Abstract
We report the case of a 38-year-old man who presented to the emergency department with fever, myalgia, nausea, vomiting, dry cough, breathlessness and abdominal pain. He was admitted due to hypoxaemia and was diagnosed with SARS-CoV-2 and was subsequently referred to the intensive care unit for intubation and mechanical ventilation. Severe rhabdomyolysis and acute kidney injury developed 4 days later and were suspected after noticing discolouration of the urine and a marked increase in plasma myoglobin levels. Treatment included hydration, forced diuresis and continuous renal replacement therapy. In addition to the coronavirus disease acute respiratory distress syndrome, he was diagnosed with possible SARS-CoV-2-induced myositis with severe rhabdomyolysis and kidney failure. The patient survived and was discharged from intensive care after 12 days, returning home 23 days after hospitalisation, fully mobilised with a partially restored kidney function.
Keywords: infectious diseases, adult intensive care
Background
Currently, there is an unprecedented surge in the demand for intensive care resources throughout the world due to the COVID-19 pandemic.1 2 Little is known about rhabdomyolysis and acute kidney injury (AKI) in the context of the COVID-19 infection in the intensive care setting.3 4 Recent reports suggest that AKI during COVID-19 infection could be associated with increased mortality.5 Due to a scarcity of reports available on the subject, we decided to present the symptoms, laboratory findings, clinical course and treatment of a patient in intensive care who developed severe rhabdomyolysis and AKI during the course of the COVID-19 infection.
Case presentation
A 38-year-old man with type 2 diabetes, gout and mild obesity presented to the emergency department with a 1-week history of fever, myalgia, nausea, vomiting, dry cough, breathlessness and abdominal pain. He was brought in by ambulance but was conscious. At the time of admission, his oxygen saturation was 91% and other vital signs were normal. The suspicion of SARS-CoV-2 infection was high at the time of admission due to the presence of the classic symptoms in combination with a history of potential SARS-CoV-2 exposure. The patient had no history of statin use, drug abuse, immobilisation or trauma (well-known causes of rhabdomyolysis) prior to admission. After laboratory verification of the SARS-CoV-2 diagnosis, the patient was transferred from a neighbouring hospital to the infectious diseases clinic. Within 24 hours of admission, the patient’s arterial oxygenation worsened, and he was admitted to the intensive care unit (ICU) for further management. Despite supplemental oxygen (15 L on reservoir mask), the respiratory rate remained high (40 per minute), and an arterial blood gas showed hypoxaemia (pO2 8.0 kPa (reference 11.1–14.4 kPa)) without hypercapnia (pCO2 5.1 (reference 4.6–6.0 kPa)). CT of the chest revealed extensive bilateral ground-glass opacities (figure 1.) Following intubation and ventilation, his vital signs stabilised. Cefotaxime was initiated at the time of hospital admission and was supplemented with metronidazole on day 5 due to a gingival infection. Analgosedation was maintained with propofol, fentanyl and clonidine throughout. Muscle relaxation was used only during the endotracheal intubation procedure.
Figure 1.
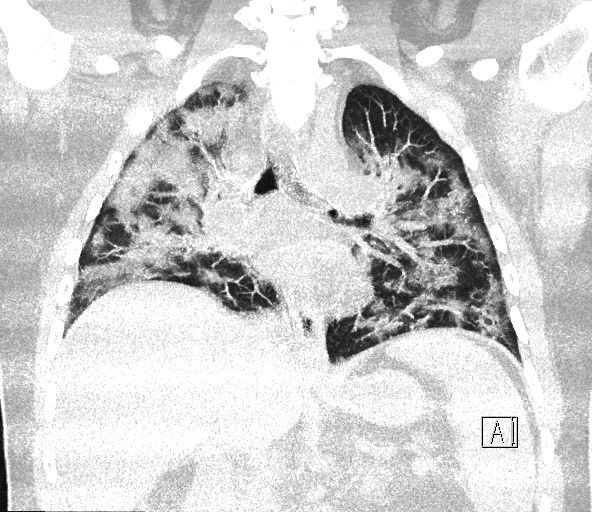
CT of the patient’s lungs showing extensive bilateral ground-glass opacities.
On day 4 in the ICU, the urine became tea-coloured, myoglobin levels gradually increased (from normal levels on admission to a peak level of >21 000 µg/L) and the kidney function deteriorated (lowest recorded relative glomerular filtration rate of 18 mL/min/1.73 m2). The rhabdomyolysis was preceded by several days of very high body temperatures (39–40°C) unresponsive to treatment with paracetamol and active external cooling. Continuous renal replacement therapy was initiated through a femoral central venous catheter on the fifth day and continued for 7 more days. The patient was treated for moderate acute respiratory distress syndrome (ARDS) and successfully weaned from the ventilator and extubated 12 days after hospitalisation. Troponin T levels increased markedly on day 12 without signs of ECG abnormalities. Myocarditis was suspected based on laboratory findings, but echocardiography revealed a normal heart function (ejection fraction >60%). Intermittent haemodialysis following intensive care was not required, and the patient was discharged from the hospital after 23 days.
Investigations
Several biochemical tests were performed throughout the intensive care period. See table 1 for an outline of key test results.
Table 1.
Biochemical test results and reference values during the patient’s admission to intensive care
| ICU admission | ICU day 5 | ICU discharge | ||
| Reference value | ||||
| B-haemoglobin | 123 | 115 | 79 | 134–170 g/L |
| B-leucocytes | 3.9 | 9.5 | 9.4 | 3.5–8.8 × 109/L |
| B-lymphocytes | 0.74 | 1.36 | 0.82 | 1.10–3.50 × 109/L |
| P-PK (INR) | 1.0 | 1.3 | 1.2 | 0.9–1.2 |
| P-APT-time | 36 | 40 | 81 | 24–33 s |
| P-D-dimer | 0.19 | 0.27 | 1.78 | <0.25 mg/L |
| S-creatinine | 51 | 161 | 278 | 60–105 µmol/L |
| P-sodium | 136 | 147 | 139 | 137–145 mmol/L |
| P-potassium | 3.6 | 4.6 | 5.3 | 3.6–4.6 mmol/L |
| S-urea | 2.7 | 12.3 | 28.7 | 3.2–8.1 mmol/L |
| S-CRP | 145 | 298 | 54 | <5 mg/L |
| P-procalcitonin | 0.16 | 9.7 | 4.3 | <0.05 µg/L |
| P-LD | 7.0 | 8.9 | 8.5 | <3.5 µkat/L |
| P-lactate (art) | 0.9 | 1.3 | 1.4 | 0.5–1.6 mmol/L |
| P-myoglobin | 44 | 11 441 | 6254 | <70 µg/L |
APT, activated partial thromboplastin; B, blood; CRP, C-reactive protein; ICU, intensive care unit; PK (INR), prothrombin complex (international normalised ratio); LD, lactate dehydrogenase; P, plasma; S, serum.
Differential diagnosis
The main differential diagnosis to the patient’s rhabdomyolysis was an acute ischaemic event in the extremities. The patient was thoroughly assessed for signs and symptoms of arterial embolisation and limb ischaemia, but no such suspicion could be verified. In addition, there was serious concern regarding the sudden, unexpected and asymptomatic increase in troponin T levels (from 8 ng/L to 444 ng/L) on the days following ICU discharge. On one hand, troponin T levels indicated significant myocardial damage, but diagnostic criteria for neither myocardial infarction nor perimyocarditis were fulfilled. On the other hand, troponin T levels may be falsely high in the setting of AKI.
Treatment
The initial treatment of the patient’s ARDS consisted of oxygen on a reservoir mask. In intensive care, endotracheal intubation followed sedation and muscle relaxation with ketamine and rocuronium, respectively. We used a lung-protective ventilation approach with a pressure-controlled mode. The patient was weaned from the ventilator using pressure-support ventilation. The patient received treatment against pulmonary and gingival infections using cefotaxime and metronidazole, respectively. Throughout intensive care, nutritional support was provided both intravenously and through a nasogastric tube. When myoglobinaemia was diagnosed on the fourth day in the ICU, an attempt was made to increase the diuresis to reduce the risk of AKI. Balanced crystalloids and albumin were administered together with furosemide, potassium canrenoate, spironolactone and mannitol. Despite these efforts, plasma myoglobin and serum creatinine continued to increase and continuous venovenous haemodiafiltration was initiated on the fifth day and continued until discharge from the ICU 7 days later. Following discharge from the ICU, the patient received physiotherapy and his muscular strength improved.
Outcome and follow-up
On the day of the hospital discharge, the patient was completely mobile and cognitively intact with a relative glomerular filtration rate of 48 mL/min/1.73 m2. Nine days after discharge, the kidney function had completely normalised.
Within 2 days of discharge from intensive care, levels of both troponin T and myoglobin rapidly normalised and testing was repeated.
Discussion
Following the rapidly evolving novel disease of the COVID-19 pandemic, new knowledge about the disease is being constantly unearthed. Thus far, only a few reports on rhabdomyolysis and AKI associated with COVID-19 have been published.3 4 Early reports from China indicate that 25% of patients with COVID-19 in critical care have AKI.6
Although type 2 diabetes is common among patients hospitalised for COVID-19, it does not appear to be an independent predictor for mortality or mechanical ventilation.7 However, obesity is prevalent in critically ill patients with COVID-19, and increasing body mass index is associated with an increased risk for death and mechanical ventilation.7
Rhabdomyolysis is characterised by the leakage of contents from the striped myocytes into the bloodstream. During massive rhabdomyolysis, AKI and subsequent renal failure are a potentially dangerous complication, particularly in the intensive care setting where it is estimated that rhabdomyolysis accounts for 7% of all AKI cases in the USA.5 8
Viral myositis and rhabdomyolysis have been associated with several viruses including the influenza A and B, coxsackie, Epstein-Barr, herpes simplex, parainfluenza, adeno, echo, cytomegalo, measles, varicella zoster, human immunodeficiency and dengue viruses.8 The potential magnitude of the myositis and rhabdomyolysis caused by the SARS-CoV-2 virus is largely unknown. Influenza A and B are the most commonly reported viruses associated with myositis. In a case series reported by the paediatric emergency department, viral myositis was a major cause of rhabdomyolysis.9
The exact mechanism by which viruses cause muscle destruction has not been established, but two possible mechanisms have been proposed: first, muscular necrosis related to the potential direct viral invasion of myocytes and second, the toxic effect on myocytes caused by the host response (cytokine release and other immunological factors).10 11
Myoglobinuria is pathognomonic of rhabdomyolysis, and evidence suggests that myoglobin impairs the glomerular filtration through several mechanisms including intrarenal vasoconstriction, ischaemic tubule injury and tubular obstruction.8 Damage to the tubular structures is augmented by hypovolaemia and an acidic urine, both relevant to the intensive care setting.12
Treatment of rhabdomyolysis during ARDS is a delicate balancing act. On one hand, hydration and forced diuresis may limit kidney damage. On the other hand, overhydration could prove deleterious to pulmonary function and gas exchange.13 Thus, early detection, close monitoring and prompt treatment are key. Therefore, we suggest regular screening of serum myoglobin levels in patients with COVID-19 admitted to intensive care.
Contemporary guidelines offer weak evidence-based treatment recommendations to prevent rhabdomyolysis-induced AKI.14 The main recommendation of these guidelines includes early crystalloid-based fluid resuscitation. The routine use of diuretics, alkalinisation, mannitol, antioxidants and renal replacement therapy is not recommended.
Our case report is limited by the lack of a definite aetiology of the rhabdomyolysis other than previously suggested viral and host-response mechanisms. Nevertheless, we believe that it is important to recognise that rhabdomyolysis could be an important complication of COVID-19. Further research designed to better understand the pathophysiological mechanisms behind COVID-19 related rhabdomyolysis would be of great value.
Patient’s perspective.
When I first became sick, symptoms were mild and it was like a common cold. Then I experienced nausea, vomiting, and pain around my hips. After a few days at home my wife had to call for an ambulance. I was able to walk to the ambulance by myself and I remember asking the ambulance crew for some water. When the doctors told me I had corona I didn’t get too worried since I am young and strong. Suddenly, I required more oxygen and I had more difficulties breathing. When I came to the ICU I was afraid and I didn’t want to be put on the ventilator. Just before they put me asleep I remember one of the doctors holding my hand telling me they would look after me, which gave me some peace of mind.
When they woke me up, I got to talk with my family over the phone and that made me very happy.
The hospital staff were like angels, I am very grateful that they took care of me so well. Now, some weeks after I was discharged, I am working part time, and enjoying meeting people again. I am not as strong as I used to be, but I am improving every week.
Learning points.
We recommend clinicians to screen for myoglobinaemia in all patients with COVID-19 on admission and throughout the intensive care period.
In the context of acute respiratory distress syndrome and pulmonary failure, the concept of hydration and forced diuresis as treatment options is likely to be undesirable.
Acute renal failure appears to be common in patients with COVID-19 in intensive care, and rhabdomyolysis could be a contributing factor to this.
Acknowledgments
The authors would like to thank www.editage.com for linguistic advice.
Footnotes
Contributors: Idea: KT and AF. Cared for the patient, data collection, writing of manuscript and proofreading of the manuscript: all authors. Literature review: KT, HW and HK.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Murthy S, Gomersall CD, Fowler RA. Care for critically ill patients with COVID-19. JAMA 2020;323:1499–500. 10.1001/jama.2020.3633 [DOI] [PubMed] [Google Scholar]
- 2.Grasselli G, Pesenti A, Cecconi M. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy. JAMA 2020;323:1545–6. 10.1001/jama.2020.4031 [DOI] [PubMed] [Google Scholar]
- 3.Jin M, Tong Q. Rhabdomyolysis as potential late complication associated with COVID-19. Emerg Infect Dis 2020;26:1618–20. 10.3201/eid2607.200445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Suwanwongse K, Shabarek N. Rhabdomyolysis as a presentation of 2019 novel coronavirus disease. Cureus 2020;12:e7561. 10.7759/cureus.7561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ronco C, Reis T. Kidney involvement in COVID-19 and rationale for extracorporeal therapies. Nat Rev Nephrol 2020;16:308–10. 10.1038/s41581-020-0284-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yu Y, Xu D, Fu S, et al. . Patients with COVID-19 in 19 ICUs in Wuhan, China: a cross-sectional study. Crit Care 2020;24:219. 10.1186/s13054-020-02939-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cariou B, Hadjadj S, Wargny M, et al. . Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia 2020:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bosch X, Poch E, Grau JM. Rhabdomyolysis and acute kidney injury. N Engl J Med 2009;361:62–72. 10.1056/NEJMra0801327 [DOI] [PubMed] [Google Scholar]
- 9.Mannix R, Tan ML, Wright R, et al. . Acute pediatric rhabdomyolysis: causes and rates of renal failure. Pediatrics 2006;118:2119–25. 10.1542/peds.2006-1352 [DOI] [PubMed] [Google Scholar]
- 10.Fodili F, van Bommel EFH. Severe rhabdomyolysis and acute renal failure following recent Coxsackie B virus infection. Neth J Med 2003;61:177–9. [PubMed] [Google Scholar]
- 11.Craighead JE, Huber SA, Sriram S. Animal models of picornavirus-induced autoimmune disease: their possible relevance to human disease. Lab Invest 1990;63:432–46. [PubMed] [Google Scholar]
- 12.Zager RA. Studies of mechanisms and protective maneuvers in myoglobinuric acute renal injury. Lab Invest 1989;60:619–29. [PubMed] [Google Scholar]
- 13.National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network, Wiedemann HP, Wheeler AP, et al. . Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006;354:2564–75. 10.1056/NEJMoa062200 [DOI] [PubMed] [Google Scholar]
- 14.Michelsen J, Cordtz J, Liboriussen L, et al. . Prevention of rhabdomyolysis-induced acute kidney injury - A DASAIM/DSIT clinical practice guideline. Acta Anaesthesiol Scand 2019;63:576–86. 10.1111/aas.13308 [DOI] [PubMed] [Google Scholar]


