-A, IL17-F, and IL-22 (which contributes to strengthen tight junction molecules and stimulate epithelial cell regeneration [176]); IgA production; and the expression of pIgR, which allows their translocation towards the gut lumen [177,178]. These cells accumulate if segmented filamentous bacteria reaches the intestinal epithelium [178,179,180].
T regulatory cells are thymic derived cells (which recognize self-antigens and control the function of autoreactive T cells) and peripheral derived T regs (which recognize food antigens in the small intestine or microbial antigens in the large intestine), where they control tolerance to innocuous non self-antigens [181].
Innate lymphoid cells, also found in the intestine, rapidly release type 1, 2, and 3 cytokines in a response to infection before the response of adaptive T cells [116,182].
There is a precise equilibrium between the gut and the resident microbiota because pattern-recognition receptors (Toll-like receptors, TLRs, and nucleotide binding oligomerization domain-like receptors) on the intestinal epithelial cells will recognize MAMPs and PAMPs crossing the intestinal barrier. In this scenario, recruited dendritic cells transport the given antigens to the mesenteric lymph nodes (MLNs) for antigen presentation. Following this step, priming and maturation of B and T lymphocytes occurs, as part of the adaptive immune response in the gut associated lymphoid tissue [183,184]. As mentioned earlier, the microbiome takes part in the maturation of several immune cells [116,117].
3.6. Gut Vascular Barrier
A sixth level of the gut barrier is the gut-vascular barrier [185] with similarities between blood-brain barrier and intestinal barrier [185,186]. This barrier should prevent the translocation of bacteria and/or microbial components across the extracellular and the intestinal epithelial barrier. The so-called gut-vascular unit includes the intestinal endothelium associated with pericytes and enteric glial cells, TJs, and adherens junctions (permeable to most of the small nutrients) [185,186,187]. Endothelial plasma membrane are isolated and carry active and passive transporters, while glial cells contribute to gut homeostasis intestinal permeability. The intact endothelium allows the free diffusion of 4 kD dextran, but 70 kD dextran is blocked. The gut-vascular barrier ultimately blocks a further dissemination of bacteria to liver and spleen, blocked by a second barrier [185]. Under certain circumstances (i.e., infection with Salmonella enterica serovar Typhimurium), the gut-vascular barrier becomes disrupted and larger substances can permeate, even in the absence of inflammation and vasodilation. This dissemination occurs through the portal circulation more than the lymphatic vessels. A marker of endothelial permeability (vesicle-associated protein-1 (PV1)) can increase during such events and bacteria can be found systemically [185]. The intestinal barrier is typically disrupted in conditions like celiac disease [185], anchylosing spondylitis [188], and NASH [87].
3.7. The Liver Barrier
In the health condition, the translocation of small amounts of microorganisms and bacterial products is an ongoing process towards mesenteric lymph nodes (MLNs) [168]. Here, the immune system is continuously stimulated, modulated to achieve immune tolerance [189,190], which allows the killing of microbes without significant inflammatory modifications at a systemic level [191,192]. Minimal amounts of bacterial mRNAs and lipopolysaccharide (LPS) [88,193,194] can permeate to the liver and help detoxifying bacterial products [195,196]. Nevertheless, the liver is usually free of bacteria [195], while acting as a second firewall for microorganisms penetrated after mucosal damage and escaped from MLNs surveillance activity [88,195,196]. At the level of the hepatic sinusoids Kupffer cells account for over the 80% of all tissue macrophages and contribute to phagocytize and kill microbes that originate from the bloodstream [88,195,197,198,199]. Liver resident macrophages also participate in the clearance of microorganisms and MAMPs and PAMPs. Kupffer cells are able to process E. coli endotoxin [200]. Kupffer cells depleted mice display delayed clearance of E. coli K-12 during bacteremia [195], while Kupffer cells perform phagocytosis and killing of green fluorescent protein expressing B.burgdorferii and antigen presentation to natural killer (NK) cells [197]. Mononuclear cells, via functional Toll-like receptor 4 (TLR4) [201,202], are activated by the lipopolysaccharide binding. The LPS-LBP complex stimulates liver resident myeloid cells via mCD14 and TLR4 [203,204,205].
4. The Main Constituents of the Ongoing Liver Damage
Several studies in animal models suggest that during liver damage, changes include composition of gut microbiota, quality/quantity of mucus, gastrointestinal motility, epithelial barrier, TJs, and intestinal immunity.
Disruption of the intestinal barrier increases the intestinal permeability, leading to filtration of damaging agents like MAMPs and PAMPs (LPS, microbial DNA, peptidoglycans, and lipopeptides), metabolic products, and whole bacteria to local MLNs, where clearance is defective [206,207,208,209]. The liver is then reached by these agents/bacteria via the mesenteric and portal circulation [88], and detrimental agents perpetuate the local damage in the liver but could also activate a systemic inflammatory response [210,211,212,213,214]. Activated Kupffer cells are essential in this respect [199,212,215,216,217].
4.1. Inflammation
Both PAMPs and TLRs interaction activate intracellular molecular pathways (MyD88-dependent or MyD88-independent) that, in turn, activate NF-κB and inflammatory cytokines including TNF-α, IL-1β, IL-6, IL-12, IL-18, chemokines like CXCL1, CXCL2, CCL2, CCL5, CCL3, CCL4, vasoactive factors like nitric oxide (NO), and production of reactive oxygen species (ROS) [218]. Liver inflammation is further enhanced following the recruitment of systemic leukocytes (CD4+ T cells, neutrophils, and monocytes) [212,215], with induction of apoptosis and necrosis of the hepatocyte [219], activation and proliferation of hepatic stellate cells (HSC), and production of transforming growth factor-β (TGFβ) with activation of fibrosis [217,220]. The following upregulation of the expression of matrix metalloproteinases (MMPs) enhance the destruction of the hepatic tissue [221,222] in tandem with increased expression of tissue inhibitors of matrix metalloproteinases (TIMPs) a step leading to inhibition of degradation of collagen fibrogenesis in the liver [221,222,223,224]. Indeed, TIMP-1 is a predictive marker for NASH [225].
4.2. Oxidative Stress
Oxidative stress contributes to liver damage [226], including liver steatosis [227], since hepatocytes are sensitive to oxidative stress-related molecules [226,228,229]. ROS can deteriorate the ongoing equilibrium of the gut-liver axis and be responsible for intestinal barrier damage. Several conditions can impair the intestinal permeability and act via abnormal redox state in the gut, including diet [230], alcohol [231], infectious [232]/primary inflammatory diseases [233], and drugs [234]. An ongoing hypoperfusion-dependent hypoxia of the intestinal mucosa leads to increased activity of xanthine oxidase, more ROS release and oxidative damage [235]. A vicious circle arises from activation of TLR of Kupffer cells and ROS generation [236], as well as activation and proliferation of hepatic stellate cells (HSC) [237]. In response to ROS, Kupffer cells produce cytokines and chemokines, and stimulate HSCs [229].
Protective mechanisms include the release of macrophagic IL-10 at the intestinal mucosa (modulation of the innate immune activation, decreased tissue damage, amelioration of integrity of the gut barrier, and decreased endotoxin absorption [238,239], and in the liver (reduced inflammations and fibrosis, and decreased activation of Kupffer cells functions [240,241]. NK cells also contribute by killing early activated and senescent HSCs, a step leading to limitation of fibrogenesis [242,243].
4.3. The Intestinal Microbiota
Gut microbiota, a super-organism hosted in the human body, can be involved in the pathogenesis of NAFLD [85]. Changes in microbiota signature might exist in NAFLD patients, but findings are difficult to be comprehensively elucidated in different NAFLD phenotypes. The difficulties in interpreting the results depend on the coexistence of complex interactions between genetic and environmental factors across several metabolic abnormalities, which also involves the liver.
For example, BMI is a determinant of gut microbiota [244]. Because NAFLD is often associated with overweight and obesity, the involvement of gut microbiota in the pathogenesis of NAFLD is a matter of active debate. Indeed, the liver has a complex vascular system. The majority of the blood flow reaches the liver from the intestine through the portal vein. Changes of intestinal immune system may affect intestinal permeability and bacterial translocation. This step might be responsible for a cascade of disorders including obesity, metabolic, and liver diseases [245,246]. Specific profiles of the gut microbiome might influence the inflammatory and fibrosis responses in NAFLD patients [247]. The combination of dysbiosis and increased intestinal permeability allows gut microorganisms to promote liver damage by release of MAMPs and PAMPs (i.e., LPS) or by products of their metabolism (i.e., ethanol, SCFAs, and trimethylamine) that enters the portal vein, which contributes with about 70% of the blood entering the liver [248,249,250]. Changes in the quality and/or quantity and/or topographic distribution of the microbiota might contribute to promote such abnormalities. This step promotes the activation of both Kupffer cells and hepatic stellate cells, for example following the influx of the LPS which acts on cell Toll-like receptor 4. Few animal studies point to this important pathway linking intestinal lumen, barrier, and liver damage. If the LPS is injected to rats, a picture of steatohepatitis develops. By contrast, anti-tumor necrosis factor antibodies improve the steatosis [251,252]. In addition, genetically obese mice develop increased intestinal permeability, a condition promoting increased portal endotoxemia [193,253]. The study of Imajo et al. [254] suggests that obesity-induced leptin plays a crucial role in NASH progression via enhanced responsivity to endotoxin (and therefore bacteria-derived products). Upregulation of CD14 in Kupffer cells and hyperreactivity against low-dose LPS occurred in high-fat diet (HFD)-induced steatosis mice, but not chow-fed-control mice. Accelerated NASH progression (with liver inflammation and fibrosis) occurred in this condition. In chow-fed mice Leptin increased hepatic expression of CD14 via STAT3 signaling and hyperreactivity against low-dose LPS without steatosis, while leptin-deficient ob/ob mice with severe steatosis had a marked decrease in hepatic CD14. In different mouse models, Henao-Meja et al. [255] investigated the role of innate immunity inflammasomes NLRP6 and NLRP3 and the protein IL-18 on NAFLD/NASH progression, with respect to the gut microbiome. Changes associated with inflammasome-deficiency influenced the configuration of the gut microbioma and were associated with worsening of hepatic steatosis and inflammation. Findings parallel the influx of TLR4 and TLR9 agonists into the portal circulation. This influx, in turn leads to enhanced hepatic tumor-necrosis factor (TNF)-alpha expression and NASH progression. Within a scenario of defective NLRP3 and NLRP6 inflammasome sensing, the microbiome appears to play a role connecting systemic auto-inflammatory and metabolic disorders. There is an association between NAFLD, SIBO, and increased endotoxemia [165,256,257,258]. The ongoing dysbiosis might encompass a condition named SIBO [259,260]. The occurrence of SIBO was higher in NAFLD [165]/NASH [257] patients than healthy controls.
NAFLD patients with advanced fibrosis show increased abundance of Escherichia coli and Bacteriodes vulgatus [261]. Obese children with NASH had greater abundance of the genus Escherichia [262]. Changes of intestinal microbiota in NAFLD may also occur because of dietary factors. A Western diet, enriched in fat, proteins of animal origin and simple sugars promotes Bacteroides abundance. By contrast, a diet rich in fiber and indigestible plant polysaccharides increases Prevotella abundance. Notably, Bacteroides genus correlates with NASH while Prevotella abundance decreased in NASH [247,263]. Ruminococcus genus abundance increases with significant liver fibrosis (score ≥F2) in humans [247]. In animal models this genus correlates with the development of metabolic impairment [264].
In obesity the ratio Firmicutes:Bacteroidetes increases, compared to lean subjects on the same diet [255]. Shift of microbiome in blood is reported in patients with liver fibrosis [265], as well as in metabolic diseases [266,267]
Yun et al. [26] had the idea of investigating the association between fecal and blood microbiome profiles, and the presence of NAFLD in obese versus lean Korean subjects. They sequenced the V3-V4 domains of the 16S rRNA genes. The 176 NAFLD subjects had distinct bacterial community with a lower biodiversity and a far distant phylotype, compared with 192 controls. In the gut, the microbiome showed decreased Desulfovibrionaceae in lean NAFLD but not in obese NAFLD. In the blood, Succinivibrionaceae showed opposite correlations in the lean vs. obese NAFLD. In addition, both gut and blood Leuconostocaceae was associated with the obese NAFLD. This study suggests that fecal and blood microbiome profiles showed different patterns between subjects with obese and lean NAFLD. Such profiles might therefore be potential biomarkers to discriminate diverse NAFLD phenotypes. In NAFLD, Raman et al. described decreased members of Firmicutes [268]. A study looked at patients with biopsy-proven NAFLD (n = 57) and reported a twofold increase in NASH in patients with Bacteroides genus counts in the 2nd and 3rd tertile, as compared with patients with lower Bacteroides counts. This latter group of patients had an abundance of Prevotella bacteria. Moreover, fibrosis could be affected by changes in microbiota, since patients with Ruminococcus counts in the third tertile had a twofold increase in stage 2 or greater fibrosis, as compared with patients with lower levels of Ruminococcus [261,269]. In another study, NAFLD patients had a lower percentage of species Bacteroidetes and higher levels of Porphyromonas and Prevotella than healthy subjects. Notably, the predisposition to NAFLD related to expression of Toll-like receptors (TLR) 4 or 9, or the receptor of tumor necrosis factor-alpha [262].
As mentioned earlier, the intestinal microbiota is essential during the process of enterohepatic circulation of primary BAs. At the level of the colon, the bile acids undergo complex biotransformation by the resident microbiota and are transformed into secondary BAs [12,89,270]. Under specific circumstances, the dysbiosis may contribute to hepatocellular injury because of increased deconjugation of BAs (i.e., production of more cytotoxic secondary bile acids) and inactivation of hepatic lipotropes (i.e., choline). Indeed, a choline-deficient diet is one way to develop NAFLD in the rat model [227,271,272,273,274].
At the extreme of the NAFLD spectrum is the so-called “cryptogenic” cirrhosis. Of note, low gut microbiota diversity exists in NAFLD-related liver cirrhosis compared to healthy controls. At the genus level, Bacteroides, Ruminococcus, Klebsiella, Prevotella, Enterococcus, Hemophilus, Lactobacillus, Streptococcus, Pseudomonas, Phascolarctobacterium, Veillonella, Atopobium, Parabacteroides, Dialister, and Christensenella were more abundant, while Methanobrevibacter and Akkermansia decreased [275]. Changes, however, might be influenced by the advanced status of disease, portal hypertension, drugs, etc.
4.4. Altered Intestinal Permeability
Changes to the aforementioned conditions might originate following impaired intestinal permeability due to disrupted of tight junctions, dysbiosis, and/or small intestinal bacterial overgrowth (SIBO). These conditions are associated with the spectrum of liver disease, i.e., alcoholic and non-alcoholic fatty liver [165,276]. Children with NAFLD showed a positive correlation between altered intestinal permeability and liver inflammation/fibrosis [256]. A metanalysis shows that patients with NAFLD more frequently exhibit altered intestinal permeability [277].
Small intestinal bacterial overgrowth (SIBO) can develop in NAFLD patients [165,278,279,280,281] along with dysbiosis [247]. Abnormal intestinal permeability might therefore be involved in this scenario [45] and act as a factor leading to liver inflammation and fibrosis. Several animal and human studies evidences point to a role for intestinal permeability in the pathogenesis of NAFLD.
In the steatotic mice endotoxin triggers liver inflammation [282]. Obese mice (C57BL/6Job/ob genetically leptin deficient and C57BL/6Jdb/db functionally deficient for the long-form leptin receptor) show increased epithelial permeability to horseradish peroxidase. TJ proteins (ZO-1 and Occludin) distribution was abnormal in both types of obese mice that also exhibited increased circulating levels of endotoxin in portal circulation and levels of circulating proinflammatory cytokines (IL-1, IL-6, INF-γ, and TNF-α) than controls. HSC became activated and displayed enhanced sensitivity to LPS and released higher levels of cytokines [193]. F11r−/− mice fed a steatogenic diet (high saturated fat, fructose and cholesterol for 8 weeks) developed a form of severe steatohepatitis with liver inflammation (hepatocyte ballooning and inflammatory cells infiltration), fibrogenesis, and increased in serum transaminases, as compared with control animals [250]. Notably, the murine gene F11r encodes the junctional adhesion molecule A (JAM-A), which is a constituent of the TJs and which modulates the epithelial barrier function, regulating IP and inflammation [283,284,285,286].
When inducing gut inflammation and gut-vascular barrier dysfunction with dextran sulfate sodium (DSS) in C57BL/6 mice fed a high-fat diet (HFD) for 12 weeks, liver fat vacuoles and leukocyte infiltration were more represented in DSS and HFD-fed mice compared to HFD-fed mice. Several other alterations included increased levels of hepatic mRNA coding for inflammatory cytokines (IL-1, IL-6, TNF-α, MCP-1), higher expression of collagen I and profibrogenic factors mRNA (TGF-β, Actin α2, tissue inhibitor of metalloproteinase-1, and plasminogen activator inhibitor-1). Additional modifications include upregulation of TLR4 and TLR 9, downregulation of ZO-1 and Claudin-1 and increased expression of PV1 [287]. Human studies are also instructive in this field and point to a role for increased intestinal permeability, disrupted TJs, and SIBO in NAFLD patients. A previous trial compared 22 patients with biopsy-proven NASH and 23 controls subjects. The authors assessed overgrowth by combined (14)C-D-xylose and lactulose breath test, intestinal permeability by a dual lactulose-rhamnose sugar test, serum endotoxin levels by limulus amoebocyte lysate assay and TNF-alpha levels by ELISA. Results showed that 50% of patients and 22% of controls (p = 0.048) had SIBO. Whereas intestinal permeability and serum endotoxin levels were similar in the two groups, endotoxin assay confirmed significantly higher TNF-α levels in patients than controls (14.2 and 7.5 pg/mL, respectively, p = 0.001) [257].
Another study compared patients with biopsy-proven NAFLD (n = 35) patients with untreated celiac disease (n = 27, as a model of intestinal hyper-permeability), and healthy subjects (n = 24). The authors used glucose hydrogen breath testing to diagnose SIBO and urinary excretion of (51)Cr-ethylene diamine tetraacetate ((51)Cr-EDTA) test to diagnose intestinal permeability. Orally administered (51)Cr-EDTA is not metabolized and is poorly absorbed (1–3%) from the gastrointestinal tract. In the presence of TJs disruption and (51)Cr-EDTA crosses the intestinal barrier through the paracellular pathway [123,288,289]. The authors also used immunohistochemical analysis of zona occludens-1 (ZO-1) expression in duodenal biopsy specimens to diagnose the integrity of TJs within the gut. NAFLD patients had significantly increased intestinal permeability and SIBO (3x) compared to the control [165]. (51)Cr-EDTA excretion levels and SIBO prevalence increased with the degree of liver steatosis. In addition, by duodenal histology NAFLD patients had reduced ZO-1 expression. Increased intestinal permeability and SIBO were independent with the severity of liver inflammation, fibrosis, and NASH.
NALFD children had increased ratio between urinary excretion of orally-administered lactulose and mannitol (L/M ratio) as a marker of intestinal permeability [256,257,290]. L/M ratio further increased in NASH patients. The increased LPS pointed to bacterial translocation while the extent of hepatic inflammation and fibrosis was proportional to the degree of intestinal permeability [256].
4.5. Products of Microbiota
Microbe-associated molecular patterns: the intestinal microbiome is an invaluable source of gases, metabolites, and bacterial products, namely MAMPs and PAMPs. Some of such metabolites deriving from the interaction of the microbiota with endogenous and exogenous substances might act as effectors of damage in the liver after crossing the portal circulation. Upon uptake in the liver, MAMPs activate local inflammatory changes mediated by patter-recognition receptors (PRRs) located on the hepatic stellate cells [291] and Kupffer cells [217]. In turn, endotoxins will activate TLR4, TLR9 (by methylated DNA) and TLR2 (by Gram-positive bacteria) [162], which represent the first step of immune response in liver disease. Further steps include the activation of further inflammatory events via activation of nuclear factor- χB (NF-χB) by the myeloid differentiation primary response protein (MYD88) enhanced hepatic tumor-necrosis factor (TNF)-alpha expression that drives NASH progression [255]. In hepatic stellate cells, fibrosis is promoted via TLR4 signaling that downregulates BMP and activin membrane-bound inhibitor homologue (BAMBI, a decoy receptor for transforming growth factor-β (TGFβ)) [218]. Further steps include the release of inflammatory cytokines, oxidative stress, and endoplasmic reticulum stress, all factors initiating and perpetuating liver damage [292]. Evidences in animals show that diets enriched in fat or deficient in choline drive steatogenic, inflammatory, and fibrogenic hits requiring TLR-4 or 9 [293,294,295]. In addition, inflammasome-deficiency-associated changes in the configuration of the gut microbiota parallel exacerbated hepatic steatosis and inflammation.
Alcohol: Dietary ethanol crosses the gastrointestinal mucosa by simple diffusion in the stomach and small intestine (about 20% and 70%, respectively) [296]. The liver is involved in ethanol metabolism pathway with enzymes that transform ethanol to acetaldehyde (enzyme alcohol dehydrogenase) and to acetate (enzyme acetaldehyde dehydrogenase) [297,298]. Most of the alcohol in the intestine is from the systemic circulation, while a smaller part originates as a product of luminal microbial fermentation, as both bacteria and enterocytes are equipped with alcohol-metabolizing enzymes [298,299]. A study showed an increase in hepatic expression of ethanol-metabolizing genes in germ-free mice and exacerbation in hepatic steatosis [300]. Acetaldehyde impairs the intestinal TJs and this, in turn, compromises the gut barrier enabling translocation of microbial products [301,302,303]. Additional effects include downregulation of the expression of intestinal AMPs [304,305] and activation of a host inflammatory immune responses [306,307,308]. Together with reduced intestinal levels of butyrate [309,310,311], changes increase intestinal permeability [312,313,314,315]. Humans display high concentrations of endogenous alcohol production [316]. Similar findings occur in animals with intestinal blind-loops [317]. Obese females with Candida albicans overgrowth have increased breath alcohol levels after a carbohydrate load [318,319]. Notably, colonic bacteria and yeast are able to produce both ethanol and acetaldehyde [320]. Even if ethanol concentrations remain low, the microbiota can oxidize ethanol to high concentrations of acetaldehyde, which is promptly absorbed into the portal blood stream. These pathways initiate histological changes similar to those occurring in NAFLD [321]. Proteobacteria, particularly Enterobacteriaceae, ferment carbohydrates to ethanol [262], and the amount produced can be remarkable [322]. A correlation exists between abundance of Proteobacteria, Enterobacteriaceae, Escherichia, liver inflammation (NASH), and serum levels of alcohol in children [262]. In another complementary study, fasting ethanol levels were positively associated with insulin resistance and were significantly higher in children with NAFLD than in controls. The authors suggest that increased blood ethanol levels in patients with NAFLD may result from insulin-dependent impairments of ADH activity in liver tissue rather than from an increased endogenous ethanol synthesis [323]. Ruminococcus ferments complex carbohydrates with production of ethanol and this property may be responsible for further liver damage [324]. Gut-derived endogenous alcohol might therefore participate in mechanism of liver damage. Increased luminal and circulating levels of ethanol, acetaldehyde, and acetate occur in non-alcoholic and alcoholic liver disease [165,325], and metabolites are independently associated with liver damage [326,327,328]. Increased ethanol production activates hepatic ethanol metabolic pathways and oxidative stress [329], contributing to the pathogenesis of NASH [262].
Bile acids: As inferred from the previous description, the gut microbiome can shape the final profile of BAs. Intestinal bacteria are responsible for the transformation of the primary BAs to the secondary BAs by deconjugation, oxidation of hydroxyl groups in 3, 7, and 12 positions, and 7-dehydroxylation [330]. A bidirectional cross-talk exists between BAs and the intestinal microbiome [331]. BAs (mainly the secondary DCA) display antimicrobial properties and can modulate the species of intestinal microbiome. The detergent effects of DCA acts on bacterial cell membranes and influences the integrity of the bacteria while modulating microbial populations [332]. In addition, BAs binding to FXR promote the production of peptides with antimicrobial effect (AMPs) (e.g., angiogenin 1, RNAase family member 4 [333,334]), which prevent gut barrier disruption and dysfunction. This process has an influence on the hydrophobicity of the BA pool and ongoing dysbiosis initiates a series of events due to unbalance between primary/secondary BAs ratio. Ultimately, changes in hepatic bile synthesis and metabolic abnormalities can arise, including FXR-dependent effect on intestinal barrier and inflammation [335], metabolic pathways [336], and carcinogenic effects [337]. Notably, FXR-deficient mice do not become responsive to diet-induced obesity [336], likely due to mechanisms related to both microbiome [334] and intestinal FXR [338]. Evidences support this complex balance between BAs and microbiota. The obstruction of bile flow is a predisposing factor for bacterial overgrowth and translocations of bacteria within the intestine. Oral administration of BAs in the mouse model can reverse this condition. The FXR-induced gene expression might modulate this pathway associated with the enteral protection and the inhibition of bacteria damage to the intestinal mucosa [333].
The abnormal intestinal microbiota is parallel to the altered bile acid homeostasis, which contributes to the metabolic dysregulation seen in NAFLD. This step can lead to impaired BA signaling in the intestine and the liver, as seen in mice on a high-fiber diet and in NAFLD patients. The increased synthesis of BAs become evident, as increased serum concentrations of primary and secondary BAs [339,340], increased hepatic bile acid synthesis, and total fecal bile acids and primary to secondary bile acid ratio [341]. Studies suggest that the microbiota, while governing the production of the secondary BAs, also influences FXR-mediated signaling in the intestine and the liver. Abundance of bacteria producing the secondary DCA might lead to a DCA-dependent suppression of FXR- and FGFR4-mediated signaling [11,339]. Notably, intestinal FXR expression decreases in mice on a high-fiber diet. Obeticholic acid restores the integrity of the gut vascular barrier and reduces the portal influx of PAMP to the liver [87].
Fatty acids: in health, the colon microbiota produces short chain fatty acids (SCFAs) acetic, propionic, and butyric acid as the main SCFAs following fermentation of indigestible carbohydrates, i.e., dietary fiber [342]. SCFAs provide the energetic substrate for colonocytes, contribute to the maintenance of the gut barrier integrity, while controlling intestinal inflammation and satiety [343,344]. SCFAs are absorbed in the intestine through the portal circulation and, in the liver, provide the necessary energy source used in gluconeogenesis and lipogenesis [248,345,346]. Moreover, at the intestinal enteroendocrine L cells, SCFAs interact with G protein coupled receptors GPR41 and GPR43 and stimulate the release of the peptide YY (PYY). This hormone slows gastric emptying and intestinal transit and helps energy absorption [347]. In addition, the release of glucagon-like peptide-1 enhances glucose-dependent insulin release [348]. Patients with type 2 diabetes mellitus exhibit reduced abundance of butyrate-producing bacteria [349], while SCFAs supplementation in patients with type 2 diabetes mellitus increased the abundance of butyrate-producing bacteria. This effects was associated with increased GLP-1 and hemoglobin A1c levels [350]. Increased levels of acetate might represent a marker of increased production of endogenous ethanol in the intestinal lumen and liver metabolism (see below). Decreased levels of butyrate might be a marker of tight junctions abnormality and increased intestinal permeability [309,312]. SCFAs appear protective in the gut. Mice fed with alcohol-enriched diet short-term and given tributyrin (the glycerol ester providing butyrate) improved the intestinal permeability and liver injury induced by ethanol [312].
Fasting-induced adipocyte factor (FIAF): The intestinal microbiota also inhibits the production and secretion of FIAF by the intestinal L cells and enterocytes. FIAF inhibits lipoprotein lipase (LPL), activates LPS, and increases triglyceride accumulation in the liver and the adipocytes [351]. Increased hepatic lipid storage activates the carbohydrate responsive element-binding protein and the sterol regulatory element-binding protein-1, perpetuating fat accumulation [352].
Choline: The macronutrient choline is processed into phosphatidylcholine (lecithin) and plays a role in the assembly and excretion of very-low density lipoprotein (VLDL) from the liver. This step prevents the formation of liver steatosis due to triglycerides [353]. Choline is also involved in the development of brain, nerve function, muscle function, and metabolic pathways [354]. Choline deficiency is associated with decreased production and release of VLDL, and therefore triglyceride accumulation in the liver [355]. In the murine model, a choline-deficient diet leads to liver steatosis and oxidative stress [227,271,273,274,356]. Intestinal bacteria can use choline for the synthesis of trimethylamine (TMA). Bacteria of the taxa Erysipelotrichia metabolize choline to TMA and this step decreases the bioavailability of choline and increases the portal influx of TMA, its conversion to trimethylamine N-oxide (TMAO), with steatogenic effects [357,358,359,360]. Suppression of intestinal microbiota in atherosclerosis-prone mice inhibited dietary-choline-enhanced atherosclerosis [355]. In addition, NAFLD patients have increased intestinal metabolism of choline, choline deficiency, and abundance of Erysipelotrichia taxa [358].
Other products: 3-(4-hydroxyphenyl) lactate correlated with specific bacterial species (Firmicutes, Bacteroidetes, and Proteobacteria), as well as hepatic fibrosis [269]. Microbiome-derived products of branched-chain and aromatic amino acid metabolism can also play a role in NAFLD. Phenylacetic acid and 3-(4-hydroxyphenyl) lactate, both related to insulin resistance. Obese, non-diabetic patients with hepatic steatosis and inflammation had low microbial gene richness, and increased microbial genetic potential for processing dietary lipids and endotoxin biosynthesis from Proteobacteria. In addition, both aromatic and branched-chain amino acid metabolism was dysregulated [361].
5. The Microbiota as a Target of Environmental Factors Involved in NAFLD
The microbiome is the first interface between the environment and almost all the metabolic, biochemical, endocrine, and signaling pathways influencing the onset and progression of NAFLD.
The gut microbiota is strongly influenced by dietary habits [362], smoking [363], ethanol intake [364], drugs (i.e., antibiotics [255,349,365,366,367], liraglutide [368], metformin [369], curcumin [370], and environmental pollutants (i.e., heavy metals, persistent organic pollutants, volatile organic compounds, pesticides [62,371,372,373,374]).
These external agents act on the microbiota affecting the ability to maintain the integrity of the mucosal barrier [106], the healthy function of epithelial intestinal cells, the efficiency of the immune system [375], and the production of local peptides and immunoglobulins with antimicrobial function [96,376,377]. Thus, abnormalities in the gut–liver axis with the microbiome as the key player leads to abnormal regulation of intestinal and metabolic homeostasis, influencing the pathogenesis of NAFLD [85]. Conversely, protective effects and a possible reversal of liver fat accumulation and lipotoxicity could be driven by favorable environmental factors modulating composition and relative abundance of specific phylotypes.
5.1. Dietary Habits and Microbiota
Dietary habits can influence the microbiota, the intestinal permeability, the vascular barrier, the enhanced influx of endotoxins in the portal vein, low-grade liver inflammation, and ultimately NAFLD/NASH, insulin resistance, and metabolic disorders [378,379]. Changes are likely due to the initial modification of the microbiota, rather than the dietary pattern per se [87]. This topic, however, is still under discussion. A Western-style diet is rich in saturated fat and sugars, and might promote injury. In mice, a high-fat diet or fiber-deprived diets change the composition of intestinal microbiota and can damage the intestinal barrier through increased intestinal permeability, reduced thickness of the mucous layer, abnormalities of tight junction proteins of the epithelial barrier and low-grade intestinal inflammation [114,380,381].
In the NAFLD animal models following high-fat diet or high-fructose intake gut permeability increased [246,382].
Both saturated fat and fructose promote the pro-inflammatory microbiome and decreases SCFAs production, which is essential for intestinal barrier function. Increased recruitment of macrophages with cytokines. Thus, TNF-α can be associated with intestinal mucosal inflammation [383,384]. Decreased expression of TJ proteins and a higher permeability of the gut barrier is another potential step [385]. In the animal model, a high-fat diet or a high-fructose diet induces endotoxemia by altering intestinal ZO-1 and occludin [193,386,387,388]. If LPS levels increase in serum due to diet, a further step can be the metabolic endotoxemia with activation of TLR-mediated low-grade liver inflammation. This is a condition potentially associated with NAFLD and NASH [379]. Of note, dysbiosis will generate an altered microbiota able to cross the intestinal epithelium at the disrupted site and to alter the TJs proteins [87]. The resulting inflammatory events lead to reduced lamina propria Treg cells, increased production of IFN-γ (by Th1 and CD8+ T cells), and increased production of IL-17 (by γδ-T cells) [381]. In an integrated study involving animals and humans, the authors reported that mice with defects in intestinal permeability develop more severe steatohepatitis after a high fructose, cholesterol diet than control mice, and colon tissues from patients with NAFLD have lower levels of JAM-A (junctional adhesion molecule), and higher levels of inflammation than subjects without NAFLD [250]. In accordance with these findings, another study found that mice genetically deficient in Jam1 on a high-fat and -fructose diet had increased intestinal permeability, endotoxemia, and hepatic inflammation [389].
In NAFLD adolescents, fructose- but not glucose-enriched drinks together with meals during 24-h increased postprandial endotoxin levels [390].
A less pro-inflammatory diet could modulate also the intestinal permeability in NAFLD patients. The Mediterranean diet is enriched in fibers, mono- and polyunsaturated fatty acids, antioxidants, polyphenols, and phytochemicals. Following this diet, SCFAs-producing bacteria might increase in the intestine due to the diet-induced prebiotic effects [391]. A small group of patients with NAFLD and increased baseline intestinal permeability ((51)Cr-EDTA) underwent a trial consisting of 16 weeks of a Mediterranean diet and 16 weeks of a low-fat diet. With both diets, intestinal permeability did not improve [392]. More studies are clearly required in this field.
5.2. Antibiotics, Probiotics, and Other Pharmacological Agents
Under certain circumstances due to disease or use of antibiotics, the function of the microbiome can be disrupted and lead to energy storage and metabolic disorders, such as obesity, diabetes, and metabolic syndrome [255,349,365,366,367]. Treatment with antibiotics leads to qualitative/quantitative modifications of the intestinal microbiome, and therefore their potential role for liver damage. For example, administration of polymyxin B was associated with improved steatosis grades in both rats and humans on total parenteral nutrition. A similar protection was documented in alcohol-exposed rats [393,394,395]. Following the intestinal bypass surgery and the associated hepatic steatosis, metronidazole administration improved the hepatic picture [396]. In children the administration of some probiotic species caused increased levels of glucagon-like peptide-1 (GLP-1) and improvement in fatty liver [397]. Administration of probiotics during four weeks to NAFLD mice improved the steatosis, hepatomegaly, and nuclear factor kappa-beta activity [398].
In an animal model (ob/ob mice fed a high fat diet), liraglutide, a drug useful in the treatment of insulin-resistance, decreased the fat content in the liver, reversed steatosis, and modified the overall composition, as well as the relative abundance of gut microbiota phylotypes involved in the pathogenesis of NAFLD (i.e., reduced Proteobacteria and increased Akkermansia muciniphila) [368].
In mice, curcumin was also able to alter the composition of a number of operational taxonomic units previously correlated with hepatic steatosis, finally resulting in an attenuation of liver fat deposition and improving the integrity of the intestinal barrier [370].
In a different animal model (C57Bl/6J mice fed fat-, fructose-, and cholesterol-rich diet), protective effects were also noticed following metformin administration in terms of changed composition of gut microbiota and integrity of the intestinal barrier [369].
5.3. Food Contamination with Chemicals of Environmental Origin, Microbiota, and NAFLD
Several studies underscore the relationships between chemicals (in particular endocrine disrupting chemicals, persistent organic pollutants, heavy metals) introduced with contaminated food or through contaminated consumer products, and alterations in gut microbiota, metabolic pathways, and hepatic fat accumulation.
In humans, urine levels of Bisphenol-A (BPA), a widely diffused plasticizer with well-known endocrine disrupting effects, have been linked with NAFLD [77], independently from the presence of type 2 diabetes [399]. In animals, dietary exposure to BPA increases lipid content and fat accumulation in the liver, and simultaneously affects the gut microbiota (i.e., increased abundance of Proteonacteria, decreased abundance of Akkermansia). Exposed animals also show increased intestinal permeability, upregulated expression of Toll-like receptor 4, increased phosphorylation of nuclear factor-kappa B in the liver and increased liver inflammation [400]. The negative effect of endocrine disruptors is also possible during early life exposure. Male offspring from mice exposed to low doses of BDE-47, a polybrominated diphenyl ether (PBDE), during pregnancy and until postnatal day 21 (through lactation) showed a worsening of high fat diet-induced obesity and liver steatosis, metabolic disturbances and alterations of gut microbiota, shifting the microbial community from Bacteroidetes (decrease) towards Firmicutes (increase) [401].
Evidences from animal and epidemiologic studies link NAFLD with persistent organic pollutants (POPs) [402]. A cross-sectional cohort study in adults without viral hepatitis, hemochromatosis, or alcoholic liver disease (NHANES), correlated ALT elevation, a proxy marker of NAFLD, with blood levels of polychlorinated biphenyls (PCBs), lead, and mercury [403]. In adult female C57BL/6 mice, the oral exposure during 6 weeks to low-dose of the polychlorinated biphenyl 126 (PCB126), a typical persistent organic pollutant, affected gut microbiota (decreased phylum levels of Firmicutes, increased Bacteroidetes), and promoted dyslipidemia and NAFLD. Notably, several specific bacterial taxa positively and significantly related to metabolic alterations [404]. Another study reported a significant alteration of the Firmicutes to Bacteroidetes ratio following PCB126 exposure and found an upregulation of Cyp1a1 gene expression (a marker of aryl hydrocarbon receptor activation), as well as increased markers of systemic and intestinal inflammation. Findings depict the intestine as a target of PCB126 toxicity [405].
In mice, the enrichment of a standard chow diet with a mixture of six commonly used pesticides (i.e., boscalid, captan, chlorpyrifos, thiofanate, thiacloprid, and ziram) at doses corresponding to the tolerable daily intake (TDI) of each pesticide promoted for 52 weeks had significant metabolic alterations. Rodents increased body weight and adiposity, developed glucose intolerance and liver steatosis, and had perturbations of gut microbiota in terms of altered urinary concentration of microbiota–related metabolites [406]. In humans, a large survey from the NHANES III study (12,732 adults) linked urinary cadmium (Cd) concentrations secondary to environmental exposure with hepatic necroinflammation, NAFLD, NASH in men, and hepatic necroinflammation in women [407]. In mice, sub-chronic (10 weeks) exposure to low doses of Cd in drinking water increased hepatic triacylglycerol, serum free fatty acid, and triglyceride levels through altered gene expression. Changes in energy metabolism were paralleled by changes (both structure and abundance) in gut microbiome, with decreased Firmicutes and γ-proteobacteria, increased serum levels of LPS and liver inflammation [408]. Another study in mice showed that low dose Cd exposure in early life promotes an increased expression of hepatic genes involved in fatty acid and lipid metabolic processes, leading to life-long metabolic consequences. In male mice, these metabolic alterations developed together with altered microbiota composition after 8 weeks (increased Bacteroidetes, reduced Firmicutes) [409].
6. Conclusions
The molecular, anatomical, and functional link between gut functions, intestinal microbiota, and liver constitutes the gut–liver axis. This axis is exposed to environmental factors, with the microbiome being the main interface. Complex interactions involve bacteria and intestinal barrier, which comes in close contact with dietary factors, food contaminants, drugs, chemicals, and bile. These elements can modulate the pathways linking the gut with the liver, leading to both local and systemic effects. The interplay is bidirectional, since BAs produced in the liver regulate microbiome composition and intestinal barrier. By contrast, intestinal products regulate BA synthesis, hepatic glucose, and lipid metabolism. Local, short-, and long-term changes of this fine interplay due to external or intrinsic factors (e.g., altered intestinal microbiome, increased gut permeability, changes in luminal levels of BAs) at the gut–liver axis disrupts the ongoing homeostasis, paving the way to local and systemic inflammation, and to several liver diseases, including NAFLD.
Changes in the liver might also affect the innate immune cells with exposure to gut derived bacterial products (endotoxin and metabolites like ethanol and trimethylamine). This step is invariably associated with liver inflammation.
According to the available evidence, the majority of environmental factors leading to abnormalities in the gut–liver axis and, in turn, to liver and metabolic dysfunction, could be managed. Future studies should therefore target the modulation of gut-liver homeostasis with both preventive and therapeutic goals.
Acknowledgments
We thank Emilio Molina Molina and Harshita Shanmugam for scientific discussion. We acknowledge the technical expertise of Paola De Benedictis, Rosa De Venuto, and Marilena D’Ambrosio.
Author Contributions
Conceptualization, A.D.C. and P.P.; literature review, A.D.C., J.B., G.G., H.H.W., and L.B.; writing—original draft preparation, A.D.C., J.B., G.G., and L.B.; writing—review and editing, D.M.D.P., G.C., and M.D.A.; supervision, D.Q.-H.W., and P.P.; funding acquisition, M.D.A. and P.P. All authors have read and agreed to the published version of the manuscript.
Funding
The present chapter is written in the context of the project FOIE GRAS, which has received funding from the European Union’s Horizon 2020 Research and Innovation program under the Marie Sklodowska-Curie Grant Agreement No. 722619.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Vernon G., Baranova A., Younossi Z.M. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment. Pharmacol. Ther. 2011;34:274–285. doi: 10.1111/j.1365-2036.2011.04724.x. [DOI] [PubMed] [Google Scholar]
- 2.European Association for the Study of the Liver. European Association for the Study of Diabetes. European Association for the Study of Obesity EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016;64:1388–1402. doi: 10.1016/j.jhep.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 3.Younossi Z.M., Blissett D., Blissett R., Henry L., Stepanova M., Younossi Y., Racila A., Hunt S., Beckerman R. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology. 2016;64:1577–1586. doi: 10.1002/hep.28785. [DOI] [PubMed] [Google Scholar]
- 4.Williams C.D., Stengel J., Asike M.I., Torres D.M., Shaw J., Contreras M., Landt C.L., Harrison S.A. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: A prospective study. Gastroenterology. 2011;140:124–131. doi: 10.1053/j.gastro.2010.09.038. [DOI] [PubMed] [Google Scholar]
- 5.Younossi Z.M., Koenig A.B., Abdelatif D., Fazel Y., Henry L., Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73–84. doi: 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 6.Younossi Z., Anstee Q.M., Marietti M., Hardy T., Henry L., Eslam M., George J., Bugianesi E. Global burden of NAFLD and NASH: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2018;15:11–20. doi: 10.1038/nrgastro.2017.109. [DOI] [PubMed] [Google Scholar]
- 7.Angulo P. Obesity and Nonalcoholic Fatty Liver Disease. Nutr. Rev. 2007;65:S57–S63. doi: 10.1301/nr.2007.jun.S57-S63. [DOI] [PubMed] [Google Scholar]
- 8.Liu C.J. Prevalence and risk factors for non-alcoholic fatty liver disease in Asian people who are not obese. J. Gastroenterol. Hepatol. 2012;27:1555–1560. doi: 10.1111/j.1440-1746.2012.07222.x. [DOI] [PubMed] [Google Scholar]
- 9.Lindenmeyer C.C., McCullough A.J. The natural history of nonalcoholic fatty liver disease—An evolving view. Clin. Liver Dis. 2018;22:11–21. doi: 10.1016/j.cld.2017.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rinella M.E., Sanyal A.J. Management of NAFLD: A stage-based approach. Nat. Rev. Gastroenterol. Hepatol. 2016;13:196–205. doi: 10.1038/nrgastro.2016.3. [DOI] [PubMed] [Google Scholar]
- 11.Tripathi A., Debelius J., Brenner D.A., Karin M., Loomba R., Schnabl B., Knight R. The gut-liver axis and the intersection with the microbiome. Nat. Rev. Gastroenterol. Hepatol. 2018;15:397–411. doi: 10.1038/s41575-018-0011-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Di Ciaula A., Garruti G., Lunardi Baccetto R., Molina-Molina E., Bonfrate L., Wang D.Q., Portincasa P. Bile acid physiology. Ann. Hepatol. 2017;16:s4–s14. doi: 10.5604/01.3001.0010.5493. [DOI] [PubMed] [Google Scholar]
- 13.Park C.C., Nguyen P., Hernandez C., Bettencourt R., Ramirez K., Fortney L., Hooker J., Sy E., Savides M.T., Alquiraish M.H., et al. Magnetic resonance elastography vs. transient elastography in detection of fibrosis and noninvasive measurement of steatosis in patients with biopsy-proven nonalcoholic fatty liver disease. Gastroenterology. 2017;152:598–607.e2. doi: 10.1053/j.gastro.2016.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Boursier J., Vergniol J., Guillet A., Hiriart J.B., Lannes A., Le Bail B., Michalak S., Chermak F., Bertrais S., Foucher J., et al. Diagnostic accuracy and prognostic significance of blood fibrosis tests and liver stiffness measurement by FibroScan in non-alcoholic fatty liver disease. J. Hepatol. 2016;65:570–578. doi: 10.1016/j.jhep.2016.04.023. [DOI] [PubMed] [Google Scholar]
- 15.Ludwig J., Viggiano T.R., McGill D.B., Oh B.J. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin. Proc. Mayo Clin. 1980;55:434–438. [PubMed] [Google Scholar]
- 16.Caldwell S.H., Oelsner D.H., Iezzoni J.C., Hespenheide E.E., Battle E.H., Driscoll C.J. Cryptogenic cirrhosis: Clinical characterization and risk factors for underlying disease. Hepatology. 1999;29:664–669. doi: 10.1002/hep.510290347. [DOI] [PubMed] [Google Scholar]
- 17.Browning J.D., Kumar K.S., Saboorian M.H., Thiele D.L. Ethnic differences in the prevalence of cryptogenic cirrhosis. Am. J. Gastroenterol. 2004;99:292–298. doi: 10.1111/j.1572-0241.2004.04059.x. [DOI] [PubMed] [Google Scholar]
- 18.Nasr P., Ignatova S., Kechagias S., Ekstedt M. Natural history of nonalcoholic fatty liver disease: A prospective follow-up study with serial biopsies. Hepatol. Commun. 2018;2:199–210. doi: 10.1002/hep4.1134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mittal S., El-Serag H.B., Sada Y.H., Kanwal F., Duan Z., Temple S., May S.B., Kramer J.R., Richardson P.A., Davila J.A. Hepatocellular carcinoma in the absence of cirrhosis in united states veterans is associated with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2016;14:124–131.e1. doi: 10.1016/j.cgh.2015.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen Z., Qin H., Qiu S., Chen G., Chen Y. Correlation of triglyceride to high-density lipoprotein cholesterol ratio with nonalcoholic fatty liver disease among the non-obese Chinese population with normal blood lipid levels: A retrospective cohort research. Lipids Health Dis. 2019;18:162. doi: 10.1186/s12944-019-1104-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ren X.Y., Shi D., Ding J., Cheng Z.Y., Li H.Y., Li J.S., Pu H.Q., Yang A.M., He C.L., Zhang J.P., et al. Total cholesterol to high-density lipoprotein cholesterol ratio is a significant predictor of nonalcoholic fatty liver: Jinchang cohort study. Lipids Health Dis. 2019;18:47. doi: 10.1186/s12944-019-0984-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Eslam M., Sanyal A.J., George J., International Consensus Panel MAFLD: A consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology. 2020;158:1999–2014.e1. doi: 10.1053/j.gastro.2019.11.312. [DOI] [PubMed] [Google Scholar]
- 23.Loomis A.K., Kabadi S., Preiss D., Hyde C., Bonato V., St Louis M., Desai J., Gill J.M., Welsh P., Waterworth D., et al. Body mass index and risk of nonalcoholic fatty liver disease: Two electronic health record prospective studies. J. Clin. Endocrinol. Metab. 2016;101:945–952. doi: 10.1210/jc.2015-3444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Younes R., Bugianesi E. NASH in lean individuals. Semin. Liver Dis. 2019;39:86–95. doi: 10.1055/s-0038-1677517. [DOI] [PubMed] [Google Scholar]
- 25.NCD Risk Factor Collaboration (NCD-RisC) Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet. 2017;390:2627–2642. doi: 10.1016/S0140-6736(17)32129-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yun Y., Kim H.N., Lee E.J., Ryu S., Chang Y., Shin H., Kim H.L., Kim T.H., Yoo K., Kim H.Y. Fecal and blood microbiota profiles and presence of nonalcoholic fatty liver disease in obese versus lean subjects. PLoS ONE. 2019;14:e0213692. doi: 10.1371/journal.pone.0213692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vecchie A., Dallegri F., Carbone F., Bonaventura A., Liberale L., Portincasa P., Fruhbeck G., Montecucco F. Obesity phenotypes and their paradoxical association with cardiovascular diseases. Eur. J. Intern. Med. 2018;48:6–17. doi: 10.1016/j.ejim.2017.10.020. [DOI] [PubMed] [Google Scholar]
- 28.Molina-Molina E., Krawczyk M., Stachowska E., Lammert F., Portincasa P. Non-alcoholic fatty liver disease in non-obese individuals: Prevalence, pathogenesis and treatment. Clin. Res. Hepatol. Gastroenterol. 2019;43:638–645. doi: 10.1016/j.clinre.2019.04.005. [DOI] [PubMed] [Google Scholar]
- 29.Loomba R., Sanyal A.J. The global NAFLD epidemic. Nat. Rev. Gastroenterol. Hepatol. 2013;10:686–690. doi: 10.1038/nrgastro.2013.171. [DOI] [PubMed] [Google Scholar]
- 30.Wang A.Y., Dhaliwal J., Mouzaki M. Lean non-alcoholic fatty liver disease. Clin. Nutr. 2019;38:975–981. doi: 10.1016/j.clnu.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 31.Younossi Z.M., Stepanova M., Negro F., Hallaji S., Younossi Y., Lam B., Srishord M. Nonalcoholic fatty liver disease in lean individuals in the United States. Medicine. 2012;91:319–327. doi: 10.1097/MD.0b013e3182779d49. [DOI] [PubMed] [Google Scholar]
- 32.Chang Y., Ryu S., Sung K.C., Cho Y.K., Sung E., Kim H.N., Jung H.S., Yun K.E., Ahn J., Shin H., et al. Alcoholic and non-alcoholic fatty liver disease and associations with coronary artery calcification: Evidence from the Kangbuk Samsung Health Study. Gut. 2019;68:1667–1675. doi: 10.1136/gutjnl-2018-317666. [DOI] [PubMed] [Google Scholar]
- 33.Younossi Z.M. Non-alcoholic fatty liver disease—A global public health perspective. J. Hepatol. 2019;70:531–544. doi: 10.1016/j.jhep.2018.10.033. [DOI] [PubMed] [Google Scholar]
- 34.Kim S.S., Cho H.J., Kim H.J., Kang D.R., Berry J.R., Kim J.H., Yang M.J., Lim S.G., Kim S., Cheong J.Y., et al. Nonalcoholic fatty liver disease as a sentinel marker for the development of diabetes mellitus in non-obese subjects. Dig. Liver Dis. 2018;50:370–377. doi: 10.1016/j.dld.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 35.Golabi P., Otgonsuren M., de Avila L., Sayiner M., Rafiq N., Younossi Z.M. Components of metabolic syndrome increase the risk of mortality in nonalcoholic fatty liver disease (NAFLD) Medicine. 2018;97:e0214. doi: 10.1097/MD.0000000000010214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wong V.W., Wong G.L., Yip G.W., Lo A.O., Limquiaco J., Chu W.C., Chim A.M., Yu C.M., Yu J., Chan F.K., et al. Coronary artery disease and cardiovascular outcomes in patients with non-alcoholic fatty liver disease. Gut. 2011;60:1721–1727. doi: 10.1136/gut.2011.242016. [DOI] [PubMed] [Google Scholar]
- 37.Assimakopoulos K., Karaivazoglou K., Tsermpini E.E., Diamantopoulou G., Triantos C. Quality of life in patients with nonalcoholic fatty liver disease: A systematic review. J. Psychosom. Res. 2018;112:73–80. doi: 10.1016/j.jpsychores.2018.07.004. [DOI] [PubMed] [Google Scholar]
- 38.Faienza M.F., Chiarito M., Molina-Molina E., Shanmugam H., Lammert F., Krawczyk M., D’Amato G., Portincasa P. Childhood obesity, cardiovascular and liver health: A growing epidemic with age. World J. Pediatr. WJP. 2020 doi: 10.1007/s12519-020-00341-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Molina-Molina E., Lunardi Baccetto R., Wang D.Q., de Bari O., Krawczyk M., Portincasa P. Exercising the hepatobiliary-gut axis. The impact of physical activity performance. Eur. J. Clin. Investig. 2018;48:e12958. doi: 10.1111/eci.12958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chalasani N., Younossi Z., Lavine J.E., Charlton M., Cusi K., Rinella M., Harrison S.A., Brunt E.M., Sanyal A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–357. doi: 10.1002/hep.29367. [DOI] [PubMed] [Google Scholar]
- 41.Chen Z., Yu Y., Cai J., Li H. Emerging Molecular Targets for Treatment of Nonalcoholic Fatty Liver Disease. Trends Endocrinol. Metab. 2019;30:903–914. doi: 10.1016/j.tem.2019.08.006. [DOI] [PubMed] [Google Scholar]
- 42.Portincasa P., Krawczyk M., Smyk W., Lammert F., Di Ciaula A. COVID-19 and nonalcoholic fatty liver disease: Two intersecting pandemics. Eur. J. Clin. Investig. 2020:e13338. doi: 10.1111/eci.13338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Martins M.J., Ascensao A., Magalhaes J., Collado M.C., Portincasa P. Molecular mechanisms of NAFLD in metabolic syndrome. BioMed Res. Int. 2015;2015:621080. doi: 10.1155/2015/621080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Day C.P., James O.F. Steatohepatitis: A tale of two “hits”? Gastroenterology. 1998;114:842–845. doi: 10.1016/S0016-5085(98)70599-2. [DOI] [PubMed] [Google Scholar]
- 45.Farrell G.C., Larter C.Z. Nonalcoholic fatty liver disease: From steatosis to cirrhosis. Hepatology. 2006;43:S99–S112. doi: 10.1002/hep.20973. [DOI] [PubMed] [Google Scholar]
- 46.Bai L., Li H. Innate immune regulatory networks in hepatic lipid metabolism. J. Mol. Med. 2019;97:593–604. doi: 10.1007/s00109-019-01765-1. [DOI] [PubMed] [Google Scholar]
- 47.Samuel V.T., Shulman G.I. Nonalcoholic fatty liver disease as a nexus of metabolic and hepatic diseases. Cell Metab. 2018;27:22–41. doi: 10.1016/j.cmet.2017.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Arab J.P., Arrese M., Trauner M. Recent insights into the pathogenesis of nonalcoholic fatty liver disease. Annu. Rev. Pathol. 2018;13:321–350. doi: 10.1146/annurev-pathol-020117-043617. [DOI] [PubMed] [Google Scholar]
- 49.Arab J.P., Karpen S.J., Dawson P.A., Arrese M., Trauner M. Bile acids and nonalcoholic fatty liver disease: Molecular insights and therapeutic perspectives. Hepatology. 2017;65:350–362. doi: 10.1002/hep.28709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Neuschwander-Tetri B.A., Loomba R., Sanyal A.J., Lavine J.E., Van Natta M.L., Abdelmalek M.F., Chalasani N., Dasarathy S., Diehl A.M., Hameed B., et al. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): A multicentre, randomised, placebo-controlled trial. Lancet. 2015;385:956–965. doi: 10.1016/S0140-6736(14)61933-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Marra F., Svegliati-Baroni G. Lipotoxicity and the gut-liver axis in NASH pathogenesis. J. Hepatol. 2018;68:280–295. doi: 10.1016/j.jhep.2017.11.014. [DOI] [PubMed] [Google Scholar]
- 52.Sunny N.E., Parks E.J., Browning J.D., Burgess S.C. Excessive hepatic mitochondrial TCA cycle and gluconeogenesis in humans with nonalcoholic fatty liver disease. Cell Metab. 2011;14:804–810. doi: 10.1016/j.cmet.2011.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lambert J.E., Ramos-Roman M.A., Browning J.D., Parks E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology. 2014;146:726–735. doi: 10.1053/j.gastro.2013.11.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Donnelly K.L., Smith C.I., Schwarzenberg S.J., Jessurun J., Boldt M.D., Parks E.J. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J. Clin. Investig. 2005;115:1343–1351. doi: 10.1172/JCI23621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Han M.S., Park S.Y., Shinzawa K., Kim S., Chung K.W., Lee J.H., Kwon C.H., Lee K.W., Lee J.H., Park C.K., et al. Lysophosphatidylcholine as a death effector in the lipoapoptosis of hepatocytes. J. Lipid Res. 2008;49:84–97. doi: 10.1194/jlr.M700184-JLR200. [DOI] [PubMed] [Google Scholar]
- 56.Mota M., Banini B.A., Cazanave S.C., Sanyal A.J. Molecular mechanisms of lipotoxicity and glucotoxicity in nonalcoholic fatty liver disease. Metab. Clin. Exp. 2016;65:1049–1061. doi: 10.1016/j.metabol.2016.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Pagadala M., Kasumov T., McCullough A.J., Zein N.N., Kirwan J.P. Role of ceramides in nonalcoholic fatty liver disease. Trends Endocrinol. Metab. 2012;23:365–371. doi: 10.1016/j.tem.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bellanti F., Mitarotonda D., Tamborra R., Blonda M., Iannelli G., Petrella A., Sanginario V., Iuliano L., Vendemiale G., Serviddio G. Oxysterols induce mitochondrial impairment and hepatocellular toxicity in non-alcoholic fatty liver disease. Free Radic. Biol. Med. 2014;75(Suppl. 1):S16–S17. doi: 10.1016/j.freeradbiomed.2014.10.594. [DOI] [PubMed] [Google Scholar]
- 59.Ioannou G.N. The role of cholesterol in the pathogenesis of NASH. Trends Endocrinol. Metab. 2016;27:84–95. doi: 10.1016/j.tem.2015.11.008. [DOI] [PubMed] [Google Scholar]
- 60.Tirosh O. Hypoxic signaling and cholesterol lipotoxicity in fatty liver disease progression. Oxidative Med. Cell. Longev. 2018;2018:2548154. doi: 10.1155/2018/2548154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ho C.M., Ho S.L., Jeng Y.M., Lai Y.S., Chen Y.H., Lu S.C., Chen H.L., Chang P.Y., Hu R.H., Lee P.H. Accumulation of free cholesterol and oxidized low-density lipoprotein is associated with portal inflammation and fibrosis in nonalcoholic fatty liver disease. J. Inflamm. 2019;16:7. doi: 10.1186/s12950-019-0211-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wahlang B., Jin J., Beier J.I., Hardesty J.E., Daly E.F., Schnegelberger R.D., Falkner K.C., Prough R.A., Kirpich I.A., Cave M.C. Mechanisms of environmental contributions to fatty liver disease. Curr. Environ. Health Rep. 2019;6:80–94. doi: 10.1007/s40572-019-00232-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fu S., Watkins S.M., Hotamisligil G.S. The role of endoplasmic reticulum in hepatic lipid homeostasis and stress signaling. Cell Metab. 2012;15:623–634. doi: 10.1016/j.cmet.2012.03.007. [DOI] [PubMed] [Google Scholar]
- 64.Perry R.J., Samuel V.T., Petersen K.F., Shulman G.I. The role of hepatic lipids in hepatic insulin resistance and type 2 diabetes. Nature. 2014;510:84–91. doi: 10.1038/nature13478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ertunc M.E., Hotamisligil G.S. Lipid signaling and lipotoxicity in metaflammation: Indications for metabolic disease pathogenesis and treatment. J. Lipid Res. 2016;57:2099–2114. doi: 10.1194/jlr.R066514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cai J., Xu M., Zhang X., Li H. Innate immune signaling in nonalcoholic fatty liver disease and cardiovascular diseases. Annu. Rev. Pathol. 2019;14:153–184. doi: 10.1146/annurev-pathmechdis-012418-013003. [DOI] [PubMed] [Google Scholar]
- 67.Cai J., Zhang X.J., Li H. The role of innate immune cells in nonalcoholic steatohepatitis. Hepatology. 2019;70:1026–1037. doi: 10.1002/hep.30506. [DOI] [PubMed] [Google Scholar]
- 68.Wang X.A., Zhang R., She Z.G., Zhang X.F., Jiang D.S., Wang T., Gao L., Deng W., Zhang S.M., Zhu L.H., et al. Interferon regulatory factor 3 constrains IKKbeta/NF-kappaB signaling to alleviate hepatic steatosis and insulin resistance. Hepatology. 2014;59:870–885. doi: 10.1002/hep.26751. [DOI] [PubMed] [Google Scholar]
- 69.Müller F.A., Sturla S.J. Human in vitro models of nonalcoholic fatty liver disease. Curr. Opin. Toxicol. 2019;16:9–16. doi: 10.1016/j.cotox.2019.03.001. [DOI] [Google Scholar]
- 70.Aung H.H., Altman R., Nyunt T., Kim J., Nuthikattu S., Budamagunta M., Voss J.C., Wilson D., Rutledge J.C., Villablanca A.C. Lipotoxic brain microvascular injury is mediated by activating transcription factor 3-dependent inflammatory and oxidative stress pathways. J. Lipid Res. 2016;57:955–968. doi: 10.1194/jlr.M061853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Le Stunff H., Coant N., Migrenne S., Magnan C. Targeting lipid sensing in the central nervous system: New therapy against the development of obesity and type 2 diabetes. Expert Opin. Ther. Targets. 2013;17:545–555. doi: 10.1517/14728222.2013.768233. [DOI] [PubMed] [Google Scholar]
- 72.Portincasa P., Wang D.Q.H. Nonalcoholic fatty liver and gallstone disease. In: Wang D.Q.H., Portincasa P., editors. Gallstones. Recent Advances in Epidemiology, Pathogenesis, Diagnosis and Management. Nova Science Publisher Inc.; New York, NY, USA: 2017. pp. 387–414. [Google Scholar]
- 73.Grattagliano I., De Bari O., Di Palo D., Montecucco F., Carbone F., Oliveira P., Wang D.Q.H., Portincasa P. Mitochondria in liver diseases. In: Oliveira P., editor. Mitochondrial Biology and Experimental Therapeutics. Springer Nature; Cham, Switzerland: 2018. pp. 91–126. [DOI] [Google Scholar]
- 74.Karlsen T.H., Lammert F., Thompson R.J. Genetics of liver disease: From pathophysiology to clinical practice. J. Hepatol. 2015;62:S6–S14. doi: 10.1016/j.jhep.2015.02.025. [DOI] [PubMed] [Google Scholar]
- 75.Krawczyk M., Portincasa P., Lammert F. PNPLA3-associated steatohepatitis: Toward a gene-based classification of fatty liver disease. Semin. Liver Dis. 2013;33:369–379. doi: 10.1055/s-0033-1358525. [DOI] [PubMed] [Google Scholar]
- 76.Albillos A., Gottardi A., Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J. Hepatol. 2019 doi: 10.1016/j.jhep.2019.10.003. [DOI] [PubMed] [Google Scholar]
- 77.Kim D., Yoo E.R., Li A.A., Cholankeril G., Tighe S.P., Kim W., Harrison S.A., Ahmed A. Elevated urinary bisphenol a levels are associated with non-alcoholic fatty liver disease among adults in the United States. Liver Int. Off. J. Int. Assoc. Study Liver. 2019;39:1335–1342. doi: 10.1111/liv.14110. [DOI] [PubMed] [Google Scholar]
- 78.Franco M.E., Fernandez-Luna M.T., Ramirez A.J., Lavado R. Metabolomic-based assessment reveals dysregulation of lipid profiles in human liver cells exposed to environmental obesogens. Toxicol. Appl. Pharmacol. 2020;398:115009. doi: 10.1016/j.taap.2020.115009. [DOI] [PubMed] [Google Scholar]
- 79.Wahlang B., Appana S., Falkner K.C., McClain C.J., Brock G., Cave M.C. Insecticide and metal exposures are associated with a surrogate biomarker for non-alcoholic fatty liver disease in the National Health and Nutrition Examination Survey 2003–2004. Environ. Sci. Pollut. Res. Int. 2020;27:6476–6487. doi: 10.1007/s11356-019-07066-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Milosevic N., Milanovic M., Sudji J., Bosic Zivanovic D., Stojanoski S., Vukovic B., Milic N., Medic Stojanoska M. Could phthalates exposure contribute to the development of metabolic syndrome and liver disease in humans? Environ. Sci. Pollut. Res. Int. 2020;27:772–784. doi: 10.1007/s11356-019-06831-2. [DOI] [PubMed] [Google Scholar]
- 81.Wang X., Yang Y., Zhu P., Wu Y., Jin Y., Yu S., Wei H., Qian M., Cao W., Xu S., et al. Prenatal exposure to diesel exhaust PM2.5 programmed non-alcoholic fatty liver disease differently in adult male offspring of mice fed normal chow and a high-fat diet. Environ. Pollut. 2019;255:113366. doi: 10.1016/j.envpol.2019.113366. [DOI] [PubMed] [Google Scholar]
- 82.Chen R., Xu Y., Xu C., Shu Y., Ma S., Lu C., Mo X. Associations between mercury exposure and the risk of nonalcoholic fatty liver disease (NAFLD) in US adolescents. Environ. Sci. Pollut. Res. Int. 2019;26:31384–31391. doi: 10.1007/s11356-019-06224-5. [DOI] [PubMed] [Google Scholar]
- 83.Ding S., Yuan C., Si B., Wang M., Da S., Bai L., Wu W. Combined effects of ambient particulate matter exposure and a high-fat diet on oxidative stress and steatohepatitis in mice. PLoS ONE. 2019;14:e0214680. doi: 10.1371/journal.pone.0214680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Xu M.X., Ge C.X., Qin Y.T., Gu T.T., Lou D.S., Li Q., Hu L.F., Feng J., Huang P., Tan J. Prolonged PM2.5 exposure elevates risk of oxidative stress-driven nonalcoholic fatty liver disease by triggering increase of dyslipidemia. Free Radic. Biol. Med. 2019;130:542–556. doi: 10.1016/j.freeradbiomed.2018.11.016. [DOI] [PubMed] [Google Scholar]
- 85.Schnabl B., Brenner D.A. Interactions between the intestinal microbiome and liver diseases. Gastroenterology. 2014;146:1513–1524. doi: 10.1053/j.gastro.2014.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Liquori G.E., Mastrodonato M., Mentino D., Scillitani G., Desantis S., Portincasa P., Ferri D. In situ characterization of O-linked glycans of Muc2 in mouse colon. Acta Histochem. 2012;114:723–732. doi: 10.1016/j.acthis.2011.12.009. [DOI] [PubMed] [Google Scholar]
- 87.Mouries J., Brescia P., Silvestri A., Spadoni I., Sorribas M., Wiest R., Mileti E., Galbiati M., Invernizzi P., Adorini L., et al. Microbiota-driven gut vascular barrier disruption is a prerequisite for non-alcoholic steatohepatitis development. J. Hepatol. 2019;71:1216–1228. doi: 10.1016/j.jhep.2019.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Brandl K., Kumar V., Eckmann L. Gut-liver axis at the frontier of host-microbial interactions. Am. J. Physiol. Gastrointest. Liver Physiol. 2017;312:G413–G419. doi: 10.1152/ajpgi.00361.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Garruti G., Di Ciaula A., Wang H.H., Wang D.Q., Portincasa P. Cross-talk between bile acids and gastro-intestinal and thermogenic hormones: Clues from bariatric surgery. Ann. Hepatol. 2017;16:S68–S82. doi: 10.5604/01.3001.0010.5499. [DOI] [PubMed] [Google Scholar]
- 90.Garruti G., Wang D.Q., Di Ciaula A., Portincasa P. Cholecystectomy: A way forward and back to metabolic syndrome? Lab. Investig. 2018;98:4–6. doi: 10.1038/labinvest.2017.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Nicoletti A., Ponziani F.R., Biolato M., Valenza V., Marrone G., Sganga G., Gasbarrini A., Miele L., Grieco A. Intestinal permeability in the pathogenesis of liver damage: From non-alcoholic fatty liver disease to liver transplantation. World J. Gastroenterol. WJG. 2019;25:4814–4834. doi: 10.3748/wjg.v25.i33.4814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Okumura R., Takeda K. Maintenance of intestinal homeostasis by mucosal barriers. Inflamm. Regen. 2018;38:5. doi: 10.1186/s41232-018-0063-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Meyer-Hoffert U., Hornef M.W., Henriques-Normark B., Axelsson L.G., Midtvedt T., Putsep K., Andersson M. Secreted enteric antimicrobial activity localises to the mucus surface layer. Gut. 2008;57:764–771. doi: 10.1136/gut.2007.141481. [DOI] [PubMed] [Google Scholar]
- 94.Savage D.C. Microbial ecology of the gastrointestinal tract. Annu. Rev. Microbiol. 1977;31:107–133. doi: 10.1146/annurev.mi.31.100177.000543. [DOI] [PubMed] [Google Scholar]
- 95.Lynch S.V., Pedersen O. The human intestinal microbiome in health and disease. N. Engl. J. Med. 2016;375:2369–2379. doi: 10.1056/NEJMra1600266. [DOI] [PubMed] [Google Scholar]
- 96.Jandhyala S.M., Talukdar R., Subramanyam C., Vuyyuru H., Sasikala M., Nageshwar Reddy D. Role of the normal gut microbiota. World J. Gastroenterol. WJG. 2015;21:8787–8803. doi: 10.3748/wjg.v21.i29.8787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kim Y.S., Ho S.B. Intestinal goblet cells and mucins in health and disease: Recent insights and progress. Curr. Gastroenterol. Rep. 2010;12:319–330. doi: 10.1007/s11894-010-0131-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Johansson M.E., Phillipson M., Petersson J., Velcich A., Holm L., Hansson G.C. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl. Acad. Sci. USA. 2008;105:15064–15069. doi: 10.1073/pnas.0803124105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Vereecke L., Beyaert R., van Loo G. Enterocyte death and intestinal barrier maintenance in homeostasis and disease. Trends Mol. Med. 2011;17:584–593. doi: 10.1016/j.molmed.2011.05.011. [DOI] [PubMed] [Google Scholar]
- 100.Tsilingiri K., Barbosa T., Penna G., Caprioli F., Sonzogni A., Viale G., Rescigno M. Probiotic and postbiotic activity in health and disease: Comparison on a novel polarised ex-vivo organ culture model. Gut. 2012;61:1007–1015. doi: 10.1136/gutjnl-2011-300971. [DOI] [PubMed] [Google Scholar]
- 101.Tsilingiri K., Rescigno M. Beneficial Microbes 4. Wageningen Academic Publishers; Wageningen, The Netherlands: 2013. Postbiotics: What else? pp. 101–107. [DOI] [PubMed] [Google Scholar]
- 102.Levy M., Blacher E., Elinav E. Microbiome, metabolites and host immunity. Curr. Opin. Microbiol. 2017;35:8–15. doi: 10.1016/j.mib.2016.10.003. [DOI] [PubMed] [Google Scholar]
- 103.Blacher E., Levy M., Tatirovsky E., Elinav E. Microbiome-modulated metabolites at the interface of host immunity. J. Immunol. 2017;198:572–580. doi: 10.4049/jimmunol.1601247. [DOI] [PubMed] [Google Scholar]
- 104.Mosca F., Gianni M.L., Rescigno M. Probiotics and Child Gastrointestinal Health. Springer; Cham, Switzerland: 2019. Can Postbiotics represent a new strategy for NEC? [Google Scholar]
- 105.Gibbins H.L., Proctor G.B., Yakubov G.E., Wilson S., Carpenter G.H. SIgA binding to mucosal surfaces is mediated by mucin-mucin interactions. PLoS ONE. 2015;10:e0119677. doi: 10.1371/journal.pone.0119677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Jakobsson H.E., Rodriguez-Pineiro A.M., Schutte A., Ermund A., Boysen P., Bemark M., Sommer F., Backhed F., Hansson G.C., Johansson M.E. The composition of the gut microbiota shapes the colon mucus barrier. EMBO Rep. 2015;16:164–177. doi: 10.15252/embr.201439263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Birchenough G.M., Nystrom E.E., Johansson M.E., Hansson G.C. A sentinel goblet cell guards the colonic crypt by triggering Nlrp6-dependent Muc2 secretion. Science. 2016;352:1535–1542. doi: 10.1126/science.aaf7419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Abreu M.T. Toll-like receptor signalling in the intestinal epithelium: How bacterial recognition shapes intestinal function. Nat. Rev. Immunol. 2010;10:131. doi: 10.1038/nri2707. [DOI] [PubMed] [Google Scholar]
- 109.Ouwerkerk J.P., de Vos W.M., Belzer C. Glycobiome: Bacteria and mucus at the epithelial interface. Best Pract. Res. Clin. Gastroenterol. 2013;27:25–38. doi: 10.1016/j.bpg.2013.03.001. [DOI] [PubMed] [Google Scholar]
- 110.Derrien M., Van Baarlen P., Hooiveld G., Norin E., Muller M., de Vos W.M. Modulation of mucosal immune response, tolerance, and proliferation in mice colonized by the mucin-degrader akkermansia muciniphila. Front. Microbiol. 2011;2:166. doi: 10.3389/fmicb.2011.00166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Dao M.C., Everard A., Aron-Wisnewsky J., Sokolovska N., Prifti E., Verger E.O., Kayser B.D., Levenez F., Chilloux J., Hoyles L., et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: Relationship with gut microbiome richness and ecology. Gut. 2016;65:426–436. doi: 10.1136/gutjnl-2014-308778. [DOI] [PubMed] [Google Scholar]
- 112.Grander C., Adolph T.E., Wieser V., Lowe P., Wrzosek L., Gyongyosi B., Ward D.V., Grabherr F., Gerner R.R., Pfister A., et al. Recovery of ethanol-induced Akkermansia muciniphila depletion ameliorates alcoholic liver disease. Gut. 2018;67:891–901. doi: 10.1136/gutjnl-2016-313432. [DOI] [PubMed] [Google Scholar]
- 113.Everard A., Belzer C., Geurts L., Ouwerkerk J.P., Druart C., Bindels L.B., Guiot Y., Derrien M., Muccioli G.G., Delzenne N.M. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA. 2013;110:9066–9071. doi: 10.1073/pnas.1219451110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Desai M.S., Seekatz A.M., Koropatkin N.M., Kamada N., Hickey C.A., Wolter M., Pudlo N.A., Kitamoto S., Terrapon N., Muller A., et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell. 2016;167:1339–1353.e21. doi: 10.1016/j.cell.2016.10.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Wrzosek L., Miquel S., Noordine M.L., Bouet S., Chevalier-Curt M.J., Robert V., Philippe C., Bridonneau C., Cherbuy C., Robbe-Masselot C., et al. Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 2013;11:61. doi: 10.1186/1741-7007-11-61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Park J.-H., Eberl G. Type 3 regulatory T cells at the interface of symbiosis. J. Microbiol. 2018;56:163–171. doi: 10.1007/s12275-018-7565-x. [DOI] [PubMed] [Google Scholar]
- 117.Belkaid Y., Hand T.W. Role of the microbiota in immunity and inflammation. Cell. 2014;157:121–141. doi: 10.1016/j.cell.2014.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Reynes B., Palou M., Rodriguez A.M., Palou A. Regulation of adaptive thermogenesis and browning by prebiotics and postbiotics. Front. Physiol. 2018;9:1908. doi: 10.3389/fphys.2018.01908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bergström J.H., Birchenough G.M.H., Katona G., Schroeder B.O., Schütte A., Ermund A., Johansson M.E.V., Hansson G.C. Gram-positive bacteria are held at a distance in the colon mucus by the lectin-like protein ZG16. Proc. Natl. Acad. Sci. USA. 2016;113:13833–13838. doi: 10.1073/pnas.1611400113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ismail A.S., Severson K.M., Vaishnava S., Behrendt C.L., Yu X., Benjamin J.L., Ruhn K.A., Hou B., DeFranco A.L., Yarovinsky F. γδ intraepithelial lymphocytes are essential mediators of host–microbial homeostasis at the intestinal mucosal surface. Proc. Natl. Acad. Sci. USA. 2011;108:8743–8748. doi: 10.1073/pnas.1019574108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Johansson M.E.V. Fast renewal of the distal colonic mucus layers by the surface goblet cells as measured by in vivo labeling of mucin glycoproteins. PLoS ONE. 2012;7:e41009. doi: 10.1371/journal.pone.0041009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mastrodonato M., Mentino D., Portincasa P., Calamita G., Liquori G.E., Ferri D. High-fat diet alters the oligosaccharide chains of colon mucins in mice. Histochem. Cell Biol. 2014;142:449–459. doi: 10.1007/s00418-014-1221-2. [DOI] [PubMed] [Google Scholar]
- 123.Ponziani F.R., Gerardi V., Gasbarrini A. Diagnosis and treatment of small intestinal bacterial overgrowth. Expert Rev. Gastroenterol. Hepatol. 2016;10:215–227. doi: 10.1586/17474124.2016.1110017. [DOI] [PubMed] [Google Scholar]
- 124.Inagaki T., Choi M., Moschetta A., Peng L., Cummins C.L., McDonald J.G., Luo G., Jones S.A., Goodwin B., Richardson J.A., et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab. 2005;2:217–225. doi: 10.1016/j.cmet.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 125.Garruti G., Wang H.H., Bonfrate L., de Bari O., Wang D.Q., Portincasa P. A pleiotropic role for the orphan nuclear receptor small heterodimer partner in lipid homeostasis and metabolic pathways. J. Lipids. 2012;2012:304292. doi: 10.1155/2012/304292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Liu H., Hu C., Zhang X., Jia W. Role of gut microbiota, bile acids and their cross-talk in the effects of bariatric surgery on obesity and type 2 diabetes. J. Diabetes Investig. 2018;9:13–20. doi: 10.1111/jdi.12687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Wahlström A., Sayin S.I., Marschall H.-U., Bäckhed F. Intestinal crosstalk between bile acids and microbiota and its impact on host metabolism. Cell Metab. 2016;24:41–50. doi: 10.1016/j.cmet.2016.05.005. [DOI] [PubMed] [Google Scholar]
- 128.Ory D.S. Nuclear receptor signaling in the control of cholesterol homeostasis: Have the orphans found a home? Circ. Res. 2004;95:660–670. doi: 10.1161/01.RES.0000143422.83209.be. [DOI] [PubMed] [Google Scholar]
- 129.De Aguiar Vallim T.Q., Tarling E.J., Edwards P.A. Pleiotropic roles of bile acids in metabolism. Cell Metab. 2013;17:657–669. doi: 10.1016/j.cmet.2013.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Luiking Y.C., Peeters T.L., Stolk M.F., Nieuwenhuijs V.B., Portincasa P., Depoortere I., van Berge Henegouwen G.P., Akkermans L.M. Motilin induces gall bladder emptying and antral contractions in the fasted state in humans. Gut. 1998;42:830–835. doi: 10.1136/gut.42.6.830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Portincasa P., Peeters T.L., van Berge-Henegouwen G.P., van Solinge W.W., Palasciano G., van Erpecum K.J. Acute intraduodenal bile salt depletion leads to strong gallbladder contraction, altered antroduodenal motility and high plasma motilin levels in humans. Neurogastroenterol. Motil. 2000;12:421–430. doi: 10.1046/j.1365-2982.2000.00217.x. [DOI] [PubMed] [Google Scholar]
- 132.Portincasa P., Di Ciaula A., Wang H.H., Palasciano G., van Erpecum K.J., Moschetta A., Wang D.Q. Coordinate regulation of gallbladder motor function in the gut-liver axis. Hepatology. 2008;47:2112–2126. doi: 10.1002/hep.22204. [DOI] [PubMed] [Google Scholar]
- 133.Jansson R., Steen G., Svanvik J. Effects of intravenous vasoactive intestinal peptide (VIP) on gallbladder function in the cat. Gastroenterology. 1978;75:47–50. doi: 10.1016/0016-5085(78)93762-9. [DOI] [PubMed] [Google Scholar]
- 134.Housset C., Chretien Y., Debray D., Chignard N. Functions of the Gallbladder. Compr. Physiol. 2016;6:1549–1577. doi: 10.1002/cphy.c150050. [DOI] [PubMed] [Google Scholar]
- 135.Choi M., Moschetta A., Bookout A.L., Peng L., Umetani M., Holmstrom S.R., Suino-Powell K., Xu H.E., Richardson J.A., Gerard R.D., et al. Identification of a hormonal basis for gallbladder filling. Nat. Med. 2006;12:1253–1255. doi: 10.1038/nm1501. [DOI] [PubMed] [Google Scholar]
- 136.Chiang J.Y. Bile acid metabolism and signaling. Compr. Physiol. 2013;3:1191–1212. doi: 10.1002/cphy.c120023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Dawson P.A., Lan T., Rao A. Bile acid transporters. J. Lipid Res. 2009;50:2340–2357. doi: 10.1194/jlr.R900012-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Bjorkhem I., Blomstrand R., Lewenhaupt A., Svensson L. Effect of lymphatic drainage on 7alpha-hydroxylation of cholesterol in rat liver. Biochem. Biophys. Res. Commun. 1978;85:532–540. doi: 10.1016/0006-291X(78)91196-8. [DOI] [PubMed] [Google Scholar]
- 139.Kemper J.K. Regulation of FXR transcriptional activity in health and disease: Emerging roles of FXR cofactors and post-translational modifications. Biochim. Biophys. Acta. 2011;1812:842–850. doi: 10.1016/j.bbadis.2010.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Goodwin B., Jones S.A., Price R.R., Watson M.A., McKee D.D., Moore L.B., Galardi C., Wilson J.G., Lewis M.C., Roth M.E., et al. A regulatory cascade of the nuclear receptors FXR, SHP-1, and LRH-1 represses bile acid biosynthesis. Mol. Cell. 2000;6:517–526. doi: 10.1016/S1097-2765(00)00051-4. [DOI] [PubMed] [Google Scholar]
- 141.Lu T.T., Makishima M., Repa J.J., Schoonjans K., Kerr T.A., Auwerx J., Mangelsdorf D.J. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol. Cell. 2000;6:507–515. doi: 10.1016/S1097-2765(00)00050-2. [DOI] [PubMed] [Google Scholar]
- 142.Nishimaki-Mogami T., Une M., Fujino T., Sato Y., Tamehiro N., Kawahara Y., Shudo K., Inoue K. Identification of intermediates in the bile acid synthetic pathway as ligands for the farnesoid X receptor. J. Lipid Res. 2004;45:1538–1545. doi: 10.1194/jlr.M400102-JLR200. [DOI] [PubMed] [Google Scholar]
- 143.Modica S., Bellafante E., Moschetta A. Master regulation of bile acid and xenobiotic metabolism via the FXR, PXR and CAR trio. Front. Biosci. (Landmark Ed.) 2008;14:4719–4745. doi: 10.2741/3563. [DOI] [PubMed] [Google Scholar]
- 144.Brighton C.A., Rievaj J., Kuhre R.E., Glass L.L., Schoonjans K., Holst J.J., Gribble F.M., Reimann F. Bile acids trigger GLP-1 release predominantly by accessing basolaterally located G Protein-coupled bile acid receptors. Endocrinology. 2015;156:3961–3970. doi: 10.1210/en.2015-1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Sinal C.J., Tohkin M., Miyata M., Ward J.M., Lambert G., Gonzalez F.J. Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell. 2000;102:731–744. doi: 10.1016/S0092-8674(00)00062-3. [DOI] [PubMed] [Google Scholar]
- 146.Pols T.W., Noriega L.G., Nomura M., Auwerx J., Schoonjans K. The bile acid membrane receptor TGR5: A valuable metabolic target. Dig. Dis. 2011;29:37–44. doi: 10.1159/000324126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Perino A., Schoonjans K. TGR5 and immunometabolism: Insights from physiology and pharmacology. Trends Pharm. Sci. 2015;36:847–857. doi: 10.1016/j.tips.2015.08.002. [DOI] [PubMed] [Google Scholar]
- 148.Li T., Chiang J.Y. Bile acid signaling in metabolic disease and drug therapy. Pharm. Rev. 2014;66:948–983. doi: 10.1124/pr.113.008201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wagner M., Zollner G., Trauner M. New molecular insights into the mechanisms of cholestasis. J. Hepatol. 2009;51:565–580. doi: 10.1016/j.jhep.2009.05.012. [DOI] [PubMed] [Google Scholar]
- 150.Kurashima Y., Kiyono H. Mucosal ecological network of epithelium and immune cells for gut homeostasis and tissue healing. Annu. Rev. Immunol. 2017;35:119–147. doi: 10.1146/annurev-immunol-051116-052424. [DOI] [PubMed] [Google Scholar]
- 151.Nevo S., Kadouri N., Abramson J. Tuft cells: From the mucosa to the thymus. Immunol. Lett. 2019;210:1–9. doi: 10.1016/j.imlet.2019.02.003. [DOI] [PubMed] [Google Scholar]
- 152.Turner J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009;9:799–809. doi: 10.1038/nri2653. [DOI] [PubMed] [Google Scholar]
- 153.Odenwald M.A., Turner J.R. The intestinal epithelial barrier: A therapeutic target? Nat. Rev. Gastroenterol. Hepatol. 2017;14:9–21. doi: 10.1038/nrgastro.2016.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Yamazaki Y., Okawa K., Yano T., Tsukita S., Tsukita S. Optimized proteomic analysis on gels of cell-cell adhering junctional membrane proteins. Biochemistry. 2008;47:5378–5386. doi: 10.1021/bi8002567. [DOI] [PubMed] [Google Scholar]
- 155.Schneeberger E.E., Lynch R.D. The tight junction: A multifunctional complex. Am. J. Physiol. Cell Physiol. 2004;286:C1213–C1228. doi: 10.1152/ajpcell.00558.2003. [DOI] [PubMed] [Google Scholar]
- 156.Van Itallie C.M., Anderson J.M. Architecture of tight junctions and principles of molecular composition. Semin. Cell Dev. Biol. 2014;36:157–165. doi: 10.1016/j.semcdb.2014.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Anderson J.M., Van Itallie C.M. Physiology and function of the tight junction. Cold Spring Harb. Perspect. Biol. 2009;1:a002584. doi: 10.1101/cshperspect.a002584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Van Itallie C.M., Holmes J., Bridges A., Gookin J.L., Coccaro M.R., Proctor W., Colegio O.R., Anderson J.M. The density of small tight junction pores varies among cell types and is increased by expression of claudin-2. J. Cell Sci. 2008;121:298–305. doi: 10.1242/jcs.021485. [DOI] [PubMed] [Google Scholar]
- 159.Taylor C.T., Dzus A.L., Colgan S.P. Autocrine regulation of epithelial permeability by hypoxia: Role for polarized release of tumor necrosis factor alpha. Gastroenterology. 1998;114:657–668. doi: 10.1016/S0016-5085(98)70579-7. [DOI] [PubMed] [Google Scholar]
- 160.Madara J.L., Stafford J. Interferon-gamma directly affects barrier function of cultured intestinal epithelial monolayers. J. Clin. Investig. 1989;83:724–727. doi: 10.1172/JCI113938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Turner J.R., Rill B.K., Carlson S.L., Carnes D., Kerner R., Mrsny R.J., Madara J.L. Physiological regulation of epithelial tight junctions is associated with myosin light-chain phosphorylation. Am. J. Physiol. 1997;273:C1378–C1385. doi: 10.1152/ajpcell.1997.273.4.C1378. [DOI] [PubMed] [Google Scholar]
- 162.Hartmann P., Haimerl M., Mazagova M., Brenner D.A., Schnabl B. Toll-like receptor 2-mediated intestinal injury and enteric tumor necrosis factor receptor I contribute to liver fibrosis in mice. Gastroenterology. 2012;143:1330–1340.e1. doi: 10.1053/j.gastro.2012.07.099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Cariello R., Federico A., Sapone A., Tuccillo C., Scialdone V.R., Tiso A., Miranda A., Portincasa P., Carbonara V., Palasciano G., et al. Intestinal permeability in patients with chronic liver diseases: Its relationship with the aetiology and the entity of liver damage. Dig. Liver Dis. 2010;42:200–204. doi: 10.1016/j.dld.2009.05.001. [DOI] [PubMed] [Google Scholar]
- 164.Assimakopoulos S.F., Tsamandas A.C., Tsiaoussis G.I., Karatza E., Triantos C., Vagianos C.E., Spiliopoulou I., Kaltezioti V., Charonis A., Nikolopoulou V.N., et al. Altered intestinal tight junctions’ expression in patients with liver cirrhosis: A pathogenetic mechanism of intestinal hyperpermeability. Eur. J. Clin. Investig. 2012;42:439–446. doi: 10.1111/j.1365-2362.2011.02609.x. [DOI] [PubMed] [Google Scholar]
- 165.Miele L., Valenza V., La Torre G., Montalto M., Cammarota G., Ricci R., Masciana R., Forgione A., Gabrieli M.L., Perotti G., et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology. 2009;49:1877–1887. doi: 10.1002/hep.22848. [DOI] [PubMed] [Google Scholar]
- 166.Bennett K.M., Walker S.L., Lo D.D. Epithelial microvilli establish an electrostatic barrier to microbial adhesion. Infect. Immun. 2014;82:2860–2871. doi: 10.1128/IAI.01681-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Di Palo D.M., Garruti G., Di Ciaula A., Molina-Molina E., Shanmugam H., De Angelis M., Portincasa P. Increased colonic permeability and lifestyles as contributing factors to obesity and liver steatosis. Nutrients. 2020;12:564. doi: 10.3390/nu12020564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Wiest R., Lawson M., Geuking M. Pathological bacterial translocation in liver cirrhosis. J. Hepatol. 2014;60:197–209. doi: 10.1016/j.jhep.2013.07.044. [DOI] [PubMed] [Google Scholar]
- 169.McDonald B.D., Jabri B., Bendelac A. Diverse developmental pathways of intestinal intraepithelial lymphocytes. Nat. Rev. Immunol. 2018;18:514–525. doi: 10.1038/s41577-018-0013-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Chieppa M., Rescigno M., Huang A.Y.C., Germain R.N. Dynamic imaging of dendritic cell extension into the small bowel lumen in response to epithelial cell TLR engagement. J. Exp. Med. 2006;203:2841–2852. doi: 10.1084/jem.20061884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Niess J.H., Brand S., Gu X., Landsman L., Jung S., McCormick B.A., Vyas J.M., Boes M., Ploegh H.L., Fox J.G., et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science. 2005;307:254–258. doi: 10.1126/science.1102901. [DOI] [PubMed] [Google Scholar]
- 172.Mazzini E., Massimiliano L., Penna G., Rescigno M. Oral tolerance can be established via gap junction transfer of fed antigens from CX3CR1+ macrophages to CD103+ dendritic cells. Immunity. 2014;40:248–261. doi: 10.1016/j.immuni.2013.12.012. [DOI] [PubMed] [Google Scholar]
- 173.Brennan P.J., Brigl M., Brenner M.B. Invariant natural killer T cells: An innate activation scheme linked to diverse effector functions. Nat. Rev. Immunol. 2013;13:101–117. doi: 10.1038/nri3369. [DOI] [PubMed] [Google Scholar]
- 174.Dias J., Leeansyah E., Sandberg J.K. Multiple layers of heterogeneity and subset diversity in human MAIT cell responses to distinct microorganisms and to innate cytokines. Proc. Natl. Acad. Sci. USA. 2017;114:E5434–E5443. doi: 10.1073/pnas.1705759114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Corbett A.J., Eckle S.B., Birkinshaw R.W., Liu L., Patel O., Mahony J., Chen Z., Reantragoon R., Meehan B., Cao H., et al. T-cell activation by transitory neo-antigens derived from distinct microbial pathways. Nature. 2014;509:361–365. doi: 10.1038/nature13160. [DOI] [PubMed] [Google Scholar]
- 176.Sandquist I., Kolls J. Update on regulation and effector functions of Th17 cells. F1000Res. 2018;7:205. doi: 10.12688/f1000research.13020.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Hirota K., Turner J.-E., Villa M., Duarte J.H., Demengeot J., Steinmetz O.M., Stockinger B. Plasticity of T H 17 cells in Peyer’s patches is responsible for the induction of T cell–dependent IgA responses. Nat. Immunol. 2013;14:372. doi: 10.1038/ni.2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Atarashi K., Tanoue T., Ando M., Kamada N., Nagano Y., Narushima S., Suda W., Imaoka A., Setoyama H., Nagamori T., et al. Th17 Cell Induction by Adhesion of Microbes to Intestinal Epithelial Cells. Cell. 2015;163:367–380. doi: 10.1016/j.cell.2015.08.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Gaboriau-Routhiau V., Rakotobe S., Lecuyer E., Mulder I., Lan A., Bridonneau C., Rochet V., Pisi A., De Paepe M., Brandi G., et al. The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity. 2009;31:677–689. doi: 10.1016/j.immuni.2009.08.020. [DOI] [PubMed] [Google Scholar]
- 180.Ivanov I.I., Atarashi K., Manel N., Brodie E.L., Shima T., Karaoz U., Wei D., Goldfarb K.C., Santee C.A., Lynch S.V., et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139:485–498. doi: 10.1016/j.cell.2009.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Sharma A., Rudra D. Emerging functions of regulatory T cells in tissue homeostasis. Front. Immunol. 2018;9:883. doi: 10.3389/fimmu.2018.00883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Wojno E.D.T., Artis D. Emerging concepts and future challenges in innate lymphoid cell biology. J. Exp. Med. 2016;213:2229–2248. doi: 10.1084/jem.20160525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Gautreaux M.D., Gelder F.B., Deitch E.A., Berg R.D. Adoptive transfer of T lymphocytes to T-cell-depleted mice inhibits Escherichia coli translocation from the gastrointestinal tract. Infect. Immun. 1995;63:3827–3834. doi: 10.1128/IAI.63.10.3827-3834.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Gautreaux M.D., Deitch E.A., Berg R.D. T lymphocytes in host defense against bacterial translocation from the gastrointestinal tract. Infect. Immun. 1994;62:2874–2884. doi: 10.1128/IAI.62.7.2874-2884.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Spadoni I., Zagato E., Bertocchi A., Paolinelli R., Hot E., Di Sabatino A., Caprioli F., Bottiglieri L., Oldani A., Viale G., et al. A gut-vascular barrier controls the systemic dissemination of bacteria. Science. 2015;350:830–834. doi: 10.1126/science.aad0135. [DOI] [PubMed] [Google Scholar]
- 186.Spadoni I., Fornasa G., Rescigno M. Organ-specific protection mediated by cooperation between vascular and epithelial barriers. Nat. Rev. Immunol. 2017;17:761–773. doi: 10.1038/nri.2017.100. [DOI] [PubMed] [Google Scholar]
- 187.Cornet A., Savidge T.C., Cabarrocas J., Deng W.L., Colombel J.F., Lassmann H., Desreumaux P., Liblau R.S. Enterocolitis induced by autoimmune targeting of enteric glial cells: A possible mechanism in Crohn’s disease? Proc. Natl. Acad. Sci. USA. 2001;98:13306–13311. doi: 10.1073/pnas.231474098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Ciccia F., Guggino G., Rizzo A., Alessandro R., Luchetti M.M., Milling S., Saieva L., Cypers H., Stampone T., Di Benedetto P., et al. Dysbiosis and zonulin upregulation alter gut epithelial and vascular barriers in patients with ankylosing spondylitis. Ann. Rheum. Dis. 2017;76:1123–1132. doi: 10.1136/annrheumdis-2016-210000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Macpherson A.J., Harris N.L. Interactions between commensal intestinal bacteria and the immune system. Nat. Rev. Immunol. 2004;4:478–485. doi: 10.1038/nri1373. [DOI] [PubMed] [Google Scholar]
- 190.Maynard C.L., Elson C.O., Hatton R.D., Weaver C.T. Reciprocal interactions of the intestinal microbiota and immune system. Nature. 2012;489:231–241. doi: 10.1038/nature11551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Macpherson A.J., Uhr T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science. 2004;303:1662–1665. doi: 10.1126/science.1091334. [DOI] [PubMed] [Google Scholar]
- 192.Macpherson A.J., Gatto D., Sainsbury E., Harriman G.R., Hengartner H., Zinkernagel R.M. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science. 2000;288:2222–2226. doi: 10.1126/science.288.5474.2222. [DOI] [PubMed] [Google Scholar]
- 193.Brun P., Castagliuolo I., Di Leo V., Buda A., Pinzani M., Palu G., Martines D. Increased intestinal permeability in obese mice: New evidence in the pathogenesis of nonalcoholic steatohepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007;292:G518–G525. doi: 10.1152/ajpgi.00024.2006. [DOI] [PubMed] [Google Scholar]
- 194.Etienne-Mesmin L., Vijay-Kumar M., Gewirtz A.T., Chassaing B. Hepatocyte Toll-Like Receptor 5 Promotes Bacterial Clearance and Protects Mice Against High-Fat Diet-Induced Liver Disease. Cell Mol. Gastroenterol. Hepatol. 2016;2:584–604. doi: 10.1016/j.jcmgh.2016.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Balmer M.L., Slack E., de Gottardi A., Lawson M.A., Hapfelmeier S., Miele L., Grieco A., Van Vlierberghe H., Fahrner R., Patuto N., et al. The liver may act as a firewall mediating mutualism between the host and its gut commensal microbiota. Sci. Transl. Med. 2014;6:237ra266. doi: 10.1126/scitranslmed.3008618. [DOI] [PubMed] [Google Scholar]
- 196.Wood N.J. Liver: The liver as a firewall—Clearance of commensal bacteria that have escaped from the gut. Nat. Rev. Gastroenterol. Hepatol. 2014;11:391. doi: 10.1038/nrgastro.2014.90. [DOI] [PubMed] [Google Scholar]
- 197.Lee W.Y., Moriarty T.J., Wong C.H., Zhou H., Strieter R.M., van Rooijen N., Chaconas G., Kubes P. An intravascular immune response to Borrelia burgdorferi involves Kupffer cells and iNKT cells. Nat. Immunol. 2010;11:295–302. doi: 10.1038/ni.1855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Knook D.L., Barkway C., Sleyster E.C. Lysosomal enzyme content of Kupffer and endothelial liver cells isolated from germfree and clean conventional rats. Infect. Immun. 1981;33:620–622. doi: 10.1128/IAI.33.2.620-622.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Schwabe R.F., Seki E., Brenner D.A. Toll-like receptor signaling in the liver. Gastroenterology. 2006;130:1886–1900. doi: 10.1053/j.gastro.2006.01.038. [DOI] [PubMed] [Google Scholar]
- 200.Fox E.S., Thomas P., Broitman S.A. Clearance of gut-derived endotoxins by the liver. Release and modification of 3H, 14C-lipopolysaccharide by isolated rat Kupffer cells. Gastroenterology. 1989;96:456–461. doi: 10.1016/0016-5085(89)91571-0. [DOI] [PubMed] [Google Scholar]
- 201.Su G.L., Klein R.D., Aminlari A., Zhang H.Y., Steinstraesser L., Alarcon W.H., Remick D.G., Wang S.C. Kupffer cell activation by lipopolysaccharide in rats: Role for lipopolysaccharide binding protein and toll-like receptor 4. Hepatology. 2000;31:932–936. doi: 10.1053/he.2000.5634. [DOI] [PubMed] [Google Scholar]
- 202.Schumann R.R., Kirschning C.J., Unbehaun A., Aberle H.P., Knope H.P., Lamping N., Ulevitch R.J., Herrmann F. The lipopolysaccharide-binding protein is a secretory class 1 acute-phase protein whose gene is transcriptionally activated by APRF/STAT/3 and other cytokine-inducible nuclear proteins. Mol. Cell. Biol. 1996;16:3490–3503. doi: 10.1128/MCB.16.7.3490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Pugin J., Schurer-Maly C.C., Leturcq D., Moriarty A., Ulevitch R.J., Tobias P.S. Lipopolysaccharide activation of human endothelial and epithelial cells is mediated by lipopolysaccharide-binding protein and soluble CD14. Proc. Natl. Acad. Sci. USA. 1993;90:2744–2748. doi: 10.1073/pnas.90.7.2744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Landmann R., Knopf H.P., Link S., Sansano S., Schumann R., Zimmerli W. Human monocyte CD14 is upregulated by lipopolysaccharide. Infect. Immun. 1996;64:1762–1769. doi: 10.1128/IAI.64.5.1762-1769.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Frey E.A., Miller D.S., Jahr T.G., Sundan A., Bazil V., Espevik T., Finlay B.B., Wright S.D. Soluble CD14 participates in the response of cells to lipopolysaccharide. J. Exp. Med. 1992;176:1665–1671. doi: 10.1084/jem.176.6.1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Lin R.S., Lee F.Y., Lee S.D., Tsai Y.T., Lin H.C., Lu R.H., Hsu W.C., Huang C.C., Wang S.S., Lo K.J. Endotoxemia in patients with chronic liver diseases: Relationship to severity of liver diseases, presence of esophageal varices, and hyperdynamic circulation. J. Hepatol. 1995;22:165–172. doi: 10.1016/0168-8278(95)80424-2. [DOI] [PubMed] [Google Scholar]
- 207.Garcia-Tsao G., Lee F.Y., Barden G.E., Cartun R., West A.B. Bacterial translocation to mesenteric lymph nodes is increased in cirrhotic rats with ascites. Gastroenterology. 1995;108:1835–1841. doi: 10.1016/0016-5085(95)90147-7. [DOI] [PubMed] [Google Scholar]
- 208.Cirera I., Bauer T.M., Navasa M., Vila J., Grande L., Taura P., Fuster J., Garcia-Valdecasas J.C., Lacy A., Suarez M.J., et al. Bacterial translocation of enteric organisms in patients with cirrhosis. J. Hepatol. 2001;34:32–37. doi: 10.1016/S0168-8278(00)00013-1. [DOI] [PubMed] [Google Scholar]
- 209.Bellot P., Garcia-Pagan J.C., Frances R., Abraldes J.G., Navasa M., Perez-Mateo M., Such J., Bosch J. Bacterial DNA translocation is associated with systemic circulatory abnormalities and intrahepatic endothelial dysfunction in patients with cirrhosis. Hepatology. 2010;52:2044–2052. doi: 10.1002/hep.23918. [DOI] [PubMed] [Google Scholar]
- 210.Ramadori G., Moriconi F., Malik I., Dudas J. Physiology and pathophysiology of liver inflammation, damage and repair. J. Physiol. Pharm. 2008;59(Suppl. 1):107–117. [PubMed] [Google Scholar]
- 211.Kudo H., Takahara T., Yata Y., Kawai K., Zhang W., Sugiyama T. Lipopolysaccharide triggered TNF-alpha-induced hepatocyte apoptosis in a murine non-alcoholic steatohepatitis model. J. Hepatol. 2009;51:168–175. doi: 10.1016/j.jhep.2009.02.032. [DOI] [PubMed] [Google Scholar]
- 212.Heymann F., Tacke F. Immunology in the liver--from homeostasis to disease. Nat. Rev. Gastroenterol. Hepatol. 2016;13:88–110. doi: 10.1038/nrgastro.2015.200. [DOI] [PubMed] [Google Scholar]
- 213.Brenner C., Galluzzi L., Kepp O., Kroemer G. Decoding cell death signals in liver inflammation. J. Hepatol. 2013;59:583–594. doi: 10.1016/j.jhep.2013.03.033. [DOI] [PubMed] [Google Scholar]
- 214.Tilg H., Moschen A.R., Szabo G. Interleukin-1 and inflammasomes in alcoholic liver disease/acute alcoholic hepatitis and nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology. 2016;64:955–965. doi: 10.1002/hep.28456. [DOI] [PubMed] [Google Scholar]
- 215.Wenfeng Z., Yakun W., Di M., Jianping G., Chuanxin W., Chun H. Kupffer cells: Increasingly significant role in nonalcoholic fatty liver disease. Ann. Hepatol. 2014;13:489–495. doi: 10.1016/S1665-2681(19)31247-5. [DOI] [PubMed] [Google Scholar]
- 216.Duffield J.S., Forbes S.J., Constandinou C.M., Clay S., Partolina M., Vuthoori S., Wu S., Lang R., Iredale J.P. Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J. Clin. Investig. 2005;115:56–65. doi: 10.1172/JCI200522675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Seki E., De Minicis S., Osterreicher C.H., Kluwe J., Osawa Y., Brenner D.A., Schwabe R.F. TLR4 enhances TGF-beta signaling and hepatic fibrosis. Nat. Med. 2007;13:1324–1332. doi: 10.1038/nm1663. [DOI] [PubMed] [Google Scholar]
- 218.Seki E., Schnabl B. Role of innate immunity and the microbiota in liver fibrosis: Crosstalk between the liver and gut. J. Physiol. 2012;590:447–458. doi: 10.1113/jphysiol.2011.219691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Wree A., Broderick L., Canbay A., Hoffman H.M., Feldstein A.E. From NAFLD to NASH to cirrhosis-new insights into disease mechanisms. Nat. Rev. Gastroenterol. Hepatol. 2013;10:627–636. doi: 10.1038/nrgastro.2013.149. [DOI] [PubMed] [Google Scholar]
- 220.Tsuchida T., Friedman S.L. Mechanisms of hepatic stellate cell activation. Nat. Rev. Gastroenterol. Hepatol. 2017;14:397–411. doi: 10.1038/nrgastro.2017.38. [DOI] [PubMed] [Google Scholar]
- 221.Roderfeld M. Matrix metalloproteinase functions in hepatic injury and fibrosis. Matrix Biol. J. Int. Soc. Matrix Biol. 2018;68–69:452–462. doi: 10.1016/j.matbio.2017.11.011. [DOI] [PubMed] [Google Scholar]
- 222.Benyon R.C., Arthur M.J. Extracellular matrix degradation and the role of hepatic stellate cells. Semin. Liver Dis. 2001;21:373–384. doi: 10.1055/s-2001-17552. [DOI] [PubMed] [Google Scholar]
- 223.Schuppan D., Ruehl M., Somasundaram R., Hahn E.G. Matrix as a modulator of hepatic fibrogenesis. Semin. Liver Dis. 2001;21:351–372. doi: 10.1055/s-2001-17556. [DOI] [PubMed] [Google Scholar]
- 224.Knittel T., Mehde M., Kobold D., Saile B., Dinter C., Ramadori G. Expression patterns of matrix metalloproteinases and their inhibitors in parenchymal and non-parenchymal cells of rat liver: Regulation by TNF-alpha and TGF-beta1. J. Hepatol. 1999;30:48–60. doi: 10.1016/S0168-8278(99)80007-5. [DOI] [PubMed] [Google Scholar]
- 225.Miele L., Forgione A., La Torre G., Vero V., Cefalo C., Racco S., Vellone V.G., Vecchio F.M., Gasbarrini G., Rapaccini G.L., et al. Serum levels of hyaluronic acid and tissue metalloproteinase inhibitor-1 combined with age predict the presence of nonalcoholic steatohepatitis in a pilot cohort of subjects with nonalcoholic fatty liver disease. Transl. Res. J. Lab. Clin. Med. 2009;154:194–201. doi: 10.1016/j.trsl.2009.06.007. [DOI] [PubMed] [Google Scholar]
- 226.Cichoz-Lach H., Michalak A. Oxidative stress as a crucial factor in liver diseases. World J. Gastroenterol. WJG. 2014;20:8082–8091. doi: 10.3748/wjg.v20.i25.8082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Grattagliano I., Caraceni P., Calamita G., Ferri D., Gargano I., Palasciano G., Portincasa P. Severe liver steatosis correlates with nitrosative and oxidative stress in rats. Eur. J. Clin. Investig. 2008;38:523–530. doi: 10.1111/j.1365-2362.2008.01963.x. [DOI] [PubMed] [Google Scholar]
- 228.Luangmonkong T., Suriguga S., Mutsaers H.A.M., Groothuis G.M.M., Olinga P., Boersema M. Targeting oxidative stress for the treatment of liver fibrosis. Rev. Physiol. Biochem. Pharmacol. 2018;175:71–102. doi: 10.1007/112_2018_10. [DOI] [PubMed] [Google Scholar]
- 229.Li S., Tan H.Y., Wang N., Zhang Z.J., Lao L., Wong C.W., Feng Y. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015;16:26087–26124. doi: 10.3390/ijms161125942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Gil-Cardoso K., Gines I., Pinent M., Ardevol A., Terra X., Blay M. A cafeteria diet triggers intestinal inflammation and oxidative stress in obese rats. Br. J. Nutr. 2017;117:218–229. doi: 10.1017/S0007114516004608. [DOI] [PubMed] [Google Scholar]
- 231.Keshavarzian A., Farhadi A., Forsyth C.B., Rangan J., Jakate S., Shaikh M., Banan A., Fields J.Z. Evidence that chronic alcohol exposure promotes intestinal oxidative stress, intestinal hyperpermeability and endotoxemia prior to development of alcoholic steatohepatitis in rats. J. Hepatol. 2009;50:538–547. doi: 10.1016/j.jhep.2008.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Van Ampting M.T., Schonewille A.J., Vink C., Brummer R.J., van der Meer R., Bovee-Oudenhoven I.M. Intestinal barrier function in response to abundant or depleted mucosal glutathione in Salmonella-infected rats. BMC Physiol. 2009;9:6. doi: 10.1186/1472-6793-9-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Novak E.A., Mollen K.P. Mitochondrial dysfunction in inflammatory bowel disease. Front. Cell Dev. Biol. 2015;3:62. doi: 10.3389/fcell.2015.00062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Utzeri E., Usai P. Role of non-steroidal anti-inflammatory drugs on intestinal permeability and nonalcoholic fatty liver disease. World J. Gastroenterol. WJG. 2017;23:3954–3963. doi: 10.3748/wjg.v23.i22.3954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Ramachandran A., Prabhu R., Thomas S., Reddy J.B., Pulimood A., Balasubramanian K.A. Intestinal mucosal alterations in experimental cirrhosis in the rat: Role of oxygen free radicals. Hepatology. 2002;35:622–629. doi: 10.1053/jhep.2002.31656. [DOI] [PubMed] [Google Scholar]
- 236.Liang S., Kisseleva T., Brenner D.A. The Role of NADPH Oxidases (NOXs) in Liver Fibrosis and the Activation of Myofibroblasts. Front. Physiol. 2016;7:17. doi: 10.3389/fphys.2016.00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Nieto N. Oxidative-stress and IL-6 mediate the fibrogenic effects of [corrected] Kupffer cells on stellate cells. Hepatology. 2006;44:1487–1501. doi: 10.1002/hep.21427. [DOI] [PubMed] [Google Scholar]
- 238.Krause P., Morris V., Greenbaum J.A., Park Y., Bjoerheden U., Mikulski Z., Muffley T., Shui J.W., Kim G., Cheroutre H., et al. IL-10-producing intestinal macrophages prevent excessive antibacterial innate immunity by limiting IL-23 synthesis. Nat. Commun. 2015;6:7055. doi: 10.1038/ncomms8055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Gomez-Hurtado I., Moratalla A., Moya-Perez A., Peiro G., Zapater P., Gonzalez-Navajas J.M., Gimenez P., Such J., Sanz Y., Frances R. Role of interleukin 10 in norfloxacin prevention of luminal free endotoxin translocation in mice with cirrhosis. J. Hepatol. 2014;61:799–808. doi: 10.1016/j.jhep.2014.05.031. [DOI] [PubMed] [Google Scholar]
- 240.Thompson K., Maltby J., Fallowfield J., McAulay M., Millward-Sadler H., Sheron N. Interleukin-10 expression and function in experimental murine liver inflammation and fibrosis. Hepatology. 1998;28:1597–1606. doi: 10.1002/hep.510280620. [DOI] [PubMed] [Google Scholar]
- 241.De Souza-Cruz S., Victoria M.B., Tarrago A.M., da Costa A.G., Pimentel J.P., Pires E.F., Araujo Lde P., Coelho-dos-Reis J.G., Gomes Mde S., Amaral L.R., et al. Liver and blood cytokine microenvironment in HCV patients is associated to liver fibrosis score: A proinflammatory cytokine ensemble orchestrated by TNF and tuned by IL-10. BMC Microbiol. 2016;16:3. doi: 10.1186/s12866-015-0610-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Melhem A., Muhanna N., Bishara A., Alvarez C.E., Ilan Y., Bishara T., Horani A., Nassar M., Friedman S.L., Safadi R. Anti-fibrotic activity of NK cells in experimental liver injury through killing of activated HSC. J. Hepatol. 2006;45:60–71. doi: 10.1016/j.jhep.2005.12.025. [DOI] [PubMed] [Google Scholar]
- 243.Krizhanovsky V., Yon M., Dickins R.A., Hearn S., Simon J., Miething C., Yee H., Zender L., Lowe S.W. Senescence of activated stellate cells limits liver fibrosis. Cell. 2008;134:657–667. doi: 10.1016/j.cell.2008.06.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Ley R.E., Turnbaugh P.J., Klein S., Gordon J.I. Microbial ecology: Human gut microbes associated with obesity. Nature. 2006;444:1022–1023. doi: 10.1038/4441022a. [DOI] [PubMed] [Google Scholar]
- 245.Serino M., Luche E., Chabo C., Amar J., Burcelin R. Intestinal microflora and metabolic diseases. Diabetes Metab. 2009;35:262–272. doi: 10.1016/j.diabet.2009.03.003. [DOI] [PubMed] [Google Scholar]
- 246.Serino M., Luche E., Gres S., Baylac A., Berge M., Cenac C., Waget A., Klopp P., Iacovoni J., Klopp C., et al. Metabolic adaptation to a high-fat diet is associated with a change in the gut microbiota. Gut. 2012;61:543–553. doi: 10.1136/gutjnl-2011-301012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Boursier J., Mueller O., Barret M., Machado M., Fizanne L., Araujo-Perez F., Guy C.D., Seed P.C., Rawls J.F., David L.A., et al. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology. 2016;63:764–775. doi: 10.1002/hep.28356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Leung C., Rivera L., Furness J.B., Angus P.W. The role of the gut microbiota in NAFLD. Nat. Rev. Gastroenterol. Hepatol. 2016;13:412–425. doi: 10.1038/nrgastro.2016.85. [DOI] [PubMed] [Google Scholar]
- 249.Minemura M., Shimizu Y. Gut microbiota and liver diseases. World J. Gastroenterol. WJG. 2015;21:1691–1702. doi: 10.3748/wjg.v21.i6.1691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Rahman K., Desai C., Iyer S.S., Thorn N.E., Kumar P., Liu Y., Smith T., Neish A.S., Li H., Tan S., et al. Loss of junctional adhesion molecule a promotes severe steatohepatitis in mice on a diet high in saturated fat, fructose, and cholesterol. Gastroenterology. 2016;151:733–746.e12. doi: 10.1053/j.gastro.2016.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Pappo I., Bercovier H., Berry E., Gallilly R., Feigin E., Freund H.R. Antitumor necrosis factor antibodies reduce hepatic steatosis during total parenteral nutrition and bowel rest in the rat. Jpn. J. Parenter. Enter. Nutr. 1995;19:80–82. doi: 10.1177/014860719501900180. [DOI] [PubMed] [Google Scholar]
- 252.Kirsch R., Clarkson V., Verdonk R.C., Marais A.D., Shephard E.G., Ryffel B., de la M Hall P. Rodent nutritional model of steatohepatitis: Effects of endotoxin (lipopolysaccharide) and tumor necrosis factor alpha deficiency. J. Gastroenterol. Hepatol. 2006;21:174–182. doi: 10.1111/j.1440-1746.2005.04220.x. [DOI] [PubMed] [Google Scholar]
- 253.Jin X., Yu C.H., Lv G.C., Li Y.M. Increased intestinal permeability in pathogenesis and progress of nonalcoholic steatohepatitis in rats. World J. Gastroenterol. WJG. 2007;13:1732–1736. doi: 10.3748/wjg.v13.i11.1732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Imajo K., Fujita K., Yoneda M., Nozaki Y., Ogawa Y., Shinohara Y., Kato S., Mawatari H., Shibata W., Kitani H., et al. Hyperresponsivity to low-dose endotoxin during progression to nonalcoholic steatohepatitis is regulated by leptin-mediated signaling. Cell Metab. 2012;16:44–54. doi: 10.1016/j.cmet.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 255.Henao-Mejia J., Elinav E., Jin C., Hao L., Mehal W.Z., Strowig T., Thaiss C.A., Kau A.L., Eisenbarth S.C., Jurczak M.J., et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature. 2012;482:179–185. doi: 10.1038/nature10809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Giorgio V., Miele L., Principessa L., Ferretti F., Villa M.P., Negro V., Grieco A., Alisi A., Nobili V. Intestinal permeability is increased in children with non-alcoholic fatty liver disease, and correlates with liver disease severity. Dig. Liver Dis. 2014;46:556–560. doi: 10.1016/j.dld.2014.02.010. [DOI] [PubMed] [Google Scholar]
- 257.Wigg A.J., Roberts-Thomson I.C., Dymock R.B., McCarthy P.J., Grose R.H., Cummins A.G. The role of small intestinal bacterial overgrowth, intestinal permeability, endotoxaemia, and tumour necrosis factor alpha in the pathogenesis of non-alcoholic steatohepatitis. Gut. 2001;48:206–211. doi: 10.1136/gut.48.2.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Cani P.D., Amar J., Iglesias M.A., Poggi M., Knauf C., Bastelica D., Neyrinck A.M., Fava F., Tuohy K.M., Chabo C., et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761–1772. doi: 10.2337/db06-1491. [DOI] [PubMed] [Google Scholar]
- 259.Gasbarrini A., Corazza G.R., Gasbarrini G., Montalto M., Di Stefano M., Basilisco G., Parodi A., Usai-Satta P., Vernia P., Anania C., et al. Methodology and indications of H2-breath testing in gastrointestinal diseases: The Rome Consensus Conference. Aliment. Pharmacol. Ther. 2009;29(Suppl. 1):1–49. doi: 10.1111/j.1365-2036.2009.03951.x. [DOI] [PubMed] [Google Scholar]
- 260.Gasbarrini A., Lauritano E.C., Gabrielli M., Scarpellini E., Lupascu A., Ojetti V., Gasbarrini G. Small intestinal bacterial overgrowth: Diagnosis and treatment. Dig. Dis. 2007;25:237–240. doi: 10.1159/000103892. [DOI] [PubMed] [Google Scholar]
- 261.Loomba R., Seguritan V., Li W., Long T., Klitgord N., Bhatt A., Dulai P.S., Caussy C., Bettencourt R., Highlander S.K., et al. Gut Microbiome-Based Metagenomic Signature for Non-invasive Detection of Advanced Fibrosis in Human Nonalcoholic Fatty Liver Disease. Cell Metab. 2017;25:1054–1062.e5. doi: 10.1016/j.cmet.2017.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Zhu L., Baker S.S., Gill C., Liu W., Alkhouri R., Baker R.D., Gill S.R. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: A connection between endogenous alcohol and NASH. Hepatology. 2013;57:601–609. doi: 10.1002/hep.26093. [DOI] [PubMed] [Google Scholar]
- 263.De Wit N.J., Afman L.A., Mensink M., Muller M. Phenotyping the effect of diet on non-alcoholic fatty liver disease. J. Hepatol. 2012;57:1370–1373. doi: 10.1016/j.jhep.2012.07.003. [DOI] [PubMed] [Google Scholar]
- 264.O’Sullivan A., He X., McNiven E.M., Haggarty N.W., Lonnerdal B., Slupsky C.M. Early diet impacts infant rhesus gut microbiome, immunity, and metabolism. J. Proteome Res. 2013;12:2833–2845. doi: 10.1021/pr4001702. [DOI] [PubMed] [Google Scholar]
- 265.Lelouvier B., Servant F., Paisse S., Brunet A.C., Benyahya S., Serino M., Valle C., Ortiz M.R., Puig J., Courtney M., et al. Changes in blood microbiota profiles associated with liver fibrosis in obese patients: A pilot analysis. Hepatology. 2016;64:2015–2027. doi: 10.1002/hep.28829. [DOI] [PubMed] [Google Scholar]
- 266.Amar J., Lange C., Payros G., Garret C., Chabo C., Lantieri O., Courtney M., Marre M., Charles M.A., Balkau B., et al. Blood microbiota dysbiosis is associated with the onset of cardiovascular events in a large general population: The D.E.S.I.R. study. PLoS ONE. 2013;8:e54461. doi: 10.1371/journal.pone.0054461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Amar J., Serino M., Lange C., Chabo C., Iacovoni J., Mondot S., Lepage P., Klopp C., Mariette J., Bouchez O., et al. Involvement of tissue bacteria in the onset of diabetes in humans: Evidence for a concept. Diabetologia. 2011;54:3055–3061. doi: 10.1007/s00125-011-2329-8. [DOI] [PubMed] [Google Scholar]
- 268.Raman M., Ahmed I., Gillevet P.M., Probert C.S., Ratcliffe N.M., Smith S., Greenwood R., Sikaroodi M., Lam V., Crotty P., et al. Fecal microbiome and volatile organic compound metabolome in obese humans with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2013;11:868–875.e3. doi: 10.1016/j.cgh.2013.02.015. [DOI] [PubMed] [Google Scholar]
- 269.Caussy C., Hsu C., Lo M.T., Liu A., Bettencourt R., Ajmera V.H., Bassirian S., Hooker J., Sy E., Richards L., et al. Link between gut-microbiome derived metabolite and shared gene-effects with hepatic steatosis and fibrosis in NAFLD. Hepatology. 2018 doi: 10.1002/hep.29892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Di Ciaula A., Wang D.Q., Molina-Molina E., Lunardi Baccetto R., Calamita G., Palmieri V.O., Portincasa P. Bile acids and cancer: Direct and environmental-dependent effects. Ann. Hepatol. 2017;16:S87–S105. doi: 10.5604/01.3001.0010.5501. [DOI] [PubMed] [Google Scholar]
- 271.Grattagliano I., Diogo C.V., Mastrodonato M., de Bari O., Persichella M., Wang D.Q., Liquori A., Ferri D., Carratu M.R., Oliveira P.J., et al. A silybin-phospholipids complex counteracts rat fatty liver degeneration and mitochondrial oxidative changes. World J. Gastroenterol. WJG. 2013;19:3007–3017. doi: 10.3748/wjg.v19.i20.3007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Mastrodonato M., Calamita G., Rossi R., Mentino D., Bonfrate L., Portincasa P., Ferri D., Liquori G.E. Altered distribution of caveolin-1 in early liver steatosis. Eur. J. Clin. Investig. 2011;41:642–651. doi: 10.1111/j.1365-2362.2010.02459.x. [DOI] [PubMed] [Google Scholar]
- 273.Pacelli C., Coluccia A., Grattagliano I., Cocco T., Petrosillo G., Paradies G., De Nitto E., Massaro A., Persichella M., Borracci P., et al. Dietary choline deprivation impairs rat brain mitochondrial function and behavioral phenotype. J. Nutr. 2010;140:1072–1079. doi: 10.3945/jn.109.116673. [DOI] [PubMed] [Google Scholar]
- 274.Petrosillo G., Portincasa P., Grattagliano I., Casanova G., Matera M., Ruggiero F.M., Ferri D., Paradies G. Mitochondrial dysfunction in rat with nonalcoholic fatty liver Involvement of complex I, reactive oxygen species and cardiolipin. Biochim. Biophys. Acta. 2007;1767:1260–1267. doi: 10.1016/j.bbabio.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 275.Ponziani F.R., Bhoori S., Castelli C., Putignani L., Rivoltini L., Del Chierico F., Sanguinetti M., Morelli D., Sterbini F.P., Petito V., et al. Hepatocellular carcinoma is associated with gut microbiota profile and inflammation in nonalcoholic fatty liver disease. Hepatology. 2019;69:107–120. doi: 10.1002/hep.30036. [DOI] [PubMed] [Google Scholar]
- 276.Philips C.A., Pande A., Shasthry S.M., Jamwal K.D., Khillan V., Chandel S.S., Kumar G., Sharma M.K., Maiwall R., Jindal A. A pilot study. Clin. Gastroenterol. Hepatol. 2017;15:600–602. doi: 10.1016/j.cgh.2016.10.029. [DOI] [PubMed] [Google Scholar]
- 277.Luther J., Garber J.J., Khalili H., Dave M., Bale S.S., Jindal R., Motola D.L., Luther S., Bohr S., Jeoung S.W., et al. Hepatic injury in nonalcoholic steatohepatitis contributes to altered intestinal permeability. Cell Mol. Gastroenterol. Hepatol. 2015;1:222–232. doi: 10.1016/j.jcmgh.2015.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Nazim M., Stamp G., Hodgson H.J. Non-alcoholic steatohepatitis associated with small intestinal diverticulosis and bacterial overgrowth. Hepatogastroenterology. 1989;36:349–351. [PubMed] [Google Scholar]
- 279.Lichtman S.N., Sartor R.B., Keku J., Schwab J.H. Hepatic inflammation in rats with experimental small intestinal bacterial overgrowth. Gastroenterology. 1990;98:414–423. doi: 10.1016/0016-5085(90)90833-M. [DOI] [PubMed] [Google Scholar]
- 280.Lichtman S.N., Keku J., Schwab J.H., Sartor R.B. Hepatic injury associated with small bowel bacterial overgrowth in rats is prevented by metronidazole and tetracycline. Gastroenterology. 1991;100:513–519. doi: 10.1016/0016-5085(91)90224-9. [DOI] [PubMed] [Google Scholar]
- 281.Kapil S., Duseja A., Sharma B.K., Singla B., Chakraborti A., Das A., Ray P., Dhiman R.K., Chawla Y. Small intestinal bacterial overgrowth and toll-like receptor signaling in patients with non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 2016;31:213–221. doi: 10.1111/jgh.13058. [DOI] [PubMed] [Google Scholar]
- 282.Diehl A.M., Li Z.P., Lin H.Z., Yang S.Q. Cytokines and the pathogenesis of non-alcoholic steatohepatitis. Gut. 2005;54:303–306. doi: 10.1136/gut.2003.024935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Vetrano S., Rescigno M., Cera M.R., Correale C., Rumio C., Doni A., Fantini M., Sturm A., Borroni E., Repici A., et al. Unique role of junctional adhesion molecule-a in maintaining mucosal homeostasis in inflammatory bowel disease. Gastroenterology. 2008;135:173–184. doi: 10.1053/j.gastro.2008.04.002. [DOI] [PubMed] [Google Scholar]
- 284.Monteiro A.C., Sumagin R., Rankin C.R., Leoni G., Mina M.J., Reiter D.M., Stehle T., Dermody T.S., Schaefer S.A., Hall R.A., et al. JAM-A associates with ZO-2, afadin, and PDZ-GEF1 to activate Rap2c and regulate epithelial barrier function. Mol. Biol. Cell. 2013;24:2849–2860. doi: 10.1091/mbc.e13-06-0298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Menard S., Cerf-Bensussan N., Heyman M. Multiple facets of intestinal permeability and epithelial handling of dietary antigens. Mucosal. Immunol. 2010;3:247–259. doi: 10.1038/mi.2010.5. [DOI] [PubMed] [Google Scholar]
- 286.Laukoetter M.G., Nava P., Lee W.Y., Severson E.A., Capaldo C.T., Babbin B.A., Williams I.R., Koval M., Peatman E., Campbell J.A., et al. JAM-A regulates permeability and inflammation in the intestine in vivo. J. Exp. Med. 2007;204:3067–3076. doi: 10.1084/jem.20071416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Cheng C., Tan J., Qian W., Zhang L., Hou X. Gut inflammation exacerbates hepatic injury in the high-fat diet induced NAFLD mouse: Attention to the gut-vascular barrier dysfunction. Life Sci. 2018;209:157–166. doi: 10.1016/j.lfs.2018.08.017. [DOI] [PubMed] [Google Scholar]
- 288.DeMeo M.T., Mutlu E.A., Keshavarzian A., Tobin M.C. Intestinal permeation and gastrointestinal disease. J. Clin. Gastroenterol. 2002;34:385–396. doi: 10.1097/00004836-200204000-00003. [DOI] [PubMed] [Google Scholar]
- 289.Arslan G., Atasever T., Cindoruk M., Yildirim I.S. (51)CrEDTA colonic permeability and therapy response in patients with ulcerative colitis. Nucl. Med. Commun. 2001;22:997–1001. doi: 10.1097/00006231-200109000-00009. [DOI] [PubMed] [Google Scholar]
- 290.Ponziani F.R., Zocco M.A., Cerrito L., Gasbarrini A., Pompili M. Bacterial translocation in patients with liver cirrhosis: Physiology, clinical consequences, and practical implications. Expert Rev. Gastroenterol. Hepatol. 2018;12:641–656. doi: 10.1080/17474124.2018.1481747. [DOI] [PubMed] [Google Scholar]
- 291.Gabele E., Muhlbauer M., Dorn C., Weiss T.S., Froh M., Schnabl B., Wiest R., Scholmerich J., Obermeier F., Hellerbrand C. Role of TLR9 in hepatic stellate cells and experimental liver fibrosis. Biochem. Biophys. Res. Commun. 2008;376:271–276. doi: 10.1016/j.bbrc.2008.08.096. [DOI] [PubMed] [Google Scholar]
- 292.Lebeaupin C., Proics E., de Bieville C.H., Rousseau D., Bonnafous S., Patouraux S., Adam G., Lavallard V.J., Rovere C., Le Thuc O., et al. ER stress induces NLRP3 inflammasome activation and hepatocyte death. Cell Death Dis. 2015;6:e1879. doi: 10.1038/cddis.2015.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Miura K., Kodama Y., Inokuchi S., Schnabl B., Aoyama T., Ohnishi H., Olefsky J.M., Brenner D.A., Seki E. Toll-like receptor 9 promotes steatohepatitis by induction of interleukin-1beta in mice. Gastroenterology. 2010;139:323–334.e7. doi: 10.1053/j.gastro.2010.03.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Saberi M., Woods N.B., de Luca C., Schenk S., Lu J.C., Bandyopadhyay G., Verma I.M., Olefsky J.M. Hematopoietic cell-specific deletion of toll-like receptor 4 ameliorates hepatic and adipose tissue insulin resistance in high-fat-fed mice. Cell Metab. 2009;10:419–429. doi: 10.1016/j.cmet.2009.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Rivera C.A., Adegboyega P., van Rooijen N., Tagalicud A., Allman M., Wallace M. Toll-like receptor-4 signaling and Kupffer cells play pivotal roles in the pathogenesis of non-alcoholic steatohepatitis. J. Hepatol. 2007;47:571–579. doi: 10.1016/j.jhep.2007.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Levitt M.D., Li R., Demaster E.G., Elson M., Furne J., Levitt D.G. Use of measurements of ethanol absorption from stomach and intestine to assess human ethanol metabolism. Am. J. Physiol. Gastrointest. Liver Physiol. 1997;273:G951–G957. doi: 10.1152/ajpgi.1997.273.4.G951. [DOI] [PubMed] [Google Scholar]
- 297.Chen P., Miyamoto Y., Mazagova M., Lee K.C., Eckmann L., Schnabl B. Microbiota Protects Mice Against Acute Alcohol-Induced Liver Injury. Alcohol. Clin. Exp. Res. 2015;39:2313–2323. doi: 10.1111/acer.12900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Ansari R.A., Husain K., Rizvi S.A. Role of Transcription Factors in Steatohepatitis and Hypertension after Ethanol: The Epicenter of Metabolism. Biomolecules. 2016;6:29. doi: 10.3390/biom6030029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Hamarneh S.R., Kim B.M., Kaliannan K., Morrison S.A., Tantillo T.J., Tao Q., Mohamed M.M.R., Ramirez J.M., Karas A., Liu W., et al. Intestinal alkaline phosphatase attenuates alcohol-induced hepatosteatosis in mice. Dig. Dis. Sci. 2017;62:2021–2034. doi: 10.1007/s10620-017-4576-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Kim D.H., Jeong D., Kang I.B., Kim H., Song K.Y., Seo K.H. Dual function of Lactobacillus kefiri DH5 in preventing high-fat-diet-induced obesity: Direct reduction of cholesterol and upregulation of PPAR-alpha in adipose tissue. Mol. Nutr. Food Res. 2017;61:1700252. doi: 10.1002/mnfr.201700252. [DOI] [PubMed] [Google Scholar]
- 301.Elamin E., Jonkers D., Juuti-Uusitalo K., van Ijzendoorn S., Troost F., Duimel H., Broers J., Verheyen F., Dekker J., Masclee A. Effects of ethanol and acetaldehyde on tight junction integrity: In vitro study in a three dimensional intestinal epithelial cell culture model. PLoS ONE. 2012;7:e35008. doi: 10.1371/journal.pone.0035008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Samak G., Aggarwal S., Rao R.K. ERK is involved in EGF-mediated protection of tight junctions, but not adherens junctions, in acetaldehyde-treated Caco-2 cell monolayers. Am. J. Physiol. Gastrointest. Liver Physiol. 2011;301:G50–G59. doi: 10.1152/ajpgi.00494.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Basuroy S., Sheth P., Mansbach C.M., Rao R.K. Acetaldehyde disrupts tight junctions and adherens junctions in human colonic mucosa: Protection by EGF and L-glutamine. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;289:G367–G375. doi: 10.1152/ajpgi.00464.2004. [DOI] [PubMed] [Google Scholar]
- 304.Yan A.W., Fouts D.E., Brandl J., Starkel P., Torralba M., Schott E., Tsukamoto H., Nelson K.E., Brenner D.A., Schnabl B. Enteric dysbiosis associated with a mouse model of alcoholic liver disease. Hepatology. 2011;53:96–105. doi: 10.1002/hep.24018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Hartmann P., Chen P., Wang H.J., Wang L., McCole D.F., Brandl K., Starkel P., Belzer C., Hellerbrand C., Tsukamoto H., et al. Deficiency of intestinal mucin-2 ameliorates experimental alcoholic liver disease in mice. Hepatology. 2013;58:108–119. doi: 10.1002/hep.26321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Park B., Lee H.R., Lee Y.J. Alcoholic liver disease: Focus on prodromal gut health. J. Dig. Dis. 2016;17:493–500. doi: 10.1111/1751-2980.12375. [DOI] [PubMed] [Google Scholar]
- 307.Wang H., Lafdil F., Kong X., Gao B. Signal transducer and activator of transcription 3 in liver diseases: A novel therapeutic target. Int. J. Biol. Sci. 2011;7:536–550. doi: 10.7150/ijbs.7.536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Mottaran E., Stewart S.F., Rolla R., Vay D., Cipriani V., Moretti M., Vidali M., Sartori M., Rigamonti C., Day C.P., et al. Lipid peroxidation contributes to immune reactions associated with alcoholic liver disease. Free Radic. Biol. Med. 2002;32:38–45. doi: 10.1016/S0891-5849(01)00757-2. [DOI] [PubMed] [Google Scholar]
- 309.Chen P., Torralba M., Tan J., Embree M., Zengler K., Starkel P., van Pijkeren J.P., DePew J., Loomba R., Ho S.B., et al. Supplementation of saturated long-chain fatty acids maintains intestinal eubiosis and reduces ethanol-induced liver injury in mice. Gastroenterology. 2015;148:203–214.e16. doi: 10.1053/j.gastro.2014.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Xie G., Zhong W., Zheng X., Li Q., Qiu Y., Li H., Chen H., Zhou Z., Jia W. Chronic ethanol consumption alters mammalian gastrointestinal content metabolites. J. Proteome Res. 2013;12:3297–3306. doi: 10.1021/pr400362z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Couch R.D., Dailey A., Zaidi F., Navarro K., Forsyth C.B., Mutlu E., Engen P.A., Keshavarzian A. Alcohol induced alterations to the human fecal VOC metabolome. PLoS ONE. 2015;10:e0119362. doi: 10.1371/journal.pone.0119362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Cresci G.A., Glueck B., McMullen M.R., Xin W., Allende D., Nagy L.E. Prophylactic tributyrin treatment mitigates chronic-binge ethanol-induced intestinal barrier and liver injury. J. Gastroenterol. Hepatol. 2017;32:1587–1597. doi: 10.1111/jgh.13731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Leclercq S., Matamoros S., Cani P.D., Neyrinck A.M., Jamar F., Starkel P., Windey K., Tremaroli V., Backhed F., Verbeke K., et al. Intestinal permeability, gut-bacterial dysbiosis, and behavioral markers of alcohol-dependence severity. Proc. Natl. Acad. Sci. USA. 2014;111:E4485–E4493. doi: 10.1073/pnas.1415174111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Arroyo V., Moreau R., Kamath P.S., Jalan R., Ginès P., Nevens F., Fernández J., To U., García-Tsao G., Schnabl B. Acute-on-chronic liver failure in cirrhosis. Nat. Rev. Dis. Primers. 2016;2:16041. doi: 10.1038/nrdp.2016.41. [DOI] [PubMed] [Google Scholar]
- 315.Cresci G.A., Bush K., Nagy L.E. Tributyrin supplementation protects mice from acute ethanol-induced gut injury. Alcohol. Clin. Exp. Res. 2014;38:1489–1501. doi: 10.1111/acer.12428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Mezey E., Imbembo A.L., Potter J.J., Rent K.C., Lombardo R., Holt P.R. Endogenous ethanol production and hepatic disease following jejunoileal bypass for morbid obesity. Am. J. Clin. Nutr. 1975;28:1277–1283. doi: 10.1093/ajcn/28.11.1277. [DOI] [PubMed] [Google Scholar]
- 317.Baraona E., Julkunen R., Tannenbaum L., Lieber C.S. Role of intestinal bacterial overgrowth in ethanol production and metabolism in rats. Gastroenterology. 1986;90:103–110. doi: 10.1016/0016-5085(86)90081-8. [DOI] [PubMed] [Google Scholar]
- 318.Kaji H., Asanuma Y., Yahara O., Shibue H., Hisamura M., Saito N., Kawakami Y., Murao M. Intragastrointestinal alcohol fermentation syndrome: Report of two cases and review of the literature. J. Forensic Sci. Soc. 1984;24:461–471. doi: 10.1016/S0015-7368(84)72325-5. [DOI] [PubMed] [Google Scholar]
- 319.Nair S., Cope K., Risby T.H., Diehl A.M. Obesity and female gender increase breath ethanol concentration: Potential implications for the pathogenesis of nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2001;96:1200–1204. doi: 10.1111/j.1572-0241.2001.03702.x. [DOI] [PubMed] [Google Scholar]
- 320.Cope K., Risby T., Diehl A.M. Increased gastrointestinal ethanol production in obese mice: Implications for fatty liver disease pathogenesis. Gastroenterology. 2000;119:1340–1347. doi: 10.1053/gast.2000.19267. [DOI] [PubMed] [Google Scholar]
- 321.Salaspuro M. Bacteriocolonic pathway for ethanol oxidation: Characteristics and implications. Ann. Med. 1996;28:195–200. doi: 10.3109/07853899609033120. [DOI] [PubMed] [Google Scholar]
- 322.Dawes E.A., Foster S.M. The formation of ethanol in Escherichia coli. Biochim. Biophys. Acta. 1956;22:253–265. doi: 10.1016/0006-3002(56)90148-2. [DOI] [PubMed] [Google Scholar]
- 323.Engstler A.J., Aumiller T., Degen C., Durr M., Weiss E., Maier I.B., Schattenberg J.M., Jin C.J., Sellmann C., Bergheim I. Insulin resistance alters hepatic ethanol metabolism: Studies in mice and children with non-alcoholic fatty liver disease. Gut. 2016;65:1564–1571. doi: 10.1136/gutjnl-2014-308379. [DOI] [PubMed] [Google Scholar]
- 324.Christopherson M.R., Dawson J.A., Stevenson D.M., Cunningham A.C., Bramhacharya S., Weimer P.J., Kendziorski C., Suen G. Unique aspects of fiber degradation by the ruminal ethanologen Ruminococcus albus 7 revealed by physiological and transcriptomic analysis. BMC Genom. 2014;15:1066. doi: 10.1186/1471-2164-15-1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Setshedi M., Wands J.R., Monte S.M. Acetaldehyde adducts in alcoholic liver disease. Oxidative Med. Cell. Longev. 2010;3:178–185. doi: 10.4161/oxim.3.3.12288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Ni Y.H., Huo L.J., Li T.T. Effect of interleukin-22 on proliferation and activation of hepatic stellate cells induced by acetaldehyde and related mechanism. Chin. J. Hepatol. 2017;25:9–14. doi: 10.3760/cma.j.issn.1007-3418.2017.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Wu X., Wang Y., Wang S., Xu R., Lv X. Purinergic P2X7 receptor mediates acetaldehyde-induced hepatic stellate cells activation via PKC-dependent GSK3beta pathway. Int. Immunopharmacol. 2017;43:164–171. doi: 10.1016/j.intimp.2016.12.017. [DOI] [PubMed] [Google Scholar]
- 328.López-Lázaro M. A local mechanism by which alcohol consumption causes cancer. Oral Oncol. 2016;62:149–152. doi: 10.1016/j.oraloncology.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 329.Baker S.S., Baker R.D., Liu W., Nowak N.J., Zhu L. Role of alcohol metabolism in non-alcoholic steatohepatitis. PLoS ONE. 2010;5:e9570. doi: 10.1371/journal.pone.0009570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 330.Ridlon J.M., Kang D.J., Hylemon P.B. Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 2006;47:241–259. doi: 10.1194/jlr.R500013-JLR200. [DOI] [PubMed] [Google Scholar]
- 331.Sayin S.I., Wahlstrom A., Felin J., Jantti S., Marschall H.U., Bamberg K., Angelin B., Hyotylainen T., Oresic M., Backhed F. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab. 2013;17:225–235. doi: 10.1016/j.cmet.2013.01.003. [DOI] [PubMed] [Google Scholar]
- 332.Yokota A., Fukiya S., Islam K.B., Ooka T., Ogura Y., Hayashi T., Hagio M., Ishizuka S. Is bile acid a determinant of the gut microbiota on a high-fat diet? Gut Microbes. 2012;3:455–459. doi: 10.4161/gmic.21216. [DOI] [PubMed] [Google Scholar]
- 333.Inagaki T., Moschetta A., Lee Y.K., Peng L., Zhao G., Downes M., Yu R.T., Shelton J.M., Richardson J.A., Repa J.J., et al. Regulation of antibacterial defense in the small intestine by the nuclear bile acid receptor. Proc. Natl. Acad. Sci. USA. 2006;103:3920–3925. doi: 10.1073/pnas.0509592103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Parseus A., Sommer N., Sommer F., Caesar R., Molinaro A., Stahlman M., Greiner T.U., Perkins R., Backhed F. Microbiota-induced obesity requires farnesoid X receptor. Gut. 2017;66:429–437. doi: 10.1136/gutjnl-2015-310283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Gadaleta R.M., van Erpecum K.J., Oldenburg B., Willemsen E.C., Renooij W., Murzilli S., Klomp L.W., Siersema P.D., Schipper M.E., Danese S., et al. Farnesoid X receptor activation inhibits inflammation and preserves the intestinal barrier in inflammatory bowel disease. Gut. 2011;60:463–472. doi: 10.1136/gut.2010.212159. [DOI] [PubMed] [Google Scholar]
- 336.Li F., Jiang C., Krausz K.W., Li Y., Albert I., Hao H., Fabre K.M., Mitchell J.B., Patterson A.D., Gonzalez F.J. Microbiome remodelling leads to inhibition of intestinal farnesoid X receptor signalling and decreased obesity. Nat. Commun. 2013;4:2384. doi: 10.1038/ncomms3384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Cao H., Xu M., Dong W., Deng B., Wang S., Zhang Y., Wang S., Luo S., Wang W., Qi Y., et al. Secondary bile acid-induced dysbiosis promotes intestinal carcinogenesis. Int. J. Cancer. 2017;140:2545–2556. doi: 10.1002/ijc.30643. [DOI] [PubMed] [Google Scholar]
- 338.Jiang C., Xie C., Li F., Zhang L., Nichols R.G., Krausz K.W., Cai J., Qi Y., Fang Z.Z., Takahashi S., et al. Intestinal farnesoid X receptor signaling promotes nonalcoholic fatty liver disease. J. Clin. Investig. 2015;125:386–402. doi: 10.1172/JCI76738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Jiao N., Baker S.S., Chapa-Rodriguez A., Liu W., Nugent C.A., Tsompana M., Mastrandrea L., Buck M.J., Baker R.D., Genco R.J., et al. Suppressed hepatic bile acid signalling despite elevated production of primary and secondary bile acids in NAFLD. Gut. 2018;67:1881–1891. doi: 10.1136/gutjnl-2017-314307. [DOI] [PubMed] [Google Scholar]
- 340.Ferslew B.C., Xie G., Johnston C.K., Su M., Stewart P.W., Jia W., Brouwer K.L., Barritt A.S.T. Altered bile acid metabolome in patients with nonalcoholic steatohepatitis. Dig. Dis. Sci. 2015;60:3318–3328. doi: 10.1007/s10620-015-3776-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Mouzaki M., Wang A.Y., Bandsma R., Comelli E.M., Arendt B.M., Zhang L., Fung S., Fischer S.E., McGilvray I.G., Allard J.P. Bile acids and dysbiosis in non-alcoholic fatty liver disease. PLoS ONE. 2016;11:e0151829. doi: 10.1371/journal.pone.0151829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Duncan S.H., Louis P., Thomson J.M., Flint H.J. The role of pH in determining the species composition of the human colonic microbiota. Environ. Microbiol. 2009;11:2112–2122. doi: 10.1111/j.1462-2920.2009.01931.x. [DOI] [PubMed] [Google Scholar]
- 343.Sawicki C.M., Livingston K.A., Obin M., Roberts S.B., Chung M., McKeown N.M. Dietary Fiber and the Human Gut Microbiota: Application of Evidence Mapping Methodology. Nutrients. 2017;9:125. doi: 10.3390/nu9020125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Koh A., De Vadder F., Kovatcheva-Datchary P., Bäckhed F. From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites. Cell. 2016;165:1332–1345. doi: 10.1016/j.cell.2016.05.041. [DOI] [PubMed] [Google Scholar]
- 345.Brussow H., Parkinson S.J. You are what you eat. Nat. Biotechnol. 2014;32:243–245. doi: 10.1038/nbt.2845. [DOI] [PubMed] [Google Scholar]
- 346.Subramanian S., Goodspeed L., Wang S., Kim J., Zeng L., Ioannou G.N., Haigh W.G., Yeh M.M., Kowdley K.V., O’Brien K.D., et al. Dietary cholesterol exacerbates hepatic steatosis and inflammation in obese LDL receptor-deficient mice. J. Lipid Res. 2011;52:1626–1635. doi: 10.1194/jlr.M016246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Musso G., Gambino R., Cassader M. Obesity, diabetes, and gut microbiota: The hygiene hypothesis expanded? Diabetes Care. 2010;33:2277–2284. doi: 10.2337/dc10-0556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Svegliati-Baroni G., Saccomanno S., Rychlicki C., Agostinelli L., De Minicis S., Candelaresi C., Faraci G., Pacetti D., Vivarelli M., Nicolini D., et al. Glucagon-like peptide-1 receptor activation stimulates hepatic lipid oxidation and restores hepatic signalling alteration induced by a high-fat diet in nonalcoholic steatohepatitis. Liver Int. Off. J. Int. Assoc. Study Liver. 2011;31:1285–1297. doi: 10.1111/j.1478-3231.2011.02462.x. [DOI] [PubMed] [Google Scholar]
- 349.Qin J., Li Y., Cai Z., Li S., Zhu J., Zhang F., Liang S., Zhang W., Guan Y., Shen D., et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490:55–60. doi: 10.1038/nature11450. [DOI] [PubMed] [Google Scholar]
- 350.Zhao L., Zhang F., Ding X., Wu G., Lam Y.Y., Wang X., Fu H., Xue X., Lu C., Ma J., et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science. 2018;359:1151–1156. doi: 10.1126/science.aao5774. [DOI] [PubMed] [Google Scholar]
- 351.Backhed F., Ding H., Wang T., Hooper L.V., Koh G.Y., Nagy A., Semenkovich C.F., Gordon J.I. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl. Acad. Sci. USA. 2004;101:15718–15723. doi: 10.1073/pnas.0407076101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Alex S., Lange K., Amolo T., Grinstead J.S., Haakonsson A.K., Szalowska E., Koppen A., Mudde K., Haenen D., Al-Lahham S., et al. Short-chain fatty acids stimulate angiopoietin-like 4 synthesis in human colon adenocarcinoma cells by activating peroxisome proliferator-activated receptor gamma. Mol. Cell. Biol. 2013;33:1303–1316. doi: 10.1128/MCB.00858-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Mehedint M.G., Zeisel S.H. Choline’s role in maintaining liver function: New evidence for epigenetic mechanisms. Curr. Opin. Clin. Nutr. Metab. Care. 2013;16:339–345. doi: 10.1097/MCO.0b013e3283600d46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Zeisel S.H., da Costa K.A. Choline: An essential nutrient for public health. Nutr. Rev. 2009;67:615–623. doi: 10.1111/j.1753-4887.2009.00246.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Wang Z., Klipfell E., Bennett B.J., Koeth R., Levison B.S., Dugar B., Feldstein A.E., Britt E.B., Fu X., Chung Y.M., et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472:57–63. doi: 10.1038/nature09922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Grattagliano I., Caraceni P., Portincasa P., Domenicali M., Palmieri V.O., Trevisani F., Bernardi M., Palasciano G. Adaptation of subcellular glutathione detoxification system to stress conditions in choline-deficient diet induced rat fatty liver. Cell Biol. Toxicol. 2003;19:355–366. doi: 10.1023/B:CBTO.0000013341.73139.fc. [DOI] [PubMed] [Google Scholar]
- 357.Dumas M.E., Barton R.H., Toye A., Cloarec O., Blancher C., Rothwell A., Fearnside J., Tatoud R., Blanc V., Lindon J.C., et al. Metabolic profiling reveals a contribution of gut microbiota to fatty liver phenotype in insulin-resistant mice. Proc. Natl. Acad. Sci. USA. 2006;103:12511–12516. doi: 10.1073/pnas.0601056103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Spencer M.D., Hamp T.J., Reid R.W., Fischer L.M., Zeisel S.H., Fodor A.A. Association between composition of the human gastrointestinal microbiome and development of fatty liver with choline deficiency. Gastroenterology. 2011;140:976–986. doi: 10.1053/j.gastro.2010.11.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Velasquez M.T., Ramezani A., Manal A., Raj D.S. Trimethylamine N-Oxide: The good, the bad and the unknown. Toxins. 2016;8:326. doi: 10.3390/toxins8110326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Chen Y.M., Liu Y., Zhou R.F., Chen X.L., Wang C., Tan X.Y., Wang L.J., Zheng R.D., Zhang H.W., Ling W.H., et al. Associations of gut-flora-dependent metabolite trimethylamine-N-oxide, betaine and choline with non-alcoholic fatty liver disease in adults. Sci. Rep. 2016;6:19076. doi: 10.1038/srep19076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Hoyles L., Fernández-Real J.-M., Federici M., Serino M., Abbott J., Charpentier J., Heymes C., Luque J.L., Anthony E., Barton R.H. Molecular phenomics and metagenomics of hepatic steatosis in non-diabetic obese women. Nat. Med. 2018;24:1070. doi: 10.1038/s41591-018-0061-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Machado M.V., Cortez-Pinto H. Diet, Microbiota, obesity, and NAFLD: A dangerous quartet. Int. J. Mol. Sci. 2016;17:481. doi: 10.3390/ijms17040481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Biedermann L., Zeitz J., Mwinyi J., Sutter-Minder E., Rehman A., Ott S.J., Steurer-Stey C., Frei A., Frei P., Scharl M., et al. Smoking cessation induces profound changes in the composition of the intestinal microbiota in humans. PLoS ONE. 2013;8:e59260. doi: 10.1371/journal.pone.0059260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Mutlu E.A., Gillevet P.M., Rangwala H., Sikaroodi M., Naqvi A., Engen P.A., Kwasny M., Lau C.K., Keshavarzian A. Colonic microbiome is altered in alcoholism. Am. J. Physiol. Gastrointest. Liver Physiol. 2012;302:G966–G978. doi: 10.1152/ajpgi.00380.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 365.Clarke S.F., Murphy E.F., Nilaweera K., Ross P.R., Shanahan F., O’Toole P.W., Cotter P.D. The gut microbiota and its relationship to diet and obesity: New insights. Gut Microbes. 2012;3:186–202. doi: 10.4161/gmic.20168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Turnbaugh P.J., Hamady M., Yatsunenko T., Cantarel B.L., Duncan A., Ley R.E., Sogin M.L., Jones W.J., Roe B.A., Affourtit J.P., et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480–484. doi: 10.1038/nature07540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Vrieze A., Van Nood E., Holleman F., Salojarvi J., Kootte R.S., Bartelsman J.F., Dallinga-Thie G.M., Ackermans M.T., Serlie M.J., Oozeer R., et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143:913–916.e917. doi: 10.1053/j.gastro.2012.06.031. [DOI] [PubMed] [Google Scholar]
- 368.Moreira G.V., Azevedo F.F., Ribeiro L.M., Santos A., Guadagnini D., Gama P., Liberti E.A., Saad M., Carvalho C. Liraglutide modulates gut microbiota and reduces NAFLD in obese mice. J. Nutr. Biochem. 2018;62:143–154. doi: 10.1016/j.jnutbio.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 369.Brandt A., Hernandez-Arriaga A., Kehm R., Sanchez V., Jin C.J., Nier A., Baumann A., Camarinha-Silva A., Bergheim I. Metformin attenuates the onset of non-alcoholic fatty liver disease and affects intestinal microbiota and barrier in small intestine. Sci. Rep. 2019;9:6668. doi: 10.1038/s41598-019-43228-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Feng W., Wang H., Zhang P., Gao C., Tao J., Ge Z., Zhu D., Bi Y. Modulation of gut microbiota contributes to curcumin-mediated attenuation of hepatic steatosis in rats. Biochim. Biophys. Acta Gen. Subj. 2017;1861:1801–1812. doi: 10.1016/j.bbagen.2017.03.017. [DOI] [PubMed] [Google Scholar]
- 371.Gao B., Chi L., Mahbub R., Bian X., Tu P., Ru H., Lu K. Multi-Omics Reveals that Lead Exposure Disturbs Gut Microbiome Development, Key Metabolites, and Metabolic Pathways. Chem. Res. Toxicol. 2017;30:996–1005. doi: 10.1021/acs.chemrestox.6b00401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 372.Gao B., Bian X., Mahbub R., Lu K. Sex-Specific effects of organophosphate diazinon on the gut microbiome and its metabolic functions. Environ. Health Perspect. 2017;125:198–206. doi: 10.1289/EHP202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Joly C., Gay-Queheillard J., Leke A., Chardon K., Delanaud S., Bach V., Khorsi-Cauet H. Impact of chronic exposure to low doses of chlorpyrifos on the intestinal microbiota in the Simulator of the Human Intestinal Microbial Ecosystem (SHIME) and in the rat. Environ. Sci. Pollut. Res. Int. 2013;20:2726–2734. doi: 10.1007/s11356-012-1283-4. [DOI] [PubMed] [Google Scholar]
- 374.Joly Condette C., Bach V., Mayeur C., Gay-Queheillard J., Khorsi-Cauet H. Chlorpyrifos exposure during perinatal period affects intestinal microbiota associated with delay of maturation of digestive tract in rats. J. Pediatr. Gastroenterol. Nutr. 2015;61:30–40. doi: 10.1097/MPG.0000000000000734. [DOI] [PubMed] [Google Scholar]
- 375.Hooper L.V., Littman D.R., Macpherson A.J. Interactions between the microbiota and the immune system. Science. 2012;336:1268–1273. doi: 10.1126/science.1223490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Thaiss C.A., Zmora N., Levy M., Elinav E. The microbiome and innate immunity. Nature. 2016;535:65–74. doi: 10.1038/nature18847. [DOI] [PubMed] [Google Scholar]
- 377.Britanova L., Diefenbach A. Interplay of innate lymphoid cells and the microbiota. Immunol. Rev. 2017;279:36–51. doi: 10.1111/imr.12580. [DOI] [PubMed] [Google Scholar]
- 378.Bischoff S.C., Barbara G., Buurman W., Ockhuizen T., Schulzke J.D., Serino M., Tilg H., Watson A., Wells J.M. Intestinal permeability—A new target for disease prevention and therapy. BMC Gastroenterol. 2014;14:189. doi: 10.1186/s12876-014-0189-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Kirpich I.A., Marsano L.S., McClain C.J. Gut-liver axis, nutrition, and non-alcoholic fatty liver disease. Clin. Biochem. 2015;48:923–930. doi: 10.1016/j.clinbiochem.2015.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Schroeder B.O., Birchenough G.M.H., Stahlman M., Arike L., Johansson M.E.V., Hansson G.C., Backhed F. Bifidobacteria or fiber protects against diet-induced microbiota-mediated colonic mucus deterioration. Cell Host Microbe. 2018;23:27–40.e7. doi: 10.1016/j.chom.2017.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Luck H., Tsai S., Chung J., Clemente-Casares X., Ghazarian M., Revelo X.S., Lei H., Luk C.T., Shi S.Y., Surendra A., et al. Regulation of obesity-related insulin resistance with gut anti-inflammatory agents. Cell Metab. 2015;21:527–542. doi: 10.1016/j.cmet.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 382.Spruss A., Kanuri G., Wagnerberger S., Haub S., Bischoff S.C., Bergheim I. Toll-like receptor 4 is involved in the development of fructose-induced hepatic steatosis in mice. Hepatology. 2009;50:1094–1104. doi: 10.1002/hep.23122. [DOI] [PubMed] [Google Scholar]
- 383.Lambertz J., Weiskirchen S., Landert S., Weiskirchen R. Fructose: A dietary sugar in crosstalk with microbiota contributing to the development and progression of non-alcoholic liver disease. Front. Immunol. 2017;8:1159. doi: 10.3389/fimmu.2017.01159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Ray K. NAFLD. Leaky guts: Intestinal permeability and NASH. Nat. Rev. Gastroenterol. Hepatol. 2015;12:123. doi: 10.1038/nrgastro.2015.15. [DOI] [PubMed] [Google Scholar]
- 385.Miele L., Marrone G., Lauritano C., Cefalo C., Gasbarrini A., Day C., Grieco A. Gut-liver axis and microbiota in NAFLD: Insight pathophysiology for novel therapeutic target. Curr. Pharm. Des. 2013;19:5314–5324. doi: 10.2174/1381612811319290011. [DOI] [PubMed] [Google Scholar]
- 386.Cani P.D., Possemiers S., Van de Wiele T., Guiot Y., Everard A., Rottier O., Geurts L., Naslain D., Neyrinck A., Lambert D.M., et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut. 2009;58:1091–1103. doi: 10.1136/gut.2008.165886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Ding S., Chi M.M., Scull B.P., Rigby R., Schwerbrock N.M., Magness S., Jobin C., Lund P.K. High-fat diet: Bacteria interactions promote intestinal inflammation which precedes and correlates with obesity and insulin resistance in mouse. PLoS ONE. 2010;5:e12191. doi: 10.1371/journal.pone.0012191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Kavanagh K., Wylie A.T., Tucker K.L., Hamp T.J., Gharaibeh R.Z., Fodor A.A., Cullen J.M. Dietary fructose induces endotoxemia and hepatic injury in calorically controlled primates. Am. J. Clin. Nutr. 2013;98:349–357. doi: 10.3945/ajcn.112.057331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Bluemel S., Wang L., Martino C., Lee S., Wang Y., Williams B., Horvath A., Stadlbauer V., Zengler K., Schnabl B. The role of intestinal c-type regenerating islet derived-3 lectins for nonalcoholic steatohepatitis. Hepatol. Commun. 2018;2:393–406. doi: 10.1002/hep4.1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Jin R., Willment A., Patel S.S., Sun X., Song M., Mannery Y.O., Kosters A., McClain C.J., Vos M.B. Fructose induced endotoxemia in pediatric nonalcoholic Fatty liver disease. Int. J. Hepatol. 2014;2014:560620. doi: 10.1155/2014/560620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Bifulco M. Mediterranean diet: The missing link between gut microbiota and inflammatory diseases. Eur. J. Clin. Nutr. 2015;69:1078. doi: 10.1038/ejcn.2015.81. [DOI] [PubMed] [Google Scholar]
- 392.Biolato M., Manca F., Marrone G., Cefalo C., Racco S., Miggiano G.A., Valenza V., Gasbarrini A., Miele L., Grieco A. Intestinal permeability after Mediterranean diet and low-fat diet in non-alcoholic fatty liver disease. World J. Gastroenterol. WJG. 2019;25:509–520. doi: 10.3748/wjg.v25.i4.509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Enomoto N., Yamashina S., Kono H., Schemmer P., Rivera C.A., Enomoto A., Nishiura T., Nishimura T., Brenner D.A., Thurman R.G. Development of a new, simple rat model of early alcohol-induced liver injury based on sensitization of Kupffer cells. Hepatology. 1999;29:1680–1689. doi: 10.1002/hep.510290633. [DOI] [PubMed] [Google Scholar]
- 394.Pappo I., Bercovier H., Berry E.M., Haviv Y., Gallily R., Freund H.R. Polymyxin B reduces total parenteral nutrition-associated hepatic steatosis by its antibacterial activity and by blocking deleterious effects of lipopolysaccharide. Jpn. J. Parenter. Enter. Nutr. 1992;16:529–532. doi: 10.1177/0148607192016006529. [DOI] [PubMed] [Google Scholar]
- 395.Pappo I., Becovier H., Berry E.M., Freund H.R. Polymyxin B reduces cecal flora, TNF production and hepatic steatosis during total parenteral nutrition in the rat. J. Surg. Res. 1991;51:106–112. doi: 10.1016/0022-4804(91)90078-Z. [DOI] [PubMed] [Google Scholar]
- 396.Drenick E.J., Fisler J., Johnson D. Hepatic steatosis after intestinal bypass—Prevention and reversal by metronidazole, irrespective of protein-calorie malnutrition. Gastroenterology. 1982;82:535–548. doi: 10.1016/S0016-5085(82)80403-4. [DOI] [PubMed] [Google Scholar]
- 397.Alisi A., Bedogni G., Baviera G., Giorgio V., Porro E., Paris C., Giammaria P., Reali L., Anania F., Nobili V. Randomised clinical trial: The beneficial effects of VSL#3 in obese children with non-alcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2014;39:1276–1285. doi: 10.1111/apt.12758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Li Z., Yang S., Lin H., Huang J., Watkins P.A., Moser A.B., Desimone C., Song X.Y., Diehl A.M. Probiotics and antibodies to TNF inhibit inflammatory activity and improve nonalcoholic fatty liver disease. Hepatology. 2003;37:343–350. doi: 10.1053/jhep.2003.50048. [DOI] [PubMed] [Google Scholar]
- 399.Dallio M., Masarone M., Errico S., Gravina A.G., Nicolucci C., Di Sarno R., Gionti L., Tuccillo C., Persico M., Stiuso P., et al. Role of bisphenol A as environmental factor in the promotion of non-alcoholic fatty liver disease: In vitro and clinical study. Aliment. Pharmacol. Ther. 2018;47:826–837. doi: 10.1111/apt.14499. [DOI] [PubMed] [Google Scholar]
- 400.Feng D., Zhang H., Jiang X., Zou J., Li Q., Mai H., Su D., Ling W., Feng X. Bisphenol A exposure induces gut microbiota dysbiosis and consequent activation of gut-liver axis leading to hepatic steatosis in CD-1 mice. Environ. Pollut. 2020;265:114880. doi: 10.1016/j.envpol.2020.114880. [DOI] [PubMed] [Google Scholar]
- 401.Wang D., Yan J., Teng M., Yan S., Zhou Z., Zhu W. In utero and lactational exposure to BDE-47 promotes obesity development in mouse offspring fed a high-fat diet: Impaired lipid metabolism and intestinal dysbiosis. Arch. Toxicol. 2018;92:1847–1860. doi: 10.1007/s00204-018-2177-0. [DOI] [PubMed] [Google Scholar]
- 402.Deierlein A.L., Rock S., Park S. Persistent Endocrine-Disrupting Chemicals and Fatty Liver Disease. Curr. Environ. Health Rep. 2017;4:439–449. doi: 10.1007/s40572-017-0166-8. [DOI] [PubMed] [Google Scholar]
- 403.Cave M., Appana S., Patel M., Falkner K.C., McClain C.J., Brock G. Polychlorinated biphenyls, lead, and mercury are associated with liver disease in American adults: NHANES 2003–2004. Environ. Health Perspect. 2010;118:1735–1742. doi: 10.1289/ehp.1002720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 404.Chi Y., Lin Y., Lu Y., Huang Q., Ye G., Dong S. Gut microbiota dysbiosis correlates with a low-dose PCB126-induced dyslipidemia and non-alcoholic fatty liver disease. Sci. Total Environ. 2019;653:274–282. doi: 10.1016/j.scitotenv.2018.10.387. [DOI] [PubMed] [Google Scholar]
- 405.Petriello M.C., Hoffman J.B., Vsevolozhskaya O., Morris A.J., Hennig B. Dioxin-like PCB 126 increases intestinal inflammation and disrupts gut microbiota and metabolic homeostasis. Environ. Pollut. 2018;242:1022–1032. doi: 10.1016/j.envpol.2018.07.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Lukowicz C., Ellero-Simatos S., Regnier M., Polizzi A., Lasserre F., Montagner A., Lippi Y., Jamin E.L., Martin J.F., Naylies C., et al. Metabolic effects of a chronic dietary exposure to a low-dose pesticide cocktail in mice: Sexual dimorphism and role of the constitutive androstane receptor. Environ. Health Perspect. 2018;126:067007. doi: 10.1289/EHP2877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Hyder O., Chung M., Cosgrove D., Herman J.M., Li Z., Firoozmand A., Gurakar A., Koteish A., Pawlik T.M. Cadmium exposure and liver disease among US adults. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract. 2013;17:1265–1273. doi: 10.1007/s11605-013-2210-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Zhang S., Jin Y., Zeng Z., Liu Z., Fu Z. Subchronic exposure of mice to cadmium perturbs their hepatic energy metabolism and gut microbiome. Chem. Res. Toxicol. 2015;28:2000–2009. doi: 10.1021/acs.chemrestox.5b00237. [DOI] [PubMed] [Google Scholar]
- 409.Ba Q., Li M., Chen P., Huang C., Duan X., Lu L., Li J., Chu R., Xie D., Song H., et al. Sex-Dependent effects of cadmium exposure in early life on gut microbiota and fat accumulation in mice. Environ. Health Perspect. 2017;125:437–446. doi: 10.1289/EHP360. [DOI] [PMC free article] [PubMed] [Google Scholar]