Abstract
The present study aimed to investigate the expression levels and clinical value of miR-365 and miR-25 in serum of patients with non-small cell lung cancer (NSCLC). Patients (180) diagnosed with NSCLC at the Affiliated Hospital of Guangdong Medical University from July 2011 to December 2013 were used as the experimental group. Volunteers (90) undergoing health examinations were used as the control group. The serum of the patients was collected after fasting in the morning. The expression levels of miR-365 and miR-25 in the serum of patients was assessed by quantitative real-time PCR (qRT-PCR), and the relationship among miR-365, miR-25 and the postoperative survival rate of NSCLC patients was analyzed. The relative expression level of miR-25 of patients with peripheral infiltration was significantly higher than that of patients without peripheral infiltration (P<0.05). There were significant differences in the relative expression level of miR-25 in different pathological grades and TNM stages, as well as with lymph node metastasis (P<0.05). The survival rate of NSCLC patients with high expression of miR-25 was significantly lower than that of NSCLC patients with low expression of miR-25 (P<0.05). The relative expression level of miR-365 of patients with peripheral infiltration was significantly lower than that of patients without peripheral infiltration (P<0.05). There were significant differences in the relative expression level of miR-365 in different pathological grades and TNM stages, as well as with lymph node metastasis (P<0.05). The survival rate of NSCLC patients with high expression of miR-365 was significantly higher than that of NSCLC patients with low expression of miR-365 (P<0.05). In conclusion, the expression levels of miR-25 and miR-365 were different in the serum of NSCLC patients, and they were closely related to certain clinical characteristics such as peripheral infiltration, pathological grade, tumor diameter, TNM stage and lymph node metastasis. Moreover, it was revealed that miR-25 and miR-365 affected the 5-year survival rate of patients. miR-25 and miR-365 could be used as important tumor markers to evaluate the prognosis of NSCLC patients.
Keywords: miR-25, miR-365, NSCLC, clinical pathology, prognosis
Introduction
Globally, the morbidity and mortality of lung cancer is ranked first among cancers (1). Non-small cell lung cancer (NSCLC) is cytological subtype of lung cancer. More than 85% of patients with lung cancer are NSCLC patients (2). According to statistics, more than 1.3 million people succumb to lung cancer every year worldwide. In recent years, medical treatments of NSCLC have been continuously improved, however, the 5-year survival rate of patients with advanced NSCLC is less than 20% (3). At present, the treatments of lung cancer include radiotherapy, surgery and chemotherapy. Numerous NSCLC patients present with advanced stage when they are diagnosed, causing difficulty in treatment, high recurrence rate of NSCLC and poor prognosis (4). In order to improve the diagnosis and treatment of NSCLC, it is crucial to identify useful biomarkers that are used for early diagnosis and prognosis monitoring of NSCLC.
MicroRNA (miRNA) is an RNA whose length is equal to the length of 19 to 24 nucleotides, which exists in a variety of tissues and blood and can regulate the expression of numerous genes in the body (5). Aberrant expression of miRNAs may lead to tumorigenesis (6) and can affect the occurrence and progression of tumors. In addition, it can also be used to monitor the prognosis of cancers (7). miRNAs that exist in blood circulation are potential medical markers of tumors (8). The expression of miR-25 has been revealed to be upregulated in numerous tumors, and play an important role in regulating tumorigenesis. According to a study by Kondo et al (9) on the expression and the clinical effects of miR-25 in patients with breast cancer, inhibiting the expression of miR-25 could significantly attenuate the proliferation ability of breast cancer cells. Furthermore, miR-25 was revealed to be a potential diagnostic factor of breast cancer. It is speculated that miR-25 may be specifically expressed in NSCLC and has a monitoring function. miR-365 is a microRNA, which is similar to miR-25. A study by Zhang et al (10) indicated that low expression of miR-365 was revealed in human gastric cancer tissues and mouse gastric cancer models. In addition, overexpression of miR-365 could significantly inhibit the proliferation ability and tumorigenic ability of gastric cancer cells. A study by Han et al (11) revealed that high expression of miR-365 was exhibited in breast cancer tissues, and miR-365 was associated with the TNM stage of patients with breast cancer, and that the proliferation and invasion abilities of breast cancer cells could be suppressed by downregulating the expression level of miR-365. In a study by Zhang et al (12), it was revealed that miR-365 played a role in suppressing breast cancer. It is unknown whether miR-365 can suppress NSCLC or not.
Based on the aforementioned, miR-365 and miR-25 have been revealed to be aberrantly expressed in various cancers and involved in the progression of these cancers. Therefore, it is surmised that these two miRNAs may also be aberrantly expressed in NSCLC and be related to the progression of the disease. However, there are few studies concerning the clinical role of miR-365 and miR-25 in NSCLC. To explore the clinical role of these two miRNAs in NSCLC, in the present study, the relationship between the expression of miR-25 and miR-365 in serum of NSCLC patients and their clinicopathological parameters was analyzed by detecting the expression of miR-25 and miR-365 in serum of NSCLC patients and comparing NSCLC patients with healthy volunteers in the control group. Furthermore, the relationship among miR-25, miR-365 and the postoperative 5-year survival rate of NSCLC patients was analyzed to identify new minimally invasive biological clinical factors for diagnosis, treatment and prognosis of NSCLC.
Materials and methods
Study subjects
In total, 180 patients, who were diagnosed with NSCLC at the Department of Pathology of Shenzhen Longhua District Central Hospital from July 2011 to December 2013, were used as the experimental group. The diagnosis of patients was based on WHO pathological histological diagnosis criteria. The study subjects had not received antitumor therapy such as radiotherapy and chemotherapy prior to enrollment in the study. The seventh edition of TNM staging diagnosis criteria for lung cancer was used, which was published by the International Association for the Study of Lung Cancer (13). Among the 180 patients, there were 108 males and 72 females. There were 82 patients >50 years old, and 98 patients were <50 years old. There were 99 patients with a history of smoking. As for the pathological grade, 56 patients were in low grade, 68 patients in middle grade, and 56 patients in high grade. There were 78 patients with lymph node metastasis and 102 patients with no lymph node metastasis. There were 88 patients with peripheral infiltration and 92 patients without peripheral infiltration. According to pathological stage, 43 patients were in stage I, 48 patients in stage II, 39 patients in stage III, and 50 patients in stage IV. The exclusion criteria was as follows: i) Patients with other tumor diseases except NSCLC; ii) patients with severe heart, liver, lung and other organ dysfunctions; iii) patients with incomplete clinical data. Volunteers (n=90), who took a health examination at the Outpatient Department during the same period, were used as the control group. These patients did not have basic diseases such as hypertension, hyperlipemia, and diabetes. General clinical data of the patients in the two groups and clinicopathological data of the patients in the experimental group were recorded. The patients, volunteers and their family members were informed, and an informed consent form was signed. This study was approved by the Medical Ethics Committee of the Affiliated Hospital of Guangdong Medical University.
Instruments and reagents
A real-time fluorescence quantitative PCR instrument (model no. 7300) was purchased from ABI; Thermo Fisher Scientific, Inc. A spectrophotometer (model no. DR5000) was purchased from Hach. A high-speed refrigerated centrifuge (model no. 5418) was purchased from Eppendorf. DEPC water was obtained from Sigma-Aldrich; Merck KGaA. A TRIzol kit was purchased from BioTeke Corporation. A reverse transcription kit was purchased from TaKaRa Bio, Inc. Internal reference primers of miR-25, miR-365 and U6 small nuclear RNA (RNU6B) were designed and synthesized by GeneCopoeia, Inc. The primer sequences are presented in Table I.
Table I.
Primer sequences of miR-25, miR-365 and U6.
| Gene | Forward primer | Reverse primer |
|---|---|---|
| miR-25 | 5′-ATCCAGTGCGTGTCGTG-3′ | 5′-TGCTCATTGCACTTGTCTC-3′ |
| miR-365 | 5′-CGTAATGCCCCTAAAAAT-3′ | 5′-GTGCAGGGTCCGAGGT-3′ |
| U6 | 5′-ATTGGAACGATACAGAGAAG-3′ | 5′-GGAACGCTTCACGAATTTG-3′ |
qRT-PCR
The peripheral blood of the patients in the experimental and control groups was collected by biochemical coagulation tubes, then placed in sterile blood collection tubes with a volume of 5 ml. Then, the serum was separated from the blood by a centrifuge and was stored in a refrigerator at −80°C. Serum samples of the patients in the two groups, which were stored in the refrigerator, were removed. Then the temperature of the serum samples was equilibrated with indoor temperature until they completely dissolved. The serum samples (500 µl) were transferred to new EP tubes. Then total RNA was extracted from the serum according to the instructions of the TRIzol serum extraction kits. Next, the concentration and purity of the extracted total RNA were detected by a DR5000 UV–VIS spectrophotometer. Lastly, 2 µl of total RNA was collected, and was reversely-transcribed into cDNA according to the instructions of the TaKaRa reverse transcription kits. cDNA was stored at −20°C. U6 was used as an internal reference gene. The reaction system was as follows: 10 µl of PCR Premix, 2 µl of upstream primers (10X), 2 µl of downstream primers (10X), 5 µl of dd water (Rnase- and Dnase-free). The PCR amplification cycle conditions were as follows: 90°C for 5 min, 90°C for 5 sec, 60°C for 30 sec, 72°C for 5 sec, 40 cycles. The PCR reaction conditions were as follows: Pre-denaturation at 94°C for 3 min. The cycle parameters were: 95°C for 60 sec, 95°C for 30 sec, 60°C for 90 sec, 40 cycles. Three replicate wells were detected for each sample miRNA. The data of the results were analyzed by 2−ΔΔCq (14).
Follow-up
The 180 patients were followed-up by telephone or visits. The follow-up was carried out trimonthly for 5 years. The deadline of the follow-up was January 2019. The overall survival period was from the first day after surgery to the date of the last follow-up or to the date of death of the patients.
Statistical analysis
SPSS 21.0 statistical software (EASYBIO) was used to analyze the data. A Chi-square test was used to compare counting data, such as sex, age and weight, between the two groups. The relative expression levels of miR-365 and miR-25 in the two groups were compared by t-test. The relationship between the relative expression levels of miR-365, miR-25 and the clinicopathological parameters of NSCLC patients was analyzed by t-test. The comparison between miR-365, miR-25 and multigroup mean values of pathological grade, TNM stage was carried out by one-way ANOVA. Then pairwise comparison was carried out by Dunnett's t-test. Kaplan-Meier curves were used to establish the survival curves of the patients with high expression and low expression of miR-365 and miR-25. A log-rank test was used to evaluate the difference of the survival curves of the patients in the two groups. When P<0.05, the difference was considered to be statistically significant.
Results
Comparison of general clinical data between the two groups
There was no significant difference between the patients in the experimental group and the subjects in the control group in terms of sex, age, height, weight, educational level, residence, exercise habits, smoking, and drinking (P>0.05; Table II).
Table II.
Comparisons between the clinical data of the patients in the experimental and control groups [n (%)].
| Characteristics | Experimental group (n=180) | Control group (n=90) | χ2 | P-value |
|---|---|---|---|---|
| Sex | 0.124 | 0.725 | ||
| Male | 108 (60.00) | 56 (62.22) | ||
| Female | 72 (40.00) | 34 (37.78) | ||
| Average age (years) | 1.069 | 0.301 | ||
| ≤50 | 98 (54.44) | 43 (47.78) | ||
| >50 | 82 (45.56) | 47 (52.22) | ||
| Height (cm) | 1.896 | 0.580 | ||
| <160 | 78 (43.33) | 50 (55.56) | ||
| ≥160 | 102 (56.67) | 40 (44.44) | ||
| Weight (kg) | 0.189 | 0.664 | ||
| <55 | 79 (43.89) | 37 (41.11) | ||
| ≥55 | 101 (56.11) | 53 (58.89) | ||
| Educational level | 1.434 | 0.231 | ||
| ≤High school | 89 (49.44) | 38 (42.22) | ||
| >High school | 91 (50.56) | 52 (57.78) | ||
| Residence | 0.119 | 0.731 | ||
| City | 90 (50.00) | 43 (47.78) | ||
| Countryside | 90 (50.00) | 47 (52.22) | ||
| Exercise habits | 2.983 | 0.084 | ||
| Yes | 76 (50.00) | 48 (53.33) | ||
| No | 104 (50.00) | 42 (46.67) | ||
| Smoking | 0.007 | 0.931 | ||
| Yes | 99 (42.22) | 49 (54.44) | ||
| No | 81 (57.78) | 41 (45.56) | ||
| Drinking | 3.023 | 0.082 | ||
| Yes | 96 (53.33) | 58 (64.44) | ||
| No | 84 (46.67) | 32 (35.56) |
Comparisons between the relative expression levels of miR-25 and miR-365 in the serum of the patients in the experimental and control groups
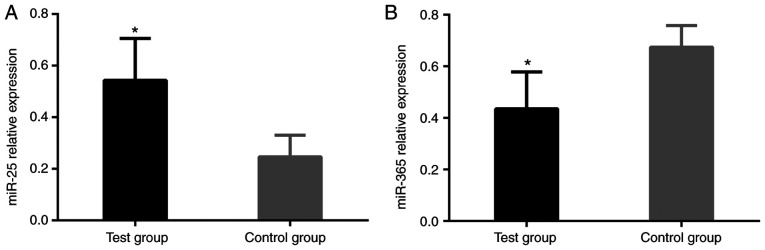
The relative expression level of miR-25 of the patients in the experimental group was significantly higher than that of the patients in the control group. The difference was statistically significant (P<0.001). The relative expression level of miR-365 of the patients in experimental group was significantly lower than that of the patients in the control group. The difference was statistically significant (P<0.001; Table III and Fig. 1).
Table III.
Comparisons between the relative expression levels of miR-25 and miR-365 in the serum of the patients in the experimental and control groups (mean ± SD).
| Group | n | Relative expression level of miR-25 | Relative expression level of miR-365 |
|---|---|---|---|
| Experimental group | 180 | 0.543±0.163 | 0.435±0.143 |
| Control group | 90 | 0.246±0.084 | 0.674±0.084 |
| t-value | 17.32 | 14.64 | |
| P-value | <0.001 | <0.001 |
Figure 1.
Expression of miR-25 and miR-365 in the experimental and control groups of NSCLC patients. (A) The expression of miR-25 of the patients in the experimental group. qRT-PCR revealed that the relative expression level of miR-25 of the patients in the experimental group was significantly higher than that of the patients in the control group. (B) The expression of miR-365 of the patients in the experimental group. qRT-PCR revealed that relative expression level of miR-365 of the patients in the experimental group was significantly lower than that of the patients in the control group. *P<0.05 vs. the control group.
Relationship between the expression of miR-25 and clinicopathological features
qRT-PCR was used to detect the expression of miR-25 in the serum of patients with various clinicopathological characteristics. There was no difference in the relative expression level of miR-25 of the patients in the experimental group although the patients had different ages, sex, history of smoking and pathological types (P>0.05). The relative expression level of miR-25 of patients with peripheral infiltration was significantly higher than that of patients without peripheral infiltration (P<0.05). There was a significant difference in the relative expression level of miR-25 in different pathological grades (P<0.05). The relative expression level of miR-25 of patients whose tumor diameter was <3.0 cm was significantly lower than that of patients whose tumor diameter was ≥3.0 cm (P<0.05). There was a significant difference in the relative expression level of miR-25 in different TNM stages (P<0.05). The relative expression level of miR-25 of patients with lymph node metastasis was significantly higher than that of patients without lymph node metastasis (P<0.05; Table IV).
Table IV.
Relationship between the relative expression level of miR-25 and clinicopathological features ( ± SD).
| Pathological parameters | n | Relative expression level of miR-25 | t/F-value | P-value |
|---|---|---|---|---|
| Sex | 1.179 | 0.240 | ||
| Male | 108 | 0.524±0.076 | ||
| Female | 72 | 0.538±0.081 | ||
| Age (years) | 0.414 | 0.679 | ||
| ≤50 | 98 | 0.546±0.082 | ||
| >50 | 82 | 0.541±0.079 | ||
| History of smoking | 0.496 | 0.621 | ||
| Yes | 99 | 0.554±0.091 | ||
| No | 81 | 0.561±0.098 | ||
| Peripheral infiltration | 8.844 | <0.001 | ||
| Yes | 88 | 0.542±0.075 | ||
| No | 92 | 0.455±0.056 | ||
| Pathological grades | 54.643 | <0.001 | ||
| High | 56 | 0.435±0.048 | ||
| Middle | 68 | 0.489±0.056a | ||
| Low | 56 | 0.547±0.065a,b | ||
| Tumor diameter | 9.069 | <0.001 | ||
| <3.0 cm | 83 | 0.485±0.076 | ||
| ≥3.0 cm | 97 | 0.597±0.088 | ||
| TNM stages | 89.623 | <0.001 | ||
| I | 43 | 0.445±0.043 | ||
| II | 48 | 0.502±0.053c | ||
| III | 39 | 0.570±0.057c,d | ||
| IV | 50 | 0.623±0.066c–e | ||
| Lymph node metastasis | 13.781 | <0.001 | ||
| Yes | 78 | 0.613±0.078 | ||
| No | 102 | 0.466±0.065 | ||
| Pathological type | 1.803 | 0.073 | ||
| Squamous carcinoma | 33 | 0.513±0.081 | ||
| Adenocarcinoma | 147 | 0.549±0.108 |
P<0.05, compared with pathological high differentiation
P<0.05, compared with pathological middle differentiation
P<0.05, compared with TNM stage I
P<0.05, compared with TNM stage II
P<0.05, compared with TNM stage III.
Relationship between the expression of miR-365 and clinicopathological features
qRT-PCR was used to detect the expression of miR-365 in the serum of patients with various clinicopathological characteristics. There was no significant difference in the relative expression level of miR-365 of the patients in the experimental group although the patients had different ages, sex, history of smoking and pathological types (P>0.05). The relative expression level of miR-365 of the patients with peripheral infiltration was significantly lower than that of patients without peripheral infiltration (P<0.05). There was a significant difference in the relative expression level of miR-365 in different pathological grades (P<0.05). The relative expression level of miR-365 of patients whose tumor diameter was <3.0 cm was significantly higher than that of patients whose tumor diameter was ≥3.0 cm (P<0.05). There was a significant difference in the relative expression level of miR-365 in different TNM stages (P<0.05). The relative expression level of miR-365 of patients with lymph node metastasis was significantly lower than that of patients without lymph node metastasis (P<0.05; Table V).
Table V.
Relationship between the relative expression level of miR-365 and clinicopathological features ( ± SD).
| Pathological parameters | n | Relative expression level of miR-365 | t/F-value | P-value |
|---|---|---|---|---|
| Sex | 0.438 | 0.662 | ||
| Male | 108 | 0.429±0.088 | ||
| Female | 72 | 0.435±0.093 | ||
| Age (years) | 0.557 | 0.579 | ||
| ≤50 | 98 | 0.438±0.076 | ||
| >50 | 82 | 0.432±0.067 | ||
| History of smoking | 0.424 | 0.672 | ||
| Yes | 99 | 0.432±0.087 | ||
| No | 81 | 0.438±0.103 | ||
| Peripheral infiltration | 8.910 | <0.001 | ||
| Yes | 88 | 0.367±0.055 | ||
| No | 92 | 0.457±0.078 | ||
| Pathological grades | 46.536 | <0.001 | ||
| High | 56 | 0.489±0.056 | ||
| Middle | 68 | 0.439±0.076a | ||
| Low | 56 | 0.377±0.045a,b | ||
| Tumor diameter | 6.363 | <0.001 | ||
| <3.0 cm | 83 | 0.484±0.087 | ||
| ≥3.0 cm | 97 | 0.398±0.094 | ||
| TNM stages | 33.257 | <0.001 | ||
| I | 43 | 0.499±0.056 | ||
| II | 48 | 0.459±0.063c | ||
| III | 39 | 0.410±0.077c,d | ||
| IV | 50 | 0.375±0.060c–e | ||
| Lymph node metastasis | 9.984 | <0.001 | ||
| Yes | 78 | 0.381±0.077 | ||
| No | 102 | 0.486±0.064 | ||
| Pathological type | 1.236 | 0.218 | ||
| Squamous carcinoma | 33 | 0.445±0.056 | ||
| Adenocarcinoma | 147 | 0.465±0.089 |
P<0.05, compared with pathological high differentiation
P<0.05, compared with pathological middle differentiation
P<0.05, compared with TNM stage I
P<0.05, compared with TNM stage II
P<0.05, compared with TNM stage III.
Survival condition of NSCLC patients
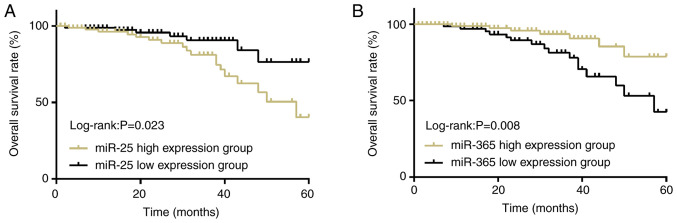
According to a critical value, which was the median of the relative expression levels of miR-25 and miR-365 in the serum of the patients in the experimental group, there were 88 patients (≥0.543) in the miR-25 high-expression group and 92 patients in miR-25 low-expression group (<0.543). There were 102 patients (≥0.435) in the miR-365 high expression group and 78 patients (<0.435) in the miR-365 low expression group. The 5-year overall survival rate of the patients in the miR-25 high-expression group [43.18% (38/88)] was significantly lower than that of the patients in the miR-25 low-expression group [75.00% (69/92); P<0.05]. The 5-year overall survival rate of the patients in the miR-365 high-expression group [76.47% (78/102)] was significantly higher than that of the patients in miR-365 low-expression group [43.59% (34/78); P<0.05; Fig. 2].
Figure 2.
The survival condition of the patients in the experimental group. (A) The 5-year overall survival rate of the patients in the miR-25 high-expression group was significantly lower than that of the patients in the miR-25 low-expression group (P<0.05). (B) The 5-year overall survival rate of the patients in the miR-365 high-expression group was significantly higher than that of the patients in the miR-365 low-expression group (P<0.05).
Discussion
In recent years, the morbidity and mortality of lung cancer have been increasing. Currently, deaths caused by lung cancer account for ~18% of cancer-related deaths worldwide, with non-small cell lung cancer accounting for the vast majority (15). Some risk factors that lead to NSCLC are smoking, air pollution, ionizing radiation, and genetic factors (16). The probability that NSCLC patients have distant metastasis and local lymph node metastasis is high. Moreover, NSCLC patients generally have no evident clinical symptoms, as a result, the early diagnosis of NSCLC patients is difficult. Numerous patients are in advanced stage (III-B-IV) and have distant metastasis and local lymph node metastasis when they are diagnosed, and are therefore not eligible to receive radical surgery (17,18). Thus, finding effective NSCLC prognostic markers is of great significance for treating patients.
Numerous studies have revealed that the aberrant expression of a great number of miRNAs is closely correlated with cancer progression. miRNAs have been revealed to be involved in the occurrence and progression of cancer by regulating the expression of its target genes and cooperating with target genes (19–22). Since miR-25 and miR-365 were identified, the medical community has been carrying out some in-depth studies on them. More and more scholars have revealed that miR-25 and miR-365 are aberrantly expressed in various cancers (23–25), and they could be used as new tumor markers. In a study by Chen et al (26), it was revealed that the expression level of miR-25 in lung cancer tissues was significantly higher than that in normal tissues. Their study confirmed that miR-25 may be an oncogene, which can control apoptosis of lung cancer cells by targetedly regulating the expression of tumor suppressor gene RGS3. In the present study, the expression of miR-25 was also increased in the serum of NSCLC patients. Possibly the progression of tumors can be suppressed by inhibiting the expression of miR-25. A study by Xiang et al (27) revealed that the relative expression level of miR-25 in lung cancer tissues was significantly higher than that in normal lung tissues, and that miR-25 could control apoptosis, metastasis and invasion of lung cancer cells by targetedly regulating FBXW7. Their study confirmed that miR-25 was involved in the occurrence and progression of lung cancer. The survival curves of NSCLC patients were analyzed in the present study. The results revealed that the higher the relative expression level of miR-25 was, the lower the 5-year survival rate of patients was, and that the expression of miR-25 was related to lymph node metastasis and peripheral infiltration. This result indicated that miR-25 may also be involved in the occurrence and progression of NSCLC. The expression of miR-365 has been revealed to be downregulated in some malignant tumors such as malignant melanoma (28), epidermal squamous cell carcinoma (29), and colon cancer (30). The expression of miR-365 was also downregulated in the serum of NSCLC patients in the present study. The result of this study was consistent with the results of other studies, and it was confirmed that miR-365 plays the same role in NSCLC. A study by Nie et al (31) revealed that low expression of miR-365 was exhibited in cancer tissues and serum of patients with pancreatic cancer, and that miR-365 was closely related to distant metastasis and clinical stage of patients with pancreatic cancer. The study also revealed that miR-365 could be used as an independent prognostic factor of the overall survival rate of patients with pancreatic cancer, and that overexpression of miR-365 could inhibit proliferation and invasion of pancreatic cancer cells. Therefore, when the expression of miR-365 was downregulated, tumor growth was observed. In the present study, the survival curves of NSCLC patients revealed that the lower the relative expression level of miR-365 was, the lower the 5-year survival rate of the patients was, which confirmed that the downregulation of miR-365 indicated worse prognosis. Therefore, the relative expression level of miR-365 of NSCLC patients with peripheral infiltration was significantly lower than that of NSCLC patients without peripheral infiltration, which indicated that miR-365 was involved in the occurrence and progression of NSCLC, and that downregulation of miR-365 indicated proliferation of cancers. miR-365 could be used as a potential molecular marker of NSCLC.
With the disclosure of the medical uses of miRNAs, it is believed that miRNAs can provide a breakthrough and improve the treatment of cancers in the near future. In the present study, miR-365 and miR-25 were revealed to be aberrantly expressed in the serum of NSCLC patients and associated with the prognosis of patients, indicating that they have the potential to be therapeutic targets for NSCLC. Previous studies have revealed that miR-25 and miR-365 are involved in tumor development by regulating their downstream target genes, for example, miR-365 could target Bcl-2 to induce the apoptosis of HCC cells (32). miR-365 could also target the volatilizing and anticancer effects of CYR61 in osteosarcoma (33). miR-25 promoted the development of liver cancer by inhibiting RhoGDI1 (34). These studies revealed that miR-365 and miR-25 play a significant role in the development of tumors. However, this study, as a clinical study, did not conduct cell experiments to explore the specific role and mechanism of miR-365 and miR-25 in NSCLC. In addition, due to the insufficient number of qualified specimens of cancer tissues and adjacent tissues obtained, their expression in tissues was not detected. It is hoped that further cell research and the increase of the number of tissue specimens can be achieved in the future.
In summary, the expression of miR-25 and miR-365 was different in the serum of NSCLC patients, and these miRNAs could be used as important tumor markers to evaluate the prognosis of NSCLC patients.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
DH conceived and designed the study and wrote the manuscript. WO analyzed the general data of the patients and compared the relative expression of miR-25 and miR-365 in the serum of patients in the experimental and control groups. HT and MP performed PCR analysis and analyzed the survival of patients. YO and ZS analyzed the relationship between miR-25, miR-365 and clinicopathological features and were responsible for the follow-up of patients. All authors read and approved the final manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of the Affiliated Hospital of Guangdong Medical University which the authors were previously affiliated to. Patients who participated in this research, signed the informed consent and had complete clinical data.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Torre LA, Siegel RL, Ward EM, Jemal A. International variation in lung cancer mortality rates and trends among women. Cancer Epidemiol Biomarkers Prev. 2014;23:1025–1036. doi: 10.1158/1055-9965.EPI-13-1220. [DOI] [PubMed] [Google Scholar]
- 2.Midha A, Dearden S, McCormack R. EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: A systematic review and global map by ethnicity (mutMapII) Am J Cancer Res. 2015;5:2892–2911. [PMC free article] [PubMed] [Google Scholar]
- 3.Zhou C, Liu D, Li J, Sun H, Zheng X, Wang S, Hong G, Mallampati S, Sun H, Zhou X, et al. Chemotherapy plus dendritic cells co-cultured with cytokine-induced killer cells versus chemotherapy alone to treat advanced non-small-cell lung cancer: A meta-analysis. Oncotarget. 2016;7:86500–86510. doi: 10.18632/oncotarget.13394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Novello S, Barlesi F, Califano R, Cufer T, Ekman S, Levra MG, Kerr K, Popat S, Reck M, Senan S, et al. Metastatic non-small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27(Suppl 5):v1–v27. doi: 10.1093/annonc/mdw326. [DOI] [PubMed] [Google Scholar]
- 5.Anastasiadou E, Jacob LS, Slack FJ. Non-coding RNA networks in cancer. Nat Rev Cancer. 2018;18:5–18. doi: 10.1038/nrc.2017.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheng CJ, Bahal R, Babar IA, Pincus Z, Barrera F, Liu C, Svoronos A, Braddock DT, Glazer PM, Engelman DM, et al. MicroRNA silencing for cancer therapy targeted to the tumour microenvironment. Nature. 2015;518:107–110. doi: 10.1038/nature13905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Christopher AF, Kaur RP, Kaur G, Kaur A, Gupta V, Bansal P. MicroRNA therapeutics: Discovering novel targets and developing specific therapy. Perspect Clin Res. 2016;7:68–74. doi: 10.4103/2229-3485.179431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mo MH, Chen L, Fu Y, Wang W, Fu SW. Cell-free circulating miRNA biomarkers in cancer. J Cancer. 2012;3:432–448. doi: 10.7150/jca.4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kondo N, Toyama T, Sugiura H, Fujii Y, Yamashita H. miR-206 Expression is down-regulated in estrogen receptor alpha-positive human breast cancer. Cancer Res. 2008;68:5004–5008. doi: 10.1158/0008-5472.CAN-08-0180. [DOI] [PubMed] [Google Scholar]
- 10.Zhang L, Liu X, Jin H, Guo X, Xia L, Chen Z, Bai M, Liu J, Shang X, Wu K, et al. miR-206 inhibits gastric cancer proliferation in part by repressing cyclinD2. Cancer Lett. 2013;332:94–101. doi: 10.1016/j.canlet.2013.01.023. [DOI] [PubMed] [Google Scholar]
- 11.Han JG, Jiang YD, Zhang CH, Yang YM, Pang D, Song YN, Zhang GQ. A novel panel of serum miR-21/miR-155/miR-365 as a potential diagnostic biomarker for breast cancer. Ann Surg Treat Res. 2017;92:55–66. doi: 10.4174/astr.2017.92.2.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang J, Zhang Z, Wang Q, Xing XJ, Zhao Y. Overexpression of microRNA-365 inhibits breast cancer cell growth and chemo-resistance through GALNT4. Eur Rev Med Pharmacol Sci. 2016;20:4710–4718. [PubMed] [Google Scholar]
- 13.Goldstraw P, Crowley J, Chansky K, Giroux DJ, Groome PA, Rami-Porta R, Postmus PE, Rusch V, Sobin L, International Association for the Study of Lung Cancer International Staging Committee; Participating Institutions The IASLC lung cancer staging project: Proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol. 2007;2:706–714. doi: 10.1097/JTO.0b013e31812f3c1a. [DOI] [PubMed] [Google Scholar]
- 14.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 15.Cheng TY, Cramb SM, Baade PD, Youlden DR, Nwogu C, Reid ME. The international epidemiology of lung cancer: Latest trends, disparities, and tumor characteristics. J Thorac Oncol. 2016;11:1653–1671. doi: 10.1016/j.jtho.2016.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Matsuura N, Koh G, Konishi C, Minamino S, Takahara Y, Harada H, Kodama K, Emoto M. Fulminant onset of insulin-dependent diabetes with positive anti-GAD antibody titers during treatment with nivolumab in a patient with NSCLC. Cancer Immunol Immunother. 2018;67:1417–1424. doi: 10.1007/s00262-018-2203-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang Y, Sun Y, Chen H. Effect of tumor size on prognosis of node-negative lung cancer with sufficient lymph node examination and no disease extension. Onco Targets Ther. 2016;9:649–653. doi: 10.2147/OTT.S98509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Helissey C, Champiat S, Soria JC. Immune checkpoint inhibitors in advanced nonsmall cell lung cancer. Curr Opin Oncol. 2015;27:108–117. doi: 10.1097/CCO.0000000000000167. [DOI] [PubMed] [Google Scholar]
- 19.Rupaimoole R, Slack FJ. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov. 2017;16:203–222. doi: 10.1038/nrd.2016.246. [DOI] [PubMed] [Google Scholar]
- 20.Yu DD, Wu Y, Shen HY, Lv MM, Chen WX, Zhang XH, Zhong SL, Tang JH, Zhao JH. Exosomes in development, metastasis and drug resistance of breast cancer. Cancer Sci. 2015;106:959–964. doi: 10.1111/cas.12715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lynch SM, McKenna MM, Walsh CP, McKenna DJ. miR-24 regulates CDKN1B/p27 expression in prostate cancer. Prostate. 2016;76:637–648. doi: 10.1002/pros.23156. [DOI] [PubMed] [Google Scholar]
- 22.Ma Z, Ma Y, Xia Q, Li Y, Li R, Chang W, Chen J, Leng Z, Tao K. MicroRNA-155 expression inversely correlates with pathologic stage of gastric cancer and it inhibits gastric cancer cell growth by targeting cyclin D1. J Cancer Res Clin Oncol. 2016;142:1201–1212. doi: 10.1007/s00432-016-2139-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zoni E, van der Horst G, van de Merbel AF, Chen L, Rane JK, Pelger RC, Collins AT, Visakorpi T, Snaar-Jagalska BE, Maitland NJ, van der Pluijm G. miR-25 modulates invasiveness and dissemination of human prostate cancer cells via regulation of αv- and α6-integrin expression. Cancer Res. 2015;75:2326–2336. doi: 10.1158/0008-5472.CAN-14-2155. [DOI] [PubMed] [Google Scholar]
- 24.Li M, Liu L, Zang W, Wang Y, Du Y, Chen X, Li P, Li J, Zhao G. miR365 overexpression promotes cell proliferation and invasion by targeting ADAMTS-1 in breast cancer. Int J Oncol. 2015;47:296–302. doi: 10.3892/ijo.2015.3015. [DOI] [PubMed] [Google Scholar]
- 25.Fassan M, Baffa R, Palazzo JP, Lloyd J, Crosariol M, Liu CG, Volinia S, Alder H, Rugge M, Croce CM, Rosenberg A. MicroRNA expression profiling of male breast cancer. Breast Cancer Res. 2009;11:R58. doi: 10.1186/bcr2348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen Z, Wu Y, Meng Q, Xia Z. Elevated microRNA-25 inhibits cell apoptosis in lung cancer by targeting RGS3. In Vitro Cell Dev Biol Anim. 2016;52:62–67. doi: 10.1007/s11626-015-9947-2. [DOI] [PubMed] [Google Scholar]
- 27.Xiang J, Hang JB, Che JM, Li HC. MiR-25 is up-regulated in non-small cell lung cancer and promotes cell proliferation and motility by targeting FBXW7. Int J Clin Exp Pathol. 2015;8:9147–9153. [PMC free article] [PubMed] [Google Scholar]
- 28.Bai J, Zhang Z, Li X, Liu H. MicroRNA-365 inhibits growth, invasion and metastasis of malignant melanoma by targeting NRP1 expression. Cancer Biomark. 2015;15:599–608. doi: 10.3233/CBM-150500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yu X, Li Z. The role of miRNAs in cutaneous squamous cell carcinoma. J Cell Mol Med. 2016;20:3–9. doi: 10.1111/jcmm.12649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhu Y, Zhao H, Rao M, Xu S. MicroRNA-365 inhibits proliferation, migration and invasion of glioma by targeting PIK3R3. Oncol Rep. 2017;37:2185–2192. doi: 10.3892/or.2017.5458. [DOI] [PubMed] [Google Scholar]
- 31.Nie J, Liu L, Zheng W, Chen L, Wu X, Xu Y, Du X, Han W. microRNA-365, down-regulated in colon cancer, inhibits cell cycle progression and promotes apoptosis of colon cancer cells by probably targeting Cyclin D1 and Bcl-2. Carcinogenesis. 2012;33:220–225. doi: 10.1093/carcin/bgr245. [DOI] [PubMed] [Google Scholar]
- 32.Li M, Yang Y, Kuang Y, Gan X, Zeng W, Liu Y, Guan H. miR-365 induces hepatocellular carcinoma cell apoptosis through targeting Bcl-2. Exp Ther Med. 2017;13:2279–2285. doi: 10.3892/etm.2017.4244. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 33.Xu Y, Chu H, Zhou Y, Wang J, Dong C, Yin R. miR-365 functions as a tumor suppressor by directly targeting CYR61 in osteosarcoma. Biomed Pharmacother. 2018;98:531–537. doi: 10.1016/j.biopha.2017.12.086. [DOI] [PubMed] [Google Scholar]
- 34.Wang C, Wang X, Su Z, Fei H, Liu X, Pan Q. miR-25 promotes hepatocellular carcinoma cell growth, migration and invasion by inhibiting RhoGDI1. Oncotarget. 2015;6:36231–36244. doi: 10.18632/oncotarget.4740. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.