Abstract
Background: To comprehensively understand the impact of sex and smoking on the efficacy of epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI) therapy in terms of overall survival (OS) in non-small-cell lung cancer (NSCLC).
Methods: PubMed, Cochrane Library, Embase, and Scopus were searched from inception to March 17, 2019. OS was analyzed based on hazard ratios (HRs) and 95% confidence intervals (CIs) and estimated using the random effects model.
Results: Our meta-analysis included 22 studies involving 11,874 patients. In the primary analysis, we found no statistically significant efficacy difference for EGFR-TKI intervention between females and males (pooled HR 0.95, 95% CI 0.87–1.04, P = 0.30) and no obvious efficacy difference between never smokers and ever smokers (pooled HR 0.91, 95% CI 0.76–1.09, P = 0.31). In the subgroup analysis of placebo control treatment, we found that female NSCLC patients who received EGFR-TKI therapy had a longer OS than male patients (pooled HR 0.86, 95% CI 0.75–1.00, P = 0.04), while smoking status showed no significant effect on the efficacy of EGFR-TKI treatment in terms of the OS of NSCLC patients in all subgroup analyses.
Conclusion: The efficacy of EGFR-TKI therapy for NSCLC patients is independent of smoking status but dependent on sex, and females have a longer OS than males.
Keywords: meta-analysis, epidermal growth factor receptor-tyrosine kinase inhibitor, overall survival, non-small-cell lung cancer, sex
Introduction
Lung cancer is one of the most common cancers in men and women, and there is no doubt that lung cancer poses the greatest threat to human life, as it results in one-quarter of all cancer deaths (1). However, non-small-cell lung cancer (NSCLC) accounts for more than 85% of lung cancer, (2) and it is well-known that there is a significant difference in the development of NSCLC between male and female patients and between patients of different smoking statuses (3, 4).
Overactivation of epidermal growth factor receptor (EGFR) tyrosine kinases is a key mechanism leading to the development of NSCLC (5). In recent years, epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) have achieved good clinical efficacy in the treatment of NSCLC. At present, three generations of EGFR-TKIs have been widely used in the clinical treatment of NSCLC, such as gefitinib, afatinib, and osimertinib, which represent first-, second-, and third-generation EGFR-TKIs, respectively (6). Of course, other new EGFR-TKIs are also on the way to development and being promoted (7). There is no doubt that EGFR-TKI therapy plays a pivotal and irreplaceable role in the treatment of patients with NSCLC.
Previous meta-analyses focused on EGFR-mutated NSCLC patients and the progression-free survival (PFS) have concluded that female and non-smoking NSCLC patients have better efficacy with EGFR-TKIs than male patients and smokers (8–10). However, as we know that EGFR-TKIs treatment also shows some kind of treatment effects for NSCLC patients with unknown or wild-type EGFR status, and the association of overall survival (OS) with EGFR-TKIs treatment in NSCLC patients. Those previous studies could be expanded.
In the meantime, we found two meta-analyses that comprehensively and thoroughly studied the effect of sex on the efficacy in terms of OS of immune checkpoint inhibitors in cancer treatment (11, 12). We were very interested in these two studies, which prompted us to re-examine the impact of sex and smoking status, the two most common and important clinical features in NSCLC, on the efficacy of EGFR-TKIs. Thus, we have done and now report a meta-analysis of the association of sex and smoking with the efficacy of EGFR-TKIs in terms of OS in NSCLC.
Methods
Search Strategy and Selection Criteria
We performed this meta-analysis according to the PRISMA guidelines (13). PubMed, Cochrane Library, Embase, and Scopus were searched from inception to March 17, 2019. Two authors independently searched the databases. The main search terms were “lung cancer,” “survival,” “hazard ratio,” “EGFR” and “randomized controlled trials.” Full details of our search strategies for the databases are shown in the Supplementary Material (Supplementary Content 1). Titles, abstracts and full-text articles were reviewed independently by two authors. Inconsistencies were discussed by all authors to reach consensus. Reference lists were also reviewed to identify additional relevant studies.
The literature inclusion criteria were as follows: randomized controlled clinical trials for NSCLC that contained any single EGFR-TKI treatment; the treatment plans in the corresponding control group did not contain any other EGFR-TKIs; the prognosis endpoint was OS, and the corresponding hazard ratios (HRs) and 95% confidence intervals (CIs) were reported according to sex and/or smoking status; and the full-text manuscripts were published in English. The exclusion criteria were as follows: retrospective studies of clinical cases; abstracts, reports, and papers from conferences; literature reviews and meta-analyses.
Data Extraction and Study Quality Assessment
Two authors independently extracted data from the included studies. Discrepancies were resolved by all authors through discussion to reach consensus. The following variables were extracted from each study: first author, publication year, EGFR mutation status, trial name, lines of therapy, EGFR-TKI intervention drug, control treatment plan, total number of patients, median age (years), median follow-up time (months), overall HR with 95% CI, HR with 95% CI according to patient sex, and HR with 95% CI according to smoking status. When duplicate publications were identified from one trial, we included only the most complete report.
The methodological quality of the included studies was assessed using the five-point Jadad score (14), which judges manuscripts according to the descriptions of randomization, blinding and withdrawals and dropouts. The details are as follows: whether randomized or not; whether randomization was described or not; whether double-blinded or not; whether blinding was described or not; and whether withdrawals and dropouts were described or not. For each of the above questions, if the answer is yes, the study gets 1 point; if the answer is no, the study gets 0 points. The quality scale ranges from 0 to 5 points for each controlled trial. A score of 2 or less indicates a low-quality study, while a score of 3–5 indicates a high-quality study.
Data Analysis
The HRs and 95% CIs were extracted from each study according to the classification of overall HR, HR in male patients, HR in female patients, HR in never smokers and HR in ever (former and/or current) smokers. For the overall HRs, we used the random effects model to calculate the pooled HR directly. For the HRs classified by sex and smoking status, we first calculated the interaction HRs and 95% CIs for each study and thereafter obtained the pooled HR using the random effects model. The heterogeneity between studies was identified using the Q-test and quantified using I2-values (11, 12). Potential publication bias was evaluated using the Egger and Begg test. To assess the differences between males and females or never smokers and ever smokers, we performed calculations using log HR to evaluate whether the variations differed from the null hypothesis by using the χ2-test (11, 12).
We performed subgroup analyses to further explore the variation of the effect of sex and smoking status on EGFR-TKI therapy efficacy. We only considered subgroups that included no less than two studies. The subgroups were EGFR status (unknown, wild-type, and mutation), lines of therapy (>1 and 1), EGFR-TKI intervention (gefitinib, erlotinib, and others), and control treatment (placebo, chemotherapy and others).
We performed all data analyses using Stata 14.0 (StataCorp LP, USA). All reported P-values are 2-sided, and a P-value of 0.05 indicated statistical significance. An HR < 1 indicated that EGFR-TKI efficacy was better than non-EGFR-TKI efficacy, EGFR-TKI efficacy in females was better than in males, and EGFR-TKI efficacy in never smokers was better than in ever smokers.
Results
Literature Search
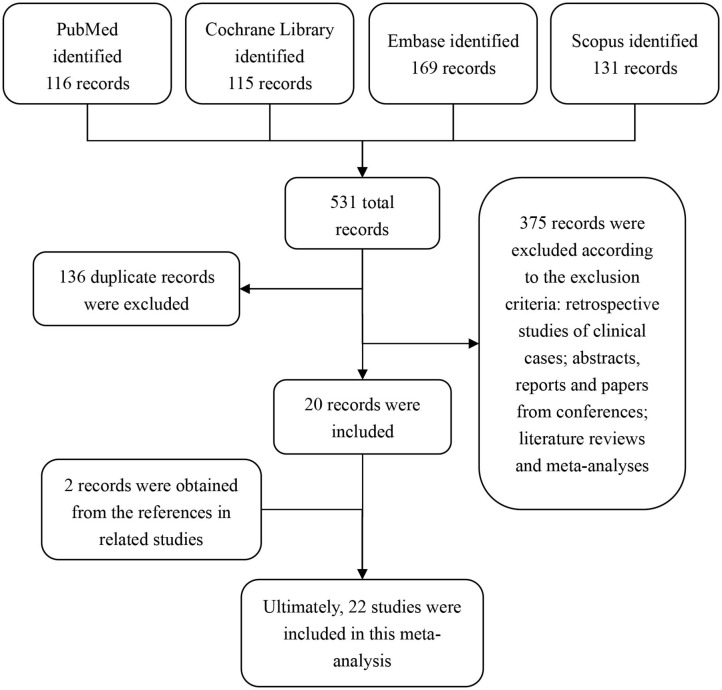
By searching our search terms in the database, we obtained 531 potential publications. A total of 136 records were excluded because of duplicated titles. By reviewing the abstract and full text, 375 records were further excluded according to our inclusion and exclusion criteria. Therefore, 20 publications were selected. In addition, by reviewing the references from these 20 selected studies, we found an additional 2 studies that were also in line with our inclusion criteria. Ultimately, 22 studies (15–36) were included in this meta-analysis (Figure 1).
Figure 1.
Flow diagram of the study selection.
Characteristics of the Identified Studies
We assessed the quality of the 22 studies by the Jadad score. The mean score was 3.64 (ranging from 3 to 5), and no study received a low-quality score (scored 2 or less), indicating that these included studies possessed high methodological quality. The Jadad scores for each study are listed in the Supplementary Table S1 in the Supplementary Material).
As shown in Table 1, for the 22 included studies, 3 involved patients with wild-type EGFR, and 5 involved patients with EGFR mutations, and the other 14 studies did not consider EGFR mutations in patients, which we defined as EGFR unknown. Eight of 22 studies were for first-line treatment, and 14 of 22 studies were for second-line or beyond treatments. Compared with placebo or standard chemotherapy, the EGFR-TKI interventions included gefitinib, erlotinib, dacomitinib, afatinib, and icotinib. In total, 11,874 patients were involved in these included trials.
Table 1.
Main characteristics and results of the 22 studies included in the meta-analysis.
| Article source | EGFR situation | Trial name | Lines of therapy | EGFR-TKI intervention | Control treatment | Total patients | Median age (years) | Median follow-up (months) | Overall HR (95% CI) | Sex of HR (95% CI) | Smoking of HR (95% CI) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | Never | Ever | ||||||||||
| Thatcher et al. (15) | Unknown | ISEL | >1 | Gefitinib | Placebo | 1,692 | 62/61 | 7.2 | 0.89 (0.77–1.02) | 0.91 (0.78-1.06) | 0.77 (0.60-0.97) | 0.67 (0.49-0.91) | 0.92 (0.80-1.05) |
| Tsao et al. (16) | Unknown | BR.21 | >1 | Erlotinib | Placebo | 731 | 62/59 | NR | 0.70 (0.58–0.85) | 0.76 (0.62–0.94) | 0.80 (0.59–1.07) | 0.42 (0.28–0.64) | 0.87 (0.71–1.05) |
| Kim et al. (17) | Unknown | INTEREST | >1 | Gefitinib | Docetaxel | 1,433 | 61/60 | 7.6 | 1.02 (0.91–1.15) | 1.08 (0.95–1.24) | 0.95 (0.78–1.17) | 0.93 (0.70–1.23) | 1.05 (0.92–1.19) |
| Maruyama et al. (18) | Unknown | V-15-32 | >1 | Gefitinib | Docetaxel | 489 | NR | 21 | 1.12 (0.89–1.40) | 1.10 (0.83–1.43) | 1.23 (0.81–1.84) | 0.93 (0.58–1.48) | 1.13 (0.87–1.45) |
| Cappuzzo et al. (19) | Unknown | SATURN | >1 | Erlotinib | Placebo | 889 | 60/60 | 11.4/11.5 | 0.81 (0.70–0.95) | 0.88 (0.74–1.05) | 0.64 (0.46–0.91) | 0.69 (0.45–1.05) | 0.84 (0.71–0.99) |
| Fukuoka et al. (20) | Unknown | IPASS | 1 | Gefitinib | Carboplatin plus paclitaxel | 1,217 | 57/57 | 17 | 0.90 (0.79–1.02) | 0.77 (0.59–1.02) | 0.93 (0.81–1.08) | 0.90 (0.78–1.03) | 0.99 (0.62–1.60) |
| Lee et al. (21) | Unknown | TOPICAL | 1 | Erlotinib | Placebo | 670 | 77/77 | NR | 0.94 (0.81–1.10) | Female vs. male 0.81 (0.59–1.01) |
Never vs. ever 0.64 (0.36–1.14) |
||
| Pérol et al. (22) | Unknown | IFCT-GFPC 0502 |
>1 | Erlotinib | Observation | 310 | 56.4/59.8 | 25.6 | 0.87 (0.68–1.13) | 0.88 (0.66–1.18) | 0.89 (0.52–1.53) | 0.83 (0.34–2.01) | 0.88 (0.68–1.15) |
| Kelly et al. (23) | Wild-type | PDX-012 | >1 | Erlotinib | Pralatrexate | 201 | 62/63 | NR | 1.19 (0.88–1.64) | 1.08 (0.75–1.56) | 1.64 (0.92–2.94) | NR | NR |
| Miller et al. (24) | Unknown | LUX-Lung 1 | >1 | Afatinib | Placebo | 585 | 58/59 | NR | 1.08 (0.86–1.35) | 1.17 (0.83–1.65) | 1.02 (0.76–1.37) | 1.20 (0.90–1.61) | 0.43 (0.31–0.61) |
| Ciuleanu et al. (25) | Unknown | TITAN | >1 | Erlotinib | Docetaxel or pemetrexed | 424 | 59/59 | 27.9/24.8 | 0.96 (0.78–1.19) | 0.86 (0.67–1.10) | 1.23 (0.78–1.94) | 0.86 (0.49–1.51) | 0.93 (0.78–1.11) |
| Garassino et al. (26) | Wild-type | TAILOR | >1 | Erlotinib | Docetaxel | 219 | 66/67 | 33 | 1.27 (0.95–1.69) | 1.18 (0.84–1.67) | 1.47 (0.84–2.56) | 1.69 (0.89–3.23) | 1.12 (0.81–1.54) |
| Inoue et al. (27) | Mutation | NEJ002 | 1 | Gefitinib | Carboplatin plus paclitaxel | 228 | NR | 23 | 0.89 (0.63–1.24) | 0.92 (0.53–1.61) | 0.88 (0.57–1.35) | 0.88 (0.57–1.37) | 0.98 (0.58–1.65) |
| Ellis et al. (28) | Unknown | NCIC CTG BR.26 |
>1 | Dacomitinib | Placebo | 720 | 63.5/65.5 | 23.4/24.4 | 1.00 (0.83–1.21) | NR | NR | 0.74 (0.56–0.98) | 1.13 (0.91–1.40) |
| Gregorc et al. (29) | Unknown | PROSE | >1 | Erlotinib | Pemetrexed or docetaxel | 263 | 66/64 | 32.4 | 1.15 (0.83–1.59) | Female vs. male 0.90 (0.64–1.27) |
Never vs. ever 0.80 (0.51–1.27) |
||
| Li et al. (30) | Wild-type | NR | >1 | Erlotinib | Pemetrexed | 123 | 54.3/55.1 | 14.7 | 1.01 (0.66–1.54) | 1.24 (0.73–2.11) | 0.64 (0.31–1.33) | 0.81 (0.34–1.90) | 1.10 (0.68–1.80) |
| Karachaliou et al. (31) | Unknown | EURTAC | 1 | Erlotinib | Cisplatin plus docetaxel or gemcitabine | 97 | NR | 49.4 | 0.71 (0.45–1.12) | Female vs. male 0.96 (0.59–1.56) | NR | NR | |
| Zhou et al. (32) | Mutation | OPTIMAL | 1 | Erlotinib | Gemcitabine plus carboplatin | 154 | 57/59 | 25.9 | 1.19 (0.83–1.71) | 1.31 (0.75–2.31) | 1.20 (0.74–1.93) | 1.44 (0.93–2.24) | 0.85 (0.44–1.64) |
| Wu et al. (33) | Mutation | ENSURE | 1 | Erlotinib | Gemcitabine plus cisplatin | 217 | 57.5/56 | 28.9/27.1 | 0.91 (0.63–1.31) | 0.86 (0.45–1.64) | 0.92 (0.59–1.44) | 0.99 (0.65–1.52) | 0.68 (0.32–1.43) |
| Yang et al. (34) | Mutation | LUX-Lung 3 and LUX-Lung 6 | 1 | Afatinib | Pemetrexed-cisplatin or gemcitabine-cisplatin | 631 | 60/59 | 41/33 | 0.81 (0.66–0.99) | 0.71 (0.51–0.99) | 0.84 (0.65–1.09) | 0.72 (0.57–0.92) | 1.02 (0.69–1.50) |
| Zhao et al. (35) | Unknown | INFORM | >1 | Gefitinib | Placebo | 296 | 55/55 | 17.8 | 0.88 (0.68–1.14) | 0.89 (0.64–1.24) | 0.93 (0.61–1.41) | 0.94 (0.65–1.35) | 0.82 (0.57–1.19) |
| Shi et al. (36) | Mutation | CONVINCE | 1 | Icotinib | Cisplatin plus pemetrexed | 285 | 56 | 39.6 | 1.03 (0.76–1.39) | 1.19 (0.69–2.04) | 0.96 (0.67–1.39) | 1.02 (0.72–1.45) | 1.20 (0.64–2.27) |
EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor; HR, hazard ratio; CI, confidence interval; NR, none reported.
In particular, Ellis et al. (28) did not report the HR for sex, and Kelly et al. (23) and Karachaliou et al. (31) did not report the HRs for smoking status that we needed. Lee et al. (21) Gregorc et al. (29) and Karachaliou et al. (31) reported the interaction HR of sex (female vs. male) in their subgroup analysis. Additionally, Lee et al. (21) and Gregorc et al. (29) reported the interaction HR of smoking status (never vs. ever smokers) in their subgroup analysis. For these interaction HRs with 95% CIs, we extracted and applied them in our meta-analysis directly (Table 1).
Primary Analysis
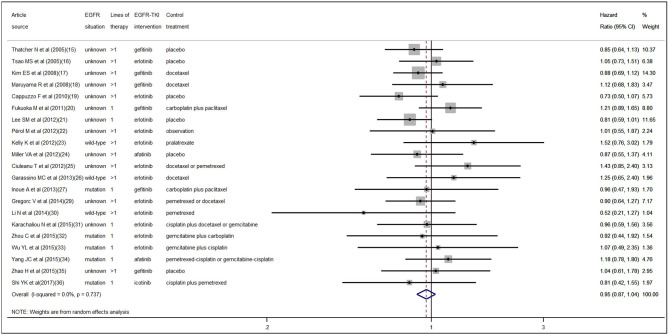
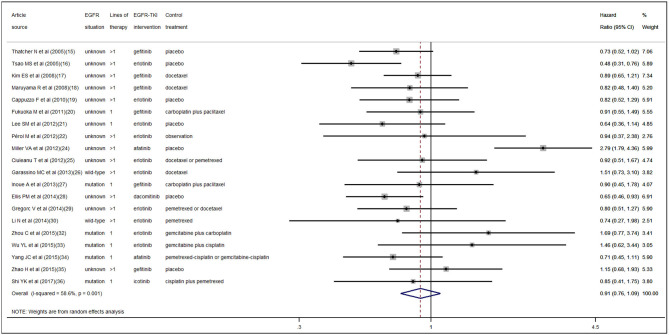
According to the pooled result of overall HRs, we found that the therapeutic effect of EGFR-TKI intervention was better than that of the control treatment (pooled HR 0.94, 95% CI 0.89–1.00, P = 0.05) in NSCLC (Figure S1 in the Supplementary Material). When pooling the interaction HRs of sex, the results showed no statistically significant efficacy difference in EGFR-TKI intervention between females and males (pooled HR 0.95, 95% CI 0.87–1.04, P = 0.30) (Figure 2). Similarly, based on the pooled interaction HR of smoking status, there was also no statistically significant efficacy difference in EGFR-TKI intervention between never smokers and ever smokers (pooled HR 0.91, 95% CI 0.76–1.09, P = 0.31) (Figure 3).
Figure 2.
Forest plot of the pooled analysis of the interaction hazard ratios of sex.
Figure 3.
Forest plot of the pooled analysis of the interaction hazard ratios of smoking status.
Heterogeneity and Publication Bias
Statistically significant interstudy heterogeneity was identified among both overall HRs (I-squared = 38.4%, P = 0.04) and smoking status interaction HRs (I-squared = 58.6%, P < 0.01) but not in sex interaction HRs (I-squared = 0.00%, P = 0.74) (Figure S1 in the Supplementary Material, Figures 2, 3). Both Egger and Begg tests indicated no evidence of publication bias.
Subgroup Analysis
We further performed subgroup analyses according to EGFR status (unknown, wild-type, and mutation), lines of therapy (>1 and 1), EGFR-TKI intervention (gefitinib, erlotinib, and others), and control treatment (placebo, chemotherapy, and others). According to the pooled interaction HRs of sex, we found a statistically significant OS advantage for females compared with males only for the EGFR-TKI intervention compared with the control placebo treatment (pooled HR 0.86, 95% CI 0.75–1.00, P = 0.04), while the other groups showed no statistically significant difference (Table 2). Furthermore, as shown in Table 3, no statistically significant difference was determined in any of the subgroups according to the pooled interaction HRs of smoking status.
Table 2.
Differences in efficacy of EGFR-TKI therapy in females and males by subgroup.
| Subgroups | Number of trials | Pooled interaction HR | P-interaction | |
|---|---|---|---|---|
| Overall | 21 | 0.95 (0.87–1.04) | 0.30 | |
| EGFR situation | Unknown | 13 | 0.94 (0.85–1.03) | 0.19 |
| Wild-type | 3 | 1.06 (0.60–1.89) | 0.83 | |
| Mutation | 5 | 1.02 (0.78–1.34) | 0.86 | |
| Lines of therapy | >1 | 13 | 0.94 (0.84–1.05) | 0.26 |
| 1 | 8 | 0.98 (0.84–1.15) | 0.82 | |
| EGFR-TKI intervention | Gefitinib | 6 | 0.97 (0.84–1.11) | 0.64 |
| Erlotinib | 12 | 0.93 (0.82–1.07) | 0.31 | |
| Others | 3 | 0.98 (0.74–1.30) | 0.91 | |
| Control treatment | Placebo | 6 | 0.86 (0.75–1.00) | 0.04 |
| Chemotherapy | 13 | 1.01 (0.89–1.14) | 0.89 | |
| Others | 2 | 1.21 (0.77–1.91) | 0.42 |
Table 3.
Differences in efficacy of EGFR-TKI therapy according to smoking status by subgroup.
| Subgroups | Number of trials | Pooled interaction HR | P-interaction | |
|---|---|---|---|---|
| Overall | 20 | 0.91 (0.76–1.09) | 0.31 | |
| EGFR situation | Unknown | 13 | 0.87 (0.69–1.10) | 0.24 |
| Wild-type | 2 | 1.15 (0.58–2.27) | 0.68 | |
| Mutation | 5 | 0.95 (0.69–1.32) | 0.77 | |
| Lines of therapy | >1 | 13 | 0.91 (0.71–1.17) | 0.46 |
| 1 | 7 | 0.87 (0.69–1.10) | 0.25 | |
| EGFR-TKI intervention | Gefitinib | 6 | 0.86 (0.73–1.03) | 0.10 |
| Erlotinib | 10 | 0.86 (0.67–1.10) | 0.23 | |
| Others | 4 | 1.03 (0.50–2.11) | 0.95 | |
| Control treatment | Placebo | 8 | 0.88 (0.59–1.29) | 0.50 |
| Chemotherapy | 12 | 0.91 (0.77–1.06) | 0.22 |
Discussion
OS and PFS are the main endpoints in clinical trials of cancer. It is well-known that PFS is not in line with OS in many cases, (37) and in such cases, cancer patients may not obtain benefit from OS even though they have an improved PFS. To reduce time, save costs, and improve drug development efficiency, an increasing number of cancer clinical trials have set the research endpoint to PFS. However, compared with PFS, OS is simple, reliable, straightforward, clear, and accurate in the evaluation of the endpoint of cancer patients. Therefore, more attention should be paid to OS. In our current study, we studied the impact of sex and smoking status on the efficacy of EGFR-TKI therapy in terms of OS in NSCLC patients and obtained meaningful findings.
We first demonstrated the advantage in OS for NSCLC patients who received EGFR-TKI intervention compared with other systemic therapies. Thereafter, we found no significant OS differences for EGFR-TKI intervention between the sexes and smoking status compared with other systemic therapies. Finally, in the subgroup analyses, when compared with placebo, we demonstrated that female NSCLC patients who received EGFR-TKI therapy had a longer OS than males. However, smoking status showed no significant effect on the efficacy of EGFR-TKI treatment in terms of the OS of NSCLC patients in all of our subgroup analyses.
In recent years, significant sex-based differences in biology, epidemiology and treatment responses have become evident (38). There are sex-related differences in the clinicopathological characteristics of NSCLC patients, and female sex is a separate advantage survival prognostic factor (4). Consistently, after adjustments for other prognostic factors, males with NSCLC have a poorer prognosis than females (39). As NSCLC is considered a sex-related disease, further investigation is warranted to advance the treatment of NSCLC patients.
In a previous meta-analysis conducted by Lee et al. (40), EGFR-TKI treatment significantly prolonged PFS for female compared with male NSCLC patients with EGFR mutations. Afterwards, another meta-analysis also performed by Lee et al. (41) further concluded that, there was no difference in OS between EGFR-TKI and chemotherapy, as well as no difference in OS between female and male EGFR mutation-positive NSCLC patients. However, in our current study, we found that NSCLC patients who received EGFR-TKI intervention had longer OS than those who received other systemic therapies, although no significant OS differences for EGFR-TKI intervention were found between the sexes. In the subgroup analysis of the placebo control group, we demonstrated that female NSCLC patients who received EGFR-TKI therapy had a longer OS than males.
For the studies on chemotherapy in patients with NSCLC, Wakelee et al. (42) reported that women had a 1.9-month statistically significant improvement in OS compared with men. Wheatley-Price et al. (43) also concluded that females had a higher response rate to chemotherapy and a longer OS than males. For our current study, when the control group was treated with chemotherapy, it significantly biased our judgment of the difference in efficacy of EGFR-TKI between the sexes. When we removed the interference of chemotherapy and other factors in the subgroup analysis and compared EGFR-TKIs with the standard placebo, we found that the efficacy of EGFR-TKIs in female patients was significantly better than that in male patients. These results indicate that there is indeed a sex difference in the efficacy of EGFR-TKIs in patients with NSCLC.
It is well-known that tobacco smoking is an important cause of the development and progression of NSCLC. The incidence of EGFR mutations in NSCLC differs according to smoking history (44). EGFR mutations are highly prevalent in never smokers with NSCLC (45). Current smoking is an independent poor prognostic factor for survival for advanced non-squamous NSCLC patients without EGFR mutations who undergo pemetrexed continuation maintenance therapy (46). In addition, according to a recently reported large population-based study, NSCLC in never smokers was found to be clinically different from smoking-associated NSCLC, and the study also concluded that the OS in never-smokers was longer than that in smokers (47).
The impact of smoking status on the efficacy of EGFR-TKIs in terms of PFS in NSCLC is contradictory according to previous meta-analyses (9, 10, 40). For the meta-analyses that studied OS, Sohn et al. (48) reported that, compared with chemotherapy or placebo, receiving EGFR-TKI therapy appeared to show longer OS among patients with NSCLC for never smokers than that seen in ever smokers. In contrast, Lee et al. (41) found no difference in OS according to smoking status for NSCLC patients who underwent EGFR-TKI treatment compared with chemotherapy. We consider this contradictory phenomenon to be due to the different inclusion criteria and the different number of included studies. However, in our current study, we found no significant OS differences for EGFR-TKI intervention compared with other systemic therapies between different smoking statuses in NSCLC patients, and further subgroup analyses also showed that smoking status had no significant effect on the efficacy of EGFR-TKI treatment.
In summary, since sex and smoking status are the two main clinical features of lung cancer, our current research has important guiding significance for the clinical treatment of lung cancer. Our results suggest that we do not need to worry that smoking status will affect the efficacy of EGFR-TKIs and that EGFR-TKIs will have better efficacy in female patients than in male patients. However, on the other hand, the efficacy of EGFR-TKIs in male patients is not so ideal, indicating that more treatment options for male lung cancer patients need to be further developed in the future.
Our current study has several limitations. First, as a meta-analysis, it relies on published results rather than the individual data of patients. Second, those excluded studies that lack published sex and smoking status subgroup analysis data may also contain potential differences. Finally, aside from sex and smoking status, differences in OS outcomes may be influenced by other non-pharmacological factors.
Conclusions
Two main conclusions can be drawn from our current meta-analysis. The first is that the efficacy of EGFR-TKI therapy for NSCLC patients is sex-dependent, and females have a longer OS advantage than males. The second point is that smoking status has no effect on the efficacy of EGFR-TKI therapy in terms of the OS of NSCLC patients.
Data Availability Statement
All datasets generated for this study are included in the article/Supplementary Material.
Author Contributions
JX and QC: concept and design. JX and LZ: acquisition, analysis or interpretation of data, and statistical analysis. JX and BH: drafting of the manuscript. JX, BH, and QC: critical revision of the manuscript for important intellectual content. BH and QC: administrative, technical or material support, and study supervision. All authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Glossary
Abbreviations
- EGFR
epidermal growth factor receptor
- EGFR-TKIs
epidermal growth factor receptor-tyrosine kinase inhibitors
- NSCLC
non-small-cell lung cancer
- OS
overall survival
- HR
hazard ratio
- CI
confidence interval
- PFS
progression-free survival.
Footnotes
Funding. This work was supported by the National Natural Science Foundation of China (Nos. 81572284 and 81770045).
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2020.01531/full#supplementary-material
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. (2019) 69:7–34. 10.3322/caac.21551 [DOI] [PubMed] [Google Scholar]
- 2.Reck M, Popat S, Reinmuth N, De Ruysscher D, Kerr KM, Peters S. Metastatic non-small-cell lung cancer (NSCLC): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. (2014) 25(Suppl. 3):iii27–39. 10.1093/annonc/mdu199 [DOI] [PubMed] [Google Scholar]
- 3.Ferketich AK, Niland JC, Mamet R, Zornosa C, D'Amico TA, Ettinger DS, et al. Smoking status and survival in the national comprehensive cancer network non-small cell lung cancer cohort. Cancer. (2013) 119:847–53. 10.1002/cncr.27824 [DOI] [PubMed] [Google Scholar]
- 4.Hsu LH, Chu NM, Liu CC, Tsai SY, You DL, Ko JS, et al. Sex-associated differences in non-small cell lung cancer in the new era: is gender an independent prognostic factor? Lung Cancer. (2009) 66:262–7. 10.1016/j.lungcan.2009.01.020 [DOI] [PubMed] [Google Scholar]
- 5.Liu TC, Jin X, Wang Y, Wang K. Role of epidermal growth factor receptor in lung cancer and targeted therapies. Am J Cancer Res. (2017) 7:187–202. [PMC free article] [PubMed] [Google Scholar]
- 6.Tan CS, Kumarakulasinghe NB, Huang YQ, Ang YLE, Choo JR, Goh BC, et al. Third generation EGFR TKIs: current data and future directions. Mol Cancer. (2018) 17:29. 10.1186/s12943-018-0778-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang S, Song Y, Liu D. EAI045: The fourth-generation EGFR inhibitor overcoming T790M and C797S resistance. Cancer Lett. (2017) 385:51–4. 10.1016/j.canlet.2016.11.008 [DOI] [PubMed] [Google Scholar]
- 8.Pinto JA, Vallejos CS, Raez LE, Mas LA, Ruiz R, Torres-Roman JS, et al. Gender and outcomes in non-small cell lung cancer: an old prognostic variable comes back for targeted therapy and immunotherapy? ESMO Open. (2018) 3:e000344. 10.1136/esmoopen-2018-000344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hasegawa Y, Ando M, Maemondo M, Yamamoto S, Isa SI, Saka H, et al. The role of smoking status on the progression-free survival of non-small cell lung cancer patients harboring activating epidermal growth factor receptor (EGFR) mutations receiving first-line EGFR tyrosine kinase inhibitor vs. platinum doublet chemotherapy: a meta-analysis of prospective randomized trials. Oncologist. (2015) 20:307–15. 10.1634/theoncologist.2014-0285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang Y, Kang S, Fang W, Hong S, Liang W, Yan Y, et al. Impact of smoking status on EGFR-TKI efficacy for advanced non–small-cell lung cancer in EGFR mutants: a meta-analysis. Clin Lung Cancer. (2015) 16:144–51.e1. 10.1016/j.cllc.2014.09.008 [DOI] [PubMed] [Google Scholar]
- 11.Wallis CJD, Butaney M, Satkunasivam R, Freedland SJ, Patel SP, Hamid O, et al. Association of patient sex with efficacy of immune checkpoint inhibitors and overall survival in advanced cancers: a systematic review and meta-analysis. JAMA Oncol. (2019) 5:529–36. 10.1001/jamaoncol.2018.5904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Conforti F, Pala L, Bagnardi V, De Pas T, Martinetti M, Viale G, et al. Cancer immunotherapy efficacy and patients' sex: a systematic review and meta-analysis. Lancet Oncol. (2018) 19:737–46. 10.1016/S1470-2045(18)30261-4 [DOI] [PubMed] [Google Scholar]
- 13.Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. (2009) 62:1006–12. 10.1016/j.jclinepi.2009.06.005 [DOI] [PubMed] [Google Scholar]
- 14.Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. (1996) 17:1–12. 10.1016/0197-2456(95)00134-4 [DOI] [PubMed] [Google Scholar]
- 15.Thatcher N, Chang A, Parikh P, Rodrigues Pereira J, Ciuleanu T, von Pawel J, et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). Lancet. (2005) 366:1527–37. 10.1016/S0140-6736(05)67625-8 [DOI] [PubMed] [Google Scholar]
- 16.Tsao MS, Sakurada A, Cutz JC, Zhu CQ, Kamel-Reid S, Squire J, et al. Erlotinib in lung cancer - molecular and clinical predictors of outcome. N Engl J Med. (2005) 353:133–44. 10.1056/NEJMoa050736 [DOI] [PubMed] [Google Scholar]
- 17.Kim ES, Hirsh V, Mok T, Socinski MA, Gervais R, Wu YL, et al. Gefitinib vs. docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomised phase III trial. Lancet. (2008) 372:1809–18. 10.1016/S0140-6736(08)61758-4 [DOI] [PubMed] [Google Scholar]
- 18.Maruyama R, Nishiwaki Y, Tamura T, Yamamoto N, Tsuboi M, Nakagawa K, et al. Phase III study, V-15–32, of gefitinib vs. docetaxel in previously treated Japanese patients with non-small-cell lung cancer. J Clin Oncol. (2008) 26:4244–52. 10.1200/JCO.2007.15.0185 [DOI] [PubMed] [Google Scholar]
- 19.Cappuzzo F, Ciuleanu T, Stelmakh L, Cicenas S, Szczesna A, Juhasz E, et al. Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: a multicentre, randomised, placebo-controlled phase 3 study. Lancet Oncol. (2010) 11:521–9. 10.1016/S1470-2045(10)70112-1 [DOI] [PubMed] [Google Scholar]
- 20.Fukuoka M, Wu YL, Thongprasert S, Sunpaweravong P, Leong SS, Sriuranpong V, et al. Biomarker analyses and final overall survival results from a phase III, randomized, open-label, first-line study of gefitinib vs. carboplatin/paclitaxel in clinically selected patients with advanced non-small-cell lung cancer in Asia (IPASS). J Clin Oncol. (2011) 29:2866–74. 10.1200/JCO.2010.33.4235 [DOI] [PubMed] [Google Scholar]
- 21.Lee SM, Khan I, Upadhyay S, Lewanski C, Falk S, Skailes G, et al. First-line erlotinib in patients with advanced non-small-cell lung cancer unsuitable for chemotherapy (TOPICAL): a double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. (2012) 13:1161–70. 10.1016/S1470-2045(12)70412-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Perol M, Chouaid C, Perol D, Barlesi F, Gervais R, Westeel V, et al. Randomized, phase III study of gemcitabine or erlotinib maintenance therapy vs. observation, with predefined second-line treatment, after cisplatin-gemcitabine induction chemotherapy in advanced non-small-cell lung cancer. J Clin Oncol. (2012) 30:3516–24. 10.1200/JCO.2011.39.9782 [DOI] [PubMed] [Google Scholar]
- 23.Kelly K, Azzoli CG, Zatloukal P, Albert I, Jiang PY, Bodkin D, et al. Randomized phase 2b study of pralatrexate vs. erlotinib in patients with stage IIIB/IV non-small-cell lung cancer (NSCLC) after failure of prior platinum-based therapy. J Thorac Oncol. (2012) 7:1041–8. 10.1097/JTO.0b013e31824cc66c [DOI] [PubMed] [Google Scholar]
- 24.Miller VA, Hirsh V, Cadranel J, Chen YM, Park K, Kim SW, et al. Afatinib vs. placebo for patients with advanced, metastatic non-small-cell lung cancer after failure of erlotinib, gefitinib, or both, and one or two lines of chemotherapy (LUX-Lung 1): a phase 2b/3 randomised trial. Lancet Oncol. (2012) 13:528–38. 10.1016/S1470-2045(12)70087-6 [DOI] [PubMed] [Google Scholar]
- 25.Ciuleanu T, Stelmakh L, Cicenas S, Miliauskas S, Grigorescu AC, Hillenbach C, et al. Efficacy and safety of erlotinib vs. chemotherapy in second-line treatment of patients with advanced, non-small-cell lung cancer with poor prognosis (TITAN): a randomised multicentre, open-label, phase 3 study. Lancet Oncol. (2012) 13:300–8. 10.1016/S1470-2045(11)70385-0 [DOI] [PubMed] [Google Scholar]
- 26.Garassino MC, Martelli O, Broggini M, Farina G, Veronese S, Rulli E, et al. Erlotinib vs. docetaxel as second-line treatment of patients with advanced non-small-cell lung cancer and wild-type EGFR tumours (TAILOR): a randomised controlled trial. Lancet Oncol. (2013) 14:981–8. 10.1016/S1470-2045(13)70310-3 [DOI] [PubMed] [Google Scholar]
- 27.Inoue A, Kobayashi K, Maemondo M, Sugawara S, Oizumi S, Isobe H, et al. Updated overall survival results from a randomized phase III trial comparing gefitinib with carboplatin-paclitaxel for chemo-naive non-small cell lung cancer with sensitive EGFR gene mutations (NEJ002). Ann Oncol. (2013) 24:54–9. 10.1093/annonc/mds214 [DOI] [PubMed] [Google Scholar]
- 28.Ellis PM, Shepherd FA, Millward M, Perrone F, Seymour L, Liu G, et al. Dacomitinib compared with placebo in pretreated patients with advanced or metastatic non-small-cell lung cancer (NCIC CTG BR.26): a double-blind, randomised, phase 3 trial. Lancet Oncol. (2014) 15:1379–88. 10.1016/S1470-2045(14)70472-3 [DOI] [PubMed] [Google Scholar]
- 29.Gregorc V, Novello S, Lazzari C, Barni S, Aieta M, Mencoboni M, et al. Predictive value of a proteomic signature in patients with non-small-cell lung cancer treated with second-line erlotinib or chemotherapy (PROSE): a biomarker-stratified, randomised phase 3 trial. Lancet Oncol. (2014) 15:713–21. 10.1016/S1470-2045(14)70162-7 [DOI] [PubMed] [Google Scholar]
- 30.Li N, Ou W, Yang H, Liu QW, Zhang SL, Wang BX, et al. A randomized phase 2 trial of erlotinib vs. pemetrexed as second-line therapy in the treatment of patients with advanced EGFR wild-type and EGFR FISH-positive lung adenocarcinoma. Cancer. (2014) 120:1379–86. 10.1002/cncr.28591 [DOI] [PubMed] [Google Scholar]
- 31.Karachaliou N, Mayo-de las Casas C, Queralt C, de Aguirre I, Melloni B, Cardenal F, et al. Association of EGFR L858R mutation in circulating free DNA with survival in the EURTAC Trial. JAMA Oncol. (2015) 1:149–57. 10.1001/jamaoncol.2014.257 [DOI] [PubMed] [Google Scholar]
- 32.Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C, et al. Final overall survival results from a randomised, phase III study of erlotinib vs. chemotherapy as first-line treatment of EGFR mutation-positive advanced non-small-cell lung cancer (OPTIMAL, CTONG-0802). Ann Oncol. (2015) 26:1877–83. 10.1093/annonc/mdv276 [DOI] [PubMed] [Google Scholar]
- 33.Wu YL, Zhou C, Liam CK, Wu G, Liu X, Zhong Z, et al. First-line erlotinib vs. gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small-cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol. (2015) 26:1883–9. 10.1093/annonc/mdv270 [DOI] [PubMed] [Google Scholar]
- 34.Yang JC, Wu YL, Schuler M, Sebastian M, Popat S, Yamamoto N, et al. Afatinib vs. cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol. (2015) 16:141–51. 10.1016/S1470-2045(14)71173-8 [DOI] [PubMed] [Google Scholar]
- 35.Zhao H, Fan Y, Ma S, Song X, Han B, Cheng Y, et al. Final overall survival results from a phase III, randomized, placebo-controlled, parallel-group study of gefitinib vs. placebo as maintenance therapy in patients with locally advanced or metastatic non-small-cell lung cancer (INFORM; C-TONG 0804). J Thorac Oncol. (2015) 10:655–64. 10.1097/JTO.0000000000000445 [DOI] [PubMed] [Google Scholar]
- 36.Shi YK, Wang L, Han BH, Li W, Yu P, Liu YP, et al. First-line icotinib vs. cisplatin/pemetrexed plus pemetrexed maintenance therapy for patients with advanced EGFR mutation-positive lung adenocarcinoma (CONVINCE): a phase 3, open-label, randomized study. Ann Oncol. (2017) 28:2443–50. 10.1093/annonc/mdx359 [DOI] [PubMed] [Google Scholar]
- 37.Amir E, Seruga B, Kwong R, Tannock IF, Ocana A. Poor correlation between progression-free and overall survival in modern clinical trials: are composite endpoints the answer? Eur J Cancer. (2012) 48:385–8. 10.1016/j.ejca.2011.10.028 [DOI] [PubMed] [Google Scholar]
- 38.Donington JS, Colson YL. Sex and gender differences in non-small cell lung cancer. Semin Thorac Cardiovasc Surg. (2011) 23:137–45. 10.1053/j.semtcvs.2011.07.001 [DOI] [PubMed] [Google Scholar]
- 39.Radkiewicz C, Dickman PW, Johansson ALV, Wagenius G, Edgren G, Lambe M. Sex and survival in non-small cell lung cancer: A nationwide cohort study. PLoS ONE. (2019) 14:e0219206. 10.1371/journal.pone.0219206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lee CK, Wu YL, Ding PN, Lord SJ, Inoue A, Zhou C, et al. Impact of specific epidermal growth factor receptor (EGFR) mutations and clinical characteristics on outcomes after treatment with EGFR tyrosine kinase inhibitors vs. chemotherapy in EGFR-mutant lung cancer: a meta-analysis. J Clin Oncol. (2015) 33:1958–65. 10.1200/JCO.2014.58.1736 [DOI] [PubMed] [Google Scholar]
- 41.Lee CK, Davies L, Wu YL, Mitsudomi T, Inoue A, Rosell R, et al. Gefitinib or Erlotinib vs chemotherapy for EGFR mutation-positive lung cancer: individual patient data meta-analysis of overall survival. J Natl Cancer Inst. (2017) 109:djw279 10.1093/jnci/djw279 [DOI] [PubMed] [Google Scholar]
- 42.Wakelee HA, Wang W, Schiller JH, Langer CJ, Sandler AB, Belani CP, et al. Survival differences by sex for patients with advanced non-small cell lung cancer on Eastern cooperative oncology group trial 1594. J Thorac Oncol. (2006) 1:441–6. 10.1097/01243894-200606000-00011 [DOI] [PubMed] [Google Scholar]
- 43.Wheatley-Price P, Blackhall F, Lee SM, Ma C, Ashcroft L, Jitlal M, et al. The influence of sex and histology on outcomes in non-small-cell lung cancer: a pooled analysis of five randomized trials. Ann Oncol. (2010) 21:2023–8. 10.1093/annonc/mdq067 [DOI] [PubMed] [Google Scholar]
- 44.Ren JH, He WS, Yan GL, Jin M, Yang KY, Wu G. EGFR mutations in non-small-cell lung cancer among smokers and non-smokers: a meta-analysis. Environ Mol Mutagen. (2012) 53:78–82. 10.1002/em.20680 [DOI] [PubMed] [Google Scholar]
- 45.Chapman AM, Sun KY, Ruestow P, Cowan DM, Madl AK. Lung cancer mutation profile of EGFR, ALK, and KRAS: meta-analysis and comparison of never and ever smokers. Lung Cancer. (2016) 102:122–34. 10.1016/j.lungcan.2016.10.010 [DOI] [PubMed] [Google Scholar]
- 46.Lin L, Zhao J, Hu J, Zou G, Huang F, Han J, et al. Current smoking has a detrimental effect on survival for epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK) negative advanced non-squamous non-small cell lung cancer (NSCLC) patients treated with pemetrexed continuation maintenance. J Cancer. (2018) 9:2140–6. 10.7150/jca.24872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lofling L, Karimi A, Sandin F, Bahmanyar S, Kieler H, Lambe M, et al. Clinical characteristics and survival in non-small cell lung cancer patients by smoking history: a population-based cohort study. Acta Oncol. (2019) 58:1618–27. 10.1080/0284186X.2019.1638521 [DOI] [PubMed] [Google Scholar]
- 48.Sohn HS, Kwon JW, Shin S, Kim HS, Kim H. Effect of smoking status on progression-free and overall survival in non-small cell lung cancer patients receiving erlotinib or gefitinib: a meta-analysis. J Clin Pharm Ther. (2015) 40:661–71. 10.1111/jcpt.12332 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All datasets generated for this study are included in the article/Supplementary Material.