Abstract
Diabetic foot ulcer (DFU) is a chronic complication that negatively affects the quality of life (QoL) of diabetic patients. In Spain, there is no specifically designed and validated instrument to assess the QoL of patients with DFU. Our aim was to adapt the Diabetic Foot Ulcer Scale-Short Form (DFS-SF) questionnaire to a Spanish population and validate it. A prospective, observational design was used. The DFS-SF was administered by personal interview. The validated SF-36 and EQ-5D generic instruments were used as reference tools. The reliability, validity, and sensitivity to changes were assessed using standard statistical methods. A sample of 141 patients with DFU was recruited. The content validity was 3.46 on average (maximum score of 4). The internal consistency of the DFS-SF subscales showed a standardized Cronbach’s α range between 0.720 and 0.948. The DFS-SF domains showed excellent reproducibility measures (intraclass correlation coefficient from 0.77–0.92). The criterion validity was good with significant correlations between each DFS-SF subscale and its corresponding SF-36 and EQ-5D subscales (p < 0.001). However, the questionnaire structure was not validated (comparative fit index = 0.844, root mean square error of approximation = 0.095, and standardized root mean square residual = 0.093). The instrument showed high sensitivity to ulcer changes over time (p < 0.001). The adapted and validated Spanish version of the DFS-SF questionnaire has good psychometric properties and shows good sensitivity to ulcer changes, although the construct validity was not optimal. The adapted questionnaire will be a useful tool specifically to assess the QoL in subjects with diabetic foot ulcers in the clinical and research settings in Spain.
Keywords: diabetic foot ulcer, type 2 diabetes, quality of life, psychometric validation, reliability, validity
1. Introduction
Diabetic foot ulcer (DFU) is a condition that has a significant impact on several aspects of everyday life [1,2]. This diabetic complication usually requires multidisciplinary approaches with often intensive and prolonged treatments, which may have a substantial impact on the quality of life (QoL) [1]. Furthermore, patients with DFU are at high-risk of cardiovascular diseases in comparison with patients without this condition due to a greater burden of the disease [2]; additionally, ulceration may lead to lower-limb amputation and is associated with an increased risk of all-cause mortality [3,4]. For these reasons, it is important to assess the QoL of these patients.
QoL is a patient-reported outcome (PRO) that has become an important physical and psychological well-being measure [1]. Some observational studies reported that DFU has a negative impact on the QoL of these patients [5,6,7]. Besides, a cross-sectional study observed that patients with DFU reported a higher perception of pain related to DFU, representing a significant clinical PRO [8]. The number and severity of foot ulcers are associated with a negative health-related QoL (HRQoL) in patients with DFU compared with those patients without this diabetic complication [9]. Moreover, physical functioning and emotional role are the most affected aspects of daily life, especially in terms of leisure activity and constraints due to the treatment received [9]. Finally, while the occurrence of a minor amputation is not associated with a poorer QoL compared with patients with active DFU [10,11], the ability to maintain mobility is related to an improved HRQoL in patients with a major amputation [10].
One major limitation of the available studies is the use of different outcome instruments to assess the relationship between QoL and DFU [10]. A systematic review of the potential measures of QoL in patients with diabetes-related foot disease showed that there is no one ideal PRO for QoL assessment, so that each instrument has its limitations [1]. The use of generic instruments could underestimate the impact of the DFU on the QoL in emotional and mental domains [10]. However, disease-specific tools such as the Diabetic Foot Ulcer Scale (DFS) have shown high sensitivity to changes related to foot health or disease severity, with few confounders [1,12]. Moreover, this questionnaire offers greater information about the impact of ulcers on the QoL and is able to detect changes in DFU [1,13]. An abbreviated version of this instrument, the DFS-short form (DFS-SF), was originally designed and validated in English and afterwards adapted and validated in other languages such as Chinese, Greek, and Polish [13,14,15,16]. The available scientific evidence shows that the DFS-SF is an instrument with good psychometric properties and replicability in patients with DFU [1,13,14,15,16].
In Spain, there is no instrument specifically designed and validated to assess the impact of DFU in the QoL of diabetic patients. The aim of the study was to translate and assess the psychometric properties of the DFS-SF in a Spanish population.
2. Experimental Section
2.1. Sample and Settings
This was a prospective, observational study conducted in subjects with DFU referred from primary care and other hospital departments. Patients were treated at the Diabetic Foot Unit primarily by a podiatrist in the context of a multidisciplinary team. The DFS-SF instrument was prospectively administered at 7 days and 4, 12, and 26 weeks after the baseline assessment. The inclusion criteria were age >18 years and new-onset DFU located below the malleoli with up to 3-months duration. The exclusion criteria included cognitive deterioration, terminal illnesses, and hospitalization. A trained researcher (D.M.-G.) performed individual interviews with all of the patients and reviewed each of their clinical records carefully to collect the following clinical variables: Age; sex; educational level; smoking habit; type of diabetes, disease duration, and diabetes-specific therapy; the presence of hypertension and dyslipidemia if they were diagnosed with the disease or were receiving for medication for any of these conditions; the presence of cardiovascular diseases (i.e., cerebrovascular disease, peripheral artery disease, and ischemic heart disease); the diagnose of diabetic retinopathy; dialysis (including both hemodialysis and peritoneal dialysis); and the use of antiplatelet agents. Glycated hemoglobin (HbA1c) was collected using the most recent value within the previous 6 months. Diabetic nephropathy was defined as an estimated glomerular filtration rate (eGFR) below 60 mL/min and/or an albumin-to-creatinine urine ratio over 30 mg/g. The diagnosis of DFU was defined according to the consensus of the International Working Group on the Diabetic Foot (IWGDF) [17]. A detailed foot examination was performed to determine the presence of previous lower-limb amputations (minor o major), deformities, or the diagnosis of Charcot foot disease and to assess the local ulcer characteristics [18,19,20]. Peripheral neuropathy was assessed using a biothensiometer (Me. Te. Da. Srl., IT) using standard procedures, as described previously [18]. Peripheral arterial disease was evaluated through the ankle-brachial index (ABI) value and classified as normal (between 0.91 and 1.30), moderate ischemia (between 0.41 and 0.90), severe ischemia (between 0 and 0.40), and non-compressible due to the presence of calcification (over 1.30) [21]. In those patients with an ABI value of 1.30 or greater, the pedal or posterior tibial pulse were explored; the diagnosis of peripheral arterial disease was defined by the presence of a non-palpable pulses. The ulcer was classified as neuropathic, ischemic and neuroischemic according to the consensus of the IWGDF [17]. Infected ulcers were clinically diagnosed by the presence of at least 2 signs or symptoms of inflammation (i.e., redness, warmth, induration, and pain/tenderness), or purulent secretions. Furthermore, systemic inflammatory findings (i.e., fever, leukocytosis, and C reactive protein) were also assessed to classify the severity of the infection [22]. All study participants signed a written informed consent form before inclusion in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the University Hospital Arnau de Vilanova.
2.2. Instruments
2.2.1. Diabetic Foot Ulcer Scale-Short Form (DFS-SF)
The DFS-SF is a disease-specific questionnaire that assesses the impact of DFU on the QoL [13]. This instrument contains 29 items based on the following 6 subscales: leisure, physical health, dependence/daily life, negative emotions, worried about ulcers/feet, and bothered by ulcer care. The score of each subscale is calculated based on a scale from 0 (poorer QoL) to 100 (higher QoL). This was validated in reference to the original extended questionnaire DFS that contains 58 items and 11 domains [12].
2.2.2. 36-Item Short-Form Health Survey (SF-36)
The SF-36 is a generic instrument that assesses the health status of the patients [23]. It is composed of 36 items that are grouped into 8 subscales: physical functioning, physical role, bodily pain, general health, vitality, social functioning, emotional role, and mental health. These subscales are weighted into physical and mental health summary scores. Each subscale is scored with a range from 0 to 100 points and is normalized using US norms; higher scores indicate a better health-related quality of life (HRQoL).
2.2.3. EuroQoL 5D Health Utility Index (EQ-5D)
EQ-5D is a generic questionnaire designed for use in different types of health states and diseases, as well as in the general populations of several countries [24]. This instrument includes 5 dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each item is divided into 3 levels: no problems, some problems, and extreme problems. A single numeric index of health status (EQ-5D index value) is defined by combining the 5 dimensions using UK weights. The questionnaire also includes a visual analogue scale (VAS) where participants are asked to indicate how they rate their current health status on a scale from 0 (minimum) to 100 points (maximum or the best imaginable health state).
2.3. Transcultural Adaptation of the DFS-SF
First, the English version was translated into Spanish by two independent translators who were experts in both languages. The 2 translated versions were later compared, and noted differences were discussed by a group of patients and a group of health-care professionals with expertise in diabetic foot disease. After this, the first Spanish version was approved and back-translated by a third English native speaker. Researchers then compared this back-translation and the original English questionnaire. The final Spanish version was assessed by the group of experts to determine its content validity using a Likert scale (File S1; available at https://www.irblleida.org/media/upload/arxius/VARIS/File%20S1_cuestionario.pdf).
2.4. Sample Size
The measure of internal consistency was determined by accepting an alpha risk of 0.05 and a beta risk of 0.2 in a two-sided test. We calculated that a sample of 124 patients would be required to designate a Cronbach’s α coefficient ≥ 0.3 as statistically significant. Anticipating a maximum dropout rate of 15%, the final required sample size resulted in 143 subjects.
2.5. Data Analysis
Data were described by using mean and standard deviation for quantitative variables and absolute and relative frequencies for qualitative variables.
The reliability was measured through internal consistency and reproducibility assessment. The internal consistency was measured by Cronbach’s α coefficient [25]. According to the standard protocol, the Cronbach value should be ≥0.70. The reproducibility was estimated using the intraclass correlation coefficient (ICC) defined by a single rater two-way mixed-effects model for quantitative variables. Baseline and first follow-up visit (at 7 days after) ratings were compared, assuming no changes for unhealed ulcers.
The validity was measured through a criterion and construct validity assessment. The criterion validity was only determined according to the concurrent validity in reference to the generic questionnaires by measuring the Pearson’s correlation coefficient of the scores of the DFS-SF subscales and comparing them with the ones of the corresponding SF-36 and EQ-5D subscales. Their values should be >0.3, which is indicative of a moderate correlation. The construct validity was assessed through a confirmatory factor analysis of the DFS-SF to test the questionnaire structure. The comparative fit index (CFI), the root mean square error of approximation (RMSEA), and the standardized root mean square residual (SRMR) were estimated. Their values should be ≥0.95, ≤0.06, and ≤0.08, respectively, as indicative of a good fit to the subscales structure.
The sensitivity to changes over time is defined as the ability of an instrument to measure a change in the state regardless of whether the change is relevant or meaningful to the decision-maker [26,27]. This was assessed through the smoothed trends from baseline until the last visit and, depending on the healing state of the ulcer, at the last available visit; changes between healed and non-healed patients were compared using the Mann–Whitney test. All analyses were performed with the R software with a significance level of 0.05 [28].
3. Results
The characteristics of the 141 participants are shown in Table 1. A high frequency of patients with neuropathy (92.9%) and macrovascular complications (89.4%) was observed. At the end of the study, 107 of the patients (75.8%) had experienced healing (Table S1).
Table 1.
Clinical and sociodemographic characteristics of the study group at baseline.
| Characteristics | Study Group (n = 141) |
|---|---|
| Age (years) | 68.3 (13.3) |
| Male (sex) | 95 (67.4) |
| Race (Caucasian) | 140 (99.3) |
| Educational level | |
| Not even primary | 57 (40.4) |
| Complete primary | 47 (33.3) |
| Secondary high cycle | 28 (19.9) |
| Graduate or higher | 9 (6.4) |
| Employed | 24 (17.0) |
| Smoking | |
| Never | 63 (44.7) |
| Current or former | 78 (55.4) |
| Type 2 diabetes | 134 (95.0) |
| BMI (kg/m2) | 29.0 (4.9) |
| HbA1c (%) | 7.5 (1.6) |
| Hypertension | 116 (82.3) |
| Dyslipidemia | 87 (61.7) |
| Microvascular complications | |
| Retinopathy | 96 (68.1) |
| Nephropathy | 51 (36.2) |
| Neuropathy | 131 (92.9) |
| Cardiovascular disease 1 | 126 (89.4) |
| Diabetes therapy | |
| OAD | 41 (29.1) |
| OAD + insulin | 57 (40.4) |
| Insulin | 36 (25.5) |
| Diet | 7 (5.0) |
| Antiplatelet agents | 94 (66.7) |
| Dialysis | 8 (5.7) |
| Ulcer type | |
| Neuropathic | 87 (61.7) |
| Ischemic | 9 (6.4) |
| Neuroischemic | 45 (31.9) |
| Infected ulcer | 83 (58.9) |
| Previous amputation | |
| Minor | 41 (29.1) |
| Major | 2 (1.4) |
| Charcot foot disease | 9 (6.4) |
Data are shown as mean (SD) for quantitative variables or n (%) for qualitative variables. 1 Cardiovascular disease included cerebrovascular disease, peripheral artery disease and ischemic heart disease. BMI, body mass index; HbA1c, glycated hemoglobin; OAD, oral antidiabetic agents.
The content validity was assessed by seven experts who rendered an average score of 3.46 (86% of the maximum score of 4). The internal consistency of the DFS-SF subscales was good (Cronbach’s α range = 0.720–0.948) (Table 2). The internal consistency of the subscales was not improved or marginally improved by item deletion. The DFS-SF domains showed reproducibility measures that ranged from good to excellent (ICC estimates = 0.77–0.92).
Table 2.
Inter-item internal consistency and reproducibility of the Diabetes Foot Ulcer-Short Form (DFS-SF) subscales.
| Dfs-Sf Subscales | Number of Items 1 | Range of Correlations 2 | Average Inter-Item Correlation | Cronbach’s Alpha | Reproducibility (ICC [95%CI]) |
|---|---|---|---|---|---|
| Leisure | 5 | 0.678–0.966 | 0.786 | 0.948 | 0.87 [0.82, 0.91] |
| Physical health | 5 | 0.195–0.641 | 0.433 | 0.792 3 | 0.78 [0.70, 0.84] |
| Worried about ulcers/feet | 4 | 0.678–0.818 | 0.760 | 0.927 | 0.92 [0.89, 0.94] |
| Dependence/daily life | 5 | 0.417–0.662 | 0.566 | 0.867 | 0.77 [0.68, 0.83] |
| Negative emotions | 6 | 0.310–0.886 | 0.553 | 0.881 4 | 0.84 [0.79, 0.89] |
| Bothered by ulcer care | 4 | 0.304–0.537 | 0.391 | 0.720 | 0.77 [0.69, 0.83] |
1 Number of items per subscale. 2 Inter-item Pearson’s correlations. 3 Improved from 0.792 to 0.794 if the second item for this subscale is deleted. 4 Improved from 0.881 to 0.892 if the fifth item for this subscale is deleted. Reproducibility was estimated by assuming no changes between the baseline and first visit (at 7 days from baseline) for unhealed ulcers. ICC, intraclass correlation coefficients; CI, confidence interval.
The DFS-SF subscales were correlated with SF-36 and EQ-5D subscales (Table 3). The Leisure and Worried about ulcers/feet subscales were moderately correlated with Social functioning (r > 0.32, p < 0.001) of the SF-36. Physical health was moderately correlated with Physical functioning, Role, and Component summary as well as Bodily pain, Social functioning, and Vitality of the SF-36 and with the EQ-5D score (r > 0.32, p < 0.001). Dependence was largely correlated with the Physical functioning, Role physical, and Overall physical components (r > 0.55, p < 0.001); also, Dependence was moderately correlated with Social functioning, Vitality, and Bodily pain of the SF-36 (r > 0.4, p < 0.001) and with the EQ-5D index value (r = 0.45, p < 0.001). The Negative emotions subscale was largely correlated with Social functioning (r > 0.52, p < 0.001) and all other SF-36 domains, the EQ-5D score, and VAS. Bothered by ulcer care was moderately correlated with Physical functioning, Role, and Component summary as well as Vitality and Social functioning (r > 0.33, p < 0.001).
Table 3.
Criterion validity of the Diabetic Foot Ulcer Scale-Short Form by Pearson’s correlation coefficients with SF-36 and EQ-5D overall and subscale scores.
| Domains. | DFS-SF Subscales | |||||
|---|---|---|---|---|---|---|
| Leisure | Physical Health | Dependence/Daily Life | Worried about Ulcers/Feet | Negative Emotions | Bothered by Ulcer Care | |
| SF-36 subscales | ||||||
| Physical functioning | 0.052 | 0.473 ** | 0.737 ** | 0.220 * | 0.398 ** | 0.370 ** |
| Role physical | 0.135 | 0.413 ** | 0.558 ** | 0.258 * | 0.445 ** | 0.376 ** |
| Bodily pain | 0.116 | 0.438 ** | 0.403 ** | 0.224 * | 0.395 ** | 0.277 * |
| General health | 0.057 | 0.165 | 0.139 | 0.183 * | 0.350 ** | 0.198 * |
| Vitality | 0.126 | 0.327 ** | 0.418 ** | 0.245 * | 0.383 ** | 0.345 ** |
| Social functioning | 0.358 ** | 0.401 ** | 0.453 ** | 0.321 ** | 0.526 ** | 0.336 ** |
| Role emotional | 0.140 | 0.271 ** | 0.221 * | 0.187 * | 0.365 ** | 0.288 * |
| Mental health | 0.079 | 0.204 * | 0.174 * | 0.256 * | 0.484 ** | 0.199 * |
| Overall physical component 1 | 0.082 | 0.482 ** | 0.674 ** | 0.229 * | 0.394 ** | 0.368 ** |
| Overall mental component 2 | 0.178 * | 0.195 * | 0.110 | 0.248 * | 0.449 ** | 0.235 * |
| EQ-5D subscales | ||||||
| VAS | 0.062 | 0.166 * | 0.223 * | 0.240 * | 0.306 ** | 0.183 * |
| EQ-5D index value | 0.039 | 0.445 ** | 0.454 ** | 0.204 * | 0.324 ** | 0.184 * |
1,2 Calculated according to the SF-36 subscales involved physical and mental roles. VAS, visual analog scale. * p < 0.05; ** p < 0.001.
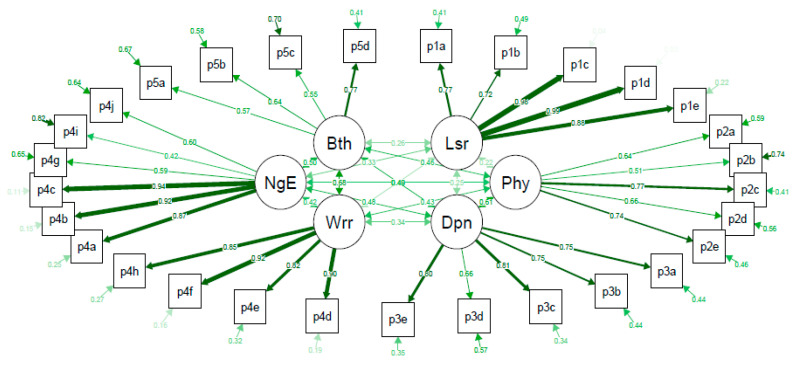
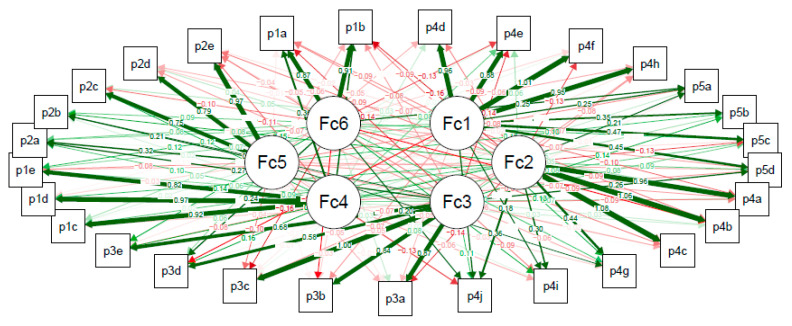
The confirmatory factor analysis of the DFS-SF showed that the questionnaire structure was not validated (Figure 1). The full set of data corresponding to Figure 1 is provided in Table S2. CFI, RMSEA, and SRMR were 0.844, 0.095, and 0.093, respectively, showing a lack of good fit to the questionnaire structure. The exploratory factor analysis of 6 factors divided the Leisure subscale into 2 subscales (questions p1a-p1b refer to hobbies and leisure activities vs. p5c-p1e that refer to holidays, weekends, and activities planning), and did not identify the Bothered by ulcer care subscale but joined all its items to the Dependence or Daily life subscale (Figure 2). The full set of data corresponding to Figure 2 is provided in Table S3.
Figure 1.
Confirmatory factor analysis of the different subscales of the Diabetes Foot Ulcer Scale-Short Form (DFS-SF). Bth, bothered by ulcer care; NgE, negative emotions; Wrr, worried about ulcers/feet; Lsr, leisure; Phy, physical health; Dpn, dependence/daily life. p1a–p5d are the items per subscale of the DFS-SF questionnaire. Each arrow between the questionnaire items and the subscale that they are measuring shows the standardized pattern coefficients for this relationship, where values closer to 1.0 (wider and darker) are indicative of better fit, and the circled arrow represented in each questionnaire item shows the residuals. The arrows connecting subscales show the pairwise correlation between them. Comparative fit index (CFI) = 0.844 (stands for comparative fit index and a value of ≥0.95 is indicative of good fit); root mean square error of approximation (RMSEA) = 0.096 (stands for the root mean square error of approximation and a value of ≤0.06 is indicative of acceptable model fit); standardized root mean square residual (SRMR) = 0.094 (stands for standardized root mean square residual and a value of ≤0.08 is indicative of an acceptable model).
Figure 2.
Exploratory factor analysis of the different subscales of the Diabetes Foot Ulcer Scale-Short Form (DFS-SF). Fc1–Fc6, Factor 1 to Factor 6 of the exploratory factor analysis. p1a–p5d are the items per subscale of the DFS-SF questionnaire. Maximum likelihood exploratory factor analysis fixing the total number of subscales in 6 with a promax rotation. Each arrow shows the loading of each questionnaire item to each subscale, where the width and color intensity of each arrow is proportional to each value. Loadings closer to 1.0 in absolute value are indicative of stronger relationships with subscales (red color is indicative of opposite relationship). The explained cumulative variance by the 6 factors was 65.5 %.
Finally, the DFS-SF was sensitive to the ulcer status changes (p < 0.001) (Table 4). Healed patients had significantly higher punctuations in all subscales in comparison with the unhealed group.
Table 4.
Summary of the descriptive analysis of sensitivity to change assessment of Diabetes Foot Ulcer Scale-Short Form (DFS-SF) subscales according to healed/unhealed group.
| Variables | Unhealed 1 (n = 34) | Healed (n = 107) | p-Overall |
|---|---|---|---|
| Change in DFS Leisure from baseline | 0.00 [0.00; 8.75] | 5.00 [0.00; 25.00] | 0.014 |
| Change in DFS Physical Health from baseline | 0.00 [−10.00; 0.00] | 20.00 [5.00; 30.00] | <0.001 |
| Change in DFS Dependence from baseline | 0.00 [0.00; 3.75] | 10.00 [0.00; 25.00] | <0.001 |
| Change in DFS Negative Emotions from baseline | 0.00 [0.00; 4.17] | 8.34 [0.00; 25.00] | 0.001 |
| Change in DFS Worried about ulcers/feet from baseline | 0.00 [0.00; 18.80] | 31.2 [9.38; 50.00] | <0.001 |
| Change in DFS Bothered by ulcer care from baseline | 0.00 [0.00; 6.25] | 18.80 [0.00; 37.50] | <0.001 |
Data are shown as median [confidence interval]. 1 We are including here all subjects that remained unhealed until the last visit, until they were lost to follow-up or experienced an unfavorable event (amputation, death).
4. Discussion
This is the first study designed to assess the psychometric properties of a specific questionnaire in patients with DFU in Spain. Our results showed that the Spanish DFS-SF had good internal consistency and criterion validity with excellent reproducibility. Although its structure was not validated, the sensitivity to ulcer changes over time was high. The internal consistency of the DFS-SF subscales was good, with a Cronbach’s α range similar to that of the original English version [13]. Furthermore, reproducibility was excellent, as observed in the English version [12,13].
The Spanish DFS-SF demonstrated good criterion validity; the overall physical and mental components of the SF-36 had moderate correlations with each of the physical and mental subscales of the DFS-SF. This is similar to the English, Greek, and Chinese studies [13,14,15]; however, the Polish version did not identify any correlation in the physical subscales [16].
Moreover, the Spanish DFS-SF demonstrated poor construct validity, which is in contrast with previously published studies [13,14,15,16]. This could be due to the cultural differences between the English and Spanish populations; in the Spanish population, the variables are distinctively distributed and grouped in comparison with other countries.
The Spanish DFS-SF showed sensitivity to ulcer changes over time as observed in the versions in other languages [7,13,16]. Furthermore, the DFS-SF is more sensitive than the generic instruments because in the latter case the impact of other conditions on the QoL may be confounded [14,16,29]. This is mainly due to the extensive use of generic instruments to measure QoL in many previous studies. Therefore, this is the first specific validated questionnaire to assess the QoL of subjects with diabetes and foot ulcers in Spain.
As mentioned previously, the optimal assessment of QoL for specific conditions, such as DFU, requires the use of instruments adequately designed for this purpose [10]. Apart from its original English version, the DFS questionnaire is currently available only in Chinese, Greek, and Polish [14,15,16]. We strongly believe that there is a great need to provide specific tools for measuring the QoL in subjects with DFU in many other countries and languages. The absence of a proper tool for this purpose in Spain was the main reason leading us to develop the current work. Fortunately, we are hereby providing the DFS-SF validated in Spain. Further, the current version uses the Spanish language, which is common to hundreds of millions worldwide, especially in South and Central America. However, we think that the current version may not be used straightforwardly in other Spanish speaking communities without a proper cultural adaptation.
This study has some limitations. Firstly, the QoL measurements could be influenced by the comorbidities present in these patients. Secondly, the test–retest was performed 7 days after treatment was started, and this may affect the reproducibility; for ethical reasons, all patients received treatment from baseline. However, this study has several strengths. We validated the DFS-SF and compared it with that of the EQ-5D, which confers more quality and precision in the validation process [13,14,15,16]. Additionally, this study reports good psychometric properties of the only diabetes-specific questionnaire of QoL used in Spanish patients with DFU. Furthermore, the prospective design allowed assessing the sensitivity to changes regarding the impact of DFU on the QoL.
5. Conclusions
In conclusion, the current version of the DFS-SF in Spanish has good psychometric properties despite the construct validity not being optimal. This questionnaire is a sensitive tool with good performance to capture ulcer changes over time. Finally, the Spanish version of the DFS-SF can be readily available for assessing the QoL in subjects with DFU in Spain. Further, this tool will be useful in clinical and research settings.
Acknowledgments
We particularly acknowledge the patients from University Hospital Arnau de Vilanova of Lleida for their collaboration.
Supplementary Materials
The following are available online at https://www.mdpi.com/2077-0383/9/8/2497/s1. File S1: Cuestionario Diabetic Foot Ulcer Scale-Short Form (DFS-SF). Table S1: Frequency of healed ulcers, amputations, and deaths of the follow-up visits. Table S2: Confirmatory factor analysis of the different subscales of the Diabetes Foot Ulcer Scale-Short Form (DFS-SF). Table S3: Exploratory factor analysis of the different subscales of the Diabetes Foot Ulcer Scale-Short Form (DFS-SF).
Author Contributions
Conceptualization, D.M.-G., M.D., N.A., J.V., and D.M.; methodology, D.M.-G., M.D., N.A., J.V., and D.M.; validation, D.M.-G., M.D., and N.A.; formal analysis, M.M.-A.; writing—Original draft preparation, D.M.-G., M.M.-A., and M.G.-C.; writing—Review and editing, J.V.-S., M.G.-C., and D.M. All authors have read and agreed to the published version of the manuscript.
Funding
CIBERDEM is an initiative from Instituto de Salud Carlos III (Plan Nacional de I + D + I and Fondo Europeo de Desarrollo Regional). M.G.-C. held a predoctoral fellowship from the Ministerio de Educación, Cultura y Deporte, FPU15/03005. This project was developed within the framework of the Doctorate in the Department of Medicine, Autonomous University of Barcelona.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- 1.Hogg F.R.A., Peach G., Price P., Thompson M.M., Hinchliffe R.J. Measures of health-related quality of life in diabetes-related foot disease: A systematic review. Diabetologia. 2012;55:552–565. doi: 10.1007/s00125-011-2372-5. [DOI] [PubMed] [Google Scholar]
- 2.Brownrigg J.R.W., Davey J., Holt P.J., Davis W.A., Thompson M.M., Ray K.K., Hinchliffe R.J. The association of ulceration of the foot with cardiovascular and all-cause mortality in patients with diabetes: A meta-analysis. Diabetologia. 2012;55:2906–2912. doi: 10.1007/s00125-012-2673-3. [DOI] [PubMed] [Google Scholar]
- 3.Prompers L., Schaper N., Apelqvist J., Edmonds M., Jude E., Mauricio D., Uccioli L., Urbancic V., Bakker K., Holstein P., et al. Prediction of outcome in individuals with diabetic foot ulcers: Focus on the differences between individuals with and without peripheral arterial disease. The EURODIALE Study. Diabetologia. 2008;51:747–755. doi: 10.1007/s00125-008-0940-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jupiter D.C., Thorud J.C., Buckley C.J., Shibuya N. The impact of foot ulceration and amputation on mortality in diabetic patients. I: From ulceration to death, a systematic review. Int. Wound J. 2016;13:892–903. doi: 10.1111/iwj.12404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sekhar M.S., Thomas R.R., Unnikrishnan M.K., Vijayanarayana K., Rodrigues G.S. Impact of diabetic foot ulcer on health-related quality of life: A cross-sectional study. Semin. Vasc. Surg. 2015;28:165–171. doi: 10.1053/j.semvascsurg.2015.12.001. [DOI] [PubMed] [Google Scholar]
- 6.Sämann A., Tajiyeva O., Müller N., Tschauner T., Hoyer H., Wolf G., Müller U.A. Prevalence of the diabetic foot syndrome at the primary care level in Germany: A cross-sectional study. Diabetes Med. 2008;25:557–563. doi: 10.1111/j.1464-5491.2008.02435.x. [DOI] [PubMed] [Google Scholar]
- 7.Spanos K., Saleptsis V., Athanasoulas A., Karathanos C., Bargiota A., Chan P., Giannoukas A.D. Factors Associated with Ulcer Healing and Quality of Life in Patients with Diabetic Foot Ulcer. Angiology. 2017;68:242–250. doi: 10.1177/0003319716651166. [DOI] [PubMed] [Google Scholar]
- 8.Ribu L., Rustøen T., Birkeland K., Hanestad B.R., Paul S.M., Miaskowski C. The Prevalence and Occurrence of Diabetic Foot Ulcer Pain and Its Impact on Health-Related Quality of Life. J. Pain. 2006;7:290–299. doi: 10.1016/j.jpain.2005.12.002. [DOI] [PubMed] [Google Scholar]
- 9.Valensi P., Girod I., Baron F., Moreau-Defarges T., Guillon P. Quality of life and clinical correlates in patients with diabetic foot ulcers. Diabetes Metab. 2005;31:263–271. doi: 10.1016/S1262-3636(07)70193-3. [DOI] [PubMed] [Google Scholar]
- 10.Wukich D.K., Raspovic K.M. Assessing Health-Related Quality of Life in Patients With Diabetic Foot Disease: Why Is It Important and How Can We Improve? The 2017 Roger E. Pecoraro Award Lecture. Diabetes Care. 2018;41:391–397. doi: 10.2337/dci17-0029. [DOI] [PubMed] [Google Scholar]
- 11.Boutoille D., Féraille A., Maulaz D., Krempf M. Quality of life with diabetes-associated foot complications: Comparison between lower-limb amputation and chronic foot ulceration. Foot Ankle Int. 2008;29:1074–1078. doi: 10.3113/FAI.2008.1074. [DOI] [PubMed] [Google Scholar]
- 12.Abetz L., Sutton M., Brady L., McNulty P., Gagnon D.D. The Diabetic Foot Ulcer Scale (DFS): A quality of life instrument for use in clinical trials. Pract. Diabetes Int. 2002;19:167–175. doi: 10.1002/pdi.356. [DOI] [Google Scholar]
- 13.Bann C.M., Fehnel S.E., Gagnon D.D. Development and Validation of the Diabetic Foot Ulcer Scale-Short Form (DFS-SF) Pharmacoeconomics. 2003;21:1277–1290. doi: 10.2165/00019053-200321170-00004. [DOI] [PubMed] [Google Scholar]
- 14.Hui L.F., Yee-Tak Fong D., Yam M., Yuk Ip W. Translation and validation of the chinese diabetic foot ulcer scale - short form. Patient. 2008;1:137–145. doi: 10.2165/01312067-200801020-00009. [DOI] [PubMed] [Google Scholar]
- 15.Kontodimopoulos N., Veniou A., Tentolouris N., Niakas D. Validity and reliability of the greek version of the diabetic foot ulcer scale-short form (DFS-SF) Hormones. 2016;15:394–403. doi: 10.14310/horm.2002.1682. [DOI] [PubMed] [Google Scholar]
- 16.Macioch T., Sobol E., Krakowiecki A., Mrozikiewicz-Rakowska B., Kasprowicz M., Hermanowski T. Health related quality of life in patients with diabetic foot ulceration - translation and Polish adaptation of Diabetic Foot Ulcer Scale short form. Health Qual. Life Outcomes. 2017;15:1–8. doi: 10.1186/s12955-017-0587-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bakker K., Apelqvist J., Lipsky B.A., Van Netten J.J., Schaper N.C. The 2015 IWGDF guidance documents on prevention and management of foot problems in diabetes: Development of an evidence-based global consensus. Diabetes. Metab. Res. Rev. 2016;32:2–6. doi: 10.1002/dmrr.2694. [DOI] [PubMed] [Google Scholar]
- 18.Dòria M., Rosado V., Pacheco L.R., Hernández M., Betriu À., Valls J., Franch-Nadal J., Fernández E., Mauricio D. Prevalence of diabetic foot disease in patients with diabetes mellitus under renal replacement therapy in Lleida, Spain. Biomed Res. Int. 2016;2016:7217586. doi: 10.1155/2016/7217586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Alcubierre N., Rubinat E., Traveset A., Martinez-Alonso M., Hernandez M., Jurjo C., Mauricio D. A prospective cross-sectional study on quality of life and treatment satisfaction in type 2 diabetic patients with retinopathy without other major late diabetic complications. Health Qual. Life Outcomes. 2014;12:131. doi: 10.1186/s12955-014-0131-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Doria M., Viadé J., Palomera E., Pérez R., Lladó M., Costa E., Huguet T., Reverter J.L., Serra-Prat M., Franch-Nadal J., et al. Short-term foot complications in Charcot neuroarthropathy: A retrospective study in tertiary care centres in Spain treatment. Endocrinol. Diabetes y Nutr. 2018;65:479–485. doi: 10.1016/j.endinu.2018.06.004. [DOI] [PubMed] [Google Scholar]
- 21.Hinchliffe R. J., Brownrigg J.R.W., Apelqvist J., Boyko E.J., Fitridge R., Mills J.L., Reekers J., Shearman C.P., Zierler R.E., Schaper N.C., et al. IWGDF guidance on the diagnosis, prognosis and management of peripheral artery disease in patients with foot ulcers in diabetes. Diabetes Metab. Res. Rev. 2016;32:37–44. doi: 10.1002/dmrr.2698. [DOI] [PubMed] [Google Scholar]
- 22.Lipsky B.A., Aragón-Sánchez J., Diggle M., Embil J., Kono S., Lavery L., Senneville E., Urbančič-Rovan V., Van Asten S., Peters E.J.G., et al. IWGDF guidance on the diagnosis and management of foot infections in persons with diabetes. Diabetes Metab. Res. Rev. 2016;32:45–74. doi: 10.1002/dmrr.2699. [DOI] [PubMed] [Google Scholar]
- 23.Ware J., Snow K., Kosinski M., Gandek B. SF-36 Health Survey Manual and Interpretation Guide. The Health Institute, New England Medical Center; Boston, MA, USA: 1993. pp. 1–29. [Google Scholar]
- 24.Ragnarson G., Apelqvist J. Health-related quality of life in patients with diabetes mellitus and foot ulcers. J. Diabetes Complicat. 2000;14:235–241. doi: 10.1016/S1056-8727(00)00133-1. [DOI] [PubMed] [Google Scholar]
- 25.Cronbach L.J. Coefficient alpha and the internal structure of tests. Psychometrika. 1951;16:297–334. doi: 10.1007/BF02310555. [DOI] [Google Scholar]
- 26.Liang M.H., Lew R.A., Stucki G., Fortin P.R., Daltroy L. Measuring clinically important changes with patient-oriented questionnaires. Med. Care. 2002;40:45–51. doi: 10.1097/00005650-200204001-00008. [DOI] [PubMed] [Google Scholar]
- 27.Liang M.H. Longitudinal construct validity: Establishment of clinical meaning in patient evaluative instruments. Med. Care. 2000;38:84–90. doi: 10.1097/00005650-200009002-00013. [DOI] [PubMed] [Google Scholar]
- 28.R Core Team R: A language and environment for statistical computing. [(accessed on 3 June 2020)]. Available online: https://www.r-project.org.
- 29.Ribu L., Birkeland K., Hanestad B.R., Moum T., Rustoen T. A longitudinal study of patients with diabetes and foot ulcers and their health-related quality of life: Wound healing and quality-of-life changes. J. Diabetes Complicat. 2008;22:400–407. doi: 10.1016/j.jdiacomp.2007.06.006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.