Abstract
Understanding which arms of the immune response are responsible for protection against SARS-CoV-2 infection is key to predicting long-term immunity and to inform vaccine design. Two studies in this issue of Cell collectively suggest that, although SARS-CoV-2 infection may blunt long-lived antibody responses, immune memory might still be achieved through virus-specific memory T cells.
Understanding which arms of the immune response are responsible for protection against SARS-CoV-2 infection is key to predicting long-term immunity and to inform vaccine design. Two studies in this issue of Cell collectively suggest that, although SARS-CoV-2 infection may blunt long-lived antibody responses, immune memory might still be achieved through virus-specific memory T cells.
Main Text
Current limited data suggest that SARS-CoV-2 induces some degree of immunity. To date, there is only one definitive report of reinfection within 4 months, proven by genetic sequencing of the virus (To et al., 2020), and in one small study rhesus macaques appeared protected against reinfection (Deng et al., 2020). Although it is early days, and this immunity may not last long, it is good news for the prospects of having an effective vaccine, which should ideally generate the type of immune response that affords protection from reinfection.
CD8+ T cells, CD4+ T cells, and B cells play important roles in the clearance of most viral infections, and immunological T and B cell memory generated after recovery is instrumental in protecting the host from severe disease upon re-exposure. However, the success of most effective vaccines to date largely hinges on the generation of a potent and lasting antibody response, by virtue of the induction of memory B cells and long-lived plasma cells that provide a continuous supply of high-affinity antibodies that circulate and survey our bloodstream and mucosal surfaces. These antibodies can bind to and neutralize the virus even at minute concentrations.
While a lot of attention has been placed in antibody-based immunity, there is increasing evidence that T cells play a major role in the resolution of COVID-19 (Chen and John Wherry, 2020), but whether SARS-CoV-2 generates long-term memory T cell responses and whether these are important for lasting immunity are still unclear. These questions are important because vaccines are generally less effective at eliciting CD8 T cell responses.
In this issue of Cell, two separate studies address the formation of long-lived immunity to SARS-CoV-2. Kaneko et al. report that severe SARS-CoV-2 infections blunt the germinal center response, which is likely to dampen the generation of long-lived antibody responses (Kaneko et al, 2020). The authors set out to establish the root cause of the reported short-lived humoral response to SARS-CoV-2, which was also characteristic of related coronaviruses causing severe infection in humans such as SARS and MERS. For SARS infections, this was thought to be caused by a lack of germinal center (GC) responses (Gu et al., 2005). GCs are transient microanatomical environments that form after antigen-activated B cells receive help from a specialized CD4 T cell subset known as follicular T helper (TFH) cells. Within GCs, B cells undergo clonal expansion and affinity maturation and receive further help from TFH cells to differentiate into memory B cells or long-lived plasma cells.
Kaneko et al. investigate GC B cell responses in individuals succumbing to SARS-CoV-2. The authors conducted extensive multicolor histological assessments of post-mortem thoracic lymph nodes and spleens. As for SARS, they found that GCs were also largely absent during the acute phase of COVID-19. The lack of GCs was accompanied by an absence of BCL6-expressing B cells or TFH cells, which are indispensable for the generation of GCs. Furthermore, an analysis of CD4 T cell composition in situ revealed an enrichment of TBET-expressing T cells with a concomitant increase of TNF-α.
The authors speculate that excessive TNF-α hampers the formation of GC responses in COVID-19 through blocking TFH cell differentiation and promoting TH1 responses. Precedents for TNF-mediated GC blockade have been reported in the context of Ehrlichia muris infection (an intracellular bacterial disease) (Popescu et al., 2019) as well as in severe malaria (Ryg-Cornejo et al., 2016). In both infection models, TNF-α blockade restores GC responses. Although TNF-α is indispensable for GC responses in vivo, this is explained by its role in lymphoid development and in establishing the architecture of secondary lymphoid organs (Pasparakis et al., 1996). Thus, the findings by Kaneko and colleagues suggest that TNF-α blockade in severe COVID-19 infection may not only prevent excessive inflammation but also enable development of long-lived, GC-derived, antibody responses.
Altogether, their data suggest that a lack of GC responses may account for the variable and often low and short-lived antibody responses observed in COVID-19 patients. Nonetheless, given that all their analyses were conducted using tissue obtained from fatal COVID-19 cases, whether GCs are also abrogated in the average milder COVID-19 infections remains unknown. It is possible that the short-lived B cell antibody responses are the result of thymus-independent (TI) B cell activation. Although the authors interpret the presence of AID+ B cells as a sign of robust T:B cell interactions, TI responses can also be isotype switched, are short lived, and can be induced by highly repetitive epitopes coating viral capsids or by activation of Toll-like receptors binding to viral nucleic acids.
In their complementary study, Sekine and colleagues conduct an extensive characterization of T cell immunity in patients suffering from COVID-19 of various degrees of disease severity and at various stages post infection (Sekine et al., 2020). They find SARS-CoV-2-specific memory T cells in most convalescent individuals, including asymptomatic cases and those with undetectable antibody responses.
In individuals with acute infection, T cells displayed an activated phenotype, whereas convalescent patients harbored SARS-CoV-2-specific CD8 T cells with a phenotype reminiscent to early differentiated memory T cells. Expression of TCF-1 by these cells suggests that they may possess a stem cell-like phenotype that endows them the ability to differentiate into multiple effector T cell subsets upon re-infection. Thus, a second SARS-CoV-2 encounter could potentially mount effective GC responses should these memory T cells give rise to TFH cells.
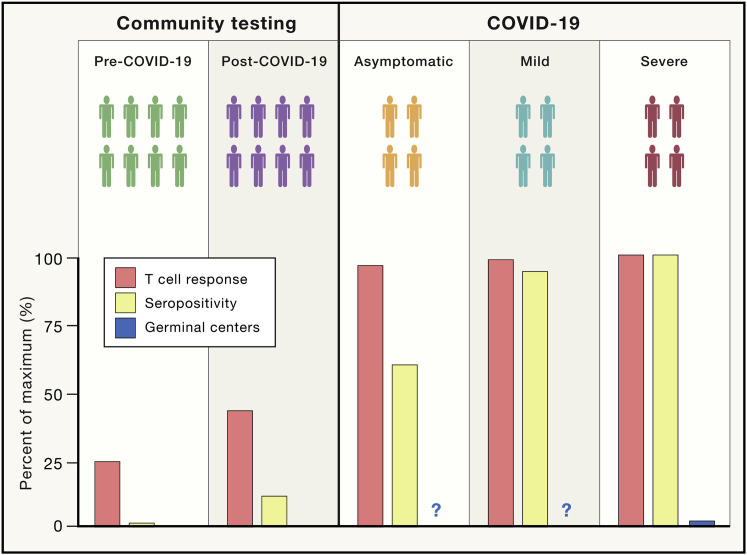
Interestingly, SARS-CoV-2-specific memory T cells were detected in exposed seronegative healthy individuals (relatives of confirmed cases), indicative of asymptomatic infection. Remarkably, ∼93% of “exposed asymptomatic” individuals mounted detectable T cell responses to SARS-CoV-2 despite only 60% of cases being seropositive (Figure 1 ). This suggests that asymptomatic infections may be more common than current data suggest and that immunosurveillance through antibody testing alone may underestimate infection prevalence or population immunity. The presence of SARS-CoV-2-specific T cells in the majority of convalescent patients is a promising sign that infection may give rise to immunity, but whether these T cells afford protection from reinfection remains to be tested.
Figure 1.
Seropositivity to SARS-CoV-2 May Underestimate COVID-19 Prevalence or Immunity
Quantification of the percentage of individuals with detectable T cell responses, serum antibodies, or germinal centers across SARS-CoV-2-exposed individuals (either asymptomatic or those with mild or severe COVID-19 symptoms) or two cohorts of seemingly healthy unexposed individuals (blood samples collected either before or during the COVID-19 pandemic). Serum antibodies and T cell responses are found in most patients with either severe or mild COVID-19. GC responses are absent in severe cases of COVID-19, but whether they are detectable in mild or asymptomatic individuals remains unknown. Most asymptomatic COVID-19 cases showed strong T cell responses even if only ∼60% of these individuals were seropositive for SARS-CoV-2.
A smaller but consistent fraction of samples collected in mid-2019 (i.e., “unexposed individuals”) also revealed SARS-CoV-2-reactive memory T cells, which was not entirely unexpected. There are currently four known strains of coronaviruses that circulate seasonally throughout the population (Moriyama et al., 2020), and extensive T cell cross reactivity across these strains of viruses has been documented (Mateus et al., 2020). Unexposed individuals that harbor cross-reactive T cells may be protected from severe disease, but whether the presence of these cells may negatively influence the generation of protective immunity remains to be tested.
On the basis of these results, it is tempting to speculate that although optimal protective immunity induced by COVID-19 infection may rely on the production of both memory T cells and GC-derived long-lived plasma cells, either response alone may confer some protection against severe disease. There is abundant evidence of redundancy and diversity in the immune system that ensures a level of protection against infection in situations in which a branch of immunity is compromised. Finding potent memory CD8+ and CD4+ T cell responses elicited during asymptomatic infections even in the absence of detectable antibody responses increases our hopes of protective immunity post-SARS-CoV-2 infection. However, robust memory CD8+ T cell responses may be difficult to recapitulate with vaccination, which will likely be more reliant on the induction of potent high-affinity neutralizing antibodies.
References
- Chen Z., John Wherry E. T cell responses in patients with COVID-19. Nat Rev Immunol. 2020;20:529–536. doi: 10.1038/s41577-020-0402-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng W., Bao L., Liu J., Xiao C., Liu J., Xue J., Lv Q., Qi F., Gao H., Yu P. Primary exposure to SARS-CoV-2 protects against reinfection in rhesus macaques. Science. 2020;369:818–823. doi: 10.1126/science.abc5343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., Zou W., Zhan J., Wang S., Xie Z. Multiple organ infection and the pathogenesis of SARS. J Exp Med. 2005;202:415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaneko N., Kuo H.-H., Boucau J., Farmer J.R., Allard-Chamard H., Mahajan V.S., Piechocka-Trocha A., Lefteri K., Osborn M., Bals J. Loss of Bcl-6-Expressing T Follicular Helper Cells and Germinal Centers in COVID-19. Cell. 2020;183:143–157. doi: 10.1016/j.cell.2020.08.025. this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mateus J., Grifoni A., Tarke A., Sidney J., Ramirez S.I., Dan J.M., Burger Z.C., Rawlings S.A., Smith D.M., Phillips E. Selective and cross-reactive SARS-CoV-2 T cell epitopes in unexposed humans. Science. 2020 doi: 10.1126/science.abd3871. Published online August 4, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriyama M., Hugentobler W.J., Iwasaki A. Seasonality of Respiratory Viral Infections. Annu Rev Virol. 2020 doi: 10.1146/annurev-virology-012420-022445. Published online March 20, 2020. [DOI] [PubMed] [Google Scholar]
- Pasparakis M., Alexopoulou L., Episkopou V., Kollias G. Immune and inflammatory responses in TNF alpha-deficient mice: a critical requirement for TNF alpha in the formation of primary B cell follicles, follicular dendritic cell networks and germinal centers, and in the maturation of the humoral immune response. J Exp Med. 1996;184:1397–1411. doi: 10.1084/jem.184.4.1397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Popescu M., Cabrera-Martinez B., Winslow G.M. TNF-α Contributes to Lymphoid Tissue Disorganization and Germinal Center B Cell Suppression during Intracellular Bacterial Infection. J Immunol. 2019;203:2415–2424. doi: 10.4049/jimmunol.1900484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryg-Cornejo V., Ioannidis L.J., Ly A., Chiu C.Y., Tellier J., Hill D.L., Preston S.P., Pellegrini M., Yu D., Nutt S.L. Severe Malaria Infections Impair Germinal Center Responses by Inhibiting T Follicular Helper Cell Differentiation. Cell Rep. 2016;14:68–81. doi: 10.1016/j.celrep.2015.12.006. [DOI] [PubMed] [Google Scholar]
- Sekine T., Perez-Potti A., Rivera-Ballesteros O., Strålin K., Gorin J.-P., Olsson A., Llewellyn-Lacey S., Kamal H., Bogdanovic G., Muschiol S. Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19. Cell. 2020;183:158–168. doi: 10.1016/j.cell.2020.08.017. this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- To K.K.-W., Hung I.F.-N., Ip J.D., Chu A.W.-H., Chan W.-M., Tam A.R., Fong C.H.-Y., Yuan S., Tsoi H.-W., Ng A.C.-K. COVID-19 re-infection by a phylogenetically distinct SARS-coronavirus-2 strain confirmed by whole genome sequencing. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa1275. Published online August 25, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]