Graphical abstract
Abbreviations: COVID-19, Coronavirus disease 2019; SARS-CoV-2, Severe acute respiratory syndrome coronavirus 2; SARS-CoV, Severe acute respiratory syndrome coronavirus; ACE, Angiotensin-converting enzyme; ACE2, Angiotensin-converting enzyme 2; ADT, Androgen deprivation therapy; WHO, World health organization; JSTOR, Journal storage; ICU, Intensive care unit; TMPRSS2, Transmembrane protein serine 2; E1, Estron; E2, Estradiol; E3, Estriol; ERs, Estrogen receptors; ERα, Estrogen receptor-alpha; ERβ, Estrogen receptor-beta; NKCs, Natural killer cells; CXCL8, C-X-C motif chemokine ligand 8; NF-kB, Nuclear factor kappa B; IFN-γ, Interferon-gamma; CCL2, C-C motif ligand 2; TLR7, Toll-like receptor 7; RNA, Ribonucleic acid; mRNA, Messenger RNA; IL-6, Interleukin 6; CXCL-1, C-X-C motif ligand 1; TLR8, Toll-like receptor 8; CD40L, CXCR3, C-X-C motif receptor 3; FcγRIIIA, Low affinity immunoglobulin gamma Fc region receptor III-A; ADCC, Antibody-dependent cellular cytotoxicity; H3N2, Influenza A virus subtype H3N2; H1N1, Influenza A virus subtype H1N1; HCoV-NL63, Human coronavirus NL63; Xp22, Chromosome X band p22; RAS, Renin-angiotensin system; ADAM10, Metalloproteinase domain-containing protein 10; ADAM17, Metalloproteinase domain-containing protein 17; PDI, Protein disulfide isomerase; ePDI, Extra-endoplasmic reticulum PDI; ROS, Reactive oxygen species
Keywords: COVID-19, SARS-CoV-2, Estrogen, Androgen, Cytokine storm, Angiotensin-Converting enzyme-2 (ACE2), Inflammation
Highlights
-
•
Effect of sex hormones on COVID-19 outcome bias in males and females.
-
•
Effect of immunological factors on COVID-19 outcome bias in males and females.
-
•
Angiotensin-converting enzyme-2 (ACE2) system and COVID-19 outcome in males and females.
-
•
Correlation between smoking and COVID-19 incidence in males and females.
-
•
Possible therapeutic options for COVID-19.
Abstract
The severe form of COVID-19 has significant sex disparities, with high fatalities commonly reported among males than females. The incidence of COVID-19 has also been higher in males compared with their female counterparts. This trend could be attributed to a better responsive and robust immune system in females. Cytokine storm is one of the pathophysiological features of severe COVID-19, and it occurs as a result of over-activation of immune cells leading to severe inflammation and tissue damage. Nevertheless, it is well modulated in females compared to their male counterparts. Severe inflammation in males is reported to facilitate progression of mild to severe COVID-19. The sex hormones, estrogens and androgens which exist in varying functional levels respectively in females and males are cited as the underlying cause for the differential immune response to COVID-19. Evidence abounds that estrogen modulate the immune system to protect females from severe inflammation and for that matter severe COVID-19. On the contrary, androgen has been implicated in over-activation of immune cells, cytokine storm and the attendant severe inflammation, which perhaps predispose males to severe COVID-19. In this review efforts are made to expand understanding and explain the possible roles of the immune system, the sex hormones and the angiotensin-converting enzyme (ACE) systems in male bias to severe COVID-19. Also, this review explores possible therapeutic avenues including androgen deprivation therapy (ADT), estrogen-based therapy, and ACE inhibitors for consideration in the fight against COVID-19.
1. Introduction
The general notion since the outbreak of COVID-19 in December 2019 has been that men are more likely than women to contract the infection [1]. The proponents of this notion have largely based their argument on the data emanated from China and Spain in the early stages of the outbreak. According to a study in early February 2020, in Wuhan, 51 % (86/168) of the hospitalized patients were males [2]. In a similar study carried out in the early part of April 2020 in Spain, males accounted for 61.0 % (2433/3987) of hospitalized patients [3]. These data support the above proposition, however, in reality, only hospitalized patients were captured in these studies, which presupposes that the patients included in these studies were severely ill. Hence, data on patients with mild symptoms who could equally be females may not have been captured in the early part of the outbreak.
Elsewhere, other studies have reported results that show a complete departure from the above assertion. For instance, the proportion of female to male active cases reported in other countries including Switzerland (53 % of female : 47 % of men), Netherlands (53 % : 47 %), Belgium (55 % : 45 %), South Korea (60 % : 40 %), Portugal (57 % : 43 %), Canada (52 % : 48 %), and Republic of Ireland (55 % : 45 %) [4] showed different trend. Available data show that the incidence of COVID-19 among males and females is not only country related, but also regionally diversified. Therefore, the conclusion is that males and females are equally likely to contract the novel coronavirus, except that, men are more likely to suffer severe COVID-19 [5]. This may be the true situation when all the available data are thoroughly interrogated. Other studies have provided data to corroborate this assertion. For instance, the World Health Organization (WHO) reported that 63 % of COVID-19 related deaths in Europe were among men [6]. Also, more than 60 % of 5700 COVID-19 patients hospitalized in New York City were men [5]. Another study has also reported that 75 % of all COVID-19 related deaths in Wuhan were males [7].
On the bases of the data emanating from various COVID-19 studies since the outbreak in December 2019, it will be appropriate to categorize the male as a risk factor for severe COVID-19. Many reasons have been adduced for this observation, spanning from lifestyle, hormonal, immunological, enzymatic to genetic variations among the sexes [8,9]. This review expands understanding on the possible biological reasons which underpin the differences observed in the outcome of COVID-19 among males and females.
2. Methodology
A literature search was conducted in PubMed, Scopus, Web of Science, ScienceDirect, JSTOR, and Google Scholar with search terms including COVID-19 positive cases, COVID-19 sex disaggregation, severe COVID-19, mild COVID-19, Country-specific COVID-19 cases, sex hormones and COVID-19, sex hormones and inflammation and COVID-19 cell invasion. A total of 332 articles were found, of which 127 were included in this review, based on their relevance to this review, reliability of research methodology, innovation and scope. Sex disaggregated data of COVID-19 from December 2019 to 3rd July 2020 was extracted from the WHO’s website, and WHO accredited databases for COVID-19 cases (the Johns Hopkins University website, the UN women count’s website and the Global Health 50/50 website) [[10], [11], [12], [13]]. The data was then cross-referenced with country-specific dedicated websites for Covid-19. Data analysis was done using Chi-Square with Yate’s correction using GraphPad Prism v.8. Analyses of these data are presented in Figs. 2, 5 and 6. This study was limited by the difficulty to obtain real-time sex-disaggregated data on hospital admissions, ICU admissions and deaths due to lack of uniform and regular update of such data on most countries’ COVID-19 dedicated websites.
Fig. 2.
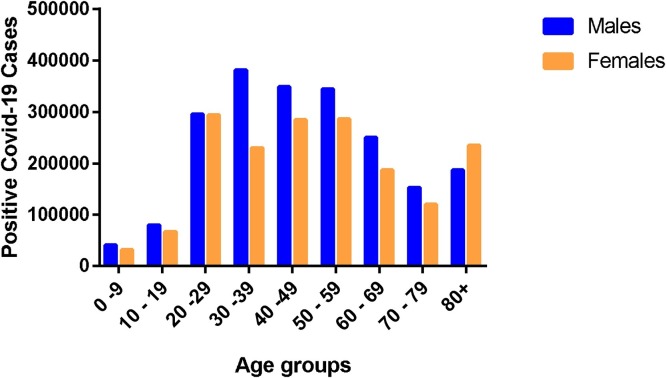
Age-group gender-disaggregated confirmed COVID-19 cases.
3. Sex hormones and COVID-19 outcome in males and females
Evidence available suggests that males are more susceptible to severe Covid-19 than females, and this disparity is largely attributed to hormonal differences [14,15]. One of the factors that directly influence the structural and functional differences among males and females is the differential production and action of the two classes of sex hormones [15,16]. Thus, estrogen in females and androgen in males [17]. There are suggestions that estrogen protects females against severe COVID-19 while androgen worsens Covid-19 outcome in males [18]. This hypothesis needs to be interrogated to prove or disprove its correctness. Hence, the effects of these hormones on the outcome of COCID-19 in males and females are discussed.
3.1. Androgen and COVID-19 outcome in males and females
The effect of androgen on COVID-19 outcome has been investigated in Italy [19]. In this study, patients with prostate cancer undergoing treatment with androgen deprivation therapy (ADT) were found to be less likely to develop severe COVID-19 compared to the non-ADT group, especially those with other cancer conditions. It is evident from this study that androgen possibly renders the virus more virulent or confers some immunological vulnerability in males, therefore exacerbating the severity of COVID-19. To further investigate the correlation between androgen levels and severe COVID-19, severe COVID-19 was assessed in patients with or without ADT. Here, prostate cancer patients receiving ADT and prostate cancer patients not on ADT were examined. As anticipated, patients on ADT recorded a significant fourfold reduced risk of severe COVID-19 compared to the patients who were not on ADT. The difference was more pronounced (fivefold risk reduction) when prostate cancer patients on ADT were compared with patients with other cancer conditions [17,19,20]. Inferring from these studies, it can be postulated that high concentration of androgen exacerbate the severity of COVID-19. This is an intriguing hypothesis that warrants further investigation to determine the scientific bases to this trend. Hence, there is the need to precipitate the biochemical and molecular bases for the correlation between androgen level and severe COVID-19.
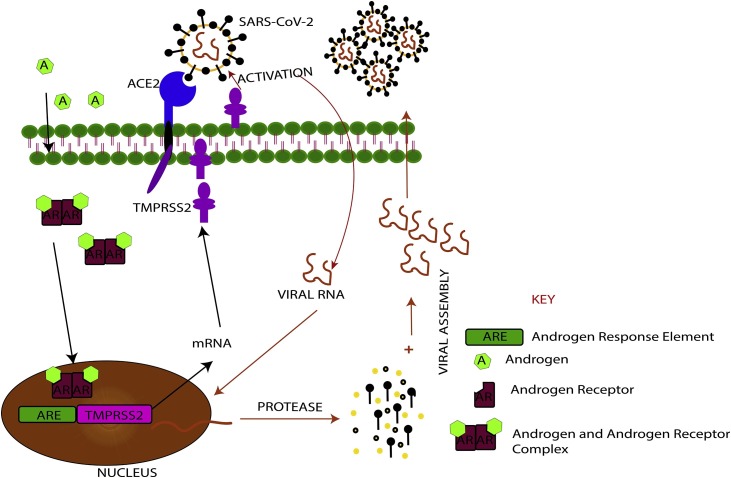
As has already been established, SARS-CoV-2 enters the human cell by binding to ACE2 through its viral spike (S) proteins, and the S protein priming by transmembrane serine protease 2 (TMPRSS2) [21,22]. The TMPRSS2 is a member of a family of proteins called type II transmembrane serine proteases involved in the development of several diseases, including viral infections such as coronavirus infections [23]. It is worth noting that TMPRSS2 is an androgen-regulated gene that is upregulated in prostate cancer condition and therefore supports tumour progression [24,25]. More important, there is evidence that the same androgen receptor regulates TMPRSS2 expression in non-prostatic tissues, including the lung [19]. Moreover, ADT is known to exert its activity by downregulating expression and activity of TMPRSS2, an indication that TMPRSS2 expression levels correlate positively with androgen expression [25].
The functional link between androgen and TMPRSS2 is enough indication that the hormone androgen is involved in the progression of mild to severe COVID-19 (Fig. 1 ). On the contrary, the female is relatively safe with respect to severe COVID-19 due to the relatively low androgen level. Per the evidence, androgen deprivation therapy (ADT) may help protect men against severe COVID-19. This important therapeutic approach could be evaluated and possibly approved as one of the therapeutic options for the management of COVID-19 as the world continues to search for a potent therapies. Notwithstanding, ADT is reported to be associated with adverse effects including, but not limited to: vasomotor complications, sexual dysfunction and gynecomastia, osteoporosis, metabolic syndrome, and depression. Additionally, neurocognitive deficits, thromboembolic disease, and cardiovascular disease have also been cited as side effects [26]. In effect, ADT can only be employed in the management of COVID-19 if these notable side effects are well managed. Managing these side effects will obviously reduce the ADT related morbidity. To this end, various interventions have been described to successfully manage these side effects. These include aerobic exercise to improve sexual and cognitive functions, the use of Denosumab to increase lumbar spine, hip and radius bone mass density, and Metformin for adverse metabolic changes [27]. More important, intermittent ADT has been found to improve early side effects, such as hot flashes, sexual function and fatigue [28]. Therefore, since application of ADT in the management of COVID-19 is expected to be short term, ADT in COVID-19 will be relatively safe. Hence, we propose that ADT be considered as treatment option for severe COVID-19. Nevertheless, prospective clinical trials are warranted to confirm the efficacy of ADT for COVID-19.
Fig. 1.
Schematic diagrams illustrating role of androgen on SARS-CoV-2 invasion of host cells.
3.2. Estrogen and COVID-19 outcome in males and females
The sex hormone estrogen is reported to protect women against diseases including COVID-19 [29,30]. Estrogen is also found in men, however, unlike women, it is produced in much lower levels, not enough to exert its protective effect in men [31]. There are three major estrogen types: estrone (E1) estradiol (E2) and estriol (E3). E1 and E2 are biologically active in non-pregnant humans while the E3 is active mainly during pregnancy [32]. The E2 is produced primarily in ovaries and testes through aromatization of testosterone [33]. Moreover, low levels are produced by the adrenal glands and certain peripheral tissues [33]. The E1, on the other hand, is derived from peripheral aromatization of androstenedione [34]. More important, E2 and E1 can be converted into each other, and both can be inactivated via hydroxylation and conjugation [35]. The E2 demonstrates between 1.25–5 times the biological potency of type E1, and the circulating amount is 1.5–4 times more than E1 in premenopausal and non-pregnant women [36]. In effect, the level of E2 is higher than E1, hence the protective effect of E2 is always higher than E1. The level of E2 in men and postmenopausal women is much lower compared to premenopausal women [37]. This explains why premenopausal women including pregnant women are well protected against the severe COVID-19 compared to men of all ages and postmenopausal women. It is worth noting that younger men are better protected than older men because of the privilege of having an appreciable level of E1 which can also be converted to E2. The E1 is derived from peripheral aromatization of androstenedione [34]. As expected, androstenedione concentration is comparatively higher in younger males than older males, therefore the production of E1 and for that matter E2 is correspondingly higher in younger males. This may account for the seemingly better protection against severe COVID-19.
In Wuhan, China, researchers successfully investigated the supposed protective function of estrogen [38]. The study demonstrated that women with low estrogen levels tended to develop severe COVID-19 than women with high levels of the hormone on hospital admission. The supposed protective function of estrogen is linked to its ability to modulate immune responses, specifically, ensuring measured immune responses from activated immune cells. In effect, estrogen is capable of preventing over-reaction of activated immune cells to infections such as COVID-19. COVID-19 associated fatalities are largely due to cytokine storm that results from the over-reaction of activated effector cells, leading to unwarranted tissues damages, and organ system dysfunction [39,40]. Estrogen is reported to play crucial role in determining the number of immune cells that are produced and activated in a given period, and therefore influences the level of response to an infection [41]. The estrogen type E2 does this by inhibiting the expression of pro-inflammatory cytokines [42,43]. In menopausal women, the E1 takes over the immunomodulatory function of E2 [44]. However, because secretion level of E1 is lower than E2, it leads to a corresponding decrease in protection in menopausal women compared to premenopausal women. Notwithstanding, menopausal women are better protected against severe COVID-19 than their male counterparts. This is because older men produce a far lower level of E1 than their female counterparts. This could also explain why older women appear to have better COVID-19 recoveries than their male counterparts (Fig. 2).
The immunomodulatory activity of estrogen in infectious diseases has been confirmed in an experimental study on SARS in mice [45]. The study demonstrated that blocking estrogen increases the number of inflammatory cells in the animals’ lungs, increasing possible tissue damages and rendering them more susceptible to severe SARS infection and by extension COVID-19. This together with other immunomodulatory activities of the estrogen supports the assertion that estrogen has a protective effect against severe COVID-19. This further presents estrogen therapy as a potential treatment option for COVID-19, and therefore should be given serious consideration. Currently, studies are ongoing to ascertain the possibility of employing short-term estrogen treatment to protect COVID-19 patients from progressing to the severe form [46,47].
The protective function of estrogen is evident in pregnant women who are naturally endowed with high estrogen levels, specifically E3 [48]. In effect, pregnant women, who are originally deemed immunocompromised, rather tend to have mild COVID-19 when they contract the virus because of the high E3 levels, attesting to the immunomodulatory activity of estrogen [15]. Originally, high-level E3 is required to influence appropriate immune responses to prevent cytokine storm in order to preserve the foetus which is perceived by the expectant mother’s immune system as non-self. Therefore, pregnant women are expected to be better protected against severe COVID-19 because of abundant level of E3. Hence, post-menopausal women with low levels of estrogen (E1) and men of all ages with low levels of E1/E2 are expected to be more susceptible to severe COVID-19 than pre-menopausal women including pregnant women.
Estrogen has two forms of receptors (ERs) ERα and ERβ, which bind to the ligands E2 and E3 to initiate gene expression [49]. It is worth noting that B and T lymphocytes, mast cells, macrophages, dendritic cells, and NK cells express ERα. Moreover, hematopoietic progenitor cells are capable of expressing both ERα and ERβ [50]. More important, there are reports that, the receptor ERα is the most effective among the two, and it is the main regulator in hematopoietic progenitor differentiation [50,51]. The direct association of estrogen, especially E2, and estrogen receptor ERα, to the activation and progenitor differentiation of key immune cells suggests that the hormone is capable of modulating immune responses. Hence, females known for producing high-level estrogen will be better protected against infections including COVID-19 compared to their male counterparts. Estrogen has also demonstrated that it can elicit an anti-inflammatory response, especially in an infectious situation such as COVID-19 [52]. For instance, there are reports that estrogen receptor (ER) ERα46 is specifically involved in activating anti-inflammatory signaling pathways in macrophages and neutrophils through the expression of C-X-C motif chemokine ligand 8 (CXCL8) [53,54]. Generally, the ER activates inhibitory responses against the transcription of pro-inflammatory genes regulated by nuclear factor kappa B (NF-kB) [55]. Moreover, an activated ER can stimulate the expression of interferon-gamma (IFN-γ) in T cells in severe inflammatory conditions [56,57]. On the other hand, there are reports that E2 exhibits regulatory function on the activity of signal transducers and activator of transcriptions [58] by inducing the expression of cytokine-inhibitor proteins in macrophages and T cells [59,60]. Again, E2 has been reported to directly downregulate the expression of CCL2 in all leukocytes to reduce their migration to the infection site. Recruitment of leukocytes to the site of infection can result in severe inflammation and tissue damage [61,62]. Hence, estrogen's ability to induce anti-inflammatory responses is an indication that hyperestrogenemic females are better protected against severe inflammation than their male counterparts.
4. Immunological factors and COVID-19 outcome in males and females
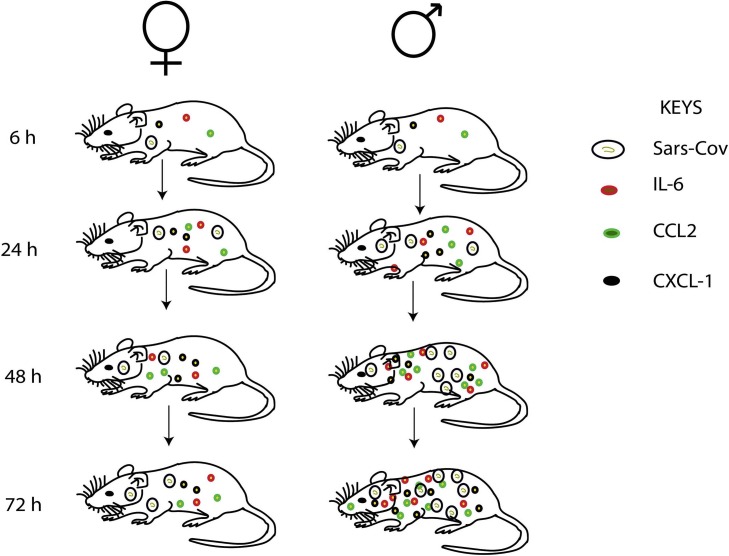
There are differences in the immune system of males and females, and these have a significant impact on the outcome of infectious diseases such as COVID-19 [63]. Females naturally have stronger immune defence and therefore are more protected from infectious diseases than their male counterparts [64]. One major factor that contributes to the differences in sex-related immune defences is the vulnerability in the sex chromosome constitution. Interestingly, some critical immune-related genes are located on the X chromosome [65]. Therefore, the immune-related proteins expressed by these genes in females are twice as those expressed by their male counterparts. Although in females, one of the X chromosomes in each cell is inactivated to balance this inequality among the sexes, genes such as Toll-like receptor 7 (TLR7) in certain immune cells are somehow able to escape this inactivation [66]. In effect, women tend to produce more of these defensive immune-related proteins and therefore are better protected against infection. Thus, the biallelic expression of such genes leads to higher immune responses and increases the resistance to infections. TLR7 has been linked to immune response against viral infections and therefore important against the severe COVID-19 [67]. Thus, TLR7 is over-expressed in innate immune cells that recognize single-strand RNA viruses which SAR-CoV-2 is a notable example [68]. These immune cells that express TLR7 include dendritic cells, circulating monocyte, macrophages and B cells [69]. And like all single-strand RNA viruses, these immune cells can recognize COVID-19. In an experimental study with male and female mice on sex-based differences in SARS-CoV infection, the scientists reported nearly identical total viral loads in both sexes at early (16 h) stages of the study [70]. However, there was a significant increase in the viral loads (2–3 folds) in the male mice compared to females after day 1 of inoculation (Fig. 3 ). Moreover, extensive hyperemia and congestion were noticed in male mice as against a nearly normal in females at day 4 [70]. This outcome corroborates the long-held assertion that COVID-19 infection in males is likely to progress to severe form compared to infected females. Furthermore, the interaction between TLR7-expressing immune cells and a single strand RNA virus can induce production of antibodies and pro-inflammatory cytokines such as IL-6 against the virus [67]. Hence, women are better protected against viral infections and for that matter the severe COVID-19 due to the over-expression of TLR7 in females compared to their male counterparts. It is quite intriguing that, the level of pro-inflammatory IL-6 post-viral infection is relatively lower in women than in their male counterparts, and hence prevents prolong inflammation, and therefore promotes good health and longevity in women [71]. An experimental study that assessed the levels and effects of inflammatory cytokines and chemokines in SARS-CoV infected male and female mice showed equal levels of the proinflammatory cytokine (IL-6) and chemokines (CCL2 and CXCL-1) in both sexes at the early stage of infection, but upregulated significantly in male mice compared to females at day 3 [70]. This outcome corroborates the assertion that there is an endogenous factor (estrogen) in females that modulates cytokine and chemokine production to prevent possible cytokine storm and prolong inflammation. The study also reported significant infiltration of neutrophils into lung of male mice right from day 1 of inoculation, and increased by 5 folds at day 4 compared to their female counterparts [70]. It can therefore be inferred that the susceptibility of male mice to SARS-CoV-2 infection correlates with increasing inflammatory cell recruitment. This explains why prolong inflammation is very common in males infected with SARS-CoV-2 compared to females, and could be one of the factors that promote severe COVID-19 in men.
Fig. 3.
Schematic diagrams illustrating differences in immune response to SARS-CoV-2 infection in male and female mice.
The over-expression of TLR7 observed in the female gender can also help to enhance the surveillance activity of the innate immune system by activating other related immune responses against the infection. Thus,the activated immune cells expressing TLR7 release interferon which further activates or recruits other relevant immune cells to enhance immune surviellance [72,73]. The interferon, therefore, helps to sustain the immune activity long enough to rid the body of the viral infection. It has been proven scientifically that interferon production is relatively higher in females compared to their male counterparts [74,75]. This could be one of the reasons why females are better protected against viral infections and for that matter severe COVID-19. Additionally, the X chromosome houses genes that code for the following proteins TLR8, CD40 L and CXCR3 over-expressed in women [76]. These proteins are reported to be involved in immune survielance against viral infection [[77], [78], [79]].
The adaptive immune system just like the innate gives better protection to females than males. Specifically, there are reports that SARS-CoV-2 activates B lymphocytes and plasma cells to produce specific neutralizing IgG [80]. The anti-SARS-CoV-2 binds and marks the SARS-CoV-2 infected cell for destruction. The virus infected cell-antibody complex is recognized by the FcgRIIIA receptor (CD16) of the effector killer cell to enhance possible phagocytosis, lysing and cell killing (ADCC) [81,82]. This activity also leads to further stimulation of innate cells and subsequent production of pro-inflammatory cytokines. The quantity and quality of antibodies produced against viral infections tend to be more in females compared to their male counterparts [83]. In an experimental study aimed at investigating the avidity of antibodies produced by influenza A virus-infected mice (male and female), the female mice compared to males, produced quality antibodies with high avidity that were able to stop reinfection and replication by preventing the virus from entering target cells. More important, antibody avidity of the females improved with increasing estrogen levels [83]. Hence, it can be hypothesized that females will be better protected against subsequent infections from the same virus after the initial exposure to the virus compared to males. The outcome of this study implies that vaccination against viral infections will be more effective in females than their male counterparts because of the difference in the quality of antibodies produced by the sexes [78]. In another study, cross-reacting antibody titer differences between the sexes were assessed by analysing antibody responses against heterosubtypic virus [84]. The study demonstrated that female mice immunized against H3N2 produced higher cross-reacting antibodies against H1N1 and vice versa than the male mice [83,84]. These suggest that females are better protected against heterologous virus challenge than males. The data support the assertion that, females are better protected by the adaptive immune system against viral infections. Therefore, females may have better immunological protection against COVID-19 from both innate and adaptive arms of the immune system, compared to their male counterparts. In effect, it will be appropriate to assert that, females recovered from COVID-19 may be the best candidates to donate plasma for the proposed convalescence therapy for COVID-19. This is because females produce relatively high-quality antibodies compared to males. Moreover, per the data from these experimental studies discussed in this review, it can be postulated that a COVID-19 vaccine may give long time protection to those who will be vaccinated, especially women. Nevertheless, individuals react differently to vaccination, hence, the duration of the vaccine protection will be subject specific.
5. Angiotensin-converting Enzyme-2 (ACE2) system and COVID-19 outcome in males and females
The gene ACE2 encodes the angiotensin-converting enzyme-2 (ACE2), the receptor for human pathogenic coronaviruses including severe acute respiratory syndrome coronavirus (SARS-CoV), HCoV-NL63, and SARS-CoV-2 [3,85]. The gene is found on the X chromosome, specifically on Xp22, and made up of approximately 18 exons and 20 introns [29]. The enzyme ACE2, a member of the renin-angiotensin system (RAS), makes it possible for these viruses to enter the target cells and ultimately produce infection [86]. ACE2 is expressed mainly on the epithelial cells of the lung, intestine, kidney, blood vessels, testes, and on the mucosa of the oral cavity [18]. This is consistent with the potential routes of viral transmission of SARS-CoV-2, as both respiratory and gastrointestinal systems share interfaces with the external environment. This may explain why high incidence of pneumonia and bronchitis, kidney and heart complications is very common in patients confirmed to have contracted the COVID-19. The SARS-CoV-2 virus, just like SARS-CoV virus has a spike (S) protein that interacts with ACE2 to access the target cells [21]. More important, ACE2, a homologue of angiotensin-converting enzyme (ACE) is also involved in the regulation of blood pressure and electrolyte homeostasis [87]. It exerts this biochemical activity by cleaving angiotensinogen II to angiotensin [88]. The resultant angiotensin then binds to the Mas receptor to induce vasodilation, anti-inflammatory, and anti-fibrotic effects in the organism [89]. Moreover, angiotensinogen, produced in the liver is cleaved by renin to form angiotensin [90]. On the contrary, the enzyme ACE is noted for its involvement in the conversion of the angiotensin I to angiotensin II [91]. Consequently, the resultant angiotensin II induces vasoconstriction, renal sodium reabsorption and potassium excretion, and aldosterone synthesis leading to blood pressure elevation, and induction of inflammatory and pro-fibrotic signal pathways [92]. Hence, the effect of renin-angiotensin-aldosterone system activation depends on the ACE/ACE2 balance, and ultimately, the balance between pro-inflammatory and pro-fibrotic, and anti-inflammatory and anti-fibrotic signal pathways [93]. It should be appreciated that ACE2 exhibits arguably two unrelated activities in the initiation and progression of COVID-19. The first is the membrane-bound ACE2 protein which serves as a binding site for the virus to access target cells [94]. In effect, the general perception is that, there is a direct correlation between the incidence of COVID-19 and the expression of ACE2 gene. The second is the well-investigated and confirmed protective role of soluble ACE2 against disease progression and development of severe infection [95], largely due to the role it plays in keeping the blood pressure normal. Additionally, the soluble ACE2 is reported to have antiviral activity [96], hence could potentially offer protection against COVID-19 infection. These two described ACE2 types obviously exhibit two contradictory fortunes. However, the link is that, the soluble ACE2 is formed from the membrane-bound ACE2.
5.1. Protective function of soluble ACE2 on severe COVID-19 in males and females
The membrane-bound ACE2 is shed from the cells by the metalloproteases A Disintegrin and metalloproteinase domain-containing protein 10 (ADAM10) and A Disintegrin and metalloproteinase domain-containing protein 17 (ADAM17) into circulation, forming the soluble ACE2 [97]. A number of studies have confirmed the assertion that the soluble ACE2 offers protection against diseases especially infections [98,99]. For instance, in a study, a circulating ACE2 protein was confirmed to have protection against influenza A (H7N9) virus-induced acute lung injuries, and that patients who were protected from the severe form of the influenza A (H7N9) had relatively higher serum ACE2 levels [100]. Additionally, in an experimental study with ACE2 gene knockout influenza A (H5N1) infected mice, the mice initially suffered severe lung damage [101], however, the condition improved when the mice were treated with human ACE2 enzyme. These studies further corroborate the assertion that soluble ACE2 protein has protective action against infectious diseases including those of viral origin. Therefore, presents soluble ACE2 enzyme as potential therapeutic option for the treatment of COVID-19. In another study, a recombinant human ACE2 enzyme proved potent by showing haemodynamic benefits in pulmonary arterial hypertension in both preclinical and clinical trials [102]. More of such recombinant constructions and expressions should be encouraged, because these protein based therapies have minimal toxicity and are functionally specific.
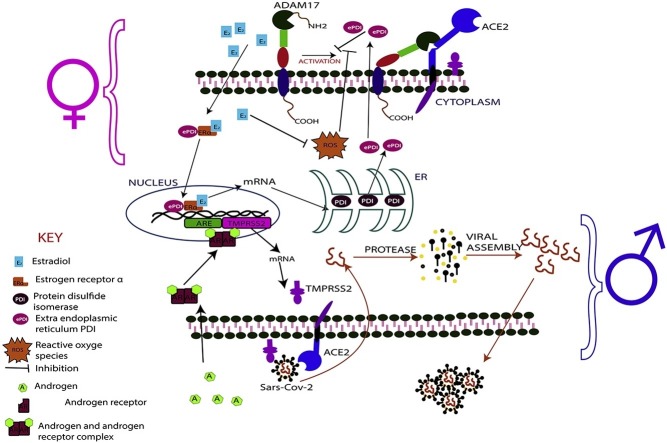
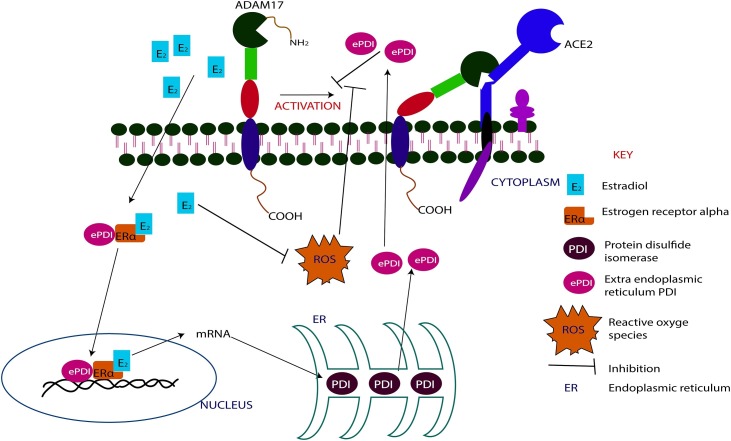
Differences in the level of soluble ACE2 have been reported among the sexes in studies elsewhere [103,104], with these studies reporting higher ACE2 levels in the serum of men compared to women [18,95]. Additionally, there are reports of high ACE2 levels in older women compared to younger women, and in older men compared to young men [95,105]. There has been a corresponding reduction in membrane-bound ACE2 in men and the elderly in most tissues [106]. This high level of soluble ACE2 in the serum, could possibly be as a results of an upregulation of metalloproteinase activity of ADAM 10/17. This biochemical activity of ADAM 10/17 creates a balance between the membrane-bound and soluble forms of the ACE2 at different fractions in the elderly and the young, and in males and females. It is worth noting that, there is a positive correlation between the levels of both estrogen and androgen, and the expression of ADAM 10/17 [107,108]. Thus, high levels of these sex hormones induce a corresponding expression of ADAM 10/17. Therefore, it can be inferred that, ADAM 10/17 is overexpressed in both sexes, and therefore should have resulted in equal and high levels of soluble ACE2 in both sexes. Nevertheless, while men have high soluble ACE2 in their serum, women have relatively lower levels of same. The disproportionate downstream activity of ADAM 10/17 in women may be explained by the over-expression of extra endoplasmic reticulum protein disulfide isomerase (ePDI) [109].
5.2. The effect of ePDI on COVID-19 outcome in males and females
Over-expressed extra endoplasmic reticulum protein disulfide isomerase is reported to inhibit the metalloproteinase activity of ADAM 10/17 by preventing the activation of the inactive form of ADAM17 [110]. To inhibit the activation of the inactive ADAM17, the ePDI first binds to ERα to form ePDI-ERα complex [110]. Subsequently, the ePDI- ERα complex binds to E2. The ePDI- ERα-E2 complex then serves as a promotor for the over-transcription of the mRNA of PDI, leading to over-expression of PDI proteins which are subsequently released from the endoplasmic reticulum into the cytoplasm as ePDI [111,112] (Fig. 4 ). These mechanisms confirm the direct influence of estrogen in the metalloproteinase activity of ADAM10/17. Thus, estrogen down-regulates the activity of ADAM10/17. Moreover, it is important to note that, high concentration of reactive oxygen species (ROS) inhibit the regulatory function of ePDI protein. [113]. However, again, E2 has been shown to inhibit the generation and activity of ROS [114,115]. Hence, the regulatory activity of ePDI is maintained as long as E2 level is high. Therefore, it will be appropriate to postulate that the E2 down-regulates the metalloproteinase activity of ADAM10/17 through the over-expression of ePDI and down-regulation of ROS activity. This further confirms the anti-inflammatory activity of estrogen, since ROS is a known promotor of prolong inflammation.
Fig. 4.
Schematic diagrams illustrating the inhibitory effects of estrogen on the metalloproteinase activity of ADAM17.
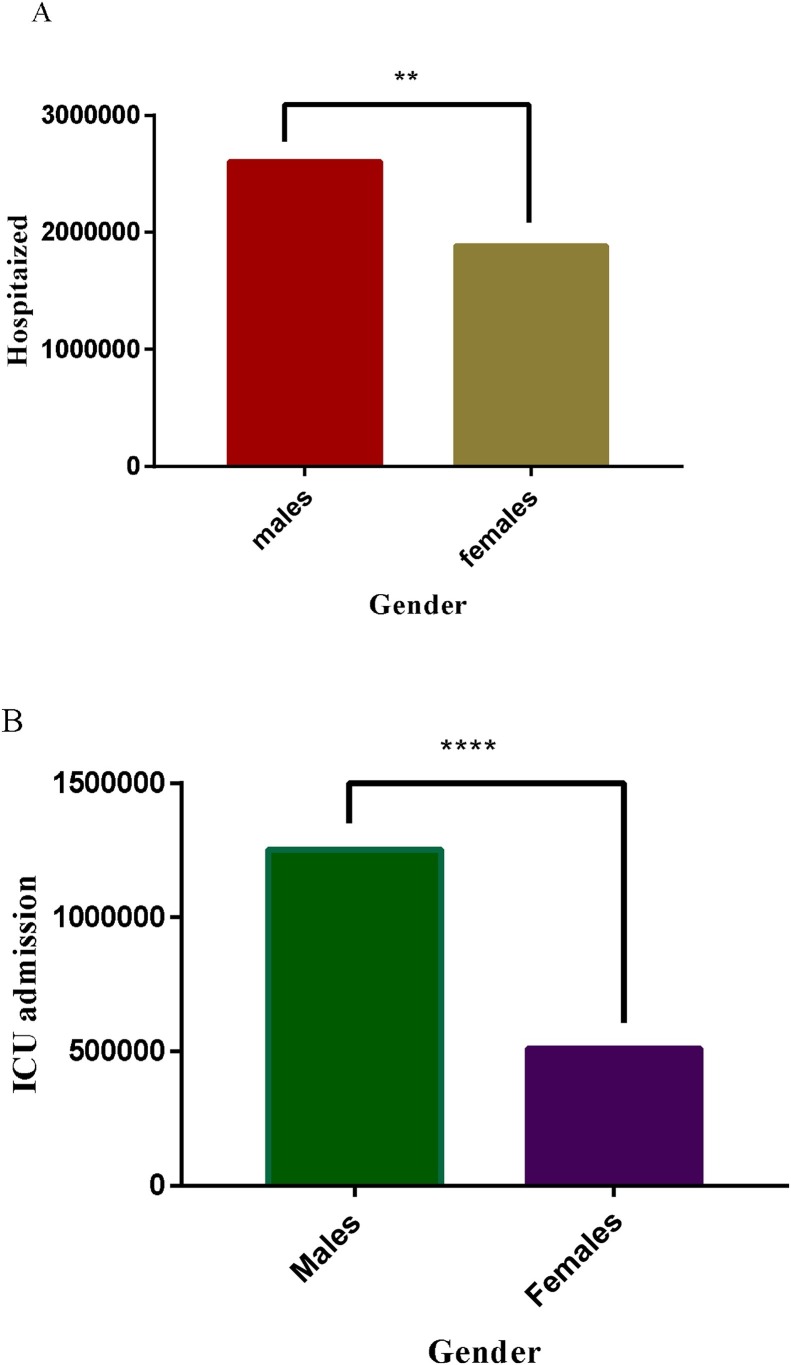
As demonstrated in Fig. 5 A (analyses of COVID-19 information retrived from websites of WHO and WHO accredited organization as of 3rd July 2020) [[10], [11], [12], [13]], the incidence of COVID-19 was significantly higher in men compared to women (p < 0.001). However, in an ideal situation, considering the biology underpinning the interaction between the virus and the host, females are supposed to have aboundant membrane-bound ACE2 due to the inhibition of the metalloproteinase activity of ADAM10/17, and hence should have rather recorded higher COVID-19 incidence than males [116,117]. Therefore, it will be appropriate to postulate that, the percentage representation between these two ACE2 forms at a particular period determines the ultimate role of ACE2 in an infectious situation. Again, it can be suggested that, the soluble ACE2 is a component in a complex regulatory system, and therefore its protective impact is dependent on other regulators such as hormonal and immunological factors. Hence, the impact of the differences in ACE2 levels among sexes and age groups is not absolute, but dependent on other functional related factors. This effect may be altered as the disease progresses and the immune system is fully activated. In effect, severe COVID-19 which is common in men compared to women may not be entirely dependent on the differences in soluble ACE2 level. For instance, soluble ACE2 is known to offer better protection against viral infections [98], hence the expectation was that men and the elderly who are known to have high soluble ACE2 levels should have had better protection against COVID-19 than women and youngsters, but the reverse is rather the case. This presupposes that there are underlying factors that modulate the activity of soluble ACE2 and other enzymes that are involved in the possible outcome of infectious diseases including COVID-19. These modulating factors include sex hormones reported to play an important role in the differences in disease outcomes observed among the sexes.
Fig. 5.
Sex related risk of severe COVID-19. **p = 0.01, ****p = 0.0001.
The hormone androgen is reported to increase plasma renin and its activity leading to transcription of angiotensinogen messenger RNA, and subsequent translation of angiotensinogen protein. This biochemical processes ultimately result in vasoconstriction and its attendant complications in severe COVID-19. The consequence of this is the predisposition of males to the severe form of the infection [118]. Thus, androgen possibly counteracts the supposed protective action of high levels of soluble ACE2. In effect, the protective role of the soluble ACE2 in males is attenuated. Estrogen, on the other hand, decreases plasma renin activity, angiotensin I receptor expression and the expression of angiotensin-converting enzyme 1, and therefore promotes vasodilation [119]. These biochemical processes help to prevent the complications associated with vasoconstriction and development of severe COVID-19 [119]. Further confirming the physiological protection against severe COVID-19 in women.
In a recent clinical trial, ACE which is functionally antagonistic to soluble ACE2 and for that matter estrogen was inhibited with ACE-inhibitor. Interestingly, the ACE-inhibitor protected the study subjects against severe COVID-19, corroborating the assertion postulated above [120]. As the world is still searching for a definitive therapy for Covid-19, ACE-inhibitors should also be considered, especially, for the treatment of males at the Intensive Care Unit (ICU). These therapeutics which are protein-based agents are largely safe. This is because protein-based agents are specific in function. Hence, present with relatively better toxicity profile [120]. Again, the application of this proposed therapy on COVID-19 will be short term. Therefore, the possibility of side effects will be minimized.
6. Other factors implicated in differential COVID-19 outcomes in males and females
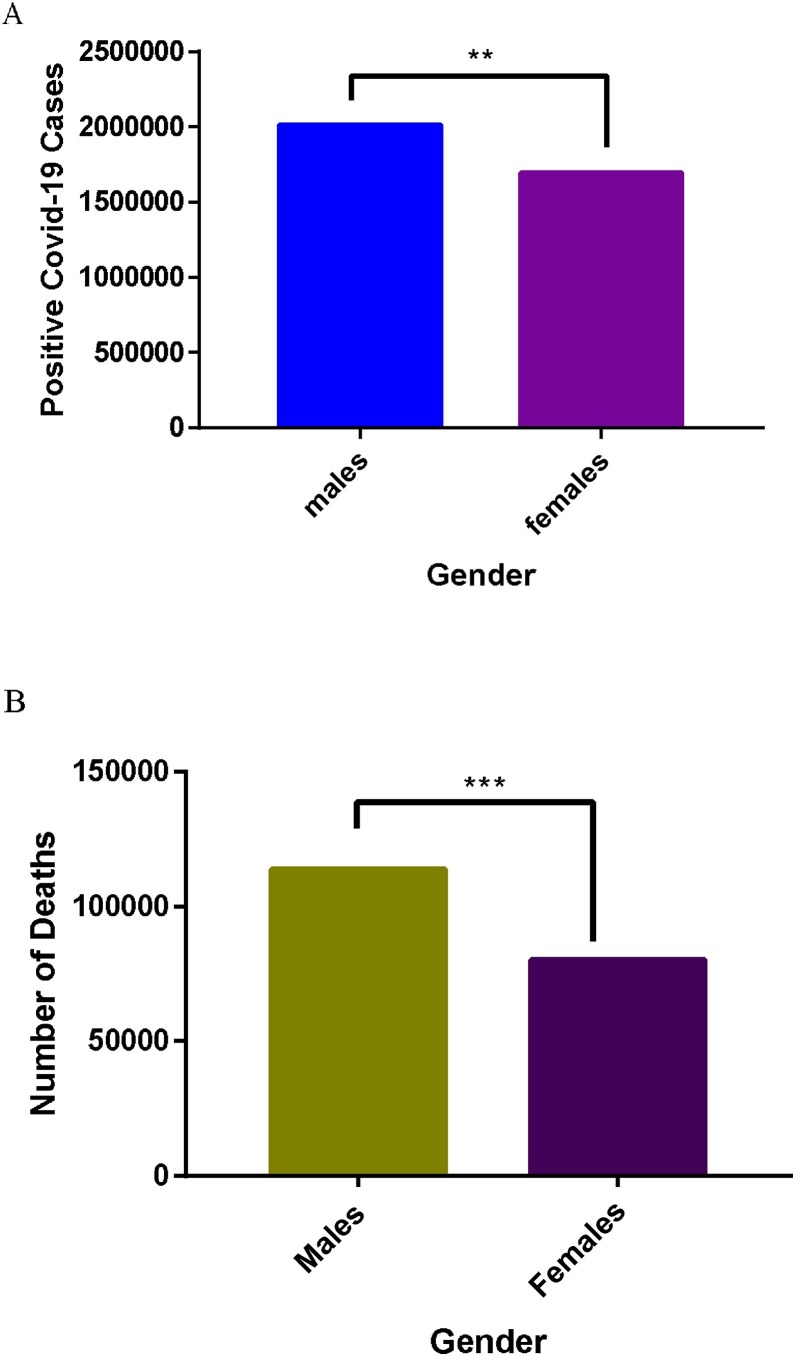
There are other factors such as personal hygiene, type of occupation, social activities, etc that contribute to high infection incidence. These factors may not be endogenous but are capable of exposing an individual to the causative agent to produce endogenous changes. For instance, females are more likely to pay particular attention to personal hygiene compared to males. In effect, women are more likely to adhere to the COVID-19 preventive protocols than males. Hence, women are able to cut down COVID-19 cases and for that matter incidence by adopting more hygienic lifestyle. Again, men are more likely than women to be involved in socialization activities such as games, sports, night clubbing etc. These activities by their nature expose patrons to the virus. Therefore, men who are in the majority at these programme, are at greater risk of contracting the virus, hence, the high incidence in men compared to women. Another important factor that influences bias COVID-19 incidence among males and females is the kind of jobs common to them. Women are more likely to be involved in office work such as executive assistant, account manager, administrative assistant, receptionist etc, and can conveniently work from home where necessary. These category of jobs scarcely expose workers to large group of people, hence, minimize the possibility of contracting the virus. On the contrary, men are mostly engaged in jobs such security officers, taxi drivers, bus and coach drvers, postal workers and courriers, cleaners, delivery drivers, sanitation officers etc. These jobs have high tendency of exposing the workers to the virus, due to the nature of these jobs and the possibility of being exposed to many people in the couse of doing their job. These factors may be part of the reasons for the high incidence of COVID-19 in males compared to their female counterparts, as illustrated in Fig. 2 (analyses of COVID-19 information retrived from websites of WHO and WHO accredited organization as of 3rd July 2020) [[10], [11], [12], [13]]. Thus, according to Fig. 2, the highest incidence of COVID-19 was found among the age group 30–39, followed by age groups 40–49 and 50–59 respectively. Interestingly, these groups form a major part of the workforce or working class in most countries. Moreover, it is worth noting that, they are more likely to be involved in social activities, because, they are the most active population, hence, the possible reason for the high incidence of COVID-19. More important, incidence of COVID-19 among the men in these groups (30–39, 40–49 and 50–59) was higher than their female counterparts. Taken all together, these data further confirm the reasons for the high COVID-19 incidence in men compared to women.
The data in Fig. 5 (analyses of COVID-19 information retrived from websites of WHO and WHO accredited organizations as of 3rd July 2020) [[10], [11], [12], [13]], showed that men hospitalized were significantly higher (p < 0.001) than their females counterparts (Fig. 5A). Given the foregoing discussion, COVID-19 patients hospitalized are likely to suffer the severe form of the infection. Hence, the male could be considered a risk factor for severe COVID-19. The data from intensive care unit (ICU) presented in Fig. 5B were the critical condition cases. As anticipated, men in critical condition were significantly higher (p < 0.0001) than their female counterparts. In effect, men are more likely to progress from severe to critical COVID-19 than their females counterparts. Furthermore, in Fig. 6B (analyses of COVID-19 information retrived from websites of WHO and WHO accredited organization as of 3rd July 2020) [[10], [11], [12], [13]] is presented data for COVID-19 deaths among males and females. As expected, COVID-19 deaths in males were significantly higher compared to their female counterparts. Inferentially, males are more likely to die from ICU admissions than females. Per the foregoing data, it is evident that males are more vulnerable to COVID-19, hence, it will be appropriate to offer commensurate care and protection to men in the fight against COVID-19, in order to reduce the nimber of fatalities.
Fig. 6.
Sex disaggregated case fatality rate of COVID-19. **p = 0.01, *** p = 0.001.
7. Correlation between smoking and COVID-19 incidence in males and females
There is a widely held assertion that smoking increases the risk of most respiratory tract infections [121]. For instance, smokers are reported to be 5-times more likely to contract influenza than non-smokers [122]. Similarly, the risk of bacterial pneumonia is 3 to 5-times higher in smokers [122]. This could be attributed to impairment in immune function due to smoking [123]. There are few reports about smoking and the associated risk with coronavirus outbreaks including Middle Eastern respiratory syndrome coronavirus (MERS) and severe respiratory syndrome coronavirus (SARS) [122]. For instance, there was a reported increased risk of mortality in smokers with MERS compared to non-smokers with MERS in a study in Korea [122]. Similarly, a review on the risk of COVID-19 and smoking, where eight studies were reviewed, reported a correlation between smoking and severe COVID-19 [121]. Hence, there is a possible association between smoking and severe COVID-19. Smoking is reported to upregulate the expression of androgen, a phenomenon associated with severe COVID-19 [124]. A study titled ‘Cigarette Smoking, Androgen Levels, and Hot Flushes in Midlife Women’, reported significantly higher androstenedione levels and a higher androgen to estrogen ratio in current smokers than never smokers [125]. A similar account has also been reported by another study on cigarette smoking and endogenous sex hormones [126]. It is worth noting that, ACE2 and TMPRSS2 known to facilitate the attachment and entry of SARS-CoV-2 into target cells are upregulated in smoking samples from both lung and oral epithelial tissue [127]. This is an indication that smoking could predispose an individual to COVID-19. Therefore, smoking should be considered a risk factor for COVID-19. More important, men smokers are more susceptible to COVID-19 than women smokers, attributable to the hormonal differences between the sexes. Thus, the TMPRSS2 which is upregulated in smokers is involved in the upregulation of androgen. Unfortunately, smoking is rather common among men than women, worsening their plight. This may partly be the reason for the high number of severe cases of COVID-19 in men compared to women.
8. Conclusion
COVID-19 is a complex and multifactorial disease that affects virtually all the organ systems in the body. The SARS-CoV-2 infection promotes chronic systemic inflammation due to the release of pro-inflammatory cytokines and prolong activation of the innate immune system. In effect, may lead to organ system dysfunction, implicated in most COVID-19 deaths, as revealed by autopsies on these bodies. This therefore puts individuals with pre-existing comorbidities at higher risk. These attributes of COVID-19 present a major public health challenge that requires decisive measures to manage. Managing the resultant chronic inflammatory responses may be as imperative as targeting the SARS-CoV-2 in the treatment of COVID-19. Hence, a definitive therapy should aim at preventing the virus from entering target cells, and modulating the resultant immune responses to prevent chronic inflammation, a common feature of severe COVID-19. The already existing antiviral agents can be screened for possible efficacy against COVID-19, as other studies focus on finding novel definitive therapies. Modulating immune response is very crucial in the management of COVID-19. This is because most COVID-19 associated complications are attributable to chronic inflammation. Thus, accounts for the COVID-19 associated organ system dysfunction. Hence, clinical screening of the existing immunomodulatory agents should be part of the processes employed to discover definitive therapy for COVID-19. In our opinion, the use of immunomodulatory drugs will be efficient in the management of COVID-19. Hence, until a definitive therapy is found, we propose that the use of immunomodulatory agents should be part of any therapeutic regimen of COVID-19. Nevertheless, this therapeutic approach should be individually tailored. Thus, the health status, age and gender of the patient should be considered in the application of this therapeuty to derive the expected outcome.
The gender disparity observed in COVID-19 vulnerability is as a result of the differences in the immune responses. The disparity in immune response among the sexes is attributable to their hormonal differences. Thus, estrogen produced at functional levels in women is known to inhibit overproduction of pro-inflammatory cytokines, hence, capable of preventing cytokine storm, implicated in severe COVID-19. On the contrary, androgen promotes the production of pro-inflammatory cytokines that promote cytokine storm, leading to physiological changes conducive for progression from mild to severe COVID-19. Therefore, androgen deprivation therapy (ADT) and estrogen-based therapy are recommended as therapy options to specifically modulate the immune response to ameliorate chronic inflammation, especially in men. Although, a number of side effects have been reported about these hormonal therapies, especially when used for a long term, they will be safe for COVID-19 therapy, because, the treatment duration is relatively short.
The enzyme ACE has been implicated in blood pressure elevation, and induction of inflammatory and pro-fibrotic signal pathways, characteristics of severe COVID-19. Therefore, therapy type that selectively inhibits the activity of ACE may facilitate faster COVID-19 recovery. Hence, recombinant ACE-inhibitors are also recommended for the treatment of severe COVID-19. Notwithstanding, the treatment should be short term to prevent side effects related to ACE functional impairment.
Discovery of an efficacious vaccine for COVID-19 will be a giant step in the fight against this pandemic. More important, data from pre-clinical and clinical trials at different stages have demonstrated great potential in the quest to discover a vaccine for COVID-19. However, just like most vaccines developed against respiratory infections of viral origin, COVID-19 vaccine may not provide long term or lifelong protection against the virus as anticipated. Hence, equal attention and resources should be devoted to finding definitive therapy for COVID-19.
Finally, there should be strong educational campaign by civil society organizations and governments against smoking. This is because, there is direct correlation between smoking and COVID-19 incidence, backed by science. Unfortunately, it is particularly worse in men smokers than women smokers.
Declaration of Competing Interest
The authors report no declarations of interest.
Author Contribution
DOA conceived, designed and wrote the manuscrip. DOA, IKB, EA did reliterature search. DOA, IKB designed and constructed the illustrative figures. DOA, IKB, AB, EA analyzed and interpreted the data. AB, SO, MTM read and edited the final manuscript. All authors read and approved the final version of the manuscript.
Acknowledgements
We appreciated the valuable contributions of Prof Zhang Juan and Prof Wang Ming of Antibody Engineering Laboratory, School of LifeScience Technology, ChinaPharmaceutical University, Nanjing, China
References
- 1.Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., Wang B., Xiang H., Cheng Z., Xiong Y. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. Jama. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Meng Y., Wu P., Lu W., Liu K., Ma K., Huang L., Cai J., Zhang H., Qin Y., Sun H. Sex-specific clinical characteristics and prognosis of coronavirus disease-19 infection in Wuhan, China: A retrospective study of 168 severe patients. PLoS Pathog. 2020;16(4) doi: 10.1371/journal.ppat.1008520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berenguer J., Ryan P., odríguez-Baño J., Jarrín I., Carratalà J., Pachón J., Yllescas M., Arribas J.R., Group C.-S.S. Characteristics and predictors of death among 4,035 consecutively hospitalized patients with COVID-19 in Spain. Clin. Microbiol. Infect. 2020 doi: 10.1016/j.cmi.2020.07.024. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kocher K.M., Délot-Vilain A., LoTempio J., Délot E.C. Paucity and disparity of publicly available sex-disaggregated data for the COVID-19 epidemic hamper evidence-based decision-making. medRxiv. 2020 doi: 10.1101/2020.04.29.20083707. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Richardson S., Hirsch J.S., Narasimhan M., Crawford J.M., McGinn T., Davidson K.W., Barnaby D.P., Becker L.B., Chelico J.D., Cohen S.L. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. Jama. 2020;323(20):1989–2104. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.W.H. Organization . 2020. Coronavirus Disease 2019 (COVID-19): Situation Report, 72. [Google Scholar]
- 7.Lawton G. Elsevier; 2020. Men Hit Harder by covid-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bwire G.M. Coronavirus: Why Men are More Vulnerable to Covid-19 Than Women? Sn Comprehensive Clinical Medicine. 2020:1. doi: 10.1007/s42399-020-00341-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bourgonje A.R., Abdulle A.E., Timens W., Hillebrands J.L., Navis G.J., Gordijn S.J., Bolling M.C., Dijkstra G., Voors A.A., Osterhaus A.D. Angiotensin‐converting enzyme‐2 (ACE2), SARS‐CoV‐2 and pathophysiology of coronavirus disease 2019 (COVID‐19) J. Pathol. 2020;251(13):228–248. doi: 10.1002/path.5471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Health Organization . 2020. WHO Corona Virus Disease (Covid-19): the Global Situation. [Google Scholar]
- 11.Global health 5050 . 2020. Tracking Differences in COVID-19 Illness and Death Among Women and Men. [Google Scholar]
- 12.John Hopkins University & Medicine . 2020. How Is the Outbreak Growing? [Google Scholar]
- 13.United Nations Women Organization . 2020. Covid-19: Emerging Gender Data and Why It Matters. [Google Scholar]
- 14.Jin J.-M., Bai P., He W., Wu F., Liu X.-F., Han D.-M., Liu S., Yang J.-K. Gender differences in patients with COVID-19: focus on severity and mortality. Front. Public Health. 2020;8:152. doi: 10.3389/fpubh.2020.00152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sharma G., Volgman A.S., Michos E.D. Sex Differences in Mortality from COVID-19 Pandemic: Are Men Vulnerable and Women Protected? JACC: Case Reports. 2020;2(9):1407–1410. doi: 10.1016/j.jaccas.2020.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Suba Z. Prevention and therapy of COVID-19 via exogenous estrogen treatment for both male and female patients. J. Pharm. Pharm. Sci. 2020;23:75–85. doi: 10.18433/jpps31069. [DOI] [PubMed] [Google Scholar]
- 17.Shen M., Shi H. Sex hormones and their receptors regulate liver energy homeostasis. Int. J. Endocrinol. 2015;2015 doi: 10.1155/2015/294278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ciaglia E., Vecchione C., Puca A.A. COVID-19 infection and circulating ACE2 levels: protective role in women and children. Front. Pediatr. 2020;8:206. doi: 10.3389/fped.2020.00206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Montopoli M., Zumerle S., Vettor R., Rugge M., Zorzi M., Catapano C.V., Carbone G., Cavalli A., Pagano F., Ragazzi E. Androgen-deprivation therapies for prostate cancer and risk of infection by SARS-CoV-2: a population-based study (n= 4532) Ann. Oncol. 2020;31(8):1040–1045. doi: 10.1016/j.annonc.2020.04.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sharifi N., Ryan C.J. Androgen hazards with COVID-19. Endocr. Relat. Cancer. 2020;27(6):E1–E3. doi: 10.1530/ERC-20-0133. [DOI] [PubMed] [Google Scholar]
- 21.Walls A.C., Park Y.-J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;281(2) doi: 10.1016/j.cell.2020.02.058. 1281-292.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li F. Structure, function, and evolution of coronavirus spike proteins. Annu. Rev. Virol. 2016;3:237–261. doi: 10.1146/annurev-virology-110615-042301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Iwata-Yoshikawa N., Okamura T., Shimizu Y., Hasegawa H., Takeda M., Nagata N.J.Jov. 2019. TMPRSS2 Contributes to Virus Spread and Immunopathology in the Airways of Murine Models After Coronavirus Infection. 93(6) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhou F., Gao S., Han D., Han W., Chen S., Patalano S., Macoska J.A., He H.H., Cai C. TMPRSS2-ERG activates NO-cGMP signaling in prostate cancer cells. Oncogene. 2019;38(22):4397–4411. doi: 10.1038/s41388-019-0730-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zoni E., Karkampouna S., Thalmann G.N., Kruithof-de Julio M., Spahn M. Emerging aspects of microRNA interaction with TMPRSS2-ERG and endocrine therapy. Mol. Cell. Endocrinol. 2018;462:9–16. doi: 10.1016/j.mce.2017.02.009. [DOI] [PubMed] [Google Scholar]
- 26.Ahmadi H., Daneshmand S. Androgen deprivation therapy: evidence‐based management of side effects. BJU Int. 2013;111(4):543–548. doi: 10.1111/j.1464-410X.2012.11774.x. [DOI] [PubMed] [Google Scholar]
- 27.Nguyen P.L., Alibhai S.M., Basaria S., D’Amico A.V., Kantoff P.W., Keating N.L., Penson D.F., Rosario D.J., Tombal B., Smith M.R. Adverse effects of androgen deprivation therapy and strategies to mitigate them. Eur. Urol. 2015;67(5):825–836. doi: 10.1016/j.eururo.2014.07.010. [DOI] [PubMed] [Google Scholar]
- 28.Moyad M.A., Newton R.U., Tunn U.W., Gruca D. Integrating diet and exercise into care of prostate cancer patients on androgen deprivation therapy. Res. Rep. Urol. 2016;8:133. doi: 10.2147/RRU.S107852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gemmati D., Bramanti B., Serino M.L., Secchiero P., Zauli G., Tisato V. COVID-19 and individual genetic Susceptibility/Receptivity: role of ACE1/ACE2 genes, immunity, inflammation and coagulation. Might the double X-Chromosome in females Be protective against SARS-CoV-2 compared to the single X-Chromosome in males? Int. J. Mol. Sci. 2020;21(10):3474. doi: 10.3390/ijms21103474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alkhouli M., Nanjundappa A., Annie F., Bates M.C., Bhatt D.L. Mayo Clinic Proceedings. Elsevier; 2020. Sex differences in COVID-19 case fatality rate: insights from a multinational registry. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Eriksson A.L., Perry J.R., Coviello A.D., Delgado G.E., Ferrucci L., Hoffman A.R., Huhtaniemi I.T., Ikram M.A., Karlsson M.K., Kleber M.E. Genetic determinants of circulating estrogen levels and evidence of a causal effect of estradiol on bone density in men. J. Clin. Endocrinol. Metab. 2018;103(3):991–1004. doi: 10.1210/jc.2017-02060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Steinman K.J., Robeck T.R., O’Brien J.K. Characterization of estrogens, testosterone, and cortisol in normal bottlenose dolphin (Tursiops truncatus) pregnancy. Gen. Comp. Endocrinol. 2016;226:102–112. doi: 10.1016/j.ygcen.2015.12.019. [DOI] [PubMed] [Google Scholar]
- 33.Siegfried K.R., Draper B.W. Elsevier; 2020. The Reproductive System, The Zebrafish in Biomedical Research; pp. 151–164. [Google Scholar]
- 34.Colao A., Di Somma C., Zhukouskaya V.V. Springer; 2018. Skeletal Tissue and Ovarian Function: Puberty and Menopause, Multidisciplinary Approach to Osteoporosis; pp. 129–142. [Google Scholar]
- 35.Pemp D., Kleider C., Schmalbach K., Hauptstein R., Geppert L.N., Köllmann C., Ickstadt K., Eckert P., Neshkova I., Jakubietz R. Qualitative and quantitative differences in estrogen biotransformation in human breast glandular and adipose tissues: implications for studies using mammary biospecimens. Arch. Toxicol. 2019;93(10):2823–2833. doi: 10.1007/s00204-019-02564-w. [DOI] [PubMed] [Google Scholar]
- 36.Perkins M.S. Stellenbosch University; Stellenbosch: 2018. Investigating the Mechanism of Action of Hormones Used in Hormone Replacement Therapy Via Estrogen Receptor Subtypes and the Influence of the Progesterone Receptor. [Google Scholar]
- 37.Subramanya V., Zhao D., Ouyang P., Lima J.A., Vaidya D., Ndumele C.E., Bluemke D.A., Shah S.J., Guallar E., Nwabuo C.C. Sex hormone levels and change in left ventricular structure among men and post-menopausal women: the Multi-Ethnic Study of Atherosclerosis (MESA) Maturitas. 2018;108:37–44. doi: 10.1016/j.maturitas.2017.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ding T., Zhang J., Wang T., Cui P., Chen Z., Jiang J., Zhou S., Dai J., Wang B., Yuan S. A Multi-hospital Study in Wuhan, China: Protective Effects of Non-menopause and Female Hormones on SARS-CoV-2 infection. medRxiv. 2020 doi: 10.1101/2020.03.26.20043943. In preparation. [DOI] [Google Scholar]
- 39.Ye Q., Wang B., Mao J. Cytokine storm in COVID-19 and treatment. J. Infect. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McGonagle D., Sharif K., O’Regan A., Bridgewood C. Interleukin-6 use in COVID-19 pneumonia related macrophage activation syndrome. Autoimmun. Rev. 2020 doi: 10.1016/j.autrev.2020.102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Drehmer M.N., Muniz Y.C.N., Marrero A.R., Löfgren S.E. Gene expression of ABHD6, a key factor in the endocannabinoid system, can Be modulated by female hormones in human immune cells. Biochem. Genet. 2019;57(1):35–45. doi: 10.1007/s10528-018-9871-8. [DOI] [PubMed] [Google Scholar]
- 42.Lou Y., Hu M., Wang Q., Yuan M., Wang N., Le F., Li L., Huang S., Wang L., Xu X. Estradiol suppresses TLR4-triggered apoptosis of decidual stromal cells and drives an anti-inflammatory TH2 shift by activating SGK1. Int. J. Biol. Sci. 2017;13(4):434. doi: 10.7150/ijbs.18278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dragin N., Nancy P., Villegas J., Roussin R., Le Panse R., Berrih-Aknin S. Balance between estrogens and proinflammatory cytokines regulates chemokine production involved in thymic germinal center formation. Sci. Rep. 2017;7(1):1–13. doi: 10.1038/s41598-017-08631-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Smy L., Straseski J.A. Measuring estrogens in women, men, and children: recent advances 2012–2017. Clin. Biochem. 2018;62:11–23. doi: 10.1016/j.clinbiochem.2018.05.014. [DOI] [PubMed] [Google Scholar]
- 45.Channappanavar R., Fett C., Mack M., Ten Eyck P.P., Meyerholz D.K., Perlman S. Sex-based differences in susceptibility to severe acute respiratory syndrome coronavirus infection. J. Immunol. 2017;198(10):4046–4053. doi: 10.4049/jimmunol.1601896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cutolo M., Smith V., Paolino S. Understanding immune effects of oestrogens to explain the reduced morbidity and mortality in female versus male COVID-19 patients. Comparisons with autoimmunity and vaccination. Clin. Exp. Rheumatol. 2020;38:383–386. [PubMed] [Google Scholar]
- 47.Al-Lami R.A., Urban R.J., Volpi E., Algburi A.M., Baillargeon J. Elsevier; 2020. Sex Hormones and Novel Corona Virus Infectious Disease (COVID-19), Mayo Clinic Proceedings. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cohn B.A., Cirillo P.M., Hopper B.R., Siiteri P.K. Third trimester estrogens and maternal breast cancer: prospective evidence. J. Clin. Endocrinol. Metab. 2017;102(10):3739–3748. doi: 10.1210/jc.2016-3476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Katzenellenbogen J.A., Mayne C.G., Katzenellenbogen B.S., Greene G.L., Chandarlapaty S. Structural underpinnings of oestrogen receptor mutations in endocrine therapy resistance. Nat. Rev. Cancer. 2018;18(6):377–388. doi: 10.1038/s41568-018-0001-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Laffont S., Seillet C., Guéry J.-C. Estrogen receptor-dependent regulation of dendritic cell development and function. Front. Immunol. 2017;8:108. doi: 10.3389/fimmu.2017.00108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chapple R.H., Hu T., Tseng Y.-J., Liu L., Kitano A., Luu V., Hoegenauer K.A., Iwawaki T., Li Q., Nakada D. ERα promotes murine hematopoietic regeneration through the Ire1α-mediated unfolded protein response. Elife. 2018;7 doi: 10.7554/eLife.31159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pelekanou V., Kampa M., Kiagiadaki F., Deli A., Theodoropoulos P., Agrogiannis G., Patsouris E., Tsapis A., Castanas E., Notas G. Estrogen anti‐inflammatory activity on human monocytes is mediated through cross‐talk between estrogen receptor ERα36 and GPR30/GPER1. J. Leukoc. Biol. 2016;99(2):333–347. doi: 10.1189/jlb.3A0914-430RR. [DOI] [PubMed] [Google Scholar]
- 53.Nguyen D.H. Springer; 2016. Macrophages, Extracellular Matrix, and Estrogens in Breast Cancer Risk, Systems Biology of Tumor Physiology; pp. 1–19. [Google Scholar]
- 54.Baci D., Bruno A., Cascini C., Gallazzi M., Mortara L., Sessa F., Pelosi G., Albini A., Noonan D.M. Acetyl-L-Carnitine downregulates invasion (CXCR4/CXCL12, MMP-9) and angiogenesis (VEGF, CXCL8) pathways in prostate cancer cells: rationale for prevention and interception strategies. J. Exp. Clin. Cancer Res. 2019;38(1):1–17. doi: 10.1186/s13046-019-1461-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zou Y.-H., Zhao L., Xu Y.-K., Bao J.-M., Liu X., Zhang J.-S., Li W., Ahmed A., Yin S., Tang G.-H. Anti-inflammatory sesquiterpenoids from the Traditional Chinese Medicine Salvia plebeia: Regulates pro-inflammatory mediators through inhibition of NF-κB and Erk1/2 signaling pathways in LPS-induced Raw264. 7 cells. J. Ethnopharmacol. 2018;210:95–106. doi: 10.1016/j.jep.2017.08.034. [DOI] [PubMed] [Google Scholar]
- 56.Mohammad I., Starskaia I., Nagy T., Guo J., Yatkin E., Väänänen K., Watford W.T., Chen Z. Estrogen receptor α contributes to T cell–mediated autoimmune inflammation by promoting T cell activation and proliferation. Sci. Signal. 2018;11(526) doi: 10.1126/scisignal.aap9415. [DOI] [PubMed] [Google Scholar]
- 57.Nagy E., Lei Y., Martínez-Martínez E., Body S.C., Schlotter F., Creager M., Assmann A., Khabbaz K., Libby P., Hansson G.K. Interferon-γ released by activated CD8+ T lymphocytes impairs the calcium resorption potential of osteoclasts in calcified human aortic valves. Am. J. Pathol. 2017;187(6):1413–1425. doi: 10.1016/j.ajpath.2017.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Papadimitriou C.A., Markaki S., Siapkaras J., Vlachos G., Efstathiou E., Grimani I., Hamilos G., Zorzou M., Dimopoulos M.-A. Hormonal therapy with letrozole for relapsed epithelial ovarian cancer. Oncology. 2004;66(2):112–117. doi: 10.1159/000077436. [DOI] [PubMed] [Google Scholar]
- 59.Jitschin R., Böttcher M., Saul D., Lukassen S., Bruns H., Loschinski R., Ekici A., Reis A., Mackensen A., Mougiakakos D. Inflammation-induced glycolytic switch controls suppressivity of mesenchymal stem cells via STAT1 glycosylation. Leukemia. 2019;33(7):1783–1796. doi: 10.1038/s41375-018-0376-6. [DOI] [PubMed] [Google Scholar]
- 60.Ghosh S., Klein R.S. Sex drives dimorphic immune responses to viral infections. J. Immunol. 2017;198(5):1782–1790. doi: 10.4049/jimmunol.1601166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Stubelius A., Andersson A., Islander U., Carlsten H. Ovarian hormones in innate inflammation. Immunobiology. 2017;222(8-9):878–883. doi: 10.1016/j.imbio.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 62.Marques P., Collado A., Martinez-Hervás S., Domingo E., Benito E., Piqueras L., Real J.T., Ascaso J.F., Sanz M.-J. Systemic inflammation in metabolic syndrome: increased platelet and leukocyte activation, and key role of CX3CL1/CX3CR1 and CCL2/CCR2 axes in arterial platelet-proinflammatory monocyte adhesion. J. Clin. Med. 2019;8(5):708. doi: 10.3390/jcm8050708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Klein S.L., Dhakal S., Ursin R.L., Deshpande S., Sandberg K., Mauvais-Jarvis F. Biological sex impacts COVID-19 outcomes. PLoS Pathog. 2020;16(6) doi: 10.1371/journal.ppat.1008570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fischinger S., Boudreau C.M., Butler A.L., Streeck H., Alter G. Seminars in Immunopathology. Springer; 2019. Sex differences in vaccine-induced humoral immunity; pp. 239–249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wang J., Syrett C.M., Kramer M.C., Basu A., Atchison M.L., Anguera M.C. Unusual maintenance of X chromosome inactivation predisposes female lymphocytes for increased expression from the inactive X. Proc. Natl. Acad. Sci. 2016;113(14):E2029–E2038. doi: 10.1073/pnas.1520113113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Souyris M., Cenac C., Azar P., Daviaud D., Canivet A., Grunenwald S., Pienkowski C., Chaumeil J., Mejía J.E., Guéry J.-C. TLR7 escapes X chromosome inactivation in immune cells. Sci. Immunol. 2018;3(19) doi: 10.1126/sciimmunol.aap8855. [DOI] [PubMed] [Google Scholar]
- 67.Conti P., Younes A. Coronavirus COV-19/SARS-CoV-2 affects women less than men: clinical response to viral infection. J. Biol. Regul. Homeost. Agents. 2020;34(2):71. doi: 10.23812/Editorial-Conti-3. [DOI] [PubMed] [Google Scholar]
- 68.Moreno-Eutimio M.A., Lopez-Macias C., Pastelin-Palacios R. Bioinformatic analysis and identification of single-stranded RNA sequences recognized by TLR7/8 in the SARS-CoV-2, SARS-CoV, and MERS-CoV genomes. Microbes Infect. 2020;22(4–5):226–229. doi: 10.1016/j.micinf.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Silva-Cardoso S.C., Affandi A.J., Spel L., Cossu M., Van Roon J.A., Boes M., Radstake T.R. CXCL4 exposure potentiates TLR-driven polarization of human monocyte-derived dendritic cells and increases stimulation of T cells. J. Immunol. 2017;199(1):253–262. doi: 10.4049/jimmunol.1602020. [DOI] [PubMed] [Google Scholar]
- 70.Raisi-Estabragh Z., McCracken C., Ardissino M., Bethell M.S., Cooper J., Cooper C., Harvey N.C., Petersen S.E. Non-white ethnicity, male sex, and higher body mass index, but not medications acting on the renin-angiotensin system are associated with coronavirus disease 2019 (covid-19) hospitalisation: review of the first 669 cases from the uk biobank. medRxiv. 2020 doi: 10.1101/2020.05.10.20096925. In press. [DOI] [Google Scholar]
- 71.Lorenz T., Demas G., Heiman J. 001 sexual activity changes patterns of inflammation in healthy women. J. Sex. Med. 2017;14(6):e351. [Google Scholar]
- 72.Tulli L., Cattaneo F., Vinot J., Baldari C.T., D’Oro U. Src family kinases regulate interferon regulatory factor 1 K63 ubiquitination following activation by TLR7/8 vaccine adjuvant in human monocytes and B cells. Front. Immunol. 2018;9:330. doi: 10.3389/fimmu.2018.00330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Deckx N., Willekens B., Wens I., Eijnde B.O., Goossens H., Van Damme P., Berneman Z.N., Cools N. Altered molecular expression of TLR-signaling pathways affects the steady-state release of IL-12p70 and IFN-α in patients with relapsing-remitting multiple sclerosis. Innate Immun. 2016;22(4):266–273. doi: 10.1177/1753425916642615. [DOI] [PubMed] [Google Scholar]
- 74.Green M.P., Harvey A.J., Spate L.D., Kimura K., Thompson J.G., Roberts R.M. The effects of 2, 4‐dinitrophenol and d‐glucose concentration on the development, sex ratio, and interferon‐tau (IFNT) production of bovine blastocysts. Mol. Reprod. Dev. 2016;83(1):50–60. doi: 10.1002/mrd.22590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Oishi I., Yoshii K., Miyahara D., Tagami T. Efficient production of human interferon beta in the white of eggs from ovalbumin gene–targeted hens. Sci. Rep. 2018;8(1):1–12. doi: 10.1038/s41598-018-28438-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Meester I., Manilla-Muñoz E., León-Cachón R.B., Paniagua-Frausto G.A., Carrión-Alvarez D., Ruiz-Rodríguez C.O., Rodríguez-Rangel X., García-Martínez J.M. SeXY chromosomes and the immune system: reflections after a comparative study. Biol. Sex Differ. 2020;11(1):3. doi: 10.1186/s13293-019-0278-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zhao H., Li D., Xie L., Cao Q., Sun Z., Li J., Hao L., Li L., Li J. HIV-derived microRNA-99 reduces phagocytic function of macrophages and enhances the release of cytokines via the TLR8 signaling pathway. Int. J. Clin. Exp. Med. 2018;11(10):10534–10541. [Google Scholar]
- 78.Wang Z., Wu T., Ma M., Zhang Z., Fu Y., Liu J., Xu J., Ding H., Han X., Chu Z. Elevated interferon‐γ–induced protein 10 and its receptor CXCR3 impair NK cell function during HIV infection. J. Leukoc. Biol. 2017;102(1):163–170. doi: 10.1189/jlb.5A1016-444R. [DOI] [PubMed] [Google Scholar]
- 79.Wang Z.-H., Liu T. MicroRNA21 meets neuronal TLR8: non-canonical functions of MicroRNA in neuropathic pain. Neurosci. Bull. 2019;35(5):949–952. doi: 10.1007/s12264-019-00366-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cao Y., Su B., Guo X., Sun W., Deng Y., Bao L., Zhu Q., Zhang X., Zheng Y., Geng C. Potent neutralizing antibodies against SARS-CoV-2 identified by high-throughput single-cell sequencing of convalescent patients’ B cells. Cell. 2020;182(1):73–84.e16. doi: 10.1016/j.cell.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Oberg H.H., Kellner C., Gonnermann D., Sebens S., Bauerschlag D., Gramatzki M., Kabelitz D., Peipp M., Wesch D. Tribody [(HER2) 2xCD16] is more effective than trastuzumab in enhancing γδ T cell and natural killer cell cytotoxicity against HER2-expressing cancer cells. Front. Immunol. 2018;9:814. doi: 10.3389/fimmu.2018.00814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Li Y., Huang K., Liu L., Qu Y., Huang Y., Wu Y., Wei J. Effects of complement and serum IgG on rituximab‑dependent natural killer cell‑mediated cytotoxicity against Raji cells. Oncol. Lett. 2018;17(1):339–347. doi: 10.3892/ol.2018.9630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Potluri T., Fink A.L., Sylvia K.E., Dhakal S., Vermillion M.S., Vom Steeg L., Deshpande S., Narasimhan H., Klein S.L. Age-associated changes in the impact of sex steroids on influenza vaccine responses in males and females. Npj Vaccines. 2019;4(1):1–12. doi: 10.1038/s41541-019-0124-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bochnowski A. Johns Hopkins University; 2016. The Effect of Donor Sex on Influenza A Infection in Primary Human Nasal Epithelial Cell Cultures. [Google Scholar]
- 85.He Y., Yang W., Liu S., Gan L., Zhang F., Mu C., Wang J., Qu L., Wang R., Deng J. Interactions between angiotensin-converting enzyme-2 polymorphisms and high salt intake increase the risk of hypertension in the Chinese Wa population. Int. J. Clin. Exp. Pathol. 2017;10(11):11159. [PMC free article] [PubMed] [Google Scholar]
- 86.Wysocki J., Schulze A., Zhao M., Haney C., Batlle D. Novel angiotensin-converting-Enzyme 2 (ACE2) truncates to target the kidney renin-angiotensin-System (RAS) Hypertension. 2019;74(Suppl_1) A099-A099. [Google Scholar]
- 87.Lin S., Pan H., Wu H., Ren D., Lu J. Role of the ACE2‑Ang‑(1‑7)‑Mas axis in blood pressure regulation and its potential as an antihypertensive in functional foods. Mol. Med. Rep. 2017;16(4):4403–4412. doi: 10.3892/mmr.2017.7168. [DOI] [PubMed] [Google Scholar]
- 88.Wu Y. Compensation of ACE2 function for possible clinical management of 2019-nCoV-induced acute lung injury. Virol. Sin. 2020:1–3. doi: 10.1007/s12250-020-00205-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.e Silva A.C.S., Miranda A.S., Rocha N.P., Teixeira A.L. Renin angiotensin system in liver diseases: Friend or foe? World J. Gastroenterol. 2017;23(19):3396. doi: 10.3748/wjg.v23.i19.3396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Vallejo-Ardila D.L., Fifis T., Burrell L.M., Walsh K., Christophi C. Renin-angiotensin inhibitors reprogram tumor immune microenvironment: a comprehensive view of the influences on anti-tumor immunity. Oncotarget. 2018;9(84):35500. doi: 10.18632/oncotarget.26174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Mascolo A., Urbanek K., De Angelis A., Sessa M., Scavone C., Berrino L., Rosano G.M.C., Capuano A., Rossi F. Angiotensin II and angiotensin 1–7: which is their role in atrial fibrillation? Heart Fail. Rev. 2020;25(2):367–380. doi: 10.1007/s10741-019-09837-7. [DOI] [PubMed] [Google Scholar]
- 92.de Farias Lelis D., de Freitas D.F., Machado A.S., Crespo T.S., Santos S.H.S. Angiotensin-(1-7), adipokines and inflammation. Metabolism. 2019;95:36–45. doi: 10.1016/j.metabol.2019.03.006. [DOI] [PubMed] [Google Scholar]
- 93.AlQudah M., Hale T.M., Czubryt M.P. Targeting the renin-angiotensin-Aldosterone system in fibrosis. Matrix Biol. 2020;91-92:92–108. doi: 10.1016/j.matbio.2020.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Li G., He X., Zhang L., Ran Q., Wang J., Xiong A., Wu D., Chen F., Sun J., Chang C. Assessing ACE2 expression patterns in lung tissues in the pathogenesis of COVID-19. J. Autoimmun. 2020 doi: 10.1016/j.jaut.2020.102463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Swärd P., Edsfeldt A., Reepalu A., Jehpsson L., Rosengren B.E., Karlsson M.K. Age and sex differences in soluble ACE2 may give insights for COVID-19. Crit. Care. 2020;24(1):1–3. doi: 10.1186/s13054-020-02942-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Batlle D., Wysocki J., Satchell K. Soluble angiotensin-converting enzyme 2: a potential approach for coronavirus infection therapy? Clin. Sci. 2020;134(5):543–545. doi: 10.1042/CS20200163. [DOI] [PubMed] [Google Scholar]
- 97.Xu J., Mukerjee S., Silva-Alves C.R., Carvalho-Galvão A., Cruz J.C., Balarini C.M., Braga V.A., Lazartigues E., França-Silva M.S. A disintegrin and metalloprotease 17 in the cardiovascular and central nervous systems. Front. Physiol. 2016;7:469. doi: 10.3389/fphys.2016.00469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kato T., Hagiyama M., Ito A. Renal ADAM10 and 17: their physiological and medical meanings. Front. Cell Dev. Biol. 2018;6:153. doi: 10.3389/fcell.2018.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhang J., Guo L., Yang L., Xu J., Zhang L., Feng L., Chen H., Wang Y. Metalloprotease ADAM17 regulates porcine epidemic diarrhea virus infection by modifying aminopeptidase N. Virology. 2018;517:24–29. doi: 10.1016/j.virol.2018.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Yang P., Gu H., Zhao Z., Wang W., Cao B., Lai C., Yang X., Zhang L., Duan Y., Zhang S. Angiotensin-converting enzyme 2 (ACE2) mediates influenza H7N9 virus-induced acute lung injury. Sci. Rep. 2014;4:7027. doi: 10.1038/srep07027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zou Z., Yan Y., Shu Y., Gao R., Sun Y., Li X., Ju X., Liang Z., Liu Q., Zhao Y. Angiotensin-converting enzyme 2 protects from lethal avian influenza A H5N1 infections. Nat. Commun. 2014;5(1):1–7. doi: 10.1038/ncomms4594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Spiekerkoetter E., Kawut S.M., de Jesus Perez V.A. New and emerging therapies for pulmonary arterial hypertension. Annu. Rev. Med. 2019;70:45–59. doi: 10.1146/annurev-med-041717-085955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sullivan J.C., Rodriguez-Miguelez P., Zimmerman M.A., Harris R.A. Differences in angiotensin (1–7) between men and women. American Journal of Physiology-Heart and Circulatory Physiology. 2015;308(9):H1171–H1176. doi: 10.1152/ajpheart.00897.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Asselta R., Paraboschi E.M., Mantovani A., Duga S. 2020. ACE2 And TMPRSS2 Variants and Expression As Candidates to Sex and Country Differences in COVID-19 Severity in Italy. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Kornilov S.A., Lucas I., Jade K., Dai C.L., Lovejoy J.C., Magis A.T. Plasma levels of soluble ACE2 are associated with sex, Metabolic Syndrome, and its biomarkers in a large cohort, pointing to a possible mechanism for increased severity in COVID-19. medRxiv. 2020 doi: 10.1101/2020.06.10.20127969. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Verdecchia P., Cavallini C., Spanevello A., Angeli F. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur. J. Intern. Med. 2020;76:14–20. doi: 10.1016/j.ejim.2020.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.McCulloch D.R., Harvey M., Herington A.C. The expression of the ADAMs proteases in prostate cancer cell lines and their regulation by dihydrotestosterone. Mol. Cell. Endocrinol. 2000;167(1–2):11–21. doi: 10.1016/s0303-7207(00)00305-1. [DOI] [PubMed] [Google Scholar]
- 108.Ren J., Nie Y., Lv M., Shen S., Tang R., Xu Y., Hou Y., Zhao S., Wang T. Estrogen upregulates MICA/B expression in human non-small cell lung cancer through the regulation of ADAM17. Cell. Mol. Immunol. 2015;12(6):768–776. doi: 10.1038/cmi.2014.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bass R., Edwards D.R. ADAMs and protein disulfide isomerase: the key to regulated cell-surface protein ectodomain shedding? Biochem. J. 2010;428(3):e3–e5. doi: 10.1042/BJ20100568. [DOI] [PubMed] [Google Scholar]
- 110.Hartmann M., Herrlich A., Herrlich P. Who decides when to cleave an ectodomain? Trends Biochem. Sci. 2013;38(3):111–120. doi: 10.1016/j.tibs.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 111.Schultz-Norton J.R., McDonald W.H., Yates J.R., Nardulli A.M. Protein disulfide isomerase serves as a molecular chaperone to maintain estrogen receptor α structure and function. Mol. Endocrinol. 2006;20(9):1982–1995. doi: 10.1210/me.2006-0006. [DOI] [PubMed] [Google Scholar]
- 112.Ejima K., Nanri H., Araki M., Uchida K., Kashimura M., Ikeda M. 17beta-estradiol induces protein thiol/disulfide oxidoreductases and protects cultured bovine aortic endothelial cells from oxidative stress. Eur. J. Endocrinol. 1999;140(6):608–613. doi: 10.1530/eje.0.1400608. [DOI] [PubMed] [Google Scholar]
- 113.Nuss J.E., Choksi K.B., DeFord J.H., Papaconstantinou J. Decreased enzyme activities of chaperones PDI and BiP in aged mouse livers. Biochem. Biophys. Res. Commun. 2008;365(2):355–361. doi: 10.1016/j.bbrc.2007.10.194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Wang J., Green P., Simpkins J. Estradiol protects against ATP depletion, mitochondrial membrane potential decline and the generation of reactive oxygen species induced by 3‐nitroproprionic acid in SK‐N‐SH human neuroblastoma cells. J. Neurochem. 2001;77(3):804–811. doi: 10.1046/j.1471-4159.2001.00271.x. [DOI] [PubMed] [Google Scholar]
- 115.Chen J.-R., Shankar K., Nagarajan S., Badger T.M., Ronis M.J. Protective effects of estradiol on ethanol-induced bone loss involve inhibition of reactive oxygen species generation in osteoblasts and downstream activation of the extracellular signal-regulated kinase/signal transducer and activator of transcription 3/receptor activator of nuclear factor-κB ligand signaling cascade. J. Pharmacol. Exp. Ther. 2008;324(1):50–59. doi: 10.1124/jpet.107.130351. [DOI] [PubMed] [Google Scholar]
- 116.Alhenc-Gelas F., Drueke T.B. Blockade of SARS-CoV-2 infection by recombinant soluble ACE2. Kidney Int. 2020;97(6):1091–1093. doi: 10.1016/j.kint.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.South A.M., Diz D.I., Chappell M.C. COVID-19, ACE2, and the cardiovascular consequences. Am. J. of Physiol. Heart Circulatory Physiol. 2020;318(5):1084–1090. doi: 10.1152/ajpheart.00217.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Backlund M. Dissertationes Scholae Doctoralis Ad Sanitatem Investigandam Universitatis Helsinkiensis. 2016. Regulation of angiotensin II type receptor by its messenger RNA-binding proteins. [Google Scholar]
- 119.Tazumi S., Yokota N., Kawakami M., Omoto S., Takamata A., Morimoto K. Effects of estrogen replacement on stress-induced cardiovascular responses via renin-angiotensin system in ovariectomized rats, American Journal of Physiology-Regulatory. Integrative Comparative Physiol. 2016;311(5):R898–R905. doi: 10.1152/ajpregu.00415.2015. [DOI] [PubMed] [Google Scholar]
- 120.South A.M., Brady T.M., Flynn J.T. ACE2, COVID-19, and ACE Inhibitor and ARB Use during the Pandemic: The Pediatric Perspective. Hypertension. 2020;76(1):16–22. doi: 10.1161/HYPERTENSIONAHA.120.15291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Grundy E.J., Suddek T., Filippidis F.T., Majeed A., Coronini-Cronberg S. Smoking, SARS-CoV-2 and COVID-19: A review of reviews considering implications for public health policy and practice. Tob. Induc. Dis. 2020;18 doi: 10.18332/tid/124788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.van Zyl-Smit R.N., Richards G., Leone F.T. Tobacco smoking and COVID-19 infection. Lancet Respir. Med. 2020;8(7):664–665. doi: 10.1016/S2213-2600(20)30239-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Vij N., Chandramani-Shivalingappa P., Van Westphal C., Hole R., Bodas M. Cigarette smoke-induced autophagy impairment accelerates lung aging, COPD-emphysema exacerbations and pathogenesis. Am. J. Physiol. Cell Physiol. 2018;314(1):C73–C87. doi: 10.1152/ajpcell.00110.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Giagulli V.A., Guastamacchia E., Magrone T., Jirillo E., Lisco G., De Pergola G., Triggiani V. Worse progression of COVID‐19 in men: Is Testosterone a key factor? Andrology. 2020 doi: 10.1111/andr.12836. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Cochran C.J., Gallicchio L., Miller S.R., Zacur H., Flaws J.A. Cigarette smoking, androgen levels, and hot flushes in midlife women. Obstet. Gynecol. 2008;112(5):1037–1044. doi: 10.1097/AOG.0b013e318189a8e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Brand J.S., Chan M.F., Dowsett M., Folkerd E., Wareham N.J., Luben R.N., van der Schouw Y.T., Khaw K.T. Cigarette smoking and endogenous sex hormones in postmenopausal women. J. Clin. Endocrinol. Metab. 2011;96(10):3184–3192. doi: 10.1210/jc.2011-1165. [DOI] [PubMed] [Google Scholar]
- 127.Chakladar J., Shende N., Li W.T., Rajasekaran M., Chang E.Y., Ongkeko W.M. Smoking-mediated upregulation of the androgen pathway leads to increased SARS-CoV-2 susceptibility. Int. J. Mol. Sci. 2020;21(10):3627. doi: 10.3390/ijms21103627. [DOI] [PMC free article] [PubMed] [Google Scholar]