Abstract
Background
Since the body of evidence addressing the coagulation derangements caused by Coronavirus disease (COVID-19) has been constantly growing, we investigated whether pre-hospitalization oral anticoagulation (OAC) or in-hospital heparin treatment could have a protective role among COVID-19 patients.
Method
In this cohort study, consecutive COVID-19 patients admitted to four different Italian Institutions were enrolled. Baseline demographic, clinical, laboratory, and radiological characteristics, as well as in-hospital treatment and outcomes were evaluated. The primary outcome was mortality.
Results
A total of 844 COVID-19 patients were enrolled as study cohort, n = 65 (7.7%) taking OACs prior to hospitalization. Regarding clinical outcomes, OAC patients developed acute hypoxemic respiratory failure (AHRF) more frequently than non-OAC patients as well as presenting a higher mortality rate (44.6% vs 19.8%, p < 0.001). At overall multivariate logistical regression, use of heparin (n = 394, 46.6%) was associated with a better chance of survival to hospital discharge (OR 0.60 [0.38–0.94], p < 0.001), in particular in patients with AHRF, with no association found with the use of OACs. In a sub-analysis, the highest mortality rate was found for AHRF patients when heparin was not administered.
Conclusion
In our cohort, OACs appeared to be ineffective in reducing mortality rate, while heparin resulted to be a useful treatment when lung disease was sufficiently severe, potentially suggesting a crucial role of microthrombosis in severe COVID-19. Due to the relatively small number of COVID-19 patients treated with OACs included in our analysis and their higher number of comorbidities, larger studies are needed in order to confirm our findings.
Keywords: Anticoagulation, Oral anticoagulants, Heparin, Microthrombosis, Coagulopathy, COVID-19
Highlights
-
•
Evidence on a prothrombotic asset caused by COVID-19 has been constantly growing.
-
•
In our cohort, OACs appeared to be ineffective in reducing mortality rate.
-
•
Heparin resulted to be a useful treatment when lung disease was sufficiently severe.
-
•
Microthrombosis may have a crucial role in COVID-19.
-
•
Due to the relatively small sample size of OAC patients included, larger studies are needed.
1. Introduction
The body of evidence addressing the coagulation derangements caused by Coronavirus disease (COVID-19) has been constantly growing. In vitro studies have shown that coronavirus infection promotes the activation of the coagulation cascade and remodeling of the extracellular matrix [1]. Additionally, COVID-19 has been associated to venous and arterial thromboembolic events in up to 21–25% of cases [[2], [3], [4], [5]], especially in intensive care unit (ICU) patients, due to additional well-known risk factors for thromboembolism (e.g. severe hypoxia, vascular damage, prolonged bed-rest, and advanced age). Therefore, it has been postulated that the excess mortality of COVID-19 may be related to the severe hypercoagulability more than to the respiratory failure per se, although its pathophysiology has not been properly understood.
Hence, we investigated whether pre-hospitalization oral anticoagulation (OAC) could have a protective role in reducing mortality among patients admitted for COVID-19 and whether, in our cohort, heparin may confirm its presumptive beneficial role [6].
2. Methods
Consecutive confirmed cases (≥ 18 years old) of COVID-19 admitted to four Italian Institutions (Luigi Sacco Hospital, Milan; Policlinico Umberto I, Rome; Spedali Civili, Brescia; Humanitas Gavazzeni Hospital, Bergamo) between February 23, 2020 and April 1, 2020 were recruited. Data were retrospectively analyzed.
A confirmed case of COVID-19 was defined by a positive result on a reverse-transcriptase-polymerase-chain-reaction assay performed on a nasopharyngeal swab, in accordance with World Health Organization (WHO) guidelines. All patients still hospitalized or with an incomplete follow-up were excluded. Acute hypoxemic respiratory failure (AHRF) was defined with PaO2/FiO2 ratio < 300. The primary outcome was mortality.
Categorical variables are reported as counts (percentage). Normality of distribution was tested for all continuous variables using a Shapiro-Wilk test. Continuous variables are reported as mean ± standard deviation (s.d.) or as median [inter-quartile range, IQR] if normally or non-normally distributed, respectively. Comparisons were performed using a student t-test, a Mann Whitney U test, a Chi Square test, or a Fisher exact test, as appropriate. A multivariate logistic regression was performed to test the association between outcomes (mortality) and cardiovascular risk factors and/or parameters of clinical interest for COVID-19 patients; strength of association is reported using odds ratios (OR). A by group survival analysis was performed using Kaplan Meier curves. Multivariate Cox regression model was utilized to determine the independent risk factors for mortality. A two-tailed p value <0.05 was deemed as statistically significant. All statistical analysis were performed using STATA version 14.0 (Stata Corp, TX, USA).
3. Results
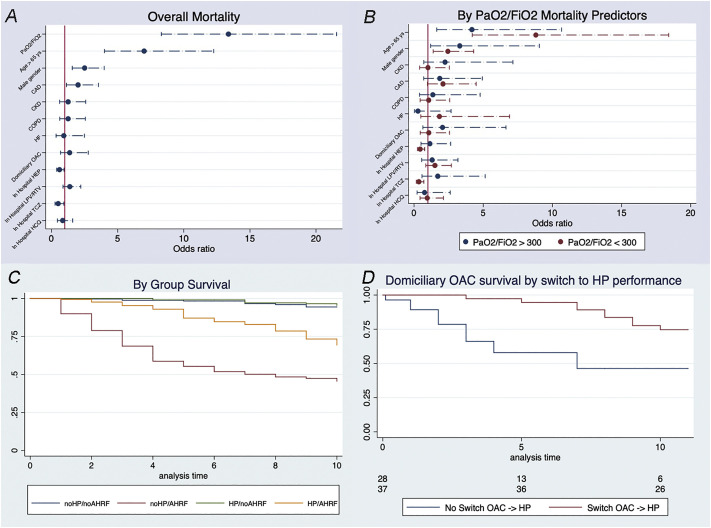
A total of 844 COVID-19 patients were enrolled as study cohort, n = 65 (7.7%) taking OACs prior to hospitalization (n = 22, 33.8% vitamin-K antagonists; n = 43, 66.2% direct OACs). Baseline clinical variables, drug therapy and clinical outcomes during hospital stay are summarized in Table 1 . OAC patients were older than non-OAC patients (76.7 ± 11.6 vs 62.3 ± 15.19 years, p < 0.001) and presented more comorbidities, such as atrial fibrillation (AF) (83.1% vs 3.1%, p < 0.001). OAC patients were more likely to have lower PaO2/FiO2 ratio (268 [148–340] vs 314 [193–386], p 0.033), worse chest x-ray severity (Taylor et al. score 3 [[2], [3], [4]] vs 2 [2,3]; p = 0.034) and lower D-dimer levels on admission (567 [239–893] vs 1017 [471–2736] ng/mL, p = 0.027). Median in-hospital stay was not significantly different in the two groups, as well as admission to ICU (3.08% vs 5.65%, p = 0.380). On the other hand, OAC patients developed AHRF and myocardial injury more often (44.6% vs 19.8%, p < 0.001; 16.92% vs 11.05%, p = 0.154) than non-OAC patients and needed non-invasive ventilation more frequently (47.7% vs 33.0%, p = 0.016). A total of 183 (21.86%) patients died, with OAC patients presenting a higher mortality rate (44.6% vs 19.8%, p < 0.001). At overall multivariate logistical regression, AHRF (OR: 13.2 [8.3–21.6], p < 0.001), age > 65 years, male gender, and history of CAD resulted independently and significantly associated with an increased risk of death during hospitalization, while, interestingly, use of heparin (n = 394, 46.6%) was associated with a better chance of survival to hospital discharge (OR 0.60 [0.38–0.94], p < 0.001), with no association with the use of OACs (Fig. 1A). A stratification of in-hospital mortality based on PaO2/FiO2 ratio is reported in Fig. 1B: as shown, the use of in-hospital heparin was protective in patients with AHRF but not in patients with PaO2/FiO2 ratio > 300 (p for interaction between heparin and PaO2/FiO2 < 300 < 0.001). The entire model has been reported in Supplementary Data. In a sub-analysis, Kaplan-Meier survival curves, stratified on OAC patients who were or were not switched to heparin during hospitalization, showed reduced survival rates in the latter group (Fig. 1C). Moreover, analyzing heparin administration in the presence or absence of AHRF (Fig. 1D), the highest mortality rate was found for AHRF patients when heparin was not administered (in particular, AHRF with vs without heparin treatment p < 0.001).
Table 1.
Baseline characteristics of the study cohort, drug therapy and clinical outcomes during hospital stay.
| Overall (n = 844) | OAC (n = 65) | No-OAC (n = 779) | p | |
|---|---|---|---|---|
| Age (years), mean ± s.d. | 63.4 ± 16.1 | 76.7 ± 11.6 | 62.3 ± 15.9 | <0.001 |
| Sex (male), n (%) | 521 (61.7) | 474 (60.9) | 47 (72.3) | 0.068 |
| Main cardiovascular risk factors, n (%) | ||||
| Hypertension | 381 (45.1) | 48 (73.9) | 333 (42.8) | <0.001 |
| Diabetes | 140 (16.6) | 17 (26.2)27 | 123 (15.8) | 0.031 |
| Dyslipidemia | 201 (23.8) | (41.5) | 174 (22.3) | <0.001 |
| Smoke | 94 (11.1) | 8 (12.3) | 86 (11.0) | 0.755 |
| Obesity | 79 (9.4) | 10 (15.4) | 69 (8.9) | 0.083 |
| Comorbidities, n (%) | ||||
| Heart failure | 78 (39 | 16 (24.6) | 23 (3) | <0.001 |
| History of atrial fibrillation | (4.6)9.2) | 54 (83.1) | 24 (3.1) | <0.001 |
| Chronic kidney disease | 63 (7.5) | 12 (18.5) | 51 (6.6) | <0.001 |
| Chronic obstructive pulmonary disease | 62 (7.4) | 17 (26.2) | 45 (5.8) | <0.001 |
| Coronary artery disease | 112 (13.3) | 19 (29.2) | 93 (11.9) | <0.001 |
| Stroke | 33 (3.9) | 10 (15.4) | 23 (2.9) | <0.001 |
| Drug therapy, n (%) | ||||
| ACE-inhibitors | 122 (14.47) | 19 (29.3) | 103 (13.24) | <0.001 |
| ARBs | 119 (14.1) | 14 (21.5) | 105 (13.75) | 0.007 |
| Beta-blockers | 142 (16.8) | 20 (30.8) | 122 (15.7) | 0.002 |
| Calcium-antagonists | 105 (12.4) | 15 (23.1) | 90 (11.6) | 0.007 |
| Diuretics | 136 (16.1) | 27 (41.5) | 109 (14.0) | <0.001 |
| VKAs | 22 (2.6) | 22 (33.8) | NA | NA |
| DOACs | 43 (5.1) | 43 (66.2) | NA | NA |
| Antiplatelets | 130 (15.4) | 19 (29.2) | 111 (14.3) | <0.001 |
| Statins | 177 (21.0) | 25 (38.5) | 152 (19.5) | <0.001 |
| AADs | 24 (2.8) | 12 (18.5) | 12 (1.5) | <0.001 |
| Laboratory findings, median [IQR] | ||||
| WBC (109/L) | 6.0 [4.7–8.3] | 6.05 [4.63–7.93] | 5.97 [4.69–8.3] | 0.873 |
| Hb (g/dl) | 13.7 [12.5–14.8] | 12.6[11.1–14.2] | 13.7 [12.6–14.8] | <0.001 |
| Platelets (109/L) | 195 [152–260] | 164[142–245] | 195 [153–260] | 0.039 |
| Creatinine (mg/dl) | 0.95 [0.79–1.15] | 1.1[0.9–1.7] | 0.93 [0.78–1.12] | <0.001 |
| D-dimer (μg/L) | 883 [420–2357] | 567[239–893] | 1017[471–2736] | 0.027 |
| LDH (U/L) | 318 [239–430] | 346.5 [284–443] | 316 [238–430] | 0.076 |
| ALT (U/L) | 29 [19–47] | 23 [17–37] | 29 [20–48] | 0.015 |
| CRP (mg/L) | 19.0 [6.4–67.7] | 20.2 [8.3–81.8] | 19 [6.1–67] | 0.427 |
| Chest radiograph severity scoring system (Taylor et. al), median [IQR] | 2 [2–3] | 3 [2–4] | 2 [2–3] | 0.034 |
| Symptom onset to admission (days), median [IQR] | 7 [3−10] | 6 [3–9.5] | 7 [3–10] | 0.811 |
| Arterial blood gas analysis | ||||
| SaO2 (%), median [IQR] | 96 [93–98] | 94 [88–96] | 96 [93–98] | 0.001 |
| paO2/FiO2, median [IQR] | 310 [190–385] | 268 [148–340] | 314 [192–386] | 0.033 |
| Drug therapy, n (%) | ||||
| Antibiotics | 373(44.2) | 35(53.8) | 338 (43.4) | 0.103 |
| Antivirals | 467 (55.3) | 41 (63.1) | 426(54.7) | 0.191 |
| Steroids | 93 (11.0) | 6 (9.2) | 87(11.2) | 0.632 |
| Hydroxychloroquine | 681(80.7) | 56 (86.1 | 625 (80.2) | 0.245 |
| Tocilizumab | 127(15.0) | 7(10.8) | 120(15.4) | 0.315 |
| Heparin | 394(46.7) | 37 (56.9) | 375 (45.8) | 0.085 |
| Myocardial injury, n (%) | 97 (11.5) | 11(16.9) | 86 (11.0) | 0.154 |
| Intensive care unit admission, n (%) | 46 (5.4) | 2(3.1) | 44(5.65) | 0.380 |
| Non-invasive ventilation, n (%) | 288(34.1) | 31 (47.7) | 257(33) | 0.016 |
| Acute respiratory distress syndrome, n (%) | 183 (21.7) | 29(44.6) | 154 (19.8) | <0.001 |
| Hospital length of stay (days), median [IQR] | 9 [5–15] | 9 [5–14] | 9 [5–15] | 0.933 |
| Deaths, n (%) | 183 (21.7) | 29(44.6) | 154(19.8) | <0.001 |
Abbreviations
AADs: antiarrhythmic drugs
ACE: angiotensin-converting enzyme
ALT: alanine aminotransferase
ARBs: angiotensin II receptor blockers
CRP: C-reactive protein
DOAC: direct anticoagulants
FiO2: fraction of inspired oxygen
Hb: hemoglobin
IQR: interquartile range
LDH: lactate dehydrogenase
NA: not available
s.d.: standard deviation
OAC: anticoagulants
paCO2: partial pressure of carbon dioxide in arterial blood
paO2: partial pressure of oxygen in arterial blood
SaO2: oxygen saturation
VKAs: vitamin k antagonists
WBC: white blood cells.
Fig. 1.
panel A-B-C-D.
Panel A-B: multivariable logistic-regression analysis showing independent overall (panel A) and by group (panel B) predictors of in-hospital death.
Panel C: Kaplan–Meier curves depicting mortality rates for OAC patients who were or were not switched to heparin during hospitalization. Log rank p for every group: No HEP/No AHRF vs No HEP/AHRF p < 0.001; No HEP/No AHRF vs HEP/No AHRF p < 0.585; No HEP/No AHRF vs HEP/AHRF p < 0.001; No HEP/AHRF vs HEP/No AHRF p < 0.001; No HEP/AHRF vs HEP/AHRF p < 0.001; HEP/No AHRF vs HEP/AHRF p < 0.001.
By group hazard ratios: No HEP/No AHRF 0.215 (CI 0.125–0.371); HEP/No AHRF 0.196 (CI 0.117–0.330); No HEP/AHRF 6.439 (CI 4.668–8.882); HEP/AHRF 0.869 (CI 1.367–2.557). All p < 0.01.
Panel D: Kaplan–Meier curves depicting mortality rates for patients who were or were not treated with heparin during hospitalization in the presence or absence of AHRF. Hazard ratio for switching from OACs to HEP: 0.213 (CI 0.079–0.575, p < 0.001).
Abbreviations
AHRF: acute hypoxemic respiratory failure
CAD: coronary artery disease
CI: confidence interval
CKD: chronic kidney disease
COPD: chronic obstructive pulmonary disease
HCQ: hydroxychloroquine
HEP: heparin
HF: heart failure
LPV/RTV: lopinavir/ritonavir
OAC: oral anticoagulant
P/F: PaO2/FiO2 ratio
TCZ: tocilizumab
ys: years.
4. Discussion
The findings of this study can be summarized as follows: (I) 7.7% of hospitalized COVID-19 patients were on OAC at hospital admission; (II) 46.6% of our cohort was treated with heparin during hospitalization, either at anticoagulant dose, if switched from domiciliary OAC (n = 37) or in case of new onset AF and pulmonary embolism, or at prophylactic dose in all other cases; (III) while OAC therapy did not show a protective effect, the subsequent use of heparin during the hospital stay seemed to be associated with survival in patients with AHRF.
Given the prothrombotic asset of COVID-19 patients, clearly demonstrated in post-mortem reports [7,8], Barnes et al. recommended VTE prophylaxis for all hospitalized COVD-19 patients and switching OAC patients to shorter acting agents (e.g., low-molecular or unfractionated heparin) during hospitalization in case of clinical deterioration, changes in renal function, or need for invasive procedures [6]. It has to be underlined though, that these recommendations were lacking in the early phase of the pandemic, and given the interactions between some antiviral drugs and OACs [9], heparins were often preferred. As with every drug, the benefits of heparin administration should be carefully evaluated against the increased risk of bleeding, to which a large proportion of patients may be unnecessarily be exposed. Indeed, Tang et al. showed that heparin administration was beneficial only in patients with high D-dimer levels and in case of sepsis-induced coagulopathy [10]. On the other hand, Whyte et al. proposed even fibrinolytic agents such as tissue-type plasminogen activator to treat COVID-19 patients with coagulopathy [11]. If such prothrombotic asset is a key feature of COVID-19, one may expect that other forms of anticoagulation may be beneficial. In contrast with the smaller cohort from Rossi et al. [12], we found no beneficial effect regarding in-hospital mortality among patients receiving OACs, whereas mortality rate was significantly reduced with the in-hospital heparin administration, but only in those patients who presented AHFR upon admission. The key physiopathological question, is whether the protective role of heparin, only in patients that are also hypoxemic, is simply due to the association between the pneumonia severity and the concurrent hypercoagulable state or the hypercoagulability is itself a cause of hypoxemia. Indeed, overt pulmonary embolism is present in a minority of the patients at admission, but microthrombi below the resolution of the contrast computed tomography (CT) may cause ventilation-perfusion mismatch and, therefore, concur to hypoxemia.
Other mechanisms, though speculative, may play a role: heparan sulfate proteoglycans are involved in severe acute respiratory syndrome coronavirus (SARS-CoV) and Human Coronavirus NL63 cell entry [13,14] so that heparin may prevent binding of spike protein to host cells, as well as interacting with SARS-CoV-2 spike S1 receptor binding domain [15]. Moreover, heparin exhibits anti-inflammatory effects [16], inhibiting inflammatory cell infiltration and dampening pro-inflammatory signals, that may be pivotal in determining different outcomes compared to OACs.
Our study has some limitations: due to the relatively small sample size of OAC patients, our findings of no mortality benefit with OACs compared to heparin may be underpowered and potentially confounded, considering proper statistical adjustments used to account for all the considered imbalances and baseline comorbidities. Additionally, the observational nature of our registry cannot prove a causal relationship between OAC and heparin administration and reduced mortality. Nevertheless, our findings confirm the need for randomized trials in support of in-hospital use of oral and parenteral anticoagulants.
5. Conclusion
In our cohort, OACs appeared to be ineffective in reducing mortality rate, even among patients with more severe AHRF, while heparin resulted to be a useful treatment when lung disease was sufficiently severe, potentially suggesting a crucial role of microthrombosis in severe COVID-19. Due to the relatively small number of COVID-19 patients treated with OACs included in our analysis and their higher number of comorbidities, larger studies are needed in order to confirm our findings. Possible mechanisms underlying the role of OAC and heparin in VTE prophylaxis and lung microthrombosis need to be further investigated with randomized trials.
Disclosures
The authors report no relationships that could be construed as a conflict of interest.
Funding
The authors received no specific funding for this work.
Authors statement
All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ijcard.2020.09.001.
Appendix A. Supplementary data
Supplementary material.
References
- 1.Gralinski L.E., Baric R.S. Molecular pathology of emerging coronavirus infections. J. Pathol. 2015;235:185–195. doi: 10.1002/path.4454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Levi M., Thachil J., Iba T., Levy J.H. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020 doi: 10.1016/s2352-3026(20)30145-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang L., Feng X., Zhang D., Jiang C., Mei H., Wang J., Zhang C., Li H., Xia X., Kong S., Liao J., Jia H., Pang X., Song Y., Tian Y., Wang B., Wu C., Yuan H., Zhang Y., Li Y., Sun W., Zhang Y., Zhu S., Wang S., Xie Y., Ge S., Zhang L., Hu Y., Xie M. Deep Vein thrombosis in hospitalized patients with coronavirus disease 2019 (COVID-19) in Wuhan, China: Prevalence, Risk factors, and outcome. Circulation. 2020 doi: 10.1161/circulationaha.120.046702. CIRCULATIONAHA.120.046702. [DOI] [PubMed] [Google Scholar]
- 4.Cui S., Chen S., Li X., Liu S., Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J. Thromb. Haemost. 2020;18:1421–1424. doi: 10.1111/jth.14830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lodigiani C., Iapichino G., Carenzo L., Cecconi M., Ferrazzi P., Sebastian T., Kucher N., Studt J.D., Sacco C., Alexia B., Sandri M.T., Barco S. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb. Res. 2020;191:9–14. doi: 10.1016/j.thromres.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barnes G.D., Burnett A., Allen A., Blumenstein M., Clark N.P., Cuker A., Dager W.E., Deitelzweig S.B., Ellsworth S., Garcia D., Kaatz S., Minichiello T. Thromboembolism and anticoagulant therapy during the COVID-19 pandemic: Interim clinical guidance from the anticoagulation forum. J. Thromb. Thrombolysis. 2020;50:72–81. doi: 10.1007/s11239-020-02138-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carsana L., Sonzogni A., Nasr A., Rossi R.S., Pellegrinelli A., Zerbi P., Rech R., Colombo R., Antinori S., Corbellino M., Galli M., Catena E., Tosoni A., Gianatti A., Nebuloni M. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study. Lancet Infect. Dis. 2020 doi: 10.1016/S1473-3099(20)30434-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ackermann M., Verleden S.E., Kuehnel M., Haverich A., Welte T., Laenger F., Vanstapel A., Werlein C., Stark H., Tzankov A., Li W.W., Li V.W., Mentzer S.J., Jonigk D. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N. Engl. J. Med. 2020 doi: 10.1056/nejmoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schiavone M., Gobbi C., Biondi-Zoccai G., D’Ascenzo F., Palazzuoli A., Gasperetti A., Mitacchione G., Viecca M., Galli M., Fedele F., Mancone M., Forleo G.B. Acute coronary syndromes and Covid-19: exploring the uncertainties. J. Clin. Med. 2020;9:1683. doi: 10.3390/jcm9061683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tang N., Bai H., Chen X., Gong J., Li D., Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020 doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Whyte C.S., Morrow G.B., Mitchell J.L., Chowdary P., Mutch N.J. Fibrinolytic abnormalities in acute respiratory distress syndrome (ARDS) and versatility of thrombolytic drugs to treat COVID-19. J. Thromb. Haemost. 2020 doi: 10.1111/jth.14872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rossi R., Coppi F., Talarico M., Boriani G. Protective role of chronic treatment with direct oral anticoagulants in elderly patients affected by interstitial pneumonia in COVID-19 era. Eur. J. Intern. Med. 2020 doi: 10.1016/j.ejim.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lang J., Yang N., Deng J., Liu K., Yang P., Zhang G., Jiang C. Inhibition of SARS pseudovirus cell entry by lactoferrin binding to heparan sulfate proteoglycans. PLoS One. 2011;6 doi: 10.1371/journal.pone.0023710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Milewska A., Nowak P., Owczarek K., Szczepanski A., Zarebski M., Hoang A., Berniak K., Wojarski J., Zeglen S., Baster Z., Rajfur Z., Pyrc K. Entry of human coronavirus NL63 into the cell. J. Virol. 2017;92 doi: 10.1128/jvi.01933-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mycroft-West C.J., Su D., Pagani I., Rudd T.R., Elli S., Guimond S.E., Miller G., Meneghetti M.C.Z., Nader H.B., Li Y., Nunes Q.M., Procter P., Mancini N., Clementi M., Bisio A., Forsyth N.R., Turnbull J.E., Guerrini M., Fernig D.G., Vicenzi E., Yates E.A., Lima M.A., Skidmore M.A. Heparin inhibits cellular invasion by SARS-CoV-2: structural dependence of the interaction of the surface protein (spike) S1 receptor binding domain with heparin. BioRxiv. 2020 doi: 10.1101/2020.04.28.066761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Young E. The anti-inflammatory effects of heparin and related compounds. Thromb. Res. 2008;122:743–752. doi: 10.1016/j.thromres.2006.10.026. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material.