Abstract
We are in the midst of the global pandemic. Though acute respiratory coronavirus (SARS-COV2) that leads to COVID-19 infects people of all ages, severe symptoms and mortality occur disproportionately in older adults. Geroscience interventions that target biological aging could decrease risk across multiple age-related diseases and improve outcomes in response to infectious disease. This offers hope for a new host-directed therapeutic approach that could (i) improve outcomes following exposure or shorten treatment regimens; (ii) reduce the chronic pathology associated with the infectious disease and subsequent comorbidity, frailty, and disability; and (iii) promote development of immunological memory that protects against relapse or improves response to vaccination. We review the possibility of this approach by examining available evidence in metformin: a generic drug with a proven safety record that will be used in a large-scale multicenter clinical trial. Though rigorous translational research and clinical trials are needed to test this empirically, metformin may improve host immune defenses and confer protection against long-term health consequences of infectious disease, age-related chronic diseases, and geriatric syndromes.
Keywords: Metformin, Aging, Geroscience, Immunity, COVID-19
Geroscience in a time of global pandemic
The promise of geroscience is that interventions that target biological aging could fortify organismal resilience and delay the onset or lessen the severity of multiple age-related diseases en masse. Never has this promise been more critical than now, in the midst of the global pandemic. Though acute respiratory coronavirus (SARS-COV2) infects people of all ages, severe symptoms and mortality occur disproportionately in older adults. In the USA, 8 out of 10 deaths occurred in persons over the age of 65 years [1], and in Britain, persons above age 80 years were ~ 180 fold more likely to die that those in their 40s [2]. Severity and complications also increase nonlinearly with age. In China, the odds of hospitalization at ages 40–49 years were 4.3% but nearly doubled to 8.2%, for 50–59-year-olds, and continued to climb for each decade of advancing age up to 18.4% in persons over the age of 80 years [3, 4].
This trend is not unique to COVID-19. It is well known that older persons generally have greater susceptibility to infections than younger adults due to age-related changes in cell-mediated immunity, underlying chronic disease, and geriatric syndromes [5, 6]. Moreover, the pattern of exponential increase in mortality rate with age in COVID-19 exhibits a doubling time estimated between 6.6 to 9 years (USA, China, Italy); this is mirrored by the exponential increase in age-related risk of mortality from all causes (doubling time of ~ 9 years, USA), and cause specific which ranges from 4.7 years (chronic obstructive pulmonary disease, COPD) to 7.1 years (diabetes mellitus, DM) [7]. This suggests contributions of age-related declines in immune and inflammatory responses in harmony with systemic age-related physiologic dysregulation and comorbidity [3, 8]. Chronically elevated inflammation and dysregulated cell–cell communication constitute examples of biological hallmarks of aging and represent both contributors to and consequences of varied chronic conditions that are independently associated with increased COVID-19 vulnerability. For example, the presence of multiple chronic conditions or comorbidities such as diabetes, chronic kidney disease, and heart failure are associated with proinflammatory cytokines and other biomarkers of biological hallmarks of aging [9–11], and have been shown to be independently associated with severe COVID-19 illness and death [12–14] [15, 16].
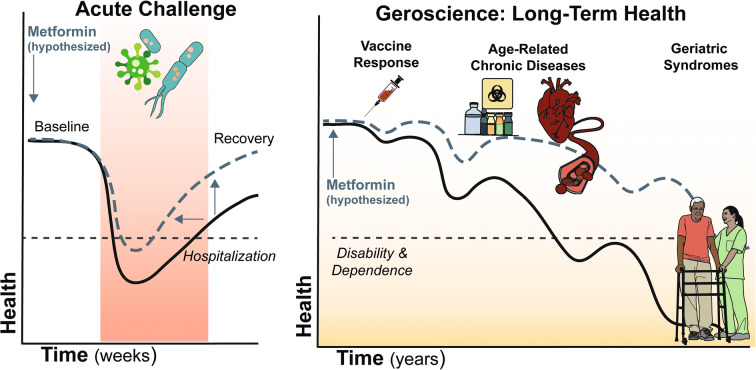
Therefore, while virus-specific treatments and vaccines are critically needed, other complementary strategies are also needed to address increased vulnerability to severe COVID-19 during this window of vulnerability as vaccines and antiviral agents are being developed. To that end, treatment of the biological underpinnings which render older adults more vulnerable may not only improve outcomes during this COVID-19 pandemic, but could also offer benefits in terms of future pandemics involving other pathogens, while also impacting positively the onset and progression of varied acute and chronic diseases [17]. Geroscience interventions that target biological aging could decrease risk across multiple age-related diseases and improve outcomes in response to infectious disease. This offers hope for a new host-directed therapeutic approach that could (i) improve outcomes following exposure or shorten treatment regimens; (ii) reduce the chronic pathology associated with the infectious disease and subsequent comorbidity, frailty, and disability; and (iii) promote development of immunological memory that protects against relapse or improves response to vaccination [18, 19] (Fig. 1).
Fig. 1.
Geroscience and immune resilience: acute and long-term health. Metformin use (blue line) initiated prior to acute illness like COVID-19 (left most panel) may improve resilience resulting in fewer events like hospitalization (dotted line), briefer recovery time, and improved return to baseline health compared to non-use (black line). Greater long-term health effects could be observed as improved response to vaccine and prevention or delay of age-related diseases and geriatric syndromes
Translational interventions in geroscience
Preclinical interventions testing programs show that biological aging pathways can be therapeutically targeted. Perhaps, the most robust intervention is caloric restriction, which has been shown to prolong the lifespan of experimental animal models such as nematodes, flies, and mice [20, 21] and may improve immune response [22]. In addition, promising pharmacological and nutraceutical candidates have been identified. In most cases, median lifespan extension is accompanied by improved health and ability of preclinical models to withstand both chronic and acute challenges [23, 24]. A select subset of these have intriguing effects in humans on overall health and immune function, including acarbose [25], and mTORC1 inhibitors rapamycin, everolimus, and RTB101 [26]. For example, mice treated with 6 weeks of rapamycin mounted a greater response to influenza vaccine than age-matched control mice [27], and two phase II studies of mTORC inhibitors demonstrated improved antibody responses to an influenza vaccine and decreased rates of upper respiratory tract infections by varied pathogens in healthy community–dwelling older adults [26, 28]. Phase III trial of mTORC1 inhibitor RTB101 did not meet its primary endpoint for prevention of respiratory illness. However, two new phase I-II trials with mTOR inhibitor sirolimus are now in progress in hospitalized patients with COVID-19 pneumonia (NCT04341675 and NCT04371640) and those with RTB101 in the nursing home setting (NCT04409327).
A critical obstacle for larger clinical trials and widespread clinical application of any drug targeting aging is safety, tolerability, and expense. So far, only a select couple of drugs can overcome these translational barriers. One such drug is metformin. Though metformin offers modest improvements in lifespan in preclinical interventions testing [29], it targets molecular and cellular drivers of aging which may exert positive effects more broadly on health span. Moreover, metformin (i) is a generic and relatively inexpensive medication, (ii) has an excellent safety record for over 5 decades, and (iii) can be generally well tolerated with long-term use in a large population of aging adults. Metformin will be tested in a first-of-its-kind trial designed to create a regulatory pathway for aging as a target for drug development, the phase III multicenter randomized double-blind placebo-controlled trial Targeting Aging with MEtformin (TAME) [30, 31]. With recruitment start coinciding with what is anticipated to be the waning of the COVID-19 global pandemic, TAME provides an excellent platform to probe preventive effects on age-related chronic disease and long-term consequences of infectious disease and vaccine response in older persons. In light of the recent global pandemic, we provide a discussion of metformin in immune aging and infectious disease focusing on clinical evidence.
A brief history of metformin and aging
Metformin is a biguanide and is currently the most widely prescribed oral hypoglycemic medication for type 2 diabetes treatment or prevention prescribed worldwide. However, its history dates back to the seventeenth century, when metformin-like guanidine compounds found in traditional herbal medicine Galega officinalis were used to treat conditions like plague, fever, and snake bites. Metformin was rediscovered in the 1940s, in a search for antimalarial agents, and in 1949, metformin was found to be helpful in treating a local influenza outbreak in the Philippines [32, 33]. Its immune effects were accompanied by the beneficial side effect of lowered blood glucose in some of the influenza patients. In the 1950s, the physician Dr. Jean Stern translated this glucose-lowering potential into diabetes treatments, though metformin was not finally approved by the Food and Drug Administration (FDA) as an antidiabetic until 1995.
Interest in metformin’s clinical effects beyond glucose regulation originated from epidemiologic studies, which observed a reduction in cardiovascular risk and all-cause mortality in individuals with diabetes treated with metformin in the Diabetes Prevention Program (DPP) and UK Prospective Diabetes Study (UKPDS) [34, 35]. Since then, there is growing interest in metformin’s clinical benefits beyond diabetes (reviewed in [30]) in diseases ranging from gestational diabetes, polycystic ovary syndrome (PCOS), and human immunodeficiency virus (HIV)–associated lipodystrophy, to dementia-type neurodegenerative disorders and certain cancers in individuals with and without type 2 diabetes.
Metformin attenuates hallmarks of biological aging
Metformin has emerged as a leading candidate in clinical trials in aging and age-related acute and chronic diseases given its cellular effects which are distinct from its effects on glucose metabolism in diabetics. Kulkarni et al. [36] recently reviewed metformin’s effects on the biological hallmarks of aging, with supporting evidence from C. elegans, drosophila, and rodents. Metformin acts through a few widely accepted cellular and molecular mechanisms that are highly interdependent and implicated metformin’s downstream effects on immune response, chronic diseases, and geriatric syndromes (Table 1). Chief among these cellular mechanisms are metformin’s actions on the mitochondrion as a weak inhibitor of complex I of the mitochondrial electron transport chain, activation of the energy sensor AMP–activated protein kinase, inhibition of mTORC1, and regulation of inflammatory cytokine signaling [30, 36]. Converging evidence also implicates lysosomes [37], gut microbiome [38], and epigenetic modulation as mediators of metformin’s actions on biological aging [39, 40]. Importantly, metformin is not proposed to exert action by any one single or distinct mechanism but through multiple mechanisms. Moreover, it is precisely this plurality of pleiotropic cellular effects that makes metformin a valuable tool to achieve downstream effects on immunoprotection, and multiple age-related diseases and geriatric conditions.
Table 1.
Summary of metformin’s mechanisms in attenuating hallmarks of aging. Metformin has been postulated to exert gerotherapeutic effects through several molecular pathways related to biological hallmarks of aging [36]
| Aging hallmarks | Metformin’s effects on key targets and pathways |
|---|---|
| Altered intercellular communication |
• Antiinflammatory response and immunomodulation ↓ pro-inflammatory cytokines (IL-6, IL-1β, CXCL1/2) [59] ↓ NF-κB and IKKα/β signaling [111] • Regulation of the gut microbiota by altering microbial folate Anti-inflammatory response via ↑ Akkermansia, ↑ Bacteroides, ↑ Butyricimonas and ↑ Parabacteroides genera [112, 113] Methionine restriction [114] |
| Deregulated nutrient sensing |
• Direct targeting of key energy sensors and modulation of nutrient sensing pathways ↑ AMPK [37, 115, 116], ↑ SIRT1 [117, 118] ↓ mTORC1 (via ↓ Rad-GTPase, ↑ TSC2, ↑ REDD1) [119–122] ↓ Insulin and IGF-1 signaling [123] |
| Genomic instability | • Genome protective effects with ↑ DNA-damage-like response and ↑ DNA repair and regulation of ATM-protein kinases [124–126] |
| Loss of proteostasis |
• Increased autophagy and rescue of protein misfolding ↑ LAMP-1 and ↑ Beclin-1 [127, 128] ↑ CEBPD-mediated autophagy [129] |
| Mitochondrial dysfunction |
• Inhibition of mitochondrial complex I of the electron transport chain • Lowering mitochondria-induced oxidative stress [130–132] ↓ Endogenous production of ROS [130–133] • Improved mitochondrial biogenesis via ↑ PGC-1α [134] |
| Stem cell exhaustion |
• Improved stem cell rejuvenation capacity [135–137] • Delayed stem cell aging [138] |
| Epigenetic alterations |
• Transcriptional regulation via histone modifications, DNA methylation and miRNAs [40] Phosphorylation of Histone Acetyl Transferases (HATs) ↓ Class II Histone Deacetylases (HDACs) ↑ DICER1 [139] |
| Telomere attrition | • Reduction of telomere shortening and ↑ telomeric repeat containing RNA (TERRA) [140] |
| Cellular senescence |
• Suppression of senescence-associated secretome ↓ p16, ↓ p21 and RNA-levels of SASP hallmarks [111, 139, 141] ↓ β-gal activity and ↓proinflammatory senescence [142] |
Mechanisms: metformin in immune response and inflammation
Metformin also exerts actions in immune modulation and inflammatory response, lending resilience to infectious diseases and acute respiratory illnesses (Fig. 2, Table 2). Metformin’s interference with key immunomodulatory mechanisms are both dependent and independent of its effects on improving in metabolic parameters. Several mechanisms may underlie metformin’s effects on immune resilience and responses to infectious disease; however, it is important to note that the effects of metformin on the immune system are dependent on the pathological context (reviewed in [41]). In the context of some cancers, metformin has been shown to be immunostimulatory with a shift favoring M1 macrophage polarization over the more antiinflammatory M2 [42, 43] and increased antigen-specific T cell responses coupled with decreased immunosuppressive T regulatory cells [44, 45]. However, this shift towards an immunostimulatory phenotype seems specific to pathological immunosuppressive conditions such as cancer. Conversely, in most other situations, metformin is able to quell aberrant inflammation and promote antiinflammatory pathways. Indeed, in the context of wound healing, metformin induces M2 polarization [46]. Additionally, during autoimmune diseases [47], graft-versus-host disease [48] and others more proinflammatory conditions, metformin is able to increase T regulatory cells and decrease T helper (Th)17 cells [47–49], an effect likely partially mediated by AMPK activation [50]. Moreover, metformin treatment in uninfected mice increases in vitro T cell IFN-γ responses as well, indicating that metformin treatment has the ability to modulate immune cells independent of current infections and potentially improve responses to future pathogens [51]. For coronaviruses, the timing of type I IFN production is critical to infection outcome: an early burst of type I IFN leads to protection, but a delay in IFN production causes an inability to control viral replication or pathogen spread, leading to cellular damage of airway epithelia and the lung parenchyma and an eventual lethal inflammatory cytokine storm [52]. Thus, metformin has the potential to improve the initial type I IFN response that is crucial for coronavirus immune responses and prevent or reduce cellular damage from exaggerated, prolonged inflammatory responses.
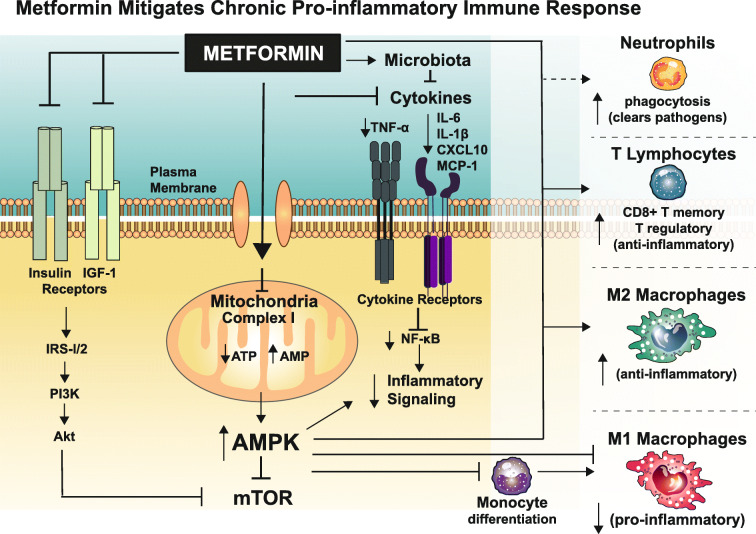
Fig. 2.
Metformin alleviates chronic proinflammatory immune signaling and restores immune response. Metformin’s cellular mechanisms include weak inhibition of complex I of the mitochondrial electron transport chain, activation of the energy sensor AMP-activated protein kinase, inhibition of the heteromultimeric protein kinase mTORC1, and suppression of elevated proinflammatory cytokines production. Converging evidence also implicates the gut microbiome which further alleviates inflammation and phagosome-lysosome fusion which induces phagocytosis of neutrophils to reduce pathogen burden. The collective result is a dampened broad proinflammatory cytokine signaling and improved immune cell activation
Table 2.
Metformin treatment in experimental models of infectious disease or acute respiratory illness
| Disease or illness | Design, species | Pathogen or exposure | Metformin dosing | Relevant result | Ref. |
|---|---|---|---|---|---|
| Acute or chronic lung illness | |||||
| ARDS | In vivo, mouse | LPS-induced lung injury model in BALB/c mice |
Prophylactic 50 mg/kg from 7 days prior to LPS injury |
- Partially reverse pulmonic injury (Pulmonary edema, vascular exudation, and neutrophil accumulation) and inflammatory cytokines -reduce LPS-induced death |
[143] |
| ARDS | In vivo, mouse | LPS-induced lung injury model in BALB/c mice |
With exposure 250 mg/kg (i.p.) 0.5 h prior to LPS injury |
- Suppress LPS-induced lung injury and inhibit markers of oxidative stress | [144] |
| ARDS | Ex vivo, human | BAL and hMDM from ARDS patients |
With exposure 500 μmol/L metformin added to culture of ARDS hMDMs + BAL |
- Increase in uptake of apoptotic neutrophils and neutrophil extracellular trap (NET) engulfment | [145] |
| Lung fibrosis | In vivo, mouse | Bleomycin lung fibrosis model |
Treatment 65 mg/kg; (i.p.) for 18 days, initiated 10-day post-bleomycin lung injury |
- Significant reductions in profibrotic markers total lung hydroxyproline, α-SMA expression. Collagen - Promotes fibrosis resolution up to 3 weeks post-exposure |
[146] |
| Bacterial infections | |||||
| L. pneumophila | In vitro, mouse, human |
- Bone marrow–derived macrophages - RAW cells (mouse) - U937 cells (human) |
Treatment 2 mM metformin 6- or 24-h postinfection |
- Suppress bacterial growth in time and concentration dependent manner | [86] |
| L. pneumophila | In vivo, mouse | L. pneumophila pneumonia model |
Prophylactic 5 mg/ml metformin from 7-days prior to infection |
- Improve survival and significant reduction in bacterial number in the lung | [86] |
| M. Tuberculosis | In vitro, human | Healthy human peripheral blood mononuclear cells (PBMC) infected with m. tuberculosis |
Treatment 3–3000 μM metformin incubation for 24 h |
- Enhanced cellular metabolism - Inhibited p70S6K and 4EBP1 - Decreased cytokine production, cellular proliferation - Increased phagocytosis activity |
[147] |
| M. Tuberculosis | In vitro, human | Lung epithelial cells and macrophages |
With exposure 2 mM and 4 mM metformin for 48-h, initiated with infection |
- Reduce bacillary loads in macrophages and lung epithelial cells | [148] |
| M. Tuberculosis | In vitro | Human monocytic cell line THP-1 and hMDMs |
With exposure 2 mM metformin in culture initiated with infection |
- Restrict mycobacterial growth by inducing mitochondrial reactive oxygen species production | [51] |
| M. Tuberculosis | In vivo, mouse | Acute and chronic M. tuberculosis |
Adjuvant treatment 500 mg/kg starting 7 or 42 days post-infection (± isoniazid or ethionamide) |
- Enhance the efficacy of conventional anti-TB drugs -improve TB-induced tissue pathology and immune response |
[51] |
| M. Tuberculosis | In vivo, mouse | BALB/c mice infected with M. tuberculosis |
Treatment 250 mg/kg for up to 6-months, initiated 6-weeks post-infection |
- No effect on lung bacillary burdens or microbiological relapse | [149] |
| P. aeruginosa | In vitro | P. aeruginosa infection across airway epithelial (Calu-3) cultures |
With exposure 0.02 or 1 mM metformin 18-h prior to inoculation |
- Reduce hyperglycemia-induced P. aeruginosa growth through airway epithelial tight junction modulation | [84] |
| P. aeruginosa | In vivo, mouse | Streptozotocin-induced diabetes |
Prophylactic 4 mg/ml (i.p.) at day −2, −1, 0 of infection |
- Reduce airway glucose and bacterial load (without change in blood glucose) | [150] |
| P. aeruginosa | In vivo, C. elegans, mouse | - P. aeruginosa PA14 infection |
With exposure 1–100 mM metformin initiated with infection |
- Dose-dependent increase resistance to P. aeruginosa PA14 infection - Increase levels of active PMK-1 - p38/PMK-1-mediated innate immunity conserved from worms to mammals |
[151] |
| S. aureus | In vitro | H441 epithelial cells |
With exposure 1 mM 18-h prior to infection |
- Increase transepithelial resistance - reduce glucose-dependent bacterial growth |
[152] |
| S. aureus | In vitro, human | Airway epithelial cells |
With exposure 0, 0.03, 0.3, or 1 mM metformin 18 h prior to inoculation |
- Reduced paracellular flux across murine tracheas. | [153] |
| S. aureus | In vivo, mouse | Wild-type, C57BL/6 or db/db, |
Prophylactic 40 mg/kg metformin 2 days prior to infection |
- Modify glucose flux across the airway epithelium - Limit hyperglycemia-induced bacterial growth |
[153] |
| Trypanosoma cruzi | In vivo, mouse | CD-1 mice on high fat diet (HFD) |
Prophylactic 50 mg/kg metformin by gavage (± HFD) 20 days prior to infection |
- Reduce mortality from 20% on HFD to 3% on HFD + metformin | [82] |
| Parasitic infection | |||||
|
Malaria P. berghei |
In vitro | Huh7 cells |
Treatment 0.04–1.25 mM metformin |
- Reduce of total parasite load (dose-dependent) | [154] |
|
Malaria P. berghei |
In vivo, mouse | C57BL/6 |
Prophylactic 500 mg/kg/day metformin, 7 days prior to infection |
- Reduce total burden of P. berghei liver infection and lessened disease severity | [155] |
|
Malaria P yoelii |
In vivo, mouse | C57BL/6 |
Treatment 5 mg/ml metformin initiated with infection or 7 days post-infection |
- Reduce of parasitemia | [156] |
| Viral infections | |||||
| HCV | In vitro | Huh7.5 cells infected with HCV particles or primary human hepatocytes |
With exposure 2–10 mM metformin + 2 μM simvastatin |
- Inhibited cell growth and HCV infection | [157] |
| HCV | In vitro | Huh-7.5 cells (HCV-susceptible subclone of Huh7 cells) |
With exposure 0–10 mM metformin for 20 h incubation |
- Anti-HCV effects in AMPK dependent and independently manner | [158] |
| HCV | In vivo, human | Patients with genotype 1 chronic HCV and insulin resistance |
Adjuvant treatment 1275–2550 mg/day metformin for 48 weeks with antiviral PEG-IFN and RBV |
- Improved sustained virological response (SVR) in females only (not overall) | [159] |
| HCV | In vivo, human | Patients with genotype 1 chronic HCV and insulin resistance |
Adjuvant treatment 1500 mg/day metformin for 48 weeks with PEG-IFN and RBV |
- Metformin adjuvant improved insulin sensitivity and increased the SVR rate | [160] |
| HCV | In vivo, human | Treatment-naïve chronic HCV patients |
Adjuvant treatment 1500 mg/day for 6-months with PEG-IFN and RBV |
- SVR rate in the metformin group was 75% versus 79% in controls (intention-to-treat) which was not significantly different. | [161] |
| HBV | In vitro | HBV-producing human hepatoma cell line HepG2.2.15 |
Treatment 0–4 mM metformin for 6 days initiated 24 h post-infection |
- Enhanced the inhibitory effects of interferon-α2b on HBsAg expression and HBV replication | [89] |
| HBV | In vitro | HepG2 and PLC/PRF/5 cells |
Combined treatment 0-200 μM metformin +20 nmol/L rapamycin for 12–48-h |
- Decrease cell viability at 12 h in Sir+Met - No change in Sir or Met monotherapy until 24 h |
[162] |
| HBV | In vivo, human | Hepatocellular carcinoma (HCC) related to HBV infection in T2DM |
Combined treatment 1700 mg/day metformin + 2 mg/day sirolimus postoperative |
- Survival in the Sir+Met group was significantly longer compared with control and monotherapy | [162] |
| HBV | In vivo, mouse | HBV × antigen transgenic (HB × Tg) mice |
Treatment 250 mg/kg/day metformin (drinking water), 2–18 months of age |
- No effect on incidence of HCC, but slightly increased hepatic cellular retinol-binding protein-I | [163] |
| HBV | Ex vivo, human | Tumor and normal liver specimens in persons with T2DM and HBV associated HCC | Prophylactic metformin as prescribed for T2DM |
- Inhibitory on HBV-associated tumorigenesis (HCC) by inhibiting tumor cell viability and promoting apoptosis - Lower recurrence rates of HBV-associated HCC |
[164] |
| HIV | In vitro |
Latently HIV-infected monocytic (THP-p89 cells) and lymphocytic (J1.1 T cell) model - acute infection models of X4- and R5-tropic viruses on Jurkat and Magi cells |
Combined treatment 100 mM metformin with bryostatin |
- Cotreatment modulates latent HIV-1 infection by purging latent virus from cellular reservoirs | [165] |
| Influenza (vaccine) | Ex vivo, human | B cells isolate (T2DM) |
Prophylactic 2000 mg /day metformin >3 years |
- Reduce B cell-intrinsic inflammation and increase antibody responses | [166] |
ARDS Acute respiratory distress syndrome, LPS lipopolysaccharide, i.p. intraperitoneal, BAL bronchoalveolar lavage, hMDM human monocyte–derived macrophages, NET neutrophil extracellular trap, PBMC peripheral blood mononuclear cells
Legionella pneumophila (L. pneumophila); Mycobacterium tuberculosis (M. Tuberculosis); tuberculosis (TB); Pseudomonas aeruginosa (P. aeruginosa); Staphylococcus aureus (S. aureus); high fat diet (HFD); Plasmodium berghei (P. berghei); Plasmodium yoelii (P. yoeii); hepatitis C virus (HCV); pegylated interferon (PEG-IFN); ribavirin (RBV); hepatitis B virus (HBV); hepatocellular carcinoma (HCC); human immunodeficiency virus (HIV)
Indeed, metformin’s effect on immune responses to chronic pathogen exposures or challenges, as well as general states with elevated inflammation, tends to be overall immunosuppressive and antiinflammatory (Fig. 2). This dysregulated immune signaling is particularly relevant to SARS-CoV-2, which causes exaggerated and aberrant host immune responses that are uniquely challenging in persons of advanced age [53]. This ‘cytokine storm’ of perpetuating proinflammatory signaling loops is thought to lead to acute respiratory distress syndrome and poorer outcomes, especially in older adults [54, 55]. Metformin improves the immune response and reduces inflammation by promoting the formation of M2 macrophages and T regulatory and CD8 memory T cells [56], and may prevent the differentiation of monocytes to macrophages via an AMPK-dependent inhibition of STAT3 [57]. In the face of chronic infection or disease, metformin also reduces genes encoding for cytokines associated with inflammation response by suppression of tumor necrosis factor-α (TNF-α) dependent NF-κB signaling and expression of interleukin (IL)-6 (IL-6), IL- β (IL-1β), MCP-1 (CCL2 chemokine ligand 2), CXCL5, and IP-10 (CXCL10) [51, 58, 59]. Collectively, this results in lower levels of circulating proinflammatory cytokines, and in a recent report in older persons with T2DM, the reduction in proinflammatory signaling was associated 5-year all-cause mortality risk [60]. Furthermore, metformin treatment diminished LPS-induced acute lung injury [61], suggesting that metformin may be able to reduce the cytokine storm-like response and prevent exaggerated lung damage due to SARS-CoV-2 as well. In addition, metformin can induce autophagy and facilitate phagosome-lysosome fusion and induces phagocytosis of neutrophils, which points to a role for metformin in killing or containing pathogens, controlling inflammation, and activating innate and adaptive immune response in the host [62].
Metformin may also reduce inflammation through gut microbiota [38, 62]. Multiple studies have shown that the glucose lowering ability of metformin is partly mediated through the gut microbiota changes [63–65]. Others have further shown that the antiinflammatory effects of metformin may also be partly mediated through the gut microbiota, even in nondiabetic conditions where the pattern of gut microbe changes with metformin are negatively correlated with a variety of inflammatory diseases [66]. While the mechanism of effect on the microbiota is not known, it seems that metformin may reduce dysfunctional gut permeability (“leaky gut”) by increasing goblet cell mass and mucin production [67], and may alleviate gut dysbiosis by increasing Akkermansia muciniphila and short-chain fatty acid-producing bacteria, such as Bacteroides and Butyricimonas [68]. Of particular interest, these alterations were still observed in aged obese mice despite the presence of preexisting age-related microbiota changes and were correlated with lower IL-6 and Il1β [67] [68]. This suggests that metformin may also be able to mitigate exaggerated inflammatory responses via changes in gut microbiota.
Metformin and resilience to severe infectious disease
COVID-19
The origin and pathogenesis of the acute respiratory disease caused by the novel coronavirus SARS-CoV2 is increasingly well understood [54]. SARS-CoV2 is an enveloped nonsegmented positive-sense RNA virus. The virus gains entrance to host cells through angiotensin-converting enzyme 2 (ACE2) receptors, and upon entry the virion releases its RNA in the cytoplasm. Translation and replication occur and new virions are then released from the host cell [69, 70]. However, key to COVID-19 pathogenesis is the attendant explosive and deleterious host immune response and immunopathogenesis [71]. This cytokine storm, or cytokine release syndrome, may occur approximately 5–8 days after symptom onset and is associated with dismal outcomes including ICU care, mechanical ventilation, prolonged hospital care, and in some cases, death. Inflammatory factors elevated during this storm include IL-6, IFNγ, TNFα, IL-1β, IL-8, MCP-1, (CXCL10), and lower T cell counts (CD8+ and CD4+ T lymphocytes) in older adults with severe COVID-19 cases [71–76]. Importantly, elevated levels of the proinflammatory factors are associated severity of COVID-19 and unfavorable outcomes like acute respiratory distress syndrome, multiple-organ failure [73, 76]. Interestingly, the list proinflammatory cytokines implicated in the cytokine storm are nearly identical to those suppressed with metformin administration.
Intriguing epidemiologic evidence indicates a potential benefit of metformin on COVID-19 health outcomes (Table 3). In a retrospective analysis of 283 T2DM patients from Wuhan, China, with confirmed COVID-19, investigators found no difference in the length of stay in hospital, but persons taking metformin had significantly lower in-hospital mortality (3 of 104, 2.9%) than those not taking metformin (22 of 179, 12.3%) [77]. Upon hospital admission, both groups were under strict glucose control and all received antiviral and supportive treatments including antibacterial and anticoagulants, with no significant differences in treatment. This report is further supported by an in-review analysis of de-identified claims data from UnitedHealth of 6256 persons hospitalized with COVID-19 [78]. Women who had at least 90 days of metformin claims in the 12 months before hospitalization were found to have ~ 21% lower odds of mortality compared with persons with T2DM but without metformin. This association was not observed for men. Though difficult to quantify, metformin’s immunoprotective effects in the context of COVID-19 may be masked if fewer persons taking metformin are being admitted to hospital.
Table 3.
Epidemiologic studies of metformin use in infectious disease
| Reference | Population | Disease status | Comparator | Follow-up | Outcome | Effect size (95% CI) |
|---|---|---|---|---|---|---|
| COVID-19 | ||||||
| Luo [77] |
China N = 283 |
COVID-19 +T2DM |
Non-use | Retro | In hospital mortality | OR 0.23 |
| Bramante [78] |
USA, women n = 3302 |
COVID-19 +T2DM |
Non-use | Retro | In hospital mortality | OR 0.792 (0.640, 0.979) |
|
US, All N = 6256 |
COVID-19 +T2DM |
Non-use | Retro | In hospital mortality | OR 0.904 (0.782, 1.045) | |
| Other respiratory illness | ||||||
| Degner [80] |
Taiwan N = 2416 |
TB +T2DM |
Non-use | All-cause mortality | HR 0.56 (0.39–0.82) | |
| Mortensen [87] |
USA, VA N = 7424* |
Pneumonia +T2DM |
Non-use | Case-control | In hospital mortality | OR 0.80 (0.72–0.88) |
| Yen [88] |
Taiwan N = 20,644* |
COPD +T2DM |
Non-use | Case-control 5 years | Hospitalized pneumonia | HR 1.17 (1.11–1.23) |
| Nonrespiratory infectious disease | ||||||
| Chen [167] | N = 135 |
Chronic HCV +HCC |
Non-use | Retro | 5-year survival rate | HR 0.24 (0.07–0.80) |
| Chen [70] |
Taiwan N = 71,824 |
HBV | Non-use | 9 years | All-cancer | HR 0.82 (0.75–0.90) |
| Chen [70] |
Taiwan N = 71,824 |
HBV | Non-use | 9 years | All-cause mortality | HR 0.56 (0.39–0.82) |
| Nkontchou [168] |
France N = 100 |
HCV Cirrhosis | Non-use | 5 years | Incident HCC | HR 0.19 (0.05–0.99 |
| Nkontchou [168] |
France N = 100 |
HCV Cirrhosis | Non-use | 5 years | Liver-related mortality | HR 0.22 (0.05–0.99) |
| Romero-Gomez [159] |
Spain N = 123 |
Chronic HCV | Placebo | 72 weeks | SVR | 52.5% (v. 42.2% Pl) |
| Sharifi [161] |
Iran N = 140 |
Chronic HCV | Placebo |
RCT, 24–48 weeks |
SVR | 75% (v. 79% Pl) |
| Yu [160] |
China N = 98 |
Chronic HCV | Placebo | 72 weeks | SVR | 59.2% (v. 38.8% Pl) |
COVID-19 Novel coronavirus-19, HBV hepatitis B virus, HCV hepatitis C virus, HCC hepatocellular carcinoma, T2DM type 2 diabetes mellitus, Retro retrospective cohort, PL placebo, SVR sustained viral response, RCT randomized clinical trial
*Cases and controls matched
Importantly, these findings are relevant only in the context of long-term treatment with metformin prior to infection. Therefore, these epidemiologic studies do not generalize to potential actions of metformin administered in hospitalized patients with active infection as a treatment adjuvant. While metformin use is generally safe, side effects and gastrointestinal discomfort do occur in a proportion of the population, and it is unknown what the additional risk of initiating metformin in hospital for those with comorbid or critical conditions upon admission would be. Basic research coupled with carefully controlled clinical trials would be required prior to translation to the clinic. Moreover, the protective effects of metformin on health outcomes due to acute infectious disease must be replicated and confirmed with rigorous prospective studies with and without overt T2DM. Finally, emerging evidence supports close investigation of both potential sexual dimorphism and mediating effects of metformin on proinflammatory components of the cytokine storm also require close investigation.
Other respiratory-related infectious disease
Evidence of metformin as a therapeutic strategy in other infectious diseases and respiratory illnesses is emerging from a range of experimental designs, model systems, and pathogens or exposures (Table 2). In a screen of 13 FDA-approved autophagy activators and AMPK modulators, metformin (i) displayed direct antimycobacterial effects; (ii) controlled the growth of drug-resistant bacterial strains, (iii) increased production of mitochondrial reactive oxygen species (mROS), and (iv) facilitated phagosome-lysosome fusion [51, 79]. In mice, use of metformin monotherapy enhanced ameliorated lung pathology and enhanced host immune responses, and in combination therapy, metformin enhanced the specific immune response and the efficacy of conventional antimycobacterial drugs [51]. Collectively, this confers host protection and lessens burden of active infection. For example, retrospective analyses of tuberculosis patients who were on metformin had fewer pulmonary cavities, less advanced disease, and lower mortality rate than those not on metformin [51, 80]. Moreover, in 2416 persons being treated for tuberculosis in Taiwan, coexisting metformin use was significantly associated with decreased mortality during treatment; strikingly, metformin users had similar mortality as patients without T2DM [80].
Laboratory studies using cells or model organisms suggest metformin could be effective against numerous other pathogens, including Trypanosoma cruzi, Trichinella spiralis, Staphylococcus aureus (S. aureus), and Pseudomonas aeruginosa (P. aeruginosa) [81, 82]. Of relevance to COVID-19, bacterial infections of the respiratory system by S. aureus and P. aeruginosa are associated with local (vs. systemic) hyperglycemia, which may be associated with respiratory tract infections in conditions like chronic obstructive pulmonary disease [83]. Interestingly, metformin’s effects on airway epithelial tight junctions [84] and bacterial load are independent of effects on blood glucose but use an AMPK- and PKCζ-mediated pathway [84, 85]. Moreover, in a murine Legionella pneumophila pneumonia model, metformin treatment improved survival of mice, which was associated with a significant reduction in bacterial number in the lung, which was mediated by mitochondrial ROS production and AMPK signaling and enhanced macrophage bactericidal activity [86]. Epidemiologic evidence are conflicting (Table 3); some studies show a net benefit of metformin use on outcomes like 30-day mortality in US veterans hospitalized with pneumonia [87], whereas a population-based Taiwanese case-control study failed to demonstrate a preventive effect of metformin on hospitalization for pneumonia in persons with T2DM and COPD [88]. Clinical trials are needed to bridge the promising preclinical evidence and epidemiologic evidence. A planned small randomized crossover placebo-controlled trial will explore effects on airway glucose and bacterial growth in nondiabetic persons with COPD (NCT03651895); however, trials with follow-up time sufficient to capture a broad range of respiratory-related outcomes may be necessary.
Chronic viral infections
Insights can also be drawn from metformin use in the context of chronic viruses, such as liver targeting hepatitis B virus (HBV) and hepatitis C virus (HCV), and human immunodeficiency virus (HIV). HBV and HCV are among the leading causes of liver disease. In HBV, approximately 90% of infections are acute, whereas 10% progress to chronic infection. Metformin-mediated decreases in hepatitis B surface antigen (HBsAg) levels in culture supernatants and in cell lysates and may work synergistically with other antivirals [89]. This will be tested in a randomized placebo-controlled trial in nondiabetic persons with chronic HBV: an investigative team in China will examine the addition of 24-week metformin to ongoing standard HBV treatment entecavir on cumulative rate of HBsAg loss (NCT04182321). Trials on long-term disease outcomes are have not been conducted, but a population-based cohort study of 71,824 HBV-infected patients in Taiwan showed that metformin use was chemoprotective; patients on metformin had a lower incidence of all-cancer, not just HBV-related liver cancer (adjusted hazard ratios, HR 0.75 (95% CI, 0.67–0.84)) [90]. This is interesting because with chronic viral exposure some of metformin’s protective effects may extend beyond the direct long-term health consequences of HBV. HCV, like SARS-CoV 2, is a ribonucleic acid (RNA) virus. HCV poses a serious health threat across all ages, but those over the age of 65 years now account for a large portion of all chronic hepatitis C infections in the USA and have the highest rate of hepatitis C-related deaths [91]. In patients with HCV and T2DM, metformin’s effects on sustained viral response (SVR) are mixed (reviewed in [81]). However, overall metformin is associated with benefit on liver injury and reduces the rate of incident hepatocellular carcinoma [92], possibly by activating type I INF signaling against HCV via activation of AMPK.
Metformin is a first-line medication for management of T2DM and insulin resistance in persons living with HIV, but metformin may have a role in HIV pathogenesis as well [93, 94]. Dysregulated metabolism is implicated in the relationship between viral proteins, immune activation, and inflammation in persons infected with HIV, and is hypothesized to dampen the protective innate and adaptive arms of immunity [95]. In persons living with HIV and T2DM in Botswana, metformin use is associated with improved CD4 lymphocyte count recovery [96]. An independent study in China suggests metformin inhibit NF-κB/p65 phosphorylation to suppress CD54 expression on CD4+ T cells, which is associated with disease progression in persons living with HIV [97]. In a recent 24-week pilot study involving 12 virally suppressed HIV-infected individuals without T2DM who were randomized to metformin or observation, metformin reduced CD4 T cell exhaustion as measured by negative immune checkpoint receptors (NCR) such as programmed cell death protein-1 (PD1) and T cell immunoreceptor with Ig and ITIM domains (TIGIT) [98]. Other trials are planned including a pilot trial in nondiabetic HIV-infected persons to evaluate metformin’s effect on various virological assays, inflammation, and disease outcomes (NCT02659306) [99, 100]; follow-up trials will be needed to uncover common mechanisms and best drug combinations [101]. Additionally, an emerging hypothesis posits that metformin may improve gut microbiota composition, which could reducing inflammation and mitigate risk of nonAIDS comorbidities [62, 102, 103].
Vaccine response
In 2014, it was reported that an mTORC1 inhibitor increased antibody titers to flu vaccination in 218 healthy elderly subjects [28]. Whether metformin improves vaccine response in nondiabetic older adults remains an active area of exploration. Recent reports are mixed. For example, a 2017 study measured in vivo and in vitro influenza vaccine responses in newly diagnosed treatment naïve T2DM patients and patients taking metformin for at least 3 years. Ongoing metformin use was associated with recovered B cell function and vaccine response compared with those who were just recently diagnosed with T2DM. Moreover, metformin administered in vitro reduced B cell intrinsic inflammation and increased antibody responses in B cells harvested from the recently diagnosed T2DM patients, resulting in B cell function similar to patients already taking metformin [104]. In contrast, a recent cross-sectional study or 67 older adults showed reduced antibody responses to influenza vaccination quantified by virus neutralizing Ab (VNA) titers and immunoglobulin (Ig) isotypes to H1N1 and H3N2 in the 11 metformin users compared with nonmetformin users [105]. Clinical trials in nondiabetic older adults are needed to resolve these seemingly discrepant results of observational studies in order to evaluate metformin as a potential adjuvant to boost response to vaccine. Two noteworthy studies on metformin and vaccine response in nondiabetic older adults are underway (Table 4). A pilot investigation is ongoing to determine whether metformin can improve the immune response to the pneumococcal conjugate vaccine (PCV-13) in older adults and if this effect is mediated by the gut microbiota (NCT03713801). An independent ongoing pilot trial will evaluate metformin’s effect on both cellular and humoral influenza vaccine responses, in healthy nondiabetic, nonprediabetic older adults (NCT03996538). While many COVID-19 vaccines are under development, it is important to note that the response to vaccines is also diminished with aging. Thus, these trials will be important to elucidate potential mechanisms to enhance protective responses in older adults if/when a COVID-19 vaccine becomes available.
Table 4.
Randomized placebo controlled trials of metformin on vaccine response, infectious, or respiratory diseases
| Clinical trial | Population | Sample size | Group | Dose (per day) | Duration | Primary (1) secondary (2) endpoints | Status |
|---|---|---|---|---|---|---|---|
| Airway Glucose in COPD | 40–75+ years nondiabetic | 39 | Placebo, metformin | 1000 mg (1 × 1000 mg) | 12 weeks |
1) Sputum glucose concentration 2) Sputum bacterial load and inflammatory marker, quality of life, pulmonary function |
Planned |
| Chronic HBV | 18–55 years HBeAg-positive chronic HBV | 60 | Entecavir + placebo, metformin | 1000 mg (2 × 500 mg XR) | 36 weeks | 1) Cumulative rate of HBsAg loss | Recruiting |
| VEME (pilot) | 65+ years nondiabetic | 26 | Placebo, metformin | 1500 mg (3 × 500 mg XR) | 20 weeks |
1) Cell-mediated immune responses to flu vaccine 2) Flu antibody titers, frailty phenotype, and T cell metabolic cellular profiles |
Ongoing |
| Impact of Metformin on Immunity | 63–89 years nondiabetic vaccine response impaired | 50 | Placebo, metformin | 1500 mg (3 × 500 mg) | 12 weeks |
1) Change in antibody responses to PCV13 2) Change in immunophenotypes |
Recruiting |
| Lilac Pilot | Nondiabetic persons living with HIV on ART | 22 | Metformin + ART | 1700 mg (2 × 850 mg) | 12 weeks |
1) Size of the HIV reservoir 2) Safety, HIV reservoir in colon biopsies, frequency of CD4+ T cells harboring inducible HIV (TILDA), change in immune activation |
Unknown |
Airway Glucose in COPD: Metformin to reduce airway glucose in COPD patients, NCT03651895
Chronic HBV: Adding Metformin to the Standard Treatment for Patients With HBeAg-Negative Chronic Hepatitis B, NCT04182321, Beijing, China
VEME (pilot): Vaccination Efficacy with Metformin in Older Adults: A Pilot Study, NCT03996538, USA
Impact of Metformin on Immunity, NCT03713801, USA
Lilac Pilot: Metformin Immunotherapy in HIV Infection, NCT02659306, Canada
Geroscience: from acute care to long-term health and prevention
Evaluating intervention effects on comorbidity and resilience to acute challenge is even more critical in the aftermath of COVID-19. The lowered overall resilience of older COVID-19 patients is easily observed in the high incidence of fatalities, hospitalizations, and potential for long-term complications in patients with advanced ages. These outcomes are associated with or exacerbated the prevalence of age-related disease, comorbidity, frailty, and loss of endogenous protective and repair mechanisms.
Most importantly, once the immediate challenge of treating acute cases of COVID-19 have passed, the need to treat these newly acquired or exacerbated long-term chronic age-related diseases in our population will be greater than ever. Efforts to treat the infectious diseases presenting in our hospitals worldwide are essential. At the same time, we must also delve deeper to understand how molecular pathways inextricably linked to biological aging, such as mTOR signaling, cellular senescence, insulin/IGF-1 signaling, and AMPK activation, are linked to immune response. This focus could provide novel host, as opposed to pathogen-directed therapies to respond to the current pandemic crisis, but more importantly aid our ability to help those affected by COVID-19 and its attendant chronic health consequences. Understanding the interaction between aging biology, viral susceptibility and immune response, and progression to comorbid chronic disease, frailty, and disability remains one of the most important biomedical issues worldwide. Geroscience represents a major strategic advancement and new therapeutic approach to considering long-term options for prevention and mitigating the anticipated swell in long-term health complications that will be left in the wake of COVID-19.
Ample evidence links metformin to lower all-cause mortality and reduced rates of multiple disease of aging, even in nondiabetic populations [106] [107] and suggest a broad geroprotective role of metformin. Clinical trials exploring the effects of metformin in nondiabetic older adults on gerocentric outcomes are underway [108] or planned [30, 31]. For example, a placebo-controlled, double-blinded clinical trial of 2-year metformin treatment for the prevention of frailty in 120 older adults with prediabetes is ongoing at University of Texas Health Science Center at San Antonio [108]. The TAME trial will evaluate metformin’s effects on incidence of any new age-related disease (cardiovascular disease, mild cognitive impairment/dementia, most cancers, or death) in 3000 older adults without diabetes [30, 31]. Both trials include collection of biospecimens and a variety of assessment measures to support ancillary investigations to complement the primary outcome. For example, in addition to an FDA-facing clinical disease composite, primary outcome TAME will include several indices of health span measures including measures of physical and cognitive function and frailty assessments, assays for key biomarkers, and biobanking of biofluids, cells, and feces for discovery and mechanistic investigations. While neither randomized controlled trial is designed to evaluate efficacy of metformin on COVID-19–related outcomes, they nonetheless represent unique and time-sensitive resources to the scientific community. These trials coincide with the pandemic, which provides an opportunity for long-term follow-up and deep phenotyping of older adults with and without exposure to COVID-19 in the course of the study and may include smaller subgroups who are symptomatic vs. asymptomatic. Fresh collection of peripheral blood mononuclear cells and tissues included in these trials provide an unprecedented opportunity for immunophenotyping; investigator-initiated studies could leverage ongoing trials to examine metformin’s effects on immune cell activation and inflammatory cytokine production. Similar to other ongoing trials or large cohort studies [109, 110], TAME will include measures to query history, symptoms, and burden of COVID-19 as exploratory assessments. Moreover, TAME’s large sample size could permit exploratory investigations on the effects of metformin on age-related diseases or functional decline in older adults with or without history of COVID-19, though such explorations would not bear the burden of proof necessary for a virus-related FDA indication. Other ancillary investigations could be envisioned to explore metformin’s effects on immune response to vaccines in older adults once a vaccine against SARS-CoV2 is developed. Ultimately, these existing or upcoming clinical trials provides a scaffolding for future investigations on long-term health effects of COVID-19 and metformin’s potential to improve resilience following immune challenge.
Concluding remarks and future
In conclusion, metformin is an attractive tool or probe for clinical trials targeting aging and to improve host immune defense and resilience in COVID-19 and infectious disease. Its immunoprotective effects are hypothesized to (i) lessen severity of unfavorable health outcomes or death in the event of exposure to infectious disease; (ii) delay or prevent long-term chronic diseases or conditions which can stem from acute challenges or viral infections; and (iii) bolster immune response to vaccine. However, this hypothesis has yet to be rigorously tested. It is time for definitive geroscience trials of not only metformin but other promising geroprotective interventions like caloric restriction, mTOR inhibitors, and senolytics. Trials informed by geroscience will offer opportunity to investigate intervention effects on vaccine response and resilience to age-related chronic diseases and geriatric syndromes and provides a unique opportunity to advance study of host-immune defense with implication for recovery from the current pandemic and unforeseen future challenges.
Authors’ contributions
JNJ is the primary author responsible for drafting, editing, figure development, and literature reviews; SG aided in literature reviews and vetting of studies listed in Table 3, and editing all manuscript drafts; ASK provided content expertise and development of Table 1; JMB provided content expertise and draft materials related to mechanisms of metformin on immune defenses and vaccine response; GAK and NB provided guidance on development, oversight, and editing throughout.
Funding
The authors acknowledge the support by the American Federation of Aging Research (JMB, Irene Diamond AFAR award), the Glenn Center for the Biology of Human Aging (NB), and the National Institutes of Health (NIH): JNJ (K01 AG059837, P30 AG021332); GAK (R33 AG061456, R21 AG063528, R56 AG060746, R35 GM124922, U01 AI124297, R21 AG060018, R01 AG058814, R01 AI142086, UH3 AG056925); NB (P30AG038072).
Data availability
Not applicable.
Compliance with ethical standards
Conflict of interest
ASK is now a Senior Scientist, Computational Genomics at AbbVie; his recent employment in Abbvie did not influence the literature reviewed. JNJ, SG, JMB, GAK, NB have no conflicts to report.
Code availability
Not applicable.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Statistics, C.N.C.f.H. COVID-19 Death Data and Resources. 2020; Available from: https://www.cdc.gov/nchs/covid19/.
- 2.The OpenSAFELY Collaborative, et al., OpenSAFELY: factors associated with COVID-19-related hospital death in the linked electronic health records of 17 million adult NHS patients. MedRxiv, 2020. 2020.05.06.20092999.
- 3.Zhou F, Yu T, du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang, D., et al., Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA, 2020. [DOI] [PMC free article] [PubMed]
- 5.Ben-Yehuda A, Weksler ME. Host resistance and the immune system. Clin Geriatr Med. 1992;8(4):701–711. [PubMed] [Google Scholar]
- 6.Yoshikawa TT. Epidemiology and unique aspects of aging and infectious diseases. Clin Infect Dis. 2000;30(6):931–933. doi: 10.1086/313792. [DOI] [PubMed] [Google Scholar]
- 7.Promislow, D.E.L., A geroscience perspective on COVID-19 mortality. J Gerontol A Biol Sci Med Sci, 2020. [DOI] [PMC free article] [PubMed]
- 8.DuGoff EH, Canudas-Romo V, Buttorff C, Leff B, Anderson GF. Multiple chronic conditions and life expectancy: a life table analysis. Med Care. 2014;52(8):688–694. doi: 10.1097/MLR.0000000000000166. [DOI] [PubMed] [Google Scholar]
- 9.Kato M, Natarajan R. Epigenetics and epigenomics in diabetic kidney disease and metabolic memory. Nat Rev Nephrol. 2019;15(6):327–345. doi: 10.1038/s41581-019-0135-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sato Y, Yanagita M. Immunology of the ageing kidney. Nat Rev Nephrol. 2019;15(10):625–640. doi: 10.1038/s41581-019-0185-9. [DOI] [PubMed] [Google Scholar]
- 11.Li H, Hastings MH, Rhee J, Trager LE, Roh JD, Rosenzweig A. Targeting age-related pathways in heart failure. Circ Res. 2020;126(4):533–551. doi: 10.1161/CIRCRESAHA.119.315889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, and the Northwell COVID-19 Research Consortium. Barnaby DP, Becker LB, Chelico JD, Cohen SL, Cookingham J, Coppa K, Diefenbach MA, Dominello AJ, Duer-Hefele J, Falzon L, Gitlin J, Hajizadeh N, Harvin TG, Hirschwerk DA, Kim EJ, Kozel ZM, Marrast LM, Mogavero JN, Osorio GA, Qiu M, Zanos TP. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323:2052. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ajaimy M, Melamed ML. COVID-19 in patients with kidney disease. Clin J Am Soc Nephrol. 2020;15(8):1087–1089. doi: 10.2215/CJN.09730620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu J, Li J, Zhu G, Zhang Y, Bi Z, Yu Y, Huang B, Fu S, Tan Y, Sun J, Li X. Clinical features of maintenance hemodialysis patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. Clin J Am Soc Nephrol. 2020;15(8):1139–1145. doi: 10.2215/CJN.04160320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guo, T., et al., Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19). JAMA Cardiol, 2020. [DOI] [PMC free article] [PubMed]
- 16.Lindner, D., et al., Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol, 2020. [DOI] [PMC free article] [PubMed]
- 17.Sierra F. Geroscience and the coronavirus pandemic: the whack-a-mole approach is not enough. J Am Geriatr Soc. 2020;68(5):951–952. doi: 10.1111/jgs.16489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yew WW, et al. Metformin as a host-directed therapeutic in tuberculosis: is there a promise? Tuberculosis (Edinb) 2019;115:76–80. doi: 10.1016/j.tube.2019.02.004. [DOI] [PubMed] [Google Scholar]
- 19.Zumla A, Wang FS, Ippolito G, Petrosillo N, Agrati C, Azhar EI, Chang C, el-Kafrawy SA, Osman M, Zitvogel L, Galle PR, Locatelli F, Gorman E, Cordon-Cardo C, O’Kane C, McAuley D, Maeurer M. Reducing mortality and morbidity in patients with severe COVID-19 disease by advancing ongoing trials of mesenchymal stromal (stem) cell (MSC) therapy - achieving global consensus and visibility for cellular host-directed therapies. Int J Infect Dis. 2020;96:431–439. doi: 10.1016/j.ijid.2020.05.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mitchell SJ, Bernier M, Mattison JA, Aon MA, Kaiser TA, Anson RM, Ikeno Y, Anderson RM, Ingram DK, de Cabo R. Daily fasting improves health and survival in male mice independent of diet composition and calories. Cell Metab. 2019;29(1):221–228. doi: 10.1016/j.cmet.2018.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hwangbo DS, et al. Mechanisms of lifespan regulation by calorie restriction and intermittent fasting in model organisms. Nutrients. 2020;12(4):1194. doi: 10.3390/nu12041194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bartley JM, Zhou X, Kuchel GA, Weinstock GM, Haynes L. Impact of age, caloric restriction, and influenza infection on mouse gut microbiome: an exploratory study of the role of age-related microbiome changes on influenza responses. Front Immunol. 2017;8:1164. doi: 10.3389/fimmu.2017.01164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yousefzadeh MJ, Zhu Y, McGowan SJ, Angelini L, Fuhrmann-Stroissnigg H, Xu M, Ling YY, Melos KI, Pirtskhalava T, Inman CL, McGuckian C, Wade EA, Kato JI, Grassi D, Wentworth M, Burd CE, Arriaga EA, Ladiges WL, Tchkonia T, Kirkland JL, Robbins PD, Niedernhofer LJ. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine. 2018;36:18–28. doi: 10.1016/j.ebiom.2018.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013;493(7432):338–345. doi: 10.1038/nature11861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M, STOP-NIDDM Trial Research Group Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: the STOP-NIDDM trial. JAMA. 2003;290(4):486–494. doi: 10.1001/jama.290.4.486. [DOI] [PubMed] [Google Scholar]
- 26.Mannick JB, et al. TORC1 inhibition enhances immune function and reduces infections in the elderly. Sci Transl Med. 2018;10(449):eaaq1564. doi: 10.1126/scitranslmed.aaq1564. [DOI] [PubMed] [Google Scholar]
- 27.Chen C, et al. mTOR regulation and therapeutic rejuvenation of aging hematopoietic stem cells. Sci Signal. 2009;2(98):ra75. doi: 10.1126/scisignal.2000559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mannick JB, et al. mTOR inhibition improves immune function in the elderly. Sci Transl Med. 2014;6(268):268ra179. doi: 10.1126/scitranslmed.3009892. [DOI] [PubMed] [Google Scholar]
- 29.Martin-Montalvo A, Mercken EM, Mitchell SJ, Palacios HH, Mote PL, Scheibye-Knudsen M, Gomes AP, Ward TM, Minor RK, Blouin MJ, Schwab M, Pollak M, Zhang Y, Yu Y, Becker KG, Bohr VA, Ingram DK, Sinclair DA, Wolf NS, Spindler SR, Bernier M, de Cabo R. Metformin improves healthspan and lifespan in mice. Nat Commun. 2013;4:2192. doi: 10.1038/ncomms3192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Barzilai N, Crandall JP, Kritchevsky SB, Espeland MA. Metformin as a tool to target aging. Cell Metab. 2016;23(6):1060–1065. doi: 10.1016/j.cmet.2016.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Justice JN, Niedernhofer L, Robbins PD, Aroda VR, Espeland MA, Kritchevsky SB, Kuchel GA, Barzilai N. Development of clinical trials to extend healthy lifespan. Cardiovasc Endocrinol Metab. 2018;7(4):80–83. doi: 10.1097/XCE.0000000000000159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.EY G. Flumamine, a new synthetic analgesic and antiflu drug. J Philippine Med Assoc. 1950;26:287–293. [PubMed] [Google Scholar]
- 33.Bailey CJ. Metformin: historical overview. Diabetologia. 2017;60(9):1566–1576. doi: 10.1007/s00125-017-4318-z. [DOI] [PubMed] [Google Scholar]
- 34.Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet, 1998. 352(9131): p. 854–65. [PubMed]
- 35.Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM, Diabetes Prevention Program Research Group Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kulkarni AS, Gubbi S, Barzilai N. Benefits of metformin in attenuating the hallmarks of aging. Cell Metab. 2020;32:15–30. doi: 10.1016/j.cmet.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang CS, Li M, Ma T, Zong Y, Cui J, Feng JW, Wu YQ, Lin SY, Lin SC. Metformin activates AMPK through the lysosomal pathway. Cell Metab. 2016;24(4):521–522. doi: 10.1016/j.cmet.2016.09.003. [DOI] [PubMed] [Google Scholar]
- 38.Pollak M. The effects of metformin on gut microbiota and the immune system as research frontiers. Diabetologia. 2017;60(9):1662–1667. doi: 10.1007/s00125-017-4352-x. [DOI] [PubMed] [Google Scholar]
- 39.Soukas AA, Hao H, Wu L. Metformin as anti-aging therapy: is it for everyone? Trends Endocrinol Metab. 2019;30(10):745–755. doi: 10.1016/j.tem.2019.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bridgeman SC, Ellison GC, Melton PE, Newsholme P, Mamotte CDS. Epigenetic effects of metformin: from molecular mechanisms to clinical implications. Diabetes Obes Metab. 2018;20(7):1553–1562. doi: 10.1111/dom.13262. [DOI] [PubMed] [Google Scholar]
- 41.Marcucci F, Romeo E, Caserta CA, Rumio C, Lefoulon F. Context-dependent pharmacological effects of metformin on the immune system. Trends Pharmacol Sci. 2020;41(3):162–171. doi: 10.1016/j.tips.2020.01.003. [DOI] [PubMed] [Google Scholar]
- 42.Ding L, Liang G, Yao Z, Zhang J, Liu R, Chen H, Zhou Y, Wu H, Yang B, He Q. Metformin prevents cancer metastasis by inhibiting M2-like polarization of tumor associated macrophages. Oncotarget. 2015;6(34):36441–36455. doi: 10.18632/oncotarget.5541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Uehara T, Eikawa S, Nishida M, Kunisada Y, Yoshida A, Fujiwara T, Kunisada T, Ozaki T, Udono H. Metformin induces CD11b+−cell-mediated growth inhibition of an osteosarcoma: implications for metabolic reprogramming of myeloid cells and anti-tumor effects. Int Immunol. 2019;31(4):187–198. doi: 10.1093/intimm/dxy079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pereira FV, Melo ACL, Low JS, de Castro ÍA, Braga TT, Almeida DC, Batista de Lima AGU, Hiyane MI, Correa-Costa M, Andrade-Oliveira V, Origassa CST, Pereira RM, Kaech SM, Rodrigues EG, Câmara NOS. Metformin exerts antitumor activity via induction of multiple death pathways in tumor cells and activation of a protective immune response. Oncotarget. 2018;9(40):25808–25825. doi: 10.18632/oncotarget.25380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Eikawa S, Nishida M, Mizukami S, Yamazaki C, Nakayama E, Udono H. Immune-mediated antitumor effect by type 2 diabetes drug, metformin. Proc Natl Acad Sci U S A. 2015;112(6):1809–1814. doi: 10.1073/pnas.1417636112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Qing L, Fu J, Wu P, Zhou Z, Yu F, Tang J. Metformin induces the M2 macrophage polarization to accelerate the wound healing via regulating AMPK/mTOR/NLRP3 inflammasome singling pathway. Am J Transl Res. 2019;11(2):655–668. [PMC free article] [PubMed] [Google Scholar]
- 47.Sun Y, Tian T, Gao J, Liu X, Hou H, Cao R, Li B, Quan M, Guo L. Metformin ameliorates the development of experimental autoimmune encephalomyelitis by regulating T helper 17 and regulatory T cells in mice. J Neuroimmunol. 2016;292:58–67. doi: 10.1016/j.jneuroim.2016.01.014. [DOI] [PubMed] [Google Scholar]
- 48.Park MJ, Lee SY, Moon SJ, Son HJ, Lee SH, Kim EK, Byun JK, Shin DY, Park SH, Yang CW, Cho ML. Metformin attenuates graft-versus-host disease via restricting mammalian target of rapamycin/signal transducer and activator of transcription 3 and promoting adenosine monophosphate-activated protein kinase-autophagy for the balance between T helper 17 and Tregs. Transl Res. 2016;173:115–130. doi: 10.1016/j.trsl.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 49.Lee SY, Moon SJ, Kim EK, Seo HB, Yang EJ, Son HJ, Kim JK, Min JK, Park SH, Cho ML. Metformin suppresses systemic autoimmunity in Roquin(san/san) mice through inhibiting B cell differentiation into plasma cells via regulation of AMPK/mTOR/STAT3. J Immunol. 2017;198(7):2661–2670. doi: 10.4049/jimmunol.1403088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Blagih J, Coulombe F, Vincent EE, Dupuy F, Galicia-Vázquez G, Yurchenko E, Raissi TC, van der Windt GJW, Viollet B, Pearce EL, Pelletier J, Piccirillo CA, Krawczyk CM, Divangahi M, Jones RG. The energy sensor AMPK regulates T cell metabolic adaptation and effector responses in vivo. Immunity. 2015;42(1):41–54. doi: 10.1016/j.immuni.2014.12.030. [DOI] [PubMed] [Google Scholar]
- 51.Singhal A, et al. Metformin as adjunct antituberculosis therapy. Sci Transl Med. 2014;6(263):263ra159. doi: 10.1126/scitranslmed.3009885. [DOI] [PubMed] [Google Scholar]
- 52.Channappanavar R, Fehr AR, Zheng J, Wohlford-Lenane C, Abrahante JE, Mack M, Sompallae R, McCray PB, Jr, Meyerholz DK, Perlman S. IFN-I response timing relative to virus replication determines MERS coronavirus infection outcomes. J Clin Invest. 2019;129(9):3625–3639. doi: 10.1172/JCI126363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nikolich-Zugich J, Knox KS, Rios CT, Natt B, Bhattacharya D, Fain MJ. SARS-CoV-2 and COVID-19 in older adults: what we may expect regarding pathogenesis, immune responses, and outcomes. Geroscience. 2020;42(2):505–514. doi: 10.1007/s11357-020-00186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Guo YR, Cao QD, Hong ZS, Tan YY, Chen SD, Jin HJ, Tan KS, Wang DY, Yan Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Mil Med Res. 2020;7(1):11. doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Meftahi GH, Jangravi Z, Sahraei H, Bahari Z. The possible pathophysiology mechanism of cytokine storm in elderly adults with COVID-19 infection: the contribution of "inflame-aging". Inflamm Res. 2020;69:825–839. doi: 10.1007/s00011-020-01372-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schuiveling M, Vazirpanah N, Radstake TRDJ, Zimmermann M, Broen JCA. Metformin, a new era for an old drug in the treatment of immune mediated disease? Curr Drug Targets. 2018;19(8):945–959. doi: 10.2174/1389450118666170613081730. [DOI] [PubMed] [Google Scholar]
- 57.Vasamsetti SB, Karnewar S, Kanugula AK, Thatipalli AR, Kumar JM, Kotamraju S. Metformin inhibits monocyte-to-macrophage differentiation via AMPK-mediated inhibition of STAT3 activation: potential role in atherosclerosis. Diabetes. 2015;64(6):2028–2041. doi: 10.2337/db14-1225. [DOI] [PubMed] [Google Scholar]
- 58.Ba W, Xu Y, Yin G, Yang J, Wang R, Chi S, Wang Y, Li C. Metformin inhibits pro-inflammatory responses via targeting nuclear factor-kappaB in HaCaT cells. Cell Biochem Funct. 2019;37(1):4–10. doi: 10.1002/cbf.3367. [DOI] [PubMed] [Google Scholar]
- 59.Cameron AR, Morrison VL, Levin D, Mohan M, Forteath C, Beall C, McNeilly AD, Balfour DJK, Savinko T, Wong AKF, Viollet B, Sakamoto K, Fagerholm SC, Foretz M, Lang CC, Rena G. Anti-inflammatory effects of metformin irrespective of diabetes status. Circ Res. 2016;119(5):652–665. doi: 10.1161/CIRCRESAHA.116.308445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tizazu AM, Nyunt MSZ, Cexus O, Suku K, Mok E, Xian CH, Chong J, Tan C, How W, Hubert S, Combet E, Fulop T, Ng TP, Larbi A. Metformin monotherapy downregulates diabetes-associated inflammatory status and impacts on mortality. Front Physiol. 2019;10:572. doi: 10.3389/fphys.2019.00572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zmijewski JW, Lorne E, Zhao X, Tsuruta Y, Sha Y, Liu G, Siegal GP, Abraham E. Mitochondrial respiratory complex I regulates neutrophil activation and severity of lung injury. Am J Respir Crit Care Med. 2008;178(2):168–179. doi: 10.1164/rccm.200710-1602OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ouyang J, Isnard S, Lin J, Fombuena B, Marette A, Routy B, Chen Y, Routy JP. Metformin effect on gut microbiota: insights for HIV-related inflammation. AIDS Res Ther. 2020;17(1):10. doi: 10.1186/s12981-020-00267-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wu H, Esteve E, Tremaroli V, Khan MT, Caesar R, Mannerås-Holm L, Ståhlman M, Olsson LM, Serino M, Planas-Fèlix M, Xifra G, Mercader JM, Torrents D, Burcelin R, Ricart W, Perkins R, Fernàndez-Real JM, Bäckhed F. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat Med. 2017;23(7):850–858. doi: 10.1038/nm.4345. [DOI] [PubMed] [Google Scholar]
- 64.Zhang X, Zhao Y, Xu J, Xue Z, Zhang M, Pang X, Zhang X, Zhao L. Modulation of gut microbiota by berberine and metformin during the treatment of high-fat diet-induced obesity in rats. Sci Rep. 2015;5:14405. doi: 10.1038/srep14405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Forslund K, et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature. 2015;528(7581):262–266. doi: 10.1038/nature15766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ma W, Chen J, Meng Y, Yang J, Cui Q, Zhou Y. Metformin alters gut microbiota of healthy mice: implication for its potential role in gut microbiota homeostasis. Front Microbiol. 2018;9:1336. doi: 10.3389/fmicb.2018.01336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ahmadi S, Razazan A, Nagpal R, Jain S, Wang B, Mishra SP, Wang S, Justice J, Ding J, McClain DA, Kritchevsky SB, Kitzman D, Yadav H. Metformin reduces aging-related leaky gut and improves cognitive function by beneficially modulating gut microbiome/goblet cell/mucin axis. J Gerontol A Biol Sci Med Sci. 2020;75(7):e9–e21. doi: 10.1093/gerona/glaa056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Shin NR, Lee JC, Lee HY, Kim MS, Whon TW, Lee MS, Bae JW. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut. 2014;63(5):727–735. doi: 10.1136/gutjnl-2012-303839. [DOI] [PubMed] [Google Scholar]
- 69.Velavan TP, Meyer CG. The COVID-19 epidemic. Tropical Med Int Health. 2020;25(3):278–280. doi: 10.1111/tmi.13383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chen Y, Liu Q, Guo D. Emerging coronaviruses: genome structure, replication, and pathogenesis. J Med Virol. 2020;92(4):418–423. doi: 10.1002/jmv.25681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Pedersen SF, Ho YC. SARS-CoV-2: a storm is raging. J Clin Invest. 2020;130(5):2202–2205. doi: 10.1172/JCI137647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun. 2020;109:102433. doi: 10.1016/j.jaut.2020.102433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ye Q, Wang B, Mao J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J Inf Secur. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Møller R, Jordan TX, Oishi K, Panis M, Sachs D, Wang TT, Schwartz RE, Lim JK, Albrecht RA, tenOever BR. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020;181(5):1036–1045. doi: 10.1016/j.cell.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Liu J, Li S, Liu J, Liang B, Wang X, Wang H, Li W, Tong Q, Yi J, Zhao L, Xiong L, Guo C, Tian J, Luo J, Yao J, Pang R, Shen H, Peng C, Liu T, Zhang Q, Wu J, Xu L, Lu S, Wang B, Weng Z, Han C, Zhu H, Zhou R, Zhou H, Chen X, Ye P, Zhu B, Wang L, Zhou W, He S, He Y, Jie S, Wei P, Zhang J, Lu Y, Wang W, Zhang L, Li L, Zhou F, Wang J, Dittmer U, Lu M, Hu Y, Yang D, Zheng X. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Moore JB, June CH. Cytokine release syndrome in severe COVID-19. Science. 2020;368(6490):473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]
- 77.Luo P, Qiu L, Liu Y, Liu XL, Zheng JL, Xue HY, Liu WH, Liu D, Li J. Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis. Am J Trop Med Hyg. 2020;103:69–72. doi: 10.4269/ajtmh.20-0375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bramante, C., et al., Observational study of metformin and risk of mortality in patients hospitalized with Covid-19. MedRxiv, 2020.
- 79.Naicker N, Sigal A, Naidoo K. Metformin as host-directed therapy for TB treatment: scoping review. Front Microbiol. 2020;11:435. doi: 10.3389/fmicb.2020.00435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Degner NR, Wang JY, Golub JE, Karakousis PC. Metformin use reverses the increased mortality associated with diabetes mellitus during tuberculosis treatment. Clin Infect Dis. 2018;66(2):198–205. doi: 10.1093/cid/cix819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Malik F, Mehdi SF, Ali H, Patel P, Basharat A, Kumar A, Ashok F, Stein J, Brima W, Malhotra P, Roth J. Is metformin poised for a second career as an antimicrobial? Diabetes Metab Res Rev. 2018;34(4):e2975. doi: 10.1002/dmrr.2975. [DOI] [PubMed] [Google Scholar]
- 82.Brima W, Eden DJ, Mehdi SF, Bravo M, Wiese MM, Stein J, Almonte V, Zhao D, Kurland I, Pessin JE, Zima T, Tanowitz HB, Weiss LM, Roth J, Nagajyothi F. The brighter (and evolutionarily older) face of the metabolic syndrome: evidence from Trypanosoma cruzi infection in CD-1 mice. Diabetes Metab Res Rev. 2015;31(4):346–359. doi: 10.1002/dmrr.2636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Mallia P, Webber J, Gill SK, Trujillo-Torralbo MB, Calderazzo MA, Finney L, Bakhsoliani E, Farne H, Singanayagam A, Footitt J, Hewitt R, Kebadze T, Aniscenko J, Padmanaban V, Molyneaux PL, Adcock IM, Barnes PJ, Ito K, Elkin SL, Kon OM, Cookson WO, Moffat MF, Johnston SL, Tregoning JS. Role of airway glucose in bacterial infections in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2018;142(3):815–823. doi: 10.1016/j.jaci.2017.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Patkee WR, et al. Metformin prevents the effects of Pseudomonas aeruginosa on airway epithelial tight junctions and restricts hyperglycaemia-induced bacterial growth. J Cell Mol Med. 2016;20(4):758–764. doi: 10.1111/jcmm.12784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kalsi KK, Garnett JP, Patkee W, Weekes A, Dockrell ME, Baker EH, Baines DL. Metformin attenuates the effect of Staphylococcus aureus on airway tight junctions by increasing PKCzeta-mediated phosphorylation of occludin. J Cell Mol Med. 2019;23(1):317–327. doi: 10.1111/jcmm.13929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kajiwara C, Kusaka Y, Kimura S, Yamaguchi T, Nanjo Y, Ishii Y, Udono H, Standiford TJ, Tateda K. Metformin mediates protection against Legionella pneumonia through activation of AMPK and mitochondrial reactive oxygen species. J Immunol. 2018;200(2):623–631. doi: 10.4049/jimmunol.1700474. [DOI] [PubMed] [Google Scholar]
- 87.Mortensen E, Anzueto A. Association of metformin and mortality for patients with diabetes who are hospitalized with pneumonia. Eur Respir J. 2018;52:PA2639. [Google Scholar]
- 88.Yen FS, Wei JCC, Yang YC, Hsu CC, Hwu CM. Respiratory outcomes of metformin use in patients with type 2 diabetes and chronic obstructive pulmonary disease. Sci Rep. 2020;10(1):10298. doi: 10.1038/s41598-020-67338-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Xun YH, Zhang YJ, Pan QC, Mao RC, Qin YL, Liu HY, Zhang YM, Yu YS, Tang ZH, Lu MJ, Zang GQ, Zhang JM. Metformin inhibits hepatitis B virus protein production and replication in human hepatoma cells. J Viral Hepat. 2014;21(8):597–603. doi: 10.1111/jvh.12187. [DOI] [PubMed] [Google Scholar]
- 90.Chen CI, Kuan CF, Fang YA, Liu SH, Liu JC, Wu LL, Chang CJ, Yang HC, Hwang J, Miser JS, Wu SY. Cancer risk in HBV patients with statin and metformin use: a population-based cohort study. Medicine (Baltimore) 2015;94(6):e462. doi: 10.1097/MD.0000000000000462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hofmeister MG, Rosenthal EM, Barker LK, Rosenberg ES, Barranco MA, Hall EW, Edlin BR, Mermin J, Ward JW, Ryerson AB. Estimating prevalence of hepatitis C virus infection in the United States, 2013-2016. Hepatology. 2019;69(3):1020–1031. doi: 10.1002/hep.30297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Harris K, Smith L. Safety and efficacy of metformin in patients with type 2 diabetes mellitus and chronic hepatitis C. Ann Pharmacother. 2013;47(10):1348–1352. doi: 10.1177/1060028013503108. [DOI] [PubMed] [Google Scholar]
- 93.Sheth SH, Larson RJ. The efficacy and safety of insulin-sensitizing drugs in HIV-associated lipodystrophy syndrome: a meta-analysis of randomized trials. BMC Infect Dis. 2010;10:183. doi: 10.1186/1471-2334-10-183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lamarca K, García Sarasola A, Vidal F, Domingo P. Drug therapies for HIV-related metabolic disorders. Expert Opin Pharmacother. 2016;17(10):1327–1338. doi: 10.1080/14656566.2016.1187133. [DOI] [PubMed] [Google Scholar]
- 95.Dagenais-Lussier X, Mouna A, Routy JP, Tremblay C, Sekaly RP, el-Far M, Grevenynghe J. Current topics in HIV-1 pathogenesis: the emergence of deregulated immuno-metabolism in HIV-infected subjects. Cytokine Growth Factor Rev. 2015;26(6):603–613. doi: 10.1016/j.cytogfr.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 96.Lai SW, Chen PC, Liao KF, Muo CH, Lin CC, Sung FC. Risk of hepatocellular carcinoma in diabetic patients and risk reduction associated with anti-diabetic therapy: a population-based cohort study. Am J Gastroenterol. 2012;107(1):46–52. doi: 10.1038/ajg.2011.384. [DOI] [PubMed] [Google Scholar]
- 97.Chen X, Chen H, Zhang Z, Fu Y, Han X, Zhang Y, Xu J, Ding H, Cui H, Dong T, Shang H, Jiang Y. Elevated CD54 expression renders CD4+ T cells susceptible to natural killer cell-mediated killing. J Infect Dis. 2019;220(12):1892–1903. doi: 10.1093/infdis/jiz413. [DOI] [PubMed] [Google Scholar]
- 98.Shikuma CM, Chew GM, Kohorn L, Souza SA, Chow D, SahBandar IN, Park EY, Hanks N, Gangcuangco LMA, Gerschenson M, Ndhlovu LC. Short communication: metformin reduces CD4 T cell exhaustion in HIV-infected adults on suppressive antiretroviral therapy. AIDS Res Hum Retrovir. 2020;36(4):303–305. doi: 10.1089/aid.2019.0078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Routy JP, Isnard S, Mehraj V, Ostrowski M, Chomont N, Ancuta P, Ponte R, Planas D, Dupuy FP, Angel JB, Lilac Study Group Effect of metformin on the size of the HIV reservoir in non-diabetic ART-treated individuals: single-arm non-randomised Lilac pilot study protocol. BMJ Open. 2019;9(4):e028444. doi: 10.1136/bmjopen-2018-028444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Doyle MA, Singer J, Lee T, Muir M, Cooper C. Improving treatment and liver fibrosis outcomes with metformin in HCV-HIV co-infected and HCV mono-infected patients with insulin resistance: study protocol for a randomized controlled trial. Trials. 2016;17(1):331. doi: 10.1186/s13063-016-1454-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hammerstad SS, et al. Diabetes and hepatitis C: a two-way association. Front Endocrinol (Lausanne) 2015;6:134. doi: 10.3389/fendo.2015.00134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Dillon SM, Kibbie J, Lee EJ, Guo K, Santiago ML, Austin GL, Gianella S, Landay AL, Donovan AM, Frank DN, McCarter MD, Wilson CC. Low abundance of colonic butyrate-producing bacteria in HIV infection is associated with microbial translocation and immune activation. AIDS. 2017;31(4):511–521. doi: 10.1097/QAD.0000000000001366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gonzalez-Hernandez LA, et al. Alterations in bacterial communities, SCFA and biomarkers in an elderly HIV-positive and HIV-negative population in western Mexico. BMC Infect Dis. 2019;19(1):234. doi: 10.1186/s12879-019-3867-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Diaz A, Romero M, Vazquez T, Lechner S, Blomberg BB, Frasca D. Metformin improves in vivo and in vitro B cell function in individuals with obesity and type-2 diabetes. Vaccine. 2017;35(20):2694–2700. doi: 10.1016/j.vaccine.2017.03.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Agarwal D, Schmader KE, Kossenkov AV, Doyle S, Kurupati R, Ertl HCJ. Immune response to influenza vaccination in the elderly is altered by chronic medication use. Immun Ageing. 2018;15:19. doi: 10.1186/s12979-018-0124-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Campbell JM, Bellman SM, Stephenson MD, Lisy K. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: a systematic review and meta-analysis. Ageing Res Rev. 2017;40:31–44. doi: 10.1016/j.arr.2017.08.003. [DOI] [PubMed] [Google Scholar]
- 107.Wang CP, Lorenzo C, Habib SL, Jo B, Espinoza SE. Differential effects of metformin on age related comorbidities in older men with type 2 diabetes. J Diabetes Complicat. 2017;31(4):679–686. doi: 10.1016/j.jdiacomp.2017.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Espinoza SE, Musi N, Wang CP, Michalek J, Orsak B, Romo T, Powers B, Conde A, Moris M, Bair-Kelps D, Li Y, Ganapathy V, Jergensen TE, Kelly LC, Jiwani R. Rationale and study design of a randomized clinical trial of metformin to prevent frailty in older adults with prediabetes. J Gerontol A Biol Sci Med Sci. 2020;75(1):102–109. doi: 10.1093/gerona/glz078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Cawthon, P., et al., Assessing the impact of the covid-19 pandemic and accompanying mitigation efforts on older adults. J Gerontol A Biol Sci Med Sci, 2020. [DOI] [PMC free article] [PubMed]
- 110.Kivi, M., I. Hansson, and P. Bjalkebring, Up and about: older adults’ wellbeing during the COVID-19 pandemic in a Swedish longitudinal study. J Gerontol B Psychol Sci Soc Sci, 2020. [DOI] [PMC free article] [PubMed]
- 111.Moiseeva O, Deschênes-Simard X, St-Germain E, Igelmann S, Huot G, Cadar AE, Bourdeau V, Pollak MN, Ferbeyre G. Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-kappaB activation. Aging Cell. 2013;12(3):489–498. doi: 10.1111/acel.12075. [DOI] [PubMed] [Google Scholar]
- 112.Bauer PV, Duca FA, Waise TMZ, Rasmussen BA, Abraham MA, Dranse HJ, Puri A, O’Brien CA, Lam TKT. Metformin alters upper small intestinal microbiota that impact a glucose-SGLT1-sensing glucoregulatory pathway. Cell Metab. 2018;27(1):101–117. doi: 10.1016/j.cmet.2017.09.019. [DOI] [PubMed] [Google Scholar]
- 113.Lee H, Ko G. Effect of metformin on metabolic improvement and gut microbiota. Appl Environ Microbiol. 2014;80(19):5935–5943. doi: 10.1128/AEM.01357-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Cabreiro F, Au C, Leung KY, Vergara-Irigaray N, Cochemé HM, Noori T, Weinkove D, Schuster E, Greene NDE, Gems D. Metformin retards aging in C. elegans by altering microbial folate and methionine metabolism. Cell. 2013;153(1):228–239. doi: 10.1016/j.cell.2013.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hawley SA, Gadalla AE, Olsen GS, Hardie DG. The antidiabetic drug metformin activates the AMP-activated protein kinase cascade via an adenine nucleotide-independent mechanism. Diabetes. 2002;51(8):2420–2425. doi: 10.2337/diabetes.51.8.2420. [DOI] [PubMed] [Google Scholar]
- 116.Onken B, Driscoll M. Metformin induces a dietary restriction-like state and the oxidative stress response to extend C. elegans Healthspan via AMPK, LKB1, and SKN-1. PLoS One. 2010;5(1):e8758. doi: 10.1371/journal.pone.0008758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Cuyas E, et al. Metformin is a direct SIRT1-activating compound: computational modeling and experimental validation. Front Endocrinol (Lausanne) 2018;9:657. doi: 10.3389/fendo.2018.00657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Canto C, et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 2009;458(7241):1056–1060. doi: 10.1038/nature07813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Kalender A, Selvaraj A, Kim SY, Gulati P, Brûlé S, Viollet B, Kemp BE, Bardeesy N, Dennis P, Schlager JJ, Marette A, Kozma SC, Thomas G. Metformin, independent of AMPK, inhibits mTORC1 in a rag GTPase-dependent manner. Cell Metab. 2010;11(5):390–401. doi: 10.1016/j.cmet.2010.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Dowling RJ, et al. Metformin inhibits mammalian target of rapamycin-dependent translation initiation in breast cancer cells. Cancer Res. 2007;67(22):10804–10812. doi: 10.1158/0008-5472.CAN-07-2310. [DOI] [PubMed] [Google Scholar]
- 121.Ben Sahra I, Regazzetti C, Robert G, Laurent K, le Marchand-Brustel Y, Auberger P, Tanti JF, Giorgetti-Peraldi S, Bost F. Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Cancer Res. 2011;71(13):4366–4372. doi: 10.1158/0008-5472.CAN-10-1769. [DOI] [PubMed] [Google Scholar]
- 122.Howell JJ, Hellberg K, Turner M, Talbott G, Kolar MJ, Ross DS, Hoxhaj G, Saghatelian A, Shaw RJ, Manning BD. Metformin inhibits hepatic mTORC1 signaling via dose-dependent mechanisms involving AMPK and the TSC complex. Cell Metab. 2017;25(2):463–471. doi: 10.1016/j.cmet.2016.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Sarfstein R, Friedman Y, Attias-Geva Z, Fishman A, Bruchim I, Werner H. Metformin downregulates the insulin/IGF-I signaling pathway and inhibits different uterine serous carcinoma (USC) cells proliferation and migration in p53-dependent or -independent manners. PLoS One. 2013;8(4):e61537. doi: 10.1371/journal.pone.0061537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Na HJ, Park JS, Pyo JH, Lee SH, Jeon HJ, Kim YS, Yoo MA. Mechanism of metformin: inhibition of DNA damage and proliferative activity in Drosophila midgut stem cell. Mech Ageing Dev. 2013;134(9):381–390. doi: 10.1016/j.mad.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 125.Lee YS, et al. Combined metformin and resveratrol confers protection against UVC-induced DNA damage in A549 lung cancer cells via modulation of cell cycle checkpoints and DNA repair. Oncol Rep. 2016;35(6):3735–3741. doi: 10.3892/or.2016.4740. [DOI] [PubMed] [Google Scholar]
- 126.Vazquez-Martin A, Oliveras-Ferraros C, Cufí S, Martin-Castillo B, Menendez JA. Metformin activates an ataxia telangiectasia mutated (ATM)/Chk2-regulated DNA damage-like response. Cell Cycle. 2011;10(9):1499–1501. doi: 10.4161/cc.10.9.15423. [DOI] [PubMed] [Google Scholar]
- 127.Wang GY, Bi YG, Liu XD, Zhao Y, Han JF, Wei M, Zhang QY. Autophagy was involved in the protective effect of metformin on hyperglycemia-induced cardiomyocyte apoptosis and Connexin43 downregulation in H9c2 cells. Int J Med Sci. 2017;14(7):698–704. doi: 10.7150/ijms.19800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Wang Y, Xu W, Yan Z, Zhao W, Mi J, Li J, Yan H. Metformin induces autophagy and G0/G1 phase cell cycle arrest in myeloma by targeting the AMPK/mTORC1 and mTORC2 pathways. J Exp Clin Cancer Res. 2018;37(1):63. doi: 10.1186/s13046-018-0731-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Tsai HH, Lai HY, Chen YC, Li CF, Huang HS, Liu HS, Tsai YS, Wang JM. Metformin promotes apoptosis in hepatocellular carcinoma through the CEBPD-induced autophagy pathway. Oncotarget. 2017;8(8):13832–13845. doi: 10.18632/oncotarget.14640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.El-Mir MY, et al. Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J Biol Chem. 2000;275(1):223–228. doi: 10.1074/jbc.275.1.223. [DOI] [PubMed] [Google Scholar]
- 131.De Haes W, et al. Metformin promotes lifespan through mitohormesis via the peroxiredoxin PRDX-2. Proc Natl Acad Sci U S A. 2014;111(24):E2501–E2509. doi: 10.1073/pnas.1321776111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Wheaton WW, Weinberg SE, Hamanaka RB, Soberanes S, Sullivan LB, Anso E, Glasauer A, Dufour E, Mutlu GM, Budigner GRS, Chandel NS. Metformin inhibits mitochondrial complex I of cancer cells to reduce tumorigenesis. Elife. 2014;3:e02242. doi: 10.7554/eLife.02242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Algire C, Moiseeva O, Deschenes-Simard X, Amrein L, Petruccelli L, Birman E, Viollet B, Ferbeyre G, Pollak MN. Metformin reduces endogenous reactive oxygen species and associated DNA damage. Cancer Prev Res (Phila) 2012;5(4):536–543. doi: 10.1158/1940-6207.CAPR-11-0536. [DOI] [PubMed] [Google Scholar]
- 134.Aatsinki SM, Buler M, Salomäki H, Koulu M, Pavek P, Hakkola J. Metformin induces PGC-1alpha expression and selectively affects hepatic PGC-1alpha functions. Br J Pharmacol. 2014;171(9):2351–2363. doi: 10.1111/bph.12585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Fang J, Yang J, Wu X, Zhang G, Li T, Wang X', Zhang H, Wang CC, Liu GH, Wang L. Metformin alleviates human cellular aging by upregulating the endoplasmic reticulum glutathione peroxidase 7. Aging Cell. 2018;17(4):e12765. doi: 10.1111/acel.12765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Neumann B, Baror R, Zhao C, Segel M, Dietmann S, Rawji KS, Foerster S, McClain CR, Chalut K, van Wijngaarden P, Franklin RJM. Metformin restores CNS remyelination capacity by rejuvenating aged stem cells. Cell Stem Cell. 2019;25(4):473–485. doi: 10.1016/j.stem.2019.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Fatt M, Hsu K, He L, Wondisford F, Miller FD, Kaplan DR, Wang J. Metformin acts on two different molecular pathways to enhance adult neural precursor proliferation/self-renewal and differentiation. Stem Cell Reports. 2015;5(6):988–995. doi: 10.1016/j.stemcr.2015.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Na HJ, Pyo JH, Jeon HJ, Park JS, Chung HY, Yoo MA. Deficiency of Atg6 impairs beneficial effect of metformin on intestinal stem cell aging in Drosophila. Biochem Biophys Res Commun. 2018;498(1):18–24. doi: 10.1016/j.bbrc.2018.02.191. [DOI] [PubMed] [Google Scholar]
- 139.Noren Hooten N, Martin-Montalvo A, Dluzen DF, Zhang Y, Bernier M, Zonderman AB, Becker KG, Gorospe M, de Cabo R, Evans MK. Metformin-mediated increase in DICER1 regulates microRNA expression and cellular senescence. Aging Cell. 2016;15(3):572–581. doi: 10.1111/acel.12469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Diman A, Boros J, Poulain F, Rodriguez J, Purnelle M, Episkopou H, Bertrand L, Francaux M, Deldicque L, Decottignies A. Nuclear respiratory factor 1 and endurance exercise promote human telomere transcription. Sci Adv. 2016;2(7):e1600031. doi: 10.1126/sciadv.1600031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Smieszek A, et al. Antioxidant and anti-senescence effect of metformin on mouse olfactory ensheathing cells (mOECs) may be associated with increased brain-derived neurotrophic factor levels-an ex vivo study. Int J Mol Sci. 2017:18(4). [DOI] [PMC free article] [PubMed]
- 142.Jiang X, et al. Metformin reduces the senescence of renal tubular epithelial cells in diabetic nephropathy via the MBNL1/miR-130a-3p/STAT3 pathway. Oxidative Med Cell Longev. 2020;2020:8708236. doi: 10.1155/2020/8708236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Yu LL, Zhu M, Huang Y, Zhao YM, Wen JJ, Yang XJ, Wu P. Metformin relieves acute respiratory distress syndrome by reducing miR-138 expression. Eur Rev Med Pharmacol Sci. 2018;22(16):5355–5363. doi: 10.26355/eurrev_201808_15737. [DOI] [PubMed] [Google Scholar]
- 144.Wang G, Song Y, Feng W, Liu L, Zhu Y, Xie X, Pan Y, Ke R, Li S, Li F, Yang L, Li M. Activation of AMPK attenuates LPS-induced acute lung injury by upregulation of PGC1alpha and SOD1. Exp Ther Med. 2016;12(3):1551–1555. doi: 10.3892/etm.2016.3465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Gregoire M, et al. Impaired efferocytosis and neutrophil extracellular trap clearance by macrophages in ARDS. Eur Respir J. 2018;52(2):1702590. doi: 10.1183/13993003.02590-2017. [DOI] [PubMed] [Google Scholar]
- 146.Rangarajan S, Bone NB, Zmijewska AA, Jiang S, Park DW, Bernard K, Locy ML, Ravi S, Deshane J, Mannon RB, Abraham E, Darley-Usmar V, Thannickal VJ, Zmijewski JW. Metformin reverses established lung fibrosis in a bleomycin model. Nat Med. 2018;24(8):1121–1127. doi: 10.1038/s41591-018-0087-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Lachmandas E, Eckold C, Böhme J, Koeken VACM, Marzuki MB, Blok B, Arts RJW, Chen J, Teng KWW, Ratter J, Smolders EJ, van den Heuvel C, Stienstra R, Dockrell HM, Newell E, Netea MG, Singhal A, Cliff JM, van Crevel R. Metformin alters human host responses to mycobacterium tuberculosis in healthy subjects. J Infect Dis. 2019;220(1):139–150. doi: 10.1093/infdis/jiz064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Rodriguez-Carlos A, Valdez-Miramontes C, Marin-Luevano P, González-Curiel I, Enciso-Moreno JA, Rivas-Santiago B. Metformin promotes mycobacterium tuberculosis killing and increases the production of human beta-defensins in lung epithelial cells and macrophages. Microbes Infect. 2020;22(3):111–118. doi: 10.1016/j.micinf.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 149.Dutta NK, Pinn ML, Karakousis PC. Metformin adjunctive therapy does not improve the sterilizing activity of the first-line antitubercular regimen in mice. Antimicrob Agents Chemother. 2017;61(8):e00652. doi: 10.1128/AAC.00652-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Gill SK, Hui K, Farne H, Garnett JP, Baines DL, Moore LSP, Holmes AH, Filloux A, Tregoning JS. Increased airway glucose increases airway bacterial load in hyperglycaemia. Sci Rep. 2016;6:27636. doi: 10.1038/srep27636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Xiao Y, Liu F, Li S, Jiang N, Yu C, Zhu X, Qin Y, Hui J, Meng L, Song C, Li XF, Liu Y. Metformin promotes innate immunity through a conserved PMK-1/p38 MAPK pathway. Virulence. 2020;11(1):39–48. doi: 10.1080/21505594.2019.1706305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Garnett JP, Braun D, McCarthy AJ, Farrant MR, Baker EH, Lindsay JA, Baines DL. Fructose transport-deficient Staphylococcus aureus reveals important role of epithelial glucose transporters in limiting sugar-driven bacterial growth in airway surface liquid. Cell Mol Life Sci. 2014;71(23):4665–4673. doi: 10.1007/s00018-014-1635-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Garnett JP, Baker EH, Naik S, Lindsay JA, Knight GM, Gill S, Tregoning JS, Baines DL. Metformin reduces airway glucose permeability and hyperglycaemia-induced Staphylococcus aureus load independently of effects on blood glucose. Thorax. 2013;68(9):835–845. doi: 10.1136/thoraxjnl-2012-203178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Ruivo MTG, Vera IM, Sales-Dias J, Meireles P, Gural N, Bhatia SN, Mota MM, Mancio-Silva L. Host AMPK is a modulator of plasmodium liver infection. Cell Rep. 2016;16(10):2539–2545. doi: 10.1016/j.celrep.2016.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Vera IM, et al. Targeting liver stage malaria with metformin. JCI Insight. 2019;4(24):e127441. doi: 10.1172/jci.insight.127441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Miyakoda M, Bayarsaikhan G, Kimura D, Akbari M, Udono H, Yui K. Metformin promotes the protection of mice infected with Plasmodium yoelii independently of gammadelta T cell expansion. Front Immunol. 2018;9:2942. doi: 10.3389/fimmu.2018.02942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Del Campo JA, et al. Simvastatin and metformin inhibit cell growth in hepatitis C virus infected cells via mTOR increasing PTEN and autophagy. PLoS One. 2018;13(1):e0191805. doi: 10.1371/journal.pone.0191805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nakashima K, Takeuchi K, Chihara K, Hotta H, Sada K. Inhibition of hepatitis C virus replication through adenosine monophosphate-activated protein kinase-dependent and -independent pathways. Microbiol Immunol. 2011;55(11):774–782. doi: 10.1111/j.1348-0421.2011.00382.x. [DOI] [PubMed] [Google Scholar]
- 159.Romero-Gomez M, et al. Treatment of insulin resistance with metformin in naive genotype 1 chronic hepatitis C patients receiving peginterferon alfa-2a plus ribavirin. Hepatology. 2009;50(6):1702–1708. doi: 10.1002/hep.23206. [DOI] [PubMed] [Google Scholar]
- 160.Yu JW, Sun LJ, Zhao YH, Kang P, Yan BZ. The effect of metformin on the efficacy of antiviral therapy in patients with genotype 1 chronic hepatitis C and insulin resistance. Int J Infect Dis. 2012;16(6):e436–e441. doi: 10.1016/j.ijid.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 161.Sharifi AH, Mohammadi M, Fakharzadeh E, Zamini H, Zaer-Rezaee H, Jabbari H, Merat S. Efficacy of adding metformin to pegylated interferon and ribavirin in treatment naive patients with chronic hepatitis C: a randomized double-blind controlled trial. Middle East J Dig Dis. 2014;6(1):13–17. [PMC free article] [PubMed] [Google Scholar]
- 162.Shen C, Peng C, Shen B, Zhu Z, Xu N, Li T, Xie J. Sirolimus and metformin synergistically inhibit hepatocellular carcinoma cell proliferation and improve long-term survival in patients with HCC related to hepatitis B virus induced cirrhosis after liver transplantation. Oncotarget. 2016;7(38):62647–62656. doi: 10.18632/oncotarget.11591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kim JH, Alam MM, Park DB, Cho M, Lee SH, Jeon YJ, Yu DY, Kim TD, Kim HY, Cho CG, Lee DH. The effect of metformin treatment on CRBP-I level and cancer development in the liver of HBx transgenic mice. Korean J Physiol Pharmacol. 2013;17(5):455–461. doi: 10.4196/kjpp.2013.17.5.455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Jiang Z, Liu H. Metformin inhibits tumorigenesis in HBV-induced hepatocellular carcinoma by suppressing HULC overexpression caused by HBX. J Cell Biochem. 2018;119(6):4482–4495. doi: 10.1002/jcb.26555. [DOI] [PubMed] [Google Scholar]
- 165.Mehla R, Bivalkar-Mehla S, Zhang R, Handy I, Albrecht H, Giri S, Nagarkatti P, Nagarkatti M, Chauhan A. Bryostatin modulates latent HIV-1 infection via PKC and AMPK signaling but inhibits acute infection in a receptor independent manner. PLoS One. 2010;5(6):e11160. doi: 10.1371/journal.pone.0011160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Moseley CE, Webster RG, Aldridge JR. Peroxisome proliferator-activated receptor and AMP-activated protein kinase agonists protect against lethal influenza virus challenge in mice. Influenza Other Respir Viruses. 2010;4(5):307–311. doi: 10.1111/j.1750-2659.2010.00155.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Chen TM, Lin CC, Huang PT, Wen CF. Metformin associated with lower mortality in diabetic patients with early stage hepatocellular carcinoma after radiofrequency ablation. J Gastroenterol Hepatol. 2011;26(5):858–865. doi: 10.1111/j.1440-1746.2011.06664.x. [DOI] [PubMed] [Google Scholar]
- 168.Nkontchou G, Cosson E, Aout M, Mahmoudi A, Bourcier V, Charif I, Ganne-Carrie N, Grando-Lemaire V, Vicaut E, Trinchet JC, Beaugrand M. Impact of metformin on the prognosis of cirrhosis induced by viral hepatitis C in diabetic patients. J Clin Endocrinol Metab. 2011;96(8):2601–2608. doi: 10.1210/jc.2010-2415. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.