Abstract
Inflammation is intimately related to the pathogenesis of numerous acute and chronic diseases like cardiovascular disease, inflammatory bowel disease, rheumatoid arthritis, and neurodegenerative diseases. Therefore anti-inflammatory therapy is a very promising strategy for the prevention and treatment of these inflammatory diseases. To overcome the shortcomings of existing anti-inflammatory agents and their traditional formulations, such as nonspecific tissue distribution and uncontrolled drug release, bioresponsive drug delivery systems have received much attention in recent years. In this review, we first provide a brief introduction of the pathogenesis of inflammation, with an emphasis on representative inflammatory cells and mediators in inflammatory microenvironments that serve as pathological fundamentals for rational design of bioresponsive carriers. Then we discuss different materials and delivery systems responsive to inflammation-associated biochemical signals, such as pH, reactive oxygen species, and specific enzymes. Also, applications of various bioresponsive drug delivery systems in the treatment of typical acute and chronic inflammatory diseases are described. Finally, crucial challenges in the future development and clinical translation of bioresponsive anti-inflammatory drug delivery systems are highlighted.
Keywords: Bioresponsive materials, Reactive oxygen species, Drug delivery, Inflammatory diseases, Nanoparticles, Microparticles, Hydrogels
Graphical abstract
Highlights
-
•
Inflammation is closely related to the pathogenesis of many acute/chronic diseases.
-
•
Low pH and high reactive oxygen species or enzymes are typical inflammatory cues.
-
•
Inflammatory cues are useful triggers for rational design of drug delivery systems.
-
•
Bioresponsive drug delivery systems are beneficial for treating inflammation.
-
•
Translation studies are necessary for anti-inflammatory bioresponsive therapies.
1. Introduction
Inflammation is an essential immune response to harmful stimuli, such as pathogens, damaged cells, chemical irritants, physical trauma, burns, radiation, and frostbite [1]. Whereas inflammation is essential for repair, remodeling, and even renewal of different tissues under a variety of noxious conditions [2], inflammatory responses need to be spatiotemporally regulated to maintain tissue or systemic homeostasis. Uncontrolled or unresolved inflammation is intimately linked to the initiation and progression of numerous acute and chronic diseases, such as acute liver/lung injury, acute bronchitis, asthma, inflammatory bowel disease (IBD), rheumatoid arthritis (RA), atherosclerosis, and neurodegenerative diseases [[3], [4], [5], [6]]. Also, inflammation is a key feature of metabolic disorders such as obesity and diabetes [7,8]. Consequently, anti-inflammation is a highly promising strategy for the prevention and treatment of acute and chronic inflammatory diseases as well as diseases associated with inflammation [5,9,10].
Historically, small molecule agents, such as glucocorticoids, nonsteroidal anti-inflammatory drugs, and antioxidants have been extensively used for the treatment of different inflammatory diseases [[11], [12], [13]]. Despite their effectiveness to varied degrees, these drugs show undesirable side effects such as osteoporosis [14], aseptic joint necrosis [15], gastrointestinal bleeding [16,17], liver/kidney injury [18], and cardiovascular risk [19]. By regulating molecular and cellular events closely associated with inflammatory responses, monoclonal antibodies to various proinflammatory cytokines and chemokines or other biomacromolecules have been clinically used or pre-clinically examined for therapy of RA [20], IBD [21,22], atherosclerotic disease [23,24], and asthma [25]. However, clinical applications of these biological agents have been considerably impaired by their limitations and adverse effects, such as high cost [26], a primary or secondary nonresponse [27], and the increased risk of severe infections and malignancies [[28], [29], [30]]. To a large degree, systemic distribution in non-target tissues and cells is mainly responsible for undesirable effects, regardless of small molecule drugs and biological therapies. Also, site-specific delivery and controlled release in inflammatory tissues/cells is critical to some newly discovered therapies, such as specialized pro-resolving mediators that have been found highly effective in the treatment of inflammatory disorders, owing to their potent activity of resolving inflammation [10,31,32].
To circumvent disadvantages of anti-inflammatory therapies based on traditional formulations, different advanced drug delivery systems have been investigated over the past decades. In this aspect, dendrimers, lipid nanoparticles, polymeric nanoparticles, micelles, nanocapsules, nanoemulsions, nanofibers, microparticles, hydrogels, and multi-compartmental delivery systems were examined for delivery of either molecular therapies or therapeutic cells for the treatment of inflammatory diseases [[33], [34], [35], [36], [37], [38]]. In addition to the totally synthesized vehicles across multiple length scales, some biomimetic particles such as exosomes, cell membrane-based nanovehicles, and cellular carriers have been developed for delivery of anti-inflammatory agents [[39], [40], [41], [42]]. The majority of these delivery systems were employed for treating inflammation in specific tissues by local delivery of various therapeutics [[33], [34], [35],[43], [44], [45], [46]]. In other cases, nanoparticles have been extensively investigated for site-specific delivery of therapeutic cargoes to inflammatory sites after systemic administration [34,37,39,41,42,[47], [48], [49], [50], [51], [52], [53]], since inflamed tissues generally show enhanced vascular permeability [54,55]. In addition, surface engineering with either molecular moieties or cell-derived membranes have been conducted for different nanovehicles to increase their targeting capability [41,[47], [48], [49], [50], [51], [52],56], in view of the fact that specific biomarkers are overexpressed in inflammatory tissues and/or cells. Furthermore, macroscale biomaterials, such as hydrogels were examined for delivery of different cells to achieve cell therapy or tissue repair at inflammatory sites [[57], [58], [59]].
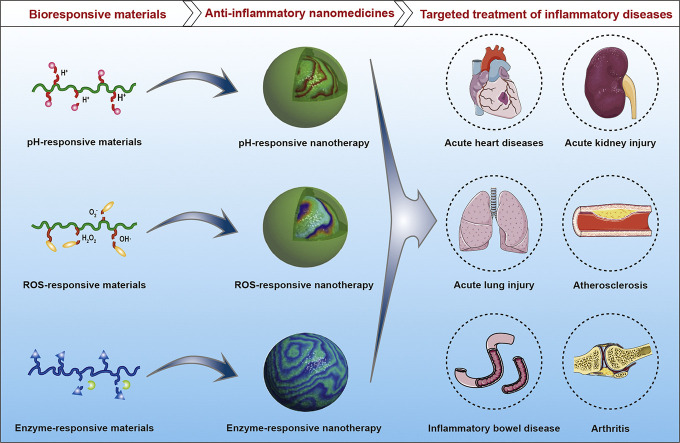
Recently, increasing evidence has demonstrated the advantages of bioresponsive delivery systems in the treatment of a diverse array of diseases, such as cancers, metabolic syndrome, and autoimmune/inflammatory diseases [33,40,[60], [61], [62], [63], [64], [65], [66]]. By more precisely regulating the cargo release behaviors at the sites of interest, bioresponsive carriers are able to considerably potentiate efficacies of different therapeutics and simultaneously reduce their side effects. In particular, biomaterials sensitive to various biochemical signals like abnormally changed pH, reactive oxygen species (ROS), and enzymes in diseased sites have been studied as stimuli-responsive delivery systems for targeted treatment of inflammatory diseases. Herein we provide a state-of-the-art review on bioresponsive drug delivery systems for the management of inflammatory diseases.
2. Inflammation and the inflammatory microenvironment
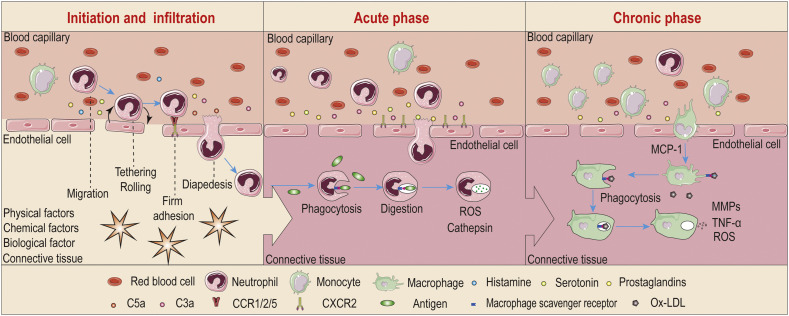
Inflammation is a biological self-response to endogenous and/or exogenous biophysicochemical stimuli, which is characterized by orchestration of a diverse array of molecular mediators and inflammatory cells (Fig. 1 ). According to the duration of inflammation, it can be classified into acute and chronic inflammatory responses. Acute inflammation, with the cardinal clinical signs of redness, heat, swelling, and pain [67], frequently lasts for several hours to days. Responding to invading pathogens, tissue injuries, or other noxious stimuli, acute inflammation is generally accompanied by rapid and dramatic accumulation of fluid, chemokines, cytokines, and inflammatory cells (particularly neutrophils) in the involved tissues/organs [68,69]. The acute inflammatory response is usually self-limiting, which can be rapidly resolved to restore tissue homeostasis. However, failure to remove a pathological insult or inability to realize resolution will lead to persistent tissue injury and a sustained inflammatory response for months to years, termed as chronic inflammation. Monocytes/macrophages are the primary contributors to the chronic inflammatory process by releasing a large number of inflammatory cytokines, which are intimately related to the pathogenesis of numerous chronic diseases. Although the initiation and progression of inflammation largely depends on types of stimuli and degrees of the body response, its common pathological and physiological features are intriguing targets for diagnosis and therapy of numerous inflammatory diseases. In the following section, we briefly introduce typical pathophysiological characteristics of the inflammatory microenvironment, which are informative for rational design of bioresponsive materials and drug delivery systems for the management of inflammation-associated diseases.
Fig. 1.
Inflammation and inflammatory microenvironments. C3a, complement 3a; C5a, complement 5a; CCR, CC-chemokine receptor; CXCR2, CXC-chemokine receptor 2; MCP-1, monocyte chemotactic protein-1; Ox-LDL, oxidized low-density lipoprotein.
2.1. Increased vascular permeability and infiltration of inflammatory cells
The most significant characteristic in the early stage of inflammation is the altered anatomy and function of the microvasculature, resulting in vasodilation, increased vascular permeability, and tissue leukocytosis [55]. The immediate responses are dominated by plasma- and cell-derived vasoactive mediators, such as fibrin, kinins, complement components (like C3a and C5a), histamine, serotonin, platelet-activating factor, and prostaglandins. These mediators can bind specific receptors on vascular endothelial cells, causing their contraction and gap formation, thereby leading to increased vascular permeability and extravasation of intravascular fluids within 30 minutes after injury. Meanwhile, these inflammatory mediators activate endothelial cells to increase the expression of different cell adhesion molecules, largely classified into four molecular families, i.e., selectins, cadherins, integrins, and immunoglobulins. These adhesion molecules expressed on endothelial cells then mediate extravasation of leukocytes from the bloodstream into the injured tissues via a precisely controlled process with sequential tethering, rolling, firm adhesion, and transmigration.
Different leukocyte subpopulations are involved in inflammatory reactions, mainly including neutrophils, eosinophils, basophils, lymphocytes, monocytes/macrophages, and mast cells. Particularly, as the most abundant leukocytes in the circulation (50-70%), neutrophils are the major cellular participant in acute inflammatory response, by carrying out their diverse functions, such as phagocytosis and digestion of pathogens, production of ROS, formation of neutrophil extracellular traps, as well as activation and regulation of innate and adaptive immunity [70,71]. In addition, neutrophils actively participate in chronic inflammatory conditions like chronic obstructive pulmonary disease [72], arthritis [73], IBD [74], and atherosclerosis [75]. Consequently, neutrophils have emerged as an exciting target for both imaging and therapeutic intervention of diseases associated with inflammation [[76], [77], [78], [79]]. Depending on the degree and persistence of injury as well as activation of different mediators, monocytes can migrate into the inflamed sites and differentiate into macrophages. The tissue-resident macrophages, together with the infiltrated macrophages and their cytokines play a key role in host defense and maintaining tissue homeostasis in the acute phase response of inflammation. Furthermore, sustained and uncontrolled macrophage responses in affected tissues are central to prolonged inflammatory and immune reactions, generally leading to chronic inflammation that is associated with a variety of infectious and noninfectious diseases, such as chronic hepatitis and atherosclerosis [4,5,80]. Macrophages have therefore been investigated as promising targets for imaging and treatment of various chronic inflammatory diseases [5,52,81]. As for mast cells, basophils, and eosinophils, they are important players in allergy, hypersensitivity, asthmatic responses, and parasite-associated inflammatory reactions [[82], [83], [84]]. Accordingly, these cells are interesting therapeutic targets for the related inflammatory diseases, despite their relatively low numbers in the circulation and local tissues. In addition to functioning as therapeutic targets, these different effector cells involved in inflammation can engulf different materials, followed by migration into inflammatory sites, making them interesting “Trojan Horses” for targeted delivery of different molecular and particulate therapeutics to inflamed sites [56,85,86].
2.2. Acidosis at inflammatory sites
Local acidification has been confirmed at inflamed sites of various diseases associated with acute and chronic inflammation, such as the peritoneum of peritonitis [87], injured arteries [88], ischemic sites [89,90], atherosclerotic plaques [91], asthmatic airways [92], rheumatic or gouty joints [93], and cerebrospinal fluid in the brain with meningitis [94]. Compared to healthy tissues, the inflamed areas generally display lower pH values. For instance, extracellular pH in the location of myocardial ischemia is approximately 6.5-6.0 [90], while pH as low as 4.2 was reported in the fracture microenvironment [95]. For patients with acute asthma, their exhaled vapor condensate showed pH 5.2, in contrast to pH 7.7 in control healthy subjects [96]. Normal synovial fluid exhibits pH 7.4-7.8, while synovial fluid in arthritic joints decreased to 6.6-7.2 [93,97,98], which is related to the intensity and state of inflammation and disease activity.
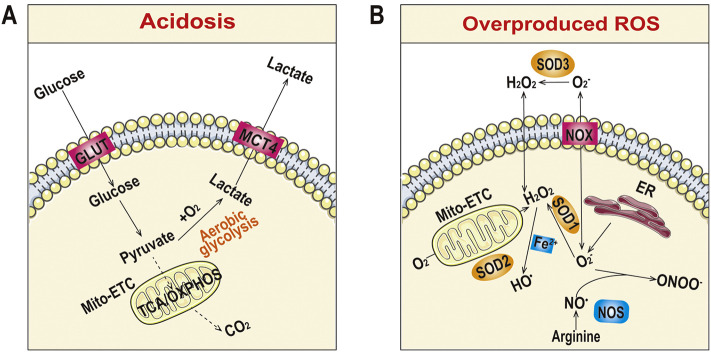
This acidosis feature (pH < 7.35) has been considered relevant to increased energy and oxygen consumption by infiltrated inflammatory cells, which shifts metabolism from aerobic to anaerobic styles in the inflamed tissues, leading to oxygen-independent glycolysis and increased lactic acid production (Fig. 2A) [99]. In the case of infection, bacterial metabolites like lactate may also contribute to acidification in the interstitial spaces [94,100]. In addition to metabolic acidosis, respiratory acidosis in inflammatory lung diseases (particularly asthma and chronic obstructive pulmonary disease) can be resulted from accidently inhaled acids such as those in fogs and air pollution [92]. The long-term exposure to acids is an important risk factor in the pathogenesis of asthma and bronchitis, which in turn leads to amplified acidosis due to altered airway pH homeostasis. By serving as an endogenous danger signal and regulating the synthesis and release of inflammatory mediators, extracellular acidosis itself can alter biological functions of different inflammatory and immune cells [99,101,102]. Whereas pH values in different inflammatory tissues were determined and compared to the corresponding normal tissues, few studies have been performed to quantify the degree of acidosis during the progression of specific inflammatory diseases. Nevertheless, some available findings revealed a first pH decrease upon the initiation of inflammation, followed by approximately sustained low pH values once inflammation persists [94]. In addition, acidosis can be alleviated by drug therapy [96]. Therefore targeting pH homeostasis might be a new therapeutic strategy to curb different inflammatory diseases [92,99,103]. Meanwhile, acidosis can be used as a very useful endogenous cue for designing delivery systems with acid-triggerable release behaviors for different therapeutics.
Fig. 2.
Typical pathological features in inflammatory tissues. (A) Acidosis. (B) Overproduced ROS. ER, endoplasmic reticulum; GLUT, glucose transporter; MCT4, monocarboxylate transport 4; Mito-ETC, mitochondrion-electron transport chain; NOX, NADPH oxidase; NOS, nitric oxide synthetase; OXPHOS, oxidative phosphorylation; SOD1, superoxide dismutase 1; SOD2, superoxide dismutase 2; SOD3, superoxide dismutase 3; TCA, tricarboxylic acid.
It should be noted that, besides these pathological conditions, varied degrees of acidic environments exist in specific organs, tissues, and cells of the human body under normal physiological conditions. For instance, the pH value along the gastrointestinal tract has a well-controlled gradient, ranging from 1.0-3.5 in the stomach to 5.5-7.5 in the intestine [104]. The normal pH range in the vagina is 3.8-4.5. In addition, subcellular compartments show different acidic microenvironments, with pH 5.0-6.5 in early and late endosomes, pH 4.5-5.0 in lysosomes, and pH 6.0-6.7 in the Golgi apparatus [63,105]. Regardless of pathological or physiological acidosis, the acidic environment has been widely studied as a trigger for site-specific drug delivery [65,[105], [106], [107]]. In particular, a large number of pH-responsive biomaterials and delivery systems have been engineered and applied for the treatment of inflammatory diseases.
2.3. Enhanced oxidative stress in inflammatory tissues
ROS are chemically reactive species containing oxygen. Typical ROS include hydrogen peroxide (H2O2), superoxide anion radical (•O2 −), hydroxyl radical (•OH), singlet oxygen (1O2), and hypochlorous acid (HOCl). Physiological levels of ROS play an important role in preserving and controlling cellular functions, such as cell differentiation, proliferation, migration, and survival. In addition, ROS are crucial bactericidal molecules for the host defense against various pathogens. However, excessive ROS, generally produced in cellular organelles such as mitochondria, peroxisomes, and endoplasmic reticulum of activated neutrophils and macrophages under inflammatory conditions (Fig. 2B), can cause oxidation of DNA, proteins, and lipids, thereby causing cell death by necrosis and/or apoptosis [108].
The ROS-mediated oxidative damage is closely related to the pathogenesis of numerous diseases associated with acute and chronic inflammation, such as acute ischemic stroke, acute liver/lung injury, acute bronchitis, acute gastroenteritis, neurodegenerative diseases, cardiovascular diseases, asthma, diabetes, IBD, and RA. In patients with IBD or Helicobacter pylori infections, their mucosal levels of ROS are 10-100 folds higher than those of the healthy population [109,110]. The injured artery of rats showed an average ROS level approximately 10 times higher than that of the normal controls [111]. In some brain diseases, the extracellular H2O2 concentration increased to 100 μM during ischemia and reperfusion, which caused modification of neuronal proteins and structural components as well as loss of cognitive function, eventually resulting in the occurrence of Alzheimer’s and Parkinson’s disease [112]. In inflammatory lung diseases, respiratory lining cells experienced 20-fold higher H2O2 levels than those in the healthy tissues [113]. Studies in animal models of acute lung injury, alcoholic liver injury, and colitis demonstrated that the ROS level generally exhibits a parabolic profile during the development of different inflammatory diseases, with the peak time dominating by the disease types and endogenous or exogenous inflammatory stimuli [[77], [78], [79]]. Consequently, the significantly increased oxidative stress in inflammatory sites is another endogenous biochemical trigger for targeted therapy of inflammatory diseases.
2.4. Overexpressed enzymes and other features under inflammatory conditions
In inflammatory diseases, a diverse array of enzymes will be up-regulated. During acute inflammation, different enzymes required for degradation of pathogens or foreign materials are generated in cytoplasmic granules of polymorphonuclear leukocytes, particularly neutrophils. For instance, lysozyme, myeloperoxidase (MPO), collagenase, elastase, and gelatinase can be released by degranulation of neutrophils [114,115], while acid hydrolases, elastases, cathepsins, and matrix metalloproteases (MMPs) will be secreted by activated monocytes/macrophages in both acute and chronic inflammatory responses [116]. The increased expression of some enzymes such as cyclooxygenase-2 directly mediates inflammation [117], and therefore being used as important therapeutic targets for the management of inflammatory disorders. In the case of infection, bacteria produce some specific enzymes like urease or azoreductase [118]. Regardless of their different sources, the inflammation-associated enzymes frequently display a notable high expression at the initial stage of inflammatory diseases [[77], [78], [79],[119], [120], [121]], which then decrease to a certain degree and maintain abnormally high levels during the whole process of inflammation. Recently, these inflammation-related enzymes have become emerging biological triggers for targeted treatment of inflammatory diseases, showing higher selectivity and sensitivity compared to the abnormal levels of pH and ROS.
In addition to the above described cellular and molecular features at inflammatory sites, other biochemical, physical, and anatomical characters exist in some inflammatory diseases. Hypoxia has been implicated in IBD, RA, cardiomyopathy, ischemia, and atherosclerosis [122,123]. The narrowing of blood vessels induced by atherosclerotic plaques, restenosis, or thrombosis can result in a significant increase in local fluid shear stress between healthy and constricted blood vessels [124,125]. Also, inflamed tissues show relatively high temperature as compared to that of the healthy tissues [126,127]. All these microenvironmental features can serve as potential targets or triggers for rational design of stimuli-responsive materials and delivery systems for a range of biomedical applications, including diagnosis, therapy, and tissue repair.
3. Materials and delivery vehicles responsive to inflammatory signals
Over the past decades, considerable advances have been made in the development of bioresponsive materials that can be processed into appealing therapeutic platforms for targeted treatment of a plethora of diseases [33,[60], [61], [62], [63], [64], [65],105,[128], [129], [130]]. In particular, pH-, ROS-, and/or enzyme-responsive materials and their delivery systems have been extensively examined for therapy of inflammation-associated diseases. Although these pathological signals are largely localized at the inflamed site, they generally show gradient distribution, with the highest level at the diseased region. Accordingly, the highest sensitivity and selectivity can be achieved at the site of action. In addition, bioresponsive nanomedicines can passively target inflammatory tissues due to enhanced vasculature permeability, and their targeting efficiency may be further enhanced by surface engineering with different targeting moieties or biomimetic strategies, thereby notably increasing sensitivity and specificity. In this section, we briefly describe different biomaterials responsive to inflammatory environments, mainly including pH-, ROS-, and enzyme-responsive materials/carriers.
3.1. pH-responsive materials and delivery systems
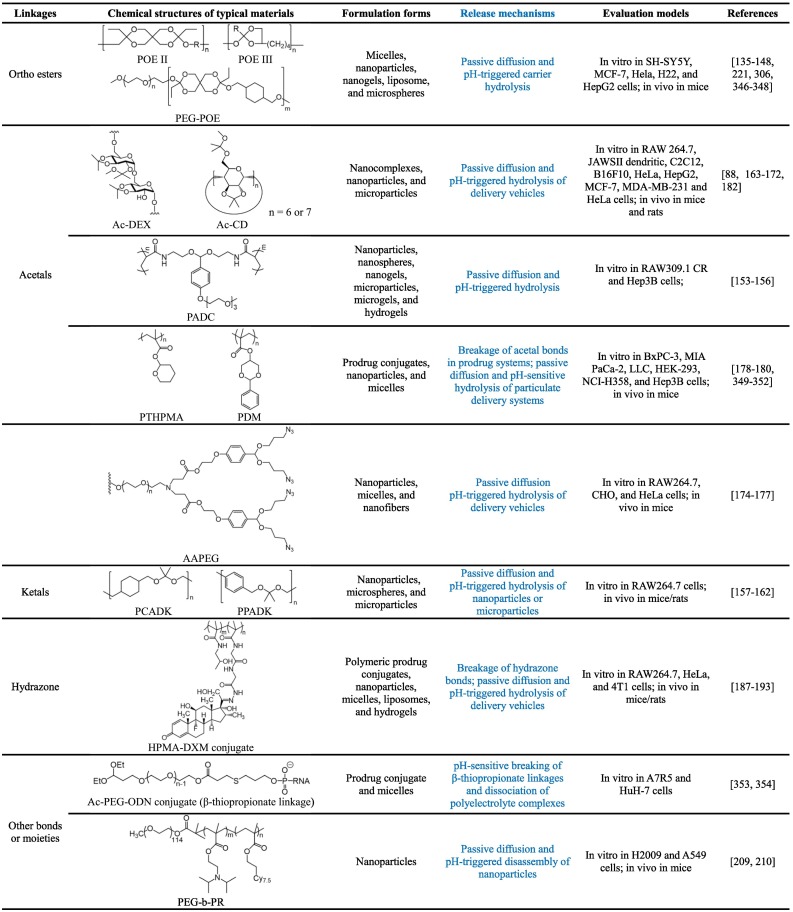
Due to the presence of acidic microenvironments under various pathophysiological conditions, pH-responsive materials have been extensively investigated for site-specific delivery of different therapeutic agents in the formulations across multiple scales [104,105,[131], [132], [133]]. The pH-sensitivity can be attributed to either hydrolysis of acid-labile bonds or protonation of ionizable groups. The acid-cleavable bonds mainly include ortho esters, acetal/ketal moieties, hydrazone/oxime linkages, β-thiopropionate linkage, silyl ether, vinyl ether, and phosphoramidate, showing accelerated hydrolysis in acidic environments (Table 1 ). As a different type of pH-sensitive materials, amine-bearing polymers, such as chitosan and its derivatives, display increased solubility at low pH, resulting from protonation of amine moieties.
Table 1.
3.1.1. Poly(ortho esters) and ortho ester-containing materials
Poly(ortho esters) (POEs) are probably the most widely and intensively studied acid-cleavable polymers, due to their excellent biocompatibility. The development of POEs can be traced back to the early 1970s [134]. Four families of POEs were developed as new bioerodible polymers by Jorge Heller and coworkers [135,136], which were specifically designed for drug delivery to avoid disadvantages of traditionally used polyesters such as poly(lactic acid) (PLA) and its copolymers. Generally, these polymers can be synthesized by the addition reaction between polyols and diketene acetals. By using diols with different degrees of chain flexibility, POEs with distinct physicochemical and mechanical properties can be obtained, such as gel-like, brittle, and hard materials [137]. For POE-based delivery systems, such as wafers and microspheres, their erosion and drug release periods may be facilely regulated from a few days to many months. Initially, different forms of POEs were fabricated to deliver small molecule drugs for the treatment of post-surgical pain, osteoarthritis, and ophthalmic disease [138], which were used later as carriers for peptides, proteins, and nucleic acids [137,139,140]. Also, block copolymers based on POEs and poly(ethylene glycol) (PEG) were synthesized and used as nanoparticles or micelles for targeted drug delivery [141].
Whereas POEs show desirable pH-sensitivity and good biocompatibility, their synthesis frequently require careful control of reaction conditions to obtain products with tailored molecular weight and defined physicochemical properties. To address this issue, ortho esters have recently been incorporated as acid-labile moieties in different polymers to synthesize pH-responsive materials. For example, ortho esters were used to prepare pH-responsive lipid conjugates for liposome formulations [142]. Pendent cyclic ortho esters were introduced in hydrophilic polymers to prepare acid-labile carrier materials for pH-triggerable drug delivery [143,144]. In addition, ortho esters have been incorporated in the polymer main chains or used as cross-linkers to construct pH-responsive nanovehicles [145,146]. By chemical functionalization of β-cyclodextrin (β-CD) with ortho esters, pH-sensitive CD derivatives were obtained, which were fabricated to pH-sensitive supramolecular nanoparticles and polypseudorotaxanes for drug delivery [147,148]. Nevertheless, for these ortho ester-containing materials, their in vivo safety profiles remain to be addressed by comprehensive in vivo studies.
3.1.2. Materials bearing acetals or ketals
Acetals/ketals are frequently used as protective groups for aldehydes and ketones in organic synthesis [149]. In drug delivery fields, both acetals and ketals were initially used as acid-cleavable agents to develop prodrugs derived from either small molecule drugs or proteins [150,151]. Studies by Fréchet’s group suggested that acetals can function as pH-sensitive linkages to fabricate diverse drug delivery systems, such as polymer-drug conjugates, micelles, nanoparticles, and microgels [[152], [153], [154]]. A similar approach was adopted by other researchers to develop pH-sensitive polymeric nanogels or hydrogels [155,156]. Of note, the hydrolysis rate of resulting systems at different pH values can be easily tailored by modulating the types of alcohol and carbonyl (aldehyde and ketone). In other studies, acetal/ketal moieties were embedded in the main chain of hydrophobic polymers to synthesize fully acid-degradable biocompatible materials that can be further processed into either nanoparticles or microparticles for site-specific delivery of different therapeutic agents [[157], [158], [159], [160], [161], [162]].
Fréchet et al. also reported facile synthesis of acid-responsive materials by acetalation of dextran using 2-methoxypropene or other functional molecules [[163], [164], [165]]. Thus obtained acetalated dextran polymers have been examined for encapsulation and pH-triggerable release of small molecules, nucleotides, and proteins in the forms of nanoparticles or microparticles [164,[166], [167], [168], [169]]. Our group demonstrated that kinetically controlled acetalation can be carried out for cyclic oligosaccharides, such as α-CD, β-CD, and their polymers, thereby affording a series of acid-labile biocompatible materials [170,171]. By regulating acetalation time, the pH-sensitive hydrolysis rate of resulting CD materials may be easily modulated. Notably, systemic in vivo studies suggested that acetalated CD materials exhibited good safety profile after administration by subcutaneous, intramuscular, intraperitoneal, or intravenous (i.v.) injection in mice, rats, and rabbits [[170], [171], [172], [173]]. In other cases, acetals were introduced as acid-labile linkages in hyperbranched polymers and dendrimers to construct different delivery systems [[174], [175], [176], [177]]. Also, linear and cyclic acetals can be incorporated in the side chains of polymers to develop acid-sensitive polymers or polymer-drug conjugates [[178], [179], [180]]. In addition to their excellent acid-sensitivity and facile synthesis procedures, most acetal- and/or ketal-derived pH-responsive materials do not generate acidic byproducts after hydrolysis, thereby avoiding significant proinflammatory effects [171,181,182]. By contrast, frequently used polyesters, such as PLA and poly(lactide-co-glycolide) (PLGA), can induce local inflammation in different tissues [183,184]. For future translation, however, in vivo safety of different types of acetal/ketal-derived materials and delivery systems need to be intensively examined in various animal models.
3.1.3. Materials with hydrazone linkages
Hydrazone linkages have been widely used in pharmaceutical sciences due to their excellent pH-sensitivity as well as synthetic flexibility and diversity [133,185,186]. Similar to other acid-labile linkages, hydrazones are generally employed to synthesize both prodrugs and delivery carriers to achieve programmed drug release in response to the intracellular or extracellular acidic microenvironment. As a decent example, Kataoka’s group synthesized an amphiphilic block copolymer in which doxorubicin was conjugated onto the polyaspartate block via hydrazone [187]. Polymeric micelles assembled by this pH-sensitive copolymer showed notable pH-dependent release profiles. By the similar strategy, other small molecule drugs, such as dexamethasone, prednisolone, isoniazid, and all-trans retinal, were conjugated onto hydrophilic polymers or inorganic nanoparticles via the hydrazone bond to construct pH-sensitive drug-polymer conjugates or prodrug nanoparticles with on-demand therapeutic release under different inflammatory conditions [[188], [189], [190], [191], [192], [193]]. As compared to pH-sensitive linkers of ortho esters, acetals, or ketals, the hydrazone bond is more useful to develop acid-triggerable prodrug systems.
On the other hand, pH-sensitive hydrazone linkages have been utilized to synthesize different pharmaceutical materials, enabling the development of diverse smart delivery systems, such as liposomes, micelles, nanoparticles, and hydrogels [[194], [195], [196], [197]]. Of note, PEG conjugates with hydrazone linkers are frequently used to fabricate long-circulating nanocarriers with acid-responsive sheddable coatings [198], which are particularly intriguing for intracellular delivery of nucleic acids. Nevertheless, these delivery platforms generally show limited control over pH-sensitive drug release, due to the presence of passive diffusion, as compared to hydrazone-derived prodrug nanoparticles and other vehicles with acid-labile matrices.
3.1.4. Materials with other acid-labile linkages
To prepare pH-sensitive delivery systems, β-thiopropionate, phosphoramidate, vinyl ether, boronate, and silyl ether were also used as acid-sensitive linkages. The acid-labile β-thiopropionate linkage was employed by Kataoka and colleagues to synthesize conjugates of PEGylated antisense oligonucleotides or siRNA, which were further assembled into polyion complex micelles for intracellular gene silencing [199]. Similarly, oligonucleotides can be covalently conjugated with PEG via an acid-cleavable phosphoramidate linkage, and the obtained conjugate is able to complex with a cationic peptide to form polyelectrolyte complex micelles for efficient intracellular delivery [200]. Likewise, phosphoramidate linkers were used to synthesize nanoformulated polymeric prodrugs for antiretroviral therapy [201,202]. In other cases, PEGylated dioleoylphosphatidylethanolamine (DOPE) conjugate with an acid-liable vinyl ether linker was used to prepare liposomes or nanoassemblies to achieve pH-triggerable drug release or gene delivery [203,204]. Also, boronate and silyl ether were used as cross-linkers to synthesize acid-sensitive biomaterials in the forms of nanoparticles, macroscale particles, sutures, or stents [205,206]. For these delivery vehicles, however, only a few studies have been conducted to investigate their pH-responsive drug release performance.
Different from the above mentioned pH-sensitive materials based on acid-labile linkages, Gao’s group designed and developed a library of tunable, ultrasensitive pH-activatable micelles self-assembled by ionizable block copolymers, in which tertiary amines serve as precisely controlled hydrophobic substituents in the ionizable hydrophobic blocks [207,208]. This type of micellar nanoparticles exhibited fast and ultrasensitive response to changes over the entire physiological arrange of pH, depending on the types of tertiary amine substituents. In addition to functioning as ultra-pH-sensitive fluorescent nanoprobes, these nanoparticles can be used for developing pH-activatable nanotherapies and nanovaccines [209,210].
3.2. ROS-responsive materials and delivery systems
In view of overproduced ROS at different diseased sites, considerable advances have been achieved in recent years in the development of ROS-responsive materials and delivery systems for on-demand drug release and targeted therapy (Table 2 ). In this section, we introduce typical ROS-responsive materials and delivery platforms.
Table 2.
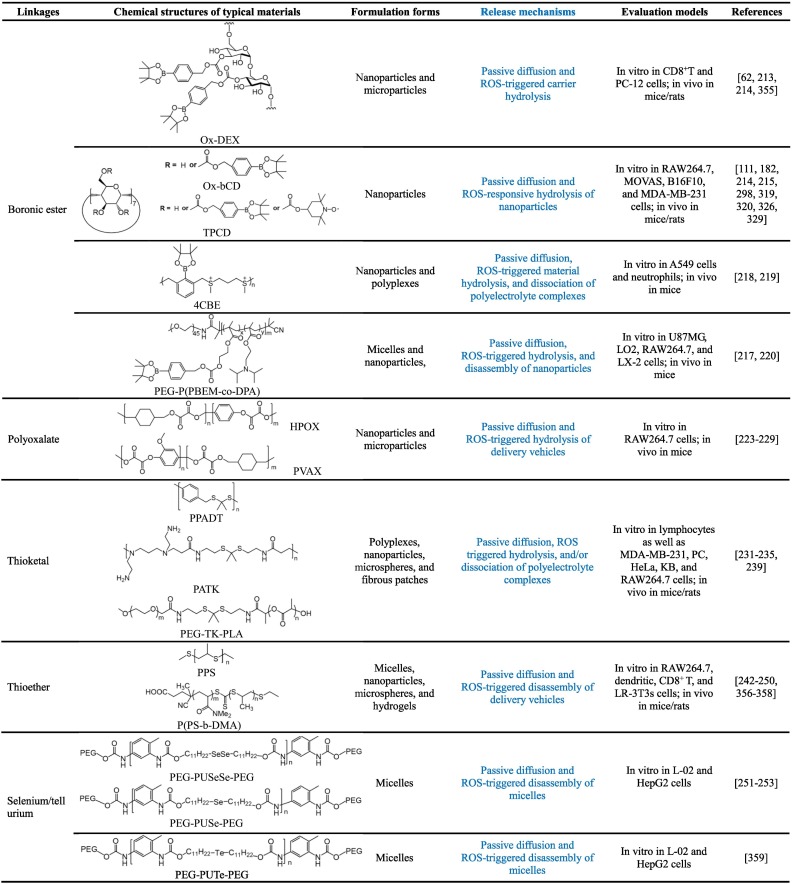
3.2.1. Boronic ester-containing materials
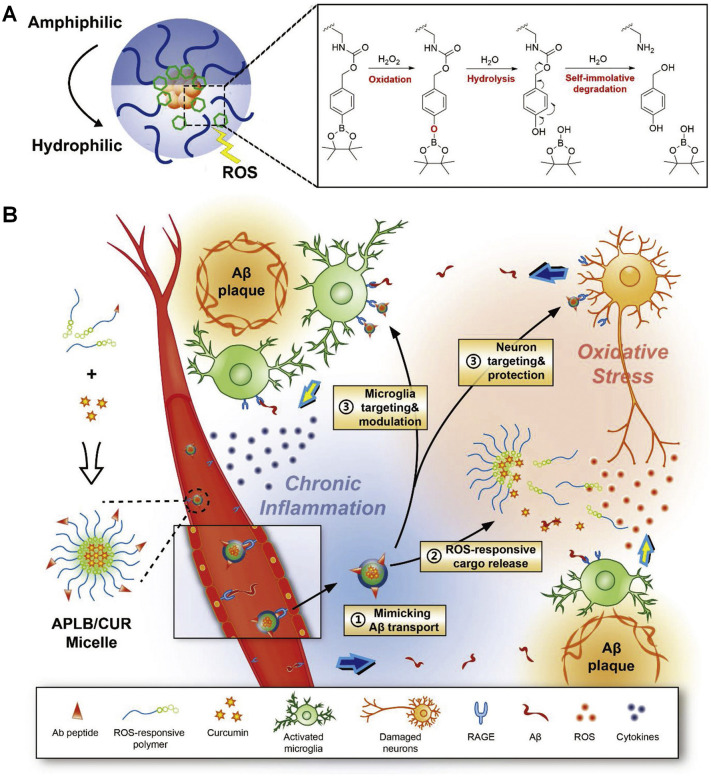
In organic synthesis, boronic acids and their derivatives are frequently used as good protective groups for diols. Since the linkages between boronic esters and the conjugated molecules can be easily cleaved under oxidative conditions, they have been extensively employed for the development of ROS-responsive fluorescent probes, prodrugs, and pharmaceutical materials [62,[211], [212], [213], [214]]. An early study by Fréchet and coworkers demonstrated successful synthesis of ROS-responsive materials by covalently conjugating an oxidation-labile unit of 4-(hydroxymethyl)phenylboronic acid pinacol ester (PBAP) onto dextran, and the functionalized polymer can be further produced into microparticles for ROS-triggerable therapeutic delivery [213]. Our studies showed that this functionalization strategy can be conducted for cyclic oligosaccharides (such as β-CD) [214,215], thereby offering ROS-responsive carrier materials. Notably, H2O2-sensitivity of resulting materials is regulatable by changing the content of PBAP or the linker type between PBAP and β-CD [215]. At a comparable content of PBAP, while materials containing either carbonate or carbamate linkages showed rapid hydrolysis in the presence of H2O2, the ester bond-based materials displayed considerably low H2O2-sensitivity. Regardless of their chemical structures, all these materials are able to be processed into nanoparticles with tunable sizes by a nanoprecipitation/self-assembly method. Furthermore, both in vitro and in vivo evaluations by our group substantiated that this type of ROS-responsive CD materials exhibited good safety profiles [214,215]. Also, phenylboronic acid or its ester was employed to functionalize hydrophilic polymers to achieve ROS-triggerable gene delivery [216,217].
In addition to functionalization of hydrophilic materials, phenylboronic acid pinacol ester was incorporated into the main or side chains of polymers to synthesize ROS-responsive materials for delivery of small molecule therapeutics and efficient gene delivery [[218], [219], [220]]. In other cases, pH/ROS dual-responsive polymers were synthesized by covalently incorporating phenylboronic acid pinacol ester into ortho ester-containing block copolymers [221]. Our recent studies suggested that pH/ROS dual-responsive nanoparticles can be easily constructed by physically combining acid-labile acetalated β-CD and PBAP-conjugated β-CD materials [88]. Of note, for ROS-responsive materials based on the boronic ester strategy, they can be easily obtained by relatively facile and effective synthesis methods, which is a considerable advantage for large-scale production and future translation. Nevertheless, hydrolysis of all the aforementioned boronic ester-containing materials can generate boronic acids that exhibit biological activities to a certain degree, therefore their possible side effects and/or synergistic effects should be took into account during the treatment of specific diseases.
3.2.2. Polyoxalate materials
Polyoxalate-based nanoparticles were initially examined for imaging of H2O2, owing to the nanomolar H2O2-sensitivity of polyoxalate materials and their specificity to H2O2 over other ROS types [222]. Later Lee et al. synthesized polyoxalate materials for H2O2-responsive therapeutic delivery [223]. As a representative example, they synthesized a bioactive polyoxalate material (i.e., HPOX) by a condensation reaction of oxalyl chloride, 1,4-cyclohexamethanol, and p-hydroxybenzyl alcohol (HBA) [224]. In the presence of ROS, HPOX can be triggerably hydrolyzed into three safe compounds, i.e., cyclohexanedimethanol, HBA, and CO2. Of note, HBA is a major bioactive component of Gastrodia elata, a widely used herbal agent for the treatment of inflammation and oxidative stress-related diseases, such as ischemic injury and coronary heart diseases. HPOX can be produced to nanoparticles by an oil-in-water solvent evaporation method, affording nanotherapies with H2O2-activatable hydrolysis and drug release properties as well as antioxidant activity [225,226]. According to the similar procedures, nanoparticles or microparticles derived from a vanillyl alcohol-containing copolyoxalate PVAX were developed as H2O2-responsive, biodegradable, and bioactive therapeutic agents for the treatment of acute oxidative injury-related diseases [[227], [228], [229]]. Despite the facile method for synthesis of this type of ROS-responsive materials, it is worth noting that oxalate should be linked with aryl moieties to afford materials with desirable H2O2-sensitivity, while only a very limited number of monomers with good biocompatibility are commercially available.
3.2.3. Thioketal-containing materials
Due to their excellent stability in both acidic and basic conditions, thioketals are also popularly used protecting groups for carbonyl in organic synthesis, which can be efficiently cleaved by ROS [230]. Taking advantage of this unique ROS-labile property, Murthy et al. synthesized a ROS-responsive polymer, i.e., poly(1,4-phenyleneacetone dimethylene thioketal) (PPADT), by condensation polymerization of 1,4-benzenedimethanethiol and 2,2-dimethoxypropane [231]. In the presence of superoxide, PPADT was hydrolyzed into small molecules. It was also found that PPADT nanoparticles were able to triggerably release the loaded fluorescent molecules in activated macrophages. Inspired by this study, different thioketal-containing polymers have been designed and synthesized as ROS-responsive materials for diverse delivery applications. For example, a thioketal polymer was synthesized by step-growth polymerization of (4,4’-bis(mercaptomethly)biphenyl and 2,2-dimethoxypropane [232]. ROS-responsive microspheres were further prepared using this polymer for inflammation-triggered local delivery of tacrolimus, a powerful immunosuppressant. Gao et al. synthesized ROS-responsive and biodegradable polyurethane by reaction of poly(ε-caprolactone) diol, 1,6-hexamethylene diisocyanate, and a thioketal-containing chain extender, which was fabricated into fibrous patches for therapeutic delivery [233]. Wang’s group reported a thioketal linkage-bridged diblock copolymer of PEG and PLA for on-demand drug delivery [234].
In other studies, the thioketal linkage was introduced into cationic polymers for ROS-responsive gene transfection [235,236]. Also, different therapeutic molecules can be covalently conjugated onto polymers via the thioketal linker to formulate prodrug nanotherapies [[237], [238], [239]]. In addition, by simultaneously incorporating thioketal and disulfide linkages in the backbone of polymers, biodegradable nanoparticles dually responsive to H2O2 and glutathione (GSH) were developed for programmable drug release [240]. It should be emphasized that for thioketal-containing materials and delivery systems, good ROS-sensitivity is only achieved at high levels of H2O2 or in the presence of other strong oxidizing agents.
3.2.4. Materials with thioether linkages and other chalcogen-containing polymers
While the above mentioned ROS-responsive materials are hydrolyzed to realize on-demand drug release, another types of ROS-sensitive materials are based on the hydrophobic to hydrophilic change after oxidation of specific groups. Organic sulfides are frequently used to synthesize ROS-responsive materials and delivery systems, since they can undergo conversion from hydrophobic sulfides to more hydrophilic sulfoxides or sulfones under oxidative conditions. Early in 2004, Hubbell’s group developed oxidation-responsive polymeric vesicles by self-assembly of triblock copolymers consisting of PEG and poly(propylene sulfide) (PPS), which can be easily synthesized by one-pot ring-opening polymerization of episulfide [241]. In the presence of H2O2, the assembled vesicles showed oxidative destabilization, resulting from the increased hydrophilicity due to the oxidation of sulfide moieties in the hydrophobic PPS block into sulfoxides/sulfones.
On the basis of this finding, different types of PPS-derived materials were synthesized to prepare ROS-responsive delivery vehicles. Duvall’s group developed ROS-responsive micelles for triggered drug release, using a copolymer of PPS and poly(N,N-dimethylacrylamide) [242]. The micelles demonstrated efficient drug release in response to H2O2 or endogenous oxidants generated in lipopolysaccharide (LPS)-induced RAW264.7 macrophages. By the living emulsion polymerization of episulfides, cross-linked polysulfide nanoparticles with oxidation-sensitivity were produced [243]. Similarly, Allen et al. fabricated PPS nanoparticles by ring-opening emulsion polymerization using activated pentaerythritol tetrathioester as a four-armed star initiator, thereby affording ROS-responsive nanoplatforms capable of loading hydrophobic molecules [244]. In addition, microspheres were directly prepared by the well-established oil-in-water emulsion solvent evaporation method using PPS [245], while hydrogels can be fabricated using PPS-containing copolymers [246]. Nanoparticles, polymersomes, microparticles, and hydrogels based on PPS-derived biomaterials have been extensively investigated as ROS-responsive delivery vehicles for small molecule drugs, proteins, and therapeutic cells, in which therapeutics can be packaged by either physical entrapment or covalent conjugation [[245], [246], [247], [248], [249], [250]].
Other chalcogen-containing polymers have also been examined as bioresponsive materials for drug delivery. In a pioneer study by Zhang and Xu et al., an amphiphilic copolymer (PEG-PUSeSe-PEG) with one block of diselenide-containing polyurethane and two PEG blocks was synthesized [251], which can assemble into micelles in aqueous solutions. Upon stimulation by either oxidants or reductants, PEG-PUSeSe-PEG micelles were disassembled, resulting from a structural dissociation of the diselenide bonds. Following studies suggested that micelles assembled from monoselenide-containing amphiphilic block copolymers also showed oxidation-responsive dissociation and drug release behaviors [252], mainly due to the structural transition of hydrophobic selenide groups into hydrophilic selenoxide or selenone groups. Subsequently, different selenium-containing polymers with diverse structures were synthesized and explored as ROS-triggerable materials for biomedical applications [253,254]. Based on the similar design principle, Xu’s group also synthesized tellurium-containing compounds or polymers to construct ROS-responsive assemblies [255]. Nevertheless, similar to thioketal-derived responsive systems, chalcogen-containing polymers show good sensitivity only at high ROS levels.
3.3. Enzyme-responsive materials and delivery systems
Overexpressed enzymes under diseased conditions have long been employed as biological signals for the design and engineering of different bioresponsive materials [[256], [257], [258]]. The responsive mechanisms mainly involve the reduction/oxidation of substrates and the formation/cleavage of chemical bonds in the presence of enzymes. Many enzymes, such as proteases, phosphatases, kinases, and oxidoreductases have been concerned in the development of stimuli-responsive biomaterials. Herein we mainly describe biomaterials sensitive to enzymes overexpressed under inflammatory conditions (Table 3 ).
Table 3.
Enzyme-responsive systems for drug delivery.
| Enzymes | Sensitive sequences or moieties | Formulation forms | Release mechanisms | Evaluation models | References |
|---|---|---|---|---|---|
| MMPs | GGRMSMPV, GPQGIAGQ, GPQGIWGQ, PLGLAG | Nanoparticles and hydrogels | Passive diffusion and enzyme-triggered hydrolysis of delivery vehicles | In vitro in B16-F10 cells; in vivo in mice, rats, and pigs | [[261], [262], [263], [264], [265], [266]] |
| Neutrophil elastase | K(ROX)AAPVRGGGK(QXL) | Hydrogels | Passive diffusion and enzyme-triggered hydrogel hydrolysis | In vitro in a reaction-diffusion model | [259] |
| Cathepsin D | Phe-Phe | Prodrugs | Enzyme-triggered breakage of peptide linkages | In vitro in HFLS cells; in vivo in mice |
[260] |
| Furin | RVRRSK | Nanocapsules | Passive diffusion and enzyme-triggered hydrolysis of nanocapsules | In vitro in HeLa and A549 cells; in vivo in mice | [267,268] |
| Protein kinase Cα | HFKKQGSFAKKK-NH2 | Nanoparticles | Passive diffusion and enzyme-triggered hydrolysis of nanoparticles | In vitro in B16 melanoma cells; in vivo in mice | [269] |
| Caspase-3 | AGVA | Nanocapsules | Passive diffusion and enzyme-triggered hydrolysis | In vitro in HeLa cells | [270] |
| MPO | Luminol | Nanoparticles | ROS- and enzyme-triggered hydrolysis of nanoparticles | In vitro in neutrophils and macrophages; in vivo in mice | [78,271] |
Extracellular enzymes like neutrophil elastase, cathepsins, and MMPs are the most frequently studied enzymatic cues in inflammation for designing responsive materials and delivery systems. Aimetti et al. developed a novel enzyme-cleavable hydrogel delivery system based on a neutrophil elastase-sensitive peptide linker (Ala-Ala-Pro-Val), its degradability was demonstrated in the presence of corresponding elastase in vitro [259]. Homma et al. found that Phe-Phe chain can serve as a cathepsin-cleavable peptide linkage to synthesize a polymeric prodrug of methotrexate-conjugated hyaluronic acid, showing effective in vivo efficacy in rats with collagen-induced arthritis [260]. By introducing MMP-cleavable peptide sequences (such as GPQGIAGQ, GPQGIWGQ, and GGRMSMPV) into hydrogels as crosslinkers, MMP-sensitive hydrogels based on either synthetic or natural polymers can be synthesized [[261], [262], [263]]. In most recent studies, the peptide PLGLAG linker was used to synthesize MMP-2/9-responsive prodrug nanotherapies [264], nanovehicles [265], and hydrogels [266].
Also, biomaterials and delivery systems responsive to intracellular enzymes, such as furin, protein kinase Cα (PKCα), and caspases have been synthesized. A study by Tang and Gu et al. demonstrated that carriers containing furin-cleavable peptide RVRRSK crosslinkers can be degraded gradually to release cargo protein along their cellular uptake pathway [267]. The same group developed a graphene-based co-delivery system, in which a cell membrane targeting anticancer protein was covalently linked with a furin-cleavable peptide to achieve sequential delivery of different therapeutics [268]. In other studies, polymers containing PKCα-specific peptide substrates were designed and prepared for targeted gene delivery [269], while caspase-3-cleavable polymeric nanocarriers were constructed using a caspase-3 substrate-based peptide linker for intracellular site-specific delivery of caspase-3 [270]. By covalently conjugating luminol onto cyclic oligosaccharides (such as α-CD and β-CD), MPO-responsive materials can be easily synthesized for imaging and therapy of inflammatory diseases [78,79,271]. Whereas enzyme-responsive delivery systems exhibit extremely higher specificity than those responsive to pH or ROS, sensitive peptides are generally required for their preparation, thereby resulting to complicated procedures and high cost in most cases.
3.4. Other bioresponsive materials and delivery systems
Besides the above mentioned biomaterials, other bioresponsive materials have also been explored for anti-inflammatory applications. In this aspect, different redox-sensitive materials and delivery systems have been developed for the treatment of different inflammatory diseases. For example, Xiao et al. synthesized a reducible cationic polymer, by Michael addition between cystamine bisacrylamide (CBA) and branched polyethylenimine (PEI, Mw = 800 Da), which was further conjugated with mannosylated PEG. The final material, named as PPM, can complex with tumor necrosis factor (TNF)-α siRNA to obtain redox-sensitive nanoparticles of 211-275 nm for colitis therapy [272]. Kim et al. designed redox-sensitive poly(oligo-L-arginine) (rsPOLA) that can form nanoplexes with endothelial nitric oxide synthase (eNOS) DNA for targeted therapy of atherosclerosis [273]. In the presence of a reducing agent β-mercaptoethanol (β-ME), eNOS/rsPOLA nanoplexes showed considerably increased diameter, resulting from reduced condensation interaction. Recently, Liu and coworkers developed an amphiphilic inulin polymer of 4-aminothiophenol-conjugated carboxymethyl inulin (i.e., ATP-CMI), which could assemble into redox-sensitive nanoparticles of about 210.18 nm [274]. In the presence of GSH, ATP-CMI nanoparticles loaded with budesonide showed notable increased drug release, due to breaking of disulfide bonds and subsequent disassembly of ATP-CMI chains. To further improve the responsive efficiency, a pH/redox-dual responsive polymeric nanoliposome system was designed for targeted delivery of a copper-liganded bioactive complex to the inflammatory site. The prepared nanoliposome displayed significantly accelerated drug release at pH 5 in a reducing environment [275].
4. Treatment of inflammatory diseases by bioresponsive drug delivery systems
To overcome limitations of traditional formulations of different anti-inflammatory drugs, bioresponsive drug delivery systems have received much attention for the treatment of inflammatory diseases in recent years. Herein we describe applications of different bioresponsive systems in the management of typical diseases associated with acute or chronic inflammation.
4.1. Therapy of acute inflammatory diseases
4.1.1. Acute cardiovascular diseases
Acute cardiovascular diseases (CVDs), such as acute heart failure, acute myocardial infarction (MI), and acute ischemic stroke, remain one of the leading causes of global death. These fatal diseases are always characterized by acute inflammatory response and elevated oxidative stress that need to be rapidly eliminated in clinical treatment [276,277]. Recently, there has been increasing interest in advanced drug delivery systems for the treatment of acute CVDs [46,49,66,[278], [279], [280], [281]]. Among them, bioresponsive materials and systems show considerable advantages in site-specific delivery and responsive release of different therapeutic agents at diseased tissues [282,283].
Taking advantage of the mildly acidic microenvironment of ischemic myocardium, Davis et al. synthesized pH-sensitive poly(cyclohexane-1,4-diyl acetone dimethylene ketal) (PCADK), which was further formulated into microparticles (~20 μm) containing a p38 mitogen-activated protein kinase (MAPK) inhibitor SB239063 [181]. Treatment by direct cardiac injection of drug-loaded microparticles (defined as PK-p38i) significantly improved cardiac dysfunction following MI, resulting from sustained and controlled release of therapeutics within the damaged myocardium. Notably, PK-p38i showed considerably higher efficacy than free drug and PLGA-based microparticles. Moreover, degradation of PCADK generates neutral and excretable compounds, different from frequently used polyesters such as PLGA that produces proinflammatory acidic byproducts after hydrolysis. Consequently, PCADK-derived delivery systems are very promising for treating MI and other inflammatory diseases. In a following study, the same group demonstrated that acid-sensitive polyketal nanoparticles (~500 nm) loaded with NOX2-NADPH oxidase siRNA can significantly attenuate NOX2 expression and recover cardiac function by inhibiting oxidative stress, after intramyocardial injection in mice with MI [162]. In other studies, pH-responsive microparticles based on acetalated dextran were examined for tunable release of a cardioprotective growth factor to the heart by local delivery [284], and peptide-functionalized acetalated dextran-derived nanoparticles (100-200 nm) were used for M2-like macrophage-mediated targeted delivery of small hydrophobic compounds to the infarcted heart for MI therapy [285]. Murry and coworkers designed a pH-responsive, injectable hydrogel [286], based on a pH- and temperature-responsive copolymer, i.e., poly(N-isopropylacrylamide-co-propylacrylic acid-co-butyl acrylate). In aqueous solution, the employed random copolymer was liquid at pH 7.4, while it formed hydrogel at pH 6.8. After injection into infarcted rat myocardium, this delivery system dramatically increased retention of basic fibroblast growth factor (bFGF). Correspondingly, 28 days of treatment with bFGF-loaded pH-responsive hydrogel significantly improved capillary and arteriolar densities in vivo, compared to bFGF in saline. Also, treatment with polymer/bFGF resulted in significant improvement in regional blood flow, whereas control groups showed no effect.
In view of overproduced ROS in ischemic myocardium, ROS-responsive delivery systems were investigated for the treatment of acute MI. For example, ROS-responsive nanoparticles self-assembled by a diblock copolymer PEG-b-PPS were used to encapsulate a natural product ginsenoside Rg3. In a rat model of myocardial ischemia-reperfusion injury, intramyocardial treatment with Rg3-loaded PEG-b-PPS nanoparticles significantly decreased the plasma levels of TNF-α, interleukin (IL)-1β, IL-6, and C-reactive protein (CRP), therefore resulting in notably improved cardiac function and reduced the infarct size [247]. In addition, fibrous patches based on polyurethane containing thioketal linkages were used as a ROS-responsive delivery platform of methylprednisolone, showing desirable effects with respect to free radical scavenging, rebuilding structures, and improving functions of infarcted myocardium after implantation in MI rats [233]. Also, captopril-loaded mesoporous silica nanoparticles of ~120 nm with H2O2-triggerable release performance were examined for therapy of heart failure in a zebrafish model [287].
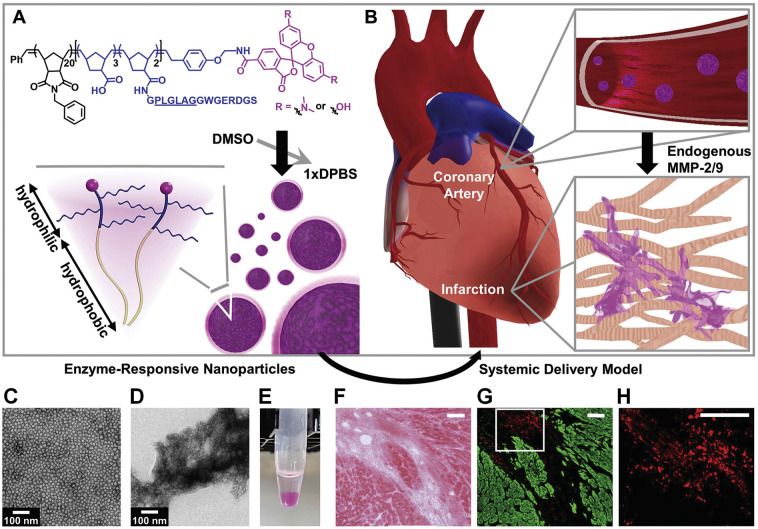
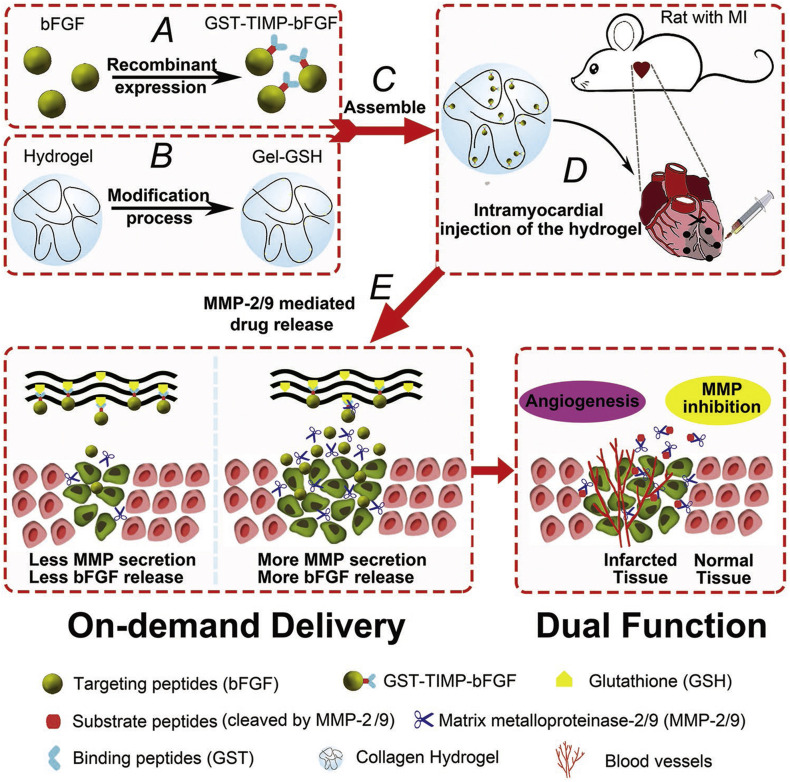
Also, abnormally increased enzymes can serve as effective endogenous biological stimuli to realize enhanced accumulation and/or triggerable release of different therapeutics for the treatment of acute CVDs. Christman’s group designed enzyme-responsive nanoparticles for targeted delivery and prolonged retention of therapeutic agents in the heart tissue after MI (Fig. 3 ) [288]. The mentioned nanoparticles with the average diameter of 20 nm were assembled by a brush peptide-polymer amphiphile (PPA) with a polynorbornene backbone bearing peptide sequences that can be specifically recognized by MMP-2 and MMP-9, while both MMP-2 and MMP-9 are endogenously expressed inflammation-related enzymes post MI. After i.v. injection, the enzyme-responsive nanoparticles efficiently accumulated in the infarct tissue through the leaky vasculature post-MI. Subsequently, up-regulated MMPs at the infarct site induced a morphological transition from discrete nanoparticles into network-like scaffolds, resulting in dramatically increased retention at the injured site for up to 28 days after injection. In another study by Gianneschi and Christman et al., they engineered peptide progelators that are water-soluble cyclic progelators showing free flow in solution until proteolytically activated by MMP-2/9 and elastase [289]. After local injection at the site of MI in rats, enzymatic cleavage of cyclic progelators generated linear self-assembling peptides, which rapidly assembled into hydrogels. Consequently, these peptides are promising structurally dynamic biomaterials for minimally invasive therapeutic delivery to the heart after MI. Nevertheless, no further experiments have been conducted using appropriate candidate drugs to demonstrate in vivo efficacies of these enzyme-responsive delivery systems in MI models. Most recently, Dai and Zhao et al. developed a GSH-modified collagen hydrogel (Gel-GSH) for MMP-2/9-responsive on-demand delivery of bFGF [266]. To this end, bFGF was fused with glutathione-S-transferase (GST) and MMP-2/9 cleavable peptide PLGLAG (TIMP), giving rise to GST-TIMP-bFGF that was loaded into Gel-GSH via a bond between GST and GSH (Fig. 4 ). After intramyocardial injection in rats post MI, substrate peptides were hydrolyzed by MMP-2/9, leading to bFGF release in the pathological microenvironment, thereby promoting vascularization and ameliorating myocardium remodeling. Similarly, MMP-responsive and injectable hydrogel was prepared for sequestration and triggerable release of MMP-2 siRNA for MI treatment in rats [195], which led to significantly improved functions, such as increased ejection fraction, stroke volume, and cardiac output, as compared to controls.
Fig. 3.
Enzyme-responsive nanoparticles for targeted accumulation and prolonged retention in heart tissue after myocardial infarction (MI). (A) Diagram of a dye-labeled brush peptide-polymer amphiphile (PPA) containing an MMP-2 and MMP-9 specific recognition sequence, shown underlined. PPAs self-assemble into nanoparticles through hydrophobic-hydrophilic interactions when dialyzed into aqueous buffer. (B) Schematic of nanoparticles freely circulating in the bloodstream upon systemic delivery. Nanoparticles enter the infarct tissue through the leaky acute MI vasculature, and up-regulated MMPs within the infarct induce the formation of an aggregate-like scaffold. (C) Responsive nanoparticles are monodisperse micelles with diameters of 15–20 nm. (D) Formation of an aggregate-like scaffold upon activation of responsive nanoparticles. (E) A corresponding image of nanoparticle solutions following activation. (F-H) Retention of responsive nanoparticles upon localized delivery. Particles were injected intramyocardially at day 7 post-MI and assessed at day 6 postinjection. (F) H&E image displays the infarct area. (G) The neighboring fluorescent section. Particles are shown in red and myocardium is shown in green. (H) A selected region from (G) (white outline) was magnified to highlight particle aggregation. Scale bars in (F-H), 100 μm. Reproduced with permission [288]. Copyright 2015, Wiley-VCH.
Fig. 4.
Schematic representations of MMP-responsive hydrogel preparation and the process of drug release in the wound bed of MI model rats. (A) Preparation of GST-TIMP-bFGF via a recombinant protein expression method. (B) GSH was loaded into the hydrogel by a chemical crosslinking process to obtain Gel-GSH. (C) GST-TIMP-bFGF was mixed with Gel-GSH, and the two components were linked with a bond between GST and GSH. (D) Intramyocardial injection of the mixed hydrogel to the wound of a rat after MI. (E) In the wound microenvironment, substrate peptides were degraded by MMP-2/9, and specific targeting peptides were released, achieving the dual functions of angiogenesis and MMP inhibition. Reproduced with permission [266]. Copyright 2019, Wiley-VCH.
4.1.2. Acute lung injury
Acute inflammation is crucially related to many life-threatening diseases in the respiratory system, such as acute lung injury (ALI) and acute pulmonary infection [290]. ALI is generally resulted from direct and indirect injury to the lungs, showing the most severe manifestation of acute respiratory distress syndrome, which affects approximately 1 million people worldwide annually [291]. Acute pulmonary infection caused by coronaviruses can lead to much more serious global health threats, like novel coronavirus disease 2019 (COVID-2019), severe acute respiratory syndrome, and Middle Eastern respiratory syndrome [292]. Over the past decades, extensive studies have demonstrated the effectiveness and advantages of advanced drug delivery systems for the treatment of pulmonary diseases [[293], [294], [295], [296]].
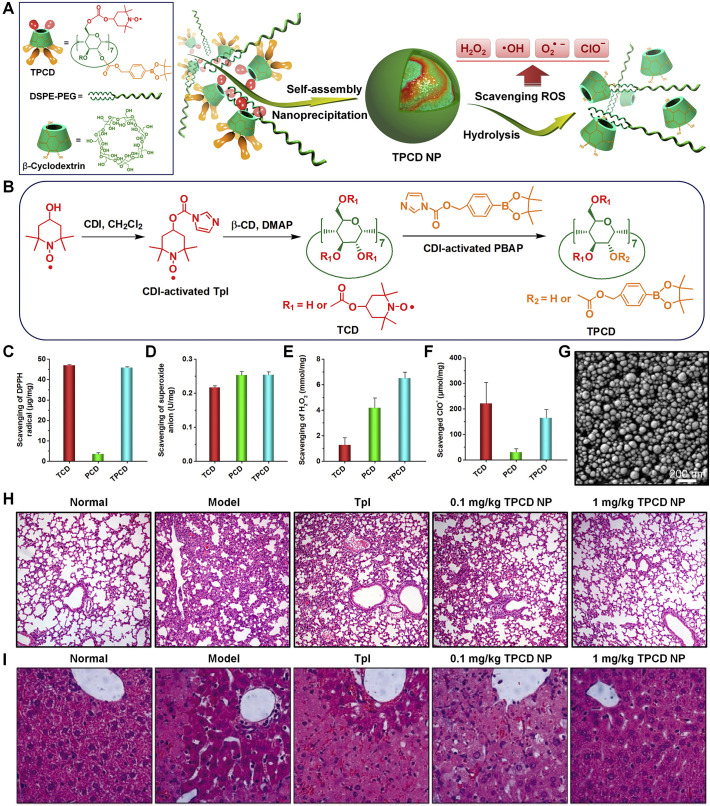
As well-documented, oxidative stress, resulting from overproduced ROS, is intimately associated with progression of ALI [297]. Therefore ROS can function as either therapeutic targets or biological stimuli for ALI therapy. Our group recently designed and developed a ROS-responsive and ROS-eliminating material (defined as TPCD) by simultaneously conjugating Tempol (Tpl) and PBAP onto β-CD (Fig. 5A-B) [298]. For synthesized TPCD bearing 2 Tpl and 5 PBAP units, it can be hydrolyzed into water-soluble products in the presence of H2O2, a typical ROS. TPCD is able to effectively scavenge superoxide anion, H2O2, radical, and hypochlorite (Fig. 5C-F), thereby serving as a broad-spectrum ROS-eliminating material. Using a nanoprecipitation/self-assembly method in the existence of lecithin and 1,2-distearoyl-snglycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000, TPCD was produced into nanoparticles with the mean diameter of 109 ± 2 nm and ζ-potential of -16 ± 0.1 mV. TPCD nanoparticles displayed desirable H2O2-responsive hydrolysis performance (Fig. 5G). By efficiently scavenging different types of ROS, TPCD nanoparticles protected RAW264.7 macrophages from H2O2-induced apoptosis. After i.v. injection, TPCD nanoparticles could accumulate in injured lungs in mice with LPS-induced ALI by passive targeting. Correspondingly, i.v. treatment with TPCD nanoparticles efficaciously mitigated symptoms relevant to ALI, as implicated by the significantly decreased lung wet/dry weight ratio, notably reduced proinflammatory cytokines (TNF-α and IL-1β) and oxidative mediators (H2O2 and MPO) in bronchoalveolar lavage fluid, remarkably decreased infiltration of neutrophils in pulmonary tissues, and improved lung tissue structure (Fig. 5H). These findings suggested that TPCD nanoparticles are promising for the treatment of pulmonary diseases related to acute inflammation and oxidative stress.
Fig. 5.
Design and preparation of broad-spectrum ROS-scavenging nanoparticles for treatment of acute lung injury (ALI). (A) Schematic illustration of engineering of a broad-spectrum ROS-scavenging material and corresponding nanoparticles. (B) The synthetic route of β-CD conjugated with Tempol (Tpl) and PBAP units (TPCD). (C–F) Comparison of scavenging capabilities of different materials for radical, superoxide anion, H2O2, and hypochlorite. (G) SEM image of TPCD NP prepared by nanoprecipitation using methanol as the solvent. (H-I) H&E-stained pathological sections of lung tissues (H) and liver tissues (I) resected from mice subjected to different treatments. Reproduced with permission [298]. Copyright 2018, Wiley-VCH.
Considering acidosis and increased expression of lipase and alkaline phosphatase in bacterial-induced ALI [299], [300], Wang et al. developed pH-responsive nanoparticles for target therapy of ALI [301,302]. To this end, infectious microenvironment-targeting and bioresponsive nanoparticles were assembled using a pH and enzymes multiple-responsive amphiphilic block copolymer consisting of biotinylated PEG-b-poly(β-amino ester)-b-PEG grafted with PEGylated lipid, which were subjected to surface modification with intercellular adhesion molecule-1 (ICAM-1) antibody [302]. The assembled nanoparticles of 120 nm in diameter were loaded with an antibiotic and an anti-inflammatory agent, giving rise to a multifunctional nanotherapy that can target infectious microenvironments and release drug molecules upon triggering by local infectious cues, as demonstrated in an acute lung bacterial infection model in mice. In vivo therapeutic studies in a sepsis mouse model revealed that i.v. treatment with the bioresponsive nanotherapy simultaneously eliminated bacteria and alleviated the host inflammatory response, as indicated by the significantly decreased levels of infiltrating leukocytes and proinflammatory cytokines (such as TNF-α, IL-1β, and IL-6) in infected lungs, and therefore it can be further developed as a mechanism-based nanotherapy for infectious diseases. In a most recent study, the same group prepared a nanotherapy assembled by an amphiphilic copolymer of poly(β-amino esters) and PEG, loading an anti-inflammatory agent TPCA-1 in the pH-sensitive core [301]. Additional decoration using anti-ICAM-1 antibody was carried out for this TPCA-1 nanotherapy to enhance lung targeting capability. In mice with ALI, i.v. injection of the TPCA-1-loaded targeting nanotherapy (~100 nm) demonstrated efficient targeting and acid-triggered drug release in inflamed lungs, thereby inhibiting lung inflammation and injury.
4.1.3. Acute kidney injury
Acute kidney injury (AKI), the most common cause of kidney dysfunction, remains a critical health issue with high morbidity and mortality worldwide [303]. AKI is mainly caused by sepsis, ischemia-reperfusion, and/or nephrotoxicity, and it is characterized by rapid progression of inflammation and oxidative stress. Different drug delivery systems have recently been explored for AKI therapy in murine models, including drug conjugates [304], micelles [305], nanoparticles [306], microspheres [307], and injectable hydrogels [308]. Of note, Ji and Du et al. designed pH-responsive and AKI-kidney targeting nanopolyplexes by electrostatic forces between anionic hyaluronic acid and cationic chitosan for effective delivery of SS-31 (a mitochondrial targeting peptide with potent antioxidant activity) to treat AKI. The developed SS-31 nanoparticles showed the mean diameter of 53 ± 0.17 nm, with ζ-potential of -19.6 ± 0.7 mV. In low pH environments, SS-31-containing nanoparticles will be broken due to dissociation of polyelectrolyte complexes, thereby allowing SS-31 release and subsequent mitochondrial targeting to achieve therapeutic effects [309]. While pH-sensitivity of this SS-31 nanotherapy was demonstrated by in vitro experiments, in vivo fluorescence imaging confirmed desirable accumulation of Cy5-labeled nanotherapy in kidneys of AKI mice after i.v. administration. Consistently, the pH-sensitive SS-31 nanotherapy effectively inhibited oxidative stress, protected mitochondrial structure, attenuated inflammatory response, and reduced apoptosis and necrosis of tubular cells after i.v. injection in AKI mice.
In view of overproduced ROS at the AKI site, the newly engineered ROS-responsive and broad-spectrum ROS-scavenging TPCD nanoparticles were employed for the treatment of acute renal injury [298]. Preliminary studies in mice with acetaminophen-induced AKI showed desirable efficacy after i.v. administration, as indicated by notably attenuated hydronephrosis and significantly decreased serum levels of creatinine (CREA) and urea (UREA). In a most recent study, a ROS-responsive drug delivery system was developed using an amphiphilic copolymer of PEG and PLGA covalently linked via the thioketal bond [310], in which mitochondrial targeting ceria nanoparticles and an anti-inflammatory drug atorvastatin were simultaneously loaded, giving rise to a nanotherapy with the average diameter of 25.5 ± 8.58 nm and ζ-potential of -0.551 ± 0.114 mV. After i.v. injection, fluorescence imaging demonstrated accumulation of the engineered nanotherapy in injured kidneys of mice with sepsis-induced AKI. In vivo studies revealed that this ROS-responsive and ROS-scavenging nanotherapy significantly decreased the levels of malondialdehyde (MDA), TNF-α, and IL-6, thereby notably reducing oxidative stress and inflammatory response. Also, i.v. treatment with this nanotherapy effectively protected the mitochondrial structure and inhibited tubular cell apoptosis and tubular necrosis in AKI mice. According to other pathologically increased biomolecules (such as MMP-2), enzyme-responsive polymeric prodrugs were synthesized for targeted therapy of AKI [311]. For different bioresponsive nanotherapies examined thus far, whereas these studies demonstrated their effectiveness in AKI treatment, the kidney targeting efficiency need to be considerably improved in future studies by delicate modulation on the particle size, shape, surface charge, and density of targeting moieties, to avoid their undesirable distribution in other non-target organs (such as the liver). For example, nanoparticles of ~75 ± 25 nm have demonstrated efficient targeting to the mesangium of the kidney [312]. Also, metabolism, renal excretion, and other safety issues should be elucidated for most nanocarriers by in-depth evaluations.
4.1.4. Acute liver injury
Similar to the above mentioned diseases associated with acute inflammation, the pathophysiological microenvironment in the liver has been deployed to treat acute liver injury, a main cause of diverse liver diseases, such as hepatic fibrosis, cirrhosis, and hepatocellular carcinoma [313]. In this context, pH-sensitive nanoparticles were formulated using ketalized maltodextrin (KMD) to achieve triggerable therapeutic delivery to the injured hepatic tissue resulting from inflammation [314]. KMD was synthesized by covalent conjugation of acid-cleavable hydrophobic groups onto maltodextrin through carbonate bonds. Using silymarin as a candidate drug, in vitro pH-dependent release profiles were found for KMD nanoparticles. Cell culture experiments revealed desirable antioxidant and anti-inflammatory activity of silymarin-loaded KMD nanoparticles. Correspondingly, this pH-responsive nanotherapy effectively suppressed acute inflammation and protected liver from acetaminophen-induced hepatic injury. Also, acid-responsive nanoparticles based on acetalated dextran were simultaneously loaded with a therapeutic compound and gold nanoparticles for targeted therapy and computer tomography imaging of acute liver failure in mice [315].
In addition, ROS-responsive delivery systems were investigated for the treatment of acute liver injury. A study by Lee and coworkers showed that H2O2-responsive polymeric prodrug PVAX microparticles containing manganese-porphyrin (a mimic of superoxide dismutase) effectively protected the liver from acetaminophen-induced injury in mice [228]. Zhou and Dong et al. prepared ROS-responsive nanoparticles by self-assembly of PEG-b-PPS, which were used for delivery of melatonin, an antioxidant molecule [316]. In vitro release of melatonin from PEG-b-PPS nanoparticles was considerably accelerated by H2O2. In a mouse model of sepsis-induced acute liver injury, the formulated ROS-responsive nanotherapy showed significantly potentiated efficacy compared to free drug after intraperitoneal injection, with respect to attenuating oxidative stress and inflammatory response as well as inhibiting liver injury. Our recent finding revealed a higher accumulation of i.v. delivered nanoparticles in the injured liver, compared to that of the healthy liver in normal mice [298]. Further, treatment of mice with acetaminophen-induced acute liver injury by ROS-responsive and ROS-eliminating TPCD nanoparticles at 1 mg/kg significantly reduced the levels of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST), in comparison to the model group treated with saline. Moreover, TPCD nanoparticles effectively suppressed neutrophil infiltration, significantly inhibited the expression of proinflammatory cytokines (TNF-α and IL-1β), and notably reduced ROS levels in hepatic tissues. Additionally, the histological abnormalities of the liver were remarkably improved after TPCD NP therapy (Fig. 5I). Given the critical role of proinflammatory cytokines in the development of acute liver failure, Yin’s group developed ROS-responsive and macrophage-targeting polyplexes to site-specifically deliver TNF-α siRNA (siTNF-α) to mannose receptor-overexpressed hepatic macrophages [317]. The polyplexes were prepared using low molecular weight PEI cross-linked with a ROS-responsive diselenide bond, which were further coated with carboxylated mannan. After i.v. administration, thus engineered siTNF-α nanotherapy showed effective active targeting to mannose receptor-overexpressed macrophages in the liver and significantly attenuated hepatic inflammation in mice with LPS/D-galactosamine-induced acute liver failure, by notably down-regulating the levels of hepatic and systemic TNF-α.
Collectively, the available findings strongly suggested that ROS-responsive drug delivery systems are particularly promising for the treatment of liver diseases related to acute injury. Notably, liver targeting can be easily achieved by most nanoparticles due to the passive tissue accumulation mediated by the mononuclear phagocyte system. Nevertheless, precision targeting of specific cell types in the liver is still challenging to realize more significant therapeutic benefits and minimize side effects.
4.2. Treatment of chronic inflammatory diseases
Chronic inflammatory diseases, such as IBD, RA, and atherosclerosis, are major causes of death worldwide among older adults. Long-term treatment with traditional anti-inflammatory agents is generally associated with notable side effects. Increasing evidence has demonstrated multiple advantages of bioresponsive drug delivery systems in the management of chronic inflammatory diseases. This section highlights applications of bioresponsive delivery strategies for the treatment of typical diseases related to chronic inflammation.
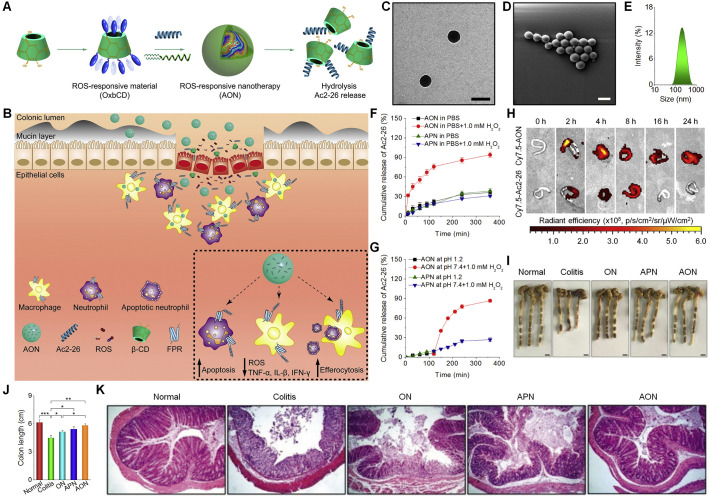
4.2.1. Inflammatory bowel disease (IBD)
IBD, including ulcerative colitis and Crohn’s disease, is a generic term for a group of chronic relapsing gastrointestinal diseases. It has been extensively demonstrated that overproduced ROS play a critical role in the pathogenesis of IBD [318], and therefore there is an increasing interest in developing ROS-responsive drug delivery systems for IBD therapy. Using a ROS-responsive polymer PPADT, Murthy et al. developed nanoparticles loaded with siRNA against TNF-α (termed as TKNs, with diameter of ~600 nm) [231]. TKNs released siRNA in response to abnormally high levels of ROS at the site of intestinal inflammation. Oral administration of TKNs significantly reduced the TNF-α mRNA level in the colon of mice with dextran sodium sulfate (DSS)-induced ulcerative colitis, thereby effectively protecting mice from colitis. Of note, TKNs showed significantly higher in vivo activity than TNF-α siRNA-containing nanoparticles derived from PLGA or a cationic lipid 1,2-dioleoyl-3-trimethylammonium-propane (DOTAP), with respect to inhibiting TNF-α mRNA in inflamed intestinal tissues. Our group developed a ROS-responsive nanotherapy mimicking superoxide dismutase/catalase [319]. This nanotherapy is consisted of an oxidation-responsive β-CD material (OxbCD) and a free radical scavenger Tpl. In addition to serving as ROS-responsive nanovehicles, OxbCD nanoparticles themselves can efficiently eliminate H2O2. Accordingly, Tpl-loaded OxbCD nanoparticles enabled scavenging of multiple major types of ROS, such as superoxide anion, hydroxyl radical, and H2O2. Interestingly, Tpl/OxbCD nanoparticles were stable at pH 1.2, while responsive release profiles were observed under colonic pH conditions, in a ROS-responsive manner. After oral administration, OxbCD nanoparticles showed considerable accumulation in the inflamed colons of mice bearing DSS-induced colitis, with significantly higher targeting efficiency than that of control PLGA nanoparticles. In mice with DSS or 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced acute colitis as well as DSS-induced chronic colitis, oral delivery of Tpl/OxbCD nanoparticles notably alleviated symptoms relevant to colitis, as exemplified by significantly decreased levels of CD98-positive intestinal epithelial cells and typical inflammatory factors, including TNF-α, IL-1β, and interferon (IFN)-γ. In all these mouse models, Tpl/OxbCD nanoparticles were more effective than Tpl/PLGA nanoparticles. In a separate study, we found that this type of H2O2-responsive and ROS-eliminating nanoparticles can potentiate in vivo antitumor activity of a ROS-responsive camptothecin-11 nanotherapy in mice with chronic colitis-associated colon cancer, by normalizing the proinflammatory microenvironment [320]. More recently, our studies showed that OxbCD nanoparticles are able to package a proresolving annexin A1-mimetic peptide Ac2-26, offering a ROS-responsive peptide nanotherapy (defined as AON) (Fig. 6A-E) [321]. AON effectively protected Ac2-26 from degradation in the gastrointestinal environment (Fig. 6F-G). Moreover, site-specific delivery and triggerable release of Ac2-26 at the inflamed colon were achieved after oral delivery of AON (Fig. 6H). In mice bearing DSS-induced acute or chronic colitis, oral treatment with AON significantly decreased the levels of proinflammatory cytokines (TNF-α, IL-1β, and IFN-γ) and oxidative stress-related molecular mediators (MDA, H2O2, and MPO) in the colonic tissues, notably reduced disease activity index, and considerably improved colonic microstructure, thereby achieving desirable therapeutic outcomes (Fig. 6I-K). In vivo efficacy of AON was further confirmed in IL-10-deficient mice that can spontaneously develop colitis. In all cases, AON showed more efficacious effects than a control PLGA nanotherapy. In addition to notably attenuated local inflammation and oxidative stress, AON treatment promoted intestinal mucosal wound healing, reshaped the gut microbiota, and enhanced short-chain fatty acid production. Importantly, AON displayed good safety profile after oral delivery at a high dose of 5 g/kg in mice. Consequently, AON is a promising ROS-responsive nanotherapy for precision therapy of IBD and other inflammatory diseases.
Fig. 6.
A proresolving peptide nanotherapy for site-specific treatment of inflammatory bowel disease. (A-B) Schematic illustration of engineering of a ROS-responsive peptide nanotherapy (AON) (A) and targeted treatment of colitis (B). (C-E) TEM (C) and SEM (D) images as well as size distribution (E) of AON. Scale bars, 200 nm. (F) In vitro release profiles of Ac2-26 nanotherapies AON and APN in 0.01 M PBS at pH 7.4 with or without 1.0 mM H2O2. (G) Ac2-26 release in buffers simulating the gastrointestinal pH conditions. (H) Selective accumulation of AON in the inflamed colons of mice with acute colitis. (I-J) Representative digital photos (I) and quantified lengths (J) of colonic tissues isolated from mice after 7 days of treatment. Scale bars, 5 mm. (K) H&E-stained histological sections of colonic tissues. AON, Ac2-26-loaded OxbCD nanoparticles; APN, Ac2-26-loaded PLGA nanoparticles; ON, the blank OxbCD nanoparticles. *p < 0.05, **p < 0.01, ***p < 0.001. Reproduced with permission [321]. Copyright 2019, Wiley-VCH.
Therapeutic advantages of bioresponsive delivery systems in IBD therapy have also been demonstrated by other studies. Siepmann et al. fabricated microbiota-sensitive 5-aminosalicylic acid pellets [322]. The resulting pellets notably mitigated manifestations relevant to TNBS-induced colitis in rats and significantly decreased inflammatory markers. Also, multi-responsive approaches were examined to deliver therapeutics for IBD management, attempting to overcome the limitations of single signal-dependent strategies. Xiao’s group designed curcumin-loaded nanoparticles derived from natural silk fibroin with surface decoration using chondroitin sulfate [323]. The generated nanoparticles displayed good bioresponsiveness upon triggering by acidity, GSH, and ROS, in activated macrophages. After oral administration or i.v. injection, this nanotherapy notably mitigated the symptoms of colitis, maintained the gut microbiota, and enhanced the survival rate of colitis mice. In addition, mannosylated bioreducible nanoparticles, pH/enzyme dual-responsive nanogels, and pH/ROS-responsive nano-in-micro composites were investigated as delivery vehicles of different drugs for targeted treatment of IBD [272,324,325].
4.2.2. Inflammatory vascular diseases
Sustained vascular inflammation has been implicated in many CVDs that remain the leading cause of global morbidity and mortality. Excessive ROS production has been found in injured arteries of many vascular diseases, such as restenosis, atherosclerosis, and aneurysm. According to local acidosis and elevated levels of ROS at the injured sites of arteries, our group developed pH- or ROS-responsive nanotherapies of 186.5 ± 2 or 253.5 ± 3.9 nm in diameter, by loading rapamycin (RAP) into nanoparticles based on acetalated β-CD or PBAP-conjugated β-CD, respectively (Fig. 7A) [111,182]. The obtained RAP nanotherapies displayed either pH- or ROS-triggerable drug release behaviors. Compared to free RAP and a nonresponsive PLGA nanotherapy, both responsive nanotherapies exhibited significantly higher anti-migration and antiproliferative effects in rat vascular smooth muscle cells (VSMCs). After i.v. injection, the related nanoparticles accumulated at the injured carotid arteries of rats with balloon angioplasty injury (Fig. 7B), which is a typical animal model of arterial restenosis. Moreover, i.v. treatment with pH- or ROS-responsive nanotherapy more significantly inhibited neointimal hyperplasia, compared to free RAP and a PLGA nanotherapy (Fig. 7C). In a most recent study, we demonstrated nanoparticles with tunable dual-sensitivity can be easily prepared by combining pH- or ROS-responsive β-CD materials [88]. In rat VSMCs, thus engineered pH/ROS dual-responsive RAP nanotherapies showed potentiated anti-migration, anti-proliferation, and cell cycle regulation capabilities, when compared with corresponding single-responsive RAP nanotherapies. Also, in vivo studies revealed enhanced therapeutic benefits of the pH/ROS dual-responsive nanotherapy, in comparison to the pH- or ROS-responsive RAP nanotherapy. Further, surface decoration of the dual-responsive nanotherapy via a type IV collagen-targeting peptide (KLWVLPKGGGC) notably potentiated its in vivo anti-restenosis efficacy, resulting from the increased accumulation in injured carotid arteries by active targeting.
Fig. 7.
Inflammation-responsive nanotherapies for targeted treatment of atherosclerosis and restenosis. (A) Design of inflammation-triggerable nanoparticles. (B) Representative ex vivo fluorescence images showing retention of Cy7.5/Ac-bCD NP at the injured site of the carotid artery in rats after intravenous injection. (C) H&E stained histological sections of carotid arteries isolated from rats subjected to various treatments. Scale bars, 200 μm (the upper panel), 50 μm (the lower panel). Reproduced with permission [111]. Copyright 2016, Elsevier. (D) Representative ex vivo fluorescence images illustrating accumulation of Cy5 fluorescent signals in the aorta of ApoE-/- mice at 24 h after intraperitoneal injection of saline, free Cy5, or Cy5/Ac-bCD NP. (E) Representative photographs of en face ORO-stained aortas from each group. (F) Quantitative analysis of the lesion area. **p < 0.01, ***p < 0.001. Reproduced with permission [182]. Copyright 2017, Elsevier. RAP, rapamycin; PLGA, poly(lactide-co-glycolide); Ac-bCD, acetalated β-cyclodextrin; Ox-bCD, oxidation-responsive β-cyclodextrin.
Atherosclerosis, a chronic inflammatory disorder characterized by the formation of atheromatous plaques in the intima of arteries, may cause severe diseases such as MI and stroke. In a previous study, we found that the above mentioned pH- or ROS-responsive RAP nanotherapy derived from chemically functionalized β-CD materials remarkably decreased the macrophage count and MMP-9 level in aortas, and therefore delayed progression of atherosclerosis and significantly enhanced stability of plaques in apolipoprotein E-deficient (ApoE-/-) mice [182], compared to a non-responsive PLGA nanotherapy (Fig. 7D-F). This enhanced anti-atherosclerotic activity of RAP by responsive nanocarriers should be mainly due to triggerable and effective release of drug molecules in atheromatous plaques, following targeting by the lymphatic translocation mediated via inflammatory infiltration of neutrophils and monocytes/macrophages that was demonstrated by ex vivo imaging in combination with flow cytometric analysis. Moreover, these responsive nanovehicles displayed good safety after long-term administration in mice. In a following study, we explored anti-atherosclerotic activity of our previously developed ROS-responsive and broad-spectrum ROS-eliminating TPCD nanoparticles [326], in view of deleterious effects of overproduced ROS on atherosclerosis. In vitro experiments revealed that TPCD nanoparticles notably suppressed foam cell formation in both macrophages and VSMCs. After i.v. administration, TPCD nanoparticles could accumulate in atherosclerotic lesions of ApoE-/- mice, mainly resulting from passive targeting via the permeable endothelium and translocation by inflammatory cells. Further in vivo therapeutic studies indicated that i.v. treatment with TPCD nanoparticles attenuated the development of atherosclerosis and stabilized plaques, as implicated by thicker fibrous caps, smaller necrotic cores, and lower macrophage counts, compared to control antioxidant drugs against atherosclerosis. The desirable in vivo efficacies of TPCD nanoparticles were largely contributed by effectively decreasing the levels of TNF-α, IL-1β, and H2O2 in the serum and aorta, which in turn led to attenuated systemic/local inflammation and oxidative stress. Since TPCD nanoparticles showed good biocompatibility in different animal models [298,326], they can be further intensively studied as a safe and effective nanotherapy for both acute and chronic inflammatory diseases. Also, ROS-responsive nanoparticles containing atorvastatin were functionalized with macrophage cell membrane [327], leading to nanotherapies with further increased in vivo anti-atherosclerotic efficacies. Most recently, we engineered a pH-responsive anti-microRNA-33 nanotherapy (defined as AAM NP), by packaging an antisense oligonucleotide against microRNA-33 (anti-miR33) into nanoparticles derived from an acetalated α-CD material and PEI (Mw = 1800 Da) for targeted therapy of atherosclerosis [328], considering lesional acidosis. AAM NP was further decorated with a peptide ligand cRGDfk targeting integrin to develop an active targeting nanotherapy RAAM NP. After i.v. delivery, both nanotherapies can be accumulated in the plaques and efficiently internalized by endothelial cells and macrophages, with RAAM NP showing considerably enhanced targeting capability. Treatment with both anti-miR33 nanotherapies significantly attenuated atherosclerosis in ApoE-/- mice and notably reduced vulnerable plaques by promoting reverse cholesterol transport and regulating adaptive immunity via modulating macrophage polarization and regulatory T cell differentiation. In other studies, redox-sensitive gene delivery nanoplatforms were examined for reducing atherosclerotic inflammation [273], by targeted delivery of eNOS plasmid to endothelial cells. These studies unambiguously demonstrated that pH-, redox-, or ROS-responsive nanoparticles are highly effective delivery vehicles for precision therapy of atherosclerosis by site-specific release of different therapeutic agents.
In an attempt to develop targeting therapies for abdominal aortic aneurysm (AAA), a leading cause of mortality and morbidity in the elderly, we recently examined the effectiveness of ROS-responsive RAP nanotherapies (Fig. 8 ) [329]. To this end, nonresponsive or ROS-responsive RAP nanotherapies were prepared by a nanoprecipitation/self-assembly method, using PLGA or OxbCD as carrier materials, respectively. While both nanotherapies remarkably inhibited calcium and inorganic phosphorus-induced calcification and cell death as well as decreased H2O2-induced ROS generation and apoptosis in VSMCs, much better results were observed for the ROS-responsive nanotherapy (OR NP). After i.v. administration in rats with CaCl2-induced AAAs, OxbCD nanoparticles displayed significantly higher accumulation in aneurysmal aortas than PLGA nanoparticles. By decreasing expressions of MMPs (MMP-2 and MMP-9) and proinflammatory cytokines (TNF-α, IL-1β, and IFN-γ), inhibiting aortic calcification and macrophage infiltration, and lowering ROS production, OR NP afforded more desirable in vivo efficacies than PLGA-based nanotherapy. Subsequently, OR NP was decorated with a cyclopeptide ligand cRGDfK that can target neovessels via binding with αvβ3 integrin (Fig. 8A). Both in vitro and in vivo studies revealed notably enhanced anti-aneurysmal efficacy of the active targeting and ROS-responsive nanotherapy ROR NP (Fig. 8B-E). Finally, functionalization with macrophage cell membrane was conducted to further increase targeting capability of ROR NP. The engineered biomimetic and bioresponsive nanotherapy more significantly prevented the development of AAAs, as indicated by the notably attenuated expansion of the aortic diameter as well as remarkably decreased calcification and elastin degradation, resulting from improved aneurysmal targeting after macrophage membrane cloaking.
Fig. 8.
A targeting nanotherapy for abdominal aortic aneurysms. (A) Schematic (left), TEM image (middle), and size distribution (right) of ROR NP. (B) Ex vivo images (left) and histograms (right) showing accumulated ROCy7.5 NP or OCy7.5 NP in aneurysmal aortas. **p < 0.01, ***p < 0.001. (C) Immunofluorescence analysis of localized ROCy5 NP in aneurysms. Scale bars, 20 μm. (D) H&E, Alizarin Red, or VVG stained histological sections of aneurysmal aortas. (E) Immunohistochemistry analyses of aneurysmal aortas. OR NP, RAP-containing OxbCD nanotherapy; ROR NP, a ROS-responsive, cRGDfK targeting RAP nanotherapy; OCy5 NP, Cy5-labeled OxbCD nanoparticles; ROCy5 NP, Cy5-labeled OxbCD nanoparticles with cRGDfK decoration; OCy7.5 NP, Cy7.5-labeled OxbCD nanoparticles; ROCy7.5 NP, Cy7.5-labeled OxbCD nanoparticles decorated with cRGDfK. Reproduced with permission [329]. Copyright 2018, Elsevier.
In other studies, ROS-responsive as well as pH- and/or thrombin-responsive nanotherapies were examined for targeted treatment of thrombus formation [330], peripheral arterial disease [226], and ischemic stroke [[331], [332], [333], [334]], offering promising therapeutic effects in different animal models. Collectively, studies by others and our group demonstrated that bioresponsive nanotherapies possess great potential for precision therapy of different inflammatory vascular diseases.
4.2.3. Rheumatoid arthritis (RA)
RA is an autoimmune and chronic inflammatory disease in the joints that may cause cartilage damage, bone erosion, and loss of joint function. To overcome significant side effects of small molecule anti-inflammatory agents, Wang’s group developed a pH-sensitive drug delivery system by conjugating dexamethasone (Dex) onto a N-(2-hydroxypropyl)methacrylamide copolymer (P-Dex) via an acid-labile hydrazone bond, which can release free dexamethasone at acidic pH [189]. This polymeric prodrug showed specific accumulation in inflamed joints of rats with adjuvant-induced arthritis (AIA). Compared to free Dex, i.v. treatment with P-Dex afforded more effective and longer-lasting anti-inflammatory effects, with notably reduced side effects. Therapeutic benefits of P-Dex were also demonstrated in a mouse model of collagen-induced arthritis (CIA) and mice with particle-induced inflammation in joints [190,335]. Based on the similar principle, Sun et al. prepared pH-sensitive micelles assembled by an amphiphilic copolymer derived from a PEG derivative with conjugated prednisolone via the hydrazone linkage (Fig. 9A) [188]. After i.v. injection in CIA mice, prodrug micelles showed significantly higher accumulation in the joints than that of free drug. Correspondingly, pH-sensitive micelles exhibited notably enhanced efficacy and simultaneously reduced myelo- and hepatotoxicity, as compared to free prednisolone. In a most recent study, Chen et al. developed macrophage-targeting pH-sensitive nanoparticles for co-delivery of two anti-inflammatory agents (Fig. 9B) [192]. All-trans-retinal was conjugated onto dextran via the hydrazone bond, followed by conjugation of a targeting unit of galactose. The assembled nanoparticles were physically loaded with triptolide, a potent anti-inflammatory drug isolated from Chinese herb. Both drugs in the assembled nanoparticles displayed acid-triggerable release profiles. In CIA mice, the nanotherapy with galactose decoration showed remarkably higher accumulation in the inflamed joints after i.v. injection, due to combined passive and active targeting effects. Further in vivo studies revealed that encapsulation into pH-responsive and targeting nanoparticles notably potentiated anti-arthritic activity of triptolide, while drug toxicity was considerably reduced. Likewise, folate receptor (FA)-targeting, acid-labile nanoparticles derived from PCADK/lipid/PLGA-PEG or PEGylated amphiphilic hyaluronan nanoparticles with calcium phosphate as a pH-responsive mineral, both containing methotrexate, were investigated for targeted RA treatment in AIA or CIA mice [336,337], respectively, giving rise to anticipated therapeutic benefits.
Fig. 9.
pH-Responsive nanotherapies for targeted treatment of rheumatoid arthritis (RA). (A) Schematic of assembly of pH-sensitive polymeric prodrug micelles for RA. Reproduced with permission [188]. Copyright 2018, Elsevier. (B) Schematic illustration of GDR-improved anti-inflammatory effect of TPT in a mouse model of CIA. GDR improves anti-inflammatory effect of TPT through several mechanisms: (1) GDR itself had anti-inflammatory property via RAR signaling; (2) GDR-mediated macrophage-targeted TPT delivery; (3) pH-sensitive nanoparticle GDR allowed the release of TPT at the intracellular regions of inflamed site, thereby enhancing anti-arthritic effect and improving safety profile of TPT. GDR, all-trans-retinal-dextran conjugate grafted with galactose; RAR, retinoic acid receptor; TPT, triptolide. Reproduced with permission [192]. Copyright 2020, Elsevier.
In addition to acid-labile nanotherapies, ROS-responsive delivery systems were examined for the RA treatment. In view of high ROS levels in diseased tissues of RA, Liu et al. fabricated ROS-responsive micelles of ~153 nm based on a vitamin E-conjugated amphiphilic copolymer derived from PLGA and PEG that were linked via a ROS-responsive diselenide bond [338], in which a candidate anti-RA drug berberine was loaded. In vitro studies based on liquid chromatography-mass spectrometry showed that ROS-responsive micelles increased both cellular and mitochondrial concentrations of berberine in RA fibroblasts nearly ten times higher than those of free berberine. After intraperitoneal injection in AIA rats, the ROS-responsive micellar nanotherapy significantly inhibited the levels of inflammatory cytokines (IL-1 and IL-6) and reduced the bone destruction, with much better efficacy than berberine. In other cases, ROS-responsive and FA-targeting liposomes and nanoparticles were investigated for targeted RA treatment [339,340], and both systems notably reinforced in vivo efficacies of loaded drugs in CIA mice.
Also, enzyme-responsive drug delivery systems have been developed for arthritis therapy. As a representative example, arthritis flare-responsive hydrogel was self-assembled by triglycerol monostearate (TG-18) to encapsulate an anti-arthritic drug triamcinolone acetonide [341]. This injectable hydrogel showed on-demand drug release profiles upon triggering by MMP-3 or MMP-9. After intra-articular injection, TG-18 hydrogel underwent disassembly correlating with arthritis severity in a mouse model of inflammatory arthritis. Moreover, local drug delivery with TG-18 hydrogel significantly reduced paw thickness and total clinical scores in mice with arthritis, while free drug did not show notable therapeutic effects, mainly due to rapid clearance from the injection site. Accordingly, bioresponsive systems can be further developed as a promising on-demand delivery strategy to potentiate anti-arthritic activity of different drugs.
4.3. Other inflammation-associated diseases
A growing body of evidence suggests that chronic inflammation and oxidative stress also play an important role in the pathophysiology of neurodegenerative diseases, such as Alzheimer’s disease (AD) and Parkinson’s disease. However, many anti-inflammatory or antioxidant therapies cannot effectively inhibit brain injury due to their limited ability to cross the blood-brain barrier (BBB) and target the disease site. Bioresponsive nanotherapies have been emerging as an effective strategy to overcome the BBB and afford the advantage of site-specific release in the impaired brain. In an elegant study, Jiang’s group synthesized a ROS-responsive amphiphilic copolymer by conjugating phenylboronic ester onto the polylysine block of PEG-polylysine, which was then functionalized with a peptide targeting receptor for advanced glycation end-products [342]. The copolymer-assembled ROS-responsive and targeting micelles were packaged with a model drug curcumin (Fig. 10 ). After i.v. injection in APPswe/PSEN1dE9 model mice, the micelles efficiently accumulated in the diseased regions by β-amyloid transportation-mediated pathway, significantly decreased the levels of proinflammatory cytokines TNF-α and IL-1β in the brain, and showed neuroprotection and microglia modulation via the synergistic effects of polymers and payloads, thereby decreasing β-amyloid plaque burdens and enhancing cognitive functions.
Fig. 10.
A ROS-responsive polymeric micelle system for targeted treatment of Alzheimer’s Disease. (A) The mechanism of ROS-induced self-immolative degradation. (B) Illustration of microglia-induced AD microenvironment and mechanisms of APLB/CUR modulation: 1) Ab peptide modified micelles mimic Aβ transportation from peripheral into brain parenchyma; 2) ROS-responsive release of ROS scavenging polymer and Aβ inhibitive curcumin; and 3) Aβ-mimicked micelle targeting to activated microglia and damaged neurons. Reproduced with permission [342]. Copyright 2019, Wiley-VCH.
Chronic inflammation also contributes to common respiratory system diseases, such as asthma, chronic obstructive pulmonary disease, and cystic fibrosis. Both nanoparticles and microparticles are promising delivery vehicles for the treatment of pulmonary diseases [293,295]. Lee et al. explored they previously developed H2O2-responsive and H2O2-scavenging HPOX nanoparticles for the treatment of asthma in a murine model [343]. After intratracheal injection in asthmatic mice induced with ovalbumin, HPOX nanoparticles significantly reduced the infiltration of inflammatory cells (macrophages and neutrophils) and expression of proinflammatory mediators (IL-4 and inducible nitric oxide synthase) as well as decreased airway remodeling. Recently, nanoparticle-embedded microgels responsive to both trypsin and neutrophil elastase were prepared by the thiol-maleimide Michael addition reaction in emulsions, using model polystyrene nanoparticles and enzyme-degradable peptide crosslinkers [344]. After aerosolized delivery, the enzyme-responsive microgels showed desirable pulmonary retention and disease-triggered degradation. Nevertheless, further therapeutic studies in specific disease models are necessary to demonstrate advantages of this novel multi-stage enzyme-responsive microgel.
Collectively, the currently available findings suggest that bioresponsive delivery systems have great potential as novel drug carriers for targeted treatment of neurodegenerative diseases and airway inflammatory diseases, although intensive and in-depth studies are necessary to evaluate their effectiveness, advantages, and benefit-risk ratio in different disease models. In addition, bioresponsive nanotherapies are able to attenuate surgical injury-induced inflammation. As demonstrated by laminectomy models in rats and rabbits [345], the aforementioned TPCD nanoparticles loaded in a poloxamer 407-derived thermosensitive hydrogel displayed notable antioxidative stress and anti-inflammatory activities by significantly reducing the levels of H2O2, MPO, TNF-α, IL-1β, and IL-6, thereby effectively reducing fibrotic tissue formation and preventing postlaminectomy epidural adhesion.
5. Concluding remarks
Over the past decades, considerable progress has been achieved in the development of bioresponsive materials and delivery systems as well as their applications in the treatment of both acute and chronic inflammatory diseases. Particularly, a large number of materials responsive to varied inflammatory microenvironments have been synthesized and evaluated in different cells and/or animal models. By triggerably releasing therapeutic molecules in response to pathologically relevant biochemical signals such as low pH, increased ROS, and overexpressed enzymes, their efficacies can be considerably enhanced, as well demonstrated in preclinical studies based on animal models of acute CVDs, acute liver injury, ALI, AKI, IBD, inflammatory vascular diseases, and RA. Despite these achievements, only a very few of bioresponsive drug delivery systems have been intensively investigated in diverse animal models of inflammatory diseases. To the best of our knowledge, currently no translation studies have been conducted for bioresponsive therapies in anti-inflammatory interventions.
Some crucial challenges should be addressed before further clinical translation studies. First, for rational design of bioresponsive drug delivery systems, the optimal biochemical signal need to be selected according to the specific disease types and diseased sites. The degree of local acidosis varies in different inflammatory diseases. Also, some normal tissues and subcellular organelles exhibit acidic environments, such as the stomach and endolysosomes, which may cause off-target drug release from pH-responsive delivery platforms, thereby impairing in vivo efficacies or generating side effects. Likewise, the ROS generation and the major type of ROS may be varied in different inflammatory diseases or at the different stages of the same disease. As for enzyme-sensitive systems, the corresponding enzymes expressed in other non-target tissues also affect their drug release behaviors. Unfortunately, these issues remain elusive, which complicates and limits the rational design and engineering of bioresponsive delivery systems with more precisely controlled release profiles. Whereas multi-responsive systems afford much more sensitivity and specificity, their development frequently involves more complicated materials synthesis and/or formulation procedures. Second, preparation processes of currently available bioresponsive delivery systems remain to be optimized to achieve desirable quality control, such as well-defined material structures, tailorable biophysicochemical properties, and good reproducibility. Cost-effective mass production is also a key issue to simultaneously ensure clinical benefits and low cost, which is especially important for the management of chronic inflammatory diseases. Third, new preclinical animal models that can fully replicate the pathophysiological microenvironment of human inflammatory diseases need to be established. Thus far, most in vivo targeting and therapeutic studies of bioresponsive delivery systems were performed in murine models of inflammatory diseases. The obtained results cannot be directly extrapolated to humans, due to the intrinsic differences between humans and other species. In this context, targeting capability and/or efficacies of currently developed anti-inflammatory delivery systems should be further examined in multiple animal models to afford more convincible results and screen translational formulations. For bioresponsive anti-inflammatory nanotherapies, their accumulation in off-target organs or tissues need to be taken into consideration. Different strategies, such as surface engineering with targeting moieties or biomimetic approaches, remain to be systemically examined to maximally enhance targeting efficiency of these nanotherapies at inflammatory sites, thereby notably minimizing their nonspecific distribution and reducing side effects. Last but not least, both acute and chronic toxicity evaluations must be carefully conducted for some promising bioresponsive materials or their delivery systems. The perfect bioresponsive delivery systems should maximize drug efficacies and simultaneously reduce or avoid drug-mediated side effects, thereby providing patients with therapeutic benefits outweighing risks.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 81971727 and 81701832), the Program for National Postdoctoral innovative talent (BX20180379), and the Program for Distinguished Young Scholars of TMMU.
References
- 1.Medzhitov R. Inflammation 2010: New adventures of an old flame. Cell. 2010;140:771–776. doi: 10.1016/j.cell.2010.03.006. [DOI] [PubMed] [Google Scholar]
- 2.Hotamisligil G.S. Inflammation, metaflammation and immunometabolic disorders. Nature. 2017;542:177. doi: 10.1038/nature21363. [DOI] [PubMed] [Google Scholar]
- 3.Heppner F.L., Ransohoff R.M., Becher B. Immune attack: The role of inflammation in Alzheimer disease. Nat. Rev. Neurosci. 2015;16:358–372. doi: 10.1038/nrn3880. [DOI] [PubMed] [Google Scholar]
- 4.Swirski F.K., Nahrendorf M. Leukocyte behavior in atherosclerosis, myocardial infarction, and heart failure. Science. 2013;339:161–166. doi: 10.1126/science.1230719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tabas I., Glass C.K. Anti-inflammatory therapy in chronic disease: Challenges and opportunities. Science. 2013;339:166–172. doi: 10.1126/science.1230720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Libby P. Atherosclerosis. Nat. Rev. Dis. Primers. 2019;5:56. doi: 10.1038/s41572-019-0106-z. [DOI] [PubMed] [Google Scholar]
- 7.Hotamisligil G.S. Inflammation and metabolic disorders. Nature. 2006;444:860–867. doi: 10.1038/nature05485. [DOI] [PubMed] [Google Scholar]
- 8.Donath M.Y., Shoelson S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011;11:98. doi: 10.1038/nri2925. [DOI] [PubMed] [Google Scholar]
- 9.Ruparelia N., Chai J.T., Fisher E.A., Choudhury R.P. Inflammatory processes in cardiovascular disease: A route to targeted therapies. Nat. Rev. Cardiol. 2017;14:133–144. doi: 10.1038/nrcardio.2016.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fullerton J.N., Gilroy D.W. Resolution of inflammation: A new therapeutic frontier. Nat. Rev. Drug Discov. 2016;15:551–567. doi: 10.1038/nrd.2016.39. [DOI] [PubMed] [Google Scholar]
- 11.Vane J.R., Botting R.M. Mechanism of action of nonsteroidal anti-inflammatory drugs. Am. J. Med. 1998;104:2S–8S. doi: 10.1016/s0002-9343(97)00203-9. [DOI] [PubMed] [Google Scholar]
- 12.Barnes P.J. How corticosteroids control inflammation: Quintiles prize lecture 2005. Br. J. Pharmacol. 2006;148:245–254. doi: 10.1038/sj.bjp.0706736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li P., Zheng Y., Chen X. Drugs for autoimmune inflammatory diseases: From small molecule compounds to anti-TNF biologics. Front. Pharmacol. 2017;8:460. doi: 10.3389/fphar.2017.00460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Canalis E. Mechanisms of glucocorticoid-induced osteoporosis. Curr. Opin. Rheumatol. 2003;15:454–457. doi: 10.1097/00002281-200307000-00013. [DOI] [PubMed] [Google Scholar]
- 15.Buchman A.L. Side effects of corticosteroid therapy. J. Clin. Gastroenterol. 2001;33:289–294. doi: 10.1097/00004836-200110000-00006. [DOI] [PubMed] [Google Scholar]
- 16.Gabriel S.E., Jaakkimainen L., Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflammatory drugs: A meta-analysis. Ann. Intern. Med. 1991;115:787–796. doi: 10.7326/0003-4819-115-10-787. [DOI] [PubMed] [Google Scholar]
- 17.Bjarnason I., Hayllar J., Macpherson A.J., Russell A.S. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology. 1993;104:1832–1847. doi: 10.1016/0016-5085(93)90667-2. [DOI] [PubMed] [Google Scholar]
- 18.Andrade R.J. Drug-induced liver injury. Nat. Rev. Dis. Primers. 2019;5:58. doi: 10.1038/s41572-019-0105-0. [DOI] [PubMed] [Google Scholar]
- 19.Howard P.A., Delafontaine P. Nonsteroidal anti-inflammatory drugs and cardiovascular risk. J. Am. Coll. Cardiol. 2004;43:519–525. doi: 10.1016/j.jacc.2003.09.043. [DOI] [PubMed] [Google Scholar]
- 20.Taylor P.C., Feldmann M. Anti-TNF biologic agents: Still the therapy of choice for rheumatoid arthritis. Nat. Rev. Rheumatol. 2009;5:578. doi: 10.1038/nrrheum.2009.181. [DOI] [PubMed] [Google Scholar]
- 21.Targan S.R. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor α for crohn's disease. N. Engl. J. Med. 1997;337:1029–1036. doi: 10.1056/NEJM199710093371502. [DOI] [PubMed] [Google Scholar]
- 22.Feagan B.G. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N. Engl. J. Med. 2013;369:699–710. doi: 10.1056/NEJMoa1215734. [DOI] [PubMed] [Google Scholar]
- 23.Ridker P.M. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 2017;377:1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 24.Kojima Y. CD47-blocking antibodies restore phagocytosis and prevent atherosclerosis. Nature. 2016;536:86–90. doi: 10.1038/nature18935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Leckie M.J. Effects of an interleukin-5 blocking monoclonal antibody on eosinophils, airway hyper-responsìveness, and the late asthmatic response. Lancet. 2000;356:2144–2148. doi: 10.1016/s0140-6736(00)03496-6. [DOI] [PubMed] [Google Scholar]
- 26.Khilfeh I. Adherence, persistence, and expenditures for high-cost anti-inflammatory drugs in rheumatoid arthritis: An exploratory study. J. Manag. Care Spec. Pharm. 2019;25:461–467. doi: 10.18553/jmcp.2019.25.4.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vermeire S., Gils A., Accossato P., Lula S., Marren A. Immunogenicity of biologics in inflammatory bowel disease. Ther. Adv. Gastroenterol. 2018;11:1–13. doi: 10.1177/1756283X17750355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Borman Z.A., Cote-Daigneault J., Colombel J.F. The risk for opportunistic infections in inflammatory bowel disease with biologics: An update. Expert Rev. Gastroenterol. Hepatol. 2018;12:1101–1108. doi: 10.1080/17474124.2018.1530983. [DOI] [PubMed] [Google Scholar]
- 29.Bonovas S. Biologic therapies and risk of infection and malignancy in patients with inflammatory bowel disease: A systematic review and network meta-analysis. Clin. Gastroenterol. Hepatol. 2016;14:1385–1397. doi: 10.1016/j.cgh.2016.04.039. [DOI] [PubMed] [Google Scholar]
- 30.Bongartz T. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: Systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA. 2006;295:2275–2285. doi: 10.1001/jama.295.19.2275. [DOI] [PubMed] [Google Scholar]
- 31.Serhan C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature. 2014;510:92–101. doi: 10.1038/nature13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Serhan C.N., Petasis N.A. Resolvins and protectins in inflammation resolution. Chem. Rev. 2011;111:5922–5943. doi: 10.1021/cr100396c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kotla N.G. Bioresponsive drug delivery systems in intestinal inflammation: State-of-the-art and future perspectives. Adv. Drug Deliv. Rev. 2019;146:248–266. doi: 10.1016/j.addr.2018.06.021. [DOI] [PubMed] [Google Scholar]
- 34.Thakur S. Novel drug delivery systems for NSAIDs in management of rheumatoid arthritis: An overview. Biomed. Pharmacother. 2018;106:1011–1023. doi: 10.1016/j.biopha.2018.07.027. [DOI] [PubMed] [Google Scholar]
- 35.Lalit K., Shivani V., Mehakjot S., Tammana C., Puneet U. Advanced drug delivery systems for transdermal delivery of non-steroidal anti-inflammatory drugs: A review. Curr. Drug Deliv. 2018;15:1087–1099. doi: 10.2174/1567201815666180605114131. [DOI] [PubMed] [Google Scholar]
- 36.Conte R., Marturano V., Peluso G., Calarco A., Cerruti P. Recent advances in nanoparticle-mediated delivery of anti-inflammatory phytocompounds. Int. J. Mol. Sci. 2017;18:709. doi: 10.3390/ijms18040709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tran T.-H., Amiji M.M. Targeted delivery systems for biological therapies of inflammatory diseases. Expert Opin. Drug Deliv. 2015;12:393–414. doi: 10.1517/17425247.2015.972931. [DOI] [PubMed] [Google Scholar]
- 38.Li C.-W., Li L.-L., Chen S., Zhang J.-X., Lu W.-L. Antioxidant nanotherapies for the treatment of inflammatory diseases. Front. Bioeng. Biotechnol. 2020;8:200. doi: 10.3389/fbioe.2020.00200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ha D., Yang N., Nadithe V. Exosomes as therapeutic drug carriers and delivery vehicles across biological membranes: Current perspectives and future challenges. Acta Pharm. Sin. B. 2016;6:287–296. doi: 10.1016/j.apsb.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yang G., Chen S., Zhang J. Bioinspired and biomimetic nanotherapies for the treatment of infectious diseases. Front. Pharmacol. 2019;10:751. doi: 10.3389/fphar.2019.00751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yan H. Engineering cell membrane-based nanotherapeutics to target inflammation. Adv. Sci. 2019;6:1900605. doi: 10.1002/advs.201900605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chu D., Dong X., Shi X., Zhang C., Wang Z. Neutrophil-based drug delivery systems. Adv. Mater. 2018;30:1706245. doi: 10.1002/adma.201706245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang S. An inflammation-targeting hydrogel for local drug delivery in inflammatory bowel disease. Sci. Transl. Med. 2015;7:300ra128. doi: 10.1126/scitranslmed.aaa5657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lamprecht A. Nanomedicines in gastroenterology and hepatology. Nat. Rev. Gastroenterol. Hepatol. 2015;12:195–204. doi: 10.1038/nrgastro.2015.37. [DOI] [PubMed] [Google Scholar]
- 45.Zhang S., Langer R., Traverso G. Nanoparticulate drug delivery systems targeting inflammation for treatment of inflammatory bowel disease. Nano Today. 2017;16:82–96. doi: 10.1016/j.nantod.2017.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mahmoudi M. Multiscale technologies for treatment of ischemic cardiomyopathy. Nat. Nanotechnol. 2017;12:845–855. doi: 10.1038/nnano.2017.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhang D., Zhang J. Surface engineering of nanomaterials with phospholipid-polyethylene glycol-derived functional conjugates for molecular imaging and targeted therapy. Biomaterials. 2020;230:119646. doi: 10.1016/j.biomaterials.2019.119646. [DOI] [PubMed] [Google Scholar]
- 48.Flores A.M. Nanoparticle therapy for vascular diseases. Arterioscler. Thromb. Vasc. Biol. 2019;39:635–646. doi: 10.1161/ATVBAHA.118.311569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dormont F., Varna M., Couvreur P. Nanoplumbers: Biomaterials to fight cardiovascular diseases. Mater. Today. 2018;21:122–143. [Google Scholar]
- 50.Luk B.T., Zhang L. Cell membrane-camouflaged nanoparticles for drug delivery. J. Control. Release. 2015;220:600–607. doi: 10.1016/j.jconrel.2015.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mulder W.J., Jaffer F.A., Fayad Z.A., Nahrendorf M. Imaging and nanomedicine in inflammatory atherosclerosis. Sci. Transl. Med. 2014;6:239sr231. doi: 10.1126/scitranslmed.3005101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.He W., Kapate N., Shields C.W., Mitragotri S. Drug delivery to macrophages: A review of targeting drugs and drug carriers to macrophages for inflammatory diseases. Adv. Drug Deliv. Rev. 2020 doi: 10.1016/j.addr.2019.1012.1001. [DOI] [PubMed] [Google Scholar]
- 53.Brusini R., Varna M., Couvreur P. Advanced nanomedicines for the treatment of inflammatory diseases. Adv. Drug Deliv. Rev. 2020 doi: 10.1016/j.addr.2020.1007.1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Nagy J.A., Benjamin L., Zeng H., Dvorak A.M., Dvorak H.F. Vascular permeability, vascular hyperpermeability and angiogenesis. Angiogenesis. 2008;11:109–119. doi: 10.1007/s10456-008-9099-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wilhelm D.L. Mechanisms responsible for increased vascular permeability in acute inflammation. Agents Actions. 1973;3:297–306. doi: 10.1007/BF01986484. [DOI] [PubMed] [Google Scholar]
- 56.Hu X., Yang G., Chen S., Luo S., Zhang J. Biomimetic and bioinspired strategies for oral drug delivery. Biomater. Sci. 2020;8:1020–1044. doi: 10.1039/c9bm01378d. [DOI] [PubMed] [Google Scholar]
- 57.Su J., Hu B.H., Lowe W.L., Jr., Kaufman D.B., Messersmith P.B. Anti-inflammatory peptide-functionalized hydrogels for insulin-secreting cell encapsulation. Biomaterials. 2010;31:308–314. doi: 10.1016/j.biomaterials.2009.09.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cruz-Acuña R. Synthetic hydrogels for human intestinal organoid generation and colonic wound repair. Nat. Cell Biol. 2017;19:1326–1335. doi: 10.1038/ncb3632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dellacherie M.O., Seo B.R., Mooney D.J. Macroscale biomaterials strategies for local immunomodulation. Nat. Rev. Mater. 2019;4:379–397. [Google Scholar]
- 60.Rajkovic O. Reactive oxygen species-responsive nanoparticles for the treatment of ischemic stroke. Adv. Ther. 2019;2:1900038. [Google Scholar]
- 61.Fu X., Hosta-Rigau L., Chandrawati R., Cui J. Multi-stimuli-responsive polymer particles, films, and hydrogels for drug delivery. Chem. 2018;4:2084–2107. [Google Scholar]
- 62.Saravanakumar G., Kim J., Kim W.J. Reactive-oxygen-species-responsive drug delivery systems: Promises and challenges. Adv. Sci. 2017;4:1600124. doi: 10.1002/advs.201600124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lu Y., Aimetti A.A., Langer R., Gu Z. Bioresponsive materials. Nat. Rev. Mater. 2016;2:16075. [Google Scholar]
- 64.Zhuang J., Gordon M.R., Ventura J., Li L., Thayumanavan S. Multi-stimuli responsive macromolecules and their assemblies. Chem. Soc. Rev. 2013;42:7421–7435. doi: 10.1039/c3cs60094g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Mura S., Nicolas J., Couvreur P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013;12:991. doi: 10.1038/nmat3776. [DOI] [PubMed] [Google Scholar]
- 66.Yang G., Song J., Zhang J. London; England: 2020. Biomimetic and Bioresponsive Nanotherapies for Inflammatory Vascular Diseases, Nanomedicine. [DOI] [PubMed] [Google Scholar]
- 67.Gabay C., Kushner I. Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 1999;340:448–454. doi: 10.1056/NEJM199902113400607. [DOI] [PubMed] [Google Scholar]
- 68.Zhang J.-M., An J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007;45:27–37. doi: 10.1097/AIA.0b013e318034194e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Rock K.L., Kono H. The inflammatory response to cell death. Annu. Rev. Pathol. 2008;3:99–126. doi: 10.1146/annurev.pathmechdis.3.121806.151456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mantovani A., Cassatella M.A., Costantini C., Jaillon S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat. Rev. Immunol. 2011;11:519–531. doi: 10.1038/nri3024. [DOI] [PubMed] [Google Scholar]
- 71.Dale D.C., Boxer L., Liles W.C. The phagocytes: Neutrophils and monocytes. Blood. 2008;112:935–945. doi: 10.1182/blood-2007-12-077917. [DOI] [PubMed] [Google Scholar]
- 72.Hoenderdos K., Condliffe A. The neutrophil in chronic obstructive pulmonary disease. Am. J. Respir. Cell Mol. Biol. 2013;48:531–539. doi: 10.1165/rcmb.2012-0492TR. [DOI] [PubMed] [Google Scholar]
- 73.Wright H.L., Moots R.J., Edwards S.W. The multifactorial role of neutrophils in rheumatoid arthritis. Nat. Rev. Rheumatol. 2014;10:593–601. doi: 10.1038/nrrheum.2014.80. [DOI] [PubMed] [Google Scholar]
- 74.Fournier B.M., Parkos C.A. The role of neutrophils during intestinal inflammation. Mucosal immunol. 2012;5:354–366. doi: 10.1038/mi.2012.24. [DOI] [PubMed] [Google Scholar]
- 75.Weber C., Zernecke A., Libby P. The multifaceted contributions of leukocyte subsets to atherosclerosis: Lessons from mouse models. Nat. Rev. Immunol. 2008;8:802–815. doi: 10.1038/nri2415. [DOI] [PubMed] [Google Scholar]
- 76.Németh T., Sperandio M., Mócsai A. Neutrophils as emerging therapeutic targets. Nat. Rev. Drug Discov. 2020;19:253–275. doi: 10.1038/s41573-019-0054-z. [DOI] [PubMed] [Google Scholar]
- 77.Xu X. A self-illuminating nanoparticle for inflammation imaging and cancer therapy. Sci. Adv. 2019;5:eaat2953. doi: 10.1126/sciadv.aat2953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Guo J. A myeloperoxidase-responsive and biodegradable luminescent material for real-time imaging of inflammatory diseases. Mater. Today. 2017;20:493–500. [Google Scholar]
- 79.Tao H. Luminescence imaging of acute liver injury by biodegradable and biocompatible nanoprobes. ACS Nano. 2020 doi: 10.1021/acsnano.1020c00539. [DOI] [PubMed] [Google Scholar]
- 80.Terrault N.A. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology. 2018;67:1560–1599. doi: 10.1002/hep.29800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Weissleder R., Nahrendorf M., Pittet M.J. Imaging macrophages with nanoparticles. Nat. Mater. 2014;13:125–138. doi: 10.1038/nmat3780. [DOI] [PubMed] [Google Scholar]
- 82.Galli S.J., Tsai M. Ige and mast cells in allergic disease. Nat. Med. 2012;18:693–704. doi: 10.1038/nm.2755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Siracusa M.C., Kim B.S., Spergel J.M., Artis D. Basophils and allergic inflammation. J. Allergy Clin. Immunol. 2013;132:789–790. doi: 10.1016/j.jaci.2013.07.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Klion A.D., Nutman T.B. The role of eosinophils in host defense against helminth parasites. J. Allergy Clin. Immunol. 2004;113:30–37. doi: 10.1016/j.jaci.2003.10.050. [DOI] [PubMed] [Google Scholar]
- 85.Zhang X. Bioinspired yeast microcapsules loaded with self-assembled nanotherapies for targeted treatment of cardiovascular disease. Mater. Today. 2017;20:301–313. [Google Scholar]
- 86.Zhou X. Yeast microcapsule-mediated targeted delivery of diverse nanoparticles for imaging and therapy via the oral route. Nano Lett. 2017;17:1056–1064. doi: 10.1021/acs.nanolett.6b04523. [DOI] [PubMed] [Google Scholar]
- 87.Sennesael J.J., De Smedt G.C., Van der Niepen P., Verbeelen D.L. The impact of peritonitis on peritoneal and systemic acid-base status of patients on continuous ambulatory peritoneal dialysis. Perit. Dial. Int. 1994;14:61–65. [PubMed] [Google Scholar]
- 88.Zhang R. A pH/ROS dual-responsive and targeting nanotherapy for vascular inflammatory diseases. Biomaterials. 2020;230:119605. doi: 10.1016/j.biomaterials.2019.119605. [DOI] [PubMed] [Google Scholar]
- 89.Nedergaard M., Kraig R.P., Tanabe J., Pulsinelli W.A. Dynamics of interstitial and intracellular pH in evolving brain infarct. Am. J. Physiol. 1991;260:R581–R588. doi: 10.1152/ajpregu.1991.260.3.R581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Garlick P.B., Radda G.K., Seeley P.J. Studies of acidosis in the ischaemic heart by phosphorus nuclear magnetic resonance. Biochem. J. 1979;184:547–554. doi: 10.1042/bj1840547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Naghavi M. pH heterogeneity of human and rabbit atherosclerotic plaques; a new insight into detection of vulnerable plaque. Atherosclerosis. 2002;164:27–35. doi: 10.1016/s0021-9150(02)00018-7. [DOI] [PubMed] [Google Scholar]
- 92.Ricciardolo F.L., Gaston B., Hunt J. Acid stress in the pathology of asthma. J. Allergy Clin. Immunol. 2004;113:610–619. doi: 10.1016/j.jaci.2003.12.034. [DOI] [PubMed] [Google Scholar]
- 93.Treuhaft P.S., MCCarty D.J. Synovial fluid pH, lactate, oxygen and carbon dioxide partial pressure in various joint diseases. Arthritis Rheum. 1971;14:475–484. doi: 10.1002/art.1780140407. [DOI] [PubMed] [Google Scholar]
- 94.Andersen N.E., Gyring J., Hansen A.J., Laursen H., Siesjo B.K. Brain acidosis in experimental pneumococcal meningitis. J. Cereb. Blood Flow Metab. 1989;9:381–387. doi: 10.1038/jcbfm.1989.57. [DOI] [PubMed] [Google Scholar]
- 95.Walters G., Pountos I., Giannoudis P.V. The cytokines and micro-environment of fracture haematoma: Current evidence. J. Tissue Eng. Regen. Med. 2018;12:e1662–e1677. doi: 10.1002/term.2593. [DOI] [PubMed] [Google Scholar]
- 96.Hunt J.F. Endogenous airway acidification. Implications for asthma pathophysiology. Am. J. Respir. Crit. Care Med. 2000;161:694–699. doi: 10.1164/ajrccm.161.3.9911005. [DOI] [PubMed] [Google Scholar]
- 97.Geborek P., Saxne T., Pettersson H., Wollheim F.A. Synovial fluid acidosis correlates with radiological joint destruction in rheumatoid arthritis knee joints. J. Rheumatol. 1989;16:468–472. [PubMed] [Google Scholar]
- 98.Cummings N.A., Nordby G.L. Measurement of synovial fluid pH in normal and arthritic knees. Arthritis Rheum. 1966;9:47–56. doi: 10.1002/art.1780090106. [DOI] [PubMed] [Google Scholar]
- 99.Kellum J.A., Song M., Li J. Science review: Extracellular acidosis and the immune response: Clinical and physiologic implications. Crit. Care. 2004;8:331–336. doi: 10.1186/cc2900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Brook I. The importance of lactic acid levels in body fluids in the detection of bacterial infections. Rev. Infect. Dis. 1981;3:470–478. doi: 10.1093/clinids/3.3.470. [DOI] [PubMed] [Google Scholar]
- 101.Rajamäki K. Extracellular acidosis is a novel danger signal alerting innate immunity via the NLRP3 inflammasome. J. Biol. Chem. 2013;288:13410–13419. doi: 10.1074/jbc.M112.426254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Erra Díaz F., Dantas E., Geffner J. Unravelling the interplay between extracellular acidosis and immune cells. Mediators Inflamm. 2018;2018:1218297. doi: 10.1155/2018/1218297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.de Nadai T.R. Metabolic acidosis treatment as part of a strategy to curb inflammation. Int. J. Inflam. 2013;2013:601424. doi: 10.1155/2013/601424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Gao W., Chan J.M., Farokhzad O.C. pH-responsive nanoparticles for drug delivery. Mol. Pharm. 2010;7:1913–1920. doi: 10.1021/mp100253e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Deirram N., Zhang C., Kermaniyan S.S., Johnston A.P.R., Such G.K. pH-responsive polymer nanoparticles for drug delivery. Macromol. Rapid Commun. 2019;40:1800917. doi: 10.1002/marc.201800917. [DOI] [PubMed] [Google Scholar]
- 106.Liu L., Yao W., Rao Y., Lu X., Gao J. pH-responsive carriers for oral drug delivery: Challenges and opportunities of current platforms. Drug Deliv. 2017;24:569–581. doi: 10.1080/10717544.2017.1279238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Tao W., Wang J., Parak W.J., Farokhzad O.C., Shi J. Nanobuffering of pH-responsive polymers: A known but sometimes overlooked phenomenon and its biological applications. ACS Nano. 2019;13:4876–4882. doi: 10.1021/acsnano.9b01696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ryter S.W. Mechanisms of cell death in oxidative stress. Antioxid. Redox Signal. 2007;9:49–89. doi: 10.1089/ars.2007.9.49. [DOI] [PubMed] [Google Scholar]
- 109.Sedghi S. Increased production of luminol enhanced chemiluminescence by the inflamed colonic mucosa in patients with ulcerative colitis. Gut. 1993;34:1191–1197. doi: 10.1136/gut.34.9.1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Simmonds N.J. Chemiluminescence assay of mucosal reactive oxygen metabolites in inflammatory bowel disease. Gastroenterology. 1992;103:186–196. doi: 10.1016/0016-5085(92)91112-h. [DOI] [PubMed] [Google Scholar]
- 111.Feng S. Nanoparticles responsive to the inflammatory microenvironment for targeted treatment of arterial restenosis. Biomaterials. 2016;105:167–184. doi: 10.1016/j.biomaterials.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 112.Uttara B., Singh A.V., Zamboni P., Mahajan R.T. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009;7:65–74. doi: 10.2174/157015909787602823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kostikas K., Papatheodorou G., Psathakis K., Panagou P., Loukides S. Oxidative stress in expired breath condensate of patients with copd. Chest. 2003;124:1373–1380. doi: 10.1378/chest.124.4.1373. [DOI] [PubMed] [Google Scholar]
- 114.Lacy P. Mechanisms of degranulation in neutrophils. Allergy Asthma Clin. Immunol. 2006;2:98–108. doi: 10.1186/1710-1492-2-3-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Witko-Sarsat V., Rieu P., Descamps-Latscha B., Lesavre P., Halbwachs-Mecarelli L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Invest. 2000;80:617–653. doi: 10.1038/labinvest.3780067. [DOI] [PubMed] [Google Scholar]
- 116.Viola A., Munari F., Sánchez-Rodríguez R., Scolaro T., Castegna A. The metabolic signature of macrophage responses. Front. Immunol. 2019;10:1462. doi: 10.3389/fimmu.2019.01462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Seibert K. Mediation of inflammation by cyclooxygenase-2. Agents Actions. Suppl. 1995;46:41–50. doi: 10.1007/978-3-0348-7276-8_5. [DOI] [PubMed] [Google Scholar]
- 118.Egorov A.M., Ulyashova M.M., Rubtsova M.Y. Bacterial enzymes and antibiotic resistance. Acta Naturae. 2018;10:33–48. [PMC free article] [PubMed] [Google Scholar]
- 119.Fingleton B. Matrix metalloproteinases as regulators of inflammatory processes. Biochim. Biophys. Acta Mol. Cell Res. 2017;1864:2036–2042. doi: 10.1016/j.bbamcr.2017.05.010. [DOI] [PubMed] [Google Scholar]
- 120.Simon L.S. Role and regulation of cyclooxygenase-2 during inflammation. Am. J. Med. 1999;106:37S–42S. doi: 10.1016/s0002-9343(99)00115-1. [DOI] [PubMed] [Google Scholar]
- 121.Minghetti L. Cyclooxygenase-2 (COX-2) in inflammatory and degenerative brain diseases. J. Neuropathol. Exp. Neurol. 2004;63:901–910. doi: 10.1093/jnen/63.9.901. [DOI] [PubMed] [Google Scholar]
- 122.Bartels K., Grenz A., Eltzschig H.K. Hypoxia and inflammation are two sides of the same coin. Proc. Natl. Acad. Sci. U. S. A. 2013;110:18351–18352. doi: 10.1073/pnas.1318345110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Taylor C.T., Colgan S.P. Regulation of immunity and inflammation by hypoxia in immunological niches. Nat. Rev. Immunol. 2017;17:774–785. doi: 10.1038/nri.2017.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Korin N. Shear-activated nanotherapeutics for drug targeting to obstructed blood vessels. Science. 2012;337:738–742. doi: 10.1126/science.1217815. [DOI] [PubMed] [Google Scholar]
- 125.Fishbein Michael C., Siegel Robert J. How big are coronary atherosclerotic plaques that rupture? Circulation. 1996;94:2662–2666. doi: 10.1161/01.cir.94.10.2662. [DOI] [PubMed] [Google Scholar]
- 126.Chanmugam A. Relative temperature maximum in wound infection and inflammation as compared with a control subject using long-wave infrared thermography. Adv. Skin Wound Care. 2017;30:406–414. doi: 10.1097/01.ASW.0000522161.13573.62. [DOI] [PubMed] [Google Scholar]
- 127.Evans S.S., Repasky E.A., Fisher D.T. Fever and the thermal regulation of immunity: The immune system feels the heat. Nat. Rev. Immunol. 2015;15:335–349. doi: 10.1038/nri3843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Tao W., He Z. ROS-responsive drug delivery systems for biomedical applications. Asian J. Pharm. 2018;13:101–112. doi: 10.1016/j.ajps.2017.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Schiffer D. Enzyme-responsive polymers for microbial infection detection. Expert Rev. Mol. Diagn. 2015;15:1125–1131. doi: 10.1586/14737159.2015.1061935. [DOI] [PubMed] [Google Scholar]
- 130.Xue F.C., Zhang J.X. Stimuli-responsive nanoparticles derived from cyclodextrin materials for drug delivery. Sci. Lett. 2015;4:195. [Google Scholar]
- 131.Kocak G., Tuncer C., Butun V. pH-responsive polymers. Polym. Chem. 2017;8:144–176. [Google Scholar]
- 132.Liu J. pH-sensitive nano-systems for drug delivery in cancer therapy. Biotechnol. Adv. 2014;32:693–710. doi: 10.1016/j.biotechadv.2013.11.009. [DOI] [PubMed] [Google Scholar]
- 133.Sonawane S.J., Kalhapure R.S., Govender T. Hydrazone linkages in pH responsive drug delivery systems. Eur. J. Pharm. Sci. 2017;99:45–65. doi: 10.1016/j.ejps.2016.12.011. [DOI] [PubMed] [Google Scholar]
- 134.Heller J., Barr J. Poly(ortho esters)from concept to reality. Biomacromolecules. 2004;5:1625–1632. doi: 10.1021/bm040049n. [DOI] [PubMed] [Google Scholar]
- 135.Heller J., Barr J., Ng S.Y., Abdellauoi K.S., Gurny R. Poly(ortho esters): Synthesis, characterization, properties and uses. Adv. Drug Deliv. Rev. 2002;54:1015–1039. doi: 10.1016/s0169-409x(02)00055-8. [DOI] [PubMed] [Google Scholar]
- 136.Heller J. Poly(ortho esters)-their development and some recent applications. Eur. J. Pharm. Biopharm. 2000;50:121–128. doi: 10.1016/s0939-6411(00)00085-0. [DOI] [PubMed] [Google Scholar]
- 137.Einmahl S. Therapeutic applications of viscous and injectable poly(ortho esters) Adv. Drug Deliv. Rev. 2001;53:45–73. doi: 10.1016/s0169-409x(01)00220-4. [DOI] [PubMed] [Google Scholar]
- 138.Heller J. Ocular delivery using poly(ortho esters) Adv. Drug Deliv. Rev. 2005;57:2053–2062. doi: 10.1016/j.addr.2005.09.007. [DOI] [PubMed] [Google Scholar]
- 139.Heller J. Polymers for controlled parenteral delivery of peptides and proteins. Adv. Drug Deliv. Rev. 1993;10:163–204. [Google Scholar]
- 140.Wang C. Molecularly engineered poly(ortho ester) microspheres for enhanced delivery of DNA vaccines. Nat. Mater. 2004;3:190–196. doi: 10.1038/nmat1075. [DOI] [PubMed] [Google Scholar]
- 141.Toncheva V., Schacht E., Ng S.Y., Barr J., Heller J. Use of block copolymers of poly(ortho esters) and poly (ethylene glycol) micellar carriers as potential tumour targeting systems. J. Drug Target. 2003;11:345–353. doi: 10.1080/10611860310001633839. [DOI] [PubMed] [Google Scholar]
- 142.Guo X., Szoka F.C., Jr. Steric stabilization of fusogenic liposomes by a low-pH sensitive PEG-diortho ester-lipid conjugate. Bioconjug. Chem. 2001;12:291–300. doi: 10.1021/bc000110v. [DOI] [PubMed] [Google Scholar]
- 143.Cheng J., Ji R., Gao S.J., Du F.S., Li Z.C. Facile synthesis of acid-labile polymers with pendent ortho esters. Biomacromolecules. 2012;13:173–179. doi: 10.1021/bm201410c. [DOI] [PubMed] [Google Scholar]
- 144.Qiao Z.Y., Zhang R., Du F.S., Liang D.H., Li Z.C. Multi-responsive nanogels containing motifs of ortho ester, oligo(ethylene glycol) and disulfide linkage as carriers of hydrophobic anti-cancer drugs. J. Control. Release. 2011;152:57–66. doi: 10.1016/j.jconrel.2011.02.029. [DOI] [PubMed] [Google Scholar]
- 145.Wang J. pH-sensitive amphiphilic triblock copolymers containing ortho ester main-chains as efficient drug delivery platforms. Mater. Sci. Eng. C. 2019;94:169–178. doi: 10.1016/j.msec.2018.09.029. [DOI] [PubMed] [Google Scholar]
- 146.Fu S. Acid-degradable poly(ortho ester urethanes) copolymers for potential drug carriers: Preparation, characterization, in vitro and in vivo evaluation. Polymer. 2017;114:1–14. [Google Scholar]
- 147.Ji R. Acid-sensitive polypseudorotaxanes based on ortho ester-modified cyclodextrin and pluronic F-127. ACS Macro Lett. 2015;4:65–69. doi: 10.1021/mz5007359. [DOI] [PubMed] [Google Scholar]
- 148.Ji R. Shell-sheddable, pH-sensitive supramolecular nanoparticles based on ortho ester-modified cyclodextrin and adamantyl PEG. Biomacromolecules. 2014;15:3531–3539. doi: 10.1021/bm500711c. [DOI] [PubMed] [Google Scholar]
- 149.Cordes E.H., Bull H.G. Mechanism and catalysis for hydrolysis of acetals, ketals, and ortho esters. Chem. Rev. 1974;74:581–603. [Google Scholar]
- 150.Tietze L.F. Development of tailor-made cytostatics activable by acid-catalyzed hydrolysis for selective tumor therapy. Angew. Chem. Int. Ed. 1990;29:782–783. [Google Scholar]
- 151.Srinivasachar K., Neville D.M. New protein crosslinking reagents that are cleaved by mild acid. Biochemistry. 1989;28:2501–2509. doi: 10.1021/bi00432a023. [DOI] [PubMed] [Google Scholar]
- 152.Gillies E.R., Goodwin A.P., Fréchet J.M.J. Acetals as pH-sensitive linkages for drug delivery. Bioconjug. Chem. 2004;15:1254–1263. doi: 10.1021/bc049853x. [DOI] [PubMed] [Google Scholar]
- 153.Murthy N. A macromolecular delivery vehicle for protein-based vaccines: Acid-degradable protein-loaded microgels. Proc. Natl. Acad. Sci. U. S. A. 2003;100:4995–5000. doi: 10.1073/pnas.0930644100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Murthy N., Thng Y.X., Schuck S., Xu M.C., Fréchet J.M. A novel strategy for encapsulation and release of proteins: Hydrogels and microgels with acid-labile acetal cross-linkers. J. Am. Chem. Soc. 2002;124:12398–12399. doi: 10.1021/ja026925r. [DOI] [PubMed] [Google Scholar]
- 155.Liu B., Thayumanavan S. Substituent effects on the pH sensitivity of acetals and ketals and their correlation with encapsulation stability in polymeric nanogels. J. Am. Chem. Soc. 2017;139:2306–2317. doi: 10.1021/jacs.6b11181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Miller K.A. Acid-triggered, acid-generating, and self-amplifying degradable polymers. J. Am. Chem. Soc. 2019;141:2838–2842. doi: 10.1021/jacs.8b07705. [DOI] [PubMed] [Google Scholar]
- 157.Paramonov S.E. Fully acid-degradable biocompatible polyacetal microparticles for drug delivery. Bioconjug. Chem. 2008;19:911–919. doi: 10.1021/bc7004472. [DOI] [PubMed] [Google Scholar]
- 158.Heffernan M.J., Murthy N. Polyketal nanoparticles: A new pH-sensitive biodegradable drug delivery vehicle. Bioconjug. Chem. 2005;16:1340–1342. doi: 10.1021/bc050176w. [DOI] [PubMed] [Google Scholar]
- 159.Lee S., Yang S.C., Heffernan M.J., Taylor W.R., Murthy N. Polyketal microparticles: A new delivery vehicle for superoxide dismutase. Bioconjug. Chem. 2007;18:4–7. doi: 10.1021/bc060259s. [DOI] [PubMed] [Google Scholar]
- 160.Yang S.C., Bhide M., Crispe I.N., Pierce R.H., Murthy N. Polyketal copolymers: A new acid-sensitive delivery vehicle for treating acute inflammatory diseases. Bioconjug. Chem. 2008;19:1164–1169. doi: 10.1021/bc700442g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Fiore V.F. Polyketal microparticles for therapeutic delivery to the lung. Biomaterials. 2010;31:810–817. doi: 10.1016/j.biomaterials.2009.09.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Somasuntharam I. Delivery of Nox2-NADPH oxidase siRNA with polyketal nanoparticles for improving cardiac function following myocardial infarction. Biomaterials. 2013;34:7790–7798. doi: 10.1016/j.biomaterials.2013.06.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bachelder E.M., Beaudette T.T., Broaders K.E., Dashe J., Fréchet J.M.J. Acetal-derivatized dextran: An acid-responsive biodegradable material for therapeutic applications. J. Am. Chem. Soc. 2008;130:10494–10495. doi: 10.1021/ja803947s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Broaders K.E., Cohen J.A., Beaudette T.T., Bachelder E.M., Fréchet J.M.J. Acetalated dextran is a chemically and biologically tunable material for particulate immunotherapy. Proc. Natl. Acad. Sci. U. S. A. 2009;106:5497. doi: 10.1073/pnas.0901592106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Cui L. Conjugation chemistry through acetals toward a dextran-based delivery system for controlled release of siRNA. J. Am. Chem. Soc. 2012;134:15840–15848. doi: 10.1021/ja305552u. [DOI] [PubMed] [Google Scholar]
- 166.Bachelder E.M., Pino E.N., Ainslie K.M. Acetalated dextran: A tunable and acid-labile biopolymer with facile synthesis and a range of applications. Chem. Rev. 2017;117:1915–1926. doi: 10.1021/acs.chemrev.6b00532. [DOI] [PubMed] [Google Scholar]
- 167.Cohen J.A. Acetal-modified dextran microparticles with controlled degradation kinetics and surface functionality for gene delivery in phagocytic and non-phagocytic cells. Adv. Mater. 2010;22:3593–3597. doi: 10.1002/adma.201000307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Volpatti L.R. Glucose-responsive nanoparticles for rapid and extended self-regulated insulin delivery. ACS Nano. 2020;14:488–497. doi: 10.1021/acsnano.9b06395. [DOI] [PubMed] [Google Scholar]
- 169.Chen N. Tunable degradation of acetalated dextran microparticles enables controlled vaccine adjuvant and antigen delivery to modulate adaptive immune responses. J. Control. Release. 2018;273:147–159. doi: 10.1016/j.jconrel.2018.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Zhang D. Engineering of biocompatible pH-responsive nanovehicles from acetalated cyclodextrins as effective delivery systems for tumor therapy. J. Biomed. Nanotechnol. 2015;11:923–941. doi: 10.1166/jbn.2015.2009. [DOI] [PubMed] [Google Scholar]
- 171.Zhang J., Jia Y., Li X., Hu Y., Li X. Facile engineering of biocompatible materials with pH-modulated degradability. Adv. Mater. 2011;23:3035–3040. doi: 10.1002/adma.201100679. [DOI] [PubMed] [Google Scholar]
- 172.He H. Cyclodextrin-derived pH-responsive nanoparticles for delivery of paclitaxel. Biomaterials. 2013;34:5344–5358. doi: 10.1016/j.biomaterials.2013.03.068. [DOI] [PubMed] [Google Scholar]
- 173.Dou Y. Sustained delivery by a cyclodextrin material-based nanocarrier potentiates antiatherosclerotic activity of rapamycin via selectively inhibiting mTORC1 in mice. J. Control. Release. 2016;235:48–62. doi: 10.1016/j.jconrel.2016.05.049. [DOI] [PubMed] [Google Scholar]
- 174.Tang Y. SiRNA crosslinked nanoparticles for the treatment of inflammation-induced liver injury. Adv. Sci. 2017;4:1600228. doi: 10.1002/advs.201600228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Huang D. Facile synthesis and self-assembly behaviour of pH-responsive degradable polyacetal dendrimers. Polym. Chem. 2016;7:6154–6158. [Google Scholar]
- 176.Wang Y.Q. Fabrication of zwitterionic and pH-responsive polyacetal dendrimers for anticancer drug delivery. Biomater. Sci. 2019;7:3238–3248. doi: 10.1039/c9bm00606k. [DOI] [PubMed] [Google Scholar]
- 177.Shenoi R.A. Branched multifunctional polyether polyketals: Variation of ketal group structure enables unprecedented control over polymer degradation in solution and within cells. J. Am. Chem. Soc. 2012;134:14945–14957. doi: 10.1021/ja305080f. [DOI] [PubMed] [Google Scholar]
- 178.Takemoto H. Polymeric modification of gemcitabine via cyclic acetal linkage for enhanced anticancer potency with negligible side effects. Biomaterials. 2020;235:119804. doi: 10.1016/j.biomaterials.2020.119804. [DOI] [PubMed] [Google Scholar]
- 179.Morinaga H., Morikawa H., Wang Y., Sudo A., Endo T. Amphiphilic copolymer having acid-labile acetal in the side chain as a hydrophobe: Controlled release of aldehyde by thermoresponsive aggregation−dissociation of polymer micelles. Macromolecules. 2009;42:2229–2235. [Google Scholar]
- 180.Griset A.P. Expansile nanoparticles: Synthesis, characterization, and in vivo efficacy of an acid-responsive polymeric drug delivery system. J. Am. Chem. Soc. 2009;131:2469–2471. doi: 10.1021/ja807416t. [DOI] [PubMed] [Google Scholar]
- 181.Sy J.C. Sustained release of a p38 inhibitor from non-inflammatory microspheres inhibits cardiac dysfunction. Nat. Mater. 2008;7:863–868. doi: 10.1038/nmat2299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Dou Y. Non-proinflammatory and responsive nanoplatforms for targeted treatment of atherosclerosis. Biomaterials. 2017;143:93–108. doi: 10.1016/j.biomaterials.2017.07.035. [DOI] [PubMed] [Google Scholar]
- 183.Shive M.S., Anderson J.M. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 1997;28:5–24. doi: 10.1016/s0169-409x(97)00048-3. [DOI] [PubMed] [Google Scholar]
- 184.Zhou J., Tsai Y.-T., Weng H., Baker D.W., Tang L. Real time monitoring of biomaterial-mediated inflammatory responses via macrophage-targeting NIR nanoprobes. Biomaterials. 2011;32:9383–9390. doi: 10.1016/j.biomaterials.2011.08.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Wahbeh J., Milkowski S. The use of hydrazones for biomedical applications. SLAS Technol. 2019;24:161–168. doi: 10.1177/2472630318822713. [DOI] [PubMed] [Google Scholar]
- 186.Ulrich S. Growing prospects of dynamic covalent chemistry in delivery applications. Acc. Chem. Res. 2019;52:510–519. doi: 10.1021/acs.accounts.8b00591. [DOI] [PubMed] [Google Scholar]
- 187.Bae Y., Fukushima S., Harada A., Kataoka K. Design of environment-sensitive supramolecular assemblies for intracellular drug delivery: Polymeric micelles that are responsive to intracellular pH change. Angew. Chem. Int. Ed. 2003;42:4640–4643. doi: 10.1002/anie.200250653. [DOI] [PubMed] [Google Scholar]
- 188.Li C. pH-sensitive polymeric micelles for targeted delivery to inflamed joints. J. Control. Release. 2017;246:133–141. doi: 10.1016/j.jconrel.2016.12.027. [DOI] [PubMed] [Google Scholar]
- 189.Wang D. Novel dexamethasone-HPMA copolymer conjugate and its potential application in treatment of rheumatoid arthritis. Arthritis Res. Ther. 2007;9:R2. doi: 10.1186/ar2106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Ren K. Macromolecular prodrug of dexamethasone prevents particle-induced peri-implant osteolysis with reduced systemic side effects. J. Control. Release. 2014;175:1–9. doi: 10.1016/j.jconrel.2013.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Ren K., Yuan H., Zhang Y., Wei X., Wang D. Macromolecular glucocorticoid prodrug improves the treatment of dextran sulfate sodium-induced mice ulcerative colitis. Clin. Immunol. 2015;160:71–81. doi: 10.1016/j.clim.2015.03.027. [DOI] [PubMed] [Google Scholar]
- 192.Li P. Synergistic effect of all-trans-retinal and triptolide encapsulated in an inflammation-targeted nanoparticle on collagen-induced arthritis in mice. J. Control. Release. 2020;319:87–103. doi: 10.1016/j.jconrel.2019.12.025. [DOI] [PubMed] [Google Scholar]
- 193.Hwang A.A. pH-responsive isoniazid-loaded nanoparticles markedly improve tuberculosis treatment in mice. Small. 2015;11:5066–5078. doi: 10.1002/smll.201500937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Xu F. High-throughput synthesis, analysis, and optimization of injectable hydrogels for protein delivery. Biomacromolecules. 2020;21:214–229. doi: 10.1021/acs.biomac.9b01132. [DOI] [PubMed] [Google Scholar]
- 195.Wang L.L. Injectable and protease-degradable hydrogel for siRNA sequestration and triggered delivery to the heart. J. Control. Release. 2018;285:152–161. doi: 10.1016/j.jconrel.2018.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Aldayel A.M. Acid-sensitive sheddable PEGylated PLGA nanoparticles increase the delivery of TNF-α siRNA in chronic inflammation sites. Mol. Ther. Nucleic Acid. 2016;5 doi: 10.1038/mtna.2016.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Sawant R.M. "Smart" drug delivery systems: Double-targeted pH-responsive pharmaceutical nanocarriers. Bioconjug. Chem. 2006;17:943–949. doi: 10.1021/bc060080h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Romberg B., Hennink W.E., Storm G. Sheddable coatings for long-circulating nanoparticles. Pharm. Res. 2008;25:55–71. doi: 10.1007/s11095-007-9348-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Oishi M., Nagasaki Y., Itaka K., Nishiyama N., Kataoka K. Lactosylated poly(ethylene glycol)-siRNA conjugate through acid-labile beta-thiopropionate linkage to construct pH-sensitive polyion complex micelles achieving enhanced gene silencing in hepatoma cells. J. Am. Chem. Soc. 2005;127:1624–1625. doi: 10.1021/ja044941d. [DOI] [PubMed] [Google Scholar]
- 200.Jeong J.H., Kim S.W., Park T.G. Novel intracellular delivery system of antisense oligonucleotide by self-assembled hybrid micelles composed of DNA/PEG conjugate and cationic fusogenic peptide. Bioconjug. Chem. 2003;14:473–479. doi: 10.1021/bc025632k. [DOI] [PubMed] [Google Scholar]
- 201.Yang L. Synthesis, nanosizing and in vitro drug release of a novel anti-HIV polymeric prodrug: Chitosan-O-isopropyl-5'-O-d4T monophosphate conjugate. Bioorg. Med. Chem. 2010;18:117–123. doi: 10.1016/j.bmc.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 202.Smith N. A long acting nanoformulated lamivudine protide. Biomaterials. 2019;223:119476. doi: 10.1016/j.biomaterials.2019.119476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Shin J., Shum P., Thompson D.H. Acid-triggered release via dePEGylation of DOPE liposomes containing acid-labile vinyl ether PEG-lipids. J. Control. Release. 2003;91:187–200. doi: 10.1016/s0168-3659(03)00232-3. [DOI] [PubMed] [Google Scholar]
- 204.Xu Z. A smart nanoassembly consisting of acid-labile vinyl ether PEG−DOPE and protamine for gene delivery: Preparation and in vitro transfection. Biomacromolecules. 2008;9:3119–3126. doi: 10.1021/bm800706f. [DOI] [PubMed] [Google Scholar]
- 205.Parrott M.C. Tunable bifunctional silyl ether cross-linkers for the design of acid-sensitive biomaterials. J. Am. Chem. Soc. 2010;132:17928–17932. doi: 10.1021/ja108568g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Li Y. Well-defined, reversible boronate crosslinked nanocarriers for targeted drug delivery in response to acidic pH values and cis-diols. Angew. Chem. Int. Ed. 2012;51:2864–2869. doi: 10.1002/anie.201107144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Wang Y. A nanoparticle-based strategy for the imaging of a broad range of tumours by nonlinear amplification of microenvironment signals. Nat. Mater. 2014;13:204–212. doi: 10.1038/nmat3819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Zhou K. Tunable, ultrasensitive pH-responsive nanoparticles targeting specific endocytic organelles in living cells. Angew. Chem. Int. Ed. 2011;50:6109–6114. doi: 10.1002/anie.201100884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Qi T. A pH-activatable nanoparticle for dual-stage precisely mitochondria-targeted photodynamic anticancer therapy. Biomaterials. 2019;213:12. doi: 10.1016/j.biomaterials.2019.05.030. [DOI] [PubMed] [Google Scholar]
- 210.Luo M. A sting-activating nanovaccine for cancer immunotherapy. Nat. Nanotechnol. 2017;12:648–654. doi: 10.1038/nnano.2017.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Yik-Sham Chung C., Timblin G.A., Saijo K., Chang C.J. Versatile histochemical approach to detection of hydrogen peroxide in cells and tissues based on puromycin staining. J. Am. Chem. Soc. 2018;140:6109–6121. doi: 10.1021/jacs.8b02279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Kim E.J. An activatable prodrug for the treatment of metastatic tumors. J. Am. Chem. Soc. 2014;136:13888–13894. doi: 10.1021/ja5077684. [DOI] [PubMed] [Google Scholar]
- 213.Broaders K.E., Grandhe S., Fréchet J.M.J. A biocompatible oxidation-triggered carrier polymer with potential in therapeutics. J. Am. Chem. Soc. 2011;133:756–758. doi: 10.1021/ja110468v. [DOI] [PubMed] [Google Scholar]
- 214.Zhang D. Biocompatible reactive oxygen species (ROS)-responsive nanoparticles as superior drug delivery vehicles. Adv. Healthc. Mater. 2015;4:69–76. doi: 10.1002/adhm.201400299. [DOI] [PubMed] [Google Scholar]
- 215.Zhang Q. Structure–property correlations of reactive oxygen species-responsive and hydrogen peroxide-eliminating materials with anti-oxidant and anti-inflammatory activities. Chem. Mater. 2017;29:8221–8238. [Google Scholar]
- 216.Liu X. Fusogenic reactive oxygen species triggered charge-reversal vector for effective gene delivery. Adv. Mater. 2016;28:1743–1752. doi: 10.1002/adma.201504288. [DOI] [PubMed] [Google Scholar]
- 217.Zheng M. ROS-responsive polymeric siRNA nanomedicine stabilized by triple interactions for the robust glioblastoma combinational RNAi therapy. Adv. Mater. 2019;31:1903277. doi: 10.1002/adma.201903277. [DOI] [PubMed] [Google Scholar]
- 218.Zhu D. Intracellularly disintegratable polysulfoniums for efficient gene delivery. Adv. Funct. Mater. 2017;27:1606826. [Google Scholar]
- 219.de Gracia Lux C. Biocompatible polymeric nanoparticles degrade and release cargo in response to biologically relevant levels of hydrogen peroxide. J. Am. Chem. Soc. 2012;134:15758–15764. doi: 10.1021/ja303372u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Lin L. Nanodrug with ROS and pH dual-sensitivity ameliorates liver fibrosis via multicellular regulation. Adv. Sci. 2020;7:1903138. doi: 10.1002/advs.201903138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Song C.-C., Ji R., Du F.-S., Liang D.-H., Li Z.-C. Oxidation-accelerated hydrolysis of the ortho ester-containing acid-labile polymers. ACS Macro Lett. 2013;2:273–277. doi: 10.1021/mz4000392. [DOI] [PubMed] [Google Scholar]
- 222.Lee D. In vivo imaging of hydrogen peroxide with chemiluminescent nanoparticles. Nat. Mater. 2007;6:765–769. doi: 10.1038/nmat1983. [DOI] [PubMed] [Google Scholar]
- 223.Park H. Antioxidant and anti-inflammatory activities of hydroxybenzyl alcohol releasing biodegradable polyoxalate nanoparticles. Biomacromolecules. 2010;11:2103–2108. doi: 10.1021/bm100474w. [DOI] [PubMed] [Google Scholar]
- 224.Kim S. Reduction of oxidative stress by p-hydroxybenzyl alcohol-containing biodegradable polyoxalate nanoparticulate antioxidant. Biomaterials. 2011;32:3021–3029. doi: 10.1016/j.biomaterials.2010.11.033. [DOI] [PubMed] [Google Scholar]
- 225.Lee D. Hydrogen peroxide-responsive copolyoxalate nanoparticles for detection and therapy of ischemia-reperfusion injury. J. Control. Release. 2013;172:1102–1110. doi: 10.1016/j.jconrel.2013.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Cho B.R. P-hydroxybenzyl alcohol-containing biodegradable nanoparticle improves functional blood flow through angiogenesis in a mouse model of hindlimb ischemia. Biomaterials. 2015;53:679–687. doi: 10.1016/j.biomaterials.2015.02.107. [DOI] [PubMed] [Google Scholar]
- 227.Lee D. H2O2-responsive molecularly engineered polymer nanoparticles as ischemia/reperfusion-targeted nanotherapeutic agents. Sci. Rep. 2013;3:2233. doi: 10.1038/srep02233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Ko E. Antioxidant polymeric prodrug microparticles as a therapeutic system for acute liver failure. Biomaterials. 2014;35:3895–3902. doi: 10.1016/j.biomaterials.2014.01.048. [DOI] [PubMed] [Google Scholar]
- 229.Park S. Therapeutic use of H2O2-responsive anti-oxidant polymer nanoparticles for doxorubicin-induced cardiomyopathy. Biomaterials. 2014;35:5944–5953. doi: 10.1016/j.biomaterials.2014.03.084. [DOI] [PubMed] [Google Scholar]
- 230.Shukla A.K., Verma M., Singh K.N. Superoxide induced deprotection of 1,3-dithiolanes: A convenient method of dedithioacetalization. Indian J. Chem. B. 2004;43:1748–1752. [Google Scholar]
- 231.Wilson D.S. Orally delivered thioketal nanoparticles loaded with TNF-α-siRNA target inflammation and inhibit gene expression in the intestines. Nat. Mater. 2010;9:923–928. doi: 10.1038/nmat2859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Regmi S. Inflammation-triggered local drug release ameliorates colitis by inhibiting dendritic cell migration and Th1/Th17 differentiation. J. Control. Release. 2019;316:138–149. doi: 10.1016/j.jconrel.2019.11.001. [DOI] [PubMed] [Google Scholar]
- 233.Yao Y. ROS-responsive polyurethane fibrous patches loaded with methylprednisolone (MP) for restoring structures and functions of infarcted myocardium in vivo. Biomaterials. 2020;232:119726. doi: 10.1016/j.biomaterials.2019.119726. [DOI] [PubMed] [Google Scholar]
- 234.Li J. Photoinduced PEG deshielding from ROS-sensitive linkage-bridged block copolymer-based nanocarriers for on-demand drug delivery. Biomaterials. 2018;170:147–155. doi: 10.1016/j.biomaterials.2018.04.015. [DOI] [PubMed] [Google Scholar]
- 235.Shim M.S., Xia Y. A reactive oxygen species (ROS)-responsive polymer for safe, efficient, and targeted gene delivery in cancer cells. Angew. Chem. Int. Ed. 2013;52:6926–6929. doi: 10.1002/anie.201209633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Wang J., He X., Shen S., Cao Z., Yang X. ROS-sensitive cross-linked polyethylenimine for red-light-activated siRNA therapy. ACS Appl. Mater. Interfaces. 2019;11:1855–1863. doi: 10.1021/acsami.8b18697. [DOI] [PubMed] [Google Scholar]
- 237.Ke W. Length effect of stimuli-responsive block copolymer prodrug filomicelles on drug delivery efficiency. J. Control. Release. 2020;318:67–77. doi: 10.1016/j.jconrel.2019.12.012. [DOI] [PubMed] [Google Scholar]
- 238.Wang S. Enhanced antitumor efficacy by a cascade of reactive oxygen species generation and drug release. Angew. Chem. Int. Ed. 2019;58:14758–14763. doi: 10.1002/anie.201908997. [DOI] [PubMed] [Google Scholar]
- 239.Wang M. High co-loading capacity and stimuli-responsive release based on cascade reaction of self-destructive polymer for improved chemo-photodynamic therapy. ACS Nano. 2019;13:7010–7023. doi: 10.1021/acsnano.9b02096. [DOI] [PubMed] [Google Scholar]
- 240.Chen D. Biodegradable, hydrogen peroxide, and glutathione dual responsive nanoparticles for potential programmable paclitaxel release. J. Am. Chem. Soc. 2018;140:7373–7376. doi: 10.1021/jacs.7b12025. [DOI] [PubMed] [Google Scholar]
- 241.Napoli A., Valentini M., Tirelli N., Müller M., Hubbell J.A. Oxidation-responsive polymeric vesicles. Nat. Mater. 2004;3:183–189. doi: 10.1038/nmat1081. [DOI] [PubMed] [Google Scholar]
- 242.Gupta M.K., Meyer T.A., Nelson C.E., Duvall C.L. Poly(PS-b-DMA) micelles for reactive oxygen species triggered drug release. J. Control. Release. 2012;162:591–598. doi: 10.1016/j.jconrel.2012.07.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Rehor A., Hubbell J.A., Tirelli N. Oxidation-sensitive polymeric nanoparticles. Langmuir. 2005;21:411–417. doi: 10.1021/la0478043. [DOI] [PubMed] [Google Scholar]
- 244.Allen B.L., Johnson J.D., Walker J.P. Encapsulation and enzyme-mediated release of molecular cargo in polysulfide nanoparticles. ACS Nano. 2011;5:5263–5272. doi: 10.1021/nn201477y. [DOI] [PubMed] [Google Scholar]
- 245.Poole K.M. ROS-responsive microspheres for on demand antioxidant therapy in a model of diabetic peripheral arterial disease. Biomaterials. 2015;41:166–175. doi: 10.1016/j.biomaterials.2014.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Gupta M.K. Cell protective, ABC triblock polymer-based thermoresponsive hydrogels with ROS-triggered degradation and drug release. J. Am. Chem. Soc. 2014;136:14896–14902. doi: 10.1021/ja507626y. [DOI] [PubMed] [Google Scholar]
- 247.Li L. Ginsenoside Rg3-loaded, reactive oxygen species-responsive polymeric nanoparticles for alleviating myocardial ischemia-reperfusion injury. J. Control. Release. 2020;317:259–272. doi: 10.1016/j.jconrel.2019.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Allen S.D. Celastrol-loaded PEG-b-PPS nanocarriers as an anti-inflammatory treatment for atherosclerosis. Biomater. Sci. 2019;7:657–668. doi: 10.1039/c8bm01224e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Wu T. Aortic plaque-targeted andrographolide delivery with oxidation-sensitive micelle effectively treats atherosclerosis via simultaneous ROS capture and anti-inflammation. Nanomedicine. 2018;14:2215–2226. doi: 10.1016/j.nano.2018.06.010. [DOI] [PubMed] [Google Scholar]
- 250.Dollinger B.R., Gupta M.K., Martin J.R., Duvall C.L. Reactive oxygen species shielding hydrogel for the delivery of adherent and nonadherent therapeutic cell types. Tissue Eng. Part A. 2017;23:1120–1131. doi: 10.1089/ten.tea.2016.0495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Ma N., Li Y., Xu H., Wang Z., Zhang X. Dual redox responsive assemblies formed from diselenide block copolymers. J. Am. Chem. Soc. 2010;132:442–443. doi: 10.1021/ja908124g. [DOI] [PubMed] [Google Scholar]
- 252.Ma N. Selenium-containing block copolymers and their oxidation-responsive aggregates. Polym. Chem. 2010;1:1609–1614. [Google Scholar]
- 253.Xu H., Cao W., Zhang X. Selenium-containing polymers: Promising biomaterials for controlled release and enzyme mimics. Acc. Chem. Res. 2013;46:1647–1658. doi: 10.1021/ar4000339. [DOI] [PubMed] [Google Scholar]
- 254.Fan Z., Xu H. Recent progress in the biological applications of reactive oxygen species-responsive polymers. Polym. Rev. 2020;60:114–143. [Google Scholar]
- 255.Cao W., Wang L., Xu H. Selenium/tellurium containing polymer materials in nanobiotechnology. Nano Today. 2015;10:717–736. [Google Scholar]
- 256.Hu Q., Katti P.S., Gu Z. Enzyme-responsive nanomaterials for controlled drug delivery. Nanoscale. 2014;6:12273–12286. doi: 10.1039/c4nr04249b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Shahriari M. Enzyme responsive drug delivery systems in cancer treatment. J. Control. Release. 2019;308:172–189. doi: 10.1016/j.jconrel.2019.07.004. [DOI] [PubMed] [Google Scholar]
- 258.Mu J., Lin J., Huang P., Chen X.Y. Development of endogenous enzyme-responsive nanomaterials for theranostics. Chem. Soc. Rev. 2018;47:5554–5573. doi: 10.1039/c7cs00663b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Aimetti A.A., Tibbitt M.W., Anseth K.S. Human neutrophil elastase responsive delivery from poly(ethylene glycol) hydrogels. Biomacromolecules. 2009;10:1484–1489. doi: 10.1021/bm9000926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Homma A. Synthesis and optimization of hyaluronic acid-methotrexate conjugates to maximize benefit in the treatment of osteoarthritis. Bioorg. Med. Chem. 2010;18:1062–1075. doi: 10.1016/j.bmc.2009.12.053. [DOI] [PubMed] [Google Scholar]
- 261.Lutolf M.P. Synthetic matrix metalloproteinase-sensitive hydrogels for the conduction of tissue regeneration: Engineering cell-invasion characteristics. Proc. Natl. Acad. Sci. U. S. A. 2003;100:5413. doi: 10.1073/pnas.0737381100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Purcell B.P. Injectable and bioresponsive hydrogels for on-demand matrix metalloproteinase inhibition. Nat. Mater. 2014;13:653–661. doi: 10.1038/nmat3922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Lutolf M.P. Repair of bone defects using synthetic mimetics of collagenous extracellular matrices. Nat. Biotechnol. 2003;21:513–518. doi: 10.1038/nbt818. [DOI] [PubMed] [Google Scholar]
- 264.Huang J. Core–shell distinct nanodrug showing on-demand sequential drug release to act on multiple cell types for synergistic anticancer therapy. ACS Nano. 2019;13:7036–7049. doi: 10.1021/acsnano.9b02149. [DOI] [PubMed] [Google Scholar]
- 265.Cheng K. Sequentially responsive therapeutic peptide assembling nanoparticles for dual-targeted cancer immunotherapy. Nano Lett. 2018;18:3250–3258. doi: 10.1021/acs.nanolett.8b01071. [DOI] [PubMed] [Google Scholar]
- 266.Fan C. Myocardial-infarction-responsive smart hydrogels targeting matrix metalloproteinase for on-demand growth factor delivery. Adv. Mater. 2019;31:1902900. doi: 10.1002/adma.201902900. [DOI] [PubMed] [Google Scholar]
- 267.Biswas A. Endoprotease-mediated intracellular protein delivery using nanocapsules. ACS Nano. 2011;5:1385–1394. doi: 10.1021/nn1031005. [DOI] [PubMed] [Google Scholar]
- 268.Jiang T. Furin-mediated sequential delivery of anticancer cytokine and small-molecule drug shuttled by graphene. Adv. Mater. 2015;27:1021–1028. doi: 10.1002/adma.201404498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Kang J.-H. Design of polymeric carriers for cancer-specific gene targeting: Utilization of abnormal protein kinase Cα activation in cancer cells. J. Am. Chem. Soc. 2008;130:14906–14907. doi: 10.1021/ja805364s. [DOI] [PubMed] [Google Scholar]
- 270.Gu Z. Protein nanocapsule weaved with enzymatically degradable polymeric network. Nano Lett. 2009;9:4533–4538. doi: 10.1021/nl902935b. [DOI] [PubMed] [Google Scholar]
- 271.Guo J. Cyclodextrin-derived intrinsically bioactive nanoparticles for treatment of acute and chronic inflammatory diseases. Adv. Mater. 2019;31:1904607. doi: 10.1002/adma.201904607. [DOI] [PubMed] [Google Scholar]
- 272.Xiao B. Mannosylated bioreducible nanoparticle-mediated macrophage-specific TNF-α RNA interference for IBD therapy. Biomaterials. 2013;34:7471–7482. doi: 10.1016/j.biomaterials.2013.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Ain U.Q., Chung H., Chung J.Y., Choi J.-H., Kim Y.-H. Amelioration of atherosclerotic inflammation and plaques via endothelial adrenoceptor-targeted eNOS gene delivery using redox-sensitive polymer bearing l-arginine. J. Control. Release. 2017;262:72–86. doi: 10.1016/j.jconrel.2017.07.019. [DOI] [PubMed] [Google Scholar]
- 274.Sun Q. Redox-sensitive nanoparticles based on 4-aminothiophenol-carboxymethyl inulin conjugate for budesonide delivery in inflammatory bowel diseases. Carbohyd. Polym. 2018;189:352–359. doi: 10.1016/j.carbpol.2017.12.021. [DOI] [PubMed] [Google Scholar]
- 275.Mavuso S. A dual pH/redox responsive copper-ligand nanoliposome bioactive complex for the treatment of chronic inflammation. Int. J. Pharm. 2016;509:348–359. doi: 10.1016/j.ijpharm.2016.05.069. [DOI] [PubMed] [Google Scholar]
- 276.Shirazi L.F., Bissett J., Romeo F., Mehta J.L. Role of inflammation in heart failure. Curr. Atheroscler. Rep. 2017;19:27. doi: 10.1007/s11883-017-0660-3. [DOI] [PubMed] [Google Scholar]
- 277.Thygesen K. Fourth universal definition of myocardial infarction (2018) Eur. Heart J. 2019;40:237–269. doi: 10.1093/eurheartj/ehy462. [DOI] [PubMed] [Google Scholar]
- 278.Lozano O., Torres-Quintanilla A., García-Rivas G. Nanomedicine for the cardiac myocyte: Where are we? J. Control. Release. 2018;271:149–165. doi: 10.1016/j.jconrel.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 279.Ibrahim A.G.E. Augmenting canonical Wnt signalling in therapeutically inert cells converts them into therapeutically potent exosome factories. Nat. Biomed. Eng. 2019;3:695–705. doi: 10.1038/s41551-019-0448-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Liu B. Cardiac recovery via extended cell-free delivery of extracellular vesicles secreted by cardiomyocytes derived from induced pluripotent stem cells. Nat. Biomed. Eng. 2018;2:293–303. doi: 10.1038/s41551-018-0229-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Evans C.W., Iyer K.S., Hool L.C. The potential for nanotechnology to improve delivery of therapy to the acute ischemic heart. Nanomedicine. 2016;11:817–832. doi: 10.2217/nnm.16.7. [DOI] [PubMed] [Google Scholar]
- 282.Guan S., Li J., Zhang K., Li J. Environmentally responsive hydrogels for repair of cardiovascular tissue. Heart Fail. Rev. 2020 doi: 10.1007/s10741-10020-09934-y. [DOI] [PubMed] [Google Scholar]
- 283.Ziegler M. A self-assembled fluorescent nanoprobe for imaging and therapy of cardiac ischemia/reperfusion injury. Adv. Ther. 2019;2:1800133. [Google Scholar]
- 284.Suarez S.L. Degradable acetalated dextran microparticles for tunable release of an engineered hepatocyte growth factor fragment. ACS Biomater. Sci. Eng. 2016;2:197–204. doi: 10.1021/acsbiomaterials.5b00335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Torrieri G. Dual-peptide functionalized acetalated dextran-based nanoparticles for sequential targeting of macrophages during myocardial infarction. Nanoscale. 2020;12:2350–2358. doi: 10.1039/c9nr09934d. [DOI] [PubMed] [Google Scholar]
- 286.Garbern J.C., Minami E., Stayton P.S., Murry C.E. Delivery of basic fibroblast growth factor with a pH-responsive, injectable hydrogel to improve angiogenesis in infarcted myocardium. Biomaterials. 2011;32:2407–2416. doi: 10.1016/j.biomaterials.2010.11.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Tan S.Y. Responsive mesoporous silica nanoparticles for sensing of hydrogen peroxide and simultaneous treatment toward heart failure. Nanoscale. 2017;9:2253–2261. doi: 10.1039/c6nr08869d. [DOI] [PubMed] [Google Scholar]
- 288.Nguyen M.M. Enzyme-responsive nanoparticles for targeted accumulation and prolonged retention in heart tissue after myocardial infarction. Adv. Mater. 2015;27:5547–5552. doi: 10.1002/adma.201502003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Carlini A.S. Enzyme-responsive progelator cyclic peptides for minimally invasive delivery to the heart post-myocardial infarction. Nat. Commun. 2019;10:1735. doi: 10.1038/s41467-019-09587-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Mizgerd J.P. Acute lower respiratory tract infection. N. Engl. J. Med. 2008;358:716–727. doi: 10.1056/NEJMra074111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Rubenfeld G.D., Herridge M.S. Epidemiology and outcomes of acute lung injury. Chest. 2007;131:554–562. doi: 10.1378/chest.06-1976. [DOI] [PubMed] [Google Scholar]
- 292.Wu F. A new coronavirus associated with human respiratory disease in china. Nature. 2020;579:265–269. doi: 10.1038/s41586-020-2008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Kuzmov A., Minko T. Nanotechnology approaches for inhalation treatment of lung diseases. J. Control. Release. 2015;219:500–518. doi: 10.1016/j.jconrel.2015.07.024. [DOI] [PubMed] [Google Scholar]
- 294.Patton J.S., Byron P.R. Inhaling medicines: Delivering drugs to the body through the lungs. Nat. Rev. Drug Discov. 2007;6:67–74. doi: 10.1038/nrd2153. [DOI] [PubMed] [Google Scholar]
- 295.El-Sherbiny I.M., Elbaz N.M., Sedki M., Elgammal A., Yacoub M.H. Magnetic nanoparticles-based drug and gene delivery systems for the treatment of pulmonary diseases. Nanomedicine (London, England) 2017;12:387–402. doi: 10.2217/nnm-2016-0341. [DOI] [PubMed] [Google Scholar]
- 296.Sadikot R.T., Kolanjiyil A.V., Kleinstreuer C., Rubinstein I. Nanomedicine for treatment of acute lung injury and acute respiratory distress syndrome. Biomed. Hub. 2017;2:1–12. doi: 10.1159/000477086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Park H.S., Kim S.R., Lee Y.C. Impact of oxidative stress on lung diseases. Respirology. 2009;14:27–38. doi: 10.1111/j.1440-1843.2008.01447.x. [DOI] [PubMed] [Google Scholar]
- 298.Li L. A broad-spectrum ROS-eliminating material for prevention of inflammation and drug-induced organ toxicity. Adv. Sci. 2018;5:1800781. doi: 10.1002/advs.201800781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Kallet R., Jasmer R., Luce J., Lin L., Marks J. The treatment of acidosis in acute lung injury with tris-hydroxymethyl aminomethane (tham) Am. J. Respir. Crit. Care Med. 2000;161:1149–1153. doi: 10.1164/ajrccm.161.4.9906031. [DOI] [PubMed] [Google Scholar]
- 300.Xiu Z.-m., Zhang Q.-b., Puppala H.L., Colvin V.L., Alvarez P.J.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012;12:4271–4275. doi: 10.1021/nl301934w. [DOI] [PubMed] [Google Scholar]
- 301.Zhang C.Y. pH-responsive nanoparticles targeted to lungs for improved therapy of acute lung inflammation/injury. ACS Appl. Mater. Interfaces. 2019;11:16380–16390. doi: 10.1021/acsami.9b04051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Zhang C.Y., Gao J., Wang Z. Bioresponsive nanoparticles targeted to infectious microenvironments for sepsis management. Adv. Mater. 2018;30:1803618. doi: 10.1002/adma.201803618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Chawla L.S., Eggers P.W., Star R.A., Kimmel P.L. Acute kidney injury and chronic kidney disease as interconnected syndromes. N. Engl. J. Med. 2014;371:58–66. doi: 10.1056/NEJMra1214243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Fu Y., Lin Q., Gong T., Sun X., Zhang Z.R. Renal-targeting triptolide-glucosamine conjugate exhibits lower toxicity and superior efficacy in attenuation of ischemia/reperfusion renal injury in rats. Acta Pharmacol. Sin. 2016;37:1467–1480. doi: 10.1038/aps.2016.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Hu J.B. E-selectin-targeted sialic acid-PEG-dexamethasone micelles for enhanced anti-inflammatory efficacy for acute kidney injury. Theranostics. 2017;7:2204–2219. doi: 10.7150/thno.19571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Yu H. Size and temporal-dependent efficacy of oltipraz-loaded PLGA nanoparticles for treatment of acute kidney injury and fibrosis. Biomaterials. 2019;219:119368. doi: 10.1016/j.biomaterials.2019.119368. [DOI] [PubMed] [Google Scholar]
- 307.Zandstra J. Microsphere-based rapamycin delivery, systemic versus local administration in a rat model of renal ischemia/reperfusion injury. Pharm. Res. 2015;32:3238–3247. doi: 10.1007/s11095-015-1700-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Zhao M. Control release of mitochondria-targeted antioxidant by injectable self-assembling peptide hydrogel ameliorated persistent mitochondrial dysfunction and inflammation after acute kidney injury. Drug Deliv. 2018;25:546–554. doi: 10.1080/10717544.2018.1440445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Liu D. Enhanced efficiency of mitochondria-targeted peptide SS-31 for acute kidney injury by pH-responsive and AKI-kidney targeted nanopolyplexes. Biomaterials. 2019;211:57–67. doi: 10.1016/j.biomaterials.2019.04.034. [DOI] [PubMed] [Google Scholar]
- 310.Yu H. ROS-responsive nano-drug delivery system combining mitochondria-targeting ceria nanoparticles with atorvastatin for acute kidney injury. Theranostics. 2020;10:2342–2357. doi: 10.7150/thno.40395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Hu J.B. An E-selectin targeting and MMP-2-responsive dextran-curcumin polymeric prodrug for targeted therapy of acute kidney injury. Biomater. Sci. 2018;6:3397–3409. doi: 10.1039/c8bm00813b. [DOI] [PubMed] [Google Scholar]
- 312.Choi C.H.J., Zuckerman J.E., Webster P., Davis M.E. Targeting kidney mesangium by nanoparticles of defined size. Proc. Natl. Acad. Sci. U. S. A. 2011;108:6656. doi: 10.1073/pnas.1103573108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Wang F.S., Fan J.G., Zhang Z., Gao B., Wang H.Y. The global burden of liver disease: The major impact of china. Hepatology. 2014;60:2099–2108. doi: 10.1002/hep.27406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Go Y. Acid-triggered echogenic nanoparticles for contrast-enhanced ultrasound imaging and therapy of acute liver failure. Biomaterials. 2018;186:22–30. doi: 10.1016/j.biomaterials.2018.09.034. [DOI] [PubMed] [Google Scholar]
- 315.Liu Z. Multifunctional nanohybrid based on porous silicon nanoparticles, gold nanoparticles, and acetalated dextran for liver regeneration and acute liver failure theranostics. Adv. Mater. 2018;30:1703393. doi: 10.1002/adma.201703393. [DOI] [PubMed] [Google Scholar]
- 316.Chen G. Reactive oxygen species-responsive polymeric nanoparticles for alleviating sepsis-induced acute liver injury in mice. Biomaterials. 2017;144:30–41. doi: 10.1016/j.biomaterials.2017.08.008. [DOI] [PubMed] [Google Scholar]
- 317.Zhang W. Macrophage-targeting and reactive oxygen species (ROS)-responsive nanopolyplexes mediate anti-inflammatory siRNA delivery against acute liver failure (ALF) Biomater. Sci. 2018;6:1986–1993. doi: 10.1039/c8bm00389k. [DOI] [PubMed] [Google Scholar]
- 318.Bhattacharyya A., Chattopadhyay R., Mitra S., Crowe S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014;94:329–354. doi: 10.1152/physrev.00040.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Zhang Q.X. A superoxide dismutase/catalase mimetic nanomedicine for targeted therapy of inflammatory bowel disease. Biomaterials. 2016;105:206–221. doi: 10.1016/j.biomaterials.2016.08.010. [DOI] [PubMed] [Google Scholar]
- 320.Zhang Q. A multifunctional nanotherapy for targeted treatment of colon cancer by simultaneously regulating tumor microenvironment. Theranostics. 2019;9:3732–3753. doi: 10.7150/thno.34377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Li C. A proresolving peptide nanotherapy for site-specific treatment of inflammatory bowel disease by regulating proinflammatory microenvironment and gut microbiota. Adv. Sci. 2019;6:1900610. doi: 10.1002/advs.201900610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Karrout Y. In vivo efficacy of microbiota-sensitive coatings for colon targeting: A promising tool for IBD therapy. J. Control. Release. 2015;197:121–130. doi: 10.1016/j.jconrel.2014.11.006. [DOI] [PubMed] [Google Scholar]
- 323.Gou S. Multi-bioresponsive silk fibroin-based nanoparticles with on-demand cytoplasmic drug release capacity for CD44-targeted alleviation of ulcerative colitis. Biomaterials. 2019;212:39–54. doi: 10.1016/j.biomaterials.2019.05.012. [DOI] [PubMed] [Google Scholar]
- 324.Knipe J.M., Strong L.E., Peppas N.A. Enzyme- and pH-responsive microencapsulated nanogels for oral delivery of siRNA to induce TNF-α knockdown in the intestine. Biomacromolecules. 2016;17:788–797. doi: 10.1021/acs.biomac.5b01518. [DOI] [PubMed] [Google Scholar]
- 325.Bertoni S. pH and reactive oxygen species-sequential responsive nano-in-micro composite for targeted therapy of inflammatory bowel disease. Adv. Funct. Mater. 2018;28:1806175. [Google Scholar]
- 326.Wang Y. Targeted therapy of atherosclerosis by a broad-spectrum reactive oxygen species scavenging nanoparticle with intrinsic anti-inflammatory activity. ACS Nano. 2018;12:8943–8960. doi: 10.1021/acsnano.8b02037. [DOI] [PubMed] [Google Scholar]
- 327.Gao C. Treatment of atherosclerosis by macrophage-biomimetic nanoparticles via targeted pharmacotherapy and sequestration of proinflammatory cytokines. Nat. Commun. 2020;11:2622. doi: 10.1038/s41467-020-16439-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Li C. Site-specific microRNA-33 antagonism by pH-responsive nanotherapies for treatment of atherosclerosis via regulating cholesterol efflux and adaptive immunity. Adv. Funct. Mater. 2020 doi: 10.1002/adfm.202002131. 2002131. [DOI] [Google Scholar]
- 329.Cheng J. A targeting nanotherapy for abdominal aortic aneurysms. J. Am. Coll. Cardiol. 2018;72:2591–2605. doi: 10.1016/j.jacc.2018.08.2188. [DOI] [PubMed] [Google Scholar]
- 330.Kang C. Fibrin-targeted and H2O2-responsive nanoparticles as a theranostics for thrombosed vessels. ACS Nano. 2017;11:6194–6203. doi: 10.1021/acsnano.7b02308. [DOI] [PubMed] [Google Scholar]
- 331.Xu J. Sequentially site-specific delivery of thrombolytics and neuroprotectant for enhanced treatment of ischemic stroke. ACS Nano. 2019;13:8577–8588. doi: 10.1021/acsnano.9b01798. [DOI] [PubMed] [Google Scholar]
- 332.Guo X. Thrombin-responsive, brain-targeting nanoparticles for improved stroke therapy. ACS Nano. 2018;12:8723–8732. doi: 10.1021/acsnano.8b04787. [DOI] [PubMed] [Google Scholar]
- 333.Mei T. Encapsulation of tissue plasminogen activator in pH-sensitive self-assembled antioxidant nanoparticles for ischemic stroke treatment-synergistic effect of thrombolysis and antioxidant. Biomaterials. 2019;215:119209. doi: 10.1016/j.biomaterials.2019.05.020. [DOI] [PubMed] [Google Scholar]
- 334.Lu Y. Microthrombus-targeting micelles for neurovascular remodeling and enhanced microcirculatory perfusion in acute ischemic stroke. Adv. Mater. 2019;31:1808361. doi: 10.1002/adma.201808361. [DOI] [PubMed] [Google Scholar]
- 335.Quan L. The evaluation of the therapeutic efficacy and side effects of a macromolecular dexamethasone prodrug in the collagen-induced arthritis mouse model. Pharm. Res. 2016;33:186–193. doi: 10.1007/s11095-015-1776-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 336.Zhao J. Multifunctional folate receptor-targeting and pH-responsive nanocarriers loaded with methotrexate for treatment of rheumatoid arthritis. Int. J. Nanomedicine. 2017;12:6735–6746. doi: 10.2147/IJN.S140992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Alam M.M. Endogenous inspired biomineral-installed hyaluronan nanoparticles as pH-responsive carrier of methotrexate for rheumatoid arthritis. J. Control. Release. 2017;252:62–72. doi: 10.1016/j.jconrel.2017.03.012. [DOI] [PubMed] [Google Scholar]
- 338.Fan X.-x. ROS-responsive berberine polymeric micelles effectively suppressed the inflammation of rheumatoid arthritis by targeting mitochondria. Nano-Micro Lett. 2020;12:76. doi: 10.1007/s40820-020-0410-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Chen M.L. Folate receptor-targeting and reactive oxygen species-responsive liposomal formulation of methotrexate for treatment of rheumatoid arthritis. Pharmaceutics. 2019;11:23. doi: 10.3390/pharmaceutics11110582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Ni R.R. Reactive oxygen species-responsive dexamethasone-loaded nanoparticles for targeted treatment of rheumatoid arthritis via suppressing the iRhom2/TNF-alpha/BAFF signaling pathway. Biomaterials. 2020;232:119730. doi: 10.1016/j.biomaterials.2019.119730. [DOI] [PubMed] [Google Scholar]
- 341.Joshi N. Towards an arthritis flare-responsive drug delivery system. Nat. Commun. 2018;9:1275. doi: 10.1038/s41467-018-03691-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Lu Y. Microenvironment remodeling micelles for Alzheimer's disease therapy by early modulation of activated microglia. Adv. Sci. 2019;6:1801586. doi: 10.1002/advs.201801586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Yoo D. Antioxidant polymeric nanoparticles as novel therapeutics for airway inflammatory diseases. Int. J. Pharm. 2013;450:87–94. doi: 10.1016/j.ijpharm.2013.04.028. [DOI] [PubMed] [Google Scholar]
- 344.Mejías J.C., Roy K. In-vitro and in-vivo characterization of a multi-stage enzyme-responsive nanoparticle-in-microgel pulmonary drug delivery system. J. Control. Release. 2019;316:393–403. doi: 10.1016/j.jconrel.2019.09.012. [DOI] [PubMed] [Google Scholar]
- 345.Wang Y. Multifunctional supramolecular hydrogel for prevention of epidural adhesion after laminectomy. ACS Nano. 2020;14:8202–8219. doi: 10.1021/acsnano.0c01658. [DOI] [PubMed] [Google Scholar]
- 346.Qiao Z.Y., Du F.S., Zhang R., Liang D.H., Li Z.C. Biocompatible thermoresponsive polymers with pendent oligo(ethylene glycol) chains and cyclic ortho ester groups. Macromolecules. 2010;43:6485–6494. [Google Scholar]
- 347.Song C.C. Toward tertiary amine-modulated acid-triggered hydrolysis of copolymers containing pendent ortho ester groups. Macromolecules. 2013;46:1093–1100. [Google Scholar]
- 348.Li S. Carboxymethyl chitosan-based nanogels via acid-labile ortho ester linkages mediated enhanced drug delivery. Int. J. Biol. Macromol. 2019;129:477–487. doi: 10.1016/j.ijbiomac.2019.02.072. [DOI] [PubMed] [Google Scholar]
- 349.Illy N. pH-sensitive poly(ethylene glycol)/poly(ethoxyethyl glycidyl ether) block copolymers: Synthesis, characterization, encapsulation, and delivery of a hydrophobic drug. Macromol. Chem. Phys. 2019;220:1900210. [Google Scholar]
- 350.Jung J., Lee I.-H., Lee E., Park J., Jon S. pH-sensitive polymer nanospheres for use as a potential drug delivery vehicle. Biomacromolecules. 2007;8:3401–3407. doi: 10.1021/bm700517z. [DOI] [PubMed] [Google Scholar]
- 351.Lee S.M., Cho J.H., Lee S.D., Kim Y.C. Nanoparticle-encapsulated p2x₇ receptor antagonist in a pH-sensitive polymer as a potential local drug delivery system to acidic inflammatory environments. Bioorg. Med. Chem. Lett. 2015;25:4197–4202. doi: 10.1016/j.bmcl.2015.08.004. [DOI] [PubMed] [Google Scholar]
- 352.Lu J. Acetals moiety contained pH-sensitive amphiphilic copolymer self-assembly used for drug carrier. Polymer. 2010;51:1709–1715. [Google Scholar]
- 353.Oishi M., Nagatsugi F., Sasaki S., Nagasaki Y., Kataoka K. Smart polyion complex micelles for targeted intracellular delivery of PEGylated antisense oligonucleotides containing acid-labile linkages. ChemBioChem. 2005;6:718–725. doi: 10.1002/cbic.200400334. [DOI] [PubMed] [Google Scholar]
- 354.Oishi M., Sasaki S., Nagasaki Y., Kataoka K. pH-responsive oligodeoxynucleotide (ODN)−poly(ethylene glycol) conjugate through acid-labile β-thiopropionate linkage: Preparation and polyion complex micelle formation. Biomacromolecules. 2003;4:1426–1432. doi: 10.1021/bm034164u. [DOI] [PubMed] [Google Scholar]
- 355.Lv W. Bioengineered boronic ester modified dextran polymer nanoparticles as reactive oxygen species responsive nanocarrier for ischemic stroke treatment. ACS Nano. 2018;12:5417–5426. doi: 10.1021/acsnano.8b00477. [DOI] [PubMed] [Google Scholar]
- 356.Hu P., Tirelli N. Scavenging ROS: Superoxide dismutase/catalase mimetics by the use of an oxidation-sensitive nanocarrier/enzyme conjugate. Bioconjug. Chem. 2012;23:438–449. doi: 10.1021/bc200449k. [DOI] [PubMed] [Google Scholar]
- 357.Scott E.A. Dendritic cell activation and T cell priming with adjuvant- and antigen-loaded oxidation-sensitive polymersomes. Biomaterials. 2012;33:6211–6219. doi: 10.1016/j.biomaterials.2012.04.060. [DOI] [PubMed] [Google Scholar]
- 358.Vanderburgh J.P. Systemic delivery of a Gli inhibitor via polymeric nanocarriers inhibits tumor-induced bone disease. J. Control. Release. 2019;311:257–272. doi: 10.1016/j.jconrel.2019.08.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Cao W., Gu Y., Meineck M., Li T., Xu H. Tellurium-containing polymer micelles: Competitive-ligand-regulated coordination responsive systems. J. Am. Chem. Soc. 2014;136:5132–5137. doi: 10.1021/ja500939m. [DOI] [PubMed] [Google Scholar]