Abstract
The novel coronavirus SARS-CoV-2 is known to cause hypoxemia and acute respiratory distress syndrome (ARDS) in a significant portion of those with severe disease. Survivors of critical illness and ARDS often experience neurocognitive impairment but, to date, there is scant literature correlating radiographic hypoxic brain injury to hypoxemia related to ARDS. In this case series, we describe three cases of hypoxic brain injury seen on magnetic resonance imaging (MRI) in patients with hypoxemia secondary to COVID-19-related ARDS. The lack of severe observed hypoxemia in two of the cases suggests that unrecognized or asymptomatic hypoxemia may play a role in hypoxic brain injury related to COVID-19.
Keywords: Hypoxic-ischemic encephalopathy, COVID-19, Acute respiratory distress syndrome, Brain MRI
Abbreviations: ARDS, Acute respiratory distress syndrome; ICU, Intensive care unit; DVT, Deep vein thrombosis; EEG, Electroencephalography; CRS, Cytokine release syndrome; CAR, Chimeric antigen receptor; ICU-AW, Intensive Care Unit-Acquired Weakness
Highlights
-
•
Cases of hypoxic ischemic brain injury in COVID-19 ARDS are described, in absence of cardiopulmonary arrest.
-
•
Lack of severe hypoxemia suggests unrecognized or asymptomatic hypoxemia is involved in hypoxic brain injury.
-
•
Other factors such as cytokine release syndrome may play a role in prolonged encephalopathy in COVID-19.
1. Introduction
The novel coronavirus SARS-CoV-2 emerged in December 2019 as a highly contagious virus, with an estimated doubling time in the early stages of 2.3–3.3 days [1]. The incidence of COVID-19 infections varies widely even within the United States, with a range of 20.6 to 915.3 cases per 100,000 in early April 2020 [2]. SARS-CoV-2 has been shown to cause critical illness in between 5 and 21% of infected patients [3,4]. Of patients with severe disease, approximately 16% had acute respiratory distress syndrome (ARDS), 14.5% required invasive mechanical ventilation, and 99% had pneumonia [4]. With the rising worldwide prevalence of COVID-19 infections, a significant proportion of the global population will require monitoring in an intensive unit care (ICU) and will develop ARDS.
ARDS is the development of an acute onset of new or worsening respiratory symptoms with radiographic evidence of bilateral opacities which cannot otherwise be explained by fluid overload or cardiac failure [5]. The hallmark of this syndrome is hypoxemia, with severity determined by the ratio of arterial oxygen tension to fractional inspired oxygen (PaO2/FiO2) and a minimum positive end expiratory pressure of five [6]. There is significant mortality and long-term morbidity associated with ARDS. Survivors of ARDS are known to develop long-term functional disability for years after the event [7]. Despite advances in treatments, the overall mortality rate is approximately 40% [8].
The prevalence of neurocognitive impairment after critical illness is well established [9,10]. In particular, survivors of ARDS seem to be at increased risk of cognitive impairment [[11], [12], [13]]. Between 70 and 78% of patients who survived ARDS had neurocognitive impairment at hospital discharge and nearly half had persistent deficits at one year in the areas of memory, processing speed, executive functioning, and attention [11,14]. While the mechanisms underlying neurocognitive dysfunction in survivors of critical illness are not well understood, many factors are thought to contribute including hypotension, sedation, delirium, hyperglycemia, and cerebral microhemorrhages [10,[14], [15], [16], [17]].
Hypoxemia and the duration of hypoxemia have been linked to poor cognitive outcomes in critically ill patients [12,14]. Brain atrophy on MRI has been reported as a consequence of critical illness and brain volume loss has been observed on computed tomography (CT) in ARDS survivors [19,20]. A recent autopsy case series found evidence of hypoxic changes in the brain tissue of patients who had succumbed to COVID-19, but there was no report of whether these patients developed ARDS, duration of mechanical ventilation if required, whether extracorporeal membrane oxygenation (ECMO) was used, presence or absence of cardiac arrest, or cause of death [21]. To date, there is scant literature linking hypoxemia to radiographic findings of hypoxic-ischemic brain injury in survivors of ARDS. In this case series, we describe three cases of hypoxic brain injury seen on MRI, along with clinical correlations, in patients with hypoxemia secondary to COVID-19 related ARDS. Notably, none of these patients experienced cardiopulmonary arrest.
2. Methods
This is a retrospective review of patients admitted to our tertiary care center with ARDS due to severe COVID-19 infection who were evaluated by the general neurology team after a consult was requested for encephalopathy or other neurologic deficit. Each patient included underwent brain MRI as part of the neurologic work up and all were found to have evidence of hypoxic-ischemic injury. Patients selected were neurologically intact prior to hospitalization with COVID-19. All patients had GCS 15 with no apparent encephalopathy prior to intubation, and all were able to provide their own history on initial presentation to the hospital. Exclusion criteria included cardiopulmonary arrest during the hospitalization or any history of hypoxic-ischemic injury prior to hospitalization. Our institution based respiratory therapy protocol dictates that all ventilated critical care patients be assessed daily for spontaneous awakening and spontaneous breathing trials, and if appropriate trials completed per a “wake up and breathe” protocol [22].
3. Cases
3.1. Case 1
32-year-old obese female with a history of asthma, obesity, gastroesophageal reflux disease, and vitamin D deficiency presented with shortness of breath and self-reported oxygen saturation of 85% measured with home oxygen monitor. She was intubated on the day of admission due to worsening hypoxia and dyspnea and met criteria for severe ARDS. Hospital course was complicated by cytokine release syndrome (treated with tocilizumab), gastrointestinal bleeding, and dysphagia requiring percutaneous endoscopic gastrostomy tube placement. She intermittently required vasopressor therapy but did not have prolonged episodes of hypotension. Table 1 summarizes degree and duration of hypoxemia. After 22 days of mechanical ventilation, she was successfully extubated.
Table 1.
Oxygenation and blood pressure characteristics.
| Case 1 | Case 2 | Case 3 | |
|---|---|---|---|
| Mean PaO2 | 116 | 98.89 | 99.78 |
| Median PaO2 | 103 | 87.5 | 91 |
| Average P/F while mechanically ventilateda | 190.99 | 216.85 | 209.86 |
| PaO2 Range | 50–264 | 59–192 | 54–289 |
| Number of PaO2 readings below 55b | 1 | 0 | 1 |
| Duration at lowest PaO2c | <6 h | <2 h | <2 h |
| SpO2 Ranged | 83%- 100% | 86%–100% | 30%–100% |
| Duration at lowest SpO2 | <6 min | <60 min | <2 min |
| Lowest MAP | 49 | 43e | 40 |
| Duration with MAP<60 | <1 h | Minutes | <40 min |
| Autonomic instabilityf | Yes | Yes | Yes |
| Maximum number of concurrent vasopressors | 1 | 3 | 1 |
| Days on ventilator | 22 | 22 | 44 |
| Days in ICU | 25 | 25 | 51 |
| Hypoglycemia | No | No | No |
P/F ratios were only included when FiO2 was provided with arterial blood gas lab draw.
Based on ARDSnet goal of PaO2 55–80 mm Hg [23].
Duration is an estimate determined by time of next ABG result demonstrating improved oxygenation.
SpO2 range obtained from vitals charted throughout admission.
MAP from blood pressure cuff, arterial MAP>60 at the time.
Autonomic instability defined as labile blood pressures requiring intermittent vasopressor therapy as well as antihypertensive therapy.
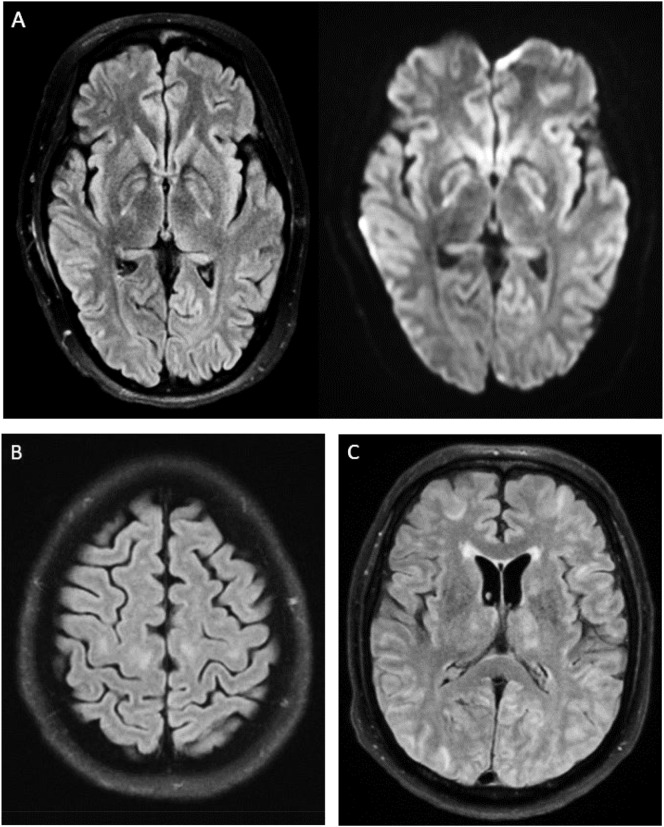
Two days after cessation of sedative medications and one day post extubation, neurology was consulted due to encephalopathy and semi-continuous right facial twitching. Given initial concern for seizures, she was started on levetiracetam with slight improvement in the movements, though the movements continued to wax and wane. Continuous video electroencephalography (EEG) showed continuous irregular generalized slow wave activity, without any seizures or epileptiform activity, verifying these movements were not seizures but rather dyskinesias or myoclonus. Aside from these movements, cranial nerves were intact, she was awake and alert but non-verbal, and had no spontaneous movements of her extremities. MRI brain demonstrated symmetric T2 FLAIR hyperintensities with mild restricted diffusion in the bilateral globus pallidi extending to the adjacent posterior limb of the internal capsules and along the cerebral peduncles, suggesting hypoxic ischemic injury (Fig. 1A).
Fig. 1.
(A) Bilateral ill-defined T2 FLAIR hyperintensities and mild restricted diffusion in the bilateral basal ganglia. (B) Bilateral T2 FLAIR hyperintensities in the perirolandic white matter. (C) Bilateral diffuse T2 FLAIR hyperintensities in the cortex, subcortical white matter, and thalami.
The suspected dyskinesias versus myoclonus were thought to be related to these radiographic changes. At time of discharge to an acute inpatient rehabilitation facility, she was alert, followed simple commands, communicated via head nodding, had minimal verbal output, and was able to flicker her toes bilaterally but had no movement of her arms. As she was noted to have more proximal than distal weakness, there was likely a component of Intensive Care Unit-Acquired Weakness (ICU-AW), in addition to resolving encephalopathy.
3.2. Case 2
42-year-old female with history of obesity and prior abdominal gunshot wound presented with shortness of breath and cough. She was intubated the day after admission due to progression of hypoxemic respiratory failure and failure of high flow oxygen supplementation. Table 1 summarizes the degree and duration of hypoxemia. Her hospital course was complicated by recurrent fevers, lower extremity deep vein thrombosis (DVT), and acute renal failure requiring renal replacement therapy. Hypotension was also present and attributed to cytokine release syndrome (CRS), which was treated with vasopressor therapy and tocilizumab. She initially failed extubation after 16 days on mechanical ventilation but was later extubated successfully after a total of 22 days on mechanical ventilation.
Five days after extubation, the patient remained poorly interactive, prompting a neurology consult. On exam she was awake and alert, non-verbal with intact cranial nerves, minimal spontaneous movement, and no motor response to noxious stimulus. MRI brain was completed and demonstrated symmetric confluent T2 hyperintensities in the subcortical white matter along bilateral perirolandic regions at the vertex, and in the corona radiata and centrum semiovale with questionable T2 hyperintensity in the bilateral lentiform and caudate nuclei (Fig. 1B).
Upon discharge to an acute inpatient rehabilitation facility she was awake, alert and oriented to person, place and time. Her motor exam was significant for profound proximal weakness, with preservation of distal extremity strength. Similar to the previous patient, there was improvement in encephalopathy and weakness likely secondary to ICU-AW.
3.3. Case 3
66-year-old male with history of diabetes presented to an outside hospital with cough, fever, diarrhea and abdominal pain. Based on outside hospital records, his oxygen saturation on presentation was 65% on room air. He was quickly placed on a non-rebreather mask with improvement in oxygen saturation to 99%. At that time, he was noted to be in mild distress but alert and oriented to person, place and time with a normal motor exam. He was intubated on the day of admission due to worsening hypoxemia (PaO2 61 on non-invasive ventilation) and transferred to our center six days later for escalation of care. ABG prior to transfer demonstrated PaO2 of 59 requiring FiO2 of 100% and PEEP of 20.
The patient's full outside hospital course was not included in available records, but oxygen saturation prior to transfer ranged from 90 to 98% and no episodes of hypotension were recorded. See Table 1 for description of hypoxemia while at our center. His course was complicated by superimposed ventilator associated pneumonia, high fevers, opioid withdrawal syndrome and acute renal failure requiring renal replacement therapy. He was initially extubated after 25 days on mechanical ventilation but then reintubated 3 days later for inability to protect his airway. He was ultimately extubated after 44 total days on a mechanical ventilation.
Neurology was consulted on day 34 of admission due to persistent encephalopathy precluding extubation. On neurologic examination while intubated and off sedation, he was noted to open eyes to verbal stimulus but did not follow any commands, had intact brainstem reflexes, and did not demonstrate purposeful or spontaneous movements. MRI brain demonstrated symmetric and diffuse cortical, subcortical, thalamic and cerebellar ill-defined T2 FLAIR hyperintensities likely consistent with hypoxic ischemic injury (Fig. 1C). There were also a few parenchymal microhemorrhages in both a central and peripheral distribution.
The patient was transferred back to the outside hospital after he no longer required ICU level care. At time of discharge from our ICU the encephalopathy was improving, he was awake, alert and oriented to person and able to follow commands. The motor exam had also improved and no abnormal movements were noted.
4. Discussion
This case series aims to describe the clinical and radiographic characteristics of patients with hypoxemia following severe COVID-19 infection. All patients included required prolonged mechanical ventilation, as well as use of vasopressor therapy due to hypotension. There were no documented episodes of hypoglycemia in any patients that were included in this study. Two of three patients had one documented PaO2 level below the ARDSNET goal of 55 mm Hg [23]. Interestingly, despite the lack of severe observed hypoxemia in two of the three patients, these patients developed hypoxic brain injury. The same two patients developed CRS requiring tocilizumab, a monoclonal antibody against the IL-6 receptor.
The development of hypoxic brain injury despite a lack of severe observed hypoxemia may reflect a proposed theory of hemoglobinopathy [24], contributing to what may present as “silent hypoxemia,” where patients with COVID-19 related pneumonia have hypoxemia with mild or absent clinical manifestations [25]. This is evidenced by the patients in case 1 and 3 who presented with low oxygen saturations, seemingly out of proportion to their symptoms. While these patients may not be initially symptomatic, perhaps prolonged unrecognized periods of hypoxemia can later lead to these findings. Hypoxemia in severe COVID-19 has been proposed as one of several mechanisms responsible for neurologic sequelae [26]. Hypoxemia likely increases risk for neurocognitive impairments, but there are a multitude of other risk factors including prolonged hypotension and large cumulative doses of sedation needed for ventilator synchrony [9].
We propose that CRS may be a contributing factor to the prolonged encephalopathy reported in two of the patients. Leukemia patients that develop CRS secondary to chimeric antigen receptor (CAR) T cell therapy often present with aphasia and encephalopathy [27]. However, it is unclear whether CAR-T cell therapy itself or CRS is more contributory. This neurotoxicity does not respond well to treatment with tocilizumab, which is thought to be due in part to the inability of tocilizumab to cross the blood brain barrier [28]. Similarly, in severe COVID-19, encephalopathy and slow neurologic recovery may be in part related to CRS, but minimal neurological improvement may be noted despite treatment with tocilizumab.
While only present in one of the patients discussed in this case series, cerebral microhemorrhages have been documented sequalae of both COVID-19 and critical illness in general [17,18]. These findings, in addition to the other factors discussed, may have contributed to the patient's prolonged encephalopathy.
Neurologic recovery of patients who develop hypoxic-ischemic brain injury is typically prolonged, with the greatest improvement seen within the first three months after injury [29]. Some degree of cognitive deficit is often permanent, though the functional impact varies widely [29]. As more patients develop and recover from severe COVID-19 infection, we can expect to see many more patients with the long-term neurocognitive sequelae that are well known to follow other critical illness and in particular ARDS. Neurorehabilitation after hospitalization will be important to improve the functioning and quality of life of these patients.
5. Conclusion
Although this study is limited in scope due to its size and retrospective design, it provides novel information regarding hypoxic-ischemic brain injury in COVID-19 related ARDS. Although the findings of hypoxic-ischemic brain injury may be generalizable to all causes of ARDS, there may be features specific to COVID-19 related ARDS. As mentioned, CRS may be involved in this relationship and perhaps potentiate susceptibility to ischemic injury. Future studies may assess in greater detail the pattern or degree of hypoxic ischemic injury on MRI and long-term neurocognitive outcomes. This case series also highlights the need for further research into COVID-19 related pneumonia and ARDS. We may need to better identify thresholds for acceptable levels of hypoxemia, as well as continue monitoring for long term neurologic and cognitive outcomes.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of Competing Interest
None.
References
- 1.Sanche S., Lin Y.T., Xu C., Romero-Severson E., Hengartner N., Ke R. High contagiousness and rapid spread of severe acute respiratory syndrome coronavirus 2. Emerg. Infect. Dis. 2020 doi: 10.3201/eid2607.200282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Geographic differences in covid-19 cases, deaths, and incidence - United States, February 12–April 7, 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020 doi: 10.15585/mmwr.mm6915e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bialek S., Boundy E., Bowen V., Chow N., Cohn A., Dowling N., Ellington S., Gierke R., Hall A., MacNeil J., Patel P., Peacock G., Pilishvili T., Razzaghi H., Reed N., Ritchey M., Sauber-Schatz E. Severe outcomes among patients with coronavirus disease 2019 (COVID-19) - United States, February 12-march 16, 2020. Morb. Mortal. Wkly. Rep. 2020 doi: 10.15585/mmwr.mm6912e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guan W.J., Ni Z.Y., Hu Y., Liang W.H., Ou C.Q., He J.X., Liu L., Shan H., Lei C.L., Hui D.S.C., Du B., Li L.J., Zeng G., Yuen K.Y., Chen R.C., Tang C.L., Wang T., Chen P.Y., Xiang J., Li S.Y., Wang J.L., Liang Z.J., Peng Y.X., Wei L., Liu Y., Hu Y.H., Peng P., Wang J.M., Liu J.Y., Chen Z., Li G., Zheng Z.J., Qiu S.Q., Luo J., Ye C.J., Zhu S.Y., Zhong N.S. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ahmad F., Koffman L. Acute respiratory distress syndrome. In: Prabhakar H., Ali Z., editors. Textb. Neuroanesthesia Neurocritical Care Vol. II - Neurocritical Care. Springer Singapore; Singapore: 2019. pp. 367–375. [DOI] [Google Scholar]
- 6.Ferguson N.D., Fan E., Camporota L., Antonelli M., Anzueto A., Beale R., Brochard L., Brower R., Esteban A., Gattinoni L., Rhodes A., Slutsky A.S., Vincent J.L., Rubenfeld G.D., Taylor Thompson B., Marco Ranieri V. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med. 2012 doi: 10.1007/s00134-012-2682-1. [DOI] [PubMed] [Google Scholar]
- 7.Herridge M.S., Tansey C.M., Matté A., Tomlinson G., Diaz-Granados N., Cooper A., Guest C.B., Mazer C.D., Mehta S., Stewart T.E., Kudlow P., Cook D., Slutsky A.S., Cheung A.M. Functional disability 5 years after acute respiratory distress syndrome. N. Engl. J. Med. 2011;364:1293–1304. doi: 10.1056/NEJMoa1011802. [DOI] [PubMed] [Google Scholar]
- 8.McNicholas B.A., Rooney G.M., Laffey J.G. Lessons to learn from epidemiologic studies in ARDS. Curr. Opin. Crit. Care. 2018;24:41–48. doi: 10.1097/MCC.0000000000000473. [DOI] [PubMed] [Google Scholar]
- 9.Hopkins R.O., Jackson J.C. Long-term neurocognitive function after critical illness. Chest. 2006 doi: 10.1378/chest.130.3.869. [DOI] [PubMed] [Google Scholar]
- 10.Pandharipande P.P., Girard T.D., Jackson J.C., Morandi A., Thompson J.L., Pun B.T., Brummel N.E., Hughes C.G., Vasilevskis E.E., Shintani A.K., Moons K.G., Geevarghese S.K., Canonico A., Hopkins R.O., Bernard G.R., Dittus R.S., Ely E.W. Long-term cognitive impairment after critical illness. N. Engl. J. Med. 2013 doi: 10.1056/NEJMoa1301372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hopkins R.O., Weaver L.K., Pope D., Orme J.F., Bigler E.D., Larson-Lohr V. Neuropsychological sequelae and impaired health status in survivors of severe acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1999 doi: 10.1164/ajrccm.160.1.9708059. [DOI] [PubMed] [Google Scholar]
- 12.Mikkelsen M.E., Christie J.D., Lanken P.N., Biester R.C., Thompson B.T., Bellamy S.L., Localio A.R., Demissie Ejigayehu, Hopkins R.O., Angus D.C. The adult respiratory distress syndrome cognitive outcomes study: long-term neuropsychological function in survivors of acute lung injury. Am. J. Respir. Crit. Care Med. 2012 doi: 10.1164/rccm.201111-2025OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sasannejad C., Ely E.W., Lahiri S. Long-term cognitive impairment after acute respiratory distress syndrome: a review of clinical impact and pathophysiological mechanisms. Crit. Care. 2019 doi: 10.1186/s13054-019-2626-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hopkins R.O., Weaver L.K., Collingridge D., Parkinson R.B., Chan K.J., Orme J.F. Two-year cognitive, emotional, and quality-of-life outcomes in acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2005 doi: 10.1164/rccm.200406-763OC. [DOI] [PubMed] [Google Scholar]
- 15.Girard T., Jackson J., Pandharipande P., Thompson J., Shintani A., Ely E. 2009. Duration of Delirium as a Predictor of Long-term Cognitive Impairment in Survivors of Critical Illness. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Porhomayon J., Joude P.A., Adlparvar G., El-Solh A.A., Nader N.D. The impact of high versus low sedation dosing strategy on cognitive dysfunction in survivors of intensive care units: a systematic review and meta-analysis. J. Cardiovasc. Thorac. Res. 2015 doi: 10.15171/jcvtr.2015.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fanou E.M., Coutinho J.M., Shannon P., Kiehl T.R., Levi M.M., Wilcox M.E., Aviv R.I., Mandell D.M. Critical illness-associated cerebral microbleeds. Stroke. 2017 doi: 10.1161/STROKEAHA.116.016289. [DOI] [PubMed] [Google Scholar]
- 18.Nicholson P., Alshafai L., Krings T. Neuroimaging findings in patients with COVID-19. Am. J. Neuroradiol. 2020 doi: 10.3174/ajnr.a6630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gunther M.L., Morandi A., Krauskopf E., Pandharipande P., Girard T.D., Jackson J.C., Thompson J., Shintani A.K., Geevarghese S., Miller R.R., Canonico A., Merkle K., Cannistraci C.J., Rogers B.P., Gatenby J. Chris, Heckers S., Gore J.C., Hopkins R.O., Ely E. Wesley. The association between brain volumes, delirium duration, and cognitive outcomes in intensive care unit survivors: the VISIONS cohort magnetic resonance imaging study. Crit. Care Med. 2012 doi: 10.1097/CCM.0b013e318250acc0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hopkins R.O., Gale S.D., Weaver L.K. Brain atrophy and cognitive impairment in survivors of acute respiratory distress syndrome. Brain Inj. 2006 doi: 10.1080/02699050500488199. [DOI] [PubMed] [Google Scholar]
- 21.Solomon I.H., Normandin E., Bhattacharyya S., Mukerji S.S., Keller K., Ali A.S., Adams G., Hornick J.L., Padera R.F., Sabeti P. Neuropathological features of Covid-19. N. Engl. J. Med. 2020 doi: 10.1056/nejmc2019373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Girard T.D., Kress J.P., Fuchs B.D., Thomason J.W., Schweickert W.D., Pun B.T., Taichman D.B., Dunn J.G., Pohlman A.S., Kinniry P.A., Jackson J.C., Canonico A.E., Light R.W., Shintani A.K., Thompson J.L., Gordon S.M., Hall J.B., Dittus R.S., Bernard G.R., Ely E.W. Efficacy and safety of a paired sedation and ventilator weaning protocol for mechanically ventilated patients in intensive care (Awakening and Breathing Controlled trial): a randomised controlled trial. Lancet. 2008 doi: 10.1016/S0140-6736(08)60105-1. [DOI] [PubMed] [Google Scholar]
- 23.Brower R.G., Matthay M.A., Morris A., Schoenfeld D., Thompson B.T., Wheeler A. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N. Engl. J. Med. 2000 doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 24.Liu W., Li H. COVID-19: attacks the 1-beta chain of hemoglobin and captures the porphyrin to inhibit human heme metabolism. ChemRxiv. 2020 doi: 10.26434/chemrxiv.11938173.v6. [DOI] [Google Scholar]
- 25.Xie J., Tong Z., Guan X., Du B., Qiu H., Slutsky A.S. Critical care crisis and some recommendations during the COVID-19 epidemic in China. Intensive Care Med. 2020 doi: 10.1007/s00134-020-05979-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guo Y.R., Cao Q.D., Hong Z.S., Tan Y.Y., Chen S.D., Jin H.J., Sen Tan K., Wang D.Y., Yan Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak- an update on the status. Mil. Med. Res. 2020 doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee D.W., Santomasso B.D., Locke F.L., Ghobadi A., Turtle C.J., Brudno J.N., Maus M.V., Park J.H., Mead E., Pavletic S., Go W.Y., Eldjerou L., Gardner R.A., Frey N., Curran K.J., Peggs K., Pasquini M., DiPersio J.F., van den Brink M.R.M., Komanduri K.V., Grupp S.A., Neelapu S.S. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol. Blood Marrow Transplant. 2019;25:625–638. doi: 10.1016/j.bbmt.2018.12.758. [DOI] [PubMed] [Google Scholar]
- 28.Shimabukuro-Vornhagen A., Gödel P., Subklewe M., Stemmler H.J., Schlößer H.A., Schlaak M., Kochanek M., Böll B., von Bergwelt-Baildon M.S. Cytokine release syndrome. J. Immunother. Cancer. 2018;6:56. doi: 10.1186/s40425-018-0343-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Anderson C.A., Arciniegas D.B. Cognitive sequelae of hypoxic-ischemic brain injury: a review. NeuroRehabilitation. 2010;26:47–63. doi: 10.3233/NRE-2010-0535. [DOI] [PubMed] [Google Scholar]