Abstract
To date, the spread of SARS-CoV-2 infection is increasing worldwide and represents a primary healthcare emergency. Although the infection can be asymptomatic, several cases develop severe pneumonia and acute respiratory distress syndrome (ARDS) characterized by high levels of pro-inflammatory cytokines, primarily interleukin (IL)-6. Based on available data, the severity of ARDS and serum levels of IL-6 are key determinants for the prognosis.
In this scenario, available in vitro and in vivo data suggested that myo-inositol is able to increase the synthesis and function of the surfactant phosphatidylinositol, acting on the phosphoinositide 3-kinase (PI3K)-regulated signaling, with amelioration of both immune system and oxygenation at the bronchoalveolar level. In addition, myo-inositol has been found able to decrease the levels of IL-6 in several experimental settings, due to an effect on the inositol-requiring enzyme 1 (IRE1)-X-box-binding protein 1 (XBP1) and on the signal transducer and activator of transcription 3 (STAT3) pathways. In this scenario, treatment with myo-inositol may be able to reduce IL-6 dependent inflammatory response and improve oxygenation in patients with severe ARDS by SARS-CoV-2. In addition, the action of myo-inositol on IRE1 endonuclease activity may also inhibit the replication of SARS-CoV-2, as was reported for the respiratory syncytial virus. Since the available data are extremely limited, if this potential therapeutic approach will be considered valid in the clinical practice, the necessary future investigations should aim to identify the best dose, administration route (oral, intravenous and/or aerosol nebulization), and cluster(s) of patients which may get beneficial effects from this treatment.
Keywords: SARS-CoV-2, Novel coronavirus, Pneumonia, Acute respiratory distress syndrome, IL-6, Inflammation, Myo-inositol, Inositol
Background
Pulmonary surfactant (PS) is a surface-active lipoprotein complex (phospho-lipoprotein) produced and secreted by type II alveolar cells, which plays a key role to allow proper oxygenation at the alveolar hydrophobic-hydrophilic interface [1]. On the one hand, PS reduces alveolar surface tension during respiration and, on the other hand, plays a key role as a defense mechanism against inhaled pathogens [2]. From the biochemical point of view, PS consists of lipids and proteins.
Lipid component of surfactant is mainly represented by phospholipids, which are primarily composed by the dipalmitoyl-phosphatidylcholine, with unsaturated phosphatidylcholine, phosphatidylglycerol (PG), and phosphatidylinositol representing lesser components. Neutral lipids and cholesterol are also present. Overall, dipalmitoyl-phosphatidylcholine, unsaturated phosphatidylcholine, and PG are the most abundant surface-active phospholipids in mature lungs, representing almost the 96% of the lipid component. Phosphatidylcholine molecules, characterized by saturated fatty acid chains, form almost 85% of the lipids in the surfactant and are organized in a monolayer in the liquid film lining the alveolus, which lowers the surface tension of that film [3]. PG represents about 11% of the lipids in the surfactant and has the function to fluidize the lipid monolayer at the interface due to the unsaturated fatty acid chains. The lipid component of surfactant is mainly assembled and packaged for secretion into secretory organelles called lamellar bodies into the type II alveolar cells, which obtain the components for these lipids by diffusion from the blood stream.
The protein component of surfactant includes the hydrophobic surfactant proteins (SPs)-B and -C, and the hydrophilic SPs-A and -D [4]. In the alveoli, these proteins are organized with the lipid components in mono- and multilayered aggregate membranes as micelles [5]. SP-A helps to regulate surfactant secretion and uptake; SP-B and SP-C facilitate adsorption and spreading of phospholipids on the liquid film lining of the alveoli. SP-D may play a role in surfactant reuptake and recycling. Additionally, SPs also regulate inflammatory responses and interact with the adaptive immune response. As a consequence, surfactant degradation or inactivation may contribute to enhanced susceptibility to lung inflammation and infection [6].
The proteins and lipids that form the surfactant have both hydrophilic and hydrophobic regions, which have a specific organization in the surfactant film to mediate the air–water interface at the alveolar surface. The hydrophilic head groups of phospholipids and the hydrophilic regions of proteins interact with water, and the hydrophobic tails and regions are facing towards the air. With this organization, the surfactant film reduces the surface tension, avoiding alveolar and lung collapse at the end of expiration, then preventing atelectasis, but also increases pulmonary compliance by recruiting collapsed airways.
Indeed, surface tension acts as “collapsing” force, which increase with the reduction of the radius of the alveoli. According to Laplace’s law, the gas pressure (P) needed to keep equilibrium between the collapsing force of surface tension (γ) and the expanding force of gas in an alveolus of radius r is expressed as follows, keeping constant the temperature and the volume of the whole system: P = 2γ/r. The normal surface tension for water at the air–liquid interface is 70 mN/m; conversely, in the lungs, it is lower due to the surfactant (25 mN/m). Nevertheless, we should consider that the surface tension decreases as temperature increases, because cohesive forces decrease with an increase of molecular thermal activity, and so it may be influenced by pathological conditions such as fever.
Moreover SPs, making variable weak bonds with lipid components by holding them longer when the interface is compressed, allow to modulate the surface tension based on the alveolar diameter. As a consequence, the surface tension varies according to the volume of air in the lungs, with the surface tension usually lower than at equilibrium during ventilation. At the end of the expiration, compressed surfactant phospholipid molecules decrease the surface tension to very low, near-zero levels. This protects lungs from atelectasis at low volumes but, at the same time, prevents time tissue damage at high volumes [7], [8]. PS greatly reducing surface tension, reduces the pressure difference needed to allow the lung to inflate, increases compliance, and allows the lungs to inflate much more easily, thereby reducing the work of breathing. In addition to maintain airway patency, the airway surfactant is expected to facilitate particle transport, decrease shear forces, and improve host resistance [9].
In this context, myo-inositol an myo-inositol-1-phosphate interact with SP-D. Specifically, the inositol ring binds the lectin calcium in the usual C-lectin mode, with two vicinal equatorial ring oxygens coordinating the calcium ion [10] and, in this way, contribute to the stability of surfactant pool sizes. Moreover, it orchestrates the interconversion between ultrastructural aggregate forms of the surfactant [11]. In addition, the correct molecular conformation of surfactant, due to the abovementioned complexes, allows robust aggregation and opsonization when binding microbial surfaces, stimulating a direct induction of microbial permeability and growth arrest [12].
As previously summarized [13], [14], [15], [16], pathways initiated by either G protein-linked receptors or receptors linked directly or indirectly to tyrosine kinases phospholipase C (PLC) [17], determine the hydrolyzes of phosphatidylinositol (4,5)-bisphosphate to form 1,4,5-triphosphate (InsP3) and 1,2-diacylglycerol. In addition, PLC can be activated by lipid products of phosphoinositide 3-kinases (PI3Ks) [18]. Robust data suggested that SP-A induces Ca2+ mobilization by activating these pathways involving PLC [19]. In this view, the activation of PI3K(s) has paramount importance as an upstream signaling molecule involved in SP-A up-regulation of PS and contributes to surfactant homeostasis at the alveolar level.
The hypothesis
Treatment with myo-inositol may be able to reduce IL-6 dependent inflammatory response and improve oxygenation in patients with severe ARDS by SARS-CoV-2. In addition, the action of myo-inositol on inositol-requiring enzyme 1 (IRE-1) endonuclease activity may also inhibit the replication of SARS-CoV-2.
Evaluation of the hypothesis
Evidence for inositol treatment in neonatal acute respiratory distress syndrome
Abnormalities in PS composition and/or reduced surfactant synthesis have been described namely in respiratory distress syndrome [20] (RDS, formerly known as hyaline membrane disease) in the infants, as well as in many similar illnesses [21]. Indeed, the absence (as well as the inadequacy) of PS in the liquid film lining of alveoli causes an increase in surface tension and alveolar collapse [22]. If not treated, such atelectasis causes an increased work of breathing, intrapulmonary shunting, ventilation-perfusion mismatch, hypoxia, and eventual respiratory failure. Surfactant deficiency is mainly due to immaturity in fetal lung and can be predicted by analysis of PS in amniotic fluid, even if current available tests suffer for a lack of sensitivity (sensitivity ranging from 30 to 70%, whereas specificity is significantly high, ranging from 95 to 99%) [23], [24]. Ontogeny of PS is tightly coupled to the length of the pregnancy, reaching a critical phase in the latest months, for the PS maturation occurs very close to term [25]. The risk of RDS decreases with increasing gestational age: 60% of babies born at fewer than 28 weeks’ gestation, 30% of babies born between 28 and 34 weeks’ gestation, and fewer than 5% of babies born after 34 weeks’ gestation develop RDS [26]. Acute respiratory distress syndrome (ARDS) has also been observed in adults, associated with different diseases [27]. ARDS usually requires mechanical ventilation and is characterized by an inflammatory condition of the airway epithelium, which involves apoptosis, recruitments of immune cells from the circulation, and protein extravasation of blood proteins into the airways [28]. In this scenario, surfactant replacement therapy has been found beneficial in the early phase of neonatal ARDS (nARDS), due to prematurity, congenital pneumonia, sepsis, meconium aspiration syndrome, bile aspiration, or pulmonary hemorrhage [29].
First attempts in RDS treatment have been made with glucocorticoids and other anti-inflammatory drugs [30]. These preliminary trials demonstrated that such compounds may, to some extent, protect premature infants from RDS, but only if they are given before birth [31], [32]. With the advent of therapies for RDS, including antenatal steroids and surfactant replacement therapy, mortality from RDS has decreased from nearly 100% to less than 10% in recent years [33]. Yet, many foetuses do not respond to glucocorticoids, particularly foetuses from multiple pregnancies [34]. Additionally, glucocorticoid-based treatment is still controversial given that glucocorticoid inhibits cell growth and tissue differentiation, thus eventually impairing alveolar maturation [35]. Natural and synthetic surfactant preparations exist, and both are effective in the treatment and prevention of RDS. Natural surfactants are derived from animal lungs (bovine or porcine) and contain phospholipids with SP-B and SP-C; first-generation synthetic surfactants contain only phospholipids without proteins [36]. A Cochrane meta-analysis comparing natural surfactant to first-generation synthetic surfactant confirmed that greater early improvement in the requirement for ventilator support, fewer pneumothoraxes, and fewer deaths associated with animal derived surfactant extract treatment. There is also a marginal decrease in the risk of bronchopulmonary dysplasia when using natural surfactant [37]. Although natural surfactants appear to be associated with higher rates of intraventricular haemorrhage, grade 3 and 4 intraventricular hemorrhage rates are not increased. The conclusion of this meta-analysis is that natural surfactants are the more desirable choice over the first-generation synthetic surfactants, which is likely due to the inclusion of the SPs in the natural surfactant. Exogenous natural surfactant given at birth of shortly after birth, expands the lung and ameliorates RDS, with a decrease in both neonatal mortality (by 45–55%) and pulmonary air lack syndromes (pneumothorax and interstitial emphysema). Composition of exogenous surfactant is of critical importance. Indeed, presence of SP-B and SP-C proteins is mandatory for surfactant activity, and their presence allows to attain better clinical results (and lower neonatal mortality), than that obtained with synthetic, protein-free surfactant [38]. Nevertheless, surfactant replacement therapy is severely hampered in the case of inflammation of the airway epithelium, which causes accelerated surfactant metabolism [28]. Failure of surfactant replacement therapy is associated with prolonged mechanical ventilation and this leads to secondary damage to the neonatal lung by induction fibrosis and collapse of alveolar-capillary in the distal areas of the lungs [39].
In human infants with RDS, a premature drop in serum inositol levels predicts a more severe course [40]. Inositol supplementation increases the saturated phosphatidylcholine/sphingomyelin ratio in the surfactant of newborns and produces a rise in serum inositol concentration. In humans, free inositol levels in sera from preterm neonates are 2–20 times higher than the levels in maternal or adult sera [41], [42]. Consistently, human milk has a high concentration of inositol, with preterm milk being the richest source, and studies in newborns suggest an endogenous synthesis of inositol during fetal life. Infants who are breast fed have higher serum inositol levels compared to those that are not at 1–2 weeks of life [43].
Robust evidence from animal models already suggested that inositol addition in in vitro condition increases the synthesized amount of surfactant phosphatidylinositol by fetal type II cells [44]. Addition of inositol supplement [45] to the feeds of glucocorticoid-treated pregnant rabbits is able to increase surfactant levels in alveolar lavage from fetuses [46]. In this model, myo-inositol decreased betamethasone-induced inhibition of lung growth and potentiated the hormone-induced increase in alveolar space saturated phosphatidylcholine; furthermore, when lung explants from 26-day-old fetuses were grown in the presence of dexamethasone and thyroxine, the addition of myo-inositol switched the acidic surfactant phospholipid from phosphatidylglycerol to phosphatidylinositol and further increased the incorporation of surfactant-associated saturated phosphatidylcholine; finally, myo-inositol increased the incorporation of dihydro-nicotinamide-adenine dinucleotide phosphate and acetate into the fatty acid moiety of surfactant phosphatidylcholine. Consistently, myo-inositol-dependent alterations in lipid synthesis may be induced in isolated type II pneumocytes by manipulation of the myo-inositol concentration in the culture medium or, as found in the Sprague-Dawley rats, by a diet supplemented with this compound [47]. In particular, accumulating evidence suggests that dietary inositol modifies the physiologic and biochemical response of the immature fetal lung to pharmacologic doses of exogenous glucocorticoids, and that combined inositol and betamethasone treatment produces a clear-cut improvement in compliance and decreases interstitial-to-air space ratios, allowing homogenous expansion of alveoli [48].
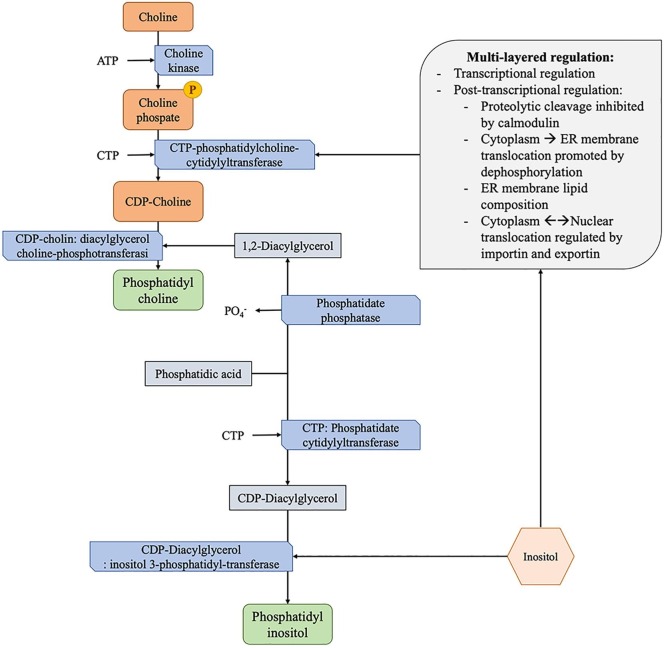
In this scenario, extracellular high content of myo-inositol in immature fetuses provides an environment that promotes both the hormone-stimulated differentiation of the lung and the production and secretion of surfactant, with consequently improved oxygenation [49]. This may be due, at least in part, to two different reasons. On the one hand, synthesis of phosphatidylinositol is dose-dependent and rate-limiting by inositol levels, and the high level of this compound allows the activation of CTP-phosphatidylcholine-cytidylyltransferase, which catalyzes the production of surfactant phosphatidylcholine (Fig. 1 ) [50], [51]. On the other hand, inositol activates phosphoinositide cycle and protein kinase C, promoting the cellular growth and differentiation in the alveolar microenvironment in a dose-dependent fashion [52].
Fig. 1.
Biosynthesis of phosphatidylcholine and phosphatidylinositol. Supplementation of inositol has been related to an improved production of phosphatidylinositol, being it the precursor. At the same time, inositol supplementation has been related to an increased production of phosphatidylcholine by modulation of CTP-phosphatidylcholine-cytidylyltransferase regulation with increased activity. CTP-phosphatidylcholine-cytidylyltransferase enzyme is the rate-limiting and regulated step in the CDP-choline pathway for the synthesis of phosphatidylcholine, and it is primarily regulated by multiple post-transcriptional mechanisms that improve the function by stabilizing the enzyme and favoring the translocation at the endoplasmic reticulum (ER) membrane.
In infants with nARDS, the food supplementation with 40 mg/kg of inositol every 6 h starting 48 h after birth correlated with a significant increase in phosphatidylcholine and phosphatidylinositol and a concomitant significant decrease in sphingomyelin and phosphatidylserine in the tracheal aspirate [53], confirming that surfactant synthesis and secretion are affected by inositol levels as in the animal models. Another study demonstrated that, compared to placebo-treated population, the intravenous administration of 80 mg/kilogram/day of inositol to premature infants with nARDS, starting 4 to 12 h after birth and subsequently every 12 h for 5 days, was associated with reduced severity and mortality due to respiratory failure, increased survival rate without bronchopulmonary dysplasia, and reduced rate of retinopathy [54].
The safety of intravenous administration of inositol was confirmed also in a population of extremely preterm infants. In a recent multicenter study, Phelps et al. [55] included 76 infants who were born between 230/7 and 296/7 gestational weeks, who were ≥ 600 g birth weight, who had no major congenital anomalies, and who had received no human milk or formula feedings since birth. The infants were allocated between 12 h and 6 days of age to receive a single low (60 mg/kg) or high (120 mg/kg) dose of 5% myo-inositol intravenously over 20 min in a 1:1:1 randomization with placebo delivered in one of two volumes to maintain masking (5% glucose). According to the data analysis, heart rate, blood pressure, and respiration did not differ between placebo and inositol infants at either dose, nor significant differences in adverse events occurred between the 3 groups. From the pharmacokinetic point of view, the central volume of distribution was 0.5115 L/kg, the clearance was 0.0679 L/kg/h, the endogenous production was 2.67 mg/kg/h, and the half-life was 5.22 h when modeled without the covariates. In addition, during the first 12 h renal inositol excretion quadrupled in the 120 mg/kg group, returning to near baseline after 48 h without any significant diuretic side-effect.
More recently, the same authors performed another multicenter trial [56], enrolling 125 infants with similar characteristics of the previous study [55]. The infants were randomized to receive 5% solution of myo-inositol intravenously at neutral pH, at the doses of 10, 40, or 80 mg/kg/day, divided q12h given over 20 min, or placebo (5% glucose for intravenous infusion and dispensed at the equivalent various volumes to maintain masking). This further large trial found that inositol, even at doses up to 80 mg/kg/day for 7–10 weeks, is well tolerated and does not increase adverse events. At 80 mg/kg/day, the pharmacokinetic analysis showed that mean serum levels reached 140 mg/L, declined after 2 weeks, converging in all groups by 6 weeks, with a mean volume of distribution 0.657 L/kg, clearance 0.058 L/kg/h, and half-life 7.90 h. Interestingly, in the inositol groups there were fewer adverse events and co-morbidities compared with the placebo group.
Potential role of inositol to counteract viral infection of the low pulmonary tract
The endoplasmic reticulum (ER) stress response is known to play a role in diverse biological processes including apoptosis, inflammation, and metabolism [57]. In addition, cellular apoptosis has been shown to be mediated by the ER stress response versus several viruses, including the severe acute respiratory syndrome coronavirus (SARS-CoV) [58]. In detail, the ER stress response depends on three main cascade pathways activated by three proteins, whose upstream signals are mediated by ER-resident transmembrane proteins: activating transcription factor 6 (ATF6), protein kinase R-like ER kinase (PERK), and IRE-1 [59]. According to recent pieces of evidence, IRE1 endonuclease activity has been demonstrated involved in the inhibition of the replication of respiratory syncytial virus [60]. This mechanism may be due, at least in part, to the destabilization of phospholipid membrane homeostasis. Indeed, ER-resident transmembrane proteins such as the protein translocon subunit Sbh1, which form part of the unfolded protein response program, are prematurely degraded by membrane stiffening at the ER [61]. Nevertheless, another study shows that influenza A viral infection activates IRE-1, and inhibiting that pathway inhibits influenza virus replication [62]. In this scenario, IRE-1 appears to have diametrically opposed effects for RSV vs influenza, and so it would be difficult to predict how it might interact with SARS-CoV-2.
Inositol’s action on the lung cancer microenvironment and IL-6 levels
Robust epidemiological data correlated cigarette smoking to both high-grade preinvasive bronchial lesions (i.e., moderate dysplasia, severe dysplasia, and carcinoma in situ) and invasive lung cancer [63]. In particular, smoking increases lung cancer risk 5- to 10-fold with a clear dose–response relationship; in addition, exposure to environmental tobacco smoke among non-smokers increases lung cancer risk by about 20% [64]. Interestingly, a diet supplemented with inositol, combined with aerosolized budesonide, has been found to inhibit the process of lung tumorigenesis in a dose-dependent fashion in a mouse model exposed to the carcinogen benzo(a)-pyrene or 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone [65]. In details, according to a robust investigation in this animal model, budesonide at 10 µg/kg body weight administered by aerosol for 20 s three times a week, plus 0.3% myo-inositol added to the diet, reduced the pulmonary tumor formation by 60%; similarly, budesonide at 25 µg/kg body weight plus 0.3% myo-inositol added to the diet reduced it by 79% [66]. In another study, diet containing both myo-inositol and dexamethasone resulted in an additive effect on the inhibition by 40% of pulmonary adenoma induced by benzo[a]pyrene in female mice [67], suggesting a potential therapeutic role of the combined treatment even after the pre-cancerous lesion formation and initiation.
The efficacy and safety of oral myo-inositol for the chemoprevention of lung cancer was also tested and confirmed in a cohort of smokers with persistent bronchial dysplasia, despite treatment with inhaled budesonide [68]. In this study, after a treatment with 18 gr/die of oral of myo-inositol mixed with juice or water divided into two doses daily, the regression of pre-existing dysplastic lesions at repeat autofluorescence bronchoscopy and biopsy was significantly increased. More recently, the same group investigated the effects of oral myo-inositol, 9 gr once/day for two weeks and then twice/day for 6 months, on bronchial dysplasia rate, Ki-67 labeling index, blood and bronchoalveolar lavage fluid levels of pro-inflammatory, oxidant/anti-oxidant biomarkers, and airway epithelial gene-expression signature for PI3K activity [69]. Confirming the previous findings [68], the complete response rate was higher in the group treated with oral myo-inositol compared to placebo; in addition, the population who showed complete response to this treatment, gene-expression signature reflective of PI3K activation within the cytologically-normal bronchial airway epithelium was significantly decreased; finally, and most importantly, oral treatment with myo-inositol has been found to significantly reduce interleukin (IL)-6 levels in bronchoalveolar lavage fluid.
One year later, the preliminary findings in a selected population with bronchial dysplasia were further investigated at a molecular level in the mouse model. In this study [70], mice with oncogenic Kras expressed in the airway epithelium (CcspCre/+; KrasLSL-G12D/+), which are known form lung premalignant lesions in a stereotypical fashion over the ten weeks following weaning, were raised on diets compounded with myo-inositol. As expected, mice treated with myo-inositol diet showed a significant decrease in the number, size, and stage of lesions as compared to those raised on control diets. Interestingly, also in this mouse model, there was a significant decrease in IL-6 levels at both proteomic and cytokine analyses.
We take the opportunity to summarize the available pieces of evidence suggesting that the strong reduction of IL-6 after treatment with myo-inositol could depend on two different mechanisms that may occur simultaneously, as we recently pointed out [71]. On the one hand, the change in signal transducer and activator of transcription 3 (STAT3) phosphorylation could reduce macrophage recruitment and their phenotype switching, with consequent reduction of IL-6-producing cell population [72]. On the other hand, the fine-regulated cross-talk between IL and 6-mediated pathways and surfactant-derived phosphatidylglycerol subfractions may account for the return to the homeostasis in the bronchoalveolar microenvironment [73], [74], [75].
These elements might have a role of paramount importance even for the adjuvant treatment of several conditions which show increased bronchoalveolar levels of IL-6, such as SARS-CoV-2 pneumonia [76], [77]. Indeed, the infections in the upper and lower respiratory tract by novel coronavirus-19 in humans causes a mild or highly acute respiratory syndrome, with consequent release of pro-inflammatory cytokines (the so-called “cytokine storm”), including IL-1β and IL-6 [78], lymphopenia (in CD4 + and CD8 + T cells) and decreased interferon (IFN)-γ expression in CD4 + T cells [79]. In addition, recent findings from the Chinese population who underwent SARS-CoV-2 infection confirmed that increased expression of IL-6 in serum is expected to predict the severity of SARS-CoV-2 pneumonia and the prognosis of patients: higher are the IL-6 serum levels, worse is the prognosis among sub-clusters of infected patients [80], [81]. This element appears very important, considering that the action of the inositol on the IRE1-X-box-binding protein 1 (XBP1) pathway is able to inhibit the IL-6 production and, in this way, reduce the prof-inflammatory cascade by activation of the IL-6-STAT3 signaling [82], [83], [84].
Conclusions and future perspective
To date, the spread of SARS-CoV-2 infection is increasing worldwide and represents a primary healthcare emergency. Although the infection can be asymptomatic, several cases develop severe pneumonia and ARDS characterized by high levels of pro-inflammatory cytokines, primarily IL-6. Based on the available data, the severity of ARDS and serum levels of IL-6 are key determinants for the prognosis of the patients.
Available robust in vitro and in vivo data suggested that myo-inositol is able to increase the synthesized amount of surfactant phosphatidylinositol and its functions, ameliorating both immune defensive system and oxygenation at the bronchoalveolar level. In addition, myo-inositol has been found able to decrease the levels of IL-6 in several experimental settings, due to a robust effect on the IRE1-XBP1 and STAT3 pathways. In this scenario, treatment with myo-inositol may be able to reduce IL-6 dependent inflammatory response and improve oxygenation in patients with severe ARDS by SARS-CoV-2. In addition, the action of myo-inositol on IRE1 endonuclease activity may also inhibit the replication of SARS-CoV-2, as was found for the respiratory syncytial virus.
Since the available data are extremely limited, if this potential therapeutic approach will be considered valid in the clinical practice, the necessary future investigations should aim to identify the best dose, administration route (oral, intravenous and/or aerosol nebulization), and cluster(s) of patients which can benefit from this treatment.
Declaration of Competing Interest
Vittori Unfer is employee at Lo.Li. Pharma s.r.l. (Rome, Italy). All the other authors have no proprietary, financial, professional or other personal interest of any nature in any product, service or company.
References
- 1.Sunde M., Pham C.L.L., Kwan A.H. Molecular Characteristics and Biological Functions of Surface-Active and Surfactant Proteins. Annu Rev Biochem. 2017;86:585–608. doi: 10.1146/annurev-biochem-061516-044847. [DOI] [PubMed] [Google Scholar]
- 2.Mindt B.C., Fritz J.H., Duerr C.U. Group 2 innate lymphoid cells in pulmonary immunity and tissue homeostasis. Front Immunol. 2018;9:840. doi: 10.3389/fimmu.2018.00840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rodriguez R.J. Respiratory Care. Daedalus Enterprises Inc.; 2003. Management of respiratory distress syndrome: An update; pp. 279–286. [PubMed] [Google Scholar]
- 4.Rynkiewicz M.J., Wu H., Cafarella T.R., Nikolaidis N.M., Head J.F., Seaton B.A. Differential ligand binding specificities of the pulmonary collectins are determined by the conformational freedom of a surface loop. Biochemistry. 2017;56:4095–4105. doi: 10.1021/acs.biochem.6b01313. [DOI] [PubMed] [Google Scholar]
- 5.Kishore U., Greenhough T.J., Waters P., Shrive A.K., Ghai R., Kamran M.F. Surfactant proteins SP-A and SP-D: Structure, function and receptors. Mol Immunol. 2006;43:1293–1315. doi: 10.1016/j.molimm.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 6.Wright JR. Host defense functions of pulmonary surfactant. In: Biology of the Neonate. Biol Neonate; 2004. p. 326–32. [DOI] [PubMed]
- 7.Schürch S., Bachofen H., Possmayer F. Surface activity in situ, in vivo, and in the captive bubble surfactometer. Comput Biochem Physiol A: Mol Integr Physiol. 2001;129:195–207. doi: 10.1016/s1095-6433(01)00316-6. [DOI] [PubMed] [Google Scholar]
- 8.Fujioka H., Halpern D., Gaver D.P. A model of surfactant-induced surface tension effects on the parenchymal tethering of pulmonary airways. J Biomech. 2013;46:319–328. doi: 10.1016/j.jbiomech.2012.11.031. [DOI] [PubMed] [Google Scholar]
- 9.Crouch E.C. Collectins and pulmonary host defense. Am J Respir Cell Mol Biol. 1998;19:177–201. doi: 10.1165/ajrcmb.19.2.140. [DOI] [PubMed] [Google Scholar]
- 10.Crouch E., McDonald B., Smith K., Roberts M., Mealy T., Seaton B. Critical role of Arg/Lys343 in the species-dependent recognition of phosphatidylinositol by pulmonary surfactant protein D. Biochemistry. 2007;46:5160–5169. doi: 10.1021/bi700037x. [DOI] [PubMed] [Google Scholar]
- 11.Ikegami M., Grant S., Korfhagen T., Scheule R.K., Whitsett J.A. Surfactant protein-D regulates the postnatal maturation of pulmonary surfactant lipid pool sizes. J Appl Physiol. 2009;106:1545–1552. doi: 10.1152/japplphysiol.91567.2008. [DOI] [PubMed] [Google Scholar]
- 12.Sawada K., Ariki S., Kojima T., Saito A., Yamazoe M., Nishitani C. Pulmonary collectins protect macrophages against pore-forming activity of Legionella pneumophila and suppress its intracellular growth. J Biol Chem. 2010;285:8434–8443. doi: 10.1074/jbc.M109.074765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Laganà A.S., Garzon S., Casarin J., Franchi M., Ghezzi F. Inositol in Polycystic Ovary Syndrome: Restoring Fertility through a Pathophysiology-Based Approach. Trends Endocrinol Metab. 2018;29:768–780. doi: 10.1016/j.tem.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 14.Facchinetti F., Appetecchia M., Aragona C., Bevilacqua A., Bezerra Espinola M.S., Bizzarri M. Experts’ opinion on Inositols in treating polycystic ovary syndrome and non-insulin dependent diabetes mellitus: a further help for human reproduction and beyond. Expert Opin Drug Metab Toxicol. 2020;16:255–274. doi: 10.1080/17425255.2020.1737675. [DOI] [PubMed] [Google Scholar]
- 15.Garzon S., Laganà A.S., Monastra G. Risk of reduced intestinal absorption of Myo-inositol caused by D-chiro-inositol or by glucose transporter inhibitors. Expert Opin Drug Metab Toxicol. 2019;15:697–703. doi: 10.1080/17425255.2019.1651839. [DOI] [PubMed] [Google Scholar]
- 16.Laganà A.S., Rossetti P., Sapia F., Chiofalo B., Buscema M., Valenti G. Evidence-Based and Patient-Oriented Inositol Treatment in Polycystic Ovary Syndrome: Changing the Perspective of the Disease. Int J Endocrinol Metab. 2017;15 doi: 10.5812/ijem.43695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bill C.A., Vines C.M., Phospholipase C. Springer; New York LLC: 2020. Advances in experimental medicine and biology; pp. 215–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhao K., Li G., Yao Y., Zhou Y., Li Z., Guo Q. Activation of phospholipase C-γ1 and translocation of phosphatidylinositol-3,4,5-trisphosphate 3-phosphatase contribute to GL-V9-induced apoptosis in human gastric cancer cells. Exp Cell Res. 2017;356:8–19. doi: 10.1016/j.yexcr.2017.03.063. [DOI] [PubMed] [Google Scholar]
- 19.Beharka A.A., Crowther J.E., McCormack F.X., Denning G.M., Lees J., Tibesar E. Pulmonary Surfactant Protein A Activates a Phosphatidylinositol 3-Kinase/Calcium Signal Transduction Pathway in Human Macrophages: Participation in the Up-Regulation of Mannose Receptor Activity. J Immunol. 2005;175:2227–2236. doi: 10.4049/jimmunol.175.4.2227. [DOI] [PubMed] [Google Scholar]
- 20.Rubarth L.B., Quinn J. Respiratory Development and Respiratory Distress Syndrome. Neonatal Netw. 2015;34:231–238. doi: 10.1891/0730-0832.34.4.231. [DOI] [PubMed] [Google Scholar]
- 21.Clements J.A., Avery M.E. Lung surfactant and neonatal respiratory distress syndrome. Am J Respir Crit Care Med. 1998;157:S59–S66. doi: 10.1164/ajrccm.157.4.nhlb1-1. [DOI] [PubMed] [Google Scholar]
- 22.Kopelman A.E., Mathew O.P. Common respiratory disorders of the newborn. Pediatr Rev. 1995;16:209–217. doi: 10.1542/pir.16-6-209. [DOI] [PubMed] [Google Scholar]
- 23.Gluck L., Kulovich M.V., Borer R.C., Brenner P.H., Anderson G.G., Spellacy W.N. Diagnosis of the respiratory distress syndrome by amniocentesis. Am J Obstet Gynecol. 1971;109:440–445. doi: 10.1016/0002-9378(71)90342-5. [DOI] [PubMed] [Google Scholar]
- 24.Hallman M., Teramo K. Measurement of the lecithin/sphingomyelin ratio and phosphatidylglycerol in aminotic fluid: an accurate method for the assessment of fetal lung maturity. Br J Obstet Gynaecol. 1981;88:806–813. doi: 10.1111/j.1471-0528.1981.tb01307.x. [DOI] [PubMed] [Google Scholar]
- 25.Bernhard W. Lung surfactant: Function and composition in the context of development and respiratory physiology. Ann Anat. 2016;208:146–150. doi: 10.1016/j.aanat.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 26.Luo J.H., Chen J., Li Q.P., Feng Z.C. Differences in clinical characteristics and therapy of neonatal acute respiratory distress syndrome (ARDS) and respiratory distress syndrome (RDS): A retrospective analysis of 925 cases. Med Sci Monit. 2019;25:4992–4998. doi: 10.12659/MSM.915213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fan E., Brodie D., Slutsky A.S. Acute respiratory distress syndrome advances in diagnosis and treatment. JAMA. 2018;319:698–710. doi: 10.1001/jama.2017.21907. [DOI] [PubMed] [Google Scholar]
- 28.Seeds M.C., Grier B.L., Suckling B.N., Safta A.M., Long D.L., Waite B.M. Secretory phospholipase A2-mediated depletion of phosphatidylglycerol in early acute respiratory distress syndrome. Am J Med Sci. 2012;343:446–451. doi: 10.1097/MAJ.0b013e318239c96c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cheifetz I.M. Pediatric ARDS. Respir Care. 2017;62:718–731. doi: 10.4187/respcare.05591. [DOI] [PubMed] [Google Scholar]
- 30.Enhörning G., Robertson B. Lung expansion in the premature rabbit fetus after tracheal deposition of surfactant. Pediatrics. 1972;50:58–66. [PubMed] [Google Scholar]
- 31.Rodriguez R.J., Martin R.J. Exogenous surfactant therapy in newborns. Respir Care Clin N Am. 1999;5:595–616. [PubMed] [Google Scholar]
- 32.Bahadue F.L., Soll R. Early versus delayed selective surfactant treatment for neonatal respiratory distress syndrome. Cochrane Database Syst Rev. 2012 doi: 10.1002/14651858.CD001456.pub2. ;11:CD001456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hallman M. Lung Surfactant, Respiratory Failure, and Genes. N Engl J Med. 2004;350:1278–1280. doi: 10.1056/NEJMp048036. [DOI] [PubMed] [Google Scholar]
- 34.Roberts D., Brown J., Medley N., Dalziel S.R. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev. 2017 doi: 10.1002/14651858.CD004454.pub3. ;3:CD004454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Halliday H., Ehrenkranz R., Doyle L. Early postnatal (<96 hours) corticosteroids for preventing chronic lung disease in preterm infants. Cochrane Database Syst Rev. 2003 doi: 10.1002/14651858.CD001146. 1:CD001146. [DOI] [PubMed] [Google Scholar]
- 36.Moya F., Maturana A. animal-derived surfactants versus past and current synthetic surfactants: current status. Clin Perinatol. 2007;34:145–177. doi: 10.1016/j.clp.2006.12.005. [DOI] [PubMed] [Google Scholar]
- 37.Ardell S., Pfister R.H., Soll R. Animal derived surfactant extract versus protein free synthetic surfactant for the prevention and treatment of respiratory distress syndrome. Cochrane Database Syst Rev. 2015 doi: 10.1002/14651858.CD000144.pub3. ;8:CD000144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Halliday H.L. Synthetic or natural surfactants. Acta Paediatr. 1997;86:233–237. doi: 10.1111/j.1651-2227.1997.tb08880.x. [DOI] [PubMed] [Google Scholar]
- 39.Polin R.A., Carlo W.A. Committee on Fetus and Newborn; American Academy of Pediatrics. Surfactant replacement therapy for preterm and term neonates with respiratory distress. Pediatrics. 2014;133:156–163. doi: 10.1542/peds.2013-3443. [DOI] [PubMed] [Google Scholar]
- 40.Hallman M., Saugstad O.D., Porreco R.P., Epstein B.L., Gluck L. Role of myoinositol in regulation of surfactant phospholipids in the newborn. Early Hum Dev. 1985;10:245–254. doi: 10.1016/0378-3782(85)90055-6. [DOI] [PubMed] [Google Scholar]
- 41.Lewin L.M., Melmed S., Passwell J.H., Yannai Y., Brish M., Orda S. Myoinositol in human neonates: Serum concentrations and renal handling. Pediatr Res. 1978;12:3–6. doi: 10.1203/00006450-197801000-00002. [DOI] [PubMed] [Google Scholar]
- 42.Burton L.E., Wells W.W. Studies on the developmental pattern of the enzymes converting glucose 6-phosphate to Myo-inositol in the rat. Dev Biol. 1974;37:35–42. doi: 10.1016/0012-1606(74)90167-5. [DOI] [PubMed] [Google Scholar]
- 43.Bromberger P., Hallman M. Myoinositol in small preterm infants: Relationship between intake and serum concentration. J Pediatr Gastroenterol Nutr. 1986;5:455–458. doi: 10.1097/00005176-198605000-00021. [DOI] [PubMed] [Google Scholar]
- 44.Batenburg J.J., Klazinga W., van Golde L.M. Regulation and location of phosphatidylglycerol and phosphatidylinositol synthesis in type II cells isolated from fetal rat lung. BBA. 1985;833:17–24. doi: 10.1016/0005-2760(85)90248-6. [DOI] [PubMed] [Google Scholar]
- 45.Batenburg J.J., Klazinga W., van Golde L.M. Regulation of phosphatidylglycerol and phosphatidylinositol synthesis in alveolar type II cells isolated from adult rat lung. FEBS Lett. 1982;147:171–174. doi: 10.1016/0014-5793(82)81035-1. [DOI] [PubMed] [Google Scholar]
- 46.Hallman M. Effect of extracellular Myo-inositol on surfactant phospholipid synthesis in the fetal rabbit lung. BBA. 1984;795:67–78. doi: 10.1016/0005-2760(84)90105-x. [DOI] [PubMed] [Google Scholar]
- 47.Bleasdale J.E. Methods for Altering the Synthesis of Phosphatidylinositol and Phosphatidylglycerol by Type II Pneumonocytes. Methods Enzymol. 1987;141:222–234. doi: 10.1016/0076-6879(87)41070-7. [DOI] [PubMed] [Google Scholar]
- 48.Anceschi M.M., Petrelli A., Zaccardo G., Barbati A., Di Renzo G.C., Cosmi E.V. Inositol and glucocorticoid in the development of lung stability in male and female rabbit fetuses. Pediatr Res. 1988;24:617–621. doi: 10.1203/00006450-198811000-00016. [DOI] [PubMed] [Google Scholar]
- 49.Beemster P., Groenen P., Steegers-Theunissen R. Involvement of inositol in reproduction. Nutr Rev. 2002;60:80–87. doi: 10.1301/00296640260042748. [DOI] [PubMed] [Google Scholar]
- 50.Mallampalli R.K., Mathur S.N., Warnock L.J., Salome R.G., Hunninghake G.W., Field F.J. Betamethasone modulation of sphingomyelin hydrolysis up-regulates CTP:cholinephosphate cytidylyltransferase activity in adult rat lung. Biochem J. 1996;318:333–341. doi: 10.1042/bj3180333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cornell R.B., Ridgway N.D. CTP:phosphocholine cytidylyltransferase: Function, regulation, and structure of an amphitropic enzyme required for membrane biogenesis. Prog Lipid Res. 2015;59:147–171. doi: 10.1016/j.plipres.2015.07.001. [DOI] [PubMed] [Google Scholar]
- 52.Gobran L.I., Xu Z.X., Rooney S.A. PKC isoforms and other signaling proteins involved in surfactant secretion in developing rat type II cells. Am J Physiol. 1998;274:L901–L907. doi: 10.1152/ajplung.1998.274.6.L901. [DOI] [PubMed] [Google Scholar]
- 53.Hallman M., Arjomaa P., Hoppu K. Inositol supplementation in respiratory distress syndrome: Relationship between serum concentration, renal excretion, and lung effluent phospholipids. J Pediatr. 1987;110:604–610. doi: 10.1016/s0022-3476(87)80561-9. [DOI] [PubMed] [Google Scholar]
- 54.Hallman M., Bry K., Hoppu K., Lappi M., Pohjavuori M. Inositol supplementation in premature infants with respiratory distress syndrome. N Engl J Med. 1992;326:1233–1239. doi: 10.1056/NEJM199205073261901. [DOI] [PubMed] [Google Scholar]
- 55.Phelps D.L., Ward R.M., Williams R.L., Watterberg K.L., Laptook A.R., Wrage L.A. Pharmacokinetics and safety of a single intravenous dose of Myo-inositol in preterm infants of 23–29 wk. Pediatr Res. 2013;74:721–729. doi: 10.1038/pr.2013.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Phelps D.L., Ward R.M., Williams R.L., Nolen T.L., Watterberg K.L., Oh W. Safety and pharmacokinetics of multiple dose Myo-inositol in preterm infants. Pediatr Res. 2016;80:209–217. doi: 10.1038/pr.2016.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hotamisligil G.S. Endoplasmic Reticulum Stress and the Inflammatory Basis of Metabolic Disease. Cell. 2010;140:900–917. doi: 10.1016/j.cell.2010.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ye Z., Wong C.K., Li P., Xie Y. A SARS-CoV protein, ORF-6, induces caspase-3 mediated, ER stress and JNK-dependent apoptosis. BBA. 2008;1780:1383–1387. doi: 10.1016/j.bbagen.2008.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kennedy D., Samali A., Jäger R. Methods for studying ER stress and UPR markers in human cells. Methods Mol Biol. 2015;1292:3–18. doi: 10.1007/978-1-4939-2522-3_1. [DOI] [PubMed] [Google Scholar]
- 60.Hassan I., Gaines K.S., Hottel W.J., Wishy R.M., Miller S.E., Powers L.S. Inositol-requiring enzyme 1 inhibits respiratory syncytial virus replication. J Biol Chem. 2014;289:7537–7546. doi: 10.1074/jbc.M113.510594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Shyu P., Jr, Ng B.S.H., Ho N., Chaw R., Seah Y.L., Marvalim C. Membrane phospholipid alteration causes chronic ER stress through early degradation of homeostatic ER-resident proteins. Sci Rep. 2019;9:8637. doi: 10.1038/s41598-019-45020-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hassan I.H., Zhang M.S., Powers L.S., Shao J.Q., Baltrusaitis J., Rutkowski D.T. Influenza A viral replication is blocked by inhibition of the inositol-requiring enzyme 1 (IRE1) stress pathway. J Biol Chem. 2012;287:4679–4689. doi: 10.1074/jbc.M111.284695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Moro-Sibilot D., Jeanmart M., Lantuejoul S., Arbib F., Laverrière M.H., Brambilla E. Cigarette smoking, preinvasive bronchial lesions, and autofluorescence bronchoscopy. Chest. 2002;122:1902–1908. doi: 10.1378/chest.122.6.1902. [DOI] [PubMed] [Google Scholar]
- 64.Schwartz A.G., Cote M.L. Epidemiology of lung cancer. Adv Exp Med Biol. 2016;893:21–41. doi: 10.1007/978-3-319-24223-1_2. [DOI] [PubMed] [Google Scholar]
- 65.Hecht S.S., Kenney P.M., Wang M., Upadhyaya P. Dose-response study of Myo-inositol as an inhibitor of lung tumorigenesis induced in A/J mice by benzo[a]pyrene and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone. Cancer Lett. 2001;167:1–6. doi: 10.1016/s0304-3835(01)00454-2. [DOI] [PubMed] [Google Scholar]
- 66.Wattenberg L.W. Chemoprevention of pulmonary carcinogenesis by brief exposures to aerosolized budesonide or beclomethasone dipropionate and by the combination of aerosolized budesonide and dietary Myo-inositol. Carcinogenesis. 2000;21:179–182. doi: 10.1093/carcin/21.2.179. [DOI] [PubMed] [Google Scholar]
- 67.Estensen R.D., Wattenberg L.W. Studies of chemopreventive effects of Myo-inositol on benzo(a)pyrene-induced neoplasia of the lung and forestomach of female a/j mice. Carcinogenesis. 1993;14:1975–1977. doi: 10.1093/carcin/14.9.1975. [DOI] [PubMed] [Google Scholar]
- 68.Lam S., McWilliams A., LeRiche J., MacAulay C., Wattenberg L., Szabo E. A phase I study of Myo-inositol for lung cancer chemoprevention. Cancer Epidemiol Biomarkers Prev. 2006;15:1526–1531. doi: 10.1158/1055-9965.EPI-06-0128. [DOI] [PubMed] [Google Scholar]
- 69.Lam S., Mandrekar S.J., Gesthalter Y., Allen Ziegler K.L., Seisler D.K., Midthun D.E. A Randomized Phase IIb Trial of Myo-inositol in Smokers with Bronchial Dysplasia. Cancer Prev Res. 2016;9:906–914. doi: 10.1158/1940-6207.CAPR-15-0254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Unver N., Delgado O., Zeleke K., Cumpian A., Tang X., Caetano M.S. Reduced IL-6 levels and tumor-associated phospho-STAT3 are associated with reduced tumor development in a mouse model of lung cancer chemoprevention with Myo-inositol. Int J Cancer. 2018;142:1405–1417. doi: 10.1002/ijc.31152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Bizzarri M., Laganà A.S., Aragona D., Unfer V. Inositol and pulmonary function. Could Myo-inositol treatment downregulate inflammation and cytokine release syndrome in SARS-CoV-2? Eur Rev Med Pharmacol Sci. 2020;24:3426–3432. doi: 10.26355/eurrev_202003_20715. [DOI] [PubMed] [Google Scholar]
- 72.Jung B.G., Wang X., Yi N., Ma J., Turner J., Samten B. Early Secreted Antigenic Target of 6-kDa of Mycobacterium tuberculosis Stimulates IL-6 Production by Macrophages through Activation of STAT3. Sci Rep. 2017;7:40984. doi: 10.1038/srep40984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Spengler D., Winoto-Morbach S., Kupsch S., Vock C., Blöchle K., Frank S. Novel therapeutic roles for surfactant-inositols and -phosphatidylglycerols in a neonatal piglet ARDS model: A translational study. Am J Physiol. 2018;314:L32–L53. doi: 10.1152/ajplung.00128.2017. [DOI] [PubMed] [Google Scholar]
- 74.Kopincova J., Mikolka P., Kolomaznik M., Kosutova P., Calkovska A., Mokra D. Selective inhibition of NF-κB and surfactant therapy in experimental meconium-induced lung injury. Physiol Res. 2017;66:S227–S236. doi: 10.33549/physiolres.933678. [DOI] [PubMed] [Google Scholar]
- 75.Steffen L., Ruppert C., Hoymann H.G., Funke M., Ebener S., Kloth C. Surfactant replacement therapy reduces acute lung injury and collapse induration-related lung remodeling in the bleomycin model. Am J Physiol. 2017;313:L313–L327. doi: 10.1152/ajplung.00033.2017. [DOI] [PubMed] [Google Scholar]
- 76.Wang W., Ye L., Ye L., Li B., Gao B., Zeng Y. Up-regulation of IL-6 and TNF-α induced by SARS-coronavirus spike protein in murine macrophages via NF-κB pathway. Virus Res. 2007;128:1–8. doi: 10.1016/j.virusres.2007.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yoshikawa T., Hill T., Li K., Peters C.J., Tseng C.-T.-K. Severe Acute Respiratory Syndrome (SARS) Coronavirus-Induced Lung Epithelial Cytokines Exacerbate SARS Pathogenesis by Modulating Intrinsic Functions of Monocyte-Derived Macrophages and Dendritic Cells. J Virol. 2009;83:3039–3048. doi: 10.1128/JVI.01792-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Conti P., Ronconi G., Caraffa A., Gallenga C.E., Ross R., Frydas I. Induction of Pro-Inflammatory Cytokines (IL-1 and IL-6) and Lung Inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): Anti-Inflammatory Strategies. J Biol Regul Homeost Agents. 2020 doi: 10.23812/CONTI-E. [DOI] [PubMed] [Google Scholar]
- 79.Pedersen S.F., Ho Y.-C. SARS-CoV-2: A Storm is Raging. J Clin Invest. 2020;130:2202–2205. doi: 10.1172/JCI137647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, et al. Clinical and immunologic features in severe and moderate Coronavirus Disease 2019. 2020;130:2620-9. [DOI] [PMC free article] [PubMed]
- 81.Chen F., Jin J., Hu J., Wang Y., Ma Z., Zhang J. Endoplasmic reticulum stress cooperates in silica nanoparticles-induced macrophage apoptosis via activation of CHOP-mediated apoptotic signaling pathway. Int J Mol Sci. 2019;20:5846. doi: 10.3390/ijms20235846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fang P., Xiang L., Huang S., Jin L., Zhou G., Zhuge L. IRE1α-XBP1 signaling pathway regulates IL-6 expression and promotes progression of hepatocellular carcinoma. Oncol Lett. 2018;16:4729–4736. doi: 10.3892/ol.2018.9176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Choi S., Snider J.M., Olakkengil N., Lambert J.M., Anderson A.K., Ross-Evans J.S. Myristate-induced endoplasmic reticulum stress requires ceramide synthases 5/6 and generation of C14-ceramide in intestinal epithelial cells. FASEB J. 2018;32:5724–5736. doi: 10.1096/fj.201800141R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhang B., Gao C., Li Y., Wang M. D-chiro-inositol enriched Fagopyrum tataricum (L.) Gaench extract alleviates mitochondrial malfunction and inhibits ER stress/JNK associated inflammation in the endothelium. J Ethnopharmacol. 2018;214:83–89. doi: 10.1016/j.jep.2017.12.002. [DOI] [PubMed] [Google Scholar]