Abstract
The recently described severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has infected millions of people, with thousands of fatalities. It has prompted global efforts in research, with focus on the pathophysiology of coronavirus disease-19 (COVID-19), and a rapid surge of publications. COVID-19 has been associated with a myriad of clinical manifestations, including the lungs, heart, kidneys, central nervous system, gastrointestinal system, skin, and blood coagulation abnormalities. The endothelium plays a key role in organ dysfunction associated with severe infection, and current data suggest that it is also involved in SARS-CoV-2-induced sepsis. This critical review aimed to address a possible unifying mechanism underlying the diverse complications of COVID-19: microvascular dysfunction, with emphasis on the renin-angiotensin system. In addition, research perspectives are suggested in order to expand understanding of the pathophysiology of the infection.
Keywords: COVID-19, Microcirculation, Renin-angiotensin system
Introduction
Seven (7) months after the first case descriptions of the coronavirus disease 19 (COVID-19) and the identification of the causal agent [1], the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has been diagnosed in almost 15,000,000 people and claimed >617,000 lives (accessed 22 July 2020 https://coronavirus.jhu.edu/map.html). Global efforts in research, including the pathophysiology of COVID-19, have been undertaken, with a rapid and rarely seen surge of publications related to a single disease, within months [2]. Accordingly, COVID-19 has been associated with a myriad of clinical manifestations, including the lungs [3], heart [4], kidneys [5], central nervous system [6], gastrointestinal system [7], skin [8], and blood coagulation abnormalities [9].
During infection, the endothelium displays an essential role in the physiological adaptive processes, adjusting blood flow to regions of increased metabolic demand and enabling the access of immune defence cells to the site of infection [10]. When activated by sepsis, dysfunctional endothelium can lead to intravascular thrombosis, amplification of the inflammatory process and disturbances in regional blood flow [11]. As a consequence, endothelial dysfunction is considered the main factor leading to organ failure in sepsis [12]. Understanding the molecular pathways involved in vascular pathophysiology during sepsis is a key step towards the development of therapy strategies.
The renin-angiotensin system (RAS) is essential for vascular homeostasis, with actions ranging from plasma volume regulation to vascular tone and inflammation [13,14]. In severe bacterial sepsis, previous studies have demonstrated RAS activation [15,16]. Severe COVID-19 has been viewed as viral sepsis, followed by exuberant cytokine production [17], and also associated with RAS imbalance.
The present review aimed to address microvascular dysfunction as a possible unifying mechanism underlying the diverse manifestations of COVID-19, with emphasis on the interactions with RAS.
COVID-19 as a Microvascular Disease
Microcirculation is well recognised as a component of the response to pathogens, which can be either adaptive or dysfunctional. The physiological microvascular response that occurs in mild to moderate infection characterises most of the classical signs of inflammation (Latin, calor, dolor, rubor, tumor) and is a direct result of cytokine-driven vasodilation at the site of pathogen inoculation [18]. Contrary to this are the extensive abnormalities of endothelial function, which may occur in sepsis, associated with disseminated intravascular coagulation (DIC) [19] and a heterogeneous vasomotor response, where hypotension and shock are associated with microvascular vasoconstriction [20]. Previous evidence of microvascular dysfunction associated with acute viral infection was provided during the influenza A (H1N1) pandemic. Salgado et al. evaluated microcirculation in critical patients with acute lung injury [21]. The degree of microvascular dysfunction correlated with the severity of the disease, as measured by the Sequential Organ Failure Assessment (SOFA) score.
The effects of inflammation on endothelial cells include increased expression of adhesion molecules, stimulated by proinflammatory cytokines and chemokines such as IL-1β and TNF-α, and C-reactive protein, that is produced in response to IL-6, amongst others [22,23]. Thrombotic phenomena, reflecting a close relationship between increased proinflammatory cytokines, may ensue endothelial dysfunction and vascular thrombosis. Patients with severe COVID-19 have displayed the ‘cytokine storm’ [24], in which proinflammatory cytokines are increased, culminating in multiorgan injury. This has been hypothesised as an exaggerated host immune system response, similar to the findings of other severe viral pneumonias such as influenza, avian influenza, and severe acute respiratory syndrome [25,26]. The vicious cycle involving cytokine overproduction and microvascular endothelial injury appears to be an essential factor leading patients with severe COVID-19 to multiple organ failure and even death [27,28]. Thus, therapeutic interventions to mitigate inflammatory organ injury have been proposed, and the beneficial role of corticosteroids for patients with COVID-19 who required respiratory support has recently been shown in the Randomised Evaluation of Covid-19 Therapy (RECOVERY) trial [29]. Interestingly, another effect of corticosteroids, via mineralocorticoid receptor binding, has also been suggested in COVID-19, with possible interplay with RAS [30].
Most current studies of COVID-19 are focussed on the macrocirculation, evaluating systemic arterial pressure, the use of vasopressors, and systemic markers of impaired tissue perfusion such as lactate. As a consequence, the reported prevalence of septic shock in adults with a diagnosis of COVID-19 ranges between 20–35%, according to the population studied, the severity of the patient sample and the definition of septic shock that is used [31,32].
Notwithstanding, mounting evidence suggests that microcirculation is a key player of SARS-CoV-2-induced pathophysiology. The angiotensin-converting enzyme type 2 (ACE2) is described as the receptor required for the viral particle cell entry [33]. This process involves priming of the viral surface spike (S) protein by the host cell serine protease TMPRSS2, as a prerequisite for cell and viral membrane fusion [33]. In addition, viral S protein has been demonstrated to induce downregulation of ACE2 [34]. These findings, together with the previous demonstration of ACE2 and TMPRSS2 expression in endothelial cells [35,36], along with other molecules involved in viral infection [37], provide a pathophysiological mechanism to support the hypothesis of direct viral infection of endothelial cells.
Recent autopsy studies have offered histological evidence of SARS-CoV-2 inside human endothelial cells. The study by Varga et al. evaluated post-mortem samples obtained from three severe COVID-19 patients. In one of them, electron microscopy revealed the presence of viral inclusions in kidney endothelial cells. In the other two patients, light microscopy results suggestive of endotheliitis (characterised by an accumulation of inflammatory cells associated with the endothelium, as well as apoptotic bodies) were found in samples recovered from the lungs, heart, kidneys, and small intestine [38]. The case-report study by Paniz-Mondolfi et al. analysed post-mortem specimens from the frontal lobe of a 74-year-old male patient with SARS-CoV-2 infection diagnosed by a positive nasopharyngeal swab test by real-time reverse transcription-polymerase chain reaction (RT-PCR). The transmission electron microscopy sections revealed the presence of viral particles within cytoplasmic vacuoles of brain capillary endothelial cells [39]. In addition, in the study by Menter et al., autopsy specimens obtained from the kidneys of two COVID-19 patients were analysed with electron microscopy, finding virus-like particles inside cytoplasmic vesicles of activated glomerular endothelial cells [40].
Recently, Damiani et al. presented the first in vivo evaluation of the microcirculation in patients with SARS-CoV-2 severe pneumonia [41]. The authors studied sublingual microcirculation, finding an inverse correlation between perfused vessel density and D-dimer levels. This study provided a direct association between human microvascular dysfunction and coagulopathy in severe COVID-19.
Thrombotic Disease in COVID-19
A second significant aspect of SARS-CoV-2-induced endothelial dysfunction is a procoagulant state. This phenomenon has important consequences on the pulmonary complications of COVID-19. Post-mortem examination of COVID-19 elderly, hypertensive, obese, and male patients has shown that impaired microcirculation was characterised by pulmonary capillary alterations and the presence of capillary microthrombi [40].
The association between microvascular disease and hypercoagulability (expressed by increased D-dimer) has previously been recognised in patients with traditional cardiovascular risk factors, including diabetes [42,43]. Additionally, hypercoagulability has been shown to accompany the progression of atherosclerosis-associated endothelial dysfunction [44]. Increased D-dimer levels are frequently found among patients with COVID-19, especially in those with severe disease, and are associated with worse prognosis, including increased risk of intensive care admission, mechanical ventilation and death [[45], [46], [47]]. For these reasons, D-dimer monitoring may be considered valuable in decision-making and patient care, as lower mortality has been reported in patients with increased D-dimer who were treated with heparin, compared with those who were not [48].
Intravascular thrombosis has been reported in the macrocirculation and microcirculation in COVID-19. A pulmonary thromboembolic disorder may play an important role in the pathophysiology of respiratory failure in COVID-19, either as large-vessel pulmonary thromboembolism (present in 81% of the critically ill patients from the series by Klok et al. [49]) or as microthrombosis. In an autopsy study of 10 patients with COVID-19, Dolhnikoff et al. observed exudative and proliferative diffuse alveolar damage, with intense epithelial viral cytopathic effects involving the alveolar and small airway epithelium, with little lymphocytic infiltration, and presence of fibrinous thrombi in small pulmonary arterioles in areas of both damaged and preserved lung parenchyma in eight out of 10 cases. Endothelial tumefaction and a large number of pulmonary megakaryocytes in the pulmonary capillaries were other indicators of activation of the coagulation cascade. These pathological observations were supportive of the concept of microthrombosis in severe COVID-19 [50].
Escher et al. [51] reported a case of a patient with COVID-19 complicated with acute respiratory distress syndrome (ARDS) and greatly increased von Willebrand factor (VWF), who improved after full anticoagulation. The increased VWF suggested massive endothelial stimulation and damage, with release of VWF. Magro et al. described five individuals with severe COVID-19 and complement-mediated microvascular injury, which was noted in the lungs and/or skin [52]. All patients had ARDS and three also had purpuric skin lesions. Of note, patients did not display classic histopathological features of ARDS, but pulmonary findings included significant deposits of complement components in the microvasculature. The pulmonary abnormalities were largely restricted to alveolar capillaries, indicating a thrombotic microvascular injury that would explain the increased dead space fraction, with respiratory failure accompanied by greater lung compliance and less pulmonary consolidation than in typical ARDS. The skin lesions showed a pattern of thrombogenic vasculopathy, with deposition of C5b-9 and C4d in both grossly involved and normally-appearing skin.
A remarkable clinical finding with important vascular implications in COVID-19 was studied by Liu et al. They described elevated plasma angiotensin II (Ang II) levels in COVID-19 patients when compared with healthy individuals, which positively correlated with more severe lung injury [53]. Although observed in a small sample of patients, these data suggest, in combination with the known procoagulant effects of Ang II (including stimulus of the expression of tissue factor [54]), a possible pathophysiological link between RAS imbalance and COVID-19-related intravascular thrombotic disease.
The proinflammatory actions of Ang II involve innate and adaptive immune responses [14]. Accordingly, mice chronically infused with Ang II display increased vascular oxidative stress and macrophage infiltration, cytokine production and endothelial dysfunction. All of these effects were prevented by an immunologic approach, by infusing the anti-inflammatory regulatory T lymphocytes (Tregs) [55].
RAS Imbalance and SARS-CoV-2 Infection
The RAS is an essential component of vascular homeostasis, where the key enzymes ACE and ACE2 display antagonistic actions. Ang II produced by ACE induces oxidative stress, cell proliferation and inflammation [56]. Conversely, Ang-(1-7), the end product of the enzymatic action of ACE2, is associated with antioxidant and antiinflammatory effects [57].
Renin-angiotensin system imbalance has previously been suggested in bacterial sepsis. Evaluating the plasma of septic patients, Boldt et al. found higher circulating levels of Ang II when compared with controls, which were suppressed with the administration of the ACE inhibitor enalaprilat [15]. These findings were expanded by Doerschug et al., who identified that increased Ang II and plasma renin activity in septic subjects were associated with microvascular dysfunction [16]. Literature investigating the association between viral infection and RAS is scarce. Evaluating patients with chronic hepatitis C infection, Powell et al. found an association between hepatic fibrosis and one polymorphism in the angiotensinogen gene, which was related to increased Ang II synthesis [58].
The modulation of RAS in sepsis has been evaluated as a potential therapy by different approaches in the last decade. In the study with enalaprilat mentioned above [15] with a small number of patients (n=40), there was no difference in survival rate compared with the control group. Akpinar et al. investigated the effects of the renin inhibitor aliskiren in bacterial sepsis-induced acute lung injury in rats [59]. The authors found a reduction in plasma Ang II, which was associated with a reduction of oxidative stress markers and cytokine expression in lung tissue, including IL-6 and TNF-α. Similar results were obtained in rodents with reduced lung injury during sepsis, which was associated with plasma decrease of IL-6 and TNF-α, using the angiotensin receptor blockers (ARBs) losartan [60] and candesartan [61]. ACE2 has also been associated with the potential beneficial effects of pharmacological interventions in RAS. Imai et al. showed that the administration of recombinant ACE2 reduced lung injury in mice during bacterial sepsis [62]. In addition, the administration of ACE inhibitors and ARBs increased the cardiac expression of ACE2 and plasma Ang-(1-7) in mice [63]. Indirect clinical evidence of the potential benefits of RAS modulation in infection was provided by a meta-analysis of 37 studies, which evaluated the effects of ACE inhibitors and ARBs in the risk of pneumonia [64]. The authors found that ACE inhibitors reduced the risk of pneumonia and pneumonia-related mortality, a benefit that was extended to high-risk patients, including those with heart failure and stroke.
Evidence suggesting a coronavirus-induced imbalance between ACE and ACE2 actions was previously provided by Kuba et al., who demonstrated, in the airways of mice that had been exposed to the related virus SARS-CoV, that ACE2 downregulation was associated with a local increase of Ang II [34]. Clinical studies with COVID-19 patients have demonstrated that blood levels of TNF-α and IL-6 are increased and positively correlate with disease severity [65]. Both cytokines are associated with endothelial barrier breakdown in sepsis [66] and reduced by Ang-(1-7) [67].
This working hypothesis of the vascular effects of SARS-CoV-2 is even more relevant in view of clinical evidence from cohorts in at least three continents identifying cardiovascular disease and particularly hypertension as important risk factors associated with worse prognosis of COVID-19 [[68], [69], [70]]. Indeed, in hypertensive patients, RAS was found to be imbalanced towards ACE activation [71], with ACE and Ang II expressed in higher levels in vascular tissues of patients exposed to cardiovascular risk factors [72].
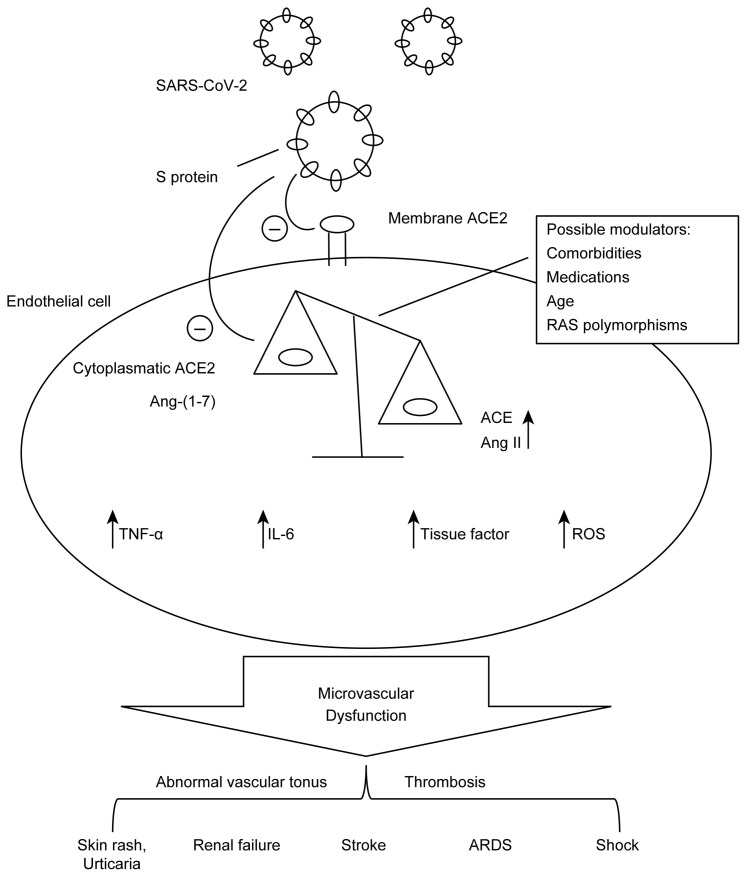
Considering the preliminary and fast growing amount of available experimental and clinical evidence, endothelial dysfunction may occur as a direct effect of the tropism of SARS-CoV-2 for vascular tissue, inducing or potentiating a previous imbalance (as in cardiovascular and metabolic disease patients) of intracellular RAS. These phenomena, together with pre-existing endothelial injury that accompanies comorbidities – such as hypertension, diabetes, coronary artery disease, and obesity – may predispose to more severe presentations, faster evolution and worse prognosis of the disease. As the ultimate result, extensive endothelial cell damage ensues, with organ dysfunction and circulatory collapse. A schematic representation of the mechanisms proposed for COVID-19 pathophysiology regarding RAS and microcirculation is shown in Figure 1 .
Figure 1.
A schematic representation of mechanisms proposed for COVID-19 pathophysiology regarding the renin-angiotensin system.
Abbreviations: ACE, angiotensin-converting enzyme; ACE2, angiotensin-converting enzyme type 2; Ang-(1-7), angiotensin-(1-7); Ang II, angiotensin II; ARDS, acute respiratory distress syndrome; IL-6, Interleukin-6; RAS, renin-angiotensin system; ROS, reactive oxygen species; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; S protein, spike protein; TNF-α, tumor necrosis factor-α.
Research Perspectives
The interplay between RAS and the microcirculation in SARS-CoV-2 infection brings up the opportunity of exciting research perspectives. It is vital to understand the dynamics of microcirculatory function during the clinical evolution of COVID-19, with particular interest in the largely unknown initial oligosymptomatic stage. Early signs of endothelial dysfunction were previously identified in human microcirculation, days before the clinical diagnosis of bacterial disease [73]. Indeed, a timely therapeutic action in the early phase is a key principle of sepsis management [74]. As a consequence, microcirculation evaluation, which can be performed at the bedside with non-invasive methods [75], may offer important clinical data regarding prognosis and treatment effects [76].
Another important issue in current comprehension of COVID-19 is to further investigate preliminary reports of young patients without known risk factors presenting severe disease, with vascular manifestations including stroke [77]. A number of pathophysiological mechanisms are proposed regarding this complication and should be subject of future research. These include direct central nervous system endothelial infection [39], cytokine-induced local microcirculatory dysfunction [78], and the development of antiphospholipid antibodies [79].
It was previously known that ACE polymorphisms are associated with vascular complications such as diabetic nephropathy [80]. Furthermore, combinations of polymorphisms of both ACE and ACE2 have been associated with the susceptibility of developing hypertension [81]. Another recent finding was reported by Bunyavanich et al., who found an age-dependent expression of ACE2 in nasal epithelial cells of COVID-19 patients, with adults displaying higher levels than children [82]. To quantitatively or qualitatively evaluate RAS-related genes and peptides in COVID-19 patients and their association with disease progression are attractive research possibilities.
The contribution of RAS in COVID-19 is also being studied with therapeutic goals. There are 33 ongoing studies currently registered in clinicaltrials.gov evaluating interventions exploring the RAS axis, including ACE inhibitors, ARBs and Ang-(1-7) as treatment strategies (accessed 22 July 2020 at https://clinicaltrials.gov/). The imbalance of Ang II/Ang-(1-7) favouring the former in face of reduction of ACE2 activity after binding of the coronavirus suggests the possibility of beneficial effects of blockade of the angiotensin type I receptor with angiotensin receptor blockers.
Conclusion
At present, the scientific community is beginning to understand the pathophysiology of COVID-19. The RAS is a powerful homeostatic system, developed in the evolutionary process hundreds of millions of years ago, allowing salt and water retention essential for vertebrates once they left their original marine environment [83], with diverse fundamental roles in health and disease. Traditionally studied in the context of chronic cardiovascular diseases, a renewed interest in RAS inflammatory and microvascular actions has been prompted by the COVID-19 pandemic. Understanding these mechanisms can lead to more effective treatments for control of this global pandemic.
Funding Sources
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing Interest Statement
The authors declare that they have no competing interests.
Acknowledgements
All above authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
References
- 1.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Haghani M., Bliemer M.C.J., Goerlandt F., Li J. The scientific literature on coronaviruses, COVID-19 and its associated safety-related research dimensions: a scientometric analysis and scoping review. Saf Sci. 2020;129:104806. doi: 10.1016/j.ssci.2020.104806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hu Q., Guan H., Sun Z., Huang L., Chen C., Ai T., et al. Early CT features and temporal lung changes in COVID-19 pneumonia in Wuhan, China. Eur J Radiol. 2020;128:109017. doi: 10.1016/j.ejrad.2020.109017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tomasoni D., Italia L., Adamo M., Inciardi R.M., Lombardi C.M., Solomon S.D., et al. COVID 19 and heart failure: from infection to inflammation and angiotensin II stimulation. Searching for evidence from a new disease. Eur J Heart Fail. 2020;22:957–966. doi: 10.1002/ejhf.1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ng J.J., Luo Y., Phua K., Choong A. Acute kidney injury in hospitalized patients with coronavirus disease 2019 (COVID-19): a meta-analysis. J Infect. 2020 doi: 10.1016/j.jinf.2020.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020 doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee I.C., Huo T.I., Huang Y.H. Gastrointestinal and liver manifestations in patients with COVID-19. J Chin Med Assoc. 2020 doi: 10.1097/JCMA.0000000000000319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J Eur Acad Dermatol Venereol. 2020 doi: 10.1111/jdv.16387. [DOI] [PubMed] [Google Scholar]
- 9.Bikdeli B., Madhavan M.V., Jimenez D., Chuich T., Dreyfus I., Driggin E., et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up. J Am Coll Cardiol. 2020 doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bermejo-Martin J.F., Martin-Fernandez M., Lopez-Mestanza C., Duque P., Almansa R. Shared features of endothelial dysfunction between sepsis and its preceding risk factors (aging and chronic disease) J Clin Med. 2018;7:400. doi: 10.3390/jcm7110400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aird W.C. The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood. 2003;101:3765–3777. doi: 10.1182/blood-2002-06-1887. [DOI] [PubMed] [Google Scholar]
- 12.Ince C., Mayeux P.R., Nguyen T., Gomez H., Kellum J.A., Ospina-Tascon G.A., et al. The endothelium in sepsis. Shock. 2016;45:259–270. doi: 10.1097/SHK.0000000000000473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ferrario C.M. Role of angiotensin II in cardiovascular disease therapeutic implications of more than a century of research. J Renin Angiotensin Aldosterone Syst. 2006;7:3–14. doi: 10.3317/jraas.2006.003. [DOI] [PubMed] [Google Scholar]
- 14.Schiffrin E.L. Immune mechanisms in hypertension and vascular injury. Clin Sci (Lond) 2014;126:267–274. doi: 10.1042/CS20130407. [DOI] [PubMed] [Google Scholar]
- 15.Boldt J., Papsdorf M., Kumle B., Piper S., Hempelmann G. Influence of angiotensin-converting enzyme inhibitor enalaprilat on endothelial-derived substances in the critically ill. Crit Care Med. 1998;26:1663–1670. doi: 10.1097/00003246-199810000-00018. [DOI] [PubMed] [Google Scholar]
- 16.Doerschug K.C., Delsing A.S., Schmidt G.A., Ashare A. Renin-angiotensin system activation correlates with microvascular dysfunction in a prospective cohort study of clinical sepsis. Crit Care. 2010;14:R24. doi: 10.1186/cc8887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ye Q., Wang B., Mao J. The pathogenesis and treatment of the ‘cytokine storm’ in COVID-19. J Infect. 2020;80:607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hatoum O.A., Binion D.G., Otterson M.F., Gutterman D.D. Acquired microvascular dysfunction in inflammatory bowel disease: loss of nitric oxide-mediated vasodilation. Gastroenterology. 2003;125:58–69. doi: 10.1016/s0016-5085(03)00699-1. [DOI] [PubMed] [Google Scholar]
- 19.Levi M., van der Poll T., Schultz M. Systemic versus localized coagulation activation contributing to organ failure in critically ill patients. Semin Immunopathol. 2011;34:167–179. doi: 10.1007/s00281-011-0283-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tyml K. Role of connexins in microvascular dysfunction during inflammation. Can J Physiol Pharmacol. 2010;89:1–12. doi: 10.1139/y10-099. [DOI] [PubMed] [Google Scholar]
- 21.Salgado D.R., Ortiz J.A., Favory R., Creteur J., Vincent J.L., De Backer D. Microcirculatory abnormalities in patients with severe influenza A (H1N1) infection. Can J Anaesth. 2010;57:940–946. doi: 10.1007/s12630-010-9365-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang C. The role of inflammatory cytokines in endothelial dysfunction. Basic Res Cardiol. 2008;103:398–406. doi: 10.1007/s00395-008-0733-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Szmitko P.E., Wang C.H., Weisel R.D., de Almeida J.R., Anderson T.J., Verma S. New markers of inflammation and endothelial cell activation: Part I. Circulation. 2003;108:1917–1923. doi: 10.1161/01.CIR.0000089190.95415.9F. [DOI] [PubMed] [Google Scholar]
- 24.Moore J.B., June C.H. Cytokine release syndrome in severe COVID-19. Science. 2020;368:473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- 25.de Jong M.D., Simmons C.P., Thanh T.T., Hien V.M., Smith G.J., Chau T.N., et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med. 2006;12:1203–1207. doi: 10.1038/nm1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wong C.K., Lam C.W., Wu A.K., Ip W.K., Lee N.L., Chan I.H., et al. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin Exp Immunol. 2004;136:95–103. doi: 10.1111/j.1365-2249.2004.02415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li H., Liu L., Zhang D., Xu J., Dai H., Tang N., et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395:1517–1520. doi: 10.1016/S0140-6736(20)30920-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Colantuoni A., Martini R., Caprari P., Ballestri M., Capecchi P.L., Gnasso A., et al. COVID-19 sepsis and microcirculation dysfunction. Front Physiol. 2020;11:747. doi: 10.3389/fphys.2020.00747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Horby P., Lim W.S., Emberson J.R., Mafham M., Bell J.L., Linsell L., et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N Engl J Med. 2020 doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liaudet L., Szabo C. Blocking mineralocorticoid receptor with spironolactone may have a wide range of therapeutic actions in severe COVID-19 disease. Crit Care. 2020;24:318. doi: 10.1186/s13054-020-03055-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang X., Yu Y., Xu J., Shu H., Xia J., Liu H., et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020 doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020 doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B., et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11:875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Aimes R.T., Zijlstra A., Hooper J.D., Ogbourne S.M., Sit M.L., Fuchs S., et al. Endothelial cell serine proteases expressed during vascular morphogenesis and angiogenesis. Thromb Haemost. 2003;89:561–572. [PubMed] [Google Scholar]
- 36.Li Y., Cao Y., Zeng Z., Liang M., Xue Y., Xi C., et al. Angiotensin-converting enzyme 2/angiotensin-(1-7)/Mas axis prevents lipopolysaccharide-induced apoptosis of pulmonary microvascular endothelial cells by inhibiting JNK/NF-kappaB pathways. Sci Rep. 2015;5:8209. doi: 10.1038/srep08209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sardu C., Gambardella J., Morelli M.B., Wang X., Marfella R., Santulli G. Hypertension, thrombosis, kidney failure, and diabetes: is COVID-19 an endothelial disease? a comprehensive evaluation of clinical and basic evidence. J Clin Med. 2020;9:1417. doi: 10.3390/jcm9051417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Paniz-Mondolfi A., Bryce C., Grimes Z., Gordon R.E., Reidy J., Lednicky J., et al. Central nervous system involvement by severe acute respiratory syndrome coronavirus -2 (SARS-CoV-2) J Med Virol. 2020 doi: 10.1002/jmv.25915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Menter T., Haslbauer J.D., Nienhold R., Savic S., Hopfer H., Deigendesch N., et al. Post-mortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings of lungs and other organs suggesting vascular dysfunction. Histopathology. 2020 doi: 10.1111/his.14134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Damiani E., Carsetti A., Casarotta E., Scorcella C., Domizi R., Adrario E., et al. Microvascular alterations in patients with SARS-COV-2 severe pneumonia. Ann Intensive Care. 2020;10:60. doi: 10.1186/s13613-020-00680-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.El Asrar M.A., Adly A.A., El Hadidy E.S., Abdelwahab M.A. D-dimer levels in type 1 and type 2 diabetic children and adolescents; relation to microvascular complications and dyslipidemia “own data and review”. Pediatr Endocrinol Rev. 2012;9:657–668. [PubMed] [Google Scholar]
- 43.Domingueti C.P., Dusse L.M., Foscolo R.B., Reis J.S., Annichino-Bizzacchi J.M., Orsi F.L., et al. Von Willebrand Factor, ADAMTS13 and D-Dimer are correlated with different levels of nephropathy in type 1 diabetes mellitus. PLoS One. 2015;10:e0132784. doi: 10.1371/journal.pone.0132784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Astrup A.S., Tarnow L., Pietraszek L., Schalkwijk C.G., Stehouwer C.D., Parving H.H., et al. Markers of endothelial dysfunction and inflammation in type 1 diabetic patients with or without diabetic nephropathy followed for 10 years: association with mortality and decline of glomerular filtration rate. Diabetes Care. 2008;31:1170–1176. doi: 10.2337/dc07-1960. [DOI] [PubMed] [Google Scholar]
- 45.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lippi G., Favaloro E.J. D-dimer is associated with severity of coronavirus disease 2019: a pooled analysis. Thromb Haemost. 2020;120:876–878. doi: 10.1055/s-0040-1709650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tang N., Bai H., Chen X., Gong J., Li D., Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18:1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Klok F.A., Kruip M., van der Meer N.J.M., Arbous M.S., Gommers D., Kant K.M., et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020 doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dolhnikoff M., Duarte-Neto A.N., de Almeida Monteiro R.A., Ferraz da Silva L.F., Pierre de Oliveira E., Nascimento Saldiva P.H., et al. Pathological evidence of pulmonary thrombotic phenomena in severe COVID-19. J Thromb Haemost. 2020 doi: 10.1111/jth.14844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Escher R., Breakey N., Lammle B. Severe COVID-19 infection associated with endothelial activation. Thromb Res. 2020;190:62. doi: 10.1016/j.thromres.2020.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Magro C., Mulvey J.J., Berlin D., Nuovo G., Salvatore S., Harp J., et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020 doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J., et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63:364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Celi A., Cianchetti S., Dell'Omo G., Pedrinelli R. Angiotensin II, tissue factor and the thrombotic paradox of hypertension. Expert Rev Cardiovasc Ther. 2010;8:1723–1729. doi: 10.1586/erc.10.161. [DOI] [PubMed] [Google Scholar]
- 55.Barhoumi T., Kasal D.A., Li M.W., Shbat L., Laurant P., Neves M.F., et al. T regulatory lymphocytes prevent angiotensin II-induced hypertension and vascular injury. Hypertension. 2011;57:469–476. doi: 10.1161/HYPERTENSIONAHA.110.162941. [DOI] [PubMed] [Google Scholar]
- 56.Panico K., Abrahao M.V., Trentin-Sonoda M., Muzi-Filho H., Vieyra A., Carneiro-Ramos M.S. Cardiac inflammation after ischemia-reperfusion of the kidney: role of the sympathetic nervous system and the renin-angiotensin system. Cell Physiol Biochem. 2019;53:587–605. doi: 10.33594/000000159. [DOI] [PubMed] [Google Scholar]
- 57.Santos R.A.S., Sampaio W.O., Alzamora A.C., Motta-Santos D., Alenina N., Bader M., et al. The ACE2/Angiotensin-(1-7)/MAS axis of the renin-angiotensin system: focus on angiotensin-(1-7) Physiol Rev. 2018;98:505–553. doi: 10.1152/physrev.00023.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Powell E.E., Edwards-Smith C.J., Hay J.L., Clouston A.D., Crawford D.H., Shorthouse C., et al. Host genetic factors influence disease progression in chronic hepatitis C. Hepatology. 2000;31:828–833. doi: 10.1053/he.2000.6253. [DOI] [PubMed] [Google Scholar]
- 59.Akpinar E., Halici Z., Cadirci E., Bayir Y., Karakus E., Calik M., et al. What is the role of renin inhibition during rat septic conditions: preventive effect of aliskiren on sepsis-induced lung injury. Naunyn Schmiedebergs Arch Pharmacol. 2014;387:969–978. doi: 10.1007/s00210-014-1014-0. [DOI] [PubMed] [Google Scholar]
- 60.Shen L., Mo H., Cai L., Kong T., Zheng W., Ye J., et al. Losartan prevents sepsis-induced acute lung injury and decreases activation of nuclear factor kappaB and mitogen-activated protein kinases. Shock. 2009;31:500–506. doi: 10.1097/SHK.0b013e318189017a. [DOI] [PubMed] [Google Scholar]
- 61.Nitescu N., Grimberg E., Guron G. Low-dose candesartan improves renal blood flow and kidney oxygen tension in rats with endotoxin-induced acute kidney dysfunction. Shock. 2008;30:166–172. doi: 10.1097/shk.0b013e31815dd780. [DOI] [PubMed] [Google Scholar]
- 62.Imai Y., Kuba K., Rao S., Huan Y., Guo F., Guan B., et al. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ferrario C.M., Jessup J., Chappell M.C., Averill D.B., Brosnihan K.B., Tallant E.A., et al. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 64.Caldeira D., Alarcao J., Vaz-Carneiro A., Costa J. Risk of pneumonia associated with use of angiotensin converting enzyme inhibitors and angiotensin receptor blockers: systematic review and meta-analysis. BMJ. 2012;345:e4260. doi: 10.1136/bmj.e4260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jose R.J., Manuel A. COVID-19 cytokine storm: the interplay between inflammation and coagulation. Lancet Respir Med. 2020 doi: 10.1016/S2213-2600(20)30216-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Radeva M.Y., Waschke J. Mind the gap: mechanisms regulating the endothelial barrier. Acta Physiol (Oxf) 2018;222 doi: 10.1111/apha.12860. [DOI] [PubMed] [Google Scholar]
- 67.Souza L.L., Costa-Neto C.M. Angiotensin-(1-7) decreases LPS-induced inflammatory response in macrophages. J Cell Physiol. 2012;227:2117–2122. doi: 10.1002/jcp.22940. [DOI] [PubMed] [Google Scholar]
- 68.Guan W.J., Ni Z.Y., Hu Y., Liang W.H., Ou C.Q., He J.X., et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020 doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Grasselli G., Zangrillo A., Zanella A., Antonelli M., Cabrini L., Castelli A., et al. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy region, Italy. JAMA. 2020 doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Goyal P., Choi J.J., Pinheiro L.C., Schenck E.J., Chen R., Jabri A., et al. Clinical characteristics of Covid-19 in New York city. N Engl J Med. 2020 doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Reja V., Goodchild A.K., Phillips J.K., Pilowsky P.M. Upregulation of angiotensin AT1 receptor and intracellular kinase gene expression in hypertensive rats. Clin Exp Pharmacol Physiol. 2006;33:690–695. doi: 10.1111/j.1440-1681.2006.04420.x. [DOI] [PubMed] [Google Scholar]
- 72.Ancion A., Tridetti J., Nguyen Trung M.L., Oury C., Lancellotti P. A review of the role of bradykinin and nitric oxide in the cardioprotective action of angiotensin-converting enzyme inhibitors: focus on perindopril. Cardiol Ther. 2019;8:179–191. doi: 10.1007/s40119-019-00150-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Weidlich K., Kroth J., Nussbaum C., Hiedl S., Bauer A., Christ F., et al. Changes in microcirculation as early markers for infection in preterm infants--an observational prospective study. Pediatr Res. 2009;66:461–465. doi: 10.1203/PDR.0b013e3181b3b1f6. [DOI] [PubMed] [Google Scholar]
- 74.Alhazzani W., Moller M.H., Arabi Y.M., Loeb M., Gong M.N., Fan E., et al. Surviving sepsis Campaign: Guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19) Crit Care Med. 2020 doi: 10.1097/CCM.0000000000004363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cracowski J.L., Roustit M. Current methods to assess human cutaneous blood flow: an updated focus on laser-based-techniques. Microcirculation. 2016;23:337–344. doi: 10.1111/micc.12257. [DOI] [PubMed] [Google Scholar]
- 76.Tibirica E., De Lorenzo A. Importance of the evaluation of systemic microvascular flow and reactivity in critically ill patients with coronavirus disease 2019 - COVID-19. Microvasc Res. 2020;131:104028. doi: 10.1016/j.mvr.2020.104028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Oxley T.J., Mocco J., Majidi S., Kellner C.P., Shoirah H., Singh I.P., et al. Large-vessel stroke as a presenting feature of COVID-19 in the young. N Engl J Med. 2020;382:e60. doi: 10.1056/NEJMc2009787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Divani A.A., Andalib S., Di Napoli M., Lattanzi S., Hussain M.S., Biller J., et al. Coronavirus disease 2019 and stroke: clinical manifestations and pathophysiological insights. J Stroke Cerebrovasc Dis. 2020;29 doi: 10.1016/j.jstrokecerebrovasdis.2020.104941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhang Y., Xiao M., Zhang S., Xia P., Cao W., Jiang W., et al. Coagulopathy and antiphospholipid antibodies in patients with Covid-19. N Engl J Med. 2020;382:e38. doi: 10.1056/NEJMc2007575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sayed-Tabatabaei F.A., Oostra B.A., Isaacs A., van Duijn C.M., Witteman J.C. ACE polymorphisms. Circ Res. 2006;98:1123–1133. doi: 10.1161/01.RES.0000223145.74217.e7. [DOI] [PubMed] [Google Scholar]
- 81.Pinheiro D.S., Santos R.S., Jardim P., Silva E.G., Reis A.A.S., Pedrino G.R., et al. The combination of ACE I/D and ACE2 G8790A polymorphisms revels susceptibility to hypertension: a genetic association study in Brazilian patients. PLoS One. 2019;14:e0221248. doi: 10.1371/journal.pone.0221248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bunyavanich S., Do A., Vicencio A. Nasal gene expression of angiotensin-converting enzyme 2 in children and adults. JAMA. 2020 doi: 10.1001/jama.2020.8707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Fournier D., Luft F.C., Bader M., Ganten D., Andrade-Navarro M.A. Emergence and evolution of the renin-angiotensin-aldosterone system. J Mol Med (Berl) 2012;90:495–508. doi: 10.1007/s00109-012-0894-z. [DOI] [PMC free article] [PubMed] [Google Scholar]